Are Chemical Reactions Closed Systems?
|
|
|
- Byron McDonald
- 6 years ago
- Views:
Transcription
1 Measuring a Chemical Reaction 1. Place about 100 ml of vinegar in a large plastic cup. Carefully weigh the cup AND the vinegar all together, and write down the total mass. (Don t worry about subtracting the mass of the cup. It won t matter.) Mass of cup PLUS vinegar g. 2. Carefully weigh about 1 teaspoon of baking soda. You should put the baking soda in a small plastic cup in order to weigh it, but first zero the balance with the empty cup on the balance before you weigh the baking soda. Write down the mass below: Mass of baking soda g. 3. How much would the cup of vinegar weigh if you added the baking soda to it (but the vinegar did not react with the baking soda)? Total mass of the cup of vinegar plus the baking soda before the reaction g. 4. Slowly pour the baking soda into the large cup of vinegar. Just add a little at a time in order to ensure that the liquid does not bubble over the top of the cup. According to the chemical equation: NaHCO 3 + HC 2 NaC 2 + H 2 O + C baking soda acetic acid sodium acetate water carbon in vinegar dioxide What is happening inside the cup? Describe this below in your own words. Make sure you explain why the liquid is bubbling. When the reaction is completely finished, weigh the cup and its contents again, and record the mass. Mass of the cup and contents after the reaction g.
2 Complete the data table below by entering your data and by gathering data from your classmates. Mass of baking soda Total mass of cup and contents before the reaction Mass of cup and contents after the reaction How much mass was lost? What is the ratio of the amount of mass lost divided by the mass of the baking soda? 5. What patterns do you notice in the data table above? What might account for the pattern(s) that you observed? (When you are finished generating a list here, turn to the next page.)
3 Counting Atoms in a Chemical Reaction The mass that you just measured after the reaction is not the same as the amount that you calculated from before the reaction. Why not? What happened to the missing mass? The mass of a single atom is very small. (For example, a one-gram piece of aluminum consists of about 22,000,000,000,000,000,000,000 = 2.2 x 1021 atoms of aluminum.) Since atomic masses are so very small, it is not convenient to measure them using the unit of grams. Instead, we use something else called an atomic mass unit (abbreviated as a.m.u.). In this unit of measurement, a hydrogen atom has a mass of 1 a.m.u. Below you will find a table of some common atoms along with their approximate masses as measured in a.m.u. Atom Hydrogen (H) Carbon (C) Oxygen (O) Sodium (Na) Mass in a.m.u The mass of one water molecule can be found with the use of this table in the following way: Questions: Water (H20) has two hydrogen atoms and one oxygen atom. Mass of H20 = 2 x (mass of H) + 1 x (mass of O) 1. Find the mass of each of the following: Baking soda (NaHCO3) Acetic acid (HC2H3O2) Carbon dioxide (CO2) = 2 x (1 a.m.u.) + 1 x (16 a.m.u.) = 18 a.m.u.
4 The reaction between baking soda and vinegar is written again below: NaHCO 3 + HC 2 NaC 2 + H 2 O + C baking soda acetic acid sodium acetate water carbon in vinegar dioxide This equation indicates that as a baking soda molecule and an acetic acid molecule react, one molecule of carbon dioxide gas is produced. As this happens in your cup, what happens to the carbon dioxide gas that is formed? Where does it go? 2. Suppose you could add one molecule of baking soda to your vinegar. Before the baking soda has time to react with the vinegar, by how much would the mass of your solution increase? 3. By how much will the mass of your solution decrease when a carbon dioxide molecule leaves the cup? 4. Fill in the blanks in this diagram using your answers to #2 and #3 above for two of the blanks. mass units baking soda mass units C 5. What fraction of the original mass of the baking soda leaves the cup? (You can be approximate here.)
5 6. What fraction of the original mass stays in the cup? (You can be approximate here, too.) 7. Use the predictions for mass change that you wrote above, but apply those predictions to a situation where you are considering measurable quantities say gram quantities. If one gram of baking soda is added to a cup of vinegar and allowed to react, what mass change do you predict will occur? 8. Throughout this activity, we assumed that each particle say baking soda or carbon dioxide--had a known mass and we could calculate this mass and make predictions based on these calculated masses. Using evidence from this experiment, how valid is this assumption?
Moles Lab Activity 1: PCU (Popcorn Counting Units)
 Moles Lab Activity 1: PCU (Popcorn Counting Units) Materials: A container of each of the following: Popcorn kernels Another type of beans A large unopened bag of popcorn Kernels Balance Safety goggles
Moles Lab Activity 1: PCU (Popcorn Counting Units) Materials: A container of each of the following: Popcorn kernels Another type of beans A large unopened bag of popcorn Kernels Balance Safety goggles
Modeling Conservation of Matter
 Modeling Conservation of Matter Imagine that you and two of your classmates want to make a strawberry banana smoothie. You lay out the ingredients: one banana, five strawberries and two scoops of ice cream.
Modeling Conservation of Matter Imagine that you and two of your classmates want to make a strawberry banana smoothie. You lay out the ingredients: one banana, five strawberries and two scoops of ice cream.
Stoichiometry Lab Report
 Stoichiometry Lab Report Touche G10 Variables Variable Dependent Controlled: Amount of NaHCO3 Theoretical Yield Vinegar Evaporation Manipulation We measure the remaining NaC 2 H 3 O 2 and compare it with
Stoichiometry Lab Report Touche G10 Variables Variable Dependent Controlled: Amount of NaHCO3 Theoretical Yield Vinegar Evaporation Manipulation We measure the remaining NaC 2 H 3 O 2 and compare it with
Goal: During this lab students will gain a quantitative understanding of limiting reagents.
 LIMITING REAGENT LAB: THE REACTION BETWEEN VINEGAR AND BAKING SODA Goal: During this lab students will gain a quantitative understanding of limiting reagents. Safety: Safety goggles should be worn at all
LIMITING REAGENT LAB: THE REACTION BETWEEN VINEGAR AND BAKING SODA Goal: During this lab students will gain a quantitative understanding of limiting reagents. Safety: Safety goggles should be worn at all
8.1 Chemical Properties and Changes. chemical property chemical change dissolving
 8.1 Chemical Properties and Changes chemical property chemical change dissolving Ability to Change 8.1 Chemical Properties and Changes In a chemical change, the properties that give a substance its identity
8.1 Chemical Properties and Changes chemical property chemical change dissolving Ability to Change 8.1 Chemical Properties and Changes In a chemical change, the properties that give a substance its identity
How many molecules are in 0.25 moles of CH 4?
 Mass Moles- Particle Particles can be atoms, molecules, ions, etc. In one mole of particles, there are 6.02x10 23 particles These particles are so small and we need so many of them to be on a human scale,
Mass Moles- Particle Particles can be atoms, molecules, ions, etc. In one mole of particles, there are 6.02x10 23 particles These particles are so small and we need so many of them to be on a human scale,
Safety: Safety goggles should be worn at all times. Students should hold the balloons on the test tubes tightly while the reaction takes place.
 LIMITING REAGENT LAB: THE REACTION BETWEEN VINEGAR AND BAKING SODA Goal: During this lab students will gain a quantitative understanding of limiting reagents. Safety: Safety goggles should be worn at all
LIMITING REAGENT LAB: THE REACTION BETWEEN VINEGAR AND BAKING SODA Goal: During this lab students will gain a quantitative understanding of limiting reagents. Safety: Safety goggles should be worn at all
Stoichiometry: Baking Soda and Vinegar Reactions Student Version
 Stoichiometry: Baking Soda and Vinegar Reactions Student Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
Stoichiometry: Baking Soda and Vinegar Reactions Student Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
Reactant A (g) Reactant B (ml) Product (ml)
 Pre-Test Key Name: Chemical Reactions Date: 1. The chart below shows the amount of gas that is produced when two reactants (a solid and a liquid) are combined. However, one of the boxes is missing information.
Pre-Test Key Name: Chemical Reactions Date: 1. The chart below shows the amount of gas that is produced when two reactants (a solid and a liquid) are combined. However, one of the boxes is missing information.
Section 1: What is a Chemical Reaction
 Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Stoichiometry World of Chemistry: Chapter 9
 Stoichiometry World of Chemistry: Chapter 9 Chocolate Chip Cookies!! 1 cup butter 1/2 cup white sugar 1 cup packed brown sugar 1 teaspoon vanilla extract 2 eggs 2 1/2 cups all-purpose flour 1 teaspoon
Stoichiometry World of Chemistry: Chapter 9 Chocolate Chip Cookies!! 1 cup butter 1/2 cup white sugar 1 cup packed brown sugar 1 teaspoon vanilla extract 2 eggs 2 1/2 cups all-purpose flour 1 teaspoon
Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version
 Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic
 Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic Key Concepts Carbon dioxide (CO 2 ) gas dissolved in water can cause water to become acidic. The acidity of water from dissolved CO 2 can
Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic Key Concepts Carbon dioxide (CO 2 ) gas dissolved in water can cause water to become acidic. The acidity of water from dissolved CO 2 can
The Chemistry Summary Sheet Chemistry is the scientific study of matter.
 The Chemistry Summary Sheet Chemistry is the scientific study of matter. ATOMIC STRUCTURE: -an atom is composed of three fundamental particles: proton, electron, neutron. -atom size mostly consists of
The Chemistry Summary Sheet Chemistry is the scientific study of matter. ATOMIC STRUCTURE: -an atom is composed of three fundamental particles: proton, electron, neutron. -atom size mostly consists of
How Much Gas is Produced?
 Chemical Dominoes Activity 3 How Much Gas is Produced? CHEM POETRY At the end of the elements trendiest street Was a beaker where just getting in was a feat; One day ol Tony, an atom of tin, Went to Club
Chemical Dominoes Activity 3 How Much Gas is Produced? CHEM POETRY At the end of the elements trendiest street Was a beaker where just getting in was a feat; One day ol Tony, an atom of tin, Went to Club
The masses of reactants and products are equal.
 Page 1 of 7 KEY CNCEPT The masses of reactants and products are equal. BEFRE, you learned Chemical reactions turn reactants into products by rearranging atoms Chemical reactions can be observed and identified
Page 1 of 7 KEY CNCEPT The masses of reactants and products are equal. BEFRE, you learned Chemical reactions turn reactants into products by rearranging atoms Chemical reactions can be observed and identified
Matter. Anything that has mass and takes up space. Solids Liquids Gases
 Introduction to Matter Matter Anything that has mass and takes up space Solids Liquids Gases Weight A measure of the gravitational force exerted on an object What is the difference between weight and mass?
Introduction to Matter Matter Anything that has mass and takes up space Solids Liquids Gases Weight A measure of the gravitational force exerted on an object What is the difference between weight and mass?
Culinary Chemistry: Stoichiometry Made Simple
 Culinary Chemistry: Stoichiometry Made Simple Background Stoichiometry is based on the law of conservation of mass which states that the mass of the reactants in a chemical reaction equals the mass of
Culinary Chemistry: Stoichiometry Made Simple Background Stoichiometry is based on the law of conservation of mass which states that the mass of the reactants in a chemical reaction equals the mass of
Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic
 Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic Name Date DEMONSTRATION 1. Your teacher blew through a straw into a universal indicator solution until it changed color. Did
Activity Sheet Chapter 6, Lesson 10 Carbon Dioxide Can Make a Solution Acidic Name Date DEMONSTRATION 1. Your teacher blew through a straw into a universal indicator solution until it changed color. Did
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions
 Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Stoichiometry: Baking Soda and Vinegar Reactions Student Advanced Version
 Stoichiometry: Baking Soda and Vinegar Reactions Student Advanced Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household
Stoichiometry: Baking Soda and Vinegar Reactions Student Advanced Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household
Chapter 6, Lesson 1: What is a Chemical Reaction?
 Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
LESSON 2. Chemical Equations. When copper sulfate reacts with ammonia, a precipitate forms and the solution color changes to deep blue.
 LESSN 2 Chemical Equations When copper sulfate reacts with ammonia, a precipitate forms and the solution color changes to deep blue. By the end of this lesson... you will be able to explain how chemical
LESSN 2 Chemical Equations When copper sulfate reacts with ammonia, a precipitate forms and the solution color changes to deep blue. By the end of this lesson... you will be able to explain how chemical
Physical or Chemical Change?
 Chemical Formulas and Equations Physical or Chemical Change? Matter can change physically or chemically; a process that produces a chemical change is a chemical reaction. Chemical Formulas and Equations
Chemical Formulas and Equations Physical or Chemical Change? Matter can change physically or chemically; a process that produces a chemical change is a chemical reaction. Chemical Formulas and Equations
2/22/2019 NEW UNIT! Chemical Interactions. Atomic Basics #19
 NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
Limiting Reactants Lab
 Name: Teacher s Name: Class: Block: Date: Partners: Limiting Reactants Lab Purpose: Through experimentation, determine the limiting reactant and the percent yield in a chemical reaction that generates
Name: Teacher s Name: Class: Block: Date: Partners: Limiting Reactants Lab Purpose: Through experimentation, determine the limiting reactant and the percent yield in a chemical reaction that generates
Physical and Chemical Changes Or How Do You Know When You ve Made Something New?
 Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Student Notes. Chemical Reactions LINK
 LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
Bellevue College CHEM& 121 Experiment: Stoichiometric Analysis of an Antacid 1
 Experiment: Stoichiometric Analysis of an Antacid 1 Introduction In this lab, you will use the concept of stoichiometry to solve two sequential problems. First, you will try to determine the products of
Experiment: Stoichiometric Analysis of an Antacid 1 Introduction In this lab, you will use the concept of stoichiometry to solve two sequential problems. First, you will try to determine the products of
LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Counting atoms
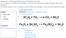 Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Lego Molecules. Standards 1.b. Students know all matter is made of atoms, which may combine to form molecules.
 Science Stars: 5 th grade Lesson Plan Lego Molecules Standards 1.b. Students know all matter is made of atoms, which may combine to form molecules. d. Students know that each element is made of one kind
Science Stars: 5 th grade Lesson Plan Lego Molecules Standards 1.b. Students know all matter is made of atoms, which may combine to form molecules. d. Students know that each element is made of one kind
Standards 8.5.c. I know chemical reactions usually liberate or absorbs heat.
 6 March 2013 Give an example of a physical change and a chemical change, and then describe how they are different from the other. Explain your answer in 2-3 sentences. Standards 8.5.c. I know chemical
6 March 2013 Give an example of a physical change and a chemical change, and then describe how they are different from the other. Explain your answer in 2-3 sentences. Standards 8.5.c. I know chemical
3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make?
 Chapter 4 Test 1. 2. 3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make? packs Work Space: 5. A kennel is moving 160
Chapter 4 Test 1. 2. 3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make? packs Work Space: 5. A kennel is moving 160
DETERMINING AND USING H
 DETERMINING AND USING H INTRODUCTION CHANGES IN CHEMISTRY Chemistry is the science that studies matter and the changes it undergoes. Changes are divided into two categories: physical and chemical. During
DETERMINING AND USING H INTRODUCTION CHANGES IN CHEMISTRY Chemistry is the science that studies matter and the changes it undergoes. Changes are divided into two categories: physical and chemical. During
SNC2D CHEMISTRY 2/23/2013. CHEMICAL REACTIONS L Conservation of Mass (P ) Activity: Measuring Mass (Part 1) Activity: Measuring Mass (Part 1)
 SNC2D CHEMISTRY CHEMICAL REACTIONS L Conservation of Mass (P.176-177) Activity: Measuring Mass (Part 1) INSTRUCTIONS (CLOSED SYSTEM) A. Measure~ 30 ml of vinegar (acetic acid) into a small Erlenmeyer flask.
SNC2D CHEMISTRY CHEMICAL REACTIONS L Conservation of Mass (P.176-177) Activity: Measuring Mass (Part 1) INSTRUCTIONS (CLOSED SYSTEM) A. Measure~ 30 ml of vinegar (acetic acid) into a small Erlenmeyer flask.
Physical & Chemical PROPERTIES
 Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED)
 CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
Conservation of Mass in Chemical Reactions Lab. Experiment Question: What happens to the total mass of substances when a chemical reaction occurs?
 Conservation of Mass in Chemical Reactions Lab Name: 5 th Grade PSI Science Score: / 5 Experiment Question: What happens to the total mass of substances when a chemical reaction occurs? Hypothesis Starters:
Conservation of Mass in Chemical Reactions Lab Name: 5 th Grade PSI Science Score: / 5 Experiment Question: What happens to the total mass of substances when a chemical reaction occurs? Hypothesis Starters:
Complete this study guide to receive 5 bonus points on your test. Only study guides that are complete will receive the bonus.
 CHEMISTRY AND PERIODIC TABLE STUDY GUIDE Assigned: Thursday, 09 10 14 Due: Thursday, 09 18 14 Test Day: Friday, 09 19 14 Complete this study guide to receive 5 bonus points on your test. Only study guides
CHEMISTRY AND PERIODIC TABLE STUDY GUIDE Assigned: Thursday, 09 10 14 Due: Thursday, 09 18 14 Test Day: Friday, 09 19 14 Complete this study guide to receive 5 bonus points on your test. Only study guides
VOCABULARY Define. 1. stoichiometry. 2. composition stoichiometry. 3. reaction stoichiometry. 4. unknown. 5. mole ratio
 CHAPTER 9 HOMEWORK 9-1 (pp. 275 279) Define. 1. stoichiometry 2. composition stoichiometry 3. reaction stoichiometry 4. unknown 5. mole ratio SKILL BUILDER On a separate sheet of paper, write five possible
CHAPTER 9 HOMEWORK 9-1 (pp. 275 279) Define. 1. stoichiometry 2. composition stoichiometry 3. reaction stoichiometry 4. unknown 5. mole ratio SKILL BUILDER On a separate sheet of paper, write five possible
Ch. 6 Chemical Composition - Notetaker (Key)
 6.1 Counting by Weighing Name: Group: Date: Ch. 6 Chemical Composition - Notetaker (Key) Chem 6 Objective: To understand the concept of average mass and explore how counting can be done by _ weighing Average
6.1 Counting by Weighing Name: Group: Date: Ch. 6 Chemical Composition - Notetaker (Key) Chem 6 Objective: To understand the concept of average mass and explore how counting can be done by _ weighing Average
Chemistry. End of Year Cornerstone Assessment
 Chemistry End of Year Cornerstone Assessment The Cornerstone Assessments were developed with support through the VDOE Mathematics and Science Partnership Grant Program NCLB Title II, Part B program by
Chemistry End of Year Cornerstone Assessment The Cornerstone Assessments were developed with support through the VDOE Mathematics and Science Partnership Grant Program NCLB Title II, Part B program by
Lab #9 Stoichiometric Relationships & Theoretical Yield
 Stoichiometric Relationships & Theoretical Yield Name: Nov. 28, 2016 Background and Purpose: Carbonates when reacted with acids will produce carbon dioxide, water and an ionic salt. In this lab, we will
Stoichiometric Relationships & Theoretical Yield Name: Nov. 28, 2016 Background and Purpose: Carbonates when reacted with acids will produce carbon dioxide, water and an ionic salt. In this lab, we will
Activity Sheet Chapter 3, Lesson 3 Density of water
 Activity Sheet Chapter 3, Lesson 3 Density of water Name Date DEMONSTRATION 1. One of your classmates lifted different amounts of water. The largest amount of water also had the most mass. You know how
Activity Sheet Chapter 3, Lesson 3 Density of water Name Date DEMONSTRATION 1. One of your classmates lifted different amounts of water. The largest amount of water also had the most mass. You know how
Moles Lab Activity 2: Elements Copper
 Materials Sample of copper Balance Pre-1982 penny Moles Lab Activity 2: Elements Copper Procedure Take the necessary measurements, and record them with units. Show all your calculations, rounding your
Materials Sample of copper Balance Pre-1982 penny Moles Lab Activity 2: Elements Copper Procedure Take the necessary measurements, and record them with units. Show all your calculations, rounding your
What Do You Think? Investigate GOALS
 Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Chapter 6 Classification of Matter
 Chapter 6 Classification of Matter Pure substances can be classified into two types elements cannot be broken down into other pure substances are made up of only one type of atom only 115 known elements
Chapter 6 Classification of Matter Pure substances can be classified into two types elements cannot be broken down into other pure substances are made up of only one type of atom only 115 known elements
Student Exploration: Chemical Changes
 Name: Date: Student Exploration: Chemical Changes Vocabulary: acid, base, catalyst, chemical change, coefficient, conservation of matter, decomposition, dissolve, double replacement, endothermic, exothermic,
Name: Date: Student Exploration: Chemical Changes Vocabulary: acid, base, catalyst, chemical change, coefficient, conservation of matter, decomposition, dissolve, double replacement, endothermic, exothermic,
Ocean Acidification in a Cup Materials
 Ocean Acidification in a Cup Ocean acidification is a problem that humans will have to deal with as we release more and more carbon dioxide into the atmosphere. This activity demonstrates how water can
Ocean Acidification in a Cup Ocean acidification is a problem that humans will have to deal with as we release more and more carbon dioxide into the atmosphere. This activity demonstrates how water can
5 th Grade Lesson Plan: Matter and Chemical Reactions
 5 th Grade Lesson Plan: Matter and Chemical Reactions Objective: Teach students that matter is neither created nor destroyed during a chemical reaction, rather, it is transformed. Identify evidence that
5 th Grade Lesson Plan: Matter and Chemical Reactions Objective: Teach students that matter is neither created nor destroyed during a chemical reaction, rather, it is transformed. Identify evidence that
Unit VI Stoichiometry. Applying Mole Town to Reactions
 Unit VI Stoichiometry Applying Mole Town to Reactions Learning Goals I can apply mole town to reactions to determine the amount of product based on the amount of a reactant. I can apply mole town to reaction
Unit VI Stoichiometry Applying Mole Town to Reactions Learning Goals I can apply mole town to reactions to determine the amount of product based on the amount of a reactant. I can apply mole town to reaction
Notes on Mole 12.notebook. October 25, CaS. Ca + S. Oct 14 6:22 PM. Oct 14 6:23 PM. Oct 14 6:24 PM. Oct 14 6:23 PM MOLE.
 How do chemists count atoms/formula units/molecules? How do we go from the atomic scale to the scale of everyday measurements (macroscopic scale)? The gateway is the mole! But before we get to the mole,
How do chemists count atoms/formula units/molecules? How do we go from the atomic scale to the scale of everyday measurements (macroscopic scale)? The gateway is the mole! But before we get to the mole,
STOICHIOMETRY. Engr. Yvonne Ligaya F. Musico 1
 STOICHIOMETRY Engr. Yvonne Ligaya F. Musico 1 Stoichiometry The study in chemistry dealing with calculations based on balanced chemical equations. The branch of chemistry dealing with mass relationships
STOICHIOMETRY Engr. Yvonne Ligaya F. Musico 1 Stoichiometry The study in chemistry dealing with calculations based on balanced chemical equations. The branch of chemistry dealing with mass relationships
Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from
 Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from http://www.csun.edu/chemteach/activities/beginninganalysis.pdf ] In this lab, you will be observing physical and chemical properties
Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from http://www.csun.edu/chemteach/activities/beginninganalysis.pdf ] In this lab, you will be observing physical and chemical properties
Stoichiometry Decomposition of Baking Soda For our soul is humbled down to the dust. Psalms 43:25
 Stoichiometry Decomposition of Baking Soda For our soul is humbled down to the dust. Psalms 43:25 Introduction A balanced chemical reaction shows products forming reactants such that the Law of Conservation
Stoichiometry Decomposition of Baking Soda For our soul is humbled down to the dust. Psalms 43:25 Introduction A balanced chemical reaction shows products forming reactants such that the Law of Conservation
Safety and Types of Fires
 Cookin' Chem Activity 2 Safety and Types of Fires CHEM POETRY Oliver learned all those Don t s and Do s, The ones that get passed down through moles of O 2 s; Each new generation did soon learn to trust
Cookin' Chem Activity 2 Safety and Types of Fires CHEM POETRY Oliver learned all those Don t s and Do s, The ones that get passed down through moles of O 2 s; Each new generation did soon learn to trust
Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from
 Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from http://www.csun.edu/chemteach/activities/beginninganalysis.pdf ] In this lab, you will be observing physical and chemical properties
Name: Date: Period: Identifying & Analyzing Powders Lab [Adapted from http://www.csun.edu/chemteach/activities/beginninganalysis.pdf ] In this lab, you will be observing physical and chemical properties
PREPARE FOR THE ACTIVITY. Activity Sheet Chapter 6, Lesson 8 ph and Color Change
 Activity Sheet Chapter 6, Lesson 8 ph and Color Change Name Date DEMONSTRATION 1. Your teacher poured green universal indicator into each of two cups. What does the change in color of the indicator solution
Activity Sheet Chapter 6, Lesson 8 ph and Color Change Name Date DEMONSTRATION 1. Your teacher poured green universal indicator into each of two cups. What does the change in color of the indicator solution
Period 11: Chemical Energy and Fossil Fuels
 Name Section Period 11: Chemical Energy and Fossil Fuels 11.1 What is the Composition of Matter? 1) Atoms a) What are the building blocks of an atom? b) Which nucleons make up an atomic nucleus? c) What
Name Section Period 11: Chemical Energy and Fossil Fuels 11.1 What is the Composition of Matter? 1) Atoms a) What are the building blocks of an atom? b) Which nucleons make up an atomic nucleus? c) What
Characteristics of Chemical Change
 Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Unit 3.3 Test: Basic Chemistry
 Unit 3.3 Test: Basic Chemistry 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 1. A student placed 2 grams of baking soda in a balloon and
Unit 3.3 Test: Basic Chemistry 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 1. A student placed 2 grams of baking soda in a balloon and
Physical Changes vs Chemical Changes Lab
 Physical Changes vs Chemical Changes Lab What was done? What can you observe? Is it a physical or chemical change and why? What else have you learned during the demonstration? #1) Crumpling Paper Notice
Physical Changes vs Chemical Changes Lab What was done? What can you observe? Is it a physical or chemical change and why? What else have you learned during the demonstration? #1) Crumpling Paper Notice
Online Resource 10. December 2016
 Designing for the Next Generation Standards: Educative Curriculum Materials and Measures of Teacher Knowledge Online Resource 10 December 2016 Toward High School Biology: Tables 1-4 provide evidence that
Designing for the Next Generation Standards: Educative Curriculum Materials and Measures of Teacher Knowledge Online Resource 10 December 2016 Toward High School Biology: Tables 1-4 provide evidence that
Lab 2-Investigating the Law of Conservation of Mass
 Name: Period: Lab 2-Investigating the Law of Conservation of Mass Objective: To corroborate the law of conservation of mass through laboratory experimentation. Background: When wood burns or water evaporates,
Name: Period: Lab 2-Investigating the Law of Conservation of Mass Objective: To corroborate the law of conservation of mass through laboratory experimentation. Background: When wood burns or water evaporates,
Chemistry 143 Experiment #11 Acid Base Titration Dr. Caddell. Titrating Acid
 Titrating Acid In this lab you will first determine the concentration of sodium hydroxide in a stock solution that you prepare. You will then use that stock sodium hydroxide solution to titrate a solution
Titrating Acid In this lab you will first determine the concentration of sodium hydroxide in a stock solution that you prepare. You will then use that stock sodium hydroxide solution to titrate a solution
Name Period Date. Lab 1: Mass of Ice Materials: beaker, ice and balance.
 Name Period Date Testing the Law of Conservation of MASS! Introduction: Does mass change in a chemical or physical reaction? In this series of experiments you will find the answer to this question. Lab
Name Period Date Testing the Law of Conservation of MASS! Introduction: Does mass change in a chemical or physical reaction? In this series of experiments you will find the answer to this question. Lab
WORKING WITH MOLES LAB STATION #1: AVERAGE MASS OF PLASTIC BAGS
 WORKING WITH MOLES LAB STATION #1: AVERAGE MASS OF PLASTIC BAGS Determine and record the mass of three different plastic bags with labels. Calculate the average mass of one plastic bag. *Bag #1 grams *Bag
WORKING WITH MOLES LAB STATION #1: AVERAGE MASS OF PLASTIC BAGS Determine and record the mass of three different plastic bags with labels. Calculate the average mass of one plastic bag. *Bag #1 grams *Bag
Unit 9 The Mole Chapter 10 of your textbook
 Unit 9 The Mole Chapter 10 of your textbook Learning Targets for Unit 9 Early Booklet E.C.: + 2 Unit 9.A Hwk. Pts.: / 36 Unit 9.A Lab Pts.: / 32 Late, Incomplete, No Work, No Units Fees? Y / N 1.1 I can
Unit 9 The Mole Chapter 10 of your textbook Learning Targets for Unit 9 Early Booklet E.C.: + 2 Unit 9.A Hwk. Pts.: / 36 Unit 9.A Lab Pts.: / 32 Late, Incomplete, No Work, No Units Fees? Y / N 1.1 I can
CHM 130 Acid-Base Titration Molarity of Acetic Acid in Vinegar
 CHM 130 Acid-Base Titration Molarity of Acetic Acid in Vinegar INTRODUCTION One of the most important techniques for chemical analysis is titration to an equivalence point. To illustrate this procedure,
CHM 130 Acid-Base Titration Molarity of Acetic Acid in Vinegar INTRODUCTION One of the most important techniques for chemical analysis is titration to an equivalence point. To illustrate this procedure,
Gas Laws. by Dawn Richardson PhD, Collin College, Preston Ridge Campus; revised by Jim Sizemore PhD, Collin College, Central Park Campus
 Gas Laws by Dawn Richardson PhD, Collin College, Preston Ridge Campus; revised by Jim Sizemore PhD, Collin College, Central Park Campus Introduction: A gas is a state of matter in which atoms or molecules
Gas Laws by Dawn Richardson PhD, Collin College, Preston Ridge Campus; revised by Jim Sizemore PhD, Collin College, Central Park Campus Introduction: A gas is a state of matter in which atoms or molecules
MEASURING THE RATE OF PHOTOSYNTHESIS
 MEASURING THE RATE OF PHOTOSYNTHESIS QUESTION: What factors affect the rate of photosynthesis in plants? BACKGROUND KNOWLEDGE Leaf disks float, normally. When the air spaces in the leaf are filled with
MEASURING THE RATE OF PHOTOSYNTHESIS QUESTION: What factors affect the rate of photosynthesis in plants? BACKGROUND KNOWLEDGE Leaf disks float, normally. When the air spaces in the leaf are filled with
Name: 1. Which of the following is probably true about 300 ml of sand and 300 ml of water?
 Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
WORKING WITH MOLES LAB. *Bag #1 *Bag #2 *Bag #3 Average
 WORKING WITH MOLES LAB In chemistry, the fundamental unit of matter is the mole. You must understand and be able to use the mole concept. In this activity, you will practice determining the number of moles,
WORKING WITH MOLES LAB In chemistry, the fundamental unit of matter is the mole. You must understand and be able to use the mole concept. In this activity, you will practice determining the number of moles,
Experiment 20: Analysis of Vinegar. Materials:
 Experiment 20: Analysis of Vinegar Materials: graduated cylinder 6 M NaOH: Dilute Sodium Hydroxide 1000 ml Florence Flask & stopper KHC 8 H 4 O 4 : Potassium Hydrogen Phthalate (KHP) 125 ml Erlenmeyer
Experiment 20: Analysis of Vinegar Materials: graduated cylinder 6 M NaOH: Dilute Sodium Hydroxide 1000 ml Florence Flask & stopper KHC 8 H 4 O 4 : Potassium Hydrogen Phthalate (KHP) 125 ml Erlenmeyer
Pure Substances and Mixtures
 Pure Substances and Mixtures Unit 1 Lesson 4 Mix It Up! Make two mixtures in transparent containers with lids. 1. Find two transparent containers with tight lids. In one container, mix either salt or sugar
Pure Substances and Mixtures Unit 1 Lesson 4 Mix It Up! Make two mixtures in transparent containers with lids. 1. Find two transparent containers with tight lids. In one container, mix either salt or sugar
A simple equation of what happens when you add baking soda to vinegar:
 What s the Matter? Matter is anything that takes up space and has mass. Mass is the stuff that matter is made of, or the amount of particles in a substance or object. Matter has physical and chemical properties
What s the Matter? Matter is anything that takes up space and has mass. Mass is the stuff that matter is made of, or the amount of particles in a substance or object. Matter has physical and chemical properties
Stoichiometry study of the relationships in a
 Note Taking Guide: Episode 801 Name Stoichiometry study of the relationships in a based on equations 2 Mg + O 2 2 MgO The in a give the for the involved in the. Ex. Problem: When elemental aluminum reacts
Note Taking Guide: Episode 801 Name Stoichiometry study of the relationships in a based on equations 2 Mg + O 2 2 MgO The in a give the for the involved in the. Ex. Problem: When elemental aluminum reacts
o Test tube In this experiment, you ll be observing the signs of chemical reactions. These include the following:
 Experiment: Chemical Reactions & Chemical s Objective In this experiment, students perform a variety of chemical reactions. For each reaction, student identify the signs that a reaction has occurred, write
Experiment: Chemical Reactions & Chemical s Objective In this experiment, students perform a variety of chemical reactions. For each reaction, student identify the signs that a reaction has occurred, write
Experiment 17 It s A Gas and More!
 Energy Energy Experiment 17 It s A Gas and More! OUTCOMES After completing this lab activity, the student should be able to: explain a simple method for distinguishing carbon dioxide gas from oxygen gas.
Energy Energy Experiment 17 It s A Gas and More! OUTCOMES After completing this lab activity, the student should be able to: explain a simple method for distinguishing carbon dioxide gas from oxygen gas.
Name Period Date. Lab: Introduction to Stoichiometry
 Name Period Date Lab: Introduction to Stoichiometry Introduction: Reactants are not always present in the exact ratio required by a balanced chemical equation. In planning any cost-effective production
Name Period Date Lab: Introduction to Stoichiometry Introduction: Reactants are not always present in the exact ratio required by a balanced chemical equation. In planning any cost-effective production
Picture This. Reading Essentials 337. Before burning. After burning
 chapter 19 Chemical Reactions section 1 Chemical Changes What You ll Learn how to identify the reactants and products in a chemical reaction how a chemical reaction follows the law of conservation of mass
chapter 19 Chemical Reactions section 1 Chemical Changes What You ll Learn how to identify the reactants and products in a chemical reaction how a chemical reaction follows the law of conservation of mass
Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds.
 Chapter 11 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
Chapter 11 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
Chem 2115 Experiment #7. Volumetric Analysis & Consumer Chemistry Standardization of an unknown solution, analysis of vinegar & antacid tablets
 Chem 2115 Experiment #7 Volumetric Analysis & Consumer Chemistry Standardization of an unknown solution, analysis of vinegar & antacid tablets OBJECTIVE: The goals of this experiment are to learn titration
Chem 2115 Experiment #7 Volumetric Analysis & Consumer Chemistry Standardization of an unknown solution, analysis of vinegar & antacid tablets OBJECTIVE: The goals of this experiment are to learn titration
Chapter 2. Section 1
 Chapter 2 Section 1 Describing Matter Properties used to describe matter can be classified as extensive or intensive Extensive property - depends on the amount of matter in a sample. Ex. Mass and Volume
Chapter 2 Section 1 Describing Matter Properties used to describe matter can be classified as extensive or intensive Extensive property - depends on the amount of matter in a sample. Ex. Mass and Volume
In this laboratory exercise we will determine the percentage Acetic Acid (CH 3 CO 2 H) in Vinegar.
 The titration of Acetic Acid in Vinegar In this laboratory exercise we will determine the percentage Acetic Acid (CH CO H) in Vinegar. We will do this by Titrating the Acetic Acid present with a Strong
The titration of Acetic Acid in Vinegar In this laboratory exercise we will determine the percentage Acetic Acid (CH CO H) in Vinegar. We will do this by Titrating the Acetic Acid present with a Strong
I hope you aren't going to tear open the package and count the nails. We agree that. mass of nails = 1340 g g = g
 The Mole Concept Counting by weighing The size of molecule is so small that it is physically difficult if not impossible to directly count out molecules. this problem is solved using a common trick. Atoms
The Mole Concept Counting by weighing The size of molecule is so small that it is physically difficult if not impossible to directly count out molecules. this problem is solved using a common trick. Atoms
Part 1: Matter. Chapter 1: Matter, Measurements, and Calculations. Sections MATTER Matter is anything that has mass and occupies space.
 Part 1: Matter Chapter 1: Matter, Measurements, and Calculations Sections 1.1-1.4 1 2 MATTER Matter is anything that has mass and occupies space. MASS Mass is a measurement of the amount of matter in an
Part 1: Matter Chapter 1: Matter, Measurements, and Calculations Sections 1.1-1.4 1 2 MATTER Matter is anything that has mass and occupies space. MASS Mass is a measurement of the amount of matter in an
More Chemical Changes
 Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Front page = 1 point, Questions on back = 1 point each, Mastery Question = 1 point DENSITY OF SOLIDS Sink or Float?
 Last name First name Date Period Front page = 1 point, Questions on back = 1 point each, Mastery Question = 1 point DENSITY OF SOLIDS Sink or? Question to investigate Why does a heavier candle float and
Last name First name Date Period Front page = 1 point, Questions on back = 1 point each, Mastery Question = 1 point DENSITY OF SOLIDS Sink or? Question to investigate Why does a heavier candle float and
General Physical Science. Chemical and Physical Properties. Chemical and Physical Properties. Chapter 13 Chemical Reactions. Physical properties
 General Physical Science Chapter 13 Chemical Reactions Chemical and Physical Properties Physical properties Observations about a substance changes that do not involve a change in the arrangement of the
General Physical Science Chapter 13 Chemical Reactions Chemical and Physical Properties Physical properties Observations about a substance changes that do not involve a change in the arrangement of the
Changes in Matter. 2. Chemical Changes
 PSC 36 Changes in Matter There are three kinds of changes that matter can undergo: hanges C l a c i. Phys. Chemical Changes 1 3. Nuc lear C hange s Important for you: understand the basic differences between
PSC 36 Changes in Matter There are three kinds of changes that matter can undergo: hanges C l a c i. Phys. Chemical Changes 1 3. Nuc lear C hange s Important for you: understand the basic differences between
Chemistry 143 Acid Base Titration Dr. Caddell. Titrating Acid
 Titrating Acid In this lab you will first determine the concentration of sodium hydroxide in a stock solution that you prepare. You will then use that stock sodium hydroxide solution to titrate a solution
Titrating Acid In this lab you will first determine the concentration of sodium hydroxide in a stock solution that you prepare. You will then use that stock sodium hydroxide solution to titrate a solution
1. Read the following article and fill in the vocabulary crossword. An Atom Apart: by Leslie Cargile
 Grade 6 Science Summer Work International School of Arts and Sciences ISAS 2012 2013 Name: 1. Read the following article and fill in the vocabulary crossword. An Atom Apart: by Leslie Cargile Have you
Grade 6 Science Summer Work International School of Arts and Sciences ISAS 2012 2013 Name: 1. Read the following article and fill in the vocabulary crossword. An Atom Apart: by Leslie Cargile Have you
Liquid X Lab. Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties?
 Names Block Date Liquid X Lab Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette to carefully place drops of each liquid
Names Block Date Liquid X Lab Station 1 The Penny Lab Water is cohesive, adhesive, and has surface tension. Does Liquid X have the same properties? 1. Use a pipette to carefully place drops of each liquid
EXTRA CREDIT REMINDER
 EXTRA CREDIT REMINDER Due Tonight at Midnight (January 21 at 11:59 pm) via email kimberlyn.jackson@hcbe.net *** Kinesthetic: If you do not know how to use Prezi you may do a power point otherwise email
EXTRA CREDIT REMINDER Due Tonight at Midnight (January 21 at 11:59 pm) via email kimberlyn.jackson@hcbe.net *** Kinesthetic: If you do not know how to use Prezi you may do a power point otherwise email
Maintaining Mass Created By: Allyn Short
 Maintaining Mass Created By: Allyn Short Focus on Inquiry The student will demonstrate that mass is conserved when substances undergo chemical and/or physical changes through experimentation and evaluation
Maintaining Mass Created By: Allyn Short Focus on Inquiry The student will demonstrate that mass is conserved when substances undergo chemical and/or physical changes through experimentation and evaluation
Section I: Synthesis reactions Synthesis reactions occur when two or more substances come together to form a single new substance.
 TYPES OF CHEMICAL REACTIONS A Laboratory Investigation Purpose: Observe the five major types of reactions. Record observations for these reactions. Complete balanced equations for these reactions. Introduction:
TYPES OF CHEMICAL REACTIONS A Laboratory Investigation Purpose: Observe the five major types of reactions. Record observations for these reactions. Complete balanced equations for these reactions. Introduction:
Copyright 2016 Edmentum - All rights reserved.
 Copyright 2016 Edmentum - All rights reserved. SI: Quiz 5 Question #1 Which of the following is true about the mass of the reactants in a chemical reaction? The total mass of the reactants in a chemical
Copyright 2016 Edmentum - All rights reserved. SI: Quiz 5 Question #1 Which of the following is true about the mass of the reactants in a chemical reaction? The total mass of the reactants in a chemical
MONDAY (12/12) TUESDAY (12/13) WEDNESDAY (12/14) THURSDAY (12/15) FRIDAY (12/16) Making Acid Rain (a lab) Quiz
 Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
Chapter 3. Mass Relationships in Chemical Reactions
 Chapter 3 Mass Relationships in Chemical Reactions In this chapter, Chemical structure and formulas in studying the mass relationships of atoms and molecules. To explain the composition of compounds and
Chapter 3 Mass Relationships in Chemical Reactions In this chapter, Chemical structure and formulas in studying the mass relationships of atoms and molecules. To explain the composition of compounds and
