CHEM 302 Organic Chemistry I Problem Set VII Chapter 7 Answers
|
|
|
- Constance Kelly
- 5 years ago
- Views:
Transcription
1 CEM 302 rganic Chemistry Problem Set V Chapter 7 Answers 1) The pk a values of acetic acid and trifluroacetic acid are 4.5 and 0.9 respectively. Based on this information, which anion, C 3 C - or CF 3 C -, is a better leaving group? The lower the pk a, the better the acid (remember pk a = -log(k a )). Therefore, since the pk a of trifluoroacetic acid is lower than that of acetic acid, would say that trifluoroacetate anion is a better leaving group that acetate. 2) Draw the Newman projection formulas of the conformations leading to both cis and trans alkenes in the following reaction: Ts C 3 C 2 - Na + ethanol heat C 3 C 2 C=CC 3 Point out which proton will be removed by the base and identify all the unfavorable interactions in the Newman projection. Use this information as your guide to predict which isomer is likely to predominate in the reaction. (N.b.: you need two Newman projection diagrams; one for the cis, and another one for the trans.) ere are the two Newman projections: Unfavorable interactions Ts Ts C 2 C 3 3 C 2 C C 3 C 3 These protons are the ones removed in base The conformer leading to the trans alkene (the one on the right) has fewer unfavorably interactions, and therefore should be more preferred; therefore, the trans alkene should be the observed product.
2 3) Explain why optically active (R) -2-iodopentane racemizes on standing in a solution of sodium iodide in acetone. n words: the iodide ion (from the N does an S N 2 on the (R)-2-iodopentane, making it the (S) enantiomer. Statistically, this process goes on, with more (R) reacting than (S) until there is an equimolar mixture of R and S. At this point, for every R which goes to S, one S goes to R you are now at a steady state which is also a racemic mixture. n pictures: - inversion inversion nitially, there is more (much more) of the iodide on the left than on the right; with time, the amount on the left decreases and the amount on the right increases. When these are equal (or very nearly so), then for each forward reaction, there is a reverse reaction. Also notice that the iodide ion is, essentially, catalytic. 4) An optically pure nitrile, shown below, was required for a research project (not mine!). the student responsible for obtaining 10 g of this material was able to find only three-quarters of this amount; the rest had to be synthesized. Both (R) and (S) enantiomers of 2-bromobutane were available, so the student decided to perform the reaction involving (R)-2-bromobutane and NaCN in DMF. The reaction proceeded as expected (that is, it went in a forward direction), and the proud student combined the original nitrile sample with 2.5 g of the newly synthesized material. Routine measure of [α] of the combined nitrile sample revealed that it was no longer optically pure. What went wrong? What was the optical purity of the sample? (This last part is a some harder question.) CN 3 C ur student needs to get the (R) enantiomer, so starting with the R-bromide was sort of foolish. is reaction is (clearly) S N 2, so he should expect to get the (S) enantiomer. n short, he picked the wrong enantiomer of the bromide with which to start. aving mixed 2.5 g of the (S) nitrile with 7.5 g ( 3 / 4 of the needed amount of 10 g) of the (R), he now has 5.0 g of pure R and 5.0 g of a racemic mixture. ur optical purity
3 can be assessed as follows: optical purity = ([ R ] [ S ]) ([ R] + [ S] ) 50%.. Doing so gives us 5.0 / 10.0 = 0.5 or 5) Each of the following reactions gives a substitution product. dentify the major product in each ease and indicates whether the reaction is likely to proceed via a bimolecular (S N 2) or a unimolecular (S N 1) mechanism. S 2 CF 3 Na acetone Mechanism is most likely an S N 2 reaction, giving as a major product: no stereochemistry specified. C 3 b) Ms A tertiary mesylate (similar to a tosylate) hmm seems to me to be (clearly) S N 1. ere s the product: C 3 Br 80% 2 ethanol c) mm. Another tertiary substrate, this one a bromide. Again, (clearly) an S N 1 reaction, with major product being:
4 d) Br NaCN DMF Some stereochemistry to keep you on your toes! Secondary substrate, decent leaving group, polar solvent sounds like ideal conditions for the S N 2 reaction. ere is the stereochemically correct product: CN e) S Na + + C 3 C 2 Br Again, nice nucleophile, unhindered halide S N 2 reaction. S 3 C C Na + + C 3 S C 3 f) An S N 2 reaction with somewhat different substrates than we ve normally seen. The second compound (dimethylsulfate) is quite similar to methanesulfonyl chloride, a
5 relative of p-toluenesulfonyl chloride. Related compounds do similar reactions. ere s the product: 3 C C C 3 6) Draw the expected product when sodium cyanide reacts with each of the following substrates. Pay attention to stereochemistry where appropriate. S C 3 b) Ts kay. We might have done one or both of these in class. Both are S N 2 reactions, and both go nicely. ere are the products. NC b) NC 7) Give the major elimination product derived from each of the following elimination reactions. Pay attention to stereochemistry where appropriate. (C 3 ) 3 C - K + (C 3 ) 3 C
6 Br C 3 - Na + C 3 b) 8) When cis-1-iodo-2-methylcyclohexane is treated with potassium tert-butoxide, the major product is 1-methylcyclohexene, a trisubstituted alkene. owever, when the corresponding trans isomers is subjected to the same conditions, the reaction progresses at a slower rate and the major product is 3-methylcyclohexene, a disubstituted alkene. Explain these observations. You may provide the arrow formalism (a mechanism) if that helps your answer. (C 3 ) 3 C - K + (C 3 ) 3 C (C 3 ) 3 C - K + (C 3 ) 3 C Whenever confront with problems similar to this one, it is a very good idea to draw more realistic looking 3-D structures (or at least get the cyclohexane in the chair conformation!) Doing so, we get: C 3 C 3 Now we can start to examine the why of the question. We can see that in the cis case, the methyl is in the preferred equatorial position, and that the iodine and the hydrogen on the carbon with the methyl are pretty nearly anti-periplanar, which is the required arrangement (conformation) for a good E 2 reaction. This reaction also yields the favored, Saytzev s product. ence, a good, fast reaction.
7 n the cis case, the ring has to flip to give a good anti-periplanar arrangement for an and the ; this flip also puts the methyl in the less preferred axial position. The ring flip is slow relatively speaking, so the reaction is slow. ere it is in pictures: C 3 C 3 Note that it is the bold hydrogen which is lost. 9) Provide effective syntheses of the following molecules from any suitable starting materials. There are a couple of decent disconnects, shown below. ll provide syntheses from both ) Na Br +
8 2) Na Ph + Br N 3 b) ere s probably the easiest: Br NaN 3 DMF
Chapter 8 Alkyl Halides and Elimination Reactions
 Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 8 Alkyl Halides and Elimination Reactions Prepared by Rabi Ann Musah State University of New York at Albany Copyright
Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 8 Alkyl Halides and Elimination Reactions Prepared by Rabi Ann Musah State University of New York at Albany Copyright
Homework problems Chapters 6 and Give the curved-arrow formalism for the following reaction: CH 3 OH + H 2 C CH +
 omework problems hapters 6 and 7 1. Give the curved-arrow formalism for the following reaction: : 3 - : 2 : 3 2-3 3 2. In each of the following sets, arrange the compounds in order of decreasing pka and
omework problems hapters 6 and 7 1. Give the curved-arrow formalism for the following reaction: : 3 - : 2 : 3 2-3 3 2. In each of the following sets, arrange the compounds in order of decreasing pka and
Nucleophilic Substitution and Elimination
 Nucleophilic Substitution and Elimination Alkyl halides react with a nucleophile in one of two ways. Either they eliminate an X to form an alkene, or they undergo a substitution with the nucleophile, Nu,
Nucleophilic Substitution and Elimination Alkyl halides react with a nucleophile in one of two ways. Either they eliminate an X to form an alkene, or they undergo a substitution with the nucleophile, Nu,
CHE 321 Summer 2012 Exam 2 Form Select the correct number of chirality centers in heroin, the illicit drug shown below.
 Multiple Choice Questions: 50 points 1. Select the correct number of chirality centers in heroin, the illicit drug shown below. A 4 B 5 C 6 D 7 E 8 F more than 8 2. Choose the correct IUPAC name for the
Multiple Choice Questions: 50 points 1. Select the correct number of chirality centers in heroin, the illicit drug shown below. A 4 B 5 C 6 D 7 E 8 F more than 8 2. Choose the correct IUPAC name for the
Basic Organic Chemistry Course code : CHEM (Pre-requisites : CHEM 11122)
 Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
+ + CH 11: Substitution and Elimination Substitution reactions
 C 11: Substitution and Elimination Substitution reactions Things to sort out: Nucleophile Electrophile -- > substrate Leaving Group S N 2 S N 1 E 1 E 2 Analysis Scheme Kinetics Reaction profile Substrates
C 11: Substitution and Elimination Substitution reactions Things to sort out: Nucleophile Electrophile -- > substrate Leaving Group S N 2 S N 1 E 1 E 2 Analysis Scheme Kinetics Reaction profile Substrates
Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides"
 Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides" t Introduction" The polarity of a carbon-halogen bond leads to the carbon having a partial positive charge"
Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides" t Introduction" The polarity of a carbon-halogen bond leads to the carbon having a partial positive charge"
Name the following compounds (include stereochemistry, cis/trans, E/Z when appropriate). Cl E- 6 chloro, 5 ethyl, 4 methyl 3-octene
 Problem Set 6 Name the following compounds (include stereochemistry, cis/trans, E/Z when appropriate). E- 6 chlo, 5 ethyl, 4 methyl 3-octene 5 methyl, 3 vinyl cyclohexene 7Z- 7 bromo 4 ethyl, 6 methyl
Problem Set 6 Name the following compounds (include stereochemistry, cis/trans, E/Z when appropriate). E- 6 chlo, 5 ethyl, 4 methyl 3-octene 5 methyl, 3 vinyl cyclohexene 7Z- 7 bromo 4 ethyl, 6 methyl
(CH 3 ) 3 COH. CH 3 ONa
 1. Rank the following compounds in the trend requested. (15 points each) a. Rank by nucleophilicity. The strongest nucleophile is 1, while the weakest nucleophile is 5. C 3 PNa (C 3 ) 3 C C 3 Na C 3 C
1. Rank the following compounds in the trend requested. (15 points each) a. Rank by nucleophilicity. The strongest nucleophile is 1, while the weakest nucleophile is 5. C 3 PNa (C 3 ) 3 C C 3 Na C 3 C
1. In the reaction shown above the nucleophile is. (a) Na (b) NaC CH (c) HC C (d) HC CH. 2. In the reaction shown above the nucleophile is
 Chemistry 247B Hanson Sample Exam 4B In each case, read each possible answer, use a process of elimination, and circle the BEST answer. If you are having trouble deciding between two answers, briefly explain
Chemistry 247B Hanson Sample Exam 4B In each case, read each possible answer, use a process of elimination, and circle the BEST answer. If you are having trouble deciding between two answers, briefly explain
CHE 275 NUCLEOPHILIC SUBSTITUTUION CHAP 8 ASSIGN. 1. Which best depicts the partial charges on methyl bromide and sodium methoxide?
 CHE 275 NUCLEPHILIC SUBSTITUTUIN CHAP 8 ASSIGN 1. Which best depicts the partial charges on methyl bromide and sodium methoxide? 2. Which of the following would be the best (most reactive) nucleophile
CHE 275 NUCLEPHILIC SUBSTITUTUIN CHAP 8 ASSIGN 1. Which best depicts the partial charges on methyl bromide and sodium methoxide? 2. Which of the following would be the best (most reactive) nucleophile
Chemistry 35 Exam 2 Answers - April 9, 2007
 Chemistry 35 Exam 2 Answers - April 9, 2007 Problem 1. (14 points) Provide the products for the reactions shown below. If stereochemistry is important, be sure to indicate stereochemistry clearly. If no
Chemistry 35 Exam 2 Answers - April 9, 2007 Problem 1. (14 points) Provide the products for the reactions shown below. If stereochemistry is important, be sure to indicate stereochemistry clearly. If no
Elimination Reactions Heating an alkyl halide with a strong base causes elimination of a. molecule of HX
 Elimination eactions eating an alkyl halide with a strong base causes elimination of a molecule of X 1. Potassium hydroxide dissolved in ethanol and the sodium salts of alcohols (such as sodium ethoxide)
Elimination eactions eating an alkyl halide with a strong base causes elimination of a molecule of X 1. Potassium hydroxide dissolved in ethanol and the sodium salts of alcohols (such as sodium ethoxide)
End-of-chapter problems from Hornback: Ch 8: 23, 25, 26, 27b,c,h,i, 28-30, 32-36, 38-40, 44, 46-48, 50, 53. Ch 9: 17d,m, 18, 19, 26, 28.
 Chem 21 Homework set 9 End-of-chapter problems from Hornback: Ch 8: 23, 25, 26, 27b,c,h,i, 28-30, 32-36, 38-40, 44, 46-48, 50, 53. Ch 9: 17d,m, 18, 19, 26, 28. 1. Draw the products of the E2 reaction of
Chem 21 Homework set 9 End-of-chapter problems from Hornback: Ch 8: 23, 25, 26, 27b,c,h,i, 28-30, 32-36, 38-40, 44, 46-48, 50, 53. Ch 9: 17d,m, 18, 19, 26, 28. 1. Draw the products of the E2 reaction of
Chapter 7 Alkenes; Elimination Reactions
 hapter 7 Alkenes; Elimination Reactions Alkenes Alkenes contain a carbon-carbon double bond. They are named as derivatives of alkanes with the suffix -ane changed to -ene. The parent alkane is the longest
hapter 7 Alkenes; Elimination Reactions Alkenes Alkenes contain a carbon-carbon double bond. They are named as derivatives of alkanes with the suffix -ane changed to -ene. The parent alkane is the longest
Lecture 18 Organic Chemistry 1
 CEM 232 rganic Chemistry I at Chicago Lecture 18 rganic Chemistry 1 Professor Duncan Wardrop March 9, 2010 1 Nucleophilicity nucleophilicity: measures the strength of the nucleophile ; more nucleophilic
CEM 232 rganic Chemistry I at Chicago Lecture 18 rganic Chemistry 1 Professor Duncan Wardrop March 9, 2010 1 Nucleophilicity nucleophilicity: measures the strength of the nucleophile ; more nucleophilic
(b) (CH 3 ) 2 CCH 2 CH 3 D 2 O. (e) A. CH 3 CCl OSO 2 CH 3 C 6 H 5 H 3 C
 278 h a p t e r 7 Further Reactions of aloalkanes 3. arbocations are stabilized by hyperconjugation: Tertiary are the most stable, followed by secondary. Primary and methyl cations are too unstable to
278 h a p t e r 7 Further Reactions of aloalkanes 3. arbocations are stabilized by hyperconjugation: Tertiary are the most stable, followed by secondary. Primary and methyl cations are too unstable to
Organic Chemistry 1 CHM 2210 Exam 3 (November 9, 2001)
 Organic Chemistry 1 CM 2210 Exam 3 (November 9, 2001) Name (print): Signature: Student ID Number: There are 14 multiple choice problems (5 points each) on this exam, however, you will only be graded out
Organic Chemistry 1 CM 2210 Exam 3 (November 9, 2001) Name (print): Signature: Student ID Number: There are 14 multiple choice problems (5 points each) on this exam, however, you will only be graded out
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions
 Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat #
 Test 4 Name Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat # Part 1. Multiple Choice. Choose the one best answer, and clearly mark that answer
Test 4 Name Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat # Part 1. Multiple Choice. Choose the one best answer, and clearly mark that answer
Organic Halogen Compounds
 8 Organic alogen ompounds APTER SUMMARY 8.1 Introduction Although organic halogen compounds are rarely found in nature, they do have a variety of commercial applications including use as insecticides,
8 Organic alogen ompounds APTER SUMMARY 8.1 Introduction Although organic halogen compounds are rarely found in nature, they do have a variety of commercial applications including use as insecticides,
CHEMISTRY MIDTERM # 3 March 31, The total number of points in this midterm is 100. The total exam time is 120 min (2 h). Good luck!
 Name: CEMISTRY 1-01 MIDTERM # March 1, 2009 The total number of points in this midterm is 100. The total exam time is 120 min (2 h). Good luck! 1. (11 pts) Mark as true (T) false (F) the following statements.
Name: CEMISTRY 1-01 MIDTERM # March 1, 2009 The total number of points in this midterm is 100. The total exam time is 120 min (2 h). Good luck! 1. (11 pts) Mark as true (T) false (F) the following statements.
PLEASE DO NOT OPEN THIS EXAM UNTIL YOU ARE INSTRUCTED TO DO SO.
 CEMISTRY 0 :5 AM Section EXAM 2 Nov 2009 Name: Note: Your exam should consist of 5 pages including the cover page and grade tabulation sheet. Skim the entire exam, and solve the easiest problems first.
CEMISTRY 0 :5 AM Section EXAM 2 Nov 2009 Name: Note: Your exam should consist of 5 pages including the cover page and grade tabulation sheet. Skim the entire exam, and solve the easiest problems first.
diene. If stereoisomeric mixtures form, indicate by drawing one and writing "+ enantiomer" and/or "+ diastereomer" in the box.
 I. (9 points) Page A. Consider how varying the conditions of a reaction can vastly influence the rate of a reaction as well as the product(s) formed. First draw the product(s) that form in the reference
I. (9 points) Page A. Consider how varying the conditions of a reaction can vastly influence the rate of a reaction as well as the product(s) formed. First draw the product(s) that form in the reference
Chapter 5. Nucleophilic aliphatic substitution mechanism. by G.DEEPA
 Chapter 5 Nucleophilic aliphatic substitution mechanism by G.DEEPA 1 Introduction The polarity of a carbon halogen bond leads to the carbon having a partial positive charge In alkyl halides this polarity
Chapter 5 Nucleophilic aliphatic substitution mechanism by G.DEEPA 1 Introduction The polarity of a carbon halogen bond leads to the carbon having a partial positive charge In alkyl halides this polarity
1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound?
 CEM 331: Chapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N C 2 C N C 2 C N 1 2 3 4 1: three sigma bonds and
CEM 331: Chapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N C 2 C N C 2 C N 1 2 3 4 1: three sigma bonds and
Organic Chemistry Practice Problems: Solutions
 rganic Chemistry Practice Problems: Solutions 1. 2. a. B, A b. D, B c. A, D d. D, A a. Resonance b. Electronegativity of fluorine atoms F F c. Neither is very acidic, but the oxygen will help stabilise
rganic Chemistry Practice Problems: Solutions 1. 2. a. B, A b. D, B c. A, D d. D, A a. Resonance b. Electronegativity of fluorine atoms F F c. Neither is very acidic, but the oxygen will help stabilise
CHAPTER 8 HW SOLUTIONS: ELIMINATIONS REACTIONS
 APTER 8 W SLUTNS: ELMNATNS REATNS S-TRANS SMERSM 1. Use a discussion and drawing of orbitals to help explain why it is generally easy to rotate around single bonds at room temperature, while it is difficult
APTER 8 W SLUTNS: ELMNATNS REATNS S-TRANS SMERSM 1. Use a discussion and drawing of orbitals to help explain why it is generally easy to rotate around single bonds at room temperature, while it is difficult
Problem Set 8: Substitution Reactions-ANSWER KEY. (b) nucleophile NH 3 H C. (d) H 3 N
 Problem Set 8: Substitution Reactions-ANSWER KEY hemistry 260 rganic hemistry 1. The answers are (1), (3) and (5). Nucleophiles generally have lone pair and/or negative charge. 2. (a) neither 4 (c) nucleophile
Problem Set 8: Substitution Reactions-ANSWER KEY hemistry 260 rganic hemistry 1. The answers are (1), (3) and (5). Nucleophiles generally have lone pair and/or negative charge. 2. (a) neither 4 (c) nucleophile
Nucleophilic Substitution & Elimination Chemistry 1
 ucleophilic Substitution & Elimination hemistry 1 What kind of mechanisms are possible? What is the major mechanism occuring? Write in ALL mechanism details (lone pairs, formal charge, curved arrows, etc.).
ucleophilic Substitution & Elimination hemistry 1 What kind of mechanisms are possible? What is the major mechanism occuring? Write in ALL mechanism details (lone pairs, formal charge, curved arrows, etc.).
REVIEW PROBLEMS Key. 1. Draw a complete orbital picture for the molecule shown below. Is this molecule chiral? Explain. H H.
 rganic hemistry II (E325) REVIEW PRBLEMS Key 1. Draw a complete orbital picture for the molecule shown below. Is this molecule chiral? Explain. 3 3 sp3 orbital p orbital sp2 orbital s orbital molecule
rganic hemistry II (E325) REVIEW PRBLEMS Key 1. Draw a complete orbital picture for the molecule shown below. Is this molecule chiral? Explain. 3 3 sp3 orbital p orbital sp2 orbital s orbital molecule
Chem 232. Problem Set 9. Question 1. D. J. Wardrop
 Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 9 Question 1. a. Rank in order of increasing number of chirality centers (1 = fewest chirality centers; 5 = most chirality). 3 C C 3 b. Rank in order
Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 9 Question 1. a. Rank in order of increasing number of chirality centers (1 = fewest chirality centers; 5 = most chirality). 3 C C 3 b. Rank in order
Chem 30A Winter Mon March 21st
 Last Name First Name MI Student ID Number: Total Score / 200 Chem 30A Winter 2005 FINAL (180 Min) Mon March 21st Course Grade ***DO NOT OPEN THIS EXAM UNTIL INSTRUCTED TO DO SO*** ONLY ANSWERS WRITTEN
Last Name First Name MI Student ID Number: Total Score / 200 Chem 30A Winter 2005 FINAL (180 Min) Mon March 21st Course Grade ***DO NOT OPEN THIS EXAM UNTIL INSTRUCTED TO DO SO*** ONLY ANSWERS WRITTEN
Department of Chemistry SUNY/Oneonta. Chem Organic Chemistry I. Examination #4 - December 9, 2002 ANSWERS
 INSTRUCTINS Department of Chemistry SUNY/neonta Chem 221 - rganic Chemistry I Examination #4 - December 9, 2002 ANSWERS This examination is in multiple choice format; the questions are in this Exam Booklet
INSTRUCTINS Department of Chemistry SUNY/neonta Chem 221 - rganic Chemistry I Examination #4 - December 9, 2002 ANSWERS This examination is in multiple choice format; the questions are in this Exam Booklet
CHAPTER 7 HW: SUBSTITUTIONS
 CHAPTER 7 HW: SUBSTTUTNS REACTN TYPES 1. assify each reaction as either a substitution, elimination, or addition reaction. Reaction Type NaH H + Na H + (cat.) H CH CH 2 H 2 CH H H + (cat.) H 2 + H 2 TERMNLGY
CHAPTER 7 HW: SUBSTTUTNS REACTN TYPES 1. assify each reaction as either a substitution, elimination, or addition reaction. Reaction Type NaH H + Na H + (cat.) H CH CH 2 H 2 CH H H + (cat.) H 2 + H 2 TERMNLGY
CHAPTER 8 HW: ELIMINATIONS REACTIONS
 APTER 8 W: ELMNATNS REATNS S-TRANS SMERSM 1. Use a discussion and drawing of orbitals to help explain why it is generally easy to rotate around single bonds at room temperature, while it is difficult to
APTER 8 W: ELMNATNS REATNS S-TRANS SMERSM 1. Use a discussion and drawing of orbitals to help explain why it is generally easy to rotate around single bonds at room temperature, while it is difficult to
1) (100 pts) 5) (20 pts) 3) (35 pts) 4) (25pts. Total (200 pts) More Tutorial at
 Name: Perm number: Question 1) (100 pts) 2) (20 pts) 3) (35 pts) 4) (25pts 5) (20 pts) Total (200 pts) Your score 1. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers
Name: Perm number: Question 1) (100 pts) 2) (20 pts) 3) (35 pts) 4) (25pts 5) (20 pts) Total (200 pts) Your score 1. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers
REACTIONS OF HALOALKANES - SUBSTITUTION AND ELIMINATION
 REACTIONS OF HALOALKANES - SUBSTITUTION AND ELIMINATION Haloalkanes (also known as halogenoalkanes and alkyl halides) are organic compounds where one of the hydrogens of an alkane or cycloalkane has been
REACTIONS OF HALOALKANES - SUBSTITUTION AND ELIMINATION Haloalkanes (also known as halogenoalkanes and alkyl halides) are organic compounds where one of the hydrogens of an alkane or cycloalkane has been
7. Haloalkanes (text )
 2009, Department of hemistry, The University of Western Ontario 7.1 7. aloalkanes (text 7.1 7.10) A. Structure and Nomenclature Like hydrogen, the halogens have a valence of one. Thus, a halogen atom can
2009, Department of hemistry, The University of Western Ontario 7.1 7. aloalkanes (text 7.1 7.10) A. Structure and Nomenclature Like hydrogen, the halogens have a valence of one. Thus, a halogen atom can
Organic Chemistry CHM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl Halides: Substitution Reactions - Chapter 6 (Wade)
 rganic Chemistry CM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl alides: Substitution Reactions - Chapter 6 (Wade) Chapter utline I. Intro to RX (6-1 - 6-7) II. Substitution Reactions A) S N 2 (6-8,
rganic Chemistry CM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl alides: Substitution Reactions - Chapter 6 (Wade) Chapter utline I. Intro to RX (6-1 - 6-7) II. Substitution Reactions A) S N 2 (6-8,
Dehydrohalogenation of Alkyl Halides E2 and E1 Reactions in Detail
 Dehydrohalogenation of Alkyl Halides E2 and E1 Reactions in Detail b-elimination Reactions Overview dehydration of alcohols: X = H; Y = OH dehydrohalogenation of alkyl halides: X = H; Y = Br, etc. X C
Dehydrohalogenation of Alkyl Halides E2 and E1 Reactions in Detail b-elimination Reactions Overview dehydration of alcohols: X = H; Y = OH dehydrohalogenation of alkyl halides: X = H; Y = Br, etc. X C
PRACTICE PROBLEMS UNIT 8
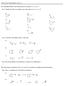 PRACTICE PROBLEMS UNIT 8 8A. Identify halides and carbocations as being 1 o, 2 o, or 3 o 8A.1 assify the following halides and carbocations as 1 o, 2 o, or 3 o. 8A.2 Circle the most stable cation in each
PRACTICE PROBLEMS UNIT 8 8A. Identify halides and carbocations as being 1 o, 2 o, or 3 o 8A.1 assify the following halides and carbocations as 1 o, 2 o, or 3 o. 8A.2 Circle the most stable cation in each
CHEM 2423 Unit 8 Homework Answers
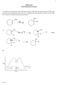 1 CEM 2423 Unit 8 omework Answers 1. Show the mechanism of the following reaction. Label the rate-limiting step. Draw the potential energy diagram for this reaction. (c) Explain the regiochemistry of this
1 CEM 2423 Unit 8 omework Answers 1. Show the mechanism of the following reaction. Label the rate-limiting step. Draw the potential energy diagram for this reaction. (c) Explain the regiochemistry of this
CHAPTER 7. Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination
 CHAPTER 7 Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 7-1 Solvolysis of Tertiary and Secondary Haloalkanes The rate of S N 2 reactions decrease dramatically
CHAPTER 7 Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 7-1 Solvolysis of Tertiary and Secondary Haloalkanes The rate of S N 2 reactions decrease dramatically
Alcohols: Contain a hydroxy group( OH) bonded to an sp 2 or sp 3 hybridized
 Lecture Notes hem 51B S. King hapter 9 Alcohols, Ethers, and Epoxides I. Introduction Alcohols, ether, and epoxides are 3 functional groups that contain σ-bonds. Alcohols: ontain a hydroxy group( ) bonded
Lecture Notes hem 51B S. King hapter 9 Alcohols, Ethers, and Epoxides I. Introduction Alcohols, ether, and epoxides are 3 functional groups that contain σ-bonds. Alcohols: ontain a hydroxy group( ) bonded
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2
 C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
Chapter 11. Solution: 11.4 What product would you expect from S N 2 reaction of 1-bromobutane with each of the following?
 hapter 11 11.1 What product would you expect to obtain from a nucleophilic substitution reaction of (S)-2-bromohexane with acetate ion, 3-2? Assume that inversion of configuration occur, and show the stereochemistry
hapter 11 11.1 What product would you expect to obtain from a nucleophilic substitution reaction of (S)-2-bromohexane with acetate ion, 3-2? Assume that inversion of configuration occur, and show the stereochemistry
Structure and Preparation of Alkenes: Elimination Reactions
 Structure and Preparation of Alkenes: Elimination Reactions Alkene Nomenclature First identify the longest continuous chain that includes the double bond. Replace the -ane ending of the corresponding unbranched
Structure and Preparation of Alkenes: Elimination Reactions Alkene Nomenclature First identify the longest continuous chain that includes the double bond. Replace the -ane ending of the corresponding unbranched
CHEM 261 HOME WORK Lecture Topics: MODULE 1: The Basics: Bonding and Molecular Structure Text Sections (N0 1.9, 9-11) Homework: Chapter 1:
 CHEM 261 HOME WORK Lecture Topics: MODULE 1: The Basics: Bonding and Molecular Structure Atomic Structure - Valence Electrons Chemical Bonds: The Octet Rule - Ionic bond - Covalent bond How to write Lewis
CHEM 261 HOME WORK Lecture Topics: MODULE 1: The Basics: Bonding and Molecular Structure Atomic Structure - Valence Electrons Chemical Bonds: The Octet Rule - Ionic bond - Covalent bond How to write Lewis
Chapter 7 Substitution Reactions
 Chapter 7 Substitution Reactions Review of Concepts Fill in the blanks below. To verify that your answers are correct, look in your textbook at the end of Chapter 7. Each of the sentences below appears
Chapter 7 Substitution Reactions Review of Concepts Fill in the blanks below. To verify that your answers are correct, look in your textbook at the end of Chapter 7. Each of the sentences below appears
Chapter 8 I. Nucleophilic Substitution (in( II. Competion with Elimination. Nucleophilic Substitution
 hapter 8 I. Nucleophilic Substitution (in( depth) II. ompetion with Elimination Nucleophilic Substitution Substrate is a sp3 hybridized carbon atom (cannot be an a vinylic halide or an aryl halide except
hapter 8 I. Nucleophilic Substitution (in( depth) II. ompetion with Elimination Nucleophilic Substitution Substrate is a sp3 hybridized carbon atom (cannot be an a vinylic halide or an aryl halide except
Chem Final Examination August 7, 2004
 Chem 281 2004-2 Final Examination August 7, 2004 Name: Student Number: Note: You are allowed to use models for this exam. Notes, textbooks and calculators are strictly prohibited. Write your final answers
Chem 281 2004-2 Final Examination August 7, 2004 Name: Student Number: Note: You are allowed to use models for this exam. Notes, textbooks and calculators are strictly prohibited. Write your final answers
CHEM 2312 practice final. Version - II
 EM 2312 practice final Version - II The following standardized final examination covers the entire introductory year of organic chemistry (EM 2311 and 2312 at Georgia Tech). The exam consists of 70 multiple
EM 2312 practice final Version - II The following standardized final examination covers the entire introductory year of organic chemistry (EM 2311 and 2312 at Georgia Tech). The exam consists of 70 multiple
1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound?
 EM 331: hapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N 2 N 2 N 1 2 3 4 2. What hybrid orbitals are used to
EM 331: hapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N 2 N 2 N 1 2 3 4 2. What hybrid orbitals are used to
Essential Organic Chemistry. Chapter 9
 Essential Organic Chemistry Paula Yurkanis Bruice Chapter 9 Substitution and Elimination Reactions of Alkyl Halides 9.1 How Alkyl Halides React Substitution Reactions One group takes the place of another.
Essential Organic Chemistry Paula Yurkanis Bruice Chapter 9 Substitution and Elimination Reactions of Alkyl Halides 9.1 How Alkyl Halides React Substitution Reactions One group takes the place of another.
Chem 3719 Example Exams. Chemistry 3719 Practice Exams
 Chem 3719 Example Exams Chemistry 3719 Practice Exams Fall 2018 Chemistry 3719, Fall 2017 Exam 1 Student Name: Y Number: This exam is worth 100 points out of a total of 700 points for Chemistry 3719/3719L.
Chem 3719 Example Exams Chemistry 3719 Practice Exams Fall 2018 Chemistry 3719, Fall 2017 Exam 1 Student Name: Y Number: This exam is worth 100 points out of a total of 700 points for Chemistry 3719/3719L.
Classes of Alkenes. Alkenes and Alkynes. Saturated compounds (alkanes): Have the maximum number of hydrogen atoms attached to each carbon atom.
 Alkenes and Alkynes Saturated compounds (alkanes): ave the maximum number of hydrogen atoms attached to each carbon atom. Unsaturated compounds: ave fewer hydrogen atoms attached to the carbon chain than
Alkenes and Alkynes Saturated compounds (alkanes): ave the maximum number of hydrogen atoms attached to each carbon atom. Unsaturated compounds: ave fewer hydrogen atoms attached to the carbon chain than
Chemistry 232 PRACTICE Midterm 3 October 2010 Your name: 1
 Chemistry 232 PRACTICE Midterm 3 October 2010 Your name: 1 You will need to be able to show picture ID to take the test. DO NOT OPEN TIS TEST UNTIL EVERYONE AS ONE I encourage following instructions: ten
Chemistry 232 PRACTICE Midterm 3 October 2010 Your name: 1 You will need to be able to show picture ID to take the test. DO NOT OPEN TIS TEST UNTIL EVERYONE AS ONE I encourage following instructions: ten
Organic Reactions Susbstitution S N. Dr. Sapna Gupta
 Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
CHEM120 - ORGANIC CHEMISTRY WORKSHEET 1
 EM120 - RGANI EMISTRY WRKSEET 1 Some of the objectives To understand and know the hybridization concept Be able to distinguish different geometries, including basic bond lengths and angles within organic
EM120 - RGANI EMISTRY WRKSEET 1 Some of the objectives To understand and know the hybridization concept Be able to distinguish different geometries, including basic bond lengths and angles within organic
Department of Chemistry SUNY/Oneonta. Chem Organic Chemistry I. Examination #4 - December 7, 1998*
 Department of hemistry SUNY/neonta hem 221 - rganic hemistry I Examination #4 - December 7, 1998* INSTRUTINS --- This examination is in multiple choice format; the questions are in this Exam Booklet and
Department of hemistry SUNY/neonta hem 221 - rganic hemistry I Examination #4 - December 7, 1998* INSTRUTINS --- This examination is in multiple choice format; the questions are in this Exam Booklet and
1. The barrier to rotation around the C-C bonds for 2-methylpropane and 2,2-dimethylpropane are shown below.
 1. The barrier to rotation around the C-C bonds for 2-methylpropane and 2,2-dimethylpropane are shown below. E Rot = 14.2 kj/mol E Rot = 19.6 kj/mol a. Why does the potential energy of a molecule increase
1. The barrier to rotation around the C-C bonds for 2-methylpropane and 2,2-dimethylpropane are shown below. E Rot = 14.2 kj/mol E Rot = 19.6 kj/mol a. Why does the potential energy of a molecule increase
1. The Substrate: CH3, 1 o, 2 o, 3 o, Allyl or Benzyl
 Putting it all together: Substitution and elimination reactions are almost always in competition with each other. In order to predict the products of a reaction, it is necessary to determine which mechanisms
Putting it all together: Substitution and elimination reactions are almost always in competition with each other. In order to predict the products of a reaction, it is necessary to determine which mechanisms
THE UNIVERSITY OF CALGARY FACULTY OF SCIENCE FINAL EXAMINATION CHEMISTRY 351 READ ALL THE INSTRUCTIONS CAREFULLY
 TE UNIVERSITY F CALGARY FACULTY F SCIENCE FINAL EXAMINATIN CEMISTRY 351 DECEMBER 22nd 1999 Time: 3 ours READ ALL TE INSTRUCTINS CAREFULLY PLEASE WRITE YUR NAME, STUDENT I.D. NUMBER N BT YUR EXAM ANSWER
TE UNIVERSITY F CALGARY FACULTY F SCIENCE FINAL EXAMINATIN CEMISTRY 351 DECEMBER 22nd 1999 Time: 3 ours READ ALL TE INSTRUCTINS CAREFULLY PLEASE WRITE YUR NAME, STUDENT I.D. NUMBER N BT YUR EXAM ANSWER
Alkyl Halides. Alkyl halides are a class of compounds where a halogen atom or atoms are bound to an sp 3 orbital of an alkyl group.
 Alkyl Halides Alkyl halides are a class of compounds where a halogen atom or atoms are bound to an sp 3 orbital of an alkyl group. CHCl 3 (Chloroform: organic solvent) CF 2 Cl 2 (Freon-12: refrigerant
Alkyl Halides Alkyl halides are a class of compounds where a halogen atom or atoms are bound to an sp 3 orbital of an alkyl group. CHCl 3 (Chloroform: organic solvent) CF 2 Cl 2 (Freon-12: refrigerant
Chapter 7 Substitution Reactions 7.1 Introduction to Substitution Reactions Substitution Reactions: two reactants exchange parts to give new products
 hapter 7 Substitution eactions 7.1 Introduction to Substitution eactions Substitution eactions: two reactants exchange parts to give new products A-B + -D A-D + B- 3 2 + Br 3 2 Br + Elimination eaction:
hapter 7 Substitution eactions 7.1 Introduction to Substitution eactions Substitution eactions: two reactants exchange parts to give new products A-B + -D A-D + B- 3 2 + Br 3 2 Br + Elimination eaction:
Retrosynthetic Analysis & Synthesis Problems
 Chem 232 D. J. Wardrop wardropd@uic.edu Retrosynthetic Analysis & Synthesis Problems As we draw towards the end of the semester, we will be paying more attention to questions regarding synthesis, which
Chem 232 D. J. Wardrop wardropd@uic.edu Retrosynthetic Analysis & Synthesis Problems As we draw towards the end of the semester, we will be paying more attention to questions regarding synthesis, which
Organic Chemistry CHM 224
 rganic Chemistry CHM 224 Exam I Review Questions This set of questions is a compilation of old exams and additional questions, and does not represent the typical length of an exam - this is WAY longer!
rganic Chemistry CHM 224 Exam I Review Questions This set of questions is a compilation of old exams and additional questions, and does not represent the typical length of an exam - this is WAY longer!
H C C C C H C H C C H H. Cl H CH 3 O. Br CH 3 OH H 3 C. H Br H
 CEMISTRY 313-61 MITERM # 3 answer key June 17, 2005 Statistics: Average: 69 pts (69%); ighest: 96 pts (96%); Lowest: 46 pts (46%) Number of students perfming at above average: 16 (53%) 1. (10 pts) Mark
CEMISTRY 313-61 MITERM # 3 answer key June 17, 2005 Statistics: Average: 69 pts (69%); ighest: 96 pts (96%); Lowest: 46 pts (46%) Number of students perfming at above average: 16 (53%) 1. (10 pts) Mark
CHE 321 Summer 2010 Exam 2 Form Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III
 CE 321 Summer 2010 Exam 2 orm 2 Multiple Choice Questions: 60 points 1. Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III (A) I only (B) II only (C) I and II only II
CE 321 Summer 2010 Exam 2 orm 2 Multiple Choice Questions: 60 points 1. Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III (A) I only (B) II only (C) I and II only II
CONCERTED sp 2 H. HO Et
 Alkyl alides Substitution and Elimination 1 Substitutions (Quick Review) 1.1 SN2 Reactions LB nucleophile "backside attack!" NERTED reaction This is fundamentally just a Lewis acid/base reaction, the Lewis
Alkyl alides Substitution and Elimination 1 Substitutions (Quick Review) 1.1 SN2 Reactions LB nucleophile "backside attack!" NERTED reaction This is fundamentally just a Lewis acid/base reaction, the Lewis
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams.
 2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
C h a p t e r S e v e n : Substitution Reactions S N 2 O H H H O H H. Br -
 C h a p t e r S e v e n : Substitution Reactions Br Br S N 2 CM 321: Summary of Important Concepts YConcepts for Chapter 7: Substitution Reactions I. Nomenclature of alkyl halides, R X A. Common name:
C h a p t e r S e v e n : Substitution Reactions Br Br S N 2 CM 321: Summary of Important Concepts YConcepts for Chapter 7: Substitution Reactions I. Nomenclature of alkyl halides, R X A. Common name:
Chapter 9. Nucleophilic Substitution and ß-Elimination
 Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Only five of the molecules below may be prepared as the sole product of allylic halogenation of the respective alkene. Circle those five.
 Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 6 Answers Question 1. nly five of the molecules below may be prepared as the sole product of allylic halogenation of the respective alkene. Circle those
Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 6 Answers Question 1. nly five of the molecules below may be prepared as the sole product of allylic halogenation of the respective alkene. Circle those
Cl Cl CH 2 OH H 3 C CH 2 CH 3 H. cis-1-(bromomethyl)-2-(2-chloroethyl)cyclobutane. Meso-1,2,3,4,butanetetraol (show as a Fischer projection)
 Page 1 2. 12 Points. (a) Name or write a structure for the following compounds (IUPAC nomenclature). Be sure to pay attention to stereochemistry! Note that most of these questions are taken directly from
Page 1 2. 12 Points. (a) Name or write a structure for the following compounds (IUPAC nomenclature). Be sure to pay attention to stereochemistry! Note that most of these questions are taken directly from
BSc. II 3 rd Semester. Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1
 BSc. II 3 rd Semester Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1 Introduction to Alkyl Halides Alkyl halides are organic molecules containing a halogen atom bonded to an
BSc. II 3 rd Semester Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1 Introduction to Alkyl Halides Alkyl halides are organic molecules containing a halogen atom bonded to an
Name: Student Number:
 Page 1 of 5 Name: Student Number: l University of Manitoba - Department of Chemistry CEM 2220 - Introductory rganic Chemistry II - Term Test 1 Thursday, February 14, 2008 This is a 2-hour test, marked
Page 1 of 5 Name: Student Number: l University of Manitoba - Department of Chemistry CEM 2220 - Introductory rganic Chemistry II - Term Test 1 Thursday, February 14, 2008 This is a 2-hour test, marked
CHEM Lecture 7
 CEM 494 Special Topics in Chemistry Illinois at Chicago CEM 494 - Lecture 7 Prof. Duncan Wardrop ctober 22, 2012 CEM 494 Special Topics in Chemistry Illinois at Chicago Preparation of Alkenes Elimination
CEM 494 Special Topics in Chemistry Illinois at Chicago CEM 494 - Lecture 7 Prof. Duncan Wardrop ctober 22, 2012 CEM 494 Special Topics in Chemistry Illinois at Chicago Preparation of Alkenes Elimination
B. A transition state represents a maximum on the reaction path diagram and can be isolated.
 Practice Hour Exam 2, Chemistry 2210, Organic Chemistry I 1. The most stable carbocation is: 2. Which of the following statements is true of transition states? A. A transition state represents a minimum
Practice Hour Exam 2, Chemistry 2210, Organic Chemistry I 1. The most stable carbocation is: 2. Which of the following statements is true of transition states? A. A transition state represents a minimum
ADDITION OF HYDROGEN HALIDES TO CONJUGATED DIENES A. 1,2- and 1,4-Additions 700 CHAPTER 15 DIENES, RESONANCE, AND AROMATICITY
 700 CAPTER 15 DIENES, RESONANCE, AND AROMATICITY 15.18 Give the structures of the starting materials that would yield each of the compounds below in Diels Alder reactions. Pay careful attention to stereochemistry,
700 CAPTER 15 DIENES, RESONANCE, AND AROMATICITY 15.18 Give the structures of the starting materials that would yield each of the compounds below in Diels Alder reactions. Pay careful attention to stereochemistry,
HALOALKANES. Structure Contain the functional group C-X where X is a halogen (F,Cl,Br or I)
 aloalkanes AS3 1 ALOALKANES Structure ontain the functional group X where X is a halogen (F,l, or I) Types aloalkanes halogen is attached to an aliphatic skeleton alkyl group aloarenes halogen is attached
aloalkanes AS3 1 ALOALKANES Structure ontain the functional group X where X is a halogen (F,l, or I) Types aloalkanes halogen is attached to an aliphatic skeleton alkyl group aloarenes halogen is attached
Chem 112A: Final Exam
 Chem 112A: Final Exam December 15th, 2010 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
Chem 112A: Final Exam December 15th, 2010 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
8.8 Unimolecular Nucleophilic Substitution S N 1
 8.8 Unimolecular Nucleophilic Substitution S N 1 A question. Tertiary alkyl halides are very unreactive in substitutions that proceed by the S N 2 mechanism. Do they undergo nucleophilic substitution at
8.8 Unimolecular Nucleophilic Substitution S N 1 A question. Tertiary alkyl halides are very unreactive in substitutions that proceed by the S N 2 mechanism. Do they undergo nucleophilic substitution at
Reactions of Alkyl Halides with Nucleophiles and Bases a substitution reaction
 Reactions of Alkyl Halides with Nucleophiles and Bases a substitution reaction Nucleophilic substitution and base induced elimination are among most widely occurring and versatile reaction types in organic
Reactions of Alkyl Halides with Nucleophiles and Bases a substitution reaction Nucleophilic substitution and base induced elimination are among most widely occurring and versatile reaction types in organic
an axial "X" is necessary for a succesful E2 reaction and also works better for S N 2
 cleophilic Substitution & Elimination hemistry 1 Templates for predicting S2 and E2 reactions primary - β α α β two different perspectives secondary - β β α β β α two different perspectives tertiary -
cleophilic Substitution & Elimination hemistry 1 Templates for predicting S2 and E2 reactions primary - β α α β two different perspectives secondary - β β α β β α two different perspectives tertiary -
CHEM 8A Summer Student ID # Organic Chemistry FINAL EXAM (400 points)
 CEM 8A Summer 2017 UCSC, Binder Name Student ID # rganic Chemistry FINAL EXAM (400 points) In each of the following problems, you will use your knowledge of organic chemistry conventions to answer the
CEM 8A Summer 2017 UCSC, Binder Name Student ID # rganic Chemistry FINAL EXAM (400 points) In each of the following problems, you will use your knowledge of organic chemistry conventions to answer the
!!!! Organic Chemistry CHM 224. Exam I Questions
 ld Exam I Questions - CHM 224 rganic Chemistry CHM 224 Exam I Questions This set of questions is a compilation of old exams, and does not represent the typical length of an exam - there are more examples,
ld Exam I Questions - CHM 224 rganic Chemistry CHM 224 Exam I Questions This set of questions is a compilation of old exams, and does not represent the typical length of an exam - there are more examples,
1-What is substitution reaction? 2-What are can Nucleophilic Substitution Reaction? 3- SN1 reaction. 4-SN2 reaction 5- mechanisms of SN1&SN2
 1-What is substitution reaction? 2-What are can Nucleophilic Substitution eaction? 3- SN1 reaction. 4-SN2 reaction 5- mechanisms of SN1&SN2 1- SUBSTITUTION EACTIONS 1-Substitution eaction In this type
1-What is substitution reaction? 2-What are can Nucleophilic Substitution eaction? 3- SN1 reaction. 4-SN2 reaction 5- mechanisms of SN1&SN2 1- SUBSTITUTION EACTIONS 1-Substitution eaction In this type
Chapter 8: Alkene Structure and Preparation via Elimination Reactions
 Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C 3 C C 3 bond length bond strength 2 C C 2 a C=C double bond is stronger than a C C single
Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C 3 C C 3 bond length bond strength 2 C C 2 a C=C double bond is stronger than a C C single
Chem 232. Problem Set 4. Table. Substrate Types and the Choice of S N 1 of S N 2
 Chem 232 D. J. Wardrop wardropd@uic.edu Problem et 4 Does the nucleophilic substitution of substrate X proceed via an N 2 or N 1 mechanism? is a perennial question in rganic Chemistry. Use the table below
Chem 232 D. J. Wardrop wardropd@uic.edu Problem et 4 Does the nucleophilic substitution of substrate X proceed via an N 2 or N 1 mechanism? is a perennial question in rganic Chemistry. Use the table below
Chapter 7 Alkenes and Alkynes I: Properties and Synthesis Elimination Reactions of Alkyl Halides"
 Chapter 7 Alkenes and Alkynes I: Properties and Synthesis Elimination Reactions of Alkyl Halides The (E)-(Z) System for Designating Alkene Diastereomers The Cahn-Ingold-Prelog convention is used to assign
Chapter 7 Alkenes and Alkynes I: Properties and Synthesis Elimination Reactions of Alkyl Halides The (E)-(Z) System for Designating Alkene Diastereomers The Cahn-Ingold-Prelog convention is used to assign
CHEM 2311 HW1. COMPLETELY FILL THE BOXES IN DARK PENCIL, i.e.:, NOT or or STRUCTURE
 EM 2311 W1 Provide answers to the following multiple choice questions on the Scantron form provided in lecture. Make sure that you put your name and bubble in your GTID # on the answer form. MPLETELY FILL
EM 2311 W1 Provide answers to the following multiple choice questions on the Scantron form provided in lecture. Make sure that you put your name and bubble in your GTID # on the answer form. MPLETELY FILL
3. (4 pts) Provide structures for the following compounds: CH 3 C H H C C - Na + acetylene 1-penten-4-yne (R)-3-butyn-2-ol sodium acetylide
 CEMTRY 313-01 MDTERM # 3 answer key November 18, 2008 tatistics: Average: 74 pts (74%); ighest: 97 pts (97%); Lowest: 38 pts (38%) Number of students perfming at above average: 35 (56%) Number of students
CEMTRY 313-01 MDTERM # 3 answer key November 18, 2008 tatistics: Average: 74 pts (74%); ighest: 97 pts (97%); Lowest: 38 pts (38%) Number of students perfming at above average: 35 (56%) Number of students
Chemistry 143 Exam #2 November 15, Name: Student Number: Section Number: TA: INSTRUCTIONS:
 Chemistry 143 Exam #2 November 15, 2017 Name: Student Number: Section Number: TA: INSTRUCTINS: This exam consists of 32 questions on 9 pages. Please make certain that your exam is complete. Write your
Chemistry 143 Exam #2 November 15, 2017 Name: Student Number: Section Number: TA: INSTRUCTINS: This exam consists of 32 questions on 9 pages. Please make certain that your exam is complete. Write your
20 Halogenoalkanes:substitution and elimination reactions
 20 alogenoalkanes:substitution and elimination reactions s to worked examples WE 20.1 Allylic halogenations (on p. 922 in Chemistry 3 ) 1-(Chloromethyl)benzene (benzyl chloride, PhC 2 ) is made on a large
20 alogenoalkanes:substitution and elimination reactions s to worked examples WE 20.1 Allylic halogenations (on p. 922 in Chemistry 3 ) 1-(Chloromethyl)benzene (benzyl chloride, PhC 2 ) is made on a large
CHAPTER 9 HW SOLUTIONS: ALCOHOLS + ETHERS
 CAPTER 9 W SLUTNS: ALCLS + ETERS ALCL + ETER NMENCLATURE. Give the UPAC name for each compound. nclude cis/trans or R/S stereochemistry if necessary. Structure C Name -methyl--heptanol -t-butylcyclopentanol
CAPTER 9 W SLUTNS: ALCLS + ETERS ALCL + ETER NMENCLATURE. Give the UPAC name for each compound. nclude cis/trans or R/S stereochemistry if necessary. Structure C Name -methyl--heptanol -t-butylcyclopentanol
Chapter 7 - Alkenes and Alkynes I
 Andrew Rosen Chapter 7 - Alkenes and Alkynes I 7.1 - Introduction - The simplest member of the alkenes has the common name of ethylene while the simplest member of the alkyne family has the common name
Andrew Rosen Chapter 7 - Alkenes and Alkynes I 7.1 - Introduction - The simplest member of the alkenes has the common name of ethylene while the simplest member of the alkyne family has the common name
75. A This is a Markovnikov addition reaction. In these reactions, the pielectrons in the alkene act as a nucleophile. The strongest electrophile will
 71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
1. Use appropriate curved arrows to indicate the complete mechanism of each of these reactions. KH (1 equiv.) + KCl THF. + HBr.
 1. Use appropriate curved arrows to indicate the complete mechanism of each of these reactions. K (1 equiv.) TF K 3 2 2 3 enantiomer While writing the mechanism, justify both the regiochemistry the relative
1. Use appropriate curved arrows to indicate the complete mechanism of each of these reactions. K (1 equiv.) TF K 3 2 2 3 enantiomer While writing the mechanism, justify both the regiochemistry the relative
