SNC2D CHEMISTRY 3/12/2013. CHEMICAL REACTIONS L Neutralization Reactions (P ) ph & Plants. ph & Plants
|
|
|
- Caroline Day
- 6 years ago
- Views:
Transcription
1 SNC2D CHEMISTRY CHEMICAL REACTIONS L Reactions (P ) ph & Plants Growing plants for a living can be risky. Many factors can affect the success of a crop, from weather conditions to the nutrient content of the soil. Soil ph is one of these factors. The ph of soil affects the growth of plants in a number of ways. For example, growers can change the colour of hydrangea flowers by changing the soil ph. NOTE! When the ph is 6.0 to 6.2, hydrangea flowers are pink. When the soil ph is 5.2 to 5.5, the flowers are blue. March 12, DCHEM - Reactions 1 ph & Plants Once soil ph is known, growers can use this information in one of two ways. Î They could plant crops that are most suited to the soil ph. For example, legumes (beans and peas) grow best at a ph of 6.2 or higher, but corn does well in soils with a ph as low as 6.0 March 12, DCHEM - Reactions 2 1
2 ph & Plants Once soil ph is known, growers can use this information in one of two ways. Ï A grower can adjust the ph of the soil to support particular plants. For example, If the soil is too acidic, adding a basic substance can increase the ph. If the soil is too basic, then adding acidic substances can lower the ph. March 12, DCHEM - Reactions 3 is a chemical reaction between an acid and a base that forms products that have a ph closer to 7 than either of the reactants. These products are usually an ionic compound (sometimes called a salt ) and water. A neutralization reaction can be summarized as follows: acid + base þ salt + water March 12, DCHEM - Reactions 4 As you know, acids form hydrogen ions (H + ) in water, and most bases release hydroxide ions (OH ). So when an acid and a base are mixed, the hydrogen ions and the hydroxide ions quickly react to produce water, which is neutral and has a ph of around 7. March 12, DCHEM - Reactions 5 2
3 NEUTRALIZATION chemical reaction in which an acid and a base react to form an ionic compound (a salt) and water acid + base þ salt + water the resulting ph is closer to 7 (neutral) March 12, DCHEM - Reactions 6 PRACTICE 1. Write (i) word equations and (ii) balanced chemical equations to represent the reaction between the following pairs of substances. (a) hydrochloric acid (HCl) and potassium hydroxide (KOH) (a) hydrochloric acid + potassium hydroxide þ potassium chloride + water 1HCl + 1KOH þ 1 KCl + 1H 2 O March 12, DCHEM - Reactions 7 PRACTICE 1. Write (i) word equations and (ii) balanced chemical equations to represent the reaction between the following pairs of substances. (b) sulphuric acid (H 2 SO 4 ) and sodium hydroxide (NaOH) (b) sulphuric acid + sodium hydroxide þ sodium sulphate + water 1H 2 SO NaOH þ 1 Na 2 SO H 2 O March 12, DCHEM - Reactions 8 3
4 PRACTICE 2. The chemical equation below represents the cleanup for the hazardous spill of a base: 1 H 3 PO KOH þ 1 K 3 PO H 2 O (a) What type of reaction is this? (a) double displacement March 12, DCHEM - Reactions 9 PRACTICE 2. The chemical equation below represents the cleanup for the hazardous spill of a base: 1 H 3 PO KOH þ 1 K 3 PO H 2 O (b) Write the general equation for this kind of reaction. (b) AB + CD þ AD + CB March 12, DCHEM - Reactions 10 PRACTICE 2. The chemical equation below represents the cleanup for the hazardous spill of a base: 1 H 3 PO KOH þ 1 K 3 PO H 2 O (c) How do you think the ph of the products compared to the ph of the initial spill? (c) ph of reactants acid (<7) and a base (>7) ph of products neutral March 12, DCHEM - Reactions 11 4
5 Applications of reactions have numerous commercial uses including: manufacturing if the ph of a solution can be changed from basic to acidic (or vice versa) a precipitate can be formed and filtered off agriculture when calcium carbonate is added to acidic soil, the soil ph becomes more basic food industry fish is a weak base so when lemon juice is added, the ph is lowered and the fishy odour is eliminated medicine when a person takes an antacid, the stomach acid becomes more basic home oven cleaner is a strong base and can be cleaned up with a solution of vinegar (acetic acid) in water environmental challenges chemical spills and acid rain March 12, DCHEM - Reactions 12 Environmental Challenges Chemical Spills One environmental challenge is chemical spills. For example, sulphuric acid and sodium hydroxide (also called caustic soda) are two of the most widely used industrial chemicals. Huge quantities of these corrosive chemicals are transported annually by truck, rail, or ship. Even with strict laws controlling how industrial chemicals are transported, accidents happen. March 12, DCHEM - Reactions 13 Environmental Challenges Chemical Spills For example, on March 31, 2007, a train was hauling L of sulphuric acid near Englehart in Northern Ontario when the train suddenly derailed and spilled some of its cargo into the Blanche River. An ecological disaster occurred dead fish washed up on shore, and local residents were warned not to allow their livestock to drink river water. March 12, DCHEM - Reactions 14 5
6 Environmental Challenges Chemical Spills The emergency response crew added calcium oxide (lime), to the river slightly upstream from the spill site. They wanted to neutralize the acid leaking from the containers. The spill team relied on a two-step neutralization reaction: Î CaO + H 2 O þ Ca(OH) 2 Ï Ca(OH) 2 + H 2 SO 4 þ H 2 O + CaSO 4 March 12, DCHEM - Reactions 15 Environmental Challenges Chemical Spills NOTE! During this process, the ph of the river water downstream was carefully monitored to ensure that just the right amount of lime was being added. March 12, DCHEM - Reactions 16 Environmental Challenges Acid Precipitation Another environmental challenge is acid precipitation. Acid precipitation is any precipitation that has a ph less than 5.6 (the ph of normal rainwater). The causes of acid precipitation are sulphur dioxide and nitrogen oxides in the atmosphere. These gases undergo chemical reactions that result in the formation of acids, which eventually fall as acid precipitation. For example, SO 2 + O 2 þ SO 3 SO 3 + H 2 O þ H 2 SO 4 March 12, DCHEM - Reactions 17 6
7 Environmental Challenges Acid Precipitation NOTE! Acid rain with a ph of 2.4 fell during a storm in New England. That s the same ph as vinegar! The ph of rain in Ontario s Muskoka-Haliburton area ranges between 3.9 and 4.4 March 12, DCHEM - Reactions 18 Environmental Challenges Acid Precipitation Acid precipitation affects us in a variety of ways: it can corrode the stone surfaces of buildings and statues and the concrete of roads and bridges (a neutralization reaction) it can corrode the iron reinforcing rods in structures (acids react with metals) it can change the ph of the soil in forests, which can cause trees and plants to die March 12, DCHEM - Reactions 19 Environmental Challenges Acid Precipitation Acid precipitation affects us in a variety of ways: it can cause the water in lakes, streams, and other freshwater bodies to become more acidic, which can cause fish and other organisms to die it is related to respiratory problems in children and people with asthma March 12, DCHEM - Reactions 20 7
8 Environmental Challenges Acid Precipitation NOTE! In Alberta and Saskatchewan most lakes are naturally protected from the effects of acid precipitation because they are surrounded by limestone (limestone reacts with excess acid to neutralize it). However, this is not true of lakes in Ontario they are situated on granite which does not react with acids and so they are at a greater risk of acidification. March 12, DCHEM - Reactions 21 Environmental Challenges Acid Precipitation Fortunately, one way to raise the ph of heavily acidified lakes is by adding lime. The process, though, is time consuming and costly. March 12, DCHEM - Reactions 22 Environmental Challenges ENVIRONMENTAL CHALLENGES chemical spills and acid precipitation (ph < 5.6) lime (calcium oxide) is used to help neutralize the effects (2 step process) but the process is time consuming and costly Î CaO + H 2 O þ Ca(OH) 2 Ï Ca(OH) 2 + H 2 SO 4 þ H 2 O + CaSO 4 March 12, DCHEM - Reactions 23 8
9 Environmental Challenges PRACTICE 3. As you just learned, one way to treat a lake polluted with acid rain is to add calcium hydroxide. Why is this only a short-term fix to the acid rain problem for the lake? unless industry changes its ways, the acid rain will continue to fall so the lake will continue to become acidic March 12, DCHEM - Reactions Why is it important to neutralize an acid spill before attempting to clean it up? it could be dangerous strong acids can cause a chemical burn March 12, DCHEM - Reactions Baking soda fizzes, releasing carbon dioxide gas, when it is added to an acid spill. (a) What evidence tells you this is a chemical change? (b) How could you use this property to tell when the acid is completely neutralized? (c) Why is baking soda a good choice for neutralizing acid spills at home? (a) fizzing a gas is produced (b) when the fizzing stops (c) available everywhere March 12, DCHEM - Reactions 26 9
10 3. Consider these compounds: HCl, KOH, NaCl, H 3 PO 4. Which could be used to raise the ph of pool water? lower the ph? Explain. to raise the ph you need a base KOH and H 3 PO 4 to lower the ph you need an acid HCl March 12, DCHEM - Reactions You can receive temporary relief from acid indigestion by using an antacid that contains a base. Why is it not a good idea to use an antacid routinely? stomach juice is meant to be acidic breaks down food but if it too basic it won t digest properly March 12, DCHEM - Reactions A student performed an experiment to test the effectiveness of two antacid tablets, brand X and brand Y. Î Each tablet was dissolved in 100 ml of water in separate beakers. Ï A sample of each solution was transferred to its own test tube. Ð Phenolphthalein(an indicator) was added to each test tube. Note: the solutions turned pink because the ph > 7 Ñ Drops of acid were added until the pink colour disappeared. Note: the antacid was neutralized at this point (ph = 7) March 12, DCHEM - Reactions 29 10
11 5. A student performed an experiment to test the effectiveness of two antacid tablets, brand X and brand Y. Ò The student obtained the following data. Antacid Size of sample (ml) Drops of acid added X Y (a) Which antacid is likely to be more effective at neutralizing acid indigestion? Explain. (a) brand Y since it took more acid to neutralize it (i.e. lower it s ph), brand Y will neutralize a greater quantity of acid than X March 12, DCHEM - Reactions A student performed an experiment to test the effectiveness of two antacid tablets, brand X and brand Y. Ò The student obtained the following data. Antacid Size of sample (ml) Drops of acid added X Y (b) If the brand X solution had a ph of 7.9, was the ph of the brand Y solution likely higher, lower, or the same? Explain. (b) higher since it took more acid to neutralize it March 12, DCHEM - Reactions 31 TEXTBOOK P.206 Q.1-3 P.216 Q.7-10,13-16 P.214 Activity B19: Neutralizing Acidic Lake... (see next page for details) NOTE! Check your work often see P.554 of your text. March 12, DCHEM - Reactions 32 11
12 Activity: Neutralizing Acidic Lake (B19/P.214) INSTRUCTIONS A. Read the activity B19: Neutralizing Acidic Lake Water Samples. B. Follow the instructions given (i.e. procedure 1 to 11). C. Answer the questions given (i.e. analysis 12-16). NOTE! This is a formal lab report. Be sure to use complete sentences, particularly when it asks you to explain, discuss, describe, Make sure you do your own work! March 12, DCHEM - Reactions 33 12
Topic 5 National Chemistry Summary Notes. Acids and Alkalis
 Topic 5 National Chemistry Summary Notes Acids and Alkalis Experiment Collect some samples of rain water LI 1 The ph Scale The ph scale is a continuous range of numbers from below 0 to above 14. Acids
Topic 5 National Chemistry Summary Notes Acids and Alkalis Experiment Collect some samples of rain water LI 1 The ph Scale The ph scale is a continuous range of numbers from below 0 to above 14. Acids
Neutralization Reactions
 Neutralization Reactions According to industry representatives, % of discarded car batteries in Ontario are recycled. This process prevents the hazardous contents of car batteries from contaminating the
Neutralization Reactions According to industry representatives, % of discarded car batteries in Ontario are recycled. This process prevents the hazardous contents of car batteries from contaminating the
! "#$%&'$!()! *+&$(,)#$! *+&$(,)#$! *+&$(,)#$!
 Learning Checkpoint, p. 197 1. A solution with a ph of 11 is basic. 2. A solution with a ph of 5 is acidic. 3. The ph of pure water is 7 (neutral).! "#$%&'$!()! *+&$(,)#$! 4. No, litmus paper does not
Learning Checkpoint, p. 197 1. A solution with a ph of 11 is basic. 2. A solution with a ph of 5 is acidic. 3. The ph of pure water is 7 (neutral).! "#$%&'$!()! *+&$(,)#$! 4. No, litmus paper does not
Neutralisation. November 29, neutral
 Neutralisation November 29, 2015 neutral STARTER Decide if the statements are true or false. 1. Vinegar is an example of an alkali. FALSE 2. Tea contains tannic acid. TRUE 3. Coke is an alkali. FALSE 4.
Neutralisation November 29, 2015 neutral STARTER Decide if the statements are true or false. 1. Vinegar is an example of an alkali. FALSE 2. Tea contains tannic acid. TRUE 3. Coke is an alkali. FALSE 4.
SNC2D CHEMISTRY 2/24/2013. CHEMICAL REACTIONS L Acids & Bases (P ) Activity: Introduction to (2DCHEM-ASG3) Introduction to Acids & Bases
 SNC2D CHEMISTRY CHEMICAL REACTIONS L Acids & Bases (P.198-201) Activity: Introduction to (2DCHEM-ASG3) INSTRUCTIONS A. Read the activity 2DCHEM - ASG3 (Introduction to Acids & Bases). B. Follow the instructions
SNC2D CHEMISTRY CHEMICAL REACTIONS L Acids & Bases (P.198-201) Activity: Introduction to (2DCHEM-ASG3) INSTRUCTIONS A. Read the activity 2DCHEM - ASG3 (Introduction to Acids & Bases). B. Follow the instructions
Classifying Substances
 Classifying Substances There are other ways you can use to classify compounds. For example, you can classify some compounds as acids or bases. You are very familiar with acids and bases because you see
Classifying Substances There are other ways you can use to classify compounds. For example, you can classify some compounds as acids or bases. You are very familiar with acids and bases because you see
Understand what acids and alkalis are, and where they are found.
 Lesson Aims- Understand what acids and alkalis are, and where they are found. Test a range of household products with litmus indicator to see whether they are acidic or alkaline. Found in citrus fruit
Lesson Aims- Understand what acids and alkalis are, and where they are found. Test a range of household products with litmus indicator to see whether they are acidic or alkaline. Found in citrus fruit
Everyday you encounter a variety of different acids and bases. Below is a list of som common acids and bases
 Acids and Bases Everyday you encounter a variety of different acids and bases. Below is a list of som common acids and bases Milk carbonic acid (H 2 CO 3 ) calcium hydroxide (Ca(OH) s ) sodium hydroxide
Acids and Bases Everyday you encounter a variety of different acids and bases. Below is a list of som common acids and bases Milk carbonic acid (H 2 CO 3 ) calcium hydroxide (Ca(OH) s ) sodium hydroxide
Unit 5 Lesson 3 Measuring ph. Copyright Houghton Mifflin Harcourt Publishing Company
 What s Your Number? What is the ph scale? The ph of a solution is a measure of how acidic or basic a solution is. A solution that has a ph value of exactly 7 is neutral neither acidic nor basic. A solution
What s Your Number? What is the ph scale? The ph of a solution is a measure of how acidic or basic a solution is. A solution that has a ph value of exactly 7 is neutral neither acidic nor basic. A solution
Have a ph value less than ph 7 Turn blue litmus indicator red Can neutralise an alkali Have a sour taste (WARNING: never taste any chemicals)
 Acids and Alkalis ACIDS (acidic solutions) Acids have the following properties: Have a ph value less than ph 7 Turn blue litmus indicator red Can neutralise an alkali Have a sour taste (WARNING: never
Acids and Alkalis ACIDS (acidic solutions) Acids have the following properties: Have a ph value less than ph 7 Turn blue litmus indicator red Can neutralise an alkali Have a sour taste (WARNING: never
Water, the SPECIAL Equilibrium
 THE ACID TEST Water, the SPECIAL Equilibrium I. Characteristics of Water A. Water are highly. B. They are in continuous. C. Always. D. Water is dense in the solid phase than in the phase. i.e. ice floats
THE ACID TEST Water, the SPECIAL Equilibrium I. Characteristics of Water A. Water are highly. B. They are in continuous. C. Always. D. Water is dense in the solid phase than in the phase. i.e. ice floats
UNIT 10 COMMON ACIDS AND ALKALIS
 ABLE EDUCATION CENTRE UNIT 10 COMMON ACIDS AND ALKALIS A Multiple-choice questions 1 Which of the following are the properties of alkalis? (1) They taste sour. (2) They are slippery. (3) They can turn
ABLE EDUCATION CENTRE UNIT 10 COMMON ACIDS AND ALKALIS A Multiple-choice questions 1 Which of the following are the properties of alkalis? (1) They taste sour. (2) They are slippery. (3) They can turn
Downloaded from
 1 X Chemistry Chapter 2 Acids, Bases and Salts Chapter Notes Top concepts: 1. Definition of acids, bases and salts: Acids Bases Salts Sour in taste Bitter in taste & soapy to touch Acid + Base Salt + Water
1 X Chemistry Chapter 2 Acids, Bases and Salts Chapter Notes Top concepts: 1. Definition of acids, bases and salts: Acids Bases Salts Sour in taste Bitter in taste & soapy to touch Acid + Base Salt + Water
Science 10. Unit 2: Chemistry. Book 5: Acid -Base Chemistry & the ph Scale. Block: Name:
 Science 10 Unit 2: Chemistry Book 5: Acid -Base Chemistry & the ph Scale Name: Block: 1 Classifying Substances There are other ways you can use to classify compounds. For example, you can classify some
Science 10 Unit 2: Chemistry Book 5: Acid -Base Chemistry & the ph Scale Name: Block: 1 Classifying Substances There are other ways you can use to classify compounds. For example, you can classify some
Name: Block: Date: Student Notes
 Name: Block: Date: LCPS Core Experience Acids and Bases Student Notes OBJECTIVES Students will: recognize some acids and bases as common and familiar household chemicals. realize that acids and bases are
Name: Block: Date: LCPS Core Experience Acids and Bases Student Notes OBJECTIVES Students will: recognize some acids and bases as common and familiar household chemicals. realize that acids and bases are
Page 1 of 14. Website: Mobile:
 Question 1: You have been provided with three test tubes. One of them contains distilled water and the other two contain an acidic solution and a basic solution, respectively. If you are given only red
Question 1: You have been provided with three test tubes. One of them contains distilled water and the other two contain an acidic solution and a basic solution, respectively. If you are given only red
Homework Acids and Alkalis
 S2 Homework Acids and Alkalis This booklet is split into the main areas of the topic. Each main area has three different spice levels which tell you about the difficulty of the homework. Each week your
S2 Homework Acids and Alkalis This booklet is split into the main areas of the topic. Each main area has three different spice levels which tell you about the difficulty of the homework. Each week your
1. Base your answer to the following question on information below and on your knowledge of chemistry.
 1. Base your answer to the following question on information below and on your knowledge of A sample of nitric acid contains both ions and ions. This sample has a ph value of 1. Write a name of the positive
1. Base your answer to the following question on information below and on your knowledge of A sample of nitric acid contains both ions and ions. This sample has a ph value of 1. Write a name of the positive
Elements and Their Oxides
 Elements and Their Oxides An oxide is a. Oxides can form when an element reacts with oxygen, often in air. This reaction can be rapid with the release of a great deal of energy, as in the combustion of
Elements and Their Oxides An oxide is a. Oxides can form when an element reacts with oxygen, often in air. This reaction can be rapid with the release of a great deal of energy, as in the combustion of
Acids & Bases Strong & weak. Thursday, October 20, 2011
 Acids & Bases Strong & weak 1 Acid Base Dissociation Acid-base reactions are equilibrium processes. The relationship between the relative concentrations of the reactants and products is a constant for
Acids & Bases Strong & weak 1 Acid Base Dissociation Acid-base reactions are equilibrium processes. The relationship between the relative concentrations of the reactants and products is a constant for
Acids and Bases. Say Thanks to the Authors Click (No sign in required)
 Acids and Bases Say Thanks to the Authors Click http://www.ck12.org/saythanks (No sign in required) To access a customizable version of this book, as well as other interactive content, visit www.ck12.org
Acids and Bases Say Thanks to the Authors Click http://www.ck12.org/saythanks (No sign in required) To access a customizable version of this book, as well as other interactive content, visit www.ck12.org
Introducing Science Summary Sheet
 Introducing Science Summary Sheet Acids in the laboratory Dilute acids You will have used some dilute acids at school, such as hydrochloric acid, sulphuric acid and nitric acid. Their bottles are labelled
Introducing Science Summary Sheet Acids in the laboratory Dilute acids You will have used some dilute acids at school, such as hydrochloric acid, sulphuric acid and nitric acid. Their bottles are labelled
Name: Date: Number: Acids
 Acids The sour taste of the lemon juice tells us that it is an acid. Acids are special kinds of chemicals. They are common in everyday life. Some are helpful, others are harmful. There are some that are
Acids The sour taste of the lemon juice tells us that it is an acid. Acids are special kinds of chemicals. They are common in everyday life. Some are helpful, others are harmful. There are some that are
Acids, Bases & Salts
 Introduction Acids, Bases & Salts Elements combine to form numerous compounds. On the basis of their chemical properties, compounds can be classified into three categories: Acids Bases Salts Acids and
Introduction Acids, Bases & Salts Elements combine to form numerous compounds. On the basis of their chemical properties, compounds can be classified into three categories: Acids Bases Salts Acids and
Unit 1: Chemistry in Action
 Unit 1: Chemistry in Action Intermediate 1 Chemistry Learning Outcomes Substances Elements Everything in the world is made from about 100 elements. Each element has a name and a symbol. Chemists have arranged
Unit 1: Chemistry in Action Intermediate 1 Chemistry Learning Outcomes Substances Elements Everything in the world is made from about 100 elements. Each element has a name and a symbol. Chemists have arranged
3/26/2011. explosion
 Chemistry Acids and Bases Year 10 Study of Chemicals and how they react Everything is made of chemicals They are made of elements. There are over 100 elements Acids are common Some are dangerous and can
Chemistry Acids and Bases Year 10 Study of Chemicals and how they react Everything is made of chemicals They are made of elements. There are over 100 elements Acids are common Some are dangerous and can
Bio 105 Lab 3: Chemistry: ph and solutions
 1 Bio 105 Lab 3: Chemistry: ph and solutions Part 1. Acid and Base Chemistry A. Introduction BIO 105 Summer 2013 Name One of the most important concepts in biology is acid/base chemistry. We are familiar
1 Bio 105 Lab 3: Chemistry: ph and solutions Part 1. Acid and Base Chemistry A. Introduction BIO 105 Summer 2013 Name One of the most important concepts in biology is acid/base chemistry. We are familiar
ACIDS & BASES. Acids & Bases 1
 ACIDS & BASES Acids and bases have real-life significance. The human body functions properly only when delicate acid-base balances are maintained and crops grow best in soil with the proper ph. In addition,
ACIDS & BASES Acids and bases have real-life significance. The human body functions properly only when delicate acid-base balances are maintained and crops grow best in soil with the proper ph. In addition,
Which other compounds react with acids to produce salts? Acids can also react with metals and carbonates to produce salts.
 Salts Textbook pages 234 243 Section 5.2 Summary Before You Read How many different uses for salts can you name? Write your answers on the lines below. What are salts? In chemistry, salts are a class of
Salts Textbook pages 234 243 Section 5.2 Summary Before You Read How many different uses for salts can you name? Write your answers on the lines below. What are salts? In chemistry, salts are a class of
The grade 5 English science unit, Acids and Bases, meets the academic content standards set in the Korean curriculum, which state students should:
 This area addresses ph among different characteristics of solutions. It will be interesting for students to classify a variety of solutions into acids and bases by using the characteristics of the solutions.
This area addresses ph among different characteristics of solutions. It will be interesting for students to classify a variety of solutions into acids and bases by using the characteristics of the solutions.
Examples of Strong Acids: Strong Acid Formula Common Source Hydrochloric Acid HCl Stomach Acid
 ACIDS AND BASES: PH AND BUFFERS PURPOSE: To determine the ph of common acids and bases using a ph meter, ph paper, and red cabbage indicator. To test the effect of adding an acid or base to a buffer solution.
ACIDS AND BASES: PH AND BUFFERS PURPOSE: To determine the ph of common acids and bases using a ph meter, ph paper, and red cabbage indicator. To test the effect of adding an acid or base to a buffer solution.
Fertilisers. Topic 12 National 5 Chemistry Summary Notes
 Fertilisers LI 1 Topic 12 National 5 Chemistry Summary Notes Chemistry is extremely important to the future of food production. As the population of the world increases, more and more efficient ways of
Fertilisers LI 1 Topic 12 National 5 Chemistry Summary Notes Chemistry is extremely important to the future of food production. As the population of the world increases, more and more efficient ways of
DOUBLE DISPLACEMENT REACTIONS. Double your pleasure, double your fun
 DOUBLE DISPLACEMENT REACTIONS Double your pleasure, double your fun Industrial processes produce unwanted by-products. Dissolved toxic metal ions-copper, mercury, and cadmium-are common leftovers in the
DOUBLE DISPLACEMENT REACTIONS Double your pleasure, double your fun Industrial processes produce unwanted by-products. Dissolved toxic metal ions-copper, mercury, and cadmium-are common leftovers in the
Acids, Bases and Salts
 Acids, Bases and Salts Synopsis Substances are classified into acids, bases and salts depending on their chemical behavior and properties. Acids are of sour taste and turn blue litmus to red. Bases are
Acids, Bases and Salts Synopsis Substances are classified into acids, bases and salts depending on their chemical behavior and properties. Acids are of sour taste and turn blue litmus to red. Bases are
Acids and Bases 2 Science Notes JC-Learn. JC-Learn. Science Notes Acids and Bases 2. 1 P a g e
 JC-Learn Science Notes Acids and Bases 2 1 P a g e Acids and Bases 2 The two most common laboratory acids are hydrochloric acid (HCl) and sulfuric acid (H2SO4). The two most common laboratory bases are
JC-Learn Science Notes Acids and Bases 2 1 P a g e Acids and Bases 2 The two most common laboratory acids are hydrochloric acid (HCl) and sulfuric acid (H2SO4). The two most common laboratory bases are
Choose the correct answer. velocity changes in every two seconds by ms -1
 Acceleration Q I Choose the correct answer 1. If a body moves with uniform velocity its acceleration is -----zero. 2. Acceleration of a body is the rate of change of ----velocity 3. The acceleration of
Acceleration Q I Choose the correct answer 1. If a body moves with uniform velocity its acceleration is -----zero. 2. Acceleration of a body is the rate of change of ----velocity 3. The acceleration of
Chemical Reactions. Chemical Reactions Chemical reactions have a standard format when written:
 0.3.notebook A chemical property is a behaviour that occurs when substances change to create a new substance. When a new substance is created, a chemical change has occurred. New colour Evidence of chemical
0.3.notebook A chemical property is a behaviour that occurs when substances change to create a new substance. When a new substance is created, a chemical change has occurred. New colour Evidence of chemical
Wherever chemical solutions are involved, ph matters. Some
 47 Acids, Bases, and the ph Scale R EA D I N G Wherever chemical solutions are involved, ph matters. Some important chemical reactions, such as those involved in corrosion of iron or digestion of food,
47 Acids, Bases, and the ph Scale R EA D I N G Wherever chemical solutions are involved, ph matters. Some important chemical reactions, such as those involved in corrosion of iron or digestion of food,
Chapter 5 Notes Science 10 Name:
 5.1 Acids and Bases Many familiar compounds are acids or bases. o Classification as acids or bases is based on chemical composition. Acids and bases can be very dangerous. o Both can be very. o NEVER try
5.1 Acids and Bases Many familiar compounds are acids or bases. o Classification as acids or bases is based on chemical composition. Acids and bases can be very dangerous. o Both can be very. o NEVER try
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED)
 CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
Acids and Bases. Acids
 1 Acids and Bases Acids Although some acids can burn and are dangerous to handle, most acids in foods are safe to eat. What acids have in common, however, is that they contain at least one hydrogen atom
1 Acids and Bases Acids Although some acids can burn and are dangerous to handle, most acids in foods are safe to eat. What acids have in common, however, is that they contain at least one hydrogen atom
MONDAY (12/12) TUESDAY (12/13) WEDNESDAY (12/14) THURSDAY (12/15) FRIDAY (12/16) Making Acid Rain (a lab) Quiz
 Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
REACTIONS OF ACIDS. J:\Science\Chemistry\Stage 1 Notes\Acids & Bases\Reactionsofacids.doc
 REACTIONS OF ACIDS 1. Acids taste sour We do not attempt to taste strong acids as they are too dangerous. They do taste sour, but then they proceed to destroy cells on your tongue and mouth. If you vomit,
REACTIONS OF ACIDS 1. Acids taste sour We do not attempt to taste strong acids as they are too dangerous. They do taste sour, but then they proceed to destroy cells on your tongue and mouth. If you vomit,
2. What characteristic of water makes it the universal solvent? Nonpolar large molecules long-chain hydrocarbon molecules polar
 PS Chemistry Chapter 22 & 23 Review Test Date Chapter 22 Suggestions for Studying Section 1 Know that a solution is made up of a solute and solvent. Be able to provide an example of a solute and a solvent.
PS Chemistry Chapter 22 & 23 Review Test Date Chapter 22 Suggestions for Studying Section 1 Know that a solution is made up of a solute and solvent. Be able to provide an example of a solute and a solvent.
Unit 13 Acids and Bases E.Q. What are the differences between acids and bases?
 Unit 13 Acids and Bases E.Q. What are the differences between acids and bases? What are Properties of Acids? They taste sour (don t try this in lab). They can conduct electricity. Can be strong or weak
Unit 13 Acids and Bases E.Q. What are the differences between acids and bases? What are Properties of Acids? They taste sour (don t try this in lab). They can conduct electricity. Can be strong or weak
HarperCollinsPublishers Licensed for home use only. Not for whiteboard or general classroom use
 BIG IDEAS Acids and alkalis You are learning to: Sort common materials into acids and alkalis Explain some properties of acids and alkalis Recognise the common chemical properties of acids and alkalis
BIG IDEAS Acids and alkalis You are learning to: Sort common materials into acids and alkalis Explain some properties of acids and alkalis Recognise the common chemical properties of acids and alkalis
Toxins 4/27/2010. Acids and Bases Lab. IV-17 to IV-22
 Toxins IV-17 to IV-22 Countless products are advertised on TV with the promise of reducing acid indigestion. a.what is acid indigestion? b.what does acid have to do with your stomach? c.how do you think
Toxins IV-17 to IV-22 Countless products are advertised on TV with the promise of reducing acid indigestion. a.what is acid indigestion? b.what does acid have to do with your stomach? c.how do you think
SNC2D CHEMISTRY 2/23/2013. CHEMICAL REACTIONS L Conservation of Mass (P ) Activity: Measuring Mass (Part 1) Activity: Measuring Mass (Part 1)
 SNC2D CHEMISTRY CHEMICAL REACTIONS L Conservation of Mass (P.176-177) Activity: Measuring Mass (Part 1) INSTRUCTIONS (CLOSED SYSTEM) A. Measure~ 30 ml of vinegar (acetic acid) into a small Erlenmeyer flask.
SNC2D CHEMISTRY CHEMICAL REACTIONS L Conservation of Mass (P.176-177) Activity: Measuring Mass (Part 1) INSTRUCTIONS (CLOSED SYSTEM) A. Measure~ 30 ml of vinegar (acetic acid) into a small Erlenmeyer flask.
MARK SCHEME for the May/June 2012 question paper for the guidance of teachers 0620 CHEMISTRY. 0620/21 Paper 2 (Core Theory), maximum raw mark 80
 UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education MARK SCHEME for the May/June 2012 question paper for the guidance of teachers 0620 CHEMISTRY
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education MARK SCHEME for the May/June 2012 question paper for the guidance of teachers 0620 CHEMISTRY
The Determination of ph of some Common Acids & Bases
 The Determination of ph of some Common Acids & Bases Introduction: An acid is a substance that when dissolved in water produces hydrogen ions, H +. Inorganic acids do not contain carbon but organic acids
The Determination of ph of some Common Acids & Bases Introduction: An acid is a substance that when dissolved in water produces hydrogen ions, H +. Inorganic acids do not contain carbon but organic acids
What is a chemical property of matter?
 What is a chemical property of matter? A. Chemical properties of matter involve a chemical change. 1. Observations of matter that involve a chemical change cause new matter to be formed. a. Chemical changes
What is a chemical property of matter? A. Chemical properties of matter involve a chemical change. 1. Observations of matter that involve a chemical change cause new matter to be formed. a. Chemical changes
7E Acids and alkalis Multiple-choice main test
 For each question, circle the correct answer. Question 1 Vinegar is an acid. Like some other acids vinegar tastes... A) salty B) sour C) sugary D) sweet Question 2 Acids may be hazardous chemicals. Concentrated
For each question, circle the correct answer. Question 1 Vinegar is an acid. Like some other acids vinegar tastes... A) salty B) sour C) sugary D) sweet Question 2 Acids may be hazardous chemicals. Concentrated
Ch. 8 - Solutions, Acids & Bases. Solution = a homogeneous mixture of 2 or more substances
 Ch. 8 - Solutions, Acids & Bases Solution = a homogeneous mixture of 2 or more substances Solute substance whose particles are dissolved in a solution Solvent substance in which the solute dissolves in
Ch. 8 - Solutions, Acids & Bases Solution = a homogeneous mixture of 2 or more substances Solute substance whose particles are dissolved in a solution Solvent substance in which the solute dissolves in
Reactions with Acids. Chemistry 10 Mrs. Page
 Reactions with Acids Chemistry 10 Mrs. Page Learning Objectives Predict the products and balance chemical equations for the reactions with acids involving; reactive metals, metal oxides, metal hydroxides,
Reactions with Acids Chemistry 10 Mrs. Page Learning Objectives Predict the products and balance chemical equations for the reactions with acids involving; reactive metals, metal oxides, metal hydroxides,
5.2. Neutralization Reactions. ph and Plants
 5.2 Neutralization Reactions Here is a summary of what you will learn in this section: Neutralization is a type of chemical reaction that occurs between an acid and a base and produces water and a salt.
5.2 Neutralization Reactions Here is a summary of what you will learn in this section: Neutralization is a type of chemical reaction that occurs between an acid and a base and produces water and a salt.
Acids Bases and Salts
 Acids Bases and Salts (By : Arvind Kumar, for class 10 X) Question 1: You have been provided with three test tubes. One of them contains distilled water and the other two contain an acidic solution and
Acids Bases and Salts (By : Arvind Kumar, for class 10 X) Question 1: You have been provided with three test tubes. One of them contains distilled water and the other two contain an acidic solution and
By All INdICATIONS (2 Hours)
 By All INdICATIONS (2 Hours) Addresses NGSS Level of Difficulty: 5 Grade Range: 6-8 OVERVIEW In this activity, students create an acid-base indicator using red cabbage extract. Students then use this indicator
By All INdICATIONS (2 Hours) Addresses NGSS Level of Difficulty: 5 Grade Range: 6-8 OVERVIEW In this activity, students create an acid-base indicator using red cabbage extract. Students then use this indicator
4.2. Double Displacement Reactions. Characteristics of Double Displacement Reactions SECTION. Key Terms
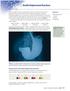 SECTION 4.2 Double Displacement Reactions X rays are highly useful for creating images of bones in the human body, but they generally do not show soft tissues clearly. To help doctors diagnose conditions
SECTION 4.2 Double Displacement Reactions X rays are highly useful for creating images of bones in the human body, but they generally do not show soft tissues clearly. To help doctors diagnose conditions
Chemical Reaction Defn: Chemical Reaction: when starting chemical species form different chemicals.
 Chemical Reaction Defn: Chemical Reaction: when starting chemical species form different chemicals. Evidence to indicate that a chemical reaction has occurred: Temperature change Different coloured materials
Chemical Reaction Defn: Chemical Reaction: when starting chemical species form different chemicals. Evidence to indicate that a chemical reaction has occurred: Temperature change Different coloured materials
... and... Complete the word equation for the reaction between sulphur and oxygen in the air. sulphur + oxygen...
 Level 5 Chemistry Questions 1. Burning fossil fuels causes air pollution. (a) Give the names of two fossil fuels.... and... Some fossil fuels contain sulphur. Complete the word equation for the reaction
Level 5 Chemistry Questions 1. Burning fossil fuels causes air pollution. (a) Give the names of two fossil fuels.... and... Some fossil fuels contain sulphur. Complete the word equation for the reaction
Acid-Base Theory. In this lecture the theory of acids and bases will be present along with web sites which discuss acids and bases
 Acid-Base Theory In this lecture the theory of acids and bases will be present along with web sites which discuss acids and bases Lecture Outline: these questions should be answered in this lecture. What
Acid-Base Theory In this lecture the theory of acids and bases will be present along with web sites which discuss acids and bases Lecture Outline: these questions should be answered in this lecture. What
ANSWERS: Acids and Bases
 1) a) Sulfuric acid + potassium hydroxide potassium ulphate + water H 2 SO 4 + 2KOH K 2 SO 4 + 2H 2 O ANSWERS: Acids and Bases b) As the KOH is added, the H 2 SO 4 is being neutralised until water is formed,
1) a) Sulfuric acid + potassium hydroxide potassium ulphate + water H 2 SO 4 + 2KOH K 2 SO 4 + 2H 2 O ANSWERS: Acids and Bases b) As the KOH is added, the H 2 SO 4 is being neutralised until water is formed,
Families of Chemical Compounds. Chapter 9
 Families of Chemical Compounds Chapter 9 Groups of Compounds Compounds are grouped based on physical and chemical properties Types: Organic, Acids, Bases, and Salts Acids and Bases Examples of Acids Aspirin
Families of Chemical Compounds Chapter 9 Groups of Compounds Compounds are grouped based on physical and chemical properties Types: Organic, Acids, Bases, and Salts Acids and Bases Examples of Acids Aspirin
NaCl (aq) + HOH (l) + (aq) + Cl (l) Eg. HCl (aq) + NH 3(aq) In both cases the acid and base react and neutralize each other.
 Acid Base Reactions An acid base reaction is called a neutralization reaction. Eg. HCl (aq) + NaOH (aq) NaCl (aq) + HOH (l) In grade 10 you learned that acid base reactions produce salt and water, however,
Acid Base Reactions An acid base reaction is called a neutralization reaction. Eg. HCl (aq) + NaOH (aq) NaCl (aq) + HOH (l) In grade 10 you learned that acid base reactions produce salt and water, however,
Global Experiment for the International Year of Chemistry. ph of the Planet
 Global Experiment for the International Year of Chemistry ph of the Planet This document contains a description for the ph of the Planet Activity that is part of the Global Experiment being conducted during
Global Experiment for the International Year of Chemistry ph of the Planet This document contains a description for the ph of the Planet Activity that is part of the Global Experiment being conducted during
(d) Bases turn red litmus blue and acids turn blue litmus red. Basic solution has a ph value more
 Page 34»Exercise» Question 1: A solution turns red litmus blue, its ph is likely to be (a) 1 (b) 4 (c) 5 (d) 10 (d) Bases turn red litmus blue and acids turn blue litmus red. Basic solution has a ph value
Page 34»Exercise» Question 1: A solution turns red litmus blue, its ph is likely to be (a) 1 (b) 4 (c) 5 (d) 10 (d) Bases turn red litmus blue and acids turn blue litmus red. Basic solution has a ph value
ACIDS & BASES PROPERTIES OF ACIDS ACIDS PROPERTIES OF ACIDS PROPERTIES OF ACIDS 11/1/2016
 SC STANDARD COVERED ACIDS & BASES Standard PS-3.7 Classify various solutions as acids or bases according to their physical properties, chemical properties (including neutralization and reaction with metals),
SC STANDARD COVERED ACIDS & BASES Standard PS-3.7 Classify various solutions as acids or bases according to their physical properties, chemical properties (including neutralization and reaction with metals),
How do Metal Carbonates and Metal Hydrogencarbonates React
 Acids, Bases, and Salts] This chapter is about: Reaction of acids and bases, how acids and bases cancel out each other s effects and many more interesting things that we use and see in our day-to-day life.
Acids, Bases, and Salts] This chapter is about: Reaction of acids and bases, how acids and bases cancel out each other s effects and many more interesting things that we use and see in our day-to-day life.
A salt is a neutral ionic compound composed of cations and anions. It is the result of an acid-base neutralisation reaction.
 Acid-base reactions When an acid and a base react, they form a salt. If the base contains hydroxide (OH ) ions, then water will also be formed. The word salt is a general term which applies to the products
Acid-base reactions When an acid and a base react, they form a salt. If the base contains hydroxide (OH ) ions, then water will also be formed. The word salt is a general term which applies to the products
An acidic substance always contains (1) in its name. An (2) is a chemical that changes colour to tell you
 Test Part A Fill in the gaps An acidic substance always contains (1) in its name. An (2) is a chemical that changes colour to tell you whether a substance is an acid or a (3). Another name for (4)/(5)
Test Part A Fill in the gaps An acidic substance always contains (1) in its name. An (2) is a chemical that changes colour to tell you whether a substance is an acid or a (3). Another name for (4)/(5)
S4 CHEMISTRY SUMMARY NOTES
 S4 CHEMISTRY SUMMARY NOTES 1. The Mole One mole of a substance = GRAM FORMULA MASS e.g. H 2 SO 4 RAM from databook pg.7 2H 2 x 1 = 2 1S 1 x 32 = 32 4O 4 x 16 = 64 98g Mass = number of moles x Mass of 1
S4 CHEMISTRY SUMMARY NOTES 1. The Mole One mole of a substance = GRAM FORMULA MASS e.g. H 2 SO 4 RAM from databook pg.7 2H 2 x 1 = 2 1S 1 x 32 = 32 4O 4 x 16 = 64 98g Mass = number of moles x Mass of 1
Experiment 8 - Double Displacement Reactions
 Experiment 8 - Double Displacement Reactions A double displacement reaction involves two ionic compounds that are dissolved in water. In a double displacement reaction, it appears as though the ions are
Experiment 8 - Double Displacement Reactions A double displacement reaction involves two ionic compounds that are dissolved in water. In a double displacement reaction, it appears as though the ions are
Acids, Bases and ph. Biology Honors. Acidic, Basic or Neutral (indicate if strong or weak acid or base) Substance ph More H + More OH - Vitamin C
 Biology Honors Name Acids, Bases and ph Substance ph More H + More OH - Acidic, Basic or Neutral (indicate if strong or weak acid or base) Vitamin C Tap water Hydrochloric acid (HCl) Sodium hydroxide (NaOH)
Biology Honors Name Acids, Bases and ph Substance ph More H + More OH - Acidic, Basic or Neutral (indicate if strong or weak acid or base) Vitamin C Tap water Hydrochloric acid (HCl) Sodium hydroxide (NaOH)
Railways (Group-D & ALP) Science Questions with solution
 Railways (Group-D & ALP) Science Questions with solution RRB Science MCQs Quiz 5 Directions: Study the following questions carefully and choose the right answer: 1. The process of dissolving an acid or
Railways (Group-D & ALP) Science Questions with solution RRB Science MCQs Quiz 5 Directions: Study the following questions carefully and choose the right answer: 1. The process of dissolving an acid or
Channa Asela
 Reproducing the following contents by amending or deleting the author s name and contact numbers is prohibited. You may email or print without any amendment. Underline the most suitable answer 1995-I-2
Reproducing the following contents by amending or deleting the author s name and contact numbers is prohibited. You may email or print without any amendment. Underline the most suitable answer 1995-I-2
Unit 5 Lesson 2 Acids, Bases, and Salts. Copyright Houghton Mifflin Harcourt Publishing Company
 Donations Accepted What are acids and bases? Acids and bases are chemicals that increase the number of ions present in a water solution when they dissolve. Lemon juice and vinegar both contain acid. Shampoo
Donations Accepted What are acids and bases? Acids and bases are chemicals that increase the number of ions present in a water solution when they dissolve. Lemon juice and vinegar both contain acid. Shampoo
Chemical Reaction Defn: Chemical Reaction: when starting chemical species form different chemicals.
 Chemistry 11 Notes on Chemical Reactions Chemical Reaction Defn: Chemical Reaction: when starting chemical species form different chemicals. Evidence to indicate that a chemical reaction has occurred:
Chemistry 11 Notes on Chemical Reactions Chemical Reaction Defn: Chemical Reaction: when starting chemical species form different chemicals. Evidence to indicate that a chemical reaction has occurred:
INTRODUCTORY CHEMISTRY Concepts and Critical Thinking Seventh Edition by Charles H. Corwin
 Lecture INTRODUCTORY CHEMISTRY Concepts and Critical Thinking Seventh Edition by Charles H. Corwin Acids and Bases Properties of Acids An acid is any substance that releases hydrogen ions, H +, into water.
Lecture INTRODUCTORY CHEMISTRY Concepts and Critical Thinking Seventh Edition by Charles H. Corwin Acids and Bases Properties of Acids An acid is any substance that releases hydrogen ions, H +, into water.
Form 4 Chapter 7: Acid and Bases
 Form 4 Chapter 7: Acid and Bases The ph Scale Properties Acids Alkalis Physical. Substances that ionized in water to produce hydrogen ions.. Sour taste.. Turn blue litmus paper red. 4. Give a ph value
Form 4 Chapter 7: Acid and Bases The ph Scale Properties Acids Alkalis Physical. Substances that ionized in water to produce hydrogen ions.. Sour taste.. Turn blue litmus paper red. 4. Give a ph value
Lesson 1: What are chemical changes?
 Lesson 1 Summary Lesson 1: What are chemical changes? Vocabulary Use with pp. 375 377 physical change a change in which the material keeps its identify chemical change a change in which one substance or
Lesson 1 Summary Lesson 1: What are chemical changes? Vocabulary Use with pp. 375 377 physical change a change in which the material keeps its identify chemical change a change in which one substance or
-log [H+][OH-] = - log [1 x ] Left hand side ( log H + ) + ( log OH - ) = ph + poh Right hand side = ( log 1) + ( log ) = 14 ph + poh = 14
![-log [H+][OH-] = - log [1 x ] Left hand side ( log H + ) + ( log OH - ) = ph + poh Right hand side = ( log 1) + ( log ) = 14 ph + poh = 14 -log [H+][OH-] = - log [1 x ] Left hand side ( log H + ) + ( log OH - ) = ph + poh Right hand side = ( log 1) + ( log ) = 14 ph + poh = 14](/thumbs/77/75028098.jpg) Autoionization of Water H 2 O H + + OH - K = [H + ][OH - ]/[H 2 O] = 1.802 x 10-16 Concentration of [H 2 O] is so HIGH autoionization is just a drop in the bucket, so [H 2 O] stays constant at 55.5 M,
Autoionization of Water H 2 O H + + OH - K = [H + ][OH - ]/[H 2 O] = 1.802 x 10-16 Concentration of [H 2 O] is so HIGH autoionization is just a drop in the bucket, so [H 2 O] stays constant at 55.5 M,
CH 13 Solutions All Practice Problems
 CH 13 Solutions All Practice Problems CH 13 Solutions Practice #1 A chemical analysis shows that a sample contains 0.0044 grams of lead in 1.00 x 10 3 grams of water. Convert the measurement to parts per
CH 13 Solutions All Practice Problems CH 13 Solutions Practice #1 A chemical analysis shows that a sample contains 0.0044 grams of lead in 1.00 x 10 3 grams of water. Convert the measurement to parts per
Aim: What is ph? Mr. M. Gonzalez. What is ph?
 Aim: Mr. M. Gonzalez Do now: What happens when you combine baking soda and vinegar? A Chemical Reaction!!! Answer The reaction between baking soda (NaHCO3) and vinegar (CH3COOH) is actually two reactions,
Aim: Mr. M. Gonzalez Do now: What happens when you combine baking soda and vinegar? A Chemical Reaction!!! Answer The reaction between baking soda (NaHCO3) and vinegar (CH3COOH) is actually two reactions,
5.1. ? Create a Quiz. Acids and Bases. Before You Read. What are acids and bases? What is ph? What are ph indicators?
 Acids and Bases Textbook pages 220 233 Section 5.1 Summary Before You Read Many acids and bases can be found in your home. Describe one acid and one base that you are familiar with. Record your answer
Acids and Bases Textbook pages 220 233 Section 5.1 Summary Before You Read Many acids and bases can be found in your home. Describe one acid and one base that you are familiar with. Record your answer
Acids and acid rock drainage
 direct environmental impacts of mining at lihir high school booklet 3 page 14 Acids and acid rock drainage Could acid from the mine damage our oceans? This leaflet explains all about acids and acid rock
direct environmental impacts of mining at lihir high school booklet 3 page 14 Acids and acid rock drainage Could acid from the mine damage our oceans? This leaflet explains all about acids and acid rock
Acids, Bases, and Salts
 Acids, Bases, and Salts 1 An acid is a substance that releases hydrogen ions when it dissociates. H +1 Cl -1 Cl -1 H +1 2 An acid is a substance that releases hydrogen ions when it dissociates in water.
Acids, Bases, and Salts 1 An acid is a substance that releases hydrogen ions when it dissociates. H +1 Cl -1 Cl -1 H +1 2 An acid is a substance that releases hydrogen ions when it dissociates in water.
Chemical Names and Formulas
 Cool Chemistry Show Activity 3 Chemical Names and Formulas GOALS In this activity you will: Predict the charges of ions of some elements. Determine the formulas of ionic compounds. Write the conventional
Cool Chemistry Show Activity 3 Chemical Names and Formulas GOALS In this activity you will: Predict the charges of ions of some elements. Determine the formulas of ionic compounds. Write the conventional
Unit 1 Chemical Changes & Structure. National 4 Unit Rates of Reaction 2. Atomic Structure and Bonding 3. Acids & Bases 4. Energy Changes - 1 -
 National 4 Unit 1 1. Rates of Reaction 2. Atomic Structure and Bonding 3. Acids & Bases 4. Energy Changes - 1 - Learning Intentions Reaction Rates & Atomic Structure Learning Intentions Tick I know the
National 4 Unit 1 1. Rates of Reaction 2. Atomic Structure and Bonding 3. Acids & Bases 4. Energy Changes - 1 - Learning Intentions Reaction Rates & Atomic Structure Learning Intentions Tick I know the
8.3 WORKING WITH SOLUTIONS 8.4 ACID BASE THEORIES 8.4 ACID BASE THEORIES PRACTICE. Applying Inquiry Skills PRACTICE. Making Connections PRACTICE
 8.3 WORKING WITH SOLUTIONS (Page 376) Applying Inquiry Skills 1. Name Use Erlenmeyer flask for temporarily containing reacting substances (specifically shaped to allow mixing of contents by swirling) and
8.3 WORKING WITH SOLUTIONS (Page 376) Applying Inquiry Skills 1. Name Use Erlenmeyer flask for temporarily containing reacting substances (specifically shaped to allow mixing of contents by swirling) and
Flushing Out the Moles in Lab: The Reaction of Calcium Chloride with Carbonate Salts
 Flushing Out the Moles in Lab: The Reaction of Calcium Chloride with Carbonate Salts Pre-lab Assignment: Reading: 1. Chapter sections 3.3, 3.4, 3.7 and 4.2 in your course text. 2. This lab handout. Questions:
Flushing Out the Moles in Lab: The Reaction of Calcium Chloride with Carbonate Salts Pre-lab Assignment: Reading: 1. Chapter sections 3.3, 3.4, 3.7 and 4.2 in your course text. 2. This lab handout. Questions:
TYPES OF CHEMICAL REACTIONS
 TYPES OF CHEMICAL REACTIONS Precipitation Reactions Compounds Soluble Ionic Compounds 1. Group 1A cations and NH 4 + 2. Nitrates (NO 3 ) Acetates (CH 3 COO ) Chlorates (ClO 3 ) Perchlorates (ClO 4 ) Solubility
TYPES OF CHEMICAL REACTIONS Precipitation Reactions Compounds Soluble Ionic Compounds 1. Group 1A cations and NH 4 + 2. Nitrates (NO 3 ) Acetates (CH 3 COO ) Chlorates (ClO 3 ) Perchlorates (ClO 4 ) Solubility
Unit 5. Chemical reactions
 Unit 5. Chemical reactions Index 1.- Physical and chemical phenomena...2 2.- What is a chemical reaction?...2 2.1. Chemical equation...2 2.2.- Balance of chemical reactions. Law of conservation of mass...3
Unit 5. Chemical reactions Index 1.- Physical and chemical phenomena...2 2.- What is a chemical reaction?...2 2.1. Chemical equation...2 2.2.- Balance of chemical reactions. Law of conservation of mass...3
Chapter 14 Acids and Bases
 Chapter 14 Acids and Bases Acids and bases are important substances in health, industry, and the environment. One of the most common characteristics of acids is their sour taste. In 1887, the Swedish chemist
Chapter 14 Acids and Bases Acids and bases are important substances in health, industry, and the environment. One of the most common characteristics of acids is their sour taste. In 1887, the Swedish chemist
Lab: Cabbages in Chemistry 3pts ec printing in COLOR / 2pts B&W
 Lab: Cabbages in Chemistry 3pts ec printing in COLOR / 2pts B&W Telltale Colors 1. In the mixing tray, place 5 drops of the chemical in 13 compartments 2. DIP test the ph paper & record the ph measurement
Lab: Cabbages in Chemistry 3pts ec printing in COLOR / 2pts B&W Telltale Colors 1. In the mixing tray, place 5 drops of the chemical in 13 compartments 2. DIP test the ph paper & record the ph measurement
Mixtures, Solubility, and Acid/Base Solutions
 Mixtures, Solubility, and Acid/Base Soluts Acid and Base Soluts What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you
Mixtures, Solubility, and Acid/Base Soluts Acid and Base Soluts What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you
(b) Calculate the concentration of the hydrochloric acid in mol/dm 3. Calculate the number of moles of hydrochloric acid reacting.
 Answer = mol/dm 3 Answer = mol/dm 3 Chemical Changes Part 4 Q1. An oven cleaner solution contained sodium hydroxide. A 25.0 cm 3 sample of the oven cleaner solution was placed in a flask. The sample was
Answer = mol/dm 3 Answer = mol/dm 3 Chemical Changes Part 4 Q1. An oven cleaner solution contained sodium hydroxide. A 25.0 cm 3 sample of the oven cleaner solution was placed in a flask. The sample was
NCERT Solutions. 95% Top Results. 12,00,000+ Hours of LIVE Learning. 1,00,000+ Happy Students. About Vedantu. Awesome Master Teachers
 Downloaded from Vedantu NCERT Solutions About Vedantu Vedantu is India s biggest LIVE online teaching platform with over 450+ best teachers from across the country. Every week we are coming up with awesome
Downloaded from Vedantu NCERT Solutions About Vedantu Vedantu is India s biggest LIVE online teaching platform with over 450+ best teachers from across the country. Every week we are coming up with awesome
Lesson Five: Acids, Bases, ph, and Buffers
 Lesson Five: Acids, Bases, ph, and Buffers Arrhenius Acids and Bases Acids and bases can be defined a number of ways. One of the oldest and most common ways is the definition according to Arrhenius, named
Lesson Five: Acids, Bases, ph, and Buffers Arrhenius Acids and Bases Acids and bases can be defined a number of ways. One of the oldest and most common ways is the definition according to Arrhenius, named
Which substance would most neutralize an acidic food? A. dairy (ph 5 7) B. water (ph 6 7) C. citrus fruit (ph 2 3) D. baking soda (ph 8 9)
 Which substance would most neutralize an acidic food? A. dairy (ph 5 7) B. water (ph 6 7) C. citrus fruit (ph 2 3) D. baking soda (ph 8 9) The table below shows the ph and reaction to litmus of four body
Which substance would most neutralize an acidic food? A. dairy (ph 5 7) B. water (ph 6 7) C. citrus fruit (ph 2 3) D. baking soda (ph 8 9) The table below shows the ph and reaction to litmus of four body
Identification of White Powders
 CSI Chemistry Activity 4 Identification of White Powders GOALS In this activity you will: Create and use a flowchart to identify an unknown entity. Identify an unknown ionic compound based on an understanding
CSI Chemistry Activity 4 Identification of White Powders GOALS In this activity you will: Create and use a flowchart to identify an unknown entity. Identify an unknown ionic compound based on an understanding
