Solid state reactions. Often defined as reactions between two solids. Here the definition is extended to any reaction involving a solid:
|
|
|
- Scot Howard
- 5 years ago
- Views:
Transcription
1 Sold state reactons Often defned as reactons between two solds Here the defnton s extended to any reacton nvolvng a sold: Sold/sold Sold/gas (Reacton, decomposton) (Sold/lqud) Intercalaton 1 2 Sold-State Reactons 2.1 Reactons Between Sold Compounds Ceramc Method Carbothermal Reducton Combuston Synthess Snterng 2.2 Sold Gas Reactons 2.3 Decomposton and Dehydraton Reactons 2.4 Intercalaton Reactons General Aspects Preparatve Methods Pllarng of Layered Compounds 2
2 Ceramc method Drect reacton of solds to form the fnal product In prncple no decomposton s nvolved. Solds do not react wth solds at room temperature even of thermodynamcs s favorable. Hgh temperature must be used. Sold-sold reactons are smple to perform, startng materals are often readly avalable at low cost and reactons are clean.e. do not nvolve other chemcal elements. Dsadvantages nclude the need for hgh temperatures, the possblty of non-homogenety, contamnaton from contaners etc. etc. 3 Synthess of YBCO, YBa 2 Cu 3 O 7-x Drect reacton between Y 2 O 3, BaO 2, CuO (Reacton between three sold components) Grnd to obtan large surface area Press nto pellets (contact) Heat n alumna boat, temperature profle: Oxdaton step 4
3 Other precursors may be used, e.g. BaCO 3, whch may be decomposed to fne graned BaO durng the reacton. ( + ntrates etc.) Decomposton must be performed n a controlled manner n order to avod volent decomposton (.e. choosng an approprate temperature) Many other ways of syntheszng YBCO: Precursor methods Sol/gel Flux Many melt-texturng methods ncludng partally meltng/fluxng 5 6
4 Sold state reactons, general aspects Nucleaton at the nterface (depends on the degree of structural rearrangement nvolved) Formaton of product layer, two reacton nterfaces Dffuson/counter dffuson through the product layer Longer dffuson paths, slower reacton rate. Smple case: lattce dffuson through a planar layer: dx/dt = k * x -1 7 Reactons often nvolve other reactons and transport mechansms. Electrons, gaseous speces, oxygen ons, defects 8
5 Dffuson s a thermally actvated process 9 Transport: Dffuson n a gradent (.e. chemcal potental gradent, Δμ) 10
6 Mass transport n an onc sold s often acheved by transport of a charged speces and counter transport of a dfferently charged on or defect n the opposte drecton. Then the electrochemcal potental s the drvng force, and not the chemcal potental or the concentraton gradent. The electrochemcal potental (η ) of speces s the sum of the chemcal potental and electrostatc potental φ: η = μ + Z Fφ Where Z s the effectve charge and F s the Faraday constant. The flux s gven by: j = c v = c B η x = c B μ x φ + ZF x 11 Implcatons of the expresson for flux: The effect of the terms may pull n opposte drectons (.e. a concentraton gradent may be countered by an electrcal feld gradent) A dfferent type of effect may occur due to the local electrcal feld between oppostely charged speces. Ths may affect the transport of an on relatve to other(s). Ths couplng results n e.g. that both Al 3+ and O 2- move n Al 2 O 3 (n contrast one would normally expect O 2- to by farly mmoble due to the large onc sze) 12
7 Example of parabolc growth: NO + Al 2 O 3 NAl 2 O 4 13 Ions n solds are not moble at low temperatures. Hgh temperatures, as a rule of thumb, at 2/3 of the meltng temperature (of one component) the dffuson s suffcent to acheve sold state reactons. Formaton of BaTO 3 by reactng BaCO 3 and TO 2 s an example of a seemngly smple reacton s more complex than expected. BaTO 3 s an mportant electroceramc, (Thermstors, capactors, optoelectronc devces, DRAMs) BaCO 3 s decomposed to reactve BaO: (Rock salt, ccp of the oxde anons, Ba 2+ n octahedral stes), TO 2 (Rutle, hcp of oxde ons, T 4+ n half of the octahedral stes) At least three stages are nvolved n formaton of BaTO 3 from BaO and TO 2. BaO react wth the surface of TO 2, formng nucle and a surface layer of BaTO 3. Reacton between BaO and BaTO 3 to form Ba 2 TO 4. NB! Ths s a necessary phase for ncreasng mgraton of Ba 2+ ons. Ba 2+ ons from the Ba-rch phase mgrate nto the TO 2 phase and form BaTO 3. 14
8 Reactons between two solds: Reacton rates depends on: Area of contact between the reactng solds,.e. surface area and densty The rate of nucleaton Rates of dffuson of ons (and other speces) Dsadvantages, e.g.: Nucleaton and dffuson related problems (hgh temperature) Formaton of undesred phases (reacton paths) (e.g. BaT 2 O 5 ) Homogeneous dstrbuton, especally for dopants, s dffcult Dffcult to montor the reacton drectly, n-stu?? Separaton of phases after synthess s dffcult Reacton wth contaners/crucbles Volatlty of one or more of the components 15 16
9 Surface area: Surface area s ncreased by crushng/mllng 1 cm3 cube: surface area 6 cm 2 1 mm cubes: surface area 60 cm 2 10μm cubes: surface area 6000 cm 2 10 nm cubes: surface area cm 2 (600m 2 ) 17 Increase dffuson rates (decrease dffuson lengths) Small partcles by grndng, ball mllng, spray dryng, freeze dryng, spray pyrolyss etc. Coolng and regrndng may be necessary Intmate mxture of reactants by coprecptaton, sol-gel processes, spray pyrolyss etc. Reducton of dffuson dstances by ncorporatng the catons n the same sold precursor. Sold state reactons nvolvng melts, molten fluxes or hgh temperature solvents. 18
10 Nucleaton Nucleaton lmted reactons may be descrbed by the Avram-Erofeyev equaton: x(t) = 1-exp(-kt n ) {or x(t) = 1-exp(-(kt) n )} N s a real number, usually between 1 and 3 (n>4 s often nterpreted as autocatalytc nucleaton) For n > 1 the curve s sgmodal Note: Ths s more emprcal for most sold state reactons. It s dffcult to talk about reacton order and actvaton energy for these reactons. 19 Nucleaton may be facltated by structural smlarty between the reactng solds, or products. In the reacton between MgO and Al 2 O 3, MgO and spnel have smlar oxde on arrangements. Spnel nucle may then easly form at the MgO surfaces. There s often a structural orentatonal relatonshp between the reactant, nucle and product. The lattce spacng (e.g. dstance between oxde ons) should not be too dfferent for orented nucleaton. 20
11 Eptactc and topotactc reactons Eptactc: structural smlarty restrcted to the surface / nterface between two crystals layer Topotactc: Structural smlarty through the crystal The ease of nucleaton depends also on the actual surface structure of the reactants. 21
Fundamental Considerations of Fuel Cells for Mobility Applications
 Fundamental Consderatons of Fuel Cells for Moblty Applcatons Davd E. Foster Engne Research Center Unversty of Wsconsn - Madson Future Engnes and Ther Fuels ERC 2011 Symposum June 9, 2011 Motvaton Reducng
Fundamental Consderatons of Fuel Cells for Moblty Applcatons Davd E. Foster Engne Research Center Unversty of Wsconsn - Madson Future Engnes and Ther Fuels ERC 2011 Symposum June 9, 2011 Motvaton Reducng
Electrochemical Equilibrium Electromotive Force
 CHM465/865, 24-3, Lecture 5-7, 2 th Sep., 24 lectrochemcal qulbrum lectromotve Force Relaton between chemcal and electrc drvng forces lectrochemcal system at constant T and p: consder Gbbs free energy
CHM465/865, 24-3, Lecture 5-7, 2 th Sep., 24 lectrochemcal qulbrum lectromotve Force Relaton between chemcal and electrc drvng forces lectrochemcal system at constant T and p: consder Gbbs free energy
Chapter 2. Electrode/electrolyte interface: ----Structure and properties
 Chapter 2 Electrode/electrolyte nterface: ----Structure and propertes Electrochemcal reactons are nterfacal reactons, the structure and propertes of electrode / electrolytc soluton nterface greatly nfluences
Chapter 2 Electrode/electrolyte nterface: ----Structure and propertes Electrochemcal reactons are nterfacal reactons, the structure and propertes of electrode / electrolytc soluton nterface greatly nfluences
Lecture 17. Membrane Separations [Ch. 14]
![Lecture 17. Membrane Separations [Ch. 14] Lecture 17. Membrane Separations [Ch. 14]](/thumbs/89/98384931.jpg) Lecture 17. embrane Separatons [Ch. 14] embrane Separaton embrane aterals embrane odules Transport n embranes -Bulk flow - Lqud dffuson n pores - Gas dffuson - onporous membranes embrane Separaton Separaton
Lecture 17. embrane Separatons [Ch. 14] embrane Separaton embrane aterals embrane odules Transport n embranes -Bulk flow - Lqud dffuson n pores - Gas dffuson - onporous membranes embrane Separaton Separaton
4.2 Chemical Driving Force
 4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
1) Silicon oxide has a typical surface potential in an aqueous medium of ϕ,0
 1) Slcon oxde has a typcal surface potental n an aqueous medum of ϕ, = 7 mv n 5 mm l at ph 9. Whch concentraton of catons do you roughly expect close to the surface? What s the average dstance between
1) Slcon oxde has a typcal surface potental n an aqueous medum of ϕ, = 7 mv n 5 mm l at ph 9. Whch concentraton of catons do you roughly expect close to the surface? What s the average dstance between
Wilbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1. Two-dimensional chemical maps as well as chemical profiles were done at 15 kv using
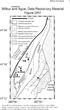 DR2006139 Wlbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1 MINERAL ANALYSES Two-dmensonal chemcal maps as well as chemcal profles were done at 15 kv usng the JEOL JXA-8600 electron mcroprobe at Yale Unversty
DR2006139 Wlbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1 MINERAL ANALYSES Two-dmensonal chemcal maps as well as chemcal profles were done at 15 kv usng the JEOL JXA-8600 electron mcroprobe at Yale Unversty
II. Equilibrium Thermodynamics. Lecture 8: The Nernst Equation
 II. Equlbrum Thermodynamcs Lecture 8: The Nernst Equaton Notes by MIT Student, re-wrtten by MZB 2014 February 27, 2014 1 Chemcal Actvty The fundamental thermodynamc quantty controllng transport and reactons
II. Equlbrum Thermodynamcs Lecture 8: The Nernst Equaton Notes by MIT Student, re-wrtten by MZB 2014 February 27, 2014 1 Chemcal Actvty The fundamental thermodynamc quantty controllng transport and reactons
CHEMICAL ENGINEERING
 Postal Correspondence GATE & PSUs -MT To Buy Postal Correspondence Packages call at 0-9990657855 1 TABLE OF CONTENT S. No. Ttle Page no. 1. Introducton 3 2. Dffuson 10 3. Dryng and Humdfcaton 24 4. Absorpton
Postal Correspondence GATE & PSUs -MT To Buy Postal Correspondence Packages call at 0-9990657855 1 TABLE OF CONTENT S. No. Ttle Page no. 1. Introducton 3 2. Dffuson 10 3. Dryng and Humdfcaton 24 4. Absorpton
Chapter 18, Part 1. Fundamentals of Atmospheric Modeling
 Overhead Sldes for Chapter 18, Part 1 of Fundamentals of Atmospherc Modelng by Mark Z. Jacobson Department of Cvl & Envronmental Engneerng Stanford Unversty Stanford, CA 94305-4020 January 30, 2002 Types
Overhead Sldes for Chapter 18, Part 1 of Fundamentals of Atmospherc Modelng by Mark Z. Jacobson Department of Cvl & Envronmental Engneerng Stanford Unversty Stanford, CA 94305-4020 January 30, 2002 Types
Gouy-Chapman model (1910) The double layer is not as compact as in Helmholtz rigid layer.
 CHE465/865, 6-3, Lecture 1, 7 nd Sep., 6 Gouy-Chapman model (191) The double layer s not as compact as n Helmholtz rgd layer. Consder thermal motons of ons: Tendency to ncrease the entropy and make the
CHE465/865, 6-3, Lecture 1, 7 nd Sep., 6 Gouy-Chapman model (191) The double layer s not as compact as n Helmholtz rgd layer. Consder thermal motons of ons: Tendency to ncrease the entropy and make the
Be true to your work, your word, and your friend.
 Chemstry 13 NT Be true to your work, your word, and your frend. Henry Davd Thoreau 1 Chem 13 NT Chemcal Equlbrum Module Usng the Equlbrum Constant Interpretng the Equlbrum Constant Predctng the Drecton
Chemstry 13 NT Be true to your work, your word, and your frend. Henry Davd Thoreau 1 Chem 13 NT Chemcal Equlbrum Module Usng the Equlbrum Constant Interpretng the Equlbrum Constant Predctng the Drecton
( ) 1/ 2. ( P SO2 )( P O2 ) 1/ 2.
 Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Chemical Equilibrium. Chapter 6 Spontaneity of Reactive Mixtures (gases) Taking into account there are many types of work that a sysem can perform
 Ths chapter deals wth chemcal reactons (system) wth lttle or no consderaton on the surroundngs. Chemcal Equlbrum Chapter 6 Spontanety of eactve Mxtures (gases) eactants generatng products would proceed
Ths chapter deals wth chemcal reactons (system) wth lttle or no consderaton on the surroundngs. Chemcal Equlbrum Chapter 6 Spontanety of eactve Mxtures (gases) eactants generatng products would proceed
...Thermodynamics. If Clausius Clapeyron fails. l T (v 2 v 1 ) = 0/0 Second order phase transition ( S, v = 0)
 If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
Thermodynamics General
 Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
1. Mean-Field Theory. 2. Bjerrum length
 1. Mean-Feld Theory Contnuum models lke the Posson-Nernst-Planck equatons are mean-feld approxmatons whch descrbe how dscrete ons are affected by the mean concentratons c and potental φ. Each on mgrates
1. Mean-Feld Theory Contnuum models lke the Posson-Nernst-Planck equatons are mean-feld approxmatons whch descrbe how dscrete ons are affected by the mean concentratons c and potental φ. Each on mgrates
Osmotic pressure and protein binding
 Osmotc pressure and proten bndng Igor R. Kuznetsov, KochLab Symposum talk 5/15/09 Today we take a closer look at one of the soluton thermodynamcs key ponts from Steve s presentaton. Here t s: d[ln(k off
Osmotc pressure and proten bndng Igor R. Kuznetsov, KochLab Symposum talk 5/15/09 Today we take a closer look at one of the soluton thermodynamcs key ponts from Steve s presentaton. Here t s: d[ln(k off
5. Diffusion. Introduction. Models of diffusion. Fourier s law of heat flux. 5 Diffusion
 5. Dffuson Introducton Numerous chemcal reactons and mcrostructural changes n solds take place through sold state dffuson. In crystallne solds, the dffuson takes place because of the presence of defects;
5. Dffuson Introducton Numerous chemcal reactons and mcrostructural changes n solds take place through sold state dffuson. In crystallne solds, the dffuson takes place because of the presence of defects;
Electrochemistry Thermodynamics
 CHEM 51 Analytcal Electrochemstry Chapter Oct 5, 016 Electrochemstry Thermodynamcs Bo Zhang Department of Chemstry Unversty of Washngton Seattle, WA 98195 Former SEAC presdent Andy Ewng sellng T-shrts
CHEM 51 Analytcal Electrochemstry Chapter Oct 5, 016 Electrochemstry Thermodynamcs Bo Zhang Department of Chemstry Unversty of Washngton Seattle, WA 98195 Former SEAC presdent Andy Ewng sellng T-shrts
Thermo-Calc Software. Modelling Multicomponent Precipitation Kinetics with CALPHAD-Based Tools. EUROMAT2013, September 8-13, 2013 Sevilla, Spain
 Modellng Multcomponent Precptaton Knetcs wth CALPHAD-Based Tools Kasheng Wu 1, Gustaf Sterner 2, Qng Chen 2, Åke Jansson 2, Paul Mason 1, Johan Bratberg 2 and Anders Engström 2 1 Inc., 2 AB EUROMAT2013,
Modellng Multcomponent Precptaton Knetcs wth CALPHAD-Based Tools Kasheng Wu 1, Gustaf Sterner 2, Qng Chen 2, Åke Jansson 2, Paul Mason 1, Johan Bratberg 2 and Anders Engström 2 1 Inc., 2 AB EUROMAT2013,
Open Systems: Chemical Potential and Partial Molar Quantities Chemical Potential
 Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Electrical double layer: revisit based on boundary conditions
 Electrcal double layer: revst based on boundary condtons Jong U. Km Department of Electrcal and Computer Engneerng, Texas A&M Unversty College Staton, TX 77843-318, USA Abstract The electrcal double layer
Electrcal double layer: revst based on boundary condtons Jong U. Km Department of Electrcal and Computer Engneerng, Texas A&M Unversty College Staton, TX 77843-318, USA Abstract The electrcal double layer
Solution Thermodynamics
 Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Name ID # For relatively dilute aqueous solutions the molality and molarity are approximately equal.
 Name ID # 1 CHEMISTRY 212, Lect. Sect. 002 Dr. G. L. Roberts Exam #1/Sprng 2000 Thursday, February 24, 2000 CLOSED BOOK EXM No notes or books allowed. Calculators may be used. tomc masses of nterest are
Name ID # 1 CHEMISTRY 212, Lect. Sect. 002 Dr. G. L. Roberts Exam #1/Sprng 2000 Thursday, February 24, 2000 CLOSED BOOK EXM No notes or books allowed. Calculators may be used. tomc masses of nterest are
Diffusion Mass Transfer
 Dffuson Mass Transfer General onsderatons Mass transfer refers to mass n transt due to a speces concentraton gradent n a mture. Must have a mture of two or more speces for mass transfer to occur. The speces
Dffuson Mass Transfer General onsderatons Mass transfer refers to mass n transt due to a speces concentraton gradent n a mture. Must have a mture of two or more speces for mass transfer to occur. The speces
Calculation Of The Interdiffusion Coefficient In The Cu Zn Diffusion Couple
 Calculaton Of The Interdffuson Coeffcent In The Cu Zn Dffuson Couple Adhurm Hoxha a and Henrch Oettel b and Detrch Heger b a Polytechnc Unversty of Trana b TU Bergakademe Freberg, Insttut für Werkstoffwssenschaft
Calculaton Of The Interdffuson Coeffcent In The Cu Zn Dffuson Couple Adhurm Hoxha a and Henrch Oettel b and Detrch Heger b a Polytechnc Unversty of Trana b TU Bergakademe Freberg, Insttut für Werkstoffwssenschaft
Problem adapted reduced models based on Reaction-Diffusion Manifolds (REDIMs)
 Problem adapted reduced models based on Reacton-Dffuson Manfolds (REDIMs) V Bykov, U Maas Thrty-Second Internatonal Symposum on ombuston, Montreal, anada, 3-8 August, 8 Problem Statement: Smulaton of reactng
Problem adapted reduced models based on Reacton-Dffuson Manfolds (REDIMs) V Bykov, U Maas Thrty-Second Internatonal Symposum on ombuston, Montreal, anada, 3-8 August, 8 Problem Statement: Smulaton of reactng
THE IGNITION PARAMETER - A quantification of the probability of ignition
 THE IGNITION PARAMETER - A quantfcaton of the probablty of ton INFUB9-2011 Topc: Modellng of fundamental processes Man author Nels Bjarne K. Rasmussen Dansh Gas Technology Centre (DGC) NBR@dgc.dk Co-author
THE IGNITION PARAMETER - A quantfcaton of the probablty of ton INFUB9-2011 Topc: Modellng of fundamental processes Man author Nels Bjarne K. Rasmussen Dansh Gas Technology Centre (DGC) NBR@dgc.dk Co-author
Chem 2A Exam 1. First letter of your last name
 Frst letter of your last name NAME: PERM# INSTRUCTIONS: Fll n your name, perm number and frst ntal of your last name above. Be sure to show all of your work for full credt. Use the back of the page f necessary.
Frst letter of your last name NAME: PERM# INSTRUCTIONS: Fll n your name, perm number and frst ntal of your last name above. Be sure to show all of your work for full credt. Use the back of the page f necessary.
Amplification and Relaxation of Electron Spin Polarization in Semiconductor Devices
 Amplfcaton and Relaxaton of Electron Spn Polarzaton n Semconductor Devces Yury V. Pershn and Vladmr Prvman Center for Quantum Devce Technology, Clarkson Unversty, Potsdam, New York 13699-570, USA Spn Relaxaton
Amplfcaton and Relaxaton of Electron Spn Polarzaton n Semconductor Devces Yury V. Pershn and Vladmr Prvman Center for Quantum Devce Technology, Clarkson Unversty, Potsdam, New York 13699-570, USA Spn Relaxaton
Supplementary Information. Fundamental Growth Principles of Colloidal Metal Nanoparticles
 Electronc Supplementary Materal (ESI) for CrystEngComm. Ths journal s The Royal Socety of Chemstry 2015 Supplementary Informaton Fundamental Growth Prncples of Collodal Metal Nanopartcles Jörg Polte* a
Electronc Supplementary Materal (ESI) for CrystEngComm. Ths journal s The Royal Socety of Chemstry 2015 Supplementary Informaton Fundamental Growth Prncples of Collodal Metal Nanopartcles Jörg Polte* a
KINETICS OF GAS HYDRATE FORMATION FROM PYROLYSIS GAS IN WATER-IN-OIL EMULSION SYSTEM
 Proceedngs of the 7th Internatonal Conference on Gas Hydrates (ICGH 211), Ednburgh, Scotland, Unted Kngdom, July 17-21, 211. KINETICS OF GAS HYDRATE FORMATION FROM PYROLYSIS GAS IN WATER-IN-OIL EMULSION
Proceedngs of the 7th Internatonal Conference on Gas Hydrates (ICGH 211), Ednburgh, Scotland, Unted Kngdom, July 17-21, 211. KINETICS OF GAS HYDRATE FORMATION FROM PYROLYSIS GAS IN WATER-IN-OIL EMULSION
University of Washington Department of Chemistry Chemistry 452/456 Summer Quarter 2014
 Lecture 16 8/4/14 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 214. Real Vapors and Fugacty Henry s Law accounts or the propertes o extremely dlute soluton. s shown n Fgure
Lecture 16 8/4/14 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 214. Real Vapors and Fugacty Henry s Law accounts or the propertes o extremely dlute soluton. s shown n Fgure
Properties of the living organism. Interaction between living organism and the enviroment. Processing informations. Definitions
 thermodynamcs energy materal Interacton between lvng organsm and the envroment Propertes of the lvng organsm Separaton from the envroment: Strctly controlled energy and materal transport. Changng n the
thermodynamcs energy materal Interacton between lvng organsm and the envroment Propertes of the lvng organsm Separaton from the envroment: Strctly controlled energy and materal transport. Changng n the
Turbulent Flow. Turbulent Flow
 http://www.youtube.com/watch?v=xoll2kedog&feature=related http://br.youtube.com/watch?v=7kkftgx2any http://br.youtube.com/watch?v=vqhxihpvcvu 1. Caothc fluctuatons wth a wde range of frequences and
http://www.youtube.com/watch?v=xoll2kedog&feature=related http://br.youtube.com/watch?v=7kkftgx2any http://br.youtube.com/watch?v=vqhxihpvcvu 1. Caothc fluctuatons wth a wde range of frequences and
Properties of the living organism. Interaction between living organism and the environment. Processing informations. Definitions
 thermodynamcs energy materal Interacton between lvng organsm and the envronment Propertes of the lvng organsm Separaton from the envroment: Strctly controlled energy and materal transport. Changng n the
thermodynamcs energy materal Interacton between lvng organsm and the envronment Propertes of the lvng organsm Separaton from the envroment: Strctly controlled energy and materal transport. Changng n the
2.If the concentration is of bulk phenomenon, it is absorption. (e.g) Absorption of Ink on the surface of a chalk.
 183101 EGIEERIG CHEMISTRY UIT 3 SURFACE CHEMISTRY TERMS AD DEFIITIOS: 1. Adsorpton: Concentraton of lqud or gaseous molecules over the surface of a sold materal s known as adsorpton. It s a surface phenomenon.
183101 EGIEERIG CHEMISTRY UIT 3 SURFACE CHEMISTRY TERMS AD DEFIITIOS: 1. Adsorpton: Concentraton of lqud or gaseous molecules over the surface of a sold materal s known as adsorpton. It s a surface phenomenon.
Lecture 12. Transport in Membranes (2)
 Lecture 12. Transport n embranes (2) odule Flow Patterns - Perfect mxng - Countercurrent flow - Cocurrent flow - Crossflow embrane Cascades External ass-transfer Resstances Concentraton Polarzaton and
Lecture 12. Transport n embranes (2) odule Flow Patterns - Perfect mxng - Countercurrent flow - Cocurrent flow - Crossflow embrane Cascades External ass-transfer Resstances Concentraton Polarzaton and
Mass transfer in multi-component mixtures
 Chapters -0 ex. 7, of 5 of boo See also Krshna & Wesselngh Chem. Eng. Sc. 5(6) 997 86-9 Mass transfer n mult-component mxtures Ron Zevenhoven Åbo Aadem Unversty Thermal and Flow Engneerng Laboratory tel.
Chapters -0 ex. 7, of 5 of boo See also Krshna & Wesselngh Chem. Eng. Sc. 5(6) 997 86-9 Mass transfer n mult-component mxtures Ron Zevenhoven Åbo Aadem Unversty Thermal and Flow Engneerng Laboratory tel.
and Statistical Mechanics Material Properties
 Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Towards the modeling of microgalvanic coupling in aluminum alloys : the choice of boundary conditions
 Presented at the COMSOL Conference 2008 Boston COMSOL Conference BOSTON November 9-11 2008 Towards the modelng of mcrogalvanc couplng n alumnum alloys : the choce of boundary condtons Ncolas Murer *1,
Presented at the COMSOL Conference 2008 Boston COMSOL Conference BOSTON November 9-11 2008 Towards the modelng of mcrogalvanc couplng n alumnum alloys : the choce of boundary condtons Ncolas Murer *1,
Diffusion Fundamentals J. Kärger, P. Heitjans, F. Grinberg, G. Schütz
 Dffuson Fundamentals J. Kärger, P. Hetjans, F. Grnberg, G. Schütz www.dffuson-fundamentals.org Oxygen dffuson through perovskte membranes Hahu Wang,*, Weshen Yang, Crstna Tablet, Jürgen Caro Unversty of
Dffuson Fundamentals J. Kärger, P. Hetjans, F. Grnberg, G. Schütz www.dffuson-fundamentals.org Oxygen dffuson through perovskte membranes Hahu Wang,*, Weshen Yang, Crstna Tablet, Jürgen Caro Unversty of
CHEMICAL REACTIONS AND DIFFUSION
 CHEMICAL REACTIONS AND DIFFUSION A.K.A. NETWORK THERMODYNAMICS BACKGROUND Classcal thermodynamcs descrbes equlbrum states. Non-equlbrum thermodynamcs descrbes steady states. Network thermodynamcs descrbes
CHEMICAL REACTIONS AND DIFFUSION A.K.A. NETWORK THERMODYNAMICS BACKGROUND Classcal thermodynamcs descrbes equlbrum states. Non-equlbrum thermodynamcs descrbes steady states. Network thermodynamcs descrbes
Nucleation kinetics in closed systems
 Nucleaton knetcs n closed systems COST Acton CM142: From molecules to crystals how do organc molecules form crystals? Zdeněk Kožíšek Insttute of Physcs of the Czech Academy of Scences, Prague, Czech Republc
Nucleaton knetcs n closed systems COST Acton CM142: From molecules to crystals how do organc molecules form crystals? Zdeněk Kožíšek Insttute of Physcs of the Czech Academy of Scences, Prague, Czech Republc
Combustion Physics (Day 1 Lecture) Chung K. Law
 Combuston Physcs (Day 1 Lecture) Chung K. Law Robert H. Goddard Professor Prnceton Unversty Prnceton-CEFRC-Combuston Insttute Summer School on Combuston June 20-24, 2016 1 Day 1: Chemcal Thermodynamcs
Combuston Physcs (Day 1 Lecture) Chung K. Law Robert H. Goddard Professor Prnceton Unversty Prnceton-CEFRC-Combuston Insttute Summer School on Combuston June 20-24, 2016 1 Day 1: Chemcal Thermodynamcs
Chapter 3 Thermochemistry of Fuel Air Mixtures
 Chapter 3 Thermochemstry of Fuel Ar Mxtures 3-1 Thermochemstry 3- Ideal Gas Model 3-3 Composton of Ar and Fuels 3-4 Combuston Stochometry t 3-5 The1 st Law of Thermodynamcs and Combuston 3-6 Thermal converson
Chapter 3 Thermochemstry of Fuel Ar Mxtures 3-1 Thermochemstry 3- Ideal Gas Model 3-3 Composton of Ar and Fuels 3-4 Combuston Stochometry t 3-5 The1 st Law of Thermodynamcs and Combuston 3-6 Thermal converson
PES 1120 Spring 2014, Spendier Lecture 6/Page 1
 PES 110 Sprng 014, Spender Lecture 6/Page 1 Lecture today: Chapter 1) Electrc feld due to charge dstrbutons -> charged rod -> charged rng We ntroduced the electrc feld, E. I defned t as an nvsble aura
PES 110 Sprng 014, Spender Lecture 6/Page 1 Lecture today: Chapter 1) Electrc feld due to charge dstrbutons -> charged rod -> charged rng We ntroduced the electrc feld, E. I defned t as an nvsble aura
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM
 ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
Electrostatic Potential from Transmembrane Currents
 Electrostatc Potental from Transmembrane Currents Let s assume that the current densty j(r, t) s ohmc;.e., lnearly proportonal to the electrc feld E(r, t): j = σ c (r)e (1) wth conductvty σ c = σ c (r).
Electrostatc Potental from Transmembrane Currents Let s assume that the current densty j(r, t) s ohmc;.e., lnearly proportonal to the electrc feld E(r, t): j = σ c (r)e (1) wth conductvty σ c = σ c (r).
Supplementary Information:
 Supplementary Informaton: Vsualzaton-based analyss of structural and dynamcal propertes of smulated hydrous slcate melt Bjaya B. Kark 1,2, Dpesh Bhattara 1, Manak Mookherjee 3 and Lars Stxrude 4 1 Department
Supplementary Informaton: Vsualzaton-based analyss of structural and dynamcal propertes of smulated hydrous slcate melt Bjaya B. Kark 1,2, Dpesh Bhattara 1, Manak Mookherjee 3 and Lars Stxrude 4 1 Department
Physics for Scientists & Engineers 2
 Equpotental Surfaces and Lnes Physcs for Scentsts & Engneers 2 Sprng Semester 2005 Lecture 9 January 25, 2005 Physcs for Scentsts&Engneers 2 1 When an electrc feld s present, the electrc potental has a
Equpotental Surfaces and Lnes Physcs for Scentsts & Engneers 2 Sprng Semester 2005 Lecture 9 January 25, 2005 Physcs for Scentsts&Engneers 2 1 When an electrc feld s present, the electrc potental has a
Gasometric Determination of NaHCO 3 in a Mixture
 60 50 40 0 0 5 15 25 35 40 Temperature ( o C) 9/28/16 Gasometrc Determnaton of NaHCO 3 n a Mxture apor Pressure (mm Hg) apor Pressure of Water 1 NaHCO 3 (s) + H + (aq) Na + (aq) + H 2 O (l) + CO 2 (g)
60 50 40 0 0 5 15 25 35 40 Temperature ( o C) 9/28/16 Gasometrc Determnaton of NaHCO 3 n a Mxture apor Pressure (mm Hg) apor Pressure of Water 1 NaHCO 3 (s) + H + (aq) Na + (aq) + H 2 O (l) + CO 2 (g)
Action Nationale de Formation en métallurgie fondamentale, Aussois, octobre 2012
 Atom Dffuson n Solds Alan Portavoce CNRS, IMNP, Faculté des Scences de Sant-Jérôme, Marselle, France Acton Natonale de Formaton en métallurge fondamentale, Aussos, -5 octobre 01 Total energy of solds Stablty
Atom Dffuson n Solds Alan Portavoce CNRS, IMNP, Faculté des Scences de Sant-Jérôme, Marselle, France Acton Natonale de Formaton en métallurge fondamentale, Aussos, -5 octobre 01 Total energy of solds Stablty
The Theory of HPLC. Band Broadening
 The Theory of HPLC Band Broadenng Wherever you see ths symbol, t s mportant to access the on-lne course as there s nteractve materal that cannot be fully shown n ths reference manual. Ams and Objectves
The Theory of HPLC Band Broadenng Wherever you see ths symbol, t s mportant to access the on-lne course as there s nteractve materal that cannot be fully shown n ths reference manual. Ams and Objectves
Thermophysical Properties of Undercooled Alloys: An Overview of the Molecular Simulation Approaches
 Int. J. Mol. Sc. 011, 1, 78-316; do:10.3390/jms101078 OPEN CCESS Revew Internatonal Journal of Molecular Scences ISSN 14-0067 www.mdp.com/journal/jms Thermophyscal Propertes of Undercooled lloys: n Overvew
Int. J. Mol. Sc. 011, 1, 78-316; do:10.3390/jms101078 OPEN CCESS Revew Internatonal Journal of Molecular Scences ISSN 14-0067 www.mdp.com/journal/jms Thermophyscal Propertes of Undercooled lloys: n Overvew
Sample Preparation. Primary Sample Preparation Techniques
 Sample Preparaton Prmary Sample Preparaton Technques Wherever you see ths symbol, t s mportant to access the on-lne course as there s nteractve materal that cannot be fully shown n ths reference manual.
Sample Preparaton Prmary Sample Preparaton Technques Wherever you see ths symbol, t s mportant to access the on-lne course as there s nteractve materal that cannot be fully shown n ths reference manual.
PRESUMABLE CAUSE OF TORNADO EVOLUTION.
 P10.7 PRESUMABLE CAUSE OF TORNAO EVOLUTION. A.Gus kov, (guskov@ssp.ac.ru) Insttute of Sold State Physcs RAS, Chernogolovka, Russa. INTROUCTION. We have obtaned the dependences of perod of eutectc pattern
P10.7 PRESUMABLE CAUSE OF TORNAO EVOLUTION. A.Gus kov, (guskov@ssp.ac.ru) Insttute of Sold State Physcs RAS, Chernogolovka, Russa. INTROUCTION. We have obtaned the dependences of perod of eutectc pattern
Crystal nucleation in sub-microemulsions
 Crystal nucleaton n sub-mcroemulsons Zdeněk Kožíšek Insttute of Physcs AS CR, Praha, Czech Republc kozsek@fzu.cz Hroshma Unversty, Japan, October 13, 211 Web page: http://www.fzu.cz/ kozsek/lectures/mcro211.pdf
Crystal nucleaton n sub-mcroemulsons Zdeněk Kožíšek Insttute of Physcs AS CR, Praha, Czech Republc kozsek@fzu.cz Hroshma Unversty, Japan, October 13, 211 Web page: http://www.fzu.cz/ kozsek/lectures/mcro211.pdf
V T for n & P = constant
 Pchem 365: hermodynamcs -SUMMARY- Uwe Burghaus, Fargo, 5 9 Mnmum requrements for underneath of your pllow. However, wrte your own summary! You need to know the story behnd the equatons : Pressure : olume
Pchem 365: hermodynamcs -SUMMARY- Uwe Burghaus, Fargo, 5 9 Mnmum requrements for underneath of your pllow. However, wrte your own summary! You need to know the story behnd the equatons : Pressure : olume
ReaxFF Potential Functions
 ReaxFF Potental Functons Supportng nformaton for the manuscrpt A ReaxFF Reactve Force Feld for Molecular Dynamcs Smulatons of Hydrocarbon Oxdaton by Kmberly Chenoweth Adr CT van Dun and Wllam A Goddard
ReaxFF Potental Functons Supportng nformaton for the manuscrpt A ReaxFF Reactve Force Feld for Molecular Dynamcs Smulatons of Hydrocarbon Oxdaton by Kmberly Chenoweth Adr CT van Dun and Wllam A Goddard
Lecture 12. Modeling of Turbulent Combustion
 Lecture 12. Modelng of Turbulent Combuston X.S. Ba Modelng of TC Content drect numercal smulaton (DNS) Statstcal approach (RANS) Modelng of turbulent non-premxed flames Modelng of turbulent premxed flames
Lecture 12. Modelng of Turbulent Combuston X.S. Ba Modelng of TC Content drect numercal smulaton (DNS) Statstcal approach (RANS) Modelng of turbulent non-premxed flames Modelng of turbulent premxed flames
5.60 Thermodynamics & Kinetics Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
Energy, Entropy, and Availability Balances Phase Equilibria. Nonideal Thermodynamic Property Models. Selecting an Appropriate Model
 Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Week3, Chapter 4. Position and Displacement. Motion in Two Dimensions. Instantaneous Velocity. Average Velocity
 Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
Appendix II Summary of Important Equations
 W. M. Whte Geochemstry Equatons of State: Ideal GasLaw: Coeffcent of Thermal Expanson: Compressblty: Van der Waals Equaton: The Laws of Thermdynamcs: Frst Law: Appendx II Summary of Important Equatons
W. M. Whte Geochemstry Equatons of State: Ideal GasLaw: Coeffcent of Thermal Expanson: Compressblty: Van der Waals Equaton: The Laws of Thermdynamcs: Frst Law: Appendx II Summary of Important Equatons
Lecture 7: Boltzmann distribution & Thermodynamics of mixing
 Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
8.022 (E&M) Lecture 4
 Topcs: 8.0 (E&M) Lecture 4 More applcatons of vector calculus to electrostatcs: Laplacan: Posson and Laplace equaton url: concept and applcatons to electrostatcs Introducton to conductors Last tme Electrc
Topcs: 8.0 (E&M) Lecture 4 More applcatons of vector calculus to electrostatcs: Laplacan: Posson and Laplace equaton url: concept and applcatons to electrostatcs Introducton to conductors Last tme Electrc
V. Electrostatics. Lecture 25: Diffuse double layer structure
 V. Electrostatcs Lecture 5: Dffuse double layer structure MIT Student Last tme we showed that whenever λ D L the electrolyte has a quas-neutral bulk (or outer ) regon at the geometrcal scale L, where there
V. Electrostatcs Lecture 5: Dffuse double layer structure MIT Student Last tme we showed that whenever λ D L the electrolyte has a quas-neutral bulk (or outer ) regon at the geometrcal scale L, where there
2016 Wiley. Study Session 2: Ethical and Professional Standards Application
 6 Wley Study Sesson : Ethcal and Professonal Standards Applcaton LESSON : CORRECTION ANALYSIS Readng 9: Correlaton and Regresson LOS 9a: Calculate and nterpret a sample covarance and a sample correlaton
6 Wley Study Sesson : Ethcal and Professonal Standards Applcaton LESSON : CORRECTION ANALYSIS Readng 9: Correlaton and Regresson LOS 9a: Calculate and nterpret a sample covarance and a sample correlaton
Understanding Electrophoretic Displays: Transient Current Characteristics of Dispersed Charges in a Non-Polar Medium
 Understandng Electrophoretc Dsplays: Transent Current Characterstcs of Dspersed Charges n a Non-Polar Medum Yoocharn Jeon, Pavel Kornlovtch, Patrca Beck, Zhang-Ln Zhou, Rchard Henze, Tm Koch HP Laboratores
Understandng Electrophoretc Dsplays: Transent Current Characterstcs of Dspersed Charges n a Non-Polar Medum Yoocharn Jeon, Pavel Kornlovtch, Patrca Beck, Zhang-Ln Zhou, Rchard Henze, Tm Koch HP Laboratores
Measurement of Radiation: Exposure. Purpose. Quantitative description of radiation
 Measurement of Radaton: Exposure George Starkschall, Ph.D. Department of Radaton Physcs U.T. M.D. Anderson Cancer Center Purpose To ntroduce the concept of radaton exposure and to descrbe and evaluate
Measurement of Radaton: Exposure George Starkschall, Ph.D. Department of Radaton Physcs U.T. M.D. Anderson Cancer Center Purpose To ntroduce the concept of radaton exposure and to descrbe and evaluate
Module 1 : The equation of continuity. Lecture 1: Equation of Continuity
 1 Module 1 : The equaton of contnuty Lecture 1: Equaton of Contnuty 2 Advanced Heat and Mass Transfer: Modules 1. THE EQUATION OF CONTINUITY : Lectures 1-6 () () () (v) (v) Overall Mass Balance Momentum
1 Module 1 : The equaton of contnuty Lecture 1: Equaton of Contnuty 2 Advanced Heat and Mass Transfer: Modules 1. THE EQUATION OF CONTINUITY : Lectures 1-6 () () () (v) (v) Overall Mass Balance Momentum
Electrochemistry High Temperature Concepts
 Electrochemstry Hgh Temperature Concepts Ngel Sammes POSTECH South Korea nsammes@postech.ac.kr 2 nd Jont European Summer School on Fuel Cell and Hydrogen Technology OK Let s Get Started Introducton to
Electrochemstry Hgh Temperature Concepts Ngel Sammes POSTECH South Korea nsammes@postech.ac.kr 2 nd Jont European Summer School on Fuel Cell and Hydrogen Technology OK Let s Get Started Introducton to
NAME and Section No. it is found that 0.6 mol of O
 NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
Formal solvers of the RT equation
 Formal solvers of the RT equaton Formal RT solvers Runge- Kutta (reference solver) Pskunov N.: 979, Master Thess Long characterstcs (Feautrer scheme) Cannon C.J.: 970, ApJ 6, 55 Short characterstcs (Hermtan
Formal solvers of the RT equaton Formal RT solvers Runge- Kutta (reference solver) Pskunov N.: 979, Master Thess Long characterstcs (Feautrer scheme) Cannon C.J.: 970, ApJ 6, 55 Short characterstcs (Hermtan
Quantum states of deuterons in palladium
 Tsuchda K. Quantum states of deuterons n palladum. n Tenth Internatonal Conference on Cold Fuson. 003. Cambrdge MA: LENR-CANR.org. Ths paper was presented at the 10th Internatonal Conference on Cold Fuson.
Tsuchda K. Quantum states of deuterons n palladum. n Tenth Internatonal Conference on Cold Fuson. 003. Cambrdge MA: LENR-CANR.org. Ths paper was presented at the 10th Internatonal Conference on Cold Fuson.
Frequency dependence of the permittivity
 Frequency dependence of the permttvty February 7, 016 In materals, the delectrc constant and permeablty are actually frequency dependent. Ths does not affect our results for sngle frequency modes, but
Frequency dependence of the permttvty February 7, 016 In materals, the delectrc constant and permeablty are actually frequency dependent. Ths does not affect our results for sngle frequency modes, but
Lecture 10. Reading: Notes and Brennan Chapter 5
 Lecture tatstcal Mechancs and Densty of tates Concepts Readng: otes and Brennan Chapter 5 Georga Tech C 645 - Dr. Alan Doolttle C 645 - Dr. Alan Doolttle Georga Tech How do electrons and holes populate
Lecture tatstcal Mechancs and Densty of tates Concepts Readng: otes and Brennan Chapter 5 Georga Tech C 645 - Dr. Alan Doolttle C 645 - Dr. Alan Doolttle Georga Tech How do electrons and holes populate
Turbulent Nonpremixed Flames
 School of Aerospace Engneerng Turbulent Nonpremxed Flames Jerry Setzman. 5 Mole Fracton.15.1.5 CH4 HO HCO x 1 Temperature Methane Flame.1..3 Dstance (cm) 15 1 5 Temperature (K) TurbulentNonpremxed -1 School
School of Aerospace Engneerng Turbulent Nonpremxed Flames Jerry Setzman. 5 Mole Fracton.15.1.5 CH4 HO HCO x 1 Temperature Methane Flame.1..3 Dstance (cm) 15 1 5 Temperature (K) TurbulentNonpremxed -1 School
INFLUENCE OF AXIAL MIXING AND PARTICLE SIZE NON-UNIFORMITY ON PERFORMANCE OF CHROMATOGRAPHIC REACTORS
 14 th European onference on Mxng Warszawa, 1-13 September 1 INFUENE OF XI MIXING N PRTIE SIZE NON-UNIFORMITY ON PERFORMNE OF HROMTOGRPHI RETORS Eugenusz Molga, Mchał ewak, nna Ostanewcz-ydzk Warsaw Unversty
14 th European onference on Mxng Warszawa, 1-13 September 1 INFUENE OF XI MIXING N PRTIE SIZE NON-UNIFORMITY ON PERFORMNE OF HROMTOGRPHI RETORS Eugenusz Molga, Mchał ewak, nna Ostanewcz-ydzk Warsaw Unversty
General Thermodynamics for Process Simulation. Dr. Jungho Cho, Professor Department of Chemical Engineering Dong Yang University
 General Thermodynamcs for Process Smulaton Dr. Jungho Cho, Professor Department of Chemcal Engneerng Dong Yang Unversty Four Crtera for Equlbra μ = μ v Stuaton α T = T β α β P = P l μ = μ l1 l 2 Thermal
General Thermodynamcs for Process Smulaton Dr. Jungho Cho, Professor Department of Chemcal Engneerng Dong Yang Unversty Four Crtera for Equlbra μ = μ v Stuaton α T = T β α β P = P l μ = μ l1 l 2 Thermal
KINETICS OF PHOTOCHEMICAL DECOMPOSITION OF SOLIDS (I) Introduction. The Rate Equation
 KINETICS OF PHOTOCHEMICL DECOMPOSITION OF SOLIDS (I) D. Ftu model whch descrbes the knetcs of photochemcal decomposton: a (S) b (S) cc (the reacton area beng located near the reactant surface) s presented.
KINETICS OF PHOTOCHEMICL DECOMPOSITION OF SOLIDS (I) D. Ftu model whch descrbes the knetcs of photochemcal decomposton: a (S) b (S) cc (the reacton area beng located near the reactant surface) s presented.
Brownian-Dynamics Simulation of Colloidal Suspensions with Kob-Andersen Type Lennard-Jones Potentials 1
 Brownan-Dynamcs Smulaton of Collodal Suspensons wth Kob-Andersen Type Lennard-Jones Potentals 1 Yuto KIMURA 2 and Mcho TOKUYAMA 3 Summary Extensve Brownan-dynamcs smulatons of bnary collodal suspenton
Brownan-Dynamcs Smulaton of Collodal Suspensons wth Kob-Andersen Type Lennard-Jones Potentals 1 Yuto KIMURA 2 and Mcho TOKUYAMA 3 Summary Extensve Brownan-dynamcs smulatons of bnary collodal suspenton
Natural Gas Conversion in Monolithic Catalysts: Interaction of Chemical Reactions and Transport Phenomena
 Natural Gas Converson VI, Studes n Surface Scence and Catalyss 136, E. Iglesa, J.J. Spvey, T.H. Flesch (eds.), p. 251-258, Elsever, 21. Natural Gas Converson n Monolthc Catalysts: Interacton of Chemcal
Natural Gas Converson VI, Studes n Surface Scence and Catalyss 136, E. Iglesa, J.J. Spvey, T.H. Flesch (eds.), p. 251-258, Elsever, 21. Natural Gas Converson n Monolthc Catalysts: Interacton of Chemcal
CHAPTER 7 ENERGY BALANCES SYSTEM SYSTEM. * What is energy? * Forms of Energy. - Kinetic energy (KE) - Potential energy (PE) PE = mgz
 SYSTM CHAPTR 7 NRGY BALANCS 1 7.1-7. SYSTM nergy & 1st Law of Thermodynamcs * What s energy? * Forms of nergy - Knetc energy (K) K 1 mv - Potental energy (P) P mgz - Internal energy (U) * Total nergy,
SYSTM CHAPTR 7 NRGY BALANCS 1 7.1-7. SYSTM nergy & 1st Law of Thermodynamcs * What s energy? * Forms of nergy - Knetc energy (K) K 1 mv - Potental energy (P) P mgz - Internal energy (U) * Total nergy,
APPENDIX F A DISPLACEMENT-BASED BEAM ELEMENT WITH SHEAR DEFORMATIONS. Never use a Cubic Function Approximation for a Non-Prismatic Beam
 APPENDIX F A DISPACEMENT-BASED BEAM EEMENT WITH SHEAR DEFORMATIONS Never use a Cubc Functon Approxmaton for a Non-Prsmatc Beam F. INTRODUCTION { XE "Shearng Deformatons" }In ths appendx a unque development
APPENDIX F A DISPACEMENT-BASED BEAM EEMENT WITH SHEAR DEFORMATIONS Never use a Cubc Functon Approxmaton for a Non-Prsmatc Beam F. INTRODUCTION { XE "Shearng Deformatons" }In ths appendx a unque development
Lecture 3 Electrostatic effects November 6, 2009
 The stablty of thn (soft) flms Lecture 3 Electrostatc effects November 6, 009 Ian Morrson 009 Revew of Lecture The dsjonng pressure s a jump n pressure at the boundary. It does not vary between the plates.
The stablty of thn (soft) flms Lecture 3 Electrostatc effects November 6, 009 Ian Morrson 009 Revew of Lecture The dsjonng pressure s a jump n pressure at the boundary. It does not vary between the plates.
,..., k N. , k 2. ,..., k i. The derivative with respect to temperature T is calculated by using the chain rule: & ( (5) dj j dt = "J j. k i.
 Suppleentary Materal Dervaton of Eq. 1a. Assue j s a functon of the rate constants for the N coponent reactons: j j (k 1,,..., k,..., k N ( The dervatve wth respect to teperature T s calculated by usng
Suppleentary Materal Dervaton of Eq. 1a. Assue j s a functon of the rate constants for the N coponent reactons: j j (k 1,,..., k,..., k N ( The dervatve wth respect to teperature T s calculated by usng
Numerical analysis on the agglomeration behavior of fine particles in plane jet
 Numercal analyss on the agglomeraton behavor of fne partcles n plane et Mn Guo 1, a, Ja L 2, Xn Su 1 and Gurong Yang 1 1 Tann Academy of Envronmental Scences, Tann, 300191, Chna; 2 The Fourth Research
Numercal analyss on the agglomeraton behavor of fne partcles n plane et Mn Guo 1, a, Ja L 2, Xn Su 1 and Gurong Yang 1 1 Tann Academy of Envronmental Scences, Tann, 300191, Chna; 2 The Fourth Research
Ph.D. Qualifying Examination in Kinetics and Reactor Design
 Knetcs and Reactor Desgn Ph.D.Qualfyng Examnaton January 2006 Instructons Ph.D. Qualfyng Examnaton n Knetcs and Reactor Desgn January 2006 Unversty of Texas at Austn Department of Chemcal Engneerng 1.
Knetcs and Reactor Desgn Ph.D.Qualfyng Examnaton January 2006 Instructons Ph.D. Qualfyng Examnaton n Knetcs and Reactor Desgn January 2006 Unversty of Texas at Austn Department of Chemcal Engneerng 1.
Over-Temperature protection for IGBT modules
 Over-Temperature protecton for IGBT modules Ke Wang 1, Yongjun Lao 2, Gaosheng Song 1, Xanku Ma 1 1 Mtsubsh Electrc & Electroncs (Shangha) Co., Ltd., Chna Room2202, Tower 3, Kerry Plaza, No.1-1 Zhongxns
Over-Temperature protecton for IGBT modules Ke Wang 1, Yongjun Lao 2, Gaosheng Song 1, Xanku Ma 1 1 Mtsubsh Electrc & Electroncs (Shangha) Co., Ltd., Chna Room2202, Tower 3, Kerry Plaza, No.1-1 Zhongxns
1st Year Thermodynamic Lectures Dr Mark R. Wormald BIBLIOGRAPHY
 M.R.W. -- Thermodynamcs 5 1st Year Thermodynamc Lectures Dr Mark R. Wormald Soluton thermodynamcs :- Lecture 1. Behavour of deal solutons (and solvents). Defnton of dealty. Solvent n dlute solutons, collgatve
M.R.W. -- Thermodynamcs 5 1st Year Thermodynamc Lectures Dr Mark R. Wormald Soluton thermodynamcs :- Lecture 1. Behavour of deal solutons (and solvents). Defnton of dealty. Solvent n dlute solutons, collgatve
ON THE CURENT DENSITY AND OVERTENSION SIGNS III. THE CASE OF THE INTERFACE BETWEEN TWO IMMISCIBLE ELECTROLYTE SOLUTIONS IN OPEN CIRCUIT CONDITION
 ON HE UREN DENSY ND OVERENSON SGNS. HE SE OF HE NERFE BEWEEN WO MMSBLE ELEROLYE SOLUONS N OPEN RU ONDON. Mhalcuc and S. Lupu abstract: For a spontaneous electrode reacton the entropy producton anhe current
ON HE UREN DENSY ND OVERENSON SGNS. HE SE OF HE NERFE BEWEEN WO MMSBLE ELEROLYE SOLUONS N OPEN RU ONDON. Mhalcuc and S. Lupu abstract: For a spontaneous electrode reacton the entropy producton anhe current
ˆ (0.10 m) E ( N m /C ) 36 ˆj ( j C m)
 7.. = = 3 = 4 = 5. The electrc feld s constant everywhere between the plates. Ths s ndcated by the electrc feld vectors, whch are all the same length and n the same drecton. 7.5. Model: The dstances to
7.. = = 3 = 4 = 5. The electrc feld s constant everywhere between the plates. Ths s ndcated by the electrc feld vectors, whch are all the same length and n the same drecton. 7.5. Model: The dstances to
Composite Hypotheses testing
 Composte ypotheses testng In many hypothess testng problems there are many possble dstrbutons that can occur under each of the hypotheses. The output of the source s a set of parameters (ponts n a parameter
Composte ypotheses testng In many hypothess testng problems there are many possble dstrbutons that can occur under each of the hypotheses. The output of the source s a set of parameters (ponts n a parameter
CALCULATION OF DIFFUSION COEFFICIENT OF ION IN MULTICOMPONENT SOLUTION FOR ION MOVEMENT IN CONCRETE
 CALCULATION OF DIFFUSION COEFFICIENT OF ION IN MULTICOMPONENT SOLUTION FOR ION MOVEMENT IN CONCRETE Worapatt RITTHICHAUY *1, Takafum SUGIYAMA * and Yukkazu TSUJI *3 ABSTRACT: A method for calculatng the
CALCULATION OF DIFFUSION COEFFICIENT OF ION IN MULTICOMPONENT SOLUTION FOR ION MOVEMENT IN CONCRETE Worapatt RITTHICHAUY *1, Takafum SUGIYAMA * and Yukkazu TSUJI *3 ABSTRACT: A method for calculatng the
PART I: MULTIPLE CHOICE (32 questions, each multiple choice question has a 2-point value, 64 points total).
 CHEMISTRY 123-07 Mdterm #2 answer key November 04, 2010 Statstcs: Average: 68 p (68%); Hghest: 91 p (91%); Lowest: 37 p (37%) Number of students performng at or above average: 58 (53%) Number of students
CHEMISTRY 123-07 Mdterm #2 answer key November 04, 2010 Statstcs: Average: 68 p (68%); Hghest: 91 p (91%); Lowest: 37 p (37%) Number of students performng at or above average: 58 (53%) Number of students
Module 3: The Whole-Process Perspective for Thermochemical Hydrogen
 "Thermodynamc Analyss of Processes for Hydrogen Generaton by Decomposton of Water" by John P. O'Connell Department of Chemcal Engneerng Unversty of Vrgna Charlottesvlle, VA 2294-4741 A Set of Energy Educaton
"Thermodynamc Analyss of Processes for Hydrogen Generaton by Decomposton of Water" by John P. O'Connell Department of Chemcal Engneerng Unversty of Vrgna Charlottesvlle, VA 2294-4741 A Set of Energy Educaton
Influence Of Operating Conditions To The Effectiveness Of Extractive Distillation Columns
 Influence Of Operatng Condtons To The Effectveness Of Extractve Dstllaton Columns N.A. Vyazmna Moscov State Unversty Of Envrnmental Engneerng, Department Of Chemcal Engneerng Ul. Staraya Basmannaya 21/4,
Influence Of Operatng Condtons To The Effectveness Of Extractve Dstllaton Columns N.A. Vyazmna Moscov State Unversty Of Envrnmental Engneerng, Department Of Chemcal Engneerng Ul. Staraya Basmannaya 21/4,
