15016 Vesicular Olivine-normative Basalt grams
|
|
|
- Eric Copeland
- 5 years ago
- Views:
Transcription
1 15016 Vesicular Olivine-normative Basalt grams Figure 1: Photograph of vesicular basalt NASA# S Cube is 1 inch. Introduction Lunar Sample is a highly-vesicular, olivinenormative, basalt with a major element composition similar to that of non-vesicular basalt (figure 1). These basalts are typical of many of the basalt samples returned from the Apollo 15, Hadley Rille site, and their composition has been studied experimentally to conclude that this volcanic magma came from a depth of greater than 250 km and are also very vesicular basalts from other locations at Apollo 15 site has been dated at 3.4 b.y. and has been exposed to cosmic rays for 300 m.y. Petrography Lunar sample is a medium-grained basalt with subhedral phenocrysts of zoned pyroxene (1-2 mm) and olivine (~1 mm) set in a matrix of subophitic intergrowths of pyroxene and plagioclase (figures 2 and 3). Vesicles (1 to 5 mm) make up about 50 % of the volume. Opaque minerals (ilmenite and ulvöspinel) frequently border the vesicles. Plagioclase platelets are sometimes hollow, with pyroxene cores. Subrounded grains of Cr-spinel are found in the pyroxene and olivine phenocrysts. Troilite and Fe-Ni metal are found in the mesostasis. Experiments show that the paragenetic sequence is Crspinel (above 1300 C), olivine (1280 C), pyroxene (1170 C) and plagioclase (below 1150 C) matching the texture of the thin sections. Brown et al. (1972) found both low-k and high-k glass in the mesostasis, and speculate on Na loss during vesiculation. The interior walls of the vesicles have been studied by Goldberg et al. (1976).
2 Figure 2: Photomicrographs of thin section 15016,146 by C 20x and 100x. Mineralogy Olivine: Olivine ranges in composition Fo (Bence and Papike 1972). Pyroxene: Pyroxene analyses are given in Bence and Papike (1972), Papike et al. (1976), and Kushiro (1973) (figure 4). Large pyroxene grains are highly zoned first towards Ca-rich, then Fe-rich, indicating rapid crystallization. Plagioclase: Plagioclase ranges An (Bence and Papike 1972). Brown et al. (1972) found reverse zoning indicating volatile loss. Ilmenite: Ilmenite was studied by Engelhardt (1979). Silica: Brown et al. (1972) found that silica in was cristobalite. Mineralogical Mode for Brown et al. Papike et al. McGee et al Olivine 6-10 vol. % Pyroxene Plagioclase Opaques Ilmenite 6 Chromite 0.1 Ulvöspinel 0.4 Mesostasis 0.3 Silica
3 Figure 3: Photomicrographs of thin section 15016,146 (crossed nicols) by C 20x and 100x. Chemistry Although the major element composition of is very similar to that of 15555, the trace element composition is higher (figure 6). Cr may also be higher in than Barker (1974) and Gibson et al. (1975) studied the gas (CO, CO 2 etc.) released by heating or mechanically crushing the sample. Goldberg et al. (1976) studied fluorine in the vesicle walls. Radiogenic age dating Murthy et al. (1973) and Evensen et al. (1973) determined the age of as 3.29 ± 0.05 b.y. by the Rb-Sr method (figure 7). Both the age and the initial Sr isotopic ratio ( 87 Sr/ 86 Sr = ± ) are similar to that of measured in the same lab. Kirsten et al. (1973) determined a plateau age of 3.38 ± 0.08 b.y. Snyder et al. (1998) determined mineral isochrons by Rb/Sr and Sm/Nd giving ages 3.34 ± 0.03 b.y. and 3.22 ± 0.07 b.y. (respectively)(figure 8). Cosmogenic isotopes and exposure ages O Kelley et al. (1972) and Eldridge et al. (1972) determined the 26 Al, 22 Na, 46 Sc, 48 V, 54 Mn and 56 Co activity by whole rock radiation counting. Aluminum 26 is saturated!
4 Di Bence abd Papike Hd 14 Lunar Basalts Kushiro A17 10 A11 TiO2 8 6 En Fo compiled by C Meyer Figure 4: Pyroxene and olivine composition of 15016, showing extreme Fe-enrichment (data from Bence and Papike 1972 and Kushiro 1973). Fs Fa A15 A MgO Figure 5: Composition diagram for lunar basalts with Kirsten et al. (1973) determined a 38 Ar exposure age of 285 m.y. Husain (1974) determined 315 m.y. Other Studies and are among the more magnesian of the Apollo 15 basalts (figure 5). Phase diagrams have been prepared from the experimental studies of Humphries et al. (1972), Kushiro (1972) (figure 9) and Kesson (1975, 1977)(figure 10). These liquids are multi-saturated at ~12 kbar and 1350 deg C, indicating that the basaltic liquid may have come from an olivinepyroxenite at greater than 250 km depth in the moon. Figure 6: Normalized rare-earth-element diagram for (Neal 2001), with soil (Wanke et al. 1973) for comparison. Gose et al. (1972) and Pearce et al. (1973) studied the magnetic properties of chips of Cisowski et al. (1975) gives a low field strength estimate for magnetization. Charette and Adams (1975) determined the reflectance spectra of was similar to that of Clayton et al. (1972) reported the isotopic composition of oxygen. Summary of Age Data for Ar/Ar Rb/Sr Pb/Pb Sm/Nd Evensen et al ± 0.05 b.y. Kirsten et al ± 0.08 Husain 1974 no plateau Anderson and Hinthorne ± 0.27 (ion probe, phosphate) Snyder et al ± ± 0.07 Caution: Original radioactive decay constants are used.
5 Figure 7: Rb/Sr isochron diagram for (from Evensen et al. 1973). Figure 9: High-pressure phase diagram for (from Kushiro 1972). Figure 10: Phase diagram for mare basalts (Kesson 1975, 1977). Figure 8: Sm/Nd and Rb/Sr isochrons for (from Snyder et al. 1997). Table 2 U ppm Th ppm K ppm Rb ppm Sr ppm Nd ppm Sm ppm technique Evensen et al IDMS Compston et al IDMS IDMS IDMS O Kelley et al counting Snyder et al IDMS
6 Table 1a. Chemical composition of reference A15PET Kushiro 72 Cuttitta 73 Laul 73 Muller 75 Taylor 73 Janghorbani 73 Baedecker 73 weight Rhodes 73 Christian mg replica SiO2 % (a) (b) 44.3 (b) TiO (a) 2.28 (b) (c ) 1.77 (d) 3 Al2O (a) 8.17 (b) (c ) 8.31 FeO (a) 22.5 (b) (c ) (d) MnO 0.33 (a) 0.33 (b) (c ) 0.27 MgO (a) (b) (c ) CaO 9.4 (a) 9.06 (b) (c ) (d) Na2O 0.21 (a) 0.24 (b) (c ) (d) K2O 0.03 (a) 0.04 (b) (c ) P2O (a) 0.25 (b) 0.06 S % 0.07 (a) 0.19 (b) sum Sc ppm (c ) 25 (d) V (c ) 140 (d) Cr 7526 (b) (c ) 4100 (d) Co (c ) 56 (d) Ni (d) 85 (e) Cu (d) Zn 1.8 (e) Ga (e) Ge ppb 28 (e) As Se Rb (d) Sr 83 (a) Y (d) Zr 95 (a) (c ) 94 (d) Nb < (d) Mo Ru Rh Pd ppb Ag ppb Cd ppb 2 (e) In ppb 0.36 (e) Sn ppb 190 (d) Sb ppb Te ppb Cs ppm 0.04 Ba (c ) (d) La < (c ) (d) Ce 14.4 (d) Pr 2 (d) Nd 9.6 (d) Sm 3.6 (c ) 3.42 (d) Eu 0.8 (c ) 0.87 (d) Gd 4.5 (d) Tb 0.59 (c ) 0.73 (d) Dy 4.55 (d) Ho 1.1 (d) Er 2.86 (d) Tm 0.4 (d) Yb (c ) 2.42 (d) Lu 0.35 (c ) 0.38 (d) Hf 2.5 (c ) 2.04 (d) Ta 0.4 (c ) W ppb Re ppb Os ppb Ir ppb 0.12 (e) Pt ppb Au ppb 0.27 (e) Th ppm 0.5 (d) U ppm (d) technique (a) XRF, (b) conventional, (c ) INAA, (d) SSMS, (e) RNAA
7 Table 1b. Chemical composition of reference Chappell 73 Helmke 73 Wolf 79 Garg 76 O Kelly 72 Evensen 73 Compston 72 Gibson 75 weight 1.04 g 923 grams SiO2 % (f) TiO (f) Al2O (f) FeO (f) MnO 0.31 (f) MgO (f) CaO 9.43 (f) Na2O 0.32 (f) K2O 0.05 (f) (I) (j) P2O (f) S % sum Sc ppm 39.1 (g) V Cr 5500 (f) 6400 (g) Co 54 (g) Ni 68 (h) Cu Zn < (h) Ga 2.7 (f) 3.2 (g) Ge ppb 4.38 (h) As Se (h) Rb 0.65 (f) (h) 0.67 (j) (j) Sr 93.3 (f) 90.7 (j) (j) Y 23 (f) Zr 86 (f) 94.7 (h) Nb 7 (f) Mo Ru Rh Pd ppb Ag ppb 0.84 (h) Cd ppb 2.05 (h) In ppb 0.34 (h) Sn ppb <60 (h) Sb ppb 3.8 (h) Te ppb 2.4 (h) Cs ppm (g) (h) Ba La 5.77 (g) Ce 15.6 (g) Pr Nd 11.4 (g) Sm 4.05 (g) Eu 0.97 (g) Gd 5.4 (g) Tb 0.9 (g) Dy 5.74 (g) Ho 1.1 (g) Er 3.1 (g) Tm Yb 2.62 (g) Lu (g) Hf 2.6 (g) 2.53 (h) Ta W ppb Re ppb (h) Os ppb <0.01 (h) Ir ppb (h) Pt ppb Au ppb (h) Th ppm 0.52 (I) U ppm 0.16 (h) 0.15 (I) technique (f) XRF (g) INAA, (h) RNAA, (I) radiation counting, (j) IDMS
8 Table 1c. Chemical composition of reference Ryder 2001 Neal 2001 weight 5 g SiO2 % 44.6 (a) TiO (a) Al2O (a) FeO (a) 21.8 (b) MnO 0.28 (a) MgO (a) CaO 9.37 (a) Na2O (a) (b) K2O (a) P2O (a) S % sum Sc ppm 40.7 (b) 42 (h) V 304 (h) Cr 6000 (a) 5840 (b) 5867 (h) Co 54.6 (b) 58 (h) Ni 72 (a) 66 (b) 75 (h) Cu 9 (a) 14 (h) Zn 18 (h) Ga 3.65 (h) Ge ppb As Se Rb 2 (a) 0.94 (h) Sr 90 (a) 100 (b) 97.2 (h) Y 26 (a) 31 (h) Zr 85 (a) 109 (h) Nb 9 (a) 7.5 (h) Mo 0.08 (h) Ru Rh Pd ppb Ag ppb Cd ppb In ppb Sn ppb Sb ppb Te ppb Cs ppm 0.02 (h) Ba 57 (b) 56 (h) La 5.22 (b) 5.9 (h) Ce 15.2 (b) 15 (h) Pr 2.24 (h) Nd 8 (b) 10.5 (h) Sm 3.67 (b) 3.42 (h) Eu 0.82 (b) 0.87 (h) Gd 4.53 (h) Tb 0.82 (b) 0.8 (h) Dy 5.1 (h) Ho 1 (h) Er 2.77 (h) Tm 0.38 (h) Yb 2.18 (b) 2.38 (h) Lu 0.31 (b) 0.31 (h) Hf 2.49 (b) 2.58 (h) Ta 0.37 (b) 0.46 (h) W ppb 0.05 (h) Re ppb Os ppb Ir ppb Pt ppb Au ppb Th ppm 0.41 (b) 0.26 (h) U ppm 0.08 (h) technique (f) XRF (g) INAA, (h) ICP-MS
9 C Meyer grams,0 313 g,1 19 g soil, g, g,0 513 g, g, g,2 PB,5,14 TS,16 80 g,17 5g,17,19,21 9g,22 PB,140 allocations,47 PB,157,20 65 g display, g sawdust, g, g,150 TS,157 PM Processing was sawn, several times, and pieces of are used for public display. There are 21 thin sections. Photo #s of S color S color S B&W S TS S group
10 References for Andersen C.A. and Hinthorne J.R. (1973b) 207 Pb/ 206 Pb ages and REE abundances in returned lunar materials by ion microprobe mass analysis (abs). Lunar Sci. IV, Lunar Planetary Institute, Houston. Barker C. (1974) Composition of the gases associated with the magmas that produced rocks and Proc. 5 th Lunar Sci. Conf Baedecker P.A., Chou C.-L., Grudewicz E.B. and Wasson J.T. (1974) Volatile and siderophile trace elements in Apollo 15 samples: Geochemical implications and characterization of the long-lived and short-lived extralunar materials. Proc. 4 th Lunar Sci. Conf Bence A.E. and Papike J.J. (1972) Pyroxenes as recorders of lunar basalt petrogenesis: Chemical trends due to crystalliquid interaction. Proc. 3 rd Lunar Sci. Conf Brown G.M., Emeleus C.H., Holland G.J., Peckett A. and Phillips R. (1972) Mineral-chemical variations in Apollo 14 and Apollo 15 basalts and granitic fractions. Proc. 3 rd Lunar Sci. Conf Butler P. (1971) Lunar Sample Catalog, Apollo 15. Curators Office, MSC Chappell B.W. and Green D.H. (1973) Chemical compositions and petrogenetic relationships in Apollo 15 mare basalts. Earth Planet. Sci. Lett. 18, Charette M.P. and Adams J.B. (1975a) Mare basalts: Characterization of compositional parameters by spectral reflectance. In Papers presented to the Conference on Origins of Mare Basalts and their Implications for Lunar Evolution, Lunar Planetary Institute, Houston. Christian R.P., Annell C.S., Carron M.K., Cuttitta F., Dwornik E.J., Ligon D.T. and Rose H.J. (1972) Chemical composition of some Apollo 15 igneous rocks. In The Apollo 15 Lunar Samples (Chamberlain and Watkins, eds.), The Lunar Science Institute, Houston. Cisowski S.M., Collinson D.W., Runcom S.K., Stephenson A. and Fuller M. (1983) A review of lunar paleointensity data and implications for the origin of lunar magnetism. Proc. 13 th Lunar Planet. Sci. Conf. A691-A704. Clayton R.N., Hurd Julie and Mayeda T.K. (1972) Oxygen isotopic compositions and oxygen concentrations of Apollo 14 and Apollo 15 rocks and soils. Proc. 3 rd Lunar Sci. Conf Compston W., de Laeter J.R. and Vernon M.J. (1972) Strontium isotope geochemistry of Apollo 15 basalts. In The Apollo 15 Lunar Samples (Chamberlain and Watkins, eds.), Lunar Science Institute, Houston. Cuttita R., Rose H.J., Annell C.S., Carron M.K., Christian R.P., Ligon D.T., Dwornik E.J., Wright T.L. and Greenland L.P. (1973) Chemistry of twenty-one igneous rocks and soils returned by the Apollo 15 mission. Proc. 4 th Lunar Sci. Conf Ehmann W.D. and Chyi L.L. (1974) Abundances of the group IVB elements, Ti, Zr, and Hf and implications of their ratios in lunar materials. Proc. 5 th Lunar Sci. Conf Eldridge J.S., O Kelley G.D. and Northcutt K.J. (1972) Concentrations of cosmogenic radionuclides in Apollo 15 rocks and soils. In The Apollo 15 Lunar Samples Lunar Sci. Institute, Houston. von Engelhardt W. (1979) Ilmenite in the crystallization sequence of lunar rocks. Proc. 10 th Lunar Sci. Conf Evensen N.M., Murthy V.R. and Coscio M.R. (1973) Rb-Sr ages of some mare basalts and the isotopic and trace element systematics in lunar fines. Proc. 4 th Lunar Sci. Conf Garg A.N. and Ehmann W.N. (1976a) Zr-Hf fractionation in chemically defined lunar rock groups. Proc. 7 th Lunar Sci. Conf Gibson E.K., Chang S., Lennon K., Moore G.W. and Pearce G.W. (1975a) Sulfur abundances and distributions in mare basalts and their source magmas. Proc. 6 th Lunar Sci. Conf Goldberg R.H., Trombrello T.A. and Burnett D.S. (1976) Flourine as a constituent in lunar magmatic gases. Proc. 7 th Lunar Sci. Conf Gose W.A., Pearce G.W., Strangway D.W. and Carnes J. (1972) Magnetism of Apollo 15 samples. In The Apollo 15 Lunar Samples Helmke P.A., Blanchard D.P., Haskin L.A., Telander K., Weiss C. and Jacobs J.W. (1973) Major and trace elements in igneous rocks from Apollo 15. The Moon 8, Husain L. (1972) 40 Ar- 39 Ar and cosmic ray exposure ages of the Apollo 15 crystalline rocks, breccias and glasses (abs). In The Apollo 15 Lunar Samples Lunar Planetary Institute, Houston. Husain L. (1974) 40 Ar- 39 Ar chronology and cosmic ray exposure ages of the Apollo 15 samples. J. Geophys. Res. 79,
11 Humphries D.J., Biggar G.M and O Hara M.J. (1972) Phase equilibria and origin of Apollo 15 basalts etc. In The Apollo 15 Lunar Samples Lunar Planetary Institute, Houston. Janghorbani M., Miller M.D., Ma M-S., Chyi L.L. and Ehmann W.D. (1973) Oxygen and other elemental abundance data for Apollo 14, 15, 16 and 17 samples. Proc. 4 th Lunar Sci. Conf Kaplan I.R., Kerridge J.F. and Petrowski C. (1976) Light element geochemistry of the Apollo 15 site. Proc. 7 th Lunar Sci. Conf Kesson S.E. (1975a) Mare basalt petrogenesis. In Papers presented to the Conference on Origins of Mare Basalts and their Implications for Lunar Evolution, Lunar Planetary Institute, Houston. Kesson S.E. (1975b) Mare basalts: melting experiments and petrogenetic interpretations. Proc. 6 th Lunar Sci. Conf Kesson S.E. (1975c) Melting experiments on synthetic mare basalts and their petrogenetic implications (abs). Lunar Sci. VI, Lunar Planetary Institute, Houston. Kesson S.E. (1977) Mare basalt petrogenesis. Phil. Trans. Roy. Soc. London A285, Kirsten T., Deubner J., Horn P., Kaneoka I., Kiko J., Schaeffer O.A. and Thio S.K. (1972) The rare gas record of Apollo 14 and 15 samples. Proc. 3 rd Lunar Sci. Conf Kushiro I. (1972) Petrology of some Apollo 15 basalts. In The Apollo 15 Lunar Samples (Chamberlain and Watkins eds.), Lunar Science Institute, Houston. Laul J.C. and Schmitt R.A. (1973b) Chemical composition of Apollo 15, 16, and 17 samples. Proc. 4 th Lunar Sci. Conf Lee D-C., Halliday A.N., Snyder G.A. and Taylor L.A. (1997) Age and origin of the Moon. Science 278, Lee D-C., Halliday A.N., Snyder G.A. and Taylor L.A. (2000) Lu-Hf systematics and evolution of the moon (abs#1288). Lunar Planet. Sci. XXXI, Lunar Planetary Institute, Houston.. Lee D.C., Halliday A.N., Leya I., Wieler R. and Weichert U. (2002) Cosmogenic tungsten and the origin and earliest differentiation of the Moon. Earth Planet. Sci. Lett. 198, LSPET (1972a) The Apollo 15 lunar samples: A preliminary description. Science 175, LSPET (1972b) Preliminary examination of lunar samples. Apollo 15 Preliminary Science Report. NASA SP-289, McGee P.E., Warner J.L. and Simonds C.H. (1977) Introduction to the Apollo Collections. Part I: Lunar Igneous Rocks. Curators Office, JSC. Muller O. (1972) Alkali and alkaline earth elements, La and U in Apollo 14 and Apollo 15 samples. In The Apollo 15 Lunar Samples Müller O. (1975) Lithophile trace and major elements in Apollo 16 and 17 lunar samples. Proc. 6 th Lunar Sci. Conf Murthy V.R., Evensen N.M., Jahn B.-M. and Coscio M.R. (1972) Apollo 14 and 15 samples: Rb-Sr ages, trace elements, and lunar evolution. Proc. 3 rd Lunar Sci. Conf Neal C.R. (2001) Interior of the moon: The presence of garnet in the primitive deep lunar mantle. J. Geophys. Res. 106, Nyquist L.E. (1977) Lunar Rb-Sr chronology. Phys. Chem. Earth 10, O Kelley G.D., Eldridge J.S. and Northcutt K.J. (1972a) Abundances of primordial radioelements K, Th, and U in Apollo 15 samples, as determined by non-destructive gamma-ray spectrometry. In The Apollo 15 Lunar Samples (Chamberlain and Watkins, eds.), Lunar Science Institute, Houston. O Kelley G.D., Eldridge J.S., Schonfeld E. and Northcutt K.J. (1972b) Primordial radionuclides and cosmogenic radionuclides in lunar samples from Apollo 15. Science 175, O Kelley G.D., Eldridge J.S., Northcutt K.J. and Schonfeld E. (1972c) Primordial radionuclides and cosmogenic radionuclides in lunar samples from Apollo 15. Proc. 3 rd Lunar Sci. Conf Papike J.J., Bence A.E. and Ward M.A. (1972) Subsolidus relations of pyroxenes from Apollo 15 basalts. In The Apollo 15 Lunar Samples Lunar Science Institute, Houston. Papike J.J. (1996) Pyroxene as a recorder of cumulate formational processes in asteroids, Moon, mars and Earth: Reading the record with the ion microprobe. Am. Mineral. 81, Papike J.J., Hodges F.N., Bence A.E., Cameron M. and Rhodes J.M. (1976) Mare basalts: Crystal chemistry,
12 mineralogy and petrology. Rev. Geophys. Space Phys. 14, Papike J.J. and Bence A.E. (1978) Lunar mare vs. terrestrial mid-ocean ridge basalts: Planetary constraints on basaltic volcanism. Geophys. Res. Lett. 5, Papike J.J. and Vaniman D.T. (1978) The lunar mare basalt suite. Geophys. Res. Lett. 5, Pearce G.W., Gose W.A. and Strangway D.W. (1973) Magnetic studies on Apollo 15 and 16 lunar samples. Proc. 4 th Lunar Sci. Conf Geologic Investigation of the Apollo 15 landing site. In Apollo 15 Preliminary Science Rpt. NASA SP-289. pages Taylor S.R., Gorton M., Muir P., Nance W., Rudowski R. and Ware N. (1972b) Composition of the lunar highlands II The Apennine Front. In The Apollo 15 Lunar Samples, Wolf R., Woodrow A. and Anders E. (1979) Lunar basalts and pristine highland rocks: Comparison of siderophile and volatile elements. Proc. 10 th Lunar Planet. Sci. Conf Rhodes J.M. and Hubbard N.J. (1973) Chemistry. classification, and petrogenesis of Apollo 15 mare basalts. Proc. 4 th Lunar Sci. Conf Ryder G. (1985) Catalog of Apollo 15 Rocks (three volumes). Curatoial Branch Pub. # 72, JSC#20787 Ryder G. and Schuraytz B.C. (2001) Chemical variations of the large Apollo 15 olivine-normative mare basalt rock samples. J. Geophys. Res. 106, E1, Schnare D.W., Day J.M.D., Norman M.D., Liu Y. and Taylor L.A. (2008) A laser-ablation ICP-MS study of Apollo 15 low-titanium olivine-normative and quartz-normative mare basalts. Geochim. Cosmochim. Acta 72, Snyder G.A., Borg L.E., Taylor L.A., Nyquist L.E. and Halliday A.N. (1998) Volcanism in the Hadley-Apennine region of the Moon: Geochronology, Nd-Sr isotopic systematics and depths of melting.(abs#1141). Lunar Planet. Sci. XXIX, Lunar Planetary Institute, Houston. Snyder G.A., Borg L.E., Lee D.C., Nyquist L.E., Taylor L.A. and Halliday A.N. (1999a) Volcanism in the Hadley- Apennine region of the Moon: Chronology, Nd-Sr-Hf isotopic systematics and petrogenesis of Apollo 15 mare basalts. Geochim. Cosmochim. Acta (say what?) Snyder G.A., Lee D.C., Taylor L.A. and Halliday A.N. (1999b) Earliest lunar volcanism: An alternative interpretation of the Apollo 14 high-al basalts from Nd-Sr- Hf isotopic studies. Meteor. & Planet. Sci. (say what?) Swann G.A., Hait M.H., Schaber G.C., Freeman V.L., Ulrich G.E., Wolfe E.W., Reed V.S. and Sutton R.L. (1971b) Preliminary description of Apollo 15 sample environments. U.S.G.S. Interagency report: 36. pp219 with maps Swann G.A., Bailey N.G., Batson R.M., Freeman V.L., Hait M.H., Head J.W., Holt H.E., Howard K.A., Irwin J.B., Larson K.B., Muehlberger W.R., Reed V.S., Rennilson J.J., Schaber G.G., Scott D.R., Silver L.T., Sutton R.L., Ulrich G.E., Wilshire H.G. and Wolfe E.W. (1972) 5. Preliminary
15556 Vesicular Olivine-normative Basalt grams
 15556 Vesicular Olivine-normative Basalt 1542.3 grams Figure 1: Photo of 15556,0. Largest vesicles are about 6 mm. NASA S87-48187. Introduction 15556 was collected about 60 meters from the edge of Hadley
15556 Vesicular Olivine-normative Basalt 1542.3 grams Figure 1: Photo of 15556,0. Largest vesicles are about 6 mm. NASA S87-48187. Introduction 15556 was collected about 60 meters from the edge of Hadley
grams g g g g Regolith Breccia
 15306 134.2 grams 15315 35.6 g 15324 32.3 g 15325 57.8 g 15330 57.8 g Regolith Breccia Figure 1a,b: Photo of dust-covered 15306. Sample is 7 cm across. S71-43064 and 067. Introduction 15306 is a regolith
15306 134.2 grams 15315 35.6 g 15324 32.3 g 15325 57.8 g 15330 57.8 g Regolith Breccia Figure 1a,b: Photo of dust-covered 15306. Sample is 7 cm across. S71-43064 and 067. Introduction 15306 is a regolith
65055 Basaltic Impact Melt grams
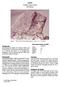 65055 Basaltic Impact Melt 500.8 grams Figure 1: Photo of 65055 showing large zap pit. Cube is 1 cm. S72-43869. Introduction According to the Apollo 16 Catalog by Ryder and Norman, 65055 is an aluminous,
65055 Basaltic Impact Melt 500.8 grams Figure 1: Photo of 65055 showing large zap pit. Cube is 1 cm. S72-43869. Introduction According to the Apollo 16 Catalog by Ryder and Norman, 65055 is an aluminous,
74235 Vitrophric Basalt 59 grams
 74235 Vitrophric Basalt 59 grams Figure 1: Photo of 74235. Cube is 1 cm. S73-16017 Figure 2: Map of station 4, Apollo 17. Transcript (while the LMP was discussing the core with CC, the CDR made a discovery)
74235 Vitrophric Basalt 59 grams Figure 1: Photo of 74235. Cube is 1 cm. S73-16017 Figure 2: Map of station 4, Apollo 17. Transcript (while the LMP was discussing the core with CC, the CDR made a discovery)
15595 and Vuggy Vitrophyric Pigeonite Basalt and grams
 and 15596 Vuggy Vitrophyric Pigeonite Basalt 237.6 and 224.8 grams Figure 1: Photo of. Sample is about 9 cm across. NASA S71-44491. Figure 2: Photo of 15596 showing surface that was exposed to micrometeorites.
and 15596 Vuggy Vitrophyric Pigeonite Basalt 237.6 and 224.8 grams Figure 1: Photo of. Sample is about 9 cm across. NASA S71-44491. Figure 2: Photo of 15596 showing surface that was exposed to micrometeorites.
,1,2,3,4,9, Impact Melt Breccia 65.4 grams
 63335 Impact Melt Breccia 65.4 grams,1,2,3,4,9,10,8 Figure 2: 63335,6. Cube is 1 cm. S75-33389.,7 Introduction 63335 is a sample chipped off of Shadow Rock (Ulrich 1973). It was collected as several fragments
63335 Impact Melt Breccia 65.4 grams,1,2,3,4,9,10,8 Figure 2: 63335,6. Cube is 1 cm. S75-33389.,7 Introduction 63335 is a sample chipped off of Shadow Rock (Ulrich 1973). It was collected as several fragments
12052 Pigeonite Basalt 1866 grams
 12052 Pigeonite Basalt 1866 grams Revised Figure 1: Photo of broken side of 12052 showing vugs. Sample is 6.5 cm high. NASA # S70-44633. Summary of Age Data for 12052 Ar/Ar Rb/Sr Nyquist 1977 (recalculated)
12052 Pigeonite Basalt 1866 grams Revised Figure 1: Photo of broken side of 12052 showing vugs. Sample is 6.5 cm high. NASA # S70-44633. Summary of Age Data for 12052 Ar/Ar Rb/Sr Nyquist 1977 (recalculated)
grams grams Regolith Breccia
 10019 297 grams 10066 60 grams Regolith Breccia Figure 1: Photo of 10019,1. Cube is 1 inch and scale is in cm. NASA S76-23354. Introduction Kramer et al. (1077) reported that 10019 and 10066 appeared to
10019 297 grams 10066 60 grams Regolith Breccia Figure 1: Photo of 10019,1. Cube is 1 inch and scale is in cm. NASA S76-23354. Introduction Kramer et al. (1077) reported that 10019 and 10066 appeared to
grams grams Crystalline matrix Breccia
 14069 24.87 grams 14070-36.56 grams Crystalline matrix Breccia Figure 1: Photo of 14069,0. Scale in cm and mm. S78-28808. 14070 Figure 2: Photo of 14070. Scale is in cm. S71-22168. Introduction 14069 and
14069 24.87 grams 14070-36.56 grams Crystalline matrix Breccia Figure 1: Photo of 14069,0. Scale in cm and mm. S78-28808. 14070 Figure 2: Photo of 14070. Scale is in cm. S71-22168. Introduction 14069 and
grams grams Poikilitic Impact Melt Breccia
 64567 13.8 grams 64569-14.3 grams Poikilitic Impact Melt Breccia Figure 1: Photo of 64567. Scale in mm. S72-55386 Mineralogical Mode by Simonds et al. (1973) 64567 64569 Plagioclase 69% 57 Pyroxene 10
64567 13.8 grams 64569-14.3 grams Poikilitic Impact Melt Breccia Figure 1: Photo of 64567. Scale in mm. S72-55386 Mineralogical Mode by Simonds et al. (1973) 64567 64569 Plagioclase 69% 57 Pyroxene 10
DRAFT Coarse-fines 569 grams
 10085 Coarse-fines 569 grams DRAFT Figure 1: Selected coarse fines from 10085. Scale is in mm. Photo from Wood et al. 1969. Introduction!0085 and 10084 were created during the Apollo 11 preliminary examination
10085 Coarse-fines 569 grams DRAFT Figure 1: Selected coarse fines from 10085. Scale is in mm. Photo from Wood et al. 1969. Introduction!0085 and 10084 were created during the Apollo 11 preliminary examination
14041, and Regolith Breccia 166.3, and 65.2 grams
 14041, and 14045 Regolith Breccia 166.3, 103.2 and 65.2 grams Figure 1a: Sample 14041-14045 on lunar surface. MET and LM in distance. AS14-68-9409. Introduction Samples 14041 14046 are fragments from a
14041, and 14045 Regolith Breccia 166.3, 103.2 and 65.2 grams Figure 1a: Sample 14041-14045 on lunar surface. MET and LM in distance. AS14-68-9409. Introduction Samples 14041 14046 are fragments from a
72335 Impact melt Breccia grams
 Impact melt Breccia 108.9 grams Figure 1: Location of on boulder #2 on landslide off of South Massif. Boulder is ~ 2-3 meters high. AS17-137-20918. Transcript Hey, that s a different rock, Gene (station
Impact melt Breccia 108.9 grams Figure 1: Location of on boulder #2 on landslide off of South Massif. Boulder is ~ 2-3 meters high. AS17-137-20918. Transcript Hey, that s a different rock, Gene (station
70035 Ilmenite Basalt 5765 grams
 Ilmenite Basalt 5765 grams Figure 1: Photograph of top surface of illustrating vugs and vesicles. Note the smooth, rounded surface shaped by micrometeorite bombardment. Small cube is 1 cm. NASA photo #
Ilmenite Basalt 5765 grams Figure 1: Photograph of top surface of illustrating vugs and vesicles. Note the smooth, rounded surface shaped by micrometeorite bombardment. Small cube is 1 cm. NASA photo #
72320 Partially Shadowed Soil (portion frozen) grams. boulder # 2 station 2 South Massif
 Partially Shadowed Soil (portion ) 106.31 grams Nansen Crater boulder # 2 station 2 South Massif Figure 1: Location of soil sample in shadow of boulder 2, at station 2, Apollo 17. NASA photo #AS17-137-20925.
Partially Shadowed Soil (portion ) 106.31 grams Nansen Crater boulder # 2 station 2 South Massif Figure 1: Location of soil sample in shadow of boulder 2, at station 2, Apollo 17. NASA photo #AS17-137-20925.
15009 Single Drive Tube Station 6
 15009 Single Drive Tube Station 6 Figure 2: Location of soil samples, trench and drive tube at station 6, Apollo 15. Figure 1: Photo of drive tube 15009 driven in all the way. AS15-86-11565. Introduction
15009 Single Drive Tube Station 6 Figure 2: Location of soil samples, trench and drive tube at station 6, Apollo 15. Figure 1: Photo of drive tube 15009 driven in all the way. AS15-86-11565. Introduction
14315 Unusual Regolith Breccia 115 grams
 Unusual Regolith Breccia 115 grams Figure 1: Photo of,0 after chipping and dusting. Sample is 5 cm across. NASA S86-36340. Introduction was collected as a grab sample from the North Boulder Field (station
Unusual Regolith Breccia 115 grams Figure 1: Photo of,0 after chipping and dusting. Sample is 5 cm across. NASA S86-36340. Introduction was collected as a grab sample from the North Boulder Field (station
DRAFT Cataclastic Dunite grams
 72415-72418 Cataclastic Dunite 58.74 grams DRAFT Figure 1: Boulder 3 at Station 2, with large dunite clast (72415-72418). The bright spot is the location of samples. A sample of the boulder matrix (72435)
72415-72418 Cataclastic Dunite 58.74 grams DRAFT Figure 1: Boulder 3 at Station 2, with large dunite clast (72415-72418). The bright spot is the location of samples. A sample of the boulder matrix (72435)
14049 Regolith Breccia grams
 Regolith Breccia 200.1 grams Figure 1: Photo of. Edge of cube is 1 inch. NASA S71-29143. Figure 2: Map of Apollo 14 showing traverse for EVA 2. Introduction The Apollo 14 regolith breccias (vitric matrix
Regolith Breccia 200.1 grams Figure 1: Photo of. Edge of cube is 1 inch. NASA S71-29143. Figure 2: Map of Apollo 14 showing traverse for EVA 2. Introduction The Apollo 14 regolith breccias (vitric matrix
grams grams Double Drive tube 61 cm
 68002 583.5 grams 68001 840.7 grams Double Drive tube 61 cm Figure 1a: Surface photo for double drive tube 68001-2. AS16-108-17684. Introduction Station 8 was the closest to South Ray Crater so premission
68002 583.5 grams 68001 840.7 grams Double Drive tube 61 cm Figure 1a: Surface photo for double drive tube 68001-2. AS16-108-17684. Introduction Station 8 was the closest to South Ray Crater so premission
62255 Anorthosite with melt 1239 grams
 62255 Anorthosite with melt 1239 grams Figure 1: Photo of 62255 showing glass splash on anorthosite. Cube is 1 cm. NASA S # S72-38309. Introduction Lunar sample 62255 is significant because it is largely
62255 Anorthosite with melt 1239 grams Figure 1: Photo of 62255 showing glass splash on anorthosite. Cube is 1 cm. NASA S # S72-38309. Introduction Lunar sample 62255 is significant because it is largely
61015 Dimict Breccia 1789 grams
 61015 Dimict Breccia 1789 grams Figure 1: Photo of 61015. NASA # S72-37758. Scale and cube in cm. Note the zap pits on the T1 surface and the residual black glass coating on the sides. Introduction Breccia
61015 Dimict Breccia 1789 grams Figure 1: Photo of 61015. NASA # S72-37758. Scale and cube in cm. Note the zap pits on the T1 surface and the residual black glass coating on the sides. Introduction Breccia
Ilmenite Basalt grams
 Ilmenite Basalt 737.6 grams Figure 1: Picute of 5 meter boulder on rim of Shorty Crater showing where was collected. Trench in forground is location of orange soil sample 74220. Photo number AS17-137-20990
Ilmenite Basalt 737.6 grams Figure 1: Picute of 5 meter boulder on rim of Shorty Crater showing where was collected. Trench in forground is location of orange soil sample 74220. Photo number AS17-137-20990
IV. Governador Valadares clinopyroxenite, 158 grams find
 IV. Governador Valadares clinopyroxenite, 158 grams find Figure IV-1. Photograph of Governador Valadares (158 grams) from Dr. Fernanda Ferrucci via Dr. Giuseppe Cavarretta. Photo taken by L. Spinozzi.
IV. Governador Valadares clinopyroxenite, 158 grams find Figure IV-1. Photograph of Governador Valadares (158 grams) from Dr. Fernanda Ferrucci via Dr. Giuseppe Cavarretta. Photo taken by L. Spinozzi.
12013 Breccia with granite 82.3 grams
 12013 Breccia with granite 82.3 grams Figure 1: Close-up photo of 12013,11. Field of view is 4.5 cm. NASA# S70-43636 Introduction 12013 is a unique lunar sample, but it did not appear to be remarkable
12013 Breccia with granite 82.3 grams Figure 1: Close-up photo of 12013,11. Field of view is 4.5 cm. NASA# S70-43636 Introduction 12013 is a unique lunar sample, but it did not appear to be remarkable
15058 Pigeonite Basalt 2672 grams
 15058 Pigeonite Basalt 2672 grams Figure 1: Photo of mare basalt 15058 (before dusting). NASA S71-44205. Sample is shaped like a brick about 6 inches long and 3 inches high. Note: see also figures 17 and
15058 Pigeonite Basalt 2672 grams Figure 1: Photo of mare basalt 15058 (before dusting). NASA S71-44205. Sample is shaped like a brick about 6 inches long and 3 inches high. Note: see also figures 17 and
10020 Ilmenite Basalt (low K) 425 grams
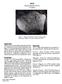 10020 Ilmenite Basalt (low K) 425 grams Figure 1: Photo of 10020 taken in the F-201 during initial processing. Sample is 6 cm across. NASA S-69-45261. Introduction 10020 is a low-k variety of fine-grained
10020 Ilmenite Basalt (low K) 425 grams Figure 1: Photo of 10020 taken in the F-201 during initial processing. Sample is 6 cm across. NASA S-69-45261. Introduction 10020 is a low-k variety of fine-grained
Element Cube Project (x2)
 Element Cube Project (x2) Background: As a class, we will construct a three dimensional periodic table by each student selecting two elements in which you will need to create an element cube. Helpful Links
Element Cube Project (x2) Background: As a class, we will construct a three dimensional periodic table by each student selecting two elements in which you will need to create an element cube. Helpful Links
DRAFT Regolith Breccia 155 grams
 12034 Regolith Breccia 155 grams DRAFT Figure 1: Lunar sample 12034 after first saw cut in 1970. NASA S75-34235. Cube is 1 cm. Introduction 12034 was collected from the bottom of the same trench as soil
12034 Regolith Breccia 155 grams DRAFT Figure 1: Lunar sample 12034 after first saw cut in 1970. NASA S75-34235. Cube is 1 cm. Introduction 12034 was collected from the bottom of the same trench as soil
10003 Ilmenite Basalt (low K) 213 grams
 10003 Ilmenite Basalt (low K) 213 grams Figure 1: Photo of 10003,25. Sample is 5 cm across. NASA S76-25545. Introduction Lunar sample 10003 is a low-k, high-ti basalt (figure 1). It is about 3.9 b.y. old,
10003 Ilmenite Basalt (low K) 213 grams Figure 1: Photo of 10003,25. Sample is 5 cm across. NASA S76-25545. Introduction Lunar sample 10003 is a low-k, high-ti basalt (figure 1). It is about 3.9 b.y. old,
67016 Feldspathic Fragmental Breccia 4262 grams
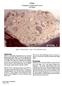 67016 Feldspathic Fragmental Breccia 4262 grams Figure 1: Photo of 67016,1. Cube is 1 inch. NASA # S81-26050. Introduction 67016 is a feldspathic fragmental breccia with both light and dark clasts (figures
67016 Feldspathic Fragmental Breccia 4262 grams Figure 1: Photo of 67016,1. Cube is 1 inch. NASA # S81-26050. Introduction 67016 is a feldspathic fragmental breccia with both light and dark clasts (figures
76215 Vesicular Micropoikilitic Impact Melt Breccia 644 grams
 76215 Vesicular Micropoikilitic Impact Melt Breccia 644 grams 76215 76215 Figure 1: Photo of station 6 boulder with location of 76215 indicated. AS17-140-21421 Figure 2: Photo of 76215. Sample is about
76215 Vesicular Micropoikilitic Impact Melt Breccia 644 grams 76215 76215 Figure 1: Photo of station 6 boulder with location of 76215 indicated. AS17-140-21421 Figure 2: Photo of 76215. Sample is about
65015 Poikilitic Impact Melt Breccia 1802 grams
 65015 Poikilitic Impact Melt Breccia 1802 grams Figure 1: Photos of 65015 showing rounded exposed surface with numerous zap pits and angular portion without zap pits protected by burial in soil. NASA photos
65015 Poikilitic Impact Melt Breccia 1802 grams Figure 1: Photos of 65015 showing rounded exposed surface with numerous zap pits and angular portion without zap pits protected by burial in soil. NASA photos
DRAFT. fillet grams grams grams (frozen) grams Vesicular Ilmenite Basalt
 71055 669.6 grams 71035 141.8 grams 71036 118.4 grams (frozen) 71037 14.39 grams Vesicular Ilmenite Basalt DRAFT 71040 71060 71055 71035 71036 71037 fillet Figure 1: This boulder of vesicular basalt from
71055 669.6 grams 71035 141.8 grams 71036 118.4 grams (frozen) 71037 14.39 grams Vesicular Ilmenite Basalt DRAFT 71040 71060 71055 71035 71036 71037 fillet Figure 1: This boulder of vesicular basalt from
Composition of the Earth and its reservoirs: Geochemical observables
 Composition of the Earth and its reservoirs: Geochemical observables Cin-Ty A. Lee Rice University MYRES-I 2004 The Earth is dynamic and heterogeneous Atmosphere Midocean Ridge Plume Ocean Crust Oceanic
Composition of the Earth and its reservoirs: Geochemical observables Cin-Ty A. Lee Rice University MYRES-I 2004 The Earth is dynamic and heterogeneous Atmosphere Midocean Ridge Plume Ocean Crust Oceanic
Solutions and Ions. Pure Substances
 Class #4 Solutions and Ions CHEM 107 L.S. Brown Texas A&M University Pure Substances Pure substance: described completely by a single chemical formula Fixed composition 1 Mixtures Combination of 2 or more
Class #4 Solutions and Ions CHEM 107 L.S. Brown Texas A&M University Pure Substances Pure substance: described completely by a single chemical formula Fixed composition 1 Mixtures Combination of 2 or more
Radiometric Dating (tap anywhere)
 Radiometric Dating (tap anywhere) Protons Neutrons Electrons Elements on the periodic table are STABLE Elements can have radioactive versions of itself called ISOTOPES!! Page 1 in your ESRT has your list!
Radiometric Dating (tap anywhere) Protons Neutrons Electrons Elements on the periodic table are STABLE Elements can have radioactive versions of itself called ISOTOPES!! Page 1 in your ESRT has your list!
Nucleus. Electron Cloud
 Atomic Structure I. Picture of an Atom Nucleus Electron Cloud II. Subatomic particles Particle Symbol Charge Relative Mass (amu) protons p + +1 1.0073 neutrons n 0 1.0087 electrons e - -1 0.00054858 Compare
Atomic Structure I. Picture of an Atom Nucleus Electron Cloud II. Subatomic particles Particle Symbol Charge Relative Mass (amu) protons p + +1 1.0073 neutrons n 0 1.0087 electrons e - -1 0.00054858 Compare
5 questions, 3 points each, 15 points total possible. 26 Fe Cu Ni Co Pd Ag Ru 101.
 Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
Atoms and the Periodic Table
 Atoms and the Periodic Table Parts of the Atom Proton Found in the nucleus Number of protons defines the element Charge +1, mass 1 Parts of the Atom Neutron Found in the nucleus Stabilizes the nucleus
Atoms and the Periodic Table Parts of the Atom Proton Found in the nucleus Number of protons defines the element Charge +1, mass 1 Parts of the Atom Neutron Found in the nucleus Stabilizes the nucleus
Summary of test results for Daya Bay rock samples. by Patrick Dobson Celia Tiemi Onishi Seiji Nakagawa
 Summary of test results for Daya Bay rock samples by Patrick Dobson Celia Tiemi Onishi Seiji Nakagawa October 2004 Summary A series of analytical tests were conducted on a suite of granitic rock samples
Summary of test results for Daya Bay rock samples by Patrick Dobson Celia Tiemi Onishi Seiji Nakagawa October 2004 Summary A series of analytical tests were conducted on a suite of granitic rock samples
EMMR25 Mineralogy: Ol + opx + chlorite + cpx + amphibole + serpentine + opaque
 GSA Data Repository 2017365 Marshall et al., 2017, The role of serpentinite derived fluids in metasomatism of the Colorado Plateau (USA) lithospheric mantle: Geology, https://doi.org/10.1130/g39444.1 Appendix
GSA Data Repository 2017365 Marshall et al., 2017, The role of serpentinite derived fluids in metasomatism of the Colorado Plateau (USA) lithospheric mantle: Geology, https://doi.org/10.1130/g39444.1 Appendix
12042 Soil 255 grams
 Soil 255 grams Figure 1: Photo of location of (Halo Crater). AS12-48-7072 Introduction are fines collected in documented bag 12. They were from the outer flank of Surveyor Crater (figure 2). Figure 2:
Soil 255 grams Figure 1: Photo of location of (Halo Crater). AS12-48-7072 Introduction are fines collected in documented bag 12. They were from the outer flank of Surveyor Crater (figure 2). Figure 2:
7) Applications of Nuclear Radiation in Science and Technique (1) Analytical applications (Radiometric titration)
 7) Applications of Nuclear Radiation in Science and Technique (1) (Radiometric titration) The radioactive material is indicator Precipitation reactions Complex formation reactions Principle of a precipitation
7) Applications of Nuclear Radiation in Science and Technique (1) (Radiometric titration) The radioactive material is indicator Precipitation reactions Complex formation reactions Principle of a precipitation
The Periodic Table of Elements
 The Periodic Table of Elements 8 Uuo Uus Uuh (9) Uup (88) Uuq (89) Uut (8) Uub (8) Rg () 0 Ds (9) 09 Mt (8) 08 Hs (9) 0 h () 0 Sg () 0 Db () 0 Rf () 0 Lr () 88 Ra () 8 Fr () 8 Rn () 8 At (0) 8 Po (09)
The Periodic Table of Elements 8 Uuo Uus Uuh (9) Uup (88) Uuq (89) Uut (8) Uub (8) Rg () 0 Ds (9) 09 Mt (8) 08 Hs (9) 0 h () 0 Sg () 0 Db () 0 Rf () 0 Lr () 88 Ra () 8 Fr () 8 Rn () 8 At (0) 8 Po (09)
CHEM 10113, Quiz 5 October 26, 2011
 CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
The Periodic Table. Periodic Properties. Can you explain this graph? Valence Electrons. Valence Electrons. Paramagnetism
 Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Chemistry 431 Practice Final Exam Fall Hours
 Chemistry 431 Practice Final Exam Fall 2018 3 Hours R =8.3144 J mol 1 K 1 R=.0821 L atm mol 1 K 1 R=.08314 L bar mol 1 K 1 k=1.381 10 23 J molecule 1 K 1 h=6.626 10 34 Js N A = 6.022 10 23 molecules mol
Chemistry 431 Practice Final Exam Fall 2018 3 Hours R =8.3144 J mol 1 K 1 R=.0821 L atm mol 1 K 1 R=.08314 L bar mol 1 K 1 k=1.381 10 23 J molecule 1 K 1 h=6.626 10 34 Js N A = 6.022 10 23 molecules mol
Worked Example of Batch Melting: Rb and Sr
 Worked Example of Batch Melting: Rb and Sr Basalt with the mode: Table 9.2. Conversion from mode to weight percent Mineral Mode Density Wt prop Wt% ol 15 3.6 54 0.18 cpx 33 3.4 112.2 0.37 plag 51 2.7 137.7
Worked Example of Batch Melting: Rb and Sr Basalt with the mode: Table 9.2. Conversion from mode to weight percent Mineral Mode Density Wt prop Wt% ol 15 3.6 54 0.18 cpx 33 3.4 112.2 0.37 plag 51 2.7 137.7
CHEM 130 Exp. 8: Molecular Models
 CHEM 130 Exp. 8: Molecular Models In this lab, we will learn and practice predicting molecular structures from molecular formulas. The Periodic Table of the Elements IA 1 H IIA IIIA IVA VA VIA VIIA 3 5
CHEM 130 Exp. 8: Molecular Models In this lab, we will learn and practice predicting molecular structures from molecular formulas. The Periodic Table of the Elements IA 1 H IIA IIIA IVA VA VIA VIIA 3 5
Part 2. Multiple choice (use answer card). 90 pts. total. 3 pts. each.
 1 Exam I CHEM 1303.001 Name (print legibly) Seat no. On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Part 1. Nomenclature. 10 pts. total. 2 pts. each. Fill in
1 Exam I CHEM 1303.001 Name (print legibly) Seat no. On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Part 1. Nomenclature. 10 pts. total. 2 pts. each. Fill in
Why all the repeating Why all the repeating Why all the repeating Why all the repeating
 Why all the repeating Why all the repeating Why all the repeating Why all the repeating Patterns What Patterns have you observed in your life? Where to Get Help If you don t understand concepts in chapter
Why all the repeating Why all the repeating Why all the repeating Why all the repeating Patterns What Patterns have you observed in your life? Where to Get Help If you don t understand concepts in chapter
Chapter 3: Stoichiometry
 Chapter 3: Stoichiometry Chem 6A Michael J. Sailor, UC San Diego 1 Announcements: Thursday (Sep 29) quiz: Bring student ID or we cannot accept your quiz! No notes, no calculators Covers chapters 1 and
Chapter 3: Stoichiometry Chem 6A Michael J. Sailor, UC San Diego 1 Announcements: Thursday (Sep 29) quiz: Bring student ID or we cannot accept your quiz! No notes, no calculators Covers chapters 1 and
The Periodic Table of the Elements
 The Periodic Table of the Elements All matter is composed of elements. All of the elements are composed of atoms. An atom is the smallest part of an element which still retains the properties of that element.
The Periodic Table of the Elements All matter is composed of elements. All of the elements are composed of atoms. An atom is the smallest part of an element which still retains the properties of that element.
Circle the letters only. NO ANSWERS in the Columns! (3 points each)
 Chemistry 1304.001 Name (please print) Exam 4 (100 points) April 12, 2017 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Chemistry 1304.001 Name (please print) Exam 4 (100 points) April 12, 2017 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
(please print) (1) (18) H IIA IIIA IVA VA VIA VIIA He (2) (13) (14) (15) (16) (17)
 CHEM 10113, Quiz 3 September 28, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
CHEM 10113, Quiz 3 September 28, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
Last 4 Digits of USC ID:
 Chemistry 05 B Practice Exam Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 8 2 4 3 9 4 0
Chemistry 05 B Practice Exam Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 8 2 4 3 9 4 0
8. Relax and do well.
 CHEM 1515 Exam II John II. Gelder October 14, 1993 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last two pages include a periodic table, a
CHEM 1515 Exam II John II. Gelder October 14, 1993 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last two pages include a periodic table, a
Made the FIRST periodic table
 Made the FIRST periodic table 1869 Mendeleev organized the periodic table based on the similar properties and relativities of certain elements Later, Henri Moseley organized the elements by increasing
Made the FIRST periodic table 1869 Mendeleev organized the periodic table based on the similar properties and relativities of certain elements Later, Henri Moseley organized the elements by increasing
Ch. 9 NOTES ~ Chemical Bonding NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics.
 Ch. 9 NOTES ~ Chemical Bonding NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. I. Review: Comparison of ionic and molecular compounds Molecular compounds Ionic
Ch. 9 NOTES ~ Chemical Bonding NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. I. Review: Comparison of ionic and molecular compounds Molecular compounds Ionic
INSTRUCTIONS: Exam III. November 10, 1999 Lab Section
 CHEM 1215 Exam III John III. Gelder November 10, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
CHEM 1215 Exam III John III. Gelder November 10, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
Earth Materials I Crystal Structures
 Earth Materials I Crystal Structures Isotopes same atomic number, different numbers of neutrons, different atomic mass. Ta ble 1-1. Su mmar y of quantu m num bers Name Symbol Values Principal n 1, 2,
Earth Materials I Crystal Structures Isotopes same atomic number, different numbers of neutrons, different atomic mass. Ta ble 1-1. Su mmar y of quantu m num bers Name Symbol Values Principal n 1, 2,
CHEM 108 (Spring-2008) Exam. 3 (105 pts)
 CHEM 08 (Spring-008) Exam. (05 pts) Name: --------------------------------------------------------------------------, CLID # -------------------------------- LAST NAME, First (Circle the alphabet segment
CHEM 08 (Spring-008) Exam. (05 pts) Name: --------------------------------------------------------------------------, CLID # -------------------------------- LAST NAME, First (Circle the alphabet segment
610B Final Exam Cover Page
 1 st Letter of Last Name NAME: 610B Final Exam Cover Page No notes or calculators of any sort allowed. You have 3 hours to complete the exam. CHEM 610B, 50995 Final Exam Fall 2003 Instructor: Dr. Brian
1 st Letter of Last Name NAME: 610B Final Exam Cover Page No notes or calculators of any sort allowed. You have 3 hours to complete the exam. CHEM 610B, 50995 Final Exam Fall 2003 Instructor: Dr. Brian
M10/4/CHEMI/SPM/ENG/TZ2/XX+ CHEMISTRY. Wednesday 12 May 2010 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES
 M10/4/CHEMI/SPM/ENG/TZ/XX+ 106116 CHEMISTRY standard level Paper 1 Wednesday 1 May 010 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so. Answer
M10/4/CHEMI/SPM/ENG/TZ/XX+ 106116 CHEMISTRY standard level Paper 1 Wednesday 1 May 010 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so. Answer
Chem Exam 1. September 26, Dr. Susan E. Bates. Name 9:00 OR 10:00
 Chem 1711 Exam 1 September 26, 2013 Dr. Susan E. Bates Name 9:00 OR 10:00 N A = 6.022 x 10 23 mol 1 I A II A III B IV B V B VI B VII B VIII I B II B III A IV A V A VI A VII A inert gases 1 H 1.008 3 Li
Chem 1711 Exam 1 September 26, 2013 Dr. Susan E. Bates Name 9:00 OR 10:00 N A = 6.022 x 10 23 mol 1 I A II A III B IV B V B VI B VII B VIII I B II B III A IV A V A VI A VII A inert gases 1 H 1.008 3 Li
Circle the letters only. NO ANSWERS in the Columns!
 Chemistry 1304.001 Name (please print) Exam 5 (100 points) April 18, 2018 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Chemistry 1304.001 Name (please print) Exam 5 (100 points) April 18, 2018 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Secondary Support Pack. be introduced to some of the different elements within the periodic table;
 Secondary Support Pack INTRODUCTION The periodic table of the elements is central to chemistry as we know it today and the study of it is a key part of every student s chemical education. By playing the
Secondary Support Pack INTRODUCTION The periodic table of the elements is central to chemistry as we know it today and the study of it is a key part of every student s chemical education. By playing the
Cosmic Building Blocks: From What is Earth Made?
 Cosmic Building Blocks: From What is Earth Made? The Sun constitutes 99.87% of the mass of the Solar system. Earth is big and important, so its composition should be similar to that of the average Solar
Cosmic Building Blocks: From What is Earth Made? The Sun constitutes 99.87% of the mass of the Solar system. Earth is big and important, so its composition should be similar to that of the average Solar
Advanced Chemistry. Mrs. Klingaman. Chapter 5: Name:
 Advanced Chemistry Mrs. Klingaman Chapter 5: The Periodic Law Name: _ Mods: Chapter 5: The Periodic Law Reading Guide 5.1 History of the Periodic Table (pgs. 125-129) 1) What did Dimitri Mendeleev notice
Advanced Chemistry Mrs. Klingaman Chapter 5: The Periodic Law Name: _ Mods: Chapter 5: The Periodic Law Reading Guide 5.1 History of the Periodic Table (pgs. 125-129) 1) What did Dimitri Mendeleev notice
8. Relax and do well.
 CHEM 1215 Exam III John III. Gelder November 11, 1998 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
CHEM 1215 Exam III John III. Gelder November 11, 1998 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
February 20, Joe Cerniglia The International Group for Historic Aircraft Recovery (TIGHAR) Job Number: S0CHG688. Dear Joe:
 February 20, 2012 Joe Cerniglia The International Group for Historic Aircraft Recovery (TIGHAR) Subject: ICP-MS Report Job Number: S0CHG688 Dear Joe: Please find enclosed the procedure report for the analysis
February 20, 2012 Joe Cerniglia The International Group for Historic Aircraft Recovery (TIGHAR) Subject: ICP-MS Report Job Number: S0CHG688 Dear Joe: Please find enclosed the procedure report for the analysis
(C) Pavel Sedach and Prep101 1
 (C) Pavel Sedach and Prep101 1 (C) Pavel Sedach and Prep101 1 (C) Pavel Sedach and Prep101 2 (C) Pavel Sedach and Prep101 2 (C) Pavel Sedach and Prep101 3 (C) Pavel Sedach and Prep101 3 (C) Pavel Sedach
(C) Pavel Sedach and Prep101 1 (C) Pavel Sedach and Prep101 1 (C) Pavel Sedach and Prep101 2 (C) Pavel Sedach and Prep101 2 (C) Pavel Sedach and Prep101 3 (C) Pavel Sedach and Prep101 3 (C) Pavel Sedach
CLASS TEST GRADE 11. PHYSICAL SCIENCES: CHEMISTRY Test 4: Matter and materials 1
 CLASS TEST GRADE PHYSICAL SCIENCES: CHEMISTRY Test 4: Matter and materials MARKS: 45 TIME: hour INSTRUCTIONS AND INFORMATION. Answer ALL the questions. 2. You may use non-programmable calculators. 3. You
CLASS TEST GRADE PHYSICAL SCIENCES: CHEMISTRY Test 4: Matter and materials MARKS: 45 TIME: hour INSTRUCTIONS AND INFORMATION. Answer ALL the questions. 2. You may use non-programmable calculators. 3. You
8. Relax and do well.
 CHEM 1314.03 Exam I John I. Gelder September 25, 1997 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
CHEM 1314.03 Exam I John I. Gelder September 25, 1997 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
PERIODIC TABLE OF THE ELEMENTS
 Useful Constants and equations: K = o C + 273 Avogadro's number = 6.022 x 10 23 d = density = mass/volume R H = 2.178 x 10-18 J c = E = h = hc/ h = 6.626 x 10-34 J s c = 2.998 x 10 8 m/s E n = -R H Z 2
Useful Constants and equations: K = o C + 273 Avogadro's number = 6.022 x 10 23 d = density = mass/volume R H = 2.178 x 10-18 J c = E = h = hc/ h = 6.626 x 10-34 J s c = 2.998 x 10 8 m/s E n = -R H Z 2
HANDOUT SET GENERAL CHEMISTRY II
 HANDOUT SET GENERAL CHEMISTRY II Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
HANDOUT SET GENERAL CHEMISTRY II Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
NAME: FIRST EXAMINATION
 1 Chemistry 64 Winter 1994 NAME: FIRST EXAMINATION THIS EXAMINATION IS WORTH 100 POINTS AND CONTAINS 4 (FOUR) QUESTIONS THEY ARE NOT EQUALLY WEIGHTED! YOU SHOULD ATTEMPT ALL QUESTIONS AND ALLOCATE YOUR
1 Chemistry 64 Winter 1994 NAME: FIRST EXAMINATION THIS EXAMINATION IS WORTH 100 POINTS AND CONTAINS 4 (FOUR) QUESTIONS THEY ARE NOT EQUALLY WEIGHTED! YOU SHOULD ATTEMPT ALL QUESTIONS AND ALLOCATE YOUR
Chemistry 2 Exam Roane State Academic Festival. Name (print neatly) School
 Name (print neatly) School There are fifteen question on this exam. Each question is weighted equally. n the answer sheet, write your name in the space provided and your answers in the blanks provided.
Name (print neatly) School There are fifteen question on this exam. Each question is weighted equally. n the answer sheet, write your name in the space provided and your answers in the blanks provided.
8. Relax and do well.
 CHEM 1014 Exam I John I. Gelder September 16, 1999 Name TA's Name Lab Section Please sign your name below to give permission to post your course scores on homework, laboratories and exams. If you do not
CHEM 1014 Exam I John I. Gelder September 16, 1999 Name TA's Name Lab Section Please sign your name below to give permission to post your course scores on homework, laboratories and exams. If you do not
CHM 101 PRACTICE TEST 1 Page 1 of 4
 CHM 101 PRACTICE TEST 1 Page 1 of 4 Please show calculations (stuffed equations) on all mathematical problems!! On the actual test, "naked answers, with no work shown, will receive no credit even if correct.
CHM 101 PRACTICE TEST 1 Page 1 of 4 Please show calculations (stuffed equations) on all mathematical problems!! On the actual test, "naked answers, with no work shown, will receive no credit even if correct.
Spin Cut-off Parameter of Nuclear Level Density and Effective Moment of Inertia
 Commun. Theor. Phys. (Beijing, China) 43 (005) pp. 709 718 c International Academic Publishers Vol. 43, No. 4, April 15, 005 Spin Cut-off Parameter of Nuclear Level Density and Effective Moment of Inertia
Commun. Theor. Phys. (Beijing, China) 43 (005) pp. 709 718 c International Academic Publishers Vol. 43, No. 4, April 15, 005 Spin Cut-off Parameter of Nuclear Level Density and Effective Moment of Inertia
Using the Periodic Table
 MATH SKILLS TRANSPARENCY WORKSHEET Using the Periodic Table 6 Use with Chapter 6, Section 6.2 1. Identify the number of valence electrons in each of the following elements. a. Ne e. O b. K f. Cl c. B g.
MATH SKILLS TRANSPARENCY WORKSHEET Using the Periodic Table 6 Use with Chapter 6, Section 6.2 1. Identify the number of valence electrons in each of the following elements. a. Ne e. O b. K f. Cl c. B g.
PHYSICAL SCIENCES GRADE : 10
 PHYSICAL SCIENCES GRADE : 0 TIME : hour TOTAL : 75 INSTRUCTIONS AND INFORMATION. Write your full name on your answer book in the appropriate place. 2. The question paper consists of SEVEN questions. Answer
PHYSICAL SCIENCES GRADE : 0 TIME : hour TOTAL : 75 INSTRUCTIONS AND INFORMATION. Write your full name on your answer book in the appropriate place. 2. The question paper consists of SEVEN questions. Answer
Guide to the Extended Step-Pyramid Periodic Table
 Guide to the Extended Step-Pyramid Periodic Table William B. Jensen Department of Chemistry University of Cincinnati Cincinnati, OH 452201-0172 The extended step-pyramid table recognizes that elements
Guide to the Extended Step-Pyramid Periodic Table William B. Jensen Department of Chemistry University of Cincinnati Cincinnati, OH 452201-0172 The extended step-pyramid table recognizes that elements
DO NOW: Retrieve your projects. We will be reviewing them again today. Textbook pg 23, answer questions 1-3. Use the section 1.2 to help you.
 DO NOW: Retrieve your projects. We will be reviewing them again today. Textbook pg, answer questions. Use the section. to help you. Chapter test is FRIDAY. The Periodic Table of Elements 8 Uuo Uus Uuh
DO NOW: Retrieve your projects. We will be reviewing them again today. Textbook pg, answer questions. Use the section. to help you. Chapter test is FRIDAY. The Periodic Table of Elements 8 Uuo Uus Uuh
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
70017 Ilmenite Basalt 2957 grams
 Ilmenite Basalt 2957 grams Figure 1: Photo of lunar basalt showing vugs and vesicles. Sample is about 10 cm across. Cube is 1 cm. NASA# S73-15723. Introduction Lunar sample is a vesicular, medium-grained,
Ilmenite Basalt 2957 grams Figure 1: Photo of lunar basalt showing vugs and vesicles. Sample is about 10 cm across. Cube is 1 cm. NASA# S73-15723. Introduction Lunar sample is a vesicular, medium-grained,
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
SCIENCE 1206 UNIT 2 CHEMISTRY. September 2017 November 2017
 SCIENCE 1206 UNIT 2 CHEMISTRY September 2017 November 2017 UNIT OUTLINE 1. Review of Grade 9 Terms & the Periodic Table Bohr diagrams Evidence for chemical reactions Chemical Tests 2. Naming & writing
SCIENCE 1206 UNIT 2 CHEMISTRY September 2017 November 2017 UNIT OUTLINE 1. Review of Grade 9 Terms & the Periodic Table Bohr diagrams Evidence for chemical reactions Chemical Tests 2. Naming & writing
6.3 Classifying Elements with the Periodic Table
 6.3 Classifying Elements with the Periodic Table The Periodic Table was developed by scientists to organize elements in such a way as to make sense of the growing information about their properties. The
6.3 Classifying Elements with the Periodic Table The Periodic Table was developed by scientists to organize elements in such a way as to make sense of the growing information about their properties. The
Atomic Emission Spectra. and. Flame Tests. Burlingame High School Chemistry
 Atomic Structure Atomic Emission Spectra and Flame Tests Flame Tests Sodium potassium lithium When electrons are excited they bump up to a higher energy level. As they bounce back down they release energy
Atomic Structure Atomic Emission Spectra and Flame Tests Flame Tests Sodium potassium lithium When electrons are excited they bump up to a higher energy level. As they bounce back down they release energy
K. 27 Co. 28 Ni. 29 Cu Rb. 46 Pd. 45 Rh. 47 Ag Cs Ir. 78 Pt.
 1 IA 1 H Hydrogen 1.01 Atomic number Element symbol Element name Atomic mass VIIIA 1 H 1.01 IIA IIIA IVA VA VIA VIIA 2 He 4.00 Metalloids 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 O 16.00 9 F
1 IA 1 H Hydrogen 1.01 Atomic number Element symbol Element name Atomic mass VIIIA 1 H 1.01 IIA IIIA IVA VA VIA VIIA 2 He 4.00 Metalloids 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 O 16.00 9 F
MANY ELECTRON ATOMS Chapter 15
 MANY ELECTRON ATOMS Chapter 15 Electron-Electron Repulsions (15.5-15.9) The hydrogen atom Schrödinger equation is exactly solvable yielding the wavefunctions and orbitals of chemistry. Howev er, the Schrödinger
MANY ELECTRON ATOMS Chapter 15 Electron-Electron Repulsions (15.5-15.9) The hydrogen atom Schrödinger equation is exactly solvable yielding the wavefunctions and orbitals of chemistry. Howev er, the Schrödinger
Chapter 12 The Atom & Periodic Table- part 2
 Chapter 12 The Atom & Periodic Table- part 2 Electrons found outside the nucleus; negatively charged Protons found in the nucleus; positive charge equal in magnitude to the electron s negative charge Neutrons
Chapter 12 The Atom & Periodic Table- part 2 Electrons found outside the nucleus; negatively charged Protons found in the nucleus; positive charge equal in magnitude to the electron s negative charge Neutrons
INSTRUCTIONS: CHEM Exam I. September 13, 1994 Lab Section
 CHEM 1314.05 Exam I John I. Gelder September 13, 1994 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
CHEM 1314.05 Exam I John I. Gelder September 13, 1994 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
60025 Ferroan Anorthosite 1836 grams
 60025 Ferroan Anorthosite 1836 grams Figure 1: 60025. NASA #S72-41586. Cube and scale are 1 cm. Note the thick black glass coating and numerous micrometeorite pits. Introduction Lunar sample 60025 is a
60025 Ferroan Anorthosite 1836 grams Figure 1: 60025. NASA #S72-41586. Cube and scale are 1 cm. Note the thick black glass coating and numerous micrometeorite pits. Introduction Lunar sample 60025 is a
Figure of rare earth elemental abundances removed due to copyright restrictions.
 Figure of rare earth elemental abundances removed due to copyright restrictions. See figure 3.1 on page 26 of Tolstikhin, Igor and Jan Kramers. The Evolution of Matter: From the Big Bang to the Present
Figure of rare earth elemental abundances removed due to copyright restrictions. See figure 3.1 on page 26 of Tolstikhin, Igor and Jan Kramers. The Evolution of Matter: From the Big Bang to the Present
[ ]:543.4(075.8) 35.20: ,..,..,.., : /... ;. 2-. ISBN , - [ ]:543.4(075.8) 35.20:34.
![[ ]:543.4(075.8) 35.20: ,..,..,.., : /... ;. 2-. ISBN , - [ ]:543.4(075.8) 35.20:34. [ ]:543.4(075.8) 35.20: ,..,..,.., : /... ;. 2-. ISBN , - [ ]:543.4(075.8) 35.20:34.](/thumbs/74/69911105.jpg) .. - 2-2009 [661.87.+661.88]:543.4(075.8) 35.20:34.2373-60..,..,..,..,.. -60 : /... ;. 2-. : -, 2008. 134. ISBN 5-98298-299-7 -., -,,. - «,, -, -», - 550800,, 240600 «-», -. [661.87.+661.88]:543.4(075.8)
.. - 2-2009 [661.87.+661.88]:543.4(075.8) 35.20:34.2373-60..,..,..,..,.. -60 : /... ;. 2-. : -, 2008. 134. ISBN 5-98298-299-7 -., -,,. - «,, -, -», - 550800,, 240600 «-», -. [661.87.+661.88]:543.4(075.8)
