Anaesthesia Gas Monitoring Štefan Trenkler
|
|
|
- Ambrose Banks
- 6 years ago
- Views:
Transcription
1 Anaesthesia Gas Monitoring Štefan Trenkler Department of Anesthesiology and Intensive Medicine P.J.Šafárik University Medical faculty Košice Košice CEEA 2015
2 Why anaesthesia gas monitoring The ability to rapidly measure concentrations of inspired and expired gas is of paramount importance in anesthetic practice. Oxygen CO 2 N 2 O Inhalational anaesthetics New technologies: low size and weight, cost Present in the most of modern op. rooms
3 Alveolar vs brain concentration Depth of anaesthesia? Brain concentration of inhalation agent?? Rapid equilibration alveolus blood brain Alveolar concentration end expiratory exhaled gas Let us measure it
4 Minimum Alveolar Concentration Alveolar (or end-expiratory) concentration at which 50% of patients will not show a motor response to a standardized surgical incision. The standard deviation of MAC is ~10%, thus 95% of patients will not respond to 1.2 MAC, and 99% will not respond to 1.3 MAC MAC-awake - the value necessary to prevent voluntary response to command. MAC-BAR - blocks autonomic response (BAR) to surgical stimulation. MAC-intubation - value that prevents response to ETI. It is about 1.3 MAC.
5 Monitoring of gases Patient safety first! Information about concentration, reached MAC, overdose, wrong agent... Gas monitoring: Standard?? ASA closed Claims Database: gas delivery equipment 2%; 21% vaporisers; mostly overdose and awareness. Most injuries preventable by using monitors.
6 Why basics of technology As anaesthetists we must be aware of: - basic physical principles involved in these monitors - their limitations - advantages - disadvantages.
7 1. Oxygen analysis Goal: to detect - hypoxia - hyperoxia - disconnection Usually placed on inspiratory arm In the past: - Mass spectroscopy - Raman spectroscopy Today two physical methods: 1. Paramagnetic analysis (Pauling sensor) 2. Electrochemical analysis fuell / galvanic cell; polarographic
8 A. Paramagnetic analyzers O 2 is a paramagnetic gas 2 unpaired electrons on outer orbit It is attracted by the rapidly changing magnetic field. O 2 and reference gas are pumped into an electromagnetic field. Between gases there is a sensitive pressure sensor. When O 2 molecules realign themselves to the magnetic field a pressure wave is generated and sensed by the sensor. This is then converted to an electrical signal proportional to the O 2 partial pressure. Displayed as Vol%.
9 Paramagnetic O 2 analyser
10 B. Electro chemical analyzers Electro chemical analyzers have two parts: - sensor containing the cathode and an anode enclosed in a membrane permeable to gases - an analyzer box with electronic circuitry, display monitor and alarm. Two physical principles are used in electrochemical analysis. 1. Galvanic / fuell cell 2. Polarographic electrode
11 Electro chemical analyzers I 1. Galvanic cell / Fuel cell The sensing electrode is a silver cathode where oxygen molecule is reduced to a hydroxide ion (O 2 OH - ) due to a chemical reaction. The hydroxide ion then reacts with a lead anode giving up electrons (battery generating a voltage proportional to the oxygen fraction). 2OH - + Pb PbO + H 2 O + 2e - The electronic current is sensed by a meter and is proportional to the partial pressure of oxygen. These require regular replacement of galvanic sensor capsule. Lifetime: depends on concentration and time exposition to oxygen
12 Fuel cell analyser
13 Electro chemical analyzers II 2. Polarographic electrode (Clark) Oxygen diffuses via a membrane and electrolytes to the cathode and a similar reaction as above takes place. The current change is proportional to the number of oxygen molecules surrounding the electrode. Main difference from fuel cell is that a polarizing voltage is applied by an external source.
14 Clark s electrode
15 Oxygen polarographic electrode Langton 2009
16
17 Anaesthesia gas analysis
18 Sensors for monitoring of gases
19 Sensors - main stream Advantages No sampling tube, no obstruction No delay in recording Suitable for neonates and children Disanvantages Weight of senser - traction on the endotracheal tube. - the newer sensors are light weight Facial burns. - eliminated with newer sensors No measurement of other gases Sensor windows may clog with secretions. - they can be replaced easily as they are disposable. Difficult to use in unusual patient positioning such as in prone positions. Need of sterilization problem. - the newer versions use disposable sensor windows Currently only CO 2 can be measured by mainstream monitors
20 Sensors - side stream Advantages Easy to connect, no weight No problems with sterilization Can be used in awake, non intubated patients Simultaneously measurement of more gases Easy to use when patient is in unusual positions (prone ) In collaboration with simultaneous oxygen administration via a nasal prong Disadvantages Return of gases in anaesthesia ( cc) Delay in recording due to movement of gases from the ET to the unit Sampling tube obstruction (water, secretion) Calibration needed
21 Anaesthesia gas analysers Size Side / main stream Breath to breath analysis Response time Accuracy One / multiple gas; specificity Water sensitivity Calibration
22 Gaseous analysis CO 2, NO 2, volatile anaesthetics 1. Mass spectrometry 2. Roman scattering 3. Refractometry 4. Piezoelectric absorption 5. Infrared absorption spectroscopy
23 Gaseous analysis CO 2, NO 2, volatile anaesthetics 1. Mass spectrometry 2. Roman scattering 3. Refractometry 4. Piezoelectric absorption 5. Infrared absorption spectroscopy
24 1. Mass spectrometry 1960 NASA programme 1970 centrally located for more theatres; later smaller size, in operating theatres 1981: Ohmeda 6000 stand alone, continuous Identification of gases according the molecule mass Side stream, vacuum chamber, ion gun, molecule fragments, trajectory bending, detection electrodes Calibration for every molecule 1980 new multigas analysers, different technologies
25 Mass spectrometry
26 Mass spectrometry
27 Monitorovanie plynov Hmotnostný spektrometer 1963
28 Mass spectrometry Advantages: - rapid response - wide range of gases (helium, nitrogen) Disadvantages: - bulk, expensive - discontinuous
29 Raman scattering Intense, coherent, monochromatic light (laser) Gas molecul will scatter the light, some will be absorbed Narrowband filters, photodetector and signal processing unit. Fast response time, multiple agents, accurate (better than IR)
30 2. Raman spectroscopy
31 3. Refractometry The refractive number of gas must be known. A light beam is split; one portion passes through a chamber containing sample gas, second passes via an identical chamber. Beams are recombined and the difference in refraction is compared to a scale. Not for breath to breath analysis. Mainly used for calibration of vaporizers.
32 4. Pizoelectric crystal adsorption A quartz crystal with surface coated with lipids vibrates at a specific frequency when stimulated with an electrical voltage. When this crystal is exposed to an inhalational agent it absorbs it and its mass changes resulting in a decrease in vibrating frequency. An electrical signal is generated which is proportional to the concentration of agent. The accuracy is 0.1% but the method is sensitive to water vapour. It is compact, fast and is used for breath by breath analysis. Only one agent can be measured and the device cannot discriminate between agents.
33 5. Infrared spectroscopy
34 Absorption bands of respiratory gases in the infrared spectrum Carbon dioxide, nitrous oxide, and anesthetic gases exhibit absorption of radiation at unique bands in the IR spectrum. CO 2 between 4.2 and 4.4 μm N 2 O between 4.4 and 4.6 μm and less strongly at 3.9 μm The volatile anesthetic at 3.3 μm and 8.0 to 13 μm
35 IR spectrometry The shorter IR spectrum range 0.76 μm - 13 μm. Measurement of the energy absorbed from a narrow band of wavelengths of IR radiation as it passes through a gas sample can be used to measure the concentrations of certain gases. Asymmetric, polyatomic, polar molecules - such as carbon dioxide, nitrous oxide, water, and the potent volatile anesthetic agents - absorb IR energy when their atoms rotate or vibrate asymmetrically; this results in a change in dipole moment, the charge distribution within the molecule. The nonpolar molecules argon, nitrogen, helium, xenon, and oxygen do not absorb IR energy. Detector: photocell, thermopiles
36 IR analysers A. Dispersive: - single optical filter, prism or diffraction to separate the component wavelenghts for each agent B. Non dispersive: - multiple narrow-band optical filters through the IR emission is passed Mostly used
37 Dispersive analyzer After passing through the gas sample, the radiation emitted by an IR source is separated or dispersed, into the component wavelengths and is arranged sequentially. By examining the entire spectrum, a plot of absorbance versus wavelength is obtained, from which the gas composition can be analyzed and quantified, provided the gases in the mixture have characteristic absorption peaks.
38 Nondispersive analyzer Radiation from the IR source is filtered to allow passage of only the specific wavelength bands, for which the gases of interest have distinct absorption peaks. The gas sample is placed between the filter and the IR detector or between the IR source and the filter IR analyzers used clinically are predominantly of the nondispersive type.
39 A simple nondispersive IR analyzer A source of IR radiation. Heating the black body causes emission of IR radiation. A sample cell, or cuvette. The gas to be analyzed is drawn through the cuvette by a sampling pump. A detector that generates an output signal. The signal is related to the intensity of the IR radiation that falls on it. A narrow-band pass filter. This filter allows only radiation at the wavelength bands of interest to pass through It is interposed between the IR source and the cuvette or between the cuvette and the detector. The intensity of radiation reaching the detector is inversely related to the concentration of the specific gas being measured.
40 Nondispersive analyser
41 Nondispersive analyser
42 Absorbance spectra
43 An infrared analyzer with multiple filters on a spinning chopper
44 Infrared Photoacoustic Spectrometer Each gas absorbs the pulsating IR energy in its absorption band, it expands and contracts at that frequency, and resulting sound waves are detected with a simple microphone.
45 Phasain IRMA mainstream probe Miniature, main stream 10 different wavelength Short answer time
46 Phasain EMMA CO 2 probe Capnometry Breathing frequency Alarms In any situation
47 Microstream. Neintubovaní pacienti
48 Comparison of analysers
49 Calibration of analysers
50 Thank you for your attention
Sub-category: Physics and Principles of Measurement Topic: Monitoring anesthetic gases and vapours Date: January 15-17, 2016 Language: English
 Course n : Course 3 Title: RESPIRATORY PHYSIOLOGY, PHYSICS AND PATHOLOGY IN RELATION TO ANAESTHESIA AND INTENSIVE CARE Sub-category: Physics and Principles of Measurement Topic: Monitoring anesthetic gases
Course n : Course 3 Title: RESPIRATORY PHYSIOLOGY, PHYSICS AND PATHOLOGY IN RELATION TO ANAESTHESIA AND INTENSIVE CARE Sub-category: Physics and Principles of Measurement Topic: Monitoring anesthetic gases
Physics of capnography
 Strona 1 z 7 Physics of capnography Bhavani Shankar Kodali MD There are five Physical methods 1. Infra-red Spectrography 2. Molecular Correlation Spectrography 3. Raman Spectrography 4. Mass Spectrography
Strona 1 z 7 Physics of capnography Bhavani Shankar Kodali MD There are five Physical methods 1. Infra-red Spectrography 2. Molecular Correlation Spectrography 3. Raman Spectrography 4. Mass Spectrography
Respiratory gas analysis
 J. A. Langton MBBS MD FRCA A. Hutton MBBS MRCP FRCA Respiratory gas analysis has now become a standard monitoring technique in anaesthesia: in theatres, intensive care unit, and for the transfer of ventilated
J. A. Langton MBBS MD FRCA A. Hutton MBBS MRCP FRCA Respiratory gas analysis has now become a standard monitoring technique in anaesthesia: in theatres, intensive care unit, and for the transfer of ventilated
Atomization. In Flame Emission
 FLAME SPECTROSCOPY The concentration of an element in a solution is determined by measuring the absorption, emission or fluorescence of electromagnetic by its monatomic particles in gaseous state in the
FLAME SPECTROSCOPY The concentration of an element in a solution is determined by measuring the absorption, emission or fluorescence of electromagnetic by its monatomic particles in gaseous state in the
LASERS. Dr D. Arun Kumar Assistant Professor Department of Physical Sciences Bannari Amman Institute of Technology Sathyamangalam
 LASERS Dr D. Arun Kumar Assistant Professor Department of Physical Sciences Bannari Amman Institute of Technology Sathyamangalam General Objective To understand the principle, characteristics and types
LASERS Dr D. Arun Kumar Assistant Professor Department of Physical Sciences Bannari Amman Institute of Technology Sathyamangalam General Objective To understand the principle, characteristics and types
 DEPARTMENT OF ELECTRONICS AND INSTRUMENTATION ENGINEERING SRM NAGAR, KATTANKULATHUR-603203 EI 2302 ANALYTICAL INSTRUMENTS QUESTION BANK UNIT I COLORIMETRY AND SPECTROPHOTOMETRY Part A 1. State Lambert
DEPARTMENT OF ELECTRONICS AND INSTRUMENTATION ENGINEERING SRM NAGAR, KATTANKULATHUR-603203 EI 2302 ANALYTICAL INSTRUMENTS QUESTION BANK UNIT I COLORIMETRY AND SPECTROPHOTOMETRY Part A 1. State Lambert
White Paper. Overview: NDIR Definition:
 Title: NDIR Technology Overview, Compliance, and Comparison to Other Generally Available Gas Measurement Technologies TSN Number: 06 File:\\MII- SRV1\Metron\Bridge_Analyzers\Customer_Service_Documentation\White_Papers\06
Title: NDIR Technology Overview, Compliance, and Comparison to Other Generally Available Gas Measurement Technologies TSN Number: 06 File:\\MII- SRV1\Metron\Bridge_Analyzers\Customer_Service_Documentation\White_Papers\06
Thermo Scientific Electrochemistry Products
 Comparing Performance of DO Methods The world leader in serving science in Water and Seawaters Thermo Scientific Electrochemistry Products Dissolved Oxygen Measurement Oxygen is essential for most of the
Comparing Performance of DO Methods The world leader in serving science in Water and Seawaters Thermo Scientific Electrochemistry Products Dissolved Oxygen Measurement Oxygen is essential for most of the
Chemistry 311: Instrumentation Analysis Topic 2: Atomic Spectroscopy. Chemistry 311: Instrumentation Analysis Topic 2: Atomic Spectroscopy
 Topic 2b: X-ray Fluorescence Spectrometry Text: Chapter 12 Rouessac (1 week) 4.0 X-ray Fluorescence Download, read and understand EPA method 6010C ICP-OES Winter 2009 Page 1 Atomic X-ray Spectrometry Fundamental
Topic 2b: X-ray Fluorescence Spectrometry Text: Chapter 12 Rouessac (1 week) 4.0 X-ray Fluorescence Download, read and understand EPA method 6010C ICP-OES Winter 2009 Page 1 Atomic X-ray Spectrometry Fundamental
Reference literature. (See: CHEM 2470 notes, Module 8 Textbook 6th ed., Chapters )
 September 17, 2018 Reference literature (See: CHEM 2470 notes, Module 8 Textbook 6th ed., Chapters 13-14 ) Reference.: https://slideplayer.com/slide/8354408/ Spectroscopy Usual Wavelength Type of Quantum
September 17, 2018 Reference literature (See: CHEM 2470 notes, Module 8 Textbook 6th ed., Chapters 13-14 ) Reference.: https://slideplayer.com/slide/8354408/ Spectroscopy Usual Wavelength Type of Quantum
Secondary Ion Mass Spectrometry (SIMS)
 CHEM53200: Lecture 10 Secondary Ion Mass Spectrometry (SIMS) Major reference: Surface Analysis Edited by J. C. Vickerman (1997). 1 Primary particles may be: Secondary particles can be e s, neutral species
CHEM53200: Lecture 10 Secondary Ion Mass Spectrometry (SIMS) Major reference: Surface Analysis Edited by J. C. Vickerman (1997). 1 Primary particles may be: Secondary particles can be e s, neutral species
Atomic Absorption Spectrophotometry. Presentation by, Mrs. Sangita J. Chandratre Department of Microbiology M. J. college, Jalgaon
 Atomic Absorption Spectrophotometry Presentation by, Mrs. Sangita J. Chandratre Department of Microbiology M. J. college, Jalgaon Defination In analytical chemistry, Atomic absorption spectroscopy is a
Atomic Absorption Spectrophotometry Presentation by, Mrs. Sangita J. Chandratre Department of Microbiology M. J. college, Jalgaon Defination In analytical chemistry, Atomic absorption spectroscopy is a
Skoog Chapter 6 Introduction to Spectrometric Methods
 Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
Questions on Instrumental Methods of Analysis
 Questions on Instrumental Methods of Analysis 1. Which one of the following techniques can be used for the detection in a liquid chromatograph? a. Ultraviolet absorbance or refractive index measurement.
Questions on Instrumental Methods of Analysis 1. Which one of the following techniques can be used for the detection in a liquid chromatograph? a. Ultraviolet absorbance or refractive index measurement.
R O Y G B V. Spin States. Outer Shell Electrons. Molecular Rotations. Inner Shell Electrons. Molecular Vibrations. Nuclear Transitions
 Spin States Molecular Rotations Molecular Vibrations Outer Shell Electrons Inner Shell Electrons Nuclear Transitions NMR EPR Microwave Absorption Spectroscopy Infrared Absorption Spectroscopy UV-vis Absorption,
Spin States Molecular Rotations Molecular Vibrations Outer Shell Electrons Inner Shell Electrons Nuclear Transitions NMR EPR Microwave Absorption Spectroscopy Infrared Absorption Spectroscopy UV-vis Absorption,
high temp ( K) Chapter 20: Atomic Spectroscopy
 high temp (2000-6000K) Chapter 20: Atomic Spectroscopy 20-1. An Overview Most compounds Atoms in gas phase high temp (2000-6000K) (AES) (AAS) (AFS) sample Mass-to-charge (ICP-MS) Atomic Absorption experiment
high temp (2000-6000K) Chapter 20: Atomic Spectroscopy 20-1. An Overview Most compounds Atoms in gas phase high temp (2000-6000K) (AES) (AAS) (AFS) sample Mass-to-charge (ICP-MS) Atomic Absorption experiment
2101 Atomic Spectroscopy
 2101 Atomic Spectroscopy Atomic identification Atomic spectroscopy refers to the absorption and emission of ultraviolet to visible light by atoms and monoatomic ions. It is best used to analyze metals.
2101 Atomic Spectroscopy Atomic identification Atomic spectroscopy refers to the absorption and emission of ultraviolet to visible light by atoms and monoatomic ions. It is best used to analyze metals.
Characterisation of vibrational modes of adsorbed species
 17.7.5 Characterisation of vibrational modes of adsorbed species Infrared spectroscopy (IR) See Ch.10. Infrared vibrational spectra originate in transitions between discrete vibrational energy levels of
17.7.5 Characterisation of vibrational modes of adsorbed species Infrared spectroscopy (IR) See Ch.10. Infrared vibrational spectra originate in transitions between discrete vibrational energy levels of
Because light behaves like a wave, we can describe it in one of two ways by its wavelength or by its frequency.
 Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
Unit 11 Instrumentation. Mass, Infrared and NMR Spectroscopy
 Unit 11 Instrumentation Mass, Infrared and NMR Spectroscopy Spectroscopic identification of organic compounds Qualitative analysis: presence but not quantity (i.e. PEDs) Quantitative analysis: quantity
Unit 11 Instrumentation Mass, Infrared and NMR Spectroscopy Spectroscopic identification of organic compounds Qualitative analysis: presence but not quantity (i.e. PEDs) Quantitative analysis: quantity
ATOMIC ABSORPTION SPECTROSCOPY (AAS) is an analytical technique that measures the concentrations of elements. It makes use of the absorption of light
 ATOMIC ABSORPTION SPECTROSCOPY (AAS) is an analytical technique that measures the concentrations of elements. It makes use of the absorption of light by these elements in order to measure their concentration.
ATOMIC ABSORPTION SPECTROSCOPY (AAS) is an analytical technique that measures the concentrations of elements. It makes use of the absorption of light by these elements in order to measure their concentration.
Absorption and scattering
 Absorption and scattering When a beam of radiation goes through the atmosphere, it encounters gas molecules, aerosols, cloud droplets, and ice crystals. These objects perturb the radiation field. Part
Absorption and scattering When a beam of radiation goes through the atmosphere, it encounters gas molecules, aerosols, cloud droplets, and ice crystals. These objects perturb the radiation field. Part
Measurements in Optics for Civil Engineers
 Measurements in Optics for Civil Engineers I. FOCAL LENGTH OF LENSES The behavior of simplest optical devices can be described by the method of geometrical optics. For convex or converging and concave
Measurements in Optics for Civil Engineers I. FOCAL LENGTH OF LENSES The behavior of simplest optical devices can be described by the method of geometrical optics. For convex or converging and concave
Fourier Transform IR Spectroscopy
 Fourier Transform IR Spectroscopy Absorption peaks in an infrared absorption spectrum arise from molecular vibrations Absorbed energy causes molecular motions which create a net change in the dipole moment.
Fourier Transform IR Spectroscopy Absorption peaks in an infrared absorption spectrum arise from molecular vibrations Absorbed energy causes molecular motions which create a net change in the dipole moment.
Classification of spectroscopic methods
 Introduction Spectroscopy is the study of the interaction between the electromagnetic radiation and the matter. Spectrophotometry is the measurement of these interactions i.e. the measurement of the intensity
Introduction Spectroscopy is the study of the interaction between the electromagnetic radiation and the matter. Spectrophotometry is the measurement of these interactions i.e. the measurement of the intensity
Electromagnetic Radiation and Scientific Instruments. PTYS April 1, 2008
 Electromagnetic Radiation and Scientific Instruments PTYS 206-2 April 1, 2008 Announcements Deep Impact 6 PM Wednesday Night Pizza, no beer Watch at home if you can t watch here. It will be discussed in
Electromagnetic Radiation and Scientific Instruments PTYS 206-2 April 1, 2008 Announcements Deep Impact 6 PM Wednesday Night Pizza, no beer Watch at home if you can t watch here. It will be discussed in
IR Spectrography - Absorption. Raman Spectrography - Scattering. n 0 n M - Raman n 0 - Rayleigh
 RAMAN SPECTROSCOPY Scattering Mid-IR and NIR require absorption of radiation from a ground level to an excited state, requires matching of radiation from source with difference in energy states. Raman
RAMAN SPECTROSCOPY Scattering Mid-IR and NIR require absorption of radiation from a ground level to an excited state, requires matching of radiation from source with difference in energy states. Raman
AN INTRODUCTION TO ATOMIC SPECTROSCOPY
 AN INTRODUCTION TO ATOMIC SPECTROSCOPY Atomic spectroscopy deals with the absorption, emission, or fluorescence by atom or elementary ions. Two regions of the spectrum yield atomic information- the UV-visible
AN INTRODUCTION TO ATOMIC SPECTROSCOPY Atomic spectroscopy deals with the absorption, emission, or fluorescence by atom or elementary ions. Two regions of the spectrum yield atomic information- the UV-visible
Spectroscopy. Page 1 of 8 L.Pillay (2012)
 Spectroscopy Electromagnetic radiation is widely used in analytical chemistry. The identification and quantification of samples using electromagnetic radiation (light) is called spectroscopy. Light has
Spectroscopy Electromagnetic radiation is widely used in analytical chemistry. The identification and quantification of samples using electromagnetic radiation (light) is called spectroscopy. Light has
MODULE 4.3 Atmospheric analysis of particulates
 MODULE 4.3 Atmospheric analysis of particulates Measurement And Characterisation Of The Particulate Content 1 Total particulate concentration 1 Composition of the particulate 1 Determination of particle
MODULE 4.3 Atmospheric analysis of particulates Measurement And Characterisation Of The Particulate Content 1 Total particulate concentration 1 Composition of the particulate 1 Determination of particle
Ms. Monika Srivastava Doctoral Scholar, AMR Group of Dr. Anurag Srivastava ABV-IIITM, Gwalior
 By Ms. Monika Srivastava Doctoral Scholar, AMR Group of Dr. Anurag Srivastava ABV-IIITM, Gwalior Unit 2 Laser acronym Laser Vs ordinary light Characteristics of lasers Different processes involved in lasers
By Ms. Monika Srivastava Doctoral Scholar, AMR Group of Dr. Anurag Srivastava ABV-IIITM, Gwalior Unit 2 Laser acronym Laser Vs ordinary light Characteristics of lasers Different processes involved in lasers
Electron Spin Resonance, Basic principle of NMR, Application of NMR in the study of Biomolecules, NMR imaging and in vivo NMR spectromicroscopy
 Electron Spin Resonance, Basic principle of NMR, Application of NMR in the study of Biomolecules, NMR imaging and in vivo NMR spectromicroscopy Mitesh Shrestha Electron Spin Resonance Electron paramagnetic
Electron Spin Resonance, Basic principle of NMR, Application of NMR in the study of Biomolecules, NMR imaging and in vivo NMR spectromicroscopy Mitesh Shrestha Electron Spin Resonance Electron paramagnetic
Atmospheric Analysis Gases. Sampling and analysis of gaseous compounds
 Atmospheric Analysis Gases Sampling and analysis of gaseous compounds Introduction - External environment (ambient air) ; global warming, acid rain, introduction of pollutants, etc - Internal environment
Atmospheric Analysis Gases Sampling and analysis of gaseous compounds Introduction - External environment (ambient air) ; global warming, acid rain, introduction of pollutants, etc - Internal environment
Ultraviolet-Visible and Infrared Spectrophotometry
 Ultraviolet-Visible and Infrared Spectrophotometry Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11451
Ultraviolet-Visible and Infrared Spectrophotometry Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11451
10/2/2008. hc λ. νλ =c. proportional to frequency. Energy is inversely proportional to wavelength And is directly proportional to wavenumber
 CH217 Fundamentals of Analytical Chemistry Module Leader: Dr. Alison Willows Electromagnetic spectrum Properties of electromagnetic radiation Many properties of electromagnetic radiation can be described
CH217 Fundamentals of Analytical Chemistry Module Leader: Dr. Alison Willows Electromagnetic spectrum Properties of electromagnetic radiation Many properties of electromagnetic radiation can be described
Photoelectric effect
 Laboratory#3 Phys4480/5480 Dr. Cristian Bahrim Photoelectric effect In 1900, Planck postulated that light is emitted and absorbed in discrete but tiny bundles of energy, E = hν, called today photons. Here
Laboratory#3 Phys4480/5480 Dr. Cristian Bahrim Photoelectric effect In 1900, Planck postulated that light is emitted and absorbed in discrete but tiny bundles of energy, E = hν, called today photons. Here
1) Introduction 2) Photo electric effect 3) Dual nature of matter 4) Bohr s atom model 5) LASERS
 1) Introduction 2) Photo electric effect 3) Dual nature of matter 4) Bohr s atom model 5) LASERS 1. Introduction Types of electron emission, Dunnington s method, different types of spectra, Fraunhoffer
1) Introduction 2) Photo electric effect 3) Dual nature of matter 4) Bohr s atom model 5) LASERS 1. Introduction Types of electron emission, Dunnington s method, different types of spectra, Fraunhoffer
FTIR Spectrometer. Basic Theory of Infrared Spectrometer. FTIR Spectrometer. FTIR Accessories
 FTIR Spectrometer Basic Theory of Infrared Spectrometer FTIR Spectrometer FTIR Accessories What is Infrared? Infrared radiation lies between the visible and microwave portions of the electromagnetic spectrum.
FTIR Spectrometer Basic Theory of Infrared Spectrometer FTIR Spectrometer FTIR Accessories What is Infrared? Infrared radiation lies between the visible and microwave portions of the electromagnetic spectrum.
Application of IR Raman Spectroscopy
 Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
CHAPTER A2 LASER DESORPTION IONIZATION AND MALDI
 Back to Basics Section A: Ionization Processes CHAPTER A2 LASER DESORPTION IONIZATION AND MALDI TABLE OF CONTENTS Quick Guide...27 Summary...29 The Ionization Process...31 Other Considerations on Laser
Back to Basics Section A: Ionization Processes CHAPTER A2 LASER DESORPTION IONIZATION AND MALDI TABLE OF CONTENTS Quick Guide...27 Summary...29 The Ionization Process...31 Other Considerations on Laser
2001 Spectrometers. Instrument Machinery. Movies from this presentation can be access at
 2001 Spectrometers Instrument Machinery Movies from this presentation can be access at http://www.shsu.edu/~chm_tgc/sounds/sound.html Chp20: 1 Optical Instruments Instrument Components Components of various
2001 Spectrometers Instrument Machinery Movies from this presentation can be access at http://www.shsu.edu/~chm_tgc/sounds/sound.html Chp20: 1 Optical Instruments Instrument Components Components of various
Spectrophotometry. Introduction
 Spectrophotometry Spectrophotometry is a method to measure how much a chemical substance absorbs light by measuring the intensity of light as a beam of light passes through sample solution. The basic principle
Spectrophotometry Spectrophotometry is a method to measure how much a chemical substance absorbs light by measuring the intensity of light as a beam of light passes through sample solution. The basic principle
Chemistry Instrumental Analysis Lecture 34. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 34 From molecular to elemental analysis there are three major techniques used for elemental analysis: Optical spectrometry Mass spectrometry X-ray spectrometry
Chemistry 4631 Instrumental Analysis Lecture 34 From molecular to elemental analysis there are three major techniques used for elemental analysis: Optical spectrometry Mass spectrometry X-ray spectrometry
Chapter 3 Electrochemical methods of Analysis-Potentiometry
 Chapter 3 Electrochemical methods of Analysis-Potentiometry Electroanalytical chemistry Contents Introduction Galvanic and electrolytic cells Salt bridge Electrode potential and cell potential Indicator
Chapter 3 Electrochemical methods of Analysis-Potentiometry Electroanalytical chemistry Contents Introduction Galvanic and electrolytic cells Salt bridge Electrode potential and cell potential Indicator
Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy
 Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy Section I Q1. Answer (i) (b) (ii) (d) (iii) (c) (iv) (c) (v) (a) (vi) (b) (vii) (b) (viii) (a) (ix)
Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy Section I Q1. Answer (i) (b) (ii) (d) (iii) (c) (iv) (c) (v) (a) (vi) (b) (vii) (b) (viii) (a) (ix)
25 Instruments for Optical Spectrometry
 25 Instruments for Optical Spectrometry 25A INSTRUMENT COMPONENTS (1) source of radiant energy (2) wavelength selector (3) sample container (4) detector (5) signal processor and readout (a) (b) (c) Fig.
25 Instruments for Optical Spectrometry 25A INSTRUMENT COMPONENTS (1) source of radiant energy (2) wavelength selector (3) sample container (4) detector (5) signal processor and readout (a) (b) (c) Fig.
Chemistry Instrumental Analysis Lecture 18. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 18 Instrumentation Radiation sources Hollow cathode lamp Most common source Consist of W anode and a cathode sealed in a glass tube filled with Ne or Ar. Hollow
Chemistry 4631 Instrumental Analysis Lecture 18 Instrumentation Radiation sources Hollow cathode lamp Most common source Consist of W anode and a cathode sealed in a glass tube filled with Ne or Ar. Hollow
VALLIAMMAI ENGINEERING COLLEGE SRM Nagar, Kattankulathur
 VALLIAMMAI ENGINEERING COLLEGE SRM Nagar, Kattankulathur 603 203 DEPARTMENT OF ELECTRONICS AND INSTRUMENTATION ENGINEERING QUESTION BANK V SEMESTER EI6501 Analytical Instruments Regulation 2013 Academic
VALLIAMMAI ENGINEERING COLLEGE SRM Nagar, Kattankulathur 603 203 DEPARTMENT OF ELECTRONICS AND INSTRUMENTATION ENGINEERING QUESTION BANK V SEMESTER EI6501 Analytical Instruments Regulation 2013 Academic
Ch. 8 Introduction to Optical Atomic Spectroscopy
 Ch. 8 Introduction to Optical Atomic Spectroscopy 8.1 3 major types of Spectrometry elemental Optical Spectrometry Ch 9, 10 Mass Spectrometry Ch 11 X-ray Spectrometry Ch 12 In this chapter theories on
Ch. 8 Introduction to Optical Atomic Spectroscopy 8.1 3 major types of Spectrometry elemental Optical Spectrometry Ch 9, 10 Mass Spectrometry Ch 11 X-ray Spectrometry Ch 12 In this chapter theories on
Design and Development of a Smartphone Based Visible Spectrophotometer for Analytical Applications
 Design and Development of a Smartphone Based Visible Spectrophotometer for Analytical Applications Bedanta Kr. Deka, D. Thakuria, H. Bora and S. Banerjee # Department of Physicis, B. Borooah College, Ulubari,
Design and Development of a Smartphone Based Visible Spectrophotometer for Analytical Applications Bedanta Kr. Deka, D. Thakuria, H. Bora and S. Banerjee # Department of Physicis, B. Borooah College, Ulubari,
Light sources. Excited gas atoms are the primaty source of radiation in combustion and discharge lamps. lamps is not continuous!
 Light sources Excited gas atoms are the primaty source of radiation in combustion and discharge lamps. Continuous spectrum: black body radiation Characteristic spectrum: emission lines Absorption lines
Light sources Excited gas atoms are the primaty source of radiation in combustion and discharge lamps. Continuous spectrum: black body radiation Characteristic spectrum: emission lines Absorption lines
Chapter 24 Photonics Question 1 Question 2 Question 3 Question 4 Question 5
 Chapter 24 Photonics Data throughout this chapter: e = 1.6 10 19 C; h = 6.63 10 34 Js (or 4.14 10 15 ev s); m e = 9.1 10 31 kg; c = 3.0 10 8 m s 1 Question 1 Visible light has a range of photons with wavelengths
Chapter 24 Photonics Data throughout this chapter: e = 1.6 10 19 C; h = 6.63 10 34 Js (or 4.14 10 15 ev s); m e = 9.1 10 31 kg; c = 3.0 10 8 m s 1 Question 1 Visible light has a range of photons with wavelengths
Mass Spectrometry in MCAL
 Mass Spectrometry in MCAL Two systems: GC-MS, LC-MS GC seperates small, volatile, non-polar material MS is detection devise (Agilent 320-MS TQ Mass Spectrometer) Full scan monitoring SIM single ion monitoring
Mass Spectrometry in MCAL Two systems: GC-MS, LC-MS GC seperates small, volatile, non-polar material MS is detection devise (Agilent 320-MS TQ Mass Spectrometer) Full scan monitoring SIM single ion monitoring
Taking fingerprints of stars, galaxies, and interstellar gas clouds
 - - Taking fingerprints of stars, galaxies, and interstellar gas clouds Absorption and emission from atoms, ions, and molecules Periodic Table of Elements The universe is mostly hydrogen H and helium He
- - Taking fingerprints of stars, galaxies, and interstellar gas clouds Absorption and emission from atoms, ions, and molecules Periodic Table of Elements The universe is mostly hydrogen H and helium He
3 - Atomic Absorption Spectroscopy
 3 - Atomic Absorption Spectroscopy Introduction Atomic-absorption (AA) spectroscopy uses the absorption of light to measure the concentration of gas-phase atoms. Since samples are usually liquids or solids,
3 - Atomic Absorption Spectroscopy Introduction Atomic-absorption (AA) spectroscopy uses the absorption of light to measure the concentration of gas-phase atoms. Since samples are usually liquids or solids,
Taking Fingerprints of Stars, Galaxies, and Other Stuff. The Bohr Atom. The Bohr Atom Model of Hydrogen atom. Bohr Atom. Bohr Atom
 Periodic Table of Elements Taking Fingerprints of Stars, Galaxies, and Other Stuff Absorption and Emission from Atoms, Ions, and Molecules Universe is mostly (97%) Hydrogen and Helium (H and He) The ONLY
Periodic Table of Elements Taking Fingerprints of Stars, Galaxies, and Other Stuff Absorption and Emission from Atoms, Ions, and Molecules Universe is mostly (97%) Hydrogen and Helium (H and He) The ONLY
Advanced Spectroscopy Laboratory
 Advanced Spectroscopy Laboratory - Raman Spectroscopy - Emission Spectroscopy - Absorption Spectroscopy - Raman Microscopy - Hyperspectral Imaging Spectroscopy FERGIELAB TM Raman Spectroscopy Absorption
Advanced Spectroscopy Laboratory - Raman Spectroscopy - Emission Spectroscopy - Absorption Spectroscopy - Raman Microscopy - Hyperspectral Imaging Spectroscopy FERGIELAB TM Raman Spectroscopy Absorption
Solution 3: A glass prism deviates the violet light most and the red light least.
 EXERCISE- 6 (A) Question 1: Name three factors on which the deviation produces by a prism depends and state how does it depend on the factors stated by you. Solution 1: The deviation produced by the prism
EXERCISE- 6 (A) Question 1: Name three factors on which the deviation produces by a prism depends and state how does it depend on the factors stated by you. Solution 1: The deviation produced by the prism
Science Drivers. Spectroscopic Sensors. In Situ Sensors. Development of autonomous and remote platforms
 Science Drivers In Situ Sensors Spectroscopic Sensors Development of autonomous and remote platforms ROVs, AUVs Cabled observatories Desire to analyze targets with discrete stability regions in the deep
Science Drivers In Situ Sensors Spectroscopic Sensors Development of autonomous and remote platforms ROVs, AUVs Cabled observatories Desire to analyze targets with discrete stability regions in the deep
The Nature of Light. We have a dual model
 Light and Atoms Properties of Light We can come to understand the composition of distant bodies by analyzing the light they emit This analysis can tell us about the composition as well as the temperature
Light and Atoms Properties of Light We can come to understand the composition of distant bodies by analyzing the light they emit This analysis can tell us about the composition as well as the temperature
atomic absorption spectroscopy general can be portable and used in-situ preserves sample simpler and less expensive
 Chapter 9: End-of-Chapter Solutions 1. The following comparison provides general trends, but both atomic absorption spectroscopy (AAS) and atomic absorption spectroscopy (AES) will have analyte-specific
Chapter 9: End-of-Chapter Solutions 1. The following comparison provides general trends, but both atomic absorption spectroscopy (AAS) and atomic absorption spectroscopy (AES) will have analyte-specific
Electromagnetic spectra
 Properties of Light Waves, particles and EM spectrum Interaction with matter Absorption Reflection, refraction and scattering Polarization and diffraction Reading foci: pp 175-185, 191-199 not responsible
Properties of Light Waves, particles and EM spectrum Interaction with matter Absorption Reflection, refraction and scattering Polarization and diffraction Reading foci: pp 175-185, 191-199 not responsible
Chapter 9. Atomic emission and Atomic Fluorescence Spectrometry Emission spectrophotometric Techniques
 Chapter 9 Atomic emission and Atomic Fluorescence Spectrometry Emission spectrophotometric Techniques Emission Spectroscopy Flame and Plasma Emission Spectroscopy are based upon those particles that are
Chapter 9 Atomic emission and Atomic Fluorescence Spectrometry Emission spectrophotometric Techniques Emission Spectroscopy Flame and Plasma Emission Spectroscopy are based upon those particles that are
Sampling for Gases and Vapors
 Sampling for Gases and Vapors EOH 466B Evaluating the Occupational Environment Spring 2008 Gases and Vapors Gases: don't exist in liquid state at STP e.g.: nitrous oxides, ozone, carbon monoxide Vapors:
Sampling for Gases and Vapors EOH 466B Evaluating the Occupational Environment Spring 2008 Gases and Vapors Gases: don't exist in liquid state at STP e.g.: nitrous oxides, ozone, carbon monoxide Vapors:
Molecular spectroscopy
 Molecular spectroscopy Origin of spectral lines = absorption, emission and scattering of a photon when the energy of a molecule changes: rad( ) M M * rad( ' ) ' v' 0 0 absorption( ) emission ( ) scattering
Molecular spectroscopy Origin of spectral lines = absorption, emission and scattering of a photon when the energy of a molecule changes: rad( ) M M * rad( ' ) ' v' 0 0 absorption( ) emission ( ) scattering
Spectrum of Radiation. Importance of Radiation Transfer. Radiation Intensity and Wavelength. Lecture 3: Atmospheric Radiative Transfer and Climate
 Lecture 3: Atmospheric Radiative Transfer and Climate Radiation Intensity and Wavelength frequency Planck s constant Solar and infrared radiation selective absorption and emission Selective absorption
Lecture 3: Atmospheric Radiative Transfer and Climate Radiation Intensity and Wavelength frequency Planck s constant Solar and infrared radiation selective absorption and emission Selective absorption
Radionuclide Imaging MII Detection of Nuclear Emission
 Radionuclide Imaging MII 3073 Detection of Nuclear Emission Nuclear radiation detectors Detectors that are commonly used in nuclear medicine: 1. Gas-filled detectors 2. Scintillation detectors 3. Semiconductor
Radionuclide Imaging MII 3073 Detection of Nuclear Emission Nuclear radiation detectors Detectors that are commonly used in nuclear medicine: 1. Gas-filled detectors 2. Scintillation detectors 3. Semiconductor
Lecture 3: Atmospheric Radiative Transfer and Climate
 Lecture 3: Atmospheric Radiative Transfer and Climate Solar and infrared radiation selective absorption and emission Selective absorption and emission Cloud and radiation Radiative-convective equilibrium
Lecture 3: Atmospheric Radiative Transfer and Climate Solar and infrared radiation selective absorption and emission Selective absorption and emission Cloud and radiation Radiative-convective equilibrium
Chemistry Instrumental Analysis Lecture 15. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 15 IR Instruments Types of Instrumentation Dispersive Spectrophotometers (gratings) Fourier transform spectrometers (interferometer) Single beam Double beam
Chemistry 4631 Instrumental Analysis Lecture 15 IR Instruments Types of Instrumentation Dispersive Spectrophotometers (gratings) Fourier transform spectrometers (interferometer) Single beam Double beam
Chapter 6 Electronic Structure of Atoms
 Chapter 6 Electronic Structure of Atoms What is the origin of color in matter? Demo: flame tests What does this have to do with the atom? Why are atomic properties periodic? 6.1 The Wave Nature of Light
Chapter 6 Electronic Structure of Atoms What is the origin of color in matter? Demo: flame tests What does this have to do with the atom? Why are atomic properties periodic? 6.1 The Wave Nature of Light
Higher -o-o-o- Past Paper questions o-o-o- 3.4 Spectra
 Higher -o-o-o- Past Paper questions 1991-2010 -o-o-o- 3.4 Spectra 1992 Q37 The diagram below shows the energy levels for the hydrogen atom. (a) Between which two energy levels would an electron transition
Higher -o-o-o- Past Paper questions 1991-2010 -o-o-o- 3.4 Spectra 1992 Q37 The diagram below shows the energy levels for the hydrogen atom. (a) Between which two energy levels would an electron transition
Introduction to Spectroscopic methods
 Introduction to Spectroscopic methods Spectroscopy: Study of interaction between light* and matter. Spectrometry: Implies a quantitative measurement of intensity. * More generally speaking electromagnetic
Introduction to Spectroscopic methods Spectroscopy: Study of interaction between light* and matter. Spectrometry: Implies a quantitative measurement of intensity. * More generally speaking electromagnetic
2.7. Raman and other Spectroscopies
 .7. Raman and other Spectroscopies The analysis and identification of the pigment chemistry of paint! Identifies radiation which is characteristic for molecular excitation modes. L. Burgio et al., Anal.
.7. Raman and other Spectroscopies The analysis and identification of the pigment chemistry of paint! Identifies radiation which is characteristic for molecular excitation modes. L. Burgio et al., Anal.
State the position of protons, neutrons and electrons in the atom
 2.1 The Atom 2.1.1 - State the position of protons, neutrons and electrons in the atom Atoms are made up of a nucleus containing positively charged protons and neutral neutrons, with negatively charged
2.1 The Atom 2.1.1 - State the position of protons, neutrons and electrons in the atom Atoms are made up of a nucleus containing positively charged protons and neutral neutrons, with negatively charged
Instrumental Analysis. Mass Spectrometry. Lecturer:! Somsak Sirichai
 303351 Instrumental Analysis Mass Spectrometry Lecturer:! Somsak Sirichai Mass Spectrometry What is Mass spectrometry (MS)? An analytic method that employs ionization and mass analysis of compounds in
303351 Instrumental Analysis Mass Spectrometry Lecturer:! Somsak Sirichai Mass Spectrometry What is Mass spectrometry (MS)? An analytic method that employs ionization and mass analysis of compounds in
3. Photons of which of the following colors of light possess the greatest amount of energy? A) blue B) green C) yellow D) red
 1. Atoms that have eight valence electrons would tend to A) be very reactive. B) be inert. C) form positive ions. D) form negative ions. 3. Photons of which of the following colors of light possess the
1. Atoms that have eight valence electrons would tend to A) be very reactive. B) be inert. C) form positive ions. D) form negative ions. 3. Photons of which of the following colors of light possess the
Production of X-rays. Radiation Safety Training for Analytical X-Ray Devices Module 9
 Module 9 This module presents information on what X-rays are and how they are produced. Introduction Module 9, Page 2 X-rays are a type of electromagnetic radiation. Other types of electromagnetic radiation
Module 9 This module presents information on what X-rays are and how they are produced. Introduction Module 9, Page 2 X-rays are a type of electromagnetic radiation. Other types of electromagnetic radiation
Option D: Medicinal Chemistry
 Option D: Medicinal Chemistry When synthesizing drugs, they end up as a mixture of many different solvents and need to be separated physical properties will help solubility difference volatility difference
Option D: Medicinal Chemistry When synthesizing drugs, they end up as a mixture of many different solvents and need to be separated physical properties will help solubility difference volatility difference
Principles of Biomedical Systems & Devices
 Principles of Biomedical Systems & Devices Lecture 17 Clinical Systems Today Week in PBS&D Clinical Laboratory Instrumentation Spectrophotometry Autoanalyzers Chromatology - Gas Chromatgraphs (Mass) Spectroscopy
Principles of Biomedical Systems & Devices Lecture 17 Clinical Systems Today Week in PBS&D Clinical Laboratory Instrumentation Spectrophotometry Autoanalyzers Chromatology - Gas Chromatgraphs (Mass) Spectroscopy
ISA QATAR : 90 th Seminar
 ANALYTICAL INSTRUMENTATION & MAINTENANCE SYSTEMS (AIMS) ISA QATAR : 90 th Seminar UTILIZATION OF LASERS IN THE FIELD OF GAS ANALYZERS Presenter: ZAHEER JUDDY Electromagnetic Spectrum Electromagnetic Spectrum
ANALYTICAL INSTRUMENTATION & MAINTENANCE SYSTEMS (AIMS) ISA QATAR : 90 th Seminar UTILIZATION OF LASERS IN THE FIELD OF GAS ANALYZERS Presenter: ZAHEER JUDDY Electromagnetic Spectrum Electromagnetic Spectrum
CHEM6416 Theory of Molecular Spectroscopy 2013Jan Spectroscopy frequency dependence of the interaction of light with matter
 CHEM6416 Theory of Molecular Spectroscopy 2013Jan22 1 1. Spectroscopy frequency dependence of the interaction of light with matter 1.1. Absorption (excitation), emission, diffraction, scattering, refraction
CHEM6416 Theory of Molecular Spectroscopy 2013Jan22 1 1. Spectroscopy frequency dependence of the interaction of light with matter 1.1. Absorption (excitation), emission, diffraction, scattering, refraction
What do we study and do?
 What do we study and do? Light comes from electrons transitioning from higher energy to lower energy levels. Wave-particle nature of light Wave nature: refraction, diffraction, interference (labs) Particle
What do we study and do? Light comes from electrons transitioning from higher energy to lower energy levels. Wave-particle nature of light Wave nature: refraction, diffraction, interference (labs) Particle
Modern physics ideas are strange! L 36 Modern Physics [2] The Photon Concept. How are x-rays produced? The uncertainty principle
![Modern physics ideas are strange! L 36 Modern Physics [2] The Photon Concept. How are x-rays produced? The uncertainty principle Modern physics ideas are strange! L 36 Modern Physics [2] The Photon Concept. How are x-rays produced? The uncertainty principle](/thumbs/88/117098787.jpg) L 36 Modern Physics [2] X-rays & gamma rays How lasers work Medical applications of lasers Applications of high power lasers Medical imaging techniques CAT scans MRI s Modern physics ideas are strange!
L 36 Modern Physics [2] X-rays & gamma rays How lasers work Medical applications of lasers Applications of high power lasers Medical imaging techniques CAT scans MRI s Modern physics ideas are strange!
FLAME PHOTOMETRY AIM INTRODUCTION
 FLAME PHOTOMETRY AIM INTRODUCTION Atomic spectroscopy is based on the absorption, emission or fluorescence process of light by atoms or elementary ions. Information for atomic scale is obtained in two
FLAME PHOTOMETRY AIM INTRODUCTION Atomic spectroscopy is based on the absorption, emission or fluorescence process of light by atoms or elementary ions. Information for atomic scale is obtained in two
PC Laboratory Raman Spectroscopy
 PC Laboratory Raman Spectroscopy Schedule: Week of September 5-9: Student presentations Week of September 19-23:Student experiments Learning goals: (1) Hands-on experience with setting up a spectrometer.
PC Laboratory Raman Spectroscopy Schedule: Week of September 5-9: Student presentations Week of September 19-23:Student experiments Learning goals: (1) Hands-on experience with setting up a spectrometer.
Lecture 0. NC State University
 Chemistry 736 Lecture 0 Overview NC State University Overview of Spectroscopy Electronic states and energies Transitions between states Absorption and emission Electronic spectroscopy Instrumentation Concepts
Chemistry 736 Lecture 0 Overview NC State University Overview of Spectroscopy Electronic states and energies Transitions between states Absorption and emission Electronic spectroscopy Instrumentation Concepts
PhET Interactive Chemistry Simulations Aligned to an Example General Chemistry Curriculum
 PhET Interactive Chemistry Simulations Aligned to an Example General Chemistry Curriculum Alignment is based on the topics and subtopics addressed by each sim. Sims that directly address the topic area
PhET Interactive Chemistry Simulations Aligned to an Example General Chemistry Curriculum Alignment is based on the topics and subtopics addressed by each sim. Sims that directly address the topic area
University of Cyprus. Reflectance and Diffuse Spectroscopy
 University of Cyprus Biomedical Imaging and Applied Optics Reflectance and Diffuse Spectroscopy Spectroscopy What is it? from the Greek: spectro = color + scope = look at or observe = measuring/recording
University of Cyprus Biomedical Imaging and Applied Optics Reflectance and Diffuse Spectroscopy Spectroscopy What is it? from the Greek: spectro = color + scope = look at or observe = measuring/recording
New advances in folded pathlength technology for Process Tunable Diode Laser Absorption Spectrometers (TDLAS)
 New advances in folded pathlength technology for Process Tunable Diode Laser Absorption Spectrometers (TDLAS) Jean-Nicolas Adami, PhD Head of Strategic Product Group Gas Analytics Mettler-Toledo GmbH,
New advances in folded pathlength technology for Process Tunable Diode Laser Absorption Spectrometers (TDLAS) Jean-Nicolas Adami, PhD Head of Strategic Product Group Gas Analytics Mettler-Toledo GmbH,
Activities at the Laboratory of the Nuclear Engineering Department of the Polytechnic University of Valencia
 7 th Workshop on European Collaboration for Higher Education and Research in Nuclear Engineering & Radiological Protection Bruxelles, Belgique 30 May - 1 June 2011 Activities at the Laboratory of the Nuclear
7 th Workshop on European Collaboration for Higher Education and Research in Nuclear Engineering & Radiological Protection Bruxelles, Belgique 30 May - 1 June 2011 Activities at the Laboratory of the Nuclear
Taking fingerprints of stars, galaxies, and interstellar gas clouds. Absorption and emission from atoms, ions, and molecules
 Taking fingerprints of stars, galaxies, and interstellar gas clouds Absorption and emission from atoms, ions, and molecules 1 Periodic Table of Elements The universe is mostly hydrogen H and helium He
Taking fingerprints of stars, galaxies, and interstellar gas clouds Absorption and emission from atoms, ions, and molecules 1 Periodic Table of Elements The universe is mostly hydrogen H and helium He
Q. Shen 1,2) and T. Toyoda 1,2)
 Photosensitization of nanostructured TiO 2 electrodes with CdSe quntum dots: effects of microstructure in substrates Q. Shen 1,2) and T. Toyoda 1,2) Department of Applied Physics and Chemistry 1), and
Photosensitization of nanostructured TiO 2 electrodes with CdSe quntum dots: effects of microstructure in substrates Q. Shen 1,2) and T. Toyoda 1,2) Department of Applied Physics and Chemistry 1), and
Lecture 7: Atomic Spectroscopy
 Lecture 7: Atomic Spectroscopy 1 Atomic spectroscopy The wavelengths of absorbance and emission from atoms in the gas phase are characteristic of atomic orbitals. 2 In the lowest energy transition, the
Lecture 7: Atomic Spectroscopy 1 Atomic spectroscopy The wavelengths of absorbance and emission from atoms in the gas phase are characteristic of atomic orbitals. 2 In the lowest energy transition, the
RINCIPLE OF COLORIMETER AND SPECTOPHOTOMETER AND VARIOUS TYPE OF ANALYSER USED IN CLINICAL BIOCHEMISTRY
 RINCIPLE OF COLORIMETER AND SPECTOPHOTOMETER AND VARIOUS TYPE OF ANALYSER USED IN CLINICAL BIOCHEMISTRY COLORIMETER What is colorimeter? Colorimetry. Principle of colorimeter. Beer's and Lambert's law.
RINCIPLE OF COLORIMETER AND SPECTOPHOTOMETER AND VARIOUS TYPE OF ANALYSER USED IN CLINICAL BIOCHEMISTRY COLORIMETER What is colorimeter? Colorimetry. Principle of colorimeter. Beer's and Lambert's law.
Lecture 6 - spectroscopy
 Lecture 6 - spectroscopy 1 Light Electromagnetic radiation can be thought of as either a wave or as a particle (particle/wave duality). For scattering of light by particles, air, and surfaces, wave theory
Lecture 6 - spectroscopy 1 Light Electromagnetic radiation can be thought of as either a wave or as a particle (particle/wave duality). For scattering of light by particles, air, and surfaces, wave theory
2.8. Raman and other Spectroscopies
 .8. Raman and other Spectroscopies The analysis and identification of the pigment chemistry of paint! Identifies radiation which is characteristic for molecular excitation modes. L. Burgio et al., Anal.
.8. Raman and other Spectroscopies The analysis and identification of the pigment chemistry of paint! Identifies radiation which is characteristic for molecular excitation modes. L. Burgio et al., Anal.
TANDEM MASS SPECTROSCOPY
 TANDEM MASS SPECTROSCOPY 1 MASS SPECTROMETER TYPES OF MASS SPECTROMETER PRINCIPLE TANDEM MASS SPECTROMETER INSTRUMENTATION QUADRAPOLE MASS ANALYZER TRIPLE QUADRAPOLE MASS ANALYZER TIME OF FLIGHT MASS ANALYSER
TANDEM MASS SPECTROSCOPY 1 MASS SPECTROMETER TYPES OF MASS SPECTROMETER PRINCIPLE TANDEM MASS SPECTROMETER INSTRUMENTATION QUADRAPOLE MASS ANALYZER TRIPLE QUADRAPOLE MASS ANALYZER TIME OF FLIGHT MASS ANALYSER
PHYSICS 116 SPECTROSCOPY: DETERMINATION OF THE WAVELENGTH OF LIGHT
 Name Date Lab Time Lab TA PHYSICS 116 SPECTROSCOPY: DETERMINATION OF THE WAVELENGTH OF LIGHT I. PURPOSE To use a diffraction grating to investigate the spectra produced by several unknown gas discharge
Name Date Lab Time Lab TA PHYSICS 116 SPECTROSCOPY: DETERMINATION OF THE WAVELENGTH OF LIGHT I. PURPOSE To use a diffraction grating to investigate the spectra produced by several unknown gas discharge
Compact Knowledge: Absorbance Spectrophotometry. Flexible. Reliable. Personal.
 L A B O R A T O R Y C O M P E T E N C E Compact Knowledge: Absorbance Spectrophotometry Flexible. Reliable. Personal. The interaction of light with molecules is an essential and well accepted technique
L A B O R A T O R Y C O M P E T E N C E Compact Knowledge: Absorbance Spectrophotometry Flexible. Reliable. Personal. The interaction of light with molecules is an essential and well accepted technique
Gaseous CEMS technology
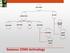 GAS CEMS EXTRACTIVE IN-SITU COLD DRY HOT WET DRY DILUTION PROBE/ POINT IN-STACK CROSS STACK OUT OF STACK Gaseous CEMS technology Probe (at stack) Heated filter Heated sample line Condenser Pump Analyzers
GAS CEMS EXTRACTIVE IN-SITU COLD DRY HOT WET DRY DILUTION PROBE/ POINT IN-STACK CROSS STACK OUT OF STACK Gaseous CEMS technology Probe (at stack) Heated filter Heated sample line Condenser Pump Analyzers
