Solution structures of potato virus X and narcissus mosaic virus from Raman optical activity
|
|
|
- Clement Cummings
- 5 years ago
- Views:
Transcription
1 Journal of General Virology (2002), 83, Printed in Great Britain... Solution structures of potato virus X and narcissus mosaic virus from Raman optical activity Ewan W. Blanch, 1 David J. Robinson, 2 Lutz Hecht, 1 Christopher D. Syme, 1 Kurt Nielsen 3 and Laurence D. Barron 1 1 Department of Chemistry, University of Glasgow, Glasgow G12 8QQ, UK 2 Scottish Crop Research Institute, Invergowrie, Dundee DD2 5DA, UK 3 Department of Chemistry, DTU 207, Technical University of Denmark, DK-2800 Lyngby, Denmark Potato virus X (PVX) and narcissus mosaic virus (NMV) were studied using vibrational Raman optical activity (ROA) in order to obtain new information on the structures of their coat protein subunits. The ROA spectra of the two intact virions are very similar to each other and similar to that of tobacco mosaic virus (TMV) studied previously, being dominated by signals characteristic of proteins with helix bundle folds. In particular, PVX and NMV show strong positive ROA bands at 1340 cm N1 assigned to hydrated α-helix and perhaps originating in surface exposed helical residues, together with less strong positive ROA intensity in the range cm N1 assigned to α-helix in a more hydrophobic environment and perhaps originating in residues at helix helix interfaces. The positive 1340 cm N1 ROA band of TMV is less intense than those of PVX and NMV, suggesting that TMV contains less hydrated α-helix. Small differences in other spectral regions reflect differences in some loop, turn and side-chain compositions and conformations among the three viruses. A pattern recognition program based on principal component analysis of ROA spectra indicates that the coat protein subunit folds of PVX and NMV may be very similar to each other and similar to that of TMV. These results suggest that PVX and NMV may have coat protein subunit structures based on folds similar to the TMV helix bundle and hence that the helical architecture of the PVX and NMV particles may be similar to that of TMV but with different structural parameters. Introduction Potato virus X (PVX) and narcissus mosaic virus (NMV) belong to the Potexvirus genus of helical plant viruses, PVX being the type species (Brunt et al., 2000). They contain genomes of single-stranded positive-sense RNA encapsidated within flexuous filamentous particles, 500 nm long and nm in diameter, with helical symmetry and a pitch of 3 4 nm. In comparison, the Tobamovirus genus of helical plant viruses, to which tobacco mosaic virus (TMV) belongs, also contain single-stranded positive-sense RNA but the particles are rigid rods 300 nm long and 18 nm in diameter with a helical pitch of 2 3 nm. Although detailed information about the genome organization, the amino acid sequences of the encoded proteins and the sequence homology between members of the Potexvirus genus is available [Brunt et al. (2000) Author for correspondence: Laurence Barron. Fax: laurence chem.gla.ac.uk and references therein], little is known about the conformation of the coat protein subunits and their packing arrangements in the virus particles. Unlike the Tobamoviruses, there are no X-ray diffraction data of sufficient resolution to show molecular conformational detail, but ultraviolet circular dichroism (UVCD) measurements suggest 45% α-helix content and 5% β-sheet for both PVX (Homer & Goodman, 1975) and NMV (Wilson et al., 1991). A model of the PVX coat protein tertiary structure has been suggested based on the predicted secondary structure content, the principles of secondary structure packing, and the known biochemical, immunological and tritium bombardment data (Baratova et al., 1992a). The lack of information about the molecular structure of potexviruses highlights the lack of physical techniques applicable to the determination of virus structure and function at the molecular level. A novel spectroscopic technique called Raman optical activity (ROA) appears to be promising for such studies and has recently been applied to intact viruses; specifically filamentous bacteriophages (Blanch et al., 1999), SGM CEB
2 E. W. Blanch and others and tobacco rattle virus (TRV) and tobacco mosaic virus (TMV) (Blanch et al., 2001a). Here we report an ROA study of the DX strain of PVX (Jones, 1982) and the MC strain of NMV (Wilson et al., 1991) from which new information about the folds of the coat proteins is deduced, mainly from a comparison with the ROA spectrum of the U1 strain of TMV. A brief review of Raman spectroscopy and ROA Since the ROA technique is unfamiliar to most virologists, a brief description may be helpful. Raman spectroscopy itself is a form of vibrational spectroscopy, complementary to infrared absorption, which provides vibrational spectra of molecules by means of inelastic scattering of visible or ultraviolet laser light. During the so-called Stokes Raman scattering event, the interaction of the molecule with the incident laser photon of energy ω, where ω is its angular frequency, can leave the molecule in an excited vibrational state of energy h ω v, with a corresponding energy loss, and hence a shift to lower angular frequency ω ω v, of the scattered photon. Therefore, by analysing the spectrum of the scattered light with a visible or ultraviolet spectrometer, a complete vibrational spectrum of the molecule may be obtained. Conventional Raman spectroscopy has a number of favourable characteristics, including the fact that water is an excellent solvent for Raman studies, which have led to many applications in biochemistry (Carey, 1982; Miura & Thomas, 1995). It has proved especially valuable in structural virology on account of its ability to provide information about both protein and nucleic acid constituents of intact virions (Thomas, 1987, 1999). ROA is a novel form of Raman spectroscopy that is sensitive to chirality, meaning handedness, in molecular structure. The two distinguishable mirror-image forms of a chiral molecule are called enantiomers. Biomolecules such as proteins and nucleic acids are chiral on account of the intrinsic chirality of their constituent amino acids (almost exclusively the L-enantiomers) and sugars (almost exclusively the D- enantiomers), respectively. Raman spectroscopy becomes sensitive to chirality by utilizing circularly polarized light: thus ROA measures small differences in the Raman spectra of chiral molecules acquired using right- and left-circularly polarized incident laser light. ROA bears the same relation to conventional Raman spectroscopy as does the widely used biochemical technique of UVCD to conventional visible and ultraviolet absorption spectroscopy. The advantage of ROA is that it provides vibrational optical activity spectra which contain much more stereochemical information than the electronic optical activity spectra provided by UVCD (just as conventional vibrational spectra, Raman scattering or infrared absorption, contain much more information about molecular structure than conventional visible and ultraviolet absorption spectra). Recent general reviews of ROA include Nafie (1997) and Barron & Hecht (2000). ROA has recently been applied to biomolecules such as proteins, carbohydrates, nucleic acids and viruses. This work has been reviewed by Barron et al. (2000). On account of its sensitivity to chirality, ROA is proving to be an incisive probe of biomolecular structure and dynamics. One reason for this is that the largest ROA signals are often associated with the most rigid chiral parts of the structure. In proteins these are usually within the peptide backbone and often give rise to ROA band patterns characteristic of the peptide backbone conformation, unlike the parent Raman spectra which are often dominated by bands from the amino acid side-chains, which often obscure the peptide backbone bands. As well as bands arising from secondary structure, protein ROA spectra also contain bands from loops and turns and so can provide information about the tertiary fold. A few ROA bands from side-chains also appear, including one for tryptophan from which the absolute stereochemistry can be deduced, even in the coat proteins of intact viruses (Blanch et al., 2001b). Carbohydrate ROA spectra provide information about anomeric preference, the pattern of OH substituents and the nature of the glycosidic link, and can detect extended secondary structure in polysaccharides. Nucleic acid ROA spectra provide information about base stacking, the mutual orientations of sugar and base rings, and the sugar phosphate backbone conformations. Methods PVX, strain DX, was propagated in Nicotiana tabacum Samsun NN, and purified as described by Abou Haidar et al. (1998). NMV, strain MC, was propagated in Chenopodium quinoa, and purified as described by Goodman (1973) for PVX, followed by sedimentation in a 10 40% sucrose gradient at r.p.m. for 140 min in a Beckman SW28 rotor. Both viruses were studied as solutions at 25 mg ml in 15 mm phosphate buffer at ph 7 4 held in small quartz microfluorescence cells at ambient temperature ( 20 C). The ROA measurements were performed using an instrument described previously (Hecht et al., 1999). It has a backscattering configuration and employs a single-grating spectrograph fitted with a backthinned charge coupled device (CCD) camera as detector and an edge filter to block the Rayleigh line. The small ROA signals are accumulated by synchronizing the Raman spectral acquisition with an electrooptic modulator that switches the polarization of the argon-ion laser beam between right- and left-circular at a suitable rate. The spectra are displayed in analogue-to-digital counter units as a function of the Stokes Raman wavenumber shift with respect to the exciting laser wavelength. The ROA spectra, which have undergone minimal smoothing (2 point FFT), are presented as circular intensity differences IR IL and the parent Raman spectra as circular intensity sums IR IL, where IR and IL are the Raman-scattered intensities in right- and left-circularly polarized incident light, respectively. The experimental conditions were as follows: laser wavelength nm; laser power at the sample 700 mw; spectral resolution 10 cm ; acquisition time 48 h. Results and Discussion The ROA spectral details and their structural assignments The backscattered Raman and ROA spectra of PVX and NMV are shown as the top and middle pairs of spectra, CEC
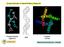
3 Potato virus X and narcissus mosaic virus Fig. 1. Backscattered Raman (I R I L ) and ROA (I R I L ) spectra of PVX (top pair), NMV (middle pair) and TMV (bottom pair) in phosphate buffer at ph 7 4. respectively, in Fig. 1. An account of most of the ROA assignments given in what follows can be found in Barron et al. (2000). The ROA spectra of the two viruses are dominated by protein bands. The general appearance of the ROA spectra of both viruses suggests that both contain a large amount of α-helix. In particular, both show an ROA couplet, negative at 1630 and positive at 1666 cm, originating in peptide amide I vibrations involving mainly C O stretching that is characteristic of α-helix. Positive protein ROA bands in the range cm together with positive bands in the range cm, originating in extended amide III vibrations involving various contributions from peptide backbone N H and C α H deformations and the C α N stretch, are also characteristic of α-helix. The former appear to arise from α-helix in a hydrophobic environment and the latter to a hydrated form of α-helix (Blanch et al., 1999). Both PVX and NMV show strong positive ROA bands at 1337 cm together with weaker positive intensity centred cm which are therefore assigned to hydrated and hydrophobic α-helix, respectively. Such an ROA band pattern is characteristic of helix bundle proteins in which most residues are surface exposed and hydrated and a smaller number are within hydrophobic interfaces between helices. One difference between the ROA spectra of the two viruses is the presence of a significant negative band at 1252 cm in the ROA spectrum of NMV assigned to β-strand, whereas PVX shows rather weaker less well-defined negative ROA here. However, both viruses show negative ROA bands at 1220 cm assigned to a hydrated form of β-strand. Differences in various weak bands reflect differences in the details of loop and turn sequences. ROA bands from the RNA are expected to be much weaker than from the protein due to the low RNA contents ( 6% of the total particle mass in each case). It is possible that the weak positive bands at 1488 and 1579 cm in the ROA spectrum of PVX arise from the nucleic acid bases since RNA and DNA often show large ROA in these regions (Bell et al., 1997, 1998) but proteins do not. Hints of similar bands appear in the ROA spectrum of NMV. More specifically, these bands may arise from guanine and adenine since bands at similar wavenumbers assigned to these bases in the viral nucleic acid are seen in the conventional transparent and resonance Raman spectra of intact viruses (Thomas, 1999), and naturally occurring nucleic acids show strong positive ROA at 1481 cm also assigned to guanine and adenine (Bell et al., 1998). The protein ROA data are consistent with the UVCD results, which predicted large amounts of α-helix with little β-sheet (Homer & Goodman, 1975; Wilson et al., 1991). Furthermore, the overall appearances of the ROA spectra of PVX and NMV are very similar to each other and to that of TMV reported recently (Blanch et al., 2001a) for which the coat protein subunits are based on four-helix bundles containing both water-exposed residues and residues at hydrophobic helix helix interfaces (Namba et al., 1989; Stubbs, 1999). One difference, however, is that the positive 1337 cm ROA bands in PVX and NMV are significantly more intense than the corresponding band in TMV, which suggests that PVX and NMV contain more hydrated α-helix than TMV. For convenience, a re-measured ROA spectrum of TMV (strain U1) of better quality than that recorded in the earlier study is shown at the bottom of Fig. 1. Our results therefore suggest that the coat protein folds of PVX and NMV are similar to that of TMV, being based on a helix bundle, but with differences of detail resulting from differences in the appended loops and turns and from the extra sequences present in PVX and NMV (the coat proteins of the DX strain of PVX, the MC strain of CED
4 E. W. Blanch and others NMV and the U1 strain of TMV contain 237, 240 and 159 amino acids, respectively). Our results do not support the model of Baratova et al. (1992a) for the PVX coat proteins, which may contain too much well-defined β-sheet (which is predicted at both the N- and C-terminal ends). However, unlike the N-terminal regions of PVX which tritium planigraphy shows are exposed, the C-terminal regions of PVX are virtually inaccessible to tritium labelling in the intact virion (Baratova et al., 1992b), but become accessible after 20 N-terminal amino acids are proteolyticaly removed (Baratova et al., 1992a). For TMV, on the other hand, tritium planigraphy shows that both N- and C-terminal regions are exposed (Goldanskii et al., 1988), as expected from the X-ray structure (Stubbs, 1999). Some of the additional 80 residues in PVX and NMV compared with TMV might constitute an N-terminal region which tends to bury the C-terminal end, but the ROA spectra suggest that this region is not organized into a β-sheet as proposed by Baratova et al. (1992a). In this connection it is worth noting that there is no similarity in primary sequence of PVX and TMV coat proteins since no alignment of the potexvirus and tobamovirus conserved primary sequences is possible and also that, according to a prediction of Sawyer et al. (1987), the N- terminal 50 residues of PVX have zero probability of being α- helix. This last prediction is unfortunate since it is tempting to assign the greater hydrated α-helix content of PVX and NMV relative to TMV to the extra sequences. TRV exhibits a strong positive ROA band at 1315 cm assigned to polyproline II (PPII)-helical structure in the additional central and C-terminal sequences which the coat proteins of this virus contain compared with TMV (Blanch et al., 2001a). However, since there is no additional positive intensity in this region of the ROA spectra of PVX and NMV compared with TMV, the additional sequences of PVX and NMV do not appear to contain much PPII structure. (All three virus ROA spectra in Fig. 1 show similar small but clear positive ROA bands at 1316 cm assigned to PPII structure.) A few ROA bands from side-chains can also be identified. ROA bands in the range cm originate in both aliphatic and aromatic side-chains. Proteins often show a tryptophan ROA band in the 1550 cm region, assigned to a W3 type vibration of the indole ring, which reflects the sign and magnitude of the torsion angle χ, (Blanch et al., 2001b). The PVX and NMV strains used here contain five and four tryptophans, respectively. The absence of significant ROA in this spectral region of both viruses therefore suggests conformational heterogeneity among these tryptophans (Blanch et al., 2001b). On the other hand, the two and six tyrosines present in PVX and NMV, respectively, could be responsible for the small but significant negative ROA bands at 1604 cm associated with Y8a,b type modes of the aromatic ring (Miura & Thomas, 1995), suggesting these sidechains have mainly fixed conformations generating ROA signals which tend to reinforce. A similar negative band is present at 1608 cm in the ROA spectrum of TMV, which contains four tyrosines. These negative Y8 ROA bands may originate in the participation of some tyrosine side-chains in subunit binding, as observed in the TMV X-ray fibre structure of Namba et al. (1989). Principal component analysis of coat protein folds We are developing a pattern recognition program, based on the multivariate statistical approach of principal component analysis (PCA), to identify protein folds from ROA spectral band patterns. The method is similar to one developed for the analysis of conventional Raman spectra of parchment (Nielsen et al., 1999) and is related to methods used for the determination of the structure of proteins from infrared vibrational circular dichroism (Pancoska et al., 1991) and UVCD (Venyaminov & Yang, 1996) spectra. From the ROA spectral data, the PCA algorithm calculates a set of sub-spectra that serve as basis functions, the algebraic combination of which with appropriate expansion coefficients can be used to reconstruct any member of the original set of experimental ROA spectra. The level of accuracy is determined by the number of basis functions used; for example, a set of ten basis functions is sufficient to reproduce adequately any member of our current set of 56 polypeptide, protein and virus ROA spectra. This set contains the ROA spectra of poly(l-lysine) and poly(l-glutamic acid) in model α-helical and unordered ( random coil ) states, 32 proteins with well-defined tertiary folds known mostly from X-ray crystallography with a few known from multidimensional solution nuclear magnetic resonance, 11 denatured or natively unfolded proteins with structures known to be mostly unordered mainly from spectroscopic techniques such as UVCD, six viruses (including TMV) with coat protein folds known from X-ray crystallography or fibre diffraction and three viruses (PVX, NMV and TRV) with unknown coat protein folds. Since the initial results are promising, we show in Fig. 2 a plot of the coefficients for the whole set of ROA spectra for the two most important basis functions. This provides a two-dimensional representation of the structural relationships among the members of the set. The proteins are colour-coded with respect to the seven different structural types listed on the figure and defined more fully in the caption. These structural types provide a useful initial classification that will be refined and enlarged in later work. Typical examples of these structural types are: alpha, human serum albumin; mainly alpha, hen lysozyme; alpha beta, hen ovalbumin; mainly beta, bovine β-lactoglobulin; beta, jack bean concanavalin A; mainly unordered, bovine β-casein; unordered, hen phosvitin. This provides a starting point for the pattern recognition method since it reveals an initial separation of the spectra into clusters corresponding to different dominant types of protein structural elements, with increasing α-helix content to the left, increasing β-sheet content to the right, and increasing disorder from bottom to top. The way in which CEE
5 Potato virus X and narcissus mosaic virus Fig. 2. Plot of the PCA coefficients for basis function 1 versus basis function 2 for a set of 56 polypeptide, protein and virus ROA spectra. PVX, NMV and TMV cluster together within the mainly alpha region. More complete definitions of the structural types are as follows: alpha, 60% α-helix with the rest mainly unordered and little or no β-sheet; mainly alpha, 35% α-helix and a small amount of β-sheet ( 5 15%); alpha beta, similar significant amounts of α-helix and β-sheet; mainly beta, 35% β-sheet and a small amount of α-helix ( 5 15%); beta, 45% β-sheet with the rest mainly unordered and little or no α-helix. PVX, NMV and TMV cluster together suggests that the folds of their coat proteins may be similar, but with those of PVX and NMV more similar to each other (since their positions are almost identical) than to that of TMV (which is shifted a little from the other two). This cluster lies within the mainly alpha region and so reinforces our conclusion above that the model of Baratova et al. (1992a) for the PVX coat proteins contains too much β-sheet. Plots of other coefficients, which will be given in a later paper, provide further discrimination between different structural types. We have shown these preliminary results here because this unbiased mathematical analysis of the ROA spectral data reinforces the visual impression of a close similarity between the coat protein folds of PVX, NMV and TMV, and because pattern recognition methods are expected to become increasingly important in future applications of ROA to structural virology. The overall structures of PVX and NMV Despite the lack of significant amino acid sequence homology to the TMV coat protein, Sawyer et al. (1987) suggested on the basis of predicted α-helical segments that the PVX coat protein exhibits an approximate twofold symmetry between the amino and carboxyl halves of the molecule such that the α-helices align themselves in pairs like in the TMV coat protein. This reinforces our conclusion from the ROA data that the PVX, NMV and TMV coat protein folds are similar. Furthermore, the low resolution X-ray fibre diffraction data available for several potexviruses are consistent with α-helices packed approximately at right angles to the viral axis, as in TMV (Stubbs, 1989). Hence the helical architecture of the PVX and NMV virus particles may be similar to that of TMV, but with different structural parameters. These different structural parameters may be associated with the small differences in the coat protein folds of PVX and NMV compared with TMV indicated by this study and which, together with their extra sequences, would lead to differences in packing of the protein subunits. We thank the BBSRC for a research grant and the EPSRC for a Research Studentship for C. D. S. The Scottish Crop Research Institute is grant-aided by the Scottish Executive Environment and Rural Affairs Department. References Abou Haidar, M. G., Xu, H. & Hefferon, K. L. (1998). Potexvirus isolation and RNA extraction. In Plant Virus Protocols, pp Edited by G. D. Foster & S. C. Taylor. Totowa, NJ: Humana Press. Baratova, L. A., Grebenshchikov, N. I., Dobrov, E. N., Gedrovich, A. V., Kashirin, I. A., Shishkov, A. V., Efimov, A. V., Ja rveku lg, L., Radavsky, Y. L. & Saarma, M. (1992a). The organization of potato virus X coat proteins in virus particles studied by tritium planigraphy and model building. Virology 188, Baratova, L. A., Grebenshchikov, N. I., Shishkov, A. V., Kashirin, I. A., Radavsky, J. L., Ja rveku lg, L. & Saarma, M. (1992b). The topography of the surface of potato virus X: tritium planigraphy and immunological analysis. Journal of General Virology 73, CEF
6 E. W. Blanch and others Barron, L. D. & Hecht, L. (2000). Vibrational Raman optical activity: from fundamentals to biochemical applications. In Circular Dichroism. Principles and Applications, pp Edited by N. Berova, K. Nakanishi & R. W. Woody. New York: John Wiley & Sons. Barron, L. D., Hecht, L., Blanch, E. W. & Bell, A. F. (2000). Solution structure and dynamics of biomolecules from Raman optical activity. Progress in Biophysics & Molecular Biology 73, Bell, A. F., Hecht, L. & Barron, L. D. (1997). Vibrational Raman optical activity as a probe of polyribonucleotide stereochemistry. Journal of the American Chemical Society 119, Bell, A. F., Hecht, L. & Barron, L. D. (1998). Vibrational Raman optical activity of DNA and RNA. Journal of the American Chemical Society 120, Blanch, E. W., Bell, A. F., Hecht, L., Day, L. A. & Barron, L. D. (1999). Raman optical activity of filamentous bacteriophages: hydration of α- helices. Journal of Molecular Biology 290, 1 7. Blanch, E. W., Robinson, D. J., Hecht, L. & Barron, L. D. (2001a). A comparison of the solution structures of tobacco rattle and tobacco mosaic viruses from Raman optical activity. Journal of General Virology 82, Blanch, E. W., Pederson, D. M., Hecht, L., Day, L. A. & Barron, L. D. (2001b). Tryptophan absolute stereochemistry in viral coat proteins from Raman optical activity. Journal of the American Chemical Society 123, Brunt, A. A., Foster, G. D., Morozov, S. Y. & Zavriev, S. K. (2000). Genus Potexvirus. InVirus Taxonomy. Seventh Report of the International Committee on Taxonomy of Viruses, pp Edited by M. H. V. van Regenmortel, C. M. Fauquet, D. H. L. Bishop, E. B. Carstens, M. K. Estes, S. M. Lemon, J. Maniloff, M. A. Mayo, D. J. McGeoch, C. R. Pringle & R. B. Wickner. San Diego: Academic Press. Carey, P. R. (1982). Biochemical Applications of Raman and Resonance Raman Spectroscopies. New York: Academic Press. Goldanskii, V. I., Kashirin, I. A., Shishkov, A. V., Baratova, L. A. & Grebenshchikov, N. I. (1988). The use of thermally activated tritium atoms for structural-biological investigations: the topography of the TMV protein-accessible surface of the virus. Journal of Molecular Biology 201, Goodman, R. M. (1973). Purification of potato virus X and the molecular weight of its coat protein subunits. John Innes Institute, 64th Annual Report, 126. Hecht, L., Barron, L. D., Blanch, E. W., Bell, A. F. & Day, L. A. (1999). Raman optical activity instrument for studies of biopolymer structure and dynamics. Journal of Raman Spectroscopy 30, Homer, R. B. & Goodman, R. M. (1975). Circular dichroism and fluorescence studies on potato virus X and its structural components. Biochimica et Biophysica Acta 378, Jones, R. A. C. (1982). Breakdown of potato virus X resistance gene Nx: selection of a group four strain from strain group three. Plant Pathology 31, Miura, T. & Thomas, G. J., Jr (1995). Raman spectroscopy of proteins and their assemblies. In Subcellular Biochemistry, vol. 24, Proteins: Structure, Function, and Engineering, pp Edited by B. B. Biswas & S. Roy. New York: Plenum Press. Nafie, L. A. (1997). Infrared and Raman vibrational optical activity. Annual Review of Physical Chemistry 48, Namba, K., Pattanayek, R. & Stubbs, G. (1989). Visualization of protein nucleic acid interactions in a virus. Refined structure of intact tobacco mosaic virus at 2 9 A by X-ray fiber diffraction. Journal of Molecular Biology 208, Nielsen, K., Garp, T., Paulsen, A. & Boghosian, S. (1999). Principal component analysis of Raman spectra of parchment. In Proceedings of the International George Papatheodorou Symposium, pp Edited by S. Boghosian, V. Dracopoulos, C. G. Kontoyannis & G. A. Voyiatzis. Greece: Patras Science Park. Pancoska, P., Yasui, S. C. & Keiderling, T. A. (1991). Statistical analysis of the vibrational circular dichroism of selected proteins and relationship to secondary structure. Biochemistry 30, Sawyer, L., Tollin, P. & Wilson, H. R. (1987). A comparison between the predicted secondary structures of potato virus X and papaya mosaic virus coat proteins. Journal of General Virology 68, Stubbs, G. (1989). Virus structure. In Prediction of Protein Structure and Principles of Protein Conformation, pp Edited by G. Fasman. New York: Plenum Press. Stubbs, G. (1999). Tobacco mosaic virus particle structure and the initiation of disassembly. Philosophical Transactions of the Royal Society B 354, Thomas, G. J., Jr. (1987). Viruses and nucleoproteins. In Biological Applications of Raman Spectroscopy, vol. 1, pp Edited by T. G. Spiro. New York: John Wiley & Sons. Thomas, G. J., Jr. (1999). Raman spectroscopy of protein and nucleic acid assemblies. Annual Review of Biophysics and Biomolecular Structure 28, Tollin, P., Wilson, H. R. & Bancroft, J. B. (1980). Further observations on the structure of particles of potato virus X. Journal of General Virology 49, Venyaminov, S. Y. & Yang, J. T. (1996). Determination of protein secondary structure. In Circular Dichroism and the Conformational Analysis of Biomolecules, pp Edited by G. D. Fasman. New York: Plenum Press. Wilson, H. R., Tollin, P., Sawyer, L., Robinson, D. J., Price, N. C. & Kelly, S. M. (1991). Secondary structures of narcissus mosaic virus coat protein. Journal of General Virology 72, Received 29 June 2001; Accepted 13 September 2001 CEG
Raman Optical Activity Comes of Age
 Raman Optical Activity Comes of Age Laurence D. Barron Department of Chemistry, University of Glasgow, Glasgow G12 8QQ, UK E-mail: laurence.barron@glasgow.ac.uk Raman optical activity (ROA) provides vibrational
Raman Optical Activity Comes of Age Laurence D. Barron Department of Chemistry, University of Glasgow, Glasgow G12 8QQ, UK E-mail: laurence.barron@glasgow.ac.uk Raman optical activity (ROA) provides vibrational
Principles of Physical Biochemistry
 Principles of Physical Biochemistry Kensal E. van Hold e W. Curtis Johnso n P. Shing Ho Preface x i PART 1 MACROMOLECULAR STRUCTURE AND DYNAMICS 1 1 Biological Macromolecules 2 1.1 General Principles
Principles of Physical Biochemistry Kensal E. van Hold e W. Curtis Johnso n P. Shing Ho Preface x i PART 1 MACROMOLECULAR STRUCTURE AND DYNAMICS 1 1 Biological Macromolecules 2 1.1 General Principles
Structural Studies of the Flexible Filamentous Plant Viruses
 Structural Studies of the Flexible Filamentous Plant Viruses Caitlin Azzo Dr. Gerald Stubbs BSCI 296 [4] Honors Thesis April 23, 2014 The Plant Viruses of Interest In the Potyviridae family: Wheat streak
Structural Studies of the Flexible Filamentous Plant Viruses Caitlin Azzo Dr. Gerald Stubbs BSCI 296 [4] Honors Thesis April 23, 2014 The Plant Viruses of Interest In the Potyviridae family: Wheat streak
Contents. xiii. Preface v
 Contents Preface Chapter 1 Biological Macromolecules 1.1 General PrincipIes 1.1.1 Macrornolecules 1.2 1.1.2 Configuration and Conformation Molecular lnteractions in Macromolecular Structures 1.2.1 Weak
Contents Preface Chapter 1 Biological Macromolecules 1.1 General PrincipIes 1.1.1 Macrornolecules 1.2 1.1.2 Configuration and Conformation Molecular lnteractions in Macromolecular Structures 1.2.1 Weak
Molecular Modeling lecture 2
 Molecular Modeling 2018 -- lecture 2 Topics 1. Secondary structure 3. Sequence similarity and homology 2. Secondary structure prediction 4. Where do protein structures come from? X-ray crystallography
Molecular Modeling 2018 -- lecture 2 Topics 1. Secondary structure 3. Sequence similarity and homology 2. Secondary structure prediction 4. Where do protein structures come from? X-ray crystallography
Electron Microscopy of Tobacco Mosaic Virus Prepared with the Aid of Negative Staining-Carbon Film Techniques
 J. gen. Virol. (1976), :3I, 265-269 Printed in Great Britain 265 Electron Microscopy of Tobacco Mosaic Virus Prepared with the Aid of Negative Staining-Carbon Film Techniques (Accepted 13 January I976)
J. gen. Virol. (1976), :3I, 265-269 Printed in Great Britain 265 Electron Microscopy of Tobacco Mosaic Virus Prepared with the Aid of Negative Staining-Carbon Film Techniques (Accepted 13 January I976)
The biomolecules of terrestrial life
 Functional groups in biomolecules Groups of atoms that are responsible for the chemical properties of biomolecules The biomolecules of terrestrial life Planets and Astrobiology (2017-2018) G. Vladilo 1
Functional groups in biomolecules Groups of atoms that are responsible for the chemical properties of biomolecules The biomolecules of terrestrial life Planets and Astrobiology (2017-2018) G. Vladilo 1
Physical principles of IR and Raman. Infrared Spectroscopy
 Physical principles of IR and Raman IR results from the absorption of energy by vibrating chemical bonds. Raman scattering results from the same types of transitions, but the selection rules are different
Physical principles of IR and Raman IR results from the absorption of energy by vibrating chemical bonds. Raman scattering results from the same types of transitions, but the selection rules are different
THE UNIVERSITY OF MANITOBA. PAPER NO: 409 LOCATION: Fr. Kennedy Gold Gym PAGE NO: 1 of 6 DEPARTMENT & COURSE NO: CHEM 4630 TIME: 3 HOURS
 PAPER NO: 409 LOCATION: Fr. Kennedy Gold Gym PAGE NO: 1 of 6 DEPARTMENT & COURSE NO: CHEM 4630 TIME: 3 HOURS EXAMINATION: Biochemistry of Proteins EXAMINER: J. O'Neil Section 1: You must answer all of
PAPER NO: 409 LOCATION: Fr. Kennedy Gold Gym PAGE NO: 1 of 6 DEPARTMENT & COURSE NO: CHEM 4630 TIME: 3 HOURS EXAMINATION: Biochemistry of Proteins EXAMINER: J. O'Neil Section 1: You must answer all of
Circular Dichroism. Principles and Applications VCH. Koji Nakanishi, Nina Berova, and Robert W. Woody EDITED BY
 Circular Dichroism Principles and Applications EDITED BY Koji Nakanishi, Nina Berova, and Robert W. Woody VCH Contents Contributors xv Chapter 1 Circular Dichroism: An Introduction 1 Günther Snatzke 1.1
Circular Dichroism Principles and Applications EDITED BY Koji Nakanishi, Nina Berova, and Robert W. Woody VCH Contents Contributors xv Chapter 1 Circular Dichroism: An Introduction 1 Günther Snatzke 1.1
Protein Structure Analysis and Verification. Course S Basics for Biosystems of the Cell exercise work. Maija Nevala, BIO, 67485U 16.1.
 Protein Structure Analysis and Verification Course S-114.2500 Basics for Biosystems of the Cell exercise work Maija Nevala, BIO, 67485U 16.1.2008 1. Preface When faced with an unknown protein, scientists
Protein Structure Analysis and Verification Course S-114.2500 Basics for Biosystems of the Cell exercise work Maija Nevala, BIO, 67485U 16.1.2008 1. Preface When faced with an unknown protein, scientists
CAP 5510 Lecture 3 Protein Structures
 CAP 5510 Lecture 3 Protein Structures Su-Shing Chen Bioinformatics CISE 8/19/2005 Su-Shing Chen, CISE 1 Protein Conformation 8/19/2005 Su-Shing Chen, CISE 2 Protein Conformational Structures Hydrophobicity
CAP 5510 Lecture 3 Protein Structures Su-Shing Chen Bioinformatics CISE 8/19/2005 Su-Shing Chen, CISE 1 Protein Conformation 8/19/2005 Su-Shing Chen, CISE 2 Protein Conformational Structures Hydrophobicity
Introduction to" Protein Structure
 Introduction to" Protein Structure Function, evolution & experimental methods Thomas Blicher, Center for Biological Sequence Analysis Learning Objectives Outline the basic levels of protein structure.
Introduction to" Protein Structure Function, evolution & experimental methods Thomas Blicher, Center for Biological Sequence Analysis Learning Objectives Outline the basic levels of protein structure.
What happens when light falls on a material? Transmission Reflection Absorption Luminescence. Elastic Scattering Inelastic Scattering
 Raman Spectroscopy What happens when light falls on a material? Transmission Reflection Absorption Luminescence Elastic Scattering Inelastic Scattering Raman, Fluorescence and IR Scattering Absorption
Raman Spectroscopy What happens when light falls on a material? Transmission Reflection Absorption Luminescence Elastic Scattering Inelastic Scattering Raman, Fluorescence and IR Scattering Absorption
Use of DLS/Raman to study the thermal unfolding process of lysozyme
 Use of DLS/Raman to study the thermal unfolding process of lysozyme A Malvern Instruments' Bioscience Development Initiative Executive summary The combination of dynamic light scattering (DLS) and Raman
Use of DLS/Raman to study the thermal unfolding process of lysozyme A Malvern Instruments' Bioscience Development Initiative Executive summary The combination of dynamic light scattering (DLS) and Raman
Basics of protein structure
 Today: 1. Projects a. Requirements: i. Critical review of one paper ii. At least one computational result b. Noon, Dec. 3 rd written report and oral presentation are due; submit via email to bphys101@fas.harvard.edu
Today: 1. Projects a. Requirements: i. Critical review of one paper ii. At least one computational result b. Noon, Dec. 3 rd written report and oral presentation are due; submit via email to bphys101@fas.harvard.edu
Nanobiotechnology. Place: IOP 1 st Meeting Room Time: 9:30-12:00. Reference: Review Papers. Grade: 40% midterm, 60% final report (oral + written)
 Nanobiotechnology Place: IOP 1 st Meeting Room Time: 9:30-12:00 Reference: Review Papers Grade: 40% midterm, 60% final report (oral + written) Midterm: 5/18 Oral Presentation 1. 20 minutes each person
Nanobiotechnology Place: IOP 1 st Meeting Room Time: 9:30-12:00 Reference: Review Papers Grade: 40% midterm, 60% final report (oral + written) Midterm: 5/18 Oral Presentation 1. 20 minutes each person
NH 2. Biochemistry I, Fall Term Sept 9, Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter
 Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Chapter 25 Organic and Biological Chemistry
 Chapter 25 Organic and Biological Chemistry Organic Chemistry The chemistry of carbon compounds. Carbon has the ability to form long chains. Without this property, large biomolecules such as proteins,
Chapter 25 Organic and Biological Chemistry Organic Chemistry The chemistry of carbon compounds. Carbon has the ability to form long chains. Without this property, large biomolecules such as proteins,
1. (5) Draw a diagram of an isomeric molecule to demonstrate a structural, geometric, and an enantiomer organization.
 Organic Chemistry Assignment Score. Name Sec.. Date. Working by yourself or in a group, answer the following questions about the Organic Chemistry material. This assignment is worth 35 points with the
Organic Chemistry Assignment Score. Name Sec.. Date. Working by yourself or in a group, answer the following questions about the Organic Chemistry material. This assignment is worth 35 points with the
Chemistry 524--Final Exam--Keiderling May 4, :30 -?? pm SES
 Chemistry 524--Final Exam--Keiderling May 4, 2011 3:30 -?? pm -- 4286 SES Please answer all questions in the answer book provided. Calculators, rulers, pens and pencils are permitted. No open books or
Chemistry 524--Final Exam--Keiderling May 4, 2011 3:30 -?? pm -- 4286 SES Please answer all questions in the answer book provided. Calculators, rulers, pens and pencils are permitted. No open books or
ion mobility spectrometry IR spectroscopy
 Debasmita Gho 29.10.2016 Introducti on Owing to its accuracy, sensitivity, and speed, mass spectrometry (MS) coupled to fragmentation techniques is the method of choice for determining the primary structure
Debasmita Gho 29.10.2016 Introducti on Owing to its accuracy, sensitivity, and speed, mass spectrometry (MS) coupled to fragmentation techniques is the method of choice for determining the primary structure
The ideal fiber pattern exhibits 4-quadrant symmetry. In the ideal pattern the fiber axis is called the meridian, the perpendicular direction is
 Fiber diffraction is a method used to determine the structural information of a molecule by using scattering data from X-rays. Rosalind Franklin used this technique in discovering structural information
Fiber diffraction is a method used to determine the structural information of a molecule by using scattering data from X-rays. Rosalind Franklin used this technique in discovering structural information
Physiochemical Properties of Residues
 Physiochemical Properties of Residues Various Sources C N Cα R Slide 1 Conformational Propensities Conformational Propensity is the frequency in which a residue adopts a given conformation (in a polypeptide)
Physiochemical Properties of Residues Various Sources C N Cα R Slide 1 Conformational Propensities Conformational Propensity is the frequency in which a residue adopts a given conformation (in a polypeptide)
Protein folding. Today s Outline
 Protein folding Today s Outline Review of previous sessions Thermodynamics of folding and unfolding Determinants of folding Techniques for measuring folding The folding process The folding problem: Prediction
Protein folding Today s Outline Review of previous sessions Thermodynamics of folding and unfolding Determinants of folding Techniques for measuring folding The folding process The folding problem: Prediction
CHAPTER 29 HW: AMINO ACIDS + PROTEINS
 CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
IR Spectrography - Absorption. Raman Spectrography - Scattering. n 0 n M - Raman n 0 - Rayleigh
 RAMAN SPECTROSCOPY Scattering Mid-IR and NIR require absorption of radiation from a ground level to an excited state, requires matching of radiation from source with difference in energy states. Raman
RAMAN SPECTROSCOPY Scattering Mid-IR and NIR require absorption of radiation from a ground level to an excited state, requires matching of radiation from source with difference in energy states. Raman
Application of Raman Spectroscopy for Noninvasive Detection of Target Compounds. Kyung-Min Lee
 Application of Raman Spectroscopy for Noninvasive Detection of Target Compounds Kyung-Min Lee Office of the Texas State Chemist, Texas AgriLife Research January 24, 2012 OTSC Seminar OFFICE OF THE TEXAS
Application of Raman Spectroscopy for Noninvasive Detection of Target Compounds Kyung-Min Lee Office of the Texas State Chemist, Texas AgriLife Research January 24, 2012 OTSC Seminar OFFICE OF THE TEXAS
INTRODUCTION TO MODERN VIBRATIONAL SPECTROSCOPY
 INTRODUCTION TO MODERN VIBRATIONAL SPECTROSCOPY MAX DIEM Department of Chemistry City University of New York Hunter College A Wiley-Interscience Publication JOHN WILEY & SONS New York Chichester Brisbane
INTRODUCTION TO MODERN VIBRATIONAL SPECTROSCOPY MAX DIEM Department of Chemistry City University of New York Hunter College A Wiley-Interscience Publication JOHN WILEY & SONS New York Chichester Brisbane
Spectroscopy Chapter 13
 Spectroscopy Chapter 13 Electromagnetic Spectrum Electromagnetic spectrum in terms of wavelength, frequency and Energy c=λν c= speed of light in a vacuum 3x108 m/s v= frequency in Hertz (Hz s-1 ) λ= wavelength
Spectroscopy Chapter 13 Electromagnetic Spectrum Electromagnetic spectrum in terms of wavelength, frequency and Energy c=λν c= speed of light in a vacuum 3x108 m/s v= frequency in Hertz (Hz s-1 ) λ= wavelength
Protein Structure. W. M. Grogan, Ph.D. OBJECTIVES
 Protein Structure W. M. Grogan, Ph.D. OBJECTIVES 1. Describe the structure and characteristic properties of typical proteins. 2. List and describe the four levels of structure found in proteins. 3. Relate
Protein Structure W. M. Grogan, Ph.D. OBJECTIVES 1. Describe the structure and characteristic properties of typical proteins. 2. List and describe the four levels of structure found in proteins. 3. Relate
Enzyme Catalysis & Biotechnology
 L28-1 Enzyme Catalysis & Biotechnology Bovine Pancreatic RNase A Biochemistry, Life, and all that L28-2 A brief word about biochemistry traditionally, chemical engineers used organic and inorganic chemistry
L28-1 Enzyme Catalysis & Biotechnology Bovine Pancreatic RNase A Biochemistry, Life, and all that L28-2 A brief word about biochemistry traditionally, chemical engineers used organic and inorganic chemistry
4 Proteins: Structure, Function, Folding W. H. Freeman and Company
 4 Proteins: Structure, Function, Folding 2013 W. H. Freeman and Company CHAPTER 4 Proteins: Structure, Function, Folding Learning goals: Structure and properties of the peptide bond Structural hierarchy
4 Proteins: Structure, Function, Folding 2013 W. H. Freeman and Company CHAPTER 4 Proteins: Structure, Function, Folding Learning goals: Structure and properties of the peptide bond Structural hierarchy
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor
 LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
Biomolecules. Energetics in biology. Biomolecules inside the cell
 Biomolecules Energetics in biology Biomolecules inside the cell Energetics in biology The production of energy, its storage, and its use are central to the economy of the cell. Energy may be defined as
Biomolecules Energetics in biology Biomolecules inside the cell Energetics in biology The production of energy, its storage, and its use are central to the economy of the cell. Energy may be defined as
2: CHEMICAL COMPOSITION OF THE BODY
 1 2: CHEMICAL COMPOSITION OF THE BODY Although most students of human physiology have had at least some chemistry, this chapter serves very well as a review and as a glossary of chemical terms. In particular,
1 2: CHEMICAL COMPOSITION OF THE BODY Although most students of human physiology have had at least some chemistry, this chapter serves very well as a review and as a glossary of chemical terms. In particular,
DOWNLOAD OR READ : INFRARED AND RAMAN SPECTROSCOPY CONCEPTS AND APPLICATIONS PDF EBOOK EPUB MOBI
 DOWNLOAD OR READ : INFRARED AND RAMAN SPECTROSCOPY CONCEPTS AND APPLICATIONS PDF EBOOK EPUB MOBI Page 1 Page 2 infrared and raman spectroscopy concepts and applications infrared and raman spectroscopy
DOWNLOAD OR READ : INFRARED AND RAMAN SPECTROSCOPY CONCEPTS AND APPLICATIONS PDF EBOOK EPUB MOBI Page 1 Page 2 infrared and raman spectroscopy concepts and applications infrared and raman spectroscopy
Circular Dichroism Studies on Turnip Rosette Virus
 J. gen. Virol. (I978), 4 r, 77-85 Printed in Great Britain 77 Circular Dichroism Studies on Turnip Rosette Virus ABIMBOLA O. DENLOYE,* R. B. HOMER,* AND R. HULLt * Department of Chemical Sciences, University
J. gen. Virol. (I978), 4 r, 77-85 Printed in Great Britain 77 Circular Dichroism Studies on Turnip Rosette Virus ABIMBOLA O. DENLOYE,* R. B. HOMER,* AND R. HULLt * Department of Chemical Sciences, University
Circular Dichroism & Optical Rotatory Dispersion. Proteins (KCsa) Polysaccharides (agarose) DNA CHEM 305. Many biomolecules are α-helical!
 Circular Dichroism & Optical Rotatory Dispersion Polysaccharides (agarose) DNA Proteins (KCsa) Many biomolecules are α-helical! How can we measure the amount and changes in amount of helical structure
Circular Dichroism & Optical Rotatory Dispersion Polysaccharides (agarose) DNA Proteins (KCsa) Many biomolecules are α-helical! How can we measure the amount and changes in amount of helical structure
BIOCHEMISTRY GUIDED NOTES - AP BIOLOGY-
 BIOCHEMISTRY GUIDED NOTES - AP BIOLOGY- ELEMENTS AND COMPOUNDS - anything that has mass and takes up space. - cannot be broken down to other substances. - substance containing two or more different elements
BIOCHEMISTRY GUIDED NOTES - AP BIOLOGY- ELEMENTS AND COMPOUNDS - anything that has mass and takes up space. - cannot be broken down to other substances. - substance containing two or more different elements
Code Course name CFU Year G6403B Info not available 3 1
 Basic aims The aim of the course is an in-depth discussion of the structureactivity relationships of the main classes of biological molecules. Strategies for synthesis, isolation and structural characterization
Basic aims The aim of the course is an in-depth discussion of the structureactivity relationships of the main classes of biological molecules. Strategies for synthesis, isolation and structural characterization
Understanding the conformational stability of protein therapeutics using Raman spectroscopy
 Understanding the conformational stability of protein therapeutics using Raman spectroscopy A Malvern Instruments' Bioscience Development Initiative Executive summary The combination of Dynamic Light Scattering
Understanding the conformational stability of protein therapeutics using Raman spectroscopy A Malvern Instruments' Bioscience Development Initiative Executive summary The combination of Dynamic Light Scattering
The topography of the surface of potato virus X: tritium planigraphy and immunological analysis
 Journal of General Virology (1992), 73, 229-235. Printed in Great Britain 229 The topography of the surface of potato virus X: tritium planigraphy and immunological analysis L. A. Baratova, 1 N. I. Grebenshchikov,
Journal of General Virology (1992), 73, 229-235. Printed in Great Britain 229 The topography of the surface of potato virus X: tritium planigraphy and immunological analysis L. A. Baratova, 1 N. I. Grebenshchikov,
Course Syllabus. Department: Science & Technology. Date: April I. Course Prefix and Number: CHM 212. Course Name: Organic Chemistry II
 Department: Science & Technology Date: April 2012 I. Course Prefix and Number: CHM 212 Course Name: Organic Chemistry II Course Syllabus Credit Hours and Contact Hours: 5 credit hours and 7 (3:3:1) contact
Department: Science & Technology Date: April 2012 I. Course Prefix and Number: CHM 212 Course Name: Organic Chemistry II Course Syllabus Credit Hours and Contact Hours: 5 credit hours and 7 (3:3:1) contact
From Amino Acids to Proteins - in 4 Easy Steps
 From Amino Acids to Proteins - in 4 Easy Steps Although protein structure appears to be overwhelmingly complex, you can provide your students with a basic understanding of how proteins fold by focusing
From Amino Acids to Proteins - in 4 Easy Steps Although protein structure appears to be overwhelmingly complex, you can provide your students with a basic understanding of how proteins fold by focusing
CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray
 CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray crystallography An example of the use of CD Modeling
CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray crystallography An example of the use of CD Modeling
Examples of Protein Modeling. Protein Modeling. Primary Structure. Protein Structure Description. Protein Sequence Sources. Importing Sequences to MOE
 Examples of Protein Modeling Protein Modeling Visualization Examination of an experimental structure to gain insight about a research question Dynamics To examine the dynamics of protein structures To
Examples of Protein Modeling Protein Modeling Visualization Examination of an experimental structure to gain insight about a research question Dynamics To examine the dynamics of protein structures To
Statistical Machine Learning Methods for Bioinformatics IV. Neural Network & Deep Learning Applications in Bioinformatics
 Statistical Machine Learning Methods for Bioinformatics IV. Neural Network & Deep Learning Applications in Bioinformatics Jianlin Cheng, PhD Department of Computer Science University of Missouri, Columbia
Statistical Machine Learning Methods for Bioinformatics IV. Neural Network & Deep Learning Applications in Bioinformatics Jianlin Cheng, PhD Department of Computer Science University of Missouri, Columbia
Chapter 19. Gene creatures, Part 1: viruses, viroids and plasmids. Prepared by Woojoo Choi
 Chapter 19. Gene creatures, Part 1: viruses, viroids and plasmids Prepared by Woojoo Choi Dead or alive? 1) In this chapter we will explore the twilight zone of biology and the gene creature who live there.
Chapter 19. Gene creatures, Part 1: viruses, viroids and plasmids Prepared by Woojoo Choi Dead or alive? 1) In this chapter we will explore the twilight zone of biology and the gene creature who live there.
PROTEIN STRUCTURE AMINO ACIDS H R. Zwitterion (dipolar ion) CO 2 H. PEPTIDES Formal reactions showing formation of peptide bond by dehydration:
 PTEI STUTUE ydrolysis of proteins with aqueous acid or base yields a mixture of free amino acids. Each type of protein yields a characteristic mixture of the ~ 20 amino acids. AMI AIDS Zwitterion (dipolar
PTEI STUTUE ydrolysis of proteins with aqueous acid or base yields a mixture of free amino acids. Each type of protein yields a characteristic mixture of the ~ 20 amino acids. AMI AIDS Zwitterion (dipolar
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. The asymmetric unit in para-iodio-phenylalanine crystal. The 50% probability ellipsoid representation was prepared using the Mercury Software. Colors are as
Supplementary Figures Supplementary Figure 1. The asymmetric unit in para-iodio-phenylalanine crystal. The 50% probability ellipsoid representation was prepared using the Mercury Software. Colors are as
Ch 3: Chemistry of Life. Chemistry Water Macromolecules Enzymes
 Ch 3: Chemistry of Life Chemistry Water Macromolecules Enzymes Chemistry Atom = smallest unit of matter that cannot be broken down by chemical means Element = substances that have similar properties and
Ch 3: Chemistry of Life Chemistry Water Macromolecules Enzymes Chemistry Atom = smallest unit of matter that cannot be broken down by chemical means Element = substances that have similar properties and
and conformational flexibility of (bio)molecules
 Raman optical activity (ROA) and conformational flexibility of (bio)molecules in solution Vladimír r Baumruk Charles University in Prague Faculty of Mathematics and Physics Institute of Physics seminář
Raman optical activity (ROA) and conformational flexibility of (bio)molecules in solution Vladimír r Baumruk Charles University in Prague Faculty of Mathematics and Physics Institute of Physics seminář
F. Piazza Center for Molecular Biophysics and University of Orléans, France. Selected topic in Physical Biology. Lecture 1
 Zhou Pei-Yuan Centre for Applied Mathematics, Tsinghua University November 2013 F. Piazza Center for Molecular Biophysics and University of Orléans, France Selected topic in Physical Biology Lecture 1
Zhou Pei-Yuan Centre for Applied Mathematics, Tsinghua University November 2013 F. Piazza Center for Molecular Biophysics and University of Orléans, France Selected topic in Physical Biology Lecture 1
Supporting Information
 1 Supporting Information 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 Materials and Methods Experiment In this study, an alloy IR probe which allowed us to get access to spectral
1 Supporting Information 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 Materials and Methods Experiment In this study, an alloy IR probe which allowed us to get access to spectral
1 Introduction FOURIER TRANSFORM INFRARED STUDIES IN SOLID EGG WHITE LYSOZYME. 1C/94/403 INTERNAL REPORT (Limited Distribution)
 1C/94/403 INTERNAL REPORT (Limited Distribution) International Atomic Energy Agency and Nations Educational Scientific and Cultural Organization INTERNATIONAL CENTRE FOR THEORETICAL PHYSICS 1 Introduction
1C/94/403 INTERNAL REPORT (Limited Distribution) International Atomic Energy Agency and Nations Educational Scientific and Cultural Organization INTERNATIONAL CENTRE FOR THEORETICAL PHYSICS 1 Introduction
Vladimír Baumruk Charles University in Prague Faculty of Mathematics and Physics Institute of Physics
 Spectral Techniques for Rapid Quantification of Protein Structure in Solution Vladimír Baumruk Charles University in Prague Faculty of Mathematics and Physics Institute of Physics primary structure Protein
Spectral Techniques for Rapid Quantification of Protein Structure in Solution Vladimír Baumruk Charles University in Prague Faculty of Mathematics and Physics Institute of Physics primary structure Protein
Supporting Information
 Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2013. Supporting Information for Adv. Mater., DOI: 10.1002/adma.201301472 Reconfigurable Infrared Camouflage Coatings from a Cephalopod
Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2013. Supporting Information for Adv. Mater., DOI: 10.1002/adma.201301472 Reconfigurable Infrared Camouflage Coatings from a Cephalopod
HOMOLOGY MODELING. The sequence alignment and template structure are then used to produce a structural model of the target.
 HOMOLOGY MODELING Homology modeling, also known as comparative modeling of protein refers to constructing an atomic-resolution model of the "target" protein from its amino acid sequence and an experimental
HOMOLOGY MODELING Homology modeling, also known as comparative modeling of protein refers to constructing an atomic-resolution model of the "target" protein from its amino acid sequence and an experimental
Analysis and Prediction of Protein Structure (I)
 Analysis and Prediction of Protein Structure (I) Jianlin Cheng, PhD School of Electrical Engineering and Computer Science University of Central Florida 2006 Free for academic use. Copyright @ Jianlin Cheng
Analysis and Prediction of Protein Structure (I) Jianlin Cheng, PhD School of Electrical Engineering and Computer Science University of Central Florida 2006 Free for academic use. Copyright @ Jianlin Cheng
Computational Molecular Modeling
 Computational Molecular Modeling Lecture 1: Structure Models, Properties Chandrajit Bajaj Today s Outline Intro to atoms, bonds, structure, biomolecules, Geometry of Proteins, Nucleic Acids, Ribosomes,
Computational Molecular Modeling Lecture 1: Structure Models, Properties Chandrajit Bajaj Today s Outline Intro to atoms, bonds, structure, biomolecules, Geometry of Proteins, Nucleic Acids, Ribosomes,
Infrared Spectroscopy
 Infrared Spectroscopy Introduction Spectroscopy is an analytical technique which helps determine structure. It destroys little or no sample. The amount of light absorbed by the sample is measured as wavelength
Infrared Spectroscopy Introduction Spectroscopy is an analytical technique which helps determine structure. It destroys little or no sample. The amount of light absorbed by the sample is measured as wavelength
BSc and MSc Degree Examinations
 Examination Candidate Number: Desk Number: BSc and MSc Degree Examinations 2018-9 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes Marking
Examination Candidate Number: Desk Number: BSc and MSc Degree Examinations 2018-9 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes Marking
Central Dogma. modifications genome transcriptome proteome
 entral Dogma DA ma protein post-translational modifications genome transcriptome proteome 83 ierarchy of Protein Structure 20 Amino Acids There are 20 n possible sequences for a protein of n residues!
entral Dogma DA ma protein post-translational modifications genome transcriptome proteome 83 ierarchy of Protein Structure 20 Amino Acids There are 20 n possible sequences for a protein of n residues!
Protein Structure Basics
 Protein Structure Basics Presented by Alison Fraser, Christine Lee, Pradhuman Jhala, Corban Rivera Importance of Proteins Muscle structure depends on protein-protein interactions Transport across membranes
Protein Structure Basics Presented by Alison Fraser, Christine Lee, Pradhuman Jhala, Corban Rivera Importance of Proteins Muscle structure depends on protein-protein interactions Transport across membranes
Introduction to Comparative Protein Modeling. Chapter 4 Part I
 Introduction to Comparative Protein Modeling Chapter 4 Part I 1 Information on Proteins Each modeling study depends on the quality of the known experimental data. Basis of the model Search in the literature
Introduction to Comparative Protein Modeling Chapter 4 Part I 1 Information on Proteins Each modeling study depends on the quality of the known experimental data. Basis of the model Search in the literature
Trpzip-based beta hairpin temperature jump IR studies enhanced by sitespecific
 Trpzip-based beta hairpin temperature jump IR studies enhanced by sitespecific isotope labeling Carsten Kretjschi 1, Karin auser 1, Rong uang 2, Tim Keiderling 2 1 Institute of Biophysics, University of
Trpzip-based beta hairpin temperature jump IR studies enhanced by sitespecific isotope labeling Carsten Kretjschi 1, Karin auser 1, Rong uang 2, Tim Keiderling 2 1 Institute of Biophysics, University of
BIOCHEMISTRY Course Outline (Fall, 2011)
 BIOCHEMISTRY 402 - Course Outline (Fall, 2011) Number OVERVIEW OF LECTURE TOPICS: of Lectures INSTRUCTOR 1. Structural Components of Proteins G. Brayer (a) Amino Acids and the Polypeptide Chain Backbone...2
BIOCHEMISTRY 402 - Course Outline (Fall, 2011) Number OVERVIEW OF LECTURE TOPICS: of Lectures INSTRUCTOR 1. Structural Components of Proteins G. Brayer (a) Amino Acids and the Polypeptide Chain Backbone...2
Analytical Technologies in Biotechnology Prof. Dr. Ashwani K Sharma Department of Biotechnology Indian Institute of Technology, Roorkee
 Analytical Technologies in Biotechnology Prof. Dr. Ashwani K Sharma Department of Biotechnology Indian Institute of Technology, Roorkee Module - 6 Spectroscopic Techniques Lecture - 2 UV-Visible Spectroscopy
Analytical Technologies in Biotechnology Prof. Dr. Ashwani K Sharma Department of Biotechnology Indian Institute of Technology, Roorkee Module - 6 Spectroscopic Techniques Lecture - 2 UV-Visible Spectroscopy
Protein Dynamics. The space-filling structures of myoglobin and hemoglobin show that there are no pathways for O 2 to reach the heme iron.
 Protein Dynamics The space-filling structures of myoglobin and hemoglobin show that there are no pathways for O 2 to reach the heme iron. Below is myoglobin hydrated with 350 water molecules. Only a small
Protein Dynamics The space-filling structures of myoglobin and hemoglobin show that there are no pathways for O 2 to reach the heme iron. Below is myoglobin hydrated with 350 water molecules. Only a small
Fluorescence Workshop UMN Physics June 8-10, 2006 Basic Spectroscopic Principles Joachim Mueller
 Fluorescence Workshop UMN Physics June 8-10, 2006 Basic Spectroscopic Principles Joachim Mueller Fluorescence, Light, Absorption, Jablonski Diagram, and Beer-Law First stab at a definition: What is fluorescence?
Fluorescence Workshop UMN Physics June 8-10, 2006 Basic Spectroscopic Principles Joachim Mueller Fluorescence, Light, Absorption, Jablonski Diagram, and Beer-Law First stab at a definition: What is fluorescence?
Full file at
 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following is an uncharged particle found in the nucleus of 1) an atom and which has
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following is an uncharged particle found in the nucleus of 1) an atom and which has
Problem Set 1
 2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
ALL LECTURES IN SB Introduction
 1. Introduction 2. Molecular Architecture I 3. Molecular Architecture II 4. Molecular Simulation I 5. Molecular Simulation II 6. Bioinformatics I 7. Bioinformatics II 8. Prediction I 9. Prediction II ALL
1. Introduction 2. Molecular Architecture I 3. Molecular Architecture II 4. Molecular Simulation I 5. Molecular Simulation II 6. Bioinformatics I 7. Bioinformatics II 8. Prediction I 9. Prediction II ALL
CONFOCHECK. Innovation with Integrity. Infrared Protein Analysis FT-IR
 CONFOCHECK Infrared Protein Analysis Innovation with Integrity FT-IR CONFOCHECK: FT-IR System for Protein Analytics FT-IR Protein Analysis Infrared spectroscopy measures molecular vibrations due to the
CONFOCHECK Infrared Protein Analysis Innovation with Integrity FT-IR CONFOCHECK: FT-IR System for Protein Analytics FT-IR Protein Analysis Infrared spectroscopy measures molecular vibrations due to the
Copyright Mark Brandt, Ph.D A third method, cryogenic electron microscopy has seen increasing use over the past few years.
 Structure Determination and Sequence Analysis The vast majority of the experimentally determined three-dimensional protein structures have been solved by one of two methods: X-ray diffraction and Nuclear
Structure Determination and Sequence Analysis The vast majority of the experimentally determined three-dimensional protein structures have been solved by one of two methods: X-ray diffraction and Nuclear
Chapter 1 1) Biological Molecules a) Only a small subset of the known elements are found in living systems i) Most abundant- C, N, O, and H ii) Less
 Chapter 1 1) Biological Molecules a) Only a small subset of the known elements are found in living systems i) Most abundant- C, N, O, and H ii) Less abundant- Ca, P, K, S, Cl, Na, and Mg b) Cells contain
Chapter 1 1) Biological Molecules a) Only a small subset of the known elements are found in living systems i) Most abundant- C, N, O, and H ii) Less abundant- Ca, P, K, S, Cl, Na, and Mg b) Cells contain
Optical Activity as a Biosignature in the Search for Extraterrestrial Life
 ptical Activity as a Biosignature in the Search for Extraterrestrial Life Andrew K. Boal AST740 21 April 2006 utline of this talk Science ow will we detect life on other planets? Some proposed biosignatures
ptical Activity as a Biosignature in the Search for Extraterrestrial Life Andrew K. Boal AST740 21 April 2006 utline of this talk Science ow will we detect life on other planets? Some proposed biosignatures
Application of IR Raman Spectroscopy
 Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Introduction to Computational Structural Biology
 Introduction to Computational Structural Biology Part I 1. Introduction The disciplinary character of Computational Structural Biology The mathematical background required and the topics covered Bibliography
Introduction to Computational Structural Biology Part I 1. Introduction The disciplinary character of Computational Structural Biology The mathematical background required and the topics covered Bibliography
Chapter 2. Chemical Principles
 Chapter 2 Chemical Principles Insert Fig CO 2 The Structure of Atoms Chemistry is the study of interactions between atoms and molecules The atom is the smallest unit of matter that enters into chemical
Chapter 2 Chemical Principles Insert Fig CO 2 The Structure of Atoms Chemistry is the study of interactions between atoms and molecules The atom is the smallest unit of matter that enters into chemical
Protein Structure Prediction II Lecturer: Serafim Batzoglou Scribe: Samy Hamdouche
 Protein Structure Prediction II Lecturer: Serafim Batzoglou Scribe: Samy Hamdouche The molecular structure of a protein can be broken down hierarchically. The primary structure of a protein is simply its
Protein Structure Prediction II Lecturer: Serafim Batzoglou Scribe: Samy Hamdouche The molecular structure of a protein can be broken down hierarchically. The primary structure of a protein is simply its
Model Worksheet Teacher Key
 Introduction Despite the complexity of life on Earth, the most important large molecules found in all living things (biomolecules) can be classified into only four main categories: carbohydrates, lipids,
Introduction Despite the complexity of life on Earth, the most important large molecules found in all living things (biomolecules) can be classified into only four main categories: carbohydrates, lipids,
HIV protease inhibitor. Certain level of function can be found without structure. But a structure is a key to understand the detailed mechanism.
 Proteins are linear polypeptide chains (one or more) Building blocks: 20 types of amino acids. Range from a few 10s-1000s They fold into varying three-dimensional shapes structure medicine Certain level
Proteins are linear polypeptide chains (one or more) Building blocks: 20 types of amino acids. Range from a few 10s-1000s They fold into varying three-dimensional shapes structure medicine Certain level
Chapter 4: Amino Acids
 Chapter 4: Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. lipid polysaccharide enzyme 1940s 1980s. Lipids membrane 1960s. Polysaccharide Are energy metabolites and many of
Chapter 4: Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. lipid polysaccharide enzyme 1940s 1980s. Lipids membrane 1960s. Polysaccharide Are energy metabolites and many of
Determining Protein Structure BIBC 100
 Determining Protein Structure BIBC 100 Determining Protein Structure X-Ray Diffraction Interactions of x-rays with electrons in molecules in a crystal NMR- Nuclear Magnetic Resonance Interactions of magnetic
Determining Protein Structure BIBC 100 Determining Protein Structure X-Ray Diffraction Interactions of x-rays with electrons in molecules in a crystal NMR- Nuclear Magnetic Resonance Interactions of magnetic
Model Mélange. Physical Models of Peptides and Proteins
 Model Mélange Physical Models of Peptides and Proteins In the Model Mélange activity, you will visit four different stations each featuring a variety of different physical models of peptides or proteins.
Model Mélange Physical Models of Peptides and Proteins In the Model Mélange activity, you will visit four different stations each featuring a variety of different physical models of peptides or proteins.
1. What is an ångstrom unit, and why is it used to describe molecular structures?
 1. What is an ångstrom unit, and why is it used to describe molecular structures? The ångstrom unit is a unit of distance suitable for measuring atomic scale objects. 1 ångstrom (Å) = 1 10-10 m. The diameter
1. What is an ångstrom unit, and why is it used to describe molecular structures? The ångstrom unit is a unit of distance suitable for measuring atomic scale objects. 1 ångstrom (Å) = 1 10-10 m. The diameter
1/23/2012. Atoms. Atoms Atoms - Electron Shells. Chapter 2 Outline. Planetary Models of Elements Chemical Bonds
 Chapter 2 Outline Atoms Chemical Bonds Acids, Bases and the p Scale Organic Molecules Carbohydrates Lipids Proteins Nucleic Acids Are smallest units of the chemical elements Composed of protons, neutrons
Chapter 2 Outline Atoms Chemical Bonds Acids, Bases and the p Scale Organic Molecules Carbohydrates Lipids Proteins Nucleic Acids Are smallest units of the chemical elements Composed of protons, neutrons
MOLECULAR LIGHT SCATTERING AND OPTICAL ACTIVITY
 MOLECULAR LIGHT SCATTERING AND OPTICAL ACTIVITY Second edition, revised and enlarged LAURENCE D. BARRON, F.R.S.E. Gardiner Professor of Chemistry, University of Glasgow 122. CAMBRIDGE UNIVERSITY PRESS
MOLECULAR LIGHT SCATTERING AND OPTICAL ACTIVITY Second edition, revised and enlarged LAURENCE D. BARRON, F.R.S.E. Gardiner Professor of Chemistry, University of Glasgow 122. CAMBRIDGE UNIVERSITY PRESS
Lecture 2 and 3: Review of forces (ctd.) and elementary statistical mechanics. Contributions to protein stability
 Lecture 2 and 3: Review of forces (ctd.) and elementary statistical mechanics. Contributions to protein stability Part I. Review of forces Covalent bonds Non-covalent Interactions: Van der Waals Interactions
Lecture 2 and 3: Review of forces (ctd.) and elementary statistical mechanics. Contributions to protein stability Part I. Review of forces Covalent bonds Non-covalent Interactions: Van der Waals Interactions
To be covered (and why) Spectroscopy of Proteins. UV-Vis Absorption. UV-Vis Absorption. Spectra
 To be covered (and why) Spectroscopy of Proteins General considerations UV-Vis Absorption quantitation Fluorescence hydrophobicity Foldedness FT-Infrared Foldedness ircular Dichroism Foldedness NMR (a
To be covered (and why) Spectroscopy of Proteins General considerations UV-Vis Absorption quantitation Fluorescence hydrophobicity Foldedness FT-Infrared Foldedness ircular Dichroism Foldedness NMR (a
Module 4 : Third order nonlinear optical processes. Lecture 28 : Inelastic Scattering Processes. Objectives
 Module 4 : Third order nonlinear optical processes Lecture 28 : Inelastic Scattering Processes Objectives In this lecture you will learn the following Light scattering- elastic and inelastic-processes,
Module 4 : Third order nonlinear optical processes Lecture 28 : Inelastic Scattering Processes Objectives In this lecture you will learn the following Light scattering- elastic and inelastic-processes,
Presenter: She Zhang
 Presenter: She Zhang Introduction Dr. David Baker Introduction Why design proteins de novo? It is not clear how non-covalent interactions favor one specific native structure over many other non-native
Presenter: She Zhang Introduction Dr. David Baker Introduction Why design proteins de novo? It is not clear how non-covalent interactions favor one specific native structure over many other non-native
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
Outline. Levels of Protein Structure. Primary (1 ) Structure. Lecture 6:Protein Architecture II: Secondary Structure or From peptides to proteins
 Lecture 6:Protein Architecture II: Secondary Structure or From peptides to proteins Margaret Daugherty Fall 2004 Outline Four levels of structure are used to describe proteins; Alpha helices and beta sheets
Lecture 6:Protein Architecture II: Secondary Structure or From peptides to proteins Margaret Daugherty Fall 2004 Outline Four levels of structure are used to describe proteins; Alpha helices and beta sheets
Bi 8 Midterm Review. TAs: Sarah Cohen, Doo Young Lee, Erin Isaza, and Courtney Chen
 Bi 8 Midterm Review TAs: Sarah Cohen, Doo Young Lee, Erin Isaza, and Courtney Chen The Central Dogma Biology Fundamental! Prokaryotes and Eukaryotes Nucleic Acid Components Nucleic Acid Structure DNA Base
Bi 8 Midterm Review TAs: Sarah Cohen, Doo Young Lee, Erin Isaza, and Courtney Chen The Central Dogma Biology Fundamental! Prokaryotes and Eukaryotes Nucleic Acid Components Nucleic Acid Structure DNA Base
Protein Structure. Hierarchy of Protein Structure. Tertiary structure. independently stable structural unit. includes disulfide bonds
 Protein Structure Hierarchy of Protein Structure 2 3 Structural element Primary structure Secondary structure Super-secondary structure Domain Tertiary structure Quaternary structure Description amino
Protein Structure Hierarchy of Protein Structure 2 3 Structural element Primary structure Secondary structure Super-secondary structure Domain Tertiary structure Quaternary structure Description amino
Biochemistry Quiz Review 1I. 1. Of the 20 standard amino acids, only is not optically active. The reason is that its side chain.
 Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
1014NSC Fundamentals of Biochemistry Semester Summary
 1014NSC Fundamentals of Biochemistry Semester Summary Griffith University, Nathan Campus Semester 1, 2014 Topics include: - Water & ph - Protein Diversity - Nucleic Acids - DNA Replication - Transcription
1014NSC Fundamentals of Biochemistry Semester Summary Griffith University, Nathan Campus Semester 1, 2014 Topics include: - Water & ph - Protein Diversity - Nucleic Acids - DNA Replication - Transcription
