Electrowetting on mineral and rock surfaces
|
|
|
- Diane Wright
- 5 years ago
- Views:
Transcription
1 GEOPHYSICAL RESEARCH LETTERS, VOL. 35, L06402, doi: /2007gl032865, 2008 Electrowetting on mineral and rock surfaces F. M. Francisca, 1 D. Fratta, 2 and H. F. Wang 3 Received 3 December 2007; revised 5 February 2008; accepted 18 February 2008; published 20 March [1] Electrowetting was investigated as a method to influence fluid movement on mineral or rock surfaces. The experimental setup was to place a small drop of water containing an electrolyte on a solid surface directly or on the same solid surface coated with a hydrophobic film. The contact angle decreased with an applied electric field until it reached a saturation value associated with a critical voltage. The electrowetting experiments were conducted again after surfactant was added to the water drop. In all cases the contact angle decreased with increasing voltage and surfactant concentration. Electrowetting enhanced the wetting effect over that of the surfactant alone both by lowering the contact angle and by promoting rupture of the hydrophobic film. Four zones of behavior were identified based on the surfactant concentration and the voltage level. The results open the possibility that electrowetting can be used to enhance mobilization of one fluid relative to another in hydrocarbon recovery or contaminant remediation, as interfacial tensions control multiphase fluid flow. Citation: Francisca, F. M., D. Fratta, and H. F. Wang (2008), Electrowetting on mineral and rock surfaces, Geophys. Res. Lett., 35, L06402, doi: /2007gl Introduction [2] In the past several decades, many techniques have been proposed and implemented to enhance oil recovery and increase the efficiency of decontamination processes [Lake, 1989; Sahimi, 1993]. Recovery and remediation techniques include the injection of high pressure fluid, steam or gas, or the addition of bacteria, brine or surfaceactive agents to promote immiscible fluid displacement [Finnerty and Singer, 1983; Stokes et al., 1986; Pennell et al., 1993; Wasan and Nikolov, 2003]. For a given pore structure and fluid pressure, the detachment, mobilization, and displacement of trapped oil depends on the contact angle and the interfacial tension [Stokes et al., 1986; Adamson and Gast, 1997]. [3] Interfacial tensions determine the shape of macroscopic drops of liquids in contact with a solid flat surface. At equilibrium, the drop satisfies the following condition (Figure 1a; Young model): cosðq 0 Þ ¼ g SV g 0 SL g LV 1 Universidad Nacional de Córdoba and Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Córdoba, Argentina. 2 Geological Engineering Program, University of Wisconsin-Madison, Madison, Wisconsin, USA. 3 Geology and Geophysics, University of Wisconsin-Madison, Madison, Wisconsin, USA. Copyright 2008 by the American Geophysical Union /08/2007GL ð1þ where q is the initial liquid-vapor and solid surface contact angle, g 0 SL is the intrinsic solid-liquid interfacial tension, g LV is the liquid-vapor interfacial tension and g SV is the solidvapor interfacial tension. The wetted part of the solid substrate is delimited by a contact line along which the three phases are in contact. The final shape of the drop depends on the interaction between the free energy change needed to expand the area of each interface and the physical-chemical interaction between phases. As a result, q 0 is a measure of the ability of each fluid phase to wet the solid surface. [4] Any interacting chemical or physical effect capable of modifying interfacial tensions induces changes in q 0 (e.g., adding surfactants or increasing the fluid temperature [Adamson and Gast, 1997]). Decreasing the solid-liquid interfacial tension g SL favors the displacement of the contact line and the liquid-gas interface [Finnerty and Singer, 1983]. The same principle applies for any two immiscible liquid phases in physical-chemical equilibrium. Of special interest to us is that the physical-chemical equilibrium is altered by applying an electric field between the solid and an electrolyte [Verjheijen and Prins, 1999; Prins et al., 2001; Quillet and Berge, 2001]. Accumulation of hydrated ions at the liquid-solid interface reduces interfacial tension [Lippmann, 1875; Rosslee and Abbott, 2000] and promotes the rupture of hydrocarbon films coating mineral surfaces [Sharma and Reiter, 1996]. The wetting of a dielectric solid surface is favored because g SL is lowered according to the equation [Mugele and Baret, 2005; Berry et al., 2006]: g SL ¼ g 0 SL 1 2 CV2 where C = e o e/d is the capacitance per unit area, e o is the permittivity of vacuum, e is the relative static permittivity of the solid phase, d is the dielectric thickness, and V is the applied voltage. Because of the influence of V on g SL,the size of the wetted area increases leading to a new equilibrium position and contact angle q (Figure 1b; Lippmann-Young model [Mugele and Baret, 2005; Berry et al., 2006]): cosðþ¼cos q ðq 0 Þþ e 0 e V 2 2 g LV d [5] This phenomenon, known as electrowetting, is independent of ionic concentration [Verjheijen and Prins, 1999; Kuo et al., 2003] but can only reduce q to a voltage-saturated value [Peykov et al., 2000; Mugele and Baret, 2005]. This saturation of the contact angle, which is sometimes explained by the ionization of the non-wetting fluid at the contact line [Vallet et al., 1999] or by the nonperfect dielectric behavior of solids [Shapiro et al., 2003], ð2þ ð3þ L of5
2 takes place at the critical voltage (V crit ) [Quinn et al., 2005]: Vcrit 2 ¼ 2d g SV g LV cosðq 0 Þ e 0 e [6] Here we show that the wettability [de Gennes, 1985; Morrow, 1990] of mineral and rock surfaces can be significantly enhanced by the process of electrowetting [Verjheijen and Prins, 1999; Quillet and Berge, 2001; Kang, 2002; Hayes and Feenstra, 2003; Kuo et al., 2003; Shapiro et al., 2003; Mugele and Baret, 2005]. We demonstrate that the combined contribution of electrowetting and detergents reduces contact angle by an additional 24% over the value obtained using surface-active agents alone. These results provide a new conceptual methodology for the improved removal of petroleum hydrocarbons from mineral surfaces. 2. Methods [7] Thin sections were prepared from six mineral (quartz, calcite, barite, plagioclase, talc, and hematite) and six rock (marble, gneiss, granite, basalt, shale, and limestone) specimens and glued to a glass slide covered with a thin film of conductive silver paint (Figure 1). The final thicknesses and surfaces were obtained by polishing the specimens with 6 mm silicon carbide grinding wheels. Before testing, all specimens were washed with deionized water, oven dried until achieving a constant weight, and then kept at room temperature for 24 hrs. Each thin section was placed on a horizontal surface with the testing surface facing up. A drop of one of three different electrolytes was placed on the testing surface with a calibrated micropipette. The two electrodes were connected to a 313A Bertan high voltage DC power supply. The electrolyte drop geometry was documented with a pixel digital camera and the contact angle was obtained using digital image processing. The right and left angles formed by the tangent line to ð4þ the drop and the solid surface were obtained by means of an image analysis algorithm and verified manually. The initial contact angle was measured after a period of stabilization of 4 min. Some specimens were coated with a hydrophobic film to analyze the effect of voltage on film rupture and wettability. All specimen surfaces were examined under a Fisher Scientific Stereomaster microscope (magnification 20 to 40) in order to verify the film rupture after testing. 3. Results [8] Here, we focus on phase interactions of electrolytes with natural mineral and rock surfaces with the goal of increasing the wetting of these materials. Figure 2 shows the influence of V on the normalized change of cos(q) for three different electrolytes over a limestone surface. A good agreement with equation (3) is observed. The increasing deviation between the model response and the experimental data at electrical potentials lower than 200V and higher than +200 V corresponds to the voltage saturation of the contact angle. [9] No significant asymmetry is observed when the polarity of the electrodes is reversed (Figure 1b). In this case, the negatively charged Cl ions move toward the steel wire, while Na + and Mg 2+ ions move toward the solid surface. The radii of these hydrated ions are: Cl = nm, Na + = nm, and Mg 2+ = nm. We observed (Figure 2) little effect of valence or hydrated size of the ions on the saturated value of the contact angle, about a 15% increase over cos(q 0 ). [10] We next conducted a series of experiments of wettability on a hydrophobic surface. Pure mineral and rock surfaces were coated with petrolatum gel (i.e., commercially Figure 1. Equilibrium position for sessile drops. (a) Young contact angle; (b) Lippmann-Young contact angle and principle of electrowetting. The two electrodes are the conductive silver paint under the solid dielectric and the stainless steel wire connected to the fluid drop. The wire diameter is 0.64 mm. Figure 2. Electrowetting phenomenon on a limestone surface. Influence of voltage on the relative change of contact angles for drops of 0.1 mol/l and 1.0 mol/l NaCl and 1.0 mol/l MgCl 2 solutions. The solid line shows the expected behavior according to the Lippmann-Young model. The limestone thickness is 400 mm and the limestone relative static permittivity is 7.3. Positive and negative voltages were tested on different electrolytes drops. Similar trends were observed on calcite, barite, and marble specimens. 2of5
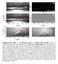
3 Figure 3. Wetting changes induced by the applied electric field: influence of the applied voltage on cos(q) for mineral and rock specimens coated with a petrolatum film. The petrolatum consists of non-polar iso- and cyclo-paraffins with some alkylated aromatic hydrocarbons. The film thickness is approximately 10 mm and the mineral and rock thicknesses are 400 mm. The solid lines show the behavior predicted by the Lippmann-Young model. The initial q measured before the application of the petrolatum film were 59.5, 55.1, 15.1, 57.9, 10.8, and 59.0 for the barite, calcite, quartz, marble, granite, and limestone specimens, respectively. The volume of the drops shown in the pictures is 4 ml. The coefficient of variation for the measured q is 3%. (a) Without surfactant: Drops are a 0.1 mol/l NaCl solution (The experimental data and the model response for the limestone specimen without the petrolatum film is shown as a reference behavior for untreated surfaces); (b) With surfactant: The liquid drop is a 0.1 mol/l NaCl solution with 2% w/w of detergent (a coconut-based anionic surfactant alcohols, (C12 14), ethoxylated, monoethers with sulphuric acid, and sodium salts ). available petroleum jelly) rendering hydrophobic specimens (q ). We observed that the initial contact angles remained constant with time and no changes in the hydrophobic film were detected (specimens were observed for 15 min under a microscope in order to verify equilibrium conditions). In Figure 3a electrowetting is responsible for the continuous reduction of q at low voltages. When electric potentials in the V range, or 900 V in the case of limestone, were applied, we observed a sharp increase in cos(q) to values higher than those predicted by equation (3). The step changes are associated with rupture of the hydrophobic film caused by the electrical attraction of hydrated ions toward the dielectric surface, which reduces q and causes an enlargement of the wetted solid surface [Hayes and Feenstra, 2003]. The rupture of the hydrophobic film was confirmed by observing the specimens under a microscope. The observed gaps show how the electrolyte liquid reaches the contaminated solid surface and solid-fluid-petrolatum contact lines develop. The higher voltage required for limestone can be attributed to a relatively high attraction between this material and the film [Basu and Sharma, 1996; Sharma and Reiter, 1996; Bergeron, 1999] and to the smoothness of the limestone surface with an absence of discontinuities (as observed under the microscope). Above the rupture voltage cos(q) increases slowly toward an asymptotic value as voltage increases. [11] A surfactant decreases g SL and g LV therefore reducing the macroscopic contact angle (when the concentration is lower than the critical micelle concentration CMC [Adamson and Gast, 1997]). We found that cos(q) at 0V increases from 0.3 to the +0.5 to +0.7 range in all petrolatum-covered mineral and rock specimens when a detergent is added to the electrolyte (Figure 3b), Also, the film ruptures a few seconds after the fluid drop is placed on the petrolatum film and the contact line needs almost 4 minutes to reach a new equilibrium position. Thereafter, a small electrowetting increase in cos(q)of0.1 is observed as V increases, which is likely produced by the accumulation of electrical charges near the solid surface and the 3of5
4 Figure 4. Schematic illustration of the simultaneous effect of surfactant and electrowetting on q. The relative importance of voltage and surfactant is determined from the change in the contact angle. The region boundaries are established by the electrowetting contact angle saturation voltage and the critical micelle concentration. The electrowetting contact angle saturation voltage increases with greater surfactant concentration (boundary between Zones I and II below CMC). consequent reduction of g SL [Lippmann, 1875; Verjheijen and Prins, 1999; Moon et al., 2002]. 4. Discussion [12] We first present a qualitative analysis of the simultaneous effect of surfactant and electrowetting on the contact angle (Figure 4). Based on over one hundred experimental runs, we identified four different zones with distinctive behavior. The region boundaries are established by the electrowetting contact angle saturation voltage [Quinn et al., 2005] and the critical micelle concentration - CMC [Adamson and Gast, 1997]. Zone I: Low voltage (V < V crit ) and low surfactant concentration, C < CMC: q is reduced by increasing the applied voltage or the surfactant concentration. Zone II:V>V crit, C < CMC: only the addition of surfactants can enhance the wetting of solid surfaces. Zone III: V<V crit, C > CMC: q is only susceptible to voltage changes. Zone IV: V>V crit, C > CMC: no further reduction in q is possible. The slope of the Zone I and Zone II boundary shows the effect of the two contributing phenomena. At surfactant concentrations less than CMC, the surfactant decreases both g SL and g LV reducing the macroscopic contact angle [Adamson and Gast, 1997], and the decrease in g SL also contributes to the reduction of q by electrowetting and the increase in the value of V crit (Equation 4 [Quinn et al., 2005]). These effects cease to contribute above the CMC and the slope of the V crit boundary changes as shown in Figure 4. [13] We can recognize these four regions in the experimental results shown in Figure 3. The tests begin in zone I and move toward zone II by increasing the applied voltage and reducing q until the contact angles reach saturation in the surfactant-free electrolyte (Figure 3a). Figure 3b shows the transition of q from zone I to III when the fluid drop is treated with the detergent and the external electric field is zero. Thereafter, as the voltage increases, q moves from zone III to IV after surpassing V crit and become saturated at a contact angle that is the combined effect of both electrowetting saturation and critical micelle concentration. [14] We have shown through experimental evidence that the combined contribution of electrowetting and surfactants helps to increase the wetted surface of natural mineral and rock surfaces. The measured contact angles decrease significantly due to the simultaneous contribution of electric voltage and detergent. The contact angle decreases 44.6% when electrowetting acts alone and it decreases 51.5% by the sole action of the detergent with respect to q 0. The decrease of q is 75.5% when the electric voltage and detergent concentrations are both increased. Hence, the additional effect due to electrowetting is equivalent to an additional decrease of 24% in the contact angle. The change in cos(q) caused by the electrowetting phenomenon shown in Figure 3b corresponds to an increase in the capillary pressure Dp c / g LV [cos(q) cos(q 0 )] of approximately 40%. Furthermore, the accumulation of electrical charges at the liquid-solid interfaces caused by the electrowetting phenomenon promotes the rupture of hydrocarbon films coating mineral surfaces [Sharma and Reiter, 1996]. These results give novel possibilities to the removal of petroleum hydrocarbons and to the wetting of mineral surfaces by electrified liquids. The combined application of electrowetting phenomena and surfactant agents could be the basis for the development of new quaternary oil recovery techniques. [15] Acknowledgments. This research was supported in part by a Fulbright-CONICET Award and the University of Wisconsin-Madison. References Adamson, A. W., and A. P. Gast (1997), Physical Chemistry of Surfaces, John Wiley, New York. Basu, S., and M. M. Sharma (1996), Measurement of critical disjoining pressure for dewetting of solid surfaces, J. Colloid Interface Sci., 181, Bergeron, V. (1999), Forces and structure in thin liquid soap films, J. Phys. I, 11, R215 R238. 4of5
5 Berry, S., J. Kedzierski, and B. Abedian (2006), Low voltage electrowetting using thin fluoropolymer films, J. Colloid Interface Sci., 303, de Gennes, P. G. (1985), Wetting: Statics and dynamics, Rev. Mod. Phys, 57, Finnerty, W. R., and M. E. Singer (1983), Microbial enhancement of oil recovery, Nature Biotechnol., 1, Hayes, R. A., and B. J. Feenstra (2003), Video-speed electronic paper based on electrowetting, Nature, 425, Kang, K. H. (2002), How electrostatic fields change contact angle in electrowetting, Langmuir, 18, 10,318 10,322. Kuo, J. S., P. Spicar-Mihalic, I. Rodriguez, and D. T. Chiu (2003), Electrowetting-induced droplet movement in an immiscible medium, Langmuir, 19, Lake, L. W. (1989), Enhanced Oil Recovery, Prentice-Hall, New York. Lippmann, G. (1875), Relations entre les phénomènes électriques et capillaires, Ann. Chim. Phys, 5, Moon, H., S. K. Cho, R. L. Garrell, and C. J. Kim (2002), Low voltage electrowetting-on-dielectric, J. Appl. Phys., 92, Morrow, N. R. (1990), Wettability and its effect on oil recovery, J. Pet. Technol., 42, Mugele, F., and J. C. Baret (2005), Electrowetting: From basic to applications, J. Phys. Condens. Matter, 17, R705 R774. Pennell, K. D., L. M. Abriola, and W. Weber (1993), Surfactant enhanced solubilization of residual dodecane in soil columns: 1. Experimental investigation, Environ. Sci. Technol., 27, Peykov, V., A. Quinn, and J. Ralston (2000), Electrowetting: A model for contact-angle saturation, Colloid Polym. Sci., 278, Prins, M. W. J., W. J. J. Welters, and J. W. Weekamp (2001), Fluid control in multichannel structures by electrocapillary pressure, Science, 291, Quillet, C., and B. Berge (2001), Electrowetting: A recent outbreak, Curr. Opinion Colloid Interface Sci., 6, Quinn, A., R. Sedev, and J. Ralson (2005), Contact angle saturation in electrowetting, J. Phys. Chem. B, 109, Rosslee, C., and N. L. Abbott (2000), Active control of interfacial properties, Curr. Opinion Colloid Interface Sci., 5, Sahimi, M. (1993), Flow phenomena in rocks: From continuum models to fractals, percolation, cellular automata, and simulated annealing, Rev. Mod. Phys., 65, Shapiro, B., H. Moon, R. L. Garrell, and C. J. Kim (2003), Equilibrium behavior of sessile drops under surface tension, applied external fields and material variations, J. Appl. Phys., 93, Sharma, A., and G. Reiter (1996), Instability of thin polymer films on coated substrates: Rupture, dewetting, and drop formation, J. Colloid Interface Sci., 178, Stokes, J. P., D. A. Weitz, J. P. Gollub, A. Dougherty, M. O. Robbins, P. M. Chaikin, and H. M. Lindsay (1986), Interfacial stability of immiscible displacement in a porous medium, Phys. Rev. Lett., 57, Vallet, M., M. Vallade, and B. Berge (1999), Limiting phenomena for the spreading of water on polymer films by electrowetting, Eur. Phys. J. B, 11, Verjheijen, H. J. J., and M. W. J. Prins (1999), Reversible electrowetting and trapping charge: Model and experiments, Langmuir, 15, Wasan, D. T., and A. D. Nikolov (2003), Spreading of nanofluids on solids, Nature, 423, F. M. Francisca, Universidad Nacional de Córdoba and Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Av. Velez Sarsfield 1611, CP 5016, Córdoba, Argentina. (ffrancis@efn.uncor.edu) D. Fratta, Geological Engineering Program, University of Wisconsin- Madison, 1415 Engineering Drive, Madison, WI 53706, USA. (fratta@ wisc.edu) H. F. Wang, Geology and Geophysics, University of Wisconsin-Madison, 1215 W Dayton St., Madison, WI 53706, USA. (wang@geology.wisc.edu) 5of5
Electrowetting on dielectrics on lubricating fluid based slippery surfaces with negligible hysteresis
 Electrowetting on dielectrics on lubricating fluid based slippery surfaces with negligible hysteresis J. Barman a, A. K. Nagarajan b and K. Khare a* a Department of Physics, Indian Institute of Technology
Electrowetting on dielectrics on lubricating fluid based slippery surfaces with negligible hysteresis J. Barman a, A. K. Nagarajan b and K. Khare a* a Department of Physics, Indian Institute of Technology
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Dispensing nano-pico droplets and liquid patterning by pyroelectrodynamic shooting P. Ferraro, S. Coppola, S. Grilli, M. Paturzo and V. Vespini Theoretical simulation of temperature
SUPPLEMENTARY INFORMATION Dispensing nano-pico droplets and liquid patterning by pyroelectrodynamic shooting P. Ferraro, S. Coppola, S. Grilli, M. Paturzo and V. Vespini Theoretical simulation of temperature
Electrowetting. space and ε l the liquid dielectric constant, Eq. (1) can be written as. γ = ε 0ε l 2d V2. (2)
 600 Electrowetting and hence a reduced flow rate in the pressure-drop direction. This reduced flow rate seems to suggest that the liquid have an apparent higher viscosity. The apparent viscosity is called
600 Electrowetting and hence a reduced flow rate in the pressure-drop direction. This reduced flow rate seems to suggest that the liquid have an apparent higher viscosity. The apparent viscosity is called
Wettability of CaCO 3 surfaces
 Colloids and Surfaces A: Physicochemical and Engineering Aspects 157 (1999) 333 340 www.elsevier.nl/locate/colsurfa Wettability of CaCO 3 surfaces Malvina G. Orkoula a,b, Petros G. Koutsoukos a,b, *, Michel
Colloids and Surfaces A: Physicochemical and Engineering Aspects 157 (1999) 333 340 www.elsevier.nl/locate/colsurfa Wettability of CaCO 3 surfaces Malvina G. Orkoula a,b, Petros G. Koutsoukos a,b, *, Michel
drops in motion Frieder Mugele the physics of electrowetting and its applications Physics of Complex Fluids University of Twente
 drops in motion the physics of electrowetting and its applications Frieder Mugele Physics of Complex Fluids niversity of Twente 1 electrowetting: the switch on the wettability voltage outline q q q q q
drops in motion the physics of electrowetting and its applications Frieder Mugele Physics of Complex Fluids niversity of Twente 1 electrowetting: the switch on the wettability voltage outline q q q q q
The Origins of Surface and Interfacial Tension
 The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
How Electrostatic Fields Change Contact Angle in Electrowetting
 038 Langmuir 2002, 8, 038-0322 How Electrostatic Fields Change Contact Angle in Electrowetting Kwan Hyoung Kang* Department of Mechanical Engineering, Pohang University of Science and Technology, San 3,
038 Langmuir 2002, 8, 038-0322 How Electrostatic Fields Change Contact Angle in Electrowetting Kwan Hyoung Kang* Department of Mechanical Engineering, Pohang University of Science and Technology, San 3,
Smart lens: tunable liquid lens for laser tracking
 Smart lens: tunable liquid lens for laser tracking Fan-Yi Lin a, Li-Yu Chu b, Yu-Shan Juan a, Sih-Ting Pan b, Shih-Kang Fan* b a Institute of Photonics Technologies, Department of Electrical Engineering,
Smart lens: tunable liquid lens for laser tracking Fan-Yi Lin a, Li-Yu Chu b, Yu-Shan Juan a, Sih-Ting Pan b, Shih-Kang Fan* b a Institute of Photonics Technologies, Department of Electrical Engineering,
Electrowetting based microliter drop tensiometer. Arun G. Banpurkar* Kevin Nichols and Frieder Mugele
 Electrowetting based microliter drop tensiometer Arun G. Banpurkar* Kevin Nichols and Frieder Mugele Physics of Complex Fluids, Faculty of Science and Technology, IMPACT and MESA+ Institute, University
Electrowetting based microliter drop tensiometer Arun G. Banpurkar* Kevin Nichols and Frieder Mugele Physics of Complex Fluids, Faculty of Science and Technology, IMPACT and MESA+ Institute, University
Supplementary Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
INVESTIGATION OF THE EFFECT OF TEMPERATURE AND PRESSURE ON INTERFACIAL TENSION AND WETTABILITY
 SCA2018_062 INVESTIGATION OF THE EFFECT OF TEMPERATURE AND PRESSURE ON INTERFACIAL TENSION AND WETTABILITY Taha M. Okasha Saudi Aramco EXPEC Advanced Research Center, Dhahran, Saudi Arabia This paper was
SCA2018_062 INVESTIGATION OF THE EFFECT OF TEMPERATURE AND PRESSURE ON INTERFACIAL TENSION AND WETTABILITY Taha M. Okasha Saudi Aramco EXPEC Advanced Research Center, Dhahran, Saudi Arabia This paper was
Solid-liquid interface
 Lecture Note #9 (Spring, 2017) Solid-liquid interface Reading: Shaw, ch. 6 Contact angles and wetting Wetting: the displacement from a surface of one fluid by another. A gas is displaced by a liquid at
Lecture Note #9 (Spring, 2017) Solid-liquid interface Reading: Shaw, ch. 6 Contact angles and wetting Wetting: the displacement from a surface of one fluid by another. A gas is displaced by a liquid at
Unique Fluid Ensemble including Silicone Oil for the Application Bull. Korean Chem. Soc. 2008, Vol. 29, No Articles
 Unique Fluid Ensemble including Silicone Oil for the Application Bull. Korean Chem. Soc. 2008, Vol. 29, No. 4 731 Articles Unique Fluid Ensemble including Silicone Oil for the Application of Optical Liquid
Unique Fluid Ensemble including Silicone Oil for the Application Bull. Korean Chem. Soc. 2008, Vol. 29, No. 4 731 Articles Unique Fluid Ensemble including Silicone Oil for the Application of Optical Liquid
Wettability of carbonate reservoir minerals under carbon storage conditions
 TCCS-9 Wettability of carbonate reservoir minerals under carbon storage conditions Dr Mihaela Stevar and Prof Martin Trusler 13 June 2017 1 Outline Background Objectives http://www.hydrobead.com/ Experimental
TCCS-9 Wettability of carbonate reservoir minerals under carbon storage conditions Dr Mihaela Stevar and Prof Martin Trusler 13 June 2017 1 Outline Background Objectives http://www.hydrobead.com/ Experimental
Direct Oil Recovery from Saturated Carbon Nanotube Sponges
 Supporting Information Direct Oil Recovery from Saturated Carbon Nanotube Sponges Xiying Li 1, Yahui Xue 1, Mingchu Zou 2, Dongxiao Zhang 3, Anyuan Cao 2,* 1, 4,*, and Huiling Duan 1 State Key Laboratory
Supporting Information Direct Oil Recovery from Saturated Carbon Nanotube Sponges Xiying Li 1, Yahui Xue 1, Mingchu Zou 2, Dongxiao Zhang 3, Anyuan Cao 2,* 1, 4,*, and Huiling Duan 1 State Key Laboratory
Electrowetting-Induced Dewetting Transitions on Superhydrophobic Surfaces
 Purdue University Purdue e-pubs Birck and NCN Publications Birck Nanotechnology Center 9-6-2011 Electrowetting-Induced Dewetting Transitions on Superhydrophobic Surfaces Niru Kumari Birck Nanotechnology
Purdue University Purdue e-pubs Birck and NCN Publications Birck Nanotechnology Center 9-6-2011 Electrowetting-Induced Dewetting Transitions on Superhydrophobic Surfaces Niru Kumari Birck Nanotechnology
Analysis and Measurement of Forces in an Electrowetting-Driven Oscillator
 Mater. es. Soc. Symp. Proc. Vol. 15 8 Materials esearch Society 15-DD8-1 Analysis and Measurement of Forces in an Electrowetting-Driven Oscillator Nathan Brad Crane 1, Alex A Volinsky 1, Vivek amadoss
Mater. es. Soc. Symp. Proc. Vol. 15 8 Materials esearch Society 15-DD8-1 Analysis and Measurement of Forces in an Electrowetting-Driven Oscillator Nathan Brad Crane 1, Alex A Volinsky 1, Vivek amadoss
Praktikum zur. Materialanalytik
 Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
An Experimental Investigation of EOR Mechanisms for Nanoparticles Fluid in Glass Micromodel
 1 / 12 An Experimental Investigation of EOR Mechanisms for Nanoparticles Fluid in Glass Micromodel Shidong Li and Ole Torsæter, Norwegian University of Science and Technology (NTNU) This paper was prepared
1 / 12 An Experimental Investigation of EOR Mechanisms for Nanoparticles Fluid in Glass Micromodel Shidong Li and Ole Torsæter, Norwegian University of Science and Technology (NTNU) This paper was prepared
Dielectrowetting: Statics and Dynamics
 Dielectrowetting: Statics and Dynamics Glen McHale University of Northumbria Carl V. Brown, Nottingham Trent University Naresh Sampara, Gary G. Wells, Michael I. Newton 8 th Intl. Electrowetting Workshop,
Dielectrowetting: Statics and Dynamics Glen McHale University of Northumbria Carl V. Brown, Nottingham Trent University Naresh Sampara, Gary G. Wells, Michael I. Newton 8 th Intl. Electrowetting Workshop,
Physics and Chemistry of Interfaces
 Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Jacco Snoeijer PHYSICS OF FLUIDS
 Jacco Snoeijer PHYSICS OF FLUIDS dynamics dynamics freezing dynamics freezing microscopics of capillarity Menu 1. surface tension: thermodynamics & microscopics 2. wetting (statics): thermodynamics & microscopics
Jacco Snoeijer PHYSICS OF FLUIDS dynamics dynamics freezing dynamics freezing microscopics of capillarity Menu 1. surface tension: thermodynamics & microscopics 2. wetting (statics): thermodynamics & microscopics
films in low voltage electrowetting
 Superior performance of multilayered fluoropolymer films in low voltage electrowetting Dimitrios P. Papageorgiou 1, Angeliki Tserepi 2, Andreas G. Boudouvis 1, Athanasios G. Papathanasiou 1* 1 School of
Superior performance of multilayered fluoropolymer films in low voltage electrowetting Dimitrios P. Papageorgiou 1, Angeliki Tserepi 2, Andreas G. Boudouvis 1, Athanasios G. Papathanasiou 1* 1 School of
Part II: Self Potential Method and Induced Polarization (IP)
 Part II: Self Potential Method and Induced Polarization (IP) Self-potential method (passive) Self-potential mechanism Measurement of self potentials and interpretation Induced polarization method (active)
Part II: Self Potential Method and Induced Polarization (IP) Self-potential method (passive) Self-potential mechanism Measurement of self potentials and interpretation Induced polarization method (active)
WETTABILITY CHANGE TO GAS-WETNESS IN POROUS MEDIA
 WETTABILITY CHANGE TO GAS-WETNESS IN POROUS MEDIA Kewen Li and Abbas Firoozabadi Reservoir Engineering Research Institute (RERI) Abstract In the petroleum literature, gas is assumed to be the non-wetting
WETTABILITY CHANGE TO GAS-WETNESS IN POROUS MEDIA Kewen Li and Abbas Firoozabadi Reservoir Engineering Research Institute (RERI) Abstract In the petroleum literature, gas is assumed to be the non-wetting
STATIC AND SPONTANEOUS ELECTROWETTING
 Brief Review Modern Physics Letters B, Vol. 19, No. 12 (2005) 549 569 c World Scientific Publishing Company STATIC AND SPONTANEOUS ELECTROWETTING LESLIE Y. YEO and HSUEH-CHIA CHANG Department of Chemical
Brief Review Modern Physics Letters B, Vol. 19, No. 12 (2005) 549 569 c World Scientific Publishing Company STATIC AND SPONTANEOUS ELECTROWETTING LESLIE Y. YEO and HSUEH-CHIA CHANG Department of Chemical
Changes of polymer material wettability by surface discharge
 Changes of polymer material wettability by surface discharge Surface discharge and material treatment Surface treatment of materials in low temperature plasma belongs to the modern and very perspective
Changes of polymer material wettability by surface discharge Surface discharge and material treatment Surface treatment of materials in low temperature plasma belongs to the modern and very perspective
Capillarity and Wetting Phenomena
 ? Pierre-Gilles de Gennes Frangoise Brochard-Wyart David Quere Capillarity and Wetting Phenomena Drops, Bubbles, Pearls, Waves Translated by Axel Reisinger With 177 Figures Springer Springer New York Berlin
? Pierre-Gilles de Gennes Frangoise Brochard-Wyart David Quere Capillarity and Wetting Phenomena Drops, Bubbles, Pearls, Waves Translated by Axel Reisinger With 177 Figures Springer Springer New York Berlin
Effect of Jatropha Bio-Surfactant on Residual Oil during Enhanced Oil Recovery Process
 Effect of Jatropha Bio-Surfactant on Residual Oil during Enhanced Oil Recovery Process 1 Ojo., T.I and 1 Fadairo., A. S, 1 Deparment of Petroleum Engineering, Covenant University, Nigeria. Abstract Surfactants
Effect of Jatropha Bio-Surfactant on Residual Oil during Enhanced Oil Recovery Process 1 Ojo., T.I and 1 Fadairo., A. S, 1 Deparment of Petroleum Engineering, Covenant University, Nigeria. Abstract Surfactants
Module 4: "Surface Thermodynamics" Lecture 21: "" The Lecture Contains: Effect of surfactant on interfacial tension. Objectives_template
 The Lecture Contains: Effect of surfactant on interfacial tension file:///e /courses/colloid_interface_science/lecture21/21_1.htm[6/16/2012 1:10:36 PM] Surface Thermodynamics: Roles of Surfactants and
The Lecture Contains: Effect of surfactant on interfacial tension file:///e /courses/colloid_interface_science/lecture21/21_1.htm[6/16/2012 1:10:36 PM] Surface Thermodynamics: Roles of Surfactants and
Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
Kinematical description of the spontaneous imbibition processes
 Proceedings of the 5th IASME / WSEAS International Conference on Fluid Mechanics and Aerodynamics, Athens, Greece, August 25-27, 2007 209 Kinematical description of the spontaneous imbibition processes
Proceedings of the 5th IASME / WSEAS International Conference on Fluid Mechanics and Aerodynamics, Athens, Greece, August 25-27, 2007 209 Kinematical description of the spontaneous imbibition processes
Frieder Mugele. Physics of Complex Fluids. University of Twente. Jacco Snoeier Physics of Fluids / UT
 coorganizers: Frieder Mugele Physics of Comple Fluids Jacco Snoeier Physics of Fluids / UT University of Twente Anton Darhuber Mesoscopic Transport Phenomena / Tu/e speakers: José Bico (ESPCI Paris) Daniel
coorganizers: Frieder Mugele Physics of Comple Fluids Jacco Snoeier Physics of Fluids / UT University of Twente Anton Darhuber Mesoscopic Transport Phenomena / Tu/e speakers: José Bico (ESPCI Paris) Daniel
Measurement of the organic saturation and organic porosity in. shale
 Measurement of the organic saturation and organic porosity in shale Qian Sang a,b, Shaojie Zhang a, Yajun Li a, Mingzhe Dong a,b Steven Bryant b a College of Petroleum Engineering, China University of
Measurement of the organic saturation and organic porosity in shale Qian Sang a,b, Shaojie Zhang a, Yajun Li a, Mingzhe Dong a,b Steven Bryant b a College of Petroleum Engineering, China University of
CHARACTERIZATION OF FLUID-ROCK INTERACTION BY ADSORPTION CALORIMETRY
 SCA2018_053 1/8 CHARACTERIZATION OF FLUID-ROCK INTERACTION BY ADSORPTION CALORIMETRY D. Korobkov 1, V. Pletneva 1, E. Dyshlyuk 2 1 Schlumberger, Moscow Research Center and 2 Dahran Carbonate Research Center
SCA2018_053 1/8 CHARACTERIZATION OF FLUID-ROCK INTERACTION BY ADSORPTION CALORIMETRY D. Korobkov 1, V. Pletneva 1, E. Dyshlyuk 2 1 Schlumberger, Moscow Research Center and 2 Dahran Carbonate Research Center
Contact angle saturation in electrowetting: Injection of ions into the surrounding media
 December 215 EPL, 112 (215) 561 doi: 1.129/295-575/112/561 www.epljournal.org Contact angle saturation in electrowetting: Injection of ions into the surrounding media Tetsuya Yamamoto 1,2,MasaoDoi 1 and
December 215 EPL, 112 (215) 561 doi: 1.129/295-575/112/561 www.epljournal.org Contact angle saturation in electrowetting: Injection of ions into the surrounding media Tetsuya Yamamoto 1,2,MasaoDoi 1 and
Interfacial Flows of Contact Line Dynamics and Liquid Displacement in a Circular Microchannel
 Proceedings of the 3 rd World Congress on Mechanical, Chemical, and Material Engineering (MCM'17) Rome, Italy June 8 10, 2017 Paper No. HTFF 159 ISSN: 2369-8136 DOI: 10.11159/htff17.159 Interfacial Flows
Proceedings of the 3 rd World Congress on Mechanical, Chemical, and Material Engineering (MCM'17) Rome, Italy June 8 10, 2017 Paper No. HTFF 159 ISSN: 2369-8136 DOI: 10.11159/htff17.159 Interfacial Flows
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai
 Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Surfaces and Interfaces Defining of interfacial region Types of interfaces: surface vs interface Surface
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Surfaces and Interfaces Defining of interfacial region Types of interfaces: surface vs interface Surface
Complete Wetting of Acrylic Solid Substrate with Silicone Oil at the Center of the Substrate
 Complete Wetting of Acrylic Solid Substrate with Silicone Oil at the Center of the Substrate Derrick O. Njobuenwu * Department of Chemical Engineering, Loughborough University Leicestershire LE11 3TU,
Complete Wetting of Acrylic Solid Substrate with Silicone Oil at the Center of the Substrate Derrick O. Njobuenwu * Department of Chemical Engineering, Loughborough University Leicestershire LE11 3TU,
Contents. Preface XI Symbols and Abbreviations XIII. 1 Introduction 1
 V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
 Studies on the Coalescence o f Liqu Title Influence of the Polarizing Potenti Phases in Aqueous Solutions (Commem to Professor Rempei Gotoh On the Oc Author(s) Matsumoto, Mutsuo Citation Bulletin of the
Studies on the Coalescence o f Liqu Title Influence of the Polarizing Potenti Phases in Aqueous Solutions (Commem to Professor Rempei Gotoh On the Oc Author(s) Matsumoto, Mutsuo Citation Bulletin of the
COMPARISON STUDY OF DOLOMITE SURFACE WETTABILITY ALTERATION BY Al2O3 AND ZrO2
 COMPARISON STUDY OF DOLOMITE SURFACE WETTABILITY ALTERATION BY Al2O3 AND ZrO2 Mohd Shahrizan Moslan, Wan Rosli Wan Sulaiman, Abdul Razak Ismail, Mohd Zaidi Jaafar and Issham Ismail Department of Petroleum
COMPARISON STUDY OF DOLOMITE SURFACE WETTABILITY ALTERATION BY Al2O3 AND ZrO2 Mohd Shahrizan Moslan, Wan Rosli Wan Sulaiman, Abdul Razak Ismail, Mohd Zaidi Jaafar and Issham Ismail Department of Petroleum
Lecture 7 Contact angle phenomena and wetting
 Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
AN EXPERIMENTAL INVESTIGATION OF BOILING HEAT CONVECTION WITH RADIAL FLOW IN A FRACTURE
 PROCEEDINGS, Twenty-Fourth Workshop on Geothermal Reservoir Engineering Stanford University, Stanford, California, January 25-27, 1999 SGP-TR-162 AN EXPERIMENTAL INVESTIGATION OF BOILING HEAT CONVECTION
PROCEEDINGS, Twenty-Fourth Workshop on Geothermal Reservoir Engineering Stanford University, Stanford, California, January 25-27, 1999 SGP-TR-162 AN EXPERIMENTAL INVESTIGATION OF BOILING HEAT CONVECTION
*blood and bones contain colloids. *milk is a good example of a colloidal dispersion.
 Chap. 3. Colloids 3.1. Introduction - Simple definition of a colloid: a macroscopically heterogeneous system where one component has dimensions in between molecules and macroscopic particles like sand
Chap. 3. Colloids 3.1. Introduction - Simple definition of a colloid: a macroscopically heterogeneous system where one component has dimensions in between molecules and macroscopic particles like sand
Synthesis of Nanoparticles using Atmospheric Microplasma Discharge
 Synthesis of Nanoparticles using Atmospheric Microplasma Discharge Ankit Bisht, G. Roshan Deen, Usman Ilyas, Y. Wang, Alireza Talebitaher, P. Lee, R.S. Rawat NSSE, National Institute of Education, Nanyang
Synthesis of Nanoparticles using Atmospheric Microplasma Discharge Ankit Bisht, G. Roshan Deen, Usman Ilyas, Y. Wang, Alireza Talebitaher, P. Lee, R.S. Rawat NSSE, National Institute of Education, Nanyang
Supplementary Information
 Supplementary Information Supplementary Figure 1 Supplementary Figure 1: Relationship between wetting and nucleation. (a) Relative decrease in the energy barrier (free energy change of heterogeneous nucleation,
Supplementary Information Supplementary Figure 1 Supplementary Figure 1: Relationship between wetting and nucleation. (a) Relative decrease in the energy barrier (free energy change of heterogeneous nucleation,
Lubricant Impregnated Nanotextured Surfaces
 Supporting Information: Enhanced Condensation on Lubricant Impregnated Nanotextured Surfaces Sushant Anand, Adam T. Paxson, Rajeev Dhiman, J. David Smith, Kripa K. Varanasi* Department of Mechanical Engineering,
Supporting Information: Enhanced Condensation on Lubricant Impregnated Nanotextured Surfaces Sushant Anand, Adam T. Paxson, Rajeev Dhiman, J. David Smith, Kripa K. Varanasi* Department of Mechanical Engineering,
PHYSICS OF FLUID SPREADING ON ROUGH SURFACES
 INTERNATIONAL JOURNAL OF NUMERICAL ANALYSIS AND MODELING Volume 5, Supp, Pages 85 92 c 2008 Institute for Scientific Computing and Information PHYSICS OF FLUID SPREADING ON ROUGH SURFACES K. M. HAY AND
INTERNATIONAL JOURNAL OF NUMERICAL ANALYSIS AND MODELING Volume 5, Supp, Pages 85 92 c 2008 Institute for Scientific Computing and Information PHYSICS OF FLUID SPREADING ON ROUGH SURFACES K. M. HAY AND
Investigations on the Evaporation of Charged Droplets on Hydrophobic Surface
 Proceedings of the International Conference on Heat Transfer and Fluid Flow Prague, Czech Republic, August 11-12, 2014 Paper No. 15 Investigations on the Evaporation of Charged Droplets on Hydrophobic
Proceedings of the International Conference on Heat Transfer and Fluid Flow Prague, Czech Republic, August 11-12, 2014 Paper No. 15 Investigations on the Evaporation of Charged Droplets on Hydrophobic
COMPARISON OF WETTABILITY AND CAPILLARY EFFECT EVALUATED BY DIFFERENT CHARACTERIZING METHODS
 18 TH INTERNATIONAL CONFERENCE ON COMPOSITE MATERIALS COMPARISON OF WETTABILITY AND CAPILLARY EFFECT EVALUATED BY DIFFERENT CHARACTERIZING METHODS S.K. Wang*, M. Li*, Y.Z. Gu, Y.X. Li and Z.G. Zhang Key
18 TH INTERNATIONAL CONFERENCE ON COMPOSITE MATERIALS COMPARISON OF WETTABILITY AND CAPILLARY EFFECT EVALUATED BY DIFFERENT CHARACTERIZING METHODS S.K. Wang*, M. Li*, Y.Z. Gu, Y.X. Li and Z.G. Zhang Key
Microfluidics 2 Surface tension, contact angle, capillary flow
 MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
Research Article Studies on a Foam System of Ultralow Interfacial Tension Applied in Daqing Oilfield after Polymer Flooding
 Chemistry Volume 213, Article ID 15274, 6 pages http://dx.doi.org/1.1155/213/15274 Research Article Studies on a Foam System of Ultralow Interfacial Tension Applied in Daqing Oilfield after Polymer Flooding
Chemistry Volume 213, Article ID 15274, 6 pages http://dx.doi.org/1.1155/213/15274 Research Article Studies on a Foam System of Ultralow Interfacial Tension Applied in Daqing Oilfield after Polymer Flooding
Simultaneous Measurement of Capillary Pressure and Dielectric Constant in Porous Media
 PIERS ONLINE, VOL. 3, NO. 4, 2007 549 Simultaneous Measurement of Capillary Pressure and Dielectric Constant in Porous Media W. J. Plug, L. M. Moreno, J. Bruining, and E. C. Slob Delft University of Technology,
PIERS ONLINE, VOL. 3, NO. 4, 2007 549 Simultaneous Measurement of Capillary Pressure and Dielectric Constant in Porous Media W. J. Plug, L. M. Moreno, J. Bruining, and E. C. Slob Delft University of Technology,
Capillary pressure as a unique function of electric permittivity and water saturation
 Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 34, L13306, doi:10.1029/2007gl029674, 2007 Capillary pressure as a unique function of electric permittivity and water saturation Willem-Jan
Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 34, L13306, doi:10.1029/2007gl029674, 2007 Capillary pressure as a unique function of electric permittivity and water saturation Willem-Jan
Low Voltage Reversible Electrowetting Exploiting Lubricated Polymer. Honeycomb Substrates. Published: Applied Physics Letters 104 (2014)
 Low Voltage Reversible Electrowetting Exploiting Lubricated Polymer Honeycomb Substrates Published: Applied Physics Letters 104 (2014) 171601. Edward Bormashenko 1, Roman Pogreb, Yelena Bormashenko 1,
Low Voltage Reversible Electrowetting Exploiting Lubricated Polymer Honeycomb Substrates Published: Applied Physics Letters 104 (2014) 171601. Edward Bormashenko 1, Roman Pogreb, Yelena Bormashenko 1,
Colloidal Particles at Liquid Interfaces: An Introduction
 1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
Evaluation of high molecular weight surfactants for application in Petroleum Industry
 Evaluation of high molecular weight surfactants for application in Petroleum Industry Mestre, C., Prieto, C., Ribeiro, F. Centro de Investigação da CEPSA, Instituto Superior Técnico Abstract The main objective
Evaluation of high molecular weight surfactants for application in Petroleum Industry Mestre, C., Prieto, C., Ribeiro, F. Centro de Investigação da CEPSA, Instituto Superior Técnico Abstract The main objective
ELECTRICAL RESISTIVITY TOMOGRAPHY
 NOTIO Association Clay Technological Centre C/ Río Cabriel s/n 45007 Toledo Tel.: 925 24 11 62 info@notio.es www.notio.es Page 1 / 7 SUMMARY 1. PHYSICAL FUNDAMENTALS OF THE... 3 1.1. ELECTRICAL BEHAVIOR
NOTIO Association Clay Technological Centre C/ Río Cabriel s/n 45007 Toledo Tel.: 925 24 11 62 info@notio.es www.notio.es Page 1 / 7 SUMMARY 1. PHYSICAL FUNDAMENTALS OF THE... 3 1.1. ELECTRICAL BEHAVIOR
Liquid lens based on electrowetting: a new adaptive component for imaging applications in consumer electronics
 Liquid lens based on electrowetting: a new adaptive component for imaging applications in consumer electronics Jerome CRASSOUS **, Claude GABAY **, Gaetan LIOGIER *, Bruno BERGE *,** * Varioptic, 8B rue
Liquid lens based on electrowetting: a new adaptive component for imaging applications in consumer electronics Jerome CRASSOUS **, Claude GABAY **, Gaetan LIOGIER *, Bruno BERGE *,** * Varioptic, 8B rue
Critical Micellization Concentration Determination using Surface Tension Phenomenon
 Critical Micellization Concentration Determination using Phenomenon 1. Introduction Surface-active agents (surfactants) were already known in ancient times, when their properties were used in everyday
Critical Micellization Concentration Determination using Phenomenon 1. Introduction Surface-active agents (surfactants) were already known in ancient times, when their properties were used in everyday
12/2/2010. Success in Surfactant EOR: Avoid the Failure Mechanisms
 Success in Surfactant EOR: Avoid the Failure Mechanisms George J. Hirasaki Petroleum Engineering, Texas A&M November 9, 2010 1 Requirements for Surfactant EOR Ultra Low IFT Mobility Control Transport Across
Success in Surfactant EOR: Avoid the Failure Mechanisms George J. Hirasaki Petroleum Engineering, Texas A&M November 9, 2010 1 Requirements for Surfactant EOR Ultra Low IFT Mobility Control Transport Across
H 2 O WHAT PROPERTIES OF WATER MAKE IT ESSENTIAL TO LIFE OF EARTH? Good solvent High Surface tension Low vapor pressure High boiling point
 Unit 9: Solutions H 2 O WHAT PROPERTIES OF WATER MAKE IT ESSENTIAL TO LIFE OF EARTH? Good solvent High Surface tension Low vapor pressure High boiling point Water is a polar molecule. It experiences hydrogen
Unit 9: Solutions H 2 O WHAT PROPERTIES OF WATER MAKE IT ESSENTIAL TO LIFE OF EARTH? Good solvent High Surface tension Low vapor pressure High boiling point Water is a polar molecule. It experiences hydrogen
A COMPARISION OF WETTABILITY AND SPONTANEOUS IMBIBITION EXPERIMENTS OF SURFACTANT SOLUTION IN SANDSTONE AND CARBONATE ROCKS
 A COMPARISION OF WETTABILITY AND SPONTANEOUS IMBIBITION EXPERIMENTS OF SURFACTANT SOLUTION IN SANDSTONE AND CARBONATE ROCKS Rebelo, David; Pereira, Maria João Email addresses: david.j.nr@hotmail.com; maria.pereira@tecnico.ulisboa.pt
A COMPARISION OF WETTABILITY AND SPONTANEOUS IMBIBITION EXPERIMENTS OF SURFACTANT SOLUTION IN SANDSTONE AND CARBONATE ROCKS Rebelo, David; Pereira, Maria João Email addresses: david.j.nr@hotmail.com; maria.pereira@tecnico.ulisboa.pt
Supplementary Figures
 Supplementary Figures 1 Supplementary Figure 1 Micro and nano-textured boiling surfaces. (a) A schematic of the textured boiling surfaces. (b) An isometric view of the square array of square micropillars.
Supplementary Figures 1 Supplementary Figure 1 Micro and nano-textured boiling surfaces. (a) A schematic of the textured boiling surfaces. (b) An isometric view of the square array of square micropillars.
Electrowetting on a semiconductor
 Electrowetting on a semiconductor Steve Arscott (a) and Matthieu Gaudet Institut d Electronique, de Microélectronique et de Nanotechnologie (IEMN), CNRS UMR8520, The University of Lille, Cité Scientifique,
Electrowetting on a semiconductor Steve Arscott (a) and Matthieu Gaudet Institut d Electronique, de Microélectronique et de Nanotechnologie (IEMN), CNRS UMR8520, The University of Lille, Cité Scientifique,
spreading of drops on soft surfaces
 Supplementary Material on Electrically modulated dynamic spreading of drops on soft surfaces Ranabir Dey 1, Ashish Daga 1, Sunando DasGupta 2,3, Suman Chakraborty 1,3 1 Department of Mechanical Engineering,
Supplementary Material on Electrically modulated dynamic spreading of drops on soft surfaces Ranabir Dey 1, Ashish Daga 1, Sunando DasGupta 2,3, Suman Chakraborty 1,3 1 Department of Mechanical Engineering,
LIQUID FILM THICKNESS OF OSCILLATING FLOW IN A MICRO TUBE
 Proceedings of the ASME/JSME 2011 8th Thermal Engineering Joint Conference AJTEC2011 March 13-17, 2011, Honolulu, Hawaii, USA AJTEC2011-44190 LIQUID FILM THICKNESS OF OSCILLATING FLOW IN A MICRO TUBE Youngbae
Proceedings of the ASME/JSME 2011 8th Thermal Engineering Joint Conference AJTEC2011 March 13-17, 2011, Honolulu, Hawaii, USA AJTEC2011-44190 LIQUID FILM THICKNESS OF OSCILLATING FLOW IN A MICRO TUBE Youngbae
CHEMISTRY Ch. 14 Notes: Mixtures and Solutions NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics.
 CHEMISTRY Ch. 14 Notes: Mixtures and Solutions NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. 14.1 notes I. Types of mixtures (mixture a physical blend of substances)
CHEMISTRY Ch. 14 Notes: Mixtures and Solutions NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. 14.1 notes I. Types of mixtures (mixture a physical blend of substances)
Derivation of continuum models for the moving contact line problem based on thermodynamic principles. Abstract
 Derivation of continuum models for the moving contact line problem based on thermodynamic principles Weiqing Ren Courant Institute of Mathematical Sciences, New York University, New York, NY 002, USA Weinan
Derivation of continuum models for the moving contact line problem based on thermodynamic principles Weiqing Ren Courant Institute of Mathematical Sciences, New York University, New York, NY 002, USA Weinan
Module 4: "Surface Thermodynamics" Lecture 22: "" The Lecture Contains: Examples on Effect of surfactant on interfacial tension. Objectives_template
 The Lecture Contains: Examples on Effect of surfactant on interfacial tension file:///e /courses/colloid_interface_science/lecture22/22_1.htm[6/16/2012 1:10:07 PM] Example Consider liquid, its vapors and
The Lecture Contains: Examples on Effect of surfactant on interfacial tension file:///e /courses/colloid_interface_science/lecture22/22_1.htm[6/16/2012 1:10:07 PM] Example Consider liquid, its vapors and
Adsorption Processes. Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad
 Adsorption Processes Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad Contents Introduction Principles of adsorption Types of adsorption Definitions Brief history Adsorption isotherms Mechanism
Adsorption Processes Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad Contents Introduction Principles of adsorption Types of adsorption Definitions Brief history Adsorption isotherms Mechanism
Colloidal dispersion
 Dispersed Systems Dispersed systems consist of particulate matter, known as the dispersed phase, distributed throughout a continuous or dispersion medium. The dispersed material may range in size from
Dispersed Systems Dispersed systems consist of particulate matter, known as the dispersed phase, distributed throughout a continuous or dispersion medium. The dispersed material may range in size from
The Wilhelmy balance. How can we measure surface tension? Surface tension, contact angles and wettability. Measuring surface tension.
 ow can we measure surface tension? Surface tension, contact angles and wettability www.wikihow.com/measure-surface-tension Measuring surface tension The Wilhelmy balance F Some methods: Wilhelmy plate
ow can we measure surface tension? Surface tension, contact angles and wettability www.wikihow.com/measure-surface-tension Measuring surface tension The Wilhelmy balance F Some methods: Wilhelmy plate
Monolayers. Factors affecting the adsorption from solution. Adsorption of amphiphilic molecules on solid support
 Monolayers Adsorption as process Adsorption of gases on solids Adsorption of solutions on solids Factors affecting the adsorption from solution Adsorption of amphiphilic molecules on solid support Adsorption
Monolayers Adsorption as process Adsorption of gases on solids Adsorption of solutions on solids Factors affecting the adsorption from solution Adsorption of amphiphilic molecules on solid support Adsorption
Interfacial Phenomena
 Physical Pharmacy Lecture 4 Interfacial Phenomena Assistant Lecturer in Pharmaceutics Overview Liquid interfaces Surface tension Interfacial tension Surface free energy Measurement of tensions Spreading
Physical Pharmacy Lecture 4 Interfacial Phenomena Assistant Lecturer in Pharmaceutics Overview Liquid interfaces Surface tension Interfacial tension Surface free energy Measurement of tensions Spreading
Instabilities in the Flow of Thin Liquid Films
 Instabilities in the Flow of Thin Liquid Films Lou Kondic Department of Mathematical Sciences Center for Applied Mathematics and Statistics New Jersey Institute of Technology Presented at Annual Meeting
Instabilities in the Flow of Thin Liquid Films Lou Kondic Department of Mathematical Sciences Center for Applied Mathematics and Statistics New Jersey Institute of Technology Presented at Annual Meeting
UvA-DARE (Digital Academic Repository) Electrokinetics in porous media Luong, D.T. Link to publication
 UvA-DARE (Digital Academic Repository) Electrokinetics in porous media Luong, D.T. Link to publication Citation for published version (APA): Luong, D. T. (2014). Electrokinetics in porous media General
UvA-DARE (Digital Academic Repository) Electrokinetics in porous media Luong, D.T. Link to publication Citation for published version (APA): Luong, D. T. (2014). Electrokinetics in porous media General
1. Resistivity of rocks
 RESISTIVITY 1) Resistivity of rocks 2) General principles of resistivity surveying 3) Field procedures, interpretation and examples 4) Summary and conclusions INDUCED POLARIZATION 1) General principles
RESISTIVITY 1) Resistivity of rocks 2) General principles of resistivity surveying 3) Field procedures, interpretation and examples 4) Summary and conclusions INDUCED POLARIZATION 1) General principles
Effects of dynamic contact angle on numerical modeling of electrowetting in parallel plate microchannels
 Microfluid Nanofluid (2010) 8:47 56 DOI 10.1007/s10404-009-0460-3 RESEARCH PAPER Effects of dynamic contact angle on numerical modeling of electrowetting in parallel plate microchannels Zahra Keshavarz-Motamed
Microfluid Nanofluid (2010) 8:47 56 DOI 10.1007/s10404-009-0460-3 RESEARCH PAPER Effects of dynamic contact angle on numerical modeling of electrowetting in parallel plate microchannels Zahra Keshavarz-Motamed
DIELECTRIC PROPERTIES OF POROUS ROCKS WITH AN APPLICATION TO SEA ICE BY LARS BACKSTROM
 DIELECTRIC PROPERTIES OF POROUS ROCKS WITH AN APPLICATION TO SEA ICE BY LARS BACKSTROM PRESENTATION GIVEN AS PARTIAL FULLFILLMENT OF THE REQUIREMENTS FOR THE CLASS GEOS 692 - FALL 2006 Contents of presentation
DIELECTRIC PROPERTIES OF POROUS ROCKS WITH AN APPLICATION TO SEA ICE BY LARS BACKSTROM PRESENTATION GIVEN AS PARTIAL FULLFILLMENT OF THE REQUIREMENTS FOR THE CLASS GEOS 692 - FALL 2006 Contents of presentation
Forces and movement of small water droplets in oil due to applied electric field
 Nordic Insulation Symposium Tampere, June 3, 23 Forces and movement of small water droplets in oil due to applied electric field A. Pedersen E. Ildstad A. Nysveen Norwegian University of Norwegian University
Nordic Insulation Symposium Tampere, June 3, 23 Forces and movement of small water droplets in oil due to applied electric field A. Pedersen E. Ildstad A. Nysveen Norwegian University of Norwegian University
Wettability of polymeric solids by aqueous solution of surfactants
 A N N A L E S U N I V E R S I T A T I S M A R I A E C U R I E - S K Ł O D O W S K A L U B L I N P O L O N I A VOL. LX, 8 SECTIO AA 2005 Wettability of polymeric solids by aqueous solution of surfactants
A N N A L E S U N I V E R S I T A T I S M A R I A E C U R I E - S K Ł O D O W S K A L U B L I N P O L O N I A VOL. LX, 8 SECTIO AA 2005 Wettability of polymeric solids by aqueous solution of surfactants
dynamics of f luids in porous media
 dynamics of f luids in porous media Jacob Bear Department of Civil Engineering Technion Israel Institute of Technology, Haifa DOVER PUBLICATIONS, INC. New York Contents Preface xvii CHAPTER 1 Introduction
dynamics of f luids in porous media Jacob Bear Department of Civil Engineering Technion Israel Institute of Technology, Haifa DOVER PUBLICATIONS, INC. New York Contents Preface xvii CHAPTER 1 Introduction
Experimental Validation of the Invariance of Electrowetting Contact Angle Saturation
 J. Adhesion Sci. Technol. (2011) DOI:10.1163/156856111X599580 brill.nl/jast Experimental Validation of the Invariance of Electrowetting Contact Angle Saturation Stéphanie Chevalliot a,b, Stein Kuiper c
J. Adhesion Sci. Technol. (2011) DOI:10.1163/156856111X599580 brill.nl/jast Experimental Validation of the Invariance of Electrowetting Contact Angle Saturation Stéphanie Chevalliot a,b, Stein Kuiper c
AN EXPERIMENTAL STUDY OF THE RELATIONSHIP BETWEEN ROCK SURFACE PROPERTIES, WETTABILITY AND OIL PRODUCTION CHARACTERISTICS
 AN EXPERIMENTAL STUDY OF THE RELATIONSHIP BETWEEN ROCK SURFACE PROPERTIES, WETTABILITY AND OIL PRODUCTION CHARACTERISTICS by Ole Torsæter, Norwegian University of Science and Technology, Trondheim Reidar
AN EXPERIMENTAL STUDY OF THE RELATIONSHIP BETWEEN ROCK SURFACE PROPERTIES, WETTABILITY AND OIL PRODUCTION CHARACTERISTICS by Ole Torsæter, Norwegian University of Science and Technology, Trondheim Reidar
Physical Chemistry of Surfaces
 Physical Chemistry of Surfaces Fifth Edition ARTHUR W. ADAMSON Department of Chemistry, University of Southern California Los Angeles, California >) A WILEY-INTERSCIENCE PUBLICATION John Wiley &. Sons,
Physical Chemistry of Surfaces Fifth Edition ARTHUR W. ADAMSON Department of Chemistry, University of Southern California Los Angeles, California >) A WILEY-INTERSCIENCE PUBLICATION John Wiley &. Sons,
Rock/water interaction in dielectric properties: Experiments with hydrophobic sandstones
 GEOPHYSICS, VOL. 60, NO. 2 (MARCH-APRIL 1995); P. 431-436, 9 FIGS., 4 TABLES. Rock/water interaction in dielectric properties: Experiments with hydrophobic sandstones Rosemary Knight* and Ana Abad ABSTRACT
GEOPHYSICS, VOL. 60, NO. 2 (MARCH-APRIL 1995); P. 431-436, 9 FIGS., 4 TABLES. Rock/water interaction in dielectric properties: Experiments with hydrophobic sandstones Rosemary Knight* and Ana Abad ABSTRACT
Wetting & Adhesion on Soft Surfaces Young s Law is dead long live Young s Law. Eric Dufresne
 Wetting & Adhesion on Soft Surfaces Young s Law is dead long live Young s Law Eric Dufresne KITP 2014 Wetting Adhesion 3mm 30 um Young s Law relates contact line geometry and material properties in equilibrium
Wetting & Adhesion on Soft Surfaces Young s Law is dead long live Young s Law Eric Dufresne KITP 2014 Wetting Adhesion 3mm 30 um Young s Law relates contact line geometry and material properties in equilibrium
Adsorption at the solid/liquid interface
 1. Ion exchanger Adsorption at the solid/liquid interface Ion exchange process means an exchange of ions between an electrolyte solution and a solid (ionite). In most cases the term is used to denote the
1. Ion exchanger Adsorption at the solid/liquid interface Ion exchange process means an exchange of ions between an electrolyte solution and a solid (ionite). In most cases the term is used to denote the
Supporting Information. Golf ball-shaped PLGA microparticles with internal pores fabricated by simple O/W emulsion
 Supplementary Material (ESI) for Chemical Communications Supporting Information Golf ball-shaped PLGA microparticles with internal pores fabricated by simple O/W emulsion Mi Ri Kim, a Seungwoo Lee, b Jung-Ki
Supplementary Material (ESI) for Chemical Communications Supporting Information Golf ball-shaped PLGA microparticles with internal pores fabricated by simple O/W emulsion Mi Ri Kim, a Seungwoo Lee, b Jung-Ki
Carbon nanotube coated snowman-like particles and their electro-responsive characteristics. Ke Zhang, Ying Dan Liu and Hyoung Jin Choi
 Supporting Information: Carbon nanotube coated snowman-like particles and their electro-responsive characteristics Ke Zhang, Ying Dan Liu and Hyoung Jin Choi Experimental Section 1.1 Materials The MWNT
Supporting Information: Carbon nanotube coated snowman-like particles and their electro-responsive characteristics Ke Zhang, Ying Dan Liu and Hyoung Jin Choi Experimental Section 1.1 Materials The MWNT
Colloid & Interface Science Case Study Model Answers
 Colloid & Interface Science Case Study Model Answers Distance Learning Course in Cosmetic Science Society of Cosmetic Scientists Common Features Formulations were examples of lyophobic colloidal systems
Colloid & Interface Science Case Study Model Answers Distance Learning Course in Cosmetic Science Society of Cosmetic Scientists Common Features Formulations were examples of lyophobic colloidal systems
Water and Aqueous Systems
 Water and Aqueous Systems The Water Molecule: a Review Water is a simple tri-atomic molecule, H 2 O Each O-H bond is highly polar, because of the high electronegativity of the oxygen (N, O, F, and Cl have
Water and Aqueous Systems The Water Molecule: a Review Water is a simple tri-atomic molecule, H 2 O Each O-H bond is highly polar, because of the high electronegativity of the oxygen (N, O, F, and Cl have
Dilute Surfactant Methods for Carbonate Formations
 Dilute Surfactant Methods for Carbonate Formations ID Number: DE-FC26-02NT 15322 Quarterly Progress Report Reporting Period Start Date: 10-1-2003 Reporting Period End Date: 12-31-2003 Submitted to the
Dilute Surfactant Methods for Carbonate Formations ID Number: DE-FC26-02NT 15322 Quarterly Progress Report Reporting Period Start Date: 10-1-2003 Reporting Period End Date: 12-31-2003 Submitted to the
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa.
 Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
A.% by mass (like % composition)
 Solutions; Colloids Key Words Solute Solvent Solubility effervescence Miscible saturated Supersaturated (metastable system)- a cooled solution contains more solute than it would at equilibrium, desolvation=
Solutions; Colloids Key Words Solute Solvent Solubility effervescence Miscible saturated Supersaturated (metastable system)- a cooled solution contains more solute than it would at equilibrium, desolvation=
Effects of Displacement Efficiency of Surfactant Flooding in High Salinity Reservoir: Interfacial Tension, Emulsification, Adsorption
 ADVANCES IN PETROLEUM EXPLORATION AND DEVELOPMENT Vol. 1, No. 1, 2011 PP. 32-39 ISSN 1925-542X [PRINT] ISSN 1925-5438 [ONLINE] www.cscanada.net www.cscanada.org YU Haiyang 1,* ; WANG Yefei 1 ; ZHANG Yan
ADVANCES IN PETROLEUM EXPLORATION AND DEVELOPMENT Vol. 1, No. 1, 2011 PP. 32-39 ISSN 1925-542X [PRINT] ISSN 1925-5438 [ONLINE] www.cscanada.net www.cscanada.org YU Haiyang 1,* ; WANG Yefei 1 ; ZHANG Yan
DLVO interaction between the spheres
 DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
Fundamentals of Droplet Flow in Microfluidics. R.B. Fair Department of Electrical and Computer Engineering Duke University Durham, N.C.
 Fundamentals of Droplet Flow in Microfluidics R.B. Fair Computer Engineering Duke University Durham, N.C., USA Outline of Presentation Review of droplet actuation mechanisms Thermocapillarity SAW Electrostatic
Fundamentals of Droplet Flow in Microfluidics R.B. Fair Computer Engineering Duke University Durham, N.C., USA Outline of Presentation Review of droplet actuation mechanisms Thermocapillarity SAW Electrostatic
