Optical spectroscopies. Rita P.Y. Chen. Electromagnetic Spectrum
|
|
|
- Neal Allen
- 5 years ago
- Views:
Transcription
1 Optical spectroscopies Rita P.Y. Chen Electromagnetic Spectrum
2 The longer the wavelength the lower the energy!!!! h = 6.63 x J s 1 ev = 1.6 x J, c= 3 x 10 8 m
3
4
5
6 Transmittance, T = I f / I o % Transmittance, %T = 100 T Absorbance, A =log 10 I 0 / I f A=log 10 1/T Absorbance O.D. units or absorbance is expressed in logarithmic terms so they are additive. An object of O.D. of 1.0 absorbs 90% of the light. Only 10? % of the light reaches detector. When another object of O.D. 1.0 placed in the path, further 90% of this light is absorbed and only? 1 % of the original light is transmitted by the second object. It is possible to express the absorbance of a mixture of substances at a particular wavelength as the sum of the absorbances of the components. The extinction coefficient indicates how efficient the molecule will absorb photons.
7 σ to σ An electron in a bonding s orbital is excited to the corresponding antibonding orbital. The energy required is large. For example, methane (which has only C-H bonds, and can only undergo σ σ* transitions) shows an absorbance maximum at 125 nm. Absorption maxima due to σ σ * transitions are not seen in typical XUV-Vis. spectra ( nm) n to σ Saturated compounds containing atoms with lone pairs (non-bonding electrons) are capable of n σ* transitions. These transitions usually need less energy than σ σ * transitions. They can be initiated by light whose wavelength is in the range nm. The number of organic functional groups with n σ* peaks in the UV region is small. X Chromophore λ max Chromophore λ max alcohols, ethers ~ 185 alkanes ~ 150 amines ~ 195 sulfur compounds ~ 195
8 n π* π π* Chromophore carbonyls λ max ~ 285 Chromophore alkenes λ max ~ 175 alkynes ~ 170 carbonyls ~ 188 n π* and π π* Transitions Molar absorbtivities from n π* transitions are relatively low, and range from 10 to 100 L mol -1 cm -1. π π* transitions normally give molar absorbtivities between 1000 and 10,000 L mol -1 cm -1. Tetraphenylcyclopentadienone
9 The table below gives a list of common solvents and the minimum wavelength from which they may be used in a 1 cm cell. Solvent acetonitrile water cyclohexane hexane methanol ethanol ether methylene chloride chloroform carbon tetrachloride Minimum Wavelength (nm) Solvent effect in absorption spectrum The solvent in which the absorbing species is dissolved also has an effect on the spectrum of the species. Peaks resulting from n π* transitions are shifted to shorter wavelengths (blue shift) with increasing solvent polarity. This arises from increased solvation of the lone pair, which lowers the energy of the n orbital. Often (but not always), the reverse (i.e. red shift) is seen for π π* transitions. This is caused by attractive polarisation forces between the solvent and the absorber, which lower the energy levels of both the excited and unexcited states. This effect is greater for the excited state, and so the energy difference between the excited and unexcited states is slightly reduced - resulting in a small red shift. This effect also influences n π* transitions but is overshadowed by the blue shift resulting from solvation of lone pairs. polarity n π* π π*
10 Circular dichroism spectroscopy
11
12 Far UV CD n -> π* ( nm) centered around 220 nm, ε = ~100 mol -1 dm 3 cm -1, involves non-bonding electrons of O of the carbonyl π -> π* centered around 190 nm ε = ~7000 mol -1 dm 3 cm -1, π -> π*dominated by the carbonyl π-bond, and also affected by the involvement of the nitrogen in the π-orbitals The intensity and energy of these transitions depends on φ and ψ (i.e., secondary structure) exiton coupling of the π->π* transitions leads to positive (π >π*)perpendicular at 192 nm and negative (π >π*)parallel at 209 nm negative at 222 nm is red shifted (n->π*)
13 [θ] α β-sheet Random coil Poly-L-proline (P2) Beer-Lambert law A= - log 10 (I in /I out ) A= ε x b x c ε = ε L -ε R differential absorbance of a 1 mol/l solution in a 1 cm cell Measured θ, ellipticity, is the rotation in degrees of a 1dmol/cm3 solution and a pathlength of 1 cm Molar ellipticity: [θ] = degrees cm 2 dmol -1 θ x 100 x Mr c x l c: mg/ml l: cm Mean residue ellipiticity: [θ] MRW = [θ] /residue number degrees cm 2 dmol -1 residue -1 ε = [θ] /3298 Litre mol -1 cm -1 or Litre (mol residue) -1 cm -1
14 Use far-uv CD to determine amounts of secondary structure in proteins generate basis sets by determining spectra of pure α-helix, β-sheet, etc. of synthetic peptides or deconvoluting CD spectra of proteins with know structures to generate basis sets of each of secondary structure poly-l-lysine {(Lys)n} can adopt 3 different conformations merely by varying the ph and temperature random coil at ph 7.0 α-helix at ph 10.8 β-form at ph 11.1 after heating to 52 C and recooling CD spectrum of unknown protein = fαsa(l) + fβsb(l) + frcsrc(l), where Sa(l), Sb(l), and SRC(l) are derived from poly-l-lysine basis spectra.
15 The disadvantage of this method is that although these basis sets are easily determined by direct measurement, they do not always agree from one lab to another. In addition, chain length and aggregation effect the basis set spectra. However, this method is usually accurate to within 10% for α-helix content. Technique Secondary Structure carboxypeptid ase α- chymotrypsin myoglobin α 23% 8% 68% X-ray β 18% 22% 0% RC + other 59% 70% 32% α 13% 12% 68% CD using (Lys) n Basis Sets β 31% 23% 5% RC + other 56% 65% 27% Protein structure IRL press
16 Far-UV CD Near-UV CD Near UV CD Wavelength Range nm nm nm nm Chromophore Contributions side chain Phe side chain Tyr side chain Trp disulphide bond
17 Thermal denaturation monitored by CD Chemical denaturation monitored by CD
18 1400 yf= x[gdnhcl] 1200 intensity at 303 nm U K fold F GdnHCl (M) yu= x[gdnhcl]
19 Fluorescence Photon emission as an electron returns from an excited state to ground state S 1 S 1 Energy hv ex hv em S 0 The electronic states of most organic molecules can be divided into singlet states and triplet states; Singlet state: All electrons in the molecule are spin-paired Triplet state: One set of electron spins is unpaired
20 Possible physical process following absorption of a photon by a molecule Fluorescence Fluorescing molecules (fluorophores) are usually aromatic Resolution of fluorescence is usually much better than absorption because you are measuring at right angles to the direction of exciting light (better signal to noise) so can work with much lower concentrations The wavelength of absorption is related to the size of the chromophores Smaller chromophores, higher energy (shorter wavelength)
21 Fluorescence Stokes Shift: The difference in wavelength between absorbed and emitted quanta. The emitted wavelength is always longer (if single photons are absorbed) or equal to the incident wavelength, due to energy conservation; the difference is absorbed as heat in the atomic lattice of the material. Fluorescnece Intensity Fluorescein molecule Stokes Shift is 25 nm 495 nm 520 nm Wavelength Stokes shift increases with solvent polarity. Fluorescence intensity is proportional to the product of ε and Q f Quantum Yield Q f = photons emitted photons absorbed = k r k r + k nr
22 Fluorescence Lifetime (τ) - is the time delay between the absorbance and the emission 1 τ = k r + k nr Many of these measurements are made possible by the fluorescence lifetime, the average time that a molecule spends in the excited state before emitting a photon and returning to the ground state. It is an important and unique feature of an excited state. Fluorescence lifetimes are very short. Most fluorescence lifetimes fall within the range of hundreds of picoseconds to hundreds of nanoseconds. The fluorescence lifetime can function as a molecular stopwatch to observe a variety of interesting molecular events. An antibody may rotate slightly within its molecular environment. A protein can change orientation. A critical binding reaction may occur. Because the time-scale of these events is similar to the fluorescence lifetime, the measurement of the fluorescence lifetime allows the researcher to peer into the molecule and observe these phenomena Fluorescence lifetime measurement Fluorescence lifetime can be measured by measuring the decay of the total fluorescence intensity following pulsed excitation which reflects the average time that a molecule remains in the singlet excited state. The fluorescence decay spectra were collected using time-correlated single-photon counting. Many dynamic events can deactivate the excited state and hence influence the lifetime, including solvent relaxation, fluctuation in macromolecular conformation, rotations of side-chains, interactions with neighboring residues, and quenching by exogeneous agents. Fluorescence lifetime of a fluorophore in a protein or nucleic acid is highly dependent upon its local environment and can vary from a few picoseconds to tens of nanoseconds.
23 Effects of molecular structure on fluorescence Extent of p-electron system : An increase in the extent of the π-electron system (i.e. degree of conjugation) leads to a shift of the absorption and fluorescence spectra to longer wavelengths and to an increase in the fluorescence quantum yield. naphthalene anthracence naphthacene
24 Substituted aromatic hydrocarbons: 1. Internal heavy atom effect: the presence of heavy atom are substitutents of aromatic molecules (e.g. Br, I) results in fluorescence quenching because of the increased probability of intersystem crossing 2. Electron-donating substituents ( -OH, -OR, -NH2, -NHR, -NR2) induce an increase in the molar absorption coefficient and a shift in both absorption and fluorescence spectra. They become broad and structureless. 3. Sulfonate: The solubility of many fluorophores is achieved by grafting sulfonate groups. It causes small red-shift in fluorescence spectrum and a slight decrease in fluorescence quantum yield. Bernard Valeur, Molecular Fluorescence, Wiley-VCH Fluorescence is very dependent on the environment. Small molecules and other compounds can absorb energy and quench (lower) fluoresence. Temperature has a big effect. Dielectric constant e.g. aqueous vs hydrophobic/polar Effect of temperature Generally, an increase in temperature results in a decrease in the fluorescence quantum yield and the life time because the non-radiative processes related to thermal agitation (collisions with solvent molecules, intramolecular vibrations and rotations ) are more efficient at higher temperature.
25 Effect of local environment Aromatic residues (especially Tryptophan) are naturally occurring fluorophores which are good probes of folding. A tryptophan side-chain in a hydrophobic core has a high fluorescence intensity (not always)and a blue-shifted (shorter wavelength) emission maximum. Exposed tryptophans (or tryptophans from unfolded proteins) have low fluorescence intensity (not always)and a red-shifted longer wavelength) emission maximum. A: extremely nonpolar environment inside the protein globule S: relatively nonpolar environment inside the protein globule, interacting with neighboring polar group I: polar but rigid environment inside the protein globule, interacting with neighboring polar group II: at protein surface, in contact with bound water or other polar group III: at protein surface, in contact with free water, typical for unfolded protein
26 Intensity Higher intensity in denatured state: There is probably a quenching group near the Trp of the native protein Fluorescence Resonance Energy Transfer (FRET) Resonance energy transfer can occur when the donor and acceptor molecules are less than 100 A of one another Energy transfer is non-radiative which means the donor is not emitting a photon which is absorbed by the acceptor DONOR ACCEPTOR Absorbance Fluorescence Absorbance Fluorescence Wavelength
27 When R > 2 R 0, E ~ 0 When R < 1/2R 0, E ~ 1 Choose 0.5 R 0 < r < 1.5 R 0 Förster Radius The distance at which energy transfer is 50% efficient (i.e., 50% of excited donors are deactivated by FRET) is defined by the Förster radius (R o ). The magnitude of R o is dependent on the spectral properties of the donor and acceptor dyes: Donor Acceptor R o (Å) Fluorescein Tetramethylrhodamine 55 IAEDANS Fluorescein 46 EDANS Dabcyl 33 Fluorescein Fluorescein 44 BODIPY FL BODIPY FL 57 Fluorescein QSY 7 and QSY 9 dyes 61
28 R 6 = R 06 (E -1-1) E = 1- (I DA /I D ) = 1- (L DA /L D ) Where I DA is the fluorescence intensity of the donor in the presence of acceptor I D is the fluorescence intensity of the donor only L DA is the fluorescence lifetime of the donor in the presence of acceptor L D is the fluorescence lifetime of the donor The critical Förster radius is 40 Å for BFP-GFP and 50 Å for CFP-YFP.
29 Fluorescence Polarization (FP) Fluorescence polarization measurements provide information on molecular orientation and mobility and processes that modulate them, including receptor ligand interactions, proteolysis, protein DNA interactions, membrane fluidity and muscle contraction Figure 1. Physical basis of fluorescence polarization assays. Dye molecules with their absorption transition vectors (arrows) aligned parallel to the electric vector of linearly polarized light (along the vertical page axis) are selectively excited. For dyes attached to small, rapidly rotating molecules, the initially photoselected orientational distribution becomes randomized prior to emission, resulting in low fluorescence polarization. Conversely, binding of the low molecular weight tracer to a large, slowly rotating molecule results in high fluorescence polarization. Fluorescence polarization therefore provides a direct readout of the extent of tracer binding to proteins, nucleic acids and other biopolymers. Polarization ratio (P) and emission anisotropy (r)
30 Fluorescence anisotropy measurement r = (IVV G IVH) /(IVV + 2G IVH) where IVV is the fluorescence intensity recorded with excitation and emission polarization in vertical position, and IVH is the fluorescence intensity recorded with the emission polarization aligned in horizontal position. The G factor is the ratio of sensitivities of detection system for vertically and horizontally polarized light G = IHV/IHH. Fluorescence polarization increases as molecular weight increases. Fluorescence polarization increases as solvent viscosity increases. Fluorescence polarization decreases as the excited state lifetime of the dye increases. Figure 2. Simulation of the relationship between molecular weight (MW) and fluorescence polarization (P). Simulations are shown for dyes with various fluorescence lifetimes ( ): 1 ns (cyanine dyes) in purple, 4 ns (fluorescein and Alexa Fluor 488 dyes) in red, 6 ns (some BODIPY dyes) in green and 20 ns (dansyl dyes) in blue. At MW = 1000, P = for = 1 ns, P = for = 4 ns, P = for = 6 ns and P = for = 20 ns. Simulations assume Po (the fundamental polarization) = 0.5 and rigid attachment of dyes to spherical carriers.
31 How does the FluoroTect System Work? The FluoroTect Green Lys in vitro Translation Labeling System uses a charged lysine trna molecule labeled with the fluorophore BODIPY -FL at the -amino acid position of lysine (Figure 1). The BODIPY -FL fluorophore, with an absorbance at 502nm and an emission at 510nm, was developed to be compatible with widely used excitation sources and common optical filter sets. The labeled FluoroTect trna is added directly to the translation reaction, and the labeled lysine is incorporated into the synthesized protein. Recently, a method using BODIPY -FL-labeled methionine was shown to be able to detect nanogram levels of protein using laser-based fluorescent gel scanning. For the FluoroTect System, lysine was chosen as the labeled amino acid because it is one of the more frequently used amino acids, comprising, on average, 6.6% of protein's amino acids. This compares to 1.7% for methionine. FTIR signal Transitions in Vibrational levels. Signals arise from transition that are localized. C=O stretch, C-H, N-H stretch, bending modes, etc. Signals result from asymmetric stretch O=C=O not active in FTIR Lower energy than electronic Less used: less intense (ε often 10-x less). Higher concentrations needed. Interference from H 2 O.
32 FTIR as a tool to study protein structure In Vibrational transitions, bond type determines vibrational frequency of transition, (cm -1 ). Diagnostic absorption bands: Amide I absorption; also has contributions from C=O stretch. ( cm -1 ) (1660 cm -1 = 6024 nm) Amide I Depends on strength of H-bonds involving C=O and N-H Also N-H stretch (3300), N-H bend ( ) Differences in H-bond geometry, and pattern of 2 structures gives characteristic amide I (& II) absorptions Sensitivity of FTIR to 2 structure Stronger H-bonds yield changes in vibrational frequencies. H-bond makes it easier to stretch along axis of C=O or N-H, but harder to bend, vs. Non-H-bond Lowest frequency is for long aggregates β-sheets over long stretches of extended structure.
33 Characteristic FTIR bands Antiparallel β-sheet and aggregates cm helix α-helix unordered β-sheet Aggregates These ranges are only guidelines, general rules. Other factors like solvent effects, distortion of 2 elements, prosthetic groups can alter frequencies Advantages of using FTIR Identify presence of protein 2 structures Detect changes in protein 2 structures Fast kinetic events (stopped flow) Good for rapidly interconverting samples (can resolve both components instead of averages). No size limit (compared to NMR). Good for membranes (even vesicles, bilayers, solids, films. 2D crystals, gels. Light scattering isn t a big problem. Samples can be suspensions or inhomogeneous dispersions. Excellent tool for studying protein aggregation, prions, amyloids, etc. (kinetics, progression). Organic solvents are good TFE, etc. for hydrophobic peptides
34 FT-IR of PrP( ) and S135-α-GalNAc PrP( ) Day 0 S135-α-GalNAc Day 0 PrP( ) Day cm -1 β-sheet S135 -α-galnac Day 14 Limitations of FTIR Width and separation of peaks gives overlap: mixtures of 2 elements can be hard to distinguish. Deconvolution is needed. Changes in spectra can be due to shift of one type of 2 structure to another, or to an increase of one type. Hard to distinguish. Deconvolution methods are not always clean. Can be difficult to quantify. Better for β-sheet than α-helix, so good complement to CD. Water interference. So FTIR works best with solids, or films.
35 Water effects on FTIR spectra Water is BIG problem: O-H bending mode at 1644 cm -1 Obscures amide I band Can subtract water signal (blank), if a small amount of water is in sample (e.g. bound to protein). But if water is solvent, O-H absorbs so strongly that you can t subtract it and still retain sample signal. Solutions - use D 2 O instead O-D stretch is lower than O-H by 400 cm -1 Slow and fast kinetics: UV-VIS, CD, FTIR,Fluorescence
36 Stopped Flow: how does it work? Fluo. A Excitation Mixer Cell DRIVE Stopping Syringe Standard stopped flow schematic B Under pneumatic drive activation, the two small volumes of solutions are driven from high performance syringes through a high efficiency mixer. The resultant mixture passes through a measurement flow cell and into a stopping syringe. Just prior to stopping, a steady state flow is achieved. As the solution fills the stopping syringe, the plunger hits a block, causing the flow to be stopped instantaneously. Using appropriate techniques, the kinetics of the reaction can be measured in the cell. Ellipticity (mdeg) Denatured 8 M Urea Refolding kinetic trace Native 0 M Urea Wavelength (nm) A(t) =ΣA i exp(-k i t) + A where A(t) is the amplitude of the change at time t, A is the amplitude at infinite time, A i is the amplitude at zero time of phase i, and k i is the rate of phase i.
37
38
CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray
 CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray crystallography An example of the use of CD Modeling
CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray crystallography An example of the use of CD Modeling
Fluorescence 2009 update
 XV 74 Fluorescence 2009 update Jablonski diagram Where does the energy go? Can be viewed like multistep kinetic pathway 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet
XV 74 Fluorescence 2009 update Jablonski diagram Where does the energy go? Can be viewed like multistep kinetic pathway 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet
To be covered (and why) Spectroscopy of Proteins. UV-Vis Absorption. UV-Vis Absorption. Spectra
 To be covered (and why) Spectroscopy of Proteins General considerations UV-Vis Absorption quantitation Fluorescence hydrophobicity Foldedness FT-Infrared Foldedness ircular Dichroism Foldedness NMR (a
To be covered (and why) Spectroscopy of Proteins General considerations UV-Vis Absorption quantitation Fluorescence hydrophobicity Foldedness FT-Infrared Foldedness ircular Dichroism Foldedness NMR (a
LABORATORY OF ELEMENTARY BIOPHYSICS
 LABORATORY OF ELEMENTARY BIOPHYSICS Experimental exercises for III year of the First cycle studies Field: Applications of physics in biology and medicine Specialization: Molecular Biophysics Fluorescence
LABORATORY OF ELEMENTARY BIOPHYSICS Experimental exercises for III year of the First cycle studies Field: Applications of physics in biology and medicine Specialization: Molecular Biophysics Fluorescence
Fluorescence (Notes 16)
 Fluorescence - 2014 (Notes 16) XV 74 Jablonski diagram Where does the energy go? Can be viewed like multistep kinetic pathway 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet
Fluorescence - 2014 (Notes 16) XV 74 Jablonski diagram Where does the energy go? Can be viewed like multistep kinetic pathway 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet
Fluorescence Workshop UMN Physics June 8-10, 2006 Basic Spectroscopic Principles Joachim Mueller
 Fluorescence Workshop UMN Physics June 8-10, 2006 Basic Spectroscopic Principles Joachim Mueller Fluorescence, Light, Absorption, Jablonski Diagram, and Beer-Law First stab at a definition: What is fluorescence?
Fluorescence Workshop UMN Physics June 8-10, 2006 Basic Spectroscopic Principles Joachim Mueller Fluorescence, Light, Absorption, Jablonski Diagram, and Beer-Law First stab at a definition: What is fluorescence?
XV 74. Flouorescence-Polarization-Circular-Dichroism- Jablonski diagram Where does the energy go?
 XV 74 Flouorescence-Polarization-Circular-Dichroism- Jablonski diagram Where does the energy go? 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet S = 0 could be any of them
XV 74 Flouorescence-Polarization-Circular-Dichroism- Jablonski diagram Where does the energy go? 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet S = 0 could be any of them
single-molecule fluorescence resonance energy transfer
 single-molecule fluorescence resonance energy transfer (2) determing the Förster radius: quantum yield, donor lifetime, spectral overlap, anisotropy michael börsch 26/05/2004 1 fluorescence (1) absorbance
single-molecule fluorescence resonance energy transfer (2) determing the Förster radius: quantum yield, donor lifetime, spectral overlap, anisotropy michael börsch 26/05/2004 1 fluorescence (1) absorbance
Chapter 17: Fundamentals of Spectrophotometry
 Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
Chapter 15 Molecular Luminescence Spectrometry
 Chapter 15 Molecular Luminescence Spectrometry Two types of Luminescence methods are: 1) Photoluminescence, Light is directed onto a sample, where it is absorbed and imparts excess energy into the material
Chapter 15 Molecular Luminescence Spectrometry Two types of Luminescence methods are: 1) Photoluminescence, Light is directed onto a sample, where it is absorbed and imparts excess energy into the material
Molecular Luminescence. Absorption Instrumentation. UV absorption spectrum. lg ε. A = εbc. monochromator. light source. Rotating mirror (beam chopper)
 Molecular Luminescence Absorption Instrumentation light source I 0 sample I detector light source Rotating mirror (beam chopper) motor b sample I detector reference I 0 UV absorption spectrum lg ε A =
Molecular Luminescence Absorption Instrumentation light source I 0 sample I detector light source Rotating mirror (beam chopper) motor b sample I detector reference I 0 UV absorption spectrum lg ε A =
Chapter 17: Fundamentals of Spectrophotometry
 Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
Spectroscopy. Page 1 of 8 L.Pillay (2012)
 Spectroscopy Electromagnetic radiation is widely used in analytical chemistry. The identification and quantification of samples using electromagnetic radiation (light) is called spectroscopy. Light has
Spectroscopy Electromagnetic radiation is widely used in analytical chemistry. The identification and quantification of samples using electromagnetic radiation (light) is called spectroscopy. Light has
Luminescence spectroscopy
 Febr. 203 Luminescence spectroscopy Biophysics 2 nd semester Józse Orbán University o Pécs, Department o Biophysics Deinitions, laws FUNDAMENTALS o SPECTROSCY review - Spectral types (absorbtion/emission
Febr. 203 Luminescence spectroscopy Biophysics 2 nd semester Józse Orbán University o Pécs, Department o Biophysics Deinitions, laws FUNDAMENTALS o SPECTROSCY review - Spectral types (absorbtion/emission
Singlet. Fluorescence Spectroscopy * LUMO
 Fluorescence Spectroscopy Light can be absorbed and re-emitted by matter luminescence (photo-luminescence). There are two types of luminescence, in this discussion: fluorescence and phosphorescence. A
Fluorescence Spectroscopy Light can be absorbed and re-emitted by matter luminescence (photo-luminescence). There are two types of luminescence, in this discussion: fluorescence and phosphorescence. A
Electronic Spectra of Complexes
 Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
UV-Vis Spectroscopy. Chem 744 Spring Gregory R. Cook, NDSU Thursday, February 14, 13
 UV-Vis Spectroscopy Chem 744 Spring 2013 UV-Vis Spectroscopy Every organic molecule absorbs UV-visible light Energy of electronic transitions saturated functionality not in region that is easily accessible
UV-Vis Spectroscopy Chem 744 Spring 2013 UV-Vis Spectroscopy Every organic molecule absorbs UV-visible light Energy of electronic transitions saturated functionality not in region that is easily accessible
CHEM Outline (Part 15) - Luminescence 2013
 CHEM 524 -- Outline (Part 15) - Luminescence 2013 XI. Molecular Luminescence Spectra (Chapter 15) Kinetic process, competing pathways fluorescence, phosphorescence, non-radiative decay Jablonski diagram
CHEM 524 -- Outline (Part 15) - Luminescence 2013 XI. Molecular Luminescence Spectra (Chapter 15) Kinetic process, competing pathways fluorescence, phosphorescence, non-radiative decay Jablonski diagram
Chem Homework Set Answers
 Chem 310 th 4 Homework Set Answers 1. Cyclohexanone has a strong infrared absorption peak at a wavelength of 5.86 µm. (a) Convert the wavelength to wavenumber.!6!1 8* = 1/8 = (1/5.86 µm)(1 µm/10 m)(1 m/100
Chem 310 th 4 Homework Set Answers 1. Cyclohexanone has a strong infrared absorption peak at a wavelength of 5.86 µm. (a) Convert the wavelength to wavenumber.!6!1 8* = 1/8 = (1/5.86 µm)(1 µm/10 m)(1 m/100
Ultraviolet-Visible Spectroscopy
 Ultraviolet-Visible Spectroscopy Introduction to UV-Visible Absorption spectroscopy from 160 nm to 780 nm Measurement of transmittance Conversion to absorbance * A=-logT=εbc Measurement of transmittance
Ultraviolet-Visible Spectroscopy Introduction to UV-Visible Absorption spectroscopy from 160 nm to 780 nm Measurement of transmittance Conversion to absorbance * A=-logT=εbc Measurement of transmittance
Fluorescence Polarization Anisotropy FPA
 Fluorescence Polarization Anisotropy FPA Optics study of light Spectroscopy = light interacts the study of the interaction between matter & electro-magnetic radiation matter Spectroscopy Atomic Spectroscopy
Fluorescence Polarization Anisotropy FPA Optics study of light Spectroscopy = light interacts the study of the interaction between matter & electro-magnetic radiation matter Spectroscopy Atomic Spectroscopy
Fluorescence Workshop UMN Physics June 8-10, 2006 Quantum Yield and Polarization (1) Joachim Mueller
 Fluorescence Workshop UMN Physics June 8-10, 2006 Quantum Yield and Polarization (1) Joachim Mueller Quantum yield, polarized light, dipole moment, photoselection, dipole radiation, polarization and anisotropy
Fluorescence Workshop UMN Physics June 8-10, 2006 Quantum Yield and Polarization (1) Joachim Mueller Quantum yield, polarized light, dipole moment, photoselection, dipole radiation, polarization and anisotropy
1. Transition dipole moment
 1. Transition dipole moment You have measured absorption spectra of aqueous (n=1.33) solutions of two different chromophores (A and B). The concentrations of the solutions were the same. The absorption
1. Transition dipole moment You have measured absorption spectra of aqueous (n=1.33) solutions of two different chromophores (A and B). The concentrations of the solutions were the same. The absorption
A very brief history of the study of light
 1. Sir Isaac Newton 1672: A very brief history of the study of light Showed that the component colors of the visible portion of white light can be separated through a prism, which acts to bend the light
1. Sir Isaac Newton 1672: A very brief history of the study of light Showed that the component colors of the visible portion of white light can be separated through a prism, which acts to bend the light
Linear and nonlinear spectroscopy
 Linear and nonlinear spectroscopy We ve seen that we can determine molecular frequencies and dephasing rates (for electronic, vibrational, or spin degrees of freedom) from frequency-domain or timedomain
Linear and nonlinear spectroscopy We ve seen that we can determine molecular frequencies and dephasing rates (for electronic, vibrational, or spin degrees of freedom) from frequency-domain or timedomain
Application of IR Raman Spectroscopy
 Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Chapter 6 Photoluminescence Spectroscopy
 Chapter 6 Photoluminescence Spectroscopy Course Code: SSCP 4473 Course Name: Spectroscopy & Materials Analysis Sib Krishna Ghoshal (PhD) Advanced Optical Materials Research Group Physics Department, Faculty
Chapter 6 Photoluminescence Spectroscopy Course Code: SSCP 4473 Course Name: Spectroscopy & Materials Analysis Sib Krishna Ghoshal (PhD) Advanced Optical Materials Research Group Physics Department, Faculty
two slits and 5 slits
 Electronic Spectroscopy 2015January19 1 1. UV-vis spectrometer 1.1. Grating spectrometer 1.2. Single slit: 1.2.1. I diffracted intensity at relative to un-diffracted beam 1.2.2. I - intensity of light
Electronic Spectroscopy 2015January19 1 1. UV-vis spectrometer 1.1. Grating spectrometer 1.2. Single slit: 1.2.1. I diffracted intensity at relative to un-diffracted beam 1.2.2. I - intensity of light
Fluorescence Spectroscopy
 Fluorescence Spectroscopy Frequency and time dependent emission Emission and Excitation fluorescence spectra Stokes Shift: influence of molecular vibrations and solvent Time resolved fluorescence measurements
Fluorescence Spectroscopy Frequency and time dependent emission Emission and Excitation fluorescence spectra Stokes Shift: influence of molecular vibrations and solvent Time resolved fluorescence measurements
PHOTOCHEMISTRY NOTES - 1 -
 - 1 - PHOTOCHEMISTRY NOTES 1 st Law (Grotthus-Draper Law) Only absorbed radiation produces chemical change. Exception inelastic scattering of X- or γ-rays (electronic Raman effect). 2 nd Law (Star-Einstein
- 1 - PHOTOCHEMISTRY NOTES 1 st Law (Grotthus-Draper Law) Only absorbed radiation produces chemical change. Exception inelastic scattering of X- or γ-rays (electronic Raman effect). 2 nd Law (Star-Einstein
Skoog Chapter 6 Introduction to Spectrometric Methods
 Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
Measurement Examples. Excitation and Emission Scans. Steady State Fluorescence Anisotropy. Kinetic Measurements
 Measurement Examples A division of Edinburgh Instruments Ltd. Excitation and Emission Scans Excitation and emission spectra are standard measurements in fluorescence spectroscopy. The figure demonstrates
Measurement Examples A division of Edinburgh Instruments Ltd. Excitation and Emission Scans Excitation and emission spectra are standard measurements in fluorescence spectroscopy. The figure demonstrates
Visible and IR Absorption Spectroscopy. Andrew Rouff and Kyle Chau
 Visible and IR Absorption Spectroscopy Andrew Rouff and Kyle Chau The Basics wavelength= (λ) original intensity= Ι o sample slab thickness= dl Final intensity= I f ε = molar extinction coefficient -di=
Visible and IR Absorption Spectroscopy Andrew Rouff and Kyle Chau The Basics wavelength= (λ) original intensity= Ι o sample slab thickness= dl Final intensity= I f ε = molar extinction coefficient -di=
Molecular spectroscopy
 Molecular spectroscopy Origin of spectral lines = absorption, emission and scattering of a photon when the energy of a molecule changes: rad( ) M M * rad( ' ) ' v' 0 0 absorption( ) emission ( ) scattering
Molecular spectroscopy Origin of spectral lines = absorption, emission and scattering of a photon when the energy of a molecule changes: rad( ) M M * rad( ' ) ' v' 0 0 absorption( ) emission ( ) scattering
Infrared Spectroscopy
 Infrared Spectroscopy Introduction Spectroscopy is an analytical technique which helps determine structure. It destroys little or no sample. The amount of light absorbed by the sample is measured as wavelength
Infrared Spectroscopy Introduction Spectroscopy is an analytical technique which helps determine structure. It destroys little or no sample. The amount of light absorbed by the sample is measured as wavelength
Molecular Luminescence Spectroscopy
 Molecular Luminescence Spectroscopy In Molecular Luminescence Spectrometry ( MLS ), molecules of the analyte in solution are excited to give a species whose emission spectrum provides information for qualitative
Molecular Luminescence Spectroscopy In Molecular Luminescence Spectrometry ( MLS ), molecules of the analyte in solution are excited to give a species whose emission spectrum provides information for qualitative
Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy
 Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy Section I Q1. Answer (i) (b) (ii) (d) (iii) (c) (iv) (c) (v) (a) (vi) (b) (vii) (b) (viii) (a) (ix)
Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy Section I Q1. Answer (i) (b) (ii) (d) (iii) (c) (iv) (c) (v) (a) (vi) (b) (vii) (b) (viii) (a) (ix)
I. Proteomics by Mass Spectrometry 1. What is an internal standard and what does it accomplish analytically?
 Name I. Proteomics by Mass Spectrometry 1. What is an internal standard and what does it accomplish analytically? Internal standards are standards added intentionally to all samples, standards and blanks.
Name I. Proteomics by Mass Spectrometry 1. What is an internal standard and what does it accomplish analytically? Internal standards are standards added intentionally to all samples, standards and blanks.
UV / Visible Spectroscopy. Click icon to add picture
 UV / Visible Spectroscopy Click icon to add picture Spectroscopy It is the branch of science that deals with the study of interaction of matter with light. OR It is the branch of science that deals with
UV / Visible Spectroscopy Click icon to add picture Spectroscopy It is the branch of science that deals with the study of interaction of matter with light. OR It is the branch of science that deals with
Principles of Physical Biochemistry
 Principles of Physical Biochemistry Kensal E. van Hold e W. Curtis Johnso n P. Shing Ho Preface x i PART 1 MACROMOLECULAR STRUCTURE AND DYNAMICS 1 1 Biological Macromolecules 2 1.1 General Principles
Principles of Physical Biochemistry Kensal E. van Hold e W. Curtis Johnso n P. Shing Ho Preface x i PART 1 MACROMOLECULAR STRUCTURE AND DYNAMICS 1 1 Biological Macromolecules 2 1.1 General Principles
Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis)
 Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis) 2 2. Circular dichroism (optical activity) CD / ORD 3 3. Fluorescence spectroscopy and energy transfer Electromagnetic Spectrum Electronic Molecular
Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis) 2 2. Circular dichroism (optical activity) CD / ORD 3 3. Fluorescence spectroscopy and energy transfer Electromagnetic Spectrum Electronic Molecular
Chapter 12 Mass Spectrometry and Infrared Spectroscopy
 Organic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 12 Mass Spectrometry and Infrared Spectroscopy Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice
Organic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 12 Mass Spectrometry and Infrared Spectroscopy Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice
4. Circular Dichroism - Spectroscopy
 4. Circular Dichroism - Spectroscopy The optical rotatory dispersion (ORD) and the circular dichroism (CD) are special variations of absorption spectroscopy in the UV and VIS region of the spectrum. The
4. Circular Dichroism - Spectroscopy The optical rotatory dispersion (ORD) and the circular dichroism (CD) are special variations of absorption spectroscopy in the UV and VIS region of the spectrum. The
Paper: 12, Organic Spectroscopy Module: 5, Applications of UV spectroscopy
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy Applications of UV-visible Spectroscopy CHE_P12_M5 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy Applications of UV-visible Spectroscopy CHE_P12_M5 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction
Solutions and Non-Covalent Binding Forces
 Chapter 3 Solutions and Non-Covalent Binding Forces 3.1 Solvent and solution properties Molecules stick together using the following forces: dipole-dipole, dipole-induced dipole, hydrogen bond, van der
Chapter 3 Solutions and Non-Covalent Binding Forces 3.1 Solvent and solution properties Molecules stick together using the following forces: dipole-dipole, dipole-induced dipole, hydrogen bond, van der
Chemistry Instrumental Analysis Lecture 3. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 3 Quantum Transitions The energy of a photon can also be transferred to an elementary particle by adsorption if the energy of the photon exactly matches the
Chemistry 4631 Instrumental Analysis Lecture 3 Quantum Transitions The energy of a photon can also be transferred to an elementary particle by adsorption if the energy of the photon exactly matches the
Infrared Spectroscopy: Identification of Unknown Substances
 Infrared Spectroscopy: Identification of Unknown Substances Suppose a white powder is one of the four following molecules. How can they be differentiated? H N N H H H H Na H H H H H A technique that is
Infrared Spectroscopy: Identification of Unknown Substances Suppose a white powder is one of the four following molecules. How can they be differentiated? H N N H H H H Na H H H H H A technique that is
Chap. 12 Photochemistry
 Chap. 12 Photochemistry Photochemical processes Jablonski diagram 2nd singlet excited state 3rd triplet excited state 1st singlet excited state 2nd triplet excited state 1st triplet excited state Ground
Chap. 12 Photochemistry Photochemical processes Jablonski diagram 2nd singlet excited state 3rd triplet excited state 1st singlet excited state 2nd triplet excited state 1st triplet excited state Ground
Reflection = EM strikes a boundary between two media differing in η and bounces back
 Reflection = EM strikes a boundary between two media differing in η and bounces back Incident ray θ 1 θ 2 Reflected ray Medium 1 (air) η = 1.00 Medium 2 (glass) η = 1.50 Specular reflection = situation
Reflection = EM strikes a boundary between two media differing in η and bounces back Incident ray θ 1 θ 2 Reflected ray Medium 1 (air) η = 1.00 Medium 2 (glass) η = 1.50 Specular reflection = situation
Introduction. The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants
 Introduction The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants Spectroscopy and the Electromagnetic Spectrum Unlike mass spectrometry,
Introduction The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants Spectroscopy and the Electromagnetic Spectrum Unlike mass spectrometry,
Advanced Pharmaceutical Analysis
 Lecture 2 Advanced Pharmaceutical Analysis IR spectroscopy Dr. Baraa Ramzi Infrared Spectroscopy It is a powerful tool for identifying pure organic and inorganic compounds. Every molecular compound has
Lecture 2 Advanced Pharmaceutical Analysis IR spectroscopy Dr. Baraa Ramzi Infrared Spectroscopy It is a powerful tool for identifying pure organic and inorganic compounds. Every molecular compound has
Circular Dichroism & Optical Rotatory Dispersion. Proteins (KCsa) Polysaccharides (agarose) DNA CHEM 305. Many biomolecules are α-helical!
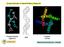 Circular Dichroism & Optical Rotatory Dispersion Polysaccharides (agarose) DNA Proteins (KCsa) Many biomolecules are α-helical! How can we measure the amount and changes in amount of helical structure
Circular Dichroism & Optical Rotatory Dispersion Polysaccharides (agarose) DNA Proteins (KCsa) Many biomolecules are α-helical! How can we measure the amount and changes in amount of helical structure
What the Einstein Relations Tell Us
 What the Einstein Relations Tell Us 1. The rate of spontaneous emission A21 is proportional to υ 3. At higher frequencies A21 >> B(υ) and all emission is spontaneous. A 21 = 8π hν3 c 3 B(ν) 2. Although
What the Einstein Relations Tell Us 1. The rate of spontaneous emission A21 is proportional to υ 3. At higher frequencies A21 >> B(υ) and all emission is spontaneous. A 21 = 8π hν3 c 3 B(ν) 2. Although
Lambert s law. Beer s law. di x / I x = -kdx (-di x = k I x dx) = - a c dx. I/I 0 = e -kl T = A = - log (T) = - log (I/I 0 )
 di x / I x = -kdx (-di x = k I x dx) Integrating this equation from x=0 ~ l (I x =I 0 ~I) gives ; ln I ln I 0 = -kl ln I/I 0 = -kl Expressing the number of photons absorbed by the slab as di x, and the
di x / I x = -kdx (-di x = k I x dx) Integrating this equation from x=0 ~ l (I x =I 0 ~I) gives ; ln I ln I 0 = -kl ln I/I 0 = -kl Expressing the number of photons absorbed by the slab as di x, and the
Chemistry Instrumental Analysis Lecture 11. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 11 Molar Absorptivities Range 0 to 10 5 Magnitude of e depends on capture cross section of the species and probability of the energy-absorbing transition. e
Chemistry 4631 Instrumental Analysis Lecture 11 Molar Absorptivities Range 0 to 10 5 Magnitude of e depends on capture cross section of the species and probability of the energy-absorbing transition. e
Analytical Technologies in Biotechnology Prof. Dr. Ashwani K Sharma Department of Biotechnology Indian Institute of Technology, Roorkee
 Analytical Technologies in Biotechnology Prof. Dr. Ashwani K Sharma Department of Biotechnology Indian Institute of Technology, Roorkee Module - 6 Spectroscopic Techniques Lecture - 2 UV-Visible Spectroscopy
Analytical Technologies in Biotechnology Prof. Dr. Ashwani K Sharma Department of Biotechnology Indian Institute of Technology, Roorkee Module - 6 Spectroscopic Techniques Lecture - 2 UV-Visible Spectroscopy
Fluorescence polarisation, anisotropy FRAP
 Fluorescence polarisation, anisotropy FRAP Reminder: fluorescence spectra Definitions! a. Emission sp. b. Excitation sp. Stokes-shift The difference (measured in nm) between the peak of the excitation
Fluorescence polarisation, anisotropy FRAP Reminder: fluorescence spectra Definitions! a. Emission sp. b. Excitation sp. Stokes-shift The difference (measured in nm) between the peak of the excitation
Advanced Analytical Chemistry
 84.514 Advanced Analytical Chemistry Part III Molecular Spectroscopy (continued) Website http://faculty.uml.edu/david_ryan/84.514 http://www.cem.msu.edu/~reusch/virtualtext/ Spectrpy/UV-Vis/spectrum.htm
84.514 Advanced Analytical Chemistry Part III Molecular Spectroscopy (continued) Website http://faculty.uml.edu/david_ryan/84.514 http://www.cem.msu.edu/~reusch/virtualtext/ Spectrpy/UV-Vis/spectrum.htm
SPECTROSCOPY MEASURES THE INTERACTION BETWEEN LIGHT AND MATTER
 SPECTROSCOPY MEASURES THE INTERACTION BETWEEN LIGHT AND MATTER c = c: speed of light 3.00 x 10 8 m/s (lamda): wavelength (m) (nu): frequency (Hz) Increasing E (J) Increasing (Hz) E = h h - Planck s constant
SPECTROSCOPY MEASURES THE INTERACTION BETWEEN LIGHT AND MATTER c = c: speed of light 3.00 x 10 8 m/s (lamda): wavelength (m) (nu): frequency (Hz) Increasing E (J) Increasing (Hz) E = h h - Planck s constant
Chapter 4 Ultraviolet and visible spectroscopy Molecular Spectrophotometry
 Chapter 4 Ultraviolet and visible spectroscopy Molecular Spectrophotometry Properties of light Electromagnetic radiation and electromagnetic spectrum Absorption of light Beer s law Limitation of Beer s
Chapter 4 Ultraviolet and visible spectroscopy Molecular Spectrophotometry Properties of light Electromagnetic radiation and electromagnetic spectrum Absorption of light Beer s law Limitation of Beer s
R O Y G B V. Spin States. Outer Shell Electrons. Molecular Rotations. Inner Shell Electrons. Molecular Vibrations. Nuclear Transitions
 Spin States Molecular Rotations Molecular Vibrations Outer Shell Electrons Inner Shell Electrons Nuclear Transitions NMR EPR Microwave Absorption Spectroscopy Infrared Absorption Spectroscopy UV-vis Absorption,
Spin States Molecular Rotations Molecular Vibrations Outer Shell Electrons Inner Shell Electrons Nuclear Transitions NMR EPR Microwave Absorption Spectroscopy Infrared Absorption Spectroscopy UV-vis Absorption,
Chemistry 524--Final Exam--Keiderling May 4, :30 -?? pm SES
 Chemistry 524--Final Exam--Keiderling May 4, 2011 3:30 -?? pm -- 4286 SES Please answer all questions in the answer book provided. Calculators, rulers, pens and pencils are permitted. No open books or
Chemistry 524--Final Exam--Keiderling May 4, 2011 3:30 -?? pm -- 4286 SES Please answer all questions in the answer book provided. Calculators, rulers, pens and pencils are permitted. No open books or
12. Structure Determination: Mass Spectrometry and Infrared Spectroscopy
 12. Structure Determination: Mass Spectrometry and Infrared Spectroscopy Determining the Structure of an Organic Compound The analysis of the outcome of a reaction requires that we know the full structure
12. Structure Determination: Mass Spectrometry and Infrared Spectroscopy Determining the Structure of an Organic Compound The analysis of the outcome of a reaction requires that we know the full structure
7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text , , 12.10)
 2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
Spectroscopy Chapter 13
 Spectroscopy Chapter 13 Electromagnetic Spectrum Electromagnetic spectrum in terms of wavelength, frequency and Energy c=λν c= speed of light in a vacuum 3x108 m/s v= frequency in Hertz (Hz s-1 ) λ= wavelength
Spectroscopy Chapter 13 Electromagnetic Spectrum Electromagnetic spectrum in terms of wavelength, frequency and Energy c=λν c= speed of light in a vacuum 3x108 m/s v= frequency in Hertz (Hz s-1 ) λ= wavelength
Fluorescence Resonance Energy Transfer (FRET) Microscopy
 Fluorescence Resonance Energy Transfer () Microscopy Mike Lorenz Optical Technology Development mlorenz@mpi-cbg.de -FLM course, May 2009 What is fluorescence? Stoke s shift Fluorescence light is always
Fluorescence Resonance Energy Transfer () Microscopy Mike Lorenz Optical Technology Development mlorenz@mpi-cbg.de -FLM course, May 2009 What is fluorescence? Stoke s shift Fluorescence light is always
/2Mα 2 α + V n (R)] χ (R) = E υ χ υ (R)
![/2Mα 2 α + V n (R)] χ (R) = E υ χ υ (R) /2Mα 2 α + V n (R)] χ (R) = E υ χ υ (R)](/thumbs/75/72875547.jpg) Spectroscopy: Engel Chapter 18 XIV 67 Vibrational Spectroscopy (Typically IR and Raman) Born-Oppenheimer approx. separate electron-nuclear Assume elect-nuclear motion separate, full wave fct. ψ (r,r) =
Spectroscopy: Engel Chapter 18 XIV 67 Vibrational Spectroscopy (Typically IR and Raman) Born-Oppenheimer approx. separate electron-nuclear Assume elect-nuclear motion separate, full wave fct. ψ (r,r) =
Fluorescence Excitation and Emission Fundamentals
 Fluorescence Excitation and Emission Fundamentals Fluorescence is a member of the ubiquitous luminescence family of processes in which susceptible molecules emit light from electronically excited states
Fluorescence Excitation and Emission Fundamentals Fluorescence is a member of the ubiquitous luminescence family of processes in which susceptible molecules emit light from electronically excited states
Lecture 5. Anisotropy decay/data analysis. Enrico Gratton
 Lecture 5. Anisotropy decay/data analysis Enrico Gratton Anisotropy decay Energy-transfer distance distributions Time resolved spectra Excited-state reactions Basic physics concept in polarization The
Lecture 5. Anisotropy decay/data analysis Enrico Gratton Anisotropy decay Energy-transfer distance distributions Time resolved spectra Excited-state reactions Basic physics concept in polarization The
( ) x10 8 m. The energy in a mole of 400 nm photons is calculated by: ' & sec( ) ( & % ) 6.022x10 23 photons' E = h! = hc & 6.
 Introduction to Spectroscopy Spectroscopic techniques are widely used to detect molecules, to measure the concentration of a species in solution, and to determine molecular structure. For proteins, most
Introduction to Spectroscopy Spectroscopic techniques are widely used to detect molecules, to measure the concentration of a species in solution, and to determine molecular structure. For proteins, most
Welcome to Organic Chemistry II
 Welcome to Organic Chemistry II Erika Bryant, Ph.D. erika.bryant@hccs.edu Class Syllabus 3 CHAPTER 12: STRUCTURE DETERMINATION 4 What is this solution Soda Tea Coffee??? 5 What is this solution Soda Tea
Welcome to Organic Chemistry II Erika Bryant, Ph.D. erika.bryant@hccs.edu Class Syllabus 3 CHAPTER 12: STRUCTURE DETERMINATION 4 What is this solution Soda Tea Coffee??? 5 What is this solution Soda Tea
Contents. xiii. Preface v
 Contents Preface Chapter 1 Biological Macromolecules 1.1 General PrincipIes 1.1.1 Macrornolecules 1.2 1.1.2 Configuration and Conformation Molecular lnteractions in Macromolecular Structures 1.2.1 Weak
Contents Preface Chapter 1 Biological Macromolecules 1.1 General PrincipIes 1.1.1 Macrornolecules 1.2 1.1.2 Configuration and Conformation Molecular lnteractions in Macromolecular Structures 1.2.1 Weak
Lecture 0. NC State University
 Chemistry 736 Lecture 0 Overview NC State University Overview of Spectroscopy Electronic states and energies Transitions between states Absorption and emission Electronic spectroscopy Instrumentation Concepts
Chemistry 736 Lecture 0 Overview NC State University Overview of Spectroscopy Electronic states and energies Transitions between states Absorption and emission Electronic spectroscopy Instrumentation Concepts
Supplemental Materials and Methods
 Supplemental Materials and Methods Time-resolved FRET (trfret) to probe for changes in the Box A/A stem upon complex assembly U3 MINI was folded and the decay of Fl fluorescence was measured at 20 ºC (see
Supplemental Materials and Methods Time-resolved FRET (trfret) to probe for changes in the Box A/A stem upon complex assembly U3 MINI was folded and the decay of Fl fluorescence was measured at 20 ºC (see
Spectroscopy in frequency and time domains
 5.35 Module 1 Lecture Summary Fall 1 Spectroscopy in frequency and time domains Last time we introduced spectroscopy and spectroscopic measurement. I. Emphasized that both quantum and classical views of
5.35 Module 1 Lecture Summary Fall 1 Spectroscopy in frequency and time domains Last time we introduced spectroscopy and spectroscopic measurement. I. Emphasized that both quantum and classical views of
Ultraviolet-Visible and Infrared Spectrophotometry
 Ultraviolet-Visible and Infrared Spectrophotometry Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11451
Ultraviolet-Visible and Infrared Spectrophotometry Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11451
ULTRAVIOLET SPECTROSCOPY or ELECTRONIC SPECTROSCOPY
 ULTRAVILET SPECTRSCPY or ELECTRNIC SPECTRSCPY S. SANKARARAMAN Department of Chemistry Indian Institute of Technology Madras Chennai 600036, INDIA Sanka@iitm.ac.in Absorption of electromagnetic radiation
ULTRAVILET SPECTRSCPY or ELECTRNIC SPECTRSCPY S. SANKARARAMAN Department of Chemistry Indian Institute of Technology Madras Chennai 600036, INDIA Sanka@iitm.ac.in Absorption of electromagnetic radiation
CHEM*3440. Photon Energy Units. Spectrum of Electromagnetic Radiation. Chemical Instrumentation. Spectroscopic Experimental Concept.
 Spectrum of Electromagnetic Radiation Electromagnetic radiation is light. Different energy light interacts with different motions in molecules. CHEM*344 Chemical Instrumentation Topic 7 Spectrometry Radiofrequency
Spectrum of Electromagnetic Radiation Electromagnetic radiation is light. Different energy light interacts with different motions in molecules. CHEM*344 Chemical Instrumentation Topic 7 Spectrometry Radiofrequency
Calculate a rate given a species concentration change.
 Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Optics and Spectroscopy
 Introduction to Optics and Spectroscopy beyond the diffraction limit Chi Chen 陳祺 Research Center for Applied Science, Academia Sinica 2015Apr09 1 Light and Optics 2 Light as Wave Application 3 Electromagnetic
Introduction to Optics and Spectroscopy beyond the diffraction limit Chi Chen 陳祺 Research Center for Applied Science, Academia Sinica 2015Apr09 1 Light and Optics 2 Light as Wave Application 3 Electromagnetic
Luminescence. Photoluminescence (PL) is luminescence that results from optically exciting a sample.
 Luminescence Topics Radiative transitions between electronic states Absorption and Light emission (spontaneous, stimulated) Excitons (singlets and triplets) Franck-Condon shift(stokes shift) and vibrational
Luminescence Topics Radiative transitions between electronic states Absorption and Light emission (spontaneous, stimulated) Excitons (singlets and triplets) Franck-Condon shift(stokes shift) and vibrational
An Introduction to Diffraction and Scattering. School of Chemistry The University of Sydney
 An Introduction to Diffraction and Scattering Brendan J. Kennedy School of Chemistry The University of Sydney 1) Strong forces 2) Weak forces Types of Forces 3) Electromagnetic forces 4) Gravity Types
An Introduction to Diffraction and Scattering Brendan J. Kennedy School of Chemistry The University of Sydney 1) Strong forces 2) Weak forces Types of Forces 3) Electromagnetic forces 4) Gravity Types
Chemistry 2. Molecular Photophysics
 Chemistry 2 Lecture 12 Molecular Photophysics Assumed knowledge Electronic states are labelled using their spin multiplicity with singlets having all electron spins paired and triplets having two unpaired
Chemistry 2 Lecture 12 Molecular Photophysics Assumed knowledge Electronic states are labelled using their spin multiplicity with singlets having all electron spins paired and triplets having two unpaired
Circular Dichroism. For students of HI Computational Structural Biology
 T H E U N I V E R S I T Y of T E X A S S C H O O L O F H E A L T H I N F O R M A T I O N S C I E N C E S A T H O U S T O N Circular Dichroism For students of HI 6001-125 Computational Structural Biology
T H E U N I V E R S I T Y of T E X A S S C H O O L O F H E A L T H I N F O R M A T I O N S C I E N C E S A T H O U S T O N Circular Dichroism For students of HI 6001-125 Computational Structural Biology
Reference literature. (See: CHEM 2470 notes, Module 8 Textbook 6th ed., Chapters )
 September 17, 2018 Reference literature (See: CHEM 2470 notes, Module 8 Textbook 6th ed., Chapters 13-14 ) Reference.: https://slideplayer.com/slide/8354408/ Spectroscopy Usual Wavelength Type of Quantum
September 17, 2018 Reference literature (See: CHEM 2470 notes, Module 8 Textbook 6th ed., Chapters 13-14 ) Reference.: https://slideplayer.com/slide/8354408/ Spectroscopy Usual Wavelength Type of Quantum
UV-Vis Absorption Experiment 5: Beer- Lambert Law and the Temperature Dependence of the Crystal Violet- Sodium Hydroxide Reaction
 1 UV-Vis Absorption Experiment 5: Beer- Lambert Law and the Temperature Dependence of the Crystal Violet- Sodium Hydroxide Reaction Overview In Part A of this experiment, the absorption behaviour of crystal
1 UV-Vis Absorption Experiment 5: Beer- Lambert Law and the Temperature Dependence of the Crystal Violet- Sodium Hydroxide Reaction Overview In Part A of this experiment, the absorption behaviour of crystal
4.3A: Electronic transitions
 Ashley Robison My Preferences Site Tools Popular pages MindTouch User Guide FAQ Sign Out If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it
Ashley Robison My Preferences Site Tools Popular pages MindTouch User Guide FAQ Sign Out If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it
CHAPTER 7 SUMMARY OF THE PRESENT WORK AND SUGGESTIONS FOR FUTURE WORK
 161 CHAPTER 7 SUMMARY OF THE PRESENT WORK AND SUGGESTIONS FOR FUTURE WORK 7.1 SUMMARY OF THE PRESENT WORK Nonlinear optical materials are required in a wide range of important applications, such as optical
161 CHAPTER 7 SUMMARY OF THE PRESENT WORK AND SUGGESTIONS FOR FUTURE WORK 7.1 SUMMARY OF THE PRESENT WORK Nonlinear optical materials are required in a wide range of important applications, such as optical
Assumed knowledge. Chemistry 2. Learning outcomes. Electronic spectroscopy of polyatomic molecules. Franck-Condon Principle (reprise)
 Chemistry 2 Lecture 11 Electronic spectroscopy of polyatomic molecules Assumed knowledge For bound excited states, transitions to the individual vibrational levels of the excited state are observed with
Chemistry 2 Lecture 11 Electronic spectroscopy of polyatomic molecules Assumed knowledge For bound excited states, transitions to the individual vibrational levels of the excited state are observed with
NH 2. Biochemistry I, Fall Term Sept 9, Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter
 Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Introduction ENERGY. Heat Electricity Electromagnetic irradiation (light)
 Photochemistry Introduction ENERGY Heat Electricity Electromagnetic irradiation (light) Vision: Triggered by a photochemical reaction Is red in the dark? The answer must be NO - Since what we see as colour
Photochemistry Introduction ENERGY Heat Electricity Electromagnetic irradiation (light) Vision: Triggered by a photochemical reaction Is red in the dark? The answer must be NO - Since what we see as colour
Fluorescence Spectroscopy
 Fluorescence Spectroscopy Steady State and Time Dependent Fluorescence Measurements Kai Wen Teng PHYS 403 Fall 15 EM Spectrum of molecules Rotational Energy Infrared Vibrational Energy Near Infrared Electronic
Fluorescence Spectroscopy Steady State and Time Dependent Fluorescence Measurements Kai Wen Teng PHYS 403 Fall 15 EM Spectrum of molecules Rotational Energy Infrared Vibrational Energy Near Infrared Electronic
Fourier Transform Infrared Spectroscopy of Metal Ligand Complexes *
 OpenStax-CNX module: m34660 1 Fourier Transform Infrared Spectroscopy of Metal Ligand Complexes * Jiebo Li Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons
OpenStax-CNX module: m34660 1 Fourier Transform Infrared Spectroscopy of Metal Ligand Complexes * Jiebo Li Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons
Structure Determination. How to determine what compound that you have? One way to determine compound is to get an elemental analysis
 Structure Determination How to determine what compound that you have? ne way to determine compound is to get an elemental analysis -basically burn the compound to determine %C, %H, %, etc. from these percentages
Structure Determination How to determine what compound that you have? ne way to determine compound is to get an elemental analysis -basically burn the compound to determine %C, %H, %, etc. from these percentages
A prevalent intraresidue hydrogen bond stabilizes proteins
 Supplementary Information A prevalent intraresidue hydrogen bond stabilizes proteins Robert W. Newberry 1 & Ronald T. Raines 1,2 * 1 Department of Chemistry and 2 Department of Biochemistry, University
Supplementary Information A prevalent intraresidue hydrogen bond stabilizes proteins Robert W. Newberry 1 & Ronald T. Raines 1,2 * 1 Department of Chemistry and 2 Department of Biochemistry, University
CHAPTER 29 HW: AMINO ACIDS + PROTEINS
 CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015,
 Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015, Course,Informa5on, BIOC%530% GraduateAlevel,discussion,of,the,structure,,func5on,,and,chemistry,of,proteins,and, nucleic,acids,,control,of,enzyma5c,reac5ons.,please,see,the,course,syllabus,and,
Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015, Course,Informa5on, BIOC%530% GraduateAlevel,discussion,of,the,structure,,func5on,,and,chemistry,of,proteins,and, nucleic,acids,,control,of,enzyma5c,reac5ons.,please,see,the,course,syllabus,and,
Modern Optical Spectroscopy
 Modern Optical Spectroscopy With Exercises and Examples from Biophysics and Biochemistry von William W Parson 1. Auflage Springer-Verlag Berlin Heidelberg 2006 Verlag C.H. Beck im Internet: www.beck.de
Modern Optical Spectroscopy With Exercises and Examples from Biophysics and Biochemistry von William W Parson 1. Auflage Springer-Verlag Berlin Heidelberg 2006 Verlag C.H. Beck im Internet: www.beck.de
Fluorescence Depolarization. Ben Berger and Ben Berger
 Fluorescence Depolarization Ben Berger and Ben Berger Small molecules rotate quickly Large molecules rotate slowly There is a time delay from the moment a molecule is excited to the time it fluoresces
Fluorescence Depolarization Ben Berger and Ben Berger Small molecules rotate quickly Large molecules rotate slowly There is a time delay from the moment a molecule is excited to the time it fluoresces
24 Introduction to Spectrochemical Methods
 24 Introduction to Spectrochemical Methods Spectroscopic method: based on measurement of the electromagnetic radiation produced or absorbed by analytes. electromagnetic radiation: include γ-ray, X-ray,
24 Introduction to Spectrochemical Methods Spectroscopic method: based on measurement of the electromagnetic radiation produced or absorbed by analytes. electromagnetic radiation: include γ-ray, X-ray,
