GEF2200 Atmosfærefysikk 2012
|
|
|
- Kathleen Watkins
- 5 years ago
- Views:
Transcription
1 GEF2200 Atmosfærefysikk 2012 Løsningsforslag til oppgavesett 4 WH (WH 2.49) The air parcel has the properties p = 1000hPa, T = 15 C and T d = 4 C. b Lifting the air parcel to p 2 = 900hPa, T 2 we still have unsaturated air, and we also have a new dew point temperature T 2d. This means that the mixing ratio is still the same, w(t 2 ) = w(t 2d ) = w(t d ) = 5.1g/kg. By reading off the diagram, the saturation mixing ratio at T 2 is w s (T 2 ) = 6.8g/kg, giving RH = 5.1/6.8 = 0.73 = 73%. The potential temperature is given by decending along the dry adiabat, giving θ = 15 C, so it is conserved. Repeating the procedure for wetbulb temperature, but stopping at p 2, we have T w = 4.7 C. Following the moist adiabat all the way to 1000hPa, we get θ w = 9.3 C, which is conserved. c Lifting the air parcel to p 3 = 800hPa, we pass the line for constant mixing ratio (5.1g/kg), which is at the LCL, so we now have saturated air. The parcel then follows the moist adiabat in acent. At p 3 we get the new values w = w s = 4.4g/kg, that is RH = 100%. For saturated air we have T = T w = T d and here T = T w = T d = 1 C. Following the moist adiabat downwards, we see that θ w = 9.3 C is still conserved, but when following the dry adiabat we get θ = 17 C, meaning that potential temperature is not conserved for moist adiabatic processes. d As already noted, LCL = 845hPa.
2 WH r (WH m) When unsaturated air is lifted, the potential temperature, the equivalent potential temperature and the mixing ratio are conserved quantities. The saturation mixing ratio on the other hand is not. Consequently the first pair of quantities are conserved. WH s (WH n) When saturated air is lifted, the only conserved quantity out of the four is the equivalent potential temperature. The potential temperature, the mixing ratio and the saturation mixing ratio are not conserved quantities when lifting saturated air. WH (WH ) The initial dew point temperature of the air is located on the skew T lnpby cooling the air parcel along the 1000 hpa isobar from the initial temperature of the air parcel to the intersection between the 1000 hpa isobar and the w s = 10 g/kg contour. The initial dew point temperature is: T d,1000 hpa = 14 C To find the final temperature of the air parcel, follow the dry adiabat from the initial location of the air parcel (the intersection between the 1000 hpa isobar and the 15 C isotherm) to the intersection between the dry adiabat and the w s = 10 g/kg contour at the lifting condensation level (LCL). As the air parcel is now saturated, further lifting must be along the saturated adiabat until the air parcel reaches the 700 hpa isobar at the intersection between the 700 hpa isobar and the w s = 6 g/kg. Itisassumedthatonly80%ofthecondensedwaterintheairparcelfalls out of the air parcel as precipitation during the lift. As the air parcel was lifted from the LCL to the 700 hpa isobar, the saturated mixing ratio decreased by 4 g/kg from 10 g/kg to 6 g/kg. 80% of the 4 g/kg, corresponding to a mixing ratio of 3.2 g/kg, falls out of the air parcel, and 20%, corresponding to a mixing ratio of.8 g/kg, remains in the air parcel.
3 Figure 1: Plot of the radiosonde values of Exercise As the air parcel starts to sink on the other side of the mountain, the saturation mixing ratio is 6 g/kg. As the air parcel sinks, the remaining water in the air parcel evaporates until there is no water left at which point the saturation mixing ratio of the air parcel is 6.8 g/kg. Up to this point, the air parcel is saturated and sinks along the saturated adiabat. Having passed this point, the air parcel will no longer be saturated and sinks along the dry adiabat until it reaches the 900 hpa isobar. The temperature of the air parcel after descending to the 900 hpa isobar is: T 900 hpa = 19.5 C WH (WH ) a Solvetheproblemusingtheskew T lnp. First,plotthesoundingonthe skew T lnp(showninfigure1). Then, toconsiderthestaticstabilityof the layers remember that for dry/unsaturated air the following is true: Γ > Γ d : the layer is unstable, Γ = Γ d : the layer is neutral, and Γ < Γ d : the layer is stable.
4 Similarly, for saturated air the following is true: Γ > Γ s : the layer is unstable, Γ = Γ s : the layer is neutral, and Γ < Γ s : the layer is stable. AB: The layer is not saturated as the dew point temperature is smaller than the temperature. The temperature decrease following the dry adiabat from p A to p B is smaller than the actual temperature decrease between A and B. Consequently: Γ > Γ d The layer is unstable BC: The layer is not saturated, as also here the dew point temperature is smaller htan the temperature of the layer. Now, the temperature decrease following the dry adiabat is equal to the temperature decrease of the layer. Consequently: Γ = Γ d The layer is neutrally stable CD: The layer is saturated as the dew point temperature equals the temperature throughout the layer. The temperature decrease in the layer equals the temperature decrease found following the saturated adiabat. Consequently: Γ = Γ s The layer is neutrally stable DE: The layer is unsaturated as the dew point temperature is smaller the temperature throughout the layer. This layer is special compared to the other layers as the temperature indreases throughout the layer yeilding a negative lapse rate Γ. Increasing temperature with height is called an inversion. Following the dry adiabat from p D to p E the temperature is found to decrease. Consequently: Γ < Γ d The layer is stable EF: The layer is again unsaturated as the dew point temperature is smaller the temperature throughout the layer. Following the dry adiabatfromp E top F yeildsatemperaturedecreasewhichislargerthanthe temperature decrease in the layer. Consequently: Γ < Γ d
5 Obs p(hpa) T(C) T d (C) θ(k) dθ/dz w s θ e (K) dθ e /dz A < 0 < 0 B < 0 C (> 0) 0 (or > 0) D > 0 < 0 E > 0 < 0 F > 0 < 0 G Table 1: Souning of WH The layer is stable FG: The layer is again unsaturated as the dew point temperature is smaller the temperature throughout the layer. Following the dry adiabat from p F to p G again yeilds a temperature decrease which is larger than the temperature decrease in the layer. Consequently: Γ < Γ d The layer is stable Alternatively, potential temperature may be calculated from the well known equation: ( ) 1000hPa R/cp θ = T (1) p and the stability of the various layer can be found by considering the sign of θ θ z. θ values and the sign of z are given in 1. Remember that according to equation (3.77) on page 91 in Wallace & Hobbs (2006), the layer is stable when θ θ z > 0, neutral when z = 0 and unstable when θ z < 0 Thus, for dry air, layer AB is unstable, BC is neutral and the rest are stable. However, if the layer is saturated we have to consider the saturated adiabat, not the dry adiabat! This is the case for layer CD. Consequently, the stability of layer CD cannot be estimated by considering θ, we must consider the equivalent potential temperature θ e.
6 There are two ways to find θ e, but we need the skew T lnpanyway: 1. To find the θ e value of say an air parcel at point A, initially locatedattheintersectionbetweenthe1000hpaisobarandthe30.0 isotherm, follow the dry adiabat until the air parcel reaches saturation at the LCL. Subsequently, follow the saturated adiabat until all water vapor in the air parcel has condensed (that is, until the dry adiabats and saturated adiabats are parallel). Then, follow the dry adiabat back to 1000 hpa. 2. We calculate it from equation (3.71) in Wallace & Hobbs (2006): ( ) Lv w s θ e = θexp (2) c p T To do this, we read off the saturation mixing ratio of water vapor (w s )forthetemperaturet ateachpoint(w s (T),notw(T) = w s (T d ), you should know the difference). L v = Jkg 1, and c p = 1004Jkg 1 K 1. The θ e -values are given in table 1. Both point C and D are saturated (T = T d ), but the equivalent potential temperature increases with height (or is almost unchanged). Therefore, this layer is approximately neutral. b Consideringconvectiveinstability,thecriterionforinstabilityisdθ e /dz < 0. Consider the stability of each layer by finding the θ e value at each pointandconsiderthesignofdθ e /dz withinthelayer. Usingtheskew T ln pyeilds: A:θ e = 80,B:θ e = 72.5,C:θ e = 70,D:θ e = 70,E:θ e = 65,F:θ e = 58, and G: θ e = 50. As the height increases within a layer, it is the change in θ e which determines the sign of dθ e /dz < 0. In layer AB dθ e /dz < 0 decreases, consequently dθ e /dz < 0 and the layer is convectively unstable. The same is true for layers BC, DE, EF and FG. In layer CD on the other hand, θ e does not change and dθ e /dz = 0. Thus layer CD is not convectively unstable.
7 Alternatively, use the θ e values in table 1. WH (WH ) Will find the entropy for converting m = 2 g = kg of ice at T 1 = 10 C to vapor at T 1 = 100 C by heating. The entopy is given by ds = dq rev T soweneedtofindexpressionsfordq rev. Notethatwedonotusespecific valuesforq rev ands here. Theheatsuppliedtochangethetemperature is given by the first law of thermodynamics: dq temp = mc px dt wherec px isthespecificheatatconstantpressureforeithericec pi orliquid water c pv. But we also have additional heating due to phase change at temperature T phase : Q phase = ml x where L x is latent heat, which has to be considered for melting and evaporation. Integrating Equation (3) is then done in four steps: S = = = S2 S 1 ds = [ T2 T2 dq temp T 1 T 273K 263K 373K + = m 273K dq rev T 1 T ] + Q phase T phase dt mc p T +ml melt 273K mc p dt T +m L vap 373K [ c pi ln L melt 273 +c pv ln L vap 373 The constants are (given at the start of the book): c pi = 2106 Jkg 1 K 1 c pv = 4218 Jkg 1 K 1 L melt = Jkg 1 L vap = Jkg 1 ] (3)
8 giving the answer S = JK 1 ( ) = 17.3 JK 1 Clearly, the phase changes are important, and as expected, since gases are more disordered than liquids or ice, the change in entropy is much larger for liquid-gas change than for ice-liquid change.
GEF2200 atmospheric physics 2018
 GEF2200 atmospheric physics 208 Solutions: thermodynamics 3 Oppgaver hentet fra boka Wallace and Hobbs (2006) er merket WH06 WH06 3.8r Unsaturated air is lifted (adiabatically): The first pair of quantities
GEF2200 atmospheric physics 208 Solutions: thermodynamics 3 Oppgaver hentet fra boka Wallace and Hobbs (2006) er merket WH06 WH06 3.8r Unsaturated air is lifted (adiabatically): The first pair of quantities
Lecture Ch. 6. Condensed (Liquid) Water. Cloud in a Jar Demonstration. How does saturation occur? Saturation of Moist Air. Saturation of Moist Air
 Lecture Ch. 6 Saturation of moist air Relationship between humidity and dewpoint Clausius-Clapeyron equation Dewpoint Temperature Depression Isobaric cooling Moist adiabatic ascent of air Equivalent temperature
Lecture Ch. 6 Saturation of moist air Relationship between humidity and dewpoint Clausius-Clapeyron equation Dewpoint Temperature Depression Isobaric cooling Moist adiabatic ascent of air Equivalent temperature
1. Water Vapor in Air
 1. Water Vapor in Air Water appears in all three phases in the earth s atmosphere - solid, liquid and vapor - and it is one of the most important components, not only because it is essential to life, but
1. Water Vapor in Air Water appears in all three phases in the earth s atmosphere - solid, liquid and vapor - and it is one of the most important components, not only because it is essential to life, but
Chapter 4 Water Vapor
 Chapter 4 Water Vapor Chapter overview: Phases of water Vapor pressure at saturation Moisture variables o Mixing ratio, specific humidity, relative humidity, dew point temperature o Absolute vs. relative
Chapter 4 Water Vapor Chapter overview: Phases of water Vapor pressure at saturation Moisture variables o Mixing ratio, specific humidity, relative humidity, dew point temperature o Absolute vs. relative
Exam 1 (Chaps. 1-6 of the notes)
 10/12/06 ATS 541 - Atmospheric Thermodynamics and Cloud Physics 1 Exam 1 (Chaps. 1-6 of the notes) ATS 541 students: Answer all questions ATS 441 students: You may delete problem 3 or problem 5 1. [10
10/12/06 ATS 541 - Atmospheric Thermodynamics and Cloud Physics 1 Exam 1 (Chaps. 1-6 of the notes) ATS 541 students: Answer all questions ATS 441 students: You may delete problem 3 or problem 5 1. [10
ATMO 551a Fall 08. Equivalent Potential Temperature
 Equivalent Potential emperature he equivalent potential temperature, θ e, is the potential temperature that would result if all of the water in the air parcel were condensed and rained out by raising the
Equivalent Potential emperature he equivalent potential temperature, θ e, is the potential temperature that would result if all of the water in the air parcel were condensed and rained out by raising the
Thermodynamics Review [?] Entropy & thermodynamic potentials Hydrostatic equilibrium & buoyancy Stability [dry & moist adiabatic]
![Thermodynamics Review [?] Entropy & thermodynamic potentials Hydrostatic equilibrium & buoyancy Stability [dry & moist adiabatic] Thermodynamics Review [?] Entropy & thermodynamic potentials Hydrostatic equilibrium & buoyancy Stability [dry & moist adiabatic]](/thumbs/81/83694823.jpg) Thermodynamics Review [?] Entropy & thermodynamic potentials Hydrostatic equilibrium & buoyancy Stability [dry & moist adiabatic] Entropy 1. (Thermodynamics) a thermodynamic quantity that changes in a
Thermodynamics Review [?] Entropy & thermodynamic potentials Hydrostatic equilibrium & buoyancy Stability [dry & moist adiabatic] Entropy 1. (Thermodynamics) a thermodynamic quantity that changes in a
Chapter 5. Atmospheric Moisture
 Chapter 5 Atmospheric Moisture hydrologic cycle--movement of water in all forms between earth & atmosphere Humidity: amount of water vapor in air vapor pressure saturation vapor pressure absolute humidity
Chapter 5 Atmospheric Moisture hydrologic cycle--movement of water in all forms between earth & atmosphere Humidity: amount of water vapor in air vapor pressure saturation vapor pressure absolute humidity
Clouds and turbulent moist convection
 Clouds and turbulent moist convection Lecture 2: Cloud formation and Physics Caroline Muller Les Houches summer school Lectures Outline : Cloud fundamentals - global distribution, types, visualization
Clouds and turbulent moist convection Lecture 2: Cloud formation and Physics Caroline Muller Les Houches summer school Lectures Outline : Cloud fundamentals - global distribution, types, visualization
Atmospheric Dynamics: lecture 3
 Atmospheric Dynamics: lecture 3 Moist convection Dew point temperature/lapse rate/lcl Equivalent potential temperature Conditional and potential instability Thermodynamic diagram CAPE Introduction to Python
Atmospheric Dynamics: lecture 3 Moist convection Dew point temperature/lapse rate/lcl Equivalent potential temperature Conditional and potential instability Thermodynamic diagram CAPE Introduction to Python
Chapter 5 - Atmospheric Moisture
 Chapter 5 - Atmospheric Moisture Understanding Weather and Climate Aguado and Burt Water Water Vapor - water in a gaseous form, not droplets. Water can also achieve solid and liquid phases on Earth Temperature
Chapter 5 - Atmospheric Moisture Understanding Weather and Climate Aguado and Burt Water Water Vapor - water in a gaseous form, not droplets. Water can also achieve solid and liquid phases on Earth Temperature
Outline. Aim. Gas law. Pressure. Scale height Mixing Column density. Temperature Lapse rate Stability. Condensation Humidity.
 Institute of Applied Physics University of Bern Outline A planetary atmosphere consists of different gases hold to the planet by gravity The laws of thermodynamics hold structure as vertical coordinate
Institute of Applied Physics University of Bern Outline A planetary atmosphere consists of different gases hold to the planet by gravity The laws of thermodynamics hold structure as vertical coordinate
1. Static Stability. (ρ V ) d2 z (1) d 2 z. = g (2) = g (3) T T = g T (4)
 NCAR (National Center for Atmospheric Research) has an excellent resource for education called COMET-MetEd. There you can find some really great tutorials on SkewT-LogP plots: visit http://www.meted.ucar.edu/mesoprim/skewt/index.htm.
NCAR (National Center for Atmospheric Research) has an excellent resource for education called COMET-MetEd. There you can find some really great tutorials on SkewT-LogP plots: visit http://www.meted.ucar.edu/mesoprim/skewt/index.htm.
Synoptic Meteorology I: Skew-T Diagrams and Thermodynamic Properties
 Synoptic Meteorology I: Skew-T Diagrams and Thermodynamic Properties For Further Reading Most information contained within these lecture notes is drawn from Chapters 1, 2, 4, and 6 of The Use of the Skew
Synoptic Meteorology I: Skew-T Diagrams and Thermodynamic Properties For Further Reading Most information contained within these lecture notes is drawn from Chapters 1, 2, 4, and 6 of The Use of the Skew
Project 3 Convection and Atmospheric Thermodynamics
 12.818 Project 3 Convection and Atmospheric Thermodynamics Lodovica Illari 1 Background The Earth is bathed in radiation from the Sun whose intensity peaks in the visible. In order to maintain energy balance
12.818 Project 3 Convection and Atmospheric Thermodynamics Lodovica Illari 1 Background The Earth is bathed in radiation from the Sun whose intensity peaks in the visible. In order to maintain energy balance
2σ e s (r,t) = e s (T)exp( rr v ρ l T ) = exp( ) 2σ R v ρ l Tln(e/e s (T)) e s (f H2 O,r,T) = f H2 O
 Formulas/Constants, Physics/Oceanography 4510/5510 B Atmospheric Physics II N A = 6.02 10 23 molecules/mole (Avogadro s number) 1 mb = 100 Pa 1 Pa = 1 N/m 2 Γ d = 9.8 o C/km (dry adiabatic lapse rate)
Formulas/Constants, Physics/Oceanography 4510/5510 B Atmospheric Physics II N A = 6.02 10 23 molecules/mole (Avogadro s number) 1 mb = 100 Pa 1 Pa = 1 N/m 2 Γ d = 9.8 o C/km (dry adiabatic lapse rate)
Clouds and atmospheric convection
 Clouds and atmospheric convection Caroline Muller CNRS/Laboratoire de Météorologie Dynamique (LMD) Département de Géosciences ENS M2 P7/ IPGP 1 What are clouds? Clouds and atmospheric convection 3 What
Clouds and atmospheric convection Caroline Muller CNRS/Laboratoire de Météorologie Dynamique (LMD) Département de Géosciences ENS M2 P7/ IPGP 1 What are clouds? Clouds and atmospheric convection 3 What
1., annual precipitation is greater than annual evapotranspiration. a. On the ocean *b. On the continents
 CHAPTER 6 HUMIDITY, SATURATION, AND STABILITY MULTIPLE CHOICE QUESTIONS 1., annual precipitation is greater than annual evapotranspiration. a. On the ocean *b. On the continents 2., annual precipitation
CHAPTER 6 HUMIDITY, SATURATION, AND STABILITY MULTIPLE CHOICE QUESTIONS 1., annual precipitation is greater than annual evapotranspiration. a. On the ocean *b. On the continents 2., annual precipitation
Introduction. Lecture 6: Water in Atmosphere. How Much Heat Is Brought Upward By Water Vapor?
 Lecture 6: Water in Atmosphere Introduction Over 70% of the planet is covered by water Water is unique in that it can simultaneously exist in all three states (solid, liquid, gas) at the same temperature
Lecture 6: Water in Atmosphere Introduction Over 70% of the planet is covered by water Water is unique in that it can simultaneously exist in all three states (solid, liquid, gas) at the same temperature
Answers to Clicker Questions
 Answers to Clicker Questions Chapter 1 What component of the atmosphere is most important to weather? A. Nitrogen B. Oxygen C. Carbon dioxide D. Ozone E. Water What location would have the lowest surface
Answers to Clicker Questions Chapter 1 What component of the atmosphere is most important to weather? A. Nitrogen B. Oxygen C. Carbon dioxide D. Ozone E. Water What location would have the lowest surface
( ) = 1005 J kg 1 K 1 ;
 Problem Set 3 1. A parcel of water is added to the ocean surface that is denser (heavier) than any of the waters in the ocean. Suppose the parcel sinks to the ocean bottom; estimate the change in temperature
Problem Set 3 1. A parcel of water is added to the ocean surface that is denser (heavier) than any of the waters in the ocean. Suppose the parcel sinks to the ocean bottom; estimate the change in temperature
Temperature and Thermodynamics, Part II. Topics to be Covered
 Teperature and Therodynaics, Part II Topics to be Covered Profiles of Teperature in the Boundary Layer Potential teperature Adiabatic Lapse Rate Theral Stratification 1/8/17 Why are We Interested in Theral
Teperature and Therodynaics, Part II Topics to be Covered Profiles of Teperature in the Boundary Layer Potential teperature Adiabatic Lapse Rate Theral Stratification 1/8/17 Why are We Interested in Theral
Simplified Microphysics. condensation evaporation. evaporation
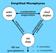 Simplified Microphysics water vapor condensation evaporation cloud droplets evaporation condensation collection rain drops fall out (precipitation) = 0 (reversible) = (irreversible) Simplified Microphysics
Simplified Microphysics water vapor condensation evaporation cloud droplets evaporation condensation collection rain drops fall out (precipitation) = 0 (reversible) = (irreversible) Simplified Microphysics
Atmospheric Composition הרכב האטמוספירה
 Atmospheric Composition הרכב האטמוספירה N 2 O 2 Trace Gases Water Vapor (H 2 O) Argon (Ar) Carbon Dioxide (CO 2 ) Neon (Ne) Helium (He) Methane (CH 4 ) Nitrous Oxide (N 2 O) Ozone (O 3 ) Nitrogen and oxygen
Atmospheric Composition הרכב האטמוספירה N 2 O 2 Trace Gases Water Vapor (H 2 O) Argon (Ar) Carbon Dioxide (CO 2 ) Neon (Ne) Helium (He) Methane (CH 4 ) Nitrous Oxide (N 2 O) Ozone (O 3 ) Nitrogen and oxygen
ATS 351, Spring 2010 Lab #9 Weather Radar - 55 points
 ATS 351, Spring 2010 Lab #9 Weather Radar - 55 points 1. (5 points) If a radar has a maximum unambiguous range of 300km, what is its PRF? (The speed of light, c, is equal to 3x10 8 m/s) 2. (5 points) Explain
ATS 351, Spring 2010 Lab #9 Weather Radar - 55 points 1. (5 points) If a radar has a maximum unambiguous range of 300km, what is its PRF? (The speed of light, c, is equal to 3x10 8 m/s) 2. (5 points) Explain
SEVERE AND UNUSUAL WEATHER
 SEVERE AND UNUSUAL WEATHER Basic Meteorological Terminology Adiabatic - Referring to a process without the addition or removal of heat. A temperature change may come about as a result of a change in the
SEVERE AND UNUSUAL WEATHER Basic Meteorological Terminology Adiabatic - Referring to a process without the addition or removal of heat. A temperature change may come about as a result of a change in the
Orographic Precipitation II: Effects of Phase Change on Orographic Flow. Richard Rotunno. National Center for Atmospheric Research, USA
 Orographic Precipitation II: Effects of Phase Change on Orographic Flow Richard Rotunno National Center for Atmospheric Research, USA Condensation D Dt vs w( x, ) vs U Large-Scale Flow 0 H L Dynamics w
Orographic Precipitation II: Effects of Phase Change on Orographic Flow Richard Rotunno National Center for Atmospheric Research, USA Condensation D Dt vs w( x, ) vs U Large-Scale Flow 0 H L Dynamics w
ATMO 551a Moist Adiabat Fall Change in internal energy: ΔU
 Enthalpy and the Moist Adiabat We have described the dry adiabat where an air parcel is lifted rapidly causing the air parcel to expand as the environmental pressure decreases and the air parcel does work
Enthalpy and the Moist Adiabat We have described the dry adiabat where an air parcel is lifted rapidly causing the air parcel to expand as the environmental pressure decreases and the air parcel does work
Radiative equilibrium Some thermodynamics review Radiative-convective equilibrium. Goal: Develop a 1D description of the [tropical] atmosphere
![Radiative equilibrium Some thermodynamics review Radiative-convective equilibrium. Goal: Develop a 1D description of the [tropical] atmosphere Radiative equilibrium Some thermodynamics review Radiative-convective equilibrium. Goal: Develop a 1D description of the [tropical] atmosphere](/thumbs/72/66535634.jpg) Radiative equilibrium Some thermodynamics review Radiative-convective equilibrium Goal: Develop a 1D description of the [tropical] atmosphere Vertical temperature profile Total atmospheric mass: ~5.15x10
Radiative equilibrium Some thermodynamics review Radiative-convective equilibrium Goal: Develop a 1D description of the [tropical] atmosphere Vertical temperature profile Total atmospheric mass: ~5.15x10
EAS270, The Atmosphere 2 nd Mid-term Exam 2 Nov. 2016
 EAS270, The Atmosphere 2 nd Mid-term Exam 2 Nov. 2016 Professor: J.D. Wilson Time available: 50 mins Value: 25% No formula sheets; no use of tablet computers etc. or cell phones. Formulae/data at back.
EAS270, The Atmosphere 2 nd Mid-term Exam 2 Nov. 2016 Professor: J.D. Wilson Time available: 50 mins Value: 25% No formula sheets; no use of tablet computers etc. or cell phones. Formulae/data at back.
ATS 351, Spring 2010 Lab #9 Weather Radar - 55 points
 ATS 351, Spring 2010 Lab #9 Weather Radar - 55 points 1. (5 points) If a radar has a maximum unambiguous range of 300km, what is its PRF? (The speed of light, c, is equal to 3x10 8 m/s) The equation to
ATS 351, Spring 2010 Lab #9 Weather Radar - 55 points 1. (5 points) If a radar has a maximum unambiguous range of 300km, what is its PRF? (The speed of light, c, is equal to 3x10 8 m/s) The equation to
Today s Lecture: Atmosphere finish primitive equations, mostly thermodynamics
 Today s Lecture: Atmosphere finish primitive equations, mostly thermodynamics Reference Peixoto and Oort, Sec. 3.1, 3.2, 3.4, 3.5 (but skip the discussion of oceans until next week); Ch. 10 Thermodynamic
Today s Lecture: Atmosphere finish primitive equations, mostly thermodynamics Reference Peixoto and Oort, Sec. 3.1, 3.2, 3.4, 3.5 (but skip the discussion of oceans until next week); Ch. 10 Thermodynamic
PHYSICAL GEOGRAPHY. By Brett Lucas
 PHYSICAL GEOGRAPHY By Brett Lucas INTRODUCTION TO ATMOSPHERIC MOISTURE Atmospheric Moisture The Nature of Water The Hydrologic Cycle Evaporation Measures of Humidity Condensation The Buoyancy of Air Precipitation
PHYSICAL GEOGRAPHY By Brett Lucas INTRODUCTION TO ATMOSPHERIC MOISTURE Atmospheric Moisture The Nature of Water The Hydrologic Cycle Evaporation Measures of Humidity Condensation The Buoyancy of Air Precipitation
Isentropic Analysis. Much of this presentation is due to Jim Moore, SLU
 Isentropic Analysis Much of this presentation is due to Jim Moore, SLU Utility of Isentropic Analysis Diagnose and visualize vertical motion - through advection of pressure and system-relative flow Depict
Isentropic Analysis Much of this presentation is due to Jim Moore, SLU Utility of Isentropic Analysis Diagnose and visualize vertical motion - through advection of pressure and system-relative flow Depict
Boundary layer equilibrium [2005] over tropical oceans
![Boundary layer equilibrium [2005] over tropical oceans Boundary layer equilibrium [2005] over tropical oceans](/thumbs/96/128963638.jpg) Boundary layer equilibrium [2005] over tropical oceans Alan K. Betts [akbetts@aol.com] Based on: Betts, A.K., 1997: Trade Cumulus: Observations and Modeling. Chapter 4 (pp 99-126) in The Physics and Parameterization
Boundary layer equilibrium [2005] over tropical oceans Alan K. Betts [akbetts@aol.com] Based on: Betts, A.K., 1997: Trade Cumulus: Observations and Modeling. Chapter 4 (pp 99-126) in The Physics and Parameterization
First Law of Thermodynamics
 First Law of Thermodynamics E int = Q + W other state variables E int is a state variable, so only depends on condition (P, V, T, ) of system. Therefore, E int only depends on initial and final states
First Law of Thermodynamics E int = Q + W other state variables E int is a state variable, so only depends on condition (P, V, T, ) of system. Therefore, E int only depends on initial and final states
Handout 12: Thermodynamics. Zeroth law of thermodynamics
 1 Handout 12: Thermodynamics Zeroth law of thermodynamics When two objects with different temperature are brought into contact, heat flows from the hotter body to a cooler one Heat flows until the temperatures
1 Handout 12: Thermodynamics Zeroth law of thermodynamics When two objects with different temperature are brought into contact, heat flows from the hotter body to a cooler one Heat flows until the temperatures
CAE 331/513 Building Science Fall 2015
 CAE 331/513 Building Science Fall 2015 Week 5: September 24, 2015 Psychrometrics (equations) Advancing energy, environmental, and sustainability research within the built environment www.built-envi.com
CAE 331/513 Building Science Fall 2015 Week 5: September 24, 2015 Psychrometrics (equations) Advancing energy, environmental, and sustainability research within the built environment www.built-envi.com
Entropy Changes & Processes
 Entropy Changes & Processes Chapter 4 of Atkins: he Second Law: he Concepts Section 4.3 Entropy of Phase ransition at the ransition emperature Expansion of the Perfect Gas Variation of Entropy with emperature
Entropy Changes & Processes Chapter 4 of Atkins: he Second Law: he Concepts Section 4.3 Entropy of Phase ransition at the ransition emperature Expansion of the Perfect Gas Variation of Entropy with emperature
4 WATER VAPOR. Contents 4.1. VAPOR PRESSURE AT SATURATION
 Copyright 2017 by Roland Stull. Practical Meteorology: An Algebra-based Survey of Atmospheric Science. v1.02b 4 WATER VAPOR Contents 4.1. Vapor Pressure at Saturation 87 HIGHER MATH Clausius-Clapeyron
Copyright 2017 by Roland Stull. Practical Meteorology: An Algebra-based Survey of Atmospheric Science. v1.02b 4 WATER VAPOR Contents 4.1. Vapor Pressure at Saturation 87 HIGHER MATH Clausius-Clapeyron
Q. Deng a, NYU Abu Dhabi G. Hernandez-Duenas b, UNAM A. Majda, Courant S. Stechmann, UW-Madison L.M. Smith, UW-Madison
 Minimal Models for Precipitating Turbulent Convection Q. Deng a, NYU Abu Dhabi G. Hernandez-Duenas b, UNAM A. Majda, Courant S. Stechmann, UW-Madison L.M. Smith, UW-Madison Contours of rain water in a
Minimal Models for Precipitating Turbulent Convection Q. Deng a, NYU Abu Dhabi G. Hernandez-Duenas b, UNAM A. Majda, Courant S. Stechmann, UW-Madison L.M. Smith, UW-Madison Contours of rain water in a
Lecture 07 February 10, 2010 Water in the Atmosphere: Part 1
 Lecture 07 February 10, 2010 Water in the Atmosphere: Part 1 About Water on the Earth: The Hydrological Cycle Review 3-states of water, phase change and Latent Heat Indices of Water Vapor Content in the
Lecture 07 February 10, 2010 Water in the Atmosphere: Part 1 About Water on the Earth: The Hydrological Cycle Review 3-states of water, phase change and Latent Heat Indices of Water Vapor Content in the
Parcel Model. Meteorology September 3, 2008
 Parcel Model Meteorology 5210 September 3, 2008 1 Governing Equations for Precipitating Convection For precipitating convection, we have the following set of equations for potential temperature, θ, mixing
Parcel Model Meteorology 5210 September 3, 2008 1 Governing Equations for Precipitating Convection For precipitating convection, we have the following set of equations for potential temperature, θ, mixing
First Law of Thermodynamics
 First Law of Thermodynamics September 11, 2013 The first law of thermodynamics is the conservation of energy applied to thermal systems. Here, we develop the principles of thermodynamics for a discrete
First Law of Thermodynamics September 11, 2013 The first law of thermodynamics is the conservation of energy applied to thermal systems. Here, we develop the principles of thermodynamics for a discrete
Chapter 6. Cloud Development and Forms
 Cloud Formation Chapter 6 Cloud Development and Forms Condensation (i.e. clouds,fog) results from: Diabatic cooling (important for fog) Adiabatic cooling (important for clouds) Clouds form due to adiabatic
Cloud Formation Chapter 6 Cloud Development and Forms Condensation (i.e. clouds,fog) results from: Diabatic cooling (important for fog) Adiabatic cooling (important for clouds) Clouds form due to adiabatic
Lecture 3: Convection
 EESC V2100 The Climate System spring 2004 Lecture 3: Convection Yochanan Kushnir Lamont Doherty Earth Observatory of Columbia University Palisades, NY 10964, USA kushnir@ldeo.columbia.edu Layers of the
EESC V2100 The Climate System spring 2004 Lecture 3: Convection Yochanan Kushnir Lamont Doherty Earth Observatory of Columbia University Palisades, NY 10964, USA kushnir@ldeo.columbia.edu Layers of the
39th International Physics Olympiad - Hanoi - Vietnam Theoretical Problem No. 3 / Solution. Solution
 Solution. For an altitude change dz, the atmospheric pressure change is : dp = ρgdz () where g is the acceleration of gravity, considered constant, ρ is the specific mass of air, which is considered as
Solution. For an altitude change dz, the atmospheric pressure change is : dp = ρgdz () where g is the acceleration of gravity, considered constant, ρ is the specific mass of air, which is considered as
CAE 331/513 Building Science Fall 2017
 CAE 331/513 Building Science Fall 2017 October 5, 2017 Psychrometrics (equations) Advancing energy, environmental, and sustainability research within the built environment www.built-envi.com Twitter: @built_envi
CAE 331/513 Building Science Fall 2017 October 5, 2017 Psychrometrics (equations) Advancing energy, environmental, and sustainability research within the built environment www.built-envi.com Twitter: @built_envi
Version 001 HW 15 Thermodynamics C&J sizemore (21301jtsizemore) 1
 Version 001 HW 15 Thermodynamics C&J sizemore 21301jtsizemore 1 This print-out should have 38 questions. Multiple-choice questions may continue on the next column or page find all choices before answering.
Version 001 HW 15 Thermodynamics C&J sizemore 21301jtsizemore 1 This print-out should have 38 questions. Multiple-choice questions may continue on the next column or page find all choices before answering.
GEF2200 vår 2017 Løsningsforslag sett 1
 GEF2200 vår 2017 Løsningsforslag sett 1 A.1.T R is the universal gas constant, with value 8.3143JK 1 mol 1. R is the gas constant for a secic gas, given by R R M (1) where M is the molecular weight of
GEF2200 vår 2017 Løsningsforslag sett 1 A.1.T R is the universal gas constant, with value 8.3143JK 1 mol 1. R is the gas constant for a secic gas, given by R R M (1) where M is the molecular weight of
EAS270, The Atmosphere Mid-term Exam 28 Oct. 2011
 EAS270, The Atmosphere Mid-term Exam 28 Oct. 2011 Professor: J.D. Wilson Time available: 50 mins Value: 20% Instructions: For each of the 30 multi-choice questions, choose the most logical option. Use
EAS270, The Atmosphere Mid-term Exam 28 Oct. 2011 Professor: J.D. Wilson Time available: 50 mins Value: 20% Instructions: For each of the 30 multi-choice questions, choose the most logical option. Use
Løsningsforslag: oppgavesett kap. 9 (2 av 3) GEF2200
 Løsningsforslag: oppgavesett kap. 9 (2 av 3) GEF2200 s.m.blichner@geo.uio.no Oppgave 1 a) The turbulent vertical flux of sensible heat (Q H ) in the atmospheric boundary layer often takes place through
Løsningsforslag: oppgavesett kap. 9 (2 av 3) GEF2200 s.m.blichner@geo.uio.no Oppgave 1 a) The turbulent vertical flux of sensible heat (Q H ) in the atmospheric boundary layer often takes place through
T ice T water T water = T ice =0 0 C. e =1
 Given 1 kg of water at 100 0 C and a very large (very very large) block of ice at 0 0 C. A reversible heat engine absorbs heat from the water and expels heat to the ice until work can no longer be extracted
Given 1 kg of water at 100 0 C and a very large (very very large) block of ice at 0 0 C. A reversible heat engine absorbs heat from the water and expels heat to the ice until work can no longer be extracted
Weather, Atmosphere and Meteorology
 S c i e n c e s Weather, Atmosphere and Meteorology Key words: Atmosphere, Ozone, Water vapor, solar radiation, Condensation, Evaporation, Humidity, Dew-Point Temperature, Cirrus Clouds, Stratus Clouds,
S c i e n c e s Weather, Atmosphere and Meteorology Key words: Atmosphere, Ozone, Water vapor, solar radiation, Condensation, Evaporation, Humidity, Dew-Point Temperature, Cirrus Clouds, Stratus Clouds,
Final Examination. Part A Answer ONLY TWELVE QUESTIONS in Part A. (Each question is 3 points)
 ATS 210 Spring Term 2001 NAME: Final Examination This is a 2 hour, closed-book examination. Calculators may be used. All answers should be written on the examination paper. Use the final sheet for any
ATS 210 Spring Term 2001 NAME: Final Examination This is a 2 hour, closed-book examination. Calculators may be used. All answers should be written on the examination paper. Use the final sheet for any
Moist Convection. Chapter 6
 Moist Convection Chapter 6 1 2 Trade Cumuli Afternoon cumulus over land 3 Cumuls congestus Convectively-driven weather systems Deep convection plays an important role in the dynamics of tropical weather
Moist Convection Chapter 6 1 2 Trade Cumuli Afternoon cumulus over land 3 Cumuls congestus Convectively-driven weather systems Deep convection plays an important role in the dynamics of tropical weather
5 Atmospheric Stability
 5 Atmospheric Stability Introduction In Chapter 4, we examined the concept of horizontal and vertical atmospheric motion, and how pressure differences create the winds that form on a variety of different
5 Atmospheric Stability Introduction In Chapter 4, we examined the concept of horizontal and vertical atmospheric motion, and how pressure differences create the winds that form on a variety of different
AT351 Lab Seven Skew-T Stability Analysis
 AT351 Lab Seven Skew-T Stability Analysis Twice a day, all around the planet, small instrument packages called radiosondes are launched into the atmosphere using balloons. These instruments record the
AT351 Lab Seven Skew-T Stability Analysis Twice a day, all around the planet, small instrument packages called radiosondes are launched into the atmosphere using balloons. These instruments record the
Moisture, Clouds, and Precipitation Earth Science, 13e Chapter 17
 Moisture, Clouds, and Precipitation Earth Science, 13e Chapter 17 Stanley C. Hatfield Southwestern Illinois College Changes of state of water, H 2 O Water is the only substance in atmosphere that exists
Moisture, Clouds, and Precipitation Earth Science, 13e Chapter 17 Stanley C. Hatfield Southwestern Illinois College Changes of state of water, H 2 O Water is the only substance in atmosphere that exists
Phase Changes and Latent Heat
 Review Questions Why can a person remove a piece of dry aluminum foil from a hot oven with bare fingers without getting burned, yet will be burned doing so if the foil is wet. Equal quantities of alcohol
Review Questions Why can a person remove a piece of dry aluminum foil from a hot oven with bare fingers without getting burned, yet will be burned doing so if the foil is wet. Equal quantities of alcohol
Numerical Example An air parcel with mass of 1 kg rises adiabatically from sea level to an altitude of 3 km. What is its temperature change?
 Numerical Example An air parcel with mass of 1 kg rises adiabatically from sea level to an altitude of 3 km. What is its temperature change? From the 1 st law, T = -g/c p z + Q/m air /c p Here, Q = 0,
Numerical Example An air parcel with mass of 1 kg rises adiabatically from sea level to an altitude of 3 km. What is its temperature change? From the 1 st law, T = -g/c p z + Q/m air /c p Here, Q = 0,
AP PHYSICS 2 WHS-CH-15 Thermodynamics Show all your work, equations used, and box in your answers!
 AP PHYSICS 2 WHS-CH-15 Thermodynamics Show all your work, equations used, and box in your answers! Nicolas Léonard Sadi Carnot (1796-1832) Sadi Carnot was a French military engineer and physicist, often
AP PHYSICS 2 WHS-CH-15 Thermodynamics Show all your work, equations used, and box in your answers! Nicolas Léonard Sadi Carnot (1796-1832) Sadi Carnot was a French military engineer and physicist, often
Chapter The transition from water vapor to liquid water is called. a. condensation b. evaporation c. sublimation d.
 Chapter-6 Multiple Choice Questions 1. The transition from water vapor to liquid water is called. a. condensation b. evaporation c. sublimation d. deposition 2. The movement of water among the great global
Chapter-6 Multiple Choice Questions 1. The transition from water vapor to liquid water is called. a. condensation b. evaporation c. sublimation d. deposition 2. The movement of water among the great global
1. Heterogeneous Systems and Chemical Equilibrium
 1. Heterogeneous Systems and Chemical Equilibrium The preceding section involved only single phase systems. For it to be in thermodynamic equilibrium, a homogeneous system must be in thermal equilibrium
1. Heterogeneous Systems and Chemical Equilibrium The preceding section involved only single phase systems. For it to be in thermodynamic equilibrium, a homogeneous system must be in thermal equilibrium
Handout 12: Thermodynamics. Zeroth law of thermodynamics
 1 Handout 12: Thermodynamics Zeroth law of thermodynamics When two objects with different temperature are brought into contact, heat flows from the hotter body to a cooler one Heat flows until the temperatures
1 Handout 12: Thermodynamics Zeroth law of thermodynamics When two objects with different temperature are brought into contact, heat flows from the hotter body to a cooler one Heat flows until the temperatures
10.2 PROCESSES 10.3 THE SECOND LAW OF THERMO/ENTROPY Student Notes
 10.2 PROCESSES 10.3 THE SECOND LAW OF THERMO/ENTROPY Student Notes I. THE FIRST LAW OF THERMODYNAMICS A. SYSTEMS AND SURROUNDING B. PV DIAGRAMS AND WORK DONE V -1 Source: Physics for the IB Diploma Study
10.2 PROCESSES 10.3 THE SECOND LAW OF THERMO/ENTROPY Student Notes I. THE FIRST LAW OF THERMODYNAMICS A. SYSTEMS AND SURROUNDING B. PV DIAGRAMS AND WORK DONE V -1 Source: Physics for the IB Diploma Study
Thermodynamics I Chapter 2 Properties of Pure Substances
 Thermodynamics I Chapter 2 Properties of Pure Substances Mohsin Mohd Sies Fakulti Kejuruteraan Mekanikal, Universiti Teknologi Malaysia Properties of Pure Substances (Motivation) To quantify the changes
Thermodynamics I Chapter 2 Properties of Pure Substances Mohsin Mohd Sies Fakulti Kejuruteraan Mekanikal, Universiti Teknologi Malaysia Properties of Pure Substances (Motivation) To quantify the changes
S = S(f) S(i) dq rev /T. ds = dq rev /T
 In 1855, Clausius proved the following (it is actually a corollary to Clausius Theorem ): If a system changes between two equilibrium states, i and f, the integral dq rev /T is the same for any reversible
In 1855, Clausius proved the following (it is actually a corollary to Clausius Theorem ): If a system changes between two equilibrium states, i and f, the integral dq rev /T is the same for any reversible
I. Convective Available Potential Energy (CAPE)
 Calculating CAPE, Lifted Index and Strength of Maximum Convective Updraft CAPE Calculation Lifted Index Calculation Maximum Updraft Strength Calculation I. Convective Available Potential Energy (CAPE)
Calculating CAPE, Lifted Index and Strength of Maximum Convective Updraft CAPE Calculation Lifted Index Calculation Maximum Updraft Strength Calculation I. Convective Available Potential Energy (CAPE)
Air stability. About. Precipitation. air in unstable equilibrium will move--up/down Fig. 5-1, p.112. Adiabatic = w/ no exchange of heat from outside!
 Air stability About clouds Precipitation A mass of moist, stable air gliding up and over these mountains condenses into lenticular clouds. Fig. 5-CO, p.110 air in unstable equilibrium will move--up/down
Air stability About clouds Precipitation A mass of moist, stable air gliding up and over these mountains condenses into lenticular clouds. Fig. 5-CO, p.110 air in unstable equilibrium will move--up/down
Atmospheric Basics Atmospheric Composition
 Atmospheric Basics Atmospheric Composition Air is a combination of many gases, each with its own unique characteristics. About 99 percent of the atmosphere is composed of nitrogen and oxygen, with the
Atmospheric Basics Atmospheric Composition Air is a combination of many gases, each with its own unique characteristics. About 99 percent of the atmosphere is composed of nitrogen and oxygen, with the
First Law of Thermodyamics U = Q + W. We can rewrite this by introducing two physical. Enthalpy, H, is the quantity U + pv, as this
 First Law of Thermodyamics U = Q + W where U is the increase in internal energy of the system, Q is the heat supplied to the system and W is the work done on the system. We can rewrite this by introducing
First Law of Thermodyamics U = Q + W where U is the increase in internal energy of the system, Q is the heat supplied to the system and W is the work done on the system. We can rewrite this by introducing
2.1 Effects of a cumulus ensemble upon the large scale temperature and moisture fields by induced subsidence and detrainment
 Atmospheric Sciences 6150 Cloud System Modeling 2.1 Effects of a cumulus ensemble upon the large scale temperature and moisture fields by induced subsidence and detrainment Arakawa (1969, 1972), W. Gray
Atmospheric Sciences 6150 Cloud System Modeling 2.1 Effects of a cumulus ensemble upon the large scale temperature and moisture fields by induced subsidence and detrainment Arakawa (1969, 1972), W. Gray
LAB 3: Atmospheric Pressure & Moisture
 Name School LAB 3: Atmospheric Pressure & Moisture Our atmosphere is a very dynamic area especially when we see what type of interactions it has with the surrounding environment. This lab will begin discussing
Name School LAB 3: Atmospheric Pressure & Moisture Our atmosphere is a very dynamic area especially when we see what type of interactions it has with the surrounding environment. This lab will begin discussing
The Atmosphere EVPP 110 Lecture Fall 2003 Dr. Largen
 1 Physical Environment: EVPP 110 Lecture Fall 2003 Dr. Largen 2 Physical Environment: Atmosphere Composition Heat transfer Atmospheric moisture Atmospheric circulation Weather and climate 3 Physical Environment:
1 Physical Environment: EVPP 110 Lecture Fall 2003 Dr. Largen 2 Physical Environment: Atmosphere Composition Heat transfer Atmospheric moisture Atmospheric circulation Weather and climate 3 Physical Environment:
The atmosphere s water
 The atmosphere s water Atmospheric Moisture and Precipitation Properties of Water The Hydrosphere and the Hydrologic Cycle Humidity The Adiabatic Process Clouds Precipitation Air Quality Main points for
The atmosphere s water Atmospheric Moisture and Precipitation Properties of Water The Hydrosphere and the Hydrologic Cycle Humidity The Adiabatic Process Clouds Precipitation Air Quality Main points for
1. Second Law of Thermodynamics
 1. Second Law of hermodynamics he first law describes how the state of a system changes in response to work it performs and heat absorbed. However, the first law cannot explain certain facts about thermal
1. Second Law of hermodynamics he first law describes how the state of a system changes in response to work it performs and heat absorbed. However, the first law cannot explain certain facts about thermal
Entropy Changes & Processes
 Entropy Changes & Processes Chapter 4 of Atkins: he Second Law: he Concepts Section 4.3, 7th edition; 3.3, 8th and 9th editions Entropy of Phase ransition at the ransition emperature Expansion of the Perfect
Entropy Changes & Processes Chapter 4 of Atkins: he Second Law: he Concepts Section 4.3, 7th edition; 3.3, 8th and 9th editions Entropy of Phase ransition at the ransition emperature Expansion of the Perfect
Honors Physics. Notes Nov 16, 20 Heat. Persans 1
 Honors Physics Notes Nov 16, 20 Heat Persans 1 Properties of solids Persans 2 Persans 3 Vibrations of atoms in crystalline solids Assuming only nearest neighbor interactions (+Hooke's law) F = C( u! u
Honors Physics Notes Nov 16, 20 Heat Persans 1 Properties of solids Persans 2 Persans 3 Vibrations of atoms in crystalline solids Assuming only nearest neighbor interactions (+Hooke's law) F = C( u! u
P sat = A exp [B( 1/ /T)] B= 5308K. A=6.11 mbar=vapor press. 0C.
![P sat = A exp [B( 1/ /T)] B= 5308K. A=6.11 mbar=vapor press. 0C. P sat = A exp [B( 1/ /T)] B= 5308K. A=6.11 mbar=vapor press. 0C.](/thumbs/79/79184286.jpg) Lecture 5. Water and water vapor in the atmosphere 14 Feb 2008 Review of buoyancy, with an unusual demonstration of Archimedes principle. Water is a polar molecule that forms hydrogen bonds. Consequently
Lecture 5. Water and water vapor in the atmosphere 14 Feb 2008 Review of buoyancy, with an unusual demonstration of Archimedes principle. Water is a polar molecule that forms hydrogen bonds. Consequently
Parcel Model. Atmospheric Sciences September 30, 2012
 Parcel Model Atmospheric Sciences 6150 September 30, 2012 1 Governing Equations for Precipitating Convection For precipitating convection, we have the following set of equations for potential temperature,
Parcel Model Atmospheric Sciences 6150 September 30, 2012 1 Governing Equations for Precipitating Convection For precipitating convection, we have the following set of equations for potential temperature,
Diabatic Processes. Diabatic processes are non-adiabatic processes such as. entrainment and mixing. radiative heating or cooling
 Diabatic Processes Diabatic processes are non-adiabatic processes such as precipitation fall-out entrainment and mixing radiative heating or cooling Parcel Model dθ dt dw dt dl dt dr dt = L c p π (C E
Diabatic Processes Diabatic processes are non-adiabatic processes such as precipitation fall-out entrainment and mixing radiative heating or cooling Parcel Model dθ dt dw dt dl dt dr dt = L c p π (C E
Summarizing, Key Point: An irreversible process is either spontaneous (ΔS universe > 0) or does not occur (ΔS universe < 0)
 Summarizing, Key Point: An irreversible process is either spontaneous (ΔS universe > 0) or does not occur (ΔS universe < 0) Key Point: ΔS universe allows us to distinguish between reversible and irreversible
Summarizing, Key Point: An irreversible process is either spontaneous (ΔS universe > 0) or does not occur (ΔS universe < 0) Key Point: ΔS universe allows us to distinguish between reversible and irreversible
PALM - Cloud Physics. Contents. PALM group. last update: Monday 21 st September, 2015
 PALM - Cloud Physics PALM group Institute of Meteorology and Climatology, Leibniz Universität Hannover last update: Monday 21 st September, 2015 PALM group PALM Seminar 1 / 16 Contents Motivation Approach
PALM - Cloud Physics PALM group Institute of Meteorology and Climatology, Leibniz Universität Hannover last update: Monday 21 st September, 2015 PALM group PALM Seminar 1 / 16 Contents Motivation Approach
Thermodynamics We Can See! Adapted from ATS 541 notes (Dr. Susan van den Heever)
 Thermodynamics We Can See! Adapted from ATS 541 notes (Dr. Susan van den Heever) What we have learned so far ~ Moist Adiabatic Latent Heating ρ ρ Condensation ~ Dry Adiabatic Cloud Types Skew- T log- P
Thermodynamics We Can See! Adapted from ATS 541 notes (Dr. Susan van den Heever) What we have learned so far ~ Moist Adiabatic Latent Heating ρ ρ Condensation ~ Dry Adiabatic Cloud Types Skew- T log- P
Thermal Energy. Practice Quiz Solutions
 Thermal Energy Practice Quiz Solutions What is thermal energy? What is thermal energy? Thermal energy is the energy that comes from heat. This heat is generated by the movement of tiny particles within
Thermal Energy Practice Quiz Solutions What is thermal energy? What is thermal energy? Thermal energy is the energy that comes from heat. This heat is generated by the movement of tiny particles within
Topic 3 &10 Review Thermodynamics
 Name: Date: Topic 3 &10 Review Thermodynamics 1. The kelvin temperature of an object is a measure of A. the total energy of the molecules of the object. B. the total kinetic energy of the molecules of
Name: Date: Topic 3 &10 Review Thermodynamics 1. The kelvin temperature of an object is a measure of A. the total energy of the molecules of the object. B. the total kinetic energy of the molecules of
Phase Diagrams. Department of Mechanical Engineering Indian Institute of Technology Kanpur Kanpur India
 Phase Diagrams 1 Increasing the temperature isobarically T-v diagram of constant-pressure phase-change processes of a pure substance at various pressures numerical values are for water. 2 Temperature -
Phase Diagrams 1 Increasing the temperature isobarically T-v diagram of constant-pressure phase-change processes of a pure substance at various pressures numerical values are for water. 2 Temperature -
Lecture Outlines PowerPoint. Chapter 17 Earth Science 11e Tarbuck/Lutgens
 Lecture Outlines PowerPoint Chapter 17 Earth Science 11e Tarbuck/Lutgens 2006 Pearson Prentice Hall This work is protected by United States copyright laws and is provided solely for the use of instructors
Lecture Outlines PowerPoint Chapter 17 Earth Science 11e Tarbuck/Lutgens 2006 Pearson Prentice Hall This work is protected by United States copyright laws and is provided solely for the use of instructors
CHAPTER 17 WORK, HEAT, & FIRST LAW OF THERMODYNAMICS
 CHAPTER 17 WORK, HEAT, and the FIRST LAW OF THERMODYNAMICS In this chapter, we will examine various thermal properties of matter, as well as several mechanisms by which energy can be transferred to and
CHAPTER 17 WORK, HEAT, and the FIRST LAW OF THERMODYNAMICS In this chapter, we will examine various thermal properties of matter, as well as several mechanisms by which energy can be transferred to and
Evaluating properties of pure substances
 Evaluating properties of pure substances Pure substance A pure substance has the same chemical composition throughout. Are the following confined in a fixed volume pure substances: Ice (single component,
Evaluating properties of pure substances Pure substance A pure substance has the same chemical composition throughout. Are the following confined in a fixed volume pure substances: Ice (single component,
EARTH SCIENCE. Prentice Hall Water in the Atmosphere Water in the Atmosphere Water in the Atmosphere.
 Prentice Hall EARTH SCIENCE Tarbuck Lutgens Water s Changes of State 1. Precipitation is any form of water that falls from a cloud. a. Examples: Snow, rain, hail, sleet 3 States of matter of water: 1.
Prentice Hall EARTH SCIENCE Tarbuck Lutgens Water s Changes of State 1. Precipitation is any form of water that falls from a cloud. a. Examples: Snow, rain, hail, sleet 3 States of matter of water: 1.
5) The amount of heat needed to raise the temperature of 1 gram of a substance by 1 C is called: Page Ref: 69
 Homework #2 Due 9/19/14 1) If the maximum temperature for a particular day is 26 C and the minimum temperature is 14 C, what would the daily mean temperature be? (Page Ref: 66) 2) How is the annual mean
Homework #2 Due 9/19/14 1) If the maximum temperature for a particular day is 26 C and the minimum temperature is 14 C, what would the daily mean temperature be? (Page Ref: 66) 2) How is the annual mean
The Clausius-Clapeyron and the Kelvin Equations
 PhD Environmental Fluid Mechanics Physics of the Atmosphere University of Trieste International Center for Theoretical Physics The Clausius-Clapeyron and the Kelvin Equations by Dario B. Giaiotti and Fulvio
PhD Environmental Fluid Mechanics Physics of the Atmosphere University of Trieste International Center for Theoretical Physics The Clausius-Clapeyron and the Kelvin Equations by Dario B. Giaiotti and Fulvio
1. Second Law of Thermodynamics
 1. Second Law of hermodynamics he first law describes how the state of a system changes in response to work it performs and heat absorbed. he second law deals with direction of thermodynamic processes
1. Second Law of hermodynamics he first law describes how the state of a system changes in response to work it performs and heat absorbed. he second law deals with direction of thermodynamic processes
S6. (a) State what is meant by an ideal gas...
 IB PHYSICS Name: DEVIL PHYSICS Period: Date: BADDEST CLASS ON CAMPUS TSOKOS CHAPTER 3 TEST REVIEW S1. Thermal energy is transferred through the glass windows of a house mainly by A. conduction. B. radiation.
IB PHYSICS Name: DEVIL PHYSICS Period: Date: BADDEST CLASS ON CAMPUS TSOKOS CHAPTER 3 TEST REVIEW S1. Thermal energy is transferred through the glass windows of a house mainly by A. conduction. B. radiation.
Lecture 10: Climate Sensitivity and Feedback
 Lecture 10: Climate Sensitivity and Feedback Human Activities Climate Sensitivity Climate Feedback 1 Climate Sensitivity and Feedback (from Earth s Climate: Past and Future) 2 Definition and Mathematic
Lecture 10: Climate Sensitivity and Feedback Human Activities Climate Sensitivity Climate Feedback 1 Climate Sensitivity and Feedback (from Earth s Climate: Past and Future) 2 Definition and Mathematic
Measuring State Parameters of the Atmosphere
 Measuring State Parameters of the Atmosphere Some Applications of Atmospheric Thermodynamics Earth Observing Laboratory, NCAR IDEAS-4 Tutorial Introduction Goals of This Presentation Present two complementary
Measuring State Parameters of the Atmosphere Some Applications of Atmospheric Thermodynamics Earth Observing Laboratory, NCAR IDEAS-4 Tutorial Introduction Goals of This Presentation Present two complementary
How to please the rulers of NPL-213 the geese
 http://www.walkingmountains. org/2015/03/reintroduction-ofthe-canada-goose/ How to please the rulers of NPL-213 the geese (Entropy and the 2 nd Law of Thermodynamics) Physics 116 2017 Tues. 3/21, Thurs
http://www.walkingmountains. org/2015/03/reintroduction-ofthe-canada-goose/ How to please the rulers of NPL-213 the geese (Entropy and the 2 nd Law of Thermodynamics) Physics 116 2017 Tues. 3/21, Thurs
METEO 431: Atmospheric Thermodynamics
 METEO 431: Atmospheric Thermodynamics Final Exam (100 points) INSTRUCTIONS: Please write as legibly as you can and make sure your thoughts are organized. I am looking for your understanding of the key
METEO 431: Atmospheric Thermodynamics Final Exam (100 points) INSTRUCTIONS: Please write as legibly as you can and make sure your thoughts are organized. I am looking for your understanding of the key
