BUFFERING MECHANISM AND SENSITIVITY TO ACID DEPOSITION OF SOILS OF AKWA IBOM STATE, NIGERIA
|
|
|
- Evelyn Gilbert
- 6 years ago
- Views:
Transcription
1 BUFFERING MECHANISM AND SENSITIVITY TO ACID DEPOSITION OF SOILS OF AKWA IBOM STATE, NIGERIA ABSTRACT Akpan *, U. S. and Udoh, B. T. Department of Soil Science and Land Resources Management, University of Uyo,Uyo, Akwa Ibom State, Nigeria The objectives of this study were to identify and classify the buffering mechanisms and sensitivity to acid deposition of soils of Akwa Ibom State. Four parent materials including coastal plain sands, beach ridge sands, river alluvium and sandstone and shale were selected for the study. In each parent material, three profile pits were sunk at representative locations.. Soil samples were collected from each genetic horizon. A total of 12 profile pits were sunk and 62 samples were generated for laboratory analysis. The study revealed that aluminium was the buffering mechanisms in beach ridge sand soils while cation exchange and aluminium were the buffering mechanisms in sandstone and shale and river alluvium soils. Also, aluminium and cation exchange were the major buffering mechanisms in coastal plain sand soils. Coastal plain sand, river alluvium and sandstone and shale soils had higher buffering capacity than beach ridge sand soils. The study also showed that beach ridge sand and sandstone and shale soils were highly sensitive to acidification. Coastal plain soil was moderately sensitive while soils of river alluvium varied from low to high in sensitivity to acid deposition depending on the composition of the depositional materials. Keywords: Buffering mechanism, soil sensitivity, soil buffering INTRODUCTION Buffering mechanisms are materials involved in the neutralization of acid or base when acid or alkaline is added to the soils (Nawaz et al., 2011). It determines the acid or base neutralizing ability of soils or buffering capacity of soil (ability of soil to resist a change in ph). Buffering mechanisms are made up of mineral and organic constituents of soils such as primary minerals (e.g. carbonate), clay and organic matter content. These mixtures of mineral and organic constituents of soils determine the capacity of the soil to resist a change in soil ph. Different soils have different composition of primary minerals, clay and organic matter content due to differences in parent materials, climate, vegetation, cropping history, time etc with attendant differences in buffering mechanisms, and different buffering capacities (Spark, 2003). In alkaline soils (ph above 7), carbonate minerals such as CaCO 3 and Na 2 CO 3 are materials involved in neutralization of acid deposition. The ability of the alkaline soil to buffer depends on the amount of these minerals present in the soil. In acid soils (ph below 5), aluminium take over the buffering role. Aluminum compound (e.g. Al(OH) 3 ) react with hydrogen ions to release aluminium ion. In other words, for every aluminium ion, three hydrogen ions can be consumed, thus ph changes at a very slow rate in comparison to the acidification reactions. In neutral to moderately acid soils (ph 5-7), the ability of the soil to buffer depends on ions exchange reactions associated with clay and organic matter. The added acid cations (H + and Al 3+ ) exchange with base cations from clay or humus exchange sites. The exchangeable base cation buffering mechanism does not actually neutralize the acidity but merely stores it in the soil reserve acidity pool (Merry, 2001). The higher the organic matter and layer silicate clay content, the higher the buffering capacity. Different soils have different degrees of responses to acid deposition (soil sensitivity to acidification) due to variation in materials involved in the neutralization process. This also account for variation in soil vulnerability to acid deposition. The most significant soil properties responsible for soil sensitivity to acidification and neutralization of acid deposition are cation exchange capacity (CEC) and base saturation (Nawaz et al., 2011). Kuylenstiema et al. (2001) used cation exchange capacity and base saturation in the determination of soil sensitivity and vulnerability to acid deposition in selected soils of Northeast region of Thailand. They observed that soils with high cation exchange capacity and high base saturation were insensitive to basic cations depletion, while soils with low cation exchange capacity and moderate base saturation were sensitive to basic cations depletion. They also observed that basic cations were not depleted in soils with very low base saturation irrespective of CEC. Soils with CEC between 0-3 cmol kg -1, base saturation is less meaningful agronomically. This is because the holding power of the soil is so low that even insignificant amount of basic cations will result in a relatively high saturation (Kuylenstiema et al., 2001). Adverse changes in soil ph can affect crop growth due to variety of reasons. Soil acidification caused a decrease in soil ph of the surface soil by replacing basic cations (Ca 2+, Mg 2+ and K + ) on soil exchange sites with hydrogen ions and the basic cations may be lost through leaching. Soil acidification also brings about upsurge in the concentrations of Al, Cu, Fe, Zn, B, Mn, Cr, and Ni up to the levels that may be phyto-toxic and reduces the availability of P through fixation. NJAFE VOL. 11 No. 3,
2 When acid rain falls on soils that cannot neutralize the acidity, surface waters become more acidic. The acid disrupt the productive cycle of fish, harms aquatic plants and micro-organisms, cause decline and dieback of forest trees and accelerates the rusting of corrugated iron sheet roofs (Stevens et al., 2009). Hence, the objectives of this study were to identify and classify the different buffering mechanisms and soil sensitivity to acid deposition, based on parent materials in Akwa Ibom State, Nigeria. MATERIALS AND METHODS Description of the study area Akwa Ibom State is located in the southeastern Nigeria and enclosed within latitudes 4 30 ' and 5 30 ' N and longitudes 7 30 ' and 8 20 ' E. The State is underlain mainly by coastal plain sands, beach ridge sands, sandstone / shale and alluvial parent materials (Petters et al., 1989). Physiographically, the landscape comprises of a low plain and riverine area with almost no portion of the State exceeding 175 m above sea level. The climate is humid tropical with annual rainfall varying from 3000 mm along the coast to about 2250 mm at the extreme north with 1-3 dry months in the year. Mean annual temperature varies between C with relative humidity of %. Field work Four parent materials were selected for the study. In each parent material, three profile pits were sunk at representative locations. The parent materials were coastal plain sand, river alluvium, beach ridge sands and sandstone/shale. A total of 12 profile pits were sited and soil samples were collected based on genetic horizons for laboratory analysis. Laboratory analysis The samples were air dried, pulverized and sieved with a 2 mm sized sieve. The samples were analyzed in the laboratory for the following properties: Particle size distribution was determined by the hydrometer method as described by Gee and Bauder (1986). Soil ph was determined in water 1:2.5 soils: water ratio using ph meter with glass electrode (Thomas, 1996). Exchangeable cations- the bases were extracted with neutral NH 4 OAC. Calcium and magnesium were determined in the extract by EDTA titration and potassium and sodium by the use of flame photometer (Udo et al., 2009). Exchangeable acidity was extracted with one normal potassium chloride solution. The exchangeable acidity and the exchangeable aluminium were determined by titration as described by Thomas (1996). The exchangeable hydrogen was obtained by subtracting exchangeable aluminium from exchangeable acidity. (Exchangeable acidity (Al +H) Exchangeable Al = Exchangeable H). Organic carbon was determined by the dichromate wet-oxidation method as described by Nelson and Sommers (1996). The values were multiplied by to obtained organic matter content. Available phosphorus was determined by the Bray-1 method as described by Kuo (1996). Total nitrogen was determined by the micro-kjeldahl digestion and distillation method as described by Bremner (1996). Cation exchange capacity was determined by method described by Sumner and Miller (1996). Percentage base saturation was calculated using the formula: summation of exchangeable bases / CEC 100 Rating of Soil Buffering Mechanisms and sensitivity to acid deposition Soil buffering mechanism and sensitivity to acid deposition of the study area were determined by comparing the soil ph, base saturation and cation exchange capacity of the study area with the soil buffering mechanism and sensitivity to acid deposition rating recommended by Nawaz et al. (2010) as shown in Tables 1 and 2. Table 1: Rating of soil buffering mechanisms Descriptive terms Class Soil ph range Buffering mechanism Extremely acid 1 <4.5 Iron range Very strongly acid Aluminium / iron range Strongly acid Aluminium range Moderately acid 1V Cation exchange Slightly acid to neutral V Silicate buffers Slightly alkaline V Silicate buffers Source: (Nawaz et al., 2010) RESULTS AND DISCUSSION Physical and chemical properties of soils of the study Area Soils of beach ridge sand parent material The minimum, maximum and mean of soil properties of beach ridge sand parent material are presented in Table 3.The sand fraction varies from 89.9 to 95.9 %, silt varies from 0.06 to 3.9 % while clay varies from 3.9 to 8.2 %, indicating sandy texture throughout the profile. The soil ph varied from 3.2 to 5.4, indicating the range of extremely to strongly acid. Organic matter varied from 1.0 to 4.6 % indicating low to high throughout the profile. NJAFE VOL. 11 No. 3,
3 Total N varies from 0.04 to 0.1%, indicating very low to moderate. Available P varied from low to moderate (3.3 to 11.9 mg kg -1 ) throughout the profile. Exchangeable Ca ( cmol kg -1 ), Mg ( cmol kg -1 ) K ( cmokg -1 ) and Na ( cmol kg -1 ) were low throughout the profile. Cation exchange capacity was very low ( cmol kg -1 ) throughout the profile. Base saturation was moderate throughout the profile ( %). Table 2: Rating of soil sensitivity to acid deposition Base saturation Cation Exchange capacity (CEC) (cmol kg -1 ) (%) < > I II II I II III II III IV III IV V V V V Source: (Nawaz et al., 2010) Key: Class I: Very high sensitivity or vulnerability II: high sensitivity or vulnerability sensitivity or vulnerability IV: Low sensitivity or vulnerability V: Very low sensitivity or vulnerability III: moderate Table 3: The mean, minimum and maximum of soil properties of beach ridge sand parent material Sand % Silt % Clay % ph Organic matter % Total nitrogen % Available phosphorus mg kg Exchangeable calcium cmol kg Exchangeable. magnesium cmol kg Exchangeable. sodium cmol kg Exchangeable potassium cmol kg Exchangeable Acidity cmol kg Effective cation exchange capacity cmol kg Base saturation % Soil of coastal plain sand parent material The minimum, maximum and mean of soil properties of coastal plain sand parent material are presented in Table 4. The sand fraction varies from 60.0 to 94.6 %, silt varies from 1.8 to 18.0 % while clay varies from 4.2 to 26.0 %, indicating sandy loam to sand texture. Soil ph varied from 4.9 to 6.5, indicating strongly to slightly acid throughout the profile. Organic matter varied from low to very high ( %). Total N varied from very low to medium ( %). Available P varied from low to high ( mg kg -1 ) throughout the profile. Exchangeable Ca ( cmol kg -1 ), Na ( cmol kg -1 ) and K ( cmol kg -1 ) were low. Exchangeable Mg ( cmol kg -1 ) varied from low to moderate. Cation exchange capacity was low throughout the profile ( cmol kg -1 ). Base saturation varied from moderate to high throughout the profile ( %). Soils of sandstone and shale parent material The minimum, maximum and mean of soil properties of sandstone and shale parent material are presented in Table 5. The sand fraction varies from 50.0 to 90.0 %, silt varies from % while the clay varies from 7.0 to 40.0%, indicating sandy clay to sand. Soil ph varied from 4.0 to 5.6, indicating very strongly acid to moderately acid throughout the profile. Organic matter varied from low to moderate ( %). Total N varied from very low to moderate ( %) throughout the profile. Available P varied from moderate to high ( mg kg -1 ). Exchangeable Ca ( cmol kg -1 ), Na ( cmol kg -1 ) and K ( cmol kg -1 ) were low throughout the profile. Exchangeable Mg varied from low to moderate ( cmol kg -1 ). Cation exchange capacity was low throughout the profile ( cmol kg -1 ). Base saturation varied from low to high ( %). Soils of alluvial parent material The minimum, maximum and mean of soil properties of alluvial parent material are presented in Table 6. The sand fraction varies from %, silt varies from % while clay varies from %, NJAFE VOL. 11 No. 3,
4 indicating clay to sandy clay loam. Soil ph varied very strongly acid to moderately acid throughout the profile ( ). Organic matter varied from low to high ( %). Total N varied from low to moderate ( %). Available P varied from low to high ( mg kg -1 ). Exchangeable Ca ( cmol kg -1 ) and Mg ( cmol kg -1 ) varied from low to moderate throughout the profile. Exchangeable Na ( cmol kg -1 ) and K ( cmol kg -1 ) were low throughout the profile. Cation exchange capacity varied from low to high ( cmol kg -1 ). Base saturation varied from very low to high ( %). Table 4: The mean, minimum and maximum of soil properties of coastal plain sand parent material Sand % Silt % Clay % ph Organic matter % Total nitrogen % Available phosphorus mg kg Exchangeable calcium cmol kg Exchangeable. magnesium cmol kg Exchangeable. sodium cmol kg Exchangeable potassium cmol kg Exchangeable Acidity cmol kg Effective cation exchange capacity cmol kg Base saturation % Table 5: The mean, minimum and maximum of soil properties of sandstone and shale parent material Sand % Silt % Clay % ph Organic matter % Total nitrogen % Available phosphorus mg kg Exchangeable calcium cmol kg Exchangeable. magnesium cmol kg Exchangeable. sodium cmol kg Exchangeable potassium cmol kg Exchangeable Acidity cmol kg Effective cation exchange capacity cmol kg Base saturation % Buffering mechanisms of soils of the study area The buffering mechanisms of soils of the study area are presented in Table 7. The ph values of soils developed from sandstone and shale and river alluvium parent materials fall within the ph range of 4.6 to 5.7 (very strongly to moderately acid). This range falls within class II- IV of the buffering mechanism rating (Table 1). This is an indication that aluminium, iron and cation exchange were the major buffering mechanism in these soils. In beach ridge sand parent material, the soil ph falls within the range of 3.2 to 5.4 (extremely to strongly acid). This range falls within class I-II. This is an indication that iron and aluminium were the major buffering mechanisms in these soils. In coastal plain sand soils, the soil ph falls within the range of 5.3 to 6.5 (strongly to slightly acid). This range falls within class III, IV and V. This is an indication that aluminium, cation exchange of kaolinitic silicate clay and organic matter were the major buffering mechanisms in these soils. The implication is that when acid is added to the soils of the study area, there will be an upsurge in the concentrations of Al, Cu, Fe, Zn, B, Mn, Cr, Ni and reduction in the availability of P through fixation. The degree of the upsurge of these elements will be highest in soils of beach ridge sand, followed by soils of alluvium, sandstone and shale and followed by soils of coastal plain sand. The trend is as followed: soils of beach ridge sand soil > soils of river alluvium = soils of sandstone and shale > soils of coastal plain sand. Soil sensitivity to acidification NJAFE VOL. 11 No. 3,
5 Based on the rating of soil sensitivity to acidification of Nawaz et al. (2010) shown in Table 2. In beach ridge sand soils, CEC was low while base saturation was moderate, indicating high sensitivity or high vulnerability to acid deposition. In coastal plain sands soils, CEC was low while base saturation varied from moderate to high, indicating moderate sensitivity or moderate vulnerability to acid deposition. Cation exchange capacity of sandstone and shale soils was low while base saturation was moderate, indicating high sensitivity or high vulnerability. In soils developed from river alluvium, CEC and base saturation varied from low to high, indicating low to high sensitivity or vulnerability depending on the composition of the depositional materials that formed the soil. The trend in soil sensitivity to acidification in the study area based on the rating was as followed: beach ridge sand > sandstone and shale > coastal plain sand. The river alluvium depends on the composition of the depositional materials that formed the soil. The high sensitivity or vulnerability to acid deposition of beach ridge sands could be attributed to the high content of oxides of Fe and Al and low clay and organic matter content (exchange sites). This is in agreement with Kuylenstiema et al, (2001) that soils with low cation exchange capacity and moderate base saturation were sensitive to base cation depletion. The moderate sensitivity to acidification of coastal plain sand and river alluvium could also be attributed to cation exchange of kaolinitic silicate clay and organic matter being the major buffering mechanisms. But the higher the organic matter and layer silicate clay content, the higher the buffering capacity and the lower the sensitivity to acidification. Table 6: The mean, minimum and maximum of soil properties of alluvial parent material Sand % Silt % Clay % ph Organic matter % Total nitrogen % Available phosphorus mg kg Exchangeable calcium cmol kg Exchangeable. magnesium cmol kg Exchangeable. sodium cmol kg Exchangeable potassium cmol kg Exchangeable Acidity cmol kg Effective cation exchange capacity cmol kg Base saturation % Table 7: Buffering mechanisms of soils of the study area Parent materials Soil ph Buffering mechanism Beach ridge sand Iron / aluminium Coastal plain sand Aluminium/cation exchange of silicate clay and organic matter Sandstone and shale Aluminium /cation exchange of silicate clay Alluvium Aluminium /cation exchange of silicate clay CONCLUSION The results of the rating of buffering mechanisms showed that iron and aluminium were the major buffering mechanisms in beach ridge sands while cation exchange and aluminium were the major buffering mechanisms in sandstone and shale and river alluvium soils. Also, aluminium and cation exchange were the major buffering mechanisms in coastal plain sand soils. The study also showed that beach ridge sand and sandstone and shale soils were highly sensitive to acidification. Coastal plain sands was moderately sensitive while soils of river alluvium varied from low to high in sensitivity to acid deposition depending on the composition of the depositional materials. REFERENCES Bremner, J. M Total Nitrogen. In: Spark, D. L. (eds.). Methods of Soil Analysis Part 3-Chemical Methods, SSSA Book Series 5, Madison, Wisconsin, USA. pp FAO Guidelines for Soil Descriptions, 3 rd edition. FAO Rome. Gee, G. W. and Bauder, J. W Particle size analysis. In: Methods of Soil Analysis, Part 1-Physical and Mineralogical Methods, SSSA Book Seria 5, Madison, Wisconsin, USA. pp NJAFE VOL. 11 No. 3,
6 Kuo, S Phosphorus. In: Spark, D. L. (eds.). Methods of Soil Analysis Part 3. Chemical Methods, SSSA Book Series 5, Madison, Wisconsin, USA. 869pp Kuylenstiema, J. C. I., Rode, H., Cinderby, S. and Hicks, K Acidification in developing countries; ecosystem sensitivity and the critical load approach on a global scale. Ambio, 30(1): Merry, R. H Acidity and alkalinity of soils. Environmental and Ecological Chemistry, Vol. 11. Encyclopaedia of Life Support Systems. pp Nawaz, R., Tripathi, N. K. and Parkpian, P Soil acidity ranking, soil sensitivity and vulnerability to acid deposition in the Northeast region of Thailand. Biology, Environment and Chemistry. Vol.1, IACSIT Press, Singapore. Nelson, D. M. and Sommers, L. E Total Carbon, Organic Carbon and Organic Matter. In:.Spark, D. L (eds.). Methods of Soil Analysis Part 3- Chemical Methods, SSSA Book Series 5, Madison, Wisconsin, USA. pp Sparks, D. L Environmental Soil Chemistry, 2 nd edition. Academic Press, Elsevier Science, Amsterdam. Petters, S. W., Usoro, E. J., Udo, E. J., Obot, U. W. and Obot, S. N Akwa Ibom State. Physical Background, Soils and Landuse and Ecological Problems.Technical Report of the Taskforce on Soils and Landuse. Govt. Printer, Uyo. 602pp. Stevens, C. J., Dis, N. B. and Gowing, D. J Regional trends in soil acidification and exchangeable metal concentration in relation to acid deposition rates. Enviro. Pollu. Vol.157. pp Sumner, M. E. and Miller, W. P Cation Exchange Capacity and Exchangeable Coefficients. In: Spark, D. L. (eds.). Methods of Soil Analysis Part 3-Chemical Methods, SSSA Book Series 5, Madison, Wisconsin, USA. pp Thomas, G. W Soil ph and Soil Acidity. In: Sparks, D. L. (eds.). Methods of Soil Analysis Part 3- Chemical Methods, SSSA Book Series 5, Madison, Wisconsin, USA. pp Udo, E. J., Ibia, T. O., Ogunwale, J. A., Ano, A. O. and Esu, I. E Manual of Soil, Plant and Water Analysis. Sibon Books Limited, Lagos.178pp. NJAFE VOL. 11 No. 3,
INFLUENCE OF PARENT MATERIAL ON ORGANO-MINERAL ASSOCIATION AND SORPTION CAPACITY OF SOILS OF AKWA IBOM STATE, NIGERIA
 International Journal of Innovative Agriculture & Biology Research 3(3):9-7, July-Sept. 5 SEAHI PUBLICATIONS, 5 www.seahipaj.org ISSN:354-934 INFLUENCE OF PARENT MATERIAL ON ORGANO-MINERAL ASSOCIATION
International Journal of Innovative Agriculture & Biology Research 3(3):9-7, July-Sept. 5 SEAHI PUBLICATIONS, 5 www.seahipaj.org ISSN:354-934 INFLUENCE OF PARENT MATERIAL ON ORGANO-MINERAL ASSOCIATION
Acid Soil. Soil Acidity and ph
 Acid Soil Soil Acidity and ph ph ph = - log (H + ) H 2 O H + + OH - (H + ) x (OH - )= K w = 10-14 measures H + activity with an electrode (in the lab), solutions (in the field) reflects the acid intensity,
Acid Soil Soil Acidity and ph ph ph = - log (H + ) H 2 O H + + OH - (H + ) x (OH - )= K w = 10-14 measures H + activity with an electrode (in the lab), solutions (in the field) reflects the acid intensity,
CLASS EXERCISE 5.1 List processes occurring in soils that cause changes in the levels of ions.
 5 SIL CHEMISTRY 5.1 Introduction A knowledge of the chemical composition of a soil is less useful than a knowledge of its component minerals and organic materials. These dictate the reactions that occur
5 SIL CHEMISTRY 5.1 Introduction A knowledge of the chemical composition of a soil is less useful than a knowledge of its component minerals and organic materials. These dictate the reactions that occur
Soil Fertility. Fundamentals of Nutrient Management June 1, Patricia Steinhilber
 Soil Fertility Fundamentals of Nutrient Management June 1, 2010 Patricia Steinhilber Ag Nutrient Management Program University of Maryland College Park Main Topics plant nutrition functional soil model
Soil Fertility Fundamentals of Nutrient Management June 1, 2010 Patricia Steinhilber Ag Nutrient Management Program University of Maryland College Park Main Topics plant nutrition functional soil model
Adsorption of ions Ion exchange CEC& AEC Factors influencing ion
 Adsorption of ions Ion exchange CEC& AEC Factors influencing ion exchange- Significance. Adsorption of ions Ion adsorption and subsequent exchange are important processes that take place between soil colloidal
Adsorption of ions Ion exchange CEC& AEC Factors influencing ion exchange- Significance. Adsorption of ions Ion adsorption and subsequent exchange are important processes that take place between soil colloidal
Be sure to show all calculations so that you can receive partial credit for your work!
 Agronomy 365T Exam 1 Spring 2004 Exam Score: Name TA Lab Hour Be sure to show all calculations so that you can receive partial credit for your work! 1) List 14 of the plant essential nutrient for plant
Agronomy 365T Exam 1 Spring 2004 Exam Score: Name TA Lab Hour Be sure to show all calculations so that you can receive partial credit for your work! 1) List 14 of the plant essential nutrient for plant
Cation Exchange Capacity, CEC
 Cation Exchange Capacity, CEC The basic building blocks of clay minerals are: silicon atoms surrounded by four oxygen atoms (tetrahedra), and aluminium atoms surrounded by six hydroxide groups (dioctahedra),
Cation Exchange Capacity, CEC The basic building blocks of clay minerals are: silicon atoms surrounded by four oxygen atoms (tetrahedra), and aluminium atoms surrounded by six hydroxide groups (dioctahedra),
FOR Soil Quality Report 2017
 Student Name: Partner Name: Laboratory Date: FOR 2505 - Soil Quality Report 2017 Objectives of this report: 10 Marks Lab Objectives Section Principles behind methods used to determine soil base cation
Student Name: Partner Name: Laboratory Date: FOR 2505 - Soil Quality Report 2017 Objectives of this report: 10 Marks Lab Objectives Section Principles behind methods used to determine soil base cation
Effect of integrated nutrient management practices on acidity and nutrient availability in acid soil
 207; 5(4): 678-682 P-ISSN: 2349 8528 E-ISSN: 232 4902 IJCS 207; 5(4): 678-682 207 JEZS Received: 03-05-207 Accepted: 04-06-207 Tupaki Lokya Agriculture Chemistry, OUAT, J Ravinder Agriculture Chemistry,
207; 5(4): 678-682 P-ISSN: 2349 8528 E-ISSN: 232 4902 IJCS 207; 5(4): 678-682 207 JEZS Received: 03-05-207 Accepted: 04-06-207 Tupaki Lokya Agriculture Chemistry, OUAT, J Ravinder Agriculture Chemistry,
Only healthy soil can grow a nutrient dense food. You are what you eat!
 Understanding How Cation Nutrients & Soil Structure are Related By Michael Martin Meléndrez Only healthy soil can grow a nutrient dense food. You are what you eat! Soil Must be able to hold onto water,
Understanding How Cation Nutrients & Soil Structure are Related By Michael Martin Meléndrez Only healthy soil can grow a nutrient dense food. You are what you eat! Soil Must be able to hold onto water,
Effect of Heat Treatment on Phosphate Sorption by Soils from Different Ecologies
 Effect of Heat Treatment on Phosphate Sorption by Soils from Different Ecologies 1* Aghedo, J.E., 2 Ukpebor, E. and 3 Oviasogie, P.O., 4 Omorogbe, S.O. 1Department of Chemistry, College of Education, Ekiadolor,
Effect of Heat Treatment on Phosphate Sorption by Soils from Different Ecologies 1* Aghedo, J.E., 2 Ukpebor, E. and 3 Oviasogie, P.O., 4 Omorogbe, S.O. 1Department of Chemistry, College of Education, Ekiadolor,
Volume Composition of a Desirable Surface Soil
 Soil Chemistry Volume Composition of a Desirable Surface Soil 50% pore space 25% air 45 to 48% mineral matter 50% solid material 25% water 2 to 5% organic matter Soil Organic Matter Soil organic matter:
Soil Chemistry Volume Composition of a Desirable Surface Soil 50% pore space 25% air 45 to 48% mineral matter 50% solid material 25% water 2 to 5% organic matter Soil Organic Matter Soil organic matter:
Gain a better understanding of soil ph and how it is measured. Understand how lime requirement is determined.
 LABORATORY 7 SOIL REACTION (ph) AND LIME REQUIREMENT I Objectives Gain a better understanding of soil ph and how it is measured. Understand how lime requirement is determined. II Introduction A Soil Reaction
LABORATORY 7 SOIL REACTION (ph) AND LIME REQUIREMENT I Objectives Gain a better understanding of soil ph and how it is measured. Understand how lime requirement is determined. II Introduction A Soil Reaction
Comparison of Selected Extractants for the Estimation of Available K in Soils of Edo State of Central Southern Nigeria
 Tropical Agricultural Research & Extension 10, 2007 Comparison of Selected Extractants for the Estimation of Available K in Soils of Edo State of Central Southern Nigeria I.O. Osemwota Department of Soil
Tropical Agricultural Research & Extension 10, 2007 Comparison of Selected Extractants for the Estimation of Available K in Soils of Edo State of Central Southern Nigeria I.O. Osemwota Department of Soil
Chunmei Chen A,B and Donald L Sparks A. Delaware, Newark, DE 19711, USA.
 Environ. Chem. 2015, 12, 64 CSIRO 2015 Supplementary material Multi-elemental scanning transmission X-ray microscopy near edge X-ray absorption fine structure spectroscopy assessment of organo mineral
Environ. Chem. 2015, 12, 64 CSIRO 2015 Supplementary material Multi-elemental scanning transmission X-ray microscopy near edge X-ray absorption fine structure spectroscopy assessment of organo mineral
FORMS AND DISTRIBUTION OF POTASSIUM IN SOILS UNDERLAIN BY THREE LITHOLOGIES SOUTHEASTERN NIGERIA
 FORMS AND DISTRIBUTION OF POTASSIUM IN SOILS UNDERLAIN BY THREE LITHOLOGIES SOUTHEASTERN NIGERIA B. N. Ndukwu, M. C. Chukwuma, C. M.Idigbor, S. N. Obasi Department of Soil Science and Technology Federal
FORMS AND DISTRIBUTION OF POTASSIUM IN SOILS UNDERLAIN BY THREE LITHOLOGIES SOUTHEASTERN NIGERIA B. N. Ndukwu, M. C. Chukwuma, C. M.Idigbor, S. N. Obasi Department of Soil Science and Technology Federal
The future of the Lowland Belizean Savannas?.
 The future of the Lowland Belizean Savannas?. Using cluster analysis to explore multivariate spatial patterns in savanna soils PETER FURLEY & SARAH BEADLE UK Belize association 15 th November 2014 Outline
The future of the Lowland Belizean Savannas?. Using cluster analysis to explore multivariate spatial patterns in savanna soils PETER FURLEY & SARAH BEADLE UK Belize association 15 th November 2014 Outline
Chapter 6 9/25/2012. Weathering, Erosion and Soils. Introduction. How Are Earth Materials Altered? Introduction. How Are Earth Materials Altered?
 Chapter 6 Introduction Rocks and minerals are disintegrated and decomposed by the processes of mechanical and chemical weathering. Weathering, Erosion and Soils This breakdown occurs because the parent
Chapter 6 Introduction Rocks and minerals are disintegrated and decomposed by the processes of mechanical and chemical weathering. Weathering, Erosion and Soils This breakdown occurs because the parent
Soil Colloidal Chemistry. Compiled and Edited by Dr. Syed Ismail, Marthwada Agril. University Parbhani,MS, India
 Soil Colloidal Chemistry Compiled and Edited by Dr. Syed Ismail, Marthwada Agril. University Parbhani,MS, India 1 The Colloidal Fraction Introduction What is a colloid? Why this is important in understanding
Soil Colloidal Chemistry Compiled and Edited by Dr. Syed Ismail, Marthwada Agril. University Parbhani,MS, India 1 The Colloidal Fraction Introduction What is a colloid? Why this is important in understanding
CHEMICAL, PHYSICAL, AND MINERALOGICAL PROPERTIES OF CERTAIN SOIL PROFILES IN THE LOWER MISSISSIPPI DELTA B. N. DRISKELL ABSTRACT
 CHEMICAL, PHYSICAL, AND MINERALOGICAL PROPERTIES OF CERTAIN SOIL PROFILES IN THE LOWER MISSISSIPPI DELTA B. N. DRISKELL Louisiana State University ABSTRACT The soils of the lower Mississippi Delta are
CHEMICAL, PHYSICAL, AND MINERALOGICAL PROPERTIES OF CERTAIN SOIL PROFILES IN THE LOWER MISSISSIPPI DELTA B. N. DRISKELL Louisiana State University ABSTRACT The soils of the lower Mississippi Delta are
Copyright SOIL STRUCTURE and CLAY MINERALS
 SOIL STRUCTURE and CLAY MINERALS Soil Structure Structure of a soil may be defined as the mode of arrangement of soil grains relative to each other and the forces acting between them to hold them in their
SOIL STRUCTURE and CLAY MINERALS Soil Structure Structure of a soil may be defined as the mode of arrangement of soil grains relative to each other and the forces acting between them to hold them in their
H + OH - H + OH - H +OH -
 Objectives SOIL CHEMISTRY Reading Materials: Brady NC, 1990. The Nature and Properties of Soils Supardi, 1989. Sifat dan Ciri Tanah Soil ph Cation exchange o Cation Exchange Capacity (CEC) o Base saturation
Objectives SOIL CHEMISTRY Reading Materials: Brady NC, 1990. The Nature and Properties of Soils Supardi, 1989. Sifat dan Ciri Tanah Soil ph Cation exchange o Cation Exchange Capacity (CEC) o Base saturation
Lecture 15: Adsorption; Soil Acidity
 Lecture 15: Adsorption; Soil Acidity Surface Complexation (Your textbook calls this adsorption ) Surface Complexation Both cations and anions can bind to sites on the external surfaces of soil minerals
Lecture 15: Adsorption; Soil Acidity Surface Complexation (Your textbook calls this adsorption ) Surface Complexation Both cations and anions can bind to sites on the external surfaces of soil minerals
Soil - the battery of your business
 Soil - the battery of your business By DAVID HANLON The basis of our farm business is the soil and increasingly all growers are focusing their attention on this vital resource. Just what state it is in
Soil - the battery of your business By DAVID HANLON The basis of our farm business is the soil and increasingly all growers are focusing their attention on this vital resource. Just what state it is in
Soil ph: Review of Concepts
 Soils and Water, Spring 008 Soil ph: Review of Concepts Acid: substance that can donate a proton Base: substance that can accept a proton HA H A HA and A - are called conjugate acid-base pairs. The strength
Soils and Water, Spring 008 Soil ph: Review of Concepts Acid: substance that can donate a proton Base: substance that can accept a proton HA H A HA and A - are called conjugate acid-base pairs. The strength
Mehlich and Modified Mehlich Buffer for Lime Requirement
 Mehlich and Modified Mehlich Buffer for Lime Requirement David H. Hardy Application and Principle The development of the Mehlich buffer was to estimate acidity actually affecting plant growth in North
Mehlich and Modified Mehlich Buffer for Lime Requirement David H. Hardy Application and Principle The development of the Mehlich buffer was to estimate acidity actually affecting plant growth in North
Agricultural Fertilizer Applicator Soil Test Results What the heck do they mean?
 Agricultural Fertilizer Applicator Soil Test Results What the heck do they mean? Northeast Ohio Pesticide Safety Education Program Lee Beers, OSU Extension Trumbull County beers.66@osu.edu 330-638-6783
Agricultural Fertilizer Applicator Soil Test Results What the heck do they mean? Northeast Ohio Pesticide Safety Education Program Lee Beers, OSU Extension Trumbull County beers.66@osu.edu 330-638-6783
Overview. Rock weathering Functions of soil Soil forming factors Soil properties
 UN-FAO A. Healthy soils are the basis for healthy food production. B. A tablespoon of normal topsoil has more microorganisms than the entire human population on Earth. C. It can take up to 1,000 years
UN-FAO A. Healthy soils are the basis for healthy food production. B. A tablespoon of normal topsoil has more microorganisms than the entire human population on Earth. C. It can take up to 1,000 years
Weathering, Soil, and Mass Movements
 Tarbuck Lutgens Weathering, Soil, and Mass Movements 5.1 Weathering Mechanical Weathering Mechanical weathering occurs when physical forces break rock into smaller and smaller pieces without changing the
Tarbuck Lutgens Weathering, Soil, and Mass Movements 5.1 Weathering Mechanical Weathering Mechanical weathering occurs when physical forces break rock into smaller and smaller pieces without changing the
Soils and Soil Minerals. Remember, most things can be too little or too much.
 Soils and Soil Minerals Remember, most things can be too little or too much. 1 2 3 Source of essential elements CO 2, O 2 from atmosphere H 2 0, O 2, minerals from soil NH 4, SO 4 can volatilize and be
Soils and Soil Minerals Remember, most things can be too little or too much. 1 2 3 Source of essential elements CO 2, O 2 from atmosphere H 2 0, O 2, minerals from soil NH 4, SO 4 can volatilize and be
Lecture 6: Soil Profiles: Diagnostic Horizons
 Lecture 6: Soil Profiles: Diagnostic Horizons Complexity in Soil Profiles Soil Horizons Soils display distinct layering O Horizon: Partially decomposed organic matter (OM) A Horizon: Near surface, mineral
Lecture 6: Soil Profiles: Diagnostic Horizons Complexity in Soil Profiles Soil Horizons Soils display distinct layering O Horizon: Partially decomposed organic matter (OM) A Horizon: Near surface, mineral
Unit E: Basic Principles of Soil Science. Lesson 2: Understanding Soil Formation
 Unit E: Basic Principles of Soil Science Lesson 2: Understanding Soil Formation Important Terms Alluvium Bedrock Chemical weathering Climate Glacial till Loess Native vegetation Organic matter Outwash
Unit E: Basic Principles of Soil Science Lesson 2: Understanding Soil Formation Important Terms Alluvium Bedrock Chemical weathering Climate Glacial till Loess Native vegetation Organic matter Outwash
Chapter 6. Weathering, Erosion, and Soil
 Chapter 6 Weathering, Erosion, and Soil Introduction Rocks and minerals disintegrate and decompose by the processes of physical and chemical weathering. This breakdown occurs because the parent material
Chapter 6 Weathering, Erosion, and Soil Introduction Rocks and minerals disintegrate and decompose by the processes of physical and chemical weathering. This breakdown occurs because the parent material
Soils. Source: Schroeder and Blum, 1992
 Soils Source: Schroeder and Blum, 1992 Literature cited: Schroeder, D. and Blum, W.E.H. 1992. Bodenkunde in Stichworten. Gebrüder Borntraeger, D-1000 Berlin. Geology and Life Conceptual model Source: Knight,
Soils Source: Schroeder and Blum, 1992 Literature cited: Schroeder, D. and Blum, W.E.H. 1992. Bodenkunde in Stichworten. Gebrüder Borntraeger, D-1000 Berlin. Geology and Life Conceptual model Source: Knight,
Running head: TITLE 1. Lab Report. Name. Academic Institution. Author Note. Class. Professor
 Running head: TITLE 1 Lab Report Name Academic Institution Author Note Class Professor March 13, 2018 TITLE 2 Soil colloids tend to have a net negative charge for two reasons: surface area and ph levels.
Running head: TITLE 1 Lab Report Name Academic Institution Author Note Class Professor March 13, 2018 TITLE 2 Soil colloids tend to have a net negative charge for two reasons: surface area and ph levels.
C2.6 Quantitative Chemistry Foundation
 C2.6 Quantitative Chemistry Foundation 1. Relative masses Use the periodic table to find the relative masses of the elements below. (Hint: The top number in each element box) Hydrogen Carbon Nitrogen Oxygen
C2.6 Quantitative Chemistry Foundation 1. Relative masses Use the periodic table to find the relative masses of the elements below. (Hint: The top number in each element box) Hydrogen Carbon Nitrogen Oxygen
ph and Liming Practices Kent Martin Stafford County 1/5/2010
 ph and Liming Practices Kent Martin Stafford County 1/5/2010 Outline What is ph Normal ph ranges Acid Soil Importance of soil ph Factors affecting soil ph Acid types and measurement Neutralizing value
ph and Liming Practices Kent Martin Stafford County 1/5/2010 Outline What is ph Normal ph ranges Acid Soil Importance of soil ph Factors affecting soil ph Acid types and measurement Neutralizing value
The Earth s Layers. Convection and Hot Spots. The Earth s Layers. The Earth s resources were determined when the planet formed.
 The Earth s resources were determined when the planet formed. Chapter 8 Earth Systems and Resources The Earth s Layers Core- the innermost zone of the planet made of nickel and iron. Mantle- above the
The Earth s resources were determined when the planet formed. Chapter 8 Earth Systems and Resources The Earth s Layers Core- the innermost zone of the planet made of nickel and iron. Mantle- above the
Soil physical and chemical properties the analogy lecture. Beth Guertal Auburn University, AL
 Soil physical and chemical properties the analogy lecture. Beth Guertal Auburn University, AL Soil Physical Properties Porosity Pore size and pore size distribution Water holding capacity Bulk density
Soil physical and chemical properties the analogy lecture. Beth Guertal Auburn University, AL Soil Physical Properties Porosity Pore size and pore size distribution Water holding capacity Bulk density
List of Equipment, Tools, Supplies, and Facilities:
 Unit D: ph of Soil Lesson 2: Identifying ph Connection With Plant Growth Student Learning Objectives: Instruction in this lesson should result in the students achieving the following objectives: 1. Explain
Unit D: ph of Soil Lesson 2: Identifying ph Connection With Plant Growth Student Learning Objectives: Instruction in this lesson should result in the students achieving the following objectives: 1. Explain
Plant Nutrition and Transport. Chapter 29
 Plant Nutrition and Transport Chapter 29 Overview: Underground Plants The success of plants depends on their ability to gather and conserve resources from their environment. The transport of materials
Plant Nutrition and Transport Chapter 29 Overview: Underground Plants The success of plants depends on their ability to gather and conserve resources from their environment. The transport of materials
Engineering Geology ECIV 3302
 Engineering Geology ECIV 3302 Instructor : Dr. Jehad Hamad 2019-2018 Chapter (5) Weathering & Soil Chapter 5: Weathering, Soil, and Mass Wasting External processes include : (1) Weathering (2) Mass wasting
Engineering Geology ECIV 3302 Instructor : Dr. Jehad Hamad 2019-2018 Chapter (5) Weathering & Soil Chapter 5: Weathering, Soil, and Mass Wasting External processes include : (1) Weathering (2) Mass wasting
SoilGen2 model: data requirements and model output
 SoilGen2 model: data requirements and model output 1. Essential plot data... 1 2. Essential soil data... 2 3. Precipitation data (for a typical year)... 2 4. Evapotranspiration and Air temperature data
SoilGen2 model: data requirements and model output 1. Essential plot data... 1 2. Essential soil data... 2 3. Precipitation data (for a typical year)... 2 4. Evapotranspiration and Air temperature data
Rocks and Weathering
 Rocks and Weathering The Effects of Weathering The process of mountain building thrusts rock up to Earth s surface. Weathering is the process that breaks down rock and other substances at Earth s surface.
Rocks and Weathering The Effects of Weathering The process of mountain building thrusts rock up to Earth s surface. Weathering is the process that breaks down rock and other substances at Earth s surface.
Chapter 8 Earth Systems and Resources
 Chapter 8 Earth Systems and Resources Earth s resources were determined when the planet formed. The Earth s Crust Layers Core: innermost zone of the planet, largely nickel and iron. Mantle: above the core,
Chapter 8 Earth Systems and Resources Earth s resources were determined when the planet formed. The Earth s Crust Layers Core: innermost zone of the planet, largely nickel and iron. Mantle: above the core,
Introduction to Soil Science and Wetlands Kids at Wilderness Camp
 Introduction to Soil Science and Wetlands Kids at Wilderness Camp Presented by: Mr. Brian Oram, PG, PASEO B.F. Environmental Consultants http://www.bfenvironmental.com and Keystone Clean Water Team http://www.pacleanwater.org
Introduction to Soil Science and Wetlands Kids at Wilderness Camp Presented by: Mr. Brian Oram, PG, PASEO B.F. Environmental Consultants http://www.bfenvironmental.com and Keystone Clean Water Team http://www.pacleanwater.org
Biosphere. All living things, plants, animals, (even you!) are part of the zone of the earth called the biosphere.
 Unit 1 Study Guide Earth s Spheres Biosphere All living things, plants, animals, (even you!) are part of the zone of the earth called the biosphere. Hydrosphere Water covers ¾ of the earth, made up mostly
Unit 1 Study Guide Earth s Spheres Biosphere All living things, plants, animals, (even you!) are part of the zone of the earth called the biosphere. Hydrosphere Water covers ¾ of the earth, made up mostly
Lecture 13 More Surface Reactions on Mineral Surfaces. & Intro to Soil Formation and Chemistry
 Lecture 13 More Surface Reactions on Mineral Surfaces & Intro to Soil Formation and Chemistry 3. charge transfer (e.g., ligand/donor sorption): Sorption involves a number of related processes that all
Lecture 13 More Surface Reactions on Mineral Surfaces & Intro to Soil Formation and Chemistry 3. charge transfer (e.g., ligand/donor sorption): Sorption involves a number of related processes that all
Weathering The effects of the physical and chemical environment on the decomposition of rocks
 Weathering The effects of the physical and chemical environment on the decomposition of rocks - Igneous rocks form at high temperatures and the constituent minerals reflect the conditions of formation.
Weathering The effects of the physical and chemical environment on the decomposition of rocks - Igneous rocks form at high temperatures and the constituent minerals reflect the conditions of formation.
MINERAL CONTENT AND DISTRIBUTION AS INDEXES OF WEATHERING IN THE OMEGA AND AHMEEK SOILS OF NORTHERN WISCONSIN
 MINERAL CONTENT AND DISTRIBUTION AS INDEXES OF WEATHERING IN THE OMEGA AND AHMEEK SOILS OF NORTHERN WISCONSIN By L. D. WHITTIG 1 AND M. L. JACKSON University of Wisconsin, Madison, Wisconsin ABSTRACT Quantitative
MINERAL CONTENT AND DISTRIBUTION AS INDEXES OF WEATHERING IN THE OMEGA AND AHMEEK SOILS OF NORTHERN WISCONSIN By L. D. WHITTIG 1 AND M. L. JACKSON University of Wisconsin, Madison, Wisconsin ABSTRACT Quantitative
Chemical Weathering and Soils
 Chemical Weathering and Soils Fresh rocks and minerals that once occupied the outermost position reached their present condition of decay through a complex of interacting physical, chemical, and biological
Chemical Weathering and Soils Fresh rocks and minerals that once occupied the outermost position reached their present condition of decay through a complex of interacting physical, chemical, and biological
Lecture 14: Cation Exchange and Surface Charging
 Lecture 14: Cation Exchange and Surface Charging Cation Exchange Cation Exchange Reactions Swapping of cations between hydrated clay interlayers and the soil solution Also occurs on organic matter functional
Lecture 14: Cation Exchange and Surface Charging Cation Exchange Cation Exchange Reactions Swapping of cations between hydrated clay interlayers and the soil solution Also occurs on organic matter functional
Lab 8 Dynamic Soil Systems I: Soil ph and Liming
 Lab 8 Dynamic Soil Systems I: Soil ph and Liming Objectives: To measure soil ph and observe conditions which change ph To distinguish between active acidity (soil solution ph) and exchangeable acidity
Lab 8 Dynamic Soil Systems I: Soil ph and Liming Objectives: To measure soil ph and observe conditions which change ph To distinguish between active acidity (soil solution ph) and exchangeable acidity
C2.6 Quantitative Chemistry Foundation
 C2.6 Quantitative Chemistry Foundation 1. Relative masses Use the periodic table to find the relative masses of the elements below. (Hint: The top number in each element box) Hydrogen Carbon Nitrogen Oxygen
C2.6 Quantitative Chemistry Foundation 1. Relative masses Use the periodic table to find the relative masses of the elements below. (Hint: The top number in each element box) Hydrogen Carbon Nitrogen Oxygen
Igneous rocks + acid volatiles = sedimentary rocks + salty oceans
 The Lithosphere Weathering physical processes chemical processes biological processes weathering rates Soil development soil formation processes types of soils and vegetation soil properties physical chemical
The Lithosphere Weathering physical processes chemical processes biological processes weathering rates Soil development soil formation processes types of soils and vegetation soil properties physical chemical
Chemistry 265 December Exam 2011 Smith-Palmer
 1 Chemistry 265 December Exam 2011 Smith-Palmer NAME: [1] 1. Define an anode [1] Define a cathode [2] What is the E o for the following reaction: Ag + + Cu Ag (s) + Cu 2+ Ag + + e - Ag (s) E o = 0.799
1 Chemistry 265 December Exam 2011 Smith-Palmer NAME: [1] 1. Define an anode [1] Define a cathode [2] What is the E o for the following reaction: Ag + + Cu Ag (s) + Cu 2+ Ag + + e - Ag (s) E o = 0.799
Lecture 29: Soil Formation
 Lecture 29: Soil Formation Factors Controlling Soil Formation 1. Parent material: Soil precursor 2. Climate: Temperature and precipitation 3. Biota: Native vegetation, microbes, soil animals, humans 4.
Lecture 29: Soil Formation Factors Controlling Soil Formation 1. Parent material: Soil precursor 2. Climate: Temperature and precipitation 3. Biota: Native vegetation, microbes, soil animals, humans 4.
Surface Processes on the Earth. Rocks, Weathering, Erosion and Soil
 Surface Processes on the Earth Rocks, Weathering, Erosion and Soil ROCKS AND ROCK CYCLE Rock types Three main types of rock Igneous Metamorphic Sedimentary Igneous Form when magma or lava cools and hardens
Surface Processes on the Earth Rocks, Weathering, Erosion and Soil ROCKS AND ROCK CYCLE Rock types Three main types of rock Igneous Metamorphic Sedimentary Igneous Form when magma or lava cools and hardens
+ 3 can also be bound to the soil complex,
 Glossary Acidification: decrease in the buffer capacity, causing the ph to decrease. Additional scope: The additional scope shows the conditions whereby the habitat type cannot be permanently maintained
Glossary Acidification: decrease in the buffer capacity, causing the ph to decrease. Additional scope: The additional scope shows the conditions whereby the habitat type cannot be permanently maintained
Soil Formation. Lesson Plan: NRES B2-4
 Soil Formation Lesson Plan: NRES B2-4 1 Anticipated Problems 1. What are five different factors that affect soil formation? 2. What are some different types of parent material that affect soils? 3. What
Soil Formation Lesson Plan: NRES B2-4 1 Anticipated Problems 1. What are five different factors that affect soil formation? 2. What are some different types of parent material that affect soils? 3. What
Feedback between nutrient availability, NPP and N release
 Feedback between nutrient availability, NPP and N release 1 Redfield ratios A typical plant = 45% C, 1.5% N, 0.2%P or C:N = 30 : 1 and C:P = 225 : 1 or C:N:P = 225 : 7.5 : 1 N:P = 7.5 : 1 Mobility of nutrients
Feedback between nutrient availability, NPP and N release 1 Redfield ratios A typical plant = 45% C, 1.5% N, 0.2%P or C:N = 30 : 1 and C:P = 225 : 1 or C:N:P = 225 : 7.5 : 1 N:P = 7.5 : 1 Mobility of nutrients
CHAPTER 3 WATER AND THE FITNESS OF THE ENVIRONMENT. Section B: The Dissociation of Water Molecules
 CHAPTER 3 WATER AND THE FITNESS OF THE ENVIRONMENT Section B: The Dissociation of Water Molecules 1. Organisms are sensitive to changes in ph 2. Acid precipitation threatens the fitness of the environment
CHAPTER 3 WATER AND THE FITNESS OF THE ENVIRONMENT Section B: The Dissociation of Water Molecules 1. Organisms are sensitive to changes in ph 2. Acid precipitation threatens the fitness of the environment
Plant nutri0on. Soils and Soil Minerals. Based on: Remember, most things can be too little or too much. 1. O 2 2. CO 2 3. H 2 O 4. inorganic minerals
 Based on: Plant nutri0on Soils and Soil Minerals 1. O 2 2. CO 2 3. H 2 O 4. inorganic minerals Too little! Just fine Too much! Remember, most things can be too little or too much. Micro Macro 1 Source
Based on: Plant nutri0on Soils and Soil Minerals 1. O 2 2. CO 2 3. H 2 O 4. inorganic minerals Too little! Just fine Too much! Remember, most things can be too little or too much. Micro Macro 1 Source
Mechanical Weathering
 Weathering is the disintegration and decomposition of material at or near the surface. Erosion is the incorporation and transportation of material by a mobile agent, usually water, wind, or ice. Geologists
Weathering is the disintegration and decomposition of material at or near the surface. Erosion is the incorporation and transportation of material by a mobile agent, usually water, wind, or ice. Geologists
Practice Questions for Lecture 5 Geology 1200
 Practice Questions for Lecture 5 Geology 1200 Use these questions to test your knowledge of Lecture5. The exams will be similar in format, except that they will deal with more than one chapter, and will
Practice Questions for Lecture 5 Geology 1200 Use these questions to test your knowledge of Lecture5. The exams will be similar in format, except that they will deal with more than one chapter, and will
WEATHERING ACCORDING TO THE CATIONIC BONDING ENERGIES OF COLLOIDS I ABSTRACT
 WEATHERING ACCORDING TO THE CATIONIC BONDING ENERGIES OF COLLOIDS I By E. R. GRAHAM University of Missouri ABSTRACT A study was made of the energy changes of several colloidal systems in relation to weathering.
WEATHERING ACCORDING TO THE CATIONIC BONDING ENERGIES OF COLLOIDS I By E. R. GRAHAM University of Missouri ABSTRACT A study was made of the energy changes of several colloidal systems in relation to weathering.
remediation measurements
 IUNG Institute of Soil Science and Plant Cultivation PULAWY, POLAND International Workshop Current developments in remediation of contaminated lands Contamination by a pyrite mine spill in Spain: effect
IUNG Institute of Soil Science and Plant Cultivation PULAWY, POLAND International Workshop Current developments in remediation of contaminated lands Contamination by a pyrite mine spill in Spain: effect
Soil horizons differ in color, texture, structure, consistence, porosity and soil reaction.
 SECTION CONTENTS: The Soil Defined The Soil Profile Descriptions Of The Three Major Horizons A Fertile Soil Soil Texture Soil Structure Soil Color Soil Organisms Organic Matter Clay and Humus The Seat
SECTION CONTENTS: The Soil Defined The Soil Profile Descriptions Of The Three Major Horizons A Fertile Soil Soil Texture Soil Structure Soil Color Soil Organisms Organic Matter Clay and Humus The Seat
( ) 4Fe( l) + 3CO 2 ( g)
 Oxidation-Reduction (Redox) Reactions The terms oxidation and reduction may sound unfamiliar, but we see the results of these processes often in our daily lives. The corrosion of metal structures and monuments,
Oxidation-Reduction (Redox) Reactions The terms oxidation and reduction may sound unfamiliar, but we see the results of these processes often in our daily lives. The corrosion of metal structures and monuments,
Reactions with Acids. Chemistry 10 Mrs. Page
 Reactions with Acids Chemistry 10 Mrs. Page Learning Objectives Predict the products and balance chemical equations for the reactions with acids involving; reactive metals, metal oxides, metal hydroxides,
Reactions with Acids Chemistry 10 Mrs. Page Learning Objectives Predict the products and balance chemical equations for the reactions with acids involving; reactive metals, metal oxides, metal hydroxides,
Soil Quality Monitoring in Estuarine Ecosystem. Sachin N Hegde Center for Ecological Science Indian Institute of Science
 Soil Quality Monitoring in Estuarine Ecosystem Sachin N Hegde Center for Ecological Science Indian Institute of Science SOIL Soil is a thin layer of earth s crust which serves as a natural medium for the
Soil Quality Monitoring in Estuarine Ecosystem Sachin N Hegde Center for Ecological Science Indian Institute of Science SOIL Soil is a thin layer of earth s crust which serves as a natural medium for the
Chapter-3 GEOGRAPHICAL LOCATION, CLIMATE AND SOIL CHARACTERISTICS OF THE STUDY SITE
 Chapter-3 GEOGRAPHICAL LOCATION, CLIMATE AND SOIL CHARACTERISTICS OF THE STUDY SITE Chapter-3 GEOGRAPHICAL LOCATION, CLIMATE AND SOIL CHARACTERISTICS OF THE STUDY SITE Assam, the eastern most state of
Chapter-3 GEOGRAPHICAL LOCATION, CLIMATE AND SOIL CHARACTERISTICS OF THE STUDY SITE Chapter-3 GEOGRAPHICAL LOCATION, CLIMATE AND SOIL CHARACTERISTICS OF THE STUDY SITE Assam, the eastern most state of
Metal-Base Cation Exchange in Cape Cod Freshwater Ecosystems
 Metal-Base Cation Exchange in Cape Cod Freshwater Ecosystems Sean O Keeffe Colorado College 13 Advisor: Ann Giblin The Ecosystem Center Marine Biological Laboratory Woods Hole, MA December 15, 2011 Abstract
Metal-Base Cation Exchange in Cape Cod Freshwater Ecosystems Sean O Keeffe Colorado College 13 Advisor: Ann Giblin The Ecosystem Center Marine Biological Laboratory Woods Hole, MA December 15, 2011 Abstract
Shirley E. Clark, Ph.D., P.E., D. WRE Robert E. Pitt, Ph.D., P.E., BCEE, D. WRE
 Shirley E. Clark, Ph.D., P.E., D. WRE Robert E. Pitt, Ph.D., P.E., BCEE, D. WRE Current PA Guidance Many guidance documents apply expected pollutant removals based on literature. However, typically presented
Shirley E. Clark, Ph.D., P.E., D. WRE Robert E. Pitt, Ph.D., P.E., BCEE, D. WRE Current PA Guidance Many guidance documents apply expected pollutant removals based on literature. However, typically presented
Horizon Differences in Selected Physical Properties of Soils formed Over Dissimilar Lithologies in South-EasternNigeria
 FUTO Journal Series (FUTOJNLS), 2015, VOL. 1, Issue 2 62 Horizon Differences in Selected Physical Properties of Soils formed Over Dissimilar Lithologies in South-EasternNigeria 1 1 2 Onweremadu, E.U.,Ahukaemere,
FUTO Journal Series (FUTOJNLS), 2015, VOL. 1, Issue 2 62 Horizon Differences in Selected Physical Properties of Soils formed Over Dissimilar Lithologies in South-EasternNigeria 1 1 2 Onweremadu, E.U.,Ahukaemere,
The Chemistry of Seawater. Unit 3
 The Chemistry of Seawater Unit 3 Water occurs naturally on earth in 3 phases: solid, liquid, or gas (liquid is most abundant) Water Phases Basic Chemistry Review What is an atom? Smallest particles of
The Chemistry of Seawater Unit 3 Water occurs naturally on earth in 3 phases: solid, liquid, or gas (liquid is most abundant) Water Phases Basic Chemistry Review What is an atom? Smallest particles of
The Lithosphere. Definition
 10/14/2014 www.komar.de The Lithosphere Ben Sullivan, Assistant Professor NRES 765, Biogeochemistry October 14th, 2014 Contact: bsullivan@cabnr.unr.edu Definition io9.com tedquarters.net Lithos = rocky;
10/14/2014 www.komar.de The Lithosphere Ben Sullivan, Assistant Professor NRES 765, Biogeochemistry October 14th, 2014 Contact: bsullivan@cabnr.unr.edu Definition io9.com tedquarters.net Lithos = rocky;
Ecoregions Glossary. 7.8B: Changes To Texas Land Earth and Space
 Ecoregions Glossary Ecoregions The term ecoregions was developed by combining the terms ecology and region. Ecology is the study of the interrelationship of organisms and their environments. The term,
Ecoregions Glossary Ecoregions The term ecoregions was developed by combining the terms ecology and region. Ecology is the study of the interrelationship of organisms and their environments. The term,
Lecture 16: Soil Acidity; Introduction to Soil Ecology
 Lecture 16: Soil Acidity; Introduction to Soil Ecology Aluminum and Soil Acidity Aluminum Toxicity in Acid Soils Tolerant Sensitive Tolerant Sensitive Plants often are sensitive to the presence of dissolved
Lecture 16: Soil Acidity; Introduction to Soil Ecology Aluminum and Soil Acidity Aluminum Toxicity in Acid Soils Tolerant Sensitive Tolerant Sensitive Plants often are sensitive to the presence of dissolved
G470/570 Regional Landscapes of Oregon Basics of Soils and Rock Weathering
 G470/570 Regional Landscapes of Oregon Basics of Soils and Rock Weathering I. SOIL BASICS A. Soils Defined 1. Soils represent the weathered mantle of unconsolidated surficial material that covers land
G470/570 Regional Landscapes of Oregon Basics of Soils and Rock Weathering I. SOIL BASICS A. Soils Defined 1. Soils represent the weathered mantle of unconsolidated surficial material that covers land
4. A hydrogen bond is formed between a hydrogen atom and a negative atom, usually a nitrogen or oxygen.
 Name Biology Summer Assignment Print, complete and bring this assignment with you on the first day of classes. Use the background information document to help you complete this assignment. Modified True/False
Name Biology Summer Assignment Print, complete and bring this assignment with you on the first day of classes. Use the background information document to help you complete this assignment. Modified True/False
It is important to recognize two distinct but overlapping uses of the term "clay":
 Soil Texture (Particle Size Analysis or Mechanical Analysis) Introduction Texture, or size distribution of mineral particles (or its associated pore volume), is one of the most important measures of a
Soil Texture (Particle Size Analysis or Mechanical Analysis) Introduction Texture, or size distribution of mineral particles (or its associated pore volume), is one of the most important measures of a
Weathering is the process that breaks down rock and other substances at Earth s surface
 Chapter 8 Notes Weathering is the process that breaks down rock and other substances at Earth s surface Factors that contribute to weathering Heat Cold Water Ice O 2 & CO 2 in the atmosphere Examples of
Chapter 8 Notes Weathering is the process that breaks down rock and other substances at Earth s surface Factors that contribute to weathering Heat Cold Water Ice O 2 & CO 2 in the atmosphere Examples of
EFFECTS OF FLUVO MARINE DEPOSITS ON SOME SOIL CHARACTERISTICS IN THE NIGER DELTA REGION OF NIGERIA
 I.J.A.B.R, VOL. 3(4) 2013: 514-518 ISSN 2250 3579 EFFECTS OF FLUVO MARINE DEPOSITS ON SOME SOIL CHARACTERISTICS IN THE NIGER DELTA REGION OF NIGERIA Egbuchua, C.N. and Enujeke, E.C. Department of Agronomy,
I.J.A.B.R, VOL. 3(4) 2013: 514-518 ISSN 2250 3579 EFFECTS OF FLUVO MARINE DEPOSITS ON SOME SOIL CHARACTERISTICS IN THE NIGER DELTA REGION OF NIGERIA Egbuchua, C.N. and Enujeke, E.C. Department of Agronomy,
NCERT Solutions for Atoms and Molecules
 1 NCERT Solutions for Atoms and Molecules Question 1. In a reaction, 5.3 g of sodium carbonate reacted with 6 g of ethanoic acid. The products were 2.2 g of carbon dioxide, 0.9 g water and 8.2 g of sodium
1 NCERT Solutions for Atoms and Molecules Question 1. In a reaction, 5.3 g of sodium carbonate reacted with 6 g of ethanoic acid. The products were 2.2 g of carbon dioxide, 0.9 g water and 8.2 g of sodium
Monument Valley, Utah. What weathering processes contributed to the development of these remarkable rock formations? Weathering Mechanisms
 Monument Valley, Utah. What weathering processes contributed to the development of these remarkable rock formations? Weathering Includes Physical, Chemical, Biological processes WEATHERING CHAPTER 7 Weathering
Monument Valley, Utah. What weathering processes contributed to the development of these remarkable rock formations? Weathering Includes Physical, Chemical, Biological processes WEATHERING CHAPTER 7 Weathering
THE UNITED REPUBLIC OF TANZANIA NATIONAL EXAMINATIONS COUNCIL CERTIFICATE OF SECONDARY EDUCATION EXAMINATION
 THE UNITED REPUBLIC OF TANZANIA NATIONAL EXAMINATIONS COUNCIL CERTIFICATE OF SECONDARY EDUCATION EXAMINATION 032/1 CHEMISTRY 1 (For Both School and Private Candidates) Time: 3 Hours Thursday, 06 th November
THE UNITED REPUBLIC OF TANZANIA NATIONAL EXAMINATIONS COUNCIL CERTIFICATE OF SECONDARY EDUCATION EXAMINATION 032/1 CHEMISTRY 1 (For Both School and Private Candidates) Time: 3 Hours Thursday, 06 th November
5072 CHEMISTRY (NEW PAPERS WITH SPA) BASIC TECHNIQUES 5067 CHEMISTRY (NEW PAPERS WITH PRACTICAL EXAM) BASIC TECHNIQUES
 5072 CHEMISTRY (NEW PAPERS WITH SPA) BASIC TECHNIQUES 5067 CHEMISTRY (NEW PAPERS WITH PRACTICAL EXAM) BASIC TECHNIQUES LEARNING OUTCOMES a) Be able to write formulae of simple compounds b) Be able to write
5072 CHEMISTRY (NEW PAPERS WITH SPA) BASIC TECHNIQUES 5067 CHEMISTRY (NEW PAPERS WITH PRACTICAL EXAM) BASIC TECHNIQUES LEARNING OUTCOMES a) Be able to write formulae of simple compounds b) Be able to write
Soil Chemistry. Dr. Shalamar Armstrong Dr. Rob Rhykerd Department of Agriculture
 Soil Chemistry Dr. Shalamar Armstrong sdarmst@ilstu.edu Dr. Rob Rhykerd rrhyker@ilstu.edu Importance of soil Feeding the world World Population & Growth Other Asia Africa India China Latin America Europe
Soil Chemistry Dr. Shalamar Armstrong sdarmst@ilstu.edu Dr. Rob Rhykerd rrhyker@ilstu.edu Importance of soil Feeding the world World Population & Growth Other Asia Africa India China Latin America Europe
SUMMARY OF ASL TEST METHODS
 SUMMARY OF ASL TEST METHODS 1. Determination of Macro and Micro Elements in Plant Tissue by Dual View Inductively Coupled Plasma Optical Emission Spectrometer This method describes multi-elemental determinations
SUMMARY OF ASL TEST METHODS 1. Determination of Macro and Micro Elements in Plant Tissue by Dual View Inductively Coupled Plasma Optical Emission Spectrometer This method describes multi-elemental determinations
Q1.Use the periodic table and the information in the table below to help you to answer the questions.
 Q1.Use the periodic table and the information in the table below to help you to answer the questions. The table shows part of an early version of the periodic table. Group 1 Group 2 Group 3 Group 4 Group
Q1.Use the periodic table and the information in the table below to help you to answer the questions. The table shows part of an early version of the periodic table. Group 1 Group 2 Group 3 Group 4 Group
Science Class 9 th ATOMS AND MOLECULES. Symbols of Atoms of Different Elements. Atomic Mass. Molecules. Ions. Mole Concept. Finish Line & Beyond
 Science Class 9 th ATOMS AND MOLECULES Symbols of Atoms of Different Elements Atomic Mass Molecules Ions Mole Concept Atom An atom is a particle of matter that uniquely defines a chemical element. An atom
Science Class 9 th ATOMS AND MOLECULES Symbols of Atoms of Different Elements Atomic Mass Molecules Ions Mole Concept Atom An atom is a particle of matter that uniquely defines a chemical element. An atom
Weathering Cycle Teacher Notes
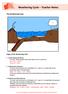 The Weathering Cycle Stages of the Weathering Cycle: 1. Carbon Dioxide and Water In clouds, carbon dioxide reacts with water to form a weak acid. H 2 O + CO 2 --> H 2 CO 3 H 2 CO 3 H + + HCO 3-2. Acid
The Weathering Cycle Stages of the Weathering Cycle: 1. Carbon Dioxide and Water In clouds, carbon dioxide reacts with water to form a weak acid. H 2 O + CO 2 --> H 2 CO 3 H 2 CO 3 H + + HCO 3-2. Acid
A few more details on clays, Soil Colloids and their properties. What expandable clays do to surface area. Smectite. Kaolinite.
 A few more details on clays, Soil Colloids and their properties What expandable clays do to surface area Kaolinite Smectite Size 0.5-5 µm External surface 10-30 m 2 /g Internal surface - Size 0.1-1 µm
A few more details on clays, Soil Colloids and their properties What expandable clays do to surface area Kaolinite Smectite Size 0.5-5 µm External surface 10-30 m 2 /g Internal surface - Size 0.1-1 µm
Weathering is the process by which rocks on or near Earth s surface break down and change. Spherical weathering near Paris, NC
 Weathering Weathering is the process by which rocks on or near Earth s surface break down and change. Spherical weathering near Paris, NC The two types of weathering are Chemical and. Mechanical Mechanical
Weathering Weathering is the process by which rocks on or near Earth s surface break down and change. Spherical weathering near Paris, NC The two types of weathering are Chemical and. Mechanical Mechanical
CHEM5. General Certificate of Education Advanced Level Examination June Unit 5 Energetics, Redox and Inorganic Chemistry
 Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials General Certificate of Education Advanced Level Examination June 2010 Question 1 2 Mark Chemistry
Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials General Certificate of Education Advanced Level Examination June 2010 Question 1 2 Mark Chemistry
GEOLOGICAL PROCESSES AND MATERIALS SGM210
 University of Pretoria GEOLOGICAL PROCESSES AND MATERIALS SGM210 2 May 2014 1 DISPERSIVE SOILS Thanks to Dr Phil Paige Green 1 Dispersive (and Erodible) Soils Phil Paige Green (H.F.W.K Elges) CSIR Built
University of Pretoria GEOLOGICAL PROCESSES AND MATERIALS SGM210 2 May 2014 1 DISPERSIVE SOILS Thanks to Dr Phil Paige Green 1 Dispersive (and Erodible) Soils Phil Paige Green (H.F.W.K Elges) CSIR Built
Lecture 2: Soil Properties and Groundwater Flow
 ENGI 7718 Environmental Geotechniques ENGI 9621 Soil Remediation Engineering Lecture 2: Soil Properties and Groundwater Flow Spring 2011 Faculty of Engineering & Applied Science 1 2.1 Soil properties 2.1.1
ENGI 7718 Environmental Geotechniques ENGI 9621 Soil Remediation Engineering Lecture 2: Soil Properties and Groundwater Flow Spring 2011 Faculty of Engineering & Applied Science 1 2.1 Soil properties 2.1.1
ATHENS 2013 SOIL ANALYSIS REPORT
 FRAMME LIFE 08 NAT//GR//000533 ATHENS 2013 SOIL ANALYSIS REPORT FRAMME - LIFE08 NAT/GR/000533 RHODES FRAMME, Fire Restoration Methodology for Mediterranean Forests - environmental safety & sustainability
FRAMME LIFE 08 NAT//GR//000533 ATHENS 2013 SOIL ANALYSIS REPORT FRAMME - LIFE08 NAT/GR/000533 RHODES FRAMME, Fire Restoration Methodology for Mediterranean Forests - environmental safety & sustainability
The Effect of Soil Parent Materials on Yield of Cocoa
 The Effect of Soil Parent Materials on Yield of Cocoa *Adzemi Mat Arshad 1 H. M. Edi Armanto 2 Haruna Yahaya Rawayau 1,3 1. Soil Science Laboratory, School of Food Science and Technology Universiti Malaysia
The Effect of Soil Parent Materials on Yield of Cocoa *Adzemi Mat Arshad 1 H. M. Edi Armanto 2 Haruna Yahaya Rawayau 1,3 1. Soil Science Laboratory, School of Food Science and Technology Universiti Malaysia
