DRAFT. fillet grams grams grams (frozen) grams Vesicular Ilmenite Basalt
|
|
|
- Claire Farmer
- 5 years ago
- Views:
Transcription
1 grams grams grams (frozen) grams Vesicular Ilmenite Basalt DRAFT fillet Figure 1: This boulder of vesicular basalt from a small crater on rim of Steno Crater had a significant fillet. It was found to have been exposed to cosmic rays and micrometeorite bombardment for ~110 m.y. Samples and 71035, and were chipped from the top knobs of this boulder. NASA surface photo AS
2 Figure 2: Three views of NASA photos S , 16164, Top is exterior surface.
3 Figure 3a, b: Thin section photos showing abundant, platy ilmenite in Dymek et al Di Hd En Fs Figure 4: Photomicrograph of thin section of (from Lofgren and Lofgren 1981). Scale bar is 1 mm. Figure 5: Thin section photo of 71055,68 showing unusual subvariolitic structure (by Neal and Taylor 1993). Field of view is 2.5 mm. Fo compiled by C Meyer Figure 6: Pyroxene and olivine composition of (from Dymek et al. 1975). Introduction Lunar basalts and 71035, and were all chipped from top of a small boulder (0.5 m) in small blocky crater (10 m) on rim of larger Steno Crater (figure 1). All samples are the same kind of high-ti basalt. The crystallization age (of 71055) was determined by Rb-Sr as 3.64 ± 0.09 b.y. The cosmic ray exposure age was found to be 110 m.y. Orientation of these samples was documented by photography (figure 35 in Wolfe et al. 1981). Thus, they have been in the Sun for 110 m.y. Sample was returned under vacuum and has been kept in a freezer since Although it has not been studied, it is the same lithology (basalt) as the other pieces of this boulder. Soil samples (259 g) and (506 g) and assorted small rock fragments were scooped from the Fa
4 Figure 7: Photo of showing exterior surface rounded by micrometeorite bombardment. NASA S Sample is 7 cm across. Mineralogical Mode for Brown et Dymek et Taylor et al al al Olivine Pyroxene Plagioclase Ilmenite Silica Figure 8: Photomicrograph of thin section 71035,30 showing ilmenite laths. Chip is about 0.8 cm. shadow area behind the boulder but this shadow would not have been permanent. They were returned in vacuum in ALSRC#1. Petrography Neal and Taylor (1993) summarize what is known about each of the samples of this basaltic boulder. Dymek et al. (1975) give a detailed petrologic description of and Warner et al. (1979) studied has not received attention, probably because it is another sample of the same material (it has been kept in a freezer since the mission).
5 Figure 9: Photo of 71036; exterior surface. NASA S Sample is 7 cm across. Figure 10: Photo of 71037; exterior surface. NASA S Sample is 2.5 cm across. Warner et al. (1979) describe as a fine-grained, high-ti basalt. Due to low trace element content they grouped with Type B Apollo 17 basalts. These samples are all very vesicular; up to 30% (figures 1, 2, 7, 9, 10). Vugs extend up to 12 mm. The texture is described as plagioclase-poikilitic (Neal and Taylor 1993). Average grain size is 1 2 mm with seriate grain size distribution. Mineralogy Olivine: Olivine in occurs as rounded cores of larger pyroxene and is Fo (Dymek et al. 1975). Pyroxene: According to Dymek et al. (1975), pyroxene in is highly complex, both chemically and texturally. The largest grains (up to 2 mm), typically composite, are composed of pale-pink (Al- and Ti-poor) to dark-pink (Al- and Ti-rich) regions arranged in parallel bands, in a radiating spehulitic pattern, or forming an hourglass structure. Pyroxene crystals are
6 Lunar Basalts A TiO A A15 Lunar Basalts A17 A MgO sample/ chondrite sample/ chondrite La Pr Sm Gd Dy Er Yb Ce Nd Eu Tb Ho Tm Lu ref A11 TiO A A12 MgO Figure 11: Chemical composition of and compared with other lunar basalts. found to have a wide range of chemical zoning (figure 6). Sung et al. (1974) found substantial Ti+3 in pyroxene. Plagioclase: Large grains of plagioclase (up to 1.5 mm) poikilitically enclose pyroxene (Dymek et al. 1975). Other plagioclase forms elongate sheaves intergrown with pyroxene (figure 5). Plagioclase composition is An Ilmenite: Elongated blades of ilmenite in have been studied by Dymek et al. (1975), von Englehardt (1979) and Muhich et al. (1990). Taylor et al. (1992) have shown how to separate the ilmenite. Chemistry Tables 1 4 and figures 12 and 13 show that the chemical composition of 71055, and are typical of Apollo 17 basalts. Additional data for Zr and Hf are given in Garg and Ehman (1976) and Hughes La Pr Sm Gd Dy Er Yb Ce Nd Eu Tb Ho Tm Lu Figure 12: Normalized rare-earth-element diagram for 71055, and (data from tables). Note similar pattern. and Schmitt (1985). Moore et al. (1974), Gibson et al. (1976), Moore and Lewis (1976) and Sill et al. (1974) determined C, S and N. Radiogenic age dating Tera et al. (1974) determined the age of by precise Rb-Sr isochron (figure 13). Murthy and Coscio (1976) merely quote Tera et al. Chen et al. (1979) and Taylor and Chen (1979) tried, but could not date by U-Th-Pb. Nyquist et al. (1976) determined the whole rock Rb and Sr isotopic composition of 71035, but did not obtain an isochron. Cosmogenic isotopes and exposure ages Rancitelli et al. (1974) determined the cosmic-rayinduced activity of (144 g). 22 Na was 92 dpm/ kg., 26 Al = 79 dpm/kg, 46 Sc = 87 dpm/kg., 54 Mn = 164 dpm/kg. and 56 Co = 279 dpm/kg. Arvidson et al. (1976) determined the cosmic ray exposure age by Kr-Kr analysis as 110 ± 7 m.y.
7 Other Studies Brecher (1974) Mayeda et al. (1975) Watson et al. (1974) Trice et al. (1974) Usselman et al. (1975) O Hara and Humphries Taylor et al. (1992) magnetic properties oxygen isotopes magnetics elastic properties experimental experimental ISRU benification Processing was returned in a sealed rock box (ALSRC #1) and placed in a freezer (-20 C). It has not been studied. A small slab was sawn from (figure 14). Summary of Age Data for Rb/Sr Tera et al ± 0.09 b.y. Chen et al Tilton and Chen 1979 Caution: Old decay constant used. U/Pb Figure 13: Picture worth a 1000 words (Tera et al. 1975).
8 Table 1. Chemical composition of reference Miller74 Brunfelt74 Boynton75 Rose 74 Philpotts74 Baedecker74 Dickenson89 weight 241 mg SiO2 % (a) (b) TiO (a) (a) (a) (b) Al2O (a) 9.31 (a) (a) 8.62 (b) FeO (a) (a) (a) 19.2 (b) MnO 0.26 (a) 0.26 (a) 0.26 (a) 0.26 (b) 0.27 (a) MgO (a) 8.79 (a) 9.04 (b) CaO 9.5 (a) (a) (a) (b) 12.2 Na2O 0.44 (a) 0.42 (a) 0.41 (a) 0.31 (b) K2O (a) 0.06 (b) (c ) P2O (b) S % sum Sc ppm 95 (a) 82 (a) 87 (b) 94 (a) 80 (d) V 129 (a) 88 (b) 364 (d) Cr 2790 (a) 2640 (a) 2805 (b) 2800 (a) 2805 (d) Co 21.6 (a) 22 (a) 51 (b) 26 (a) 18 (d) Ni <10 (a) 43 (b) 2 (d) Cu 4.4 (a) 31 (b) Zn 3 (a) 1.9 (d) Ga 3 (a) 8.1 (b) 4.27 (d) 22 (d) Ge ppb 3.3 (d) 2.4 (d) As Se Rb 0.9 (a) 0.9 (b) (c ) Sr 104 (a) 170 (b) 121 (c ) Y 69 (b) Zr 223 (b) Nb 27 (b) Mo Ru Rh Pd ppb Ag ppb Cd ppb <1.5 (d) In ppb 4.7 (d) Sn ppb Sb ppb Te ppb Cs ppm 0.07 (a) Ba 39 (a) 315 (b) 62.4 (c ) 63 (d) La 4.67 (a) 4.6 (a) 4.6 (d) Ce 13.4 (a) 22 (a) 15.6 (c ) 23 (a) 13 (d) Pr Nd 17 (c ) 33 (d) Sm 7.05 (a) 6.1 (a) 6.72 (c ) 6 (d) Eu 1.49 (a) 1.8 (a) 1.36 (c ) 1.5 (a) 1.3 (d) Gd Tb 1.74 (a) 2 (a) 2.1 (a) 1.6 (d) Dy 14.3 (a) 13 (c ) Ho Er 7.74 (c ) 0.87 (d) Tm Yb 5.4 (a) 6.3 (a) 5.4 (b) 7.75 (c ) 5.7 (a) 6.4 (d) Lu 1.1 (a) 1.1 (a) 1 (d) Hf 6.6 (a) 6 (a) 7 (a) Ta 1.54 (a) 1.6 (a) 1.3 (a) W ppb (a) Re ppb Os ppb Ir ppb 1.1 (d) Pt ppb Au ppb (d) Th ppm 0.32 (a) U ppm (a) technique: (a) INAA, (b) microchemical, (c ) IDMS, (d) RNAA
9 Table 2. Chemical composition of reference Rhodes76 weight SiO2 % (a) TiO (a) Al2O (a) FeO (a) MnO 0.29 (a) MgO 7.98 (a) CaO (a) Na2O 0.38 (a) K2O 0.03 (a) P2O5 0.1 (a) S % 0.15 (a) sum Sc ppm 87 (b) V Cr 2669 (a) Co 19 (b) Ni Cu Zn Ga Ge ppb As Se Rb 0.41 (c ) Sr 130 (c ) Y Zr Nb Mo Ru Rh Pd ppb Ag ppb Cd ppb In ppb Sn ppb Sb ppb Te ppb Cs ppm Ba 66.3 (c ) La 5.77 (c ) Ce 18.7 (c ) Pr Nd 18.8 (c ) Sm 7.5 (c ) Eu 1.5 (c ) Gd 12.1 (c ) Tb Dy 13.6 (c ) Ho Er 8.27 (c ) Tm Yb 7.71 (c ) Lu 1.14 (b) Hf 7 (b) Ta W ppb Re ppb Os ppb Ir ppb Pt ppb Au ppb Th ppm U ppm technique: (a) XRF, (b) INAA, (c ) IDMS Table 3. Chemical composition of Ma 1979 reference Warner79 weight 0.54 g SiO2 % TiO (a) Al2O3 8.9 (a) FeO 19.4 (a) MnO (a) MgO 7 (a) CaO 11.2 (a) Na2O (a) K2O (a) P2O5 S % sum Sc ppm 85 (a) V 73 (a) Cr 2121 (a) Co 20 (a) Ni Cu Zn Ga Ge ppb As Se Rb Sr Y Zr Nb Mo Ru Rh Pd ppb Ag ppb Cd ppb In ppb Sn ppb Sb ppb Te ppb Cs ppm Ba La 6.1 (a) Ce 21 (a) Pr Nd 23 (a) Sm 8.1 (a) Eu 1.54 (a) Gd Tb 1.9 (a) Dy 13 (a) Ho Er Tm Yb 7.4 (a) Lu 1.02 (a) Hf 7 (a) Ta 1.7 (a) W ppb Re ppb Os ppb Ir ppb Pt ppb Au ppb Th ppm U ppm technique: (a) INAA
10 B1,10,26,9,7,8 Figure 14: Group photo after cutting slab from Small cube is 1 cm. NASA S This is the top, exposed lunar surface. CMeyer g saw cuts 1973,7 29 g,8 394 g,26,9 slab 27.5 g 15 g, g,14 25 g,18 PB, g,11 PB, g,64,72,71 TS,78 TS C Meyer g CMeyer g, g,3 PB, g,1 PB,28,29,30 TS,4,5 TS
11 References Arvidson R., Drozd R., Guiness E., Hohenberg C., Morgan C., Morrison R. and Oberbeck V. (1976) Cosmic ray exposure ages of Apollo 17 samples and the age of Tycho. Proc. 7 th Lunar Sci. Conf Baedecker P.A., Chou C.L., Sundberg L.L. and Wasson J.T. (1974) Volatile and siderophile trace elements in the soils and rocks of Taurus-Littrow. Proc. 5 th Lunar Sci. Conf Boynton W.V., Baedecker P.A., Chou C.L., Robinson K.L. and Wasson J.T. (1975) Mixing and transport of lunar surface materials: Evidence obtained by the determination of lithophile, siderophile and volatile elements. Proc. 6 th Lunar Sci. Conf Brecher A. (1974) Inferences from comparative magnetic studies of some Apollo 17 basalts, breccias and soils. LS V, Brown G.M., Peckett A., Emeleus C.H., Phillips R. and Pinsent R.H. (1975) Petrology and mineralogy of Apollo 17 mare basalts. Proc. 6 th Lunar Sci. Conf Brunfelt A.O., Heier K.S., Nilssen B., Steinnes E. and Sundvoll B. (1974) Elemental composition of Apollo 17 fines and rocks. Proc. 5 th Lunar Sci. Conf Chen J.H., Tilton G.R. and Mattinson J.M. (1979) Lead isotope systematics of three Taurus-Littrow mare basalts. LPS X, Dickinson T., Taylor G.J., Keil K. and Bild R.W. (1989) Germanium abundances in lunar basalts: Evidence of mantle metasomatism. Proc. 19 th Lunar Planet. Sci. Conf Dymek R.F., Albee A.L. and Chodos A.A. (1975) Comparative mineralogy and petrology of Apollo 17 mare basalts: Samples 70215, 71055, 74255, Proc. 6 th Lunar Sci. Conf Engelhardt W. von (1979) Ilmenite in the crystallization sequence of lunar rocks. Proc. 10 th Lunar Planet. Sci. Conf Garg A.N. and Ehmann W.N. (1976) Zr-Hf fractionation in chemically defined lunar rock groups. Proc. 7 th Lunar Sci. Conf Gibson E.K., Morris R.V. and Usselman T.M. (1976) Sulfur in the Apollo 17 basalts and their source regions. Proc. 7 th Lunar Sci. Conf Hughes S.S. and Schmitt R.A. (1985) Zr-Hf-Ta fractionation during lunar evolution. Proc. 16 th Lunar Planet. Sci. Conf. D31-D45. LSPET (1973) Preliminary examination of lunar samples. Apollo 17 Prelim. Sci. Rept. NASA SP-330 Ma M.-S., Schmitt R.A., Warner R.D., Taylor G.J. and Keil K. (1979) Composition, petrology and genesis of Apollo 17 high-ti mare basalts. LPS X, Mayeda T.K., Shearer J. and Clayton R.N. (1975) Oxygen isotope fractionation of Apollo 17 rocks. Proc. 6 th Lunar Sci. Conf Miller M.D., Pacer R.A., Ma M.-S., Hawke B.R., Lockart G.L. and Ehmann W.D. (1974) Composition studies of the lunar regolith at the Apollo 17 site. Proc. 5 th Lunar Sci. Conf Moore C.B., Lewis C.F. and Cripe J.D. (1974) Total carbon and sulfur contents of Apollo 17 lunar samples. Proc. 5 th Lunar Sci. Conf Moore C.B and Lewis C.F. (1976) Total nitrogen contents of Apollo 15, 16 and 17 lunar rocks and soils. LS VII, Muhich T., Vaniman D. and Heiken G. (1990) Ilmenite in high-ti Apollo 17 basalts: Variations in composition with degree of exsolution. LPSC XXI, Muthy V.R. and Coscio C. (1976) Rb-Sr ages and isotopic systematics of some Serenitatis mare basalts. Proc. 7 th Lunar Sci. Conf Neal C.R. and Taylor L.A. (1993) Catalog of Apollo 17 Rocks. Vol. 2 Nyquist L.E., Bansal B.M. and Wiesmann H. (1976) Sr isotopic constraints on the petrogenesis of Apollo 17 mare basalts. Proc. 7 th Lunar Sci. Conf O Hara M.J. and Humphries D.J. (1975) Armalcolite crystallization, phenocryst assemblages, eruption conditions and origin of eleven high titanium basalts from Taurus Littrow. LS VI, Philpotts J.A., Schumann S., Kouns C.W., Lum R.K.L and Winzer S. (1974) Origin of Apollo 17 rocks and soils. Proc. 5 th Lunar Sci. Conf Rancitelli L.A., Perkins R.W., Fekix W.D., and Wogman N.A. (1974) Solar flare and lunar surface process characterization of Apollo 17 site. Proc. 5 th Lunar Sci. Conf
12 Rhodes J.M., Hubbard N.J., Wiesmann H., Rodgers K.V., Brannon J.C. and Bansal B.M. (1976) Chemistry, classification and petrogenesis of Apollo 17 mare basalts. Proc. 7 th Lunar Sci. Conf Rose H.J., Cuttitta F., Berman S., Brown F.W., Carron M.K., Christian R.P., Dwornik E.J. and Greenland L.P. (1974) Chemical composition of rocks and soils at Taurus-Littrow. Proc. 5 th Lunar Sci. Conf Sill G.T., Nagy B., Nagy L.A., Hamilton P.B, McEwan W.S. and Urey H.C. (1974) Carbon compounds in Apollo 17 lunar samples: Implications of cometary contribution to breccia 78155? LS V, Sung C.-M., Abu-Eid R.M. and Burns R.G. (1974) Ti+3/ Ti+4 ratios in lunar pyroxenes: implications to depth of origin of mare basalt magma. Proc. 5 th Lunar Sci. Conf Taylor L.A., McKay D.S., Patchen A., Wentworth S., Oder R. and Jerde E. (1992) Magnetic beneficiation of high-ti mare basalts: Petrographic analysis. LPSC XXVIII, Tera F., Papanastassiou D.A. and Wasserburg G.J. (1974) The lunar time scale and a summary of isotopic evidence for a terminal lunar cataclysm. LS V, Tilton G.R. and Chen J.H. (1979) Lead isotope systematics of three Apollo 17 mare basalts. Proc. 10 th Lunar Planet. Sci. Conf Trice R., Warren N. and Anderson O.L. (1974) Rock elastic properties and near-surface structure of Taurus-Littrow. Proc. 5 th Lunar Sci. Conf Usselman T.M., Lofgren G.E., Donaldson C.H. and Williams R.J. (1975) Experimentally reproduced textures and mineral chemistries of high-titanium mare basalts. Proc. 6 th Lunar Sci. Conf Warner R.D., Taylor G.J., Conrad G.H., Northrup H.R., Barker S., Keil K., Ma M.-S. and Schmitt R. (1979) Apollo 17 high Ti mare basalts: New bulk composition data, magma types and petrogenesis. Proc. 10 th Lunar Planet. Sci. Conf Watson D.E., Larson E.E. and Reynolds R.L. (1974) Microscopic and thermomagnetic analysis of Apollo 17 breccia and basalt: feasibility of obtaining meaningful paleointensities of the lunar magnetic field. LS V, Wolfe E.W., Bailey N.G., Lucchitta B.K., Muehlberger W.R., Scott D.H., Sutton R.L. and Wilshire H.G. (1981) The geologic investigation of the Taurus-Littrow Valley: Apollo 17 landing site. USGS Prof. Paper 1080 Table 4 U ppm Th ppm K ppm Rb ppm Sr ppm Nd ppm Sm ppm technique Rancitelli et al counting LSPET counting Brunfelt et al INAA Tilton and Chen idms
74235 Vitrophric Basalt 59 grams
 74235 Vitrophric Basalt 59 grams Figure 1: Photo of 74235. Cube is 1 cm. S73-16017 Figure 2: Map of station 4, Apollo 17. Transcript (while the LMP was discussing the core with CC, the CDR made a discovery)
74235 Vitrophric Basalt 59 grams Figure 1: Photo of 74235. Cube is 1 cm. S73-16017 Figure 2: Map of station 4, Apollo 17. Transcript (while the LMP was discussing the core with CC, the CDR made a discovery)
72320 Partially Shadowed Soil (portion frozen) grams. boulder # 2 station 2 South Massif
 Partially Shadowed Soil (portion ) 106.31 grams Nansen Crater boulder # 2 station 2 South Massif Figure 1: Location of soil sample in shadow of boulder 2, at station 2, Apollo 17. NASA photo #AS17-137-20925.
Partially Shadowed Soil (portion ) 106.31 grams Nansen Crater boulder # 2 station 2 South Massif Figure 1: Location of soil sample in shadow of boulder 2, at station 2, Apollo 17. NASA photo #AS17-137-20925.
72335 Impact melt Breccia grams
 Impact melt Breccia 108.9 grams Figure 1: Location of on boulder #2 on landslide off of South Massif. Boulder is ~ 2-3 meters high. AS17-137-20918. Transcript Hey, that s a different rock, Gene (station
Impact melt Breccia 108.9 grams Figure 1: Location of on boulder #2 on landslide off of South Massif. Boulder is ~ 2-3 meters high. AS17-137-20918. Transcript Hey, that s a different rock, Gene (station
DRAFT Cataclastic Dunite grams
 72415-72418 Cataclastic Dunite 58.74 grams DRAFT Figure 1: Boulder 3 at Station 2, with large dunite clast (72415-72418). The bright spot is the location of samples. A sample of the boulder matrix (72435)
72415-72418 Cataclastic Dunite 58.74 grams DRAFT Figure 1: Boulder 3 at Station 2, with large dunite clast (72415-72418). The bright spot is the location of samples. A sample of the boulder matrix (72435)
65055 Basaltic Impact Melt grams
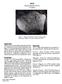
 65055 Basaltic Impact Melt 500.8 grams Figure 1: Photo of 65055 showing large zap pit. Cube is 1 cm. S72-43869. Introduction According to the Apollo 16 Catalog by Ryder and Norman, 65055 is an aluminous,
65055 Basaltic Impact Melt 500.8 grams Figure 1: Photo of 65055 showing large zap pit. Cube is 1 cm. S72-43869. Introduction According to the Apollo 16 Catalog by Ryder and Norman, 65055 is an aluminous,
Ilmenite Basalt grams
 Ilmenite Basalt 737.6 grams Figure 1: Picute of 5 meter boulder on rim of Shorty Crater showing where was collected. Trench in forground is location of orange soil sample 74220. Photo number AS17-137-20990
Ilmenite Basalt 737.6 grams Figure 1: Picute of 5 meter boulder on rim of Shorty Crater showing where was collected. Trench in forground is location of orange soil sample 74220. Photo number AS17-137-20990
,1,2,3,4,9, Impact Melt Breccia 65.4 grams
 63335 Impact Melt Breccia 65.4 grams,1,2,3,4,9,10,8 Figure 2: 63335,6. Cube is 1 cm. S75-33389.,7 Introduction 63335 is a sample chipped off of Shadow Rock (Ulrich 1973). It was collected as several fragments
63335 Impact Melt Breccia 65.4 grams,1,2,3,4,9,10,8 Figure 2: 63335,6. Cube is 1 cm. S75-33389.,7 Introduction 63335 is a sample chipped off of Shadow Rock (Ulrich 1973). It was collected as several fragments
grams grams Regolith Breccia
 10019 297 grams 10066 60 grams Regolith Breccia Figure 1: Photo of 10019,1. Cube is 1 inch and scale is in cm. NASA S76-23354. Introduction Kramer et al. (1077) reported that 10019 and 10066 appeared to
10019 297 grams 10066 60 grams Regolith Breccia Figure 1: Photo of 10019,1. Cube is 1 inch and scale is in cm. NASA S76-23354. Introduction Kramer et al. (1077) reported that 10019 and 10066 appeared to
14041, and Regolith Breccia 166.3, and 65.2 grams
 14041, and 14045 Regolith Breccia 166.3, 103.2 and 65.2 grams Figure 1a: Sample 14041-14045 on lunar surface. MET and LM in distance. AS14-68-9409. Introduction Samples 14041 14046 are fragments from a
14041, and 14045 Regolith Breccia 166.3, 103.2 and 65.2 grams Figure 1a: Sample 14041-14045 on lunar surface. MET and LM in distance. AS14-68-9409. Introduction Samples 14041 14046 are fragments from a
DRAFT Coarse-fines 569 grams
 10085 Coarse-fines 569 grams DRAFT Figure 1: Selected coarse fines from 10085. Scale is in mm. Photo from Wood et al. 1969. Introduction!0085 and 10084 were created during the Apollo 11 preliminary examination
10085 Coarse-fines 569 grams DRAFT Figure 1: Selected coarse fines from 10085. Scale is in mm. Photo from Wood et al. 1969. Introduction!0085 and 10084 were created during the Apollo 11 preliminary examination
15556 Vesicular Olivine-normative Basalt grams
 15556 Vesicular Olivine-normative Basalt 1542.3 grams Figure 1: Photo of 15556,0. Largest vesicles are about 6 mm. NASA S87-48187. Introduction 15556 was collected about 60 meters from the edge of Hadley
15556 Vesicular Olivine-normative Basalt 1542.3 grams Figure 1: Photo of 15556,0. Largest vesicles are about 6 mm. NASA S87-48187. Introduction 15556 was collected about 60 meters from the edge of Hadley
70035 Ilmenite Basalt 5765 grams
 Ilmenite Basalt 5765 grams Figure 1: Photograph of top surface of illustrating vugs and vesicles. Note the smooth, rounded surface shaped by micrometeorite bombardment. Small cube is 1 cm. NASA photo #
Ilmenite Basalt 5765 grams Figure 1: Photograph of top surface of illustrating vugs and vesicles. Note the smooth, rounded surface shaped by micrometeorite bombardment. Small cube is 1 cm. NASA photo #
14315 Unusual Regolith Breccia 115 grams
 Unusual Regolith Breccia 115 grams Figure 1: Photo of,0 after chipping and dusting. Sample is 5 cm across. NASA S86-36340. Introduction was collected as a grab sample from the North Boulder Field (station
Unusual Regolith Breccia 115 grams Figure 1: Photo of,0 after chipping and dusting. Sample is 5 cm across. NASA S86-36340. Introduction was collected as a grab sample from the North Boulder Field (station
grams grams Poikilitic Impact Melt Breccia
 64567 13.8 grams 64569-14.3 grams Poikilitic Impact Melt Breccia Figure 1: Photo of 64567. Scale in mm. S72-55386 Mineralogical Mode by Simonds et al. (1973) 64567 64569 Plagioclase 69% 57 Pyroxene 10
64567 13.8 grams 64569-14.3 grams Poikilitic Impact Melt Breccia Figure 1: Photo of 64567. Scale in mm. S72-55386 Mineralogical Mode by Simonds et al. (1973) 64567 64569 Plagioclase 69% 57 Pyroxene 10
15595 and Vuggy Vitrophyric Pigeonite Basalt and grams
 and 15596 Vuggy Vitrophyric Pigeonite Basalt 237.6 and 224.8 grams Figure 1: Photo of. Sample is about 9 cm across. NASA S71-44491. Figure 2: Photo of 15596 showing surface that was exposed to micrometeorites.
and 15596 Vuggy Vitrophyric Pigeonite Basalt 237.6 and 224.8 grams Figure 1: Photo of. Sample is about 9 cm across. NASA S71-44491. Figure 2: Photo of 15596 showing surface that was exposed to micrometeorites.
grams grams Double Drive tube 61 cm
 68002 583.5 grams 68001 840.7 grams Double Drive tube 61 cm Figure 1a: Surface photo for double drive tube 68001-2. AS16-108-17684. Introduction Station 8 was the closest to South Ray Crater so premission
68002 583.5 grams 68001 840.7 grams Double Drive tube 61 cm Figure 1a: Surface photo for double drive tube 68001-2. AS16-108-17684. Introduction Station 8 was the closest to South Ray Crater so premission
grams grams Crystalline matrix Breccia
 14069 24.87 grams 14070-36.56 grams Crystalline matrix Breccia Figure 1: Photo of 14069,0. Scale in cm and mm. S78-28808. 14070 Figure 2: Photo of 14070. Scale is in cm. S71-22168. Introduction 14069 and
14069 24.87 grams 14070-36.56 grams Crystalline matrix Breccia Figure 1: Photo of 14069,0. Scale in cm and mm. S78-28808. 14070 Figure 2: Photo of 14070. Scale is in cm. S71-22168. Introduction 14069 and
grams g g g g Regolith Breccia
 15306 134.2 grams 15315 35.6 g 15324 32.3 g 15325 57.8 g 15330 57.8 g Regolith Breccia Figure 1a,b: Photo of dust-covered 15306. Sample is 7 cm across. S71-43064 and 067. Introduction 15306 is a regolith
15306 134.2 grams 15315 35.6 g 15324 32.3 g 15325 57.8 g 15330 57.8 g Regolith Breccia Figure 1a,b: Photo of dust-covered 15306. Sample is 7 cm across. S71-43064 and 067. Introduction 15306 is a regolith
61015 Dimict Breccia 1789 grams
 61015 Dimict Breccia 1789 grams Figure 1: Photo of 61015. NASA # S72-37758. Scale and cube in cm. Note the zap pits on the T1 surface and the residual black glass coating on the sides. Introduction Breccia
61015 Dimict Breccia 1789 grams Figure 1: Photo of 61015. NASA # S72-37758. Scale and cube in cm. Note the zap pits on the T1 surface and the residual black glass coating on the sides. Introduction Breccia
Element Cube Project (x2)
 Element Cube Project (x2) Background: As a class, we will construct a three dimensional periodic table by each student selecting two elements in which you will need to create an element cube. Helpful Links
Element Cube Project (x2) Background: As a class, we will construct a three dimensional periodic table by each student selecting two elements in which you will need to create an element cube. Helpful Links
12052 Pigeonite Basalt 1866 grams
 12052 Pigeonite Basalt 1866 grams Revised Figure 1: Photo of broken side of 12052 showing vugs. Sample is 6.5 cm high. NASA # S70-44633. Summary of Age Data for 12052 Ar/Ar Rb/Sr Nyquist 1977 (recalculated)
12052 Pigeonite Basalt 1866 grams Revised Figure 1: Photo of broken side of 12052 showing vugs. Sample is 6.5 cm high. NASA # S70-44633. Summary of Age Data for 12052 Ar/Ar Rb/Sr Nyquist 1977 (recalculated)
76215 Vesicular Micropoikilitic Impact Melt Breccia 644 grams
 76215 Vesicular Micropoikilitic Impact Melt Breccia 644 grams 76215 76215 Figure 1: Photo of station 6 boulder with location of 76215 indicated. AS17-140-21421 Figure 2: Photo of 76215. Sample is about
76215 Vesicular Micropoikilitic Impact Melt Breccia 644 grams 76215 76215 Figure 1: Photo of station 6 boulder with location of 76215 indicated. AS17-140-21421 Figure 2: Photo of 76215. Sample is about
12013 Breccia with granite 82.3 grams
 12013 Breccia with granite 82.3 grams Figure 1: Close-up photo of 12013,11. Field of view is 4.5 cm. NASA# S70-43636 Introduction 12013 is a unique lunar sample, but it did not appear to be remarkable
12013 Breccia with granite 82.3 grams Figure 1: Close-up photo of 12013,11. Field of view is 4.5 cm. NASA# S70-43636 Introduction 12013 is a unique lunar sample, but it did not appear to be remarkable
Radiometric Dating (tap anywhere)
 Radiometric Dating (tap anywhere) Protons Neutrons Electrons Elements on the periodic table are STABLE Elements can have radioactive versions of itself called ISOTOPES!! Page 1 in your ESRT has your list!
Radiometric Dating (tap anywhere) Protons Neutrons Electrons Elements on the periodic table are STABLE Elements can have radioactive versions of itself called ISOTOPES!! Page 1 in your ESRT has your list!
IV. Governador Valadares clinopyroxenite, 158 grams find
 IV. Governador Valadares clinopyroxenite, 158 grams find Figure IV-1. Photograph of Governador Valadares (158 grams) from Dr. Fernanda Ferrucci via Dr. Giuseppe Cavarretta. Photo taken by L. Spinozzi.
IV. Governador Valadares clinopyroxenite, 158 grams find Figure IV-1. Photograph of Governador Valadares (158 grams) from Dr. Fernanda Ferrucci via Dr. Giuseppe Cavarretta. Photo taken by L. Spinozzi.
The Periodic Table. Periodic Properties. Can you explain this graph? Valence Electrons. Valence Electrons. Paramagnetism
 Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
The Periodic Table of Elements
 The Periodic Table of Elements 8 Uuo Uus Uuh (9) Uup (88) Uuq (89) Uut (8) Uub (8) Rg () 0 Ds (9) 09 Mt (8) 08 Hs (9) 0 h () 0 Sg () 0 Db () 0 Rf () 0 Lr () 88 Ra () 8 Fr () 8 Rn () 8 At (0) 8 Po (09)
The Periodic Table of Elements 8 Uuo Uus Uuh (9) Uup (88) Uuq (89) Uut (8) Uub (8) Rg () 0 Ds (9) 09 Mt (8) 08 Hs (9) 0 h () 0 Sg () 0 Db () 0 Rf () 0 Lr () 88 Ra () 8 Fr () 8 Rn () 8 At (0) 8 Po (09)
Oriented Ilmenite Basalt 1493 grams
 74275 Oriented Ilmenite Basalt 1493 grams 74275 Figure 1: Location of basalt sample 74275 at Shorty Crater - also see 74220. (falce color) AS17-137-20990 Introduction 74275 is a fine-grained, high-ti mare
74275 Oriented Ilmenite Basalt 1493 grams 74275 Figure 1: Location of basalt sample 74275 at Shorty Crater - also see 74220. (falce color) AS17-137-20990 Introduction 74275 is a fine-grained, high-ti mare
15009 Single Drive Tube Station 6
 15009 Single Drive Tube Station 6 Figure 2: Location of soil samples, trench and drive tube at station 6, Apollo 15. Figure 1: Photo of drive tube 15009 driven in all the way. AS15-86-11565. Introduction
15009 Single Drive Tube Station 6 Figure 2: Location of soil samples, trench and drive tube at station 6, Apollo 15. Figure 1: Photo of drive tube 15009 driven in all the way. AS15-86-11565. Introduction
DRAFT Regolith Breccia 155 grams
 12034 Regolith Breccia 155 grams DRAFT Figure 1: Lunar sample 12034 after first saw cut in 1970. NASA S75-34235. Cube is 1 cm. Introduction 12034 was collected from the bottom of the same trench as soil
12034 Regolith Breccia 155 grams DRAFT Figure 1: Lunar sample 12034 after first saw cut in 1970. NASA S75-34235. Cube is 1 cm. Introduction 12034 was collected from the bottom of the same trench as soil
Chemistry 431 Practice Final Exam Fall Hours
 Chemistry 431 Practice Final Exam Fall 2018 3 Hours R =8.3144 J mol 1 K 1 R=.0821 L atm mol 1 K 1 R=.08314 L bar mol 1 K 1 k=1.381 10 23 J molecule 1 K 1 h=6.626 10 34 Js N A = 6.022 10 23 molecules mol
Chemistry 431 Practice Final Exam Fall 2018 3 Hours R =8.3144 J mol 1 K 1 R=.0821 L atm mol 1 K 1 R=.08314 L bar mol 1 K 1 k=1.381 10 23 J molecule 1 K 1 h=6.626 10 34 Js N A = 6.022 10 23 molecules mol
14049 Regolith Breccia grams
 Regolith Breccia 200.1 grams Figure 1: Photo of. Edge of cube is 1 inch. NASA S71-29143. Figure 2: Map of Apollo 14 showing traverse for EVA 2. Introduction The Apollo 14 regolith breccias (vitric matrix
Regolith Breccia 200.1 grams Figure 1: Photo of. Edge of cube is 1 inch. NASA S71-29143. Figure 2: Map of Apollo 14 showing traverse for EVA 2. Introduction The Apollo 14 regolith breccias (vitric matrix
Atoms and the Periodic Table
 Atoms and the Periodic Table Parts of the Atom Proton Found in the nucleus Number of protons defines the element Charge +1, mass 1 Parts of the Atom Neutron Found in the nucleus Stabilizes the nucleus
Atoms and the Periodic Table Parts of the Atom Proton Found in the nucleus Number of protons defines the element Charge +1, mass 1 Parts of the Atom Neutron Found in the nucleus Stabilizes the nucleus
Solutions and Ions. Pure Substances
 Class #4 Solutions and Ions CHEM 107 L.S. Brown Texas A&M University Pure Substances Pure substance: described completely by a single chemical formula Fixed composition 1 Mixtures Combination of 2 or more
Class #4 Solutions and Ions CHEM 107 L.S. Brown Texas A&M University Pure Substances Pure substance: described completely by a single chemical formula Fixed composition 1 Mixtures Combination of 2 or more
70017 Ilmenite Basalt 2957 grams
 Ilmenite Basalt 2957 grams Figure 1: Photo of lunar basalt showing vugs and vesicles. Sample is about 10 cm across. Cube is 1 cm. NASA# S73-15723. Introduction Lunar sample is a vesicular, medium-grained,
Ilmenite Basalt 2957 grams Figure 1: Photo of lunar basalt showing vugs and vesicles. Sample is about 10 cm across. Cube is 1 cm. NASA# S73-15723. Introduction Lunar sample is a vesicular, medium-grained,
PERIODIC TABLE OF THE ELEMENTS
 Useful Constants and equations: K = o C + 273 Avogadro's number = 6.022 x 10 23 d = density = mass/volume R H = 2.178 x 10-18 J c = E = h = hc/ h = 6.626 x 10-34 J s c = 2.998 x 10 8 m/s E n = -R H Z 2
Useful Constants and equations: K = o C + 273 Avogadro's number = 6.022 x 10 23 d = density = mass/volume R H = 2.178 x 10-18 J c = E = h = hc/ h = 6.626 x 10-34 J s c = 2.998 x 10 8 m/s E n = -R H Z 2
62255 Anorthosite with melt 1239 grams
 62255 Anorthosite with melt 1239 grams Figure 1: Photo of 62255 showing glass splash on anorthosite. Cube is 1 cm. NASA S # S72-38309. Introduction Lunar sample 62255 is significant because it is largely
62255 Anorthosite with melt 1239 grams Figure 1: Photo of 62255 showing glass splash on anorthosite. Cube is 1 cm. NASA S # S72-38309. Introduction Lunar sample 62255 is significant because it is largely
CHEM 10113, Quiz 5 October 26, 2011
 CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
5 questions, 3 points each, 15 points total possible. 26 Fe Cu Ni Co Pd Ag Ru 101.
 Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
Nucleus. Electron Cloud
 Atomic Structure I. Picture of an Atom Nucleus Electron Cloud II. Subatomic particles Particle Symbol Charge Relative Mass (amu) protons p + +1 1.0073 neutrons n 0 1.0087 electrons e - -1 0.00054858 Compare
Atomic Structure I. Picture of an Atom Nucleus Electron Cloud II. Subatomic particles Particle Symbol Charge Relative Mass (amu) protons p + +1 1.0073 neutrons n 0 1.0087 electrons e - -1 0.00054858 Compare
CLASS TEST GRADE 11. PHYSICAL SCIENCES: CHEMISTRY Test 4: Matter and materials 1
 CLASS TEST GRADE PHYSICAL SCIENCES: CHEMISTRY Test 4: Matter and materials MARKS: 45 TIME: hour INSTRUCTIONS AND INFORMATION. Answer ALL the questions. 2. You may use non-programmable calculators. 3. You
CLASS TEST GRADE PHYSICAL SCIENCES: CHEMISTRY Test 4: Matter and materials MARKS: 45 TIME: hour INSTRUCTIONS AND INFORMATION. Answer ALL the questions. 2. You may use non-programmable calculators. 3. You
CHEM 130 Exp. 8: Molecular Models
 CHEM 130 Exp. 8: Molecular Models In this lab, we will learn and practice predicting molecular structures from molecular formulas. The Periodic Table of the Elements IA 1 H IIA IIIA IVA VA VIA VIIA 3 5
CHEM 130 Exp. 8: Molecular Models In this lab, we will learn and practice predicting molecular structures from molecular formulas. The Periodic Table of the Elements IA 1 H IIA IIIA IVA VA VIA VIIA 3 5
10020 Ilmenite Basalt (low K) 425 grams
 10020 Ilmenite Basalt (low K) 425 grams Figure 1: Photo of 10020 taken in the F-201 during initial processing. Sample is 6 cm across. NASA S-69-45261. Introduction 10020 is a low-k variety of fine-grained
10020 Ilmenite Basalt (low K) 425 grams Figure 1: Photo of 10020 taken in the F-201 during initial processing. Sample is 6 cm across. NASA S-69-45261. Introduction 10020 is a low-k variety of fine-grained
If anything confuses you or is not clear, raise your hand and ask!
 CHM 1045 Dr. Light s Section December 10, 2002 FINAL EXAM Name (please print) Recitation Section Meeting Time This exam consists of six pages. Make sure you have one of each. Print your name at the top
CHM 1045 Dr. Light s Section December 10, 2002 FINAL EXAM Name (please print) Recitation Section Meeting Time This exam consists of six pages. Make sure you have one of each. Print your name at the top
Ch. 9 NOTES ~ Chemical Bonding NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics.
 Ch. 9 NOTES ~ Chemical Bonding NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. I. Review: Comparison of ionic and molecular compounds Molecular compounds Ionic
Ch. 9 NOTES ~ Chemical Bonding NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. I. Review: Comparison of ionic and molecular compounds Molecular compounds Ionic
Last 4 Digits of USC ID:
 Chemistry 05 B Practice Exam Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 8 2 4 3 9 4 0
Chemistry 05 B Practice Exam Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 8 2 4 3 9 4 0
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
NAME: FIRST EXAMINATION
 1 Chemistry 64 Winter 1994 NAME: FIRST EXAMINATION THIS EXAMINATION IS WORTH 100 POINTS AND CONTAINS 4 (FOUR) QUESTIONS THEY ARE NOT EQUALLY WEIGHTED! YOU SHOULD ATTEMPT ALL QUESTIONS AND ALLOCATE YOUR
1 Chemistry 64 Winter 1994 NAME: FIRST EXAMINATION THIS EXAMINATION IS WORTH 100 POINTS AND CONTAINS 4 (FOUR) QUESTIONS THEY ARE NOT EQUALLY WEIGHTED! YOU SHOULD ATTEMPT ALL QUESTIONS AND ALLOCATE YOUR
Chapter 3: Stoichiometry
 Chapter 3: Stoichiometry Chem 6A Michael J. Sailor, UC San Diego 1 Announcements: Thursday (Sep 29) quiz: Bring student ID or we cannot accept your quiz! No notes, no calculators Covers chapters 1 and
Chapter 3: Stoichiometry Chem 6A Michael J. Sailor, UC San Diego 1 Announcements: Thursday (Sep 29) quiz: Bring student ID or we cannot accept your quiz! No notes, no calculators Covers chapters 1 and
(C) Pavel Sedach and Prep101 1
 (C) Pavel Sedach and Prep101 1 (C) Pavel Sedach and Prep101 1 (C) Pavel Sedach and Prep101 2 (C) Pavel Sedach and Prep101 2 (C) Pavel Sedach and Prep101 3 (C) Pavel Sedach and Prep101 3 (C) Pavel Sedach
(C) Pavel Sedach and Prep101 1 (C) Pavel Sedach and Prep101 1 (C) Pavel Sedach and Prep101 2 (C) Pavel Sedach and Prep101 2 (C) Pavel Sedach and Prep101 3 (C) Pavel Sedach and Prep101 3 (C) Pavel Sedach
Part 2. Multiple choice (use answer card). 90 pts. total. 3 pts. each.
 1 Exam I CHEM 1303.001 Name (print legibly) Seat no. On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Part 1. Nomenclature. 10 pts. total. 2 pts. each. Fill in
1 Exam I CHEM 1303.001 Name (print legibly) Seat no. On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Part 1. Nomenclature. 10 pts. total. 2 pts. each. Fill in
02/05/09 Last 4 Digits of USC ID: Dr. Jessica Parr
 Chemistry 05 B First Letter of PLEASE PRINT YOUR NAME IN BLOCK LETTERS Exam last Name Name: 02/05/09 Last 4 Digits of USC ID: Dr. Jessica Parr Lab TA s Name: Question Points Score Grader 2 2 9 3 9 4 2
Chemistry 05 B First Letter of PLEASE PRINT YOUR NAME IN BLOCK LETTERS Exam last Name Name: 02/05/09 Last 4 Digits of USC ID: Dr. Jessica Parr Lab TA s Name: Question Points Score Grader 2 2 9 3 9 4 2
Chem Exam 1. September 26, Dr. Susan E. Bates. Name 9:00 OR 10:00
 Chem 1711 Exam 1 September 26, 2013 Dr. Susan E. Bates Name 9:00 OR 10:00 N A = 6.022 x 10 23 mol 1 I A II A III B IV B V B VI B VII B VIII I B II B III A IV A V A VI A VII A inert gases 1 H 1.008 3 Li
Chem 1711 Exam 1 September 26, 2013 Dr. Susan E. Bates Name 9:00 OR 10:00 N A = 6.022 x 10 23 mol 1 I A II A III B IV B V B VI B VII B VIII I B II B III A IV A V A VI A VII A inert gases 1 H 1.008 3 Li
Guide to the Extended Step-Pyramid Periodic Table
 Guide to the Extended Step-Pyramid Periodic Table William B. Jensen Department of Chemistry University of Cincinnati Cincinnati, OH 452201-0172 The extended step-pyramid table recognizes that elements
Guide to the Extended Step-Pyramid Periodic Table William B. Jensen Department of Chemistry University of Cincinnati Cincinnati, OH 452201-0172 The extended step-pyramid table recognizes that elements
PHYSICAL SCIENCES GRADE : 10
 PHYSICAL SCIENCES GRADE : 0 TIME : hour TOTAL : 75 INSTRUCTIONS AND INFORMATION. Write your full name on your answer book in the appropriate place. 2. The question paper consists of SEVEN questions. Answer
PHYSICAL SCIENCES GRADE : 0 TIME : hour TOTAL : 75 INSTRUCTIONS AND INFORMATION. Write your full name on your answer book in the appropriate place. 2. The question paper consists of SEVEN questions. Answer
Essential Chemistry for Biology
 1 Chapter 2 Essential Chemistry for Biology Biology and Society: More Precious than Gold A drought is a period of abnormally dry weather that changes the environment and one of the most devastating disasters.
1 Chapter 2 Essential Chemistry for Biology Biology and Society: More Precious than Gold A drought is a period of abnormally dry weather that changes the environment and one of the most devastating disasters.
SCIENCE 1206 UNIT 2 CHEMISTRY. September 2017 November 2017
 SCIENCE 1206 UNIT 2 CHEMISTRY September 2017 November 2017 UNIT OUTLINE 1. Review of Grade 9 Terms & the Periodic Table Bohr diagrams Evidence for chemical reactions Chemical Tests 2. Naming & writing
SCIENCE 1206 UNIT 2 CHEMISTRY September 2017 November 2017 UNIT OUTLINE 1. Review of Grade 9 Terms & the Periodic Table Bohr diagrams Evidence for chemical reactions Chemical Tests 2. Naming & writing
9/20/2017. Elements are Pure Substances that cannot be broken down into simpler substances by chemical change (contain Only One Type of Atom)
 CAPTER 6: TE PERIODIC TABLE Elements are Pure Substances that cannot be broken down into simpler substances by chemical change (contain Only One Type of Atom) The Periodic Table (Mendeleev) In 1872, Dmitri
CAPTER 6: TE PERIODIC TABLE Elements are Pure Substances that cannot be broken down into simpler substances by chemical change (contain Only One Type of Atom) The Periodic Table (Mendeleev) In 1872, Dmitri
Why all the repeating Why all the repeating Why all the repeating Why all the repeating
 Why all the repeating Why all the repeating Why all the repeating Why all the repeating Patterns What Patterns have you observed in your life? Where to Get Help If you don t understand concepts in chapter
Why all the repeating Why all the repeating Why all the repeating Why all the repeating Patterns What Patterns have you observed in your life? Where to Get Help If you don t understand concepts in chapter
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
10003 Ilmenite Basalt (low K) 213 grams
 10003 Ilmenite Basalt (low K) 213 grams Figure 1: Photo of 10003,25. Sample is 5 cm across. NASA S76-25545. Introduction Lunar sample 10003 is a low-k, high-ti basalt (figure 1). It is about 3.9 b.y. old,
10003 Ilmenite Basalt (low K) 213 grams Figure 1: Photo of 10003,25. Sample is 5 cm across. NASA S76-25545. Introduction Lunar sample 10003 is a low-k, high-ti basalt (figure 1). It is about 3.9 b.y. old,
DO NOW: Retrieve your projects. We will be reviewing them again today. Textbook pg 23, answer questions 1-3. Use the section 1.2 to help you.
 DO NOW: Retrieve your projects. We will be reviewing them again today. Textbook pg, answer questions. Use the section. to help you. Chapter test is FRIDAY. The Periodic Table of Elements 8 Uuo Uus Uuh
DO NOW: Retrieve your projects. We will be reviewing them again today. Textbook pg, answer questions. Use the section. to help you. Chapter test is FRIDAY. The Periodic Table of Elements 8 Uuo Uus Uuh
The Periodic Table of the Elements
 The Periodic Table of the Elements All matter is composed of elements. All of the elements are composed of atoms. An atom is the smallest part of an element which still retains the properties of that element.
The Periodic Table of the Elements All matter is composed of elements. All of the elements are composed of atoms. An atom is the smallest part of an element which still retains the properties of that element.
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Modified from: Larry Scheffler Lincoln High School IB Chemistry 1-2.1
 Modified from: Larry Scheffler Lincoln High School IB Chemistry 1-2.1 The development of the periodic table brought a system of order to what was otherwise an collection of thousands of pieces of information.
Modified from: Larry Scheffler Lincoln High School IB Chemistry 1-2.1 The development of the periodic table brought a system of order to what was otherwise an collection of thousands of pieces of information.
7) Applications of Nuclear Radiation in Science and Technique (1) Analytical applications (Radiometric titration)
 7) Applications of Nuclear Radiation in Science and Technique (1) (Radiometric titration) The radioactive material is indicator Precipitation reactions Complex formation reactions Principle of a precipitation
7) Applications of Nuclear Radiation in Science and Technique (1) (Radiometric titration) The radioactive material is indicator Precipitation reactions Complex formation reactions Principle of a precipitation
(please print) (1) (18) H IIA IIIA IVA VA VIA VIIA He (2) (13) (14) (15) (16) (17)
 CHEM 10113, Quiz 3 September 28, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
CHEM 10113, Quiz 3 September 28, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
M10/4/CHEMI/SPM/ENG/TZ2/XX+ CHEMISTRY. Wednesday 12 May 2010 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES
 M10/4/CHEMI/SPM/ENG/TZ/XX+ 106116 CHEMISTRY standard level Paper 1 Wednesday 1 May 010 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so. Answer
M10/4/CHEMI/SPM/ENG/TZ/XX+ 106116 CHEMISTRY standard level Paper 1 Wednesday 1 May 010 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so. Answer
MANY ELECTRON ATOMS Chapter 15
 MANY ELECTRON ATOMS Chapter 15 Electron-Electron Repulsions (15.5-15.9) The hydrogen atom Schrödinger equation is exactly solvable yielding the wavefunctions and orbitals of chemistry. Howev er, the Schrödinger
MANY ELECTRON ATOMS Chapter 15 Electron-Electron Repulsions (15.5-15.9) The hydrogen atom Schrödinger equation is exactly solvable yielding the wavefunctions and orbitals of chemistry. Howev er, the Schrödinger
Using the Periodic Table
 MATH SKILLS TRANSPARENCY WORKSHEET Using the Periodic Table 6 Use with Chapter 6, Section 6.2 1. Identify the number of valence electrons in each of the following elements. a. Ne e. O b. K f. Cl c. B g.
MATH SKILLS TRANSPARENCY WORKSHEET Using the Periodic Table 6 Use with Chapter 6, Section 6.2 1. Identify the number of valence electrons in each of the following elements. a. Ne e. O b. K f. Cl c. B g.
HANDOUT SET GENERAL CHEMISTRY II
 HANDOUT SET GENERAL CHEMISTRY II Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
HANDOUT SET GENERAL CHEMISTRY II Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
8. Relax and do well.
 CHEM 1515 Exam II John II. Gelder October 14, 1993 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last two pages include a periodic table, a
CHEM 1515 Exam II John II. Gelder October 14, 1993 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last two pages include a periodic table, a
CHM 101 PRACTICE TEST 1 Page 1 of 4
 CHM 101 PRACTICE TEST 1 Page 1 of 4 Please show calculations (stuffed equations) on all mathematical problems!! On the actual test, "naked answers, with no work shown, will receive no credit even if correct.
CHM 101 PRACTICE TEST 1 Page 1 of 4 Please show calculations (stuffed equations) on all mathematical problems!! On the actual test, "naked answers, with no work shown, will receive no credit even if correct.
Circle the letters only. NO ANSWERS in the Columns!
 Chemistry 1304.001 Name (please print) Exam 5 (100 points) April 18, 2018 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Chemistry 1304.001 Name (please print) Exam 5 (100 points) April 18, 2018 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Faculty of Natural and Agricultural Sciences Chemistry Department. Semester Test 1. Analytical Chemistry CMY 283. Time: 120 min Marks: 100 Pages: 6
 Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 120 min
Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 120 min
1 Genesis 1:1. Chapter 10 Matter. Lesson. Genesis 1:1 In the beginning God created the heavens and the earth. (NKJV)
 1 Genesis 1:1 Genesis 1:1 In the beginning God created the heavens and the earth. (NKJV) 1 Vocabulary Saturated having all the solute that can be dissolved at that temperature Neutron a particle with no
1 Genesis 1:1 Genesis 1:1 In the beginning God created the heavens and the earth. (NKJV) 1 Vocabulary Saturated having all the solute that can be dissolved at that temperature Neutron a particle with no
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
INSTRUCTIONS: CHEM Exam I. September 13, 1994 Lab Section
 CHEM 1314.05 Exam I John I. Gelder September 13, 1994 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
CHEM 1314.05 Exam I John I. Gelder September 13, 1994 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
Chemistry 2 Exam Roane State Academic Festival. Name (print neatly) School
 Name (print neatly) School There are fifteen question on this exam. Each question is weighted equally. n the answer sheet, write your name in the space provided and your answers in the blanks provided.
Name (print neatly) School There are fifteen question on this exam. Each question is weighted equally. n the answer sheet, write your name in the space provided and your answers in the blanks provided.
Speed of light c = m/s. x n e a x d x = 1. 2 n+1 a n π a. He Li Ne Na Ar K Ni 58.
 Physical Chemistry II Test Name: KEY CHEM 464 Spring 18 Chapters 7-11 Average = 1. / 16 6 questions worth a total of 16 points Planck's constant h = 6.63 1-34 J s Speed of light c = 3. 1 8 m/s ħ = h π
Physical Chemistry II Test Name: KEY CHEM 464 Spring 18 Chapters 7-11 Average = 1. / 16 6 questions worth a total of 16 points Planck's constant h = 6.63 1-34 J s Speed of light c = 3. 1 8 m/s ħ = h π
Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
HANDOUT SET GENERAL CHEMISTRY I
 HANDOUT SET GENERAL CHEMISTRY I Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
HANDOUT SET GENERAL CHEMISTRY I Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
INSTRUCTIONS: Exam III. November 10, 1999 Lab Section
 CHEM 1215 Exam III John III. Gelder November 10, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
CHEM 1215 Exam III John III. Gelder November 10, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
610B Final Exam Cover Page
 1 st Letter of Last Name NAME: 610B Final Exam Cover Page No notes or calculators of any sort allowed. You have 3 hours to complete the exam. CHEM 610B, 50995 Final Exam Fall 2003 Instructor: Dr. Brian
1 st Letter of Last Name NAME: 610B Final Exam Cover Page No notes or calculators of any sort allowed. You have 3 hours to complete the exam. CHEM 610B, 50995 Final Exam Fall 2003 Instructor: Dr. Brian
8. Relax and do well.
 CHEM 1314.03 Exam I John I. Gelder September 25, 1997 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
CHEM 1314.03 Exam I John I. Gelder September 25, 1997 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
Spin Cut-off Parameter of Nuclear Level Density and Effective Moment of Inertia
 Commun. Theor. Phys. (Beijing, China) 43 (005) pp. 709 718 c International Academic Publishers Vol. 43, No. 4, April 15, 005 Spin Cut-off Parameter of Nuclear Level Density and Effective Moment of Inertia
Commun. Theor. Phys. (Beijing, China) 43 (005) pp. 709 718 c International Academic Publishers Vol. 43, No. 4, April 15, 005 Spin Cut-off Parameter of Nuclear Level Density and Effective Moment of Inertia
Chapter 12 The Atom & Periodic Table- part 2
 Chapter 12 The Atom & Periodic Table- part 2 Electrons found outside the nucleus; negatively charged Protons found in the nucleus; positive charge equal in magnitude to the electron s negative charge Neutrons
Chapter 12 The Atom & Periodic Table- part 2 Electrons found outside the nucleus; negatively charged Protons found in the nucleus; positive charge equal in magnitude to the electron s negative charge Neutrons
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
PART 1 Introduction to Theory of Solids
 Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:1 Trim:165 240MM TS: Integra, India PART 1 Introduction to Theory of Solids Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:2
Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:1 Trim:165 240MM TS: Integra, India PART 1 Introduction to Theory of Solids Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:2
30 Zn(s) 45 Rh. Pd(s) Ag(s) Cd(s) In(s) Sn(s) white. 77 Ir. Pt(s) Au. Hg(l) Tl. 109 Mt. 111 Uuu. 112 Uub. 110 Uun. 65 Tb. 62 Sm. 64 Gd. 63 Eu.
 Enthalpy changes: experimentally it is much easier to measure heat flow at const pressure - this is enthalpy q p = )H : also nearly all chemical reactions are done at constant pressure. Enthalpy (heat)
Enthalpy changes: experimentally it is much easier to measure heat flow at const pressure - this is enthalpy q p = )H : also nearly all chemical reactions are done at constant pressure. Enthalpy (heat)
FINAL EXAM April 26, 2004
 CM 1045 (11:15 am Lecture) Dr. Light FINAL EXAM April 26, 2004 Name (please print) Check your recitation section: Sec. 21 5:30-6:20 pm (Popovic) Sec. 24 3:30-4:20 pm (Giunta) Sec. 22 6:30-7:20 pm (Popovic)
CM 1045 (11:15 am Lecture) Dr. Light FINAL EXAM April 26, 2004 Name (please print) Check your recitation section: Sec. 21 5:30-6:20 pm (Popovic) Sec. 24 3:30-4:20 pm (Giunta) Sec. 22 6:30-7:20 pm (Popovic)
K. 27 Co. 28 Ni. 29 Cu Rb. 46 Pd. 45 Rh. 47 Ag Cs Ir. 78 Pt.
 1 IA 1 H Hydrogen 1.01 Atomic number Element symbol Element name Atomic mass VIIIA 1 H 1.01 IIA IIIA IVA VA VIA VIIA 2 He 4.00 Metalloids 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 O 16.00 9 F
1 IA 1 H Hydrogen 1.01 Atomic number Element symbol Element name Atomic mass VIIIA 1 H 1.01 IIA IIIA IVA VA VIA VIIA 2 He 4.00 Metalloids 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 O 16.00 9 F
CHEM 10123/10125, Exam 2
 CHEM 10123/10125, Exam 2 March 7, 2012 (50 minutes) Name (please print) Please box your answers, and remember that significant figures, phases (for chemical equations), and units do count! 1. (13 points)
CHEM 10123/10125, Exam 2 March 7, 2012 (50 minutes) Name (please print) Please box your answers, and remember that significant figures, phases (for chemical equations), and units do count! 1. (13 points)
8. Relax and do well.
 CHEM 1215 Exam III John III. Gelder November 11, 1998 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
CHEM 1215 Exam III John III. Gelder November 11, 1998 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
 lectures accompanying the book: Solid State Physics: An Introduction, by Philip ofmann (2nd edition 2015, ISBN-10: 3527412824, ISBN-13: 978-3527412822, Wiley-VC Berlin. www.philiphofmann.net 1 Bonds between
lectures accompanying the book: Solid State Physics: An Introduction, by Philip ofmann (2nd edition 2015, ISBN-10: 3527412824, ISBN-13: 978-3527412822, Wiley-VC Berlin. www.philiphofmann.net 1 Bonds between
Made the FIRST periodic table
 Made the FIRST periodic table 1869 Mendeleev organized the periodic table based on the similar properties and relativities of certain elements Later, Henri Moseley organized the elements by increasing
Made the FIRST periodic table 1869 Mendeleev organized the periodic table based on the similar properties and relativities of certain elements Later, Henri Moseley organized the elements by increasing
Atomic Structure & Interatomic Bonding
 Atomic Structure & Interatomic Bonding Chapter Outline Review of Atomic Structure Atomic Bonding Atomic Structure Atoms are the smallest structural units of all solids, liquids & gases. Atom: The smallest
Atomic Structure & Interatomic Bonding Chapter Outline Review of Atomic Structure Atomic Bonding Atomic Structure Atoms are the smallest structural units of all solids, liquids & gases. Atom: The smallest
1 of 5 14/10/ :21
 X-ray absorption s, characteristic X-ray lines... 4.2.1 Home About Table of Contents Advanced Search Copyright Feedback Privacy You are here: Chapter: 4 Atomic and nuclear physics Section: 4.2 Absorption
X-ray absorption s, characteristic X-ray lines... 4.2.1 Home About Table of Contents Advanced Search Copyright Feedback Privacy You are here: Chapter: 4 Atomic and nuclear physics Section: 4.2 Absorption
Secondary Support Pack. be introduced to some of the different elements within the periodic table;
 Secondary Support Pack INTRODUCTION The periodic table of the elements is central to chemistry as we know it today and the study of it is a key part of every student s chemical education. By playing the
Secondary Support Pack INTRODUCTION The periodic table of the elements is central to chemistry as we know it today and the study of it is a key part of every student s chemical education. By playing the
[ ]:543.4(075.8) 35.20: ,..,..,.., : /... ;. 2-. ISBN , - [ ]:543.4(075.8) 35.20:34.
![[ ]:543.4(075.8) 35.20: ,..,..,.., : /... ;. 2-. ISBN , - [ ]:543.4(075.8) 35.20:34. [ ]:543.4(075.8) 35.20: ,..,..,.., : /... ;. 2-. ISBN , - [ ]:543.4(075.8) 35.20:34.](/thumbs/74/69911105.jpg) .. - 2-2009 [661.87.+661.88]:543.4(075.8) 35.20:34.2373-60..,..,..,..,.. -60 : /... ;. 2-. : -, 2008. 134. ISBN 5-98298-299-7 -., -,,. - «,, -, -», - 550800,, 240600 «-», -. [661.87.+661.88]:543.4(075.8)
.. - 2-2009 [661.87.+661.88]:543.4(075.8) 35.20:34.2373-60..,..,..,..,.. -60 : /... ;. 2-. : -, 2008. 134. ISBN 5-98298-299-7 -., -,,. - «,, -, -», - 550800,, 240600 «-», -. [661.87.+661.88]:543.4(075.8)
