Name Class Date. Copyright Houghton Mifflin Harcourt Publishing Company. When working with this Lab Report file, remember to save often.
|
|
|
- Britton Little
- 5 years ago
- Views:
Transcription
1 Name Class Date Copyright Houghton Mifflin Harcourt Publishing Company Virtual Lab Identifying Elements When working with this Lab Report file, remember to save often. PROBLEM Chemistry is often used to identify likely sources of material for tools and products. Whenever any such source is found, confirmation of the identity of the elements available provides useful data for evaluating the trade-offs between its economic viability and environmental sustainability. Simple tests such as flame tests provide a rapid way to separate promising sources of an element from those that aren t likely to be feasible. More complex and expensive tests such as those provided through atomic emission spectroscopy are often required as follow-ups, because some flame test results for different elements have similar colors. In this virtual lab simulation, you are challenged to use both approaches to confirm the identity of the metal elements contained in samples of ores found in volcanic deposits. Background The high temperatures found under Earth s surface are capable of melting a wide variety of different minerals into magma. When this molten material reaches the surface as lava, it begins to cool. Because different minerals harden at different temperatures, certain types of eruptions have patterns of cooling that result in some minerals separating from others, concentrating certain metals in some areas. Flame tests and spectroscopy are among the tests used to identify and evaluate such promising mineral deposits. The Bohr model of the atom, which describes electrons at specific energy levels and sublevels outside of the nucleus, provides an explanation for how both flame tests and emission spectroscopy work. When an electron within an atom absorbs energy, the electron moves from one energy level to a higher one. After some time, it will return to a lower energy level, releasing energy in the form of electromagnetic radiation. Because each type of atom has only certain allowed energy levels, only certain amounts of energy will be released, corresponding to certain wavelengths and frequencies of electromagnetic radiation. The emission spectrum is a pattern formed by all of the possible wavelengths and frequencies. The color of the flame test results from the combination of those wavelengths and frequencies that are in the visible light part of the electromagnetic spectrum. When a flame test is performed, the most effective approach is to use a small amount of a solution of a metal compound. For this reason, metal ores and other samples are typically dissolved in an acidic solution. Such an approach usually provides a long-lasting and vivid color change to the flame. While it is possible in some cases to perform flame tests using powdered metal, the color, if visible, lasts only a very short time. Flame-test results for some metals are shown in the table that follows.
2 Flame Colors for Some Metals Found In Volcanic Deposits Arsenic, As blue Molybdenum, Mo yellowish green Calcium, Ca yellowish red Potassium, K lilac to red Copper, Cu blue or green Selenium, Se blue Lead, Pb blue Sodium, Na yellow Lithium, Li red or magenta Zinc, Zn bluish green or whitish green PLANNING Goal Identify an element based on its flame test and emission spectrum. Objectives Relate electron transitions within an atom to emission of specific frequencies of light. Explain the color of a flame test based on the emission of light. Demonstrate how to identify an unknown element by comparing its detailed emission spectrum to a library of standard, known spectra. Evaluate how well emission spectra could work in identifying a mixture of elements. Materials This virtual lab requires: A computer or tablet capable of running the simulation. A sound card with speakers or headphones to play the audio. Electrical Safety Exercise caution when working with electrical equipment such as computers. Do not use electrical equipment with frayed or twisted wires. The area under and around electrical equipment should be dry; cords should not lie in puddles of spilled liquid. Reality Check The processes of performing flame tests and checking atomic emission spectra have been simplified for the purposes of this lab. An advantage of the virtual lab is that the process can be redone over and over without running out of mineral samples. The procedures shown in this lab are not suitable for a real-world experiment. Raw samples of mineral ores are likely to contain enough trace impurities to make positive identification through spectroscopy more complicated than depicted.
3 Your Plan 1. Adjust or replace the following sample procedure to suit your plan for performing the virtual lab. Be sure to provide more detail about exactly how you plan to accomplish the second step. a. Choose sample. b. Prepare sample for the flame test using the tools and reagents available. First, use the hammer to collect some powder from the sample. Place the powder in the beaker, and dissolve it in acid. Dip the wire loop in the solution. c. Insert the wire loop with solution into the flame and observe and record its color. d. Compare your results to other flame-test results to narrow the possibilities for the metal. e. Observe the full emission spectrum of the sample, recording the color and wavelength of some of the key lines. OBSERVATIONS (PERFORM) Select one of these data table options and adjust it to suit your plan for performing this activity. Alternatively, you can delete all of these tables and set up your own approach for recording data. Once a table fits your plan, you can copy and paste it into this Lab Report as many times as you will need it for your multiple trials. Data Table Option A Flame test color: Other observations: Data Table Option B Sample Likely metal 1: Flame test color Other observations Likely metal 2: Likely metal 3:
4 ANALYSIS AND INTERPRETATIONS Emission Spectra Comparisons Sample Match with actual results likely metal 1:? (Y/N) color 1 wavelength 1 color 2 wavelength 2 color 3 wavelength 3 color 4 wavelength 4 color 5 wavelength 5 color 6 wavelength 6 Match with likely metal 2:? (Y/N) Match with likely metal 3:? (Y/N)
5 2. Interpreting Flame Tests Given the results of your flame test, were you able to narrow the possibilities? List which metals are likeliest in your data table(s). 3. Evaluating Methods Explain why you usually can t use just a single spectral line to confirm the identity of an element. 4. Analyzing Spectrometry Results Using the table above, compare some of the spectral lines from your unknown sample to the known samples of metals that were the most likely candidates for your unknown. SUMMARY AND CONCLUSION 5. Results Were you able to identify the unknown? Explain why or why not. 6. Reflections Magnesium metal burns with a bright white flame. What conclusions can you draw about the electron transitions that can take place for a magnesium atom? 7. Reflections Would you be able to use a similar approach to identify a mixture of two or three elements with emission spectrum analysis? Why or why not? 8. Conclusions How did the conclusions you could draw from the flame-test results differ from the conclusions you could draw from the emission spectrometry results?
6 EXTEND If you will be performing additional trials, copy and paste the tables from above into this Lab Report as many times as you will need them. 9. Relating Wavelength to Energy Level Differences During your work with the model set of energy levels, if you were trying to get a line to move from the green part of the spectrum toward the orange or red end of the spectrum, would you drag its energy level to be closer to or farther from the lowest energy level? Explain your answer. 10. Evaluating Limitations of Models The model set of energy levels only included levels for n = 2 through n = 6. If n = 7 had been included, in what part of the electromagnetic spectrum would you expect its transition lines to have been found? Why? Copyright Houghton Mifflin Harcourt Publishing Company
EM SPECTRUM, WAVELENGTH, FREQUENCY, AND ENERGY WORKSHEET
 EM SPECTRUM, WAVELENGTH, FREQUENCY, AND ENERGY WORKSHEET 1.) Look at the EM spectrum below to answer this question. As you move across the visible light spectrum from red to violet (A) Does the wavelength
EM SPECTRUM, WAVELENGTH, FREQUENCY, AND ENERGY WORKSHEET 1.) Look at the EM spectrum below to answer this question. As you move across the visible light spectrum from red to violet (A) Does the wavelength
Lab: Excited Electrons
 Part A: EMISSION SPECTROSCOPY Lab: Excited Electrons According to the Bohr atomic model, electrons orbit the nucleus within specific energy levels. These levels are defined by unique amounts of energy.
Part A: EMISSION SPECTROSCOPY Lab: Excited Electrons According to the Bohr atomic model, electrons orbit the nucleus within specific energy levels. These levels are defined by unique amounts of energy.
EXPERIMENT 3 Flame Tests & Electron Configuration
 EXPERIMENT 3 Flame Tests & Electron Configuration INTRODUCTION Many elements produce colors in the flame when heated. The origin of this phenomenon lies in the arrangement, or configuration of the electrons
EXPERIMENT 3 Flame Tests & Electron Configuration INTRODUCTION Many elements produce colors in the flame when heated. The origin of this phenomenon lies in the arrangement, or configuration of the electrons
Duncan. Electrons, Energy, & the Electromagnetic Spectrum Notes Simplified, 2-D Bohr Model: Figure 1. Figure 2. Figure 3
 Electrons, Energy, & the Electromagnetic Spectrum Notes Simplified, 2-D Bohr Model: Figure 1 Figure 2 Figure 3 Light Calculation Notes Here s how the type/form of EM radiation can be determined The amount
Electrons, Energy, & the Electromagnetic Spectrum Notes Simplified, 2-D Bohr Model: Figure 1 Figure 2 Figure 3 Light Calculation Notes Here s how the type/form of EM radiation can be determined The amount
Title: FLAME TESTS. sodium chloride. calcium nitrate. potassium nitrate. strontium nitrate. copper(ii) nitrate. lithium nitrate. nitrate.
 Title: FLAME TESTS Target In this lab students will learn about atomic energy levels, emissions spectroscopy and flame tests for element identification. Students will identify the unknown elements from
Title: FLAME TESTS Target In this lab students will learn about atomic energy levels, emissions spectroscopy and flame tests for element identification. Students will identify the unknown elements from
Experiment 3 Electromagnetic Radiation and Atom Interaction
 Experiment 3 Electromagnetic Radiation and Atom Interaction B OBJECTIVES To be familiar with the relationship between emission line spectra and the energy levels of electrons in various atoms. B INTRODUCTION
Experiment 3 Electromagnetic Radiation and Atom Interaction B OBJECTIVES To be familiar with the relationship between emission line spectra and the energy levels of electrons in various atoms. B INTRODUCTION
Chapter 7. Quantum Theory and Atomic Structure
 Chapter 7 Quantum Theory and Atomic Structure Outline 1. The Nature of Light 2. Atomic Spectra 3. The Wave-Particle Duality of Matter and Energy 4. The Quantum-Mechanical Model of the Atom 3 September
Chapter 7 Quantum Theory and Atomic Structure Outline 1. The Nature of Light 2. Atomic Spectra 3. The Wave-Particle Duality of Matter and Energy 4. The Quantum-Mechanical Model of the Atom 3 September
MODERN ATOMIC THEORY AND THE PERIODIC TABLE
 C10 04/19/2013 13:34:14 Page 114 CHAPTER 10 MODERN ATOMIC THEORY AND THE PERIODIC TABLE SOLUTIONS TO REVIEW QUESTIONS 1. Wavelength is defined as the distance between consecutive peaks in a wave. It is
C10 04/19/2013 13:34:14 Page 114 CHAPTER 10 MODERN ATOMIC THEORY AND THE PERIODIC TABLE SOLUTIONS TO REVIEW QUESTIONS 1. Wavelength is defined as the distance between consecutive peaks in a wave. It is
Regents Chemistry PRACTICE PACKET. Unit 2: Atomic Theory
 *STUDENT* *STUDENT* Regents Chemistry PRACTICE PACKET Unit 2: Atomic Theory 2 Copyright 2015 Tim Dolgos Name History of Atomic Theory Period Fill in the missing information in the chart below: Name of
*STUDENT* *STUDENT* Regents Chemistry PRACTICE PACKET Unit 2: Atomic Theory 2 Copyright 2015 Tim Dolgos Name History of Atomic Theory Period Fill in the missing information in the chart below: Name of
Atomic Spectra: Energy, Light, and the Electron
 Atomic Spectra: Energy, Light, and the Electron Introduction: An atom consists of a nucleus, containing protons and neutrons, and tiny electrons, which move around the nucleus. Picture a beehive where
Atomic Spectra: Energy, Light, and the Electron Introduction: An atom consists of a nucleus, containing protons and neutrons, and tiny electrons, which move around the nucleus. Picture a beehive where
Forensics Lab Flame Tests
 Forensics Lab Flame Tests Name Per Due Date Introduction The fundamental particles that make up the building blocks of matter are known as atoms, each of which is shown on the periodic table of the elements.
Forensics Lab Flame Tests Name Per Due Date Introduction The fundamental particles that make up the building blocks of matter are known as atoms, each of which is shown on the periodic table of the elements.
Chemistry 212 ATOMIC SPECTROSCOPY
 Chemistry 212 ATOMIC SPECTROSCOPY The emission and absorption of light energy of particular wavelengths by atoms and molecules is a common phenomenon. The emissions/absorptions are characteristic for each
Chemistry 212 ATOMIC SPECTROSCOPY The emission and absorption of light energy of particular wavelengths by atoms and molecules is a common phenomenon. The emissions/absorptions are characteristic for each
Atomic Spectra Introduction
 Atomic Spectra Introduction: Light and all other electromagnetic radiation is energy that is emitted in the form of waves. Thus light behaves like a wave, and the energy of light varies with the wavelength
Atomic Spectra Introduction: Light and all other electromagnetic radiation is energy that is emitted in the form of waves. Thus light behaves like a wave, and the energy of light varies with the wavelength
Western Carolina University
 CHEM 132 Lab 03 Chemistry 132 Lab 03 Flame Test and Electron Configuration Prelaboratory Exercise Go to Chem21Labs.com and complete the on-line prelab by answering the questions below. The prelab will
CHEM 132 Lab 03 Chemistry 132 Lab 03 Flame Test and Electron Configuration Prelaboratory Exercise Go to Chem21Labs.com and complete the on-line prelab by answering the questions below. The prelab will
.Lab25. FlameTests A A. Pre-Lab Discussion. Purpose. Equipment. Materials. Safety,I II-n
 '~.,,;. Name.:," _'._..--".-_-+-_-,~_,---_---:-~_._.'Date Class"_-'--.,"'-' FlameTests.Lab25 Text reference: Chapter 13, PP: 339-343 ~ Pre-Lab Discussion. The normal electron conflgurationof atoms or ions
'~.,,;. Name.:," _'._..--".-_-+-_-,~_,---_---:-~_._.'Date Class"_-'--.,"'-' FlameTests.Lab25 Text reference: Chapter 13, PP: 339-343 ~ Pre-Lab Discussion. The normal electron conflgurationof atoms or ions
Academic Chemistry Chapter 5 Electrons in Atoms Notes
 Academic Chemistry Chapter 5 Electrons in Atoms Notes Name Date ATOMIC MODEL See Atomic Model Timeline worksheet for specifics. ELECTRONS Quantum Mechanical (QM) Model- This is the currently accepted model
Academic Chemistry Chapter 5 Electrons in Atoms Notes Name Date ATOMIC MODEL See Atomic Model Timeline worksheet for specifics. ELECTRONS Quantum Mechanical (QM) Model- This is the currently accepted model
Today is Thursday, March (!) 1 st, 2018
 In This Lesson: Atomic Emissions (Lesson 2 of 4) Stuff You Need: Calculator Today is Thursday, March (!) 1 st, 2018 Pre-Class: [choose one] What is white light? How are fireworks made to be different colors?
In This Lesson: Atomic Emissions (Lesson 2 of 4) Stuff You Need: Calculator Today is Thursday, March (!) 1 st, 2018 Pre-Class: [choose one] What is white light? How are fireworks made to be different colors?
1. The elements on the periodic table are arranged by increasing A) atomic weight. B) atomic number. C) valence electrons. D) mass number.
 1. The elements on the periodic table are arranged by increasing A) atomic weight. B) atomic number. C) valence electrons. D) mass number. 2. Periodic law states which of the following? A) Physical and
1. The elements on the periodic table are arranged by increasing A) atomic weight. B) atomic number. C) valence electrons. D) mass number. 2. Periodic law states which of the following? A) Physical and
Chapter 2 Atoms and Elements. 2.4 The Atom
 Chapter 2 Atoms and Elements 2.4 The Atom Atoms Dalton s Atomic Theory Are tiny particles of matter. Of an element are similar and different from other elements. Of two or more different elements combine
Chapter 2 Atoms and Elements 2.4 The Atom Atoms Dalton s Atomic Theory Are tiny particles of matter. Of an element are similar and different from other elements. Of two or more different elements combine
Name Date Period Unit 3 Review: Electrons and the periodic table
 Name Date Period Unit 3 Review: Electrons and the periodic table G Chem; Coleman SHOW YOUR WORK ON ANY AND ALL CALCULATIONS. SIG FIGS MATTER. UNITS MATTER. General Questions: 1. Use the following terms
Name Date Period Unit 3 Review: Electrons and the periodic table G Chem; Coleman SHOW YOUR WORK ON ANY AND ALL CALCULATIONS. SIG FIGS MATTER. UNITS MATTER. General Questions: 1. Use the following terms
Name Chemistry-PAP Period. Notes: Electrons. Light travels through space as a wave. Waves have three primary characteristics:
 Name Chemistry-PAP Period The Wave Nature of Light Notes: Electrons Light travels through space as a wave. Waves have three primary characteristics: Wavelength (λ): the distance between 2 consecutive crests
Name Chemistry-PAP Period The Wave Nature of Light Notes: Electrons Light travels through space as a wave. Waves have three primary characteristics: Wavelength (λ): the distance between 2 consecutive crests
Emission Spectroscopy
 Objectives Emission Spectroscopy Observe spectral lines from a hydrogen gas discharge tube Determine the initial and final energy levels for the electronic transitions associated with the visible portion
Objectives Emission Spectroscopy Observe spectral lines from a hydrogen gas discharge tube Determine the initial and final energy levels for the electronic transitions associated with the visible portion
Atomic Theory. H. Cannon, C. Clapper and T. Guillot Klein High School
 Atomic Theory Unit 3 Development of the Atomic Theory 1. Where is the mass of the atom concentrated? 2. What is located in the nucleus? 3. What is the negative particle that orbits the nucleus? 4. What
Atomic Theory Unit 3 Development of the Atomic Theory 1. Where is the mass of the atom concentrated? 2. What is located in the nucleus? 3. What is the negative particle that orbits the nucleus? 4. What
What are the energies (J) and wavelengths (in nm) for these colors? Color Energy wavelength. Rev. F11 Page 1 of 5
 Exp. 8 Pre Lab ASSIGNMENT Name: Lab Section Score: / 10 (1) Use the equations [see the discussion on the next page] to calculate the energies of the 6 lowest states for the hydrogen atom, and enter your
Exp. 8 Pre Lab ASSIGNMENT Name: Lab Section Score: / 10 (1) Use the equations [see the discussion on the next page] to calculate the energies of the 6 lowest states for the hydrogen atom, and enter your
Spectroscopy of Atoms and Molecules
 CHEM 121L General Chemistry Laboratory Revision 2.1 Spectroscopy of Atoms and Molecules Learn about the Interaction of Photons with Atoms and Molecules. Learn about the Electronic Structure of Atoms. Learn
CHEM 121L General Chemistry Laboratory Revision 2.1 Spectroscopy of Atoms and Molecules Learn about the Interaction of Photons with Atoms and Molecules. Learn about the Electronic Structure of Atoms. Learn
UNIT 4 - Electron Configuration
 Name: Mods: UNIT 4 - Electron Configuration Date Agenda Homework Wed 10/29 Thurs 10/30 Fri 10/31 Mon 11/3 Tues 11/4 Wed 11/5 Go over test PPT Notes - Wavelength and Frequency (slides 1-9) Go over Worksheet
Name: Mods: UNIT 4 - Electron Configuration Date Agenda Homework Wed 10/29 Thurs 10/30 Fri 10/31 Mon 11/3 Tues 11/4 Wed 11/5 Go over test PPT Notes - Wavelength and Frequency (slides 1-9) Go over Worksheet
Electron Energy and Light
 Why? Electron Energy and Light How does light reveal the behavior of electrons in an atom? From fireworks to stars, the color of light is useful in finding out what s in matter. The emission of light by
Why? Electron Energy and Light How does light reveal the behavior of electrons in an atom? From fireworks to stars, the color of light is useful in finding out what s in matter. The emission of light by
Introduction to light Light is a form of energy called electromagnetic radiation. A chart of the electromagnetic spectrum is shown below.
 Experiment: Spectroscopy Introduction to light Light is a form of energy called electromagnetic radiation. A chart of the electromagnetic spectrum is shown below. Radiowave Microwave Infrared Visible Ultraviolet
Experiment: Spectroscopy Introduction to light Light is a form of energy called electromagnetic radiation. A chart of the electromagnetic spectrum is shown below. Radiowave Microwave Infrared Visible Ultraviolet
Spectroscopy Lab. Name: Period: Mr. Palermo- Regent s Chemistry. Background Info:
 Mr. Palermo- Regent s Chemistry Background Info: Spectroscopy Lab Bohr proposed that an electron is found only in specific circular paths, or orbits, around the nucleus. As a result, each orbit has a fixed
Mr. Palermo- Regent s Chemistry Background Info: Spectroscopy Lab Bohr proposed that an electron is found only in specific circular paths, or orbits, around the nucleus. As a result, each orbit has a fixed
Contents. Atomic Structure. 2(a) The Periodic Table. 2(b) Amount of Substance. 2(c) 2(d) Bonding and Structure. Enthalpy Changes.
 1 C h e m i s t r y Contents 2(a) 2(b) 2(c) 2(d) 2(e) Atomic Structure The Periodic Table Amount of Substance Bonding and Structure Enthalpy Changes 3 14 18 28 39 2 C h e m i s t r y 2(a) Atomic Structure
1 C h e m i s t r y Contents 2(a) 2(b) 2(c) 2(d) 2(e) Atomic Structure The Periodic Table Amount of Substance Bonding and Structure Enthalpy Changes 3 14 18 28 39 2 C h e m i s t r y 2(a) Atomic Structure
Chapter 8 Chemical Reactions
 Chapter 8 Chemical Reactions 8.1 Chemical Reactions Evidence of a Chemical Change Chemical reactions involve rearrangement and exchange of atoms to produce new molecules Remember: matter can neither be
Chapter 8 Chemical Reactions 8.1 Chemical Reactions Evidence of a Chemical Change Chemical reactions involve rearrangement and exchange of atoms to produce new molecules Remember: matter can neither be
Atomic Theory. Unit 3 Development of the Atomic Theory
 Atomic Theory Unit 3 Development of the Atomic Theory 1. Where is the mass of the atom concentrated? 2. What is located in the nucleus? 3. What is the negative particle that orbits the nucleus? 4. What
Atomic Theory Unit 3 Development of the Atomic Theory 1. Where is the mass of the atom concentrated? 2. What is located in the nucleus? 3. What is the negative particle that orbits the nucleus? 4. What
Paradigm Shift: Development of Current Atomic Theory Spectroscopy and Energy Levels in Atoms. OR, Show me the Electrons!
 Lecture 2 362 January 16, 2019 Paradigm Shift: Development of Current Atomic Theory Spectroscopy and Energy Levels in Atoms OR, Show me the Electrons! Color Red Yellow White Green Blue Violet Metal Flame
Lecture 2 362 January 16, 2019 Paradigm Shift: Development of Current Atomic Theory Spectroscopy and Energy Levels in Atoms OR, Show me the Electrons! Color Red Yellow White Green Blue Violet Metal Flame
Classifying Chemical Reactions
 1 Classifying Chemical Reactions Analyzing and Predicting Products Introduction The power of chemical reactions to transform our lives is visible all around us-in our cars, even in our bodies. Chemists
1 Classifying Chemical Reactions Analyzing and Predicting Products Introduction The power of chemical reactions to transform our lives is visible all around us-in our cars, even in our bodies. Chemists
Warm-up For sulfur: 1. How many valence electrons does it have? 2. What ion does this typically form? 3. Write the electron configuration for the ion.
 Warm-up For sulfur: 1. How many valence electrons does it have? 2. What ion does this typically form? 3. Write the electron configuration for the ion. Nucleus Contains 99.9% of the mass of an atom Found
Warm-up For sulfur: 1. How many valence electrons does it have? 2. What ion does this typically form? 3. Write the electron configuration for the ion. Nucleus Contains 99.9% of the mass of an atom Found
Higher -o-o-o- Past Paper questions o-o-o- 3.4 Spectra
 Higher -o-o-o- Past Paper questions 1991-2010 -o-o-o- 3.4 Spectra 1992 Q37 The diagram below shows the energy levels for the hydrogen atom. (a) Between which two energy levels would an electron transition
Higher -o-o-o- Past Paper questions 1991-2010 -o-o-o- 3.4 Spectra 1992 Q37 The diagram below shows the energy levels for the hydrogen atom. (a) Between which two energy levels would an electron transition
Atomic Emission and Molecular Absorption Spectra
 Atomic Emission and Molecular Absorption Spectra v062513_6pm Objective: The student will observe the atomic emission spectra of hydrogen using a spectroscope, determine the identity of an unknown metal
Atomic Emission and Molecular Absorption Spectra v062513_6pm Objective: The student will observe the atomic emission spectra of hydrogen using a spectroscope, determine the identity of an unknown metal
Identification of an Unknown Compound through Mass Correlations
 EXPERIMENT Identification of an Unknown Compound through Mass Correlations PURPOSE To carry out a series of decomposition reactions for five different unknown, and use stoichiometry in order to identify
EXPERIMENT Identification of an Unknown Compound through Mass Correlations PURPOSE To carry out a series of decomposition reactions for five different unknown, and use stoichiometry in order to identify
Emission of Light: Discharge Lamps & Flame Tests 1
 Emission of Light: Discharge Lamps & Flame Tests 1 Objectives At the end of this activity you should be able to: o Describe how discharge lamps emit photons following electrical excitation of gaseous atoms.
Emission of Light: Discharge Lamps & Flame Tests 1 Objectives At the end of this activity you should be able to: o Describe how discharge lamps emit photons following electrical excitation of gaseous atoms.
Pre-Lab Exercises Lab 2: Spectroscopy
 Pre-Lab Exercises Lab 2: Spectroscopy 1. Which color of visible light has the longest wavelength? Name Date Section 2. List the colors of visible light from highest frequency to lowest frequency. 3. Does
Pre-Lab Exercises Lab 2: Spectroscopy 1. Which color of visible light has the longest wavelength? Name Date Section 2. List the colors of visible light from highest frequency to lowest frequency. 3. Does
Chemistry: Hood River Valley High School Unit 3 Note Packet and Goals. Description A1. Marble Lab. Nailon Isotope Lab A2.
 Chemistry: Hood River Valley High School Unit 3 Note Packet and Goals Name: Period: Unit 3 Atomic Structure and Periodic Table Unit Goals- As you work through this unit, you should be able to: 1. describe
Chemistry: Hood River Valley High School Unit 3 Note Packet and Goals Name: Period: Unit 3 Atomic Structure and Periodic Table Unit Goals- As you work through this unit, you should be able to: 1. describe
Lesson Title: High School Spectroscopy Lab Amount of time for this lesson =60 minutes
 Lesson Title: High School Spectroscopy Lab Amount of time for this lesson =60 minutes 1. Standards and Safety and Materials: A. Standards Wyoming State Standards SC8.2.3 Students clearly and accurately
Lesson Title: High School Spectroscopy Lab Amount of time for this lesson =60 minutes 1. Standards and Safety and Materials: A. Standards Wyoming State Standards SC8.2.3 Students clearly and accurately
Chemistry Semester One Exam Review
 Chemistry Semester One Exam Review Name: 1. Compare physical and chemical changes in matter. 2. State the law on conservation of mass. 3. On which type of mixture(s) does the Tyndall Effect scatter light?
Chemistry Semester One Exam Review Name: 1. Compare physical and chemical changes in matter. 2. State the law on conservation of mass. 3. On which type of mixture(s) does the Tyndall Effect scatter light?
Unit 4: Chemical Changes (Higher Content)
 Metals react with oxygen to produce metal oxides. E.g. Copper + Oxygen > Copper Oxide The reactions are oxidation reactions because the metals gain oxygen. Reactivity of Metals Metal Extraction Metals
Metals react with oxygen to produce metal oxides. E.g. Copper + Oxygen > Copper Oxide The reactions are oxidation reactions because the metals gain oxygen. Reactivity of Metals Metal Extraction Metals
Chemistry Final Exam Sample Items
 Chemistry Final Exam Sample Items 1. Which best describes the current atomic theory? a. Atoms consist of electrons circling in definite orbits around a positive nucleus. b. Atoms are composed of electrons
Chemistry Final Exam Sample Items 1. Which best describes the current atomic theory? a. Atoms consist of electrons circling in definite orbits around a positive nucleus. b. Atoms are composed of electrons
Modern Atomic Theory and Electron Configurations
 Chem 101 Modern Atomic Theory and Electron Configurations Lectures 8 and 9 Types of Electromagnetic Radiation Electromagnetic radiation is given off by atoms when they have been excited by any form of
Chem 101 Modern Atomic Theory and Electron Configurations Lectures 8 and 9 Types of Electromagnetic Radiation Electromagnetic radiation is given off by atoms when they have been excited by any form of
Bohr Diagram, Lewis Structures, Valence Electrons Review 1. What is the maximum number of electrons you can fit in each energy level or shell?
 AP Chemistry Ms. Ye Name Date Block Bohr Diagram, Lewis Structures, Valence Electrons Review 1. What is the maximum number of electrons you can fit in each energy level or shell? 1 st shell 2 nd shell
AP Chemistry Ms. Ye Name Date Block Bohr Diagram, Lewis Structures, Valence Electrons Review 1. What is the maximum number of electrons you can fit in each energy level or shell? 1 st shell 2 nd shell
2 SPECTROSCOPIC ANALYSIS
 2 SPECTROSCOPIC ANALYSIS 2.1 Introduction Chemical analysis falls into two basic categories: qualitative what is present quantitative how much is present Spectroscopy is capable of both types of analysis,
2 SPECTROSCOPIC ANALYSIS 2.1 Introduction Chemical analysis falls into two basic categories: qualitative what is present quantitative how much is present Spectroscopy is capable of both types of analysis,
2. Why do the discharge tubes get hot after running for a period of time?
 EXPERIIMENT #2 FLAME TESTS Note: Experiments #2 and #3 can be completed together in a single lab period (90 minutes). A combined data sheet for the two labs can be found after Experiment #3. Discussion:
EXPERIIMENT #2 FLAME TESTS Note: Experiments #2 and #3 can be completed together in a single lab period (90 minutes). A combined data sheet for the two labs can be found after Experiment #3. Discussion:
What are the isotopes of hydrogen? H, 2 H, and 3 H
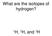 What are the isotopes of hydrogen? 1 H, 2 H, and 3 H The average mass of a boron atom is 10.81. Assuming you were able to isolate only one boron atom, the chance that you would randomly get one with a
What are the isotopes of hydrogen? 1 H, 2 H, and 3 H The average mass of a boron atom is 10.81. Assuming you were able to isolate only one boron atom, the chance that you would randomly get one with a
Experiment 24: Spectroscopy
 Experiment 24: Spectroscopy Figure 24.1: Spectroscopy EQUIPMENT High Voltage Power Supply Incandescent Light Source (3) Gas Discharge Tubes: 1. Helium 2. Hydrogen 3. Unknown Element Spectrometer Felt (1)
Experiment 24: Spectroscopy Figure 24.1: Spectroscopy EQUIPMENT High Voltage Power Supply Incandescent Light Source (3) Gas Discharge Tubes: 1. Helium 2. Hydrogen 3. Unknown Element Spectrometer Felt (1)
ELECTRONS LAB name: 80/1200
 EECTRONS B name: 80/1200 Objective: To use flame tests + bright line spectra to identify elements and compounds. Electrons once were in plum pudding, then flying randomly around the nucleus, until Niels
EECTRONS B name: 80/1200 Objective: To use flame tests + bright line spectra to identify elements and compounds. Electrons once were in plum pudding, then flying randomly around the nucleus, until Niels
Electronic Structure of Atoms
 Electronic Structure of Atoms Electrons inhabit regions of space known as orbitals. Heisenberg Uncertainty Principle NOTE Impossible to define with absolute precision, at the same time, both the position
Electronic Structure of Atoms Electrons inhabit regions of space known as orbitals. Heisenberg Uncertainty Principle NOTE Impossible to define with absolute precision, at the same time, both the position
To observe flame test colors produced by ions in solution.
 Flame Tests PURPOSE To determine the identities of ions in two solutions of unknown composition by comparing the colors they produce in flame tests with colors produced by solutions of known composition.
Flame Tests PURPOSE To determine the identities of ions in two solutions of unknown composition by comparing the colors they produce in flame tests with colors produced by solutions of known composition.
Electronic configuration
 Electronic configuration The energy of electrons The electrons of an atom all have the same charge and the same mass, but each electron has a different amount of energy. Electrons that have the lowest
Electronic configuration The energy of electrons The electrons of an atom all have the same charge and the same mass, but each electron has a different amount of energy. Electrons that have the lowest
Atomic Theory C &03
 Atomic Theory Part One: Flame Tests Part Two: Atomic Spectra Part Three: Applications of Spectra (optional) C12-2-02 &03 This activity will focus on the visible portion of the electromagnetic spectrum.
Atomic Theory Part One: Flame Tests Part Two: Atomic Spectra Part Three: Applications of Spectra (optional) C12-2-02 &03 This activity will focus on the visible portion of the electromagnetic spectrum.
CHAPTER 1: Chemistry, An Introduction
 CHAPTER 1: Chemistry, An Introduction science: the study of nature to explain what one observes 1.4 THE SCIENTIFIC METHOD: How Chemists Think Applying the Scientific Method 1. Make an observation, and
CHAPTER 1: Chemistry, An Introduction science: the study of nature to explain what one observes 1.4 THE SCIENTIFIC METHOD: How Chemists Think Applying the Scientific Method 1. Make an observation, and
Unit 7: The Periodic Table
 Unit 7: The Periodic Table Name Class Website: http://pilarz.weebly.com PS:7 TLW categorize elements of the periodic table according to common properties and explain how elements differ in structural parts
Unit 7: The Periodic Table Name Class Website: http://pilarz.weebly.com PS:7 TLW categorize elements of the periodic table according to common properties and explain how elements differ in structural parts
Atomic Structure and Periodicity
 Atomic Structure and Periodicity Atoms and isotopes: Isotopes-#p + same for all but mass number is different b/c of # n o Average atomic mass is weighted average of all the isotopes for an element Average
Atomic Structure and Periodicity Atoms and isotopes: Isotopes-#p + same for all but mass number is different b/c of # n o Average atomic mass is weighted average of all the isotopes for an element Average
The relationship between these aspects is described by the following equation: E = hν =
 1 Learning Outcomes EXPERIMENT A10: LINE SPECTRUM Upon completion of this lab, the student will be able to: 1) Examine the line spectrum of the hydrogen atom. 2) Calculate the frequency and energy of the
1 Learning Outcomes EXPERIMENT A10: LINE SPECTRUM Upon completion of this lab, the student will be able to: 1) Examine the line spectrum of the hydrogen atom. 2) Calculate the frequency and energy of the
EXPERIMENT 4 THE N-BOTTLE PROBLEM
 EXPERIMENT 4 THE N-BOTTLE PROBLEM INTRODUCTION The purpose of this experiment is to use your knowledge about acid-base and precipitation reactions that occur in aqueous solutions to identify the ionic
EXPERIMENT 4 THE N-BOTTLE PROBLEM INTRODUCTION The purpose of this experiment is to use your knowledge about acid-base and precipitation reactions that occur in aqueous solutions to identify the ionic
Color. 3. Why are the color labels in the table above plural (i.e., Reds rather than Red )?
 NS D3 Electron Energy and Light Name From fireworks to stars, the color of light is useful in finding out what s in matter. The emission of light by hydrogen and other atoms has played a key role in understanding
NS D3 Electron Energy and Light Name From fireworks to stars, the color of light is useful in finding out what s in matter. The emission of light by hydrogen and other atoms has played a key role in understanding
The Structure of the Atom
 CHAPTER 5 The Structure of the Atom 5.4 Light and Spectroscopy 460 370 BC 1808 1870 1897 1910 1925 Today Democritus Atomism Dalton Modern atomic theory Crookes Cathode rays Thomson Discovery of the electron
CHAPTER 5 The Structure of the Atom 5.4 Light and Spectroscopy 460 370 BC 1808 1870 1897 1910 1925 Today Democritus Atomism Dalton Modern atomic theory Crookes Cathode rays Thomson Discovery of the electron
CRHS Academic Chemistry Unit 4 Electrons. Notes. Key Dates
 Name Period CRHS Academic Chemistry Unit 4 Electrons Notes Key Dates Quiz Date Exam Date Lab Dates Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry Website: https://cincochem.pbworks.com
Name Period CRHS Academic Chemistry Unit 4 Electrons Notes Key Dates Quiz Date Exam Date Lab Dates Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry Website: https://cincochem.pbworks.com
Atomic Theory: Spectroscopy and Flame Tests
 Atomic Theory: Spectroscopy and Flame Tests Introduction Light energy is also known as electromagnetic (EM) radiation. The light that we observe with our eyes, visible light, is just a small portion of
Atomic Theory: Spectroscopy and Flame Tests Introduction Light energy is also known as electromagnetic (EM) radiation. The light that we observe with our eyes, visible light, is just a small portion of
1 WHAT IS SPECTROSCOPY?
 1 WHAT IS SPECTROSCOPY? 1.1 The Nature Of Electromagnetic Radiation Anyone who has been sunburnt will know that light packs a punch: in scientific terms, it contains considerable amounts of energy. All
1 WHAT IS SPECTROSCOPY? 1.1 The Nature Of Electromagnetic Radiation Anyone who has been sunburnt will know that light packs a punch: in scientific terms, it contains considerable amounts of energy. All
P O G I L E L E C T R O N E N E R G Y A N D L I G H T
 South Pasadena Honors Chemistry Name 9 Atomic Structure Period Date Why? P O G I L E L E C T R O N E N E R G Y A N D L I G H T How does light reveal the behavior of electrons in an atom? From fireworks
South Pasadena Honors Chemistry Name 9 Atomic Structure Period Date Why? P O G I L E L E C T R O N E N E R G Y A N D L I G H T How does light reveal the behavior of electrons in an atom? From fireworks
EXPERIMENT 7. Determination of Sodium by Flame Atomic-Emission Spectroscopy
 EXPERIMENT 7 Determination of Sodium by Flame Atomic-Emission Spectroscopy USE ONLY DEIONIZED WATER (NOT DISTILLED WATER!) THROUGHOUT THE ENTIRE EXPERIMENT Distilled water actually has too much sodium
EXPERIMENT 7 Determination of Sodium by Flame Atomic-Emission Spectroscopy USE ONLY DEIONIZED WATER (NOT DISTILLED WATER!) THROUGHOUT THE ENTIRE EXPERIMENT Distilled water actually has too much sodium
A) neutron C) Magnesium reacts with an acid. C) gamma radiation D) a proton
 1. Which statement describes a chemical property of the element magnesium? A) Magnesium is malleable. B) Magnesium conducts electricity. C) Magnesium reacts with an acid. D) Magnesium has a high boiling
1. Which statement describes a chemical property of the element magnesium? A) Magnesium is malleable. B) Magnesium conducts electricity. C) Magnesium reacts with an acid. D) Magnesium has a high boiling
Atomic Theory: Spectroscopy and Flame Tests
 Atomic Theory: Spectroscopy and Flame Tests Introduction Light energy is also known as electromagnetic (EM) radiation. The light that we observe with our eyes, visible light, is just a small portion of
Atomic Theory: Spectroscopy and Flame Tests Introduction Light energy is also known as electromagnetic (EM) radiation. The light that we observe with our eyes, visible light, is just a small portion of
Complete the following chart: (assume the overall charge on all atoms = 0...except the last one. #of Protons. #of Neutrons. He 4
 Name: Period: Complete the following chart: (assume the overall charge on all atoms = 0....except the last one Element Symbol Atomic Number #of Protons #of Neutrons Hydrogen 1 Mass Numbra- (amu) #of Electrons
Name: Period: Complete the following chart: (assume the overall charge on all atoms = 0....except the last one Element Symbol Atomic Number #of Protons #of Neutrons Hydrogen 1 Mass Numbra- (amu) #of Electrons
Page 2. Q1.A student investigated food dyes using paper chromatography. This is the method used.
 Q1.A student investigated food dyes using paper chromatography. This is the method used. 1. Put a spot of food colouring X on the start line. 2. Put spots of four separate dyes, A, B, C and D, on the start
Q1.A student investigated food dyes using paper chromatography. This is the method used. 1. Put a spot of food colouring X on the start line. 2. Put spots of four separate dyes, A, B, C and D, on the start
Modern Atomic Theory. Chapter Rutherford s Atom Electromagnetic Radiation. Rutherford showed: Questions left unanswered:
 Copyright 2004 by Houghton Mifflin Company. Modern Atomic Theory Chapter 10 All rights reserved. 1 10.1 Rutherford s Atom Rutherford showed: Atomic nucleus is composed of protons (positive) and neutrons
Copyright 2004 by Houghton Mifflin Company. Modern Atomic Theory Chapter 10 All rights reserved. 1 10.1 Rutherford s Atom Rutherford showed: Atomic nucleus is composed of protons (positive) and neutrons
Lab 1 Write-Up: Flame Tests. Determine the Identity of unknown substances using the Flame Test
 Roy1 Baylee Roy Chemistry Honors Mrs. Lyles 26 September 2014 Lab 1 Write-Up: Flame Tests Purpose Determine the Identity of unknown substances using the Flame Test Procedure In this lab you will take a
Roy1 Baylee Roy Chemistry Honors Mrs. Lyles 26 September 2014 Lab 1 Write-Up: Flame Tests Purpose Determine the Identity of unknown substances using the Flame Test Procedure In this lab you will take a
Name That Salt. The six salts used in this experiment are:
 Name That Salt Learning Objectives: In this experiment there are six unidentified salts labelled Salt 1 through Salt 6. Each team will be given one of these salts and their job is to determine the identity
Name That Salt Learning Objectives: In this experiment there are six unidentified salts labelled Salt 1 through Salt 6. Each team will be given one of these salts and their job is to determine the identity
Sample Copyright. Academic Group ATOMIC STRUCTURE 1. Topics covered in this chapter:
 ATOMIC STRUCTURE Topics covered in this chapter:. Structure of the Atom.2 Atomic Number, Mass Number.3 Isotopes.4 The Mass Spectrometer.5 Atomic Structure and Light Spectra.6 Electron Arrangements in Atoms.7
ATOMIC STRUCTURE Topics covered in this chapter:. Structure of the Atom.2 Atomic Number, Mass Number.3 Isotopes.4 The Mass Spectrometer.5 Atomic Structure and Light Spectra.6 Electron Arrangements in Atoms.7
KWL CHART--ELECTRONS
 KWL CHART--ELECTRONS WHAT DO I ALREADY KNOW ABOUT ELECTRONS? WHAT DO I WANT TO KNOW CONCERNING ELECTRONS? WHAT HAVE I LEARNED TODAY ABOUT ELECTRONS? GPS STANDARD SC3. Students will use the modern atomic
KWL CHART--ELECTRONS WHAT DO I ALREADY KNOW ABOUT ELECTRONS? WHAT DO I WANT TO KNOW CONCERNING ELECTRONS? WHAT HAVE I LEARNED TODAY ABOUT ELECTRONS? GPS STANDARD SC3. Students will use the modern atomic
Atomic Theory: Spectroscopy and Flame Tests
 Atomic Theory: Spectroscopy and Flame Tests Introduction Light energy is also known as electromagnetic (EM) radiation. The light that we observe with our eyes, visible light, is just a small portion of
Atomic Theory: Spectroscopy and Flame Tests Introduction Light energy is also known as electromagnetic (EM) radiation. The light that we observe with our eyes, visible light, is just a small portion of
Collegiate Institute for Math and Science Day 57: December 9, 2016 Room 427
 Unit 2: Atomic Concepts Outline Name: Period: Date: 1. The modern model of the atom has evolved over a long period of time through the work of many scientists. Dalton s Model: Elements are made of atoms
Unit 2: Atomic Concepts Outline Name: Period: Date: 1. The modern model of the atom has evolved over a long period of time through the work of many scientists. Dalton s Model: Elements are made of atoms
Chapter 4 The Structure of the Atom
 Chapter 4 The Structure of the Atom Read pg. 86-97 4.1 Early Theories of Matter The Philosophers Democritus Artistotle - Artistotle s influence so great and the science so primitive (lacking!) his denial
Chapter 4 The Structure of the Atom Read pg. 86-97 4.1 Early Theories of Matter The Philosophers Democritus Artistotle - Artistotle s influence so great and the science so primitive (lacking!) his denial
SPECTROSCOPY: A KEY TO ELEMENTAL IDENTITY
 AME PARTERS SECTIO DATE SPECTROSCOPY: A KEY TO ELEMETAL IDETITY This activity is designed to introduce visible light spectroscopy as a means of identifying elements and providing information on atomic
AME PARTERS SECTIO DATE SPECTROSCOPY: A KEY TO ELEMETAL IDETITY This activity is designed to introduce visible light spectroscopy as a means of identifying elements and providing information on atomic
What is a theory? An organized system of accepted knowledge that applies in a variety of circumstances to explain a specific set of phenomena
 Atomic Structure What is a theory? An organized system of accepted knowledge that applies in a variety of circumstances to explain a specific set of phenomena Early Theories Democritus: 4 B.C.: atom He
Atomic Structure What is a theory? An organized system of accepted knowledge that applies in a variety of circumstances to explain a specific set of phenomena Early Theories Democritus: 4 B.C.: atom He
Partner: Judy 6 October An Activity Series
 Partner: Judy 6 October 2011 An Activity Series Purpose: The purpose of this lab is to verify the activity series of five metals and three halogens. This can be done by reacting the metal with various
Partner: Judy 6 October 2011 An Activity Series Purpose: The purpose of this lab is to verify the activity series of five metals and three halogens. This can be done by reacting the metal with various
for free past papers visit
 NAME SCHOOL ------------------------------- INDEX NO. ----------------------------- SIGNATURE -------------------------- DATE ---------------------------------- 233/2 CHEMISTRYPAPER 2 (Theory) TIME: 2
NAME SCHOOL ------------------------------- INDEX NO. ----------------------------- SIGNATURE -------------------------- DATE ---------------------------------- 233/2 CHEMISTRYPAPER 2 (Theory) TIME: 2
Year 10 Track 3 CHEMISTRY Time: 1:30min
 Year 10 Track 3 CHEMISTRY Time: 1:30min Name: Class: Useful Data: A copy of the Periodic Table is printed below. Relative atomic mass may be taken as: Cu = 63.5 1 Faraday = 96, 500C Q = It Instructions
Year 10 Track 3 CHEMISTRY Time: 1:30min Name: Class: Useful Data: A copy of the Periodic Table is printed below. Relative atomic mass may be taken as: Cu = 63.5 1 Faraday = 96, 500C Q = It Instructions
NAME INDEX NUMBER CANDIDATE SIGNATURE DATE.. 233/1/ CHEMISTRY PAPER 1 (THEORY) JULY/AUGUST HOURS
 NAME INDEX NUMBER CANDIDATE SIGNATURE DATE.. 233/1/ CHEMISTRY PAPER 1 (THEORY) JULY/AUGUST 2018 2 HOURS INSTRUCTIONS TO CANDIDATES. (a) Write your name and index number in the spaces provided above. (b)
NAME INDEX NUMBER CANDIDATE SIGNATURE DATE.. 233/1/ CHEMISTRY PAPER 1 (THEORY) JULY/AUGUST 2018 2 HOURS INSTRUCTIONS TO CANDIDATES. (a) Write your name and index number in the spaces provided above. (b)
Chem 161. Dr. Jasmine Bryant
 Chem 161 Dr. Jasmine Bryant Chapter 1: Matter, Energy, and the Origins of the Universe Problems: 1.1-1.10, 1.14-1.22, 1.24-1.93, 1.96 Science: study of nature that results in a logical explanation of the
Chem 161 Dr. Jasmine Bryant Chapter 1: Matter, Energy, and the Origins of the Universe Problems: 1.1-1.10, 1.14-1.22, 1.24-1.93, 1.96 Science: study of nature that results in a logical explanation of the
Observation of Atomic Spectra
 Observation of Atomic Spectra Introduction In this experiment you will observe and measure the wavelengths of different colors of light emitted by atoms. You will first observe light emitted from excited
Observation of Atomic Spectra Introduction In this experiment you will observe and measure the wavelengths of different colors of light emitted by atoms. You will first observe light emitted from excited
Atomic Spectra for Atoms and Ions. Light is made up of different wavelengths
 Atomic Spectra for Atoms and Ions What will you be doing in lab next week? Recording the line spectra of several different substances in discharge tubes. Recording the line spectra of several ions from
Atomic Spectra for Atoms and Ions What will you be doing in lab next week? Recording the line spectra of several different substances in discharge tubes. Recording the line spectra of several ions from
EXPERIMENTS. Testing products of combustion: Reducing Copper(III) Oxide to Copper. Page 4
 APPARATUS Page 2 APPARATUS Page 3 Reducing Copper(III) Oxide to Copper EXPERIMENTS Page 4 Testing products of combustion: EXPERIMENTS Showing that oxygen and water is needed for rusting iron Page 5 Showing
APPARATUS Page 2 APPARATUS Page 3 Reducing Copper(III) Oxide to Copper EXPERIMENTS Page 4 Testing products of combustion: EXPERIMENTS Showing that oxygen and water is needed for rusting iron Page 5 Showing
Chemistry 40S Atomic Structure (This unit has been adapted from https://bblearn.merlin.mb.ca)
 Chemistry 40S Atomic Structure (This unit has been adapted from https://bblearn.merlin.mb.ca) Name: 1 2 Lesson 1: The Nature of Light Goals: Describe light in terms of electromagnetic energy. Describe
Chemistry 40S Atomic Structure (This unit has been adapted from https://bblearn.merlin.mb.ca) Name: 1 2 Lesson 1: The Nature of Light Goals: Describe light in terms of electromagnetic energy. Describe
Atomic Spectra & Electron Energy Levels
 CHM151LL: ATOMIC SPECTRA & ELECTRON ENERGY LEVELS 1 Atomic Spectra & Electron Energy Levels OBJECTIVES: To measure the wavelength of visible light emitted by excited atoms to calculate the energy of that
CHM151LL: ATOMIC SPECTRA & ELECTRON ENERGY LEVELS 1 Atomic Spectra & Electron Energy Levels OBJECTIVES: To measure the wavelength of visible light emitted by excited atoms to calculate the energy of that
Making Ionic Compounds Lab#
 Making Ionic Compounds Lab# Elements combine to form compounds. If energy is released as the compound is formed, the resulting product is more stable than the reacting elements. In this lab you will react
Making Ionic Compounds Lab# Elements combine to form compounds. If energy is released as the compound is formed, the resulting product is more stable than the reacting elements. In this lab you will react
Physical Changes and Chemical Reactions
 Physical Changes and Chemical Reactions Gezahegn Chaka, Ph.D., and Sudha Madhugiri, Ph.D., Collin College Department of Chemistry Objectives Introduction To observe physical and chemical changes. To identify
Physical Changes and Chemical Reactions Gezahegn Chaka, Ph.D., and Sudha Madhugiri, Ph.D., Collin College Department of Chemistry Objectives Introduction To observe physical and chemical changes. To identify
How many ml of M sodium hydroxide is required to completely react with 15.0 ml of 2.00 M sulfuric acid?
 147 EXAMPLE PROBLEM: How many ml of 0.250 M sodium hydroxide is required to completely react with 15.0 ml of 2.00 M sulfuric acid? 1 - Convert 15.0 ml to moles using concentration (2.00 mol acid per liter).
147 EXAMPLE PROBLEM: How many ml of 0.250 M sodium hydroxide is required to completely react with 15.0 ml of 2.00 M sulfuric acid? 1 - Convert 15.0 ml to moles using concentration (2.00 mol acid per liter).
5E Essential Lesson-SC.8.P.8.6. Element Name: Hydrogen (H) Element Name: Helium (He) Number of orbitals: 1. Number of valence electrons: 2
 Element Name: Hydrogen (H) Number of orbitals: 1 Number of protons: 1 Atomic Mass: 1.01 AMU Properties: gas, bonds with other elements, flammable Element Name: Helium (He) Number of orbitals: 1 Number
Element Name: Hydrogen (H) Number of orbitals: 1 Number of protons: 1 Atomic Mass: 1.01 AMU Properties: gas, bonds with other elements, flammable Element Name: Helium (He) Number of orbitals: 1 Number
E J The electron s energy difference between the second and third levels is J. = J
 The wavelength of light emitted is 654 nm. = c f c 3.00 10 8 m/s f c 3.00 108 m 1s 6.54 10 7 m f 4.59 4.59 10 14 z 1 s 10 14 The frequency of the light emitted is 4.59 10 14 z. E hf h 6.63 10 34 J/z E
The wavelength of light emitted is 654 nm. = c f c 3.00 10 8 m/s f c 3.00 108 m 1s 6.54 10 7 m f 4.59 4.59 10 14 z 1 s 10 14 The frequency of the light emitted is 4.59 10 14 z. E hf h 6.63 10 34 J/z E
Chemistry 40S Atomic Structure (This unit has been adapted from https://bblearn.merlin.mb.ca)
 Chemistry 40S Atomic Structure (This unit has been adapted from https://bblearn.merlin.mb.ca) Name: 1 Lesson 1: The Nature of Light Goals: Describe light in terms of electromagnetic energy. Describe the
Chemistry 40S Atomic Structure (This unit has been adapted from https://bblearn.merlin.mb.ca) Name: 1 Lesson 1: The Nature of Light Goals: Describe light in terms of electromagnetic energy. Describe the
