Gouy-Chapman model (1910) The double layer is not as compact as in Helmholtz rigid layer.
|
|
|
- Megan Cameron
- 6 years ago
- Views:
Transcription
1 CHE465/865, 6-3, Lecture 1, 7 nd Sep., 6 Gouy-Chapman model (191) The double layer s not as compact as n Helmholtz rgd layer. Consder thermal motons of ons: Tendency to ncrease the entropy and make the layer dffuse (whle electrostatc attracton tends to keep ons close to nterface and make the layer more rgd). etal Soluton Dstrbuton of ons: determned by electrostatcs and stat. mechancs Assumptons: Ions: pont ons, hghly moble Solvent: delectrc contnuum etal: perfect conductor σ x Dstrbuton of ons wth dstance x from nterface: apply Boltzmann statstcs zf n = n exp n : partcle densty (#partcles/cm3) Regons of large ϕ ( x) S ( ϕ ( x) ϕ ) RT : reduced densty of postve ons, enhanced densty of anons (compared to bulk).
2 Charge densty nvolvng all onc speces: ( ) ρ x = n z e wth e = zf = n z e exp S ( ϕ ( x) ϕ ) RT Galvan potental n soluton obeys Posson s equaton: d ϕ dx ( x) ρ ( x) = Resultng expressons s the Posson-Boltzmann equaton: d ϕ dx ( x) 1 z Fϕ = n z q x = x εε εε S exp, wher e ϕ ( ) ϕ ( ) ϕ RT Note: Ths equaton s precsely equal to the Debye-Hückel theory of onc nteractons n dlute solutons, whch determnes the charge dstrbuton around a central on. -19 C Next: Let s consder a z-z-electrolyte (*) ρ ( x) ( ) ϕ ( ) zfϕ x zf x = n zq exp exp RT RT
3 Debye-Hückel approxmaton Let s further assume that the potental varaton s small, zf ϕ RT ( x) << 1.e. potental varatons are ϕ << 5 mv at room-t. Ths wellknown approxmaton corresponds to the so-called Debye-Hückel approxmaton. If we use ths approxmaton n the charge dstrbuton (*) of the z-z electrolyte and then nsert ths charge dstrbuton nto the Posson-Boltzmann equaton, then we arrve at the lnear Posson-Boltzmann equaton: d ϕ dx = κ ϕ Here, κ s the nverse Debye-length, ( zf ) 1/ c 1 κ = = εε RT L D, c : bulk electrolyte concentraton (far away from nterface) κ or ts nverse L D are mportant characterstcs of the electrolyte. Debye-length L D : quanttatve measure of wdth of space charge regon wthn whch ϕ ( x) decreases from ϕ to tremendous mportance n electrochemstry and bology. S ϕ
4 The followng effects are mportant to remember: Larger electrolyte concentraton c smaller.a.w: the double layer becomes less dffuse Hgher temperature larger L D.a.w.: the double layer becomes more dffuse L D Potental and charge dstrbuton n the electrolyte soluton are gven n Debye-Hückel approxmaton by: σ ϕ = εε κ ( x) exp ( κ x) ( x) = exp ( x) ρ σ κ κ.e. they are exponental functons of the dstance from the nterface,.e. the excess surface charge densty σ on the metal s balanced by an exponentally decayng space-charge layer n soluton. The double layer capacty n ths approxmaton s gven by C d,dh εε = εε κ = L.e. t s gven by the plate capactor formula wth the Debye length as the effectve plate separaton. D
5 Table: Debye length at varous electrolyte concentratons (1-1 electrolyte) c / mol l L D / Å Hgher concentratons: steeper potental drop n soluton narrower space charge regon, smaller L D hgher double layer capactes, C d,dh In practce: The Debye-Hückel (DH) )approxmaton s not vald at large electrolyte concentraton. It works well as long as the potental varaton does not exceed ϕ 8 mv.
6 General Case: Nonlnear Posson-Boltzmann-equaton For a z-z-electrolyte wth concentraton c the Posson- Boltzmann equaton has an explct soluton. Ths wll be added as an appendx. Here only the man results wll be gven. Potental dstrbuton n soluton etal Soluton ϕ (nterface) ϕ, = ϕ - ϕ S ϕ S (bulk) σ x The relaton between potental and charge densty of the dffuse layer s gven by dϕ 1/ zfϕ, = = ( 8RT c ) snh RT σ εε εε dx x= Dfferentatng ths expresson gves the dfferental capacty n the Gouy-Chapman model: σ εε zfϕ, d,gc = = cosh, where ϕ, = pzc ϕ, LD RT C E E
7 The varaton of ths capactance wth electrode potental E s thus gven by C d,gc Ths model predcts correctly the mnmum n capacty at the pzc. For large E, t predcts an unlmted rse of the capacty. E-E pzc Why would the DL capacty rse unlmtedly n ths approach? Whch assumpton s responsble for ths unphyscal behavour? It s assumed that ons are pont charges, whch could approach the electrode surface arbtrarly close wth ncreasng E. Ths leads to a very small charge separaton (consder the plate capactor as an analogue!). The capacty rses wthout lmt. What s the soluton out of ths capacty catastrophe??? Ions have a fnte sze! Arbtrarly close approach s not possble. Ions are stopped from approachng the electrode at dstances that correspond to ther rad. A further refnement of the double layer models takes ths effect nto account.
8 The Stern odel (194) accounts for fnte sze of ons combnes the Helmholtz and Gouy- Chapmann models two parts of double layer: (a) compact layer ( rgd layer ) of ons at dstance of closes approach (b) dffuse layer. The compact layer, x < x H, s chargefree (lnear varaton of potental)! etal σ compact layer x H dffuse layer Soluton athematcally: two capactors n seres wth total capacty C C C x εε H = + = + L εε cosh z d dff,h dff,gc, Far from E pzc (large ϕ, = E Epzc ): D Fϕ RT Cdff,H << Cdff,GC Cd Cdff,H Helmholtz, rgd Close to E pzc (small ϕ, = E Epzc ): Cdff,H >> Cdff,GC Cd Cdff,GC Gouy-Chapman, dffuse Remember: the smaller guy always wns (well, not always, but here)!
9 Effect of electrolyte concentraton: smaller electrolyte concentraton smaller κ (L D larger), double s more dffuse! C dff,gc becomes more mportant The Stern model reproduces gross features of real systems. Potental dstrbuton n nterfacal layer and capacty varaton wth E are shown n the fgures below. What happens upon ncreasng the electrolyte concentraton? Potental dstrbuton Double layer capactance charge free moble ons
10 Further refnement: Grahame model (1947) Some ons (usually anons) loose hydraton shell smaller rad! Ions of smaller rad could approach the electrode closer. Dstngush three dfferent regons! Inner Helmholtz plane (IHP): through centers of small, partally solvated ons Outer Helmholtz plane (OHP): through centers of fully solvated ons Outsde OHP: dffuse layer
11 Ths s the best model so far. It shows good correspondence to the expermental data. Why are the potental dstrbutons (n (b)) dfferent for postve and negatve E? Ths s due to a dfference between anons and catons. Anons have a less rgd solvaton shell. They become more easly desolvated. Thus, desolvated anons could form the IHP. Ths happens at postve E. Catons keep ther solvaton shell. They, thus, cannot approach the electrode closer than to the OHP.
12 Appendx: Electrocapllary measurements Only applcable to lqud electrodes (metals), based on measurement of surface tenson (Lppmann s method). The method was developed specfcally for mercury electrodes. Ths s a null-pont technque,.e. the measurement of a physcal property s performed when the system has reached an equlbrum state. Such measurements are rather accurate. Balanced forces: Surface tenson of mercury n the capllary counterbalances the force of gravty π r γ cosθ = π r ρ hg c c Hg
13 r c s the capllary radus, γ s the surface tenson, θ s the contact angle h s the heght of the capllary column of Hg. In the measurement, the contact angle θ s measured wth a mcroscope. Above relaton then provdes values of the surface tenson γ. As the expermental parameter, the electrode potental E can be vared. What happens upon changng E? When the system s balanced (reachng a new equlbrum mnmum of Gbbs free energy!), the change n surface energy s exactly balanced by the electrcal work, A γ = QdE s d where A s s the surface area and Q s the excess charge on the surface. Therefore, the charge densty on the Hg electrode s gven by Q dγ σ = =. As de It s, thus, possble to measure the surface charge as a functon of E.
Chapter 2. Electrode/electrolyte interface: ----Structure and properties
 Chapter 2 Electrode/electrolyte nterface: ----Structure and propertes Electrochemcal reactons are nterfacal reactons, the structure and propertes of electrode / electrolytc soluton nterface greatly nfluences
Chapter 2 Electrode/electrolyte nterface: ----Structure and propertes Electrochemcal reactons are nterfacal reactons, the structure and propertes of electrode / electrolytc soluton nterface greatly nfluences
V. Electrostatics. Lecture 25: Diffuse double layer structure
 V. Electrostatcs Lecture 5: Dffuse double layer structure MIT Student Last tme we showed that whenever λ D L the electrolyte has a quas-neutral bulk (or outer ) regon at the geometrcal scale L, where there
V. Electrostatcs Lecture 5: Dffuse double layer structure MIT Student Last tme we showed that whenever λ D L the electrolyte has a quas-neutral bulk (or outer ) regon at the geometrcal scale L, where there
1) Silicon oxide has a typical surface potential in an aqueous medium of ϕ,0
 1) Slcon oxde has a typcal surface potental n an aqueous medum of ϕ, = 7 mv n 5 mm l at ph 9. Whch concentraton of catons do you roughly expect close to the surface? What s the average dstance between
1) Slcon oxde has a typcal surface potental n an aqueous medum of ϕ, = 7 mv n 5 mm l at ph 9. Whch concentraton of catons do you roughly expect close to the surface? What s the average dstance between
1. Mean-Field Theory. 2. Bjerrum length
 1. Mean-Feld Theory Contnuum models lke the Posson-Nernst-Planck equatons are mean-feld approxmatons whch descrbe how dscrete ons are affected by the mean concentratons c and potental φ. Each on mgrates
1. Mean-Feld Theory Contnuum models lke the Posson-Nernst-Planck equatons are mean-feld approxmatons whch descrbe how dscrete ons are affected by the mean concentratons c and potental φ. Each on mgrates
Electrical double layer: revisit based on boundary conditions
 Electrcal double layer: revst based on boundary condtons Jong U. Km Department of Electrcal and Computer Engneerng, Texas A&M Unversty College Staton, TX 77843-318, USA Abstract The electrcal double layer
Electrcal double layer: revst based on boundary condtons Jong U. Km Department of Electrcal and Computer Engneerng, Texas A&M Unversty College Staton, TX 77843-318, USA Abstract The electrcal double layer
Electrochemical Equilibrium Electromotive Force
 CHM465/865, 24-3, Lecture 5-7, 2 th Sep., 24 lectrochemcal qulbrum lectromotve Force Relaton between chemcal and electrc drvng forces lectrochemcal system at constant T and p: consder Gbbs free energy
CHM465/865, 24-3, Lecture 5-7, 2 th Sep., 24 lectrochemcal qulbrum lectromotve Force Relaton between chemcal and electrc drvng forces lectrochemcal system at constant T and p: consder Gbbs free energy
1st Year Thermodynamic Lectures Dr Mark R. Wormald BIBLIOGRAPHY
 M.R.W. -- Thermodynamcs 5 1st Year Thermodynamc Lectures Dr Mark R. Wormald Soluton thermodynamcs :- Lecture 1. Behavour of deal solutons (and solvents). Defnton of dealty. Solvent n dlute solutons, collgatve
M.R.W. -- Thermodynamcs 5 1st Year Thermodynamc Lectures Dr Mark R. Wormald Soluton thermodynamcs :- Lecture 1. Behavour of deal solutons (and solvents). Defnton of dealty. Solvent n dlute solutons, collgatve
Appendix II Summary of Important Equations
 W. M. Whte Geochemstry Equatons of State: Ideal GasLaw: Coeffcent of Thermal Expanson: Compressblty: Van der Waals Equaton: The Laws of Thermdynamcs: Frst Law: Appendx II Summary of Important Equatons
W. M. Whte Geochemstry Equatons of State: Ideal GasLaw: Coeffcent of Thermal Expanson: Compressblty: Van der Waals Equaton: The Laws of Thermdynamcs: Frst Law: Appendx II Summary of Important Equatons
ˆ (0.10 m) E ( N m /C ) 36 ˆj ( j C m)
 7.. = = 3 = 4 = 5. The electrc feld s constant everywhere between the plates. Ths s ndcated by the electrc feld vectors, whch are all the same length and n the same drecton. 7.5. Model: The dstances to
7.. = = 3 = 4 = 5. The electrc feld s constant everywhere between the plates. Ths s ndcated by the electrc feld vectors, whch are all the same length and n the same drecton. 7.5. Model: The dstances to
Lecture 3 Electrostatic effects November 6, 2009
 The stablty of thn (soft) flms Lecture 3 Electrostatc effects November 6, 009 Ian Morrson 009 Revew of Lecture The dsjonng pressure s a jump n pressure at the boundary. It does not vary between the plates.
The stablty of thn (soft) flms Lecture 3 Electrostatc effects November 6, 009 Ian Morrson 009 Revew of Lecture The dsjonng pressure s a jump n pressure at the boundary. It does not vary between the plates.
Week3, Chapter 4. Position and Displacement. Motion in Two Dimensions. Instantaneous Velocity. Average Velocity
 Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
PES 1120 Spring 2014, Spendier Lecture 6/Page 1
 PES 110 Sprng 014, Spender Lecture 6/Page 1 Lecture today: Chapter 1) Electrc feld due to charge dstrbutons -> charged rod -> charged rng We ntroduced the electrc feld, E. I defned t as an nvsble aura
PES 110 Sprng 014, Spender Lecture 6/Page 1 Lecture today: Chapter 1) Electrc feld due to charge dstrbutons -> charged rod -> charged rng We ntroduced the electrc feld, E. I defned t as an nvsble aura
Review of Classical Thermodynamics
 Revew of Classcal hermodynamcs Physcs 4362, Lecture #1, 2 Syllabus What s hermodynamcs? 1 [A law] s more mpressve the greater the smplcty of ts premses, the more dfferent are the knds of thngs t relates,
Revew of Classcal hermodynamcs Physcs 4362, Lecture #1, 2 Syllabus What s hermodynamcs? 1 [A law] s more mpressve the greater the smplcty of ts premses, the more dfferent are the knds of thngs t relates,
Electrostatic Potential from Transmembrane Currents
 Electrostatc Potental from Transmembrane Currents Let s assume that the current densty j(r, t) s ohmc;.e., lnearly proportonal to the electrc feld E(r, t): j = σ c (r)e (1) wth conductvty σ c = σ c (r).
Electrostatc Potental from Transmembrane Currents Let s assume that the current densty j(r, t) s ohmc;.e., lnearly proportonal to the electrc feld E(r, t): j = σ c (r)e (1) wth conductvty σ c = σ c (r).
Introduction to Statistical Methods
 Introducton to Statstcal Methods Physcs 4362, Lecture #3 hermodynamcs Classcal Statstcal Knetc heory Classcal hermodynamcs Macroscopc approach General propertes of the system Macroscopc varables 1 hermodynamc
Introducton to Statstcal Methods Physcs 4362, Lecture #3 hermodynamcs Classcal Statstcal Knetc heory Classcal hermodynamcs Macroscopc approach General propertes of the system Macroscopc varables 1 hermodynamc
Mass Transfer Processes
 Mass Transfer Processes S. Majd Hassanzadeh Department of Earth Scences Faculty of Geoscences Utrecht Unversty Outlne: 1. Measures of Concentraton 2. Volatlzaton and Dssoluton 3. Adsorpton Processes 4.
Mass Transfer Processes S. Majd Hassanzadeh Department of Earth Scences Faculty of Geoscences Utrecht Unversty Outlne: 1. Measures of Concentraton 2. Volatlzaton and Dssoluton 3. Adsorpton Processes 4.
Differentiating Gaussian Processes
 Dfferentatng Gaussan Processes Andrew McHutchon Aprl 17, 013 1 Frst Order Dervatve of the Posteror Mean The posteror mean of a GP s gven by, f = x, X KX, X 1 y x, X α 1 Only the x, X term depends on the
Dfferentatng Gaussan Processes Andrew McHutchon Aprl 17, 013 1 Frst Order Dervatve of the Posteror Mean The posteror mean of a GP s gven by, f = x, X KX, X 1 y x, X α 1 Only the x, X term depends on the
Chapter 18, Part 1. Fundamentals of Atmospheric Modeling
 Overhead Sldes for Chapter 18, Part 1 of Fundamentals of Atmospherc Modelng by Mark Z. Jacobson Department of Cvl & Envronmental Engneerng Stanford Unversty Stanford, CA 94305-4020 January 30, 2002 Types
Overhead Sldes for Chapter 18, Part 1 of Fundamentals of Atmospherc Modelng by Mark Z. Jacobson Department of Cvl & Envronmental Engneerng Stanford Unversty Stanford, CA 94305-4020 January 30, 2002 Types
If two volatile and miscible liquids are combined to form a solution, Raoult s law is not obeyed. Use the experimental data in Table 9.
 9.9 Real Solutons Exhbt Devatons from Raoult s Law If two volatle and mscble lquds are combned to form a soluton, Raoult s law s not obeyed. Use the expermental data n Table 9.3: Physcal Chemstry 00 Pearson
9.9 Real Solutons Exhbt Devatons from Raoult s Law If two volatle and mscble lquds are combned to form a soluton, Raoult s law s not obeyed. Use the expermental data n Table 9.3: Physcal Chemstry 00 Pearson
Week 9 Chapter 10 Section 1-5
 Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Physics 607 Exam 1. ( ) = 1, Γ( z +1) = zγ( z) x n e x2 dx = 1. e x2
 Physcs 607 Exam 1 Please be well-organzed, and show all sgnfcant steps clearly n all problems. You are graded on your wor, so please do not just wrte down answers wth no explanaton! Do all your wor on
Physcs 607 Exam 1 Please be well-organzed, and show all sgnfcant steps clearly n all problems. You are graded on your wor, so please do not just wrte down answers wth no explanaton! Do all your wor on
Supplementary Notes for Chapter 9 Mixture Thermodynamics
 Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Solution Thermodynamics
 Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Answers Problem Set 2 Chem 314A Williamsen Spring 2000
 Answers Problem Set Chem 314A Wllamsen Sprng 000 1) Gve me the followng crtcal values from the statstcal tables. a) z-statstc,-sded test, 99.7% confdence lmt ±3 b) t-statstc (Case I), 1-sded test, 95%
Answers Problem Set Chem 314A Wllamsen Sprng 000 1) Gve me the followng crtcal values from the statstcal tables. a) z-statstc,-sded test, 99.7% confdence lmt ±3 b) t-statstc (Case I), 1-sded test, 95%
Open Systems: Chemical Potential and Partial Molar Quantities Chemical Potential
 Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
The equation of motion of a dynamical system is given by a set of differential equations. That is (1)
 Dynamcal Systems Many engneerng and natural systems are dynamcal systems. For example a pendulum s a dynamcal system. State l The state of the dynamcal system specfes t condtons. For a pendulum n the absence
Dynamcal Systems Many engneerng and natural systems are dynamcal systems. For example a pendulum s a dynamcal system. State l The state of the dynamcal system specfes t condtons. For a pendulum n the absence
Grand canonical Monte Carlo simulations of bulk electrolytes and calcium channels
 Grand canoncal Monte Carlo smulatons of bulk electrolytes and calcum channels Thess of Ph.D. dssertaton Prepared by: Attla Malascs M.Sc. n Chemstry Supervsor: Dr. Dezső Boda Unversty of Pannona Insttute
Grand canoncal Monte Carlo smulatons of bulk electrolytes and calcum channels Thess of Ph.D. dssertaton Prepared by: Attla Malascs M.Sc. n Chemstry Supervsor: Dr. Dezső Boda Unversty of Pannona Insttute
Measurement of Radiation: Exposure. Purpose. Quantitative description of radiation
 Measurement of Radaton: Exposure George Starkschall, Ph.D. Department of Radaton Physcs U.T. M.D. Anderson Cancer Center Purpose To ntroduce the concept of radaton exposure and to descrbe and evaluate
Measurement of Radaton: Exposure George Starkschall, Ph.D. Department of Radaton Physcs U.T. M.D. Anderson Cancer Center Purpose To ntroduce the concept of radaton exposure and to descrbe and evaluate
Frequency dependence of the permittivity
 Frequency dependence of the permttvty February 7, 016 In materals, the delectrc constant and permeablty are actually frequency dependent. Ths does not affect our results for sngle frequency modes, but
Frequency dependence of the permttvty February 7, 016 In materals, the delectrc constant and permeablty are actually frequency dependent. Ths does not affect our results for sngle frequency modes, but
Understanding Electrophoretic Displays: Transient Current Characteristics of Dispersed Charges in a Non-Polar Medium
 Understandng Electrophoretc Dsplays: Transent Current Characterstcs of Dspersed Charges n a Non-Polar Medum Yoocharn Jeon, Pavel Kornlovtch, Patrca Beck, Zhang-Ln Zhou, Rchard Henze, Tm Koch HP Laboratores
Understandng Electrophoretc Dsplays: Transent Current Characterstcs of Dspersed Charges n a Non-Polar Medum Yoocharn Jeon, Pavel Kornlovtch, Patrca Beck, Zhang-Ln Zhou, Rchard Henze, Tm Koch HP Laboratores
Physics 114 Exam 2 Spring Name:
 Physcs 114 Exam Sprng 013 Name: For gradng purposes (do not wrte here): Queston 1. 1... 3. 3. Problem Answer each of the followng questons. Ponts for each queston are ndcated n red wth the amount beng
Physcs 114 Exam Sprng 013 Name: For gradng purposes (do not wrte here): Queston 1. 1... 3. 3. Problem Answer each of the followng questons. Ponts for each queston are ndcated n red wth the amount beng
CHAPTER 5 NUMERICAL EVALUATION OF DYNAMIC RESPONSE
 CHAPTER 5 NUMERICAL EVALUATION OF DYNAMIC RESPONSE Analytcal soluton s usually not possble when exctaton vares arbtrarly wth tme or f the system s nonlnear. Such problems can be solved by numercal tmesteppng
CHAPTER 5 NUMERICAL EVALUATION OF DYNAMIC RESPONSE Analytcal soluton s usually not possble when exctaton vares arbtrarly wth tme or f the system s nonlnear. Such problems can be solved by numercal tmesteppng
Snce h( q^; q) = hq ~ and h( p^ ; p) = hp, one can wrte ~ h hq hp = hq ~hp ~ (7) the uncertanty relaton for an arbtrary state. The states that mnmze t
 8.5: Many-body phenomena n condensed matter and atomc physcs Last moded: September, 003 Lecture. Squeezed States In ths lecture we shall contnue the dscusson of coherent states, focusng on ther propertes
8.5: Many-body phenomena n condensed matter and atomc physcs Last moded: September, 003 Lecture. Squeezed States In ths lecture we shall contnue the dscusson of coherent states, focusng on ther propertes
NUMERICAL DIFFERENTIATION
 NUMERICAL DIFFERENTIATION 1 Introducton Dfferentaton s a method to compute the rate at whch a dependent output y changes wth respect to the change n the ndependent nput x. Ths rate of change s called the
NUMERICAL DIFFERENTIATION 1 Introducton Dfferentaton s a method to compute the rate at whch a dependent output y changes wth respect to the change n the ndependent nput x. Ths rate of change s called the
2 Finite difference basics
 Numersche Methoden 1, WS 11/12 B.J.P. Kaus 2 Fnte dfference bascs Consder the one- The bascs of the fnte dfference method are best understood wth an example. dmensonal transent heat conducton equaton T
Numersche Methoden 1, WS 11/12 B.J.P. Kaus 2 Fnte dfference bascs Consder the one- The bascs of the fnte dfference method are best understood wth an example. dmensonal transent heat conducton equaton T
Physics 114 Exam 2 Fall 2014 Solutions. Name:
 Physcs 114 Exam Fall 014 Name: For gradng purposes (do not wrte here): Queston 1. 1... 3. 3. Problem Answer each of the followng questons. Ponts for each queston are ndcated n red. Unless otherwse ndcated,
Physcs 114 Exam Fall 014 Name: For gradng purposes (do not wrte here): Queston 1. 1... 3. 3. Problem Answer each of the followng questons. Ponts for each queston are ndcated n red. Unless otherwse ndcated,
Robert Eisberg Second edition CH 09 Multielectron atoms ground states and x-ray excitations
 Quantum Physcs 量 理 Robert Esberg Second edton CH 09 Multelectron atoms ground states and x-ray exctatons 9-01 By gong through the procedure ndcated n the text, develop the tme-ndependent Schroednger equaton
Quantum Physcs 量 理 Robert Esberg Second edton CH 09 Multelectron atoms ground states and x-ray exctatons 9-01 By gong through the procedure ndcated n the text, develop the tme-ndependent Schroednger equaton
Interconnect Modeling
 Interconnect Modelng Modelng of Interconnects Interconnect R, C and computaton Interconnect models umped RC model Dstrbuted crcut models Hgher-order waveform n dstrbuted RC trees Accuracy and fdelty Prepared
Interconnect Modelng Modelng of Interconnects Interconnect R, C and computaton Interconnect models umped RC model Dstrbuted crcut models Hgher-order waveform n dstrbuted RC trees Accuracy and fdelty Prepared
Copyright 2017 by Taylor Enterprises, Inc., All Rights Reserved. Adjusted Control Limits for U Charts. Dr. Wayne A. Taylor
 Taylor Enterprses, Inc. Adjusted Control Lmts for U Charts Copyrght 207 by Taylor Enterprses, Inc., All Rghts Reserved. Adjusted Control Lmts for U Charts Dr. Wayne A. Taylor Abstract: U charts are used
Taylor Enterprses, Inc. Adjusted Control Lmts for U Charts Copyrght 207 by Taylor Enterprses, Inc., All Rghts Reserved. Adjusted Control Lmts for U Charts Dr. Wayne A. Taylor Abstract: U charts are used
PHY688, Statistical Mechanics
 Department of Physcs & Astronomy 449 ESS Bldg. Stony Brook Unversty January 31, 2017 Nuclear Astrophyscs James.Lattmer@Stonybrook.edu Thermodynamcs Internal Energy Densty and Frst Law: ε = E V = Ts P +
Department of Physcs & Astronomy 449 ESS Bldg. Stony Brook Unversty January 31, 2017 Nuclear Astrophyscs James.Lattmer@Stonybrook.edu Thermodynamcs Internal Energy Densty and Frst Law: ε = E V = Ts P +
( ) 1/ 2. ( P SO2 )( P O2 ) 1/ 2.
 Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
CHAPTER 14 GENERAL PERTURBATION THEORY
 CHAPTER 4 GENERAL PERTURBATION THEORY 4 Introducton A partcle n orbt around a pont mass or a sphercally symmetrc mass dstrbuton s movng n a gravtatonal potental of the form GM / r In ths potental t moves
CHAPTER 4 GENERAL PERTURBATION THEORY 4 Introducton A partcle n orbt around a pont mass or a sphercally symmetrc mass dstrbuton s movng n a gravtatonal potental of the form GM / r In ths potental t moves
Homework 4. 1 Electromagnetic surface waves (55 pts.) Nano Optics, Fall Semester 2015 Photonics Laboratory, ETH Zürich
 Homework 4 Contact: frmmerm@ethz.ch Due date: December 04, 015 Nano Optcs, Fall Semester 015 Photoncs Laboratory, ETH Zürch www.photoncs.ethz.ch The goal of ths problem set s to understand how surface
Homework 4 Contact: frmmerm@ethz.ch Due date: December 04, 015 Nano Optcs, Fall Semester 015 Photoncs Laboratory, ETH Zürch www.photoncs.ethz.ch The goal of ths problem set s to understand how surface
Physics 181. Particle Systems
 Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
Inductance Calculation for Conductors of Arbitrary Shape
 CRYO/02/028 Aprl 5, 2002 Inductance Calculaton for Conductors of Arbtrary Shape L. Bottura Dstrbuton: Internal Summary In ths note we descrbe a method for the numercal calculaton of nductances among conductors
CRYO/02/028 Aprl 5, 2002 Inductance Calculaton for Conductors of Arbtrary Shape L. Bottura Dstrbuton: Internal Summary In ths note we descrbe a method for the numercal calculaton of nductances among conductors
Fundamental Considerations of Fuel Cells for Mobility Applications
 Fundamental Consderatons of Fuel Cells for Moblty Applcatons Davd E. Foster Engne Research Center Unversty of Wsconsn - Madson Future Engnes and Ther Fuels ERC 2011 Symposum June 9, 2011 Motvaton Reducng
Fundamental Consderatons of Fuel Cells for Moblty Applcatons Davd E. Foster Engne Research Center Unversty of Wsconsn - Madson Future Engnes and Ther Fuels ERC 2011 Symposum June 9, 2011 Motvaton Reducng
STATISTICAL MECHANICS
 STATISTICAL MECHANICS Thermal Energy Recall that KE can always be separated nto 2 terms: KE system = 1 2 M 2 total v CM KE nternal Rgd-body rotaton and elastc / sound waves Use smplfyng assumptons KE of
STATISTICAL MECHANICS Thermal Energy Recall that KE can always be separated nto 2 terms: KE system = 1 2 M 2 total v CM KE nternal Rgd-body rotaton and elastc / sound waves Use smplfyng assumptons KE of
Canonical transformations
 Canoncal transformatons November 23, 2014 Recall that we have defned a symplectc transformaton to be any lnear transformaton M A B leavng the symplectc form nvarant, Ω AB M A CM B DΩ CD Coordnate transformatons,
Canoncal transformatons November 23, 2014 Recall that we have defned a symplectc transformaton to be any lnear transformaton M A B leavng the symplectc form nvarant, Ω AB M A CM B DΩ CD Coordnate transformatons,
COMPUTER AIDED ANALYSIS OF THE MOS CAPACITOR IN LOW FREQUENCY CONDITIONS
 IN 843-688 centfc Bulletn of the Electrcal Engneerng aculty Year 4 No.4 (8) OMPUTER IE NYI O THE MO PITOR IN OW REQUENY ONITION G. PREU epartment of Electroncs, Telecommuncatons and Energy Engneerng, Valaha
IN 843-688 centfc Bulletn of the Electrcal Engneerng aculty Year 4 No.4 (8) OMPUTER IE NYI O THE MO PITOR IN OW REQUENY ONITION G. PREU epartment of Electroncs, Telecommuncatons and Energy Engneerng, Valaha
Problem Points Score Total 100
 Physcs 450 Solutons of Sample Exam I Problem Ponts Score 1 8 15 3 17 4 0 5 0 Total 100 All wor must be shown n order to receve full credt. Wor must be legble and comprehensble wth answers clearly ndcated.
Physcs 450 Solutons of Sample Exam I Problem Ponts Score 1 8 15 3 17 4 0 5 0 Total 100 All wor must be shown n order to receve full credt. Wor must be legble and comprehensble wth answers clearly ndcated.
2016 Wiley. Study Session 2: Ethical and Professional Standards Application
 6 Wley Study Sesson : Ethcal and Professonal Standards Applcaton LESSON : CORRECTION ANALYSIS Readng 9: Correlaton and Regresson LOS 9a: Calculate and nterpret a sample covarance and a sample correlaton
6 Wley Study Sesson : Ethcal and Professonal Standards Applcaton LESSON : CORRECTION ANALYSIS Readng 9: Correlaton and Regresson LOS 9a: Calculate and nterpret a sample covarance and a sample correlaton
Chapter 13: Multiple Regression
 Chapter 13: Multple Regresson 13.1 Developng the multple-regresson Model The general model can be descrbed as: It smplfes for two ndependent varables: The sample ft parameter b 0, b 1, and b are used to
Chapter 13: Multple Regresson 13.1 Developng the multple-regresson Model The general model can be descrbed as: It smplfes for two ndependent varables: The sample ft parameter b 0, b 1, and b are used to
Linear Approximation with Regularization and Moving Least Squares
 Lnear Approxmaton wth Regularzaton and Movng Least Squares Igor Grešovn May 007 Revson 4.6 (Revson : March 004). 5 4 3 0.5 3 3.5 4 Contents: Lnear Fttng...4. Weghted Least Squares n Functon Approxmaton...
Lnear Approxmaton wth Regularzaton and Movng Least Squares Igor Grešovn May 007 Revson 4.6 (Revson : March 004). 5 4 3 0.5 3 3.5 4 Contents: Lnear Fttng...4. Weghted Least Squares n Functon Approxmaton...
Mass transfer in multi-component mixtures
 Chapters -0 ex. 7, of 5 of boo See also Krshna & Wesselngh Chem. Eng. Sc. 5(6) 997 86-9 Mass transfer n mult-component mxtures Ron Zevenhoven Åbo Aadem Unversty Thermal and Flow Engneerng Laboratory tel.
Chapters -0 ex. 7, of 5 of boo See also Krshna & Wesselngh Chem. Eng. Sc. 5(6) 997 86-9 Mass transfer n mult-component mxtures Ron Zevenhoven Åbo Aadem Unversty Thermal and Flow Engneerng Laboratory tel.
Module 3: Element Properties Lecture 1: Natural Coordinates
 Module 3: Element Propertes Lecture : Natural Coordnates Natural coordnate system s bascally a local coordnate system whch allows the specfcaton of a pont wthn the element by a set of dmensonless numbers
Module 3: Element Propertes Lecture : Natural Coordnates Natural coordnate system s bascally a local coordnate system whch allows the specfcaton of a pont wthn the element by a set of dmensonless numbers
Introduction to Vapor/Liquid Equilibrium, part 2. Raoult s Law:
 CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
Supplementary Information. Fundamental Growth Principles of Colloidal Metal Nanoparticles
 Electronc Supplementary Materal (ESI) for CrystEngComm. Ths journal s The Royal Socety of Chemstry 2015 Supplementary Informaton Fundamental Growth Prncples of Collodal Metal Nanopartcles Jörg Polte* a
Electronc Supplementary Materal (ESI) for CrystEngComm. Ths journal s The Royal Socety of Chemstry 2015 Supplementary Informaton Fundamental Growth Prncples of Collodal Metal Nanopartcles Jörg Polte* a
Gravitational Acceleration: A case of constant acceleration (approx. 2 hr.) (6/7/11)
 Gravtatonal Acceleraton: A case of constant acceleraton (approx. hr.) (6/7/11) Introducton The gravtatonal force s one of the fundamental forces of nature. Under the nfluence of ths force all objects havng
Gravtatonal Acceleraton: A case of constant acceleraton (approx. hr.) (6/7/11) Introducton The gravtatonal force s one of the fundamental forces of nature. Under the nfluence of ths force all objects havng
4.2 Chemical Driving Force
 4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
Numerical Investigation of Electroosmotic Flow. in Convergent/Divergent Micronozzle
 Appled Mathematcal Scences, Vol. 5, 2011, no. 27, 1317-1323 Numercal Investgaton of Electroosmotc Flow n Convergent/Dvergent Mcronozzle V. Gnanaraj, V. Mohan, B. Vellakannan Thagarajar College of Engneerng
Appled Mathematcal Scences, Vol. 5, 2011, no. 27, 1317-1323 Numercal Investgaton of Electroosmotc Flow n Convergent/Dvergent Mcronozzle V. Gnanaraj, V. Mohan, B. Vellakannan Thagarajar College of Engneerng
Kernel Methods and SVMs Extension
 Kernel Methods and SVMs Extenson The purpose of ths document s to revew materal covered n Machne Learnng 1 Supervsed Learnng regardng support vector machnes (SVMs). Ths document also provdes a general
Kernel Methods and SVMs Extenson The purpose of ths document s to revew materal covered n Machne Learnng 1 Supervsed Learnng regardng support vector machnes (SVMs). Ths document also provdes a general
MAGNETISM MAGNETIC DIPOLES
 MAGNETISM We now turn to magnetsm. Ths has actually been used for longer than electrcty. People were usng compasses to sal around the Medterranean Sea several hundred years BC. However t was not understood
MAGNETISM We now turn to magnetsm. Ths has actually been used for longer than electrcty. People were usng compasses to sal around the Medterranean Sea several hundred years BC. However t was not understood
Econ107 Applied Econometrics Topic 3: Classical Model (Studenmund, Chapter 4)
 I. Classcal Assumptons Econ7 Appled Econometrcs Topc 3: Classcal Model (Studenmund, Chapter 4) We have defned OLS and studed some algebrac propertes of OLS. In ths topc we wll study statstcal propertes
I. Classcal Assumptons Econ7 Appled Econometrcs Topc 3: Classcal Model (Studenmund, Chapter 4) We have defned OLS and studed some algebrac propertes of OLS. In ths topc we wll study statstcal propertes
and Statistical Mechanics Material Properties
 Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Lecture 7: Boltzmann distribution & Thermodynamics of mixing
 Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
1. Mathematical models of the chromatographic process
 1. athematcal models of the chromatographc process - What determnes retenton tme n L? - What causes pea broadenng n L? - Why are the L peas often asymmetrc? - Why s partton chromatography much more popular
1. athematcal models of the chromatographc process - What determnes retenton tme n L? - What causes pea broadenng n L? - Why are the L peas often asymmetrc? - Why s partton chromatography much more popular
This chapter illustrates the idea that all properties of the homogeneous electron gas (HEG) can be calculated from electron density.
 1 Unform Electron Gas Ths chapter llustrates the dea that all propertes of the homogeneous electron gas (HEG) can be calculated from electron densty. Intutve Representaton of Densty Electron densty n s
1 Unform Electron Gas Ths chapter llustrates the dea that all propertes of the homogeneous electron gas (HEG) can be calculated from electron densty. Intutve Representaton of Densty Electron densty n s
Lecture 8 Modal Analysis
 Lecture 8 Modal Analyss 16.0 Release Introducton to ANSYS Mechancal 1 2015 ANSYS, Inc. February 27, 2015 Chapter Overvew In ths chapter free vbraton as well as pre-stressed vbraton analyses n Mechancal
Lecture 8 Modal Analyss 16.0 Release Introducton to ANSYS Mechancal 1 2015 ANSYS, Inc. February 27, 2015 Chapter Overvew In ths chapter free vbraton as well as pre-stressed vbraton analyses n Mechancal
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM
 ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
MEASUREMENT OF MOMENT OF INERTIA
 1. measurement MESUREMENT OF MOMENT OF INERTI The am of ths measurement s to determne the moment of nerta of the rotor of an electrc motor. 1. General relatons Rotatng moton and moment of nerta Let us
1. measurement MESUREMENT OF MOMENT OF INERTI The am of ths measurement s to determne the moment of nerta of the rotor of an electrc motor. 1. General relatons Rotatng moton and moment of nerta Let us
Complex Numbers. x = B B 2 4AC 2A. or x = x = 2 ± 4 4 (1) (5) 2 (1)
 Complex Numbers If you have not yet encountered complex numbers, you wll soon do so n the process of solvng quadratc equatons. The general quadratc equaton Ax + Bx + C 0 has solutons x B + B 4AC A For
Complex Numbers If you have not yet encountered complex numbers, you wll soon do so n the process of solvng quadratc equatons. The general quadratc equaton Ax + Bx + C 0 has solutons x B + B 4AC A For
Chapter 2: Electric Energy and Capacitance
 Chapter : Electrc Energy and Capactance Potental One goal of physcs s to dentfy basc forces n our world, such as the electrc force as studed n the prevous lectures. Expermentally, we dscovered that the
Chapter : Electrc Energy and Capactance Potental One goal of physcs s to dentfy basc forces n our world, such as the electrc force as studed n the prevous lectures. Expermentally, we dscovered that the
PART I: MULTIPLE CHOICE (32 questions, each multiple choice question has a 2-point value, 64 points total).
 CHEMISTRY 123-07 Mdterm #2 answer key November 04, 2010 Statstcs: Average: 68 p (68%); Hghest: 91 p (91%); Lowest: 37 p (37%) Number of students performng at or above average: 58 (53%) Number of students
CHEMISTRY 123-07 Mdterm #2 answer key November 04, 2010 Statstcs: Average: 68 p (68%); Hghest: 91 p (91%); Lowest: 37 p (37%) Number of students performng at or above average: 58 (53%) Number of students
Global Sensitivity. Tuesday 20 th February, 2018
 Global Senstvty Tuesday 2 th February, 28 ) Local Senstvty Most senstvty analyses [] are based on local estmates of senstvty, typcally by expandng the response n a Taylor seres about some specfc values
Global Senstvty Tuesday 2 th February, 28 ) Local Senstvty Most senstvty analyses [] are based on local estmates of senstvty, typcally by expandng the response n a Taylor seres about some specfc values
Numerical Heat and Mass Transfer
 Master degree n Mechancal Engneerng Numercal Heat and Mass Transfer 06-Fnte-Dfference Method (One-dmensonal, steady state heat conducton) Fausto Arpno f.arpno@uncas.t Introducton Why we use models and
Master degree n Mechancal Engneerng Numercal Heat and Mass Transfer 06-Fnte-Dfference Method (One-dmensonal, steady state heat conducton) Fausto Arpno f.arpno@uncas.t Introducton Why we use models and
Temperature. Chapter Heat Engine
 Chapter 3 Temperature In prevous chapters of these notes we ntroduced the Prncple of Maxmum ntropy as a technque for estmatng probablty dstrbutons consstent wth constrants. In Chapter 9 we dscussed the
Chapter 3 Temperature In prevous chapters of these notes we ntroduced the Prncple of Maxmum ntropy as a technque for estmatng probablty dstrbutons consstent wth constrants. In Chapter 9 we dscussed the
Solutions of execrises
 Solutons of execrses 1) XPS spectrum analyss The fgure below shows an XPS spectrum measured on the surface of a clean nsoluble homo-polyether. Usng the formulas and tables n ths document, answer the followng
Solutons of execrses 1) XPS spectrum analyss The fgure below shows an XPS spectrum measured on the surface of a clean nsoluble homo-polyether. Usng the formulas and tables n ths document, answer the followng
Psychology 282 Lecture #24 Outline Regression Diagnostics: Outliers
 Psychology 282 Lecture #24 Outlne Regresson Dagnostcs: Outlers In an earler lecture we studed the statstcal assumptons underlyng the regresson model, ncludng the followng ponts: Formal statement of assumptons.
Psychology 282 Lecture #24 Outlne Regresson Dagnostcs: Outlers In an earler lecture we studed the statstcal assumptons underlyng the regresson model, ncludng the followng ponts: Formal statement of assumptons.
Thermal-Fluids I. Chapter 18 Transient heat conduction. Dr. Primal Fernando Ph: (850)
 hermal-fluds I Chapter 18 ransent heat conducton Dr. Prmal Fernando prmal@eng.fsu.edu Ph: (850) 410-6323 1 ransent heat conducton In general, he temperature of a body vares wth tme as well as poston. In
hermal-fluds I Chapter 18 ransent heat conducton Dr. Prmal Fernando prmal@eng.fsu.edu Ph: (850) 410-6323 1 ransent heat conducton In general, he temperature of a body vares wth tme as well as poston. In
Lecture Notes on Linear Regression
 Lecture Notes on Lnear Regresson Feng L fl@sdueducn Shandong Unversty, Chna Lnear Regresson Problem In regresson problem, we am at predct a contnuous target value gven an nput feature vector We assume
Lecture Notes on Lnear Regresson Feng L fl@sdueducn Shandong Unversty, Chna Lnear Regresson Problem In regresson problem, we am at predct a contnuous target value gven an nput feature vector We assume
Energy, Entropy, and Availability Balances Phase Equilibria. Nonideal Thermodynamic Property Models. Selecting an Appropriate Model
 Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Transfer Functions. Convenient representation of a linear, dynamic model. A transfer function (TF) relates one input and one output: ( ) system
 Transfer Functons Convenent representaton of a lnear, dynamc model. A transfer functon (TF) relates one nput and one output: x t X s y t system Y s The followng termnology s used: x y nput output forcng
Transfer Functons Convenent representaton of a lnear, dynamc model. A transfer functon (TF) relates one nput and one output: x t X s y t system Y s The followng termnology s used: x y nput output forcng
Gasometric Determination of NaHCO 3 in a Mixture
 60 50 40 0 0 5 15 25 35 40 Temperature ( o C) 9/28/16 Gasometrc Determnaton of NaHCO 3 n a Mxture apor Pressure (mm Hg) apor Pressure of Water 1 NaHCO 3 (s) + H + (aq) Na + (aq) + H 2 O (l) + CO 2 (g)
60 50 40 0 0 5 15 25 35 40 Temperature ( o C) 9/28/16 Gasometrc Determnaton of NaHCO 3 n a Mxture apor Pressure (mm Hg) apor Pressure of Water 1 NaHCO 3 (s) + H + (aq) Na + (aq) + H 2 O (l) + CO 2 (g)
II. Equilibrium Thermodynamics. Lecture 8: The Nernst Equation
 II. Equlbrum Thermodynamcs Lecture 8: The Nernst Equaton Notes by MIT Student, re-wrtten by MZB 2014 February 27, 2014 1 Chemcal Actvty The fundamental thermodynamc quantty controllng transport and reactons
II. Equlbrum Thermodynamcs Lecture 8: The Nernst Equaton Notes by MIT Student, re-wrtten by MZB 2014 February 27, 2014 1 Chemcal Actvty The fundamental thermodynamc quantty controllng transport and reactons
Math1110 (Spring 2009) Prelim 3 - Solutions
 Math 1110 (Sprng 2009) Solutons to Prelm 3 (04/21/2009) 1 Queston 1. (16 ponts) Short answer. Math1110 (Sprng 2009) Prelm 3 - Solutons x a 1 (a) (4 ponts) Please evaluate lm, where a and b are postve numbers.
Math 1110 (Sprng 2009) Solutons to Prelm 3 (04/21/2009) 1 Queston 1. (16 ponts) Short answer. Math1110 (Sprng 2009) Prelm 3 - Solutons x a 1 (a) (4 ponts) Please evaluate lm, where a and b are postve numbers.
Comparative Studies of Law of Conservation of Energy. and Law Clusters of Conservation of Generalized Energy
 Comparatve Studes of Law of Conservaton of Energy and Law Clusters of Conservaton of Generalzed Energy No.3 of Comparatve Physcs Seres Papers Fu Yuhua (CNOOC Research Insttute, E-mal:fuyh1945@sna.com)
Comparatve Studes of Law of Conservaton of Energy and Law Clusters of Conservaton of Generalzed Energy No.3 of Comparatve Physcs Seres Papers Fu Yuhua (CNOOC Research Insttute, E-mal:fuyh1945@sna.com)
Relaxation in water /spin ice models
 Relaxaton n water /spn ce models Ivan A. Ryzhkn Insttute of Sold State Physcs of Russan Academy of Scences, Chernogolovka, Moscow Dstrct, 1443 Russa Outlne specfcty of approach quaspartcles Jaccard s theory
Relaxaton n water /spn ce models Ivan A. Ryzhkn Insttute of Sold State Physcs of Russan Academy of Scences, Chernogolovka, Moscow Dstrct, 1443 Russa Outlne specfcty of approach quaspartcles Jaccard s theory
8.022 (E&M) Lecture 4
 Topcs: 8.0 (E&M) Lecture 4 More applcatons of vector calculus to electrostatcs: Laplacan: Posson and Laplace equaton url: concept and applcatons to electrostatcs Introducton to conductors Last tme Electrc
Topcs: 8.0 (E&M) Lecture 4 More applcatons of vector calculus to electrostatcs: Laplacan: Posson and Laplace equaton url: concept and applcatons to electrostatcs Introducton to conductors Last tme Electrc
Chapter 8. Potential Energy and Conservation of Energy
 Chapter 8 Potental Energy and Conservaton of Energy In ths chapter we wll ntroduce the followng concepts: Potental Energy Conservatve and non-conservatve forces Mechancal Energy Conservaton of Mechancal
Chapter 8 Potental Energy and Conservaton of Energy In ths chapter we wll ntroduce the followng concepts: Potental Energy Conservatve and non-conservatve forces Mechancal Energy Conservaton of Mechancal
V T for n & P = constant
 Pchem 365: hermodynamcs -SUMMARY- Uwe Burghaus, Fargo, 5 9 Mnmum requrements for underneath of your pllow. However, wrte your own summary! You need to know the story behnd the equatons : Pressure : olume
Pchem 365: hermodynamcs -SUMMARY- Uwe Burghaus, Fargo, 5 9 Mnmum requrements for underneath of your pllow. However, wrte your own summary! You need to know the story behnd the equatons : Pressure : olume
Important Instructions to the Examiners:
 Summer 0 Examnaton Subject & Code: asc Maths (70) Model Answer Page No: / Important Instructons to the Examners: ) The Answers should be examned by key words and not as word-to-word as gven n the model
Summer 0 Examnaton Subject & Code: asc Maths (70) Model Answer Page No: / Important Instructons to the Examners: ) The Answers should be examned by key words and not as word-to-word as gven n the model
Correlation and Regression. Correlation 9.1. Correlation. Chapter 9
 Chapter 9 Correlaton and Regresson 9. Correlaton Correlaton A correlaton s a relatonshp between two varables. The data can be represented b the ordered pars (, ) where s the ndependent (or eplanator) varable,
Chapter 9 Correlaton and Regresson 9. Correlaton Correlaton A correlaton s a relatonshp between two varables. The data can be represented b the ordered pars (, ) where s the ndependent (or eplanator) varable,
Joint Statistical Meetings - Biopharmaceutical Section
 Iteratve Ch-Square Test for Equvalence of Multple Treatment Groups Te-Hua Ng*, U.S. Food and Drug Admnstraton 1401 Rockvlle Pke, #200S, HFM-217, Rockvlle, MD 20852-1448 Key Words: Equvalence Testng; Actve
Iteratve Ch-Square Test for Equvalence of Multple Treatment Groups Te-Hua Ng*, U.S. Food and Drug Admnstraton 1401 Rockvlle Pke, #200S, HFM-217, Rockvlle, MD 20852-1448 Key Words: Equvalence Testng; Actve
π e ax2 dx = x 2 e ax2 dx or x 3 e ax2 dx = 1 x 4 e ax2 dx = 3 π 8a 5/2 (a) We are considering the Maxwell velocity distribution function: 2πτ/m
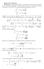 Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
Kinematics of Fluids. Lecture 16. (Refer the text book CONTINUUM MECHANICS by GEORGE E. MASE, Schaum s Outlines) 17/02/2017
 17/0/017 Lecture 16 (Refer the text boo CONTINUUM MECHANICS by GEORGE E. MASE, Schaum s Outlnes) Knematcs of Fluds Last class, we started dscussng about the nematcs of fluds. Recall the Lagrangan and Euleran
17/0/017 Lecture 16 (Refer the text boo CONTINUUM MECHANICS by GEORGE E. MASE, Schaum s Outlnes) Knematcs of Fluds Last class, we started dscussng about the nematcs of fluds. Recall the Lagrangan and Euleran
Osmotic pressure and protein binding
 Osmotc pressure and proten bndng Igor R. Kuznetsov, KochLab Symposum talk 5/15/09 Today we take a closer look at one of the soluton thermodynamcs key ponts from Steve s presentaton. Here t s: d[ln(k off
Osmotc pressure and proten bndng Igor R. Kuznetsov, KochLab Symposum talk 5/15/09 Today we take a closer look at one of the soluton thermodynamcs key ponts from Steve s presentaton. Here t s: d[ln(k off
STATISTICAL MECHANICAL ENSEMBLES 1 MICROSCOPIC AND MACROSCOPIC VARIABLES PHASE SPACE ENSEMBLES. CHE 524 A. Panagiotopoulos 1
 CHE 54 A. Panagotopoulos STATSTCAL MECHACAL ESEMBLES MCROSCOPC AD MACROSCOPC ARABLES The central queston n Statstcal Mechancs can be phrased as follows: f partcles (atoms, molecules, electrons, nucle,
CHE 54 A. Panagotopoulos STATSTCAL MECHACAL ESEMBLES MCROSCOPC AD MACROSCOPC ARABLES The central queston n Statstcal Mechancs can be phrased as follows: f partcles (atoms, molecules, electrons, nucle,
PHYS 705: Classical Mechanics. Newtonian Mechanics
 1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
STAT 511 FINAL EXAM NAME Spring 2001
 STAT 5 FINAL EXAM NAME Sprng Instructons: Ths s a closed book exam. No notes or books are allowed. ou may use a calculator but you are not allowed to store notes or formulas n the calculator. Please wrte
STAT 5 FINAL EXAM NAME Sprng Instructons: Ths s a closed book exam. No notes or books are allowed. ou may use a calculator but you are not allowed to store notes or formulas n the calculator. Please wrte
3. Be able to derive the chemical equilibrium constants from statistical mechanics.
 Lecture #17 1 Lecture 17 Objectves: 1. Notaton of chemcal reactons 2. General equlbrum 3. Be able to derve the chemcal equlbrum constants from statstcal mechancs. 4. Identfy how nondeal behavor can be
Lecture #17 1 Lecture 17 Objectves: 1. Notaton of chemcal reactons 2. General equlbrum 3. Be able to derve the chemcal equlbrum constants from statstcal mechancs. 4. Identfy how nondeal behavor can be
