ATMO 551a Moist Adiabat Fall Change in internal energy: ΔU
|
|
|
- Kory Davis
- 5 years ago
- Views:
Transcription
1 Enthalpy and the Moist Adiabat We have described the dry adiabat where an air parcel is lifted rapidly causing the air parcel to expand as the environmental pressure decreases and the air parcel does work on the environment but no heat is exchanged with the environment. As a result some of the air parcel s internal energy is converted to doing work on the environment. The result is the dry adiabatic temperature gradient dt dz = g (1) C p Now we discuss the effect of transferring the latent of water vapor to increase the temperature of the air. This includes discussion about various measures of energy or heat, and highlight what the concept of enthalpy is. Recall that when heat enters or is added to a parcel, this heat can go to the internal energy of the parcel, as well as to work the parcel does on the environment. Remember that energy added to the system goes into some combination of the bulk energy of the system, E B, (which is large scale mechanical energy: kinetic plus potential energy) plus the internal energy of the system, U. We write this as Δ( U + E B ) = ΔQ + ΔW (2) Heat added: ΔQ SYSTEM Work done by surroundings: ΔW Change in internal energy: ΔU Change in bulk energy: ΔE B where ΔW is defined as positive when the environment does work on the parcel (not when the parcel does work on the environment). Remember also that ΔW =-p ΔV. Now we ignore changes in bulk energy and express the properties of the parcel in specific units (i.e. per unit mass), yielding dq = du dw = du + p d(1/ρ) Note that q here is the heat per unit mass, not the specific humidity. Remember that when heat is added to the system (=air parcel) when volume (i.e. density) is kept constant, the only place for the energy to go is into the kinetic energy of the molecules: dq v = du = c v dt. If we consider heating up the gas from some absolute zero temperature, we can define the total specific internal energy as 1 Kursinski 10/18/10
2 u = c v T where we have assumed c v is independent of T and that no phase changes have occurred. We ll address the effects of phase changes shortly. Now, when heat is added to the air parcel when it is held at constant pressure (which is the typical situation in the atmosphere) we can rewrite d(1/ρ) = d(r*t/p m A ), and end up with dq p = c v dt + (R*/m a ) dt = c p dt Here, there is a work term, because as the gas heats up at constant pressure, it expands and does work on the environment. To keep track of this process, we define the enthalpy, h h = u + p/ρ = c p T Note that Enthalpy is not a measure of the energy contained in a parcel. It is a hybrid that contains both the internal energy and some of the energy that the air parcel transferred to the environment that the environment stores to make room for the parcel. In a hydrostatic atmosphere, this second form of energy is stored as the extra gravitational potential of the overriding air that has been lifted to make room for the expanded parcel. Now consider the most general form of dq again, where we take advantage of our expression for enthalpy dq = du dw = du + pd(1/ρ) = du + p d(r*t/pm a ) = dh ρ -1 dp In hydrostatic equilibrium, we have dq = dh + gdz = dh + dφ We call the term h + Φ the dry static energy, and keep track of it with the potential temperature θ = T + Φ/c p = T + Γ d z What goes into the dry static energy is the gravitational potential energy of the parcel, the internal energy of the parcel, and the gravitational potential of the air overlying the parcel caused by the parcel s presence (volume). We keep track of dry static energy because it is constant 2 Kursinski 10/18/10
3 during adiabatic processes, when dq = 0. Dry static energy goes to zero as a parcel is adiabatically dropped to the ground and then cooled at constant pressure to zero. Moist static energy This has all been review, but I m now using the notation and terms in W+H to be consistent. Now let s consider what happens when we throw phase changes into the mix. Water vapor releases heat when it condenses. This is called the latent heat of vaporization in W+H, although you will also see it called the latent heat of condensation. The amount of heat released is a weak function of temperature, with a value of about 2.5x10 6 J/kg at 0 C, and 2.25 x 10 6 J/kg at 100 C. The amount of heat released depends on the amount of mass of water vapor condensed or in units of specific heat, we have dq = -L v dm v dq = -L v dq v Note that there s a minus sign because vapor content goes down when condensation occurs. Relating this to the heating of our parcel, we have dq = dh + dφ = -L v dq v If there is no heat exchange besides this latent process, we can write 0 = dh + dφ + L v dq v This is called a moist adiabatic process the only heat exchange is the conversion of latent heat of vapor into sensible heat as the vapor condenses. Since no heat or mass actually crosses the boundary of our parcel, this is really considered an interconversion of internal enrgy. We can even consider the un-condensed vapor as a form of internal energy, not as a form of heat. In this case, we define the moist static energy of a parcel as, h + Φ + L v q v To keep track of moist static energy, we define the equivalent potential temperature θ e = θ + (L v /c p )q v θ e is always larger than θ, as it includes the internal potential energy of the vapor. It is also conserved when cloud water condenses or evaporates. You can think of equivalent potential temperature as the temperature a parcel would have if you first condensed all the vapor out, and then dropped it to surface pressure adiabatically. Exercise: What is the potential temperature outside right now? (T = 34.7; Td = 1.6; p = 926 mb) In the general adiabatic case for a rising air parcel, dz > 0, dt < 0 and dq < 0. We need an additional constraint on the problem and this is provided by the assertion that dq = dq s (T), which is the same thing as saying that RH will max out at 100%. (We will learn later that this is merely an approximation, but it is a good one for coarse purposes). For RH to equal 100% as 3 Kursinski 10/18/10
4 temperature is decreasing, there must be a corresponding decrease in saturation vapor pressure, which is achieved by condensation. The saturation specific humidity is the ratio of the saturation density to air density. q s = ρ s /ρ a The saturation density is obtained from the saturation vapor pressure using the ideal gas law. ρ s = n sv M w n sv = e s (T)/(R*T) Using the ideal gas law to relate air density to air pressure, we get ρ a = pm w /(R*T) These lead to the following simplification for q s q s = (M w /M A )(e s (T)/p) Now our goal is to find dq s, which is more easily manipulated through its log dq s = q s dlnq s = q s (dlne s dlnp) [Note that we have conveniently neglected the sensitivity of molecular weight of air to vapor pressure, which will contribute a very minor term.] The first term shows that saturation mixing ratio will increase if saturation vapor pressure increases. Note the second term that shows that saturation specific humidity will increase with a decrease in ambient pressure. This might seem counterintuitive, since we know that q doesn t change with changes in pressure. However, q s is a different beast than q. Whereas q keeps track of the mass of vapor in a given mass of air, q s tracks the mass of water vapor that would be in equilibrium with a flat surface of water per unit mass of air. As pressure goes up and down, saturation vapor pressure doesn t notice. It is only sensitive to temperature. However, the dry air mass is fluctuating with pressure, causing the constant saturation vapor pressure to be a relatively larger fraction of the air pressure as pressure goes down. To explore this further, consider a chamber that is partially filled with water and is held at temperature T. In equilibrium, the total pressure will be p d + e s (T), where p d is the pressure due to the dry air. Now we evacuate the chamber of half the air. At this very instant the pressure will be p d /2 + e s (T)/2, and q will be the same as before since there has been no condensation or evaporation (yet). But the water surface will still have a vapor pressure of e s (T). So over time, water will evaporate and q will increase to a new, higher equilibrium value, qs(t,p f ). Now we go back to the moist parcel that is ascending adiabatically. From Clausius-Clapyron, we have dlne s = L c M w /(R*T 2 ) dt And hydrostatic pressure for ideal gases can be written as: dlnp = -dz/h 4 Kursinski 10/18/10
5 Where H is the scale height of the atmosphere = R*T/(M A g). Thus our expression for dq s can be written in terms of dt and dz, which are the same independent variables used in our dry adiabatic equation. dq s = q s (L c M w /(R*T 2 ) dt + dz/h) Subbing all this into the energy conservation equation yields, 0 = c p dt + gdz + L c q s [L c M w /(R*T 2 ) dt + dz/h] Now we can solve for dt/dz = -Γ M, which is called the moist adiabatic lapse rate dt/dz = - (g + L c q s /H) / [c p + L c q s (L c M w /(R*T 2 ))] At this point, it is useful to note the g/c p is the dry adiabatic lapse rate, Γ D, and that H is a function of gravity. After some algebra, we get, Γ M = Γ D qsm A 1+ R * T qs M 1+ c R * T p W 2 When all is said and done, we see that there are two terms that make the moist adiabatic lapse rate different from the dry value. The numerator contains a term that came from the pressure dependence of saturation specific humidity (as discussed above). The denominator has a term that came from the temperature dependence of saturation specific humidity through the Clausius Clapyron relation, and this is the dominant term. While both terms are important to getting the right value, the CC term dominates. At 0 C and mean sea level pressure (MSL) the denominator is 1.68 and the numerator is 1.12, with that the moist lapse rate is (6.5 K/km), about 2/3 the value of the dry lapse rate. At 15 C, the numerator is and the denominator increases to 2.630, for a moist lapse rate of 4.9 half the value of the dry lapse rate. This addition of the 2 nd term in the denominator signifies that that both kinetic and latent energy sources are used to do work on the environment as it expands, thus slowing the cooling relative to the case when only kinetic energy is used. The modifier in the numerator accounts for the evaporation (and hence cooling) that happens independently of temperature as air depressurizes, due to the drop in vapor pressure at fixed specific humidity. Because it represents a cooling with height, it increases the magnitude of the lapse rate, but only slightly relative to the CC effect. 5 Kursinski 10/18/10
ATMO 551a Fall 08. Equivalent Potential Temperature
 Equivalent Potential emperature he equivalent potential temperature, θ e, is the potential temperature that would result if all of the water in the air parcel were condensed and rained out by raising the
Equivalent Potential emperature he equivalent potential temperature, θ e, is the potential temperature that would result if all of the water in the air parcel were condensed and rained out by raising the
1. Water Vapor in Air
 1. Water Vapor in Air Water appears in all three phases in the earth s atmosphere - solid, liquid and vapor - and it is one of the most important components, not only because it is essential to life, but
1. Water Vapor in Air Water appears in all three phases in the earth s atmosphere - solid, liquid and vapor - and it is one of the most important components, not only because it is essential to life, but
df dz = dp dt Essentially, this is just a statement of the first law in one of the forms we derived earlier (expressed here in W m 3 ) dq p dt dp
 A problem with using entropy as a variable is that it is not a particularly intuitive concept. The mechanics of using entropy for evaluating system evolution is well developed, but it sometimes feels a
A problem with using entropy as a variable is that it is not a particularly intuitive concept. The mechanics of using entropy for evaluating system evolution is well developed, but it sometimes feels a
Thermodynamics Review [?] Entropy & thermodynamic potentials Hydrostatic equilibrium & buoyancy Stability [dry & moist adiabatic]
![Thermodynamics Review [?] Entropy & thermodynamic potentials Hydrostatic equilibrium & buoyancy Stability [dry & moist adiabatic] Thermodynamics Review [?] Entropy & thermodynamic potentials Hydrostatic equilibrium & buoyancy Stability [dry & moist adiabatic]](/thumbs/81/83694823.jpg) Thermodynamics Review [?] Entropy & thermodynamic potentials Hydrostatic equilibrium & buoyancy Stability [dry & moist adiabatic] Entropy 1. (Thermodynamics) a thermodynamic quantity that changes in a
Thermodynamics Review [?] Entropy & thermodynamic potentials Hydrostatic equilibrium & buoyancy Stability [dry & moist adiabatic] Entropy 1. (Thermodynamics) a thermodynamic quantity that changes in a
ATMO/OPTI 656b Spring 09. Physical properties of the atmosphere
 The vertical structure of the atmosphere. Physical properties of the atmosphere To first order, the gas pressure at the bottom of an atmospheric column balances the downward force of gravity on the column.
The vertical structure of the atmosphere. Physical properties of the atmosphere To first order, the gas pressure at the bottom of an atmospheric column balances the downward force of gravity on the column.
Outline. Aim. Gas law. Pressure. Scale height Mixing Column density. Temperature Lapse rate Stability. Condensation Humidity.
 Institute of Applied Physics University of Bern Outline A planetary atmosphere consists of different gases hold to the planet by gravity The laws of thermodynamics hold structure as vertical coordinate
Institute of Applied Physics University of Bern Outline A planetary atmosphere consists of different gases hold to the planet by gravity The laws of thermodynamics hold structure as vertical coordinate
Atmospheric Thermodynamics
 Atmospheric Thermodynamics R. Wordsworth February 12, 2015 1 Objectives Derive hydrostatic equation Derive dry and moist adiabats Understand how the theory relates to observed properties of real atmospheres
Atmospheric Thermodynamics R. Wordsworth February 12, 2015 1 Objectives Derive hydrostatic equation Derive dry and moist adiabats Understand how the theory relates to observed properties of real atmospheres
Radiative equilibrium Some thermodynamics review Radiative-convective equilibrium. Goal: Develop a 1D description of the [tropical] atmosphere
![Radiative equilibrium Some thermodynamics review Radiative-convective equilibrium. Goal: Develop a 1D description of the [tropical] atmosphere Radiative equilibrium Some thermodynamics review Radiative-convective equilibrium. Goal: Develop a 1D description of the [tropical] atmosphere](/thumbs/72/66535634.jpg) Radiative equilibrium Some thermodynamics review Radiative-convective equilibrium Goal: Develop a 1D description of the [tropical] atmosphere Vertical temperature profile Total atmospheric mass: ~5.15x10
Radiative equilibrium Some thermodynamics review Radiative-convective equilibrium Goal: Develop a 1D description of the [tropical] atmosphere Vertical temperature profile Total atmospheric mass: ~5.15x10
Quasi-equilibrium transitions
 Quasi-equilibrium transitions We have defined a two important equilibrium conditions. he first is one in which there is no heating, or the system is adiabatic, and dh/ =0, where h is the total enthalpy
Quasi-equilibrium transitions We have defined a two important equilibrium conditions. he first is one in which there is no heating, or the system is adiabatic, and dh/ =0, where h is the total enthalpy
P sat = A exp [B( 1/ /T)] B= 5308K. A=6.11 mbar=vapor press. 0C.
![P sat = A exp [B( 1/ /T)] B= 5308K. A=6.11 mbar=vapor press. 0C. P sat = A exp [B( 1/ /T)] B= 5308K. A=6.11 mbar=vapor press. 0C.](/thumbs/79/79184286.jpg) Lecture 5. Water and water vapor in the atmosphere 14 Feb 2008 Review of buoyancy, with an unusual demonstration of Archimedes principle. Water is a polar molecule that forms hydrogen bonds. Consequently
Lecture 5. Water and water vapor in the atmosphere 14 Feb 2008 Review of buoyancy, with an unusual demonstration of Archimedes principle. Water is a polar molecule that forms hydrogen bonds. Consequently
2σ e s (r,t) = e s (T)exp( rr v ρ l T ) = exp( ) 2σ R v ρ l Tln(e/e s (T)) e s (f H2 O,r,T) = f H2 O
 Formulas/Constants, Physics/Oceanography 4510/5510 B Atmospheric Physics II N A = 6.02 10 23 molecules/mole (Avogadro s number) 1 mb = 100 Pa 1 Pa = 1 N/m 2 Γ d = 9.8 o C/km (dry adiabatic lapse rate)
Formulas/Constants, Physics/Oceanography 4510/5510 B Atmospheric Physics II N A = 6.02 10 23 molecules/mole (Avogadro s number) 1 mb = 100 Pa 1 Pa = 1 N/m 2 Γ d = 9.8 o C/km (dry adiabatic lapse rate)
Exam 1 (Chaps. 1-6 of the notes)
 10/12/06 ATS 541 - Atmospheric Thermodynamics and Cloud Physics 1 Exam 1 (Chaps. 1-6 of the notes) ATS 541 students: Answer all questions ATS 441 students: You may delete problem 3 or problem 5 1. [10
10/12/06 ATS 541 - Atmospheric Thermodynamics and Cloud Physics 1 Exam 1 (Chaps. 1-6 of the notes) ATS 541 students: Answer all questions ATS 441 students: You may delete problem 3 or problem 5 1. [10
Chapter 4 Water Vapor
 Chapter 4 Water Vapor Chapter overview: Phases of water Vapor pressure at saturation Moisture variables o Mixing ratio, specific humidity, relative humidity, dew point temperature o Absolute vs. relative
Chapter 4 Water Vapor Chapter overview: Phases of water Vapor pressure at saturation Moisture variables o Mixing ratio, specific humidity, relative humidity, dew point temperature o Absolute vs. relative
First Law of Thermodynamics
 First Law of Thermodynamics September 11, 2013 The first law of thermodynamics is the conservation of energy applied to thermal systems. Here, we develop the principles of thermodynamics for a discrete
First Law of Thermodynamics September 11, 2013 The first law of thermodynamics is the conservation of energy applied to thermal systems. Here, we develop the principles of thermodynamics for a discrete
Lecture Ch. 6. Condensed (Liquid) Water. Cloud in a Jar Demonstration. How does saturation occur? Saturation of Moist Air. Saturation of Moist Air
 Lecture Ch. 6 Saturation of moist air Relationship between humidity and dewpoint Clausius-Clapeyron equation Dewpoint Temperature Depression Isobaric cooling Moist adiabatic ascent of air Equivalent temperature
Lecture Ch. 6 Saturation of moist air Relationship between humidity and dewpoint Clausius-Clapeyron equation Dewpoint Temperature Depression Isobaric cooling Moist adiabatic ascent of air Equivalent temperature
Parcel Model. Meteorology September 3, 2008
 Parcel Model Meteorology 5210 September 3, 2008 1 Governing Equations for Precipitating Convection For precipitating convection, we have the following set of equations for potential temperature, θ, mixing
Parcel Model Meteorology 5210 September 3, 2008 1 Governing Equations for Precipitating Convection For precipitating convection, we have the following set of equations for potential temperature, θ, mixing
Meteorology 6150 Cloud System Modeling
 Meteorology 6150 Cloud System Modeling Steve Krueger Spring 2009 1 Fundamental Equations 1.1 The Basic Equations 1.1.1 Equation of motion The movement of air in the atmosphere is governed by Newton s Second
Meteorology 6150 Cloud System Modeling Steve Krueger Spring 2009 1 Fundamental Equations 1.1 The Basic Equations 1.1.1 Equation of motion The movement of air in the atmosphere is governed by Newton s Second
ATMO/OPTI 656b Spring 08. Physical Properties of the Atmosphere
 Physical Properties of the Atmosphere Thin as a piece of paper The atmosphere is a very thin layer above the solid Earth and its oceans. This is true of the atmospheres of all of the terrestrial planets.
Physical Properties of the Atmosphere Thin as a piece of paper The atmosphere is a very thin layer above the solid Earth and its oceans. This is true of the atmospheres of all of the terrestrial planets.
Today s Lecture: Atmosphere finish primitive equations, mostly thermodynamics
 Today s Lecture: Atmosphere finish primitive equations, mostly thermodynamics Reference Peixoto and Oort, Sec. 3.1, 3.2, 3.4, 3.5 (but skip the discussion of oceans until next week); Ch. 10 Thermodynamic
Today s Lecture: Atmosphere finish primitive equations, mostly thermodynamics Reference Peixoto and Oort, Sec. 3.1, 3.2, 3.4, 3.5 (but skip the discussion of oceans until next week); Ch. 10 Thermodynamic
Parcel Model. Atmospheric Sciences September 30, 2012
 Parcel Model Atmospheric Sciences 6150 September 30, 2012 1 Governing Equations for Precipitating Convection For precipitating convection, we have the following set of equations for potential temperature,
Parcel Model Atmospheric Sciences 6150 September 30, 2012 1 Governing Equations for Precipitating Convection For precipitating convection, we have the following set of equations for potential temperature,
Clouds associated with cold and warm fronts. Whiteman (2000)
 Clouds associated with cold and warm fronts Whiteman (2000) Dalton s law of partial pressures! The total pressure exerted by a mixture of gases equals the sum of the partial pressure of the gases! Partial
Clouds associated with cold and warm fronts Whiteman (2000) Dalton s law of partial pressures! The total pressure exerted by a mixture of gases equals the sum of the partial pressure of the gases! Partial
ρ x + fv f 'w + F x ρ y fu + F y Fundamental Equation in z coordinate p = ρrt or pα = RT Du uv tanφ Dv Dt + u2 tanφ + vw a a = 1 p Dw Dt u2 + v 2
 Fundamental Equation in z coordinate p = ρrt or pα = RT Du uv tanφ + uw Dt a a = 1 p ρ x + fv f 'w + F x Dv Dt + u2 tanφ + vw a a = 1 p ρ y fu + F y Dw Dt u2 + v 2 = 1 p a ρ z g + f 'u + F z Dρ Dt + ρ
Fundamental Equation in z coordinate p = ρrt or pα = RT Du uv tanφ + uw Dt a a = 1 p ρ x + fv f 'w + F x Dv Dt + u2 tanφ + vw a a = 1 p ρ y fu + F y Dw Dt u2 + v 2 = 1 p a ρ z g + f 'u + F z Dρ Dt + ρ
2 Equations of Motion
 2 Equations of Motion system. In this section, we will derive the six full equations of motion in a non-rotating, Cartesian coordinate 2.1 Six equations of motion (non-rotating, Cartesian coordinates)
2 Equations of Motion system. In this section, we will derive the six full equations of motion in a non-rotating, Cartesian coordinate 2.1 Six equations of motion (non-rotating, Cartesian coordinates)
Clouds and turbulent moist convection
 Clouds and turbulent moist convection Lecture 2: Cloud formation and Physics Caroline Muller Les Houches summer school Lectures Outline : Cloud fundamentals - global distribution, types, visualization
Clouds and turbulent moist convection Lecture 2: Cloud formation and Physics Caroline Muller Les Houches summer school Lectures Outline : Cloud fundamentals - global distribution, types, visualization
Buoyancy and Coriolis forces
 Chapter 2 Buoyancy and Coriolis forces In this chapter we address several topics that we need to understand before starting on our study of geophysical uid dynamics. 2.1 Hydrostatic approximation Consider
Chapter 2 Buoyancy and Coriolis forces In this chapter we address several topics that we need to understand before starting on our study of geophysical uid dynamics. 2.1 Hydrostatic approximation Consider
1. Static Stability. (ρ V ) d2 z (1) d 2 z. = g (2) = g (3) T T = g T (4)
 NCAR (National Center for Atmospheric Research) has an excellent resource for education called COMET-MetEd. There you can find some really great tutorials on SkewT-LogP plots: visit http://www.meted.ucar.edu/mesoprim/skewt/index.htm.
NCAR (National Center for Atmospheric Research) has an excellent resource for education called COMET-MetEd. There you can find some really great tutorials on SkewT-LogP plots: visit http://www.meted.ucar.edu/mesoprim/skewt/index.htm.
Simplified Microphysics. condensation evaporation. evaporation
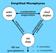 Simplified Microphysics water vapor condensation evaporation cloud droplets evaporation condensation collection rain drops fall out (precipitation) = 0 (reversible) = (irreversible) Simplified Microphysics
Simplified Microphysics water vapor condensation evaporation cloud droplets evaporation condensation collection rain drops fall out (precipitation) = 0 (reversible) = (irreversible) Simplified Microphysics
References: Parcel Theory. Vertical Force Balance. ESCI Cloud Physics and Precipitation Processes Lesson 3 - Stability and Buoyancy Dr.
 References: ESCI 340 - Cloud Physics and Precipitation Processes Lesson 3 - Stability and Buoyancy Dr. DeCaria Glossary of Meteorology, 2nd ed., American Meteorological Society A Short Course in Cloud
References: ESCI 340 - Cloud Physics and Precipitation Processes Lesson 3 - Stability and Buoyancy Dr. DeCaria Glossary of Meteorology, 2nd ed., American Meteorological Society A Short Course in Cloud
Atsc final Equations: page 1/6
 Atsc. 405 2012 final Equations: page 1/6 Answer each of these 7 questions (note weight). Show all your work on all questions (needed for partial credit). Be sure to put your name on any detached pages.
Atsc. 405 2012 final Equations: page 1/6 Answer each of these 7 questions (note weight). Show all your work on all questions (needed for partial credit). Be sure to put your name on any detached pages.
1/18/2011. Conservation of Momentum Conservation of Mass Conservation of Energy Scaling Analysis ESS227 Prof. Jin-Yi Yu
 Lecture 2: Basic Conservation Laws Conservation Law of Momentum Newton s 2 nd Law of Momentum = absolute velocity viewed in an inertial system = rate of change of Ua following the motion in an inertial
Lecture 2: Basic Conservation Laws Conservation Law of Momentum Newton s 2 nd Law of Momentum = absolute velocity viewed in an inertial system = rate of change of Ua following the motion in an inertial
Measuring State Parameters of the Atmosphere
 Measuring State Parameters of the Atmosphere Some Applications of Atmospheric Thermodynamics Earth Observing Laboratory, NCAR IDEAS-4 Tutorial Introduction Goals of This Presentation Present two complementary
Measuring State Parameters of the Atmosphere Some Applications of Atmospheric Thermodynamics Earth Observing Laboratory, NCAR IDEAS-4 Tutorial Introduction Goals of This Presentation Present two complementary
Naraine Persaud, Entry Code ME-11 1
 Naraine Persaud, Entry Code ME-11 1 Persaud, N. 2005. Adiabatic cooling. In: Water Encyclopedia Volume 4: Oceanography; Meteorology; Physics and Chemistry; Water Law; and Water History, Art, and Culture.
Naraine Persaud, Entry Code ME-11 1 Persaud, N. 2005. Adiabatic cooling. In: Water Encyclopedia Volume 4: Oceanography; Meteorology; Physics and Chemistry; Water Law; and Water History, Art, and Culture.
Hence. The second law describes the direction of energy transfer in spontaneous processes
 * Heat and Work The first law of thermodynamics states that: Although energy has many forms, the total quantity of energy is constant. When energy disappears in one form, it appears simultaneously in other
* Heat and Work The first law of thermodynamics states that: Although energy has many forms, the total quantity of energy is constant. When energy disappears in one form, it appears simultaneously in other
The Clausius-Clapeyron and the Kelvin Equations
 PhD Environmental Fluid Mechanics Physics of the Atmosphere University of Trieste International Center for Theoretical Physics The Clausius-Clapeyron and the Kelvin Equations by Dario B. Giaiotti and Fulvio
PhD Environmental Fluid Mechanics Physics of the Atmosphere University of Trieste International Center for Theoretical Physics The Clausius-Clapeyron and the Kelvin Equations by Dario B. Giaiotti and Fulvio
Introduction. Lecture 6: Water in Atmosphere. How Much Heat Is Brought Upward By Water Vapor?
 Lecture 6: Water in Atmosphere Introduction Over 70% of the planet is covered by water Water is unique in that it can simultaneously exist in all three states (solid, liquid, gas) at the same temperature
Lecture 6: Water in Atmosphere Introduction Over 70% of the planet is covered by water Water is unique in that it can simultaneously exist in all three states (solid, liquid, gas) at the same temperature
Atmospheric Dynamics: lecture 2
 Atmospheric Dynamics: lecture 2 Topics Some aspects of advection and the Coriolis-effect (1.7) Composition of the atmosphere (figure 1.6) Equation of state (1.8&1.9) Water vapour in the atmosphere (1.10)
Atmospheric Dynamics: lecture 2 Topics Some aspects of advection and the Coriolis-effect (1.7) Composition of the atmosphere (figure 1.6) Equation of state (1.8&1.9) Water vapour in the atmosphere (1.10)
Dynamic Meteorology: lecture 2
 Dynamic Meteorology: lecture 2 Sections 1.3-1.5 and Box 1.5 Potential temperature Radiatively determined temperature (boxes 1.1-1.4) Buoyancy (-oscillations) and static instability, Brunt-Vaisala frequency
Dynamic Meteorology: lecture 2 Sections 1.3-1.5 and Box 1.5 Potential temperature Radiatively determined temperature (boxes 1.1-1.4) Buoyancy (-oscillations) and static instability, Brunt-Vaisala frequency
ATMOS 5130 Lecture 9. Enthalpy Conservation Property The Second Law and Its Consequences Entropy
 ATMOS 5130 Lecture 9 Enthalpy Conservation Property The Second Law and Its Consequences Entropy CLASS Presentation Form group of 2 students Present ~20 minute presentation (~ 10 minute each person) Focus
ATMOS 5130 Lecture 9 Enthalpy Conservation Property The Second Law and Its Consequences Entropy CLASS Presentation Form group of 2 students Present ~20 minute presentation (~ 10 minute each person) Focus
Measuring State Parameters of the Atmosphere
 Measuring State Parameters of the Atmosphere Some Applications of Atmospheric Thermodynamics Earth Observing Laboratory, NCAR IDEAS-4 Tutorial Introduction Goals of This Presentation Present two complementary
Measuring State Parameters of the Atmosphere Some Applications of Atmospheric Thermodynamics Earth Observing Laboratory, NCAR IDEAS-4 Tutorial Introduction Goals of This Presentation Present two complementary
Rocket propulsion Prof. K. Ramamurthi Department of Mechanical Engineering Indian Institute of Technology, Madras. Lecture 09 Theory of Nozzles
 Rocket propulsion Prof. K. Ramamurthi Department of Mechanical Engineering Indian Institute of Technology, Madras Lecture 09 Theory of Nozzles (Refer Slide Time: 00:14) Good morning. We will develop the
Rocket propulsion Prof. K. Ramamurthi Department of Mechanical Engineering Indian Institute of Technology, Madras Lecture 09 Theory of Nozzles (Refer Slide Time: 00:14) Good morning. We will develop the
Project 3 Convection and Atmospheric Thermodynamics
 12.818 Project 3 Convection and Atmospheric Thermodynamics Lodovica Illari 1 Background The Earth is bathed in radiation from the Sun whose intensity peaks in the visible. In order to maintain energy balance
12.818 Project 3 Convection and Atmospheric Thermodynamics Lodovica Illari 1 Background The Earth is bathed in radiation from the Sun whose intensity peaks in the visible. In order to maintain energy balance
1. The vertical structure of the atmosphere. Temperature profile.
 Lecture 4. The structure of the atmosphere. Air in motion. Objectives: 1. The vertical structure of the atmosphere. Temperature profile. 2. Temperature in the lower atmosphere: dry adiabatic lapse rate.
Lecture 4. The structure of the atmosphere. Air in motion. Objectives: 1. The vertical structure of the atmosphere. Temperature profile. 2. Temperature in the lower atmosphere: dry adiabatic lapse rate.
GEF2200 Atmosfærefysikk 2012
 GEF2200 Atmosfærefysikk 2012 Løsningsforslag til oppgavesett 4 WH06 3.46 (WH 2.49) The air parcel has the properties p = 1000hPa, T = 15 C and T d = 4 C. b Lifting the air parcel to p 2 = 900hPa, T 2 we
GEF2200 Atmosfærefysikk 2012 Løsningsforslag til oppgavesett 4 WH06 3.46 (WH 2.49) The air parcel has the properties p = 1000hPa, T = 15 C and T d = 4 C. b Lifting the air parcel to p 2 = 900hPa, T 2 we
First-Order Differential Equations
 CHAPTER 1 First-Order Differential Equations 1. Diff Eqns and Math Models Know what it means for a function to be a solution to a differential equation. In order to figure out if y = y(x) is a solution
CHAPTER 1 First-Order Differential Equations 1. Diff Eqns and Math Models Know what it means for a function to be a solution to a differential equation. In order to figure out if y = y(x) is a solution
GEF2200 atmospheric physics 2018
 GEF2200 atmospheric physics 208 Solutions: thermodynamics 3 Oppgaver hentet fra boka Wallace and Hobbs (2006) er merket WH06 WH06 3.8r Unsaturated air is lifted (adiabatically): The first pair of quantities
GEF2200 atmospheric physics 208 Solutions: thermodynamics 3 Oppgaver hentet fra boka Wallace and Hobbs (2006) er merket WH06 WH06 3.8r Unsaturated air is lifted (adiabatically): The first pair of quantities
Conservation of Mass Conservation of Energy Scaling Analysis. ESS227 Prof. Jin-Yi Yu
 Lecture 2: Basic Conservation Laws Conservation of Momentum Conservation of Mass Conservation of Energy Scaling Analysis Conservation Law of Momentum Newton s 2 nd Law of Momentum = absolute velocity viewed
Lecture 2: Basic Conservation Laws Conservation of Momentum Conservation of Mass Conservation of Energy Scaling Analysis Conservation Law of Momentum Newton s 2 nd Law of Momentum = absolute velocity viewed
Physics Nov Cooling by Expansion
 Physics 301 19-Nov-2004 25-1 Cooling by Expansion Now we re going to change the subject and consider the techniques used to get really cold temperatures. Of course, the best way to learn about these techniques
Physics 301 19-Nov-2004 25-1 Cooling by Expansion Now we re going to change the subject and consider the techniques used to get really cold temperatures. Of course, the best way to learn about these techniques
Atmospheric Physics. August 10, 2017
 Atmospheric Physics August 10, 2017 1 Contents I Equilibrium Thermodynamics 6 1 Molecular Thermodynamics 6 1.1 Atmospheric application: water vapor and dry air.................. 10 2 Intensive and extensive
Atmospheric Physics August 10, 2017 1 Contents I Equilibrium Thermodynamics 6 1 Molecular Thermodynamics 6 1.1 Atmospheric application: water vapor and dry air.................. 10 2 Intensive and extensive
Kelvin Effect. Covers Reading Material in Chapter 10.3 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics
 Kelvin Effect Covers Reading Material in Chapter 10.3 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics Vapor Pressure (e) e < e # e = e # Vapor Pressure e > e # Relative humidity RH =
Kelvin Effect Covers Reading Material in Chapter 10.3 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics Vapor Pressure (e) e < e # e = e # Vapor Pressure e > e # Relative humidity RH =
Temperature and Thermodynamics, Part II. Topics to be Covered
 Teperature and Therodynaics, Part II Topics to be Covered Profiles of Teperature in the Boundary Layer Potential teperature Adiabatic Lapse Rate Theral Stratification 1/8/17 Why are We Interested in Theral
Teperature and Therodynaics, Part II Topics to be Covered Profiles of Teperature in the Boundary Layer Potential teperature Adiabatic Lapse Rate Theral Stratification 1/8/17 Why are We Interested in Theral
ATMO551a Fall Vertical Structure of Earth s Atmosphere
 Vertical Structure of Earth s Atmosphere Thin as a piece of paper The atmosphere is a very thin layer above the solid Earth and its oceans. This is true of the atmospheres of all of the terrestrial planets.
Vertical Structure of Earth s Atmosphere Thin as a piece of paper The atmosphere is a very thin layer above the solid Earth and its oceans. This is true of the atmospheres of all of the terrestrial planets.
First Law of Thermodyamics U = Q + W. We can rewrite this by introducing two physical. Enthalpy, H, is the quantity U + pv, as this
 First Law of Thermodyamics U = Q + W where U is the increase in internal energy of the system, Q is the heat supplied to the system and W is the work done on the system. We can rewrite this by introducing
First Law of Thermodyamics U = Q + W where U is the increase in internal energy of the system, Q is the heat supplied to the system and W is the work done on the system. We can rewrite this by introducing
Thermodynamic Energy Equation
 Thermodynamic Energy Equation The temperature tendency is = u T x v T y w T z + dt dt (1) where dt/dt is the individual derivative of temperature. This temperature change experienced by the air parcel
Thermodynamic Energy Equation The temperature tendency is = u T x v T y w T z + dt dt (1) where dt/dt is the individual derivative of temperature. This temperature change experienced by the air parcel
Classical Thermodynamics. Dr. Massimo Mella School of Chemistry Cardiff University
 Classical Thermodynamics Dr. Massimo Mella School of Chemistry Cardiff University E-mail:MellaM@cardiff.ac.uk The background The field of Thermodynamics emerged as a consequence of the necessity to understand
Classical Thermodynamics Dr. Massimo Mella School of Chemistry Cardiff University E-mail:MellaM@cardiff.ac.uk The background The field of Thermodynamics emerged as a consequence of the necessity to understand
Applied Thermodynamics for Marine Systems Prof. P. K. Das Department of Mechanical Engineering Indian Institute of Technology, Kharagpur
 Applied Thermodynamics for Marine Systems Prof. P. K. Das Department of Mechanical Engineering Indian Institute of Technology, Kharagpur Lecture - 21 Psychometric Processes Good afternoon, yesterday we
Applied Thermodynamics for Marine Systems Prof. P. K. Das Department of Mechanical Engineering Indian Institute of Technology, Kharagpur Lecture - 21 Psychometric Processes Good afternoon, yesterday we
What is thermodynamics? and what can it do for us?
 What is thermodynamics? and what can it do for us? The overall goal of thermodynamics is to describe what happens to a system (anything of interest) when we change the variables that characterized the
What is thermodynamics? and what can it do for us? The overall goal of thermodynamics is to describe what happens to a system (anything of interest) when we change the variables that characterized the
4.1 LAWS OF MECHANICS - Review
 4.1 LAWS OF MECHANICS - Review Ch4 9 SYSTEM System: Moving Fluid Definitions: System is defined as an arbitrary quantity of mass of fixed identity. Surrounding is everything external to this system. Boundary
4.1 LAWS OF MECHANICS - Review Ch4 9 SYSTEM System: Moving Fluid Definitions: System is defined as an arbitrary quantity of mass of fixed identity. Surrounding is everything external to this system. Boundary
Entropy and the Second Law of Thermodynamics
 Entropy and the Second Law of Thermodynamics Reading Problems 7-1 7-3 7-88, 7-131, 7-135 7-6 7-10 8-24, 8-44, 8-46, 8-60, 8-73, 8-99, 8-128, 8-132, 8-1 8-10, 8-13 8-135, 8-148, 8-152, 8-166, 8-168, 8-189
Entropy and the Second Law of Thermodynamics Reading Problems 7-1 7-3 7-88, 7-131, 7-135 7-6 7-10 8-24, 8-44, 8-46, 8-60, 8-73, 8-99, 8-128, 8-132, 8-1 8-10, 8-13 8-135, 8-148, 8-152, 8-166, 8-168, 8-189
Lecture 9: Climate Sensitivity and Feedback Mechanisms
 Lecture 9: Climate Sensitivity and Feedback Mechanisms Basic radiative feedbacks (Plank, Water Vapor, Lapse-Rate Feedbacks) Ice albedo & Vegetation-Climate feedback Cloud feedback Biogeochemical feedbacks
Lecture 9: Climate Sensitivity and Feedback Mechanisms Basic radiative feedbacks (Plank, Water Vapor, Lapse-Rate Feedbacks) Ice albedo & Vegetation-Climate feedback Cloud feedback Biogeochemical feedbacks
Convective Heat and Mass Transfer Prof. A.W. Date Department of Mechanical Engineering Indian Institute of Technology, Bombay
 Convective Heat and Mass Transfer Prof. A.W. Date Department of Mechanical Engineering Indian Institute of Technology, Bombay Module No. # 01 Lecture No. # 32 Stefan Flow Model We are now familiar with
Convective Heat and Mass Transfer Prof. A.W. Date Department of Mechanical Engineering Indian Institute of Technology, Bombay Module No. # 01 Lecture No. # 32 Stefan Flow Model We are now familiar with
4. Atmospheric transport. Daniel J. Jacob, Atmospheric Chemistry, Harvard University, Spring 2017
 4. Atmospheric transport Daniel J. Jacob, Atmospheric Chemistry, Harvard University, Spring 2017 Forces in the atmosphere: Gravity g Pressure-gradient ap = ( 1/ ρ ) dp / dx for x-direction (also y, z directions)
4. Atmospheric transport Daniel J. Jacob, Atmospheric Chemistry, Harvard University, Spring 2017 Forces in the atmosphere: Gravity g Pressure-gradient ap = ( 1/ ρ ) dp / dx for x-direction (also y, z directions)
Lecture 7. Science A-30 February 21, 2008 Air may be forced to move up or down in the atmosphere by mechanical forces (wind blowing over an obstacle,
 Lecture 7. Science A-30 February 21, 2008 Air may be forced to move up or down in the atmosphere by mechanical forces (wind blowing over an obstacle, like a mountain) or by buoyancy forces. Air that is
Lecture 7. Science A-30 February 21, 2008 Air may be forced to move up or down in the atmosphere by mechanical forces (wind blowing over an obstacle, like a mountain) or by buoyancy forces. Air that is
ESCI 485 Air/Sea Interaction Lesson 1 Stresses and Fluxes Dr. DeCaria
 ESCI 485 Air/Sea Interaction Lesson 1 Stresses and Fluxes Dr DeCaria References: An Introduction to Dynamic Meteorology, Holton MOMENTUM EQUATIONS The momentum equations governing the ocean or atmosphere
ESCI 485 Air/Sea Interaction Lesson 1 Stresses and Fluxes Dr DeCaria References: An Introduction to Dynamic Meteorology, Holton MOMENTUM EQUATIONS The momentum equations governing the ocean or atmosphere
EART164: PLANETARY ATMOSPHERES
 EART16: PLANETARY ATMOSPHERES Francis Nimmo Last Week How do planets form? They accrete from the solar nebula (dust+gas) They may subsequently migrate Where do atmospheres come from? Primary, secondary,
EART16: PLANETARY ATMOSPHERES Francis Nimmo Last Week How do planets form? They accrete from the solar nebula (dust+gas) They may subsequently migrate Where do atmospheres come from? Primary, secondary,
Liquid water static energy page 1/8
 Liquid water static energy age 1/8 1) Thermodynamics It s a good idea to work with thermodynamic variables that are conserved under a known set of conditions, since they can act as assive tracers and rovide
Liquid water static energy age 1/8 1) Thermodynamics It s a good idea to work with thermodynamic variables that are conserved under a known set of conditions, since they can act as assive tracers and rovide
Planetary Atmospheres
 Planetary Atmospheres Structure Composition Meteorology Clouds Photochemistry Atmospheric Escape EAS 4803/8803 - CP 20:1 Cloud formation Saturated Vapor Pressure: Maximum amount of water vapor partial
Planetary Atmospheres Structure Composition Meteorology Clouds Photochemistry Atmospheric Escape EAS 4803/8803 - CP 20:1 Cloud formation Saturated Vapor Pressure: Maximum amount of water vapor partial
Global Energy Balance: Greenhouse Effect
 Global Energy Balance: Greenhouse Effect Atmospheric Composition & Structure Physical Causes of Greenhouse Effects Chapter 3: 44 48. Atmospheric Composition Why does water vapor vary so much? Saturation
Global Energy Balance: Greenhouse Effect Atmospheric Composition & Structure Physical Causes of Greenhouse Effects Chapter 3: 44 48. Atmospheric Composition Why does water vapor vary so much? Saturation
2.1 Effects of a cumulus ensemble upon the large scale temperature and moisture fields by induced subsidence and detrainment
 Atmospheric Sciences 6150 Cloud System Modeling 2.1 Effects of a cumulus ensemble upon the large scale temperature and moisture fields by induced subsidence and detrainment Arakawa (1969, 1972), W. Gray
Atmospheric Sciences 6150 Cloud System Modeling 2.1 Effects of a cumulus ensemble upon the large scale temperature and moisture fields by induced subsidence and detrainment Arakawa (1969, 1972), W. Gray
An alternative, less empirical approach (though still full of brazen assumptions) is the following:
 ERTH 500: More Notes on Final Project: Dr. Dave Dempsey Earth Systems II Modeling the Dept. of (Spring 2016) Cenozoic Icehouse Earth Earth & Climate Sciences More notes on Upslope/Monsoon Precipitation
ERTH 500: More Notes on Final Project: Dr. Dave Dempsey Earth Systems II Modeling the Dept. of (Spring 2016) Cenozoic Icehouse Earth Earth & Climate Sciences More notes on Upslope/Monsoon Precipitation
ATMO 551a Fall The Carnot Cycle
 What is a arnot ycle and Why do we care The arnot ycle arnot was a French engineer who was trying to understand how to extract usable mechanical work from a heat engine, that is an engine where a gas or
What is a arnot ycle and Why do we care The arnot ycle arnot was a French engineer who was trying to understand how to extract usable mechanical work from a heat engine, that is an engine where a gas or
Numerical Example An air parcel with mass of 1 kg rises adiabatically from sea level to an altitude of 3 km. What is its temperature change?
 Numerical Example An air parcel with mass of 1 kg rises adiabatically from sea level to an altitude of 3 km. What is its temperature change? From the 1 st law, T = -g/c p z + Q/m air /c p Here, Q = 0,
Numerical Example An air parcel with mass of 1 kg rises adiabatically from sea level to an altitude of 3 km. What is its temperature change? From the 1 st law, T = -g/c p z + Q/m air /c p Here, Q = 0,
Thermodynamics Introduction and Basic Concepts
 Thermodynamics Introduction and Basic Concepts by Asst. Prof. Channarong Asavatesanupap Mechanical Engineering Department Faculty of Engineering Thammasat University 2 What is Thermodynamics? Thermodynamics
Thermodynamics Introduction and Basic Concepts by Asst. Prof. Channarong Asavatesanupap Mechanical Engineering Department Faculty of Engineering Thammasat University 2 What is Thermodynamics? Thermodynamics
Chapter 5 - Atmospheric Moisture
 Chapter 5 - Atmospheric Moisture Understanding Weather and Climate Aguado and Burt Water Water Vapor - water in a gaseous form, not droplets. Water can also achieve solid and liquid phases on Earth Temperature
Chapter 5 - Atmospheric Moisture Understanding Weather and Climate Aguado and Burt Water Water Vapor - water in a gaseous form, not droplets. Water can also achieve solid and liquid phases on Earth Temperature
w = -nrt hot ln(v 2 /V 1 ) nrt cold ln(v 1 /V 2 )[sincev/v 4 3 = V 1 /V 2 ]
![w = -nrt hot ln(v 2 /V 1 ) nrt cold ln(v 1 /V 2 )[sincev/v 4 3 = V 1 /V 2 ] w = -nrt hot ln(v 2 /V 1 ) nrt cold ln(v 1 /V 2 )[sincev/v 4 3 = V 1 /V 2 ]](/thumbs/85/91320285.jpg) Chemistry 433 Lecture 9 Entropy and the Second Law NC State University Spontaneity of Chemical Reactions One might be tempted based on the results of thermochemistry to predict that all exothermic reactions
Chemistry 433 Lecture 9 Entropy and the Second Law NC State University Spontaneity of Chemical Reactions One might be tempted based on the results of thermochemistry to predict that all exothermic reactions
Needs work : define boundary conditions and fluxes before, change slides Useful definitions and conservation equations
 Needs work : define boundary conditions and fluxes before, change slides 1-2-3 Useful definitions and conservation equations Turbulent Kinetic energy The fluxes are crucial to define our boundary conditions,
Needs work : define boundary conditions and fluxes before, change slides 1-2-3 Useful definitions and conservation equations Turbulent Kinetic energy The fluxes are crucial to define our boundary conditions,
LECTURE 5 THE CONTENTS OF THIS LECTURE ARE AS FOLLOWS: 1.0 SOME OF THE THEORETICAL CONCEPTS INVOLVED IN HEAT FLOW. 1.1 Sensible Heat Flow
 LECTURE 5 THE CONTENTS OF THIS LECTURE ARE AS FOLLOWS: 1.0 SOME OF THE THEORETICAL CONCEPTS INVOLVED IN HEAT FLOW 1.1 Sensible Heat Flow 1.2 Latent heat Flow 1.3 Humidity 1.4 Thermostat Condition in the
LECTURE 5 THE CONTENTS OF THIS LECTURE ARE AS FOLLOWS: 1.0 SOME OF THE THEORETICAL CONCEPTS INVOLVED IN HEAT FLOW 1.1 Sensible Heat Flow 1.2 Latent heat Flow 1.3 Humidity 1.4 Thermostat Condition in the
Köhler Curve. Covers Reading Material in Chapter 10.3 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics
 Köhler Curve Covers Reading Material in Chapter 10.3 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics Review of Kelvin Effect Gibbs Energy for formation of a drop G = G &'()*+, G ).'+
Köhler Curve Covers Reading Material in Chapter 10.3 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics Review of Kelvin Effect Gibbs Energy for formation of a drop G = G &'()*+, G ).'+
Atmospheric Sciences 321. Science of Climate. Lecture 13: Surface Energy Balance Chapter 4
 Atmospheric Sciences 321 Science of Climate Lecture 13: Surface Energy Balance Chapter 4 Community Business Check the assignments HW #4 due Wednesday Quiz #2 Wednesday Mid Term is Wednesday May 6 Practice
Atmospheric Sciences 321 Science of Climate Lecture 13: Surface Energy Balance Chapter 4 Community Business Check the assignments HW #4 due Wednesday Quiz #2 Wednesday Mid Term is Wednesday May 6 Practice
CHEMICAL ENGINEERING THERMODYNAMICS. Andrew S. Rosen
 CHEMICAL ENGINEERING THERMODYNAMICS Andrew S. Rosen SYMBOL DICTIONARY 1 TABLE OF CONTENTS Symbol Dictionary... 3 1. Measured Thermodynamic Properties and Other Basic Concepts... 5 1.1 Preliminary Concepts
CHEMICAL ENGINEERING THERMODYNAMICS Andrew S. Rosen SYMBOL DICTIONARY 1 TABLE OF CONTENTS Symbol Dictionary... 3 1. Measured Thermodynamic Properties and Other Basic Concepts... 5 1.1 Preliminary Concepts
Composition, Structure and Energy. ATS 351 Lecture 2 September 14, 2009
 Composition, Structure and Energy ATS 351 Lecture 2 September 14, 2009 Composition of the Atmosphere Atmospheric Properties Temperature Pressure Wind Moisture (i.e. water vapor) Density Temperature A measure
Composition, Structure and Energy ATS 351 Lecture 2 September 14, 2009 Composition of the Atmosphere Atmospheric Properties Temperature Pressure Wind Moisture (i.e. water vapor) Density Temperature A measure
Lecture 3: Convection
 EESC V2100 The Climate System spring 2004 Lecture 3: Convection Yochanan Kushnir Lamont Doherty Earth Observatory of Columbia University Palisades, NY 10964, USA kushnir@ldeo.columbia.edu Layers of the
EESC V2100 The Climate System spring 2004 Lecture 3: Convection Yochanan Kushnir Lamont Doherty Earth Observatory of Columbia University Palisades, NY 10964, USA kushnir@ldeo.columbia.edu Layers of the
Step 1. Step 2. g l = g v. dg = 0 We have shown that over a plane surface of water. g v g l = ρ v R v T ln e/e sat. this can be rewritten
 The basic question is what makes the existence of a droplet thermodynamically preferable to the existence only of water vapor. We have already derived an expression for the saturation vapor pressure over
The basic question is what makes the existence of a droplet thermodynamically preferable to the existence only of water vapor. We have already derived an expression for the saturation vapor pressure over
Diffusional Growth of Liquid Phase Hydrometeros.
 Diffusional Growth of Liquid Phase Hydrometeros. I. Diffusional Growth of Liquid Phase Hydrometeors A. Basic concepts of diffusional growth. 1. To understand the diffusional growth of a droplet, we must
Diffusional Growth of Liquid Phase Hydrometeros. I. Diffusional Growth of Liquid Phase Hydrometeors A. Basic concepts of diffusional growth. 1. To understand the diffusional growth of a droplet, we must
Lecture 10: Climate Sensitivity and Feedback
 Lecture 10: Climate Sensitivity and Feedback Human Activities Climate Sensitivity Climate Feedback 1 Climate Sensitivity and Feedback (from Earth s Climate: Past and Future) 2 Definition and Mathematic
Lecture 10: Climate Sensitivity and Feedback Human Activities Climate Sensitivity Climate Feedback 1 Climate Sensitivity and Feedback (from Earth s Climate: Past and Future) 2 Definition and Mathematic
Basic Thermodynamics Prof. S.K Som Department of Mechanical Engineering Indian Institute of Technology, Kharagpur
 Basic Thermodynamics Prof. S.K Som Department of Mechanical Engineering Indian Institute of Technology, Kharagpur Lecture - 17 Properties of Pure Substances-I Good morning to all of you. We were discussing
Basic Thermodynamics Prof. S.K Som Department of Mechanical Engineering Indian Institute of Technology, Kharagpur Lecture - 17 Properties of Pure Substances-I Good morning to all of you. We were discussing
Chapter 4. Energy Analysis of Closed Systems
 Chapter 4 Energy Analysis of Closed Systems The first law of thermodynamics is an expression of the conservation of energy principle. Energy can cross the boundaries of a closed system in the form of heat
Chapter 4 Energy Analysis of Closed Systems The first law of thermodynamics is an expression of the conservation of energy principle. Energy can cross the boundaries of a closed system in the form of heat
First Law CML 100, IIT Delhi SS. The total energy of the system. Contribution from translation + rotation + vibrations.
 Internal Energy he total energy of the system. Contribution from translation + rotation + vibrations. Equipartition theorem for the translation and rotational degrees of freedom. 1/ k B Work Path function,
Internal Energy he total energy of the system. Contribution from translation + rotation + vibrations. Equipartition theorem for the translation and rotational degrees of freedom. 1/ k B Work Path function,
1 One-dimensional analysis
 One-dimensional analysis. Introduction The simplest models for gas liquid flow systems are ones for which the velocity is uniform over a cross-section and unidirectional. This includes flows in a long
One-dimensional analysis. Introduction The simplest models for gas liquid flow systems are ones for which the velocity is uniform over a cross-section and unidirectional. This includes flows in a long
Radiative-Convective Models. The Hydrological Cycle Hadley Circulation. Manabe and Strickler (1964) Course Notes chapter 5.1
 Climate Modeling Lecture 8 Radiative-Convective Models Manabe and Strickler (1964) Course Notes chapter 5.1 The Hydrological Cycle Hadley Circulation Prepare for Mid-Term (Friday 9 am) Review Course Notes
Climate Modeling Lecture 8 Radiative-Convective Models Manabe and Strickler (1964) Course Notes chapter 5.1 The Hydrological Cycle Hadley Circulation Prepare for Mid-Term (Friday 9 am) Review Course Notes
Theory. Humidity h of an air-vapor mixture is defined as the mass ratio of water vapor and dry air,
 Theory Background In a cooling tower with open water circulation, heat is removed from water because of the material and heat exchange between the water and the ambient air. The cooling tower is a special
Theory Background In a cooling tower with open water circulation, heat is removed from water because of the material and heat exchange between the water and the ambient air. The cooling tower is a special
Isobaric Coordinates
 METR 402: Atmos Dynamics I Fall 20 Isobaric Coordinates Dr. Dave Dempsey Dept. of Geosciences, SFSU In coordinate systems applied to the earth, the vertical coordinate describes position in the vertical
METR 402: Atmos Dynamics I Fall 20 Isobaric Coordinates Dr. Dave Dempsey Dept. of Geosciences, SFSU In coordinate systems applied to the earth, the vertical coordinate describes position in the vertical
MME 2010 METALLURGICAL THERMODYNAMICS II. Fundamentals of Thermodynamics for Systems of Constant Composition
 MME 2010 METALLURGICAL THERMODYNAMICS II Fundamentals of Thermodynamics for Systems of Constant Composition Thermodynamics addresses two types of problems: 1- Computation of energy difference between two
MME 2010 METALLURGICAL THERMODYNAMICS II Fundamentals of Thermodynamics for Systems of Constant Composition Thermodynamics addresses two types of problems: 1- Computation of energy difference between two
Name... Class... Date... Specific heat capacity and specific latent heat
 Specific heat capacity and specific latent heat Specification references: P3.2.2 Temperature changes in a system and specific heat capacity P3.2.3 Changes of heat and specific latent heat Aims This is
Specific heat capacity and specific latent heat Specification references: P3.2.2 Temperature changes in a system and specific heat capacity P3.2.3 Changes of heat and specific latent heat Aims This is
Thermodynamics part III.
 Thermodynamics part III. a.) Fenomenological thermodynamics macroscopic description b.) Molecular thermodynamics microscopic description b1.) kinetical gas theory b2.) statistical thermodynamics Laws of
Thermodynamics part III. a.) Fenomenological thermodynamics macroscopic description b.) Molecular thermodynamics microscopic description b1.) kinetical gas theory b2.) statistical thermodynamics Laws of
Atmospheric Composition הרכב האטמוספירה
 Atmospheric Composition הרכב האטמוספירה N 2 O 2 Trace Gases Water Vapor (H 2 O) Argon (Ar) Carbon Dioxide (CO 2 ) Neon (Ne) Helium (He) Methane (CH 4 ) Nitrous Oxide (N 2 O) Ozone (O 3 ) Nitrogen and oxygen
Atmospheric Composition הרכב האטמוספירה N 2 O 2 Trace Gases Water Vapor (H 2 O) Argon (Ar) Carbon Dioxide (CO 2 ) Neon (Ne) Helium (He) Methane (CH 4 ) Nitrous Oxide (N 2 O) Ozone (O 3 ) Nitrogen and oxygen
1 Introduction to Governing Equations 2 1a Methodology... 2
 Contents 1 Introduction to Governing Equations 2 1a Methodology............................ 2 2 Equation of State 2 2a Mean and Turbulent Parts...................... 3 2b Reynolds Averaging.........................
Contents 1 Introduction to Governing Equations 2 1a Methodology............................ 2 2 Equation of State 2 2a Mean and Turbulent Parts...................... 3 2b Reynolds Averaging.........................
1. Heterogeneous Systems and Chemical Equilibrium
 1. Heterogeneous Systems and Chemical Equilibrium The preceding section involved only single phase systems. For it to be in thermodynamic equilibrium, a homogeneous system must be in thermal equilibrium
1. Heterogeneous Systems and Chemical Equilibrium The preceding section involved only single phase systems. For it to be in thermodynamic equilibrium, a homogeneous system must be in thermal equilibrium
Physics 53. Thermal Physics 1. Statistics are like a bikini. What they reveal is suggestive; what they conceal is vital.
 Physics 53 Thermal Physics 1 Statistics are like a bikini. What they reveal is suggestive; what they conceal is vital. Arthur Koestler Overview In the following sections we will treat macroscopic systems
Physics 53 Thermal Physics 1 Statistics are like a bikini. What they reveal is suggestive; what they conceal is vital. Arthur Koestler Overview In the following sections we will treat macroscopic systems
Chapter 5. On-line resource
 Chapter 5 The water-air heterogeneous system On-line resource on-line analytical system that portrays the thermodynamic properties of water vapor and many other gases http://webbook.nist.gov/chemistry/fluid/
Chapter 5 The water-air heterogeneous system On-line resource on-line analytical system that portrays the thermodynamic properties of water vapor and many other gases http://webbook.nist.gov/chemistry/fluid/
GEF2200 vår 2017 Løsningsforslag sett 1
 GEF2200 vår 2017 Løsningsforslag sett 1 A.1.T R is the universal gas constant, with value 8.3143JK 1 mol 1. R is the gas constant for a secic gas, given by R R M (1) where M is the molecular weight of
GEF2200 vår 2017 Løsningsforslag sett 1 A.1.T R is the universal gas constant, with value 8.3143JK 1 mol 1. R is the gas constant for a secic gas, given by R R M (1) where M is the molecular weight of
