Amino Acids and Peptides
|
|
|
- Clare Hines
- 6 years ago
- Views:
Transcription
1 Amino Acids Amino Acids and Peptides Amino acid a compound that contains both an amino group and a carboxyl group α-amino acid an amino acid in which the amino group is on the carbon adjacent to the carboxyl group although α-amino acids are commonly written in the unionized form, they are - more properly R- C-C written in R- the C-C zwitterion (internal salt) form 2 3 unionized form zwitterion Page 2 Chirality of Amino Acids Chirality of Amino Acids With the exception of glycine, all protein-derived amino acids have at least one stereocenter (the α-carbon) and are chiral the vast majority of α-amino acids have the L- configuration at the α-carbon Comparison of the stereochemistry of alanine and glyceraldehyde (Fischer projection formulas) the naturally occurring form C - 3 C - 3 C - 3 C - 3 C 3 D-Alanine C 3 L-Alanine C 3 C 3 D-Alanine L-Alanine (Fischer projections) the naturally occurring form C C 2 D-Glyceraldehyde C C 2 L-Glyceraldehyde Page 3 Page 4 onpolar side chains (predominant form at p 7.0) glycine (gly, G) phenylalanine (phe, F) - alanine (ala, A) C 3 - tryptophan (trp, W) valine (val, V) (C 3 ) 2 C leucine (leu, L) (C 3 ) 2 CC 2 - isoleucine (ile, I) proline (Pro, P) C 3 C 2 C( C 3 )- methionine (met, M) C 3 C 2 C 2 - Polar side chains (predominant form at p 7.0) asparagine (asn, ) serine (ser, ) 2 CC 2 - glutamine (glu, G) 2 CC 2 C 2 - C 2 - threonine (thr, T) C 3 C- Page 5 Page 6 1
2 Acidic side chains (predominant form at p 7.0) aspartic acid (asp, D) glutamic acid (glu, E) - CC CC 2 C 2 - cysteine (cys, C) tyrosine (tyr, Y) C 2 - C 2 - Basic side chains (predominant form at p 7.0) arginine (arg, R) histidine (his, ) 2 2 CC 2 C 2 C 2 - C 2 - lysine (lys, K) 3 C 2 C 2 C 2 C 2 - Page 7 Page 8 Uncommon Amino Acids ote these structural features 1. All 20 are α-amino acids 2. For 19 of the 20, the α-amino group is primary; for proline, it is secondary 3. With the exception of glycine, the α-carbon of each is a stereocenter 4. Isoleucine and threonine contain a second stereocenter 5. The sulfhydryl group (pk a 8.3) of cysteine, the imidazole group (pk a 6.0) of histidine, and the phenolic hydroxyl (pk Page 9 a 10.1) of phenylalanine are partially ionized at p 7.0, but the ionic form is not Each example is derived from a common amino acid by the modification shown in color I I 3 C - C - C - 3 I I 3 ydroxylysine ydroxyproline Thyroxine hydroxylysine and hydroxyproline are found only in a few connective tissues such as collagen thyroxine is found only in the thyroid gland Page 10 Ionization of Amino Acids 1 charge 0 charge -1 charge 3 C pka = C - 2 C - pk a = 9.69 Isoelectric zwitterion Titration of Amino Acids Figure (a) Titration of alanine with a 2 charge 3 C pk a = charge 3 C - 0 charge 3 C - pk a = 6.04 pk a = charge 2 C - Page 11 Isoelectric zwitterion Page 12 2
3 Titration of Amino Acids Acidity α-c Groups Figure (b) Titration of histidine with a The average an α-carboxyl group is 2.19, which makes them considerably stronger acids than acetic acid (pk a 4.76) the greater acidity of the amino acid carboxyl group is due to the electron-withdrawing inductive effect of the - 3 group The ammonium ion has an electron-withdrawing inductive effect RCC 2 pk a = 2.19 RCC Page 13 Page 14 Acidity α- 3 groups Basicity Guanidine Group The average value of pk a for an α- 3 group is 9.47, compared with a value of for a 2 alkylammonium ion pk a = 9.47 RCC - 2 RCC pk a = C 3 CC 3 2 C 3 CC The side chain of arginine is a considerably stronger base than an aliphatic amine basicity of the guanido group is attributed to the large resonance stabilization of the protonated form relative 2 to the neutral 2 form 2 R C 2 R C R C 3 2 R C 2 pk a = Page 15 Page 16 Basicity Imidazole Group Ionization vs p The imidazole group on the side chain of histidine is a heterocyclic aromatic amine C 2 CC - 2 C 2 CC Given the value of each functional group, we can calculate the ratio of each acid to its conjugate base as a function of p Consider the ionization of an α-c pk a = 2.00 α C α C this lone pair is not a part of the aromatic sextet; it is the proton acceptor C 2 CC pk a 6.04 writing the acid ionization constant and rearranging terms gives [ α-c - ] K a = [ 3 ] [ α-c - ] K = a or [ α-c ] [ α-c ] [ 3 ] Page 17 Page 18 3
4 Ionization vs p Ionization vs p substituting the value of K a (1 x 10-2 ) for the hydrogen [ α-c ion - concentration ] K = a 1.00 at p x 10-2 = 7.0 = ( x ) gives [ α-c ] [ 3 ] 1.00 x 10-7 We can also calculate the ratio of acid to conjugate base for an α- 3 group; for this calculation, assume a value 10.0 for pk a α 3 2 pk a = α 2 3 at p 7.0, the α-carboxyl group is virtually 100% in the ionized or conjugate base form, and has a net charge of -1 we can repeat this calculation at any p and determine the ratio of [α C - ]to[α C] and Page 19 writing the acid ionization constant and rearranging gives [ α- 2 ] K [ α- = a 3 ] [ 3 ] Page 20 Ionization vs p enderson-asselbalch substituting values for K a of an α- 3 group and the hydrogen ion concentration at p 7.0 gives [ α- 2 ] K a 1.00 x [ α- = 3 ] [ 3 = = 1.00 x 10-3 ] 1.00 x 10-7 at p 7.0, the ratio of α- 2 to α- 3 is approximately 1 to 1000 at this p, an α-amino group is 99.9% in the acid or protonated form and has a charge of 1 Page 21 We have calculated the ratio of acid to conjugate base for an α-carboxyl group and an α-amino group at p 7.0 We can do this for any weak acid and its conjugate base at any p using the enderson-asselbalch equation Page 22 [conjugate base] p = pk a log [weak acid] enderson-asselbalch Isoelectric p using the enderson-asselbalch equation, we can calculate the percent of charged or uncharged form present and the net charge on 100% 86% 99% 100% 88% 100% serine at p 3.0, 7.0, and C-C- 3 - C-C C-C- - C 2 C 2 C 2 p 3.0 p 7.0 p 10.0 et charge 1 et charge 0 et charge -1 Isoelectric p, pi the p at which the majority of molecules of a compound in solution have no net charge the pi for glycine, for example, falls midway between the pk a values for the carboxyl and amino groups pi = 1 2 (pk a α C p K a α 3 ) = 1 ( ) = Page 23 given in the following tables are isoelectric p values for the 20 protein-derived amino acids Page 24 4
5 Isoelectric p (pi) Table 3.2 pk a and pi of α-amino acids Isoelectric p (pi) Table 3.2 (cont'd) onpolar & polar side chains α C α 3 alanine asparagine glutamine glycine isoleucine leucine methionine phenylalanine proline serine threonine tryptophan valine Page 25 ide Chain pi Page 26 Acidic ide Chains α C α C α 3 α 3 ide Chain aspartic acid glutamic acid cysteine tyrosine Basic ide Chains ide Chain arginine histidine lysine pi pi Electrophoresis Electrophoresis(Process) Electrophoresis the process of separating compounds on the basis of their electric charge electrophoresis of amino acids can be carried out using paper, starch, agar, certain plastics, and cellulose acetate as solid supports in paper electrophoresis, a paper strip saturated with an aqueous buffer of predetermined p serves as a bridge between two electrode vessels Page 27 a sample of amino acids is applied as a spot (the origin) on the solid support strip an electric potential is applied to the electrode vessels and amino acids migrate toward the electrode with charge opposite their own molecules with a high charge density move faster than those with a low charge density molecules at their isoelectric point remain at the origin after separation is complete, the strip is dried and developed to make the separated amino acids Page 28 Polypeptides In 1902, Emil Fischer proposed that proteins are long chains of amino acids joined by amide bonds to which he gave the name peptide bonds Peptide bond the special name given to the amide bond between the α- carboxyl group of one amino acid and the α-amino group of another erylalanine (er-ala) 2 C C 3 erine (er) Alanine (Ala) peptide bond 2 C 3 - C3 erylalanine (er-ala) Page 29 Page 30 5
6 Peptides Geometry of Peptide Bond peptide the name given to a short polymer of amino acids joined by peptide bonds; they are classified by the number of amino acids in the chain dipeptide a molecule containing two amino acids joined by a peptide bond tripeptide a molecule containing three amino acids joined by peptide bonds polypeptide a macromolecule containing many amino acids joined by peptide bonds protein a biological macromolecule of molecular Page 31 the four atoms of a peptide bond and the two alpha carbons joined to it lie in a plane with bond angles of 120 about C and to account for this geometry, Linus Pauling proposed that a peptide bond is most accurately represented as a hybrid of two contributing structures - C α C α the hybrid has C considerable C- double C bond character and C α rotation about the Cpeptide α bond is restricted Page 32 (1) (2) Writing Peptides ome mall Peptides By convention, peptides are written from the left, beginning with the free - 3 group and ending with the free -C - group the repeat pattern, starting from the -terminal amino acid, is ---> α-carbon ---> carbonyl carbon etc. -terminal amino acid 3 peptide bonds er-met-asn - 2 C-terminal amino acid 3 -C- C--C-C-C 3 C 2 C 2 C - C 6 5 L-Aspartyl-L-phenylalanine methyl ester (Aspartame) 3 -C 2 -C 2 -C-- C-C - C 2 β-alanyl-l-histidine (Carnosine) Page 33 Page 34 Glutathione Enkephalins - 3 Glutathione, G (reduced form) - 2e - oxidation 2e - reduction Glutathione, G-G (oxidized form) - - A disulfide bond Leucine enkephalin Tyr-Gly-Gly-Phe-Leu = Y-G-G-F-L Methionine enkephalin Tyr-Gly-Gly-Phe-Met = Y-G-G-F-M Page 35 Page 36 6
7 xytocin & Vasopressin 3 -Cys-Tyr-Ile Gln Cys-Asn Pro-Leu-Gly-C- 2 xytocin 3 -Cys-Tyr-Phe Gln Cys-Asn Pro-Arg-Gly-C- 2 Vasopressin End Page 37 Page 38 7
Biochemistry by Mary K. Campbell & Shawn O. Farrell 8th. Ed. 2016
 3 Biochemistry by Mary K. Campbell & Shawn. Farrell 8th. Ed. 2016 3-1 3 Amino Acids & Peptides 3-2 3 Learning bjectives 1. What are amino acids, and what is their threedimensional structure? 2. What are
3 Biochemistry by Mary K. Campbell & Shawn. Farrell 8th. Ed. 2016 3-1 3 Amino Acids & Peptides 3-2 3 Learning bjectives 1. What are amino acids, and what is their threedimensional structure? 2. What are
Proteins: Characteristics and Properties of Amino Acids
 SBI4U:Biochemistry Macromolecules Eachaminoacidhasatleastoneamineandoneacidfunctionalgroupasthe nameimplies.thedifferentpropertiesresultfromvariationsinthestructuresof differentrgroups.thergroupisoftenreferredtoastheaminoacidsidechain.
SBI4U:Biochemistry Macromolecules Eachaminoacidhasatleastoneamineandoneacidfunctionalgroupasthe nameimplies.thedifferentpropertiesresultfromvariationsinthestructuresof differentrgroups.thergroupisoftenreferredtoastheaminoacidsidechain.
Chemistry Chapter 22
 hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
Chemical Properties of Amino Acids
 hemical Properties of Amino Acids Protein Function Make up about 15% of the cell and have many functions in the cell 1. atalysis: enzymes 2. Structure: muscle proteins 3. Movement: myosin, actin 4. Defense:
hemical Properties of Amino Acids Protein Function Make up about 15% of the cell and have many functions in the cell 1. atalysis: enzymes 2. Structure: muscle proteins 3. Movement: myosin, actin 4. Defense:
A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of
 Questions with Answers- Amino Acids & Peptides A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of 5.0-6.5 (Questions 1-4) 1. Which
Questions with Answers- Amino Acids & Peptides A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of 5.0-6.5 (Questions 1-4) 1. Which
Solutions In each case, the chirality center has the R configuration
 CAPTER 25 669 Solutions 25.1. In each case, the chirality center has the R configuration. C C 2 2 C 3 C(C 3 ) 2 D-Alanine D-Valine 25.2. 2 2 S 2 d) 2 25.3. Pro,, Trp, Tyr, and is, Trp, Tyr, and is Arg,
CAPTER 25 669 Solutions 25.1. In each case, the chirality center has the R configuration. C C 2 2 C 3 C(C 3 ) 2 D-Alanine D-Valine 25.2. 2 2 S 2 d) 2 25.3. Pro,, Trp, Tyr, and is, Trp, Tyr, and is Arg,
CHEM J-9 June 2014
 CEM1611 2014-J-9 June 2014 Alanine (ala) and lysine (lys) are two amino acids with the structures given below as Fischer projections. The pk a values of the conjugate acid forms of the different functional
CEM1611 2014-J-9 June 2014 Alanine (ala) and lysine (lys) are two amino acids with the structures given below as Fischer projections. The pk a values of the conjugate acid forms of the different functional
Chapter 4: Amino Acids
 Chapter 4: Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. lipid polysaccharide enzyme 1940s 1980s. Lipids membrane 1960s. Polysaccharide Are energy metabolites and many of
Chapter 4: Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. lipid polysaccharide enzyme 1940s 1980s. Lipids membrane 1960s. Polysaccharide Are energy metabolites and many of
Read more about Pauling and more scientists at: Profiles in Science, The National Library of Medicine, profiles.nlm.nih.gov
 2018 Biochemistry 110 California Institute of Technology Lecture 2: Principles of Protein Structure Linus Pauling (1901-1994) began his studies at Caltech in 1922 and was directed by Arthur Amos oyes to
2018 Biochemistry 110 California Institute of Technology Lecture 2: Principles of Protein Structure Linus Pauling (1901-1994) began his studies at Caltech in 1922 and was directed by Arthur Amos oyes to
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
1. Amino Acids and Peptides Structures and Properties
 1. Amino Acids and Peptides Structures and Properties Chemical nature of amino acids The!-amino acids in peptides and proteins (excluding proline) consist of a carboxylic acid ( COOH) and an amino ( NH
1. Amino Acids and Peptides Structures and Properties Chemical nature of amino acids The!-amino acids in peptides and proteins (excluding proline) consist of a carboxylic acid ( COOH) and an amino ( NH
Principles of Biochemistry
 Principles of Biochemistry Fourth Edition Donald Voet Judith G. Voet Charlotte W. Pratt Chapter 4 Amino Acids: The Building Blocks of proteins (Page 76-90) Chapter Contents 1- Amino acids Structure: 2-
Principles of Biochemistry Fourth Edition Donald Voet Judith G. Voet Charlotte W. Pratt Chapter 4 Amino Acids: The Building Blocks of proteins (Page 76-90) Chapter Contents 1- Amino acids Structure: 2-
Lecture'18:'April'2,'2013
 CM'224' 'rganic'chemistry'ii Spring'2013,'Des'Plaines' 'Prof.'Chad'Landrie 2 3 N cysteine (Cys) S oxidation S S 3 N cystine N 3 Lecture'18:'April'2,'2013 Disaccharides+&+Polysaccharides Amino+acids++(26.1926.3)
CM'224' 'rganic'chemistry'ii Spring'2013,'Des'Plaines' 'Prof.'Chad'Landrie 2 3 N cysteine (Cys) S oxidation S S 3 N cystine N 3 Lecture'18:'April'2,'2013 Disaccharides+&+Polysaccharides Amino+acids++(26.1926.3)
Practice Midterm Exam 200 points total 75 minutes Multiple Choice (3 pts each 30 pts total) Mark your answers in the space to the left:
 MITES ame Practice Midterm Exam 200 points total 75 minutes Multiple hoice (3 pts each 30 pts total) Mark your answers in the space to the left: 1. Amphipathic molecules have regions that are: a) polar
MITES ame Practice Midterm Exam 200 points total 75 minutes Multiple hoice (3 pts each 30 pts total) Mark your answers in the space to the left: 1. Amphipathic molecules have regions that are: a) polar
Chapter 3 - Amino Acids
 hapter 3 Amino Acids αamino Acids are the main constituents of proteins. But they also can function as neurotransmitters (glutamate, γaminobutyric acid), hormones (thyroxine; see right), as bacterial cell
hapter 3 Amino Acids αamino Acids are the main constituents of proteins. But they also can function as neurotransmitters (glutamate, γaminobutyric acid), hormones (thyroxine; see right), as bacterial cell
Exam I Answer Key: Summer 2006, Semester C
 1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
PROTEIN STRUCTURE AMINO ACIDS H R. Zwitterion (dipolar ion) CO 2 H. PEPTIDES Formal reactions showing formation of peptide bond by dehydration:
 PTEI STUTUE ydrolysis of proteins with aqueous acid or base yields a mixture of free amino acids. Each type of protein yields a characteristic mixture of the ~ 20 amino acids. AMI AIDS Zwitterion (dipolar
PTEI STUTUE ydrolysis of proteins with aqueous acid or base yields a mixture of free amino acids. Each type of protein yields a characteristic mixture of the ~ 20 amino acids. AMI AIDS Zwitterion (dipolar
UNIT TWELVE. a, I _,o "' I I I. I I.P. l'o. H-c-c. I ~o I ~ I / H HI oh H...- I II I II 'oh. HO\HO~ I "-oh
 UNT TWELVE PROTENS : PEPTDE BONDNG AND POLYPEPTDES 12 CONCEPTS Many proteins are important in biological structure-for example, the keratin of hair, collagen of skin and leather, and fibroin of silk. Other
UNT TWELVE PROTENS : PEPTDE BONDNG AND POLYPEPTDES 12 CONCEPTS Many proteins are important in biological structure-for example, the keratin of hair, collagen of skin and leather, and fibroin of silk. Other
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I Dr. Glen B Legge
 EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
Exam III. Please read through each question carefully, and make sure you provide all of the requested information.
 09-107 onors Chemistry ame Exam III Please read through each question carefully, and make sure you provide all of the requested information. 1. A series of octahedral metal compounds are made from 1 mol
09-107 onors Chemistry ame Exam III Please read through each question carefully, and make sure you provide all of the requested information. 1. A series of octahedral metal compounds are made from 1 mol
Translation. A ribosome, mrna, and trna.
 Translation The basic processes of translation are conserved among prokaryotes and eukaryotes. Prokaryotic Translation A ribosome, mrna, and trna. In the initiation of translation in prokaryotes, the Shine-Dalgarno
Translation The basic processes of translation are conserved among prokaryotes and eukaryotes. Prokaryotic Translation A ribosome, mrna, and trna. In the initiation of translation in prokaryotes, the Shine-Dalgarno
CHAPTER 29 HW: AMINO ACIDS + PROTEINS
 CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell
 Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell Mathematics and Biochemistry University of Wisconsin - Madison 0 There Are Many Kinds Of Proteins The word protein comes
Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell Mathematics and Biochemistry University of Wisconsin - Madison 0 There Are Many Kinds Of Proteins The word protein comes
Potentiometric Titration of an Amino Acid. Introduction
 NAME: Course: DATE Sign-Off: Performed: Potentiometric Titration of an Amino Acid Introduction In previous course-work, you explored the potentiometric titration of a weak acid (HOAc). In this experiment,
NAME: Course: DATE Sign-Off: Performed: Potentiometric Titration of an Amino Acid Introduction In previous course-work, you explored the potentiometric titration of a weak acid (HOAc). In this experiment,
Periodic Table. 8/3/2006 MEDC 501 Fall
 Periodic Table 8/3/2006 MEDC 501 Fall 2006 1 rbitals Shapes of rbitals s - orbital p -orbital 8/3/2006 MEDC 501 Fall 2006 2 Ionic Bond - acl Electronic Structure 11 a :: 1s 2 2s 2 2p x2 2p y2 2p z2 3s
Periodic Table 8/3/2006 MEDC 501 Fall 2006 1 rbitals Shapes of rbitals s - orbital p -orbital 8/3/2006 MEDC 501 Fall 2006 2 Ionic Bond - acl Electronic Structure 11 a :: 1s 2 2s 2 2p x2 2p y2 2p z2 3s
NAME. EXAM I I. / 36 September 25, 2000 Biochemistry I II. / 26 BICH421/621 III. / 38 TOTAL /100
 EXAM I I. / 6 September 25, 2000 Biochemistry I II. / 26 BIH421/621 III. / 8 TOTAL /100 I. MULTIPLE HOIE (6 points) hoose the BEST answer to the question by circling the appropriate letter. 1. An amino
EXAM I I. / 6 September 25, 2000 Biochemistry I II. / 26 BIH421/621 III. / 8 TOTAL /100 I. MULTIPLE HOIE (6 points) hoose the BEST answer to the question by circling the appropriate letter. 1. An amino
7.05 Spring 2004 February 27, Recitation #2
 Recitation #2 Contact Information TA: Victor Sai Recitation: Friday, 3-4pm, 2-132 E-mail: sai@mit.edu ffice ours: Friday, 4-5pm, 2-132 Unit 1 Schedule Recitation/Exam Date Lectures covered Recitation #2
Recitation #2 Contact Information TA: Victor Sai Recitation: Friday, 3-4pm, 2-132 E-mail: sai@mit.edu ffice ours: Friday, 4-5pm, 2-132 Unit 1 Schedule Recitation/Exam Date Lectures covered Recitation #2
Protein Structure Bioinformatics Introduction
 1 Swiss Institute of Bioinformatics Protein Structure Bioinformatics Introduction Basel, 27. September 2004 Torsten Schwede Biozentrum - Universität Basel Swiss Institute of Bioinformatics Klingelbergstr
1 Swiss Institute of Bioinformatics Protein Structure Bioinformatics Introduction Basel, 27. September 2004 Torsten Schwede Biozentrum - Universität Basel Swiss Institute of Bioinformatics Klingelbergstr
CHMI 2227 EL. Biochemistry I. Test January Prof : Eric R. Gauthier, Ph.D.
 CHMI 2227 EL Biochemistry I Test 1 26 January 2007 Prof : Eric R. Gauthier, Ph.D. Guidelines: 1) Duration: 55 min 2) 14 questions, on 7 pages. For 70 marks (5 marks per question). Worth 15 % of the final
CHMI 2227 EL Biochemistry I Test 1 26 January 2007 Prof : Eric R. Gauthier, Ph.D. Guidelines: 1) Duration: 55 min 2) 14 questions, on 7 pages. For 70 marks (5 marks per question). Worth 15 % of the final
Content : Properties of amino acids.. Separation and Analysis of Amino Acids
 قسم الكيمياء الحيوية.دولت على سالمه د. استاذ الكيمياء الحيوية ٢٠١٥-٢٠١٤ المحاضرة الثانية 1 Content : Properties of amino acids.. Separation and Analysis of Amino Acids 2 3 Physical Properties of Amino
قسم الكيمياء الحيوية.دولت على سالمه د. استاذ الكيمياء الحيوية ٢٠١٥-٢٠١٤ المحاضرة الثانية 1 Content : Properties of amino acids.. Separation and Analysis of Amino Acids 2 3 Physical Properties of Amino
BENG 183 Trey Ideker. Protein Sequencing
 BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
INTRODUCTION. Amino acids occurring in nature have the general structure shown below:
 Biochemistry I Laboratory Amino Acid Thin Layer Chromatography INTRODUCTION The primary importance of amino acids in cell structure and metabolism lies in the fact that they serve as building blocks for
Biochemistry I Laboratory Amino Acid Thin Layer Chromatography INTRODUCTION The primary importance of amino acids in cell structure and metabolism lies in the fact that they serve as building blocks for
BCH 4053 Exam I Review Spring 2017
 BCH 4053 SI - Spring 2017 Reed BCH 4053 Exam I Review Spring 2017 Chapter 1 1. Calculate G for the reaction A + A P + Q. Assume the following equilibrium concentrations: [A] = 20mM, [Q] = [P] = 40fM. Assume
BCH 4053 SI - Spring 2017 Reed BCH 4053 Exam I Review Spring 2017 Chapter 1 1. Calculate G for the reaction A + A P + Q. Assume the following equilibrium concentrations: [A] = 20mM, [Q] = [P] = 40fM. Assume
Viewing and Analyzing Proteins, Ligands and their Complexes 2
 2 Viewing and Analyzing Proteins, Ligands and their Complexes 2 Overview Viewing the accessible surface Analyzing the properties of proteins containing thousands of atoms is best accomplished by representing
2 Viewing and Analyzing Proteins, Ligands and their Complexes 2 Overview Viewing the accessible surface Analyzing the properties of proteins containing thousands of atoms is best accomplished by representing
ORGANIC - BROWN 8E CH AMINO ACIDS AND PROTEINS.
 !! www.clutchprep.com CONCEPT: INTRODUCTION TO PROTEINS Proteins are polypeptides that have some biological function. Peptides are composed of polymers of monomeric units called α-amino acids The 20 most
!! www.clutchprep.com CONCEPT: INTRODUCTION TO PROTEINS Proteins are polypeptides that have some biological function. Peptides are composed of polymers of monomeric units called α-amino acids The 20 most
BIS Office Hours
 BIS103-001 001 ffice ours TUE (2-3 pm) Rebecca Shipman WED (9:30-10:30 am) TUE (12-1 pm) Stephen Abreu TUR (12-1 pm) FRI (9-11 am) Steffen Abel Lecture 2 Topics Finish discussion of thermodynamics (ΔG,
BIS103-001 001 ffice ours TUE (2-3 pm) Rebecca Shipman WED (9:30-10:30 am) TUE (12-1 pm) Stephen Abreu TUR (12-1 pm) FRI (9-11 am) Steffen Abel Lecture 2 Topics Finish discussion of thermodynamics (ΔG,
Section Week 3. Junaid Malek, M.D.
 Section Week 3 Junaid Malek, M.D. Biological Polymers DA 4 monomers (building blocks), limited structure (double-helix) RA 4 monomers, greater flexibility, multiple structures Proteins 20 Amino Acids,
Section Week 3 Junaid Malek, M.D. Biological Polymers DA 4 monomers (building blocks), limited structure (double-helix) RA 4 monomers, greater flexibility, multiple structures Proteins 20 Amino Acids,
Dental Biochemistry Exam The total number of unique tripeptides that can be produced using all of the common 20 amino acids is
 Exam Questions for Dental Biochemistry Monday August 27, 2007 E.J. Miller 1. The compound shown below is CH 3 -CH 2 OH A. acetoacetate B. acetic acid C. acetaldehyde D. produced by reduction of acetaldehyde
Exam Questions for Dental Biochemistry Monday August 27, 2007 E.J. Miller 1. The compound shown below is CH 3 -CH 2 OH A. acetoacetate B. acetic acid C. acetaldehyde D. produced by reduction of acetaldehyde
Protein structure. Protein structure. Amino acid residue. Cell communication channel. Bioinformatics Methods
 Cell communication channel Bioinformatics Methods Iosif Vaisman Email: ivaisman@gmu.edu SEQUENCE STRUCTURE DNA Sequence Protein Sequence Protein Structure Protein structure ATGAAATTTGGAAACTTCCTTCTCACTTATCAGCCACCT...
Cell communication channel Bioinformatics Methods Iosif Vaisman Email: ivaisman@gmu.edu SEQUENCE STRUCTURE DNA Sequence Protein Sequence Protein Structure Protein structure ATGAAATTTGGAAACTTCCTTCTCACTTATCAGCCACCT...
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor
 LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
Lecture 15: Realities of Genome Assembly Protein Sequencing
 Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
NH 2. Biochemistry I, Fall Term Sept 9, Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter
 Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Protein Structure Marianne Øksnes Dalheim, PhD candidate Biopolymers, TBT4135, Autumn 2013
 Protein Structure Marianne Øksnes Dalheim, PhD candidate Biopolymers, TBT4135, Autumn 2013 The presentation is based on the presentation by Professor Alexander Dikiy, which is given in the course compedium:
Protein Structure Marianne Øksnes Dalheim, PhD candidate Biopolymers, TBT4135, Autumn 2013 The presentation is based on the presentation by Professor Alexander Dikiy, which is given in the course compedium:
A Plausible Model Correlates Prebiotic Peptide Synthesis with. Primordial Genetic Code
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 A Plausible Model Correlates Prebiotic Peptide Synthesis with Primordial Genetic Code Jianxi Ying,
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 A Plausible Model Correlates Prebiotic Peptide Synthesis with Primordial Genetic Code Jianxi Ying,
Properties of Amino Acids
 Biochemistry Department Date:19/9/ 2017 Properties of Amino Acids Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology 1 Intended Learning Outcomes (ILOs) By the end of this lecture,
Biochemistry Department Date:19/9/ 2017 Properties of Amino Acids Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology 1 Intended Learning Outcomes (ILOs) By the end of this lecture,
Peptides And Proteins
 Kevin Burgess, May 3, 2017 1 Peptides And Proteins from chapter(s) in the recommended text A. Introduction B. omenclature And Conventions by amide bonds. on the left, right. 2 -terminal C-terminal triglycine
Kevin Burgess, May 3, 2017 1 Peptides And Proteins from chapter(s) in the recommended text A. Introduction B. omenclature And Conventions by amide bonds. on the left, right. 2 -terminal C-terminal triglycine
12/6/12. Dr. Sanjeeva Srivastava IIT Bombay. Primary Structure. Secondary Structure. Tertiary Structure. Quaternary Structure.
 Dr. anjeeva rivastava Primary tructure econdary tructure Tertiary tructure Quaternary tructure Amino acid residues α Helix Polypeptide chain Assembled subunits 2 1 Amino acid sequence determines 3-D structure
Dr. anjeeva rivastava Primary tructure econdary tructure Tertiary tructure Quaternary tructure Amino acid residues α Helix Polypeptide chain Assembled subunits 2 1 Amino acid sequence determines 3-D structure
5) An amino acid that doesn't exist in proteins: - Tyrosine - Tryptophan - Cysteine - ornithine* 6) How many tripeptides can be formed from using one
 Biochemistry First 1) An amino acid that does not have L & D: - proline - serine - Glycine* - valine 2) An amino acid that exists in beta-bends and turns: - Gly* - Pro - Arg - Asp 3) An amino acid that
Biochemistry First 1) An amino acid that does not have L & D: - proline - serine - Glycine* - valine 2) An amino acid that exists in beta-bends and turns: - Gly* - Pro - Arg - Asp 3) An amino acid that
NAME IV. /22. I. MULTIPLE CHOICE. (48 points; 2 pts each) Choose the BEST answer to the question by circling the appropriate letter.
 NAME Exam I I. /48 September 25, 2017 Biochemistry I II. / 4 BI/CH 421/621 III. /26 IV. /22 TOTAL /100 I. MULTIPLE CHOICE. (48 points; 2 pts each) Choose the BEST answer to the question by circling the
NAME Exam I I. /48 September 25, 2017 Biochemistry I II. / 4 BI/CH 421/621 III. /26 IV. /22 TOTAL /100 I. MULTIPLE CHOICE. (48 points; 2 pts each) Choose the BEST answer to the question by circling the
Note that side chains serve as a) stabilizers of protein structure, b) reactive centers, and c) micro-environments. * + H 3 N-C-COOH H 2 N-C-COO -
 BIOCEMISTRY I AMINO ACIDS I. Amino Acid Structure One of the most important macromolecules (chains of distinct molecular units) in the biosphere is protein. Proteins are needed for catalysis, reaction,
BIOCEMISTRY I AMINO ACIDS I. Amino Acid Structure One of the most important macromolecules (chains of distinct molecular units) in the biosphere is protein. Proteins are needed for catalysis, reaction,
Studies Leading to the Development of a Highly Selective. Colorimetric and Fluorescent Chemosensor for Lysine
 Supporting Information for Studies Leading to the Development of a Highly Selective Colorimetric and Fluorescent Chemosensor for Lysine Ying Zhou, a Jiyeon Won, c Jin Yong Lee, c * and Juyoung Yoon a,
Supporting Information for Studies Leading to the Development of a Highly Selective Colorimetric and Fluorescent Chemosensor for Lysine Ying Zhou, a Jiyeon Won, c Jin Yong Lee, c * and Juyoung Yoon a,
Content : Properties of amino acids.. Separation and Analysis of Amino Acids
 قسم الكيمياء الحيوية.دولت على سالمه د استاذ الكيمياء الحيوية ٢٠١٥-٢٠١٤ المحاضرة الثانية Content : Properties of amino acids.. Separation and Analysis of Amino Acids 2 -3 A. Physical properties 1. Solubility:
قسم الكيمياء الحيوية.دولت على سالمه د استاذ الكيمياء الحيوية ٢٠١٥-٢٠١٤ المحاضرة الثانية Content : Properties of amino acids.. Separation and Analysis of Amino Acids 2 -3 A. Physical properties 1. Solubility:
Basic Principles of Protein Structures
 Basic Principles of Protein Structures Proteins Proteins: The Molecule of Life Proteins: Building Blocks Proteins: Secondary Structures Proteins: Tertiary and Quartenary Structure Proteins: Geometry Proteins
Basic Principles of Protein Structures Proteins Proteins: The Molecule of Life Proteins: Building Blocks Proteins: Secondary Structures Proteins: Tertiary and Quartenary Structure Proteins: Geometry Proteins
Dental Biochemistry EXAM I
 Dental Biochemistry EXAM I August 29, 2005 In the reaction below: CH 3 -CH 2 OH -~ ethanol CH 3 -CHO acetaldehyde A. acetoacetate is being produced B. ethanol is being oxidized to acetaldehyde C. acetaldehyde
Dental Biochemistry EXAM I August 29, 2005 In the reaction below: CH 3 -CH 2 OH -~ ethanol CH 3 -CHO acetaldehyde A. acetoacetate is being produced B. ethanol is being oxidized to acetaldehyde C. acetaldehyde
Review of General & Organic Chemistry
 Review of General & Organic Chemistry Diameter of a nucleus is only about 10-15 m. Diameter of an atom is only about 10-10 m. Fig 3.1 The structure of an atom Periodic Table, shown below, is a representation
Review of General & Organic Chemistry Diameter of a nucleus is only about 10-15 m. Diameter of an atom is only about 10-10 m. Fig 3.1 The structure of an atom Periodic Table, shown below, is a representation
BI/CH421 Biochemistry I Exam 1 09/29/2014
 Part I: Multiple choice for each question, circle the choice that best answers the question. Do not write the letter in the margin to indicate your answer, circle it. 3 points each. 1. For a reaction with
Part I: Multiple choice for each question, circle the choice that best answers the question. Do not write the letter in the margin to indicate your answer, circle it. 3 points each. 1. For a reaction with
Sheet #3 Dr. Mamoun Ahram 06/07/2014. Peptide Bonds
 Peptide Bonds Features of peptide bond: Zigzag structure : when you look at it you notice that it s going up,down,up Notice the figure below A structure that consumes the least amount of energy to form
Peptide Bonds Features of peptide bond: Zigzag structure : when you look at it you notice that it s going up,down,up Notice the figure below A structure that consumes the least amount of energy to form
Part II => PROTEINS and ENZYMES. 2.1 AMINO ACIDS 2.1a Nomenclature 2.1b Stereochemistry 2.1c Derivatives
 Part II => PROTEINS and ENZYMES 2.1 AMINO ACIDS 2.1a Nomenclature 2.1b Stereochemistry 2.1c Derivatives Section 2.1a: Nomenclature Synopsis 2.1a - Proteins (or polypeptides) are polymers made up of building
Part II => PROTEINS and ENZYMES 2.1 AMINO ACIDS 2.1a Nomenclature 2.1b Stereochemistry 2.1c Derivatives Section 2.1a: Nomenclature Synopsis 2.1a - Proteins (or polypeptides) are polymers made up of building
Conformational Analysis
 Conformational Analysis C01 3 C C 3 is the most stable by 0.9 kcal/mole C02 K eq = K 1-1 * K 2 = 0.45-1 * 0.048 = 0.11 C04 The intermediate in the reaction of 2 has an unfavorable syn-pentane interaction,
Conformational Analysis C01 3 C C 3 is the most stable by 0.9 kcal/mole C02 K eq = K 1-1 * K 2 = 0.45-1 * 0.048 = 0.11 C04 The intermediate in the reaction of 2 has an unfavorable syn-pentane interaction,
Structures in equilibrium at point A: Structures in equilibrium at point B: (ii) Structure at the isoelectric point:
 ame 21 F10-Final Exam Page 2 I. (42 points) (1) (16 points) The titration curve for L-lysine is shown below. Provide (i) the main structures in equilibrium at each of points A and B indicated below and
ame 21 F10-Final Exam Page 2 I. (42 points) (1) (16 points) The titration curve for L-lysine is shown below. Provide (i) the main structures in equilibrium at each of points A and B indicated below and
Biochemistry Quiz Review 1I. 1. Of the 20 standard amino acids, only is not optically active. The reason is that its side chain.
 Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
Next Step MCAT Super Review: Amino Acids
 Next Step Welcome to Super Review! Introduction AA Study Strategies AA Content Review Peptide Bond AA Separation & Purification MCAT Passage What Next? How Can Next Step Help? Introduction to Super Review
Next Step Welcome to Super Review! Introduction AA Study Strategies AA Content Review Peptide Bond AA Separation & Purification MCAT Passage What Next? How Can Next Step Help? Introduction to Super Review
Proton Acidity. (b) For the following reaction, draw the arrowhead properly to indicate the position of the equilibrium: HA + K + B -
 Proton Acidity A01 Given that acid A has a pk a of 15 and acid B has a pk a of 10, then: (a) Which of the two acids is stronger? (b) For the following reaction, draw the arrowhead properly to indicate
Proton Acidity A01 Given that acid A has a pk a of 15 and acid B has a pk a of 10, then: (a) Which of the two acids is stronger? (b) For the following reaction, draw the arrowhead properly to indicate
CHEMISTRY ATAR COURSE DATA BOOKLET
 CHEMISTRY ATAR COURSE DATA BOOKLET 2018 2018/2457 Chemistry ATAR Course Data Booklet 2018 Table of contents Periodic table of the elements...3 Formulae...4 Units...4 Constants...4 Solubility rules for
CHEMISTRY ATAR COURSE DATA BOOKLET 2018 2018/2457 Chemistry ATAR Course Data Booklet 2018 Table of contents Periodic table of the elements...3 Formulae...4 Units...4 Constants...4 Solubility rules for
C CH 3 N C COOH. Write the structural formulas of all of the dipeptides that they could form with each other.
 hapter 25 Biochemistry oncept heck 25.1 Two common amino acids are 3 2 N alanine 3 2 N threonine Write the structural formulas of all of the dipeptides that they could form with each other. The carboxyl
hapter 25 Biochemistry oncept heck 25.1 Two common amino acids are 3 2 N alanine 3 2 N threonine Write the structural formulas of all of the dipeptides that they could form with each other. The carboxyl
Molecular Selective Binding of Basic Amino Acids by a Water-soluble Pillar[5]arene
![Molecular Selective Binding of Basic Amino Acids by a Water-soluble Pillar[5]arene Molecular Selective Binding of Basic Amino Acids by a Water-soluble Pillar[5]arene](/thumbs/77/75727507.jpg) Electronic supplementary information Molecular Selective Binding of Basic Amino Acids y a Water-solule Pillar[5]arene Chunju Li,* a, Junwei Ma, a Liu Zhao, a Yanyan Zhang, c Yihua Yu, c Xiaoyan Shu, a
Electronic supplementary information Molecular Selective Binding of Basic Amino Acids y a Water-solule Pillar[5]arene Chunju Li,* a, Junwei Ma, a Liu Zhao, a Yanyan Zhang, c Yihua Yu, c Xiaoyan Shu, a
Protein Struktur (optional, flexible)
 Protein Struktur (optional, flexible) 22/10/2009 [ 1 ] Andrew Torda, Wintersemester 2009 / 2010, AST nur für Informatiker, Mathematiker,.. 26 kt, 3 ov 2009 Proteins - who cares? 22/10/2009 [ 2 ] Most important
Protein Struktur (optional, flexible) 22/10/2009 [ 1 ] Andrew Torda, Wintersemester 2009 / 2010, AST nur für Informatiker, Mathematiker,.. 26 kt, 3 ov 2009 Proteins - who cares? 22/10/2009 [ 2 ] Most important
The Structure of Enzymes!
 The Structure of Enzymes Levels of Protein Structure 0 order amino acid composition Primary Secondary Motifs Tertiary Domains Quaternary ther sequence repeating structural patterns defined by torsion angles
The Structure of Enzymes Levels of Protein Structure 0 order amino acid composition Primary Secondary Motifs Tertiary Domains Quaternary ther sequence repeating structural patterns defined by torsion angles
The Structure of Enzymes!
 The Structure of Enzymes Levels of Protein Structure 0 order amino acid composition Primary Secondary Motifs Tertiary Domains Quaternary ther sequence repeating structural patterns defined by torsion angles
The Structure of Enzymes Levels of Protein Structure 0 order amino acid composition Primary Secondary Motifs Tertiary Domains Quaternary ther sequence repeating structural patterns defined by torsion angles
Lecture 14 - Cells. Astronomy Winter Lecture 14 Cells: The Building Blocks of Life
 Lecture 14 Cells: The Building Blocks of Life Astronomy 141 Winter 2012 This lecture describes Cells, the basic structural units of all life on Earth. Basic components of cells: carbohydrates, lipids,
Lecture 14 Cells: The Building Blocks of Life Astronomy 141 Winter 2012 This lecture describes Cells, the basic structural units of all life on Earth. Basic components of cells: carbohydrates, lipids,
Biomolecules: lecture 9
 Biomolecules: lecture 9 - understanding further why amino acids are the building block for proteins - understanding the chemical properties amino acids bring to proteins - realizing that many proteins
Biomolecules: lecture 9 - understanding further why amino acids are the building block for proteins - understanding the chemical properties amino acids bring to proteins - realizing that many proteins
1014NSC Fundamentals of Biochemistry Semester Summary
 1014NSC Fundamentals of Biochemistry Semester Summary Griffith University, Nathan Campus Semester 1, 2014 Topics include: - Water & ph - Protein Diversity - Nucleic Acids - DNA Replication - Transcription
1014NSC Fundamentals of Biochemistry Semester Summary Griffith University, Nathan Campus Semester 1, 2014 Topics include: - Water & ph - Protein Diversity - Nucleic Acids - DNA Replication - Transcription
QUESTION 1 Which two functional groups react to form the peptide link found in proteins?
 QUESTION 1 Which two functional groups react to form the peptide link found in proteins? NH and NH and NH2 and and NH2 A 2 B 2 C D OH COOH OH COOH QUESTION 2 The elements present in proteins are A B C
QUESTION 1 Which two functional groups react to form the peptide link found in proteins? NH and NH and NH2 and and NH2 A 2 B 2 C D OH COOH OH COOH QUESTION 2 The elements present in proteins are A B C
SEQUENCE ALIGNMENT BACKGROUND: BIOINFORMATICS. Prokaryotes and Eukaryotes. DNA and RNA
 SEQUENCE ALIGNMENT BACKGROUND: BIOINFORMATICS 1 Prokaryotes and Eukaryotes 2 DNA and RNA 3 4 Double helix structure Codons Codons are triplets of bases from the RNA sequence. Each triplet defines an amino-acid.
SEQUENCE ALIGNMENT BACKGROUND: BIOINFORMATICS 1 Prokaryotes and Eukaryotes 2 DNA and RNA 3 4 Double helix structure Codons Codons are triplets of bases from the RNA sequence. Each triplet defines an amino-acid.
B O C 4 H 2 O O. NOTE: The reaction proceeds with a carbonium ion stabilized on the C 1 of sugar A.
 hbcse 33 rd International Page 101 hemistry lympiad Preparatory 05/02/01 Problems d. In the hydrolysis of the glycosidic bond, the glycosidic bridge oxygen goes with 4 of the sugar B. n cleavage, 18 from
hbcse 33 rd International Page 101 hemistry lympiad Preparatory 05/02/01 Problems d. In the hydrolysis of the glycosidic bond, the glycosidic bridge oxygen goes with 4 of the sugar B. n cleavage, 18 from
CHEM 3653 Exam # 1 (03/07/13)
 1. Using phylogeny all living organisms can be divided into the following domains: A. Bacteria, Eukarya, and Vertebrate B. Archaea and Eukarya C. Bacteria, Eukarya, and Archaea D. Eukarya and Bacteria
1. Using phylogeny all living organisms can be divided into the following domains: A. Bacteria, Eukarya, and Vertebrate B. Archaea and Eukarya C. Bacteria, Eukarya, and Archaea D. Eukarya and Bacteria
Protein Structure. Role of (bio)informatics in drug discovery. Bioinformatics
 Bioinformatics Protein Structure Principles & Architecture Marjolein Thunnissen Dep. of Biochemistry & Structural Biology Lund University September 2011 Homology, pattern and 3D structure searches need
Bioinformatics Protein Structure Principles & Architecture Marjolein Thunnissen Dep. of Biochemistry & Structural Biology Lund University September 2011 Homology, pattern and 3D structure searches need
PROTEIN SECONDARY STRUCTURE PREDICTION: AN APPLICATION OF CHOU-FASMAN ALGORITHM IN A HYPOTHETICAL PROTEIN OF SARS VIRUS
 Int. J. LifeSc. Bt & Pharm. Res. 2012 Kaladhar, 2012 Research Paper ISSN 2250-3137 www.ijlbpr.com Vol.1, Issue. 1, January 2012 2012 IJLBPR. All Rights Reserved PROTEIN SECONDARY STRUCTURE PREDICTION:
Int. J. LifeSc. Bt & Pharm. Res. 2012 Kaladhar, 2012 Research Paper ISSN 2250-3137 www.ijlbpr.com Vol.1, Issue. 1, January 2012 2012 IJLBPR. All Rights Reserved PROTEIN SECONDARY STRUCTURE PREDICTION:
Q.8. Isomers are the compounds that must have same
 Choose the single correct answer for each of the following questions: (1 Mark each). Q. 1. ow many lone pairs are present in C 3 --C 3 i) 3 ii) 2 iii) 1 iv) 4 Q.2. When any hydrocarbon is added to water,
Choose the single correct answer for each of the following questions: (1 Mark each). Q. 1. ow many lone pairs are present in C 3 --C 3 i) 3 ii) 2 iii) 1 iv) 4 Q.2. When any hydrocarbon is added to water,
ANSWERS TO CASE STUDIES Chapter 2: Drug Design and Relationship of Functional Groups to Pharmacologic Activity
 ANSWERS TO CASE STUDIES Chapter 2: Drug Design and Relationship of Functional Groups to Pharmacologic Activity Absorption/Acid-Base Case (p. 42) Question #1: Drug Cetirizine Clemastin e Functional groups
ANSWERS TO CASE STUDIES Chapter 2: Drug Design and Relationship of Functional Groups to Pharmacologic Activity Absorption/Acid-Base Case (p. 42) Question #1: Drug Cetirizine Clemastin e Functional groups
ADSORPTION OF TWENTY BIO-AMINO ACIDS BY NATURAL ALLOPHANE AND IMOGOLITE
 ADSORPTION OF TWENTY BIO-AMINO ACIDS BY NATURAL ALLOPHANE AND IMOGOLITE Hashizume Hideo National Institute for Materials Science, 1-1 Namiki, Tsukuba, Ibaraki 305-0044, Japan Fax: +81-29-860-4667 E-mail:
ADSORPTION OF TWENTY BIO-AMINO ACIDS BY NATURAL ALLOPHANE AND IMOGOLITE Hashizume Hideo National Institute for Materials Science, 1-1 Namiki, Tsukuba, Ibaraki 305-0044, Japan Fax: +81-29-860-4667 E-mail:
Problem Set 1
 2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
Collision Cross Section: Ideal elastic hard sphere collision:
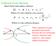 Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
6. Compounds with amine groups
 6. ompounds with amine groups 6.1. lassifying amines and amides 6.2 Properties of amines 6.3 Amine mechanisms 6.4 Amino acids 6.5 Amino acids and p 6.6 Amine preparation Answers ompounds with amine groups
6. ompounds with amine groups 6.1. lassifying amines and amides 6.2 Properties of amines 6.3 Amine mechanisms 6.4 Amino acids 6.5 Amino acids and p 6.6 Amine preparation Answers ompounds with amine groups
Protein Struktur. Biologen und Chemiker dürfen mit Handys spielen (leise) go home, go to sleep. wake up at slide 39
 Protein Struktur Biologen und Chemiker dürfen mit Handys spielen (leise) go home, go to sleep wake up at slide 39 Andrew Torda, Wintersemester 2016/ 2017 Andrew Torda 17.10.2016 [ 1 ] Proteins - who cares?
Protein Struktur Biologen und Chemiker dürfen mit Handys spielen (leise) go home, go to sleep wake up at slide 39 Andrew Torda, Wintersemester 2016/ 2017 Andrew Torda 17.10.2016 [ 1 ] Proteins - who cares?
1-How can we distinguish between D-amino acid and L-amino acid?
 AMINO ACIDS (2) Questions from the previous lecture : 1-How can we distinguish between D-amino acid and L-amino acid? According to the location of the amino group in the amino acid, the R group is above,
AMINO ACIDS (2) Questions from the previous lecture : 1-How can we distinguish between D-amino acid and L-amino acid? According to the location of the amino group in the amino acid, the R group is above,
Rotamers in the CHARMM19 Force Field
 Appendix A Rotamers in the CHARMM19 Force Field The people may be made to follow a path of action, but they may not be made to understand it. Confucius (551 BC - 479 BC) ( ) V r 1 (j),r 2 (j),r 3 (j),...,r
Appendix A Rotamers in the CHARMM19 Force Field The people may be made to follow a path of action, but they may not be made to understand it. Confucius (551 BC - 479 BC) ( ) V r 1 (j),r 2 (j),r 3 (j),...,r
26.7 Laboratory Synthesis of Peptides
 S Hornback_Ch26_1123-1161 12/15/04 8:18 PM Page 1148 1148 CHAPTER 26 AMI ACIDS, PEPTIDES, AD PRTEIS A chain B chain Gly Ile Val Glu Intramolecular disulfide bridge Gln Cys S S Cys Thr Ser Ile Cys Ser Leu
S Hornback_Ch26_1123-1161 12/15/04 8:18 PM Page 1148 1148 CHAPTER 26 AMI ACIDS, PEPTIDES, AD PRTEIS A chain B chain Gly Ile Val Glu Intramolecular disulfide bridge Gln Cys S S Cys Thr Ser Ile Cys Ser Leu
Buffer solutions المحاليل المنظمة
 Buffer solutions المحاليل المنظمة Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Understanding ph balance The human
Buffer solutions المحاليل المنظمة Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Understanding ph balance The human
Packing of Secondary Structures
 7.88 Lecture Notes - 4 7.24/7.88J/5.48J The Protein Folding and Human Disease Professor Gossard Retrieving, Viewing Protein Structures from the Protein Data Base Helix helix packing Packing of Secondary
7.88 Lecture Notes - 4 7.24/7.88J/5.48J The Protein Folding and Human Disease Professor Gossard Retrieving, Viewing Protein Structures from the Protein Data Base Helix helix packing Packing of Secondary
Chemistry 224 Bioorganic Chemistry Friday, Sept. 29, This Exam is closed book and closed notes. Please show all your work!
 page 1 of 6 hemistry 224 ame Bioorganic hemistry Friday, ept. 29, 2000 Exam 1 100 points This Exam is closed book and closed notes Please show all your work! tereochemistry counts as indicated! eatness
page 1 of 6 hemistry 224 ame Bioorganic hemistry Friday, ept. 29, 2000 Exam 1 100 points This Exam is closed book and closed notes Please show all your work! tereochemistry counts as indicated! eatness
BIOL/BIOC Come to the PASS workshop with your mock exam complete. During the workshop you can work with other students to review your work.
 It is most beneficial to you to write this mock midterm UNDER EXAM CONDITIONS. This means: Complete the midterm in 1.5 hour(s). Work on your own. Keep your notes and textbook closed. Attempt every question.
It is most beneficial to you to write this mock midterm UNDER EXAM CONDITIONS. This means: Complete the midterm in 1.5 hour(s). Work on your own. Keep your notes and textbook closed. Attempt every question.
المرحلة الثانية- قسم علوم الحياة الجامعة المستنصرية
 1 Biochemistry Amino acids For nd year students, Biology Department, College of Sciences, Mustansiriyah University 018-019 الكيمياء حياتية ٢ االحماض االمينية المرحلة الثانية- قسم علوم الحياة الجامعة المستنصرية
1 Biochemistry Amino acids For nd year students, Biology Department, College of Sciences, Mustansiriyah University 018-019 الكيمياء حياتية ٢ االحماض االمينية المرحلة الثانية- قسم علوم الحياة الجامعة المستنصرية
Tamer Barakat. Razi Kittaneh. Mohammed Bio. Diala Abu-Hassan
 14 Tamer Barakat Razi Kittaneh Mohammed Bio Diala Abu-Hassan Protein structure: We already know that when two amino acids bind, a dipeptide is formed which is considered to be an oligopeptide. When more
14 Tamer Barakat Razi Kittaneh Mohammed Bio Diala Abu-Hassan Protein structure: We already know that when two amino acids bind, a dipeptide is formed which is considered to be an oligopeptide. When more
Practice Final for Chem 104
 Practice Final for Chem 104 These questions cover the material that will be covered on the Final Exam. The distribution of the questions is about the same as the Final Exam. The forms of the questions
Practice Final for Chem 104 These questions cover the material that will be covered on the Final Exam. The distribution of the questions is about the same as the Final Exam. The forms of the questions
1) Which of the following represents the breaking of a noncovalent interaction? Topic: The Nature of Noncovalent Interactions
 Multiple Choice Questions 1) Which of the following represents the breaking of a noncovalent interaction? A) hydrolysis of an ester B) dissolving of salt crystals C) ionization of water D) decomposition
Multiple Choice Questions 1) Which of the following represents the breaking of a noncovalent interaction? A) hydrolysis of an ester B) dissolving of salt crystals C) ionization of water D) decomposition
Discussion Section (Day, Time): TF:
 ame: Chemistry 27 Professor Gavin MacBeath arvard University Spring 2004 Final Exam Thursday, May 28, 2004 2:15 PM - 5:15 PM Discussion Section (Day, Time): Directions: TF: 1. Do not write in red ink.
ame: Chemistry 27 Professor Gavin MacBeath arvard University Spring 2004 Final Exam Thursday, May 28, 2004 2:15 PM - 5:15 PM Discussion Section (Day, Time): Directions: TF: 1. Do not write in red ink.
Towards Understanding the Origin of Genetic Languages
 Towards Understanding the Origin of Genetic Languages Why do living organisms use 4 nucleotide bases and 20 amino acids? Apoorva Patel Centre for High Energy Physics and Supercomputer Education and Research
Towards Understanding the Origin of Genetic Languages Why do living organisms use 4 nucleotide bases and 20 amino acids? Apoorva Patel Centre for High Energy Physics and Supercomputer Education and Research
Separation of Large and Small Peptides by Supercritical Fluid Chromatography and Detection by Mass Spectrometry
 Separation of Large and Small Peptides by Supercritical Fluid Chromatography and Detection by Mass Spectrometry Application Note Biologics and Biosimilars Author Edgar Naegele Agilent Technologies, Inc.
Separation of Large and Small Peptides by Supercritical Fluid Chromatography and Detection by Mass Spectrometry Application Note Biologics and Biosimilars Author Edgar Naegele Agilent Technologies, Inc.
Conceptual toolkit: the molecular principles for understanding proteins
 onceptual toolkit: the molecular principles for understanding proteins 1 Introductory note This chapter is intended to provide a concise summary of the principles with which you should be familiar in order
onceptual toolkit: the molecular principles for understanding proteins 1 Introductory note This chapter is intended to provide a concise summary of the principles with which you should be familiar in order
