Reading: Finish reading Ch. 9 if you haven't already HW - finish smartwork above and begin working on competition problems from the Ch. 9 Handout.
|
|
|
- Opal Fisher
- 5 years ago
- Views:
Transcription
1 Ch. 8 Smartwork available - due 10 AM - don't forget to check for carbocation rearrangements! Ch. 9 Smartwork available - due Saturday 11:59 PM Final Exam (Monday 8 AM here) will be comprehensive/cumulative - slightly heavier on the Ch. 8/9 material than on other material. The exam is written by me and is up to 50 multiple choice questions (MC only). Some Ch. 8/9 old test questions available on Exam website. Be sure to utilize the Study Guides available on the website. Model kit allowed. No calculator needed. Last SI Sessions tonight & tomorrow night. Final Exam Cram is Wed 12/6 from 7-9 PM in Wiggins library 3rd floor My office hours this week: Today 12-1 PM, Tuesday 12-1 PM, Wednesday 1:30-2:30 PM, Friday 11 am - 12 pm, or by appointment. Reading: Finish reading Ch. 9 if you haven't already HW - finish smartwork above and begin working on competition problems from the Ch. 9 Handout.
2 The First Part of the Frontier Orbital Interactions of an E2 Step Ref: Karty, Interchapter 1, p.394
3 The Second Part of the Frontier Orbital Interactions of an E2 Step Ref: Karty, Interchapter 1, p.394
4 Basic Conditions
5 Reasonable Mech. In Basic Cond.
6 Acidic Conditions
7 Reasonable Mechanism for Acidic Cond.
8 Favor E2, Hoffman product Strong, Bulky Bases
9 Water Leaving Group Mechanism The leaving group leaves as H 2 O, not HO. Being uncharged, H 2 O is much more stable than HO, and is an excellent leaving group.
10 S N 1 Mechanisms/Rate Determining Steps What favors each mechanism? S N 2 E1 E2
11 Chapter 9 Cheat Sheet Nucelophile vs. Base: Great Leaving Groups: Rule of thumb - the weaker (more stable) the base, the better the LG ALSO WATER AND ALCOHOLS OR OTHER NEUTRAL MOLECULES ARE GREAT LEAVING GROUPS Solvents:
12 Chapter 9: Competition between S N 1, S N 2, E1, and E2 A decision pathway 1. First consider the substrate. If the carbon with the leaving group is: a. Methyl (0 ), the reaction will be S N 2. b. Primary (1 ), most likely S N 2. Exceptions: a) if a strong, bulky base, E2 would be favored. b) if allylic or benzylic primary, then could be S N 1 or E1. c. Secondary (2 ) can t rule any mechanism out, but usually S N 2 or E2 unless the base or nucleophile is a weak one, though allylic or benzylic may encourage more S N 1/E1. d. Tertiary (3 ), rule S N2 out (too sterically hindered for S N2), but all others are fair game. Consider base/nuc strength and temperature. Other substrate-related issues: S N2 is fastest with the LEAST substituted substrates fastest with 0 or 1 (sterics) E2 is fastest with 3 substrate because of + charge buildup at the carbon during the transition state. S N 1 and E1 are fastest w/ tertiary, benzylic or allylic substrates. 2. Second, consider the quality & strength of the other reagent (the nucleophile or base the reagent that is not the substrate w/leaving group). Note that some reagents can act as nucleophiles or bases. a. General Nucleophile Strength: Stronger Nucleophiles favor S N 2 (PUSH off the leaving group). Weaker nucleophiles favor S N 1. Nucleophile strength is solvent-dependent b. Base Strength: We are familiar with base strength from Ch. 6 - review the pka Chart as necessary. Strong bases favor E2 (attack the substrate with the leaving group still attached) over E1. Weak bases favor E1. The bases below, or any other very strong, bulky bases tend to favor E2 and the Hofmann (less substituted) product over the Zaitsev (more substituted) product (regiochemistry effect)
13 3. If there is a reagent that can act as both a strong base and a strong nucleophile, and the substrate is secondary (2 ), the reaction will likely favor the E2 mechanism, unless a low or moderate temperature is indicated (more on temp next). 4. Consider temperature. If all else is equal, heat favors elimination over substitution. ( S rxn is more positive for an elimination reaction than for a substitution reaction.) 5. Consider the Solvent. 6. Consider the Leaving Group (LG). The best leaving groups are neutral molecules or are stable with a negative charge. S N 1 and E1 reactions need good leaving groups. S N 2 and E2 reactions can work with good or moderate leaving groups. All reactions work better with better leaving groups, though. 7. Consider the concentration. A high concentration of the base/nucleophile favors the bimolecular reactions (S N 2 and E2) because the base or nucleophile are part of the rate law for these reactions. Low concentration of the base/nuc favors the unimolecular reactions (S N 1 and E1), because the base or nucleophile are not part of the rate law for these reactions. Concentration is not very often indicated, so it is a little unusual to use it as a determining factor.
14 Organic Chemistry I - Ch. 9 S N 2/S N 1/E2/E1 Competition Problems Draw the Major Product(s) for each reaction, given correct stereo- and regio- chemistry, as appropriate 1) 2) 3) 4) 5) 6) 7)
15 8) 9) 10) 11) 12) 13)
Organic Chemistry CHM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl Halides: Substitution Reactions - Chapter 6 (Wade)
 rganic Chemistry CM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl alides: Substitution Reactions - Chapter 6 (Wade) Chapter utline I. Intro to RX (6-1 - 6-7) II. Substitution Reactions A) S N 2 (6-8,
rganic Chemistry CM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl alides: Substitution Reactions - Chapter 6 (Wade) Chapter utline I. Intro to RX (6-1 - 6-7) II. Substitution Reactions A) S N 2 (6-8,
1. The Substrate: CH3, 1 o, 2 o, 3 o, Allyl or Benzyl
 Putting it all together: Substitution and elimination reactions are almost always in competition with each other. In order to predict the products of a reaction, it is necessary to determine which mechanisms
Putting it all together: Substitution and elimination reactions are almost always in competition with each other. In order to predict the products of a reaction, it is necessary to determine which mechanisms
Reactions of Alkyl Halides with Nucleophiles and Bases a substitution reaction
 Reactions of Alkyl Halides with Nucleophiles and Bases a substitution reaction Nucleophilic substitution and base induced elimination are among most widely occurring and versatile reaction types in organic
Reactions of Alkyl Halides with Nucleophiles and Bases a substitution reaction Nucleophilic substitution and base induced elimination are among most widely occurring and versatile reaction types in organic
Chapter 8. Substitution reactions of Alkyl Halides
 Chapter 8. Substitution reactions of Alkyl Halides There are two types of possible reaction in organic compounds in which sp 3 carbon is bonded to an electronegative atom or group (ex, halides) 1. Substitution
Chapter 8. Substitution reactions of Alkyl Halides There are two types of possible reaction in organic compounds in which sp 3 carbon is bonded to an electronegative atom or group (ex, halides) 1. Substitution
Reaction mechanism 1/17/19. HCl. HCl. 3 radical can form here. Br 2. hint!
 If the reagents or reaction conditions favor radicals, then it is likely to be a radical reaction. Look for hints like peroxide, AIBN, NBS, hn (UV light). 3 radical can form here Reaction mechanism How
If the reagents or reaction conditions favor radicals, then it is likely to be a radical reaction. Look for hints like peroxide, AIBN, NBS, hn (UV light). 3 radical can form here Reaction mechanism How
Glendale Community College Chemistry 105 Exam. 3 Lecture Notes Chapters 6 & 7
 Sevada Chamras, Ph.D. Glendale Community College Chemistry 105 Exam. 3 Lecture Notes Chapters 6 & 7 Description: Examples: 3 Major Types of Organic Halides: 1. Alkyl Halides: Chapter 6 (Part 1/2) : Alkyl
Sevada Chamras, Ph.D. Glendale Community College Chemistry 105 Exam. 3 Lecture Notes Chapters 6 & 7 Description: Examples: 3 Major Types of Organic Halides: 1. Alkyl Halides: Chapter 6 (Part 1/2) : Alkyl
The Electrophile. S N 2 and E2 least stable most stable least hindered most hindered. S N 1 and E1. > x > >
 The Electrophile 1 Recall that electrophile means electron- loving. When considering substitution and elimination reactions we must consider the carbon attached to the leaving group. Is it a primary, secondary,
The Electrophile 1 Recall that electrophile means electron- loving. When considering substitution and elimination reactions we must consider the carbon attached to the leaving group. Is it a primary, secondary,
REACTION AND SYNTHESIS REVIEW
 REACTION AND SYNTHESIS REVIEW A STUDENT SHOULD BE ABLE TO PREDICT PRODUCTS, IDENTIFY REACTANTS, GIVE REACTION CONDITIONS, PROPOSE SYNTHESES, AND PROPOSE MECHANISMS (AS LISTED BELOW). REVIEW THE MECHANISM
REACTION AND SYNTHESIS REVIEW A STUDENT SHOULD BE ABLE TO PREDICT PRODUCTS, IDENTIFY REACTANTS, GIVE REACTION CONDITIONS, PROPOSE SYNTHESES, AND PROPOSE MECHANISMS (AS LISTED BELOW). REVIEW THE MECHANISM
REACTIONS OF HALOALKANES - SUBSTITUTION AND ELIMINATION
 REACTIONS OF HALOALKANES - SUBSTITUTION AND ELIMINATION Haloalkanes (also known as halogenoalkanes and alkyl halides) are organic compounds where one of the hydrogens of an alkane or cycloalkane has been
REACTIONS OF HALOALKANES - SUBSTITUTION AND ELIMINATION Haloalkanes (also known as halogenoalkanes and alkyl halides) are organic compounds where one of the hydrogens of an alkane or cycloalkane has been
Preparation of Alkyl Halides, R-X. Reaction of alkanes with Cl 2 & Br 2 (F 2 is too reactive, I 2 is unreactive): R + X X 2.
 Preparation of Alkyl alides, R-X Reaction of alkanes with Cl 2 & Br 2 (F 2 is too reactive, I 2 is unreactive): UV R + X 2 R X or heat + X This mechanism involves a free radical chain reaction. A chain
Preparation of Alkyl alides, R-X Reaction of alkanes with Cl 2 & Br 2 (F 2 is too reactive, I 2 is unreactive): UV R + X 2 R X or heat + X This mechanism involves a free radical chain reaction. A chain
Nucleophilic Substitution and Elimination
 Nucleophilic Substitution and Elimination Alkyl halides react with a nucleophile in one of two ways. Either they eliminate an X to form an alkene, or they undergo a substitution with the nucleophile, Nu,
Nucleophilic Substitution and Elimination Alkyl halides react with a nucleophile in one of two ways. Either they eliminate an X to form an alkene, or they undergo a substitution with the nucleophile, Nu,
11. Nucleophilic Substitution Reactions
 11. Nucleophilic Substitution Reactions A. Introduction It would be beneficial if you review the chapter on substitution reactions in your textbook prior to lab. This is Ch. 11 in the 9 th edition McMurry
11. Nucleophilic Substitution Reactions A. Introduction It would be beneficial if you review the chapter on substitution reactions in your textbook prior to lab. This is Ch. 11 in the 9 th edition McMurry
Chapter 11: Nucleophilic Substitution and Elimination Walden Inversion
 hapter 11: Nucleophilic Substitution and Elimination Walden Inversion (S)-(-) Malic acid [a] D = -2.3 Ag 2, 2 Pl 5 l Ag 2, 2 ()-2-hlorosuccinic acid l (-)-2-hlorosuccinic acid Pl 5 ()-() Malic acid [a]
hapter 11: Nucleophilic Substitution and Elimination Walden Inversion (S)-(-) Malic acid [a] D = -2.3 Ag 2, 2 Pl 5 l Ag 2, 2 ()-2-hlorosuccinic acid l (-)-2-hlorosuccinic acid Pl 5 ()-() Malic acid [a]
CHAPTER 7. Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination
 CHAPTER 7 Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 7-1 Solvolysis of Tertiary and Secondary Haloalkanes The rate of S N 2 reactions decrease dramatically
CHAPTER 7 Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 7-1 Solvolysis of Tertiary and Secondary Haloalkanes The rate of S N 2 reactions decrease dramatically
Basic Organic Chemistry Course code : CHEM (Pre-requisites : CHEM 11122)
 Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
CHEM 243 ORGANIC CHEMISTRY I Fall 2018 Exam II Information and Study Guide
 CHEM 243 ORGANIC CHEMISTRY I Fall 2018 Exam II Information and Study Guide page 1 CHEM 243 Exam II is scheduled for Monday, November 5 in DMF 477 & 481 (lab rooms). You must take the exam during your normal
CHEM 243 ORGANIC CHEMISTRY I Fall 2018 Exam II Information and Study Guide page 1 CHEM 243 Exam II is scheduled for Monday, November 5 in DMF 477 & 481 (lab rooms). You must take the exam during your normal
Sn1 or Sn2 Reactions: A Guide to Deciding Which Reaction is Occurring
 Sn1 or Sn2 Reactions: A Guide to Deciding Which Reaction is Occurring The following is a discussion of the approach you should use in order to determine if a chemical reaction occurs via a Sn1 or Sn2 mechanism.
Sn1 or Sn2 Reactions: A Guide to Deciding Which Reaction is Occurring The following is a discussion of the approach you should use in order to determine if a chemical reaction occurs via a Sn1 or Sn2 mechanism.
Chapter 8: Alkene Structure and Preparation via Elimination Reactions
 1. Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C bond length bond strength 3 C C 3 3 C C 3 3 C C 3 3 C 2 C C 2 3 C a C=C double bond
1. Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C bond length bond strength 3 C C 3 3 C C 3 3 C C 3 3 C 2 C C 2 3 C a C=C double bond
Give the major organic product(s) of the following reaction.
 Give the major organic product(s) of the following reaction. 1) NaNO2, HCl 0 o C 2) CuCN 2016-09-23 Q1 A D B C E F There is no reaction or the correct product is not listed here. 5 of 7 Give the major
Give the major organic product(s) of the following reaction. 1) NaNO2, HCl 0 o C 2) CuCN 2016-09-23 Q1 A D B C E F There is no reaction or the correct product is not listed here. 5 of 7 Give the major
Chapter 5. Nucleophilic aliphatic substitution mechanism. by G.DEEPA
 Chapter 5 Nucleophilic aliphatic substitution mechanism by G.DEEPA 1 Introduction The polarity of a carbon halogen bond leads to the carbon having a partial positive charge In alkyl halides this polarity
Chapter 5 Nucleophilic aliphatic substitution mechanism by G.DEEPA 1 Introduction The polarity of a carbon halogen bond leads to the carbon having a partial positive charge In alkyl halides this polarity
Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides"
 Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides" t Introduction" The polarity of a carbon-halogen bond leads to the carbon having a partial positive charge"
Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides" t Introduction" The polarity of a carbon-halogen bond leads to the carbon having a partial positive charge"
Methyl > primary > secondary >> tertiary
 Lecture 1 Notes September 6, 2012 Welcome to CHM 427. For some of you, this will be the last organic chemistry class that you will ever take. So I see it as my personal mission to make sure that you know
Lecture 1 Notes September 6, 2012 Welcome to CHM 427. For some of you, this will be the last organic chemistry class that you will ever take. So I see it as my personal mission to make sure that you know
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2
 C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
Substitution and Elimination reactions
 PART 3 Substitution and Elimination reactions Chapter 8. Substitution reactions of RX 9. Elimination reactions of RX 10. Substit n/elimin n of other comp ds 11. Organometallic comp ds 12. Radical reactions
PART 3 Substitution and Elimination reactions Chapter 8. Substitution reactions of RX 9. Elimination reactions of RX 10. Substit n/elimin n of other comp ds 11. Organometallic comp ds 12. Radical reactions
CONCERTED sp 2 H. HO Et
 Alkyl alides Substitution and Elimination 1 Substitutions (Quick Review) 1.1 SN2 Reactions LB nucleophile "backside attack!" NERTED reaction This is fundamentally just a Lewis acid/base reaction, the Lewis
Alkyl alides Substitution and Elimination 1 Substitutions (Quick Review) 1.1 SN2 Reactions LB nucleophile "backside attack!" NERTED reaction This is fundamentally just a Lewis acid/base reaction, the Lewis
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams.
 2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions
 Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
Chapter 8: Alkene Structure and Preparation via Elimination Reactions
 Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C 3 C C 3 bond length bond strength 2 C C 2 a C=C double bond is stronger than a C C single
Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C 3 C C 3 bond length bond strength 2 C C 2 a C=C double bond is stronger than a C C single
BSc. II 3 rd Semester. Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1
 BSc. II 3 rd Semester Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1 Introduction to Alkyl Halides Alkyl halides are organic molecules containing a halogen atom bonded to an
BSc. II 3 rd Semester Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1 Introduction to Alkyl Halides Alkyl halides are organic molecules containing a halogen atom bonded to an
Chemistry Organic Chemistry II, Spring 2018
 Chemistry 2320 Organic Chemistry II, Spring 2018 Instructor: Dr. Tom Chang Office: Widtsoe 337 Phone: 797-3545 Email: tom.chang@usu.edu Meeting Time/Place: MWF 10:30-11:20 am, Eccles Business Building
Chemistry 2320 Organic Chemistry II, Spring 2018 Instructor: Dr. Tom Chang Office: Widtsoe 337 Phone: 797-3545 Email: tom.chang@usu.edu Meeting Time/Place: MWF 10:30-11:20 am, Eccles Business Building
Chapter 11, Part 1: Polar substitution reactions involving alkyl halides
 hapter 11, Part 1: Polar substitution reactions involving alkyl halides Overview: The nature of alkyl halides and other groups with electrophilic sp 3 hybridized leads them to react with nucleophiles and
hapter 11, Part 1: Polar substitution reactions involving alkyl halides Overview: The nature of alkyl halides and other groups with electrophilic sp 3 hybridized leads them to react with nucleophiles and
Elimination Reactions Heating an alkyl halide with a strong base causes elimination of a. molecule of HX
 Elimination eactions eating an alkyl halide with a strong base causes elimination of a molecule of X 1. Potassium hydroxide dissolved in ethanol and the sodium salts of alcohols (such as sodium ethoxide)
Elimination eactions eating an alkyl halide with a strong base causes elimination of a molecule of X 1. Potassium hydroxide dissolved in ethanol and the sodium salts of alcohols (such as sodium ethoxide)
Homework problems Chapters 6 and Give the curved-arrow formalism for the following reaction: CH 3 OH + H 2 C CH +
 omework problems hapters 6 and 7 1. Give the curved-arrow formalism for the following reaction: : 3 - : 2 : 3 2-3 3 2. In each of the following sets, arrange the compounds in order of decreasing pka and
omework problems hapters 6 and 7 1. Give the curved-arrow formalism for the following reaction: : 3 - : 2 : 3 2-3 3 2. In each of the following sets, arrange the compounds in order of decreasing pka and
Elimination reactions
 Chapter 9 Elimination reactions E2 and E1 reactions Competition between S N and E Elimination reactions Ch 9 #2 elimination and/or substitution 2 mechanisms ~ E2 and E1 E2: bimolecular elimination rxn
Chapter 9 Elimination reactions E2 and E1 reactions Competition between S N and E Elimination reactions Ch 9 #2 elimination and/or substitution 2 mechanisms ~ E2 and E1 E2: bimolecular elimination rxn
Chapter 8: Nucleophilic Substitution 8.1: Functional Group Transformation By Nucleophilic Substitution
 hapter 8: Nucleophilic Substitution 8.1: Functional Group Transformation By Nucleophilic Substitution Nu: = l,, I Nu - Nucleophiles are Lewis bases (electron-pair donor) Nucleophiles are often negatively
hapter 8: Nucleophilic Substitution 8.1: Functional Group Transformation By Nucleophilic Substitution Nu: = l,, I Nu - Nucleophiles are Lewis bases (electron-pair donor) Nucleophiles are often negatively
Classes of Alkenes. Alkenes and Alkynes. Saturated compounds (alkanes): Have the maximum number of hydrogen atoms attached to each carbon atom.
 Alkenes and Alkynes Saturated compounds (alkanes): ave the maximum number of hydrogen atoms attached to each carbon atom. Unsaturated compounds: ave fewer hydrogen atoms attached to the carbon chain than
Alkenes and Alkynes Saturated compounds (alkanes): ave the maximum number of hydrogen atoms attached to each carbon atom. Unsaturated compounds: ave fewer hydrogen atoms attached to the carbon chain than
11. Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations
 11. Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations Based on McMurry s Organic Chemistry, 6 th edition 2003 Ronald Kluger Department of Chemistry University of Toronto Alkyl Halides
11. Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations Based on McMurry s Organic Chemistry, 6 th edition 2003 Ronald Kluger Department of Chemistry University of Toronto Alkyl Halides
Elimination Reactions The E2 Mechanism
 Elimination Reactions The E2 Mechanism The E2 Mechanism X X- B: B- δ- B:- δ+ R 1 δ- R 2 δ+ X δ- The E2 Mechanism R 3 R 4 transition state Free energy (G) Eact B:- B R 1 R 2 X R 1 R 2 R 3 R 4 R 4 R 3 X:-
Elimination Reactions The E2 Mechanism The E2 Mechanism X X- B: B- δ- B:- δ+ R 1 δ- R 2 δ+ X δ- The E2 Mechanism R 3 R 4 transition state Free energy (G) Eact B:- B R 1 R 2 X R 1 R 2 R 3 R 4 R 4 R 3 X:-
1-What is substitution reaction? 2-What are can Nucleophilic Substitution Reaction? 3- SN1 reaction. 4-SN2 reaction 5- mechanisms of SN1&SN2
 1-What is substitution reaction? 2-What are can Nucleophilic Substitution eaction? 3- SN1 reaction. 4-SN2 reaction 5- mechanisms of SN1&SN2 1- SUBSTITUTION EACTIONS 1-Substitution eaction In this type
1-What is substitution reaction? 2-What are can Nucleophilic Substitution eaction? 3- SN1 reaction. 4-SN2 reaction 5- mechanisms of SN1&SN2 1- SUBSTITUTION EACTIONS 1-Substitution eaction In this type
ORGANIC - CLUTCH CH. 8 - ELIMINATION REACTIONS.
 !! www.clutchprep.com CONCEPT: THE E2 MECHANISM A strong nucleophile reacts with an inaccessible leaving group to produce beta-elimination in one-step. E2 Properties (Circle One) Nucleophile = Strong /
!! www.clutchprep.com CONCEPT: THE E2 MECHANISM A strong nucleophile reacts with an inaccessible leaving group to produce beta-elimination in one-step. E2 Properties (Circle One) Nucleophile = Strong /
c. Cl H Page 1 of 7 major P (E > Z and more substituted over less substituted alkene) LG must be axial are the same Cl -
 CEM 109A 1. Predict the products of the following reactions (a-c E2, d-f E1 KEY focuses only on elimination products, in most cases there will also be substitution products.) a. - LG must be axial - are
CEM 109A 1. Predict the products of the following reactions (a-c E2, d-f E1 KEY focuses only on elimination products, in most cases there will also be substitution products.) a. - LG must be axial - are
Chapter 9. Nucleophilic Substitution and ß-Elimination
 Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Organic Reactions Susbstitution S N. Dr. Sapna Gupta
 Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Ch. 2 Power Points below (including plenty that I would have shown today, but did not get to).
 Lewis Structure + Functional Group Quiz Average: 77. The quizzes are easier than the tests. If your score was 60-50 or below, need to do more practice problems/studying and/or adjust your class/work/life
Lewis Structure + Functional Group Quiz Average: 77. The quizzes are easier than the tests. If your score was 60-50 or below, need to do more practice problems/studying and/or adjust your class/work/life
Elimination Reactions. Chapter 6 1
 Elimination Reactions Chapter 6 1 E1 Mechanism Step 1: halide ion leaves, forming a carbocation. Step 2: Base abstracts H + from adjacent carbon forming the double bond. Chapter 6 2 E1 Energy Diagram E1:
Elimination Reactions Chapter 6 1 E1 Mechanism Step 1: halide ion leaves, forming a carbocation. Step 2: Base abstracts H + from adjacent carbon forming the double bond. Chapter 6 2 E1 Energy Diagram E1:
Learning Guide for Chapter 10 - Alkyl Halides II
 Learning Guide for Chapter 10 - Alkyl Halides II I. Elimination Reactions of Alkyl Halides Introduction Mechanisms Beta hydrogens, constitutional isomers, and stereoisomers E2 vs E1 Strong and Weak Bases
Learning Guide for Chapter 10 - Alkyl Halides II I. Elimination Reactions of Alkyl Halides Introduction Mechanisms Beta hydrogens, constitutional isomers, and stereoisomers E2 vs E1 Strong and Weak Bases
7. Haloalkanes (text )
 2009, Department of hemistry, The University of Western Ontario 7.1 7. aloalkanes (text 7.1 7.10) A. Structure and Nomenclature Like hydrogen, the halogens have a valence of one. Thus, a halogen atom can
2009, Department of hemistry, The University of Western Ontario 7.1 7. aloalkanes (text 7.1 7.10) A. Structure and Nomenclature Like hydrogen, the halogens have a valence of one. Thus, a halogen atom can
Chapter 8 Outline: Alkenes: Structure and Preparation via β-elimination
 Chapter 8 Outline: Alkenes: Structure and Preparation via β-elimination 1. What is β elimination? 2. Alkenes: structure, steroisomerism and stability 3. Elimination Reactions o E2 Mechanism o E1 Mechanism
Chapter 8 Outline: Alkenes: Structure and Preparation via β-elimination 1. What is β elimination? 2. Alkenes: structure, steroisomerism and stability 3. Elimination Reactions o E2 Mechanism o E1 Mechanism
Some Arrow-Pushing Guidelines (Section 1.14) 1. Arrows follow electron movement.
 Chem 350 Jasperse Ch. 1 Notes 1 Note: The headers and associated chapters don t actually jive with the textbook we are using this summer. But otherwise this highlights a lot of the chemistry from Organic
Chem 350 Jasperse Ch. 1 Notes 1 Note: The headers and associated chapters don t actually jive with the textbook we are using this summer. But otherwise this highlights a lot of the chemistry from Organic
Initials: 1. Chem 633: Advanced Organic Chemistry 2016 Final Exam
 Initials: 1 ame: Chem 633: Advanced rganic Chemistry 2016 Final Exam This exam is closed note, closed book. Please answer the following questions clearly and concisely. In general, use pictures and less
Initials: 1 ame: Chem 633: Advanced rganic Chemistry 2016 Final Exam This exam is closed note, closed book. Please answer the following questions clearly and concisely. In general, use pictures and less
Chapter 7 Substitution Reactions 7.1 Introduction to Substitution Reactions Substitution Reactions: two reactants exchange parts to give new products
 hapter 7 Substitution eactions 7.1 Introduction to Substitution eactions Substitution eactions: two reactants exchange parts to give new products A-B + -D A-D + B- 3 2 + Br 3 2 Br + Elimination eaction:
hapter 7 Substitution eactions 7.1 Introduction to Substitution eactions Substitution eactions: two reactants exchange parts to give new products A-B + -D A-D + B- 3 2 + Br 3 2 Br + Elimination eaction:
CHE 171: Mechanistic Organic Chemistry I
 CHE 171: Mechanistic Organic Chemistry I Syllabus, Autumn Quarter 2003 Instructor: Dr. Matthew R. Dintzner Office hours: Mondays 9:00-11:30 AM, Tuesdays 10:00-12:30, or by appointment Office, Lab: O'Connell
CHE 171: Mechanistic Organic Chemistry I Syllabus, Autumn Quarter 2003 Instructor: Dr. Matthew R. Dintzner Office hours: Mondays 9:00-11:30 AM, Tuesdays 10:00-12:30, or by appointment Office, Lab: O'Connell
Lab 11 Guide: Nucleophilic Substitution (Nov 10 16)
 Lab 11 Guide: Nucleophilic Substitution (Nov 10 16) Nucleophilic Substitution of Alkyl Halides, Exp. 17A, B, and C, pages 187-192 in Taber This week you will be doing examining real life S N 1 and S N
Lab 11 Guide: Nucleophilic Substitution (Nov 10 16) Nucleophilic Substitution of Alkyl Halides, Exp. 17A, B, and C, pages 187-192 in Taber This week you will be doing examining real life S N 1 and S N
FOURTH EXAMINATION. (1)..20 pts... (2)..24 pts... (3)..18 pts... (4)..12 pts... (5)..12 pts... (6).. 6 pts... (7).. 8 pts... Bonus 4 pts...
 Name: CHEM 331 FURTH EXAMINATIN All answers should be written on the exam in the spaces provided. Clearly indicate your answers in the spaces provided; if I have to guess as to what or where your answer
Name: CHEM 331 FURTH EXAMINATIN All answers should be written on the exam in the spaces provided. Clearly indicate your answers in the spaces provided; if I have to guess as to what or where your answer
10. Organohalides. Based on McMurry s Organic Chemistry, 7 th edition
 10. Organohalides Based on McMurry s Organic Chemistry, 7 th edition What Is an Alkyl Halide An organic compound containing at least one carbonhalogen bond (C-X) X (F, Cl, Br, I) replaces H Can contain
10. Organohalides Based on McMurry s Organic Chemistry, 7 th edition What Is an Alkyl Halide An organic compound containing at least one carbonhalogen bond (C-X) X (F, Cl, Br, I) replaces H Can contain
Give the major product(s) of the following reaction.
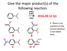 Give the major product(s) of the following reaction. HNO3 H2SO4 2016-09-12 Q1 A B E. There is no reaction or the correct product is not listed here. C D Give the major product(s) of the following reaction.
Give the major product(s) of the following reaction. HNO3 H2SO4 2016-09-12 Q1 A B E. There is no reaction or the correct product is not listed here. C D Give the major product(s) of the following reaction.
Walden discovered a series of reactions that could interconvert (-)-malic acid and (+)-malic acid.
 Chapter 11: Reactions of alkyl halides: nucleophilic substitutions and eliminations Alkyl halides are polarized in the C-X bond, making carbon δ+ (electrophilic). A nucleophilecan attack this carbon, displacing
Chapter 11: Reactions of alkyl halides: nucleophilic substitutions and eliminations Alkyl halides are polarized in the C-X bond, making carbon δ+ (electrophilic). A nucleophilecan attack this carbon, displacing
PAPER No. 05: TITLE: ORGANIC CHEMISTRY-II MODULE No. 12: TITLE: S N 1 Reactions
 Subject hemistry Paper o and Title Module o and Title Module Tag 05, ORGAI EMISTRY-II 12, S 1 Reactions E_P5_M12 EMISTRY PAPER o. 05: TITLE: ORGAI EMISTRY-II TABLE OF OTETS 1. Learning Outcomes 2. Introduction
Subject hemistry Paper o and Title Module o and Title Module Tag 05, ORGAI EMISTRY-II 12, S 1 Reactions E_P5_M12 EMISTRY PAPER o. 05: TITLE: ORGAI EMISTRY-II TABLE OF OTETS 1. Learning Outcomes 2. Introduction
PAPER No. : 5; Organic Chemistry-II MODULE No. : 13; Mixed S N 1 and S N 2 Reactions
 Subject Chemistry Paper No and Title Module No and Title Module Tag 5; Organic Chemistry-II 13; Mixed S N 1 and S N 2 Reactions CHE_P5_M13 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. Nature
Subject Chemistry Paper No and Title Module No and Title Module Tag 5; Organic Chemistry-II 13; Mixed S N 1 and S N 2 Reactions CHE_P5_M13 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. Nature
LECTURE #13 Tues., Oct.18, Midterm exam: Tues.Oct.25 during class Ch.1, , 7.10, 2, Sections
 CEM 221 section 01 LECTURE #13 Tues., Oct.18, 2005 Midterm exam: Tues.Oct.25 during class Ch.1, 7.2-7.5, 7.10, 2, 3.1-3.5 ASSGNED READNGS: TODAY S CLASS: Sections 4.1 4.6 NEXT CLASS: rest of Ch.4 http://artsandscience.concordia.ca/facstaff/p-r/rogers
CEM 221 section 01 LECTURE #13 Tues., Oct.18, 2005 Midterm exam: Tues.Oct.25 during class Ch.1, 7.2-7.5, 7.10, 2, 3.1-3.5 ASSGNED READNGS: TODAY S CLASS: Sections 4.1 4.6 NEXT CLASS: rest of Ch.4 http://artsandscience.concordia.ca/facstaff/p-r/rogers
REACTIONS OF ALCOHOLS Created by: Mohammad Heidarian
 Nucleophilic Substitution, β- Elimination, and Oxidation reactions are the main type of reactions associated with alcohols. Nucleophilic Substitution: A reaction in which a nucleophile replaces a leaving
Nucleophilic Substitution, β- Elimination, and Oxidation reactions are the main type of reactions associated with alcohols. Nucleophilic Substitution: A reaction in which a nucleophile replaces a leaving
UCF - ORGANIC CHEMISTRY 1 - PROF. DAOUDI UCF PROF. DAOUDI EXAM 3 REVIEW.
 UCF PROF. DAOUDI EXAM 3 REVIEW www.clutchprep.com 1 PRACTICE: Identify the most stable and the least stable alkene PRACTICE: Create the full arrow pushing mechanism which shows all intermediates and all
UCF PROF. DAOUDI EXAM 3 REVIEW www.clutchprep.com 1 PRACTICE: Identify the most stable and the least stable alkene PRACTICE: Create the full arrow pushing mechanism which shows all intermediates and all
Physical Properties: Structure:
 Nomenclature: Functional group suffix = -ol Functional group prefix = hydroxy- Primary, secondary or tertiary? Alcohols are described as primary (1 o ), secondary (2 o ) or tertiary (3 o ) depending on
Nomenclature: Functional group suffix = -ol Functional group prefix = hydroxy- Primary, secondary or tertiary? Alcohols are described as primary (1 o ), secondary (2 o ) or tertiary (3 o ) depending on
Loudon Chapter 9 Review: Reactions of Alkyl Halides Jacquie Richardson, CU Boulder Last updated 5/1/2016
 Chapter 9 covers reactions you can do with alkyl halides. For the most part, these break down into two categories: substitution and elimination. Substitution results in replacing the halogen with some
Chapter 9 covers reactions you can do with alkyl halides. For the most part, these break down into two categories: substitution and elimination. Substitution results in replacing the halogen with some
Essential Organic Chemistry. Chapter 9
 Essential Organic Chemistry Paula Yurkanis Bruice Chapter 9 Substitution and Elimination Reactions of Alkyl Halides 9.1 How Alkyl Halides React Substitution Reactions One group takes the place of another.
Essential Organic Chemistry Paula Yurkanis Bruice Chapter 9 Substitution and Elimination Reactions of Alkyl Halides 9.1 How Alkyl Halides React Substitution Reactions One group takes the place of another.
Announcements Monday, September 18
 Announcements Monday, September 18 WeBWorK 1.4, 1.5 are due on Wednesday at 11:59pm. The first midterm is on this Friday, September 22. Midterms happen during recitation. The exam covers through 1.5. About
Announcements Monday, September 18 WeBWorK 1.4, 1.5 are due on Wednesday at 11:59pm. The first midterm is on this Friday, September 22. Midterms happen during recitation. The exam covers through 1.5. About
Lecture 21 Organic Chemistry 1
 CEM 232 Organic Chemistry I at Chicago Lecture 21 Organic Chemistry 1 Professor Duncan Wardrop March 30, 2010 1 Self Test Question Ethanolysis of alkyl halide 1 gives ether 2 as one product; however, several
CEM 232 Organic Chemistry I at Chicago Lecture 21 Organic Chemistry 1 Professor Duncan Wardrop March 30, 2010 1 Self Test Question Ethanolysis of alkyl halide 1 gives ether 2 as one product; however, several
Outline for Today. Monday, Nov. 26. Chapter 11: Intermolecular Forces and Liquids. Intermolecular Foces. Comparing States of Matter
 Outline for Today Monday, Nov. 26 Chapter 11: Intermolecular Forces and Liquids Intermolecular Foces Comparing States of Matter Properties of Liquids Phase Changes!1 Exam 3 Reminders When: Wednesday at
Outline for Today Monday, Nov. 26 Chapter 11: Intermolecular Forces and Liquids Intermolecular Foces Comparing States of Matter Properties of Liquids Phase Changes!1 Exam 3 Reminders When: Wednesday at
Facebook: UCI ORganic Chemistry Peer Tutoring King 51 Fall 2017
 1 Organic Chemistry 51A Professor King Final Exam Review Session December 7 th, 2017 Will Cabanela (rcabanel@uci.edu) Amanda Pinski (apinski@uci.edu) Zachary Valley (zvalley@uci.edu) http://sites.uci.edu/ochemtutors
1 Organic Chemistry 51A Professor King Final Exam Review Session December 7 th, 2017 Will Cabanela (rcabanel@uci.edu) Amanda Pinski (apinski@uci.edu) Zachary Valley (zvalley@uci.edu) http://sites.uci.edu/ochemtutors
+ + CH 11: Substitution and Elimination Substitution reactions
 C 11: Substitution and Elimination Substitution reactions Things to sort out: Nucleophile Electrophile -- > substrate Leaving Group S N 2 S N 1 E 1 E 2 Analysis Scheme Kinetics Reaction profile Substrates
C 11: Substitution and Elimination Substitution reactions Things to sort out: Nucleophile Electrophile -- > substrate Leaving Group S N 2 S N 1 E 1 E 2 Analysis Scheme Kinetics Reaction profile Substrates
Chapter 7 - Alkenes and Alkynes I
 Andrew Rosen Chapter 7 - Alkenes and Alkynes I 7.1 - Introduction - The simplest member of the alkenes has the common name of ethylene while the simplest member of the alkyne family has the common name
Andrew Rosen Chapter 7 - Alkenes and Alkynes I 7.1 - Introduction - The simplest member of the alkenes has the common name of ethylene while the simplest member of the alkyne family has the common name
12AL Experiment 11 (3 days): Nucleophilic Substitution Reactions
 12AL Experiment 11 (3 days): Nucleophilic Substitution Reactions Instructor note: Day 1 (half of the class); Day 2 (other half); Day 3 (everyone to finish up any separation & purification steps etc). Initial
12AL Experiment 11 (3 days): Nucleophilic Substitution Reactions Instructor note: Day 1 (half of the class); Day 2 (other half); Day 3 (everyone to finish up any separation & purification steps etc). Initial
Chemistry 2030 Introduction to Organic Chemistry Fall Semester 2015 Dr. Rainer Glaser
 Chemistry 2030 Introduction to Organic Chemistry Fall Semester 2015 Dr. Rainer Glaser Examination #3 Nucleophilic Substitutions & Eliminations, Alcohols, and Ethers Thursday, November 5, 2015, 8:25-9:15
Chemistry 2030 Introduction to Organic Chemistry Fall Semester 2015 Dr. Rainer Glaser Examination #3 Nucleophilic Substitutions & Eliminations, Alcohols, and Ethers Thursday, November 5, 2015, 8:25-9:15
CHEM*2700 ORGANIC CHEMISTRY I (Spring/Summer Semester 2009) Information Sheet and Course Outline
 CHEM*2700 ORGANIC CHEMISTRY I (Spring/Summer Semester 2009) Information Sheet and Course Outline Instructor: Professor William Tam Office: MacN 332 Phone: 824-4120 (Ext.52268) E-mail: wtam@uoguelph.ca
CHEM*2700 ORGANIC CHEMISTRY I (Spring/Summer Semester 2009) Information Sheet and Course Outline Instructor: Professor William Tam Office: MacN 332 Phone: 824-4120 (Ext.52268) E-mail: wtam@uoguelph.ca
OChem 1 Mechanism Flashcards. Dr. Peter Norris, 2018
 OChem 1 Mechanism Flashcards Dr. Peter Norris, 2018 Mechanism Basics Chemical change involves bonds forming and breaking; a mechanism describes those changes using curved arrows to describe the electrons
OChem 1 Mechanism Flashcards Dr. Peter Norris, 2018 Mechanism Basics Chemical change involves bonds forming and breaking; a mechanism describes those changes using curved arrows to describe the electrons
OChem 1 Mechanism Flashcards. Dr. Peter Norris, 2015
 OChem 1 Mechanism Flashcards Dr. Peter Norris, 2015 Mechanism Basics Chemical change involves bonds forming and breaking; a mechanism describes those changes using curved arrows to describe the electrons
OChem 1 Mechanism Flashcards Dr. Peter Norris, 2015 Mechanism Basics Chemical change involves bonds forming and breaking; a mechanism describes those changes using curved arrows to describe the electrons
Organic Halogen Compounds
 8 Organic alogen ompounds APTER SUMMARY 8.1 Introduction Although organic halogen compounds are rarely found in nature, they do have a variety of commercial applications including use as insecticides,
8 Organic alogen ompounds APTER SUMMARY 8.1 Introduction Although organic halogen compounds are rarely found in nature, they do have a variety of commercial applications including use as insecticides,
Homework - Chapter 9 Chem 2310
 Homework - Chapter 9 Chem 2310 me:. ntroduction to organic halides 1. Draw a line structure for a compound with the following description: an alkyl halide with the formula C 5 H 11 a vinyl halide with
Homework - Chapter 9 Chem 2310 me:. ntroduction to organic halides 1. Draw a line structure for a compound with the following description: an alkyl halide with the formula C 5 H 11 a vinyl halide with
Learning Guide for Chapter 7 - Organic Reactions I
 Learning Guide for Chapter 7 - rganic Reactions I I. Introduction to Reactions II. Principles of Kinetics III. Principles of Thermodynamics IV. Nucleophiles and Electrophiles V. Acids and Bases What a
Learning Guide for Chapter 7 - rganic Reactions I I. Introduction to Reactions II. Principles of Kinetics III. Principles of Thermodynamics IV. Nucleophiles and Electrophiles V. Acids and Bases What a
Learning Guide for Chapter 7 - Organic Reactions I
 Learning Guide for Chapter 7 - rganic Reactions I I. Introduction to Reactions II. Principles of Kinetics III. Principles of Thermodynamics IV. cleophiles and Electrophiles V. Acids and Bases What a chemical
Learning Guide for Chapter 7 - rganic Reactions I I. Introduction to Reactions II. Principles of Kinetics III. Principles of Thermodynamics IV. cleophiles and Electrophiles V. Acids and Bases What a chemical
Chemistry 330 Fall 2015 Organic Chemistry I
 Chemistry 330 Fall 2015 Organic Chemistry I Instructor: John G. Kodet Contact Information: Office: Faraday Hall 335 Email: jkodet@niu.edu Office Hours: MW 2:00-3:00 pm, and by appointment Lecture: MWF
Chemistry 330 Fall 2015 Organic Chemistry I Instructor: John G. Kodet Contact Information: Office: Faraday Hall 335 Email: jkodet@niu.edu Office Hours: MW 2:00-3:00 pm, and by appointment Lecture: MWF
Classes of Halides. Chapter 6 Alkyl Halides: Nucleophilic Substitution and Elimination. Polarity and Reactivity. Classes of Alkyl Halides
 rganic hemistry, 5 th Edition L. G. Wade, Jr. hapter 6 Alkyl alides: Nucleophilic Substitution and Elimination lasses of alides Alkyl: alogen, X, is directly bonded to sp 3 carbon. Vinyl: X is bonded to
rganic hemistry, 5 th Edition L. G. Wade, Jr. hapter 6 Alkyl alides: Nucleophilic Substitution and Elimination lasses of alides Alkyl: alogen, X, is directly bonded to sp 3 carbon. Vinyl: X is bonded to
CHE2060 Lecture 5: Acid-base chemistry. CHE2060 Lecture 5: Acid-base chemistry
 CHE2060 Lecture 5: Acid-base chemistry 5.1 Acids & bases: overview & basics 5.2 Acid & base strength 5.3 Equilibrium acid-base reactions 5.4 The leveling effect of solvents 5.5 Estimation of acidity by
CHE2060 Lecture 5: Acid-base chemistry 5.1 Acids & bases: overview & basics 5.2 Acid & base strength 5.3 Equilibrium acid-base reactions 5.4 The leveling effect of solvents 5.5 Estimation of acidity by
6-2 This exercise is worked out on page 220 as "Working with Concepts".
 Copyright 2009 James K Whitesell 6-1 Although we can approach this exercise from a chemical perspective, one can also teach a non-chemist how to derive the answer once the name of the starting material
Copyright 2009 James K Whitesell 6-1 Although we can approach this exercise from a chemical perspective, one can also teach a non-chemist how to derive the answer once the name of the starting material
Homework for Chapter 7 Chem 2310
 omework for Chapter 7 Chem 2310 Name I. Introduction to Reactions 1. Explain why the following fits the definition of a chemical reaction. C 3 Na C 3 Na 2. Using the chemical reaction above, give all compounds
omework for Chapter 7 Chem 2310 Name I. Introduction to Reactions 1. Explain why the following fits the definition of a chemical reaction. C 3 Na C 3 Na 2. Using the chemical reaction above, give all compounds
Give a common name for the following compound.
 Give a common name for the following compound. A. Benzyl Phenoate B. Phenyl Phenoate C. Benzyl Benzoate D. Phenyl Benzoate E. Phenyl Phenylacetate F. Phenyl Phenylethanoate G. Phenyl Benzylacetate 2016-10-07
Give a common name for the following compound. A. Benzyl Phenoate B. Phenyl Phenoate C. Benzyl Benzoate D. Phenyl Benzoate E. Phenyl Phenylacetate F. Phenyl Phenylethanoate G. Phenyl Benzylacetate 2016-10-07
Alkyl Halides. Alkyl halides are a class of compounds where a halogen atom or atoms are bound to an sp 3 orbital of an alkyl group.
 Alkyl Halides Alkyl halides are a class of compounds where a halogen atom or atoms are bound to an sp 3 orbital of an alkyl group. CHCl 3 (Chloroform: organic solvent) CF 2 Cl 2 (Freon-12: refrigerant
Alkyl Halides Alkyl halides are a class of compounds where a halogen atom or atoms are bound to an sp 3 orbital of an alkyl group. CHCl 3 (Chloroform: organic solvent) CF 2 Cl 2 (Freon-12: refrigerant
Homework - Review of Chem 2310
 omework - Review of Chem 2310 Chapter 1 - Atoms and Molecules Name 1. What is organic chemistry? 2. Why is there an entire one year course devoted to the study of organic compounds? 3. Give 4 examples
omework - Review of Chem 2310 Chapter 1 - Atoms and Molecules Name 1. What is organic chemistry? 2. Why is there an entire one year course devoted to the study of organic compounds? 3. Give 4 examples
Give the major organic product(s) of the following reaction.
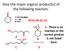 Give the major organic product(s) of the following reaction. 1) CH3CH2MgBr 2) H3O 2016-09-26 Q1 + E. There is no reaction or the correct product A B is not listed here. C D Explanation CH3CH2MgBr 1) CH3CH2MgBr
Give the major organic product(s) of the following reaction. 1) CH3CH2MgBr 2) H3O 2016-09-26 Q1 + E. There is no reaction or the correct product A B is not listed here. C D Explanation CH3CH2MgBr 1) CH3CH2MgBr
Chapter 7 Alkenes; Elimination Reactions
 hapter 7 Alkenes; Elimination Reactions Alkenes Alkenes contain a carbon-carbon double bond. They are named as derivatives of alkanes with the suffix -ane changed to -ene. The parent alkane is the longest
hapter 7 Alkenes; Elimination Reactions Alkenes Alkenes contain a carbon-carbon double bond. They are named as derivatives of alkanes with the suffix -ane changed to -ene. The parent alkane is the longest
Statics - TAM 210 & TAM 211. Spring 2018
 Statics - TAM 210 & TAM 211 Spring 2018 Course distribution Required TAM 210 TAM 211 Aerospace Engineering 31 1 Agricultural & Biological 12 3 Bioengineering 2 6 Civil Engineering 41 Engineering Mechanics
Statics - TAM 210 & TAM 211 Spring 2018 Course distribution Required TAM 210 TAM 211 Aerospace Engineering 31 1 Agricultural & Biological 12 3 Bioengineering 2 6 Civil Engineering 41 Engineering Mechanics
(b) (CH 3 ) 2 CCH 2 CH 3 D 2 O. (e) A. CH 3 CCl OSO 2 CH 3 C 6 H 5 H 3 C
 278 h a p t e r 7 Further Reactions of aloalkanes 3. arbocations are stabilized by hyperconjugation: Tertiary are the most stable, followed by secondary. Primary and methyl cations are too unstable to
278 h a p t e r 7 Further Reactions of aloalkanes 3. arbocations are stabilized by hyperconjugation: Tertiary are the most stable, followed by secondary. Primary and methyl cations are too unstable to
Chapter 8 Alkyl Halides and Elimination Reactions
 Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 8 Alkyl Halides and Elimination Reactions Prepared by Rabi Ann Musah State University of New York at Albany Copyright
Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 8 Alkyl Halides and Elimination Reactions Prepared by Rabi Ann Musah State University of New York at Albany Copyright
Chapter 11 - Alcohols and Ethers 1
 Andrew Rosen Chapter 11 - Alcohols and Ethers 1 11.1 - Structure and Nomenclature - The common naming calls alcohols as alkyl alcohols (eg: methyl alcohol) - The common names of ethers have the groups
Andrew Rosen Chapter 11 - Alcohols and Ethers 1 11.1 - Structure and Nomenclature - The common naming calls alcohols as alkyl alcohols (eg: methyl alcohol) - The common names of ethers have the groups
ORGANIC - EGE 5E CH. 7 - NUCLEOPHILIC SUBSTITUTION AND ELIMINATION REACTIONS
 !! www.clutchprep.com CONCEPT: INTRODUCTION TO SUBSTITUTION Previously, we discussed the various ways that acids could react with bases: Recall that in these mechanisms, electrons always travel from density
!! www.clutchprep.com CONCEPT: INTRODUCTION TO SUBSTITUTION Previously, we discussed the various ways that acids could react with bases: Recall that in these mechanisms, electrons always travel from density
Learning Guide for Chapter 15 - Alcohols (II)
 Learning Guide for Chapter 15 - Alcohols (II) I. Introduction to alcohol reactivity II. Reactions of alcohols with acids III. Reactions of alcohols with electrophiles alogenated phosphorus and sulfur compounds
Learning Guide for Chapter 15 - Alcohols (II) I. Introduction to alcohol reactivity II. Reactions of alcohols with acids III. Reactions of alcohols with electrophiles alogenated phosphorus and sulfur compounds
an axial "X" is necessary for a succesful E2 reaction and also works better for S N 2
 cleophilic Substitution & Elimination hemistry 1 Templates for predicting S2 and E2 reactions primary - β α α β two different perspectives secondary - β β α β β α two different perspectives tertiary -
cleophilic Substitution & Elimination hemistry 1 Templates for predicting S2 and E2 reactions primary - β α α β two different perspectives secondary - β β α β β α two different perspectives tertiary -
Announcements Wednesday, September 20
 Announcements Wednesday, September 20 WeBWorK 1.4, 1.5 are due on Wednesday at 11:59pm. The first midterm is on this Friday, September 22. Midterms happen during recitation. The exam covers through 1.5.
Announcements Wednesday, September 20 WeBWorK 1.4, 1.5 are due on Wednesday at 11:59pm. The first midterm is on this Friday, September 22. Midterms happen during recitation. The exam covers through 1.5.
More tutorial at
 Answer all the questions. 1) (12 pts) Assign (R) or (S) to all the chiral centers in the following molecules and mark the priority of each group around a chiral center (just the top chiral carbon in molecule
Answer all the questions. 1) (12 pts) Assign (R) or (S) to all the chiral centers in the following molecules and mark the priority of each group around a chiral center (just the top chiral carbon in molecule
Dehydrohalogenation of Alkyl Halides E2 and E1 Reactions in Detail
 Dehydrohalogenation of Alkyl Halides E2 and E1 Reactions in Detail b-elimination Reactions Overview dehydration of alcohols: X = H; Y = OH dehydrohalogenation of alkyl halides: X = H; Y = Br, etc. X C
Dehydrohalogenation of Alkyl Halides E2 and E1 Reactions in Detail b-elimination Reactions Overview dehydration of alcohols: X = H; Y = OH dehydrohalogenation of alkyl halides: X = H; Y = Br, etc. X C
