Apical and Lateral Cell Protrusions Interconnect Epithelial Cells in Live Drosophila Wing Imaginal Discs
|
|
|
- Gwendoline Brown
- 5 years ago
- Views:
Transcription
1 DEVELOPMENTAL DYNAMICS 236: , 2007 RESEARCH ARTICLE Apical and Lateral Cell Protrusions Interconnect Epithelial Cells in Live Drosophila Wing Imaginal Discs Fabio Demontis and Christian Dahmann* Communication among cells by means of the exchange of signaling cues is important for tissue and organ development. Recent reports indicate that one way that signaling cues can be delivered is by movement along cellular protrusions interconnecting cells. Here, by using confocal laser scanning microscopy and three-dimensional rendering, we describe in Drosophila melanogaster wing imaginal discs lateral protrusions interconnecting cells of the columnar epithelium. Moreover, we identified protrusions of the apical surface of columnar cells that reached and apparently contacted cells of the overlying squamous epithelium. Both apical and lateral protrusions could be visualized by expression of Tkv-GFP, a green fluorescent protein (GFP) -tagged version of a receptor of the Dpp/BMP4 signaling molecule, and the endosome marker GFP-Rab5. Our results demonstrate a previously unexpected richness of cellular protrusions within wing imaginal discs and support the view that cellular protrusions may provide a means for exchanging signaling cues between cells. Developmental Dynamics 236: , Wiley-Liss, Inc. Key words: Drosophila; cell protrusion; microvilli; Prominin-like; Tkv; Rab5 Accepted 10 August 2007 INTRODUCTION Cell protrusions are outward extensions of the cell plasma membrane made by many different cell types and organisms. Important cellular processes involve cell protrusions, including nutrient resorption (Louvard et al., 1992), mechanosensing (Frolenkov et al., 2004), photosensing (Corbeil et al., 2001; Pellikka et al., 2002), establishment of cell adhesion (Vasioukhin et al., 2000), cell migration (Fulga and Rorth, 2002), fusion of epithelial sheets (Jacinto et al., 2000; Martin- Blanco et al., 2000), wound healing (Wood et al., 2002), axon guidance (Ritzenthaler et al., 2000), and cell-tocell communication (reviewed in Rorth, 2003). Intercellular communication is important for the growth and patterning of tissues and organs and cell protrusions may mediate this communication by one of several mechanisms. Signaling mediated by cell protrusions can occur by means of transport and local release of signalling molecules, shedding of vesicles (Marzesco et al., 2005), display of receptors (Tomlinson et al., 1987; Hsiung et al., 2005) and membrane-tethered ligands on the protrusion (De Joussineau et al., 2003), or by establishing direct organelle exchange between connected cells (Rustom et al., 2004; reviewed in Demontis, 2004). The wing imaginal discs of Drosophila melanogaster, which will give rise to the wings and parts of the body wall of adult flies, provide an attractive system for studying cell protrusions within a developing tissue. In wing imaginal discs, a monolayer of columnar and squamous epithelial cells is arranged in a sac-like structure with the apical membranes facing an inter- Max Planck Institute of Molecular Cell Biology and Genetics, Dresden, Germany The Supplementary Material referred to in this article can be found at Grant sponsor: Deutsche Forschungsgemeinschaft; Grant number: DA586/4-1. Dr. Demontis present address is Harvard Medical School, Department of Genetics, 77 Avenue Louis Pasteur, Boston, MA *Correspondence to: Christian Dahmann, Max Planck Institute of Molecular Cell Biology and Genetics, Pfotenhauerstrasse 108, Dresden, Germany. dahmann@mpi-cbg.de DOI /dvdy Published online 13 September 2007 in Wiley InterScience ( Wiley-Liss, Inc.
2 PROTRUSIONS INTERCONNECT EPITHELIAL CELLS 3409 Fig. 1. Apical cell protrusions connect the columnar epithelium to the squamous epithelium in wing imaginal discs. A: Scheme of the Drosophila wing imaginal disc and location of apical cell protrusions. When viewed onto the plane of the epithelium (XY), cells of the squamous epithelium (s.e.) have a larger circumference than those of the columnar epithelium (c.e.). In cross-section (XZ), cells of the squamous and columnar epithelia are apposed with their apical membranes facing a lumen. Apical cell protrusions (Pr.) arise from the columnar epithelium and are directed toward the squamous epithelium. B,B : XZ view of a three-dimensional (3D) rendered, living wing imaginal disc expressing CD8 green fluorescent protein (GFP; green) in the dorsal compartment (ap-gal4, UAS-CD8-GFP) and stained with the lipophilic dye FM4-64 (red). In B, only the CD8-GFP channel is shown. Apical cell protrusions arise from the columnar epithelium (bottom) and extend toward the squamous epithelium (top). C,D: Apical views onto the columnar epithelium of the 3D-rendered tissue show that these apical protrusions are abundant and present over the entire tissue with no apparent regional preference. E H : The 3D rendering of a living wing imaginal disc expressing CD8-GFP (green) in cell clones (act5c GAL4, UAS-CD8-GFP) and stained with the lipophilic dye FM4-64 (red). Different views of the tissue are provided. E,E : An XY view of the columnar epithelium from the basal side shows cell clones belonging to the columnar epithelium that express CD8-GFP. F,F : An XY view of the squamous epithelium and the underlying columnar epithelium shows apical protrusions extending from the columnar cells. G H : Tilting of the 3D-rendered tissue shows that the apical protrusions of columnar cells extend to the level of the squamous epithelium, detected by the presence of a squamous cell expressing CD8-GFP (asterisk). I,I : A higher magnification XY view of apical protrusions at the level of the squamous epithelium is shown. Apical protrusions display enlarged terminal tips (arrowhead), local bulges (arrows), and are sometimes branched (asterisk). In addition, some apical protrusions were bent in their terminal tract when contacting the squamous epithelium. Scale bar 10 m in B D,E H, 5 m in I,I. nal lumen (Fig. 1A; reviewed in Cohen, 1993). Signaling among columnar cells as well as between columnar and squamous cells is important for growth and patterning of wing imaginal discs (reviewed in Ramirez-Weber and Kornberg, 2000; Gibson and Schubiger, 2001). Several protrusions have been identified in imaginal discs. The apical side of imaginal disc cells is decorated with microvilli (Poodry and Schneiderman, 1970; Ursprung, 1972) and filopodia have been described at their basal side (Eaton et al., 1995). In addition, three more kinds of protrusions have been observed, all of which have been implicated in exchanging signaling molecules important for the growth and patterning of wing imaginal disc cells. Cells at the periphery of the columnar cell sheet have long, planar filopodia-like protrusions, termed cytonemes, which connect to cells within the center of the columnar cell sheet (Ramirez-Weber and Kornberg, 1999). Columnar cells also display apical cell protrusions that are a few cell diameters long and that are involved in the process of lateral inhibition (De Joussineau et al., 2003). Furthermore, a subpopulation of squamous cells forms microtubulecontaining protrusions that extend through the lumen toward columnar cells (Cho et al., 2000; Gibson and Schubiger, 2000). A comprehensive analysis of the types of cell protrusions present in the wing imaginal disc has not been reported. It is therefore conceivable that we are currently underestimating the number and sort of cell protrusions present in this tissue. The identification and characterization of additional cell protrusions could be hampered by fixation of the tissue, which is known to result, at least in some cases, in the profound alteration or destruction of cell protrusions (Ramirez-Weber and Kornberg, 1999). Here, we have identified cellular protrusions of wing imaginal discs by expressing a membrane-bound form of green fluorescent protein (GFP) in columnar cells. Live, unfixed imaginal discs were then analyzed by laser scanning microscopy and three-dimensional (3D) rendering. We have identified and characterized two kinds of previously unreported protrusions. Protrusions of the apical plasma membrane that extended through the imaginal disc lumen and apparently contacted the squamous cells, and protrusions of the lateral plasma membrane that extended in between neighboring columnar cells. Both
3 3410 DEMONTIS AND DAHMANN kinds of protrusions could be identified with GFP-actin, a GFP-tagged version of the Dpp receptor Thickveins (Tkv), and the endosomal marker GFP-Rab5. Implications for signaling among wing imaginal disc cells are discussed. RESULTS Apical Protrusions of the Columnar Epithelium Contact the Apposing Squamous Epithelium in Live Drosophila Wing Imaginal Discs To identify and analyze cellular protrusions of columnar wing imaginal disc cells, we used the ap-gal4 driver to express CD8-GFP, a transmembrane protein routinely used to mark plasma membranes (Lee and Luo, 1999), in columnar cells of the dorsal compartment of wing imaginal discs. ap-gal4 is not active in the squamous wing imaginal disc cells, limiting CD8-GFP expression to the columnar cells. Third instar larvae were dissected, transferred to culture medium containing the lipophilic dye FM4-64 to stain plasma membranes of all wing imaginal discs cells, and imaged by confocal microscopy. Stacks of XY confocal sections were collected and rendered in three dimensions (see the Experimental Procedures section). We have identified protrusions extending from the apical plasma membrane and protrusions extending from the lateral plasma membrane of columnar cells. We will first describe the apical protrusions and in subsequent paragraphs the lateral protrusions. The apical surface of the columnar epithelium displayed many cell protrusions, ranging in length from 1 to 10 m (Fig. 1B D and Supplementary Movie S1, which can be viewed at jpages/ /suppmat). These apical protrusions appeared to be a common attribute of all wing imaginal disc columnar cells, with an average density of (S.D.) apical protrusions per cell (n 48). Apical protrusions projected through the lumen of the wing imaginal disc toward the squamous epithelium (scheme in Fig. 1A). To investigate in more detail the morphology of these apical protrusions, we expressed CD8-GFP in clones of cells. The 3D rendering showed that these cell protrusions arose from CD8-GFP expressing cells in the columnar epithelium (Fig. 1E,E ), passed through the lumen that separated the columnar from the squamous epithelium (Fig. 1G,G ), and reached and apparently contacted the squamous epithelium (Fig. 1F,F,H,H and Supplementary Movies S2,3). Most of these protrusions were linear, elongated structures; however, some protrusions were branched, especially in the terminal tracts. Sometimes, conversely, two protrusions initially distinct came in close contact with one another and apparently coalesced to form a single structure (Fig. 1I). Remarkably, most of these protrusions had an enlarged and roughly spherical terminal tip (Fig. 1F,I,I ) whose diameter of approximately 1 m exceeded the normal protrusion diameter of less than 0.2 m. Sometimes this enlarged terminal tip was the only part of the protrusion that contacted the squamous epithelium; however, in other cases, the terminal tract of the protrusion, preceding the tip, also seemed to contact the squamous epithelium (Fig. 1I,I ). Some apical protrusions had local bulges or widenings, which may have been due to vesicles inside the protrusion (Fig. 1I,I ), as previously reported for other types of cell protrusions (Ramirez-Weber and Kornberg, 1999; Rustom et al., 2004; Hsiung et al., 2005). We conclude that wing imaginal disc columnar cells display apical protrusions that contact the squamous epithelium and that have specific morphological features. Apical Protrusions Contain Actin Many cellular protrusions contain an actin-based core (reviewed in Rorth, 2003). To test whether apical protrusions of columnar cells also contain actin, we used ap-gal4 to express a GFP-actin fusion protein in the dorsal compartment of wing imaginal discs. Confocal micrographs through the columnar epithelium detected GFP-actin in the cortical area of dorsal columnar cells, as expected (Fig. 2B B ). Confocal micrographs taken at the level of the squamous epithelium of live wing imaginal discs showed GFPactin in dot-like structures in close proximity to the squamous epithelial cells (Fig. 2A A ), similar to the CD8- GFP labeled tips of apical protrusions (Fig. 1F,F ). The 3D rendering of the collected micrographs identified GFPactin on apical protrusions of columnar cells that extended across the lumen and that came in close contact with the apposing squamous epithelium (Fig. 2C E, Supplementary Movie S4), resembling CD8-GFP labeled apical protrusions. We conclude that the apical protrusions of columnar cells contain actin. Drosophila Prominin-like GFP Appears to Be Present on Apical Protrusions of Columnar Cells Microvilli are protrusions of the apical membrane of polarized epithelial cells, including wing imaginal disc cells, containing an actin-based core. To test whether the CD8-GFP labeled protrusions may correspond to microvilli, we investigated whether a marker for microvilli would identify CD8-GFP like apical protrusions. Members of the Prominin family of pentaspan transmembrane proteins are among the most specific markers of microvilli (Corbeil et al., 2001). In mammalian cells, endogenous Prominin-1 and a Prominin-2-GFP fusion protein selectively localize to microvilli (Weigmann et al., 1997; Fargeas et al., 2003). To date, two members of the prominin family, Prominin and Prominin-like, have been identified in the Drosophila melanogaster genome (Fargeas et al., 2003; Zelhof et al., 2006). To test whether Promininlike localizes to apical protrusions, we fused Drosophila Prominin-like to GFP and expressed the fusion protein using the GAL4/UAS system in a subset of columnar wing imaginal disc cells. By analyzing live wing imaginal discs expressing the Prominin-like- GFP fusion protein, we noticed, however, that the GFP fluorescence was undetectable (data not shown), possibly because the proper folding of the GFP molecule was impaired in the context of the fusion protein, as described for other GFP-fusion proteins
4 PROTRUSIONS INTERCONNECT EPITHELIAL CELLS 3411 Fig. 2. Green fluorescent protein (GFP) -actin localizes to apical protrusions of the wing disc columnar epithelium. A E: Different views of live wing imaginal disc expressing GFP-actin (green) in the dorsal compartment (ap-gal4, UAS-GFP-actin) and stained with the lipophilic dye FM4-64 (red). A B : Micrographs of single confocal XY sections through the squamous (A A ) and columnar (B B ) epithelium are shown. The dorsal compartment is to the top. Dot-like GFP-actin containing structures are detected in the squamous cells overlying the dorsal compartment of the columnar epithelium (A,A ). These structures presumably correspond to the cell protrusions arising from columnar epithelial cells (B B ) and presumably contact the squamous epithelial cells (red, A,A ). C E: Different views of the three-dimensional (3D) rendered tissue are shown. In C,C, a XZ view is depicted with the dorsal compartment to the right. GFP-actin labeled protrusions project from the columnar cells (bottom) toward squamous epithelium (top). In C, only the GFP-actin channel is shown. D E: Apical views onto the columnar epithelium of the 3D-rendered tissue show that these apical protrusions are abundant and present over the entire tissue with no apparent regional preference. Scale bars 10 m. (Pedelacq et al., 2006; and references therein). We therefore tested whether Prominin-like-GFP localized to apical protrusions of wing imaginal disc cells by immunostaining of fixed wing imaginal discs. To this purpose, we expressed either CD8-GFP or Prominin-like-GFP in the dorsal compartment of wing imaginal discs and compared the localization of these two transmembrane proteins by immunostaining against GFP. Zonula adherens, which localize to the apical region of the lateral plasma membrane, and the more basal region of the lateral plasma membrane were identified using antibodies against E-cadherin and Fasciclin III, respectively. Consistent with our analysis of live wing imaginal discs expressing CD8-GFP, optical cross-sections (XZ) showed that CD8- GFP localized to the apical and lateral membrane of expressing cells and that CD8-GFP was enriched apical to the zonula adherens (Fig. 3A,A ). By contrast to live wing imaginal discs, the lumen between the squamous epithelium and columnar epithelium could not be clearly resolved by confocal microscopy in the fixed tissue. We therefore analyzed XY confocal sections that, because they were slightly tilted relative to the plane of the squamous and columnar epithelia, displayed the squamous cells, the apical surface of the columnar cells, and the lumen in between the two epithelia (Fig. 3G). In these images, the larger circumference distinguishes squamous cells from columnar cells. We detected CD8-GFP in dot-like structures at the interface between the squamous epithelium and the columnar epithelium in the dorsal compartment, but not in the non CD8-GFP-expressing ventral compartment (Fig. 3B,B,C). These dot-like structures presumably corresponded to cross-sections of the apical protrusions that we have identified in live wing imaginal discs (Fig. 1). To test whether Prominin-like-GFP localizes to apical cell protrusions in fixed wing imaginal discs, we compared its localization with that of CD8-GFP expressing, fixed wing imaginal discs. In XZ confocal micrographs, Prominin-like-GFP localized to the plasma membrane apical to the zonula adherens and was mainly excluded from the lateral plasma membrane (Fig. 3D,D ). A weaker but reproducible staining of Prominin-like- GFP was also detected on the basal plasma membrane (Supplementary Figure S1). In apical XY confocal micrographs, we detected Prominin-like- GFP in dot-like structures at the interface between the squamous epithelium and the columnar epithelium in the dorsal compartment (Fig. 3E,E,F), similar to CD8-GFP in fixed wing imaginal discs (Fig. 3B,B,C). No such dot-like structures were detected in the ventral compartment. The localization of Prominin-like-GFP is consistent with CD8-GFP and Prominin-like-GFP identifying the same apical protrusions. Because members
5 3412 DEMONTIS AND DAHMANN of the Prominin family are markers for microvilli, this finding indicates that apical protrusions may correspond to microvilli. Lateral Protrusions of the Columnar Epithelium Extend in Between Cells Within the Same Epithelium In addition to apical protrusions, expression of CD8-GFP in clones of cells also identified previously uncharacterized protrusions that extended from the lateral plasma membrane of columnar cells (Fig. 4A A ). These lateral protrusions extended in between neighboring cells, usually one or two cells apart from the expressing cells. The lateral protrusions had a diameter of less than 0.2 m and appeared to be randomly distributed along the lateral side of the cell. On average, approximately (S.D.) lateral protrusions per cell (n 77) were detected on the apical most 8 m of the lateral plasma membrane. Of interest, we noticed that some of the lateral protrusions displayed an enlarged terminal tip (Fig. 4A ), similar to the apical protrusions. Prominin-like-GFP was not detected on the lateral membrane (Supplementary Figure S1), suggesting that Prominin-GFP is excluded from lateral protrusions. This finding indicates that lateral protrusions might have a distinct nature from the apical protrusions. By 3D-rendering of micrographs serially acquired along the Z-axis (see the Experimental Procedures section), we observed that some lateral protrusions extended in between neighboring cells parallel to the plane of the epithelium (XY) as well as perpendicular to the plane of the epithelium (XZ), that is parallel to the lateral membrane of columnar cells (Fig. 4B B,D E ; see Fig. 4C for a scheme). To test whether these lateral protrusions contained, like the apical protrusions, actin, we expressed a GFP-actin fusion protein in clones of few cells. As shown in Figure 4F,F, GFP-actin identified lateral cell protrusions of columnar epithelial cells that closely resembled the CD8-GFP labeled lateral protrusions, indicating that the lateral protrusions contained actin. Tkv-GFP Identifies Apical and Basal Protrusions of Columnar Wing Imaginal Disc Cells The growth and patterning of wing imaginal discs requires the communication between cells through the exchange of secreted signaling molecules. The location and connectivity of apical protrusions (columnar cells to squamous cells) and lateral protrusions (columnar cells to columnar cells) makes them well suited to play a role in the exchange of such signaling molecules. Dpp is a member of the transforming growth factor- family of secreted molecules that is important for the growth and patterning of wing imaginal discs (Spencer et al., 1982; Padgett et al., 1987; Capdevila and Guerrero, 1994; Zecca et al., 1995) and that has been shown to be present in the lumen between columnar cells and squamous cells (Gibson et al., 2002). To test whether apical or lateral protrusions could mediate the communication between cells by displaying a receptor for Dpp, we expressed a GFP-tagged version of the Dpp receptor Thickveins (Tkv; Hsiung et al., 2005) in the dorsal compartment of columnar wing imaginal disc cells. XY confocal sections through the lumen of live wing imaginal discs were analyzed. Most of the Tkv-GFP fluorescence was detected on the nonprotruding plasma membrane of expressing cells. In addition, Tkv-GFP was also present on dot-like structures. These dot-like structures were costained with the lipophilic dye FM4-64 and presumably represent cross-sections of apical protrusions (Fig. 5A B ). To test this further, we rendered the tissue in three dimensions. As shown in Figure 5C, Tkv- GFP labeled protrusions of the apical plasma membrane projected toward the squamous epithelium, indicating that Tkv-GFP was present on apical protrusions (see also Supplementary Movies S5 and S6). To test whether Tkv-GFP was also present on lateral protrusions, we expressed Tkv-GFP in clones of a few cells. As shown in Figure 5D D, Tkv- GFP identified protrusions of the lateral membrane that resembled CD8-GFP labeled lateral protrusions. Thus, we detected Tkv-GFP on both apical and lateral protrusions of wing disc columnar cells. The presence of Tkv-GFP on apical and lateral protrusions is consistent with the notion that these cell protrusions could mediate communication between cells. GFP-Rab5 Compartments Are Detected Inside Both Apical and Lateral Protrusions Our analysis of CD8-GFP labeled apical protrusions of live wing imaginal disc cells revealed bulges that exceeded the normal protrusion diameter of 0.2 m (Fig. 1I,I ), suggesting that vesicles may be transported inside these cell protrusions. To test this hypothesis, we expressed GFP-Rab5 (Wucherpfennig et al., 2003), a marker of early endosomes (Chavrier et al., 1990), in clones of a few cells and analyzed its localization in live wing imaginal discs. GFP-Rab5 compartments were detected in XY confocal micrographs corresponding to the luminal space or to the squamous epithelium overlying the expressing cells (Fig. 6A A ). FM4-64 labeled plasma membrane was encircling GFP-Rab5 compartments (Fig. 6A A ), suggesting that GFP-Rab5 compartments were inside apical protrusions. In addition, by analyzing XY micrographs corresponding to the lateral membrane of columnar cells, we observed the presence of distinct GFP- Rab5 compartments in lateral protrusions (Fig. 6B F ). Thus, both apical and lateral protrusions contain GFP- Rab5 compartments. DISCUSSION Cell protrusions are a common attribute of most cell types and play fundamental roles in development and disease. Here, we identify to-date uncharacterized protrusions of the apical and lateral plasma membrane in the Drosophila melanogaster wing imaginal disc columnar epithelium. We provide evidence that both apical and lateral protrusions contain actin and, furthermore, that apical protrusions might be microvilli. We find a receptor for the Dpp signaling molecule, Tkv- GFP, as well as the endosomal marker Rab5-GFP, on lateral and apical protrusions, consistent with the view that
6 PROTRUSIONS INTERCONNECT EPITHELIAL CELLS 3413 these protrusions are involved in cellto-cell communication. Apical Protrusions Connect Columnar Cells With Squamous Cells Microvilli are a paradigm for apical cell protrusions of epithelia (e.g., Heintzelman and Mooseker, 1992). Several electron microscopic studies have described microvilli on the apical surface of columnar wing imaginal disc cells (Schlichting et al., 2006; Ur- Fig. 3. Fig. 4. Fig. 3. CD8-green fluorescent protein (GFP) and Prominin-like-GFP localize to apical punctuate structures of the columnar epithelium in fixed wing imaginal discs. A F: Immunostaining of wing imaginal disc expressing CD8-GFP (A C, ap-gal4, UAS-CD8-GFP) or a Promininlike-GFP fusion protein (D F, ap-gal4, UASprominin-like-GFP) in the dorsal compartment stained for GFP (green), DE-Cadherin (DE-Cad, blue), and Fasciclin III (FasIII, red). A,A,D,D : XZ views of the dorsal compartment (apical to the top) show that both CD8-GFP and Promininlike-GFP are enriched on the apical side of columnar epithelial cells. B,B,E,E : Apical XY sections (dorsal compartment to the top) are shown. C and F are higher magnifications of the boxed areas shown in B and E. CD8-GFP and Prominin-like-GFP localize to apical punctuate structures of the wing disc columnar epithelium. Because the confocal sections are slightly tilted, both columnar and squamous epithelial cells are depicted in different parts of the micrographs. G: A cartoon depicting the position of the XY sections shown in B,B,E,E. Scale bars 20 m in A B,D E ; 5 m in C,F. Fig. 4. Lateral protrusions of columnar epithelial cells extend in between neighboring cells and contain green fluorescent protein (GFP) -actin. A A : XY micrographs of a live wing imaginal disc expressing CD8-GFP (green) in clones of columnar cells (act5c GAL4, UAS-CD8-GFP) and stained with the lipophilic dye FM4-64 (red). Protrusions (A, asterisks) arising from the lateral membrane of columnar cells extend in between neighboring cells. B,B : Three-dimensional (3D) rendering of the columnar epithelium containing CD8-GFP expressing clones. Only the GFP channel (white) is shown. Basolateral (B) and basal (B ) views of the 3D-rendered tissue are depicted. Yellow arrows indicate the X-axis and the Y-axis and red arrows the Z- axis. Lateral protrusions arise from the lateral membrane and can extend along the Z-axis. C: Scheme of protrusions arising from the lateral membrane of wing disc columnar cells. D E : Higher magnifications of the 3D-rendered tissue shown in B. F F : XY-micrographs of a live wing imaginal disc expressing GFPactin (green) in clones of columnar cells (act5c GAL4, UAS-GFP-actin) and stained with the lipophilic dye FM4-64 (red). Protrusions arising from the lateral membrane of columnar cells extend in between neighboring cells. Scale bars 5 m in A A,F F, 10 m in B,B.
7 3414 DEMONTIS AND DAHMANN Fig. 5. A green fluorescent protein (GFP) -tagged form of the Dpp receptor Thickveins (Tkv) localizes to both apical and lateral protrusions of the wing disc columnar epithelium. A C : Different views of a live wing imaginal disc expressing Tkv-GFP (green) in the dorsal compartment (ap-gal4, UAS-tkv-GFP) and stained with the lipophilic dye FM4-64 (red). A B : Micrographs of single confocal XY sections through the lumen (A A ) and columnar epithelium (B B ) are shown. The dorsal compartment is to the top. FM4-64 labels within the lumen dots that correspond to cross-sections of apical protrusions (A). Tkv-GFP colocalizes with some of these dots (A ). C,C : A XZ view of the threedimensional (3D) -rendered tissue is depicted with the dorsal compartment to the front. Tkv-GFP labeled protrusions project from the columnar cells (bottom) toward the squamous epithelium (top). In C, only the Tkv-GFP channel is shown. Tkv-GFP expressing cells are shorter and the lumen between columnar epithelium and squamous epithelium wider presumably as a consequence of the role of Dpp signaling in maintaining proper columnar architecture (Gibson and Perrimon, 2005; Shen and Dahmann, 2005). Under this experimental condition, FM4-64 staining identifies within the dorsal wing disc pouch protrusions of the squamous epithelium projecting toward the columnar epithelium (red, see also Supplementary Figure S2). D D : XY micrographs of live wing imaginal discs expressing Tkv-GFP (green) in clones of columnar cells (act5c GAL4, UAS-tkv-GFP) and stained with the lipophilic dye FM4-64 (red). Tkv-GFP labeled protrusions arising from the lateral membrane of columnar cells extend in between neighboring cells. Scale bars 10 m in A C, 2 m in D D. Fig. 6. Rab5 green fluorescent protein (GFP) vesicles are detected on apical and lateral protrusions. A F : XY micrographs of live wing imaginal discs expressing GFP-Rab5 (green) in clones of columnar cells (act5c GAL4, UAS-GFP-Rab5) and stained with the lipophilic dye FM4-64 (red). A A : A micrograph of an apical protrusion contacting the squamous epithelium, identified by FM4-64 staining (red). GFP-Rab5 compartments are detected at the tip of apical protrusions. B B : Micrograph of the columnar cell clone from which the apical protrusion shown in A arises. A lateral protrusion extends in between neighboring cells and contains a GFP-Rab5 compartment. C F : Serial XY sections along the Z-axis. GFP- Rab5 is only detected on cellular extensions in some sections, but not others, indicating that these structures are cell protrusions. Numbers to the right indicate the distance from the squamous epithelium along the Z-axis. Scale bars 2.5 m in A B, 5 m in C F.
8 PROTRUSIONS INTERCONNECT EPITHELIAL CELLS 3415 sprung, 1972; Poodry and Schneiderman, 1970). As seen in electron microscopic preparations, microvilli are numerous, short (on average approximately 500 nm long) protrusions extending into the lumen of wing imaginal discs. Using CD8-GFP, we have identified protrusions that extend from the apical surface of columnar wing imaginal disc cells. Unlike the microvilli identified by electron microscopy, these protrusions are long (up to 10 m), extend through the entire lumen getting in close contact to the overlaying squamous cells, and display enlarged terminal tips (Fig. 1). Despite these apparent differences, several observations suggest that apical CD8-GFP labeled protrusions and microvilli are one and the same structure. First, both kinds of protrusions are extensions of the apical plasma membrane. Second, both kinds of protrusions contain actin. Third, the two kinds of protrusions have a comparable diameter of less than 0.2 m. Finally, our data are consistent with the notion that apical protrusions contain the microvilli marker Prominin-like. It is therefore conceivable that the morphological differences between microvilli, as seen in electron microscopic preparations, and CD8-GFP labeled protrusions, as seen in live preparations, are a consequence of the technique used to visualize the protrusions. If CD8-GFP labeled protrusions were microvilli, this would indicate that microvilli of columnar cells extended through the entire lumen and got in close contact to the overlying squamous epithelium. Analysis of the Drosophila follicular epithelium has provided evidence that also in this epithelium microvilli can contact overlying cells, in this case the oocyte (Mahowald, 1972; Mahowald and Kambysellis, 1980). Microvilli of follicle cells projecting toward the oocyte have also been described in amphibians and mammals (Dantzer, 1985; Villecco et al., 2002; Makabe et al., 2006), suggesting that follicle cell microvilli might establish connections with oocytes in many animal species. Considering that, over the past decades, microvilli have been identified in a plethora of epithelia and organisms, we speculate that microvilli might connect apposed epithelia during different developmental stages in several organisms. This raises therefore the intriguing possibility that microvilli, in addition to increasing the surface area of cells, also serve additional functions, for example, to mediate the communication or cohesion of cells from apposed cell layers. What is the function of apical protrusions? Cells of the squamous and columnar epithelium exchange signals that are important for the growth and patterning of wing imaginal discs (Cho et al., 2000; Gibson and Schubiger, 2000; Gibson et al., 2002; Pallavi and Shashidhara, 2003, 2005). Cells of the squamous epithelium display long microtubule-based protrusions directed toward the columnar epithelium (Cho et al., 2000; Gibson and Schubiger, 2000). It has been proposed that these protrusions could mediate the exchange of instructive signals between the two apposed epithelia that are important for proliferation and patterning of imaginal discs (Cho et al., 2000; Gibson and Schubiger, 2000). However, these protrusions were detected mainly in the prospective hinge and notum regions, but not in the pouch region of the wing imaginal disc that gives rise to the adult wing (Gibson and Schubiger, 2000). It is therefore uncertain whether squamous wing imaginal disc cells could also exchange signals with the underlying columnar pouch cells through protrusions. Our results now open up the possibility that the apical protrusions of columnar cells, which are present on all columnar cells, fulfill the role in transmitting signals between the two apposed epithelia. Other possible functions for these protrusions include the provision of physical links between the two epithelia, which might be important for maintaining the disc-like shape of the wing imaginal disc. Tools to specifically ablate these protrusions will be ultimately required to address the function of these apical protrusions. Lateral Protrusions Interconnect Cells Within the Columnar Epithelium The development of the wing imaginal disc and other epithelia relies on short-range cell interactions among columnar cells (e.g., Diaz-Benjumea and Cohen, 1995; Rulifson and Blair, 1995; Zecca et al., 1995; Neumann and Cohen, 1996; Milan et al., 2001, 2002). Short-range cell interactions could be mediated by the spread of signaling molecules through the extracellular space in between cells (reviewed in Teleman et al., 2001; Entchev and Gonzalez-Gaitan, 2002; Vincent and Dubois, 2002). In addition, shortrange cell interactions could be mediated by specialized cellular structures. Gap junctions are present between neighboring cells of the columnar epithelium of imaginal discs (Agrell, 1968; Poodry and Schneiderman, 1970). However, exchange of signals by means of gap junctions is limited to molecules of small size ( 1 kda; Loewenstein, 1981). Electron microscopy studies have further identified cytoplasmic bridges in the columnar epithelium of imaginal discs (Poodry and Schneiderman, 1970). Cytoplasmatic bridges are short cellular protrusions of the basolateral membrane that connect neighboring cells and establish direct membrane and cytoplasmic continuity between connected cells, indicating that local exchange of information could occur by means of these cellular structures (Poodry and Schneiderman, 1970). However, cytoplasmic bridges are similar to remnants of cytokinesis (Poodry and Schneiderman, 1970), suggesting that they only connect sister cells and would, therefore, not be of major importance for pattern formation in imaginal discs, which has been shown to be independent of cell lineage (Bryant and Schneiderman, 1969; Garcia-Bellido and Merriam, 1969). Finally, recent reports suggest that short-range communication in the wing imaginal disc can occur by means of cellular protrusions (De Joussineau et al., 2003). We observed in live wing imaginal discs that columnar cells have protrusions of the lateral membrane that extend in between the lateral plasma membranes of neighboring cells. In common with the apical protrusions, these lateral protrusions also displayed enlarged terminal tips (Fig. 4D D ), suggesting that this might be a common morphological feature of cell protrusions in live tissue. The localization and direction of these protrusions is consistent with a role in mediating some of the short-range sig-
9 3416 DEMONTIS AND DAHMANN naling that has been observed in wing imaginal discs. In principle, connections established by lateral protrusions could result in a global interaction network facilitating the exchange of survival information and positional cues. Moreover, the lateral protrusions may also contribute to longrange signaling between cells, for example, by increasing the rate of transport of signaling molecules. The apical and lateral protrusions described here differ from cytonemes and protrusions emanating from sensory organ precursor cells, two kinds of cellular protrusions that were recently described in wing imaginal discs (Ramirez-Weber and Kornberg, 1999; Hsiung et al., 2005; De Joussineau et al., 2003). Apical protrusions project toward the overlying squamous epithelium, whereas cytonemes and sensory organ precursor cell protrusions project toward columnar cells. Similar to the lateral protrusions described here, sensory organ precursor cell protrusions are short protrusions interconnecting cells in the columnar epithelium. However, sensory organ precursor cell protrusions were mainly detected at the basal side of cells or apical to the zonula adherens (De Joussineau et al., 2003). In contrast, the lateral protrusions observed here mainly emanate from below the zonula adherens. The lateral protrusions also differ from cytonemes present in the wing imaginal disc pouch in that they are shorter (3 5 m instead of 20 m in length) and that they apparently emanate in all directions and are not, as cytonemes, directed toward the anteroposterior or dorsoventral compartment boundaries. Apical and Lateral Protrusions and Intercellular Signaling Several recent studies have identified signaling molecules or their receptors associated with various cellular protrusions, providing evidence that these cellular protrusions are important for mediating intercellular signaling. Molecules associated with protrusions include Sevenless, a receptor for the Boss ligand (Tomlinson et al., 1987); Delta, a Notch ligand (De Joussineau et al., 2003); Scabrous, a signaling molecule (Chou and Chien, 2002); and Tkv, a receptor for Dpp (Hsiung et al., 2005) on cellular protrusions in Drosophila and members of the erbb/her family, receptors for EGF and related ligands, on extensions in mammalian cells (Lidke et al., 2004). We extend these findings by showing that Tkv-GFP can also localize to apical and lateral cell protrusions. We note, however, that we do not find Tkv-GFP enriched on these protrusions compared with the nonprotruding plasma membrane, nor do we find Tkv-GFP in these protrusions on motile punctae, as previously described for cytonemes (Hsiung et al., 2005). A further indication that apical and lateral protrusions might be involved in mediating signals between cells is our finding that Rab5-GFP localizes to these protrusions. One way by which cellular protrusions convey instructive cues is by endocytosis and trafficking of signaling complexes on endosomes. The early endosomal protein Rab5a localizes, for example, to axons and dendrites of neurons (de Hoop et al., 1994) and is important for coupling clathrin-dependent endocytosis to axonal retrograde transport (Deinhardt et al., 2006). Furthermore, several signaling molecules are associated with endosomes (reviewed in Miaczynska et al., 2004). It will therefore be interesting to learn whether endocytosis takes place from the plasma membrane of apical and lateral protrusions and whether endosomes in these protrusions associate with signaling complexes. EXPERIMENTAL PROCEDURES Molecular Cloning and Fly Stocks To generate the UAS-prominin-like- GFP transgene, the enhanced GFP (EGFP) coding sequence was polymerase chain reaction (PCR) amplified with primers 5 -CGGCTCGAGATG- GTGAGCAAGGGCGAGGAGCTG-3 and 5 -CTAGTCTAGATTACTTGTA- CAGCTCGTCCATGCC-3 (the underlined sequences are XhoI and XbaI restriction sites used for cloning) using pegfp-n1 (Clontech) as template (nn ), and cloned in the puast vector (Brand and Perrimon, 1993). Drosophila melanogaster promininlike (CG7740) coding sequence (nucleotides of AF197345) and 5 - UTR (Fargeas et al., 2003) were PCR amplified from cdna clone LD16666 (kindly provided by Denis Corbeil) with primers: 5 -CCGCTCGAGTCG- ATCGGATAAATTTGGAATAGAAAA- GGC-3 and 5 -CCGCTCGAGATCCT- GCTCGGAGGCACCCGGATAG-3 (the underlined sequences are XhoI restriction sites used for cloning). The PCR product was digested with XhoI and cloned in frame in the puast-egfp vector. The correct nucleotide sequences of the cloned PCR products were confirmed by sequencing before injection into y w embryos to obtain transgenic flies. Additional fly stocks used in this work include ap-gal4 (Calleja et al., 1996), act5c CD2 GAL4 (Pignoni and Zipursky, 1997), UAS-CD8-GFP (Lee and Luo, 1999), UAS-tkv-GFP (Hsiung et al., 2005), UAS-GFP-Rab5 (Wucherpfennig et al., 2003), and UAS-GFP-actin (Verkhusha et al., 1999). Marked clones composed of few cells were generated by Flp-mediated mitotic recombination (Golic and Lindquist, 1989) subjecting third-instar larvae to a 34 C heat-shock for 30 min at 20 hr before dissection. Immunohistochemistry Late third-instar larvae were dissected in ice-cold Ringer s solution and fixed for 40 min at room temperature in PEM solution (0.1 M PIPES, 2 mm MgSO 4, 1 mm EGTA) with 4% formaldehyde and 0.1% Triton X-100. Larval carcasses were washed in PBT (phosphate buffered saline, 0.1% bovine serum albumin, 0.1% Triton X-100) and incubated with appropriate primary antibodies for 1 hr at room temperature. Subsequently, larval carcasses were washed in PBT, blocked with PBT containing 5% heatinactivated goat serum, and incubated with fluorophore-conjugated secondary antibodies for 1 hr at room temperature. Wing imaginal discs were then washed, dissected out of the carcasses, and mounted in 50% glycerol, 0.1 M sodium carbonate ph 9, and PPDA (p-phenylene diamine, Sigma, Taufkirchen). Stained tissues were observed with a ZEISS Laser Scan-
10 PROTRUSIONS INTERCONNECT EPITHELIAL CELLS 3417 ning Microscope 510 Meta (Carl Zeiss AG, Jena). Primary antibodies used in this study were mouse anti-fasciclin III 7G10 (1:100, Developmental Studies Hybridoma Bank), rabbit anti- GFP (1:2,000, Clontech ), and rat anti DE-Cad (DCAD1 and DCAD2, 1:100; (Oda et al., 1994). Secondary antibodies used were Alexa- 488-, Alexa-594- (Molecular Probes, Eugene, OR), and Cy5- (Jackson ImmunoResearch, West Grove, PA) conjugated anti-rabbit, anti-mouse, or anti-rat IgG (1:200). Laser Scanning Microscopy of Live Wing Imaginal Discs and 3D Rendering Wing imaginal discs were dissected in Ringer s solution from third-instar larvae and processed as previously described (Greco et al., 2001). Briefly, live wing imaginal discs were transferred to a microscope glass-slide with a chamber delimited by double-sided adhesive tape containing either Shields and Sang s M3 culture medium (Shields and Sang, 1970) or Ringer s solution with 9 M FM4-64 (Molecular Probes). Wing imaginal discs were oriented with the squamous epithelium facing the coverslip. The wing imaginal discs were observed immediately using a ZEISS LSM 510 laser scanning confocal microscope (Carl Zeiss AG), usually using a water immersion 63 objective. Stacks of closely spaced (0.05 m) sections were taken starting from the squamous epithelium progressing toward the basolateral side of the columnar epithelial cells. Confocal sections were then exported as TIF files and assembled in a library for processing using the 3D rendering software Volocity v2.5 (Improvision, Lexington, MA). Quicktime movies and single snapshots were then obtained from the 3D rendering of the tissue. Cytonemes were not observed in our preparations as their identification requires a slight flattening of imaginal discs (Ramirez-Weber and Kornberg, 1999; Hsiung et al., 2005). All images shown here refer to the pouch region of wing imaginal discs. ACKNOWLEDGMENTS We thank Konrad Basler, Marcos González-Gaitán, Suzanne Eaton, Tom Kornberg, and the Bloomington Drosophila Stock Center for fly stocks; Tadashi Uemura and the Developmental Studies Hybridoma Bank for antibodies; and Denis Corbeil for the cdna clone LD We also thank Suzanne Eaton and Andrew Oates for critical comments on the manuscript. This work was supported by the Max Planck Society and a grant from the Deutsche Forschungsgemeinschaft (C.D.). REFERENCES Agrell IPS Differentiation of the membrane systems in the cells of imaginal disks. Z Zellforsch 88: Brand AH, Perrimon N Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118: Bryant PJ, Schneiderman HA Cell lineage, growth, and determination in the imaginal leg discs of Drosophila melanogaster. Dev Biol 20: Calleja M, Moreno E, Pelaz S, Morata G Visualization of gene expression in living adult Drosophila. Science 274: Capdevila J, Guerrero I Targeted expression of the signaling molecule decapentaplegic induces pattern duplications and growth alterations in Drosophila wings. EMBO J 13: Chavrier P, Parton RG, Hauri HP, Simons K, Zerial M Localization of low molecular weight GTP binding proteins to exocytic and endocytic compartments. Cell 62: Cho KO, Chern J, Izaddoost S, Choi KW Novel signaling from the peripodial membrane is essential for eye disc patterning in Drosophila. Cell 103: Chou YH, Chien CT Scabrous controls ommatidial rotation in the Drosophila compound eye. Dev Cell 3: Cohen SM Imaginal disc development. In: Bate M, Martinez Arias A, editors. The development of Drosophila melanogaster. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press. Corbeil D, Roper K, Fargeas CA, Joester A, Huttner WB Prominin: a story of cholesterol, plasma membrane protrusions and human pathology. Traffic 2: Dantzer V Electron microscopy of the initial stages of placentation in the pig. Anat Embryol (Berl) 172: de Hoop MJ, Huber LA, Stenmark H, Williamson E, Zerial M, Parton RG, Dotti CG The involvement of the small GTP-binding protein Rab5a in neuronal endocytosis. Neuron 13: De Joussineau C, Soule J, Martin M, Anguille C, Montcourrier P, Alexandre D Delta-promoted filopodia mediate long-range lateral inhibition in Drosophila. Nature 426: Deinhardt K, Salinas S, Verastegui C, Watson R, Worth D, Hanrahan S, Bucci C, Schiavo G Rab5 and Rab7 control endocytic sorting along the axonal retrograde transport pathway. Neuron 52: Demontis F Nanotubes make big science. PLoS Biol 2:E215. Diaz-Benjumea FJ, Cohen SM Serrate signals through Notch to establish a Wingless-dependent organizer at the dorsal/ventral compartment boundary of the Drosophila wing. Development 121: Eaton S, Auvinen P, Luo L, Jan YN, Simons K CDC42 and Rac1 control different actin-dependent processes in the Drosophila wing disc epithelium. J Cell Biol 131: Entchev EV, Gonzalez-Gaitan MA Morphogen gradient formation and vesicular trafficking. Traffic 3: Fargeas CA, Florek M, Huttner WB, Corbeil D Characterization of prominin-2, a new member of the prominin family of pentaspan membrane glycoproteins. J Biol Chem 278: Frolenkov GI, Belyantseva IA, Friedman TB, Griffith AJ Genetic insights into the morphogenesis of inner ear hair cells. Nat Rev Genet 5: Fulga TA, Rorth P Invasive cell migration is initiated by guided growth of long cellular extensions. Nat Cell Biol 4: Garcia-Bellido A, Merriam JR Cell lineage of the imaginal discs in Drosophila gynandromorphs. J Exp Zool 170: Gibson MC, Perrimon N Extrusion and death of DPP/BMP-compromised epithelial cells in the developing Drosophila wing. Science 307: Gibson MC, Schubiger G Peripodial cells regulate proliferation and patterning of Drosophila imaginal discs. Cell 103: Gibson MC, Schubiger G Drosophila peripodial cells, more than meets the eye? Bioessays 23: Gibson MC, Lehman DA, Schubiger G Lumenal transmission of decapentaplegic in Drosophila imaginal discs. Dev Cell 3: Golic KG, Lindquist S The FLP recombinase of yeast catalyzes site-specific recombination in the Drosophila genome. Cell 59: Greco V, Hannus M, Eaton S Argosomes: a potential vehicle for the spread ofmorphogensthroughepithelia.cell106: Heintzelman MB, Mooseker MS Assembly of the intestinal brush border cytoskeleton. Curr Top Dev Biol 26: Hsiung F, Ramirez-Weber FA, Iwaki DD, Kornberg TB Dependence of Drosophila wing imaginal disc cytonemes on Decapentaplegic. Nature 437: Jacinto A, Wood W, Balayo T, Turmaine M, Martinez-Arias A, Martin P Dy-
11 3418 DEMONTIS AND DAHMANN namic actin-based epithelial adhesion and cell matching during Drosophila dorsal closure. Curr Biol 10: Lee T, Luo L Mosaic analysis with a repressible cell marker for studies of gene function in neuronal morphogenesis. Neuron 22: Lidke DS, Nagy P, Heintzmann R, Arndt- Jovin DJ, Post JN, Grecco HE, Jares- Erijman EA, Jovin TM Quantum dot ligands provide new insights into erbb/her receptor-mediated signal transduction. Nat Biotechnol 22: Loewenstein WR Junctional intercellular communication: the cell-to-cell membrane channel. Physiol Rev 61: Louvard D, Kedinger M, Hauri HP The differentiating intestinal epithelial cell: establishment and maintenance of functions through interactions between cellular structures. Annu Rev Cell Biol 8: Mahowald AP Ultrastructural observations on oogenesis in Drosophila. J Morphol 137: Mahowald AP, Kambysellis MP Oogenesis. In: Ashburner M, Wright TRF, editors. The genetics and biology of Drosophila. New York: Academic Press. p Makabe S, Naguro T, Stallone T Oocyte-follicle cell interactions during ovarian follicle development, as seen by high resolution scanning and transmission electron microscopy in humans. Microsc Res Tech 69: Martin-Blanco E, Pastor-Pareja JC, Garcia-Bellido A JNK and decapentaplegic signaling control adhesiveness and cytoskeleton dynamics during thorax closure in Drosophila. Proc Natl Acad Sci U S A 97: Marzesco AM, Janich P, Wilsch-Brauninger M, Dubreuil V, Langenfeld K, Corbeil D, Huttner WB Release of extracellular membrane particles carrying the stem cell marker prominin-1 (CD133) from neural progenitors and other epithelial cells. J Cell Sci 118: Miaczynska M, Pelkmans L, Zerial M Not just a sink: endosomes in control of signal transduction. Curr Opin Cell Biol 16: Milan M, Weihe U, Perez L, Cohen SM The LRR proteins capricious and Tartan mediate cell interactions during DV boundary formation in the Drosophila wing. Cell 106: Milan M, Perez L, Cohen SM Shortrange cell interactions and cell survival in the Drosophila wing. Dev Cell 2: Neumann CJ, Cohen SM A hierarchy of cross-regulation involving Notch, wingless, vestigial and cut organizes the dorsal/ventral axis of the Drosophila wing. Development 122: Oda H, Uemura T, Harada Y, Iwai Y, Takeichi M A Drosophila homolog of cadherin associated with armadillo and essential for embryonic cell cell adhesion. Dev Biol 165: Padgett RW, St Johnston RD, Gelbart WM A transcript from a Drosophila pattern gene predicts a protein homologous to the transforming growth factorbeta family. Nature 325: Pallavi SK, Shashidhara LS Egfr/ Ras pathway mediates interactions between peripodial and disc proper cells in Drosophila wing discs. Development 130: Pallavi SK, Shashidhara LS Signaling interactions between squamous and columnar epithelia of the Drosophila wing disc. J Cell Sci 118: Pedelacq JD, Cabantous S, Tran T, Terwilliger TC, Waldo GS Engineering and characterization of a superfolder green fluorescent protein. Nat Biotechnol 24: Pellikka M, Tanentzapf G, Pinto M, Smith C, McGlade CJ, Ready DF, Tepass U Crumbs, the Drosophila homologue of human CRB1/RP12, is essential for photoreceptor morphogenesis. Nature 416: Pignoni F, Zipursky SL Induction of Drosophila eye development by decapentaplegic. Development 124: Poodry CA, Schneiderman HA The ultrastructure of the developing leg of Drosophila melanogaster. Wilhelm Roux Arch 166:1 44. Ramirez-Weber FA, Kornberg TB Cytonemes: cellular processes that project to the principal signaling center indrosophilaimaginaldiscs. Cell97: Ramirez-Weber FA, Kornberg TB Signaling reaches to new dimensions in Drosophila imaginal discs. Cell 103: Ritzenthaler S, Suzuki E, Chiba A Postsynaptic filopodia in muscle cells interact with innervating motoneuron axons. Nat Neurosci 3: Rorth P Communication by touch: role of cellular extensions in complex animals. Cell 112: Rulifson EJ, Blair SS Notch regulates wingless expression and is not required for reception of the paracrine wingless signal during wing margin neurogenesis in Drosophila. Development 121: Rustom A, Saffrich R, Markovic I, Walther P, Gerdes HH Nanotubular highways for intercellular organelle transport. Science 303: Schlichting K, Wilsch-Brauninger M, Demontis F, Dahmann C Cadherin Cad99C is required for normal microvilli morphology in Drosophila follicle cells. J Cell Sci 119: Shen J, Dahmann C Extrusion of cells with inappropriate Dpp signaling from Drosophila wing disc epithelia. Science 307: Shields G, Sang JH Characteristics of five cell types appearing during in vitro culture of embryonic material from Drosophila melanogaster. J Embryol Exp Morphol 23: Spencer FA, Hoffmann FM, Gelbart WM Decapentaplegic: a gene complex affecting morphogenesis in Drosophila melanogaster. Cell 28: Teleman AA, Strigini M, Cohen SM Shaping morphogen gradients. Cell 105: Tomlinson A, Bowtell DD, Hafen E, Rubin GM Localization of the sevenless protein, a putative receptor for positional information, in the eye imaginal disc of Drosophila. Cell 51: Ursprung H The fine structure of imaginal disks. In: The biology of imaginal disks. New York: Springer Verlag. Vasioukhin V, Bauer C, Yin M, Fuchs E Directed actin polymerization is the driving force for epithelial cell cell adhesion. Cell 100: Verkhusha VV, Tsukita S, Oda H Actin dynamics in lamellipodia of migrating border cells in the Drosophila ovary revealed by a GFP-actin fusion protein. FEBS Lett 445: Villecco EI, Genta SB, Sanchez Riera AN, Sanchez SS Ultrastructural characteristics of the follicle cell-oocyte interface in the oogenesis of Ceratophrys cranwelli. Zygote 10: Vincent JP, Dubois L Morphogen transport along epithelia, an integrated trafficking problem. Dev Cell 3: Weigmann A, Corbeil D, Hellwig A, Huttner WB Prominin, a novel microvilli-specific polytopic membrane protein of the apical surface of epithelial cells, is targeted to plasmalemmal protrusions of non-epithelial cells. Proc Natl Acad Sci U S A 94: Wood W, Jacinto A, Grose R, Woolner S, Gale J, Wilson C, Martin P Wound healing recapitulates morphogenesis in Drosophila embryos. Nat Cell Biol 4: Wucherpfennig T, Wilsch-Brauninger M, Gonzalez-Gaitan M Role of Drosophila Rab5 during endosomal trafficking at the synapse and evoked neurotransmitter release. J Cell Biol 161: Zecca M, Basler K, Struhl G Sequential organizing activities of engrailed, hedgehog and decapentaplegic in the Drosophilawing. Development121: Zelhof AC, Hardy RW, Becker A, Zuker CS Transforming the architecture of compound eyes. Nature 443:
The Imperatives of Context and Contour for Morphogen Dispersion
 2252 Biophysical Journal Volume 103 December 2012 2252 2256 The Imperatives of Context and Contour for Morphogen Dispersion Thomas B. Kornberg* Cardiovascular Research Institute, University of California,
2252 Biophysical Journal Volume 103 December 2012 2252 2256 The Imperatives of Context and Contour for Morphogen Dispersion Thomas B. Kornberg* Cardiovascular Research Institute, University of California,
Developmental Biology
 Developmental Biology 334 (2009) 161 173 Contents lists available at ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology Wingless signaling and the control of cell
Developmental Biology 334 (2009) 161 173 Contents lists available at ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology Wingless signaling and the control of cell
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins
 13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
Supplementary Materials for
 www.sciencemag.org/cgi/content/full/science.1244624/dc1 Supplementary Materials for Cytoneme-Mediated Contact-Dependent Transport of the Drosophila Decapentaplegic Signaling Protein Sougata Roy, Hai Huang,
www.sciencemag.org/cgi/content/full/science.1244624/dc1 Supplementary Materials for Cytoneme-Mediated Contact-Dependent Transport of the Drosophila Decapentaplegic Signaling Protein Sougata Roy, Hai Huang,
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila
 Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila Melissa M. Rolls and Chris Q. Doe, Inst. Neurosci and Inst. Mol. Biol., HHMI, Univ. Oregon, Eugene, Oregon 97403 Correspondence
Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila Melissa M. Rolls and Chris Q. Doe, Inst. Neurosci and Inst. Mol. Biol., HHMI, Univ. Oregon, Eugene, Oregon 97403 Correspondence
Segment boundary formation in Drosophila embryos
 Segment boundary formation in Drosophila embryos Development 130, August 2003 Camilla W. Larsen, Elizabeth Hirst, Cyrille Alexandre and Jean Paul Vincent 1. Introduction: - Segment boundary formation:
Segment boundary formation in Drosophila embryos Development 130, August 2003 Camilla W. Larsen, Elizabeth Hirst, Cyrille Alexandre and Jean Paul Vincent 1. Introduction: - Segment boundary formation:
Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo
 Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo (magenta), Fas2 (blue) and GFP (green) in overview (A) and
Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo (magenta), Fas2 (blue) and GFP (green) in overview (A) and
Cell Biology Review. The key components of cells that concern us are as follows: 1. Nucleus
 Cell Biology Review Development involves the collective behavior and activities of cells, working together in a coordinated manner to construct an organism. As such, the regulation of development is intimately
Cell Biology Review Development involves the collective behavior and activities of cells, working together in a coordinated manner to construct an organism. As such, the regulation of development is intimately
Nature Neuroscience: doi: /nn.2662
 Supplementary Figure 1 Atlastin phylogeny and homology. (a) Maximum likelihood phylogenetic tree based on 18 Atlastin-1 sequences using the program Quicktree. Numbers at internal nodes correspond to bootstrap
Supplementary Figure 1 Atlastin phylogeny and homology. (a) Maximum likelihood phylogenetic tree based on 18 Atlastin-1 sequences using the program Quicktree. Numbers at internal nodes correspond to bootstrap
Cell biology: Death drags down the neighbourhood
 Cell biology: Death drags down the neighbourhood The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Vasquez,
Cell biology: Death drags down the neighbourhood The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Vasquez,
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1 Sns and Duf co-localise in embryonic nephrocytes a-c, Wild-type stage 11 (a),14 (b) and 16 (c) embryos stained with anti-duf (green) and anti-sns (red). Higher magnification images
Supplementary Figure 1 Sns and Duf co-localise in embryonic nephrocytes a-c, Wild-type stage 11 (a),14 (b) and 16 (c) embryos stained with anti-duf (green) and anti-sns (red). Higher magnification images
Dpp signaling promotes the cuboidal-to-columnar shape transition of Drosophila wing disc epithelia by regulating Rho1
 1362 Research Article Dpp signaling promotes the cuboidal-to-columnar shape transition of Drosophila wing disc epithelia by regulating Rho1 Thomas J. Widmann and Christian Dahmann* Max Planck Institute
1362 Research Article Dpp signaling promotes the cuboidal-to-columnar shape transition of Drosophila wing disc epithelia by regulating Rho1 Thomas J. Widmann and Christian Dahmann* Max Planck Institute
purpose of this Chapter is to highlight some problems that will likely provide new
 119 Chapter 6 Future Directions Besides our contributions discussed in previous chapters to the problem of developmental pattern formation, this work has also brought new questions that remain unanswered.
119 Chapter 6 Future Directions Besides our contributions discussed in previous chapters to the problem of developmental pattern formation, this work has also brought new questions that remain unanswered.
Cells to Tissues. Peter Takizawa Department of Cell Biology
 Cells to Tissues Peter Takizawa Department of Cell Biology From one cell to ensembles of cells. Multicellular organisms require individual cells to work together in functional groups. This means cells
Cells to Tissues Peter Takizawa Department of Cell Biology From one cell to ensembles of cells. Multicellular organisms require individual cells to work together in functional groups. This means cells
Neurite formation & neuronal polarization
 Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 An immature neuron in cell culture first sprouts
Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 An immature neuron in cell culture first sprouts
Axis Specification in Drosophila
 Developmental Biology Biology 4361 Axis Specification in Drosophila November 2, 2006 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Developmental Biology Biology 4361 Axis Specification in Drosophila November 2, 2006 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Neurite formation & neuronal polarization. The cytoskeletal components of neurons have characteristic distributions and associations
 Mechanisms of neuronal migration & Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 The cytoskeletal
Mechanisms of neuronal migration & Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 The cytoskeletal
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/301/ra98/dc1 Supplementary Materials for Regulation of Epithelial Morphogenesis by the G Protein Coupled Receptor Mist and Its Ligand Fog Alyssa J. Manning,
www.sciencesignaling.org/cgi/content/full/6/301/ra98/dc1 Supplementary Materials for Regulation of Epithelial Morphogenesis by the G Protein Coupled Receptor Mist and Its Ligand Fog Alyssa J. Manning,
Chapter 4 Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays.
 Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays. The data described in chapter 3 presented evidence that endogenous
Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays. The data described in chapter 3 presented evidence that endogenous
Cell-Cell Communication in Development
 Biology 4361 - Developmental Biology Cell-Cell Communication in Development October 2, 2007 Cell-Cell Communication - Topics Induction and competence Paracrine factors inducer molecules Signal transduction
Biology 4361 - Developmental Biology Cell-Cell Communication in Development October 2, 2007 Cell-Cell Communication - Topics Induction and competence Paracrine factors inducer molecules Signal transduction
10/2/2015. Chapter 4. Determination and Differentiation. Neuroanatomical Diversity
 Chapter 4 Determination and Differentiation Neuroanatomical Diversity 1 Neurochemical diversity: another important aspect of neuronal fate Neurotransmitters and their receptors Excitatory Glutamate Acetylcholine
Chapter 4 Determination and Differentiation Neuroanatomical Diversity 1 Neurochemical diversity: another important aspect of neuronal fate Neurotransmitters and their receptors Excitatory Glutamate Acetylcholine
Pattern formation: Wingless on the move Robert Howes and Sarah Bray
 R222 Dispatch Pattern formation: Wingless on the move Robert Howes and Sarah Bray Wingless is a key morphogen in Drosophila. Although it is evident that Wingless acts at a distance from its site of synthesis,
R222 Dispatch Pattern formation: Wingless on the move Robert Howes and Sarah Bray Wingless is a key morphogen in Drosophila. Although it is evident that Wingless acts at a distance from its site of synthesis,
Drosophila wing. Temporal regulation of Apterous activity during development of the. Marco Milán and Stephen M. Cohen* SUMMARY
 Development 127, 3069-3078 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV2547 3069 Temporal regulation of Apterous activity during development of the Drosophila wing Marco Milán
Development 127, 3069-3078 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV2547 3069 Temporal regulation of Apterous activity during development of the Drosophila wing Marco Milán
Lecture 3 13/11/2018
 Lecture 3 13/11/2018 1 Plasma membrane ALL cells have a cell membrane made of proteins and lipids. protein channel Cell Membrane Layer 1 Layer 2 lipid bilayer protein pump Lipid bilayer allows water, carbon
Lecture 3 13/11/2018 1 Plasma membrane ALL cells have a cell membrane made of proteins and lipids. protein channel Cell Membrane Layer 1 Layer 2 lipid bilayer protein pump Lipid bilayer allows water, carbon
Cells. Steven McLoon Department of Neuroscience University of Minnesota
 Cells Steven McLoon Department of Neuroscience University of Minnesota 1 Microscopy Methods of histology: Treat the tissue with a preservative (e.g. formaldehyde). Dissect the region of interest. Embed
Cells Steven McLoon Department of Neuroscience University of Minnesota 1 Microscopy Methods of histology: Treat the tissue with a preservative (e.g. formaldehyde). Dissect the region of interest. Embed
SUPPLEMENTARY INFORMATION
 GP2 Type I-piliated bacteria FAE M cell M cell pocket idc T cell mdc Generation of antigenspecific T cells Induction of antigen-specific mucosal immune response Supplementary Figure 1 Schematic diagram
GP2 Type I-piliated bacteria FAE M cell M cell pocket idc T cell mdc Generation of antigenspecific T cells Induction of antigen-specific mucosal immune response Supplementary Figure 1 Schematic diagram
Chapter 11. Development: Differentiation and Determination
 KAP Biology Dept Kenyon College Differential gene expression and development Mechanisms of cellular determination Induction Pattern formation Chapter 11. Development: Differentiation and Determination
KAP Biology Dept Kenyon College Differential gene expression and development Mechanisms of cellular determination Induction Pattern formation Chapter 11. Development: Differentiation and Determination
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11589 Supplementary Figure 1 Ciona intestinalis and Petromyzon marinus neural crest expression domain comparison. Cartoon shows dorsal views of Ciona mid gastrula (left) and Petromyzon
doi:10.1038/nature11589 Supplementary Figure 1 Ciona intestinalis and Petromyzon marinus neural crest expression domain comparison. Cartoon shows dorsal views of Ciona mid gastrula (left) and Petromyzon
C. elegans L1 cell adhesion molecule functions in axon guidance
 C. elegans L1 cell adhesion molecule functions in axon guidance Biorad Lihsia Chen Dept. of Genetics, Cell Biology & Development Developmental Biology Center C. elegans embryogenesis Goldstein lab, UNC-Chapel
C. elegans L1 cell adhesion molecule functions in axon guidance Biorad Lihsia Chen Dept. of Genetics, Cell Biology & Development Developmental Biology Center C. elegans embryogenesis Goldstein lab, UNC-Chapel
Chapter 6: A Tour of the Cell
 AP Biology Reading Guide Fred and Theresa Holtzclaw Chapter 6: A Tour of the Cell Name Period Chapter 6: A Tour of the Cell Concept 6.1 To study cells, biologists use microscopes and the tools of biochemistry
AP Biology Reading Guide Fred and Theresa Holtzclaw Chapter 6: A Tour of the Cell Name Period Chapter 6: A Tour of the Cell Concept 6.1 To study cells, biologists use microscopes and the tools of biochemistry
Genetic characterization of the Drosophila homologue of coronin
 Research Article 1911 Genetic characterization of the Drosophila homologue of coronin V. Bharathi, S. K. Pallavi, R. Bajpai, B. S. Emerald and L. S. Shashidhara* Centre for Cellular and Molecular Biology,
Research Article 1911 Genetic characterization of the Drosophila homologue of coronin V. Bharathi, S. K. Pallavi, R. Bajpai, B. S. Emerald and L. S. Shashidhara* Centre for Cellular and Molecular Biology,
Supplementary Figure 1. Real time in vivo imaging of SG secretion. (a) SGs from Drosophila third instar larvae that express Sgs3-GFP (green) and
 Supplementary Figure 1. Real time in vivo imaging of SG secretion. (a) SGs from Drosophila third instar larvae that express Sgs3-GFP (green) and Lifeact-Ruby (red) were imaged in vivo to visualize secretion
Supplementary Figure 1. Real time in vivo imaging of SG secretion. (a) SGs from Drosophila third instar larvae that express Sgs3-GFP (green) and Lifeact-Ruby (red) were imaged in vivo to visualize secretion
Axis Specification in Drosophila
 Developmental Biology Biology 4361 Axis Specification in Drosophila November 6, 2007 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Developmental Biology Biology 4361 Axis Specification in Drosophila November 6, 2007 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Cell Cell Communication in Development
 Biology 4361 Developmental Biology Cell Cell Communication in Development June 25, 2008 Cell Cell Communication Concepts Cells in developing organisms develop in the context of their environment, including
Biology 4361 Developmental Biology Cell Cell Communication in Development June 25, 2008 Cell Cell Communication Concepts Cells in developing organisms develop in the context of their environment, including
Model for the regulation of size in the wing imaginal disc of Drosophila.
 Model for the regulation of size in the wing imaginal disc of Drosophila. Tinri Aegerter-Wilmsen 1,2, Christof M. Aegerter 3, Ernst Hafen 1,2, Konrad Basler 4* For animal development it is necessary that
Model for the regulation of size in the wing imaginal disc of Drosophila. Tinri Aegerter-Wilmsen 1,2, Christof M. Aegerter 3, Ernst Hafen 1,2, Konrad Basler 4* For animal development it is necessary that
Massachusetts Institute of Technology Harvard Medical School Brigham and Women s Hospital VA Boston Healthcare System 2.79J/3.96J/BE.
 Massachusetts Institute of Technology Harvard Medical School Brigham and Women s Hospital VA Boston Healthcare System 2.79J/3.96J/BE.441/HST522J INTEGRINS I.V. Yannas, Ph.D. and M. Spector, Ph.D. Regulator
Massachusetts Institute of Technology Harvard Medical School Brigham and Women s Hospital VA Boston Healthcare System 2.79J/3.96J/BE.441/HST522J INTEGRINS I.V. Yannas, Ph.D. and M. Spector, Ph.D. Regulator
Supplementary Information
 Supplementary Information Supplementary Figure 1. JAK/STAT in early wing development (a-f) Wing primordia of second instar larvae of the indicated genotypes labeled to visualize expression of upd mrna
Supplementary Information Supplementary Figure 1. JAK/STAT in early wing development (a-f) Wing primordia of second instar larvae of the indicated genotypes labeled to visualize expression of upd mrna
Axis Specification in Drosophila
 Developmental Biology Biology 4361 Axis Specification in Drosophila July 9, 2008 Drosophila Development Overview Fertilization Cleavage Gastrulation Drosophila body plan Oocyte formation Genetic control
Developmental Biology Biology 4361 Axis Specification in Drosophila July 9, 2008 Drosophila Development Overview Fertilization Cleavage Gastrulation Drosophila body plan Oocyte formation Genetic control
A re-evaluation of the contributions of Apterous and Notch to the dorsoventral lineage restriction boundary in the Drosophila wing
 Development 130, 553-562 2003 The Company of Biologists Ltd doi:10.1242/dev.00276 553 A re-evaluation of the contributions of Apterous and Notch to the dorsoventral lineage restriction boundary in the
Development 130, 553-562 2003 The Company of Biologists Ltd doi:10.1242/dev.00276 553 A re-evaluation of the contributions of Apterous and Notch to the dorsoventral lineage restriction boundary in the
Developmental processes Differential gene expression Introduction to determination The model organisms used to study developmental processes
 Date Title Topic(s) Learning Outcomes: Sept 28 Oct 3 1. What is developmental biology and why should we care? 2. What is so special about stem cells and gametes? Developmental processes Differential gene
Date Title Topic(s) Learning Outcomes: Sept 28 Oct 3 1. What is developmental biology and why should we care? 2. What is so special about stem cells and gametes? Developmental processes Differential gene
Under the Radar Screen: How Bugs Trick Our Immune Defenses
 Under the Radar Screen: How Bugs Trick Our Immune Defenses Session 2: Phagocytosis Marie-Eve Paquet and Gijsbert Grotenbreg Whitehead Institute for Biomedical Research Salmonella Gram negative bacteria
Under the Radar Screen: How Bugs Trick Our Immune Defenses Session 2: Phagocytosis Marie-Eve Paquet and Gijsbert Grotenbreg Whitehead Institute for Biomedical Research Salmonella Gram negative bacteria
The neuron as a secretory cell
 The neuron as a secretory cell EXOCYTOSIS ENDOCYTOSIS The secretory pathway. Transport and sorting of proteins in the secretory pathway occur as they pass through the Golgi complex before reaching the
The neuron as a secretory cell EXOCYTOSIS ENDOCYTOSIS The secretory pathway. Transport and sorting of proteins in the secretory pathway occur as they pass through the Golgi complex before reaching the
Advanced Higher Biology. Unit 1- Cells and Proteins 2c) Membrane Proteins
 Advanced Higher Biology Unit 1- Cells and Proteins 2c) Membrane Proteins Membrane Structure Phospholipid bilayer Transmembrane protein Integral protein Movement of Molecules Across Membranes Phospholipid
Advanced Higher Biology Unit 1- Cells and Proteins 2c) Membrane Proteins Membrane Structure Phospholipid bilayer Transmembrane protein Integral protein Movement of Molecules Across Membranes Phospholipid
 DOI: 10.1038/ncb2819 Gαi3 / Actin / Acetylated Tubulin Gαi3 / Actin / Acetylated Tubulin a a Gαi3 a Actin Gαi3 WT Gαi3 WT Gαi3 WT b b Gαi3 b Actin Gαi3 KO Gαi3 KO Gαi3 KO # # Figure S1 Loss of protein
DOI: 10.1038/ncb2819 Gαi3 / Actin / Acetylated Tubulin Gαi3 / Actin / Acetylated Tubulin a a Gαi3 a Actin Gαi3 WT Gαi3 WT Gαi3 WT b b Gαi3 b Actin Gαi3 KO Gαi3 KO Gαi3 KO # # Figure S1 Loss of protein
Chapter 6: A Tour of the Cell
 Chapter 6: A Tour of the Cell 1. The study of cells has been limited by their small size, and so they were not seen and described until 1665, when Robert Hooke first looked at dead cells from an oak tree.
Chapter 6: A Tour of the Cell 1. The study of cells has been limited by their small size, and so they were not seen and described until 1665, when Robert Hooke first looked at dead cells from an oak tree.
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2647 Figure S1 Other Rab GTPases do not co-localize with the ER. a, Cos-7 cells cotransfected with an ER luminal marker (either KDEL-venus or mch-kdel) and mch-tagged human Rab5 (mch-rab5,
DOI: 10.1038/ncb2647 Figure S1 Other Rab GTPases do not co-localize with the ER. a, Cos-7 cells cotransfected with an ER luminal marker (either KDEL-venus or mch-kdel) and mch-tagged human Rab5 (mch-rab5,
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10923 Supplementary Figure 1 Ten-a and Ten-m antibody and cell type specificities. a c, Representative single confocal sections of a Drosophila NMJ stained with antibodies to Ten-a (red),
doi:10.1038/nature10923 Supplementary Figure 1 Ten-a and Ten-m antibody and cell type specificities. a c, Representative single confocal sections of a Drosophila NMJ stained with antibodies to Ten-a (red),
Drosophila Life Cycle
 Drosophila Life Cycle 1 Early Drosophila Cleavage Nuclei migrate to periphery after 10 nuclear divisions. Cellularization occurs when plasma membrane folds in to divide nuclei into cells. Drosophila Superficial
Drosophila Life Cycle 1 Early Drosophila Cleavage Nuclei migrate to periphery after 10 nuclear divisions. Cellularization occurs when plasma membrane folds in to divide nuclei into cells. Drosophila Superficial
Bio 127 Section I Introduction to Developmental Biology. Cell Cell Communication in Development. Developmental Activities Coordinated in this Way
 Bio 127 Section I Introduction to Developmental Biology Cell Cell Communication in Development Gilbert 9e Chapter 3 It has to be EXTREMELY well coordinated for the single celled fertilized ovum to develop
Bio 127 Section I Introduction to Developmental Biology Cell Cell Communication in Development Gilbert 9e Chapter 3 It has to be EXTREMELY well coordinated for the single celled fertilized ovum to develop
Regulation and signaling. Overview. Control of gene expression. Cells need to regulate the amounts of different proteins they express, depending on
 Regulation and signaling Overview Cells need to regulate the amounts of different proteins they express, depending on cell development (skin vs liver cell) cell stage environmental conditions (food, temperature,
Regulation and signaling Overview Cells need to regulate the amounts of different proteins they express, depending on cell development (skin vs liver cell) cell stage environmental conditions (food, temperature,
Neurite initiation. Neurite formation begins with a bud that sprouts from the cell body. One or several neurites can sprout at a time.
 Neurite initiation. Neuronal maturation initiation f-actin polarization and maturation tubulin stage 1: "spherical" neuron stage 2: neurons extend several neurites stage 3: one neurite accelerates its
Neurite initiation. Neuronal maturation initiation f-actin polarization and maturation tubulin stage 1: "spherical" neuron stage 2: neurons extend several neurites stage 3: one neurite accelerates its
Endocytosis of Hedgehog through Dispatched Regulates Long-Range Signaling
 Article Endocytosis of Hedgehog through Dispatched Regulates Long-Range Signaling Graphical Abstract Authors Gisela D Angelo, Tamás Matusek, Sandrine Pizette, Pascal P. Thérond Correspondence dangelo@unice.fr
Article Endocytosis of Hedgehog through Dispatched Regulates Long-Range Signaling Graphical Abstract Authors Gisela D Angelo, Tamás Matusek, Sandrine Pizette, Pascal P. Thérond Correspondence dangelo@unice.fr
Lecture 7. Development of the Fruit Fly Drosophila
 BIOLOGY 205/SECTION 7 DEVELOPMENT- LILJEGREN Lecture 7 Development of the Fruit Fly Drosophila 1. The fruit fly- a highly successful, specialized organism a. Quick life cycle includes three larval stages
BIOLOGY 205/SECTION 7 DEVELOPMENT- LILJEGREN Lecture 7 Development of the Fruit Fly Drosophila 1. The fruit fly- a highly successful, specialized organism a. Quick life cycle includes three larval stages
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2215 Figure S1 Number of egfp-vps4a bursts versus cellular expression levels. The total number of egfp-vps4a bursts, counted at the end of each movie (frame 2000, after 1h 28 min) are plotted
DOI: 10.1038/ncb2215 Figure S1 Number of egfp-vps4a bursts versus cellular expression levels. The total number of egfp-vps4a bursts, counted at the end of each movie (frame 2000, after 1h 28 min) are plotted
Reading. Lecture VI. Making Connections 9/17/12. Bio 3411 Lecture VI. Making Connections. Bio 3411 Monday September 17, 2012
 Lecture VI. Making Connections Bio 3411 Monday September 17, 2012!! 1! Reading NEUROSCIENCE: 5 th ed, pp!507?536! 4 th ed, pp 577-609 Bentley, D., & Caudy, M. (1983). Nature, 304(5921), 62-65. Dickson,
Lecture VI. Making Connections Bio 3411 Monday September 17, 2012!! 1! Reading NEUROSCIENCE: 5 th ed, pp!507?536! 4 th ed, pp 577-609 Bentley, D., & Caudy, M. (1983). Nature, 304(5921), 62-65. Dickson,
targets. clustering show that different complex pathway
 Supplementary Figure 1. CLICR allows clustering and activation of cytoplasmic protein targets. (a, b) Upon light activation, the Cry2 (red) and LRP6c (green) components co-cluster due to the heterodimeric
Supplementary Figure 1. CLICR allows clustering and activation of cytoplasmic protein targets. (a, b) Upon light activation, the Cry2 (red) and LRP6c (green) components co-cluster due to the heterodimeric
Figure 1. Automated 4D detection and tracking of endosomes and polarity markers.
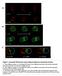 A t = 0 z t = 90 B t = 0 z t = 90 C t = 0-90 t = 0-180 t = 0-300 Figure 1. Automated 4D detection and tracking of endosomes and polarity markers. A: Three different z planes of a dividing SOP shown at
A t = 0 z t = 90 B t = 0 z t = 90 C t = 0-90 t = 0-180 t = 0-300 Figure 1. Automated 4D detection and tracking of endosomes and polarity markers. A: Three different z planes of a dividing SOP shown at
Chapter 3: Cells. Lectures by Mark Manteuffel, St. Louis Community College
 Chapter 3: Cells Lectures by Mark Manteuffel, St. Louis Community College Learning Objectives Be able to describe: what a cell is & two main classes of cells. structure & functions of cell membranes. how
Chapter 3: Cells Lectures by Mark Manteuffel, St. Louis Community College Learning Objectives Be able to describe: what a cell is & two main classes of cells. structure & functions of cell membranes. how
Supporting Information
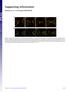 Supporting Information Fleissner et al. 10.1073/pnas.0907039106 Fig. S1. (A) MAK-2-GFP localized to CATs tips is not bound by membrane. his-3::pccg1 mak-2-gfp; mak-2 strain labeled with membrane dye FM4
Supporting Information Fleissner et al. 10.1073/pnas.0907039106 Fig. S1. (A) MAK-2-GFP localized to CATs tips is not bound by membrane. his-3::pccg1 mak-2-gfp; mak-2 strain labeled with membrane dye FM4
Different cell types exhibit different degrees of complexity in
 Distinct roles of Bazooka and Stardust in the specification of Drosophila photoreceptor membrane architecture Yang Hong, Larry Ackerman, Lily Yeh Jan, and Yuh-Nung Jan* Howard Hughes Medical Institute
Distinct roles of Bazooka and Stardust in the specification of Drosophila photoreceptor membrane architecture Yang Hong, Larry Ackerman, Lily Yeh Jan, and Yuh-Nung Jan* Howard Hughes Medical Institute
Zimmerman AP Biology CBHS South Name Chapter 7&8 Guided Reading Assignment 1) What is resolving power and why is it important in biology?
 Zimmerman AP Biology CBHS South Name Chapter 7&8 Guided Reading Assignment 1) What is resolving power and why is it important in biology? 2) How does an electron microscope work and what is the difference
Zimmerman AP Biology CBHS South Name Chapter 7&8 Guided Reading Assignment 1) What is resolving power and why is it important in biology? 2) How does an electron microscope work and what is the difference
Signal Transduction. Dr. Chaidir, Apt
 Signal Transduction Dr. Chaidir, Apt Background Complex unicellular organisms existed on Earth for approximately 2.5 billion years before the first multicellular organisms appeared.this long period for
Signal Transduction Dr. Chaidir, Apt Background Complex unicellular organisms existed on Earth for approximately 2.5 billion years before the first multicellular organisms appeared.this long period for
Chemical aspects of the cell. Shape and structure of the cell
 Chemical aspects of the cell Shape and structure of the cell Cellular composition https://www.studyblue.com/ 2 Cellular composition Set of videos with basic information: Cell characteristics: https://www.youtube.com/watch?v=urujd5nexc8
Chemical aspects of the cell Shape and structure of the cell Cellular composition https://www.studyblue.com/ 2 Cellular composition Set of videos with basic information: Cell characteristics: https://www.youtube.com/watch?v=urujd5nexc8
Molecular Cell Biology 5068 In Class Exam 1 September 30, Please print your name:
 Molecular Cell Biology 5068 In Class Exam 1 September 30, 2014 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
Molecular Cell Biology 5068 In Class Exam 1 September 30, 2014 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
Conclusions. The experimental studies presented in this thesis provide the first molecular insights
 C h a p t e r 5 Conclusions 5.1 Summary The experimental studies presented in this thesis provide the first molecular insights into the cellular processes of assembly, and aggregation of neural crest and
C h a p t e r 5 Conclusions 5.1 Summary The experimental studies presented in this thesis provide the first molecular insights into the cellular processes of assembly, and aggregation of neural crest and
Cell-Cell Communication in Development
 Biology 4361 - Developmental Biology Cell-Cell Communication in Development June 23, 2009 Concepts Cell-Cell Communication Cells develop in the context of their environment, including: - their immediate
Biology 4361 - Developmental Biology Cell-Cell Communication in Development June 23, 2009 Concepts Cell-Cell Communication Cells develop in the context of their environment, including: - their immediate
Establishing positional information through gradient dynamics
 Extra view Fly 4:4, 273-277; October/November/December 2010; 2010 Landes Bioscience Extra view Establishing positional information through gradient dynamics A lesson from the Hedgehog signaling pathway
Extra view Fly 4:4, 273-277; October/November/December 2010; 2010 Landes Bioscience Extra view Establishing positional information through gradient dynamics A lesson from the Hedgehog signaling pathway
Epithelial Polarity. Gerard Apodaca Luciana I. Gallo. Colloquium series on Building BloCks of the Cell: Cell structure and function
 Colloquium series on Building BloCks of the Cell: Cell structure and function Series Editor: Ivan Robert Nabi Epithelial Polarity Gerard Apodaca Luciana I. Gallo life sciences Morgan & Claypool life SCIEnCES
Colloquium series on Building BloCks of the Cell: Cell structure and function Series Editor: Ivan Robert Nabi Epithelial Polarity Gerard Apodaca Luciana I. Gallo life sciences Morgan & Claypool life SCIEnCES
Neural development its all connected
 Neural development its all connected How do you build a complex nervous system? How do you build a complex nervous system? 1. Learn how tissue is instructed to become nervous system. Neural induction 2.
Neural development its all connected How do you build a complex nervous system? How do you build a complex nervous system? 1. Learn how tissue is instructed to become nervous system. Neural induction 2.
Chapter 4 Active Reading Guide A Tour of the Cell
 Name: AP Biology Mr. Croft Chapter 4 Active Reading Guide A Tour of the Cell Section 1 1. The study of cells has been limited by their small size, and so they were not seen and described until 1665, when
Name: AP Biology Mr. Croft Chapter 4 Active Reading Guide A Tour of the Cell Section 1 1. The study of cells has been limited by their small size, and so they were not seen and described until 1665, when
Compartments and the control of growth in the Drosophila wing imaginal disc
 Access Development the most First recent posted version epress online at online on http://dev.biologists.org/lookup/doi/10.1242/dev.02618 11 October publication 2006 as date 10.1242/dev.02618 11 October
Access Development the most First recent posted version epress online at online on http://dev.biologists.org/lookup/doi/10.1242/dev.02618 11 October publication 2006 as date 10.1242/dev.02618 11 October
Article. Physical Mechanisms Shaping the Drosophila Dorsoventral Compartment Boundary
 Current Biology, 967 976, June 5, ª Elsevier Ltd All rights reserved DOI.6/j.cub...7 Physical Mechanisms Shaping the Drosophila Dorsoventral Compartment Boundary Article Maryam Aliee,, Jens-Christian Röper,,,
Current Biology, 967 976, June 5, ª Elsevier Ltd All rights reserved DOI.6/j.cub...7 Physical Mechanisms Shaping the Drosophila Dorsoventral Compartment Boundary Article Maryam Aliee,, Jens-Christian Röper,,,
Development of Drosophila
 Development of Drosophila Hand-out CBT Chapter 2 Wolpert, 5 th edition March 2018 Introduction 6. Introduction Drosophila melanogaster, the fruit fly, is found in all warm countries. In cooler regions,
Development of Drosophila Hand-out CBT Chapter 2 Wolpert, 5 th edition March 2018 Introduction 6. Introduction Drosophila melanogaster, the fruit fly, is found in all warm countries. In cooler regions,
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
Developmental Biology Lecture Outlines
 Developmental Biology Lecture Outlines Lecture 01: Introduction Course content Developmental Biology Obsolete hypotheses Current theory Lecture 02: Gametogenesis Spermatozoa Spermatozoon function Spermatozoon
Developmental Biology Lecture Outlines Lecture 01: Introduction Course content Developmental Biology Obsolete hypotheses Current theory Lecture 02: Gametogenesis Spermatozoa Spermatozoon function Spermatozoon
Concept 6.1 To study cells, biologists use microscopes and the tools of biochemistry
 Name Period Chapter 6: A Tour of the Cell Concept 6.1 To study cells, biologists use microscopes and the tools of biochemistry 1. The study of cells has been limited by their small size, and so they were
Name Period Chapter 6: A Tour of the Cell Concept 6.1 To study cells, biologists use microscopes and the tools of biochemistry 1. The study of cells has been limited by their small size, and so they were
Amneh Auben. Abdulrahman Jabr. Diala Abu-Hassan
 21 Amneh Auben Abdulrahman Jabr Diala Abu-Hassan Matrix polysaccharides Extracellular matrix (ECM): It s a collection of components that fills the spaces outside the cell or between the cells. ---------
21 Amneh Auben Abdulrahman Jabr Diala Abu-Hassan Matrix polysaccharides Extracellular matrix (ECM): It s a collection of components that fills the spaces outside the cell or between the cells. ---------
Introduction to cells
 Almen Cellebiologi Introduction to cells 1. Unity and diversity of cells 2. Microscopes and visualization of cells 3. Prokaryotic cells, eubacteria and archaea 4. Eucaryotic cells, nucleus, mitochondria
Almen Cellebiologi Introduction to cells 1. Unity and diversity of cells 2. Microscopes and visualization of cells 3. Prokaryotic cells, eubacteria and archaea 4. Eucaryotic cells, nucleus, mitochondria
Supplemental table S7.
 Supplemental table S7. GO terms significantly enriched in significantly up-regulated genes of the microarray. K: number of genes from the input cluster in the given category. F: number of total genes in
Supplemental table S7. GO terms significantly enriched in significantly up-regulated genes of the microarray. K: number of genes from the input cluster in the given category. F: number of total genes in
MOLECULAR CONTROL OF EMBRYONIC PATTERN FORMATION
 MOLECULAR CONTROL OF EMBRYONIC PATTERN FORMATION Drosophila is the best understood of all developmental systems, especially at the genetic level, and although it is an invertebrate it has had an enormous
MOLECULAR CONTROL OF EMBRYONIC PATTERN FORMATION Drosophila is the best understood of all developmental systems, especially at the genetic level, and although it is an invertebrate it has had an enormous
Axon Guidance. Multiple decision points along a growing axon s trajectory Different types of axon guidance cues:
 Axon Guidance Multiple decision points along a growing axon s trajectory Different types of axon guidance cues: Contact mediated - requires direct contact by growth cone Long range - growth cone responds
Axon Guidance Multiple decision points along a growing axon s trajectory Different types of axon guidance cues: Contact mediated - requires direct contact by growth cone Long range - growth cone responds
Enduring understanding 1.A: Change in the genetic makeup of a population over time is evolution.
 The AP Biology course is designed to enable you to develop advanced inquiry and reasoning skills, such as designing a plan for collecting data, analyzing data, applying mathematical routines, and connecting
The AP Biology course is designed to enable you to develop advanced inquiry and reasoning skills, such as designing a plan for collecting data, analyzing data, applying mathematical routines, and connecting
CELB40060 Membrane Trafficking in Animal Cells. Prof. Jeremy C. Simpson. Lecture 2 COPII and export from the ER
 CELB40060 Membrane Trafficking in Animal Cells Prof. Jeremy C. Simpson Lecture 2 COPII and export from the ER Today s lecture... The COPII coat - localisation and subunits Formation of the COPII coat at
CELB40060 Membrane Trafficking in Animal Cells Prof. Jeremy C. Simpson Lecture 2 COPII and export from the ER Today s lecture... The COPII coat - localisation and subunits Formation of the COPII coat at
Shavenbaby Couples Patterning to Epidermal Cell Shape Control. Chanut-Delalande H, Fernandes I, Roch F, Payre F, Plaza S (2006) PLoS Biol 4(9): e290
 Shavenbaby Couples Patterning to Epidermal Cell Shape Control. Chanut-Delalande H, Fernandes I, Roch F, Payre F, Plaza S (2006) PLoS Biol 4(9): e290 Question (from Introduction): How does svb control the
Shavenbaby Couples Patterning to Epidermal Cell Shape Control. Chanut-Delalande H, Fernandes I, Roch F, Payre F, Plaza S (2006) PLoS Biol 4(9): e290 Question (from Introduction): How does svb control the
CHAPTER 3. Cell Structure and Genetic Control. Chapter 3 Outline
 CHAPTER 3 Cell Structure and Genetic Control Chapter 3 Outline Plasma Membrane Cytoplasm and Its Organelles Cell Nucleus and Gene Expression Protein Synthesis and Secretion DNA Synthesis and Cell Division
CHAPTER 3 Cell Structure and Genetic Control Chapter 3 Outline Plasma Membrane Cytoplasm and Its Organelles Cell Nucleus and Gene Expression Protein Synthesis and Secretion DNA Synthesis and Cell Division
The function of engrailed and the specification of Drosophila wing pattern
 Development 121, 3447-3456 (1995) Printed in Great Britain The Company of Biologists Limited 1995 3447 The function of engrailed and the specification of Drosophila wing pattern Isabel Guillén 1, *, Jose
Development 121, 3447-3456 (1995) Printed in Great Britain The Company of Biologists Limited 1995 3447 The function of engrailed and the specification of Drosophila wing pattern Isabel Guillén 1, *, Jose
Chapter 18 Lecture. Concepts of Genetics. Tenth Edition. Developmental Genetics
 Chapter 18 Lecture Concepts of Genetics Tenth Edition Developmental Genetics Chapter Contents 18.1 Differentiated States Develop from Coordinated Programs of Gene Expression 18.2 Evolutionary Conservation
Chapter 18 Lecture Concepts of Genetics Tenth Edition Developmental Genetics Chapter Contents 18.1 Differentiated States Develop from Coordinated Programs of Gene Expression 18.2 Evolutionary Conservation
mirror controls planar polarity and equator formation through repression of fringe expression and through control of cell affinities
 Development 126, 5857-5866 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV5359 5857 mirror controls planar polarity and equator formation through repression of fringe expression
Development 126, 5857-5866 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV5359 5857 mirror controls planar polarity and equator formation through repression of fringe expression
The majority of cells in the nervous system arise during the embryonic and early post
 Introduction Introduction The majority of cells in the nervous system arise during the embryonic and early post natal period. These cells are derived from population of neural stem cells first shown by
Introduction Introduction The majority of cells in the nervous system arise during the embryonic and early post natal period. These cells are derived from population of neural stem cells first shown by
Introduction to Cellular Communication *
 OpenStax-CNX module: m53235 1 Introduction to Cellular Communication * Steven Telleen This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0 1 Why Cells Communicate
OpenStax-CNX module: m53235 1 Introduction to Cellular Communication * Steven Telleen This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0 1 Why Cells Communicate
Chapter 7: Membrane Structure and Function
 Chapter 7: Membrane Structure and Function 7.1 Cellular membranes are fluid mosaics of lipids and proteins 1. The large molecules of all living things fall into just four main classes. Name them. 2. Explain
Chapter 7: Membrane Structure and Function 7.1 Cellular membranes are fluid mosaics of lipids and proteins 1. The large molecules of all living things fall into just four main classes. Name them. 2. Explain
Cellular Neuroanatomy II The Prototypical Neuron: Neurites. Reading: BCP Chapter 2
 Cellular Neuroanatomy II The Prototypical Neuron: Neurites Reading: BCP Chapter 2 Major Internal Features of a Neuron The neuron is the functional unit of the nervous system. A typical neuron has a soma
Cellular Neuroanatomy II The Prototypical Neuron: Neurites Reading: BCP Chapter 2 Major Internal Features of a Neuron The neuron is the functional unit of the nervous system. A typical neuron has a soma
CELL BIOLOGY. Which of the following cell structures does not have membranes? A. Ribosomes B. Mitochondria C. Chloroplasts D.
 1 CELL BIOLOGY PROKARYOTIC and EUKARYOTIC SP/1. SP/2. SP/4. Plant and animal cells both have A. ribosomes, cell walls and mitochondria. B. Golgi apparatus, chromosomes and mitochondria. C. Golgi apparatus,
1 CELL BIOLOGY PROKARYOTIC and EUKARYOTIC SP/1. SP/2. SP/4. Plant and animal cells both have A. ribosomes, cell walls and mitochondria. B. Golgi apparatus, chromosomes and mitochondria. C. Golgi apparatus,
Exam 1 ID#: October 4, 2007
 Biology 4361 Name: KEY Exam 1 ID#: October 4, 2007 Multiple choice (one point each) (1-25) 1. The process of cells forming tissues and organs is called a. morphogenesis. b. differentiation. c. allometry.
Biology 4361 Name: KEY Exam 1 ID#: October 4, 2007 Multiple choice (one point each) (1-25) 1. The process of cells forming tissues and organs is called a. morphogenesis. b. differentiation. c. allometry.
A A A A B B1
 LEARNING OBJECTIVES FOR EACH BIG IDEA WITH ASSOCIATED SCIENCE PRACTICES AND ESSENTIAL KNOWLEDGE Learning Objectives will be the target for AP Biology exam questions Learning Objectives Sci Prac Es Knowl
LEARNING OBJECTIVES FOR EACH BIG IDEA WITH ASSOCIATED SCIENCE PRACTICES AND ESSENTIAL KNOWLEDGE Learning Objectives will be the target for AP Biology exam questions Learning Objectives Sci Prac Es Knowl
1. What are the three general areas of the developing vertebrate limb? 2. What embryonic regions contribute to the developing limb bud?
 Study Questions - Lecture 17 & 18 1. What are the three general areas of the developing vertebrate limb? The three general areas of the developing vertebrate limb are the proximal stylopod, zeugopod, and
Study Questions - Lecture 17 & 18 1. What are the three general areas of the developing vertebrate limb? The three general areas of the developing vertebrate limb are the proximal stylopod, zeugopod, and
Cellular Neuroanatomy I The Prototypical Neuron: Soma. Reading: BCP Chapter 2
 Cellular Neuroanatomy I The Prototypical Neuron: Soma Reading: BCP Chapter 2 Functional Unit of the Nervous System The functional unit of the nervous system is the neuron. Neurons are cells specialized
Cellular Neuroanatomy I The Prototypical Neuron: Soma Reading: BCP Chapter 2 Functional Unit of the Nervous System The functional unit of the nervous system is the neuron. Neurons are cells specialized
Principles of Experimental Embryology
 Biology 4361 Developmental Biology Principles of Experimental Embryology September 19, 2006 Major Research Questions How do forces outside the embryo affect its development? (Environmental Developmental
Biology 4361 Developmental Biology Principles of Experimental Embryology September 19, 2006 Major Research Questions How do forces outside the embryo affect its development? (Environmental Developmental
BE 159: Signal Transduction and Mechanics in Morphogenesis
 BE 159: Signal Transduction and Mechanics in Morphogenesis Justin Bois Caltech Winter, 2018 2018 Justin Bois. This work is licensed under a Creative Commons Attribution License CC-BY 4.0. 5 Delta-Notch
BE 159: Signal Transduction and Mechanics in Morphogenesis Justin Bois Caltech Winter, 2018 2018 Justin Bois. This work is licensed under a Creative Commons Attribution License CC-BY 4.0. 5 Delta-Notch
