Counterion Condensation of a Polyelectrolyte within a Micro/Nanochannel
|
|
|
- Myles Boyd
- 5 years ago
- Views:
Transcription
1 International Journal of Chemical Engineering and Applications, Vol. 7, No., April 6 Counterion Condensation of a Polyelectrolyte within a Micro/Nanochannel Cheng-Hsuan Huang, Yu-Fan Lee, and Eric Lee It also found many novel applications in different fields, such as drag delivery, phase separation in pharmacy and cosmetics, cell fixation in biotechnology, and the fabrication of soft materials in nanotechnology [4], [5]. Because of their fundamental importance in biology and biochemistry, polyelectrolytes have been the obect of a continued interest since the early days of polymer science. Electrostatic behavior is a key factor when investigating of a charged polyelectrolyte and that is fundamentally different from the conventional small particles. When the ions concentration in the bulk solution increases, the double layer surrounding the particle is suppressed and more counterions are driven into the interior of the porous particle. This phenomenon is referred to as the shielding effect by some researchers [6], as the strong Coulomb repulsive interaction between fixed charges is somehow shielded by the existence of counterions in between. Note that this shielding effect leads to the well-known counterion condensation, where the thermal energy of the counterions is insufficient for them to escape the electric field generated by the large number of charges on the macroion backbone [7]. The occurrence of shielding effect leads to a neutralization of the total charge of macroions, which is directly related to some important phenomena in biology, such as DNA condensation [8] and RNA folding [9]. DNA condensation, defined as the packing of random coiled DNA into small ordered structures, has been studied extensively. According to Zimm and Le Bret [], counterion condensation is a phenomenon which can be solidly accounted for based on the Poisson-Boltzmann equation, so the shielding effect here can be viewed as a conceptual extension of the physical condensation of counterions. Hsu and Lee [] studied a uniformly distributed charged polyelectrolyte by Poisson-Boltzmann equation, and their result successfully described the observed counterion condensation for a giant folded DNA, which cannot be interpreted by the famous theory proposed by Manning []. Yeh and Hsu [3] further proposed a ph-regulated model to show the ions (H+ and OH-) dissociated by water have significant influence on the effective charge of a polyelectrolyte. Thanks to the advances of the micro-/nanofabrication technology, microdevices with even smaller features can be produced now and the electrokinetic technique can be further downscaled to tens or hundreds of nanometers, allowing manipulation of even smaller biological particles, such as virus, bacteria, DNA, chromosomes, and other macromolecules [4]. Similar phenomena are observed in synthetic hydrogels or polymeric ultrafiltration membrane, where the porous membrane can be viewed as the numerous tiny micro/nanopores [5]. For example, the DNA sequencing Abstract Counterion condensation phenomenon of a highly charged polyelectrolyte is directly related to DNA (DeoxyriboNucleic Acid) condensation and RNA (RiboNucleic Acid) folding, which are research topics of vital interest in areas like biological and medical sciences as well as electrokinetic separations. With the advances of the modern micro/nanofabrication technology, practical applications are normally conducted in corresponding micro/nanochannels. As a result, a fundamental understanding of the counterion condensation phenomenon within a micro/nanochannel is crucial for successful operations utilizing it. We provide a theoretical analysis here with the polyelectrolye modelled as a charged porous sphere, and the full nonlinear Poisson equation is adopted to describe the interaction between the counterions in the suspending electrolyte solution and the backbone macroion of the polyelectrolyte itself. The solution is obtained numerically with a pseudo-spectral method based on Chebyshev polynomials. The degree of counterion condensation is expressed as a function of various parameters of interest, such as the charge condition of the polyelectrolyte, the ratio of the particle to pore radii, and the ionic strength of the suspending electrolyte solution. Results presented here provide useful information relevant in various practical applications such as biosensors, DNA stretching, and gene delivery. Index Terms Boundary confinement effect, counterion condensation, nanostructure, polyelectrolyte. I. INTRODUCTION Polyelectrolyte is a term first introduced by Fuoss [] to describe a substance of high molecular weight which is simultaneously an electrolyte. It is a macromolecule bearing a number of dissociable functional groups which can dissociate into charged polymer chains (macroions) and small counter-ions. Many organic and biological entities are polyelectrolytes, such as proteins, polysaccharides, DNA, and so on. Synthetic polymers in industry such as PAA (polyacrylic acid), PMA (polymethacrylic acid), and PAMAM (polyamidoamine) are polyelectrolytes as well []. The broad spectrum of variability of polyelectrolytes opens a lot of practical applications, for example, gene delivery has been accomplished using a variety of positively charged polyelectrolyte to form DNA complexes and transfect cultured cells with lower toxicities and higher efficiencies [3]. Manuscript received June, 5; revised August, 5. This work is partially supported by the Ministry of Science and Technology, Republic of China. C. H. Huang was with the Department of Chemical Engineering, National Taiwan University, Taipei, Taiwan. He is now with Taiwan Semiconductor Manufacturing Company ( hsuanhzb@tsmc.com). Y. F. Lee and E. Lee are with the Department of Chemical Engineering, National Taiwan University, Taipei, Taiwan ( r35438@ntu.edu.tw, ericlee@ntu.edu.tw). DOI:.7763/IJCEA.6.V
2 International Journal of Chemical Engineering and Applications, Vol. 7, No., April 6 technique utilize gel-electrophoresis very often, and Ma and Bloomfield [6] used agarose gel electrophoresis to measure the effective charge neutralization of DNA by counterions of different structure and valence, including Na+, Mg+, and Co(NH3)63+. In their measurements demonstrate that gel electrophoresis is a simple and surprisingly effective method to study the relative binding of counterions to DNA. Moreover, DNA mapping approach with DNA stretching technique has also gained much attention due to its low cost and efficiency [7]-[9]. Among this technique, the screening effect of Coulomb interaction is a crucial factor of the elongation of DNA, which has been explained in terms of self-avoidance. Therefore, it is important to consider aforementioned electrostatic phenomena to develop a comprehensive model of polyelectrolytes in micro/nanochannels. A series of researches results regarding colloidal particle conducting electrophoretic motion within a cylindrical channel has been reported in the literature [], []. To investigate the electrical properties of a polyelectrolyte, it has been common to treat a polyelectrolyte macromolecule as a charged colloid, and Overbeek [] indicated there are three possible ways to model a polyelectrolyte. Among them, both the coiled cylinder and the porous sphere models have been adopted by various research groups with success in different situations. In reality, according to a recent experimental study [3] on DNA electrophoresis, the actual image of a DNA molecule in free solution, obtained via an optical microscopy, is of a 3-D blob conformation closely resembling a porous sphere. Moreover, as indicated by Salieb-Beugelaar et al. [4], a DNA molecule do not have to deform when it enters a nanostructure of the dimensions larger than twice the radius of gyration, where the nanostructure can be a capillary, nanochannel, nanoslit or a pore matrix (gel). In addition, experimental observation of some specific protein particles by optical microscope demonstrates a porous 3-D blob shape as well, reported by Bell and Dunnill [4]. As a result, it is appropriate to treat the general polyelectrolytes in an electrolyte solution as charged porous spheres, say, 3-D blob shape. In this study, we investigated the counterion condensation phenomenon of a polyelectrolyte within a micro/nanochannel by solving the full nonlinear Poisson-Boltzmann equation, and the polyelectrolyte modeled as a charged porous sphere with a uniformly distributed constant fixed charge density. In microfluidic operations involving polyelectrolytes such as proteins, the boundary effect becomes important, so we focus on the impact on the effective charge density due to the presence of the micro/nanochannel. Parameters of electrokinetic interest are examined, such as the double layer thickness of the particle, the electric characteristic properties of the porous particle, the ratio of pore-to-particle radii, and so on. A pseudo-spectral method [5] based on Chebyshev polynomials is used to solve the resulting general electrokinetic equations numerically. This study provides important fundamental information about the counterion condensation or the shielding effect of charged porous sphere like polyelectrolytes in micro- and nanochannels, which is essential to the successful designs and operations like biosensors, LOC devices and DNA stretching channels frequently utilized in biochemical, biological, and other fields involving soft matters such as colloids and bio-related materials. Fig.. Schematic representation of the problem considered where a porous particle of radius a is placed on the axis of a long cylindrical pore of radius Rb. II. THEORY A. Full Nonlinear Poisson Boltzmann Equation As illustrated in Fig., we consider a porous spherical particle of radius a with fixed volumetric charge density ρfix in the center of a cylindrical channel of radius Rb. The cylindrical channel is filled with an aqueous solution containing z : z electrolytes, where z and z are the valences of cations and anions. The electroneutrality in the bulk liquid phase requires that n = n/α, where n and n being respectively the bulk concentrations of cations and anions and α = -z/z. The porous particle is penetrable for ions and fluid and carries functional groups which may dissociate to generate counterions. We assume that the electric permittivity ε takes the same value both inside and outside the polyelectrolyte particle. Spherical coordinates (r,, ) is adopted to describe the liquid phase and the porous structure, and the cylindrical coordinates (R,,Z) is applied to region of the cylindrical channel. The origin of the spherical coordinate is located at the center of the porous particle and the symmetric nature of the problem suggests that only half of the (r, ) domain needs to be considered. Based on Gauss's law, the electric potential and the space charge density ρ are related by Poisson equation: z en fix fix z en () () where, n, z, and e are the Laplace operator, the number concentration of the ionic species, the valence of the ionic species, and the elementary charge respectively. By setting the electric potential at zero where the ion concentration n equals its bulk value n, we further assume that the distribution of ions obeys Boltzmann distribution, viz., z e n n exp kbt (3) where kb is the Boltzmann constant and T is the absolute temperature. From ()-(3), we obtain the following Poisson-Boltzmann equation, 97
3 International Journal of Chemical Engineering and Applications, Vol. 7, No., April 6 z e fix n exp k BT (4) z e n exp k BT (5) A pseudo-spectral method based on Chebyshev polynomials is adopted here to solve the above equations associated with corresponding boundary conditions. To simplify the treatment, subsequent discussions are based on scaled symbols, i.e., the governing equations are rewritten in dimensionless form. The following symbols are chosen for the characteristic variables: the radius of porous particle, a, the thermal electric potential per valence of cations, (defined as kbt/ze), and the bulk concentration of the cations, n. Corresponding dimensionless variables are listed as follows: r* = r/a, R* = R/a, Z* = Z/a, n* = n/n, * = /, and Qfix = ρfixa/ε. In terms of dimensionless variables, the electric potential can be expressed as follows: B. Neutralization Fraction The driven-in space charges of counterions within the particle are regarded as if they are close enough to induce the physical condensation, hence neutralize the total amount of fixed charges to an effective value, Qeff, defined as z e z e a exp * exp * Q fix a * * exp * exp * *, * * (6) () n* r * sin d dr Q fix 4 *3 r 3 z Qeff z,. r * r * sin exp e* exp e* d dr * 4 r *3 a 3 r (3) Q fix * (7) The fraction of the fixed charges neutralized by these counterions is defined as a neutralization fraction: where κ- is the Debye screening length given by kbt / n (ez ) (8) (4) Q fix Qfix=5 The electric potential * remains symmetry at the center of the porous particle owing to the spherical symmetry. The cylindrical wall (R* = Rb* = Rb/a) is assumed chargeless. In addition, the axial symmetry condition requires that */ θ = at θ =. Infinitely far away from the sphere, the presence of the colloid should not be felt, hence the electric field * depending only on R*, the radius of the micro-/nanochannel. In other words, * = *(R*), where *(R*) is the electric potential distribution within the pore without the presence of a particle. Note that in practice, however, one cannot assume that both the inlet and outlet are infinitely far away from the sphere. Instead, a finite value of L/a is selected, with one-dimensional electric potential profile applied at both inlet (Z* = -L/a) and outlet (Z* = L/a). The solution is then considered to be convergent once the mobility thus calculated does not change with increasing value of L/a anymore. According to our calculation experience, L/a = 6 is long enough to ustify the convergence. Hence Z* = L/a = 6 is chosen here to be the outlet location throughout our analysis. The boundary conditions needed for the corresponding two-dimensional problem with the presence of a spherical particle are thus complete, and listed as follows:, r * Qeff.8.6 Rb* a Fig.. Neutralization fraction of fixed charges, θ, of a polyelectrolyte as a function of κa at different values of Rb* with Qfix = 5. Bold dashed: corresponding to results of a polyelectrolyte immersed in an unbounded electrolyte solution Rb*=..4 * *, * * R*, r*, (9) Qfix=5. R R* b, a L Z, a * () - - a Fig. 3. Boundary factor of fixed charges, Γ, of a polyelectrolyte as a function of κa at different values of Rb* with Qfix = 5. () 98
4 International Journal of Chemical Engineering and Applications, Vol. 7, No., April 6 counterions are driven into the interior of the porous sphere, hence neutralizes the fixed charges there, leading to a larger fraction. Up to 8% amount of charges inside the porous sphere are neutralized when the electric double layer is thin (κa = ). Due to the reduction of the effective charges of the porous particle, the gradient of electrical potential near the particle surface decrease, hence depressing an interior induced electric field. This is referred to as the abovementioned shielding effect [6], [7], [8]. The degree of the neutralization fraction depends on Rb* as well. The presence of the cylindrical channel squeezes the double layer surrounding the particle when it is thick hence affects the neutralization fraction by means of the electrostatic interaction. This phenomenon, however, tends to diminish when the double layer gets thinner, as shown in Fig.. Moreover, the smaller the radius of the cylindrical channel is, the lower the neutralization fraction of porous particle charges. To illustrate the influence of the radius of the cylindrical channel more specifically, we define the boundary factor Γ as: Fig. 4. Contour plots of the net amount of space charges, ρ*, with Qfix = 5, κa =. and (a) Rb* =.5. (b) Rb* =. III. RESULTS AND DISCUSSION A computer program developed by the authors employing a pseudo-spectral method is used to carry out all the numerical computations in this study. Standard mesh refinement test is conducted successfully to ensure the solution is grid-independent, a critical check for the convergence of a numerical solution in general. The accuracy and reliability of the present scheme is further verified by a direct comparison with the numerical results available in the literature for limiting cases, as reported by Hsu and Lee [] where a porous sphere with fixed volumetric charge density ρfix immersed in an unbounded electrolyte solution. We compare our calculation results with Hsu s predictions for the corresponding contour of the electric potential, setting the pore-to-particle radius, measured by Rb*, to be close to infinity, or very large (Say, Rb* = 5) to approach an unbounded case. The agreement is excellent. Less than.% deviations are observed overall. We thus conclude our calculation results are both accurate and reliable. Note that the dimensionless fixed charge density, Qfix (=ρfixa/ ) is function of the particle radius a. The actual range of Qfix in practice can go over several orders of magnitude as a result, depending on the specific particle radius a of interest. Nagasawa et al. [6] reported a Na-PVS polyelectrolyte system with Qfix smaller than. In reality, As far as the counterion condensation is concerned, Qfix = 5 is high enough to represent the electrostatic behavior of a porous particle in general. Moreover, to conform to the practical application and emphasize the boundary confinement effect of a cylindrical pore, the radius of channel is confined to the same order of magnitude of the porous particle size. We first investigate the impact on neutralization fraction of polyelectrolyte charges from the boundary confinement effect. Fig. depicts the neutralization fraction θ as a function of κa at various pore-to-particle radii, Rb*. Note that the bold-faced dashed line represents the results of Hsu and Lee in, and that κa represents the reciprocal of the double layer thickness in dimensionless form. Unlike a rigid particle with constant surface potential, the charged condition of a porous sphere is very sensitive to the concentration of the surrounding electrolyte ions in the solution. The higher the κa is, the more (5) where represents the neutralization fraction of a porous sphere immersed in an unbounded electrolyte solution, which corresponds to the bold dashed line in Fig.. Rebuilded form Fig., Fig. 3 depicting boundary factor Γ as a function of κa at various pore-to-particle radii, Rb*. Fig. 3 shows that when the channel is narrow, say (Rb* =.), the boundary affects up to 95% reduction of the neutralization fraction with κa =.. On the other hand, when the channel is wide, say (Rb* = ), the boundary affects the neutralization fraction weakly so that the boundary factor Γ almost approaches, which means the boundary effect is not significant. Fig. 5. Neutralization fraction of fixed charges, θ, of a polyelectrolyte as a function of Qfix at different values of Rb* with (a) κa =.. (b) κa =.. To explore the mechanism behind it, we present the contour plots of the net amount of space charges. As shown in Fig. 4, two representative values of pore-to-particle radii (Rb* =.5 and ) are adopted to compare their space charges (ρ*) distribution. This is because the confining boundary effect squeezes not only the original double layer surrounding the particle but also the extra coions outside the double layer, hence increasing the effective charge density of the porous 99
5 International Journal of Chemical Engineering and Applications, Vol. 7, No., April 6 more significant. As shown in Fig. 6, the effective charge density of a polyelectrolyte, Qeff, is depicted as a function of Qfix at different double layer thickness. When Qfix is low, straight lines indicating linear relationship between the effective charge density and the fixed charge density is observed, corresponding to the results of the Debye-Huckel approximation reported by Hermans and Fuita [9], valid for the low electric potential situation. When the Qfix goes higher, however, the nonlinear behavior takes place leading to the counterion condensation eventually which reduces the effective charge density significantly. By observing the transition from linear behavior to non-linear behavior, we can pinpoint approximately a critical value of fixed charge density corresponding to the occurrence of counterion condensation. Note also that this critical value is a function of κa as well, a parameter that can be manipulated by the experimentalists. The corresponding case of wider cylindrical wall is shown as well for comparison purpose in Fig. 7. The neutralization of fixed charge density of porous particle is essentially not affected by the presence of the nearby cylindrical channel, if the double layer is very thin (κa = ). On the other hand, the thicker the double layer, indicated by small values of κa, the more profound the boundary confinement effect in terms of effective charges reduction. This is understandable as the electrostatic impact from the presence of the micro/nanochannel is directly related to the degree of deformation of the double layer. sphere instead. Indeed the electrostatic behavior in a nanostructure is fundamentally different from the one in a microstructure. Up to 5% reduction of dimensionless space charge density ρ* is observed for narrower channel (Rb* =.5) to wider one (Rb* = ) at the center of the porous particle, which means less effective counterion inside the particle and hence leads to low neutralization fraction. This has direct impact in practical applications of biochemistry. For instance, by control of the neutralization of the total charges of macroions, people can manipulate the DNA condensation and RNA folding experimentally, which plays an important role in DNA vaccines and gene delivery. 8 R*b =.5 6 a =. Qeff a =. 4 a = a = 4 Qfix 6 8 Fig. 6. Variation of the effective charge density, Qeff, of the porous particle as a function of fixed charge density at various values of κa with Rb* =.5. Dashed lines: results valid for low electric potential only taking no account of counterion condensation. Closed circles correspond to the approximate critical values of fixed charge density where the counterion condensation takes place. To shed more light on the influence of the fixed charge density on particle s charge neutralization, we illustrate the neutralization fraction as a function of Qfix at various levels of pore-to-particle radii Rb*. Considering the effect of the electric double layer thickness on θ, we fixed the κa value for (a) κa =. and (b) κa =. respectively to represent thicker and thinner double layer thickness, as shown in Fig. 4. The neutralization fraction increases with increasing fixed charge density of porous particle monotonically. For smaller Qfix, the fixed charges of porous particle are too low to overcome strong Coulomb repulsive interaction between counterions, hence unable to attract more counterions and leading to θ approaches. When Qfix goes higher, however, more counterions would be attracted into porous sphere and neutralize more fixed charges, thus increasing the value of θ. It should be noted when Qfix gets very large, the neutralization fraction eventually approaches an upper limit value. This implies that the effect of counterions on the charge neutralization approaches a finite value with increasing Qfix. Similar phenomenon is observed as well in Fig. 5, where narrower cylindrical pore leads to smaller neutralization fraction due to more coions squeezing into porous particle. Moreover, comparing Fig. 5(a) and (b), it can be found when the concentration of electrolyte solution increases (higher κa), the neutralization fraction raise accordingly regardless of pore-to-particle radii. This is because the higher the κa is, the more counterions would be, hence neutralizing more fixed charge on porous particle and leading to the shielding effect Fig. 7. Variation of the effective charge density, Qeff, of the porous particle as a function of fixed charge density at various values of κa with Rb* =. Dashed lines: results valid for low electric potential only taking no account of counterion condensation. Closed circles correspond to the approximate critical values of fixed charge density where the counterion condensation takes place. IV. CONCLUSION Excellent agreement with numerical data of the corresponding limiting case indicates the reliability of this analysis, as well as the success of using the charged porous sphere to model a polyelectrolyte system. In this study, we introduce a general model to evaluate the neutralization fraction quantitatively for a highly charged polyelectrolyte, even if severe counterion condensation has already takes place, such as a giant DNA in the extremely folded compact state. Additionally, the narrower pore-to-particle radii, the more extra coions swept inside the porous sphere due to the
6 International Journal of Chemical Engineering and Applications, Vol. 7, No., April 6 confining boundary effect, hence decreasing the neutralization fraction of the porous sphere. Indeed the electrostatic behavior in a nanostructure is fundamentally different from the one in a microstructure. This has direct impact in practical applications of biochemistry. For instance, by the control the neutralization of the total charges of macroions, people can manipulate the DNA condensation and RNA folding experimentally, which plays an important role in DNA vaccines and gene delivery. ACKNOWLEDGMENT This work is partially supported by the Ministry of Science and Technology, Republic of China. REFERENCES [] R. M. Fuoss, "Polyelectrolytes," Science, vol. 8, pp , 948. [] F. B. Surani and Y. Qiao, "Energy absorption of a polyacrylic acid partial sodium salt-modified nanoporous system," Journal of Materials Research, vol., pp , 6. [3] C. C. Lee, J. A. MacKay, J. M. Fréchet, and F. C. Szoka, "Designing dendrimers for biological applications," Nature Biotechnology, vol. 3, pp , 5. [4] M. Shimomura and T. Sawadaishi, "Bottom-up strategy of materials fabrication: A new trend in nanotechnology of soft materials," Current Opinion in Colloid & Interface Science, vol. 6, pp. -6,. [5] A. B. Lowe and C. L. McCormick, Polyelectrolytes and Polyzwitterions: Synthesis, Properties, and Applications, 6. [6] H. Ohshima and T. Kondo, "Electrophoretic mobility and Donnan potential of a large colloidal particle with a surface charge layer," Journal of Colloid and Interface Science, vol. 6, pp. 35-3, 987. [7] U. Böhme and U. Scheler, "Counterion condensation and effective charge of poly (styrenesulfonate)," Advances in Colloid and Interface Science, vol. 58, pp ,. [8] E. Nordmeier, Macromolecular Symposia, Wiley Online Library, pp [9] V. L. Murthy and G. D. Rose, "Is counterion delocalization responsible for collapse in RNA folding?" Biochemistry, vol. 39, pp ,. [] M. L. Bret and B. H. Zimm, "Distribution of counterions around a cylindrical polyelectrolyte and Manning's condensation theory," Biopolymers, vol. 3, pp. 87-3, 984. [] H. P. Hsu and E. Lee, "Counterion condensation of a polyelectrolyte," Electrochemistry Communications, vol. 5, pp. 59-6,. [] G. S. Manning, "Limiting laws and counterion condensation in polyelectrolyte solutions I. Colligative properties," The Journal of Chemical Physics, vol. 5, pp , 969. [3] L. H. Yeh, J. P. Hsu, S. Qian, and S. Tseng, "Counterion condensation in ph-regulated polyelectrolytes," Electrochemistry Communications, vol. 9, pp. 97-,. [4] G. Salieb-Beugelaar, K. Dorfman, A. Van den Berg, and J. Eikel, "Electrophoretic separation of DNA in gels and nanostructures," Lab on a Chip, vol. 9, pp , 9. [5] P. Dechadilok and W. M. Deen, "Electrostatic and electrokinetic effects on hindered diffusion in pores," Journal of Membrane Science, vol. 336, pp. 7-6, 9. [6] C. Ma and V. A. Bloomfield, "Gel electrophoresis measurement of counterion condensation on DNA," Biopolymers, vol. 35, pp. -6, 995. [7] W. Reisner et al., "Nanoconfinement-enhanced conformational response of single DNA molecules to changes in ionic environment," Physical Review Letters, vol. 99, p. 583, 7. [8] C. Zhang, F. Zhang, J. A. van Kan, and J. R. Van der Maarel, "Effects of electrostatic screening on the conformation of single DNA molecules confined in a nanochannel," The Journal of Chemical Physics, vol. 8, p. 59, 8. [9] Y. Kim et al., "Nanochannel confinement: DNA stretch approaching full contour length," Lab on a Chip, vol., pp. 7-79,. [] C. H. Huang, H. P. Hsu, and E. Lee, "Electrophoretic motion of a charged porous sphere within micro-and nanochannels," Physical Chemistry Chemical Physics, vol. 4, pp ,. [] S. Tseng, C. Y. Lin, J. P. Hsu, and L. H. Yeh, "Electrophoresis of deformable polyelectrolytes in a nanofluidic channel," Langmuir, vol. 9, pp , 3. [] J. T. G. Overbeek, "Polyelectrolytes, past, present and future," Pure and Applied Chemistry, vol. 46, pp. 9-, 976. [3] J. Mannion and H. Craighead, "Nanofluidic structures for single biomolecule fluorescent detection," Biopolymers, vol. 85, pp. 3-43, 7. [4] D. Bell and P. Dunnill, "The influence of precipitation reactor configuration on the centrifugal recovery of isoelectric soya protein precipitate," Biotechnology and Bioengineering, vol. 4, pp , 98. [5] M. Y. Hussaini and T. A. Zang, "Spectral methods in fluid dynamics," Annual Review of Fluid Mechanics, vol. 9, pp , 987. [6] M. Nagasawa, A. Soda, and I. Kagawa, "Electrophoresis of polyelectrolyte in salt solutions," Journal of Polymer Science, vol. 3, pp , 958. [7] N. Imai and K. Iwasa, "Theory of electrophoresis of polyelectrolytes," Israel Journal of Chemistry, vol., pp. 3-33, 973. [8] Y. Y. He and E. Lee, "Electrophoresis in concentrated dispersions of charged porous spheres," Chemical Engineering Science, vol. 63, pp , 8. [9] J. Hermans and H. Fuita, "Electrophoresis of charged polymer molecules with partial free drainage," Proc. K. Ned. Akad. Wet., Ser. B: Phys. Sci., vol. 58, pp. 8-87, 955. Cheng-Hsuan Huang received his PhD degree in from National Taiwan University under the supervision of Prof. Eric Lee. Currently, he works in Taiwan Semiconductor Manufacturing Company in Taiwan. Yu-Fan Lee received his bachelor degree of chemical engineering from National Taiwan University in 4. Now, he studies for a master degree under the supervision of Prof. Eric Lee, and works as a teaching assistant of Intermediate Numerical Methods Course in National Taiwan University. Eric Lee is a professor of the Department of Chemical Engineering at National Taiwan University, Taipei, Taiwan. His research interests include the fluid mechanics of systems containing polymers, such as polymer solutions, polymer melts, and other non-newtonian fluids. The general electrokinetic behavior of colloidal particles of sub-micron or nano-scale dimensions is covered as well, focusing on the electrophoretic motion above all. The impact of air-water interface as well as particle migrations in gel or porous media is of particular interest in recent years.
Electrolyte Concentration Dependence of Ion Transport through Nanochannels
 Electrolyte Concentration Dependence of Ion Transport through Nanochannels Murat Bakirci mbaki001@odu.edu Yunus Erkaya yerka001@odu.edu ABSTRACT The magnitude of current through a conical nanochannel filled
Electrolyte Concentration Dependence of Ion Transport through Nanochannels Murat Bakirci mbaki001@odu.edu Yunus Erkaya yerka001@odu.edu ABSTRACT The magnitude of current through a conical nanochannel filled
 Number of pages in the question paper : 05 Number of questions in the question paper : 48 Modeling Transport Phenomena of Micro-particles Note: Follow the notations used in the lectures. Symbols have their
Number of pages in the question paper : 05 Number of questions in the question paper : 48 Modeling Transport Phenomena of Micro-particles Note: Follow the notations used in the lectures. Symbols have their
International Journal of Engineering & Technology IJET-IJENS Vol:18 No:03 1
 International Journal of Engineering & Technology IJET-IJENS Vol:18 No:03 1 Analytical Derivation of Diffusio-osmosis Electric Potential and Velocity Distribution of an Electrolyte in a Fine Capillary
International Journal of Engineering & Technology IJET-IJENS Vol:18 No:03 1 Analytical Derivation of Diffusio-osmosis Electric Potential and Velocity Distribution of an Electrolyte in a Fine Capillary
2 Structure. 2.1 Coulomb interactions
 2 Structure 2.1 Coulomb interactions While the information needed for reproduction of living systems is chiefly maintained in the sequence of macromolecules, any practical use of this information must
2 Structure 2.1 Coulomb interactions While the information needed for reproduction of living systems is chiefly maintained in the sequence of macromolecules, any practical use of this information must
8.592J HST.452J: Statistical Physics in Biology
 Assignment # 4 8.592J HST.452J: Statistical Physics in Biology Coulomb Interactions 1. Flory Theory: The Coulomb energy of a ball of charge Q and dimension R in d spacial dimensions scales as Q 2 E c.
Assignment # 4 8.592J HST.452J: Statistical Physics in Biology Coulomb Interactions 1. Flory Theory: The Coulomb energy of a ball of charge Q and dimension R in d spacial dimensions scales as Q 2 E c.
Electrophoretic Mobility within a Confining Well
 Electrophoretic Mobility within a Confining Well Tyler N. Shendruk Martin Bertrand Gary W. Slater University of Ottawa December 5, 2013 Free-Solution Electrophoresis: free-draining polyelectrolytes Free-Draining
Electrophoretic Mobility within a Confining Well Tyler N. Shendruk Martin Bertrand Gary W. Slater University of Ottawa December 5, 2013 Free-Solution Electrophoresis: free-draining polyelectrolytes Free-Draining
Electrophoretic Light Scattering Overview
 Electrophoretic Light Scattering Overview When an electric field is applied across an electrolytic solution, charged particles suspended in the electrolyte are attracted towards the electrode of opposite
Electrophoretic Light Scattering Overview When an electric field is applied across an electrolytic solution, charged particles suspended in the electrolyte are attracted towards the electrode of opposite
Potential changes of the cross section for rectangular microchannel with different aspect ratios
 Korean J. Chem. Eng., 24(1), 186-190 (2007) SHORT COMMUNICATION Potential changes of the cross section for rectangular microchannel with different aspect ratios Hyo Song Lee, Ki Ho Kim, Jae Keun Yu, Soon
Korean J. Chem. Eng., 24(1), 186-190 (2007) SHORT COMMUNICATION Potential changes of the cross section for rectangular microchannel with different aspect ratios Hyo Song Lee, Ki Ho Kim, Jae Keun Yu, Soon
al., 2000) to extend the previous numerical model to account for the effect of interfacial charges. This extension allows the analysis of the electrok
 The Journal of Engineering and Exact Sciences - JCEC ISSN: 2446-9416 Vol. 03 N. 03 (2017) 294 319 doi: https://doi.org/10.18540/2446941603032017294 OPEN ACCESS SIMULATION OF A PERFECTLY DIELECTRIC DROP
The Journal of Engineering and Exact Sciences - JCEC ISSN: 2446-9416 Vol. 03 N. 03 (2017) 294 319 doi: https://doi.org/10.18540/2446941603032017294 OPEN ACCESS SIMULATION OF A PERFECTLY DIELECTRIC DROP
Electrophoretic Deposition. - process in which particles, suspended in a liquid medium, migrate in an electric field and deposit on an electrode
 Electrophoretic Deposition - process in which particles, suspended in a liquid medium, migrate in an electric field and deposit on an electrode no redox differs from electrolytic in several ways deposit
Electrophoretic Deposition - process in which particles, suspended in a liquid medium, migrate in an electric field and deposit on an electrode no redox differs from electrolytic in several ways deposit
Nanopores and Nanofluidics for Single DNA Studies Derek Stein Department of Physics Brown University
 Nanopores and Nanofluidics for Single DNA Studies Derek Stein Department of Physics Brown University Overview Motivation: biological, physical, and technological The program: develop and characterize new
Nanopores and Nanofluidics for Single DNA Studies Derek Stein Department of Physics Brown University Overview Motivation: biological, physical, and technological The program: develop and characterize new
Effect of charged boundary on electrophoresis: Sphere in spherical cavity at arbitrary potential and double-layer thickness
 Journal of Colloid and Interface Science 314 (2007) 256 263 www.elsevier.com/locate/jcis Effect of charged boundary on electrophoresis: Sphere in spherical cavity at arbitrary potential and double-layer
Journal of Colloid and Interface Science 314 (2007) 256 263 www.elsevier.com/locate/jcis Effect of charged boundary on electrophoresis: Sphere in spherical cavity at arbitrary potential and double-layer
V = 2ze 2 n. . a. i=1
 IITS: Statistical Physics in Biology Assignment # 3 KU Leuven 5/29/2013 Coulomb Interactions & Polymers 1. Flory Theory: The Coulomb energy of a ball of charge Q and dimension R in d spacial dimensions
IITS: Statistical Physics in Biology Assignment # 3 KU Leuven 5/29/2013 Coulomb Interactions & Polymers 1. Flory Theory: The Coulomb energy of a ball of charge Q and dimension R in d spacial dimensions
Lecture 5: Macromolecules, polymers and DNA
 1, polymers and DNA Introduction In this lecture, we focus on a subfield of soft matter: macromolecules and more particularly on polymers. As for the previous chapter about surfactants and electro kinetics,
1, polymers and DNA Introduction In this lecture, we focus on a subfield of soft matter: macromolecules and more particularly on polymers. As for the previous chapter about surfactants and electro kinetics,
Dependence of Potential and Ion Distribution on Electrokinetic Radius in Infinite and Finite-length Nano-channels
 Presented at the COMSOL Conference 2008 Boston Dependence of Potential and Ion Distribution on Electrokinetic Radius in Infinite and Finite-length Nano-channels Jarrod Schiffbauer *,1, Josh Fernandez 2,
Presented at the COMSOL Conference 2008 Boston Dependence of Potential and Ion Distribution on Electrokinetic Radius in Infinite and Finite-length Nano-channels Jarrod Schiffbauer *,1, Josh Fernandez 2,
Multimedia : Boundary Lubrication Podcast, Briscoe, et al. Nature , ( )
 3.05 Nanomechanics of Materials and Biomaterials Thursday 04/05/07 Prof. C. Ortiz, MITDMSE I LECTURE 14: TE ELECTRICAL DOUBLE LAYER (EDL) Outline : REVIEW LECTURE #11 : INTRODUCTION TO TE ELECTRICAL DOUBLE
3.05 Nanomechanics of Materials and Biomaterials Thursday 04/05/07 Prof. C. Ortiz, MITDMSE I LECTURE 14: TE ELECTRICAL DOUBLE LAYER (EDL) Outline : REVIEW LECTURE #11 : INTRODUCTION TO TE ELECTRICAL DOUBLE
Origin of the Electrophoretic Force on DNA in Nanopores. Biological and Soft Systems - Cavendish Laboratory
 Origin of the Electrophoretic Force on DNA in Nanopores Ulrich F. Keyser Biological and Soft Systems - Cavendish Laboratory Acknowledgements Delft Cees Dekker, Nynke H. Dekker, Serge G. Lemay R. Smeets,
Origin of the Electrophoretic Force on DNA in Nanopores Ulrich F. Keyser Biological and Soft Systems - Cavendish Laboratory Acknowledgements Delft Cees Dekker, Nynke H. Dekker, Serge G. Lemay R. Smeets,
Module : 9 Electrophoretic Separation Methods
 Module : 9 Electrophoretic Separation Methods Dr. Sirshendu De Professor, Department of Chemical Engineering Indian Institute of Technology, Kharagpur e-mail: sde@che.iitkgp.ernet.in Keywords: Separation
Module : 9 Electrophoretic Separation Methods Dr. Sirshendu De Professor, Department of Chemical Engineering Indian Institute of Technology, Kharagpur e-mail: sde@che.iitkgp.ernet.in Keywords: Separation
Lecture 3 Charged interfaces
 Lecture 3 Charged interfaces rigin of Surface Charge Immersion of some materials in an electrolyte solution. Two mechanisms can operate. (1) Dissociation of surface sites. H H H H H M M M +H () Adsorption
Lecture 3 Charged interfaces rigin of Surface Charge Immersion of some materials in an electrolyte solution. Two mechanisms can operate. (1) Dissociation of surface sites. H H H H H M M M +H () Adsorption
From Polymer Gel Nanoparticles to Nanostructured Bulk Gels
 From Polymer Gel Nanoparticles to Nanostructured Bulk Gels Zhibing Hu Departments of Physics and Chemistry, University of North Texas Denton, TX 76203, U. S. A. Phone: 940-565 -4583, FAX: 940-565-4824,
From Polymer Gel Nanoparticles to Nanostructured Bulk Gels Zhibing Hu Departments of Physics and Chemistry, University of North Texas Denton, TX 76203, U. S. A. Phone: 940-565 -4583, FAX: 940-565-4824,
SELF-ASSEMBLY AND NANOTECHNOLOGY A Force Balance Approach
 SELF-ASSEMBLY AND NANOTECHNOLOGY A Force Balance Approach Yoon S. Lee Scientific Information Analyst Chemical Abstracts Service A Division of the American Chemical Society Columbus, Ohio WILEY A JOHN WILEY
SELF-ASSEMBLY AND NANOTECHNOLOGY A Force Balance Approach Yoon S. Lee Scientific Information Analyst Chemical Abstracts Service A Division of the American Chemical Society Columbus, Ohio WILEY A JOHN WILEY
Soft Matter and Biological Physics
 Dr. Ulrich F. Keyser - ufk20 (at) cam.ac.uk Soft Matter and Biological Physics Question Sheet Michaelmas 2011 Version: November 2, 2011 Question 0: Sedimentation Initially consider identical small particles
Dr. Ulrich F. Keyser - ufk20 (at) cam.ac.uk Soft Matter and Biological Physics Question Sheet Michaelmas 2011 Version: November 2, 2011 Question 0: Sedimentation Initially consider identical small particles
Impacts of Electroosmosis Forces on Surface-Tension- Driven Micro-Pumps
 Proceedings of the World Congress on Mechanical, Chemical, and Material Engineering (MCM 2015) Barcelona, Spain July 20-21, 2015 Paper No. 290 Impacts of Electroosmosis Forces on Surface-Tension- Driven
Proceedings of the World Congress on Mechanical, Chemical, and Material Engineering (MCM 2015) Barcelona, Spain July 20-21, 2015 Paper No. 290 Impacts of Electroosmosis Forces on Surface-Tension- Driven
Numerical Modeling of the Bistability of Electrolyte Transport in Conical Nanopores
 Numerical Modeling of the Bistability of Electrolyte Transport in Conical Nanopores Long Luo, Robert P. Johnson, Henry S. White * Department of Chemistry, University of Utah, Salt Lake City, UT 84112,
Numerical Modeling of the Bistability of Electrolyte Transport in Conical Nanopores Long Luo, Robert P. Johnson, Henry S. White * Department of Chemistry, University of Utah, Salt Lake City, UT 84112,
Application of COMSOL Multiphysics in Nanoscale Electrokinetic Transport
 High Performance Computing Day Application of COMSOL Multiphysics in Nanoscale Electrokinetic Transport Selcuk Atalay, PhD Candidate Advisor: Prof. Shizhi Qian Institute of Micro/Nanotechnology Aerospace
High Performance Computing Day Application of COMSOL Multiphysics in Nanoscale Electrokinetic Transport Selcuk Atalay, PhD Candidate Advisor: Prof. Shizhi Qian Institute of Micro/Nanotechnology Aerospace
Confinement of polymer chains and gels
 Confinement of polymer chains and gels Nefeli Georgoulia - Student number: 70732831 1 Introduction Confinement of polymer chains is significant in industrial as well as biological applications. For this
Confinement of polymer chains and gels Nefeli Georgoulia - Student number: 70732831 1 Introduction Confinement of polymer chains is significant in industrial as well as biological applications. For this
*blood and bones contain colloids. *milk is a good example of a colloidal dispersion.
 Chap. 3. Colloids 3.1. Introduction - Simple definition of a colloid: a macroscopically heterogeneous system where one component has dimensions in between molecules and macroscopic particles like sand
Chap. 3. Colloids 3.1. Introduction - Simple definition of a colloid: a macroscopically heterogeneous system where one component has dimensions in between molecules and macroscopic particles like sand
Applied Surfactants: Principles and Applications
 Applied Surfactants: Principles and Applications Tadros, Tharwat F. ISBN-13: 9783527306299 Table of Contents Preface. 1 Introduction. 1.1 General Classification of Surface Active Agents. 1.2 Anionic Surfactants.
Applied Surfactants: Principles and Applications Tadros, Tharwat F. ISBN-13: 9783527306299 Table of Contents Preface. 1 Introduction. 1.1 General Classification of Surface Active Agents. 1.2 Anionic Surfactants.
Key Concepts for section IV (Electrokinetics and Forces)
 Key Concepts for section IV (Electrokinetics and Forces) 1: Debye layer, Zeta potential, Electrokinetics 2: Electrophoresis, Electroosmosis 3: Dielectrophoresis 4: InterDebye layer force, VanDer Waals
Key Concepts for section IV (Electrokinetics and Forces) 1: Debye layer, Zeta potential, Electrokinetics 2: Electrophoresis, Electroosmosis 3: Dielectrophoresis 4: InterDebye layer force, VanDer Waals
Protein separation and characterization
 Address:800 S Wineville Avenue, Ontario, CA 91761,USA Website:www.aladdin-e.com Email USA: tech@aladdin-e.com Email EU: eutech@aladdin-e.com Email Asia Pacific: cntech@aladdin-e.com Protein separation
Address:800 S Wineville Avenue, Ontario, CA 91761,USA Website:www.aladdin-e.com Email USA: tech@aladdin-e.com Email EU: eutech@aladdin-e.com Email Asia Pacific: cntech@aladdin-e.com Protein separation
 Number of pages in the question paper : 06 Number of questions in the question paper : 48 Modeling Transport Phenomena of Micro-particles Note: Follow the notations used in the lectures. Symbols have their
Number of pages in the question paper : 06 Number of questions in the question paper : 48 Modeling Transport Phenomena of Micro-particles Note: Follow the notations used in the lectures. Symbols have their
Particle Characterization Laboratories, Inc.
 Analytical services Particle size analysis Dynamic Light Scattering Static Light Scattering Sedimentation Diffraction Zeta Potential Analysis Single Point Titration Isoelectric point determination Aqueous
Analytical services Particle size analysis Dynamic Light Scattering Static Light Scattering Sedimentation Diffraction Zeta Potential Analysis Single Point Titration Isoelectric point determination Aqueous
Analytical Technologies in Biotechnology Prof. Dr. Ashwani K. Sharma Department of Biotechnology Indian Institute of Technology, Roorkee
 Analytical Technologies in Biotechnology Prof. Dr. Ashwani K. Sharma Department of Biotechnology Indian Institute of Technology, Roorkee Module - 4 Electrophoresis Lecture - 1 Basis Concept in Electrophoresis
Analytical Technologies in Biotechnology Prof. Dr. Ashwani K. Sharma Department of Biotechnology Indian Institute of Technology, Roorkee Module - 4 Electrophoresis Lecture - 1 Basis Concept in Electrophoresis
Bchem 675 Lecture 9 Electrostatics-Lecture 2 Debye-Hückel: Continued Counter ion condensation
 Bchem 675 Lecture 9 Electrostatics-Lecture 2 Debye-Hückel: Continued Counter ion condensation Ion:ion interactions What is the free energy of ion:ion interactions ΔG i-i? Consider an ion in a solution
Bchem 675 Lecture 9 Electrostatics-Lecture 2 Debye-Hückel: Continued Counter ion condensation Ion:ion interactions What is the free energy of ion:ion interactions ΔG i-i? Consider an ion in a solution
The effect of surface dipoles and of the field generated by a polarization gradient on the repulsive force
 Journal of Colloid and Interface Science 263 (2003) 156 161 www.elsevier.com/locate/jcis The effect of surface dipoles and of the field generated by a polarization gradient on the repulsive force Haohao
Journal of Colloid and Interface Science 263 (2003) 156 161 www.elsevier.com/locate/jcis The effect of surface dipoles and of the field generated by a polarization gradient on the repulsive force Haohao
Proteins polymer molecules, folded in complex structures. Konstantin Popov Department of Biochemistry and Biophysics
 Proteins polymer molecules, folded in complex structures Konstantin Popov Department of Biochemistry and Biophysics Outline General aspects of polymer theory Size and persistent length of ideal linear
Proteins polymer molecules, folded in complex structures Konstantin Popov Department of Biochemistry and Biophysics Outline General aspects of polymer theory Size and persistent length of ideal linear
arxiv: v1 [cond-mat.soft] 16 Nov 2014
![arxiv: v1 [cond-mat.soft] 16 Nov 2014 arxiv: v1 [cond-mat.soft] 16 Nov 2014](/thumbs/91/106587590.jpg) Polyelectrolytes polarization in non-uniform electric fields Farnoush Farahpour 1, Fathollah Varnik 2, and Mohammad Reza Ejtehadi 1 1 Sharif University of Technology, Department of Physics, P.O. Box 11155-9161,
Polyelectrolytes polarization in non-uniform electric fields Farnoush Farahpour 1, Fathollah Varnik 2, and Mohammad Reza Ejtehadi 1 1 Sharif University of Technology, Department of Physics, P.O. Box 11155-9161,
Electrokinetic assembly and manipulation II Lecture by Chung, Jae-Hyun
 Electrokinetic assembly and manipulation II Lecture by Chung, Jae-Hyun Chung, Jae-Hyun, Mechanical Engineering, University of Washington Liu, Wing Kam, Mechanical Engineering, Northwestern University Liu,
Electrokinetic assembly and manipulation II Lecture by Chung, Jae-Hyun Chung, Jae-Hyun, Mechanical Engineering, University of Washington Liu, Wing Kam, Mechanical Engineering, Northwestern University Liu,
Chapter 12 - Modern Materials
 Chapter 12 - Modern Materials 12.1 Semiconductors Inorganic compounds that semiconduct tend to have chemical formulas related to Si and Ge valence electron count of four. Semiconductor conductivity can
Chapter 12 - Modern Materials 12.1 Semiconductors Inorganic compounds that semiconduct tend to have chemical formulas related to Si and Ge valence electron count of four. Semiconductor conductivity can
Molecular Driving Forces
 Molecular Driving Forces Statistical Thermodynamics in Chemistry and Biology SUBGfittingen 7 At 216 513 073 / / Ken A. Dill Sarina Bromberg With the assistance of Dirk Stigter on the Electrostatics chapters
Molecular Driving Forces Statistical Thermodynamics in Chemistry and Biology SUBGfittingen 7 At 216 513 073 / / Ken A. Dill Sarina Bromberg With the assistance of Dirk Stigter on the Electrostatics chapters
Inorganic compounds that semiconduct tend to have an average of 4 valence electrons, and their conductivity may be increased by doping.
 Chapter 12 Modern Materials 12.1 Semiconductors Inorganic compounds that semiconduct tend to have an average of 4 valence electrons, and their conductivity may be increased by doping. Doping yields different
Chapter 12 Modern Materials 12.1 Semiconductors Inorganic compounds that semiconduct tend to have an average of 4 valence electrons, and their conductivity may be increased by doping. Doping yields different
Lecture 18: Microfluidic MEMS, Applications
 MECH 466 Microelectromechanical Systems University of Victoria Dept. of Mechanical Engineering Lecture 18: Microfluidic MEMS, Applications 1 Overview Microfluidic Electrokinetic Flow Basic Microfluidic
MECH 466 Microelectromechanical Systems University of Victoria Dept. of Mechanical Engineering Lecture 18: Microfluidic MEMS, Applications 1 Overview Microfluidic Electrokinetic Flow Basic Microfluidic
Initial position, x p (0)/L
 .4 ) xp().2 ) ( 2L 2 xp Dc ( Displacement, /L.2.4.5.5 Initial position, x p ()/L Supplementary Figure Computed displacements of (red) positively- and (blue) negatively-charged particles at several CO 2
.4 ) xp().2 ) ( 2L 2 xp Dc ( Displacement, /L.2.4.5.5 Initial position, x p ()/L Supplementary Figure Computed displacements of (red) positively- and (blue) negatively-charged particles at several CO 2
Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015,
 Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015, Course,Informa5on, BIOC%530% GraduateAlevel,discussion,of,the,structure,,func5on,,and,chemistry,of,proteins,and, nucleic,acids,,control,of,enzyma5c,reac5ons.,please,see,the,course,syllabus,and,
Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015, Course,Informa5on, BIOC%530% GraduateAlevel,discussion,of,the,structure,,func5on,,and,chemistry,of,proteins,and, nucleic,acids,,control,of,enzyma5c,reac5ons.,please,see,the,course,syllabus,and,
Exchange of Counterions in DNA Condensation. Abstract
 Exchange of Counterions in DNA Condensation Yoshihiro Murayama and Masaki Sano Department of Physics, University of Tokyo, Tokyo 113-0033, Japan Abstract We measured the fluorescence intensity of DNA-bound
Exchange of Counterions in DNA Condensation Yoshihiro Murayama and Masaki Sano Department of Physics, University of Tokyo, Tokyo 113-0033, Japan Abstract We measured the fluorescence intensity of DNA-bound
ELECTROVISCOUS FLOW THROUGH A MICROFLUIDIC T-JUNCTION
 Ninth International Conference on CFD in the Minerals and Process Industries CSIRO, Melbourne, Australia 10-12 December 2012 ELECTROVISCOUS FLOW THROUGH A MICROFLUIDIC T-JUNCTION Joseph D. BERRY 1, Malcolm
Ninth International Conference on CFD in the Minerals and Process Industries CSIRO, Melbourne, Australia 10-12 December 2012 ELECTROVISCOUS FLOW THROUGH A MICROFLUIDIC T-JUNCTION Joseph D. BERRY 1, Malcolm
Regulació electrostàtica de canals microfluídics i porus biològics. Jordi Faraudo Institut de Ciència de Materials de Barcelona
 Regulació electrostàtica de canals microfluídics i porus biològics Jordi Faraudo Institut de Ciència de Materials de Barcelona A few (interesting?) examples of nanofluidic devices Electrostatic regulation
Regulació electrostàtica de canals microfluídics i porus biològics Jordi Faraudo Institut de Ciència de Materials de Barcelona A few (interesting?) examples of nanofluidic devices Electrostatic regulation
26.542: COLLOIDAL NANOSCIENCE & NANOSCALE ENGINEERING Fall 2013
 26.542: COLLOIDAL NANOSCIENCE & NANOSCALE ENGINEERING Fall 2013 Classes: Thurs, 6-9 pm; Ball Hall Room 208 Professor: Dr. B. Budhlall Office: Ball Hall 203B, Phone: 978-934-3414 Email: Bridgette_Budhlall@uml.edu
26.542: COLLOIDAL NANOSCIENCE & NANOSCALE ENGINEERING Fall 2013 Classes: Thurs, 6-9 pm; Ball Hall Room 208 Professor: Dr. B. Budhlall Office: Ball Hall 203B, Phone: 978-934-3414 Email: Bridgette_Budhlall@uml.edu
ISO Colloidal systems Methods for zetapotential. Part 1: Electroacoustic and electrokinetic phenomena
 INTERNATIONAL STANDARD ISO 13099-1 First edition 2012-06-15 Colloidal systems Methods for zetapotential determination Part 1: Electroacoustic and electrokinetic phenomena Systèmes colloïdaux Méthodes de
INTERNATIONAL STANDARD ISO 13099-1 First edition 2012-06-15 Colloidal systems Methods for zetapotential determination Part 1: Electroacoustic and electrokinetic phenomena Systèmes colloïdaux Méthodes de
Electrostatic Forces & The Electrical Double Layer
 Electrostatic Forces & The Electrical Double Layer Dry Clay Swollen Clay Repulsive electrostatics control swelling of clays in water LiquidSolid Interface; Colloids Separation techniques such as : column
Electrostatic Forces & The Electrical Double Layer Dry Clay Swollen Clay Repulsive electrostatics control swelling of clays in water LiquidSolid Interface; Colloids Separation techniques such as : column
Aqueous solutions. Solubility of different compounds in water
 Aqueous solutions Solubility of different compounds in water The dissolution of molecules into water (in any solvent actually) causes a volume change of the solution; the size of this volume change is
Aqueous solutions Solubility of different compounds in water The dissolution of molecules into water (in any solvent actually) causes a volume change of the solution; the size of this volume change is
Supporting Information for Lysozyme Adsorption in ph-responsive Hydrogel Thin-Films: The non-trivial Role of Acid-Base Equilibrium
 Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 215 Supporting Information for Lysozyme Adsorption in ph-responsive Hydrogel Thin-Films: The non-trivial
Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 215 Supporting Information for Lysozyme Adsorption in ph-responsive Hydrogel Thin-Films: The non-trivial
Colloidal Suspension Rheology Chapter 1 Study Questions
 Colloidal Suspension Rheology Chapter 1 Study Questions 1. What forces act on a single colloidal particle suspended in a flowing fluid? Discuss the dependence of these forces on particle radius. 2. What
Colloidal Suspension Rheology Chapter 1 Study Questions 1. What forces act on a single colloidal particle suspended in a flowing fluid? Discuss the dependence of these forces on particle radius. 2. What
Lectures 11-13: Electrostatics of Salty Solutions
 Lectures 11-13: Electrostatics of Salty Solutions Lecturer: Brigita Urbanc Office: 1-909 (E-mail: brigita@drexel.edu) Course website: www.physics.drexel.edu/~brigita/courses/biophys_011-01/ 1 Water as
Lectures 11-13: Electrostatics of Salty Solutions Lecturer: Brigita Urbanc Office: 1-909 (E-mail: brigita@drexel.edu) Course website: www.physics.drexel.edu/~brigita/courses/biophys_011-01/ 1 Water as
The Poisson-Nernst. Nernst-Planck (PNP) system for ion transport. Tai-Chia Lin National Taiwan University
 The Poisson-Nernst Nernst-Planck (PNP) system for ion transport Tai-Chia Lin National Taiwan University 3rd OCAMI-TIMS Workshop in Japan, Osaka, March 13-16, 2011 Background Ion transport is crucial in
The Poisson-Nernst Nernst-Planck (PNP) system for ion transport Tai-Chia Lin National Taiwan University 3rd OCAMI-TIMS Workshop in Japan, Osaka, March 13-16, 2011 Background Ion transport is crucial in
Colloid Chemistry. La chimica moderna e la sua comunicazione Silvia Gross.
 Colloid Chemistry La chimica moderna e la sua comunicazione Silvia Gross Istituto Dipartimento di Scienze di e Scienze Tecnologie Chimiche Molecolari ISTM-CNR, Università Università degli Studi degli Studi
Colloid Chemistry La chimica moderna e la sua comunicazione Silvia Gross Istituto Dipartimento di Scienze di e Scienze Tecnologie Chimiche Molecolari ISTM-CNR, Università Università degli Studi degli Studi
INTERMOLECULAR AND SURFACE FORCES
 INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
Supporting Information. DNA Manipulation and Separation in Sub-Lithographic Scale Nanowire Array
 Supporting Information DNA Manipulation and Separation in Sub-Lithographic Scale Nanowire Array Takao Yasui a *, Sakon Rahong b, Koki Motoyama a, Takeshi Yanagida b *, Qiong Wu a, Noritada Kaji a, Masaki
Supporting Information DNA Manipulation and Separation in Sub-Lithographic Scale Nanowire Array Takao Yasui a *, Sakon Rahong b, Koki Motoyama a, Takeshi Yanagida b *, Qiong Wu a, Noritada Kaji a, Masaki
CH676 Physical Chemistry: Principles and Applications. CH676 Physical Chemistry: Principles and Applications
 CH676 Physical Chemistry: Principles and Applications History of Nanotechnology: Time Line Democritus in ancient Greece: concept of atom 1900 : Rutherford : discovery of atomic nucleus The first TEM was
CH676 Physical Chemistry: Principles and Applications History of Nanotechnology: Time Line Democritus in ancient Greece: concept of atom 1900 : Rutherford : discovery of atomic nucleus The first TEM was
Stability of colloidal systems
 Stability of colloidal systems Colloidal stability DLVO theory Electric double layer in colloidal systems Processes to induce charges at surfaces Key parameters for electric forces (ζ-potential, Debye
Stability of colloidal systems Colloidal stability DLVO theory Electric double layer in colloidal systems Processes to induce charges at surfaces Key parameters for electric forces (ζ-potential, Debye
Liquid Chromatography
 Liquid Chromatography 1. Introduction and Column Packing Material 2. Retention Mechanisms in Liquid Chromatography 3. Method Development 4. Column Preparation 5. General Instrumental aspects 6. Detectors
Liquid Chromatography 1. Introduction and Column Packing Material 2. Retention Mechanisms in Liquid Chromatography 3. Method Development 4. Column Preparation 5. General Instrumental aspects 6. Detectors
Electrostatic Interactions in Mixtures of Cationic and Anionic Biomolecules: Bulk Structures and Induced Surface Pattern Formation
 Electrostatic Interactions in Mixtures of Cationic and Anionic Biomolecules: Bulk Structures and Induced Surface Pattern Formation Monica Olvera de la Cruz F. J. Solis, P. Gonzalez- Mozuleos (theory) E.
Electrostatic Interactions in Mixtures of Cationic and Anionic Biomolecules: Bulk Structures and Induced Surface Pattern Formation Monica Olvera de la Cruz F. J. Solis, P. Gonzalez- Mozuleos (theory) E.
Engineering Nanomedical Systems. Zeta Potential
 BME 695 Engineering Nanomedical Systems Lecture 7 Zeta Potential James F. Leary, Ph.D. SVM Endowed Professor of Nanomedicine Professor of Basic Medical Sciences and Biomedical Engineering Member: Purdue
BME 695 Engineering Nanomedical Systems Lecture 7 Zeta Potential James F. Leary, Ph.D. SVM Endowed Professor of Nanomedicine Professor of Basic Medical Sciences and Biomedical Engineering Member: Purdue
An electrokinetic LB based model for ion transport and macromolecular electrophoresis
 An electrokinetic LB based model for ion transport and macromolecular electrophoresis Raffael Pecoroni Supervisor: Michael Kuron July 8, 2016 1 Introduction & Motivation So far an mesoscopic coarse-grained
An electrokinetic LB based model for ion transport and macromolecular electrophoresis Raffael Pecoroni Supervisor: Michael Kuron July 8, 2016 1 Introduction & Motivation So far an mesoscopic coarse-grained
Untangling the Mechanics of Entangled Biopolymers
 Untangling the Mechanics of Entangled Biopolymers Rae M. Robertson-Anderson Physics Department University of San Diego students/postdocs: Cole Chapman, PhD Tobias Falzone, PhD Stephanie Gorczyca, USD 16
Untangling the Mechanics of Entangled Biopolymers Rae M. Robertson-Anderson Physics Department University of San Diego students/postdocs: Cole Chapman, PhD Tobias Falzone, PhD Stephanie Gorczyca, USD 16
arxiv: v1 [cond-mat.soft] 17 Sep 2016
![arxiv: v1 [cond-mat.soft] 17 Sep 2016 arxiv: v1 [cond-mat.soft] 17 Sep 2016](/thumbs/76/73264929.jpg) Polyelectrolyte Polypeptide Scaling Laws Via Mechanical and Dielectric Relaxation Measurements Jorge Monreal University of South Florida arxiv:1609.05358v1 [cond-mat.soft] 17 Sep 2016 Dated: March 1, 2018
Polyelectrolyte Polypeptide Scaling Laws Via Mechanical and Dielectric Relaxation Measurements Jorge Monreal University of South Florida arxiv:1609.05358v1 [cond-mat.soft] 17 Sep 2016 Dated: March 1, 2018
Quick guide to selecting columns and standards for Gel Permeation Chromatography and Size Exclusion Chromatography SELECTION GUIDE
 Quick guide to selecting columns and standards for Gel Permeation Chromatography and Size Exclusion Chromatography SELECTION GUIDE Introduction Gel permeation chromatography (GPC) and size exclusion chromatography
Quick guide to selecting columns and standards for Gel Permeation Chromatography and Size Exclusion Chromatography SELECTION GUIDE Introduction Gel permeation chromatography (GPC) and size exclusion chromatography
1. Poisson-Boltzmann 1.1. Poisson equation. We consider the Laplacian. which is given in spherical coordinates by (2)
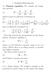 1. Poisson-Boltzmann 1.1. Poisson equation. We consider the Laplacian operator (1) 2 = 2 x + 2 2 y + 2 2 z 2 which is given in spherical coordinates by (2) 2 = 1 ( r 2 ) + 1 r 2 r r r 2 sin θ θ and in
1. Poisson-Boltzmann 1.1. Poisson equation. We consider the Laplacian operator (1) 2 = 2 x + 2 2 y + 2 2 z 2 which is given in spherical coordinates by (2) 2 = 1 ( r 2 ) + 1 r 2 r r r 2 sin θ θ and in
Journal of Colloid and Interface Science
 Journal of Colloid and Interface Science 35 (2011) 78 82 Contents lists available at ScienceDirect Journal of Colloid and Interface Science www.elsevier.com/locate/jcis Diffusion through colloidosome shells
Journal of Colloid and Interface Science 35 (2011) 78 82 Contents lists available at ScienceDirect Journal of Colloid and Interface Science www.elsevier.com/locate/jcis Diffusion through colloidosome shells
Surface Forces & Liquid Films (Answers to Exercise Problems)
 //5 Surface Forces & Liquid Films (nswers to Exercise Problems) Wuge H. Briscoe wuge.briscoe@bris.ac.uk URL: wugebrisco7.wix.com/briscoegroup Exercise : van der Waals forces & liquid films When octane
//5 Surface Forces & Liquid Films (nswers to Exercise Problems) Wuge H. Briscoe wuge.briscoe@bris.ac.uk URL: wugebrisco7.wix.com/briscoegroup Exercise : van der Waals forces & liquid films When octane
CBE2006(applied biology) is not included in major credits but included in graduation credits.
 Major The freshmen in all the engineering departments are required to take Seminar for Freshmen (HSS3014, 1 credits) course to be opened in the first semester of 1st grade. (applied from the class 2014)
Major The freshmen in all the engineering departments are required to take Seminar for Freshmen (HSS3014, 1 credits) course to be opened in the first semester of 1st grade. (applied from the class 2014)
Insulator-Based Dielectrophoretic Manipulation of DNA in a Microfluidic Device
 Insulator-Based Dielectrophoretic Manipulation of DNA in a Microfluidic Device Lin Gan 07/17/2015 1 Motivation Photo credit: (1) commons.wikimedia.org (2) h-a-y-s-t-a-c-k.net (3) joshmayo.com 2 Motivation
Insulator-Based Dielectrophoretic Manipulation of DNA in a Microfluidic Device Lin Gan 07/17/2015 1 Motivation Photo credit: (1) commons.wikimedia.org (2) h-a-y-s-t-a-c-k.net (3) joshmayo.com 2 Motivation
Charged Interfaces & electrokinetic
 Lecture Note #7 Charged Interfaces & electrokinetic phenomena Reading: Shaw, ch. 7 Origin of the charge at colloidal surfaces 1. Ionization Proteins acquire their charge by ionization of COOH and NH 2
Lecture Note #7 Charged Interfaces & electrokinetic phenomena Reading: Shaw, ch. 7 Origin of the charge at colloidal surfaces 1. Ionization Proteins acquire their charge by ionization of COOH and NH 2
Electroviscous Effects in Low Reynolds Number Flow through a Microfluidic Contraction with Rectangular Cross-Section
 PROCEEDINGS OF WORLD ACADEMY OF SCIENCE, ENGINEERING AND TECHNOLOGY VOLUME 3 JULY 28 ISSN 37-6884 Electroviscous Effects in Low Reynolds Number Flow through a Microfluidic Contraction with Rectangular
PROCEEDINGS OF WORLD ACADEMY OF SCIENCE, ENGINEERING AND TECHNOLOGY VOLUME 3 JULY 28 ISSN 37-6884 Electroviscous Effects in Low Reynolds Number Flow through a Microfluidic Contraction with Rectangular
Module 8: "Stability of Colloids" Lecture 38: "" The Lecture Contains: Calculation for CCC (n c )
 The Lecture Contains: Calculation for CCC (n c ) Relation between surface charge and electrostatic potential Extensions to DLVO theory file:///e /courses/colloid_interface_science/lecture38/38_1.htm[6/16/2012
The Lecture Contains: Calculation for CCC (n c ) Relation between surface charge and electrostatic potential Extensions to DLVO theory file:///e /courses/colloid_interface_science/lecture38/38_1.htm[6/16/2012
Polarizability-Dependent Induced-Charge. Electroosmotic Flow of Dielectric Particles and. Its Applications
 Polarizability-Dependent Induced-Charge Electroosmotic Flow of Dielectric Particles and Its Applications by Fang Zhang A thesis presented to the University of Waterloo in fulfillment of the thesis requirement
Polarizability-Dependent Induced-Charge Electroosmotic Flow of Dielectric Particles and Its Applications by Fang Zhang A thesis presented to the University of Waterloo in fulfillment of the thesis requirement
SEPARATION BY BARRIER
 SEPARATION BY BARRIER SEPARATION BY BARRIER Phase 1 Feed Barrier Phase 2 Separation by barrier uses a barrier which restricts and/or enhances the movement of certain chemical species with respect to other
SEPARATION BY BARRIER SEPARATION BY BARRIER Phase 1 Feed Barrier Phase 2 Separation by barrier uses a barrier which restricts and/or enhances the movement of certain chemical species with respect to other
V. Electrostatics Lecture 24: Diffuse Charge in Electrolytes
 V. Electrostatics Lecture 24: Diffuse Charge in Electrolytes MIT Student 1. Poisson-Nernst-Planck Equations The Nernst-Planck Equation is a conservation of mass equation that describes the influence of
V. Electrostatics Lecture 24: Diffuse Charge in Electrolytes MIT Student 1. Poisson-Nernst-Planck Equations The Nernst-Planck Equation is a conservation of mass equation that describes the influence of
Nanophysics: Main trends
 Nano-opto-electronics Nanophysics: Main trends Nanomechanics Main issues Light interaction with small structures Molecules Nanoparticles (semiconductor and metallic) Microparticles Photonic crystals Nanoplasmonics
Nano-opto-electronics Nanophysics: Main trends Nanomechanics Main issues Light interaction with small structures Molecules Nanoparticles (semiconductor and metallic) Microparticles Photonic crystals Nanoplasmonics
Single action pressing (from top)
 www.komage.de Single action pressing (from top) Double action pressing with fixed die Typical course of the pressure during pressing and ejection (Single action) Upper punch Pressure Lower punch Time Green
www.komage.de Single action pressing (from top) Double action pressing with fixed die Typical course of the pressure during pressing and ejection (Single action) Upper punch Pressure Lower punch Time Green
A Microfluidic Electroosmotic Mixer and the Effect of Potential and Frequency on its Mixing Efficiency
 Proceedings of the 009 IEEE International Conference on Systems, Man, and Cybernetics San Antonio, TX, USA - October 009 A Microfluidic Electroosmotic Mixer and the Effect of Potential and Frequency on
Proceedings of the 009 IEEE International Conference on Systems, Man, and Cybernetics San Antonio, TX, USA - October 009 A Microfluidic Electroosmotic Mixer and the Effect of Potential and Frequency on
Foundations of. Colloid Science SECOND EDITION. Robert J. Hunter. School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS
 Foundations of Colloid Science SECOND EDITION Robert J. Hunter School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS CONTENTS 1 NATURE OF COLLOIDAL DISPERSIONS 1.1 Introduction 1 1.2 Technological
Foundations of Colloid Science SECOND EDITION Robert J. Hunter School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS CONTENTS 1 NATURE OF COLLOIDAL DISPERSIONS 1.1 Introduction 1 1.2 Technological
Contents. Preface XI Symbols and Abbreviations XIII. 1 Introduction 1
 V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
Electrostatic Field Calculations for Electrophoresis Using Surfaces
 Mater. Res. Soc. Symp. Proc. Vol. 1061 2008 Materials Research Society 1061-MM09-25 Electrostatic Field Calculations for Electrophoresis Using Surfaces Perumal Ramasamy 1, Raafat M Elmaghrabi 2, and Gary
Mater. Res. Soc. Symp. Proc. Vol. 1061 2008 Materials Research Society 1061-MM09-25 Electrostatic Field Calculations for Electrophoresis Using Surfaces Perumal Ramasamy 1, Raafat M Elmaghrabi 2, and Gary
South pacific Journal of Technology and Science
 Zetasizer Technique Dr. Nagham Mahmood Aljamali Abstract: Assist. Professor in Organic Chemistry, Chemistry Department.,College of Education.,Kufa University.,IRAQ. In this review study., zetasizer technique
Zetasizer Technique Dr. Nagham Mahmood Aljamali Abstract: Assist. Professor in Organic Chemistry, Chemistry Department.,College of Education.,Kufa University.,IRAQ. In this review study., zetasizer technique
Anindya Aparajita, Ashok K. Satapathy* 1.
 μflu12-2012/24 NUMERICAL ANALYSIS OF HEAT TRANSFER CHARACTERISTICS OF COMBINED ELECTROOSMOTIC AND PRESSURE-DRIVEN FULLY DEVELOPED FLOW OF POWER LAW NANOFLUIDS IN MICROCHANNELS Anindya Aparajita, Ashok
μflu12-2012/24 NUMERICAL ANALYSIS OF HEAT TRANSFER CHARACTERISTICS OF COMBINED ELECTROOSMOTIC AND PRESSURE-DRIVEN FULLY DEVELOPED FLOW OF POWER LAW NANOFLUIDS IN MICROCHANNELS Anindya Aparajita, Ashok
Electrostatic Double Layer Force: Part III
 NPTEL Chemical Engineering Interfacial Engineering Module 3: Lecture 4 Electrostatic Double Layer Force: Part III Dr. Pallab Ghosh Associate Professor Department of Chemical Engineering IIT Guwahati, Guwahati
NPTEL Chemical Engineering Interfacial Engineering Module 3: Lecture 4 Electrostatic Double Layer Force: Part III Dr. Pallab Ghosh Associate Professor Department of Chemical Engineering IIT Guwahati, Guwahati
Electroosmotic Flow Mixing in a Microchannel
 *c: Unmarked Revised manuscript Click here to view linked References 1 1 1 0 1 0 1 0 1 0 1 0 1 Effects of Ionic Concentration Gradient on Electroosmotic Flow Mixing in a Microchannel Ran Peng and Dongqing
*c: Unmarked Revised manuscript Click here to view linked References 1 1 1 0 1 0 1 0 1 0 1 0 1 Effects of Ionic Concentration Gradient on Electroosmotic Flow Mixing in a Microchannel Ran Peng and Dongqing
arxiv: v1 [cond-mat.soft] 11 Apr 2015
![arxiv: v1 [cond-mat.soft] 11 Apr 2015 arxiv: v1 [cond-mat.soft] 11 Apr 2015](/thumbs/89/98626519.jpg) Electrophoretic Mobility of Polyelectrolytes within a Confining Well Tyler N. Shendruk, 1 Martin Bertrand, and Gary W. Slater 1 The Rudolf Peierls Centre for Theoretical Physics, Department of Physics,
Electrophoretic Mobility of Polyelectrolytes within a Confining Well Tyler N. Shendruk, 1 Martin Bertrand, and Gary W. Slater 1 The Rudolf Peierls Centre for Theoretical Physics, Department of Physics,
Ionic Behavior in Highly Concentrated Aqueous Solutions Nanoconfined between Discretely Charged Silicon Surfaces
 Ionic Behavior in Highly Concentrated Aqueous Solutions Nanoconfined between Discretely Charged Silicon Surfaces Yinghua Qiu, Jian Ma and Yunfei Chen* Jiangsu Key Laboratory for Design and Manufacture
Ionic Behavior in Highly Concentrated Aqueous Solutions Nanoconfined between Discretely Charged Silicon Surfaces Yinghua Qiu, Jian Ma and Yunfei Chen* Jiangsu Key Laboratory for Design and Manufacture
II. The physico-chemical properties of proteins
 II. The physico-chemical properties of proteins Proteins differ by there physical and chemical properties: Molecular mass Total electrical charge Termolability Solubility Molecular weight of the proteins
II. The physico-chemical properties of proteins Proteins differ by there physical and chemical properties: Molecular mass Total electrical charge Termolability Solubility Molecular weight of the proteins
Interfacial forces and friction on the nanometer scale: A tutorial
 Interfacial forces and friction on the nanometer scale: A tutorial M. Ruths Department of Chemistry University of Massachusetts Lowell Presented at the Nanotribology Tutorial/Panel Session, STLE/ASME International
Interfacial forces and friction on the nanometer scale: A tutorial M. Ruths Department of Chemistry University of Massachusetts Lowell Presented at the Nanotribology Tutorial/Panel Session, STLE/ASME International
SECOND PUBLIC EXAMINATION. Honour School of Physics Part C: 4 Year Course. Honour School of Physics and Philosophy Part C C7: BIOLOGICAL PHYSICS
 2757 SECOND PUBLIC EXAMINATION Honour School of Physics Part C: 4 Year Course Honour School of Physics and Philosophy Part C C7: BIOLOGICAL PHYSICS TRINITY TERM 2013 Monday, 17 June, 2.30 pm 5.45 pm 15
2757 SECOND PUBLIC EXAMINATION Honour School of Physics Part C: 4 Year Course Honour School of Physics and Philosophy Part C C7: BIOLOGICAL PHYSICS TRINITY TERM 2013 Monday, 17 June, 2.30 pm 5.45 pm 15
Continuous agglomerate model for identifying the solute- indifferent part of colloid nanoparticle's surface charge
 Journal of Physics: Conference Series PAPER OPEN ACCESS Continuous agglomerate model for identifying the solute- indifferent part of colloid nanoparticle's surface charge To cite this article: A V Alfimov
Journal of Physics: Conference Series PAPER OPEN ACCESS Continuous agglomerate model for identifying the solute- indifferent part of colloid nanoparticle's surface charge To cite this article: A V Alfimov
Introduction to the calculators in the Zetasizer software
 Introduction to the calculators in the Zetasizer software PARTICLE SIZE ZETA POTENTIAL MOLECULAR WEIGHT MOLECULAR SIZE Introduction The calculators are a series of tools in the Zetasizer software that
Introduction to the calculators in the Zetasizer software PARTICLE SIZE ZETA POTENTIAL MOLECULAR WEIGHT MOLECULAR SIZE Introduction The calculators are a series of tools in the Zetasizer software that
TECHNOLOGIES THAT TRANSFORM POLLUTANTS TO INNOCUOUS COMPONENTS: CHEMICAL AND PHYSICOCHEMICAL METHODS
 TECHNOLOGIES THAT TRANSFORM POLLUTANTS TO INNOCUOUS COMPONENTS: CHEMICAL AND PHYSICOCHEMICAL METHODS HUANG Xia Tsinghua University, Beijing, P.R. China Keywords: Pollutants, Innocuous Components, Chemical
TECHNOLOGIES THAT TRANSFORM POLLUTANTS TO INNOCUOUS COMPONENTS: CHEMICAL AND PHYSICOCHEMICAL METHODS HUANG Xia Tsinghua University, Beijing, P.R. China Keywords: Pollutants, Innocuous Components, Chemical
WJEC England GCSE Chemistry. Topic 5: Bonding, structure and properties. Notes. (Content in bold is for Higher Tier only)
 WJEC England GCSE Chemistry Topic 5: Bonding, structure and properties Notes (Content in bold is for Higher Tier only) Chemical bonds Compounds - substances in which 2 or more elements are chemically combined.
WJEC England GCSE Chemistry Topic 5: Bonding, structure and properties Notes (Content in bold is for Higher Tier only) Chemical bonds Compounds - substances in which 2 or more elements are chemically combined.
Some Commonly Neglected Issues Affect DEP Applications
 Some Commonly Neglected Issues Affect DEP Applications Vandana Pandian 1, Jozef Brcka 3, Jacques Faguet 3, Eric Lee 3, Guigen Zhang 1,2,4 * 1 Department of Bioengineering, Clemson University, Clemson,
Some Commonly Neglected Issues Affect DEP Applications Vandana Pandian 1, Jozef Brcka 3, Jacques Faguet 3, Eric Lee 3, Guigen Zhang 1,2,4 * 1 Department of Bioengineering, Clemson University, Clemson,
NUMERICAL INVESTIGATION OF THERMOCAPILLARY INDUCED MOTION OF A LIQUID SLUG IN A CAPILLARY TUBE
 Proceedings of the Asian Conference on Thermal Sciences 2017, 1st ACTS March 26-30, 2017, Jeju Island, Korea ACTS-P00786 NUMERICAL INVESTIGATION OF THERMOCAPILLARY INDUCED MOTION OF A LIQUID SLUG IN A
Proceedings of the Asian Conference on Thermal Sciences 2017, 1st ACTS March 26-30, 2017, Jeju Island, Korea ACTS-P00786 NUMERICAL INVESTIGATION OF THERMOCAPILLARY INDUCED MOTION OF A LIQUID SLUG IN A
Electrical double layer
 Electrical double layer Márta Berka és István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry http://dragon.unideb.hu/~kolloid/ 7. lecture Adsorption of strong electrolytes from
Electrical double layer Márta Berka és István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry http://dragon.unideb.hu/~kolloid/ 7. lecture Adsorption of strong electrolytes from
