CHEMICAL ENGINEERING
|
|
|
- Oscar Lamb
- 6 years ago
- Views:
Transcription
1 Postal Correspondence GATE & PSUs -MT To Buy Postal Correspondence Packages call at
2 TABLE OF CONTENT S. No. Ttle Page no. 1. Introducton 3 2. Dffuson Dryng and Humdfcaton Absorpton and Strppng Dstllaton Extracton and Leachng Adsorpton 64 Annexure - A Important Formulae used n Mass Transfer 66 Annexure - B Practce Set 72 Annexure - C Gate Based Questons 86 2
3 Chapter-1 Introducton The process of mass transfer occurs due to concentraton dfference of mxture components. It occurs from a regon of hgher concentraton to a regon of lower concentraton. Concentraton dfference s the pllar of mass transfer. Varous separaton technques such as dstllaton, gas adsorpton, lqud extracton, dryng, crystallzaton, etc. are studed n Mass transfer, where operaton may occur sothermally or non-sothermally. Mass transfer operaton may occur smultaneously wth heat transfer, for example; Dryng, Humdfcaton, Dstllaton, crystallzaton etc. Mass transfer operaton may occur n one drecton example, gas absorpton. The contactng can be done n sx ways namely, Gas-Gas, GasLqud, Gas-sold, Lqud-Lqud and Lqud-Sold and Sold-Sold. (a) Lqud-Vapour Dstllaton. (b) Lqud-gas Gas absorpton, strppng, Humdfcaton and Dehumdfcaton. (c) Lqud-Sold Crystallzaton, Leachng, Adsorpton. (d) Lqud- Lqud Extracton. (e) Sold-Gas Adsorpton, Dryng. The equllbrum between phases s attaned after suffcently long tme. Snce Mass transfer occurs due to both molecular dffuson and turbulence, the detaled study of factors affectng mass transfer s studed. At the phase nterface there s no resstance due to thermodynamc equllbrum (T, P, ) at nterface. Rate of mass transfer s measured by devaton from equllbrum.e. hgher the devaton, hgher s the drvng force Some Important Defntons: 1) Dstllaton: Dstllaton s a vapour-lqud operaton n whch the mxture components are separated by use of thermal energy. When lqud mxture s heated, dfferent components exert dfferent vapour pressure, expressed n terms of relatve volatlty. Ths pressure dfference results n separaton of components n such a way that the top product contans hgher amount of lght component and bottom products contans hgher amounts of heaver component, as shown n fgure 1.1. A dstllaton example s separaton of crude petroleum nto gasolne, kerosene, etc. Fg Schematc dagram of a dstllaton column 3
4 2) Absorpton and Strppng: Gas absorpton s a gas-lqud operaton n whch one or more consttuents of a gas mxture are separated by usng a sutable lqud solvent.e. component moves from gas phase to lqud phase as shown n fgure 1.2. Example of gas absorpton methods s ammona washng from ammona-ar mxture by means of water. Strppng s opposte of absorpton.e. a component moves from lqud phase to gas phase. 3) Fg Schematc dagram of a Lqud-Lqud Extractor Crystallzaton: It s lqud-sold operaton n whch we obtan unform crystals of good purty. The saturated lqud s subjected to changes n temperature and pressure n such a way that crystals get separated from the feed lquor as shown n the fgure ) Fg Schematc dagram of an absorpton column Lqud-Lqud Extracton: It s a lqud-lqud operaton, also called as solvent extracton, n whch components of a lqud mxture are separated by treatng t wth sutable solvent whch dssolves one or more consttuents of mxture more preferably. It s an effcent separaton process n cases where separaton s ether not possble or not economcal by usng dstllaton eg. Separaton of components of an azeotropc mxture. 4) Fg Schematc dagram of a crystallzer Dryng: Dryng s gas-sold operaton n whch a relatvely small amount of water s removed from sold materal, by contactng t wth a contnuous stream of gas (ar) as shown n fgure 1.5. Fg Schematc dagram of a dryer 1.2. Important concepts to remember: 1) Ideal gas law : PV nrt Where, P Pressure (kpa), V Volume (m3) 4
5 n number of moles (kmol), T Temperature (K) m2 kpa R Unversal gas constant kmol K v or psa ) and partal pressure (p A, pb ) : The vapour pressure of a lqud 2) Vapour Pressure (p A s defned as the absolute pressure at whch the lqud and ts vapour are n equllbrum at gven temperature n a closed vessel. The partal pressure of a gas component that s present n a gaseous mxture, s a pressure that would be exerted by that component f t alone were present n the same volume and at the same temperature. p A x A p Av 3) Dalton s law : Dalton s law mathematcally s gven by: P pa + p B, Where P s the total pressure exerted by gaseous mxture. pa and pb are the partal pressures of component gases A and B respectvely. 4) More Volatle Component: More volatle component s lower bolng pont component or the component wth hgher vapour pressure at a gven temperature. It s also called as the lghter component. 5) Less Volatle Component: In a bnary system, t s the component wth hgher bolng pont or lower vapour pressure at a gven temperature. It s also called as the heaver component. 6) Concentraton: 6.1. Mass Concentraton (Also known as mascon): It s defned as the mass of consttuent dvded by the volume of mxture. It s denoted by or. m where, m mass of consttuent, V volume of mxture. V For a pure chemcal the mass concentraton equals ts densty. For Bnary system havng components A and B, mass densty ( ρ ) of soluton s gven byρ A + ρ B ρ, where ρ A and ρ B are mass concentraton s of A and B respectvely Molar concentraton (Molarty): Molar concentraton s defned as the number of moles of speces A per unt volume of the soluton. 3 Mathematcally, It s gven as c A (kmol/m ) na ρ A V MA where, n A moles of speces A, MA molecular weght of component A For a bnary system of A and B, the total molar concentraton of the soluton s gven by C ca +cb 6.3. Mass Fracton (x A') : 5
6 WA, where WA and WB are gven weghts of A and B respectvely x A' WA + WB 6.4. Mole Fracton (x A ) : The mole fracton (xa) of speces A can be defned as the rato of number of moles of component to the total no. of moles. Mathematcally, for a bnary system, x A na na + nb Note: x A + x B 1 and x' A + x' B 1 In gas phase the concentratons are expressed n terms of partal pressures. In case of an deal gas p A V n A RT or ca na pa V RT Where, p A partal pressure of speces A n the mxture n A number of moles of A V molar volume of mxture T absolute temperature (K) R unversal gas constant xa n terms of p A s gven as: xa ca pa /RT pa C P/RT P Where P s the total pressure exerted by the gas mxture. C ca + cb pa pb P + RT RT RT 7) Velocty of Speces: Dfferent chemcal speces are movng at dfferent velocty n a dffusng mxture. The bulk velocty of the mxture would be some sort of an average velocty. For a mxture of n speces local mass average velocty u s defned as n ρ u u 1 n ρ Where u s velocty of th speces. 1 In case of bnary system, u ρ A u A +ρ B u B ρ n c U Local molar average velocty U of mxture s gven by U 1n c 1 6
7 c U +c U In a bnary system, U A A B B C 8) Flux: Flux s a vector quantty. The amount of speces (mass or molar unts) that crosses a unt area per unt tme s called flux (mass transfer flux or molar flux respectvely) of a speces Mass Flux: The mass flux of speces s defned as the mass of speces that passes through a unt area per unt tme. It s also defned as mass flow rate per unt area. () Mass flux relatve to fxed coordnate s gven by, ρ u For a bnary system, the mass flux of A and B relatve to statonary coordnate are A A u A and B ρ Bu B respectvely () Mass flux relatve to the mass average velocty u s gven by, j (u u ) For a bnary system, the mass flux of A and B relatve to mass average velocty are- ja ρ A (u A u) and jb ρ B(u B u) respectvely 8.2. Molar Flux: Molar flux s defned as moles of speces that passes through a unt area per unt tme. It s also defned as molar flow rate per unt area. () Molar flux relatve to the statonary coordnate s gven by, N c U For a bnary system, the molar fluxes of A and B wth respect to statonary coordnates are- N A ca U A and N B c c BU B respectvely () Molar flux relatve to molar average velocty U (also known as bulk velocty) s gven by, J c (U U) For a bnary system, the molar fluxes of A and B wth respect to an observer movng wth bulk velocty, J A c A (U A U) and J B c B (U B U) respectvely 1.3. Dmensonless numbers used n mass transfer: (1) Sherwood number (Sh) k'l convectve mass transport Sh, k' Mass transfer coeffcent DAB molecular mass transport Where L s the characterstcs length (2) Schmdt Number (Sc) Sc 1 Momentum dffusvty ν D AB DAB Mass dffusvty (3) Prandtl Number (Pr) 7
8 Pr C p Momentum dffusvty α K Thermal dffusvty (4) Lews Number (Le) Le K 1 Momentum dffusvty α Sc Mass dffusvty Pr DAB ρcp DAB (5) Reynolds Number (Re) ρvd Inertal force Re μ Vscous force (6) Stanton number (St) k' Sh St υ ReSc (7) Peclet number (Pe) Pe ReSc 1. Calculate the equllbrum composton of the lqud and vapour phase for a mxture of methyl alcohol and water at a temperature of 400 K and under a pressure of 45 kpa. Assume that both lqud and vapour behave deally Data : Vapour pressure of water at 400 K 15 kpa Vapour pressure of methanol at 400 K 60 kpa Soluton: Let x1 and y1 be mole fracton of methyl alcohol n lqud and vapour respectvely. p1 partal pressure of methyl alcohol p1 p1v x1 60x1 p2 water partal pressure p2 p2v x x1 and we know total pressure P p1 p x1 +15(1 x1) x1 15x x1 x y1 p1 p1x P P 45 Hence at equllbrum: Lqud phase 0.67 mole fracton of methyl alcohol 8
9 Vapour phase 0.89 mole fracton of methyl alcohol. 2. A mxture of benzene and toluene bols at 380 K under a pressure of kpa. Determne the composton of bolng lqud assumng that mxture obeys Raoult s law. At 380 K the vapour pressure of benzene s 160 kpa and the toluene s 70 kpa. Soluton: Let mole fracton of benzene n lqud xa P kpa (gven data) pa 160 kpa p B 70 kpa xa P p vb p va p vb xa Mole fracton of benzene n bolng lqud Hence composton of the bolng lqud 34.8 mole% 9
10 10
If two volatile and miscible liquids are combined to form a solution, Raoult s law is not obeyed. Use the experimental data in Table 9.
 9.9 Real Solutons Exhbt Devatons from Raoult s Law If two volatle and mscble lquds are combned to form a soluton, Raoult s law s not obeyed. Use the expermental data n Table 9.3: Physcal Chemstry 00 Pearson
9.9 Real Solutons Exhbt Devatons from Raoult s Law If two volatle and mscble lquds are combned to form a soluton, Raoult s law s not obeyed. Use the expermental data n Table 9.3: Physcal Chemstry 00 Pearson
modeling of equilibrium and dynamic multi-component adsorption in a two-layered fixed bed for purification of hydrogen from methane reforming products
 modelng of equlbrum and dynamc mult-component adsorpton n a two-layered fxed bed for purfcaton of hydrogen from methane reformng products Mohammad A. Ebrahm, Mahmood R. G. Arsalan, Shohreh Fatem * Laboratory
modelng of equlbrum and dynamc mult-component adsorpton n a two-layered fxed bed for purfcaton of hydrogen from methane reformng products Mohammad A. Ebrahm, Mahmood R. G. Arsalan, Shohreh Fatem * Laboratory
Assignment 4. Adsorption Isotherms
 Insttute of Process Engneerng Assgnment 4. Adsorpton Isotherms Part A: Compettve adsorpton of methane and ethane In large scale adsorpton processes, more than one compound from a mxture of gases get adsorbed,
Insttute of Process Engneerng Assgnment 4. Adsorpton Isotherms Part A: Compettve adsorpton of methane and ethane In large scale adsorpton processes, more than one compound from a mxture of gases get adsorbed,
Thermodynamics General
 Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Energy, Entropy, and Availability Balances Phase Equilibria. Nonideal Thermodynamic Property Models. Selecting an Appropriate Model
 Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Diffusion Mass Transfer
 Dffuson Mass Transfer General onsderatons Mass transfer refers to mass n transt due to a speces concentraton gradent n a mture. Must have a mture of two or more speces for mass transfer to occur. The speces
Dffuson Mass Transfer General onsderatons Mass transfer refers to mass n transt due to a speces concentraton gradent n a mture. Must have a mture of two or more speces for mass transfer to occur. The speces
Mass transfer in multi-component mixtures
 Chapters -0 ex. 7, of 5 of boo See also Krshna & Wesselngh Chem. Eng. Sc. 5(6) 997 86-9 Mass transfer n mult-component mxtures Ron Zevenhoven Åbo Aadem Unversty Thermal and Flow Engneerng Laboratory tel.
Chapters -0 ex. 7, of 5 of boo See also Krshna & Wesselngh Chem. Eng. Sc. 5(6) 997 86-9 Mass transfer n mult-component mxtures Ron Zevenhoven Åbo Aadem Unversty Thermal and Flow Engneerng Laboratory tel.
Lecture 17. Membrane Separations [Ch. 14]
![Lecture 17. Membrane Separations [Ch. 14] Lecture 17. Membrane Separations [Ch. 14]](/thumbs/89/98384931.jpg) Lecture 17. embrane Separatons [Ch. 14] embrane Separaton embrane aterals embrane odules Transport n embranes -Bulk flow - Lqud dffuson n pores - Gas dffuson - onporous membranes embrane Separaton Separaton
Lecture 17. embrane Separatons [Ch. 14] embrane Separaton embrane aterals embrane odules Transport n embranes -Bulk flow - Lqud dffuson n pores - Gas dffuson - onporous membranes embrane Separaton Separaton
Adsorption: A gas or gases from a mixture of gases or a liquid (or liquids) from a mixture of liquids is bound physically to the surface of a solid.
 Searatons n Chemcal Engneerng Searatons (gas from a mxture of gases, lquds from a mxture of lquds, solds from a soluton of solds n lquds, dssolved gases from lquds, solvents from gases artally/comletely
Searatons n Chemcal Engneerng Searatons (gas from a mxture of gases, lquds from a mxture of lquds, solds from a soluton of solds n lquds, dssolved gases from lquds, solvents from gases artally/comletely
Mass Transfer Processes
 Mass Transfer Processes S. Majd Hassanzadeh Department of Earth Scences Faculty of Geoscences Utrecht Unversty Outlne: 1. Measures of Concentraton 2. Volatlzaton and Dssoluton 3. Adsorpton Processes 4.
Mass Transfer Processes S. Majd Hassanzadeh Department of Earth Scences Faculty of Geoscences Utrecht Unversty Outlne: 1. Measures of Concentraton 2. Volatlzaton and Dssoluton 3. Adsorpton Processes 4.
Name: SID: Discussion Session:
 Name: SID: Dscusson Sesson: Chemcal Engneerng Thermodynamcs 141 -- Fall 007 Thursday, November 15, 007 Mdterm II SOLUTIONS - 70 mnutes 110 Ponts Total Closed Book and Notes (0 ponts) 1. Evaluate whether
Name: SID: Dscusson Sesson: Chemcal Engneerng Thermodynamcs 141 -- Fall 007 Thursday, November 15, 007 Mdterm II SOLUTIONS - 70 mnutes 110 Ponts Total Closed Book and Notes (0 ponts) 1. Evaluate whether
Chapter 3 Thermochemistry of Fuel Air Mixtures
 Chapter 3 Thermochemstry of Fuel Ar Mxtures 3-1 Thermochemstry 3- Ideal Gas Model 3-3 Composton of Ar and Fuels 3-4 Combuston Stochometry t 3-5 The1 st Law of Thermodynamcs and Combuston 3-6 Thermal converson
Chapter 3 Thermochemstry of Fuel Ar Mxtures 3-1 Thermochemstry 3- Ideal Gas Model 3-3 Composton of Ar and Fuels 3-4 Combuston Stochometry t 3-5 The1 st Law of Thermodynamcs and Combuston 3-6 Thermal converson
Introduction to Vapor/Liquid Equilibrium, part 2. Raoult s Law:
 CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
CHEMICAL REACTIONS AND DIFFUSION
 CHEMICAL REACTIONS AND DIFFUSION A.K.A. NETWORK THERMODYNAMICS BACKGROUND Classcal thermodynamcs descrbes equlbrum states. Non-equlbrum thermodynamcs descrbes steady states. Network thermodynamcs descrbes
CHEMICAL REACTIONS AND DIFFUSION A.K.A. NETWORK THERMODYNAMICS BACKGROUND Classcal thermodynamcs descrbes equlbrum states. Non-equlbrum thermodynamcs descrbes steady states. Network thermodynamcs descrbes
Supplementary Notes for Chapter 9 Mixture Thermodynamics
 Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Module 1 : The equation of continuity. Lecture 1: Equation of Continuity
 1 Module 1 : The equaton of contnuty Lecture 1: Equaton of Contnuty 2 Advanced Heat and Mass Transfer: Modules 1. THE EQUATION OF CONTINUITY : Lectures 1-6 () () () (v) (v) Overall Mass Balance Momentum
1 Module 1 : The equaton of contnuty Lecture 1: Equaton of Contnuty 2 Advanced Heat and Mass Transfer: Modules 1. THE EQUATION OF CONTINUITY : Lectures 1-6 () () () (v) (v) Overall Mass Balance Momentum
Open Systems: Chemical Potential and Partial Molar Quantities Chemical Potential
 Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
CHE-201. I n t r o d u c t i o n t o Chemical E n g i n e e r i n g. C h a p t e r 6. Multiphase Systems
 I n t r o d u c t o n t o Chemcal E n g n e e r n g CHE-201 I N S T R U C T O R : D r. N a b e e l S a l m b o - G h a n d e r C h a t e r 6 Multhase Systems Introductory Statement: Phase s a state of
I n t r o d u c t o n t o Chemcal E n g n e e r n g CHE-201 I N S T R U C T O R : D r. N a b e e l S a l m b o - G h a n d e r C h a t e r 6 Multhase Systems Introductory Statement: Phase s a state of
Week3, Chapter 4. Position and Displacement. Motion in Two Dimensions. Instantaneous Velocity. Average Velocity
 Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
4.2 Chemical Driving Force
 4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
LNG CARGO TRANSFER CALCULATION METHODS AND ROUNDING-OFFS
 CARGO TRANSFER CALCULATION METHODS AND ROUNDING-OFFS CONTENTS 1. Method for determnng transferred energy durng cargo transfer. Calculatng the transferred energy.1 Calculatng the gross transferred energy.1.1
CARGO TRANSFER CALCULATION METHODS AND ROUNDING-OFFS CONTENTS 1. Method for determnng transferred energy durng cargo transfer. Calculatng the transferred energy.1 Calculatng the gross transferred energy.1.1
Chapter 18, Part 1. Fundamentals of Atmospheric Modeling
 Overhead Sldes for Chapter 18, Part 1 of Fundamentals of Atmospherc Modelng by Mark Z. Jacobson Department of Cvl & Envronmental Engneerng Stanford Unversty Stanford, CA 94305-4020 January 30, 2002 Types
Overhead Sldes for Chapter 18, Part 1 of Fundamentals of Atmospherc Modelng by Mark Z. Jacobson Department of Cvl & Envronmental Engneerng Stanford Unversty Stanford, CA 94305-4020 January 30, 2002 Types
10.34 Numerical Methods Applied to Chemical Engineering Fall Homework #3: Systems of Nonlinear Equations and Optimization
 10.34 Numercal Methods Appled to Chemcal Engneerng Fall 2015 Homework #3: Systems of Nonlnear Equatons and Optmzaton Problem 1 (30 ponts). A (homogeneous) azeotrope s a composton of a multcomponent mxture
10.34 Numercal Methods Appled to Chemcal Engneerng Fall 2015 Homework #3: Systems of Nonlnear Equatons and Optmzaton Problem 1 (30 ponts). A (homogeneous) azeotrope s a composton of a multcomponent mxture
Lecture 12. Transport in Membranes (2)
 Lecture 12. Transport n embranes (2) odule Flow Patterns - Perfect mxng - Countercurrent flow - Cocurrent flow - Crossflow embrane Cascades External ass-transfer Resstances Concentraton Polarzaton and
Lecture 12. Transport n embranes (2) odule Flow Patterns - Perfect mxng - Countercurrent flow - Cocurrent flow - Crossflow embrane Cascades External ass-transfer Resstances Concentraton Polarzaton and
( ) 1/ 2. ( P SO2 )( P O2 ) 1/ 2.
 Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Solution Thermodynamics
 CH2351 Chemcal Engneerng Thermodynamcs II Unt I, II www.msubbu.n Soluton Thermodynamcs www.msubbu.n Dr. M. Subramanan Assocate Professor Department of Chemcal Engneerng Sr Svasubramanya Nadar College of
CH2351 Chemcal Engneerng Thermodynamcs II Unt I, II www.msubbu.n Soluton Thermodynamcs www.msubbu.n Dr. M. Subramanan Assocate Professor Department of Chemcal Engneerng Sr Svasubramanya Nadar College of
Number Average Molar Mass. Mass Average Molar Mass. Z-Average Molar Mass
 17 Molar mass: There are dfferent ways to report a molar mass lke (a) Number average molar mass, (b) mass average molar mass, (c) Vscosty average molar mass, (d) Z- Average molar mass Number Average Molar
17 Molar mass: There are dfferent ways to report a molar mass lke (a) Number average molar mass, (b) mass average molar mass, (c) Vscosty average molar mass, (d) Z- Average molar mass Number Average Molar
Solution Thermodynamics
 Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Prediction of Ultrasonic Velocity in Binary Mixtures of a Nuclear Extractant and Monocarboxylic Acids using Several Theoretical Models
 Predcton of ltrasonc Velocty n Bnary Mxtures of a Nuclear Extractant and Monocarboxylc Acds usng Several Theoretcal Models R. K. Mshra 1, B. Dala 1*, N. Swan 2 and S.K. Dash 3 1 BSH, Gandh Insttute of
Predcton of ltrasonc Velocty n Bnary Mxtures of a Nuclear Extractant and Monocarboxylc Acds usng Several Theoretcal Models R. K. Mshra 1, B. Dala 1*, N. Swan 2 and S.K. Dash 3 1 BSH, Gandh Insttute of
Thermodynamics II. Department of Chemical Engineering. Prof. Kim, Jong Hak
 Thermodynamcs II Department of Chemcal Engneerng Prof. Km, Jong Hak Soluton Thermodynamcs : theory Obectve : lay the theoretcal foundaton for applcatons of thermodynamcs to gas mxture and lqud soluton
Thermodynamcs II Department of Chemcal Engneerng Prof. Km, Jong Hak Soluton Thermodynamcs : theory Obectve : lay the theoretcal foundaton for applcatons of thermodynamcs to gas mxture and lqud soluton
KINETICS OF GAS HYDRATE FORMATION FROM PYROLYSIS GAS IN WATER-IN-OIL EMULSION SYSTEM
 Proceedngs of the 7th Internatonal Conference on Gas Hydrates (ICGH 211), Ednburgh, Scotland, Unted Kngdom, July 17-21, 211. KINETICS OF GAS HYDRATE FORMATION FROM PYROLYSIS GAS IN WATER-IN-OIL EMULSION
Proceedngs of the 7th Internatonal Conference on Gas Hydrates (ICGH 211), Ednburgh, Scotland, Unted Kngdom, July 17-21, 211. KINETICS OF GAS HYDRATE FORMATION FROM PYROLYSIS GAS IN WATER-IN-OIL EMULSION
Influence Of Operating Conditions To The Effectiveness Of Extractive Distillation Columns
 Influence Of Operatng Condtons To The Effectveness Of Extractve Dstllaton Columns N.A. Vyazmna Moscov State Unversty Of Envrnmental Engneerng, Department Of Chemcal Engneerng Ul. Staraya Basmannaya 21/4,
Influence Of Operatng Condtons To The Effectveness Of Extractve Dstllaton Columns N.A. Vyazmna Moscov State Unversty Of Envrnmental Engneerng, Department Of Chemcal Engneerng Ul. Staraya Basmannaya 21/4,
NAME and Section No. it is found that 0.6 mol of O
 NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
General Thermodynamics for Process Simulation. Dr. Jungho Cho, Professor Department of Chemical Engineering Dong Yang University
 General Thermodynamcs for Process Smulaton Dr. Jungho Cho, Professor Department of Chemcal Engneerng Dong Yang Unversty Four Crtera for Equlbra μ = μ v Stuaton α T = T β α β P = P l μ = μ l1 l 2 Thermal
General Thermodynamcs for Process Smulaton Dr. Jungho Cho, Professor Department of Chemcal Engneerng Dong Yang Unversty Four Crtera for Equlbra μ = μ v Stuaton α T = T β α β P = P l μ = μ l1 l 2 Thermal
Chemical Equilibrium. Chapter 6 Spontaneity of Reactive Mixtures (gases) Taking into account there are many types of work that a sysem can perform
 Ths chapter deals wth chemcal reactons (system) wth lttle or no consderaton on the surroundngs. Chemcal Equlbrum Chapter 6 Spontanety of eactve Mxtures (gases) eactants generatng products would proceed
Ths chapter deals wth chemcal reactons (system) wth lttle or no consderaton on the surroundngs. Chemcal Equlbrum Chapter 6 Spontanety of eactve Mxtures (gases) eactants generatng products would proceed
Estimation of the composition of the liquid and vapor streams exiting a flash unit with a supercritical component
 Department of Energ oltecnco d Mlano Va Lambruschn - 05 MILANO Eercses of Fundamentals of Chemcal rocesses rof. Ganpero Gropp Eercse 8 Estmaton of the composton of the lqud and vapor streams etng a unt
Department of Energ oltecnco d Mlano Va Lambruschn - 05 MILANO Eercses of Fundamentals of Chemcal rocesses rof. Ganpero Gropp Eercse 8 Estmaton of the composton of the lqud and vapor streams etng a unt
Lecture. Polymer Thermodynamics 0331 L Chemical Potential
 Prof. Dr. rer. nat. habl. S. Enders Faculty III for Process Scence Insttute of Chemcal Engneerng Department of Thermodynamcs Lecture Polymer Thermodynamcs 033 L 337 3. Chemcal Potental Polymer Thermodynamcs
Prof. Dr. rer. nat. habl. S. Enders Faculty III for Process Scence Insttute of Chemcal Engneerng Department of Thermodynamcs Lecture Polymer Thermodynamcs 033 L 337 3. Chemcal Potental Polymer Thermodynamcs
PETE 310 Lectures # 24 & 25 Chapter 12 Gas Liquid Equilibrium
 ETE 30 Lectures # 24 & 25 Chapter 2 Gas Lqud Equlbrum Thermal Equlbrum Object A hgh T, Object B low T Intal contact tme Intermedate tme. Later tme Mechancal Equlbrum ressure essels Vale Closed Vale Open
ETE 30 Lectures # 24 & 25 Chapter 2 Gas Lqud Equlbrum Thermal Equlbrum Object A hgh T, Object B low T Intal contact tme Intermedate tme. Later tme Mechancal Equlbrum ressure essels Vale Closed Vale Open
I wish to publish my paper on The International Journal of Thermophysics. A Practical Method to Calculate Partial Properties from Equation of State
 I wsh to publsh my paper on The Internatonal Journal of Thermophyscs. Ttle: A Practcal Method to Calculate Partal Propertes from Equaton of State Authors: Ryo Akasaka (correspondng author) 1 and Takehro
I wsh to publsh my paper on The Internatonal Journal of Thermophyscs. Ttle: A Practcal Method to Calculate Partal Propertes from Equaton of State Authors: Ryo Akasaka (correspondng author) 1 and Takehro
Principles of Food and Bioprocess Engineering (FS 231) Solutions to Example Problems on Heat Transfer
 Prncples of Food and Boprocess Engneerng (FS 31) Solutons to Example Problems on Heat Transfer 1. We start wth Fourer s law of heat conducton: Q = k A ( T/ x) Rearrangng, we get: Q/A = k ( T/ x) Here,
Prncples of Food and Boprocess Engneerng (FS 31) Solutons to Example Problems on Heat Transfer 1. We start wth Fourer s law of heat conducton: Q = k A ( T/ x) Rearrangng, we get: Q/A = k ( T/ x) Here,
McCabe-Thiele Diagrams for Binary Distillation
 McCabe-Thele Dagrams for Bnary Dstllaton Tore Haug-Warberg Dept. of Chemcal Engneerng August 31st, 2005 F V 1 V 2 L 1 V n L n 1 V n+1 L n V N L N 1 L N L 0 VN+1 Q < 0 D Q > 0 B FIGURE 1: Smplfed pcture
McCabe-Thele Dagrams for Bnary Dstllaton Tore Haug-Warberg Dept. of Chemcal Engneerng August 31st, 2005 F V 1 V 2 L 1 V n L n 1 V n+1 L n V N L N 1 L N L 0 VN+1 Q < 0 D Q > 0 B FIGURE 1: Smplfed pcture
Exercises of Fundamentals of Chemical Processes
 Department of Energ Poltecnco d Mlano a Lambruschn 4 2056 MILANO Exercses of undamentals of Chemcal Processes Prof. Ganpero Gropp Exercse 7 ) Estmaton of the composton of the streams at the ext of an sothermal
Department of Energ Poltecnco d Mlano a Lambruschn 4 2056 MILANO Exercses of undamentals of Chemcal Processes Prof. Ganpero Gropp Exercse 7 ) Estmaton of the composton of the streams at the ext of an sothermal
VAPOR LIQUID EQUILIBRIUM DATA GENERATION FOR ACETIC ACID AND p-xylene AT ATMOSPHERIC PRESSURE
 Int. J. Chem. Sc.: 14(3), 2016, 1511-1519 ISSN 0972-768X www.sadgurupublcatons.com VAPOR LIQUID EQUILIBRIUM DATA GENERATION FOR ACETIC ACID AND p-xylene AT ATMOSPHERIC PRESSURE PAWAN KIRAN MALI *,a and
Int. J. Chem. Sc.: 14(3), 2016, 1511-1519 ISSN 0972-768X www.sadgurupublcatons.com VAPOR LIQUID EQUILIBRIUM DATA GENERATION FOR ACETIC ACID AND p-xylene AT ATMOSPHERIC PRESSURE PAWAN KIRAN MALI *,a and
Gasometric Determination of NaHCO 3 in a Mixture
 60 50 40 0 0 5 15 25 35 40 Temperature ( o C) 9/28/16 Gasometrc Determnaton of NaHCO 3 n a Mxture apor Pressure (mm Hg) apor Pressure of Water 1 NaHCO 3 (s) + H + (aq) Na + (aq) + H 2 O (l) + CO 2 (g)
60 50 40 0 0 5 15 25 35 40 Temperature ( o C) 9/28/16 Gasometrc Determnaton of NaHCO 3 n a Mxture apor Pressure (mm Hg) apor Pressure of Water 1 NaHCO 3 (s) + H + (aq) Na + (aq) + H 2 O (l) + CO 2 (g)
A Modulated Hydrothermal (MHT) Approach for the Facile. Synthesis of UiO-66-Type MOFs
 Supplementary Informaton A Modulated Hydrothermal (MHT) Approach for the Facle Synthess of UO-66-Type MOFs Zhgang Hu, Yongwu Peng, Zx Kang, Yuhong Qan, and Dan Zhao * Department of Chemcal and Bomolecular
Supplementary Informaton A Modulated Hydrothermal (MHT) Approach for the Facle Synthess of UO-66-Type MOFs Zhgang Hu, Yongwu Peng, Zx Kang, Yuhong Qan, and Dan Zhao * Department of Chemcal and Bomolecular
ChE 512: Topic 1 Reactions at a fluid non-porous solid interface. P.A. Ramachandran
 he 512: Topc 1 Reactons at a flud non-porous sold nterface P.. Ramachandran rama@wustl.edu OUTLIE External Transport: Flm oncept Mass transfer coeffcents Effect of transport on reacton multaneous heat
he 512: Topc 1 Reactons at a flud non-porous sold nterface P.. Ramachandran rama@wustl.edu OUTLIE External Transport: Flm oncept Mass transfer coeffcents Effect of transport on reacton multaneous heat
#64. ΔS for Isothermal Mixing of Ideal Gases
 #64 Carnot Heat Engne ΔS for Isothermal Mxng of Ideal Gases ds = S dt + S T V V S = P V T T V PV = nrt, P T ds = v T = nr V dv V nr V V = nrln V V = - nrln V V ΔS ΔS ΔS for Isothermal Mxng for Ideal Gases
#64 Carnot Heat Engne ΔS for Isothermal Mxng of Ideal Gases ds = S dt + S T V V S = P V T T V PV = nrt, P T ds = v T = nr V dv V nr V V = nrln V V = - nrln V V ΔS ΔS ΔS for Isothermal Mxng for Ideal Gases
Wilbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1. Two-dimensional chemical maps as well as chemical profiles were done at 15 kv using
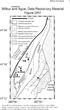 DR2006139 Wlbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1 MINERAL ANALYSES Two-dmensonal chemcal maps as well as chemcal profles were done at 15 kv usng the JEOL JXA-8600 electron mcroprobe at Yale Unversty
DR2006139 Wlbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1 MINERAL ANALYSES Two-dmensonal chemcal maps as well as chemcal profles were done at 15 kv usng the JEOL JXA-8600 electron mcroprobe at Yale Unversty
Vapor-Liquid Equilibria for Water+Hydrochloric Acid+Magnesium Chloride and Water+Hydrochloric Acid+Calcium Chloride Systems at Atmospheric Pressure
 Chnese J. Chem. Eng., 4() 76 80 (006) RESEARCH OES Vapor-Lqud Equlbra for Water+Hydrochlorc Acd+Magnesum Chlorde and Water+Hydrochlorc Acd+Calcum Chlorde Systems at Atmospherc Pressure ZHAG Yng( 张颖 ) and
Chnese J. Chem. Eng., 4() 76 80 (006) RESEARCH OES Vapor-Lqud Equlbra for Water+Hydrochlorc Acd+Magnesum Chlorde and Water+Hydrochlorc Acd+Calcum Chlorde Systems at Atmospherc Pressure ZHAG Yng( 张颖 ) and
CinChE Problem-Solving Strategy Chapter 4 Development of a Mathematical Model. formulation. procedure
 nhe roblem-solvng Strategy hapter 4 Transformaton rocess onceptual Model formulaton procedure Mathematcal Model The mathematcal model s an abstracton that represents the engneerng phenomena occurrng n
nhe roblem-solvng Strategy hapter 4 Transformaton rocess onceptual Model formulaton procedure Mathematcal Model The mathematcal model s an abstracton that represents the engneerng phenomena occurrng n
Process Modeling. Improving or understanding chemical process operation is a major objective for developing a dynamic process model
 Process Modelng Improvng or understandng chemcal process operaton s a major objectve for developng a dynamc process model Balance equatons Steady-state balance equatons mass or energy mass or energy enterng
Process Modelng Improvng or understandng chemcal process operaton s a major objectve for developng a dynamc process model Balance equatons Steady-state balance equatons mass or energy mass or energy enterng
Three-Phase Distillation in Packed Towers: Short-Cut Modelling and Parameter Tuning
 European Symposum on Computer Arded Aded Process Engneerng 15 L. Pugjaner and A. Espuña (Edtors) 2005 Elsever Scence B.V. All rghts reserved. Three-Phase Dstllaton n Packed Towers: Short-Cut Modellng and
European Symposum on Computer Arded Aded Process Engneerng 15 L. Pugjaner and A. Espuña (Edtors) 2005 Elsever Scence B.V. All rghts reserved. Three-Phase Dstllaton n Packed Towers: Short-Cut Modellng and
Design Equations. ν ij r i V R. ν ij r i. Q n components. = Q f c jf Qc j + Continuous Stirred Tank Reactor (steady-state and constant phase)
 Desgn Equatons Batch Reactor d(v R c j ) dt = ν j r V R n dt dt = UA(T a T) r H R V R ncomponents V R c j C pj j Plug Flow Reactor d(qc j ) dv = ν j r 2 dt dv = R U(T a T) n r H R Q n components j c j
Desgn Equatons Batch Reactor d(v R c j ) dt = ν j r V R n dt dt = UA(T a T) r H R V R ncomponents V R c j C pj j Plug Flow Reactor d(qc j ) dv = ν j r 2 dt dv = R U(T a T) n r H R Q n components j c j
Basic concept of reactive flows. Basic concept of reactive flows Combustion Mixing and reaction in high viscous fluid Application of Chaos
 Introducton to Toshhsa Ueda School of Scence for Open and Envronmental Systems Keo Unversty, Japan Combuston Mxng and reacton n hgh vscous flud Applcaton of Chaos Keo Unversty 1 Keo Unversty 2 What s reactve
Introducton to Toshhsa Ueda School of Scence for Open and Envronmental Systems Keo Unversty, Japan Combuston Mxng and reacton n hgh vscous flud Applcaton of Chaos Keo Unversty 1 Keo Unversty 2 What s reactve
Application of Activity Coefficient Models in VLE Determination for Azeotropic System Using Othmer Type Ebulliometer
 Applcaton of Actvty Coeffcent Models n VLE Determnaton for Azeotropc System Usng Othmer Type Ebullometer Manojumar M.S, B. Svapraash 2 Department of Chemcal Engneerng, Annamala Unversty, Annamala Nagar-
Applcaton of Actvty Coeffcent Models n VLE Determnaton for Azeotropc System Usng Othmer Type Ebullometer Manojumar M.S, B. Svapraash 2 Department of Chemcal Engneerng, Annamala Unversty, Annamala Nagar-
The ChemSep Book. Harry A. Kooijman Consultant. Ross Taylor Clarkson University, Potsdam, New York University of Twente, Enschede, The Netherlands
 The ChemSep Book Harry A. Koojman Consultant Ross Taylor Clarkson Unversty, Potsdam, New York Unversty of Twente, Enschede, The Netherlands Lbr Books on Demand www.bod.de Copyrght c 2000 by H.A. Koojman
The ChemSep Book Harry A. Koojman Consultant Ross Taylor Clarkson Unversty, Potsdam, New York Unversty of Twente, Enschede, The Netherlands Lbr Books on Demand www.bod.de Copyrght c 2000 by H.A. Koojman
PART I: MULTIPLE CHOICE (32 questions, each multiple choice question has a 2-point value, 64 points total).
 CHEMISTRY 123-07 Mdterm #2 answer key November 04, 2010 Statstcs: Average: 68 p (68%); Hghest: 91 p (91%); Lowest: 37 p (37%) Number of students performng at or above average: 58 (53%) Number of students
CHEMISTRY 123-07 Mdterm #2 answer key November 04, 2010 Statstcs: Average: 68 p (68%); Hghest: 91 p (91%); Lowest: 37 p (37%) Number of students performng at or above average: 58 (53%) Number of students
Electrical double layer: revisit based on boundary conditions
 Electrcal double layer: revst based on boundary condtons Jong U. Km Department of Electrcal and Computer Engneerng, Texas A&M Unversty College Staton, TX 77843-318, USA Abstract The electrcal double layer
Electrcal double layer: revst based on boundary condtons Jong U. Km Department of Electrcal and Computer Engneerng, Texas A&M Unversty College Staton, TX 77843-318, USA Abstract The electrcal double layer
INTRODUCTION TO CHEMICAL PROCESS SIMULATORS
 INTRODUCTION TO CHEMICAL PROCESS SIMULATORS DWSIM Chemcal Process Smulator A. Carrero, N. Qurante, J. Javaloyes October 2016 Introducton to Chemcal Process Smulators Contents Monday, October 3 rd 2016
INTRODUCTION TO CHEMICAL PROCESS SIMULATORS DWSIM Chemcal Process Smulator A. Carrero, N. Qurante, J. Javaloyes October 2016 Introducton to Chemcal Process Smulators Contents Monday, October 3 rd 2016
...Thermodynamics. If Clausius Clapeyron fails. l T (v 2 v 1 ) = 0/0 Second order phase transition ( S, v = 0)
 If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
MASS TRANSFER Lesson 1 BY DR. ARI SEPPÄLÄ AALTO UNIVERSITY
 SS TRNSFER 2015 Lesson 1 BY DR. RI SEPPÄLÄ LTO UNIVERSITY Structure of the course 6 lessons by r Seälä and Tuula Noonen 5 excercse lessons (one homework roblem n each excercse) by Votto Kotaho (1/6 onts)
SS TRNSFER 2015 Lesson 1 BY DR. RI SEPPÄLÄ LTO UNIVERSITY Structure of the course 6 lessons by r Seälä and Tuula Noonen 5 excercse lessons (one homework roblem n each excercse) by Votto Kotaho (1/6 onts)
The influence of non-ideal vapor-liquid-equilibrium on vaporization of multicomponent hydrocarbon fuels
 ICLASS 202, 2 th Trennal Internatonal Conference on Lqud Atomzaton and Spray Systems, Hedelberg, Germany, September 2-6, 202 The nfluence of non-deal vapor-lqud-equlbrum on vaporzaton of multcomponent
ICLASS 202, 2 th Trennal Internatonal Conference on Lqud Atomzaton and Spray Systems, Hedelberg, Germany, September 2-6, 202 The nfluence of non-deal vapor-lqud-equlbrum on vaporzaton of multcomponent
Chemical Engineering Department University of Washington
 Chemcal Engneerng Department Unversty of Washngton ChemE 60 - Exam I July 4, 003 - Mass Flow Rate of Steam Through a Turbne (5 onts) Steam enters a turbne at 70 o C and.8 Ma and leaves at 00 ka wth a qualty
Chemcal Engneerng Department Unversty of Washngton ChemE 60 - Exam I July 4, 003 - Mass Flow Rate of Steam Through a Turbne (5 onts) Steam enters a turbne at 70 o C and.8 Ma and leaves at 00 ka wth a qualty
y i x P vap 10 A T SOLUTION TO HOMEWORK #7 #Problem
 SOLUTION TO HOMEWORK #7 #roblem 1 10.1-1 a. In order to solve ths problem, we need to know what happens at the bubble pont; at ths pont, the frst bubble s formed, so we can assume that all of the number
SOLUTION TO HOMEWORK #7 #roblem 1 10.1-1 a. In order to solve ths problem, we need to know what happens at the bubble pont; at ths pont, the frst bubble s formed, so we can assume that all of the number
Solutions to Exercises in Astrophysical Gas Dynamics
 1 Solutons to Exercses n Astrophyscal Gas Dynamcs 1. (a). Snce u 1, v are vectors then, under an orthogonal transformaton, u = a j u j v = a k u k Therefore, u v = a j a k u j v k = δ jk u j v k = u j
1 Solutons to Exercses n Astrophyscal Gas Dynamcs 1. (a). Snce u 1, v are vectors then, under an orthogonal transformaton, u = a j u j v = a k u k Therefore, u v = a j a k u j v k = δ jk u j v k = u j
Airflow and Contaminant Simulation with CONTAM
 Arflow and Contamnant Smulaton wth CONTAM George Walton, NIST CHAMPS Developers Workshop Syracuse Unversty June 19, 2006 Network Analogy Electrc Ppe, Duct & Ar Wre Ppe, Duct, or Openng Juncton Juncton
Arflow and Contamnant Smulaton wth CONTAM George Walton, NIST CHAMPS Developers Workshop Syracuse Unversty June 19, 2006 Network Analogy Electrc Ppe, Duct & Ar Wre Ppe, Duct, or Openng Juncton Juncton
Chapter One Mixture of Ideal Gases
 herodynacs II AA Chapter One Mxture of Ideal Gases. Coposton of a Gas Mxture: Mass and Mole Fractons o deterne the propertes of a xture, we need to now the coposton of the xture as well as the propertes
herodynacs II AA Chapter One Mxture of Ideal Gases. Coposton of a Gas Mxture: Mass and Mole Fractons o deterne the propertes of a xture, we need to now the coposton of the xture as well as the propertes
Investigation of High-Pressure Phase Equilibrium with the Observation of the Cloud Point
 Investgaton of Hgh-Pressure Phase Equlbrum wth the Observaton of the Cloud Pont Introducton In the last 2 decades supercrtcal solvents have had a more sgnfcant role as the solvents n chemcal reactons.
Investgaton of Hgh-Pressure Phase Equlbrum wth the Observaton of the Cloud Pont Introducton In the last 2 decades supercrtcal solvents have had a more sgnfcant role as the solvents n chemcal reactons.
Electrochemical Equilibrium Electromotive Force
 CHM465/865, 24-3, Lecture 5-7, 2 th Sep., 24 lectrochemcal qulbrum lectromotve Force Relaton between chemcal and electrc drvng forces lectrochemcal system at constant T and p: consder Gbbs free energy
CHM465/865, 24-3, Lecture 5-7, 2 th Sep., 24 lectrochemcal qulbrum lectromotve Force Relaton between chemcal and electrc drvng forces lectrochemcal system at constant T and p: consder Gbbs free energy
Lecture 11. Transport in Membranes (1)
 ecture 11. Transport n embranes (1) ass Transfer n embranes uk Fow qud Dffuson through ores Gas Dffuson through orous embranes Transport through onporous embranes - Souton-dffuson for qud mxtures - Souton-dffuson
ecture 11. Transport n embranes (1) ass Transfer n embranes uk Fow qud Dffuson through ores Gas Dffuson through orous embranes Transport through onporous embranes - Souton-dffuson for qud mxtures - Souton-dffuson
3) Thermodynamic equation to characterize interfaces
 3) Thermodynamc equaton to characterze nterfaces 3.1) Gbbs Model Realty: rapd contnuous change of chemcal and thermodynamc propertes Replaced by model (constant propertes up to the surface) uv bulk uv
3) Thermodynamc equaton to characterze nterfaces 3.1) Gbbs Model Realty: rapd contnuous change of chemcal and thermodynamc propertes Replaced by model (constant propertes up to the surface) uv bulk uv
Robert Eisberg Second edition CH 09 Multielectron atoms ground states and x-ray excitations
 Quantum Physcs 量 理 Robert Esberg Second edton CH 09 Multelectron atoms ground states and x-ray exctatons 9-01 By gong through the procedure ndcated n the text, develop the tme-ndependent Schroednger equaton
Quantum Physcs 量 理 Robert Esberg Second edton CH 09 Multelectron atoms ground states and x-ray exctatons 9-01 By gong through the procedure ndcated n the text, develop the tme-ndependent Schroednger equaton
Week 9 Chapter 10 Section 1-5
 Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Module 3 LOSSY IMAGE COMPRESSION SYSTEMS. Version 2 ECE IIT, Kharagpur
 Module 3 LOSSY IMAGE COMPRESSION SYSTEMS Verson ECE IIT, Kharagpur Lesson 6 Theory of Quantzaton Verson ECE IIT, Kharagpur Instructonal Objectves At the end of ths lesson, the students should be able to:
Module 3 LOSSY IMAGE COMPRESSION SYSTEMS Verson ECE IIT, Kharagpur Lesson 6 Theory of Quantzaton Verson ECE IIT, Kharagpur Instructonal Objectves At the end of ths lesson, the students should be able to:
Name ID # For relatively dilute aqueous solutions the molality and molarity are approximately equal.
 Name ID # 1 CHEMISTRY 212, Lect. Sect. 002 Dr. G. L. Roberts Exam #1/Sprng 2000 Thursday, February 24, 2000 CLOSED BOOK EXM No notes or books allowed. Calculators may be used. tomc masses of nterest are
Name ID # 1 CHEMISTRY 212, Lect. Sect. 002 Dr. G. L. Roberts Exam #1/Sprng 2000 Thursday, February 24, 2000 CLOSED BOOK EXM No notes or books allowed. Calculators may be used. tomc masses of nterest are
Lecture 5.8 Flux Vector Splitting
 Lecture 5.8 Flux Vector Splttng 1 Flux Vector Splttng The vector E n (5.7.) can be rewrtten as E = AU (5.8.1) (wth A as gven n (5.7.4) or (5.7.6) ) whenever, the equaton of state s of the separable form
Lecture 5.8 Flux Vector Splttng 1 Flux Vector Splttng The vector E n (5.7.) can be rewrtten as E = AU (5.8.1) (wth A as gven n (5.7.4) or (5.7.6) ) whenever, the equaton of state s of the separable form
University of Washington Department of Chemistry Chemistry 452/456 Summer Quarter 2014
 Lecture 12 7/25/14 ERD: 7.1-7.5 Devoe: 8.1.1-8.1.2, 8.2.1-8.2.3, 8.4.1-8.4.3 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 2014 A. Free Energy and Changes n Composton: The
Lecture 12 7/25/14 ERD: 7.1-7.5 Devoe: 8.1.1-8.1.2, 8.2.1-8.2.3, 8.4.1-8.4.3 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 2014 A. Free Energy and Changes n Composton: The
Chapter 11 Structure of Matter 133 Answers to the Conceptual Questions
 hapter 11 Structure of Matter 1 Answers to the onceptual Questons 1. These models gave detaled explanatons for events that had already occurred but lacked any predctve power.. Many examples apply.. Although
hapter 11 Structure of Matter 1 Answers to the onceptual Questons 1. These models gave detaled explanatons for events that had already occurred but lacked any predctve power.. Many examples apply.. Although
Be true to your work, your word, and your friend.
 Chemstry 13 NT Be true to your work, your word, and your frend. Henry Davd Thoreau 1 Chem 13 NT Chemcal Equlbrum Module Usng the Equlbrum Constant Interpretng the Equlbrum Constant Predctng the Drecton
Chemstry 13 NT Be true to your work, your word, and your frend. Henry Davd Thoreau 1 Chem 13 NT Chemcal Equlbrum Module Usng the Equlbrum Constant Interpretng the Equlbrum Constant Predctng the Drecton
Thermodynamics Second Law Entropy
 Thermodynamcs Second Law Entropy Lana Sherdan De Anza College May 8, 2018 Last tme the Boltzmann dstrbuton (dstrbuton of energes) the Maxwell-Boltzmann dstrbuton (dstrbuton of speeds) the Second Law of
Thermodynamcs Second Law Entropy Lana Sherdan De Anza College May 8, 2018 Last tme the Boltzmann dstrbuton (dstrbuton of energes) the Maxwell-Boltzmann dstrbuton (dstrbuton of speeds) the Second Law of
Problem Set #6 solution, Chem 340, Fall 2013 Due Friday, Oct 11, 2013 Please show all work for credit
 Problem Set #6 soluton, Chem 340, Fall 2013 Due Frday, Oct 11, 2013 Please show all work for credt To hand n: Atkns Chap 3 Exercses: 3.3(b), 3.8(b), 3.13(b), 3.15(b) Problems: 3.1, 3.12, 3.36, 3.43 Engel
Problem Set #6 soluton, Chem 340, Fall 2013 Due Frday, Oct 11, 2013 Please show all work for credt To hand n: Atkns Chap 3 Exercses: 3.3(b), 3.8(b), 3.13(b), 3.15(b) Problems: 3.1, 3.12, 3.36, 3.43 Engel
University Physics AI No. 10 The First Law of Thermodynamics
 Unversty hyscs I No he Frst Law o hermodynamcs lass Number Name Ihoose the orrect nswer Whch o the ollowng processes must volate the rst law o thermodynamcs? (here may be more than one answer!) (,B,D )
Unversty hyscs I No he Frst Law o hermodynamcs lass Number Name Ihoose the orrect nswer Whch o the ollowng processes must volate the rst law o thermodynamcs? (here may be more than one answer!) (,B,D )
CHAPTER 3 MODELLING OF DISTILLATION COLUMN
 4 CHAPTER 3 MODEIG O DISTIATIO COUM In ths chapter, the bass of dstllaton, need for dstllaton control and dfferent control technques are descrbed. Model of W and Skogestad column s presented. euro model
4 CHAPTER 3 MODEIG O DISTIATIO COUM In ths chapter, the bass of dstllaton, need for dstllaton control and dfferent control technques are descrbed. Model of W and Skogestad column s presented. euro model
Adiabatic Sorption of Ammonia-Water System and Depicting in p-t-x Diagram
 Adabatc Sorpton of Ammona-Water System and Depctng n p-t-x Dagram J. POSPISIL, Z. SKALA Faculty of Mechancal Engneerng Brno Unversty of Technology Techncka 2, Brno 61669 CZECH REPUBLIC Abstract: - Absorpton
Adabatc Sorpton of Ammona-Water System and Depctng n p-t-x Dagram J. POSPISIL, Z. SKALA Faculty of Mechancal Engneerng Brno Unversty of Technology Techncka 2, Brno 61669 CZECH REPUBLIC Abstract: - Absorpton
Ionization fronts in HII regions
 Ionzaton fronts n HII regons Intal expanson of HII onzaton front s supersonc, creatng a shock front. Statonary frame: front advances nto neutral materal In frame where shock front s statonary, neutral
Ionzaton fronts n HII regons Intal expanson of HII onzaton front s supersonc, creatng a shock front. Statonary frame: front advances nto neutral materal In frame where shock front s statonary, neutral
Solid state reactions. Often defined as reactions between two solids. Here the definition is extended to any reaction involving a solid:
 Sold state reactons Often defned as reactons between two solds Here the defnton s extended to any reacton nvolvng a sold: Sold/sold Sold/gas (Reacton, decomposton) (Sold/lqud) Intercalaton 1 2 Sold-State
Sold state reactons Often defned as reactons between two solds Here the defnton s extended to any reacton nvolvng a sold: Sold/sold Sold/gas (Reacton, decomposton) (Sold/lqud) Intercalaton 1 2 Sold-State
Module 1 : The equation of continuity. Lecture 5: Conservation of Mass for each species. & Fick s Law
 Module : The equato of cotuty Lecture 5: Coservato of Mass for each speces & Fck s Law NPTEL, IIT Kharagpur, Prof. Sakat Chakraborty, Departmet of Chemcal Egeerg 2 Basc Deftos I Mass Trasfer, we usually
Module : The equato of cotuty Lecture 5: Coservato of Mass for each speces & Fck s Law NPTEL, IIT Kharagpur, Prof. Sakat Chakraborty, Departmet of Chemcal Egeerg 2 Basc Deftos I Mass Trasfer, we usually
Spin-rotation coupling of the angularly accelerated rigid body
 Spn-rotaton couplng of the angularly accelerated rgd body Loua Hassan Elzen Basher Khartoum, Sudan. Postal code:11123 E-mal: louaelzen@gmal.com November 1, 2017 All Rghts Reserved. Abstract Ths paper s
Spn-rotaton couplng of the angularly accelerated rgd body Loua Hassan Elzen Basher Khartoum, Sudan. Postal code:11123 E-mal: louaelzen@gmal.com November 1, 2017 All Rghts Reserved. Abstract Ths paper s
Appendix II Summary of Important Equations
 W. M. Whte Geochemstry Equatons of State: Ideal GasLaw: Coeffcent of Thermal Expanson: Compressblty: Van der Waals Equaton: The Laws of Thermdynamcs: Frst Law: Appendx II Summary of Important Equatons
W. M. Whte Geochemstry Equatons of State: Ideal GasLaw: Coeffcent of Thermal Expanson: Compressblty: Van der Waals Equaton: The Laws of Thermdynamcs: Frst Law: Appendx II Summary of Important Equatons
Dynamic Model of Dividing Wall Column for Separation of Ternary System
 R. K. Dohare, K. Sngh, R. Kumar, S. Upadhyaya, S. Gupta Dynamc Model of Dvdng Wall Column for Separaton of Ternary System Raeev Kumar Dohare a, Kalash Sngh a, Raesh Kumar a, Sushant Upadhyaya a, Sourabh
R. K. Dohare, K. Sngh, R. Kumar, S. Upadhyaya, S. Gupta Dynamc Model of Dvdng Wall Column for Separaton of Ternary System Raeev Kumar Dohare a, Kalash Sngh a, Raesh Kumar a, Sushant Upadhyaya a, Sourabh
Substance and heat transfer in multistage systems
 aa Ivanovc-Kneevc Internatonal Journal of Appled Physcs http://www.aras.org/aras/ournals/ap Substance and heat transfer n multstage systems AJA IVANOVIC-KNEZEVIC Faculty of Technology and etallurgy, The
aa Ivanovc-Kneevc Internatonal Journal of Appled Physcs http://www.aras.org/aras/ournals/ap Substance and heat transfer n multstage systems AJA IVANOVIC-KNEZEVIC Faculty of Technology and etallurgy, The
MATH 5630: Discrete Time-Space Model Hung Phan, UMass Lowell March 1, 2018
 MATH 5630: Dscrete Tme-Space Model Hung Phan, UMass Lowell March, 08 Newton s Law of Coolng Consder the coolng of a well strred coffee so that the temperature does not depend on space Newton s law of collng
MATH 5630: Dscrete Tme-Space Model Hung Phan, UMass Lowell March, 08 Newton s Law of Coolng Consder the coolng of a well strred coffee so that the temperature does not depend on space Newton s law of collng
(1) The saturation vapor pressure as a function of temperature, often given by the Antoine equation:
 CE304, Sprng 2004 Lecture 22 Lecture 22: Topcs n Phase Equlbra, part : For the remander of the course, we wll return to the subject of vapor/lqud equlbrum and ntroduce other phase equlbrum calculatons
CE304, Sprng 2004 Lecture 22 Lecture 22: Topcs n Phase Equlbra, part : For the remander of the course, we wll return to the subject of vapor/lqud equlbrum and ntroduce other phase equlbrum calculatons
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM
 ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
Modeling of CO 2 Cut in CBM Production
 Modelng of CO Cut n CBM Producton A study by: - Kamal Morad - Davd Dunn - ous Mattar Fekete Assocates Inc. Mult-Component Gas n Coalbed Methane Coalbed Methane has dfferent characterstcs from conventonal
Modelng of CO Cut n CBM Producton A study by: - Kamal Morad - Davd Dunn - ous Mattar Fekete Assocates Inc. Mult-Component Gas n Coalbed Methane Coalbed Methane has dfferent characterstcs from conventonal
University of Washington Department of Chemistry Chemistry 452/456 Summer Quarter 2014
 Lecture 16 8/4/14 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 214. Real Vapors and Fugacty Henry s Law accounts or the propertes o extremely dlute soluton. s shown n Fgure
Lecture 16 8/4/14 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 214. Real Vapors and Fugacty Henry s Law accounts or the propertes o extremely dlute soluton. s shown n Fgure
V T for n & P = constant
 Pchem 365: hermodynamcs -SUMMARY- Uwe Burghaus, Fargo, 5 9 Mnmum requrements for underneath of your pllow. However, wrte your own summary! You need to know the story behnd the equatons : Pressure : olume
Pchem 365: hermodynamcs -SUMMARY- Uwe Burghaus, Fargo, 5 9 Mnmum requrements for underneath of your pllow. However, wrte your own summary! You need to know the story behnd the equatons : Pressure : olume
Pulse Coded Modulation
 Pulse Coded Modulaton PCM (Pulse Coded Modulaton) s a voce codng technque defned by the ITU-T G.711 standard and t s used n dgtal telephony to encode the voce sgnal. The frst step n the analog to dgtal
Pulse Coded Modulaton PCM (Pulse Coded Modulaton) s a voce codng technque defned by the ITU-T G.711 standard and t s used n dgtal telephony to encode the voce sgnal. The frst step n the analog to dgtal
A New Steps Sequence Table to Improve the Performance of the Jam Petrochemical H 2 -PSA Industrial Plant
 The 14 th Natnal Chemcal Engneerng Congress (IChEC 01) Sharf Unversty of Technology, Tehran, Iran, 16-18 October, 01 A New Steps Sequence Table to Improve the Performance of the Jam Petrochemcal H -PSA
The 14 th Natnal Chemcal Engneerng Congress (IChEC 01) Sharf Unversty of Technology, Tehran, Iran, 16-18 October, 01 A New Steps Sequence Table to Improve the Performance of the Jam Petrochemcal H -PSA
1. Mathematical models of the chromatographic process
 1. athematcal models of the chromatographc process - What determnes retenton tme n L? - What causes pea broadenng n L? - Why are the L peas often asymmetrc? - Why s partton chromatography much more popular
1. athematcal models of the chromatographc process - What determnes retenton tme n L? - What causes pea broadenng n L? - Why are the L peas often asymmetrc? - Why s partton chromatography much more popular
