Chemical Reactions. Chapter Test A. Multiple Choice. 1 Pearson Education, Inc., or its affiliates. All rights reserved.
|
|
|
- Franklin McDowell
- 5 years ago
- Views:
Transcription
1 Name Date Class Chemical Reactions Chapter Test A Multiple Choice Write the letter of the correct answer on the line at the left. 1. Chemistry is a. a characteristic of a substance that can be observed without changing the substance into another substance. b. the study of matter and how matter changes. c. anything that has mass and takes up space. d. a rapid reaction between oxygen and a substance called a fuel. 2. The only sure evidence for a chemical reaction is a. the formation of a gas. b. a color change. c. the production of one or more new substances. d. changes in properties. 3. A material used to decrease the rate of a chemical reaction is a(n) a. inhibitor. b. catalyst. c. enzyme. d. fuel. 4. CaCO 3 represents a chemical a. symbol. b. formula. c. subscript. d. reaction. 5. Which is the correct chemical equation for the following statement? Sodium reacts with oxygen in a 2 to 1 ratio to produce sodium oxide. a. 4 Na + O 2 2 Na 2 O b. 2 Na + O 2 2 NaO c. Na 2 O + O 2 2 NaO d. Na 2 + O 2 Na 2 O 2 6. The principle of conservation of mass is true a. only for reactions that take place in open systems. b. only for reactions that take place in closed systems. c. for reactions in both open and closed systems. d. for no reactions at all. 7. In chemical reactions, what does the principle of conservation of mass mean? a. Matter is not created or destroyed. b. The total mass of the reactants is greater than the total mass of the products. c. The total mass of the reactants is less than the total mass of the products. d. Matter is not changed. 1
2 Name Date Class 8. Chemicals that act as biological catalysts by speeding up reactions in living things are called a. inhibitors. b. enzymes. c. fuels. d. reactants. 9. Which of the following is an example of how to supply activation energy to begin a reaction? a. Cool the reaction flask in an b. Add a catalyst. ice bath. c. Heat the reaction flask on a d. Add an inhibitor. hot plate. 10. A bottle of hydrogen peroxide that eventually turns into a bottle of water and oxygen gas is an example of a a. synthesis reaction. b. decomposition reaction. c. replacement reaction. d. precipitate reaction. Completion Fill in the line to complete each statement. 11. A precipitate is a(n) that forms from solution during a chemical reaction. 12. In a chemical equation, the substances written on the right side of the arrow are called. 13. The _ is the minimum amount of energy that must be added to start a chemical reaction. 14. A mistake in the design of an experiment that makes a certain result more likely is called experimental. 15. A reaction in which one element replaces another in a compound or in which two elements in different compounds trade places is called a(n) reaction. True or False If the statement is true, write true. If it is false, change the underlined word or words to make the statement true. 16. Physical changes occur when existing bonds break and new bonds form. 17. The amount of matter in a chemical reaction changes. 18. Synthesis is the process of making a more complex compound from simpler substances. 19. Subjective reasoning relies on gathering and evaluating evidence. 20. The rate of a chemical reaction is independent of temperature. 21. A chemical reaction may be detected by observing changes in the properties of matter. 2
3 Name Date Class Using Science Skills Use the diagram to answer each question. 22. When the candle was lit, a pool of liquid wax formed in the area around the wick, and then spilled over the side and solidified. Does this observation refer to a physical change or a chemical change? Explain. 23. If the products formed from the burning candle are mostly carbon (C), carbon dioxide (CO 2 ), and water (H 2 O), what elements were in the reactants? How do you know? 24. Is the reaction that occurs in the diagram endothermic or exothermic? Explain. 3
4 Name Date Class Essay Write an answer for each of the following questions on a separate sheet of paper. 25. Explain which bonds are broken and which bonds are formed during the reaction of magnesium and oxygen to form magnesium oxide. Identify which substances are compounds and which are elements. 2 Mg + O 2 2 MgO 26. For each situation described below, tell whether the reaction is endothermic or exothermic. Explain your reasoning in terms of the energy of the reactants and products. A) A log burns in a fireplace, giving off gases and leaving behind ash. B) When vinegar and baking soda are mixed, new substances form and the solution becomes cooler. Using Science Skills Use the diagram to answer each question. 27. What type of chemical reaction is represented in the diagram? Explain. 28. What is the purpose of the burning splint in the reaction? 29. What is the first rule you should follow if you have an accident while doing this lab? 4
5 Name Date Class Essay Write an answer for each of the following questions on a separate sheet of paper. 30. Relate the writing and balancing of a chemical equation to the principle of conservation of mass. 31. Describe the difference between a synthesis reaction and a decomposition reaction. 32. Explain each of the following in terms of factors that affect the rate of a chemical reaction. A) Chemical reactions in living things would not be possible without the enzymes found in cells. B) Wood burns at a steady rate, but sawdust may explode if a spark ignites it. (Sawdust is tiny particles of wood made from sawing wood.) 5
Choose the answer that best completes each statement.
 Name Date CHAPTER TEST: CHEMICAL BONDS AND EQUATIONS Multiple Choice Choose the answer that best completes each statement. 1. Magnesium bromide is an ionic compound with the chemical formula MgBr2. What
Name Date CHAPTER TEST: CHEMICAL BONDS AND EQUATIONS Multiple Choice Choose the answer that best completes each statement. 1. Magnesium bromide is an ionic compound with the chemical formula MgBr2. What
Chemical Reactions Chapter 2 L book pages L44 - L73. examples?
 Name: Period: Chemical Reactions Chapter 2 L book pages L44 - L73 Vocabulary Word What is this? (definition) What are some examples? What does it look like? (draw a picture or diagram) Physical property
Name: Period: Chemical Reactions Chapter 2 L book pages L44 - L73 Vocabulary Word What is this? (definition) What are some examples? What does it look like? (draw a picture or diagram) Physical property
Chapter Introduction Lesson 1 Understanding Chemical Reactions Lesson 2 Types of Chemical Reactions Lesson 3 Energy Changes and Chemical Reactions
 Chapter Introduction Lesson 1 Understanding Chemical Reactions Lesson 2 Types of Chemical Reactions Lesson 3 Energy Changes and Chemical Reactions Chapter Wrap-Up Changes in Matter A physical change does
Chapter Introduction Lesson 1 Understanding Chemical Reactions Lesson 2 Types of Chemical Reactions Lesson 3 Energy Changes and Chemical Reactions Chapter Wrap-Up Changes in Matter A physical change does
Chemical formula - tells you how many atoms of each element are in a compound example: CO 2 (carbon dioxide) has one carbon atom and two oxygen atoms
 Chemical Reactions Chemical formula - tells you how many atoms of each element are in a compound example: CO 2 (carbon dioxide) has one carbon atom and two oxygen atoms 2 points Chemical Equation - a short,
Chemical Reactions Chemical formula - tells you how many atoms of each element are in a compound example: CO 2 (carbon dioxide) has one carbon atom and two oxygen atoms 2 points Chemical Equation - a short,
Physical Change - alters the form or appearance of a substance but does not change it into a new, different substance
 Chemical Reactions Physical Change - alters the form or appearance of a substance but does not change it into a new, different substance Chemical Change - forms one or more new substances with properties
Chemical Reactions Physical Change - alters the form or appearance of a substance but does not change it into a new, different substance Chemical Change - forms one or more new substances with properties
Ch & 12 Test. Name: Class: Date: Multiple Choice Identify the choice that best completes the statement or answers the question.
 Name: Class: Date: Ch 10 11 & 12 Test Multiple Choice Identify the choice that best completes the statement or answers the question. 1. How many valence electrons does Argon, a noble gas, have? a. 1 b.
Name: Class: Date: Ch 10 11 & 12 Test Multiple Choice Identify the choice that best completes the statement or answers the question. 1. How many valence electrons does Argon, a noble gas, have? a. 1 b.
Describing Chemical Reactions
 Describing Chemical Reactions This section explains how to show chemical reactions with symbols. It also states the principle of conservation of mass, and identifies three categories of chemical reactions.
Describing Chemical Reactions This section explains how to show chemical reactions with symbols. It also states the principle of conservation of mass, and identifies three categories of chemical reactions.
Describing Chemical Reactions
 Name Date Class Chemical Reactions Guided Reading and Study Describing Chemical Reactions This section explains how to show chemical reactions with symbols. It also states the principle of conservation
Name Date Class Chemical Reactions Guided Reading and Study Describing Chemical Reactions This section explains how to show chemical reactions with symbols. It also states the principle of conservation
Chemical Reactions Chapter 17
 Chemical Reactions Chapter 17 I. Physical Change Changes which affect the size or shape of the substance BUT NOT its chemical properties or formulas. e.g.; ice melting. Water is still water II. Chemical
Chemical Reactions Chapter 17 I. Physical Change Changes which affect the size or shape of the substance BUT NOT its chemical properties or formulas. e.g.; ice melting. Water is still water II. Chemical
Chapter 14 Study Questions Name: Sci Class:
 Chapter 14 Study Questions Name: Sci Class: Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. Balancing a chemical equation so that the same
Chapter 14 Study Questions Name: Sci Class: Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. Balancing a chemical equation so that the same
Section 1: What is a Chemical Reaction
 Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Chapter 7: Chemical Reactions
 Chapter 7: Chemical Reactions Physical Science C. Goodman, Doral Academy Charter High School, 2012-2013 Based on a presentation by Holt, Rinehart and Winston Section 7.1 Essential questions 1. What is
Chapter 7: Chemical Reactions Physical Science C. Goodman, Doral Academy Charter High School, 2012-2013 Based on a presentation by Holt, Rinehart and Winston Section 7.1 Essential questions 1. What is
Physical Change - alters the form or appearance of a substance but does not change it into a new, different substance
 Chemical Reactions Physical Change - alters the form or appearance of a substance but does not change it into a new, different substance Chemical Change (aka chemical reaction) - forms one or more new
Chemical Reactions Physical Change - alters the form or appearance of a substance but does not change it into a new, different substance Chemical Change (aka chemical reaction) - forms one or more new
Chemical Reactions. Section 7.1: Nature of Reactions
 Chemical Reactions Section 7.1: Nature of Reactions When do chemical reactions take place? What is the role of energy in chemical reactions? 1 Chemical Reactions It is a change in matter that produces
Chemical Reactions Section 7.1: Nature of Reactions When do chemical reactions take place? What is the role of energy in chemical reactions? 1 Chemical Reactions It is a change in matter that produces
Student Exploration: Chemical Changes
 Name: Date: Student Exploration: Chemical Changes Vocabulary: acid, base, catalyst, chemical change, coefficient, conservation of matter, decomposition, dissolve, double replacement, endothermic, exothermic,
Name: Date: Student Exploration: Chemical Changes Vocabulary: acid, base, catalyst, chemical change, coefficient, conservation of matter, decomposition, dissolve, double replacement, endothermic, exothermic,
Student Notes. Chemical Reactions LINK
 LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
Observing Chemical Change - 5.1
 Observing Chemical Change - 5.1 Vocabulary: Physical change - Chemical change - Reactant Product Precipitate Exothermic reaction - Endothermic reaction - Properties of matter: Two kinds of properties of
Observing Chemical Change - 5.1 Vocabulary: Physical change - Chemical change - Reactant Product Precipitate Exothermic reaction - Endothermic reaction - Properties of matter: Two kinds of properties of
Chapter 7 Chemical Reactions
 Chapter 7 Chemical Reactions Chemical Equation --> is a representation of a chemical reaction in which the reactants and products are expressed as formulas Reactants --> substances that undergo change
Chapter 7 Chemical Reactions Chemical Equation --> is a representation of a chemical reaction in which the reactants and products are expressed as formulas Reactants --> substances that undergo change
Physical or Chemical Change?
 Chemical Formulas and Equations Physical or Chemical Change? Matter can change physically or chemically; a process that produces a chemical change is a chemical reaction. Chemical Formulas and Equations
Chemical Formulas and Equations Physical or Chemical Change? Matter can change physically or chemically; a process that produces a chemical change is a chemical reaction. Chemical Formulas and Equations
Chemical Formulas and Equations
 Chemical Formulas and Equations 8.5D recognize that chemical formulas are used to identify substances and determine the number of atoms of each element in chemical formulas containing substances; 8.5F
Chemical Formulas and Equations 8.5D recognize that chemical formulas are used to identify substances and determine the number of atoms of each element in chemical formulas containing substances; 8.5F
1 The Nature of Chemical Reactions
 CHAPTER 7 1 The Nature of Chemical Reactions SECTION Chemical Reactions KEY IDEAS As you read this section, keep these questions in mind: What happens in a chemical reaction? What is the role of energy
CHAPTER 7 1 The Nature of Chemical Reactions SECTION Chemical Reactions KEY IDEAS As you read this section, keep these questions in mind: What happens in a chemical reaction? What is the role of energy
Chemical Symbols & Formulas
 + Chemical Symbols & Formulas + Chemical Symbols All elements in the Periodic Table have symbols that are recognized world wide. It does not matter which country you are in or the language you speak. Example:
+ Chemical Symbols & Formulas + Chemical Symbols All elements in the Periodic Table have symbols that are recognized world wide. It does not matter which country you are in or the language you speak. Example:
Section 1 Chemical Changes
 Chemical Reactions Section 1 Chemical Changes What You ll Learn: How to identify the reactants & products in a chemical reaction How a chemical reaction follows the law of conservation of mass How chemists
Chemical Reactions Section 1 Chemical Changes What You ll Learn: How to identify the reactants & products in a chemical reaction How a chemical reaction follows the law of conservation of mass How chemists
Chemical Reactions. Chemical changes are occurring around us all the time
 Chemical changes are occurring around us all the time Food cooking Fuel being burned in a car s engine Oxygen being used in the human body The starting materials are called reactants The ending materials
Chemical changes are occurring around us all the time Food cooking Fuel being burned in a car s engine Oxygen being used in the human body The starting materials are called reactants The ending materials
Characteristics & Types of Chemical Reactions
 Characteristics & Types of Chemical Reactions What is a chemical reaction? Another name for a chemical change Bonds break and atoms of one or more substances are rearranged and new bonds form New substance(s)
Characteristics & Types of Chemical Reactions What is a chemical reaction? Another name for a chemical change Bonds break and atoms of one or more substances are rearranged and new bonds form New substance(s)
What does rate of reaction mean?
 Junior Science What does rate of reaction mean? It is not how much of a product is made, but instead how quickly a reaction takes place. The speed of a reaction is called the rate of the reaction. What
Junior Science What does rate of reaction mean? It is not how much of a product is made, but instead how quickly a reaction takes place. The speed of a reaction is called the rate of the reaction. What
PHYSICAL & CHEMICAL CHANGES
 PHYSICAL & CHEMICAL CHANGES The characteristics of a substance are called its properties. Physical properties are those that describe the matter without changing its composition. Examples are density,
PHYSICAL & CHEMICAL CHANGES The characteristics of a substance are called its properties. Physical properties are those that describe the matter without changing its composition. Examples are density,
Chapter 2. Section 1
 Chapter 2 Section 1 Describing Matter Properties used to describe matter can be classified as extensive or intensive Extensive property - depends on the amount of matter in a sample. Ex. Mass and Volume
Chapter 2 Section 1 Describing Matter Properties used to describe matter can be classified as extensive or intensive Extensive property - depends on the amount of matter in a sample. Ex. Mass and Volume
Pre-Lab Exercises Lab 3: Chemical Properties
 Pre-Lab Exercises Lab 3: Chemical Properties 1. How is a chemical property different from a physical property? Name Date Section 2. How is a chemical change different from a physical change? 3. Give two
Pre-Lab Exercises Lab 3: Chemical Properties 1. How is a chemical property different from a physical property? Name Date Section 2. How is a chemical change different from a physical change? 3. Give two
Chemical Reactions. Chemical Reaction occur when atoms collide. Reaction rates change from increasing or decreasing collisions
 Chemical Reactions Questions 1. What is a chemical reaction 2. What evidence suggests that a chemical reaction has taken place? 3. What factors can affect the rate of a chemical reaction (how fast the
Chemical Reactions Questions 1. What is a chemical reaction 2. What evidence suggests that a chemical reaction has taken place? 3. What factors can affect the rate of a chemical reaction (how fast the
CHAPTER 11: CHEMICAL REACTIONS. Mrs. Brayfield
 CHAPTER 11: CHEMICAL REACTIONS Mrs. Brayfield WRITING EQUATIONS Write the chemical equation for the following: Magnesium metal reacts with hydrochloric acid to form aqueous magnesium chloride and hydrogen
CHAPTER 11: CHEMICAL REACTIONS Mrs. Brayfield WRITING EQUATIONS Write the chemical equation for the following: Magnesium metal reacts with hydrochloric acid to form aqueous magnesium chloride and hydrogen
Chemical Equations. Physical Science
 Chemical Equations Physical Science Chemical Equations A chemical equation is a shorthand way of expressing a chemical reaction. CHEMICAL REACTIONS Reactants: Zn + I 2 Product: Zn I 2 Chemical reactions
Chemical Equations Physical Science Chemical Equations A chemical equation is a shorthand way of expressing a chemical reaction. CHEMICAL REACTIONS Reactants: Zn + I 2 Product: Zn I 2 Chemical reactions
Chemical reactions. C2- Topic 5
 Chemical reactions C2- Topic 5 What is a chemical reaction? A chemical reaction is a change that takes place when one or more substances (called reactants) form one or more new substances (called products)
Chemical reactions C2- Topic 5 What is a chemical reaction? A chemical reaction is a change that takes place when one or more substances (called reactants) form one or more new substances (called products)
Name Class Date. Complete each of the following sentences by choosing the correct term from the word bank.
 Skills Worksheet Chapter Review USING KEY TERMS Complete each of the following sentences by choosing the correct term from the word bank. subscript exothermic reaction inhibitor synthesis reaction coefficient
Skills Worksheet Chapter Review USING KEY TERMS Complete each of the following sentences by choosing the correct term from the word bank. subscript exothermic reaction inhibitor synthesis reaction coefficient
4 Energy and Rates of Chemical Reactions
 CHAPTER 14 4 and Rates of Chemical Reactions SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: How is energy involved in a chemical reaction?
CHAPTER 14 4 and Rates of Chemical Reactions SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: How is energy involved in a chemical reaction?
Chemical Bonds & Reactions
 Chemical Bonds & Reactions Chemical Bond A force of attraction that holds two atoms together Has a significant effect on chemical and physical properties of compounds involves the valence electrons Valence
Chemical Bonds & Reactions Chemical Bond A force of attraction that holds two atoms together Has a significant effect on chemical and physical properties of compounds involves the valence electrons Valence
The Atmosphere Exam questions
 The Atmosphere Exam questions 2012 - Higher What substance is formed when carbon is burned in oxygen? Give the effect of this substance on moist litmus paper. The table gives the % by volume of five gases/
The Atmosphere Exam questions 2012 - Higher What substance is formed when carbon is burned in oxygen? Give the effect of this substance on moist litmus paper. The table gives the % by volume of five gases/
Observations. Direct Observations Those observations we make ourselves
 Observations Direct Observations Those observations we make ourselves Indirect Observations Those observations that we figure out from direct observations Observing Milk Consumption The cafeteria is considering
Observations Direct Observations Those observations we make ourselves Indirect Observations Those observations that we figure out from direct observations Observing Milk Consumption The cafeteria is considering
The masses of reactants and products are equal.
 Page 1 of 7 KEY CNCEPT The masses of reactants and products are equal. BEFRE, you learned Chemical reactions turn reactants into products by rearranging atoms Chemical reactions can be observed and identified
Page 1 of 7 KEY CNCEPT The masses of reactants and products are equal. BEFRE, you learned Chemical reactions turn reactants into products by rearranging atoms Chemical reactions can be observed and identified
Chapter 6 and 7 Study Guide Reactions and Bonds
 Name_ Per. Block _ Multiple Choice: Chapter 6 and 7 Study Guide Reactions and Bonds 1. Copper is a good conductor of electricity because its electrons A. are positively charged B. are free to move and
Name_ Per. Block _ Multiple Choice: Chapter 6 and 7 Study Guide Reactions and Bonds 1. Copper is a good conductor of electricity because its electrons A. are positively charged B. are free to move and
The rate of a chemical reaction is the speed at which the reactants are used up or the speed at which new products are formed.
 IGCSE CHEMISTRY RVISION NOTES TOPIC 7: Chemical Reactions Rates of reaction Rate is a measure of how fast a reaction goes, its speed. The rate of a chemical reaction is the speed at which the reactants
IGCSE CHEMISTRY RVISION NOTES TOPIC 7: Chemical Reactions Rates of reaction Rate is a measure of how fast a reaction goes, its speed. The rate of a chemical reaction is the speed at which the reactants
Physical science Review
 Name: Topics: Physical science Review 1. Organization of matter 2. Particle model of solids liquids and gases 3. Characteristic vs. Non characteristic properties 4. Changes of matter 5. Law of conservation
Name: Topics: Physical science Review 1. Organization of matter 2. Particle model of solids liquids and gases 3. Characteristic vs. Non characteristic properties 4. Changes of matter 5. Law of conservation
Chemical Bonds & Reactions
 Chemical Bonds & Reactions Chemical Bonding Do you understand how it works? What do you think when I pull out a bag of candy? I want that candy cause I don t have any! Does everyone think the same thing?
Chemical Bonds & Reactions Chemical Bonding Do you understand how it works? What do you think when I pull out a bag of candy? I want that candy cause I don t have any! Does everyone think the same thing?
Changes in Matter Study Guide
 Changes in Matter Study Guide Define the following and give a real world example: 1. : A change in matter that requires the substances to absorb energy Example: Photosynthesis (the plants absorb light
Changes in Matter Study Guide Define the following and give a real world example: 1. : A change in matter that requires the substances to absorb energy Example: Photosynthesis (the plants absorb light
material organization.notebook September 09, 2016
 Organization of Matter 1 Review: CPs and NCPs Non Characteristic Properties: A physical or chemical property that is NOT UNIQUE to one particular substance Characteristic Properties: A physical or chemical
Organization of Matter 1 Review: CPs and NCPs Non Characteristic Properties: A physical or chemical property that is NOT UNIQUE to one particular substance Characteristic Properties: A physical or chemical
9-1 The Nature of Chemical Reactions
 9-1 The Nature of Chemical Reactions What are chemical reactions and why do they occur? A chemical reaction is a process in which one or more substances are converted into new substances with different
9-1 The Nature of Chemical Reactions What are chemical reactions and why do they occur? A chemical reaction is a process in which one or more substances are converted into new substances with different
Unit 5: Diversity of Matter
 21 21 Table of Contents Unit 5: Diversity of Matter Chapter 21: Chemical Reactions 21.1: Chemical Changes 21.2: Chemical Equations 21.3: Classifying Chemical Reactions 21.4: Chemical Reactions and Energy
21 21 Table of Contents Unit 5: Diversity of Matter Chapter 21: Chemical Reactions 21.1: Chemical Changes 21.2: Chemical Equations 21.3: Classifying Chemical Reactions 21.4: Chemical Reactions and Energy
Understanding Equations
 Chemical Reactions Chemical reaction: a process of chemically changing both the physical and chemical properties of a substance to a new substance with different physical and chemical properties. Video
Chemical Reactions Chemical reaction: a process of chemically changing both the physical and chemical properties of a substance to a new substance with different physical and chemical properties. Video
Section 1 Forming New Substances
 Section 1 Forming New Substances Key Concept During chemical reactions, atoms rearrange to form new substances that have different properties than the original substances had. What You Will Learn Four
Section 1 Forming New Substances Key Concept During chemical reactions, atoms rearrange to form new substances that have different properties than the original substances had. What You Will Learn Four
Matter: anything that has volume or mass Matter is made up of atoms and molecules.
 What is Matter??? Matter: anything that has volume or mass Matter is made up of atoms and molecules. 1 Atom: smallest unit of matter. Atom is the simplest form of an element. (Ex: 4 Carbon atoms = 4 C)
What is Matter??? Matter: anything that has volume or mass Matter is made up of atoms and molecules. 1 Atom: smallest unit of matter. Atom is the simplest form of an element. (Ex: 4 Carbon atoms = 4 C)
composition of matter, and the changes that matter undergoes. Examples of Uses of Chemistry in Everyday Life
 Name Matter and Change: Unit Objective Study Guide Date Due Directions: Write your answers to the following questions in the space provided. For problem solving, all of the work leading up to the final
Name Matter and Change: Unit Objective Study Guide Date Due Directions: Write your answers to the following questions in the space provided. For problem solving, all of the work leading up to the final
Chapter 17: Chemical Reactions
 Chapter 17: Chemical Reactions Section 1: Chemical Formulas and Equations Chemical Equations Balancing Chemical Equations Law of Conservation of Mass Evidences of Chemical Reactions Symbols to represent
Chapter 17: Chemical Reactions Section 1: Chemical Formulas and Equations Chemical Equations Balancing Chemical Equations Law of Conservation of Mass Evidences of Chemical Reactions Symbols to represent
Balancing Equations. Chemical reactions occur when bonds (between the electrons of atoms) are formed or broken Chemical reactions involve
 Balancing Equations Chemical reactions occur when bonds (between the electrons of atoms) are formed or broken Chemical reactions involve changes in the chemical composition of matter the making of new
Balancing Equations Chemical reactions occur when bonds (between the electrons of atoms) are formed or broken Chemical reactions involve changes in the chemical composition of matter the making of new
Chemical Reactions & Equations
 Chemical Reactions & Equations + Evidence of Chemical Reactions Release of a gas Example: bubbles formed when magnesium and hydrochloric acid were mixed Color change Example: color went from clear to yellowish
Chemical Reactions & Equations + Evidence of Chemical Reactions Release of a gas Example: bubbles formed when magnesium and hydrochloric acid were mixed Color change Example: color went from clear to yellowish
Chemical reactions. Section Describing chemical reactions
 Chemical reactions Section 11.1 - Describing chemical reactions What are everyday examples of chemical reactions? How do you write a chemical equation in words? Chemical equations are like a recipe, telling
Chemical reactions Section 11.1 - Describing chemical reactions What are everyday examples of chemical reactions? How do you write a chemical equation in words? Chemical equations are like a recipe, telling
Safety and Types of Fires
 Cookin' Chem Activity 2 Safety and Types of Fires CHEM POETRY Oliver learned all those Don t s and Do s, The ones that get passed down through moles of O 2 s; Each new generation did soon learn to trust
Cookin' Chem Activity 2 Safety and Types of Fires CHEM POETRY Oliver learned all those Don t s and Do s, The ones that get passed down through moles of O 2 s; Each new generation did soon learn to trust
Careful observations led to the discovery of the conservation of mass.
 Chapter 7, Section 2 Key Concept: The masses of reactants and products are equal. BEFORE, you learned Chemical reactions turn reactants into products by rearranging atoms Chemical reactions can be observed
Chapter 7, Section 2 Key Concept: The masses of reactants and products are equal. BEFORE, you learned Chemical reactions turn reactants into products by rearranging atoms Chemical reactions can be observed
Introduction to Chemical Reactions. Making new substances
 Introduction to Chemical Reactions Making new substances Main Ideas Chemical Reactions are represented by Chemical Equations. Chemical Equations are balanced to show the same number of atoms of each element
Introduction to Chemical Reactions Making new substances Main Ideas Chemical Reactions are represented by Chemical Equations. Chemical Equations are balanced to show the same number of atoms of each element
CHEMICAL EQUATIONS. REACTION CONDITIONS - give conditions necessary for chemical reaction to occur. May be:
 112 CHEMICAL EQUATIONS REACTION CONDITIONS - give conditions necessary for chemical reaction to occur. May be: - apply heat - catalysts - substances that will help reaction proceed faster - other conditions,
112 CHEMICAL EQUATIONS REACTION CONDITIONS - give conditions necessary for chemical reaction to occur. May be: - apply heat - catalysts - substances that will help reaction proceed faster - other conditions,
Chemical Reactions and Equations
 Name Date Chemical Reactions and Equations What happens to atoms and energy during a chemical reaction? Before You Read Before you read the chapter, think about what you know about chemical reactions Record
Name Date Chemical Reactions and Equations What happens to atoms and energy during a chemical reaction? Before You Read Before you read the chapter, think about what you know about chemical reactions Record
Chapter 7 TEST CHEMICAL REACTIONS PART 1: MULTIPLE CHOICE.
 Chapter 7 TEST CHEMICAL REACTIONS PART 1: MULTIPLE CHOICE. 1. Which of these is not a sign of a chemical reaction? a. A gas is given off. c. Heat is released. b. The material dissolves. d. A color change
Chapter 7 TEST CHEMICAL REACTIONS PART 1: MULTIPLE CHOICE. 1. Which of these is not a sign of a chemical reaction? a. A gas is given off. c. Heat is released. b. The material dissolves. d. A color change
Elements, Compounds Mixtures Physical and Chemical Changes
 Elements, Compounds Mixtures Physical and Chemical Changes Fundamentals of Chemistry 1 Classification of Matter Matter is any substance having distinct physical characteristics and chemical properties.
Elements, Compounds Mixtures Physical and Chemical Changes Fundamentals of Chemistry 1 Classification of Matter Matter is any substance having distinct physical characteristics and chemical properties.
Every living and nonliving things is made up of matter. MATTER: anything that has mass & takes up space. What does all matter have in common?
 the basics Every living and nonliving things is made up of matter MATTER: anything that has mass & takes up space What does all matter have in common? Smallest unit of matter ALL matter is made of particles
the basics Every living and nonliving things is made up of matter MATTER: anything that has mass & takes up space What does all matter have in common? Smallest unit of matter ALL matter is made of particles
1 Forming New Substances
 CHAPTER 9 1 Forming New Substances SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a What are exothermic reactions? What are
CHAPTER 9 1 Forming New Substances SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a What are exothermic reactions? What are
Sulfuric acid is hazardous: Safety glasses are REQUIRED during this experiment.
 CHEMICAL COMPOSITION Life exists on Earth because of the abundant presence of liquid water. While other planets have water, it may be primarily found as either a gas, as on Venus, or as a solid, such as
CHEMICAL COMPOSITION Life exists on Earth because of the abundant presence of liquid water. While other planets have water, it may be primarily found as either a gas, as on Venus, or as a solid, such as
Semester 1 Review Questions for Exam
 Semester 1 Review Questions for Exam Completion Complete each sentence or statement. 1. The part of an ecosystem where an organism lives and feeds is called the organism s 2. All the biotic and abiotic
Semester 1 Review Questions for Exam Completion Complete each sentence or statement. 1. The part of an ecosystem where an organism lives and feeds is called the organism s 2. All the biotic and abiotic
Physical and Chemical change: Conservation of matter *
 OpenStax-CNX module: m38711 1 Physical and Chemical change: Conservation of matter * Free High School Science Texts Project This work is produced by OpenStax-CNX and licensed under the Creative Commons
OpenStax-CNX module: m38711 1 Physical and Chemical change: Conservation of matter * Free High School Science Texts Project This work is produced by OpenStax-CNX and licensed under the Creative Commons
Activation Energy is the minimum amount of energy needed to start a chemical reaction.
 5.3 Controlling Chemical Reactions Vocabulary: Activation energy Concentration Catalyst Enzyme Inhibitor - How do reactions get started? Chemical reactions won t begin until the reactants have enough energy.
5.3 Controlling Chemical Reactions Vocabulary: Activation energy Concentration Catalyst Enzyme Inhibitor - How do reactions get started? Chemical reactions won t begin until the reactants have enough energy.
Lesson #6: Chemical Reaction Types
 Lesson #6: Chemical Types Type #1 The Synthesis In this type of reaction 2 or more elements or compounds combine to form a more complex compound Two or more things become one bigger thing (ex) 2Mg + O
Lesson #6: Chemical Types Type #1 The Synthesis In this type of reaction 2 or more elements or compounds combine to form a more complex compound Two or more things become one bigger thing (ex) 2Mg + O
Science 1206 Chemistry Unit #10
 Science 1206 Chemistry Unit #10 Introduction to Chemical Equations Communicating Chemical Changes Chemical change occurs when matter changes into a new and different form. The new form has a new make up
Science 1206 Chemistry Unit #10 Introduction to Chemical Equations Communicating Chemical Changes Chemical change occurs when matter changes into a new and different form. The new form has a new make up
Representing Chemical Change
 Representing Chemical Change As we have already mentioned, a number of changes can occur when elements react with one another. These changes may either be physical or chemical. One way of representing
Representing Chemical Change As we have already mentioned, a number of changes can occur when elements react with one another. These changes may either be physical or chemical. One way of representing
Important Examples of Chemical Change
 Important Examples of Chemical Change chemical change: the change that occurs when substances react in a chemical reaction to create different substances When was the last time you watched an awesome display
Important Examples of Chemical Change chemical change: the change that occurs when substances react in a chemical reaction to create different substances When was the last time you watched an awesome display
CHEMICAL REACTIONS 3/1/2012 PARTS OF A CHEMICAL EQUATION. Reactants: Zn + I 2 Product: ZnI 2. Indicators of chemical reactions
 CHEMICAL REACTIONS Reactants: Zn + I 2 Product: ZnI 2 Indicators of chemical reactions Emission of light or heat Production of a gas Formation of a precipitate Color change Emission of odor PARTS OF A
CHEMICAL REACTIONS Reactants: Zn + I 2 Product: ZnI 2 Indicators of chemical reactions Emission of light or heat Production of a gas Formation of a precipitate Color change Emission of odor PARTS OF A
Conservation The Law of Conservation of Matter states that matter cannot be created or destroyed in ordinary chemical reactions. This means that no
 Chemical Reactions Conservation The Law of Conservation of Matter states that matter cannot be created or destroyed in ordinary chemical reactions. This means that no atoms can be lost or gained. This
Chemical Reactions Conservation The Law of Conservation of Matter states that matter cannot be created or destroyed in ordinary chemical reactions. This means that no atoms can be lost or gained. This
Name Date Class STUDY GUIDE FOR CONTENT MASTERY
 Chemical Reactions Section 10.1 Reactions and Equations In your textbook, read about evidence of chemical reactions. For each statement, write yes if evidence of a chemical reaction is present. Write no
Chemical Reactions Section 10.1 Reactions and Equations In your textbook, read about evidence of chemical reactions. For each statement, write yes if evidence of a chemical reaction is present. Write no
What is Matter??? Ma#er: anything that has volume or mass Ma2er is made up of atoms and molecules.
 What is Matter??? Ma#er: anything that has volume or mass Ma2er is made up of atoms and molecules. Atom: smallest unit of ma2er. Atom is the simplest form of an element. (Ex: 4 Carbon atoms = 4 C) *Element:
What is Matter??? Ma#er: anything that has volume or mass Ma2er is made up of atoms and molecules. Atom: smallest unit of ma2er. Atom is the simplest form of an element. (Ex: 4 Carbon atoms = 4 C) *Element:
8.1 Chemical Properties and Changes. chemical property chemical change dissolving
 8.1 Chemical Properties and Changes chemical property chemical change dissolving Ability to Change 8.1 Chemical Properties and Changes In a chemical change, the properties that give a substance its identity
8.1 Chemical Properties and Changes chemical property chemical change dissolving Ability to Change 8.1 Chemical Properties and Changes In a chemical change, the properties that give a substance its identity
Jeddah Knowledge International School
 Jeddah Knowledge International School Science Revision Pack 2016-2017 Quarter 1 Grade 9 Name: Section: Science Revision Pack Grade Nine Quarter One Page 1 Revision Pack Grade 9 Chemistry Question 1: Circle
Jeddah Knowledge International School Science Revision Pack 2016-2017 Quarter 1 Grade 9 Name: Section: Science Revision Pack Grade Nine Quarter One Page 1 Revision Pack Grade 9 Chemistry Question 1: Circle
Which of the following is NOT an example of a chemical reaction? A. Milk turning sour B. Food being digested C. A match burning D.
 Chemical Reactions Which of the following is NOT an example of a chemical reaction? A. Milk turning sour B. Food being digested C. A match burning D. Ice melting Which of the following is NOT an example
Chemical Reactions Which of the following is NOT an example of a chemical reaction? A. Milk turning sour B. Food being digested C. A match burning D. Ice melting Which of the following is NOT an example
Chemistry Chapter 1 Test Review
 Chemistry Chapter 1 Test Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. A chemical can be defined as a. a toxic substance. b. an unnatural additive
Chemistry Chapter 1 Test Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. A chemical can be defined as a. a toxic substance. b. an unnatural additive
2/22/2019 NEW UNIT! Chemical Interactions. Atomic Basics #19
 NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
Change Matter around us goes through many changes. E.g. Water evapourates, bread is toasted for your breakfast Changes are divided into two
 Change Matter around us goes through many changes. E.g. Water evapourates, bread is toasted for your breakfast Changes are divided into two categories: Physical changes Chemical changes Physical Changes
Change Matter around us goes through many changes. E.g. Water evapourates, bread is toasted for your breakfast Changes are divided into two categories: Physical changes Chemical changes Physical Changes
Chemical Changes. Lavoisier and the Conservation of Mass
 1 Chemical Changes Lavoisier and the Conservation of Mass Chemical reactions are taking place all around you and even within you. A chemical reaction is a change in which one or more substances are converted
1 Chemical Changes Lavoisier and the Conservation of Mass Chemical reactions are taking place all around you and even within you. A chemical reaction is a change in which one or more substances are converted
Chapter 6, Lesson 1: What is a Chemical Reaction?
 Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
1. Review chemical vs. physical reactions 2. Chemical reactions video 3. Useful chemical reactions research assignment
 Science 24: Day 10 1. Review chemical vs. physical reactions 2. Chemical reactions video 3. Useful chemical reactions research assignment https://www.youtube.com/watch?v=fofpjj7v414 Which reaction was
Science 24: Day 10 1. Review chemical vs. physical reactions 2. Chemical reactions video 3. Useful chemical reactions research assignment https://www.youtube.com/watch?v=fofpjj7v414 Which reaction was
CHEMICAL REACTIONS. Discuss breaking/forming bonds 10/29/2012. Products Reactants
 CHEMICAL REACTIONS - Page 321 Products Reactants Reactants: Zn + I 2 Product: Zn I 2 Discuss breaking/forming bonds Chemical Reaction or Change process in which one or more substances are converted into
CHEMICAL REACTIONS - Page 321 Products Reactants Reactants: Zn + I 2 Product: Zn I 2 Discuss breaking/forming bonds Chemical Reaction or Change process in which one or more substances are converted into
Chemical Reactions BASICS
 Chemical Reactions BASICS There are 5 simple reactions in this chemistry class (but more are coming later in the year). They are synthesis, decomposition, single replacement, double replacement, and combustion.
Chemical Reactions BASICS There are 5 simple reactions in this chemistry class (but more are coming later in the year). They are synthesis, decomposition, single replacement, double replacement, and combustion.
Balancing CHEMICAL EQUATIONS
 Balancing CHEMICAL EQUATIONS CHEMICAL REACTIONS involves a chemical change in the identity of one or more chemical species Ex. Rusting of iron (Fe): chemical rxn btwn water and iron involve the breaking
Balancing CHEMICAL EQUATIONS CHEMICAL REACTIONS involves a chemical change in the identity of one or more chemical species Ex. Rusting of iron (Fe): chemical rxn btwn water and iron involve the breaking
Q1. Methane and oxygen react together to produce carbon dioxide and water.
 Q1. Methane and oxygen react together to produce carbon dioxide and water. The methane gas will not burn in oxygen until a flame is applied, but once lit it continues to burn. (a) Explain why energy must
Q1. Methane and oxygen react together to produce carbon dioxide and water. The methane gas will not burn in oxygen until a flame is applied, but once lit it continues to burn. (a) Explain why energy must
Chemistry Part 2: We re Not Done Yet!
 Chemistry Part 2: We re Not Done Yet! WOW! Learning about atoms sure was fun! Let s continue our fascinating exploration of chemicals. What is a Chemical? Chemical: A substance having a constant composition
Chemistry Part 2: We re Not Done Yet! WOW! Learning about atoms sure was fun! Let s continue our fascinating exploration of chemicals. What is a Chemical? Chemical: A substance having a constant composition
Matter. Matter is anything that takes up space and has mass.
 Matter Matter is anything that takes up space and has mass. States of Matter Matter exists in three states: O Solid O Examples: rock, wood, chalk O Liquid O Examples: water, milk O Gas O Examples: oxygen,
Matter Matter is anything that takes up space and has mass. States of Matter Matter exists in three states: O Solid O Examples: rock, wood, chalk O Liquid O Examples: water, milk O Gas O Examples: oxygen,
5. Researching the properties of particular materials and understand why they are used for particular products.
 1. The difference between a physical and chemical change. 2. How to identify whether a physical or chemical change has taken place. 3. Understand what reactants and products are. 4. Writing and understanding
1. The difference between a physical and chemical change. 2. How to identify whether a physical or chemical change has taken place. 3. Understand what reactants and products are. 4. Writing and understanding
Science 10: Chemistry II Practice Test
 Science 10: Chemistry II Practice Test Name: Multiple Choice: Circle the letter of the best answer. You may refer to a periodic table and an ion chart. 1. What type of reaction is the following? silver
Science 10: Chemistry II Practice Test Name: Multiple Choice: Circle the letter of the best answer. You may refer to a periodic table and an ion chart. 1. What type of reaction is the following? silver
Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds.
 Chapter 9 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
Chapter 9 Chemical Reactions Notes Chemical Reactions: Chemical reactions are processes in which the atoms of one or more substances are rearranged to form different chemical compounds. How to tell if
ICP Final Exam Review - Part 2
 ICP Final Exam Review - Part 2 Modified True/False Indicate whether the statement is true or false. If false, change the identified word or phrase to make the statement true. 1. A combustion reaction occurs
ICP Final Exam Review - Part 2 Modified True/False Indicate whether the statement is true or false. If false, change the identified word or phrase to make the statement true. 1. A combustion reaction occurs
Topic 1 Topic 2 Topic 3 Topic 4 Topic 5
 Topic 1 Topic 2 Topic 3 Topic 4 Topic 5 10 10 10 10 10 20 20 20 20 20 30 30 30 30 30 40 40 40 40 40 50 50 50 50 50 Question 1-10 Oxygen gas is written as O 2 in chemical equations. What term is used to
Topic 1 Topic 2 Topic 3 Topic 4 Topic 5 10 10 10 10 10 20 20 20 20 20 30 30 30 30 30 40 40 40 40 40 50 50 50 50 50 Question 1-10 Oxygen gas is written as O 2 in chemical equations. What term is used to
Crash Course on Nucleus
 II. Chemistry Crash Course on Nucleus A. Atomic Theory 1. Atom a. Most basic unit of matter - building block b. Can t be broken down further and still retain its properties c. Identified by the number
II. Chemistry Crash Course on Nucleus A. Atomic Theory 1. Atom a. Most basic unit of matter - building block b. Can t be broken down further and still retain its properties c. Identified by the number
LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Counting atoms
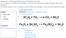 Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
STEMscopedia: PHYSICAL AND CHEMICAL PROPERTIES AND CHANGES 8P1CD
 Reflect STEMscopedia: PHYSICAL AND CHEMICAL 8P1CD What do ice cream, root beer, and carbon dioxide gas have in common? Not only do these ingredients combine to make a good treat on a hot summer day, but
Reflect STEMscopedia: PHYSICAL AND CHEMICAL 8P1CD What do ice cream, root beer, and carbon dioxide gas have in common? Not only do these ingredients combine to make a good treat on a hot summer day, but
by Cyndee Crawford October 2014
 7-5 Chemical Nature of Matter PART 2 by Cyndee Crawford October 2014 7-5 The student will demonstrate an understanding of the classifications and properties of matter and the changes that matter undergoes.
7-5 Chemical Nature of Matter PART 2 by Cyndee Crawford October 2014 7-5 The student will demonstrate an understanding of the classifications and properties of matter and the changes that matter undergoes.
