CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray
|
|
|
- Bennett Bates
- 5 years ago
- Views:
Transcription
1 CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray crystallography
2 An example of the use of CD Modeling an extracellular loop of the κ opioid receptor second extracellular loop involved in binding to dynorphin Biochemistry (2002) 41, 61-68
3 No secondary structure of the peptide in water but substantial α-helix in the presence of phospholipids (DPC,DHPC,DMPC) or in the solvent hexafluoro-2-propanol 33 amino acid peptide interaction of the peptide with the lipid induces α helix Biochemistry (2002) 41, 61-68
4 Modeling an extracellular loop of the κ receptor combination of CD, fluorescence and NMR yields the model Biochemistry (2002) 41, 61-68
5 Human Carbonic Anhydrase II: an example showing induced CD of tryptophans in a protein in the near UV 7 tryptophans 7% α 28% β Biochem (1994) 33, )
6 CD spectra of Human Carbonic Anhydrase II A: near-uv CD spectrum of HCAII pwt in 0 M GuHCL (solid) and 5 M GuHCL(dashed) B: The Far-UV CD spectrum of HCAII pwt in 0 M GuHCL (solid) and 5 M GuHCL (dashed) Near UV from 7 tryptophans Far UV consistent with α (7%), β (28%) (mol wt) [θ] = θ obs [ ] C (mg/ml) (10)(l cm) deg cm 2 dmol Units of molar ellipticity Biochem 33,
7 Contribution to CD from each tryptophan is different: monitor of tertiary structure. determine CD of sitedirected mutants eliminating each tryptophan and determining the difference spectrum Biochem 33,
8 Fluorescence Spectroscopy and SEX white light Fluorescent Plumage of Parrots is a Sexual Signal! UV illumination Science (2002) 295, 92
9 Fluorescence Spectroscopy Decay from excited state back to ground state with emission of a photon ex Absorption (10-15 sec) ground competition Fluorescence (10-8 sec) Non-radiative decay Fluorescence properties depend on what happens to the molecule during the ~10-8 sec during which it is excited -dynamic properties of biomolecules are probed by fluorescence
10 Example of what can happen to the excited state: collision with another molecule X* + Y XY k In 10-8 sec, a small molecule can diffuse 50Å (water at 25 o C) So an excited state (X*) can be perturbed by a collision with molecule (Y) that is within this distance X* 50Å Y Y
11 Within about sec, all excited state molecules lose heat to decay to the lowest vibronic level of the first excited state S 2 S heat sec: internal conversion heat loss to solvent 0 3 S ABS light heat 10-8 sec: larger energy gap results in slower relaxation to ground electronic state 0
12 The rate of internal conversion within the excited state manifold is due to loss of energy to the solvent via collisions and is dependent on the rate of collisions with the solvent collision rate = k coll [solvent] Collisions with solvent molecules ~10 10 M -1 sec -1 55M for water In 55 M solvent, the rate of collisions of a single molecule is sec -1 S 2 S 1 Due to small energy gaps, collisions are very effective so that within sec all excited state molecules are in the lowest vibrational state of S 1 S 0 However, the non-radiative loss of energy to the ground state, Internal Conversion to S 0,is much slower due to the large energy gap
13 Phosphorescence results from emission of a photon from the lowest triplet state (electron spins aligned) S sec: internal conversion heat S T sec: spin change, singlet to triplet intersystem crossing ABS fluorescence 3 S phosphorescence 10-6 to 10-2 sec: triplet to singlet 0
14 What can you monitor by fluorescence? k f k I (all non-radiative modes) Competition between rate of fluorescence and rates of non-radiative modes Faster non-radiative de-excitation less fluorescence Many processes occur to a molecule during its excited state lifetime that influence the fluorescence: (1) collisions (quenching) accessibility (2) energy transfer distance (3) solvent relaxation solvent polarity (4) chromophore rotation molecular size/viscosity
15 Steady state measurement Most common methods of measuring fluorescence source (continous) monochrometer polarizer sample polarizer detector for measuring absorption monochrometer Dynamic methods detector for fluorescence Pulse spectroscopy: Measure the intensity of emitted light after a very brief pulse of light (nsec duration) -measure fluorescence lifetime Also: Phase and Modulation Spectroscopy See Ann. Rev. Biophys Bioeng. (1984) 13,
16 Excitation Spectrum Dependence of the emission intensity on the excitation wavelength, measured at a constant emission wavelength Emission Spectrum Wavelength dependence of the emission intensity, measured with a constant excitation wavelength ε 0-0 F λ
17 What parameters are measured? (1) Excitation Spectrum (2) Emission Spectrum (3) Quantum Yield (Q f ) (4) Lifetime (τ) (5) Polarization (Anisotropy) S 1 S o λ ex λ em Excitation spectrum Measure fluorescence as a function of excitation wavelength at fixed λ em Identical to the absorption spectrum Since all emission comes from the same energy level (S υ =0 ), the only thing that changes with λ ex is the probability of absorption F(λ ex ) ε(λ ex ) ε 0-0 F λ
18 Emission spectrum Measure fluorescence at fixed λ ex as a function of λ em Emission spectrum is always red-shifted (lower energy) compared to the absorption spectrum S S o ε 0-0 F λ
19 Fluorescence: Mirror-Image Rule S (0 2 ) (0 1 ) (0 0 ) (0 2 ) ε (0 1 ) (0 0 ) (0 0 ) F (1 0 ) (2 0 ) (2 0 ) S o (1 0 ) (0 0 ) Higher energy λ Lower energy Franck-Condon Overlap Factors: Prob (0 2 ) Prob (2 0 ) etc ε 0-0 F λ
20 Fluorescence: Self-Overlap Thermal population of υ = 1 state results in some molecules having blue-shifted emission Solvent-induced heterogeneity also results in self-overlap Thermal population of υ = 1 state results in some molecules with red-shifted absorbance Stokes Shift 0-0 ε F λ
21 The ground state and excited state of molecules can have very different properties Example 1: β-naphthol OH pk a (S o ) = 10.8 pk a (S 1 ) = 3.5 Deprotonates upon excitation at ph 7 Example 2: Formaldehyde changes geometry and permanent dipole upon excitation H H C O C S 0, planar 20 0 S 1, non-planar O
22 Science (2000) 290, An example where the excited state has different properties than the ground state The case of the Blue-Fluorescent Antibodies raised against trans-stilbene
23 Antibodies binds to trans-stilbene: The protein inhibits thermal relaxation from the excited state Science (2000) 290,
24 Ground State of stilbene: 45 kcal/mol energy barrier to twist from the trans to the cis conformation high energy transition state has a 90 o twist Science (2000) 290,
25 Excited state isomerizes very easily low energy barrier to 90 o twist (electron is in antibonding π * orbital) But the 90 o twisted state is the main pathway for thermal relaxation, so most of the energy is lost as heat after absorption from either cis or trans stilbene only 2% of molecules fluoresce trans cis Science (2000) 290,
26 Antibody binding to trans-stilbene: Locks the stilbene in the trans-configuration 70% of excited molecules that absorb a photon fluoresce (only 2% for stilbene in solution) trans Science (2000) 290,
27 Stokes shift Difference in wavelengths of the peaks of the excitation and emission spectra Due to solvent relaxation around the alteration in the electronic redistribution in the excited state. Stokes Shift 0-0 ε F λ The Stokes shift is a measurement of the environment around the fluorophore
28 Solvent relaxation and the Stokes Shift S 1 ε sec sec F 10-8 sec S 1 S 0 S 0 Absorption ~ sec Solvent relaxation ~ sec Fluorescence ~ 10-8 sec Permanent Dipoles of solvent re-orient to adjust to altered dipole of fluorophore upon excitation Requires: dipole-dipole interaction (1) solvent polarity (dielectric, ε) Stokes Shift 0-0 ε F (2) mobility of solvent (reorientation of solvent dipoles) λ
29 PRODAN µ δ δ + µ* Dipole moment of excited state
30 Direction of the permanent dipole is the same, but magnitude is greater in the excited state PRODAN In cyclohexane ε = 2 PRODAN in cyclohexane (1), chlorobenzene (2), dimethylformamide (3), ethanol (4) and water (5).
31 Stokes shift: example of tryptophan fluorescence tryptophan In non-polar solvents In polar solvents λ em ~ 325nm λ em ~ 360nm (1) Protein denaturation tryptophan emission often is red-shifted (2) buried vs exposed tryptophans in native proteins can be distinguished ~ 325nm ~ 360nm
32 Fluorescence from the 7 Tryptophan Residues of Human Carbonic Anhydrase II % total fluorescence W97 : 52% W245 : 38% 5 of the 7 tryptophans have little fluorescence due to enhanced thermal relaxation (quenching) from interactions with other amino acids such as histidines ET: energy transfer from one tryptophan to another q: quenching - non-radiative relaxation to ground state Biochem (1995) 34,
33 Human Carbonic Anhydrase II Trp245 Trp97 Biochem (1994) 33, )
34 W97 and W245 Fluorescence W97 : buried : blue shifted spectrum W245 : exposed to surface : red shifted spectrum Biochem (1995) 34,
XV 74. Flouorescence-Polarization-Circular-Dichroism- Jablonski diagram Where does the energy go?
 XV 74 Flouorescence-Polarization-Circular-Dichroism- Jablonski diagram Where does the energy go? 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet S = 0 could be any of them
XV 74 Flouorescence-Polarization-Circular-Dichroism- Jablonski diagram Where does the energy go? 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet S = 0 could be any of them
Fluorescence (Notes 16)
 Fluorescence - 2014 (Notes 16) XV 74 Jablonski diagram Where does the energy go? Can be viewed like multistep kinetic pathway 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet
Fluorescence - 2014 (Notes 16) XV 74 Jablonski diagram Where does the energy go? Can be viewed like multistep kinetic pathway 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet
What the Einstein Relations Tell Us
 What the Einstein Relations Tell Us 1. The rate of spontaneous emission A21 is proportional to υ 3. At higher frequencies A21 >> B(υ) and all emission is spontaneous. A 21 = 8π hν3 c 3 B(ν) 2. Although
What the Einstein Relations Tell Us 1. The rate of spontaneous emission A21 is proportional to υ 3. At higher frequencies A21 >> B(υ) and all emission is spontaneous. A 21 = 8π hν3 c 3 B(ν) 2. Although
Fluorescence 2009 update
 XV 74 Fluorescence 2009 update Jablonski diagram Where does the energy go? Can be viewed like multistep kinetic pathway 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet
XV 74 Fluorescence 2009 update Jablonski diagram Where does the energy go? Can be viewed like multistep kinetic pathway 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet
Singlet. Fluorescence Spectroscopy * LUMO
 Fluorescence Spectroscopy Light can be absorbed and re-emitted by matter luminescence (photo-luminescence). There are two types of luminescence, in this discussion: fluorescence and phosphorescence. A
Fluorescence Spectroscopy Light can be absorbed and re-emitted by matter luminescence (photo-luminescence). There are two types of luminescence, in this discussion: fluorescence and phosphorescence. A
Chapter 17: Fundamentals of Spectrophotometry
 Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
Chemistry 2. Molecular Photophysics
 Chemistry 2 Lecture 12 Molecular Photophysics Assumed knowledge Electronic states are labelled using their spin multiplicity with singlets having all electron spins paired and triplets having two unpaired
Chemistry 2 Lecture 12 Molecular Photophysics Assumed knowledge Electronic states are labelled using their spin multiplicity with singlets having all electron spins paired and triplets having two unpaired
Fluorescence Polarization Anisotropy FPA
 Fluorescence Polarization Anisotropy FPA Optics study of light Spectroscopy = light interacts the study of the interaction between matter & electro-magnetic radiation matter Spectroscopy Atomic Spectroscopy
Fluorescence Polarization Anisotropy FPA Optics study of light Spectroscopy = light interacts the study of the interaction between matter & electro-magnetic radiation matter Spectroscopy Atomic Spectroscopy
Chapter 17: Fundamentals of Spectrophotometry
 Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis)
 Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis) 2 2. Circular dichroism (optical activity) CD / ORD 3 3. Fluorescence spectroscopy and energy transfer Electromagnetic Spectrum Electronic Molecular
Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis) 2 2. Circular dichroism (optical activity) CD / ORD 3 3. Fluorescence spectroscopy and energy transfer Electromagnetic Spectrum Electronic Molecular
Lecture 5. Anisotropy decay/data analysis. Enrico Gratton
 Lecture 5. Anisotropy decay/data analysis Enrico Gratton Anisotropy decay Energy-transfer distance distributions Time resolved spectra Excited-state reactions Basic physics concept in polarization The
Lecture 5. Anisotropy decay/data analysis Enrico Gratton Anisotropy decay Energy-transfer distance distributions Time resolved spectra Excited-state reactions Basic physics concept in polarization The
single-molecule fluorescence resonance energy transfer
 single-molecule fluorescence resonance energy transfer (2) determing the Förster radius: quantum yield, donor lifetime, spectral overlap, anisotropy michael börsch 26/05/2004 1 fluorescence (1) absorbance
single-molecule fluorescence resonance energy transfer (2) determing the Förster radius: quantum yield, donor lifetime, spectral overlap, anisotropy michael börsch 26/05/2004 1 fluorescence (1) absorbance
Molecular Luminescence. Absorption Instrumentation. UV absorption spectrum. lg ε. A = εbc. monochromator. light source. Rotating mirror (beam chopper)
 Molecular Luminescence Absorption Instrumentation light source I 0 sample I detector light source Rotating mirror (beam chopper) motor b sample I detector reference I 0 UV absorption spectrum lg ε A =
Molecular Luminescence Absorption Instrumentation light source I 0 sample I detector light source Rotating mirror (beam chopper) motor b sample I detector reference I 0 UV absorption spectrum lg ε A =
Fluorescence Spectroscopy
 Fluorescence Spectroscopy Frequency and time dependent emission Emission and Excitation fluorescence spectra Stokes Shift: influence of molecular vibrations and solvent Time resolved fluorescence measurements
Fluorescence Spectroscopy Frequency and time dependent emission Emission and Excitation fluorescence spectra Stokes Shift: influence of molecular vibrations and solvent Time resolved fluorescence measurements
PHOTOCHEMISTRY NOTES - 1 -
 - 1 - PHOTOCHEMISTRY NOTES 1 st Law (Grotthus-Draper Law) Only absorbed radiation produces chemical change. Exception inelastic scattering of X- or γ-rays (electronic Raman effect). 2 nd Law (Star-Einstein
- 1 - PHOTOCHEMISTRY NOTES 1 st Law (Grotthus-Draper Law) Only absorbed radiation produces chemical change. Exception inelastic scattering of X- or γ-rays (electronic Raman effect). 2 nd Law (Star-Einstein
CHAPTER 13 Molecular Spectroscopy 2: Electronic Transitions
 CHAPTER 13 Molecular Spectroscopy 2: Electronic Transitions I. General Features of Electronic spectroscopy. A. Visible and ultraviolet photons excite electronic state transitions. ε photon = 120 to 1200
CHAPTER 13 Molecular Spectroscopy 2: Electronic Transitions I. General Features of Electronic spectroscopy. A. Visible and ultraviolet photons excite electronic state transitions. ε photon = 120 to 1200
Assumed knowledge. Chemistry 2. Learning outcomes. Electronic spectroscopy of polyatomic molecules. Franck-Condon Principle (reprise)
 Chemistry 2 Lecture 11 Electronic spectroscopy of polyatomic molecules Assumed knowledge For bound excited states, transitions to the individual vibrational levels of the excited state are observed with
Chemistry 2 Lecture 11 Electronic spectroscopy of polyatomic molecules Assumed knowledge For bound excited states, transitions to the individual vibrational levels of the excited state are observed with
Excited State Processes
 Excited State Processes Photophysics Fluorescence (singlet state emission) Phosphorescence (triplet state emission) Internal conversion (transition to singlet gr. state) Intersystem crossing (transition
Excited State Processes Photophysics Fluorescence (singlet state emission) Phosphorescence (triplet state emission) Internal conversion (transition to singlet gr. state) Intersystem crossing (transition
Chap. 12 Photochemistry
 Chap. 12 Photochemistry Photochemical processes Jablonski diagram 2nd singlet excited state 3rd triplet excited state 1st singlet excited state 2nd triplet excited state 1st triplet excited state Ground
Chap. 12 Photochemistry Photochemical processes Jablonski diagram 2nd singlet excited state 3rd triplet excited state 1st singlet excited state 2nd triplet excited state 1st triplet excited state Ground
What dictates the rate of radiative or nonradiative excited state decay?
 What dictates the rate of radiative or nonradiative excited state decay? Transitions are faster when there is minimum quantum mechanical reorganization of wavefunctions. This reorganization energy includes
What dictates the rate of radiative or nonradiative excited state decay? Transitions are faster when there is minimum quantum mechanical reorganization of wavefunctions. This reorganization energy includes
LABORATORY OF ELEMENTARY BIOPHYSICS
 LABORATORY OF ELEMENTARY BIOPHYSICS Experimental exercises for III year of the First cycle studies Field: Applications of physics in biology and medicine Specialization: Molecular Biophysics Fluorescence
LABORATORY OF ELEMENTARY BIOPHYSICS Experimental exercises for III year of the First cycle studies Field: Applications of physics in biology and medicine Specialization: Molecular Biophysics Fluorescence
Luminescence. Photoluminescence (PL) is luminescence that results from optically exciting a sample.
 Luminescence Topics Radiative transitions between electronic states Absorption and Light emission (spontaneous, stimulated) Excitons (singlets and triplets) Franck-Condon shift(stokes shift) and vibrational
Luminescence Topics Radiative transitions between electronic states Absorption and Light emission (spontaneous, stimulated) Excitons (singlets and triplets) Franck-Condon shift(stokes shift) and vibrational
Generation of light Light sources
 Generation of light Light sources Black-body radiation Luminescence Luminescence Laser Repetition Types of energy states in atoms and molecules are independent (not coupled) Energy states are non-continuous,
Generation of light Light sources Black-body radiation Luminescence Luminescence Laser Repetition Types of energy states in atoms and molecules are independent (not coupled) Energy states are non-continuous,
Mie vs Rayleigh. Sun
 Mie vs Rayleigh Sun Chemists Probe Various Energy Levels of Molecules With Appropiate Energy Radiation It is convenient (and accurate enough for our purposes) to treat a molecule or system of molecules
Mie vs Rayleigh Sun Chemists Probe Various Energy Levels of Molecules With Appropiate Energy Radiation It is convenient (and accurate enough for our purposes) to treat a molecule or system of molecules
CHEM Outline (Part 15) - Luminescence 2013
 CHEM 524 -- Outline (Part 15) - Luminescence 2013 XI. Molecular Luminescence Spectra (Chapter 15) Kinetic process, competing pathways fluorescence, phosphorescence, non-radiative decay Jablonski diagram
CHEM 524 -- Outline (Part 15) - Luminescence 2013 XI. Molecular Luminescence Spectra (Chapter 15) Kinetic process, competing pathways fluorescence, phosphorescence, non-radiative decay Jablonski diagram
To be covered (and why) Spectroscopy of Proteins. UV-Vis Absorption. UV-Vis Absorption. Spectra
 To be covered (and why) Spectroscopy of Proteins General considerations UV-Vis Absorption quantitation Fluorescence hydrophobicity Foldedness FT-Infrared Foldedness ircular Dichroism Foldedness NMR (a
To be covered (and why) Spectroscopy of Proteins General considerations UV-Vis Absorption quantitation Fluorescence hydrophobicity Foldedness FT-Infrared Foldedness ircular Dichroism Foldedness NMR (a
Linear and nonlinear spectroscopy
 Linear and nonlinear spectroscopy We ve seen that we can determine molecular frequencies and dephasing rates (for electronic, vibrational, or spin degrees of freedom) from frequency-domain or timedomain
Linear and nonlinear spectroscopy We ve seen that we can determine molecular frequencies and dephasing rates (for electronic, vibrational, or spin degrees of freedom) from frequency-domain or timedomain
Chemistry Instrumental Analysis Lecture 3. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 3 Quantum Transitions The energy of a photon can also be transferred to an elementary particle by adsorption if the energy of the photon exactly matches the
Chemistry 4631 Instrumental Analysis Lecture 3 Quantum Transitions The energy of a photon can also be transferred to an elementary particle by adsorption if the energy of the photon exactly matches the
Department of Chemistry Physical Chemistry Göteborg University
 Department of Chemistry Physical Chemistry Göteborg University &RQVWUXFWLRQRIDSXOVHGG\HODVHU 3OHDVHREVHUYHWKDWWKHVDIHW\SUHFDXWLRQVRQSDJHPXVW EHIROORZHGRWKHUZLVHWKHUHLVDULVNRIH\HGDPDJH Renée Andersson -97,
Department of Chemistry Physical Chemistry Göteborg University &RQVWUXFWLRQRIDSXOVHGG\HODVHU 3OHDVHREVHUYHWKDWWKHVDIHW\SUHFDXWLRQVRQSDJHPXVW EHIROORZHGRWKHUZLVHWKHUHLVDULVNRIH\HGDPDJH Renée Andersson -97,
Modern Optical Spectroscopy
 Modern Optical Spectroscopy With Exercises and Examples from Biophysics and Biochemistry von William W Parson 1. Auflage Springer-Verlag Berlin Heidelberg 2006 Verlag C.H. Beck im Internet: www.beck.de
Modern Optical Spectroscopy With Exercises and Examples from Biophysics and Biochemistry von William W Parson 1. Auflage Springer-Verlag Berlin Heidelberg 2006 Verlag C.H. Beck im Internet: www.beck.de
Principles of Physical Biochemistry
 Principles of Physical Biochemistry Kensal E. van Hold e W. Curtis Johnso n P. Shing Ho Preface x i PART 1 MACROMOLECULAR STRUCTURE AND DYNAMICS 1 1 Biological Macromolecules 2 1.1 General Principles
Principles of Physical Biochemistry Kensal E. van Hold e W. Curtis Johnso n P. Shing Ho Preface x i PART 1 MACROMOLECULAR STRUCTURE AND DYNAMICS 1 1 Biological Macromolecules 2 1.1 General Principles
Chem 344 Final Exam Tuesday, Dec. 11, 2007, 3-?? PM
 Chem 344 Final Exam Tuesday, Dec. 11, 2007, 3-?? PM Closed book exam, only pencils and calculators permitted. You may bring and use one 8 1/2 x 11" paper with anything on it. No Computers. Put all of your
Chem 344 Final Exam Tuesday, Dec. 11, 2007, 3-?? PM Closed book exam, only pencils and calculators permitted. You may bring and use one 8 1/2 x 11" paper with anything on it. No Computers. Put all of your
Chapter 6 Photoluminescence Spectroscopy
 Chapter 6 Photoluminescence Spectroscopy Course Code: SSCP 4473 Course Name: Spectroscopy & Materials Analysis Sib Krishna Ghoshal (PhD) Advanced Optical Materials Research Group Physics Department, Faculty
Chapter 6 Photoluminescence Spectroscopy Course Code: SSCP 4473 Course Name: Spectroscopy & Materials Analysis Sib Krishna Ghoshal (PhD) Advanced Optical Materials Research Group Physics Department, Faculty
Chemistry 304B, Spring 1999 Lecture 5 1. UV Spectroscopy:
 Chemistry 304B, Spring 1999 Lecture 5 1 Ultraviolet spectroscopy; UV Spectroscopy: Infrared spectroscopy; Nuclear magnetic resonance spectroscopy General basis of spectroscopy: Shine light at a collection
Chemistry 304B, Spring 1999 Lecture 5 1 Ultraviolet spectroscopy; UV Spectroscopy: Infrared spectroscopy; Nuclear magnetic resonance spectroscopy General basis of spectroscopy: Shine light at a collection
Chapter 15 Molecular Luminescence Spectrometry
 Chapter 15 Molecular Luminescence Spectrometry Two types of Luminescence methods are: 1) Photoluminescence, Light is directed onto a sample, where it is absorbed and imparts excess energy into the material
Chapter 15 Molecular Luminescence Spectrometry Two types of Luminescence methods are: 1) Photoluminescence, Light is directed onto a sample, where it is absorbed and imparts excess energy into the material
two slits and 5 slits
 Electronic Spectroscopy 2015January19 1 1. UV-vis spectrometer 1.1. Grating spectrometer 1.2. Single slit: 1.2.1. I diffracted intensity at relative to un-diffracted beam 1.2.2. I - intensity of light
Electronic Spectroscopy 2015January19 1 1. UV-vis spectrometer 1.1. Grating spectrometer 1.2. Single slit: 1.2.1. I diffracted intensity at relative to un-diffracted beam 1.2.2. I - intensity of light
Chem 442 Review of Spectroscopy
 Chem 44 Review of Spectroscopy General spectroscopy Wavelength (nm), frequency (s -1 ), wavenumber (cm -1 ) Frequency (s -1 ): n= c l Wavenumbers (cm -1 ): n =1 l Chart of photon energies and spectroscopies
Chem 44 Review of Spectroscopy General spectroscopy Wavelength (nm), frequency (s -1 ), wavenumber (cm -1 ) Frequency (s -1 ): n= c l Wavenumbers (cm -1 ): n =1 l Chart of photon energies and spectroscopies
Lecture 3: Light absorbance
 Lecture 3: Light absorbance Perturbation Response 1 Light in Chemistry Light Response 0-3 Absorbance spectrum of benzene 2 Absorption Visible Light in Chemistry S 2 S 1 Fluorescence http://www.microscopyu.com
Lecture 3: Light absorbance Perturbation Response 1 Light in Chemistry Light Response 0-3 Absorbance spectrum of benzene 2 Absorption Visible Light in Chemistry S 2 S 1 Fluorescence http://www.microscopyu.com
1. Transition dipole moment
 1. Transition dipole moment You have measured absorption spectra of aqueous (n=1.33) solutions of two different chromophores (A and B). The concentrations of the solutions were the same. The absorption
1. Transition dipole moment You have measured absorption spectra of aqueous (n=1.33) solutions of two different chromophores (A and B). The concentrations of the solutions were the same. The absorption
Optical spectroscopies. Rita P.Y. Chen. Electromagnetic Spectrum
 Optical spectroscopies Rita P.Y. Chen Electromagnetic Spectrum The longer the wavelength the lower the energy!!!! h = 6.63 x 10-34 J s 1 ev = 1.6 x 10-19 J, c= 3 x 10 8 m Transmittance, T = I f / I
Optical spectroscopies Rita P.Y. Chen Electromagnetic Spectrum The longer the wavelength the lower the energy!!!! h = 6.63 x 10-34 J s 1 ev = 1.6 x 10-19 J, c= 3 x 10 8 m Transmittance, T = I f / I
Advanced Organic Chemistry Chm 512/412 Spring Handout I Photochemistry Part 1. Photophysical Processes Quenching Alkene cis-trans Isomerization
 Advanced rganic Chemistry Chm 512/412 Spring 2010 Handout I Photochemistry Part 1 Photophysical Processes Quenching Alkene cis-trans Isomerization Importance of Photochemistry/Photophysics rganic Synthesis
Advanced rganic Chemistry Chm 512/412 Spring 2010 Handout I Photochemistry Part 1 Photophysical Processes Quenching Alkene cis-trans Isomerization Importance of Photochemistry/Photophysics rganic Synthesis
Fluorescence polarisation, anisotropy FRAP
 Fluorescence polarisation, anisotropy FRAP Reminder: fluorescence spectra Definitions! a. Emission sp. b. Excitation sp. Stokes-shift The difference (measured in nm) between the peak of the excitation
Fluorescence polarisation, anisotropy FRAP Reminder: fluorescence spectra Definitions! a. Emission sp. b. Excitation sp. Stokes-shift The difference (measured in nm) between the peak of the excitation
Reflection = EM strikes a boundary between two media differing in η and bounces back
 Reflection = EM strikes a boundary between two media differing in η and bounces back Incident ray θ 1 θ 2 Reflected ray Medium 1 (air) η = 1.00 Medium 2 (glass) η = 1.50 Specular reflection = situation
Reflection = EM strikes a boundary between two media differing in η and bounces back Incident ray θ 1 θ 2 Reflected ray Medium 1 (air) η = 1.00 Medium 2 (glass) η = 1.50 Specular reflection = situation
Molecular spectroscopy
 Molecular spectroscopy Origin of spectral lines = absorption, emission and scattering of a photon when the energy of a molecule changes: rad( ) M M * rad( ' ) ' v' 0 0 absorption( ) emission ( ) scattering
Molecular spectroscopy Origin of spectral lines = absorption, emission and scattering of a photon when the energy of a molecule changes: rad( ) M M * rad( ' ) ' v' 0 0 absorption( ) emission ( ) scattering
Introduction to Fluorescence Spectroscopies I. Theory
 March 22, 2006; Revised January 26, 2011 for CHMY 374 Adapted for CHMY 362 November 13, 2011 Edited by Lauren Woods December 2, 2011 17mar14, P.Callis; 1feb17 P. Callis, 29jan18 P. Callis Introduction
March 22, 2006; Revised January 26, 2011 for CHMY 374 Adapted for CHMY 362 November 13, 2011 Edited by Lauren Woods December 2, 2011 17mar14, P.Callis; 1feb17 P. Callis, 29jan18 P. Callis Introduction
A very brief history of the study of light
 1. Sir Isaac Newton 1672: A very brief history of the study of light Showed that the component colors of the visible portion of white light can be separated through a prism, which acts to bend the light
1. Sir Isaac Newton 1672: A very brief history of the study of light Showed that the component colors of the visible portion of white light can be separated through a prism, which acts to bend the light
Fluorescence spectra of 1-Pyrenebutyric acid, concentration dependence and dynamic quenching phenomena
 Practicum Spectroscopy Fall 2010 Fluorescence spectra of 1-Pyrenebutyric acid, concentration dependence and dynamic quenching phenomena LUM Jorge Ferreiro, study degree Chemistry, 5 th semester, fjorge@student.ethz.ch
Practicum Spectroscopy Fall 2010 Fluorescence spectra of 1-Pyrenebutyric acid, concentration dependence and dynamic quenching phenomena LUM Jorge Ferreiro, study degree Chemistry, 5 th semester, fjorge@student.ethz.ch
Fluorescence Workshop UMN Physics June 8-10, 2006 Basic Spectroscopic Principles Joachim Mueller
 Fluorescence Workshop UMN Physics June 8-10, 2006 Basic Spectroscopic Principles Joachim Mueller Fluorescence, Light, Absorption, Jablonski Diagram, and Beer-Law First stab at a definition: What is fluorescence?
Fluorescence Workshop UMN Physics June 8-10, 2006 Basic Spectroscopic Principles Joachim Mueller Fluorescence, Light, Absorption, Jablonski Diagram, and Beer-Law First stab at a definition: What is fluorescence?
NPTEL/IITM. Molecular Spectroscopy Lectures 1 & 2. Prof.K. Mangala Sunder Page 1 of 15. Topics. Part I : Introductory concepts Topics
 Molecular Spectroscopy Lectures 1 & 2 Part I : Introductory concepts Topics Why spectroscopy? Introduction to electromagnetic radiation Interaction of radiation with matter What are spectra? Beer-Lambert
Molecular Spectroscopy Lectures 1 & 2 Part I : Introductory concepts Topics Why spectroscopy? Introduction to electromagnetic radiation Interaction of radiation with matter What are spectra? Beer-Lambert
Discussion Session prior to the Second Examination: Sunday evening April 13 6 to 8 pm. 161 Noyes Laboratory
 Discussion Session prior to the Second Examination: Sunday evening April 13 6 to 8 pm 161 Noyes Laboratory Determination of the Stokes Radius by measuring the Rotational Diffusion Coefficient: D rot D
Discussion Session prior to the Second Examination: Sunday evening April 13 6 to 8 pm 161 Noyes Laboratory Determination of the Stokes Radius by measuring the Rotational Diffusion Coefficient: D rot D
Fluorescence Excitation and Emission Fundamentals
 Fluorescence Excitation and Emission Fundamentals Fluorescence is a member of the ubiquitous luminescence family of processes in which susceptible molecules emit light from electronically excited states
Fluorescence Excitation and Emission Fundamentals Fluorescence is a member of the ubiquitous luminescence family of processes in which susceptible molecules emit light from electronically excited states
UV-vis (Electronic) Spectra Ch.13 Atkins, Ch.19 Engel
 XV 74 UV-vis (Electronic) Spectra-2014 -Ch.13 Atkins, Ch.19 Engel Most broadly used analytical tech / especially bio-applic. inexpensive optics / solvent & cell usually not problem intense transitions
XV 74 UV-vis (Electronic) Spectra-2014 -Ch.13 Atkins, Ch.19 Engel Most broadly used analytical tech / especially bio-applic. inexpensive optics / solvent & cell usually not problem intense transitions
The Excited State of β-naphtol
 The Excited State of β-naphtol laboratory experiment in chemical physics The Excited State of β-naphtol ntroduction n this laboratory we will use the absorption and fluorescence spectroscopy to study the
The Excited State of β-naphtol laboratory experiment in chemical physics The Excited State of β-naphtol ntroduction n this laboratory we will use the absorption and fluorescence spectroscopy to study the
Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy
 Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy Section I Q1. Answer (i) (b) (ii) (d) (iii) (c) (iv) (c) (v) (a) (vi) (b) (vii) (b) (viii) (a) (ix)
Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy Section I Q1. Answer (i) (b) (ii) (d) (iii) (c) (iv) (c) (v) (a) (vi) (b) (vii) (b) (viii) (a) (ix)
Chapter 11. Basics in spin-orbit couplings
 1- The Jablonski diagram (or the state diagram of diamagnetic molecules) 2- Various natures of excited states and basics in molecular orbitals 3- Vibronic coupling and the Franck-Condon term 4- Excited
1- The Jablonski diagram (or the state diagram of diamagnetic molecules) 2- Various natures of excited states and basics in molecular orbitals 3- Vibronic coupling and the Franck-Condon term 4- Excited
Contents. xiii. Preface v
 Contents Preface Chapter 1 Biological Macromolecules 1.1 General PrincipIes 1.1.1 Macrornolecules 1.2 1.1.2 Configuration and Conformation Molecular lnteractions in Macromolecular Structures 1.2.1 Weak
Contents Preface Chapter 1 Biological Macromolecules 1.1 General PrincipIes 1.1.1 Macrornolecules 1.2 1.1.2 Configuration and Conformation Molecular lnteractions in Macromolecular Structures 1.2.1 Weak
Presenter: She Zhang
 Presenter: She Zhang Introduction Dr. David Baker Introduction Why design proteins de novo? It is not clear how non-covalent interactions favor one specific native structure over many other non-native
Presenter: She Zhang Introduction Dr. David Baker Introduction Why design proteins de novo? It is not clear how non-covalent interactions favor one specific native structure over many other non-native
Fluorescence Workshop UMN Physics June 8-10, 2006 Quantum Yield and Polarization (1) Joachim Mueller
 Fluorescence Workshop UMN Physics June 8-10, 2006 Quantum Yield and Polarization (1) Joachim Mueller Quantum yield, polarized light, dipole moment, photoselection, dipole radiation, polarization and anisotropy
Fluorescence Workshop UMN Physics June 8-10, 2006 Quantum Yield and Polarization (1) Joachim Mueller Quantum yield, polarized light, dipole moment, photoselection, dipole radiation, polarization and anisotropy
Chem 325 NMR Intro. The Electromagnetic Spectrum. Physical properties, chemical properties, formulas Shedding real light on molecular structure:
 Physical properties, chemical properties, formulas Shedding real light on molecular structure: Wavelength Frequency ν Wavelength λ Frequency ν Velocity c = 2.998 10 8 m s -1 The Electromagnetic Spectrum
Physical properties, chemical properties, formulas Shedding real light on molecular structure: Wavelength Frequency ν Wavelength λ Frequency ν Velocity c = 2.998 10 8 m s -1 The Electromagnetic Spectrum
Quantum Chemistry. NC State University. Lecture 5. The electronic structure of molecules Absorption spectroscopy Fluorescence spectroscopy
 Quantum Chemistry Lecture 5 The electronic structure of molecules Absorption spectroscopy Fluorescence spectroscopy NC State University 3.5 Selective absorption and emission by atmospheric gases (source:
Quantum Chemistry Lecture 5 The electronic structure of molecules Absorption spectroscopy Fluorescence spectroscopy NC State University 3.5 Selective absorption and emission by atmospheric gases (source:
10. 6 Photochemistry. Out-class reading: Levine, pp photochemistry
 Out-class reading: Levine, pp. 800-804 photochemistry 6.1 Brief introduction of light 1) Photochemistry The branch of chemistry which deals with the study of chemical reaction initiated by light. 2) Energy
Out-class reading: Levine, pp. 800-804 photochemistry 6.1 Brief introduction of light 1) Photochemistry The branch of chemistry which deals with the study of chemical reaction initiated by light. 2) Energy
Circular Dichroism & Optical Rotatory Dispersion. Proteins (KCsa) Polysaccharides (agarose) DNA CHEM 305. Many biomolecules are α-helical!
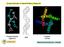 Circular Dichroism & Optical Rotatory Dispersion Polysaccharides (agarose) DNA Proteins (KCsa) Many biomolecules are α-helical! How can we measure the amount and changes in amount of helical structure
Circular Dichroism & Optical Rotatory Dispersion Polysaccharides (agarose) DNA Proteins (KCsa) Many biomolecules are α-helical! How can we measure the amount and changes in amount of helical structure
Fluorescence Depolarization. Ben Berger and Ben Berger
 Fluorescence Depolarization Ben Berger and Ben Berger Small molecules rotate quickly Large molecules rotate slowly There is a time delay from the moment a molecule is excited to the time it fluoresces
Fluorescence Depolarization Ben Berger and Ben Berger Small molecules rotate quickly Large molecules rotate slowly There is a time delay from the moment a molecule is excited to the time it fluoresces
Rotational Brownian motion; Fluorescence correlation spectroscpy; Photobleaching and FRET. David A. Case Rutgers, Spring 2009
 Rotational Brownian motion; Fluorescence correlation spectroscpy; Photobleaching and FRET David A. Case Rutgers, Spring 2009 Techniques based on rotational motion What we studied last time probed translational
Rotational Brownian motion; Fluorescence correlation spectroscpy; Photobleaching and FRET David A. Case Rutgers, Spring 2009 Techniques based on rotational motion What we studied last time probed translational
Chapter 2 Energy Transfer Review
 Chapter 2 Energy Transfer Review In this chapter, we discuss the basic concepts of excitation energy transfer, making the distinction between radiative and nonradiative, and giving a brief overview on
Chapter 2 Energy Transfer Review In this chapter, we discuss the basic concepts of excitation energy transfer, making the distinction between radiative and nonradiative, and giving a brief overview on
BMB/Bi/Ch 173 Winter 2018
 BMB/Bi/Ch 173 Winter 2018 Homework Set 8.1 (100 Points) Assigned 2-27-18, due 3-6-18 by 10:30 a.m. TA: Rachael Kuintzle. Office hours: SFL 220, Friday 3/2 4:00-5:00pm and SFL 229, Monday 3/5 4:00-5:30pm.
BMB/Bi/Ch 173 Winter 2018 Homework Set 8.1 (100 Points) Assigned 2-27-18, due 3-6-18 by 10:30 a.m. TA: Rachael Kuintzle. Office hours: SFL 220, Friday 3/2 4:00-5:00pm and SFL 229, Monday 3/5 4:00-5:30pm.
Electronic Spectra of Complexes
 Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
Last Updated: April 22, 2012 at 7:49pm
 Page 1 Electronic Properties of d 6 π Coordination Compounds The metal-to-ligand charge transfer (MLCT) excited states of d 6 π coordination compounds have emerged as the most efficient for both solar
Page 1 Electronic Properties of d 6 π Coordination Compounds The metal-to-ligand charge transfer (MLCT) excited states of d 6 π coordination compounds have emerged as the most efficient for both solar
Wavelength λ Velocity v. Electric Field Strength Amplitude A. Time t or Distance x time for 1 λ to pass fixed point. # of λ passing per s ν= 1 p
 Introduction to Spectroscopy (Chapter 6) Electromagnetic radiation (wave) description: Wavelength λ Velocity v Electric Field Strength 0 Amplitude A Time t or Distance x Period p Frequency ν time for 1
Introduction to Spectroscopy (Chapter 6) Electromagnetic radiation (wave) description: Wavelength λ Velocity v Electric Field Strength 0 Amplitude A Time t or Distance x Period p Frequency ν time for 1
 UV-Visible, Fluorescence and CD Spectroscopy Of Proteins Sepideh hkhorasanizadeh h Department of Biochemistry and Molecular Genetics Jordan Hall 6042 http://en.wikipedia.org/wiki/uv-vis_spectroscopy http://en.wikipedia.org/wiki/fluorescence_spectroscopy
UV-Visible, Fluorescence and CD Spectroscopy Of Proteins Sepideh hkhorasanizadeh h Department of Biochemistry and Molecular Genetics Jordan Hall 6042 http://en.wikipedia.org/wiki/uv-vis_spectroscopy http://en.wikipedia.org/wiki/fluorescence_spectroscopy
Molecular Luminescence Spectroscopy
 Molecular Luminescence Spectroscopy In Molecular Luminescence Spectrometry ( MLS ), molecules of the analyte in solution are excited to give a species whose emission spectrum provides information for qualitative
Molecular Luminescence Spectroscopy In Molecular Luminescence Spectrometry ( MLS ), molecules of the analyte in solution are excited to give a species whose emission spectrum provides information for qualitative
Problem 1. Anthracene and a chiral derivative of anthracene
 Molecular Photophysics 330 Physical rganic Chemistry 6C50 Thursday November 5 004, 4.00-7.00 h This exam consists of four problems that have an equal weight in the final score Most problems are composed
Molecular Photophysics 330 Physical rganic Chemistry 6C50 Thursday November 5 004, 4.00-7.00 h This exam consists of four problems that have an equal weight in the final score Most problems are composed
Skoog Chapter 6 Introduction to Spectrometric Methods
 Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
General Considerations 1
 General Considerations 1 Absorption or emission of electromagnetic radiation results in a permanent energy transfer from the emitting object or to the absorbing medium. This permanent energy transfer can
General Considerations 1 Absorption or emission of electromagnetic radiation results in a permanent energy transfer from the emitting object or to the absorbing medium. This permanent energy transfer can
wbt Λ = 0, 1, 2, 3, Eq. (7.63)
 7.2.2 Classification of Electronic States For all diatomic molecules the coupling approximation which best describes electronic states is analogous to the Russell- Saunders approximation in atoms The orbital
7.2.2 Classification of Electronic States For all diatomic molecules the coupling approximation which best describes electronic states is analogous to the Russell- Saunders approximation in atoms The orbital
Lab 11: Must what goes in be the same as what comes out? Spectroscopy & Fluorescence in Chlorophyll.
 Lab 11: Must what goes in be the same as what comes out? Spectroscopy & Fluorescence in Chlorophyll. Introduction to Fluorescence: Fluorescence is one of the possible mechanisms for emission of light by
Lab 11: Must what goes in be the same as what comes out? Spectroscopy & Fluorescence in Chlorophyll. Introduction to Fluorescence: Fluorescence is one of the possible mechanisms for emission of light by
Introduction... Theory Influence of Excitation Pulse Shape...
 1. Fluorescence Anisotropy: Theory and Applications Robert F. Steiner 1.1. 1.2. 1.3. 1.4. 1.5. 1.6. Introduction... Theory... 1.2.1. Meaning of Anisotropy... 1.2.2. Influence of Excitation Pulse Shape...
1. Fluorescence Anisotropy: Theory and Applications Robert F. Steiner 1.1. 1.2. 1.3. 1.4. 1.5. 1.6. Introduction... Theory... 1.2.1. Meaning of Anisotropy... 1.2.2. Influence of Excitation Pulse Shape...
Electronic Excitation by UV/Vis Spectroscopy :
 SPECTROSCOPY Light interacting with matter as an analytical tool III Pharm.D Department of Pharmaceutical Analysis SRM College Of Pharmacy,Katankulathur Electronic Excitation by UV/Vis Spectroscopy : X-ray:
SPECTROSCOPY Light interacting with matter as an analytical tool III Pharm.D Department of Pharmaceutical Analysis SRM College Of Pharmacy,Katankulathur Electronic Excitation by UV/Vis Spectroscopy : X-ray:
PART VI : MOLECULAR LUMINESCENCE SPECTROSCOPY (Recommendations 1985)
 PART VI : MOLECULAR LUMINESCENCE SPECTROSCOPY (Recommendations 1985) 1. INTRODUCTION This document does not aim to be completely self-contained since many of the terms and units needed for describing Molecular
PART VI : MOLECULAR LUMINESCENCE SPECTROSCOPY (Recommendations 1985) 1. INTRODUCTION This document does not aim to be completely self-contained since many of the terms and units needed for describing Molecular
Aula 5 e 6 Transferência de Energia e Transferência de Elétron Caminhos de espécies fotoexcitadas
 Fotoquímica Aula 5 e 6 Transferência de Energia e Transferência de Elétron Prof. Amilcar Machulek Junior IQ/USP - CEPEMA Caminhos de espécies fotoexcitadas 1 Diagrama de Jablonski S 2 Relaxation (τ < 1ps)
Fotoquímica Aula 5 e 6 Transferência de Energia e Transferência de Elétron Prof. Amilcar Machulek Junior IQ/USP - CEPEMA Caminhos de espécies fotoexcitadas 1 Diagrama de Jablonski S 2 Relaxation (τ < 1ps)
Luminescence spectroscopy
 Febr. 203 Luminescence spectroscopy Biophysics 2 nd semester Józse Orbán University o Pécs, Department o Biophysics Deinitions, laws FUNDAMENTALS o SPECTROSCY review - Spectral types (absorbtion/emission
Febr. 203 Luminescence spectroscopy Biophysics 2 nd semester Józse Orbán University o Pécs, Department o Biophysics Deinitions, laws FUNDAMENTALS o SPECTROSCY review - Spectral types (absorbtion/emission
Chapter 3. Electromagnetic Theory, Photons. and Light. Lecture 7
 Lecture 7 Chapter 3 Electromagnetic Theory, Photons. and Light Sources of light Emission of light by atoms The electromagnetic spectrum see supplementary material posted on the course website Electric
Lecture 7 Chapter 3 Electromagnetic Theory, Photons. and Light Sources of light Emission of light by atoms The electromagnetic spectrum see supplementary material posted on the course website Electric
Protein Dynamics. The space-filling structures of myoglobin and hemoglobin show that there are no pathways for O 2 to reach the heme iron.
 Protein Dynamics The space-filling structures of myoglobin and hemoglobin show that there are no pathways for O 2 to reach the heme iron. Below is myoglobin hydrated with 350 water molecules. Only a small
Protein Dynamics The space-filling structures of myoglobin and hemoglobin show that there are no pathways for O 2 to reach the heme iron. Below is myoglobin hydrated with 350 water molecules. Only a small
Isothermal experiments characterize time-dependent aggregation and unfolding
 1 Energy Isothermal experiments characterize time-dependent aggregation and unfolding Technical ote Introduction Kinetic measurements have, for decades, given protein scientists insight into the mechanisms
1 Energy Isothermal experiments characterize time-dependent aggregation and unfolding Technical ote Introduction Kinetic measurements have, for decades, given protein scientists insight into the mechanisms
1 Basic Optical Principles
 13 1 Basic Optical Principles 1.1 Introduction To understand important optical methods used to investigate biomolecules, such as fluorescence polarization anisotropy, Förster resonance energy transfer,
13 1 Basic Optical Principles 1.1 Introduction To understand important optical methods used to investigate biomolecules, such as fluorescence polarization anisotropy, Förster resonance energy transfer,
( ) x10 8 m. The energy in a mole of 400 nm photons is calculated by: ' & sec( ) ( & % ) 6.022x10 23 photons' E = h! = hc & 6.
 Introduction to Spectroscopy Spectroscopic techniques are widely used to detect molecules, to measure the concentration of a species in solution, and to determine molecular structure. For proteins, most
Introduction to Spectroscopy Spectroscopic techniques are widely used to detect molecules, to measure the concentration of a species in solution, and to determine molecular structure. For proteins, most
I 2 Vapor Absorption Experiment and Determination of Bond Dissociation Energy.
 I 2 Vapor Absorption Experiment and Determination of Bond Dissociation Energy. What determines the UV-Vis (i.e., electronic transitions) band appearance? Usually described by HOMO LUMO electron jump LUMO
I 2 Vapor Absorption Experiment and Determination of Bond Dissociation Energy. What determines the UV-Vis (i.e., electronic transitions) band appearance? Usually described by HOMO LUMO electron jump LUMO
Today: general condition for threshold operation physics of atomic, vibrational, rotational gain media intro to the Lorentz model
 Today: general condition for threshold operation physics of atomic, vibrational, rotational gain media intro to the Lorentz model Laser operation Simplified energy conversion processes in a laser medium:
Today: general condition for threshold operation physics of atomic, vibrational, rotational gain media intro to the Lorentz model Laser operation Simplified energy conversion processes in a laser medium:
Optics and Spectroscopy
 Introduction to Optics and Spectroscopy beyond the diffraction limit Chi Chen 陳祺 Research Center for Applied Science, Academia Sinica 2015Apr09 1 Light and Optics 2 Light as Wave Application 3 Electromagnetic
Introduction to Optics and Spectroscopy beyond the diffraction limit Chi Chen 陳祺 Research Center for Applied Science, Academia Sinica 2015Apr09 1 Light and Optics 2 Light as Wave Application 3 Electromagnetic
24 Introduction to Spectrochemical Methods
 24 Introduction to Spectrochemical Methods Spectroscopic method: based on measurement of the electromagnetic radiation produced or absorbed by analytes. electromagnetic radiation: include γ-ray, X-ray,
24 Introduction to Spectrochemical Methods Spectroscopic method: based on measurement of the electromagnetic radiation produced or absorbed by analytes. electromagnetic radiation: include γ-ray, X-ray,
118/218 FINAL TAKE HOME due Monday, March 19, Chemistry room 4129
 118/218 FINAL TAKE HOME due Monday, March 19, Chemistry room 4129 [1] In the stilbene photoisomerization paper on the website handouts page refer to Figure 3. (a) How does the wavelength and timing of
118/218 FINAL TAKE HOME due Monday, March 19, Chemistry room 4129 [1] In the stilbene photoisomerization paper on the website handouts page refer to Figure 3. (a) How does the wavelength and timing of
Chemistry Instrumental Analysis Lecture 11. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 11 Molar Absorptivities Range 0 to 10 5 Magnitude of e depends on capture cross section of the species and probability of the energy-absorbing transition. e
Chemistry 4631 Instrumental Analysis Lecture 11 Molar Absorptivities Range 0 to 10 5 Magnitude of e depends on capture cross section of the species and probability of the energy-absorbing transition. e
Excited States in Organic Light-Emitting Diodes
 Excited States in Organic Light-Emitting Diodes The metal-to-ligand charge transfer (MLCT) excited states of d 6 π coordination compounds have emerged as the most efficient for solar harvesting and sensitization
Excited States in Organic Light-Emitting Diodes The metal-to-ligand charge transfer (MLCT) excited states of d 6 π coordination compounds have emerged as the most efficient for solar harvesting and sensitization
Chem Homework Set Answers
 Chem 310 th 4 Homework Set Answers 1. Cyclohexanone has a strong infrared absorption peak at a wavelength of 5.86 µm. (a) Convert the wavelength to wavenumber.!6!1 8* = 1/8 = (1/5.86 µm)(1 µm/10 m)(1 m/100
Chem 310 th 4 Homework Set Answers 1. Cyclohexanone has a strong infrared absorption peak at a wavelength of 5.86 µm. (a) Convert the wavelength to wavenumber.!6!1 8* = 1/8 = (1/5.86 µm)(1 µm/10 m)(1 m/100
1. Photoreduction of Benzophenone in 2-Propanol
 1. Photoreduction of Benzophenone in 2-Propanol Topic: photochemistry, photophysics, kinetics, physical-organic chemistry Level: undergraduate physical chemistry Time: 2 x 2 hours (separated by ~24 hours)
1. Photoreduction of Benzophenone in 2-Propanol Topic: photochemistry, photophysics, kinetics, physical-organic chemistry Level: undergraduate physical chemistry Time: 2 x 2 hours (separated by ~24 hours)
Rice/TCU REU on Computational Neuroscience. Fundamentals of Molecular Imaging
 Rice/TCU REU on Computational Neuroscience Fundamentals of Molecular Imaging June 3, 2008 Neal Waxham 713-500-5621 m.n.waxham@uth.tmc.edu Objectives Brief discussion of optical resolution and lasers as
Rice/TCU REU on Computational Neuroscience Fundamentals of Molecular Imaging June 3, 2008 Neal Waxham 713-500-5621 m.n.waxham@uth.tmc.edu Objectives Brief discussion of optical resolution and lasers as
C101-E145 TALK LETTER. Vol. 17
 C101-E145 TALK LETTER Vol. 17 UV-VIS Spectroscopy and Fluorescence Spectroscopy (Part 1 of 2) ------- 02 Applications: Spectrofluorophotometers Used in a Variety of Fields ------- 06 Q&A: Is there a way
C101-E145 TALK LETTER Vol. 17 UV-VIS Spectroscopy and Fluorescence Spectroscopy (Part 1 of 2) ------- 02 Applications: Spectrofluorophotometers Used in a Variety of Fields ------- 06 Q&A: Is there a way
Introduction ENERGY. Heat Electricity Electromagnetic irradiation (light)
 Photochemistry Introduction ENERGY Heat Electricity Electromagnetic irradiation (light) Vision: Triggered by a photochemical reaction Is red in the dark? The answer must be NO - Since what we see as colour
Photochemistry Introduction ENERGY Heat Electricity Electromagnetic irradiation (light) Vision: Triggered by a photochemical reaction Is red in the dark? The answer must be NO - Since what we see as colour
I 2 Vapor Absorption Experiment and Determination of Bond Dissociation Energy.
 I 2 Vapor Absorption Experiment and Determination of Bond Dissociation Energy. What determines the UV-Vis (i.e., electronic transitions) band appearance? Usually described by HOMO LUMO electron jump LUMO
I 2 Vapor Absorption Experiment and Determination of Bond Dissociation Energy. What determines the UV-Vis (i.e., electronic transitions) band appearance? Usually described by HOMO LUMO electron jump LUMO
Introduction to Spectroscopic methods
 Introduction to Spectroscopic methods Spectroscopy: Study of interaction between light* and matter. Spectrometry: Implies a quantitative measurement of intensity. * More generally speaking electromagnetic
Introduction to Spectroscopic methods Spectroscopy: Study of interaction between light* and matter. Spectrometry: Implies a quantitative measurement of intensity. * More generally speaking electromagnetic
