SoilGen2 model: data requirements and model output
|
|
|
- Dylan Bradley
- 5 years ago
- Views:
Transcription
1 SoilGen2 model: data requirements and model output 1. Essential plot data Essential soil data Precipitation data (for a typical year) Evapotranspiration and Air temperature data (for a typical year) Climate and vegetation data (timeseries) Weathering rates and initial cation pools locked in primary minerals Chemical equilibrium constants and activation energy Timing for C-inputs per vegetation type in a typical year History of atmospheric pco2-levels (time series) Bioturbation history as intensity with depth (time series) Fertilization history (time series) Event history Process constants Model outputs... 4 The below table links the data categories to files. The yellow-marked data groups are the most essential to be able to run SoilGen in a local context. group (numbers refer to list below) File m/v 1. Essential plot data Input via user interface or <SoilGenBatch.txt> M 2. Essential soil data 3. Precipitation data for typical year Soil input file coupled via user interface or <SoilGenBatch.txt> M 4. Evapotranspiration and Air temperature data for a typical year 5. Climate and vegetation data (time series) File coupled via user interface (default: <climate.txt> ) V 6. Initial status minerals File coupled via user interface or <SoilGenBatch.txt> M 7. Chemical equilibrium constants and activation energy File coupled via user interface or <SoilGenBatch.txt> M 8. Timing for C-inputs per vegetation type in a typical year File coupled via user interface or <SoilGenBatch.txt> M 9. Atmospheric pco 2-levels (time series) File coupled via user interface V 10. Bioturbation intensity with depth (time series) File coupled via user interface (default: <BioturbINT.txt> ) V 11. Fertilization data (time series) File coupled via user interface (default: <Fertilization.txt> ) V 12. Event data (erosion, deposition, water table, change in hydraulic File coupled via user interface (default: <Events.txt> ) V characteristics, plowing, SlashBurn) 13. Various process constants File modifiable via user interface (default: <SoftConstants.txt> ) V M=mandatory file; V=not mandatory Detailed information can be found in the user guide at 1. Essential plot data Slope angle (degrees) Upslope bearing (degrees) Downwind bearing (degrees) Latitude (degrees) 1
2 2. Essential soil data Per soil layer: Starting depth Ending depth cm Initial Clay% Initial Silt% Initial Sand% Initial OC% Mass% solid fraction, should sum to 100% Initial Bulk density kg dm -3 Initial moisture content dm 3 dm -3 Initial temperature C Initial calcite content Initial gypsum content Mass fraction mineral phase Initial pco 2 bar Initial CEC ol + kg -1 Mg/Ca Ca/Na Ca/K Ca/Al Ca/H Selectivity Coefficients Initial amounts in solution Ca, Mg, K, Na, Al Cl, SO4, Alkalinity ol dm 3 Initial amount at exchange complex Ca, Mg, K, Na, H, Al ol + kg Precipitation data (for a typical year) Per Julian day Fraction of day where precipitation starts Amount of precipitation Surface flux density d -1 Water composition Ca, Mg, Na, K, Cl, SO4, Alkalinity, Al ol dm Evapotranspiration and Air temperature data (for a typical year) Per week Potential evapotranspiration (sum for the week) Mean daily temperature C (calculated over the week) Mean daily amplitude C (calculated over the week) 5. Climate and vegetation data (timeseries) For specific years along the timeline (missing values will be interpolated) YearBP P EP JanT JulT PlantsInC(t/ha,y) ManureInC(t/ha,y) Vegetation Period Annual precipitation Annual potential evapotranspiration Average January temperature C Average July temperature C Annual C-input (root and leaf litter or crop residue) of plants kg.1000 ha -1 y -1 Annual C-input as organic manure (SoilGen2.25+: NPP can be estimated in user interface) kg.1000 ha -1 y -1 Vegetation type (4 types possible) 6. Weathering rates and initial cation pools locked in primary minerals SoilGen2.24: the profile is assumed to be homogeneous for primary minerals! Weathering rate Ca from primary minerals m 3 mol -1 c y -1 Weathering rate Mg from primary minerals m 3 mol -1 c y -1 Weathering rate K from primary minerals m 3 mol -1 c y -1 Weathering rate Na from primary minerals m 3 mol -1 c y -1 2
3 coefficient for congruent weathering Anorthite (Ca) with respect to Al - coefficient for congruent weathering Chlorite (Mg) with respect to Al - coefficient for congruent weathering Microcline (K) with respect to Al - coefficient for congruent weathering Albite (Na) with respect to Al - Initial free" Al; initial Gibsite pool) Initial Ca (e.g. from total analysis corrected for Ca not in primary silicate minerals) Initial Mg (e.g. from total analysis corrected for Mg not in primary silicate minerals) Initial K (e.g. from total analysis corrected for K not in primary silicate minerals) Initial Na (e.g. from total analysis corrected for Na not in primary silicate minerals) SoilGen2.25 and later: (per layer) Units Albite (NaAlSi3O8) K-feldspar (KAlSi3O8) Muscovite (KAl3Si3O10(OH)2) Biotite (KMg1.5Fe1.5AlSi3O10(OH)2) Quartz (SiO2) Chlorite As Clinochlore (Mg5Al2Si3O10 (OH)8) Anorthite (CaAl2Si2O8) Fosterite (Mg2SiO4) Augite As Orthoferrosilite (FeSiO3) Kaolinite (Al2Si2O5(OH)4) Illite (Si3.4O10(OH)2Mg0.02Al2.38Ca0.01K0.44) Montmorillonite (Si4O10(OH)2Mg0.33Al1.67Ca Mg K0.0825Na0.0825) Hornblende (Ca2Mg4AlSi6.7AlO22.4) Fayalite (Fe2SiO4) Gibbsite (Al(OH)3) Amorphite (mineral with input composition in Amorphite.txt) massfraction Otherite (mineral with input composition in Otherite.txt) massfraction 7. Chemical equilibrium constants and activation energy Henrys law constant for CO2 at 25 C and activation energy - and kj mol -1 Stability constant of water at 25 C and activation energy - and kj mol -1 First dissociation constant of H2CO3 at 25 C and activation energy - and kj mol -1 Second dissociation constant of H2CO3 at 25 C and activation energy - and kj mol -1 Stability constant of CaCO3 at 25 C and activation energy - and kj mol -1 Stability constant of CaHCO3+ at 25 C and activation energy - and kj mol -1 Stability constant of CaOH+ at 25 C and activation energy - and kj mol -1 Stability constant of CaSO4 at 25 C and activation energy - and kj mol -1 Stability constant of MgCO3 at 25 C and activation energy - and kj mol -1 Stability constant of MgHCO3+ at 25 C and activation energy - and kj mol -1 Stability constant of MgOH+ at 25 C and activation energy - and kj mol -1 Stability constant of MgSO4 at 25 C and activation energy - and kj mol -1 Stability constant of NaSO4- at 25 C and activation energy - and kj mol -1 Stability constant of NaCO3- at 25 C and activation energy - and kj mol -1 Stability constant of KSO4- at 25 C and activation energy - and kj mol -1 Solubility product of gypsum at 25 C and activation energy - and kj mol -1 Solubility product of calcite at 25 C and activation energy - and kj mol -1 Solubility product of Gibbsite at 25 C and activation energy - and kj mol Timing for C-inputs per vegetation type in a typical year initial profile data: distribution C over various pools (%) (all soil compartments) % Per month in a typical year for each one of 4 vegetation types: Percentage of the plant residues (litter) input to the soil % Percentage of the manure input to the soil % Per vegetation type Maximum rooting depth 3
4 9. History of atmospheric pco2-levels (time series) Year Before Present pco 2 bar 10. Bioturbation history as intensity with depth (time series) Year= simulation year (oldest year = 1); Upper depth of bioturbation (usually 0: the soil surface) Depth of maximal bioturbation Lower depth of bioturbation Magnitude of bioturbation at upper depth Bioturbset.txt: ppt Magnitude of (maximal) bioturbation at depth of maximal bioturbation Bioturbset.txt: ppt Magnitude of bioturbation at lower depth of bioturbation (usually 0). Bioturbset.txt: ppt 11. Fertilization history (time series) Year Before Present Ca Mg Na K Cl SO4 HCO3 CO3 mol m Event history Per year one option of the following (or nothing) Simulation year, Deposition, nr compartments added, Clay Silt OC Units as in soil data file BulkDensity MoistureContent Calcite Gypsum pco2 Ca-sol Mg-sol Na-sol K- sol Cl-sol SO4-sol Alkalinity Al-sol Org3-sol HOrg-sol H2Org-sol DOC Ca-exch Mg-exch Na-exch K-exch H-exch Al-exch CEC Mg/Ca-select Ca/Na-select Ca/K-select Ca/Al-select Ca/H-select (longer list including minerals for SoilGen2.25+) Simulation year, Erosion, nr compartments removed Simulation year, WaterTable, depth as nr compartments Simulation year, VGNChange, nr of compartments involved, <filename> <filename> points to a file with Van Genuchten parameters that may have been measured in a certain year Simulation year, Plowing, nr of compartments involved, mixed by plowing Simulation year, SlashBurn, nr of compartments involved, c lost to atmosphere (=CO2), c lost to charcoal (=IOM), c lost to humus 13. Process constants This file is only to be changed when the model is to be calibrated. See the user manual at Model outputs Soil variable Units Soil variable Units Water Chemistry 4
5 Potential (kpa) Cation Exchange Capacity (ol+ / kg soil) Water content (cm.3 / cm.3) exchangeable cations (K, Na, (ol+ / kg soil) Mg, Ca, Al) Flux () Base Saturation (%) Evapotranspiration () Ions in solution (K, Na, Ca, Mg, Al, HCO3, CO3, Cl, SO4) Plant-available water (cm.3 / cm.3) ph, ph1:5 (-log mol / l) Mineralogy and weathering indices Alkalinity (ol- / l) Elements in minerals (mol / m2) Sodium Adsorption Ratio (-) (Ca, Mg, Na, K, Fe, Si, Al) Minerals (17 predefined, (kg / m2) EC (ms / m) plus 2 user- defined minerals) CIA (% Nesbitt+Young1982) ESP (%) IndexB (- Kronberg+Nesbitt1981) Biotics CIW (% Harnois1988) Roots (fraction of total) CPA (% BuggleEtAl2011) OC (mass % solid fraction) PIA (% FedoEtAl1995) Decomposable Plant Material (Mg / ha) DPM WI (- PriceEtAl1991) Resistant Plant Material RPM (Mg / ha) Total Reserve Of (cmol+ / kg soil) Biomass BIO (Mg / ha) Bases Physics Humus HUM (Mg / ha) bulk density (kg / dm.3) Inert Organic Matter IOM (Mg / ha) Clay (mass% fine earth) pco2 (bar) Silt (mass% fine earth) delta14c (per mil) Sand (mass% fine earth) 14C-age (14C years before simulation year) Porosity (volume fraction) 14C-per mil contributions per (14C per mil / C in pool) pool (DPM, RPM, BIO, HUM, IOM) Clay (volume%) Clay Dispersion (-) Indicator Temperature ( C) 5
Adsorption of ions Ion exchange CEC& AEC Factors influencing ion
 Adsorption of ions Ion exchange CEC& AEC Factors influencing ion exchange- Significance. Adsorption of ions Ion adsorption and subsequent exchange are important processes that take place between soil colloidal
Adsorption of ions Ion exchange CEC& AEC Factors influencing ion exchange- Significance. Adsorption of ions Ion adsorption and subsequent exchange are important processes that take place between soil colloidal
Acid Soil. Soil Acidity and ph
 Acid Soil Soil Acidity and ph ph ph = - log (H + ) H 2 O H + + OH - (H + ) x (OH - )= K w = 10-14 measures H + activity with an electrode (in the lab), solutions (in the field) reflects the acid intensity,
Acid Soil Soil Acidity and ph ph ph = - log (H + ) H 2 O H + + OH - (H + ) x (OH - )= K w = 10-14 measures H + activity with an electrode (in the lab), solutions (in the field) reflects the acid intensity,
The Lithosphere. Definition
 10/14/2014 www.komar.de The Lithosphere Ben Sullivan, Assistant Professor NRES 765, Biogeochemistry October 14th, 2014 Contact: bsullivan@cabnr.unr.edu Definition io9.com tedquarters.net Lithos = rocky;
10/14/2014 www.komar.de The Lithosphere Ben Sullivan, Assistant Professor NRES 765, Biogeochemistry October 14th, 2014 Contact: bsullivan@cabnr.unr.edu Definition io9.com tedquarters.net Lithos = rocky;
organisms CaCO 3 + H 2 O + CO 2 shallow water
 Weathering and Reverse weathering Step I:Weathering of igneous rocks 1. Igneous rocks are mainly composed of Al, Si and O 2 with minor and varying quantities of Na, K, Ca and Mg composing pheldspar minerals
Weathering and Reverse weathering Step I:Weathering of igneous rocks 1. Igneous rocks are mainly composed of Al, Si and O 2 with minor and varying quantities of Na, K, Ca and Mg composing pheldspar minerals
(4) Give an example of important reactions that are responsible for the composition of river water.
 Lecture 12 Global Biogeochemical Cycles (1) If rivers are the chief source of the dissolved salts in seawater, why is seawater not simply a concentrated version of average composition of all rivers? The
Lecture 12 Global Biogeochemical Cycles (1) If rivers are the chief source of the dissolved salts in seawater, why is seawater not simply a concentrated version of average composition of all rivers? The
Soil Colloidal Chemistry. Compiled and Edited by Dr. Syed Ismail, Marthwada Agril. University Parbhani,MS, India
 Soil Colloidal Chemistry Compiled and Edited by Dr. Syed Ismail, Marthwada Agril. University Parbhani,MS, India 1 The Colloidal Fraction Introduction What is a colloid? Why this is important in understanding
Soil Colloidal Chemistry Compiled and Edited by Dr. Syed Ismail, Marthwada Agril. University Parbhani,MS, India 1 The Colloidal Fraction Introduction What is a colloid? Why this is important in understanding
Continent-Ocean Interaction: Role of Weathering
 Institute of Astrophysics and Geophysics (Build. B5c) Room 0/13 email: Guy.Munhoven@ulg.ac.be Phone: 04-3669771 28th February 2018 Organisation of the Lecture 1 Carbon cycle processes time scales modelling:
Institute of Astrophysics and Geophysics (Build. B5c) Room 0/13 email: Guy.Munhoven@ulg.ac.be Phone: 04-3669771 28th February 2018 Organisation of the Lecture 1 Carbon cycle processes time scales modelling:
Copyright SOIL STRUCTURE and CLAY MINERALS
 SOIL STRUCTURE and CLAY MINERALS Soil Structure Structure of a soil may be defined as the mode of arrangement of soil grains relative to each other and the forces acting between them to hold them in their
SOIL STRUCTURE and CLAY MINERALS Soil Structure Structure of a soil may be defined as the mode of arrangement of soil grains relative to each other and the forces acting between them to hold them in their
Effect of chemical composition to large scale CO 2 Injection in Morrow Sandstone, Farnsworth Hydrocarbon Field, Texas, USA
 Effect of chemical composition to large scale CO 2 Injection in Morrow Sandstone, Farnsworth Hydrocarbon Field, Texas, USA Bulbul Ahmmed Martin Appold Department of Geological Sciences University of Missouri-Columbia
Effect of chemical composition to large scale CO 2 Injection in Morrow Sandstone, Farnsworth Hydrocarbon Field, Texas, USA Bulbul Ahmmed Martin Appold Department of Geological Sciences University of Missouri-Columbia
Sedimentary Rocks and Processes
 Sedimentary Rocks and Processes Weathering Sedimentary Processes Breakdown of pre-existing rock by physical and chemical processes Transport Movement of sediments from environments of relatively high potential
Sedimentary Rocks and Processes Weathering Sedimentary Processes Breakdown of pre-existing rock by physical and chemical processes Transport Movement of sediments from environments of relatively high potential
Lecture 4 What Controls the Composition of Seawater
 Lecture 4 What Controls the Composition of Seawater Seawater is salty! Why? What controls the composition of seawater? Do Chemical Equilibrium reactions control the composition of the Ocean? What is meant
Lecture 4 What Controls the Composition of Seawater Seawater is salty! Why? What controls the composition of seawater? Do Chemical Equilibrium reactions control the composition of the Ocean? What is meant
Capabilities of TOUGH Codes for Modeling Geologic Sequestration and Leakage of CO 2
 Capabilities of TOUGH Codes for Modeling Geologic Sequestration and Leakage of CO 2 Karsten Pruess Earth Sciences Division Lawrence Berkeley National Laboratory Presented at Workshop on Leakage Modeling
Capabilities of TOUGH Codes for Modeling Geologic Sequestration and Leakage of CO 2 Karsten Pruess Earth Sciences Division Lawrence Berkeley National Laboratory Presented at Workshop on Leakage Modeling
Lecture 6. Physical Properties. Solid Phase. Particle Composition
 Lecture 6 Physical Properties Solid Phase Particle Composition 1 Questions What are tetrahedrons and octahedrons? How do silica tetrahedra bonds affect mineral weathering? Difference between primary and
Lecture 6 Physical Properties Solid Phase Particle Composition 1 Questions What are tetrahedrons and octahedrons? How do silica tetrahedra bonds affect mineral weathering? Difference between primary and
Volume Composition of a Desirable Surface Soil
 Soil Chemistry Volume Composition of a Desirable Surface Soil 50% pore space 25% air 45 to 48% mineral matter 50% solid material 25% water 2 to 5% organic matter Soil Organic Matter Soil organic matter:
Soil Chemistry Volume Composition of a Desirable Surface Soil 50% pore space 25% air 45 to 48% mineral matter 50% solid material 25% water 2 to 5% organic matter Soil Organic Matter Soil organic matter:
Igneous rocks + acid volatiles = sedimentary rocks + salty oceans
 The Lithosphere Weathering physical processes chemical processes biological processes weathering rates Soil development soil formation processes types of soils and vegetation soil properties physical chemical
The Lithosphere Weathering physical processes chemical processes biological processes weathering rates Soil development soil formation processes types of soils and vegetation soil properties physical chemical
CO 2 -water-rock reactivity at hydrothermal temperatures: The BigRig2 experiment
 CO 2 -water-rock reactivity at hydrothermal temperatures: The BigRig2 experiment C.A. ROCHELLE 1 *, K. BATEMAN 1, A. LACINSKA 1, D. WAGNER 1, J. LIONS 2 AND I. GAUS 2 1 British Geological Survey, Keyworth,
CO 2 -water-rock reactivity at hydrothermal temperatures: The BigRig2 experiment C.A. ROCHELLE 1 *, K. BATEMAN 1, A. LACINSKA 1, D. WAGNER 1, J. LIONS 2 AND I. GAUS 2 1 British Geological Survey, Keyworth,
Soil ph: Review of Concepts
 Soils and Water, Spring 008 Soil ph: Review of Concepts Acid: substance that can donate a proton Base: substance that can accept a proton HA H A HA and A - are called conjugate acid-base pairs. The strength
Soils and Water, Spring 008 Soil ph: Review of Concepts Acid: substance that can donate a proton Base: substance that can accept a proton HA H A HA and A - are called conjugate acid-base pairs. The strength
Weathering and Soils
 OCN 401-17 Aug. 29, 2016 KCR Weathering and Soils Biogeochemistry Chapter 4: The Lithosphere Introduction: the context Rock Weathering Soil Chemical Reactions Soil Development (see text) Weathering Rates
OCN 401-17 Aug. 29, 2016 KCR Weathering and Soils Biogeochemistry Chapter 4: The Lithosphere Introduction: the context Rock Weathering Soil Chemical Reactions Soil Development (see text) Weathering Rates
Lecture 15: Adsorption; Soil Acidity
 Lecture 15: Adsorption; Soil Acidity Surface Complexation (Your textbook calls this adsorption ) Surface Complexation Both cations and anions can bind to sites on the external surfaces of soil minerals
Lecture 15: Adsorption; Soil Acidity Surface Complexation (Your textbook calls this adsorption ) Surface Complexation Both cations and anions can bind to sites on the external surfaces of soil minerals
CLASS EXERCISE 5.1 List processes occurring in soils that cause changes in the levels of ions.
 5 SIL CHEMISTRY 5.1 Introduction A knowledge of the chemical composition of a soil is less useful than a knowledge of its component minerals and organic materials. These dictate the reactions that occur
5 SIL CHEMISTRY 5.1 Introduction A knowledge of the chemical composition of a soil is less useful than a knowledge of its component minerals and organic materials. These dictate the reactions that occur
Soil Fertility. Fundamentals of Nutrient Management June 1, Patricia Steinhilber
 Soil Fertility Fundamentals of Nutrient Management June 1, 2010 Patricia Steinhilber Ag Nutrient Management Program University of Maryland College Park Main Topics plant nutrition functional soil model
Soil Fertility Fundamentals of Nutrient Management June 1, 2010 Patricia Steinhilber Ag Nutrient Management Program University of Maryland College Park Main Topics plant nutrition functional soil model
Sedimentary Geology. Strat and Sed, Ch. 1 1
 Sedimentary Geology Strat and Sed, Ch. 1 1 Sedimentology vs. Stratigraphy Sedimentology is the study of the origin and classification of sediments and sedimentary rocks Mostly the physical and chemical
Sedimentary Geology Strat and Sed, Ch. 1 1 Sedimentology vs. Stratigraphy Sedimentology is the study of the origin and classification of sediments and sedimentary rocks Mostly the physical and chemical
INTRODUCTION TO GEOCHEMICAL AND REACTIVE TRANSPORT MODELING. Ondra Sracek
 INTRODUCTION TO GEOCHEMICAL AND REACTIVE TRANSPORT MODELING Ondra Sracek Principal types of geochemical modeling: 1. Speciation (for exemple, for lead); 2. Inverse geochemical modeling; 3. Direct geochemical
INTRODUCTION TO GEOCHEMICAL AND REACTIVE TRANSPORT MODELING Ondra Sracek Principal types of geochemical modeling: 1. Speciation (for exemple, for lead); 2. Inverse geochemical modeling; 3. Direct geochemical
Lab 8 Dynamic Soil Systems I: Soil ph and Liming
 Lab 8 Dynamic Soil Systems I: Soil ph and Liming Objectives: To measure soil ph and observe conditions which change ph To distinguish between active acidity (soil solution ph) and exchangeable acidity
Lab 8 Dynamic Soil Systems I: Soil ph and Liming Objectives: To measure soil ph and observe conditions which change ph To distinguish between active acidity (soil solution ph) and exchangeable acidity
Gain a better understanding of soil ph and how it is measured. Understand how lime requirement is determined.
 LABORATORY 7 SOIL REACTION (ph) AND LIME REQUIREMENT I Objectives Gain a better understanding of soil ph and how it is measured. Understand how lime requirement is determined. II Introduction A Soil Reaction
LABORATORY 7 SOIL REACTION (ph) AND LIME REQUIREMENT I Objectives Gain a better understanding of soil ph and how it is measured. Understand how lime requirement is determined. II Introduction A Soil Reaction
The Lithosphere. Definition
 10/12/2015 www.komar.de The Lithosphere Ben Sullivan, Assistant Professor NRES 765, Biogeochemistry October 14th, 2015 Contact: bsullivan@cabnr.unr.edu Definition io9.com tedquarters.net Lithos = rocky;
10/12/2015 www.komar.de The Lithosphere Ben Sullivan, Assistant Professor NRES 765, Biogeochemistry October 14th, 2015 Contact: bsullivan@cabnr.unr.edu Definition io9.com tedquarters.net Lithos = rocky;
Create custom rock (Rock1) and fluid (Fluid1) compositions. 1. Copy the folder Module3 to your project folder located in Library\Gems3\projects.
 MODULE 3: GREISEN ALTERATION In this tutorial we will use the GEMS project file Module3 in the examples to model the reaction path of a leucogranite during greisenization and evaluate the solubility of
MODULE 3: GREISEN ALTERATION In this tutorial we will use the GEMS project file Module3 in the examples to model the reaction path of a leucogranite during greisenization and evaluate the solubility of
Cation Exchange Capacity, CEC
 Cation Exchange Capacity, CEC The basic building blocks of clay minerals are: silicon atoms surrounded by four oxygen atoms (tetrahedra), and aluminium atoms surrounded by six hydroxide groups (dioctahedra),
Cation Exchange Capacity, CEC The basic building blocks of clay minerals are: silicon atoms surrounded by four oxygen atoms (tetrahedra), and aluminium atoms surrounded by six hydroxide groups (dioctahedra),
T6 soil base cation weathering rates
 T6 soil base cation weathering rates julian aherne :: trent university FORFLUX :: biogeochemistry of irish forests [RSF 07510] Advisory Group Meeting [5 6 December 2011] objective (a) to determine the
T6 soil base cation weathering rates julian aherne :: trent university FORFLUX :: biogeochemistry of irish forests [RSF 07510] Advisory Group Meeting [5 6 December 2011] objective (a) to determine the
Alleviating Soil Acidity through Organic Matter Management. Malcolm E. Sumner, University of Georgia Marcos A Pavan IAPAR
 Alleviating Soil Acidity through Organic Matter Management Malcolm E. Sumner, University of Georgia Marcos A Pavan IAPAR Introduction Most work on topsoil acidity amelioration Mechanical lime incorporation
Alleviating Soil Acidity through Organic Matter Management Malcolm E. Sumner, University of Georgia Marcos A Pavan IAPAR Introduction Most work on topsoil acidity amelioration Mechanical lime incorporation
2.2 Acid mine drainage
 PROBLEM SOLVING 2. CHEMICAL REACTIONS AND EQUILIBRIA 1 2.2 Acid mine drainage Problem 2.2 The weathering of iron sulphide minerals produces acidified water, leading to major environmental problems from
PROBLEM SOLVING 2. CHEMICAL REACTIONS AND EQUILIBRIA 1 2.2 Acid mine drainage Problem 2.2 The weathering of iron sulphide minerals produces acidified water, leading to major environmental problems from
Be sure to show all calculations so that you can receive partial credit for your work!
 Agronomy 365T Exam 1 Spring 2004 Exam Score: Name TA Lab Hour Be sure to show all calculations so that you can receive partial credit for your work! 1) List 14 of the plant essential nutrient for plant
Agronomy 365T Exam 1 Spring 2004 Exam Score: Name TA Lab Hour Be sure to show all calculations so that you can receive partial credit for your work! 1) List 14 of the plant essential nutrient for plant
Weathering and Soils
 OCN 401-11 Oct. 13, 2011 KCR Weathering and Soils Biogeochemistry Chapter 4: The Lithosphere Introduction: the context Rock Weathering Soil Chemical Reactions Soil Development (see text) Weathering Rates
OCN 401-11 Oct. 13, 2011 KCR Weathering and Soils Biogeochemistry Chapter 4: The Lithosphere Introduction: the context Rock Weathering Soil Chemical Reactions Soil Development (see text) Weathering Rates
Groundwater chemistry
 Read: Ch. 3, sections 1, 2, 3, 5, 7, 9; Ch. 7, sections 2, 3 PART 14 Groundwater chemistry Introduction Matter present in water can be divided into three categories: (1) Suspended solids (finest among
Read: Ch. 3, sections 1, 2, 3, 5, 7, 9; Ch. 7, sections 2, 3 PART 14 Groundwater chemistry Introduction Matter present in water can be divided into three categories: (1) Suspended solids (finest among
Tailings and Mineral Carbonation: The Potential for Atmospheric CO 2 Sequestration
 Tailings and Mineral Carbonation: The Potential for Atmospheric CO 2 Sequestration H. Andrew Rollo Lorax Environmental Services Ltd. Heather. E. Jamieson Department of Geological Sciences and Geological
Tailings and Mineral Carbonation: The Potential for Atmospheric CO 2 Sequestration H. Andrew Rollo Lorax Environmental Services Ltd. Heather. E. Jamieson Department of Geological Sciences and Geological
Lecture 29: Soil Formation
 Lecture 29: Soil Formation Factors Controlling Soil Formation 1. Parent material: Soil precursor 2. Climate: Temperature and precipitation 3. Biota: Native vegetation, microbes, soil animals, humans 4.
Lecture 29: Soil Formation Factors Controlling Soil Formation 1. Parent material: Soil precursor 2. Climate: Temperature and precipitation 3. Biota: Native vegetation, microbes, soil animals, humans 4.
FOR Soil Quality Report 2017
 Student Name: Partner Name: Laboratory Date: FOR 2505 - Soil Quality Report 2017 Objectives of this report: 10 Marks Lab Objectives Section Principles behind methods used to determine soil base cation
Student Name: Partner Name: Laboratory Date: FOR 2505 - Soil Quality Report 2017 Objectives of this report: 10 Marks Lab Objectives Section Principles behind methods used to determine soil base cation
Monument Valley, Utah. What weathering processes contributed to the development of these remarkable rock formations? Weathering Mechanisms
 Monument Valley, Utah. What weathering processes contributed to the development of these remarkable rock formations? Weathering Includes Physical, Chemical, Biological processes WEATHERING CHAPTER 7 Weathering
Monument Valley, Utah. What weathering processes contributed to the development of these remarkable rock formations? Weathering Includes Physical, Chemical, Biological processes WEATHERING CHAPTER 7 Weathering
WEATHERING. Weathering breakdown of rock materials Erosion transport of broken-down materials
 WEATHERING the interacting physical, chemical & biological processes that progressively alter the original lithologic character of rocks to produce secondary minerals (e.g. clays) & unconsolidated regolith
WEATHERING the interacting physical, chemical & biological processes that progressively alter the original lithologic character of rocks to produce secondary minerals (e.g. clays) & unconsolidated regolith
Lecture 13 More Surface Reactions on Mineral Surfaces. & Intro to Soil Formation and Chemistry
 Lecture 13 More Surface Reactions on Mineral Surfaces & Intro to Soil Formation and Chemistry 3. charge transfer (e.g., ligand/donor sorption): Sorption involves a number of related processes that all
Lecture 13 More Surface Reactions on Mineral Surfaces & Intro to Soil Formation and Chemistry 3. charge transfer (e.g., ligand/donor sorption): Sorption involves a number of related processes that all
Modelling the Reactivity of Multi-Mineral Systems Application to the Prediction of Copper Heap Leach Drain Down Chemistry
 Modelling the Reactivity of Multi-Mineral Systems Application to the Prediction of Copper Heap Leach Drain Down Chemistry David Bird, Julien Declercq and Rob Bowell Outline 1. Introduction 2. Kinetic database
Modelling the Reactivity of Multi-Mineral Systems Application to the Prediction of Copper Heap Leach Drain Down Chemistry David Bird, Julien Declercq and Rob Bowell Outline 1. Introduction 2. Kinetic database
Water Quality Evaluation and Limits
 Cooling Tower Institute Annual Conference February 7-10, 2010 Houston, TX, USA Water Quality Evaluation and Limits Robert J. Ferguson French Creek Software, Inc. Kimberton, PA 19442 Part 1: Evaluation
Cooling Tower Institute Annual Conference February 7-10, 2010 Houston, TX, USA Water Quality Evaluation and Limits Robert J. Ferguson French Creek Software, Inc. Kimberton, PA 19442 Part 1: Evaluation
Only healthy soil can grow a nutrient dense food. You are what you eat!
 Understanding How Cation Nutrients & Soil Structure are Related By Michael Martin Meléndrez Only healthy soil can grow a nutrient dense food. You are what you eat! Soil Must be able to hold onto water,
Understanding How Cation Nutrients & Soil Structure are Related By Michael Martin Meléndrez Only healthy soil can grow a nutrient dense food. You are what you eat! Soil Must be able to hold onto water,
Weathering and Erosion
 Weathering and Erosion Weathering the disintegration and decomposition of material at the surface Erosion the transportation of weathered material by water, wind, or ice Weathering Two kinds of weathering
Weathering and Erosion Weathering the disintegration and decomposition of material at the surface Erosion the transportation of weathered material by water, wind, or ice Weathering Two kinds of weathering
Soil physical and chemical properties the analogy lecture. Beth Guertal Auburn University, AL
 Soil physical and chemical properties the analogy lecture. Beth Guertal Auburn University, AL Soil Physical Properties Porosity Pore size and pore size distribution Water holding capacity Bulk density
Soil physical and chemical properties the analogy lecture. Beth Guertal Auburn University, AL Soil Physical Properties Porosity Pore size and pore size distribution Water holding capacity Bulk density
Chemistry of Tiling and Crusting
 Chemistry of Tiling and Crusting Tom DeSutter Assistant Professor of Soil Science NDSU Soil and Soil/Water Training 25 January 2012 What is Dispersion? Soil particles are repelled away from each other
Chemistry of Tiling and Crusting Tom DeSutter Assistant Professor of Soil Science NDSU Soil and Soil/Water Training 25 January 2012 What is Dispersion? Soil particles are repelled away from each other
BUFFERING MECHANISM AND SENSITIVITY TO ACID DEPOSITION OF SOILS OF AKWA IBOM STATE, NIGERIA
 BUFFERING MECHANISM AND SENSITIVITY TO ACID DEPOSITION OF SOILS OF AKWA IBOM STATE, NIGERIA ABSTRACT Akpan *, U. S. and Udoh, B. T. Department of Soil Science and Land Resources Management, University
BUFFERING MECHANISM AND SENSITIVITY TO ACID DEPOSITION OF SOILS OF AKWA IBOM STATE, NIGERIA ABSTRACT Akpan *, U. S. and Udoh, B. T. Department of Soil Science and Land Resources Management, University
The Effect of Soil Parent Materials on Yield of Cocoa
 The Effect of Soil Parent Materials on Yield of Cocoa *Adzemi Mat Arshad 1 H. M. Edi Armanto 2 Haruna Yahaya Rawayau 1,3 1. Soil Science Laboratory, School of Food Science and Technology Universiti Malaysia
The Effect of Soil Parent Materials on Yield of Cocoa *Adzemi Mat Arshad 1 H. M. Edi Armanto 2 Haruna Yahaya Rawayau 1,3 1. Soil Science Laboratory, School of Food Science and Technology Universiti Malaysia
EXAMPLE PROBLEMS. 1. Example 1 - Column Infiltration
 EXAMPLE PROBLEMS The module UNSATCHEM is developed from the variably saturated solute transport model HYDRUS-1D [Šimůnek et al., 1997], and thus the water flow and solute transport parts of the model have
EXAMPLE PROBLEMS The module UNSATCHEM is developed from the variably saturated solute transport model HYDRUS-1D [Šimůnek et al., 1997], and thus the water flow and solute transport parts of the model have
A few more details on clays, Soil Colloids and their properties. What expandable clays do to surface area. Smectite. Kaolinite.
 A few more details on clays, Soil Colloids and their properties What expandable clays do to surface area Kaolinite Smectite Size 0.5-5 µm External surface 10-30 m 2 /g Internal surface - Size 0.1-1 µm
A few more details on clays, Soil Colloids and their properties What expandable clays do to surface area Kaolinite Smectite Size 0.5-5 µm External surface 10-30 m 2 /g Internal surface - Size 0.1-1 µm
1/31/2013. Weathering Includes Physical, Chemical, Biological processes. Weathering Mechanisms. Wind abrasion forming Ventifacts
 Monument Valley, Utah. What weathering processes contributed to the development of these remarkable rock formations? Weathering Includes Physical, Chemical, Biological processes Weathering Mechanisms Physical
Monument Valley, Utah. What weathering processes contributed to the development of these remarkable rock formations? Weathering Includes Physical, Chemical, Biological processes Weathering Mechanisms Physical
Soils and Soil Minerals. Remember, most things can be too little or too much.
 Soils and Soil Minerals Remember, most things can be too little or too much. 1 2 3 Source of essential elements CO 2, O 2 from atmosphere H 2 0, O 2, minerals from soil NH 4, SO 4 can volatilize and be
Soils and Soil Minerals Remember, most things can be too little or too much. 1 2 3 Source of essential elements CO 2, O 2 from atmosphere H 2 0, O 2, minerals from soil NH 4, SO 4 can volatilize and be
Mid-Term #1 (125 points total)
 Ocean 520 Name: Chemical Oceanography 20 October 2009 Fall 2009 Points are in parentheses (show all your work) (use back if necessary) MidTerm #1 (125 points total) 1. Doney et al (2008) Ocean Acidification
Ocean 520 Name: Chemical Oceanography 20 October 2009 Fall 2009 Points are in parentheses (show all your work) (use back if necessary) MidTerm #1 (125 points total) 1. Doney et al (2008) Ocean Acidification
Model Study of Formation of the Cap Rocks for Geothermal System Using ChemTOUGH2
 Proceedings World Geothermal Congress 2005 Antalya, Turkey, 24-29 April 2005 Model Study of Formation of the Cap Rocks for Geothermal System Using ChemTOUGH2 Sato, T. +, Sato, M. +. Ueda, A. ++, Kato,
Proceedings World Geothermal Congress 2005 Antalya, Turkey, 24-29 April 2005 Model Study of Formation of the Cap Rocks for Geothermal System Using ChemTOUGH2 Sato, T. +, Sato, M. +. Ueda, A. ++, Kato,
The Production of Sediment. Contents. Weathering. Chapters 1, 3
 The Production of Sediment Chapters 1, 3 Contents Weathering Physical, chemical, biogeochemical processes Rates Products Carbon cycle and global change Erosion/Soils Sediment Texture Weathering General
The Production of Sediment Chapters 1, 3 Contents Weathering Physical, chemical, biogeochemical processes Rates Products Carbon cycle and global change Erosion/Soils Sediment Texture Weathering General
WEATHERING AND MASS MOVEMENTS. 1. Introduction 2. Physical/mechanical weathering 3. Chemical weathering 4. Mass movement processes
 WEATHERING AND MASS MOVEMENTS 1. Introduction 2. Physical/mechanical weathering 3. Chemical weathering 4. Mass movement processes Mean annual temperature ( C) Hot Cold TEMP. -10 0 10 20 Strong chemical
WEATHERING AND MASS MOVEMENTS 1. Introduction 2. Physical/mechanical weathering 3. Chemical weathering 4. Mass movement processes Mean annual temperature ( C) Hot Cold TEMP. -10 0 10 20 Strong chemical
Global Carbon Cycle - I
 Global Carbon Cycle - I OCN 401 - Biogeochemical Systems Reading: Schlesinger, Chapter 11 1. Overview of global C cycle 2. Global C reservoirs Outline 3. The contemporary global C cycle 4. Fluxes and residence
Global Carbon Cycle - I OCN 401 - Biogeochemical Systems Reading: Schlesinger, Chapter 11 1. Overview of global C cycle 2. Global C reservoirs Outline 3. The contemporary global C cycle 4. Fluxes and residence
Lecture 16 Guest Lecturer this week. Prof. Greg Ravizza
 Lecture 16 Guest Lecturer this week. Prof. Greg Ravizza General Concepts for Natural Controls on Fresh Water Composition 1. Generalizations about freshwater compositions 2. Conservative/nonconservative
Lecture 16 Guest Lecturer this week. Prof. Greg Ravizza General Concepts for Natural Controls on Fresh Water Composition 1. Generalizations about freshwater compositions 2. Conservative/nonconservative
LAB 2: SILICATE MINERALS
 GEOLOGY 640: Geology through Global Arts and Artifacts LAB 2: SILICATE MINERALS FRAMEWORK SILICATES The framework silicates quartz and feldspar are the most common minerals in Earth s crust. Quartz (SiO
GEOLOGY 640: Geology through Global Arts and Artifacts LAB 2: SILICATE MINERALS FRAMEWORK SILICATES The framework silicates quartz and feldspar are the most common minerals in Earth s crust. Quartz (SiO
Global Carbon Cycle - I Systematics: Reservoirs and Fluxes
 OCN 401-10 Nov. 16, 2010 KCR Global Carbon Cycle - I Systematics: Reservoirs and Fluxes The Global carbon cycle Reservoirs: biomass on land in the oceans, atmosphere, soil and rocks, waters Processes:
OCN 401-10 Nov. 16, 2010 KCR Global Carbon Cycle - I Systematics: Reservoirs and Fluxes The Global carbon cycle Reservoirs: biomass on land in the oceans, atmosphere, soil and rocks, waters Processes:
Redox, ph, pe OUTLINE 9/12/17. Equilibrium? Finish last lecture Mineral stability Aquatic chemistry oxidation and reduction: redox
 Redox, ph, pe Equilibrium? OUTLINE Finish last lecture Mineral stability Aquatic chemistry oxidation and reduction: redox Reading: White p555-563 1 Question of the day? So what about the CO 2 system? CO
Redox, ph, pe Equilibrium? OUTLINE Finish last lecture Mineral stability Aquatic chemistry oxidation and reduction: redox Reading: White p555-563 1 Question of the day? So what about the CO 2 system? CO
ph and Liming Practices Kent Martin Stafford County 1/5/2010
 ph and Liming Practices Kent Martin Stafford County 1/5/2010 Outline What is ph Normal ph ranges Acid Soil Importance of soil ph Factors affecting soil ph Acid types and measurement Neutralizing value
ph and Liming Practices Kent Martin Stafford County 1/5/2010 Outline What is ph Normal ph ranges Acid Soil Importance of soil ph Factors affecting soil ph Acid types and measurement Neutralizing value
Flocculation and Dispersion
 Flocculation and Dispersion Flocculation is the process, where the individual particles of clay are coagulated to form floccular aggregates. The degree and permanence of flocculation depend upon the nature
Flocculation and Dispersion Flocculation is the process, where the individual particles of clay are coagulated to form floccular aggregates. The degree and permanence of flocculation depend upon the nature
Chapter 7: Anion and molecular retention
 I. Anions and molecules of importance in soils Anions of major importance to agricultural soils and soil chemistry are: H 2 PO - 4, HPO 2-4, SO 2-4, HCO - 3, NO - 3, Cl -, F - and OH -. Also, micronutrients
I. Anions and molecules of importance in soils Anions of major importance to agricultural soils and soil chemistry are: H 2 PO - 4, HPO 2-4, SO 2-4, HCO - 3, NO - 3, Cl -, F - and OH -. Also, micronutrients
WEATHERING. Turning Rock to Sediment and Solutions 10/22/2012
 WEATHERING Turning Rock to Sediment and Solutions Igneous rocks form at high temperatures; at the Earth s surface they are chemically unstable and will begin to disintegrate and decompose in a process
WEATHERING Turning Rock to Sediment and Solutions Igneous rocks form at high temperatures; at the Earth s surface they are chemically unstable and will begin to disintegrate and decompose in a process
How Does Redox Status Influence Exchangeable Potassium In Soil?
 How Does Redox Status Influence Exchangeable Potassium In Soil? Michael L. Thompson Taslima Stephen Iowa State University 1 Why do we care about exchangeable K? K is required by all plants. A soil may
How Does Redox Status Influence Exchangeable Potassium In Soil? Michael L. Thompson Taslima Stephen Iowa State University 1 Why do we care about exchangeable K? K is required by all plants. A soil may
MARLET Serge (*),VALLÈS Vincent (**),LAFOLIE François (**) CONDOM Nicolas (*)
 Scientific registration n 537 Symposium n 29 Présentation : Oral Hydrogeochemical modeling: a suitable approachto predict the effect of irrigation on soil salinity,sodicity and alkalinity Modélisation
Scientific registration n 537 Symposium n 29 Présentation : Oral Hydrogeochemical modeling: a suitable approachto predict the effect of irrigation on soil salinity,sodicity and alkalinity Modélisation
CE4501 Environmental Engineering Chemical Processes, Fall 2008 Problem Set 6
 CE4501 Environmental Engineering Chemical Processes, Fall 2008 Problem Set 6 Due: Friday, 11/21 by 5 p.m. Solutions will be posted on the Web. Problem sets will be graded for completeness, and one problem
CE4501 Environmental Engineering Chemical Processes, Fall 2008 Problem Set 6 Due: Friday, 11/21 by 5 p.m. Solutions will be posted on the Web. Problem sets will be graded for completeness, and one problem
Chapter 6 9/25/2012. Weathering, Erosion and Soils. Introduction. How Are Earth Materials Altered? Introduction. How Are Earth Materials Altered?
 Chapter 6 Introduction Rocks and minerals are disintegrated and decomposed by the processes of mechanical and chemical weathering. Weathering, Erosion and Soils This breakdown occurs because the parent
Chapter 6 Introduction Rocks and minerals are disintegrated and decomposed by the processes of mechanical and chemical weathering. Weathering, Erosion and Soils This breakdown occurs because the parent
Processed Food Production. Consistent product Long shelf life Low cost
 How is the production of wheat connected to Earth and environmental sciences? Processed Food Production Consistent product Long shelf life Low cost Twinkie Ingredients Enriched bleached wheat flour [flour,
How is the production of wheat connected to Earth and environmental sciences? Processed Food Production Consistent product Long shelf life Low cost Twinkie Ingredients Enriched bleached wheat flour [flour,
Geology 560, Prof. Thomas Johnson, Fall 2003 Unit II: Chemical reactions: Guide Questions
 Geology 560, Prof. Thomas Johnson, Fall 2003 Unit II: Chemical reactions: Guide Questions Goals: Refresh your memory on the basics of chemical reactions Ponder the chaos that underlies chemical reactions;
Geology 560, Prof. Thomas Johnson, Fall 2003 Unit II: Chemical reactions: Guide Questions Goals: Refresh your memory on the basics of chemical reactions Ponder the chaos that underlies chemical reactions;
Husein Ajwa, Emeritus Department of Plant Sciences UC Davis
 Husein Ajwa, Emeritus Department of Plant Sciences UC Davis HUSEIN AJWA Plant Sciences Department, UC Davis 1636 East Alisal St., Salinas, CA 93905 Office: (831) 755-2823, Fax: (831) 755-2898 email: haajwa@ucdavis.edu
Husein Ajwa, Emeritus Department of Plant Sciences UC Davis HUSEIN AJWA Plant Sciences Department, UC Davis 1636 East Alisal St., Salinas, CA 93905 Office: (831) 755-2823, Fax: (831) 755-2898 email: haajwa@ucdavis.edu
GEOLOGICAL PROCESSES AND MATERIALS SGM210
 University of Pretoria GEOLOGICAL PROCESSES AND MATERIALS SGM210 2 May 2014 1 DISPERSIVE SOILS Thanks to Dr Phil Paige Green 1 Dispersive (and Erodible) Soils Phil Paige Green (H.F.W.K Elges) CSIR Built
University of Pretoria GEOLOGICAL PROCESSES AND MATERIALS SGM210 2 May 2014 1 DISPERSIVE SOILS Thanks to Dr Phil Paige Green 1 Dispersive (and Erodible) Soils Phil Paige Green (H.F.W.K Elges) CSIR Built
Weathering Cycle Teacher Notes
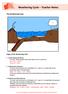 The Weathering Cycle Stages of the Weathering Cycle: 1. Carbon Dioxide and Water In clouds, carbon dioxide reacts with water to form a weak acid. H 2 O + CO 2 --> H 2 CO 3 H 2 CO 3 H + + HCO 3-2. Acid
The Weathering Cycle Stages of the Weathering Cycle: 1. Carbon Dioxide and Water In clouds, carbon dioxide reacts with water to form a weak acid. H 2 O + CO 2 --> H 2 CO 3 H 2 CO 3 H + + HCO 3-2. Acid
Lecture 6: Soil Profiles: Diagnostic Horizons
 Lecture 6: Soil Profiles: Diagnostic Horizons Complexity in Soil Profiles Soil Horizons Soils display distinct layering O Horizon: Partially decomposed organic matter (OM) A Horizon: Near surface, mineral
Lecture 6: Soil Profiles: Diagnostic Horizons Complexity in Soil Profiles Soil Horizons Soils display distinct layering O Horizon: Partially decomposed organic matter (OM) A Horizon: Near surface, mineral
I.S : What s in it and the role of the Geologist
 Institute of Geologists of Ireland Pyrite Course I.S. 398-1: What s in it and the role of the Geologist Michael L.J. Maher 4 December, 2013 Responsibilities of Geologist You re only the messenger! Classification
Institute of Geologists of Ireland Pyrite Course I.S. 398-1: What s in it and the role of the Geologist Michael L.J. Maher 4 December, 2013 Responsibilities of Geologist You re only the messenger! Classification
Chapter 6. Weathering, Erosion, and Soil
 Chapter 6 Weathering, Erosion, and Soil Introduction Rocks and minerals disintegrate and decompose by the processes of physical and chemical weathering. This breakdown occurs because the parent material
Chapter 6 Weathering, Erosion, and Soil Introduction Rocks and minerals disintegrate and decompose by the processes of physical and chemical weathering. This breakdown occurs because the parent material
Weathering and mineral equilibria. Seminar at NGU 23 May 2016 Håkon Rueslåtten
 Weathering and mineral equilibria Seminar at NGU 23 May 2016 Håkon Rueslåtten Weathering is the breakdown of rocks and minerals that are exposed to surface processes (climatically controlled). Water is
Weathering and mineral equilibria Seminar at NGU 23 May 2016 Håkon Rueslåtten Weathering is the breakdown of rocks and minerals that are exposed to surface processes (climatically controlled). Water is
Wednesday week 12. These ions move through the soil to streams and eventually to the ocean. In the ocean; CaCO 3 + H 2 O + CO 2 H 2 O + H 2 O
 Wednesday week 12 I. Control of CO 2 content of atmosphere by the ocean H 4 SiO 4 A. Consider a hypothetical planet with a crust made of single mineral (Wallastonite) CaSiO3. We could use the composition
Wednesday week 12 I. Control of CO 2 content of atmosphere by the ocean H 4 SiO 4 A. Consider a hypothetical planet with a crust made of single mineral (Wallastonite) CaSiO3. We could use the composition
Lecture 14: Cation Exchange and Surface Charging
 Lecture 14: Cation Exchange and Surface Charging Cation Exchange Cation Exchange Reactions Swapping of cations between hydrated clay interlayers and the soil solution Also occurs on organic matter functional
Lecture 14: Cation Exchange and Surface Charging Cation Exchange Cation Exchange Reactions Swapping of cations between hydrated clay interlayers and the soil solution Also occurs on organic matter functional
LAB 2 IDENTIFYING MATERIALS FOR MAKING SOILS: ROCK AND PARENT MATERIALS
 LAB 2 IDENTIFYING MATERIALS FOR MAKING SOILS: ROCK AND PARENT MATERIALS Learning outcomes The student is able to: 1. understand and identify rocks 2. understand and identify parent materials 3. recognize
LAB 2 IDENTIFYING MATERIALS FOR MAKING SOILS: ROCK AND PARENT MATERIALS Learning outcomes The student is able to: 1. understand and identify rocks 2. understand and identify parent materials 3. recognize
Mechanical Weathering
 Weathering is the disintegration and decomposition of material at or near the surface. Erosion is the incorporation and transportation of material by a mobile agent, usually water, wind, or ice. Geologists
Weathering is the disintegration and decomposition of material at or near the surface. Erosion is the incorporation and transportation of material by a mobile agent, usually water, wind, or ice. Geologists
Weathering: the disintegration, or breakdown of rock material
 Weathering: the disintegration, or breakdown of rock material Mechanical Weathering: no change in chemical composition--just disintegration into smaller pieces Chemical Weathering: breakdown as a result
Weathering: the disintegration, or breakdown of rock material Mechanical Weathering: no change in chemical composition--just disintegration into smaller pieces Chemical Weathering: breakdown as a result
Sedimentology & Stratigraphy. Thanks to Rob Viens for slides
 Sedimentology & Stratigraphy Thanks to Rob Viens for slides Sedimentology The study of the processes that erode, transport and deposit sediments Sedimentary Petrology The study of the characteristics and
Sedimentology & Stratigraphy Thanks to Rob Viens for slides Sedimentology The study of the processes that erode, transport and deposit sediments Sedimentary Petrology The study of the characteristics and
APPENDIX F PHOTOGRAPHS OF CORE SAMPLES
 EKATI DIAMOND MINE LONG LAKE CONTAINMENT FACILITY INVESTIGATION 2013 EBA FILE: E14103013-01.002 MAY 2014 ISSUED FOR USE APPENDIX F PHOTOGRAPHS OF CORE SAMPLES LLCF Geotechnical Site Investigation Report_IFU.docx
EKATI DIAMOND MINE LONG LAKE CONTAINMENT FACILITY INVESTIGATION 2013 EBA FILE: E14103013-01.002 MAY 2014 ISSUED FOR USE APPENDIX F PHOTOGRAPHS OF CORE SAMPLES LLCF Geotechnical Site Investigation Report_IFU.docx
Lecture series outline
 Water Quality 2 Lecture series outline Lecture 1a Water uses, water quality guidelines; physical, chemical and biological parameters Lecture 1b The origins of the constituents of water quality through
Water Quality 2 Lecture series outline Lecture 1a Water uses, water quality guidelines; physical, chemical and biological parameters Lecture 1b The origins of the constituents of water quality through
Sintering behavior of feldspar rocks
 Research Inventy: International Journal Of Engineering And Science Vol.4, Issue 10 (October2014), PP 49-55 Issn (e): 2278-4721, Issn (p):2319-6483, www.researchinventy.com Sintering behavior of feldspar
Research Inventy: International Journal Of Engineering And Science Vol.4, Issue 10 (October2014), PP 49-55 Issn (e): 2278-4721, Issn (p):2319-6483, www.researchinventy.com Sintering behavior of feldspar
Basalt-Atmosphere Interactions on Venus - The Rocks Perspective. Allan Treiman Susanne Schwenzer LPI
 Basalt-Atmosphere Interactions on Venus - The Rocks Perspective Allan Treiman Susanne Schwenzer LPI Plan of Talk Hoped to have more results - sorry! So, a selection of research problems on Venus rock-atmosphere
Basalt-Atmosphere Interactions on Venus - The Rocks Perspective Allan Treiman Susanne Schwenzer LPI Plan of Talk Hoped to have more results - sorry! So, a selection of research problems on Venus rock-atmosphere
TWO COMPONENT (BINARY) PHASE DIAGRAMS. Experimental Determination of 2-Component Phase Diagrams
 Page 1 of 12 EENS 211 Earth Materials Tulane University Prof. Stephen A. Nelson TWO COMPONENT (BINARY) PHASE DIAGRAMS This document last updated on 08-Oct-2003 Experimental Determination of 2-Component
Page 1 of 12 EENS 211 Earth Materials Tulane University Prof. Stephen A. Nelson TWO COMPONENT (BINARY) PHASE DIAGRAMS This document last updated on 08-Oct-2003 Experimental Determination of 2-Component
Chapter 3: Acid Base Equilibria. HCl + KOH KCl + H 2 O acid + base salt + water
 Chapter 3: Acid Base Equilibria HCl + KOH KCl + H 2 O acid + base salt + water What is an acid? The Arrhenius concept proposed that acids are substances that produce hydrogen ions (H + ) in aqueous solutions.
Chapter 3: Acid Base Equilibria HCl + KOH KCl + H 2 O acid + base salt + water What is an acid? The Arrhenius concept proposed that acids are substances that produce hydrogen ions (H + ) in aqueous solutions.
Soil Water Atmosphere Plant (SWAP) Model: I. INTRODUCTION AND THEORETICAL BACKGROUND
 Soil Water Atmosphere Plant (SWAP) Model: I. INTRODUCTION AND THEORETICAL BACKGROUND Reinder A.Feddes Jos van Dam Joop Kroes Angel Utset, Main processes Rain fall / irrigation Transpiration Soil evaporation
Soil Water Atmosphere Plant (SWAP) Model: I. INTRODUCTION AND THEORETICAL BACKGROUND Reinder A.Feddes Jos van Dam Joop Kroes Angel Utset, Main processes Rain fall / irrigation Transpiration Soil evaporation
Global Carbon Cycle - I
 Global Carbon Cycle - I Reservoirs and Fluxes OCN 401 - Biogeochemical Systems 13 November 2012 Reading: Schlesinger, Chapter 11 Outline 1. Overview of global C cycle 2. Global C reservoirs 3. The contemporary
Global Carbon Cycle - I Reservoirs and Fluxes OCN 401 - Biogeochemical Systems 13 November 2012 Reading: Schlesinger, Chapter 11 Outline 1. Overview of global C cycle 2. Global C reservoirs 3. The contemporary
Lect. 2: Chemical Water Quality
 The Islamic University of Gaza Faculty of Engineering Civil Engineering Department M.Sc. Water Resources Water Quality Management (ENGC 6304) Lect. 2: Chemical Water Quality ١ Chemical water quality parameters
The Islamic University of Gaza Faculty of Engineering Civil Engineering Department M.Sc. Water Resources Water Quality Management (ENGC 6304) Lect. 2: Chemical Water Quality ١ Chemical water quality parameters
Table 1. Solubility of gypsum and anhydrite adopted in this research and the predicted SI values
 Supporting information: Table 1. Solubility of gypsum and anhydrite adopted in this research and the predicted SI values Sample T ID # ( o C) 16, 65, 74 P NaCl CaSO 4 nh 2 O SI System (psi) (mol/kg H 2
Supporting information: Table 1. Solubility of gypsum and anhydrite adopted in this research and the predicted SI values Sample T ID # ( o C) 16, 65, 74 P NaCl CaSO 4 nh 2 O SI System (psi) (mol/kg H 2
TABLE DR1. COMBINED EMPA AND LA-ICP-MS DATA FOR WATERSHED 2 AND 34 MINERALS USED IN THE MASS BALANCE CALCULATIONS
 TABLE DR1. COMBINED EMPA AND LA-ICP-MS DATA FOR WATERSHED 2 AND 34 MINERALS USED IN THE MASS BALANCE CALCULATIONS Allanite Plagioclase C17-3 C17-3 C17-3 C17-3 Mean C17-3 C17-3 C17-3 C17-3 Mean E1C E1R
TABLE DR1. COMBINED EMPA AND LA-ICP-MS DATA FOR WATERSHED 2 AND 34 MINERALS USED IN THE MASS BALANCE CALCULATIONS Allanite Plagioclase C17-3 C17-3 C17-3 C17-3 Mean C17-3 C17-3 C17-3 C17-3 Mean E1C E1R
STUDIES ON THE SORPTION OF PHOSPHATE ON SOME SOILS OF INDIA SATURATED WITH DIFFERENT CATIONS
 I.J.S.N., VOL. 2(2) 211: 327-333 ISSN 2229 6441 STUDIES ON THE SORPTION OF PHOSPHATE ON SOME SOILS OF INDIA SATURATED WITH DIFFERENT CATIONS Bansal, O. P. Chemistry Department, D.S. College, Aligarh-221
I.J.S.N., VOL. 2(2) 211: 327-333 ISSN 2229 6441 STUDIES ON THE SORPTION OF PHOSPHATE ON SOME SOILS OF INDIA SATURATED WITH DIFFERENT CATIONS Bansal, O. P. Chemistry Department, D.S. College, Aligarh-221
INCREASING TREND IN DUST CLOUD INTRUSIONS FROM THE SAHARA OVER ISRAEL. Department of Geophysics and Planetary Sciences, Tel Aviv University
 INCREASING TREND IN DUST CLOUD INTRUSIONS FROM THE SAHARA OVER ISRAEL Eliezer Ganor, Amnon Stupp, Pinhas Alpert, Isabella Osetinsky Department of Geophysics and Planetary Sciences, Tel Aviv University
INCREASING TREND IN DUST CLOUD INTRUSIONS FROM THE SAHARA OVER ISRAEL Eliezer Ganor, Amnon Stupp, Pinhas Alpert, Isabella Osetinsky Department of Geophysics and Planetary Sciences, Tel Aviv University
1. Water in Soils: Infiltration and Redistribution
 Contents 1 Water in Soils: Infiltration and Redistribution 1 1a Material Properties of Soil..................... 2 1b Soil Water Flow........................... 4 i Incorporating K - θ and ψ - θ Relations
Contents 1 Water in Soils: Infiltration and Redistribution 1 1a Material Properties of Soil..................... 2 1b Soil Water Flow........................... 4 i Incorporating K - θ and ψ - θ Relations
Soil Mechanics Prof. B.V.S. Viswanadham Department of Civil Engineering Indian Institute of Technology, Bombay Lecture 3
 Soil Mechanics Prof. B.V.S. Viswanadham Department of Civil Engineering Indian Institute of Technology, Bombay Lecture 3 In the previous lecture we have studied about definitions of volumetric ratios and
Soil Mechanics Prof. B.V.S. Viswanadham Department of Civil Engineering Indian Institute of Technology, Bombay Lecture 3 In the previous lecture we have studied about definitions of volumetric ratios and
Classification of Igneous Rocks
 Classification of Igneous Rocks Textures: Glassy- no crystals formed Aphanitic- crystals too small to see by eye Phaneritic- can see the constituent minerals Fine grained- < 1 mm diameter Medium grained-
Classification of Igneous Rocks Textures: Glassy- no crystals formed Aphanitic- crystals too small to see by eye Phaneritic- can see the constituent minerals Fine grained- < 1 mm diameter Medium grained-
