Standards 8.5.c. I know chemical reactions usually liberate or absorbs heat.
|
|
|
- Dennis Nelson
- 6 years ago
- Views:
Transcription
1 6 March 2013 Give an example of a physical change and a chemical change, and then describe how they are different from the other. Explain your answer in 2-3 sentences.
2 Standards 8.5.c. I know chemical reactions usually liberate or absorbs heat.
3 When chemical reactions occur, the connections between atoms must be broken and rearranged. In order to do this, Heat is involved in all chemical reactions.
4 Endothermic Within-Heating Endo = Enter Therm/ic = Heat Exothermic Outside-Heating Exo = Exit Therm/ic = Heat
5 Endothermic Reactions in which the system absorbs heat from the surrounding. Exothermic Reactions in which the system releases heat to the surrounding.
6 Endothermic It feels: Cold It feels: Exothermic Hot
7 Endothermic Examples: Cooking an egg Baking a bread Exothermic Examples: Candle flame Rusting Iron
8 Exothermic Reaction
9 Endothermic Reaction
10 Heat (Thermal Energy) in phase change is not the same as Heat of Reaction. Phase Change = Physical Change Chemical Reaction = Chemical Change
11 Example Cooking Egg Baking of cake Activation of cold pack Non-Example Heat of melting Heat of vaporization Heat of sublimation
12 Example Candle burning Explosion of dynamite Activation of yeast Non-Example Heat of deposition Heat of freezing Heat of condensation
13 6 March 2013 In your own understanding, what causes steel to rust. Explain your answer in 2-3 sentences.
14 Minimum amount of energy needed to start a chemical reaction All reactions require a certain amount of activation energy.
15 Surface Area Temperature Concentration Presence of catalyst or inhibitor
16 Factors Effects Surface Area The greater the area exposed, speed up the reaction. Speeding up reactions are both useful and harmful. Useful: chewing food to make it easy to digest. Harmful: gas fumes or dust particles ignites easily.
17 Temperature Increasing the temperature, increases the movement of the particles and energy. Decreasing the temperature, decreases the movement of the particles and energy. Something good to remember: We store food in the refrigerator to lower the temperature and slowdown the reaction in spoiling food.
18 Concentration It is the amount of stuff(substance) in a given volume. Example: Adding more sugar in coffee increases the concentration of sugar (sweeter). Adding more lemon juice in lemonade increases the concentration of acidity (sourness).
19 Catalyst A material added to reduce the activation energy. Example: Lead and Sulfur as fuel additive Enzymes are examples of catalyst found in the human body.
20 Inhibitors A material added to reduce the rate of reaction. Examples: Food preservatives Lubricant/protective coating
21 Is popping popcorn a physical change or a chemical change? Explain your answer. Due: March 01, 2013
22 Objective: Observe a chemical reaction. Observe an endothermic reaction.
23 1.3 grams citric acid (H3C6H5O7) 1.5 grams Baking Soda = Sodium Bicarbonate (NaHCO3) Zip-lock bag Small cup Electronic scale Graduated cylinder Weighing plate 20 ml H 2 O
24 H 3 C 6 H 5 O 7 + 3NaHCO 3 Citric acid + sodium bicarbonate CO 2 + 3H 2 O + NaC 6 H 5 O 7 carbon + water + sodium citrate dioxide
25 Procedure: Place weighing plat on scale press zero so that scale doesn t measure the mass of the weighing plate. Measure 1.5g sodium bicarbonate (baking soda) onto the plate. Add to plastic bag. Measure 1.3g citric acid onto plate. Add to plastic bag. Using the graduated cylinder, measure 20 ml of H 2 O. Pour the water into the small cup. Place the cup inside the plastic bag standing up. Be careful not to spill the water! Carefully squeeze the air out of the bag and zip the bag tightly shut. Tip the cup over to spill the water and mix ingredients with your hands. Record your observations below in the data section. Clean your lab station and answer discussion questions below.
26 Reactants Elements in Reactant Observations Citric Acid Sodium Bicarbonate
27 What do you SEE happening during the reaction? What do you FEEL happening during the reaction?
28 1. Do you think this was a physical or chemical reaction? What is your evidence? 2. What do you think was produced in this reaction? What is your evidence? 3. From what you observed and felt during the reaction do you think energy was going into the reaction (being used up) or out of the reaction (heat is being produced)? What is your evidence? 4. From what you observed in today s lab, what do you think an endothermic reaction is? 5. What is one question you have about this experiment? (write AT LEAST 1 question)
29 Objective: Observe a chemical reaction. Observe an exothermic reaction.
30 Materials: 1.5 grams yeast 20 ml hydrogen peroxide (H 2 O 2 ) Zip-lock bag Small cup Electronic scale Graduated cylinder Weighing plate
31 Procedures 1. Place weighing plat on scale press zero so that scale doesn t measure the mass of the weighing plate. 2. Measure 1.5g yeast onto the plate. Add to plastic bag. 3. Using the graduated cylinder, measure 20 ml of H 2 O 2. Pour the hydrogen peroxide into the small cup. 4. Place the cup inside the plastic bag standing up. Be careful not to spill! 5. Carefully squeeze the air out of the bag and zip the bag tightly shut. 6. Tip the cup over to spill the hydrogen peroxide and mix ingredients with your hands. 7. Record your observations below in the data section. 8. Clean your lab station and answer discussion questions below.
32 Reactants Hydrogen Peroxide Elements in Reactant Observations Yeast (this is a type of cell and has LOTS of elements)
33 What do you SEE happening during the reaction? What do you FEEL happening during the reaction?
34 1. Do you think this was a physical or chemical reaction? What is your evidence? 2. What do you think was produced in this reaction? What is your evidence? 3. From what you observed and felt during the reaction do you think energy was going into the reaction (being used up) or out of the reaction (heat is being produced)? What is your evidence? 4. From what you observed in today s lab, what do you think an exothermic reaction is? 5. What is one question you have about this experiment? (write AT LEAST 1 question)
35 How can you tell if a chemical reaction is endothermic or exothermic? What are common daily events involving endothermic or exothermic reactions? Name at least 2 examples per type, then support your answer by paraphrasing the concepts you learned? Why are reactions (exothermic and endothermic) important to our existence?
36 Sugar dissolving in tea Chemical Change Physical Change
37 Logs burning Chemical Change Physical Change
38 Breaking water up by separating it into hydrogen and oxygen Chemical Change Physical Change
39 Cutting paper Chemical Change Physical Change
40 Crushing an aspirin Chemical Change Physical Change
41 Metal rusting Chemical Change Physical Change
42 An egg rotting Chemical Change Physical Change
43 An egg breaking Chemical Change Physical Change
Physical or Chemical Change?
 Chemical Formulas and Equations Physical or Chemical Change? Matter can change physically or chemically; a process that produces a chemical change is a chemical reaction. Chemical Formulas and Equations
Chemical Formulas and Equations Physical or Chemical Change? Matter can change physically or chemically; a process that produces a chemical change is a chemical reaction. Chemical Formulas and Equations
Today is: Monday, October 17th
 Today is: Monday, October 17th Get out your homework! 10/17/2016 #motivationmonday (This is Week 11 Warm Ups!) 1. It s a new quarter, which means a fresh start! What can you do to stay motivated this quarter?
Today is: Monday, October 17th Get out your homework! 10/17/2016 #motivationmonday (This is Week 11 Warm Ups!) 1. It s a new quarter, which means a fresh start! What can you do to stay motivated this quarter?
1 Forming New Substances
 CHAPTER 9 1 Forming New Substances SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a What are exothermic reactions? What are
CHAPTER 9 1 Forming New Substances SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a What are exothermic reactions? What are
Activation Energy is the minimum amount of energy needed to start a chemical reaction.
 5.3 Controlling Chemical Reactions Vocabulary: Activation energy Concentration Catalyst Enzyme Inhibitor - How do reactions get started? Chemical reactions won t begin until the reactants have enough energy.
5.3 Controlling Chemical Reactions Vocabulary: Activation energy Concentration Catalyst Enzyme Inhibitor - How do reactions get started? Chemical reactions won t begin until the reactants have enough energy.
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions
 Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Section 1: What is a Chemical Reaction
 Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Chemical Reaction Lab Bagged Chemical Reactions
 Learning Target: The student experiments and determines that the rates of reaction among atoms and molecules depend on the concentration, pressure, and temperature of the reactants and the presence or
Learning Target: The student experiments and determines that the rates of reaction among atoms and molecules depend on the concentration, pressure, and temperature of the reactants and the presence or
Experiment 17 It s A Gas and More!
 Energy Energy Experiment 17 It s A Gas and More! OUTCOMES After completing this lab activity, the student should be able to: explain a simple method for distinguishing carbon dioxide gas from oxygen gas.
Energy Energy Experiment 17 It s A Gas and More! OUTCOMES After completing this lab activity, the student should be able to: explain a simple method for distinguishing carbon dioxide gas from oxygen gas.
Physical and Chemical Changes
 Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
LESSON 1: DESCRIBING MATTER pg.5. Chemistry = Is the study of matter & how matter changes. Liquid/Solid/Gas
 Chemistry..CHAPTER 1: INTRO TO MATTER LESSON 1: DESCRIBING MATTER pg.5 Chemistry = Is the study of matter & how matter changes A. Matter = anything that has mass & takes up space à You, air, plastic, metal,
Chemistry..CHAPTER 1: INTRO TO MATTER LESSON 1: DESCRIBING MATTER pg.5 Chemistry = Is the study of matter & how matter changes A. Matter = anything that has mass & takes up space à You, air, plastic, metal,
Observing Chemical Change - 5.1
 Observing Chemical Change - 5.1 Vocabulary: Physical change - Chemical change - Reactant Product Precipitate Exothermic reaction - Endothermic reaction - Properties of matter: Two kinds of properties of
Observing Chemical Change - 5.1 Vocabulary: Physical change - Chemical change - Reactant Product Precipitate Exothermic reaction - Endothermic reaction - Properties of matter: Two kinds of properties of
3 Chemical Properties
 CHAPTER 7 3 Chemical Properties SECTION The Properties of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What are chemical properties of matter? What
CHAPTER 7 3 Chemical Properties SECTION The Properties of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What are chemical properties of matter? What
MiSP CHEMICAL REACTIONS, L3 Teacher Guide. Introduction
 MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
3.3. Physical and Chemical Changes. Before You Read. What is a physical change? What is a chemical change?
 Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
4 Energy and Rates of Chemical Reactions
 CHAPTER 14 4 and Rates of Chemical Reactions SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: How is energy involved in a chemical reaction?
CHAPTER 14 4 and Rates of Chemical Reactions SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: How is energy involved in a chemical reaction?
1 The Nature of Chemical Reactions
 CHAPTER 7 1 The Nature of Chemical Reactions SECTION Chemical Reactions KEY IDEAS As you read this section, keep these questions in mind: What happens in a chemical reaction? What is the role of energy
CHAPTER 7 1 The Nature of Chemical Reactions SECTION Chemical Reactions KEY IDEAS As you read this section, keep these questions in mind: What happens in a chemical reaction? What is the role of energy
Page 1 / 12. Chemistry Exam. Name: Matter Properties, Structure. Question 1 (1 point) The atomic number of an atom is. A. The mass of the atom.
 Chemistry Exam Matter Properties, Structure Name: Question 1 (1 point) The atomic number of an atom is A. The mass of the atom. B. The number of protons added to the number of neutrons in the nucleus.
Chemistry Exam Matter Properties, Structure Name: Question 1 (1 point) The atomic number of an atom is A. The mass of the atom. B. The number of protons added to the number of neutrons in the nucleus.
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 6, LESSON 1: WHAT IS A CHEMICAL REACTION? MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact
The Next Generation Science Standards (NGSS) CHAPTER 6, LESSON 1: WHAT IS A CHEMICAL REACTION? MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact
Student Notes. Chemical Reactions LINK
 LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
2/22/2019 NEW UNIT! Chemical Interactions. Atomic Basics #19
 NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds.
 Chapter 11 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
Chapter 11 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
6.1- Chemical vs. Physical - Pre-Lab Questions
 6.1- Chemical vs. Physical - Pre-Lab Questions Name: Instructor: Date: Section/Group: 1. Using the procedures for each station provided as a guide, predict which properties you will be looking for in each
6.1- Chemical vs. Physical - Pre-Lab Questions Name: Instructor: Date: Section/Group: 1. Using the procedures for each station provided as a guide, predict which properties you will be looking for in each
Chemistry is the study of matter, its properties and its changes or transformations. Feb 19 11:08 AM
 What is Chemistry? Chemistry is the study of matter, its properties and its changes or transformations. Matter is anything that has mass and takes up space. All matter has physical and chemical properties.
What is Chemistry? Chemistry is the study of matter, its properties and its changes or transformations. Matter is anything that has mass and takes up space. All matter has physical and chemical properties.
Moles Lab Activity 1: PCU (Popcorn Counting Units)
 Moles Lab Activity 1: PCU (Popcorn Counting Units) Materials: A container of each of the following: Popcorn kernels Another type of beans A large unopened bag of popcorn Kernels Balance Safety goggles
Moles Lab Activity 1: PCU (Popcorn Counting Units) Materials: A container of each of the following: Popcorn kernels Another type of beans A large unopened bag of popcorn Kernels Balance Safety goggles
Sulfuric acid is hazardous: Safety glasses are REQUIRED during this experiment.
 CHEMICAL COMPOSITION Life exists on Earth because of the abundant presence of liquid water. While other planets have water, it may be primarily found as either a gas, as on Venus, or as a solid, such as
CHEMICAL COMPOSITION Life exists on Earth because of the abundant presence of liquid water. While other planets have water, it may be primarily found as either a gas, as on Venus, or as a solid, such as
Physical & Chemical PROPERTIES
 Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Are Chemical Reactions Closed Systems?
 Measuring a Chemical Reaction 1. Place about 100 ml of vinegar in a large plastic cup. Carefully weigh the cup AND the vinegar all together, and write down the total mass. (Don t worry about subtracting
Measuring a Chemical Reaction 1. Place about 100 ml of vinegar in a large plastic cup. Carefully weigh the cup AND the vinegar all together, and write down the total mass. (Don t worry about subtracting
Observations. Direct Observations Those observations we make ourselves
 Observations Direct Observations Those observations we make ourselves Indirect Observations Those observations that we figure out from direct observations Observing Milk Consumption The cafeteria is considering
Observations Direct Observations Those observations we make ourselves Indirect Observations Those observations that we figure out from direct observations Observing Milk Consumption The cafeteria is considering
Chemical Energy Conversions. Vanderbilt Student Volunteers for Science VINSE/VSVS Rural
 Chemical Energy Conversions Vanderbilt Student Volunteers for Science 2018-2019 VINSE/VSVS Rural Important!!! Please use this resource to reinforce your understanding of the lesson! Make sure you have
Chemical Energy Conversions Vanderbilt Student Volunteers for Science 2018-2019 VINSE/VSVS Rural Important!!! Please use this resource to reinforce your understanding of the lesson! Make sure you have
Physical and Chemical Changes & Properties of Matter
 Physical and Chemical Changes & Properties of Matter S8P1. Obtain, evaluate, and communicate information about the structure and properties of matter. 8P1CD Physical And Chemical Properties And Changes
Physical and Chemical Changes & Properties of Matter S8P1. Obtain, evaluate, and communicate information about the structure and properties of matter. 8P1CD Physical And Chemical Properties And Changes
Signs of Chemical Reactions
 Signs of Chemical Reactions Tell the difference between changes which are PHYSICAL and those which involve a CHEMICAL reaction Doing six different experiments and observing the changes which
Signs of Chemical Reactions Tell the difference between changes which are PHYSICAL and those which involve a CHEMICAL reaction Doing six different experiments and observing the changes which
Why do cold packs get cold?
 Assignment #5 Temperature in Reactions LO: To determine which solute dissolves most endothermically and exothermically in water. EQ: What makes an endothermic reaction feel cold? (explain using bonds and
Assignment #5 Temperature in Reactions LO: To determine which solute dissolves most endothermically and exothermically in water. EQ: What makes an endothermic reaction feel cold? (explain using bonds and
Period 11: Chemical Energy and Fossil Fuels
 Name Section Period 11: Chemical Energy and Fossil Fuels 11.1 What is the Composition of Matter? 1) Atoms a) What are the building blocks of an atom? b) Which nucleons make up an atomic nucleus? c) What
Name Section Period 11: Chemical Energy and Fossil Fuels 11.1 What is the Composition of Matter? 1) Atoms a) What are the building blocks of an atom? b) Which nucleons make up an atomic nucleus? c) What
Chapter 6, Lesson 1: What is a Chemical Reaction?
 Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Signs of Chemical Reactions
 Signs of Chemical Reactions Tell the difference between changes which are PHYSICAL and those which involve a CHEMICAL reaction Doing six different experiments and observing the changes which
Signs of Chemical Reactions Tell the difference between changes which are PHYSICAL and those which involve a CHEMICAL reaction Doing six different experiments and observing the changes which
Section 1 Chemical Changes
 Chemical Reactions Section 1 Chemical Changes What You ll Learn: How to identify the reactants & products in a chemical reaction How a chemical reaction follows the law of conservation of mass How chemists
Chemical Reactions Section 1 Chemical Changes What You ll Learn: How to identify the reactants & products in a chemical reaction How a chemical reaction follows the law of conservation of mass How chemists
8.5E: Chemical Reactions
 Reflect Have you ever seen fireworks explode in the sky? If so, you may have observed a variety of different colors and shapes in the sky and watched with amazement as the colors changed overhead. These
Reflect Have you ever seen fireworks explode in the sky? If so, you may have observed a variety of different colors and shapes in the sky and watched with amazement as the colors changed overhead. These
Unit 10 Chemical Reactions. Mrs. Valentine Physical Science
 Unit 10 Chemical Reactions Mrs. Valentine Physical Science Section 1 Evidence for Chemical Reactions We see chemical reactions all around us. We are even doing some right now in our bodies. Picture yourself
Unit 10 Chemical Reactions Mrs. Valentine Physical Science Section 1 Evidence for Chemical Reactions We see chemical reactions all around us. We are even doing some right now in our bodies. Picture yourself
Physical and Chemical change: Conservation of matter *
 OpenStax-CNX module: m38711 1 Physical and Chemical change: Conservation of matter * Free High School Science Texts Project This work is produced by OpenStax-CNX and licensed under the Creative Commons
OpenStax-CNX module: m38711 1 Physical and Chemical change: Conservation of matter * Free High School Science Texts Project This work is produced by OpenStax-CNX and licensed under the Creative Commons
Changes in Matter. Introduction to Chemistry
 Changes in Matter Introduction to Chemistry Classifying Matter Matter: is anything that has mass and volume. Volume: the amount of space that something takes up Property: a characteristic of a material
Changes in Matter Introduction to Chemistry Classifying Matter Matter: is anything that has mass and volume. Volume: the amount of space that something takes up Property: a characteristic of a material
Name: 1. Which of the following is probably true about 300 ml of sand and 300 ml of water?
 Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Chemistry Part 2: We re Not Done Yet!
 Chemistry Part 2: We re Not Done Yet! WOW! Learning about atoms sure was fun! Let s continue our fascinating exploration of chemicals. What is a Chemical? Chemical: A substance having a constant composition
Chemistry Part 2: We re Not Done Yet! WOW! Learning about atoms sure was fun! Let s continue our fascinating exploration of chemicals. What is a Chemical? Chemical: A substance having a constant composition
Physical and Chemical Properties. Book K
 Physical and Chemical Properties Book K Warm Up: As you view the video clip, list examples of physical and chemical changes. Physical Change Chemical Change 1. 1. 2. 2. 3. 3. 4. 4. Warm Up: As you view
Physical and Chemical Properties Book K Warm Up: As you view the video clip, list examples of physical and chemical changes. Physical Change Chemical Change 1. 1. 2. 2. 3. 3. 4. 4. Warm Up: As you view
Name Period Date. Lab 1: Mass of Ice Materials: beaker, ice and balance.
 Name Period Date Testing the Law of Conservation of MASS! Introduction: Does mass change in a chemical or physical reaction? In this series of experiments you will find the answer to this question. Lab
Name Period Date Testing the Law of Conservation of MASS! Introduction: Does mass change in a chemical or physical reaction? In this series of experiments you will find the answer to this question. Lab
Changes in Matter Study Guide
 Changes in Matter Study Guide Define the following and give a real world example: 1. : A change in matter that requires the substances to absorb energy Example: Photosynthesis (the plants absorb light
Changes in Matter Study Guide Define the following and give a real world example: 1. : A change in matter that requires the substances to absorb energy Example: Photosynthesis (the plants absorb light
A simple equation of what happens when you add baking soda to vinegar:
 What s the Matter? Matter is anything that takes up space and has mass. Mass is the stuff that matter is made of, or the amount of particles in a substance or object. Matter has physical and chemical properties
What s the Matter? Matter is anything that takes up space and has mass. Mass is the stuff that matter is made of, or the amount of particles in a substance or object. Matter has physical and chemical properties
Unit 6M.3: Changing materials
 Unit 6M.3: Adding materials to water. Chemical reactions Skill you will use: Classifying Observing Predicting By the end of this unit you should: Know that when substances are added to water, some will
Unit 6M.3: Adding materials to water. Chemical reactions Skill you will use: Classifying Observing Predicting By the end of this unit you should: Know that when substances are added to water, some will
STEMscopedia: PHYSICAL AND CHEMICAL PROPERTIES AND CHANGES 8P1CD
 Reflect STEMscopedia: PHYSICAL AND CHEMICAL 8P1CD What do ice cream, root beer, and carbon dioxide gas have in common? Not only do these ingredients combine to make a good treat on a hot summer day, but
Reflect STEMscopedia: PHYSICAL AND CHEMICAL 8P1CD What do ice cream, root beer, and carbon dioxide gas have in common? Not only do these ingredients combine to make a good treat on a hot summer day, but
Chemical Reactions. Chapter Test A. Multiple Choice. 1 Pearson Education, Inc., or its affiliates. All rights reserved.
 Name Date Class Chemical Reactions Chapter Test A Multiple Choice Write the letter of the correct answer on the line at the left. 1. Chemistry is a. a characteristic of a substance that can be observed
Name Date Class Chemical Reactions Chapter Test A Multiple Choice Write the letter of the correct answer on the line at the left. 1. Chemistry is a. a characteristic of a substance that can be observed
Name Date. Physical and Chemical Changes
 Name Date Physical and Chemical Changes Physical Changes Physical Change: A change in which no new substances form Do not change the type of matter an object is made of You change the shape, size or more
Name Date Physical and Chemical Changes Physical Changes Physical Change: A change in which no new substances form Do not change the type of matter an object is made of You change the shape, size or more
Alternative Reaction Pathways
 Section 1 Energy and Entropy: Alternative Reaction Pathways What Do You See? Learning Outcomes In this section you will Apply the engineering-design process to scientific and everyday situations. Generate
Section 1 Energy and Entropy: Alternative Reaction Pathways What Do You See? Learning Outcomes In this section you will Apply the engineering-design process to scientific and everyday situations. Generate
6 th Grade Introduction to Chemistry
 Lesson 1 (Describing Matter) 6 th Grade Introduction to Chemistry Matter anything that has mass and takes up space All the stuff in the natural world is matter. Chapter 1: Introduction to Matter Chemistry
Lesson 1 (Describing Matter) 6 th Grade Introduction to Chemistry Matter anything that has mass and takes up space All the stuff in the natural world is matter. Chapter 1: Introduction to Matter Chemistry
Physical Changes vs Chemical Changes Lab
 Physical Changes vs Chemical Changes Lab What was done? What can you observe? Is it a physical or chemical change and why? What else have you learned during the demonstration? #1) Crumpling Paper Notice
Physical Changes vs Chemical Changes Lab What was done? What can you observe? Is it a physical or chemical change and why? What else have you learned during the demonstration? #1) Crumpling Paper Notice
Understanding Equations
 Chemical Reactions Chemical reaction: a process of chemically changing both the physical and chemical properties of a substance to a new substance with different physical and chemical properties. Video
Chemical Reactions Chemical reaction: a process of chemically changing both the physical and chemical properties of a substance to a new substance with different physical and chemical properties. Video
Physical and Chemical Properties of Matter
 Physical and Chemical Properties of Matter Please grab a sheet from the front and get out scissors. You will also need a glue stick and piece of construction paper. Think-Pair-Share on the following questions.
Physical and Chemical Properties of Matter Please grab a sheet from the front and get out scissors. You will also need a glue stick and piece of construction paper. Think-Pair-Share on the following questions.
4. Every CHANGE in matter includes a change in, which is conserved in a chemical reaction and. TRANSFORMED from one form to another.
 Part C: Lesson 1.4 - Read pages 20-29 to answer these questions. Add to notes - 1. A physical change alters the form or appearance of matter, such as Change in size, a change in SIZE or. SHAPE shape, or
Part C: Lesson 1.4 - Read pages 20-29 to answer these questions. Add to notes - 1. A physical change alters the form or appearance of matter, such as Change in size, a change in SIZE or. SHAPE shape, or
What Do You Think? Investigate GOALS. Part A: Freezing Water
 Activity 5 Freezing Water GOALS In this activity you will: Determine the freezing point of water. Show graphically what happens to the temperature as water is cooled to freezing and while it is freezing.
Activity 5 Freezing Water GOALS In this activity you will: Determine the freezing point of water. Show graphically what happens to the temperature as water is cooled to freezing and while it is freezing.
LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EXPERIMENTS
 LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EPERIMENTS PURPOSE: To practice the scientific method of observation, hypothesis, experimentation, & conclusion. SAFETY CONCERNS: Always wear
LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EPERIMENTS PURPOSE: To practice the scientific method of observation, hypothesis, experimentation, & conclusion. SAFETY CONCERNS: Always wear
Matter and Change. Monday Tuesday Wednesday Thursday Friday. Textbook Activity. Meet in B122. Hw: pg 7 Hw: pg 4. Matter Review
 Matter and Change Monday Tuesday Wednesday Thursday Friday SEPTEMBER 1 2 3 4 5 NO SCHOOL Matter & Change Classifying Matter Textbook Activity Meet in B122 Physical and Chemical Properties and Changes Indiana
Matter and Change Monday Tuesday Wednesday Thursday Friday SEPTEMBER 1 2 3 4 5 NO SCHOOL Matter & Change Classifying Matter Textbook Activity Meet in B122 Physical and Chemical Properties and Changes Indiana
Foundations of Chemistry
 Foundations of Chemistry Physical Changes Key Concepts How can a change in energy affect the state of matter? What happens when something dissolves? What is meant by conservation of mass? What do you think?
Foundations of Chemistry Physical Changes Key Concepts How can a change in energy affect the state of matter? What happens when something dissolves? What is meant by conservation of mass? What do you think?
Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds.
 Chapter 9 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
Chapter 9 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
Chemical Reactions and Equations
 Chemical Reactions and Equations Changes and Chemical Reactions Key Concepts Why do chemical reactions always involve a change in? What is the difference between an endothermic reaction and an exothermic
Chemical Reactions and Equations Changes and Chemical Reactions Key Concepts Why do chemical reactions always involve a change in? What is the difference between an endothermic reaction and an exothermic
MATTER. Physical Science 2nd Semester NAME: CLASS PERIOD: TEACHER: HW POINTS EARNED LAB POINTS EARNED. PAGE NUMBERS Learning Targets: Matter 1-2
 MATTER Physical Science 2nd Semester ASSIGNMENT PAGE NUMBERS Learning Targets: Matter 1-2 NAME: CLASS PERIOD: TEACHER: DUE DATE HW POINTS EARNED LAB POINTS EARNED Density Lab 3-4 Density Calculations Worksheet
MATTER Physical Science 2nd Semester ASSIGNMENT PAGE NUMBERS Learning Targets: Matter 1-2 NAME: CLASS PERIOD: TEACHER: DUE DATE HW POINTS EARNED LAB POINTS EARNED Density Lab 3-4 Density Calculations Worksheet
Lesson 5: Oxygen, Oxidation, and Combustion
 Lesson 5: Oxidation, and Students are introduced to exothermic reactions that involve oxidation. Main Concept: Oxygen is a highly reactive element involved in chemical reactions that release heat energy.
Lesson 5: Oxidation, and Students are introduced to exothermic reactions that involve oxidation. Main Concept: Oxygen is a highly reactive element involved in chemical reactions that release heat energy.
Physical Changes can be observed without changing the identity of the substance (often states of matter changes).
 Physical Changes can be observed without changing the identity of the substance (often states of matter changes). Remember: States of matter changes are physical changes. The identify of the substance
Physical Changes can be observed without changing the identity of the substance (often states of matter changes). Remember: States of matter changes are physical changes. The identify of the substance
Supplemental Material
 Supplemental Material Chemical and Physical Change Assessment CPCA γ-version This assessment can be fashioned into a quiz booklet using the following cover page. At the end of the quiz is a customized
Supplemental Material Chemical and Physical Change Assessment CPCA γ-version This assessment can be fashioned into a quiz booklet using the following cover page. At the end of the quiz is a customized
Bags of Reactions Chemistry I Acc
 Introduction: Bags of Reactions Chemistry I Acc Plop, plop, fizz, fizz, oh, what a relief it is. claims the old TV ad for a popular antacid. Just what is in the tablet that is relieving the upset stomach?
Introduction: Bags of Reactions Chemistry I Acc Plop, plop, fizz, fizz, oh, what a relief it is. claims the old TV ad for a popular antacid. Just what is in the tablet that is relieving the upset stomach?
5.3. Changes in Matter. Physical Change
 5.3 Changes in Matter LEARNING TIP Read the first two paragraphs on this page. Write a one sentence summary to remember important information. For the rest of Section 5.3, read each part and pause to think
5.3 Changes in Matter LEARNING TIP Read the first two paragraphs on this page. Write a one sentence summary to remember important information. For the rest of Section 5.3, read each part and pause to think
7th Grade Chemical Reactions and Energy
 Slide 1 / 119 Slide 2 / 119 7th Grade Chemical Reactions and Energy 2015-10-27 www.njctl.org Slide 3 / 119 Table of Contents: Chemical Reactions and Definitions of Energy What is a change and what are
Slide 1 / 119 Slide 2 / 119 7th Grade Chemical Reactions and Energy 2015-10-27 www.njctl.org Slide 3 / 119 Table of Contents: Chemical Reactions and Definitions of Energy What is a change and what are
1 Three States of Matter
 CHAPTER 3 1 Three States of Matter SECTION States of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What is matter made of? What are the three most common
CHAPTER 3 1 Three States of Matter SECTION States of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What is matter made of? What are the three most common
Atoms. - Proton - Neutron. - Electron
 Chemistry of Life Atoms - The basic unit of matter is called an Atom - Atoms are incredibly small, but despite its extremely small size, an atom contains subatomic particles that are even smaller - Three
Chemistry of Life Atoms - The basic unit of matter is called an Atom - Atoms are incredibly small, but despite its extremely small size, an atom contains subatomic particles that are even smaller - Three
Chapter 2. Section 1
 Chapter 2 Section 1 Describing Matter Properties used to describe matter can be classified as extensive or intensive Extensive property - depends on the amount of matter in a sample. Ex. Mass and Volume
Chapter 2 Section 1 Describing Matter Properties used to describe matter can be classified as extensive or intensive Extensive property - depends on the amount of matter in a sample. Ex. Mass and Volume
Factors in Reaction Rates
 Section 6 Factors in Reaction Rates What Do You See? Learning Outcomes In this section you will Discover conditions that make a reaction proceed faster or slower. Discuss explanations for why this happens
Section 6 Factors in Reaction Rates What Do You See? Learning Outcomes In this section you will Discover conditions that make a reaction proceed faster or slower. Discuss explanations for why this happens
1. Review chemical vs. physical reactions 2. Chemical reactions video 3. Useful chemical reactions research assignment
 Science 24: Day 10 1. Review chemical vs. physical reactions 2. Chemical reactions video 3. Useful chemical reactions research assignment https://www.youtube.com/watch?v=fofpjj7v414 Which reaction was
Science 24: Day 10 1. Review chemical vs. physical reactions 2. Chemical reactions video 3. Useful chemical reactions research assignment https://www.youtube.com/watch?v=fofpjj7v414 Which reaction was
Volume. measures how much space matter takes up. solubility. The amount of mass for an object is called. matter
 measures how much space matter takes up. Volume is the resistance of an object to sink, and it depends on. The ability of a substance to dissolve into another substance is called. The amount of mass for
measures how much space matter takes up. Volume is the resistance of an object to sink, and it depends on. The ability of a substance to dissolve into another substance is called. The amount of mass for
Chemical Reactions and Energy. Classwork and Homework. Slide 1 / 75. Slide 2 / 75. Slide 3 / 75. Unit Preview Classwork
 New Jersey Center for Teaching and Learning Slide 1 / 75 Progressive Science Initiative This material is made freely available at www.njctl.org and is intended for the non-commercial use of students and
New Jersey Center for Teaching and Learning Slide 1 / 75 Progressive Science Initiative This material is made freely available at www.njctl.org and is intended for the non-commercial use of students and
Chemical Reactions and Energy. Classwork and Homework. Slide 1 / 75. Slide 2 / 75. Slide 3 / 75. Slide 4 / 75. Slide 5 / 75.
 Slide 1 / 75 Slide 2 / 75 New Jersey Center for Teaching and Learning Progressive Science Initiative This material is made freely available at www.njctl.org and is intended for the non-commercial use of
Slide 1 / 75 Slide 2 / 75 New Jersey Center for Teaching and Learning Progressive Science Initiative This material is made freely available at www.njctl.org and is intended for the non-commercial use of
LESSON 2. Chemical Equations. When copper sulfate reacts with ammonia, a precipitate forms and the solution color changes to deep blue.
 LESSN 2 Chemical Equations When copper sulfate reacts with ammonia, a precipitate forms and the solution color changes to deep blue. By the end of this lesson... you will be able to explain how chemical
LESSN 2 Chemical Equations When copper sulfate reacts with ammonia, a precipitate forms and the solution color changes to deep blue. By the end of this lesson... you will be able to explain how chemical
Suggest one advantage and one disadvantage of using butane compared to charcoal as a source of heat [2]
![Suggest one advantage and one disadvantage of using butane compared to charcoal as a source of heat [2] Suggest one advantage and one disadvantage of using butane compared to charcoal as a source of heat [2]](/thumbs/78/78230619.jpg) High Demand Questions QUESTIONSHEET 1 A gas barbecue uses butane (C 4 H 10 ) as a fuel. The butane is stored as a liquid in a container. When the burner is turned on, the liquid turns to a gas and is lit
High Demand Questions QUESTIONSHEET 1 A gas barbecue uses butane (C 4 H 10 ) as a fuel. The butane is stored as a liquid in a container. When the burner is turned on, the liquid turns to a gas and is lit
Chemical Symbols & Formulas
 + Chemical Symbols & Formulas + Chemical Symbols All elements in the Periodic Table have symbols that are recognized world wide. It does not matter which country you are in or the language you speak. Example:
+ Chemical Symbols & Formulas + Chemical Symbols All elements in the Periodic Table have symbols that are recognized world wide. It does not matter which country you are in or the language you speak. Example:
Matter. Anything that has mass and takes up space. Solids Liquids Gases
 Introduction to Matter Matter Anything that has mass and takes up space Solids Liquids Gases Weight A measure of the gravitational force exerted on an object What is the difference between weight and mass?
Introduction to Matter Matter Anything that has mass and takes up space Solids Liquids Gases Weight A measure of the gravitational force exerted on an object What is the difference between weight and mass?
Transformation of Matter: Physical and Chemical Changes
 Transformation of Matter: Physical and Chemical Changes What does it mean to transform? Transform: change in form, appearance, or makeup What kinds of things transform? How can it be transformed? How
Transformation of Matter: Physical and Chemical Changes What does it mean to transform? Transform: change in form, appearance, or makeup What kinds of things transform? How can it be transformed? How
Limiting Reactants Lab
 Name: Teacher s Name: Class: Block: Date: Partners: Limiting Reactants Lab Purpose: Through experimentation, determine the limiting reactant and the percent yield in a chemical reaction that generates
Name: Teacher s Name: Class: Block: Date: Partners: Limiting Reactants Lab Purpose: Through experimentation, determine the limiting reactant and the percent yield in a chemical reaction that generates
Lesson 1: What are chemical changes?
 Lesson 1 Summary Lesson 1: What are chemical changes? Vocabulary Use with pp. 375 377 physical change a change in which the material keeps its identify chemical change a change in which one substance or
Lesson 1 Summary Lesson 1: What are chemical changes? Vocabulary Use with pp. 375 377 physical change a change in which the material keeps its identify chemical change a change in which one substance or
LESSON 1. Chemical Reactions. Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions.
 LESSON 1 Chemical Reactions Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions. By the end of this lesson... you will be able to explain ways
LESSON 1 Chemical Reactions Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions. By the end of this lesson... you will be able to explain ways
PROPERTIES OF MATTER Review Stations
 PROPERTIES OF MATTER Review Stations PROPERTIES OF MATTER STATION 1 Where did the water droplets on the outside of this cup come from? Answer: The droplets came from the air surrounding the cup. What phase
PROPERTIES OF MATTER Review Stations PROPERTIES OF MATTER STATION 1 Where did the water droplets on the outside of this cup come from? Answer: The droplets came from the air surrounding the cup. What phase
Important Examples of Chemical Change
 Important Examples of Chemical Change chemical change: the change that occurs when substances react in a chemical reaction to create different substances When was the last time you watched an awesome display
Important Examples of Chemical Change chemical change: the change that occurs when substances react in a chemical reaction to create different substances When was the last time you watched an awesome display
Reaction Rates and Equilibrium
 CHAPTER 7 14 SECTION Chemical Reactions Reaction Rates and Equilibrium KEY IDEAS As you read this section, keep these questions in mind: How can you increase the rate of a reaction? What does a catalyst
CHAPTER 7 14 SECTION Chemical Reactions Reaction Rates and Equilibrium KEY IDEAS As you read this section, keep these questions in mind: How can you increase the rate of a reaction? What does a catalyst
PROPERTIES OF MATTER STATION 1. Where did the water droplets on the outside of this cup come from? What phase change is this?
 PROPERTIES OF MATTER STATION 1 Where did the water droplets on the outside of this cup come from? What phase change is this? PROPERTIES OF MATTER STATION 2 Make a copy of this graph on your paper. Label
PROPERTIES OF MATTER STATION 1 Where did the water droplets on the outside of this cup come from? What phase change is this? PROPERTIES OF MATTER STATION 2 Make a copy of this graph on your paper. Label
Liquid X Lab. Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties?
 Names Block Date Liquid X Lab Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette to carefully place drops of each liquid
Names Block Date Liquid X Lab Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette to carefully place drops of each liquid
CHEM1301. F2014 Chapter 1 and 3
 CHEM1301 F2014 Chapter 1 and 3 Chemistry:Why am I here? Chemistry The science that seeks to understand the behavior of matter by studying the BEHAVIOR of atoms and molecules. Not just a cause and effect
CHEM1301 F2014 Chapter 1 and 3 Chemistry:Why am I here? Chemistry The science that seeks to understand the behavior of matter by studying the BEHAVIOR of atoms and molecules. Not just a cause and effect
Chemical Reactions. 1. What is the difference between a physical change and a chemical change
 What is a Chemical Reaction Chemical Reactions 1. What is the difference between a physical change and a chemical change 2. Give an example of a physical change and a chemical change using paper. 3. What
What is a Chemical Reaction Chemical Reactions 1. What is the difference between a physical change and a chemical change 2. Give an example of a physical change and a chemical change using paper. 3. What
Exothermic and Endothermic Reactions
 Exothermic and Endothermic Reactions 1 of 24 Boardworks Ltd 2016 Exothermic and Endothermic Reactions 2 of 24 Boardworks Ltd 2016 Energy conservation 3 of 24 Boardworks Ltd 2016 Energy cannot be created
Exothermic and Endothermic Reactions 1 of 24 Boardworks Ltd 2016 Exothermic and Endothermic Reactions 2 of 24 Boardworks Ltd 2016 Energy conservation 3 of 24 Boardworks Ltd 2016 Energy cannot be created
Classification of Matter
 Matter in Chemistry Classification of Matter Elements: substances that are made from one type of atom only. Compounds: substances that are made from more than one type of atom chemically bonded together.
Matter in Chemistry Classification of Matter Elements: substances that are made from one type of atom only. Compounds: substances that are made from more than one type of atom chemically bonded together.
Science Module 2: Basic Chemistry. Curriculum Project. Basic Chemistry - 1
 Science Module 2: Basic Chemistry Curriculum Project Basic Chemistry - 1 Contents Chapter 1. Matter Chapter 2. Particles in Matter Chapter 3. Chemical Reactions Chapter 4. Acids, Bases, Salts 1. Matter
Science Module 2: Basic Chemistry Curriculum Project Basic Chemistry - 1 Contents Chapter 1. Matter Chapter 2. Particles in Matter Chapter 3. Chemical Reactions Chapter 4. Acids, Bases, Salts 1. Matter
Chemistry: The Science of Matter. Ch 1
 Chemistry: The Science of Matter Ch 1 Chemistry Science that investigates & explains the structure and properties of matter Matter Anything that takes up space and has mass Mass Amount of matter that
Chemistry: The Science of Matter Ch 1 Chemistry Science that investigates & explains the structure and properties of matter Matter Anything that takes up space and has mass Mass Amount of matter that
LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Counting atoms
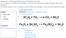 Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Today is: Friday, October 21st
 Today is: Friday, October 21st 10/21/2016 #followyourdreamsfriday 1. What are some of your biggest goals in life? 2. Which of the following is an example of a chemical change? Why? Justify your answer.
Today is: Friday, October 21st 10/21/2016 #followyourdreamsfriday 1. What are some of your biggest goals in life? 2. Which of the following is an example of a chemical change? Why? Justify your answer.
ANSWER KEY. Introduction to Matter
 Chapter Project Worksheet 1 1. The object to be massed is placed on one side of the balance. Objects with known masses are placed on the other side of the arm until the balance arm is level. 2. Sample
Chapter Project Worksheet 1 1. The object to be massed is placed on one side of the balance. Objects with known masses are placed on the other side of the arm until the balance arm is level. 2. Sample
Post-Show. Chemistry. Periodic Table of the Elements. After the Show. Traveling Science Shows
 Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
