ProbSeq a fragmentation model for interpretation of electrospray tandem mass spectrometry data
|
|
|
- Osborn Dawson
- 6 years ago
- Views:
Transcription
1 Comparative and Functional Genomics Comp Funct Genom 2004; 5: Published online in Wiley InterScience ( DOI: /cfg.370 Conference Paper ProbSeq a fragmentation model for interpretation of electrospray tandem mass spectrometry data John Skilling 1, Richard Denny 2, Keith Richardson 2 *, Phillip Young 2,Thérèse McKenna 2, Iain Campuzano 2 and Mark Ritchie 2 1 Maximum Entropy Data Consultants Ltd, Tresawsan, Killaha East, Kenmare, Ireland 2 Waters Corporation MS Technologies Centre, Floats Road, Wythenshawe, Manchester M23 9LZ, UK *Correspondence to: Keith Richardson, Waters Corporation MS Technologies Centre, Floats Road, Wythenshawe, Manchester M23 9LZ, UK. keith richardson@waters.com Abstract We describe a probabilistic peptide fragmentation model for use in protein databank searching and de novo sequencing of electrospray tandem mass spectrometry data. A probabilistic framework for tuning of the model using a range of well-characterized samples are introduced. We present preliminary results of our tuning efforts. Copyright 2004 John Wiley & Sons, Ltd. Received: 31 October 2003 Revised: 18 November 2003 Accepted: 26 November 2003 Introduction Sequencing of peptides from their collisioninduced fragments is a well-established technique (Papayannopoulos, 1995). A biological sample, which may contain an unknown protein or a mixture of proteins, is subjected to an enzymatic digest (usually tryptic) resulting in a mixture of peptides. This mixture is analysed using liquid chromatography and electrospray quadrupole timeof-flight (Q-TOF) mass spectrometry. A Q-TOF (Morris et al., 1996) mass spectrometer consists of a quadrupole mass analyser, a collision cell filled with an inert gas, and a time-of-flight analyser. The quadrupole allows the mass:charge range of peptides which enter the collision cell to be selected accurately. The Q-TOF can operate in two modes. In wide-bandpass, low-energy mode, all peptides are allowed to pass into the collision cell and the voltage applied to the collision cell is low, allowing the peptides to pass through intact into the TOF stage. In narrow-bandpass, high-energy mode, the quadrupole is tuned to pass only a small mass:charge range around a single peptide. In this mode, the collision cell voltage is relatively high, causing the peptide to fragment as it is pulled through the gas. The final, TOF stage of the instrument allows the peptide ion or fragment ion masses to be determined to an accuracy of around five parts per million on a well-calibrated instrument. The decision to switch mode is made in real time using a number of possible strategies, and the collision energy can be adjusted to make useful fragmentation of each peptide more likely. Peptides fragment in complex ways, and their fragments in turn have complex representations in mass spectra. Manual interpretation of these spectra is a skilled and laborious job, and a modern experiment can produce many hundreds. Around 1998 (see Skilling, 2000a, 2000b), we began to use the ProbSeq fragmentation model in combination with Markov chain methods to interpret this data. Commonly, we are required to answer two questions: How much can we infer Copyright 2004 John Wiley & Sons, Ltd.
2 62 J. Skilling et al. about the sequence of the peptide which gave rise to a single spectrum? and Given many spectra, what can we infer about the mixture of proteins that may be present?. To address these questions in a robust, probabilistic way, we appeal to Bayes theorem: Pr(Sequence Spectrum, I) Prior Likelihood = Evidence Pr(Sequence I) Pr(Spectrum Sequence, I) =. Pr(Spectrum I) (1) The Likelihood Pr(Spectrum Sequence, I) on the RHS of the above equation encodes our incomplete knowledge about peptide fragmentation. It is the probability that the peptide under consideration could give rise to the observed spectrum. The Prior Pr(Sequence I) is a probability that we assign to each candidate sequence before we look at the data. At this level, the Evidence is a normalization factor, but it can be used to objectively compare different likelihood functions and other background assumptions. I contains information about the context in which this calculation is being used, and it happens that it is different for each of the above questions. Our likelihood function is called ProbSeq and it is, in itself, a Bayesian calculation which involves a good deal of prior information about peptide fragmentation. The rest of this paper introduces a new framework which we can use to improve the model. We regard it to be a strength of the Bayesian approach that we can treat this as an inference problem at every level (Skilling, 1998). Materials and methods We decided to base the tuning of the likelihood function on human assignments of sequences to spectra. The samples chosen consisted of tryptic digests of pyruvate kinase, glyceraldehyde 3- phosphate dehydrogenase, alcohol dehydrogenase and β-lactoglobulin. The solvent was 0.1% formic acid in acetonitrile:water, 1:1. The samples were infused directly into a nanolockspray source. The instrument used was a Waters Q-TOF ULTIMA operating in positive ion mode. The collision energy was ev. The acquired spectra were analysed manually to provide a list of verified sequences. The data was processed using the MaxEnt3 deconvolution algorithm to remove isotope series and to resolve overlapping isotope clusters and multiple charge states. The data was split into two sets. The first, tuning, set was used to tune the fragmentation model, while the second, validation, set was reserved for assessment of the resulting parameters. The chemical structure and fragmentation modes for a typical peptide are shown in Figure 1. The types of fragments that are observed depends on the collision energy used. The calculation of the likelihood is based on a probabilistic summation over all of the possible ways that a peptide could fragment and give rise to trial masses: Pr(Spectrum Sequence,,I) = Frag Pr(Spectrum Frag) Pr(Frag Sequence,,I) (2) where is a database containing probabilistic information about peptide fragmentation and Frag is a particular fragmentation pattern. Experience tells us that some patterns of fragmentation are more likely than others, e.g. y ion series are correlated: if y n is present, there are better than even odds that y n+1 is also present. This is the information that is encoded in the last term in equation (2). For a peptide of length n amino acids, the sum in equation (2) contains 2 n terms for y ions alone and 2 6n terms if a,b,c,x,y,z-ions are all considered with extra factors for various losses. It would be impractical to do this summation explicitly for each trial sequence. Remarkably, using a Markov chain allows the summation to be performed in O(n) steps. Considering only y ions, the chain is structured as follows: Pr(y 1 on) = p 1 and Pr(y r on) = p r Pr(y r 1 on) + q r Pr(y r 1 off). (3) As mentioned above, the probability p that the y series stays on is expected to be greater than 0.5, and the probability q that it switches on is likely to
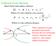
3 ProbSeq a fragmentation model for electrospray tandem MS data interpretation 63 a b c N-terminus NH 2 OH C-terminus HOOC C=NH NH H 3 C CH 3 CH 3 CH 2 HN N CH 2 (CH 2 ) 3 CH CH 2 HC CH 3 CH 2 CH 2 H 2 N CH CO NH CH CO NH CH CO NH CH CO NH CH CO NH CH CO N CO NH CH COOH x y z D R V Y I H P F Figure 1. The chemical structure of a peptide. The boldface letters are one-letter amino acid codes. Also indicated are the three backbone fragmentation modes. At the collision energies used in this study, the b, y mode dominates. a and z ions are also commonly observed. Losses can also occur from terminal and side-chain groups be less than 0.5. It is also to be expected that the values of p and q at a particular point in the chain should depend on the amino acid sequence of the peptide under consideration. At each step along the chain, the probability that a y ion will be observed is influenced by whether or not its predecessor was observed and we do as many summations as are locally available. When we reach the end of the chain, we have accumulated Pr(Spectrum Sequence,,I), with a computational cost of only O(n). As well as the Markov chain parameters which describe the expected appearance of series of b and y ions, each amino acid may undergo a number of losses, may exhibit a propensity for cleavage to occur on the C- or N-terminal side and may appear isolated from the rest of the sequence as an immonium ion. The maximum number of probabilities required to encode this description is seven per amino acid. This leads to our fragmentation model,, having over 100 tuneable parameters. Some examples are given in Table 1. Reasonable values for some of these can be arrived at by consulting the literature (Papayannopoulos, 1995), and others can be estimated by manually inspecting many fragmentation spectra. We decided to treat the tuning of the ProbSeq parameters as another exercise in Bayesian inference. In order to do this we needed to define prior distributions for our parameters and a likelihood function for comparison against the data. Having a Table 1. Some examples of tuneable likelihood parameters. The first column contains probabilities that apply globally, while the second column contains probabilities that may be different for each amino acid Global Pr(y n+1 on y n on) Pr(y n+1 off y n on) Pr(y H 2 O) Amino Acid Pr(break left) Pr(break right) Pr(Immonium) list of sequences validated against known spectra, the tuning likelihood becomes: Pr(Spectra Sequences,,I) = Pr(Spectrum Sequence,,I) (4) Spectra We take the prior distribution for each of the probabilities in the ProbSeq model,, to be uniform on (0, 1). We are left with the problem of how to explore the >100-dimensional parameter space. We validate the result by checking whether a chosen set of parameters,, does or does not enable a de novo sequence determination to recover the correct, known, sequence. This has the advantage of tuning our parameters directly upon our own implementation software. De novo sequencing involves the exploration of a large space of peptide sequences that are consistent
4 64 J. Skilling et al. with the intact mass of an unknown peptide. At least some of the candidate sequences must be compared with the associated fragmentation spectrum. The number of possible trial sequences grows exponentially with precursor mass with: log 2 n M 25 7 (5) where n is the number of trial sequences and M is the nominal mass of the precursor. The implementation used in the Waters proteomics product, ProteinLynx Global Server, does not use an exhaustive search but simulates this by sampling from the space of possible peptide sequences through a terminated Markov Chain Monte Carlo (MCMC) algorithm. Initially, an ensemble of trial solutions is constructed by sampling from a prior distribution. The prior probability of a trial peptide is based on the natural abundances of its constituent amino acids and the preference for C-terminal residues, if appropriate, for a particular digestion reagent. In order to proceed, new trial sequences must be generated. We employ transition engines that change the state of an ensemble member, with a probability of transition, T, defined by: T (i j ) T (j i) = Prior(j ) Prior(i) (6) where i and j represent two possible solutions in the space. Transitions of this type will eventually converge on occupancies of states which match the prior distribution. The various transition schemes employed by the de novo algorithm are outlined in Table 2. The data must be introduced via the likelihood function (ProbSeq in this case), as we wish to progress from the prior to the posterior. The transition probabilities are therefore combined with acceptance probabilities, A, where: A(i j )T (i j ) Prior(j )Likelihood(j ) = A(j i)t (j i) Prior(i)Likelihood(i) = Posterior(j ) (7) Posterior(i) so that: A(i j ) A(j i) = Likelihood(j ) Likelihood(i) (8) Table 2. The MCMC transitions for peptide sequencing: reversal of a contiguous subsequence with randomly chosen end-points, rotation of a contiguous subsequence with randomly chosen end-points, permutation of a contiguous subsequence with randomly chosen end-points, replacement of a contiguous subsequence with randomly chosen end-points, exchange of the C-terminus and N-terminus ends of two sequences to preserve nominal mass. The last transition is an example of a genetic algorithm Type Before After Reversal XXXARQEIKXXX XXXKIEQRAXXX Rotation XXXARQEIKXXX XXXQEIKARXXX Permutation XXXARQEIKXXX XXXIQRKAEXXX Replacement XXXNEQXXX XXXEKGGXXX Exchange EKGG-DQCYKR, NEH-YKDQCR NEH-DQCYKR, EKGG-YKDQCR This is the usual Metropolis Hastings approach of detailed balancing (Metropolis et al., 1953; Hastings, 1970). In order to improve convergence, the likelihood is introduced in a modified form, by raising it to some power 0 λ 1. Here, λ is analogous to an inverse temperature and by raising it slowly from 0 to 1 (i.e. by slow cooling), we implement simulated annealing (Kirkpatrick et al., 1983). We use simulated annealing for both de novo exploration and exploring the ProbSeq parameter space, (see Table 3 for an outline of the commonalities). The result of a de novo exploration is a number of candidate peptide sequences that may account for the fragmentation spectrum and precursor mass, accompanied by a posterior probability. In order to assess whether the results of tuning the ProbSeq likelihood parameters afford greater discrimination in favour of the correct sequences, we can perform de novo searches and inspect the ranks and posterior probabilities of the correct sequences. Indeed, Table 3. Comparison between tuning and sequencing, which are both inference problems that can be explored using MCMC methods Probseq tuning De novo sequencing Likelihood Pr(Spectrum ) Pr(Spectrum Sequence) Space [0, 1] n -unit hypercube Discrete space of sequences Prior Uniform Given by amino acid abundance
5 ProbSeq a fragmentation model for electrospray tandem MS data interpretation 65 we could have replaced equation (4) in favour of a likelihood for the tuning parameters which incorporated de novo searches in order to involve this discrimination directly in the tuning process. However, the exploration of the parameter space would have become prohibitively time-consuming. Results and Discussion Although cysteine and tryptophan are under-represented in the tuning dataset, the probabilities for immonium production allow us to make general comparisons with observations described in the literature. Papayannopoulos (1995) indicates that arginine, lysine, leucine/isoleucine, cysteine, histidine, phenylalanine, tyrosine and tryptophan may give diagnostically important immonium ions (see Table 4). Our results indicate that arginine, leucine/ isoleucine, cysteine, histidine, phenylalanine, tyrosine and tryptophan are likely to give stronger signals, although the strengths reported may be sensitive to the configuration of the instrument. The results of performing de novo searches on the tuning and validation datasets are summarized in Tables 5 and 6. There is a noticeable improvement in the results of de novo sequencing in the tuning dataset: the correct sequence is ranked first on eight occasions with the tuned prior as opposed to six with the original prior out of a total of 39 sequences. With the tuning dataset, 17 correct sequences appeared in the top 10 with the tuned prior, against 10 for the original prior. The improvement is less clear in the validation dataset but is still present, with 11 correct for the tuned prior and nine for the original prior out of 31 sequences; 17 correct sequences appeared in the top 10 for both the tuned and original priors. The improvements gained by tuning the prior probabilities of the ProbSeq model are slight. This is to be expected as, when accurately massmeasured data are available, the information in the data dominates the prior. The quality of our results is not critically dependent on specific parameter settings, which is reassuring. We may have gained some improvement for marginal data but this remains to be seen, perhaps through tuning and validation with a more extensive library of data. Overall, tuning has resulted in the softening of some probabilities, e.g. those for immonium Table 4. Listed for each amino acid are occurrences in the validated sequences throughout the tuning dataset, occurrences in different spectra, mean probability of immonium ion production and its standard deviation as sampled from the posterior distribution during the MCMC exploration Amino acid Occurrences Spectra Pr(Immonium) Standard deviation Alanine Arginine Asparagine Aspartic acid Cysteine Glutamine Glutamic acid Glycine Histidine (Iso)leucine Lysine Methionine Phenylalanine Proline Serine Threonine Tryptophan Tyrosine Valine Amino acids with immonium ions, described as diagnostically important. Leucine and Isoleucine were not considered to be distinguishable. The probabilities of immonium production were set to zero for alanine and glycine throughout the tuning process.
6 66 J. Skilling et al. Table 5. Result of tuning on tuning dataset Correct sequence Rank Probability (%) Top seq (untuned) Rank Probability (%) Top seq (tuned) SLGGEVFLDFTK SLNEVFDLFTK SLGGEVFDLFTK LPLVGGHEGAGVVVGMGENVK 374 <0.01 LPLVGGHEKRVVGMGENVK 56 <0.01 LPLVGGHEGAGVVVGFAVNVK ANGTTVLVGMPAGAK >999 <0.01 KNTTVLVGMKPAK ANGTTVLVGMPAGAK TPEVDDEALEKFDK 387 <0.01 TPEVQPPQASTHTDK >999 <0.01 TPEVKMTNSVTDADK VLVLDTDYKK 371 <0.01 LVQEGQAYTGQ >999 <0.01 LVQQGEAYASQ TKLPAVFK >999 <0.01 LLLYLLR >999 <0.01 LRFRRR TPEVDDEALEK >999 <0.01 dpttwppeek >999 <0.01 TPEPPPMMAFK SLAMAASDLSLLDAQSAPLR >999 <0.01 SLAMAASDLSLLDALGGVLGR 174 <0.01 SLTTAASDLSLLDAKSALPR VLVLDTDYK 253 <0.01 VLDELWYK >999 <0.01 LVEDLWYK TPEVDDEALEKFDK >999 <0.01 TPSFEPASETEVVDK >999 <0.01 SLGMNPSFGGEVPTDK VAGTWYSLAMAASDLSLLDAQSAPLR >999 <0.01 LPTGRSLLRRGKSDLSLNGLGLNNR >999 <0.01 RWPLRRWSKRDLSLDGTQALPR LDALNENK >999 <0.01 QPTWEQK >999 <0.01 QPTWEGAK LSFNPTQLEEQCHL >999 <0.01 FNRTHDKQGLPdGK >999 <0.01 EFALPTRNHMTWR SLAMAASDLSLLDAQSAPLR >999 <0.01 LSMGRAMLSLLDAKSALPR >999 <0.01 SLNWDLKLLSSDDSALPR ALPMHLR LAPMHLR ALPMHLR TPEVDDEALEKFDK TPEVDDEALEKFDK TPEVDDEALEKFDK TPEVDDEALEKFDK >999 <0.01 TPEVDDEAKLEFDK TPEVDDEAKLEFDK VLVLDTDYKK 499 <0.01 VLVLdGLKYK VLVLPAYKYK SLAMAASDLSLLDAQSAPLR >999 <0.01 SLAASLKADLLSDLSAADLR SLAMAASLDSLLDAQSAPLR TKLPAVFK KTLPAVFK KTLPAVFK TPEVDDEALEK TPEVDDEALEK TPEVDDEALEK VLVLDTDYK VLVLPSFYK VLVLPSFYK VYVEELKPTPEGDLELLLQK VYVEELQPTPWPFLLLLAGK >999 <0.01 VYVEELQPTPWPFLLLLQK VAGTWYSLAMAASDLSLLDAQSAPLR >999 <0.01 LLLRYLRRRLLANYKRELYGK >999 <0.01 DSDLdDDLQFRFLRGLKLLLLR LDALNENK LDALNENK LDALNENK VVGLSTLPELYEK VVGLSTLPELYEK VVGLSTLPELYEK EKDLVGAVLK 443 <0.01 GLSPMVKVLK 378 <0.01 GLSPMVKVLK EALDFFAR EALDFFAR EALDFFAR YVVDTSK >999 <0.01 YPTDTSK YPTDTSK GVLFYESHGK GVLFYESHGK GVLFYESHGK ANELLLNVK >999 <0.01 GQELLLNVK NAELLLNVK SLSLVGSYVGNR SLSLVGSYVAAGK LSSLVGSYVGNR VLGLDGGEGKEELFR >999 <0.01 VLGLDRGVVdVQFR VLGLDGRVdVVQFR VLGLDGGEGKEELFR >999 <0.01 VLGLDMVLMLdALLG >999 <0.01 VLGLDMVLEYLALGL ATDGGAHGVLNVSVSEAALEASTR >999 <0.01 ESAAVHVAAKGASVSEAALEASTR >999 <0.01 KFSGMKLAASASVSEAALEKMR LPLVGGHEGAGVVVGMGENVK >999 <0.01 LPLVHGGEKGVVVGMGEGRK >999 <0.01 LPLVGGHEKGVVVGMGEGRK DLPVPKPK DLPVKPPK DLPVKPPK LGDYAGLK 89 <0.01 YATQLLK 45 <0.01 AYTGALLK ATDGGAHGVLNVSVSEAALEASTR >999 <0.01 AGVGGMHGVLRGVKLLMAMKMR >999 <0.01 VVdLHGVLHLFRLMAMMRK d, denotes Carboxyamidomethyl cysteine.
7 ProbSeq a fragmentation model for electrospray tandem MS data interpretation 67 Table 6. Result of tuning on validation dataset Correct sequence Rank Probability (%) Top seq (untuned) Rank Probability (%) Top seq (tuned) GAAQNLLPASTGAAK GAAQNLLQFALSH AGAQNLLKFALSH LVLNGHALTLFQER LVLNGHALTLFAGER LVLNGHALTLFQER VVDLMVHMASKE VVPMMVHMASKE 76 <0.01 VVPMMVHMASEK LVLNGHALTLFQERDPSNLK 335 <0.01 LVLNGVGVHGNEWHFKGdLK 0 <0.01 LVLRGMFELMEQLFHdLK VLLSAPSADAPMFVMGVNHEK 0 <0.01 VLLSAPSADAPMEGWWPRAGAGHDK 0 <0.01 LVLSAPSADAPMFVYDYHEAASHK YDDLKR YDDLKR YDDLKR DAGAGLALNDHFVK 0 <0.01 GEQVAALQNGESPK ADGAGLALGGNAESPK YDSTHGHFK YDSTHGHFK YDSTHGHFK VLLSAPSADAPMFVMGVNHEK 0 <0.01 APSDALQNGAQEDVFADAHGVT VLLSAPASDAPMFVMGVNHEK DAGAGLALNDHFVK ADGAGLALNDHFVK ADGAGLALNDHFVK LVLNGHALTLFQERDPSNLK 0 <0.01 LVLNGHALdLdLDLLLGVGGR 0 <0.01 LVLNGHALQWGWQRLGKdK VLHDNFGLVEGLMTTVHALTATQK 0 <0.01 DLAVNNGVLVEGLMTTVHALFDLGP 0 <0.01 EVGGNNLVLVEGLMTTVHALTHYK LEKPAKYDDLK 0 <0.01 LEKTGdKELLK 0 <0.01 YFVPPNTQLLK VVDLMVHMASKE VVPMMVHMASKE VVDLMVHMASKE APLLAVTR APLLAVTR APLLAVTR VNLAMNVGK VNLAMNVGK VNLAMNVGK LDLDSAPLTAR LDLDSAPLTAR LDLDSAPLTAR RFDELLEASDGLMVAR >999 <0.01 FDRELLEASDGLMVAR QNSAFLLEASDGLMVAR GVNLPGAAVDLPAVSEK >999 <0.01 GVNLPQAVDLFGLFF >999 <0.01 GVNLPQAVDLLWRR GDLGLELPAEK GLDGLELPAEK GLDGLELPAEK LTLDNAYMEK LTLDNAYMEK LTLDNAYMEK MQHLLAR MKHLLAR MKHLLAR VYVDDGLLSLQVK VYVDDAVLSLQVK VYVDPGMLSLQVK KLFEELAR >999 <0.01 KLFNGSLAR >999 <0.01 KLFNELNK GSGTAEVELK GSTGAEVELK GSGTAPMELK LFEELAR LFEELAR LFEELAR RPVAVALDTK RPVAVALDTK RPVAVALDTK FDELLEASDGLMVAR DFELLEAMAGLMGLR DFELLEAMAAVMGLR GDYPLEAVR GDYPLEAVR GDYPLEAVR FGVEQDVDMVFASF >999 <0.01 VGDQGDFQSSVFASF >999 <0.01 GVTNPTMFGGGTFASF MNFSHGTHEYHAETLK >999 <0.01 MNEHVDADQTFAQMHK 505 <0.01 MNMVLHDAGAHGDQFMK
8 68 J. Skilling et al. production, and confirmation for others in the original set. However, the results are not markedly different and we conclude that a single set of reasonable parameters will suffice to give good results over a range of instruments and the details of their configurations. We have put in place a flexible, automated scheme for tuning the prior used in the calculation of the likelihood Pr(Spectrum Sequence), used in both de novo sequencing and databank searching applications, to which new data can easily be added and the fragmentation characteristics observed from new instruments and experiments accommodated. References 1. Hastings WK Biometrika 57: Kirkpatrick S, Gelatt CD, Vecchi MP Science 220: Morris HR, Paxton T, Dell A et al Rapid Commun Mass Spectrom 10: Metropolis N, Rosenbluth AW, Rosenbluth MN et al J Chem Phys 21: Papayannopoulos IA Mass Spectrom Rev 14(1): Skilling J US Patent No Skilling J US Patent No Skilling J JMicrosc190: Acknowledgements KR would like to thank the organizers of the MIPNETS meeting held in Liverpool, UK, in June 2003 for providing the opportunity to present and discuss this work.
Proteomics. November 13, 2007
 Proteomics November 13, 2007 Acknowledgement Slides presented here have been borrowed from presentations by : Dr. Mark A. Knepper (LKEM, NHLBI, NIH) Dr. Nathan Edwards (Center for Bioinformatics and Computational
Proteomics November 13, 2007 Acknowledgement Slides presented here have been borrowed from presentations by : Dr. Mark A. Knepper (LKEM, NHLBI, NIH) Dr. Nathan Edwards (Center for Bioinformatics and Computational
Protein Identification Using Tandem Mass Spectrometry. Nathan Edwards Informatics Research Applied Biosystems
 Protein Identification Using Tandem Mass Spectrometry Nathan Edwards Informatics Research Applied Biosystems Outline Proteomics context Tandem mass spectrometry Peptide fragmentation Peptide identification
Protein Identification Using Tandem Mass Spectrometry Nathan Edwards Informatics Research Applied Biosystems Outline Proteomics context Tandem mass spectrometry Peptide fragmentation Peptide identification
Proteome Informatics. Brian C. Searle Creative Commons Attribution
 Proteome Informatics Brian C. Searle searleb@uw.edu Creative Commons Attribution Section structure Class 1 Class 2 Homework 1 Mass spectrometry and de novo sequencing Database searching and E-value estimation
Proteome Informatics Brian C. Searle searleb@uw.edu Creative Commons Attribution Section structure Class 1 Class 2 Homework 1 Mass spectrometry and de novo sequencing Database searching and E-value estimation
INTRODUCTION. Amino acids occurring in nature have the general structure shown below:
 Biochemistry I Laboratory Amino Acid Thin Layer Chromatography INTRODUCTION The primary importance of amino acids in cell structure and metabolism lies in the fact that they serve as building blocks for
Biochemistry I Laboratory Amino Acid Thin Layer Chromatography INTRODUCTION The primary importance of amino acids in cell structure and metabolism lies in the fact that they serve as building blocks for
Lecture 15: Realities of Genome Assembly Protein Sequencing
 Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
Sequence comparison: Score matrices. Genome 559: Introduction to Statistical and Computational Genomics Prof. James H. Thomas
 Sequence comparison: Score matrices Genome 559: Introduction to Statistical and omputational Genomics Prof James H Thomas Informal inductive proof of best alignment path onsider the last step in the best
Sequence comparison: Score matrices Genome 559: Introduction to Statistical and omputational Genomics Prof James H Thomas Informal inductive proof of best alignment path onsider the last step in the best
Sequence comparison: Score matrices. Genome 559: Introduction to Statistical and Computational Genomics Prof. James H. Thomas
 Sequence comparison: Score matrices Genome 559: Introduction to Statistical and omputational Genomics Prof James H Thomas FYI - informal inductive proof of best alignment path onsider the last step in
Sequence comparison: Score matrices Genome 559: Introduction to Statistical and omputational Genomics Prof James H Thomas FYI - informal inductive proof of best alignment path onsider the last step in
Sequence comparison: Score matrices
 Sequence comparison: Score matrices http://facultywashingtonedu/jht/gs559_2013/ Genome 559: Introduction to Statistical and omputational Genomics Prof James H Thomas FYI - informal inductive proof of best
Sequence comparison: Score matrices http://facultywashingtonedu/jht/gs559_2013/ Genome 559: Introduction to Statistical and omputational Genomics Prof James H Thomas FYI - informal inductive proof of best
PROTEIN STRUCTURE AMINO ACIDS H R. Zwitterion (dipolar ion) CO 2 H. PEPTIDES Formal reactions showing formation of peptide bond by dehydration:
 PTEI STUTUE ydrolysis of proteins with aqueous acid or base yields a mixture of free amino acids. Each type of protein yields a characteristic mixture of the ~ 20 amino acids. AMI AIDS Zwitterion (dipolar
PTEI STUTUE ydrolysis of proteins with aqueous acid or base yields a mixture of free amino acids. Each type of protein yields a characteristic mixture of the ~ 20 amino acids. AMI AIDS Zwitterion (dipolar
Proteins: Characteristics and Properties of Amino Acids
 SBI4U:Biochemistry Macromolecules Eachaminoacidhasatleastoneamineandoneacidfunctionalgroupasthe nameimplies.thedifferentpropertiesresultfromvariationsinthestructuresof differentrgroups.thergroupisoftenreferredtoastheaminoacidsidechain.
SBI4U:Biochemistry Macromolecules Eachaminoacidhasatleastoneamineandoneacidfunctionalgroupasthe nameimplies.thedifferentpropertiesresultfromvariationsinthestructuresof differentrgroups.thergroupisoftenreferredtoastheaminoacidsidechain.
Collision Cross Section: Ideal elastic hard sphere collision:
 Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
BENG 183 Trey Ideker. Protein Sequencing
 BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
A rapid and highly selective colorimetric method for direct detection of tryptophan in proteins via DMSO acceleration
 A rapid and highly selective colorimetric method for direct detection of tryptophan in proteins via DMSO acceleration Yanyan Huang, Shaoxiang Xiong, Guoquan Liu, Rui Zhao Beijing National Laboratory for
A rapid and highly selective colorimetric method for direct detection of tryptophan in proteins via DMSO acceleration Yanyan Huang, Shaoxiang Xiong, Guoquan Liu, Rui Zhao Beijing National Laboratory for
Lecture 14 - Cells. Astronomy Winter Lecture 14 Cells: The Building Blocks of Life
 Lecture 14 Cells: The Building Blocks of Life Astronomy 141 Winter 2012 This lecture describes Cells, the basic structural units of all life on Earth. Basic components of cells: carbohydrates, lipids,
Lecture 14 Cells: The Building Blocks of Life Astronomy 141 Winter 2012 This lecture describes Cells, the basic structural units of all life on Earth. Basic components of cells: carbohydrates, lipids,
(Bio)chemical Proteomics. Alex Kentsis October, 2013
 (Bio)chemical Proteomics Alex Kentsis October, 2013 http://alexkentsis.net A brief history of chemical proteomics 1907: Eduard Buchner, demonstration of cell-free alcohol fermentation (i.e. enzymes) 1946:
(Bio)chemical Proteomics Alex Kentsis October, 2013 http://alexkentsis.net A brief history of chemical proteomics 1907: Eduard Buchner, demonstration of cell-free alcohol fermentation (i.e. enzymes) 1946:
Computational Methods for Mass Spectrometry Proteomics
 Computational Methods for Mass Spectrometry Proteomics Eidhammer, Ingvar ISBN-13: 9780470512975 Table of Contents Preface. Acknowledgements. 1 Protein, Proteome, and Proteomics. 1.1 Primary goals for studying
Computational Methods for Mass Spectrometry Proteomics Eidhammer, Ingvar ISBN-13: 9780470512975 Table of Contents Preface. Acknowledgements. 1 Protein, Proteome, and Proteomics. 1.1 Primary goals for studying
Discussion Section (Day, Time): TF:
 ame: Chemistry 27 Professor Gavin MacBeath arvard University Spring 2004 Final Exam Thursday, May 28, 2004 2:15 PM - 5:15 PM Discussion Section (Day, Time): Directions: TF: 1. Do not write in red ink.
ame: Chemistry 27 Professor Gavin MacBeath arvard University Spring 2004 Final Exam Thursday, May 28, 2004 2:15 PM - 5:15 PM Discussion Section (Day, Time): Directions: TF: 1. Do not write in red ink.
All Proteins Have a Basic Molecular Formula
 All Proteins Have a Basic Molecular Formula Homa Torabizadeh Abstract This study proposes a basic molecular formula for all proteins. A total of 10,739 proteins belonging to 9 different protein groups
All Proteins Have a Basic Molecular Formula Homa Torabizadeh Abstract This study proposes a basic molecular formula for all proteins. A total of 10,739 proteins belonging to 9 different protein groups
Atomic masses. Atomic masses of elements. Atomic masses of isotopes. Nominal and exact atomic masses. Example: CO, N 2 ja C 2 H 4
 High-Resolution Mass spectrometry (HR-MS, HRAM-MS) (FT mass spectrometry) MS that enables identifying elemental compositions (empirical formulas) from accurate m/z data 9.05.2017 1 Atomic masses (atomic
High-Resolution Mass spectrometry (HR-MS, HRAM-MS) (FT mass spectrometry) MS that enables identifying elemental compositions (empirical formulas) from accurate m/z data 9.05.2017 1 Atomic masses (atomic
Range of Certified Values in Reference Materials. Range of Expanded Uncertainties as Disseminated. NMI Service
 Calibration and Capabilities Amount of substance,, Russian Federation (Ural Scientific and Research Institiute Metrology, Rosstandart) (D.I. Mendeleyev Institute Metrology, Rosstandart) The uncertainty
Calibration and Capabilities Amount of substance,, Russian Federation (Ural Scientific and Research Institiute Metrology, Rosstandart) (D.I. Mendeleyev Institute Metrology, Rosstandart) The uncertainty
Protein Secondary Structure Prediction
 part of Bioinformatik von RNA- und Proteinstrukturen Computational EvoDevo University Leipzig Leipzig, SS 2011 the goal is the prediction of the secondary structure conformation which is local each amino
part of Bioinformatik von RNA- und Proteinstrukturen Computational EvoDevo University Leipzig Leipzig, SS 2011 the goal is the prediction of the secondary structure conformation which is local each amino
Protein Structure Bioinformatics Introduction
 1 Swiss Institute of Bioinformatics Protein Structure Bioinformatics Introduction Basel, 27. September 2004 Torsten Schwede Biozentrum - Universität Basel Swiss Institute of Bioinformatics Klingelbergstr
1 Swiss Institute of Bioinformatics Protein Structure Bioinformatics Introduction Basel, 27. September 2004 Torsten Schwede Biozentrum - Universität Basel Swiss Institute of Bioinformatics Klingelbergstr
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I Dr. Glen B Legge
 EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
Mass Spectrometry Based De Novo Peptide Sequencing Error Correction
 Mass Spectrometry Based De Novo Peptide Sequencing Error Correction by Chenyu Yao A thesis presented to the University of Waterloo in fulfillment of the thesis requirement for the degree of Master of Mathematics
Mass Spectrometry Based De Novo Peptide Sequencing Error Correction by Chenyu Yao A thesis presented to the University of Waterloo in fulfillment of the thesis requirement for the degree of Master of Mathematics
A Logic-Based Approach to Polymer Sequence Analysis
 A Logic-Based Approach to Polymer Sequence Analysis Renato Bruni Dep. of Electronic and Information Engineering, University of Perugia, Via G. Duranti, 93-06125 Perugia - Italy. E-mail: renato.bruni@diei.unipg.it
A Logic-Based Approach to Polymer Sequence Analysis Renato Bruni Dep. of Electronic and Information Engineering, University of Perugia, Via G. Duranti, 93-06125 Perugia - Italy. E-mail: renato.bruni@diei.unipg.it
LC-MS Based Metabolomics
 LC-MS Based Metabolomics Analysing the METABOLOME 1. Metabolite Extraction 2. Metabolite detection (with or without separation) 3. Data analysis Metabolite Detection GC-MS: Naturally volatile or made volatile
LC-MS Based Metabolomics Analysing the METABOLOME 1. Metabolite Extraction 2. Metabolite detection (with or without separation) 3. Data analysis Metabolite Detection GC-MS: Naturally volatile or made volatile
1. Amino Acids and Peptides Structures and Properties
 1. Amino Acids and Peptides Structures and Properties Chemical nature of amino acids The!-amino acids in peptides and proteins (excluding proline) consist of a carboxylic acid ( COOH) and an amino ( NH
1. Amino Acids and Peptides Structures and Properties Chemical nature of amino acids The!-amino acids in peptides and proteins (excluding proline) consist of a carboxylic acid ( COOH) and an amino ( NH
Identification of Human Hemoglobin Protein Variants Using Electrospray Ionization-Electron Transfer Dissociation Mass Spectrometry
 Identification of Human Hemoglobin Protein Variants Using Electrospray Ionization-Electron Transfer Dissociation Mass Spectrometry Jonathan Williams Waters Corporation, Milford, MA, USA A P P L I C AT
Identification of Human Hemoglobin Protein Variants Using Electrospray Ionization-Electron Transfer Dissociation Mass Spectrometry Jonathan Williams Waters Corporation, Milford, MA, USA A P P L I C AT
A Plausible Model Correlates Prebiotic Peptide Synthesis with. Primordial Genetic Code
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 A Plausible Model Correlates Prebiotic Peptide Synthesis with Primordial Genetic Code Jianxi Ying,
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 A Plausible Model Correlates Prebiotic Peptide Synthesis with Primordial Genetic Code Jianxi Ying,
CHEMISTRY ATAR COURSE DATA BOOKLET
 CHEMISTRY ATAR COURSE DATA BOOKLET 2018 2018/2457 Chemistry ATAR Course Data Booklet 2018 Table of contents Periodic table of the elements...3 Formulae...4 Units...4 Constants...4 Solubility rules for
CHEMISTRY ATAR COURSE DATA BOOKLET 2018 2018/2457 Chemistry ATAR Course Data Booklet 2018 Table of contents Periodic table of the elements...3 Formulae...4 Units...4 Constants...4 Solubility rules for
MS/MS of Peptides Manual Sequencing of Protonated Peptides
 S/S of Peptides anual Sequencing of Protonated Peptides Árpád Somogyi Associate irector CCIC, ass Spectrometry and Proteomics Laboratory SU July 11, 2018 Peptides Product Ion Scan Product ion spectra contain
S/S of Peptides anual Sequencing of Protonated Peptides Árpád Somogyi Associate irector CCIC, ass Spectrometry and Proteomics Laboratory SU July 11, 2018 Peptides Product Ion Scan Product ion spectra contain
Discussion Section (Day, Time):
 Chemistry 27 Spring 2005 Exam 1 Chemistry 27 Professor Gavin MacBeath arvard University Spring 2005 our Exam 1 Friday, February 25, 2005 11:07 AM 12:00 PM Discussion Section (Day, Time): TF: Directions:
Chemistry 27 Spring 2005 Exam 1 Chemistry 27 Professor Gavin MacBeath arvard University Spring 2005 our Exam 1 Friday, February 25, 2005 11:07 AM 12:00 PM Discussion Section (Day, Time): TF: Directions:
Periodic Table. 8/3/2006 MEDC 501 Fall
 Periodic Table 8/3/2006 MEDC 501 Fall 2006 1 rbitals Shapes of rbitals s - orbital p -orbital 8/3/2006 MEDC 501 Fall 2006 2 Ionic Bond - acl Electronic Structure 11 a :: 1s 2 2s 2 2p x2 2p y2 2p z2 3s
Periodic Table 8/3/2006 MEDC 501 Fall 2006 1 rbitals Shapes of rbitals s - orbital p -orbital 8/3/2006 MEDC 501 Fall 2006 2 Ionic Bond - acl Electronic Structure 11 a :: 1s 2 2s 2 2p x2 2p y2 2p z2 3s
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor
 LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
Part 4 The Select Command and Boolean Operators
 Part 4 The Select Command and Boolean Operators http://cbm.msoe.edu/newwebsite/learntomodel Introduction By default, every command you enter into the Console affects the entire molecular structure. However,
Part 4 The Select Command and Boolean Operators http://cbm.msoe.edu/newwebsite/learntomodel Introduction By default, every command you enter into the Console affects the entire molecular structure. However,
CHAPTER 29 HW: AMINO ACIDS + PROTEINS
 CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
Enzyme Catalysis & Biotechnology
 L28-1 Enzyme Catalysis & Biotechnology Bovine Pancreatic RNase A Biochemistry, Life, and all that L28-2 A brief word about biochemistry traditionally, chemical engineers used organic and inorganic chemistry
L28-1 Enzyme Catalysis & Biotechnology Bovine Pancreatic RNase A Biochemistry, Life, and all that L28-2 A brief word about biochemistry traditionally, chemical engineers used organic and inorganic chemistry
C h a p t e r 2 A n a l y s i s o f s o m e S e q u e n c e... methods use different attributes related to mis sense mutations such as
 C h a p t e r 2 A n a l y s i s o f s o m e S e q u e n c e... 2.1Introduction smentionedinchapter1,severalmethodsareavailabletoclassifyhuman missensemutationsintoeitherbenignorpathogeniccategoriesandthese
C h a p t e r 2 A n a l y s i s o f s o m e S e q u e n c e... 2.1Introduction smentionedinchapter1,severalmethodsareavailabletoclassifyhuman missensemutationsintoeitherbenignorpathogeniccategoriesandthese
The Select Command and Boolean Operators
 The Select Command and Boolean Operators Part of the Jmol Training Guide from the MSOE Center for BioMolecular Modeling Interactive version available at http://cbm.msoe.edu/teachingresources/jmol/jmoltraining/boolean.html
The Select Command and Boolean Operators Part of the Jmol Training Guide from the MSOE Center for BioMolecular Modeling Interactive version available at http://cbm.msoe.edu/teachingresources/jmol/jmoltraining/boolean.html
PROTEIN SECONDARY STRUCTURE PREDICTION: AN APPLICATION OF CHOU-FASMAN ALGORITHM IN A HYPOTHETICAL PROTEIN OF SARS VIRUS
 Int. J. LifeSc. Bt & Pharm. Res. 2012 Kaladhar, 2012 Research Paper ISSN 2250-3137 www.ijlbpr.com Vol.1, Issue. 1, January 2012 2012 IJLBPR. All Rights Reserved PROTEIN SECONDARY STRUCTURE PREDICTION:
Int. J. LifeSc. Bt & Pharm. Res. 2012 Kaladhar, 2012 Research Paper ISSN 2250-3137 www.ijlbpr.com Vol.1, Issue. 1, January 2012 2012 IJLBPR. All Rights Reserved PROTEIN SECONDARY STRUCTURE PREDICTION:
Module No. 31: Peptide Synthesis: Definition, Methodology & applications
 PAPER 9: TECHNIQUES USED IN MOLECULAR BIOPHYSICS I Module No. 31: Peptide Synthesis: Definition, Methodology & applications Objectives: 1. Introduction 2. Synthesis of peptide 2.1. N-terminal protected
PAPER 9: TECHNIQUES USED IN MOLECULAR BIOPHYSICS I Module No. 31: Peptide Synthesis: Definition, Methodology & applications Objectives: 1. Introduction 2. Synthesis of peptide 2.1. N-terminal protected
Viewing and Analyzing Proteins, Ligands and their Complexes 2
 2 Viewing and Analyzing Proteins, Ligands and their Complexes 2 Overview Viewing the accessible surface Analyzing the properties of proteins containing thousands of atoms is best accomplished by representing
2 Viewing and Analyzing Proteins, Ligands and their Complexes 2 Overview Viewing the accessible surface Analyzing the properties of proteins containing thousands of atoms is best accomplished by representing
Principles of Biochemistry
 Principles of Biochemistry Fourth Edition Donald Voet Judith G. Voet Charlotte W. Pratt Chapter 4 Amino Acids: The Building Blocks of proteins (Page 76-90) Chapter Contents 1- Amino acids Structure: 2-
Principles of Biochemistry Fourth Edition Donald Voet Judith G. Voet Charlotte W. Pratt Chapter 4 Amino Acids: The Building Blocks of proteins (Page 76-90) Chapter Contents 1- Amino acids Structure: 2-
Protein structure. Protein structure. Amino acid residue. Cell communication channel. Bioinformatics Methods
 Cell communication channel Bioinformatics Methods Iosif Vaisman Email: ivaisman@gmu.edu SEQUENCE STRUCTURE DNA Sequence Protein Sequence Protein Structure Protein structure ATGAAATTTGGAAACTTCCTTCTCACTTATCAGCCACCT...
Cell communication channel Bioinformatics Methods Iosif Vaisman Email: ivaisman@gmu.edu SEQUENCE STRUCTURE DNA Sequence Protein Sequence Protein Structure Protein structure ATGAAATTTGGAAACTTCCTTCTCACTTATCAGCCACCT...
Chapter 5. Complexation of Tholins by 18-crown-6:
 5-1 Chapter 5. Complexation of Tholins by 18-crown-6: Identification of Primary Amines 5.1. Introduction Electrospray ionization (ESI) is an excellent technique for the ionization of complex mixtures,
5-1 Chapter 5. Complexation of Tholins by 18-crown-6: Identification of Primary Amines 5.1. Introduction Electrospray ionization (ESI) is an excellent technique for the ionization of complex mixtures,
Studies Leading to the Development of a Highly Selective. Colorimetric and Fluorescent Chemosensor for Lysine
 Supporting Information for Studies Leading to the Development of a Highly Selective Colorimetric and Fluorescent Chemosensor for Lysine Ying Zhou, a Jiyeon Won, c Jin Yong Lee, c * and Juyoung Yoon a,
Supporting Information for Studies Leading to the Development of a Highly Selective Colorimetric and Fluorescent Chemosensor for Lysine Ying Zhou, a Jiyeon Won, c Jin Yong Lee, c * and Juyoung Yoon a,
Amino Acids and Peptides
 Amino Acids Amino Acids and Peptides Amino acid a compound that contains both an amino group and a carboxyl group α-amino acid an amino acid in which the amino group is on the carbon adjacent to the carboxyl
Amino Acids Amino Acids and Peptides Amino acid a compound that contains both an amino group and a carboxyl group α-amino acid an amino acid in which the amino group is on the carbon adjacent to the carboxyl
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
A study of matrix energy during peptide formation through chemical graphs
 International Journal of Mathematics and Soft Computing Vol.3, No.2 (2013), 11-15. ISSN Print : 2249-3328 ISSN Online: 2319-5215 A study of matrix energy during peptide formation through chemical graphs
International Journal of Mathematics and Soft Computing Vol.3, No.2 (2013), 11-15. ISSN Print : 2249-3328 ISSN Online: 2319-5215 A study of matrix energy during peptide formation through chemical graphs
Separation of Large and Small Peptides by Supercritical Fluid Chromatography and Detection by Mass Spectrometry
 Separation of Large and Small Peptides by Supercritical Fluid Chromatography and Detection by Mass Spectrometry Application Note Biologics and Biosimilars Author Edgar Naegele Agilent Technologies, Inc.
Separation of Large and Small Peptides by Supercritical Fluid Chromatography and Detection by Mass Spectrometry Application Note Biologics and Biosimilars Author Edgar Naegele Agilent Technologies, Inc.
Hypergraphs, Metabolic Networks, Bioreaction Systems. G. Bastin
 Hypergraphs, Metabolic Networks, Bioreaction Systems. G. Bastin PART 1 : Metabolic flux analysis and minimal bioreaction modelling PART 2 : Dynamic metabolic flux analysis of underdetermined networks 2
Hypergraphs, Metabolic Networks, Bioreaction Systems. G. Bastin PART 1 : Metabolic flux analysis and minimal bioreaction modelling PART 2 : Dynamic metabolic flux analysis of underdetermined networks 2
Chromatography 1 of 26 Boardworks Ltd 2016
 Chromatography 1 of 26 Boardworks Ltd 2016 Chromatography 2 of 26 Boardworks Ltd 2016 What is chromatography? 3 of 26 Boardworks Ltd 2016 Different instrumental methods can be used to analyse and identify
Chromatography 1 of 26 Boardworks Ltd 2016 Chromatography 2 of 26 Boardworks Ltd 2016 What is chromatography? 3 of 26 Boardworks Ltd 2016 Different instrumental methods can be used to analyse and identify
via Tandem Mass Spectrometry and Propositional Satisfiability De Novo Peptide Sequencing Renato Bruni University of Perugia
 De Novo Peptide Sequencing via Tandem Mass Spectrometry and Propositional Satisfiability Renato Bruni bruni@diei.unipg.it or bruni@dis.uniroma1.it University of Perugia I FIMA International Conference
De Novo Peptide Sequencing via Tandem Mass Spectrometry and Propositional Satisfiability Renato Bruni bruni@diei.unipg.it or bruni@dis.uniroma1.it University of Perugia I FIMA International Conference
How did they form? Exploring Meteorite Mysteries
 Exploring Meteorite Mysteries Objectives Students will: recognize that carbonaceous chondrite meteorites contain amino acids, the first step towards living plants and animals. conduct experiments that
Exploring Meteorite Mysteries Objectives Students will: recognize that carbonaceous chondrite meteorites contain amino acids, the first step towards living plants and animals. conduct experiments that
Accurate Mass Measurement for Intact Proteins using ESI-oa-TOF. Application Note. Donghui Yi and Christine Miller Agilent Technologies
 Accurate Mass Measurement for Intact Proteins using ESI-oa-TOF Application Note Donghui Yi and Christine Miller Jon D. Williams, GlaxoSmithKline Introduction Mass spectrometry (MS) has become a core technology
Accurate Mass Measurement for Intact Proteins using ESI-oa-TOF Application Note Donghui Yi and Christine Miller Jon D. Williams, GlaxoSmithKline Introduction Mass spectrometry (MS) has become a core technology
The Calculation of Physical Properties of Amino Acids Using Molecular Modeling Techniques (II)
 1046 Bull. Korean Chem. Soc. 2004, Vol. 25, No. 7 Myung-Jae Lee and Ui-Rak Kim The Calculation of Physical Properties of Amino Acids Using Molecular Modeling Techniques (II) Myung-Jae Lee and Ui-Rak Kim
1046 Bull. Korean Chem. Soc. 2004, Vol. 25, No. 7 Myung-Jae Lee and Ui-Rak Kim The Calculation of Physical Properties of Amino Acids Using Molecular Modeling Techniques (II) Myung-Jae Lee and Ui-Rak Kim
12/6/12. Dr. Sanjeeva Srivastava IIT Bombay. Primary Structure. Secondary Structure. Tertiary Structure. Quaternary Structure.
 Dr. anjeeva rivastava Primary tructure econdary tructure Tertiary tructure Quaternary tructure Amino acid residues α Helix Polypeptide chain Assembled subunits 2 1 Amino acid sequence determines 3-D structure
Dr. anjeeva rivastava Primary tructure econdary tructure Tertiary tructure Quaternary tructure Amino acid residues α Helix Polypeptide chain Assembled subunits 2 1 Amino acid sequence determines 3-D structure
NPTEL VIDEO COURSE PROTEOMICS PROF. SANJEEVA SRIVASTAVA
 LECTURE-25 Quantitative proteomics: itraq and TMT TRANSCRIPT Welcome to the proteomics course. Today we will talk about quantitative proteomics and discuss about itraq and TMT techniques. The quantitative
LECTURE-25 Quantitative proteomics: itraq and TMT TRANSCRIPT Welcome to the proteomics course. Today we will talk about quantitative proteomics and discuss about itraq and TMT techniques. The quantitative
Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell
 Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell Mathematics and Biochemistry University of Wisconsin - Madison 0 There Are Many Kinds Of Proteins The word protein comes
Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell Mathematics and Biochemistry University of Wisconsin - Madison 0 There Are Many Kinds Of Proteins The word protein comes
Translation. A ribosome, mrna, and trna.
 Translation The basic processes of translation are conserved among prokaryotes and eukaryotes. Prokaryotic Translation A ribosome, mrna, and trna. In the initiation of translation in prokaryotes, the Shine-Dalgarno
Translation The basic processes of translation are conserved among prokaryotes and eukaryotes. Prokaryotic Translation A ribosome, mrna, and trna. In the initiation of translation in prokaryotes, the Shine-Dalgarno
De Novo Peptide Identification Via Mixed-Integer Linear Optimization And Tandem Mass Spectrometry
 17 th European Symposium on Computer Aided Process Engineering ESCAPE17 V. Plesu and P.S. Agachi (Editors) 2007 Elsevier B.V. All rights reserved. 1 De Novo Peptide Identification Via Mixed-Integer Linear
17 th European Symposium on Computer Aided Process Engineering ESCAPE17 V. Plesu and P.S. Agachi (Editors) 2007 Elsevier B.V. All rights reserved. 1 De Novo Peptide Identification Via Mixed-Integer Linear
Figure S1. Interaction of PcTS with αsyn. (a) 1 H- 15 N HSQC NMR spectra of 100 µm αsyn in the absence (0:1, black) and increasing equivalent
 Figure S1. Interaction of PcTS with αsyn. (a) 1 H- 15 N HSQC NMR spectra of 100 µm αsyn in the absence (0:1, black) and increasing equivalent concentrations of PcTS (100 µm, blue; 500 µm, green; 1.5 mm,
Figure S1. Interaction of PcTS with αsyn. (a) 1 H- 15 N HSQC NMR spectra of 100 µm αsyn in the absence (0:1, black) and increasing equivalent concentrations of PcTS (100 µm, blue; 500 µm, green; 1.5 mm,
A Theoretical Inference of Protein Schemes from Amino Acid Sequences
 A Theoretical Inference of Protein Schemes from Amino Acid Sequences Angel Villahoz-Baleta angel_villahozbaleta@student.uml.edu ABSTRACT Proteins are based on tri-dimensional dispositions generated from
A Theoretical Inference of Protein Schemes from Amino Acid Sequences Angel Villahoz-Baleta angel_villahozbaleta@student.uml.edu ABSTRACT Proteins are based on tri-dimensional dispositions generated from
Problem Set 1
 2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
Chemistry Chapter 22
 hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
Effective Strategies for Improving Peptide Identification with Tandem Mass Spectrometry
 Effective Strategies for Improving Peptide Identification with Tandem Mass Spectrometry by Xi Han A thesis presented to the University of Waterloo in fulfillment of the thesis requirement for the degree
Effective Strategies for Improving Peptide Identification with Tandem Mass Spectrometry by Xi Han A thesis presented to the University of Waterloo in fulfillment of the thesis requirement for the degree
[ instrument specifications ]
![[ instrument specifications ] [ instrument specifications ]](/thumbs/95/122987442.jpg) Designed for leading researchers working at the limits of conventional mass spectrometry capabilities who need to further characterize and define their samples the Waters SYNAPT High Definition MS (HDMS
Designed for leading researchers working at the limits of conventional mass spectrometry capabilities who need to further characterize and define their samples the Waters SYNAPT High Definition MS (HDMS
1. Prepare the MALDI sample plate by spotting an angiotensin standard and the test sample(s).
 Analysis of a Peptide Sequence from a Proteolytic Digest by MALDI-TOF Post-Source Decay (PSD) and Collision-Induced Dissociation (CID) Standard Operating Procedure Purpose: The following procedure may
Analysis of a Peptide Sequence from a Proteolytic Digest by MALDI-TOF Post-Source Decay (PSD) and Collision-Induced Dissociation (CID) Standard Operating Procedure Purpose: The following procedure may
ZAHID IQBAL WARRAICH
 Q1 Chromatography is an important analytical technique in chemistry. There is a number of techniques under the general heading of chromatography. (a) Paper and gas chromatography rely on partition to separate
Q1 Chromatography is an important analytical technique in chemistry. There is a number of techniques under the general heading of chromatography. (a) Paper and gas chromatography rely on partition to separate
INTRODUCTION OVERVIEW
 COMPARISON OF CCS(N 2 ) MEASUREMENTS OBTAINED FROM TWO DIFFERENT T-WAVE ION S WITH DIRECT MEASUREMENTS USING A DRIFT TUBE ION Kevin Giles, Martin Palmer, Keith Richardson, Nick Tomczyk Waters MS Technologies
COMPARISON OF CCS(N 2 ) MEASUREMENTS OBTAINED FROM TWO DIFFERENT T-WAVE ION S WITH DIRECT MEASUREMENTS USING A DRIFT TUBE ION Kevin Giles, Martin Palmer, Keith Richardson, Nick Tomczyk Waters MS Technologies
Read more about Pauling and more scientists at: Profiles in Science, The National Library of Medicine, profiles.nlm.nih.gov
 2018 Biochemistry 110 California Institute of Technology Lecture 2: Principles of Protein Structure Linus Pauling (1901-1994) began his studies at Caltech in 1922 and was directed by Arthur Amos oyes to
2018 Biochemistry 110 California Institute of Technology Lecture 2: Principles of Protein Structure Linus Pauling (1901-1994) began his studies at Caltech in 1922 and was directed by Arthur Amos oyes to
Structures in equilibrium at point A: Structures in equilibrium at point B: (ii) Structure at the isoelectric point:
 ame 21 F10-Final Exam Page 2 I. (42 points) (1) (16 points) The titration curve for L-lysine is shown below. Provide (i) the main structures in equilibrium at each of points A and B indicated below and
ame 21 F10-Final Exam Page 2 I. (42 points) (1) (16 points) The titration curve for L-lysine is shown below. Provide (i) the main structures in equilibrium at each of points A and B indicated below and
Very recently Roussis and Proulx clearly demonstrated
 APPLICATION NOTE Deconvolution of Isobaric Interferences in Mass Spectra Juris Meija and Joseph A. Caruso Department of Chemistry, University of Cincinnati, Cincinnati, Ohio, USA The concept of isobar
APPLICATION NOTE Deconvolution of Isobaric Interferences in Mass Spectra Juris Meija and Joseph A. Caruso Department of Chemistry, University of Cincinnati, Cincinnati, Ohio, USA The concept of isobar
Introduction to Proteomics: Fragmentation of protonated peptides and manual sequencing
 Introduction to Proteomics: ragmentation of protonated peptides and manual sequencing Árpád Somogyi CCIC SP SU Summer Workshop S/S hod using ESI Ion rap (Bottom up) 1 An alternative strategy for complex
Introduction to Proteomics: ragmentation of protonated peptides and manual sequencing Árpád Somogyi CCIC SP SU Summer Workshop S/S hod using ESI Ion rap (Bottom up) 1 An alternative strategy for complex
Choosing the metabolomics platform
 GBS 748 Choosing the metabolomics platform Stephen Barnes, PhD 4 7117; sbarnes@uab.edu So, I have my samples what s next? You ve collected your samples and you may have extracted them Protein precipitation
GBS 748 Choosing the metabolomics platform Stephen Barnes, PhD 4 7117; sbarnes@uab.edu So, I have my samples what s next? You ve collected your samples and you may have extracted them Protein precipitation
Mass spectrometry has been used a lot in biology since the late 1950 s. However it really came into play in the late 1980 s once methods were
 Mass spectrometry has been used a lot in biology since the late 1950 s. However it really came into play in the late 1980 s once methods were developed to allow the analysis of large intact (bigger than
Mass spectrometry has been used a lot in biology since the late 1950 s. However it really came into play in the late 1980 s once methods were developed to allow the analysis of large intact (bigger than
Electrospray Ionization High-Resolution Ion Mobility Spectrometry for the Detection of Organic Compounds, 1. Amino Acids
 Anal. Chem. 2001, 73, 3028-3034 Electrospray Ionization High-Resolution Ion Mobility Spectrometry for the Detection of Organic Compounds, 1. Amino Acids Luther W. Beegle and Isik Kanik* Jet Propulsion
Anal. Chem. 2001, 73, 3028-3034 Electrospray Ionization High-Resolution Ion Mobility Spectrometry for the Detection of Organic Compounds, 1. Amino Acids Luther W. Beegle and Isik Kanik* Jet Propulsion
LECTURE-13. Peptide Mass Fingerprinting HANDOUT. Mass spectrometry is an indispensable tool for qualitative and quantitative analysis of
 LECTURE-13 Peptide Mass Fingerprinting HANDOUT PREAMBLE Mass spectrometry is an indispensable tool for qualitative and quantitative analysis of proteins, drugs and many biological moieties to elucidate
LECTURE-13 Peptide Mass Fingerprinting HANDOUT PREAMBLE Mass spectrometry is an indispensable tool for qualitative and quantitative analysis of proteins, drugs and many biological moieties to elucidate
Tutorial 1: Setting up your Skyline document
 Tutorial 1: Setting up your Skyline document Caution! For using Skyline the number formats of your computer have to be set to English (United States). Open the Control Panel Clock, Language, and Region
Tutorial 1: Setting up your Skyline document Caution! For using Skyline the number formats of your computer have to be set to English (United States). Open the Control Panel Clock, Language, and Region
Biological Mass Spectrometry
 Biochemistry 412 Biological Mass Spectrometry February 13 th, 2007 Proteomics The study of the complete complement of proteins found in an organism Degrees of Freedom for Protein Variability Covalent Modifications
Biochemistry 412 Biological Mass Spectrometry February 13 th, 2007 Proteomics The study of the complete complement of proteins found in an organism Degrees of Freedom for Protein Variability Covalent Modifications
Bayesian Protein Structure Prediction
 This is page 1 Printer: Opaque this Bayesian Protein Structure Prediction Scott C. Schmidler Jun S. Liu Douglas L. Brutlag ABSTRACT An important role for statisticians in the age of the Human Genome Project
This is page 1 Printer: Opaque this Bayesian Protein Structure Prediction Scott C. Schmidler Jun S. Liu Douglas L. Brutlag ABSTRACT An important role for statisticians in the age of the Human Genome Project
Modeling Mass Spectrometry-Based Protein Analysis
 Chapter 8 Jan Eriksson and David Fenyö Abstract The success of mass spectrometry based proteomics depends on efficient methods for data analysis. These methods require a detailed understanding of the information
Chapter 8 Jan Eriksson and David Fenyö Abstract The success of mass spectrometry based proteomics depends on efficient methods for data analysis. These methods require a detailed understanding of the information
A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility
 (P&S Ch 5; Fer Ch 2, 9; Palm Ch 10,11; Zub Ch 9) A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility B.
(P&S Ch 5; Fer Ch 2, 9; Palm Ch 10,11; Zub Ch 9) A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility B.
Solutions In each case, the chirality center has the R configuration
 CAPTER 25 669 Solutions 25.1. In each case, the chirality center has the R configuration. C C 2 2 C 3 C(C 3 ) 2 D-Alanine D-Valine 25.2. 2 2 S 2 d) 2 25.3. Pro,, Trp, Tyr, and is, Trp, Tyr, and is Arg,
CAPTER 25 669 Solutions 25.1. In each case, the chirality center has the R configuration. C C 2 2 C 3 C(C 3 ) 2 D-Alanine D-Valine 25.2. 2 2 S 2 d) 2 25.3. Pro,, Trp, Tyr, and is, Trp, Tyr, and is Arg,
Biological Mass Spectrometry: Proteomics. Table of Contents Introduction 2 Information on how to use the module 3
 Biological Mass Spectrometry: Proteomics Table of Contents Introduction 2 Information on how to use the module 3 Section 1. Proteins and Proteomics 5 Section 1A. What is a protein? 5 Section 1B. What is
Biological Mass Spectrometry: Proteomics Table of Contents Introduction 2 Information on how to use the module 3 Section 1. Proteins and Proteomics 5 Section 1A. What is a protein? 5 Section 1B. What is
Potentiometric Titration of an Amino Acid. Introduction
 NAME: Course: DATE Sign-Off: Performed: Potentiometric Titration of an Amino Acid Introduction In previous course-work, you explored the potentiometric titration of a weak acid (HOAc). In this experiment,
NAME: Course: DATE Sign-Off: Performed: Potentiometric Titration of an Amino Acid Introduction In previous course-work, you explored the potentiometric titration of a weak acid (HOAc). In this experiment,
Patrick: An Introduction to Medicinal Chemistry 5e Chapter 03
 01) Which of the following statements is not true regarding the active site of an enzyme? a. An active site is normally on the surface of an enzyme. b. An active site is normally hydrophobic in nature.
01) Which of the following statements is not true regarding the active site of an enzyme? a. An active site is normally on the surface of an enzyme. b. An active site is normally hydrophobic in nature.
BCH 4053 Exam I Review Spring 2017
 BCH 4053 SI - Spring 2017 Reed BCH 4053 Exam I Review Spring 2017 Chapter 1 1. Calculate G for the reaction A + A P + Q. Assume the following equilibrium concentrations: [A] = 20mM, [Q] = [P] = 40fM. Assume
BCH 4053 SI - Spring 2017 Reed BCH 4053 Exam I Review Spring 2017 Chapter 1 1. Calculate G for the reaction A + A P + Q. Assume the following equilibrium concentrations: [A] = 20mM, [Q] = [P] = 40fM. Assume
Exam III. Please read through each question carefully, and make sure you provide all of the requested information.
 09-107 onors Chemistry ame Exam III Please read through each question carefully, and make sure you provide all of the requested information. 1. A series of octahedral metal compounds are made from 1 mol
09-107 onors Chemistry ame Exam III Please read through each question carefully, and make sure you provide all of the requested information. 1. A series of octahedral metal compounds are made from 1 mol
Asymmetrical features of mass spectral peaks produced by quadrupole mass filters
 RAPID COMMUNICATIONS IN MASS SPECTROMETRY Rapid Commun. Mass Spectrom. 2003; 17: 1051 1055 Published online in Wiley InterScience (www.interscience.wiley.com). DOI: 10.1002/rcm.1017 Asymmetrical features
RAPID COMMUNICATIONS IN MASS SPECTROMETRY Rapid Commun. Mass Spectrom. 2003; 17: 1051 1055 Published online in Wiley InterScience (www.interscience.wiley.com). DOI: 10.1002/rcm.1017 Asymmetrical features
Improved 6- Plex TMT Quantification Throughput Using a Linear Ion Trap HCD MS 3 Scan Jane M. Liu, 1,2 * Michael J. Sweredoski, 2 Sonja Hess 2 *
 Improved 6- Plex TMT Quantification Throughput Using a Linear Ion Trap HCD MS 3 Scan Jane M. Liu, 1,2 * Michael J. Sweredoski, 2 Sonja Hess 2 * 1 Department of Chemistry, Pomona College, Claremont, California
Improved 6- Plex TMT Quantification Throughput Using a Linear Ion Trap HCD MS 3 Scan Jane M. Liu, 1,2 * Michael J. Sweredoski, 2 Sonja Hess 2 * 1 Department of Chemistry, Pomona College, Claremont, California
Lecture 8: Mass Spectrometry
 intensity Lecture 8: Mass Spectrometry Relative abundance m/z 1 Ethylbenzene experiment CH 2 CH 3 + m/z = 106 CH 2 + m/z = 91 C 8 H 10 MW = 106 CH + m/z = 77 + 2 2 What information can we get from MS spectrum?
intensity Lecture 8: Mass Spectrometry Relative abundance m/z 1 Ethylbenzene experiment CH 2 CH 3 + m/z = 106 CH 2 + m/z = 91 C 8 H 10 MW = 106 CH + m/z = 77 + 2 2 What information can we get from MS spectrum?
Protein analysis using mass spectrometry
 Protein analysis using mass spectrometry Michael Stadlmeier 2017/12/18 Literature http://www.carellgroup.de/teaching/master 3 What is Proteomics? The proteome is: the entire set of proteins in a given
Protein analysis using mass spectrometry Michael Stadlmeier 2017/12/18 Literature http://www.carellgroup.de/teaching/master 3 What is Proteomics? The proteome is: the entire set of proteins in a given
Lecture 8: Mass Spectrometry
 intensity Lecture 8: Mass Spectrometry Relative abundance m/z 1 Ethylbenzene CH 2 CH 3 + m/z = 106 CH 2 + m/z = 91 C 8 H 10 MW = 106 CH + m/z = 77 + 2 2 What information can be obtained from a MS spectrum?
intensity Lecture 8: Mass Spectrometry Relative abundance m/z 1 Ethylbenzene CH 2 CH 3 + m/z = 106 CH 2 + m/z = 91 C 8 H 10 MW = 106 CH + m/z = 77 + 2 2 What information can be obtained from a MS spectrum?
Biochemistry Quiz Review 1I. 1. Of the 20 standard amino acids, only is not optically active. The reason is that its side chain.
 Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
Hemoglobin (Hb) exists in the blood cells of
 Prediction of Product Ion Isotope Ratios in the Tandem Electrospray Ionization Mass Spectra from the Second Isotope of Tryptic Peptides: Identification of the Variant 131 Gln Glu, Hemoglobin Camden Brian
Prediction of Product Ion Isotope Ratios in the Tandem Electrospray Ionization Mass Spectra from the Second Isotope of Tryptic Peptides: Identification of the Variant 131 Gln Glu, Hemoglobin Camden Brian
Generation Date: 12/07/2015 Generated By: Tristan Wiley Title: Bio I Winter Packet
 Generation Date: 12/07/2015 Generated By: Tristan Wiley Title: Bio I Winter Packet 1. Many natural ecosystems have been destroyed by human activity. To better manage our remaining natural ecosystems, we
Generation Date: 12/07/2015 Generated By: Tristan Wiley Title: Bio I Winter Packet 1. Many natural ecosystems have been destroyed by human activity. To better manage our remaining natural ecosystems, we
Patrick: An Introduction to Medicinal Chemistry 5e Chapter 04
 01) Which of the following statements is not true about receptors? a. Most receptors are proteins situated inside the cell. b. Receptors contain a hollow or cleft on their surface which is known as a binding
01) Which of the following statements is not true about receptors? a. Most receptors are proteins situated inside the cell. b. Receptors contain a hollow or cleft on their surface which is known as a binding
National Nutrient Database for Standard Reference Release 28 slightly revised May, 2016
 National base for Standard Reference Release 28 slightly revised May, 206 Full Report (All s) 005, Cheese, cottage, lowfat, 2% milkfat Report Date: February 23, 208 02:7 EST values and weights are for
National base for Standard Reference Release 28 slightly revised May, 206 Full Report (All s) 005, Cheese, cottage, lowfat, 2% milkfat Report Date: February 23, 208 02:7 EST values and weights are for
Mass Spectrometry of Proteins
 University of South Bohemia Faculty of Science Mass Spectrometry of Proteins Biological Chemistry Bachelor Thesis Author: Tamara Buchberger Supervisor: Prof. RNDr. Libor Grubhoffer, CSc. May, 2011 Annotation
University of South Bohemia Faculty of Science Mass Spectrometry of Proteins Biological Chemistry Bachelor Thesis Author: Tamara Buchberger Supervisor: Prof. RNDr. Libor Grubhoffer, CSc. May, 2011 Annotation
