TRANSPORT PHENOMENON FICK S LAW OF DIFFUSION ATP-POWERED PUMPS -II-
|
|
|
- Arron Walters
- 5 years ago
- Views:
Transcription
1 TRANSPORT PHENOMENON FICK S LAW OF DIFFUSION ATP-POWERED PUMPS -II- Yalçın İŞLER, PhD Izmir Katip Celebi University Department of Biomedical Engineering islerya@yahoo.com
2 DIFFUSION Diffusion describes the spread of particles through random motion from the regions of higher concentration to the regions of lower concentration. After a period of time the particles are distributed randomly. At the end of a certain period, the average travelled distances by the particles will be zero. Because the number of particles moving in one direction is almost equal to the number of particles moving in the reverse direction
3 Diffusion The root mean square of the distances travelled by particles is always a positive. x rms x n x
4 Diffusion After a period of time, particles reach such locations along the motion axis that the graph of the number of particles and their distances from the starting point gives a "GAUSS" curve. N is the number of particles between x and x+δx
5 Diffusion In addition to diffusion particles there may be other dissolved particles in the solution. In this case, the distribution rate of the particles, (travelled distance per unit of time) will be changed. High energy particles will lead to scattering of the low energy particles
6 Diffusion The Boltzmann Equation describes the relationship between the particles in gases and solutions. the number of particles (N y ) which have E y energy and the number of particles (N d ) which have E d energy ln N N (E kt k= Boltzmann constant = 1,38 x10-3 J/ K T= absolute temperature ( K) y d y E d )
7 Diffusion The kinetic energy of a particle is: If this formula is written in Boltzmann Equation; ln N N y d will be obtained. If the low energy level E d =0, then V d =0. Therefore, instead of N d, N 0 could be used and equation will be as follows; ln N N y 0 ( mv y kt mv y kt mv d )
8 Diffusion This new equation is also a Gaussion function. Each vertical line in the graph, represents the ratio of the number of the particles whose velocities are between v and v+δv and the number of stable particles.
9 Diffusion According to the kinetic theory of gases, there is a relationship between the kinetic energy of a particle and the absolute temperature of the system. E 1 mv 3 kt k= Boltzmann constant Because the movement has 3 dimension, velocity has 3 dimension (V x, V y, V z ). Therefore, the kinetic energy in one dimension is the 1/3 of the total kinetic energy of the system
10 Diffusion The movement of particles (molecules or ions) in aqueous solutions is more complex- than the movements of molecules in gases. Because in these systems, particles are under the effect of friction forces. Friction force is proportional to the particle velocity and has opposite direction. For example, a particle which has V x velocity in x-axis is under the effect of the following friction force: F x fv x f= Friction coefficient
11 Diffusion Friction coefficient for the spherical molecules is: f 6r r viscosity coefficient of the system radius of the molecule
12 Diffusion By the help of velocity, distance and energy equations, the following formula will be obtained: kt x t1 This is the random walk f of particles in one dimension. In 3 dimension the equation will be: r x y z kt f t 1 kt f t 1 kt f t 1 6kT f t 1
13 Diffusion Diffusion coefficient (D) D kt f kt 6r Diffusion coefficient (D) will vary depending on the absolute temperature, the viscosity of the medium size and shape of the particle Random walk equations in 3 dimensions could be written in terms of diffusion coefficient as follows: x Dt s r 4Dt 6Dt
14 Diffusion - Example Question: How long will it take for a water molecule to diffuse 0.01 m in three dimensions? Diffusion coefficient of a water molecule in room temperature is: D=x10-9 m /s
15 Diffusion - Example Soluton: r 6Dt (0.01) t t second (.31hour)
16 Fick s Law of Diffusion Because the particles in a solution move randomly, the probability of movement in all directions is equal. If the molecules are highly concentrated in a region, the number of molecules leaving the area will be more than the number of the molecules arriving this region. Thus, there will be a net flow of molecules from highly concentrated region to the low concentrated region. This phenomenon is called "diffusion".
17 Flow of molecules in one dimension Highly concentrated region A = Cross-section area of the pipe Low concentrated region
18 Fick s Law of Diffusion c 1 is the concentration in x 1 and c is the concentration in x A is the cross-section area of the pipe Net amount of particle (Δm) which flow from x 1 to x in Δt time is: m (Ac1 Ac) or t m A(c1 c) t With a proportionality constant the equation will be: m t ka(c1 c) kac
19 Fick s Law of Diffusion If the x 1 and x planes are too far apart from each other, then the amount of flowing particles between these planes will be very low. On the other hand, if these two planes are very close to each other, then the amount of flowing particles will be very high. Therefore, k constant in Fick s Diffusion equation is inversely proportional with (x -x 1 )=Δx. k D x k is referred to Fick's diffusion constant. D is the diffusion coefficient.
20 Fick s Law of Diffusion Put k equation in the place of the Fick s Diffusion equation and: m D c m kac Ac DA 1 t x x t A c J D x D c x J: Diffusion flux: the amount of substance per unit area per unit time Fick s first law postulates that the flux goes from regions of high concentration to regions of low concentration, with a magnitude that is proportional to the concentration gradient (spatial derivative).
21 Fick s Law of Diffusion c t D c x Fick's second law predicts how diffusion causes the concentration to change with time If the inial conditions are considered as; all the particles being in position x = 0 at time t=0 and in time particles walk away in both directions then the solution of the equation will be as follows; c 1 (4Dt) 0,5 e x 4Dt
22 Fick s Law of Diffusion (a) The Concentration and (b) The concentration gradient change in time and with distance
23 References Prof. Dr. Gürbüz Çelebi. Biyofizik. Tıp ve Diş Hekimliği Öğrencileri için. Barış Yayınları Fakülteler Kitabevi, Cilt I, III.Baskı, İzmir, 005. Prof. Dr. Ferit Pehlivan. Biyofizik. Hacettepe Taş. 4. Tıpkı Basım, Ankara,
Biophysics I. DIFFUSION
 Biophysics I. DIFFUSION Experiment add a droplet of ink to a glass of water Observation: the stain spreads and eventually colours the entire fluid add a droplet of ink to HOT and COLD water Observation:
Biophysics I. DIFFUSION Experiment add a droplet of ink to a glass of water Observation: the stain spreads and eventually colours the entire fluid add a droplet of ink to HOT and COLD water Observation:
t = no of steps of length s
 s t = no of steps of length s Figure : Schematic of the path of a diffusing molecule, for example, one in a gas or a liquid. The particle is moving in steps of length s. For a molecule in a liquid the
s t = no of steps of length s Figure : Schematic of the path of a diffusing molecule, for example, one in a gas or a liquid. The particle is moving in steps of length s. For a molecule in a liquid the
Biology 3550 Physical Principles in Biology Fall Semester 2017 Mid-Term Exam 6 October 2017
 100 points total Please write your name on each page. In your answers you should: Biology 3550 Physical Principles in Biology Fall Semester 2017 Mid-Term Exam 6 October 2017 Show your work or provide an
100 points total Please write your name on each page. In your answers you should: Biology 3550 Physical Principles in Biology Fall Semester 2017 Mid-Term Exam 6 October 2017 Show your work or provide an
University of Washington Department of Chemistry Chemistry 453 Winter Quarter 2005
 Lecture /4/ University of Washington Department of Chemistry Chemistry 4 Winter Quarter A. Polymer Properties of DNA When a linear polymer like DNA becomes long enough it can no longer be treated as a
Lecture /4/ University of Washington Department of Chemistry Chemistry 4 Winter Quarter A. Polymer Properties of DNA When a linear polymer like DNA becomes long enough it can no longer be treated as a
BAE 820 Physical Principles of Environmental Systems
 BAE 820 Physical Principles of Environmental Systems Estimation of diffusion Coefficient Dr. Zifei Liu Diffusion mass transfer Diffusion mass transfer refers to mass in transit due to a species concentration
BAE 820 Physical Principles of Environmental Systems Estimation of diffusion Coefficient Dr. Zifei Liu Diffusion mass transfer Diffusion mass transfer refers to mass in transit due to a species concentration
Module 5: Rise and Fall of the Clockwork Universe. You should be able to demonstrate and show your understanding of:
 OCR B Physics H557 Module 5: Rise and Fall of the Clockwork Universe You should be able to demonstrate and show your understanding of: 5.2: Matter Particle model: A gas consists of many very small, rapidly
OCR B Physics H557 Module 5: Rise and Fall of the Clockwork Universe You should be able to demonstrate and show your understanding of: 5.2: Matter Particle model: A gas consists of many very small, rapidly
Chapter 14. The Ideal Gas Law and Kinetic Theory
 Chapter 14 The Ideal Gas Law and Kinetic Theory 14.1 Molecular Mass, the Mole, and Avogadro s Number To facilitate comparison of the mass of one atom with another, a mass scale know as the atomic mass
Chapter 14 The Ideal Gas Law and Kinetic Theory 14.1 Molecular Mass, the Mole, and Avogadro s Number To facilitate comparison of the mass of one atom with another, a mass scale know as the atomic mass
FORMULA SHEET. General formulas:
 FORMULA SHEET You may use this formula sheet during the Advanced Transport Phenomena course and it should contain all formulas you need during this course. Note that the weeks are numbered from 1.1 to
FORMULA SHEET You may use this formula sheet during the Advanced Transport Phenomena course and it should contain all formulas you need during this course. Note that the weeks are numbered from 1.1 to
Biomolecular hydrodynamics
 Biomolecular hydrodynamics Chem 341, Fall, 2014 1 Frictional coefficients Consider a particle moving with velocity v under the influence of some external force F (say a graviational or electrostatic external
Biomolecular hydrodynamics Chem 341, Fall, 2014 1 Frictional coefficients Consider a particle moving with velocity v under the influence of some external force F (say a graviational or electrostatic external
Diffusion - The Heat Equation
 Chapter 6 Diffusion - The Heat Equation 6.1 Goal Understand how to model a simple diffusion process and apply it to derive the heat equation in one dimension. We begin with the fundamental conservation
Chapter 6 Diffusion - The Heat Equation 6.1 Goal Understand how to model a simple diffusion process and apply it to derive the heat equation in one dimension. We begin with the fundamental conservation
Perfect Gases Transport Phenomena
 Perfect Gases Transport Phenomena We have been able to relate quite a few macroscopic properties of gasses such as P, V, T to molecular behaviour on microscale. We saw how macroscopic pressure is related
Perfect Gases Transport Phenomena We have been able to relate quite a few macroscopic properties of gasses such as P, V, T to molecular behaviour on microscale. We saw how macroscopic pressure is related
Lecture 18 Molecular Motion and Kinetic Energy
 Physical Principles in Biology Biology 3550 Fall 2017 Lecture 18 Molecular Motion and Kinetic Energy Monday, 2 October c David P. Goldenberg University of Utah goldenberg@biology.utah.edu Fick s First
Physical Principles in Biology Biology 3550 Fall 2017 Lecture 18 Molecular Motion and Kinetic Energy Monday, 2 October c David P. Goldenberg University of Utah goldenberg@biology.utah.edu Fick s First
Nicholas J. Giordano. Chapter 10 Fluids
 Nicholas J. Giordano www.cengage.com/physics/giordano Chapter 10 Fluids Fluids A fluid may be either a liquid or a gas Some characteristics of a fluid Flows from one place to another Shape varies according
Nicholas J. Giordano www.cengage.com/physics/giordano Chapter 10 Fluids Fluids A fluid may be either a liquid or a gas Some characteristics of a fluid Flows from one place to another Shape varies according
Physics 1501 Lecture 35
 Physics 1501: Lecture 35 Todays Agenda Announcements Homework #11 (Dec. 2) and #12 (Dec. 9): 2 lowest dropped Honors students: see me after the class! Todays topics Chap.16: Temperature and Heat» Latent
Physics 1501: Lecture 35 Todays Agenda Announcements Homework #11 (Dec. 2) and #12 (Dec. 9): 2 lowest dropped Honors students: see me after the class! Todays topics Chap.16: Temperature and Heat» Latent
Similarities and differences:
 How does the system reach equilibrium? I./9 Chemical equilibrium I./ Equilibrium electrochemistry III./ Molecules in motion physical processes, non-reactive systems III./5-7 Reaction rate, mechanism, molecular
How does the system reach equilibrium? I./9 Chemical equilibrium I./ Equilibrium electrochemistry III./ Molecules in motion physical processes, non-reactive systems III./5-7 Reaction rate, mechanism, molecular
University of Washington Department of Chemistry Chemistry 453 Winter Quarter 2013
 Lecture 1 3/13/13 University of Washington Department of Chemistry Chemistry 53 Winter Quarter 013 A. Definition of Viscosity Viscosity refers to the resistance of fluids to flow. Consider a flowing liquid
Lecture 1 3/13/13 University of Washington Department of Chemistry Chemistry 53 Winter Quarter 013 A. Definition of Viscosity Viscosity refers to the resistance of fluids to flow. Consider a flowing liquid
Molecular Weight & Energy Transport
 Molecular Weight & Energy Transport 7 September 20 Goals Review mean molecular weight Practice working with diffusion Mean Molecular Weight. We will frequently use µ,, and (the mean molecular weight per
Molecular Weight & Energy Transport 7 September 20 Goals Review mean molecular weight Practice working with diffusion Mean Molecular Weight. We will frequently use µ,, and (the mean molecular weight per
Mechanics and Heat. Chapter 5: Work and Energy. Dr. Rashid Hamdan
 Mechanics and Heat Chapter 5: Work and Energy Dr. Rashid Hamdan 5.1 Work Done by a Constant Force Work Done by a Constant Force A force is said to do work if, when acting on a body, there is a displacement
Mechanics and Heat Chapter 5: Work and Energy Dr. Rashid Hamdan 5.1 Work Done by a Constant Force Work Done by a Constant Force A force is said to do work if, when acting on a body, there is a displacement
Conceptual Model of Diffusion
 Conceptual Model of Diffusion This section introduces the concept of molecular diffusion based on the random path of molecules. The theory and animation demonstrate how, through Brownian motion, a Gaussian
Conceptual Model of Diffusion This section introduces the concept of molecular diffusion based on the random path of molecules. The theory and animation demonstrate how, through Brownian motion, a Gaussian
Chapter 14. The Ideal Gas Law and Kinetic Theory
 Chapter 14 The Ideal Gas Law and Kinetic Theory 14.1 Molecular Mass, the Mole, and Avogadro s Number The atomic number of an element is the # of protons in its nucleus. Isotopes of an element have different
Chapter 14 The Ideal Gas Law and Kinetic Theory 14.1 Molecular Mass, the Mole, and Avogadro s Number The atomic number of an element is the # of protons in its nucleus. Isotopes of an element have different
must move around the sphere : l 3 dt dt d dy
 Name: KEY SC/BPHS090 First Term Test (Lew 9 October 014 Student ID: Be sure to write your name and student ID above. Read the three questions carefully, think, then write your answers in the lined space
Name: KEY SC/BPHS090 First Term Test (Lew 9 October 014 Student ID: Be sure to write your name and student ID above. Read the three questions carefully, think, then write your answers in the lined space
Lecture 5 Entropy and Disorder
 Lecture 5 Entropy and Disorder Third law S (T) = S(0) + C P ln T Background information: 2 1 ds = n Cp ln T 2 /T 1 nrlnp 2 /P 1 The change in entropy can be calculated using graphical method. In all these
Lecture 5 Entropy and Disorder Third law S (T) = S(0) + C P ln T Background information: 2 1 ds = n Cp ln T 2 /T 1 nrlnp 2 /P 1 The change in entropy can be calculated using graphical method. In all these
Computational Neuroscience. Session 2-1
 Computational Neuroscience. Session 2-1 Dr. Marco A Roque Sol 06/04/2018 All living cells exhibit an electrical potential difference between the inner and outer surface of the cytoplasmic membrane. This
Computational Neuroscience. Session 2-1 Dr. Marco A Roque Sol 06/04/2018 All living cells exhibit an electrical potential difference between the inner and outer surface of the cytoplasmic membrane. This
Solids (cont.) Describe the movement of particles in a solid and the forces between them.
 Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
Kinetic Theory. T.R. Lemberger, Feb Calculation of Pressure for an Ideal Gas. Area, A.
 Kinetic Theory. T.R. Lemberger, Feb. 2007 In the simplest model, the equilibrium distribution of gas particle velocities is this: all particles move with the same speed, c. The equipartition theorem requires:
Kinetic Theory. T.R. Lemberger, Feb. 2007 In the simplest model, the equilibrium distribution of gas particle velocities is this: all particles move with the same speed, c. The equipartition theorem requires:
CHAPTER 19 Molecules in Motion. 1. Matter transport is driven by concentration gradient.
 CHAPTER 19 Molecules in Motion I. Transport in Gases. A. Fick s Law of Diffusion. 1. Matter transport is driven by concentration gradient. Flux (matter) = J m = D dn dz N = number density of particles
CHAPTER 19 Molecules in Motion I. Transport in Gases. A. Fick s Law of Diffusion. 1. Matter transport is driven by concentration gradient. Flux (matter) = J m = D dn dz N = number density of particles
Introduction to Physiology II: Control of Cell Volume and Membrane Potential
 Introduction to Physiology II: Control of Cell Volume and Membrane Potential J. P. Keener Mathematics Department Math Physiology p.1/23 Basic Problem The cell is full of stuff: Proteins, ions, fats, etc.
Introduction to Physiology II: Control of Cell Volume and Membrane Potential J. P. Keener Mathematics Department Math Physiology p.1/23 Basic Problem The cell is full of stuff: Proteins, ions, fats, etc.
Topics covered so far:
 Topics covered so far: Chap 1: The kinetic theory of gases P, T, and the Ideal Gas Law Chap 2: The principles of statistical mechanics 2.1, The Boltzmann law (spatial distribution) 2.2, The distribution
Topics covered so far: Chap 1: The kinetic theory of gases P, T, and the Ideal Gas Law Chap 2: The principles of statistical mechanics 2.1, The Boltzmann law (spatial distribution) 2.2, The distribution
Plasma Physics Prof. Vijayshri School of Sciences, IGNOU. Lecture No. # 38 Diffusion in Plasmas
 Plasma Physics Prof. Vijayshri School of Sciences, IGNOU Lecture No. # 38 Diffusion in Plasmas In today s lecture, we will be taking up the topic diffusion in plasmas. Diffusion, why do you need to study
Plasma Physics Prof. Vijayshri School of Sciences, IGNOU Lecture No. # 38 Diffusion in Plasmas In today s lecture, we will be taking up the topic diffusion in plasmas. Diffusion, why do you need to study
QuickCheck. Collisions between molecules. Collisions between molecules
 Collisions between molecules We model molecules as rigid spheres of radius r as shown at the right. The mean free path of a molecule is the average distance it travels between collisions. The average time
Collisions between molecules We model molecules as rigid spheres of radius r as shown at the right. The mean free path of a molecule is the average distance it travels between collisions. The average time
Physics 9 Spring 2011 Midterm 1 Solutions
 Physics 9 Spring 2011 Midterm 1 s For the midterm, you may use one sheet of notes with whatever you want to put on it, front and back. Please sit every other seat, and please don t cheat! If something
Physics 9 Spring 2011 Midterm 1 s For the midterm, you may use one sheet of notes with whatever you want to put on it, front and back. Please sit every other seat, and please don t cheat! If something
E6 PROPERTIES OF GASES Flow-times, density, phase changes, solubility
 E6 PROPERTIES OF GASES Flow-times, density, phase changes, solubility Introduction Kinetic-Molecular Theory The kinetic energy of an object is dependent on its mass and its speed. The relationship, given
E6 PROPERTIES OF GASES Flow-times, density, phase changes, solubility Introduction Kinetic-Molecular Theory The kinetic energy of an object is dependent on its mass and its speed. The relationship, given
Cell Processes: Diffusion Part 1: Fick s First Law
 MathBench- Australia Diffusion Part 1 December 2015 page 1 Cell Processes: Diffusion Part 1: Fick s First Law URL: http://mathbench.org.au/cellular-processes/introduction-to-diffusion/ Measuring Movement
MathBench- Australia Diffusion Part 1 December 2015 page 1 Cell Processes: Diffusion Part 1: Fick s First Law URL: http://mathbench.org.au/cellular-processes/introduction-to-diffusion/ Measuring Movement
Final exam. Please write your name on the exam and keep an ID card ready. You may use a calculator (but no computer or smart phone) and a dictionary.
 Biophysics of Macromolecules Prof. D. Braun and Prof. J. Lipfert SS 2015 Final exam Final exam Name: Student number ( Matrikelnummer ): Please write your name on the exam and keep an ID card ready. You
Biophysics of Macromolecules Prof. D. Braun and Prof. J. Lipfert SS 2015 Final exam Final exam Name: Student number ( Matrikelnummer ): Please write your name on the exam and keep an ID card ready. You
Carriers Concentration and Current in Semiconductors
 Carriers Concentration and Current in Semiconductors Carrier Transport Two driving forces for carrier transport: electric field and spatial variation of the carrier concentration. Both driving forces lead
Carriers Concentration and Current in Semiconductors Carrier Transport Two driving forces for carrier transport: electric field and spatial variation of the carrier concentration. Both driving forces lead
First Law of Thermodynamics Second Law of Thermodynamics Mechanical Equivalent of Heat Zeroth Law of Thermodynamics Thermal Expansion of Solids
 Slide 1 / 66 1 What is the name of the following statement: "When two systems are in thermal equilibrium with a third system, then they are in thermal equilibrium with each other"? A B C D E First Law
Slide 1 / 66 1 What is the name of the following statement: "When two systems are in thermal equilibrium with a third system, then they are in thermal equilibrium with each other"? A B C D E First Law
Collisions between molecules
 Collisions between molecules We model molecules as rigid spheres of radius r as shown at the right. The mean free path of a molecule is the average distance it travels between collisions. The average time
Collisions between molecules We model molecules as rigid spheres of radius r as shown at the right. The mean free path of a molecule is the average distance it travels between collisions. The average time
ENGR 292 Fluids and Thermodynamics
 ENGR 292 Fluids and Thermodynamics Scott Li, Ph.D., P.Eng. Mechanical Engineering Technology Camosun College Timeline Last week, Reading Break Feb.21: Thermodynamics 1 Feb.24: Midterm Review (Fluid Statics
ENGR 292 Fluids and Thermodynamics Scott Li, Ph.D., P.Eng. Mechanical Engineering Technology Camosun College Timeline Last week, Reading Break Feb.21: Thermodynamics 1 Feb.24: Midterm Review (Fluid Statics
C C C C 2 C 2 C 2 C + u + v + (w + w P ) = D t x y z X. (1a) y 2 + D Z. z 2
 This chapter provides an introduction to the transport of particles that are either more dense (e.g. mineral sediment) or less dense (e.g. bubbles) than the fluid. A method of estimating the settling velocity
This chapter provides an introduction to the transport of particles that are either more dense (e.g. mineral sediment) or less dense (e.g. bubbles) than the fluid. A method of estimating the settling velocity
Transport (kinetic) phenomena: diffusion, electric conductivity, viscosity, heat conduction...
 Transport phenomena 1/16 Transport (kinetic) phenomena: diffusion, electric conductivity, viscosity, heat conduction... Flux of mass, charge, momentum, heat,...... J = amount (of quantity) transported
Transport phenomena 1/16 Transport (kinetic) phenomena: diffusion, electric conductivity, viscosity, heat conduction... Flux of mass, charge, momentum, heat,...... J = amount (of quantity) transported
Phys /7/13. November 7, 2013 Physics 131 Prof. E. F. Redish. Theme Music: Duke Ellington Take the A Train. Cartoon: Bill Amend FoxTrot
 November 7, 2013 Physics 131 Prof. E. F. Redish Theme Music: Duke Ellington Take the A Train Cartoon: Bill Amend FoxTrot 1 Tension: The Spring A spring changes its length in response to pulls (or pushes)
November 7, 2013 Physics 131 Prof. E. F. Redish Theme Music: Duke Ellington Take the A Train Cartoon: Bill Amend FoxTrot 1 Tension: The Spring A spring changes its length in response to pulls (or pushes)
17-1 Ideal Gases. Gases are the easiest state of matter to describe - All ideal gases exhibit similar behavior.
 17-1 Ideal Gases Gases are the easiest state of matter to describe - All ideal gases exhibit similar behavior. An ideal gas is one that is thin enough, that the interactions between molecules can be ignored.
17-1 Ideal Gases Gases are the easiest state of matter to describe - All ideal gases exhibit similar behavior. An ideal gas is one that is thin enough, that the interactions between molecules can be ignored.
Part I.
 Part I bblee@unimp . Introduction to Mass Transfer and Diffusion 2. Molecular Diffusion in Gasses 3. Molecular Diffusion in Liquids Part I 4. Molecular Diffusion in Biological Solutions and Gels 5. Molecular
Part I bblee@unimp . Introduction to Mass Transfer and Diffusion 2. Molecular Diffusion in Gasses 3. Molecular Diffusion in Liquids Part I 4. Molecular Diffusion in Biological Solutions and Gels 5. Molecular
Kinetic Model of Gases
 Kinetic Model of Gases Section 1.3 of Atkins, 6th Ed, 24.1 of Atkins, 7th Ed. 21.1 of Atkins, 8th Ed., and 20.1 of Atkins, 9th Ed. Basic Assumptions Molecular Speeds RMS Speed Maxwell Distribution of Speeds
Kinetic Model of Gases Section 1.3 of Atkins, 6th Ed, 24.1 of Atkins, 7th Ed. 21.1 of Atkins, 8th Ed., and 20.1 of Atkins, 9th Ed. Basic Assumptions Molecular Speeds RMS Speed Maxwell Distribution of Speeds
Lecture 24. Ideal Gas Law and Kinetic Theory
 Lecture 4 Ideal Gas Law and Kinetic Theory Today s Topics: Ideal Gas Law Kinetic Theory of Gases Phase equilibria and phase diagrams Ideal Gas Law An ideal gas is an idealized model for real gases that
Lecture 4 Ideal Gas Law and Kinetic Theory Today s Topics: Ideal Gas Law Kinetic Theory of Gases Phase equilibria and phase diagrams Ideal Gas Law An ideal gas is an idealized model for real gases that
Non equilibrium thermodynamics: foundations, scope, and extension to the meso scale. Miguel Rubi
 Non equilibrium thermodynamics: foundations, scope, and extension to the meso scale Miguel Rubi References S.R. de Groot and P. Mazur, Non equilibrium Thermodynamics, Dover, New York, 1984 J.M. Vilar and
Non equilibrium thermodynamics: foundations, scope, and extension to the meso scale Miguel Rubi References S.R. de Groot and P. Mazur, Non equilibrium Thermodynamics, Dover, New York, 1984 J.M. Vilar and
Center of Mass, Improper Integrals
 Unit #14 : Center of Mass, Improper Integrals Goals: Apply the slicing integral approach to computing more complex totals calculations, including center of mass. Learn how to evaluate integrals involving
Unit #14 : Center of Mass, Improper Integrals Goals: Apply the slicing integral approach to computing more complex totals calculations, including center of mass. Learn how to evaluate integrals involving
Appendix 4. Appendix 4A Heat Capacity of Ideal Gases
 Appendix 4 W-143 Appendix 4A Heat Capacity of Ideal Gases We can determine the heat capacity from the energy content of materials as a function of temperature. The simplest material to model is an ideal
Appendix 4 W-143 Appendix 4A Heat Capacity of Ideal Gases We can determine the heat capacity from the energy content of materials as a function of temperature. The simplest material to model is an ideal
6. Transport phenomena. Basel, 2008
 6. Transport phenomena Basel, 2008 6. Transport phenomena 1. Introduction 2. Phenomenological equations for transport properties 3. Transport properties of a perfect gas 4. Diffusion in a fluid 5. Measurement
6. Transport phenomena Basel, 2008 6. Transport phenomena 1. Introduction 2. Phenomenological equations for transport properties 3. Transport properties of a perfect gas 4. Diffusion in a fluid 5. Measurement
Rate of change of velocity. a=dv/dt. Acceleration is a vector quantity.
 9.7 CENTRIFUGATION The centrifuge is a widely used instrument in clinical laboratories for the separation of components. Various quantities are used for the description and the calculation of the separation
9.7 CENTRIFUGATION The centrifuge is a widely used instrument in clinical laboratories for the separation of components. Various quantities are used for the description and the calculation of the separation
2 Equations of Motion
 2 Equations of Motion system. In this section, we will derive the six full equations of motion in a non-rotating, Cartesian coordinate 2.1 Six equations of motion (non-rotating, Cartesian coordinates)
2 Equations of Motion system. In this section, we will derive the six full equations of motion in a non-rotating, Cartesian coordinate 2.1 Six equations of motion (non-rotating, Cartesian coordinates)
Mechanical Engineering. Postal Correspondence Course HEAT TRANSFER. GATE, IES & PSUs
 Heat Transfer-ME GATE, IES, PSU 1 SAMPLE STUDY MATERIAL Mechanical Engineering ME Postal Correspondence Course HEAT TRANSFER GATE, IES & PSUs Heat Transfer-ME GATE, IES, PSU 2 C O N T E N T 1. INTRODUCTION
Heat Transfer-ME GATE, IES, PSU 1 SAMPLE STUDY MATERIAL Mechanical Engineering ME Postal Correspondence Course HEAT TRANSFER GATE, IES & PSUs Heat Transfer-ME GATE, IES, PSU 2 C O N T E N T 1. INTRODUCTION
Lecture 6 Molecular motion and Transport p roperties properties
 Lecture 6 Molecular motion and Transport properties Molecular motion The Aim: Describe the migration of properties through the matter using simple random motion picture Within this lecture: Transport properties
Lecture 6 Molecular motion and Transport properties Molecular motion The Aim: Describe the migration of properties through the matter using simple random motion picture Within this lecture: Transport properties
Physics of biological membranes, diffusion, osmosis Dr. László Nagy
 Physics of biological membranes, diffusion, osmosis Dr. László Nagy -Metabolic processes and transport processes. - Macrotransport : transport of large amount of material : through vessel systems : in
Physics of biological membranes, diffusion, osmosis Dr. László Nagy -Metabolic processes and transport processes. - Macrotransport : transport of large amount of material : through vessel systems : in
K n. III. Gas flow. 1. The nature of the gas : Knudsen s number. 2. Relative flow : Reynold s number R = ( dimensionless )
 III. Gas flow. The nature of the gas : Knudsen s number K n λ d 2. Relative flow : U ρ d η U : stream velocity ρ : mass density Reynold s number R ( dimensionless ) 3. Flow regions - turbulent : R > 2200
III. Gas flow. The nature of the gas : Knudsen s number K n λ d 2. Relative flow : U ρ d η U : stream velocity ρ : mass density Reynold s number R ( dimensionless ) 3. Flow regions - turbulent : R > 2200
Introduction to Mass Transfer
 Introduction to Mass Transfer Introduction Three fundamental transfer processes: i) Momentum transfer ii) iii) Heat transfer Mass transfer Mass transfer may occur in a gas mixture, a liquid solution or
Introduction to Mass Transfer Introduction Three fundamental transfer processes: i) Momentum transfer ii) iii) Heat transfer Mass transfer Mass transfer may occur in a gas mixture, a liquid solution or
Ⅰ. Vacuum Technology. 1. Introduction Vacuum empty in Greek Torricelli Vacuum 2 ) 760 mm. mbar 2 ) Enclosure. To pump. Thin Film Technology
 Ⅰ. Vacuum Technology. Introduction Vacuum empty in Greek 643 Torricelli Vacuum 76 mm Hg Enclosure F ma 76mmHg 76Torr atm (76cm) (3.6g / cm 3 ) (98.665cm/sec ).33 6 dynes/ cm.33bar.33 5 ascal( N / m ).4696
Ⅰ. Vacuum Technology. Introduction Vacuum empty in Greek 643 Torricelli Vacuum 76 mm Hg Enclosure F ma 76mmHg 76Torr atm (76cm) (3.6g / cm 3 ) (98.665cm/sec ).33 6 dynes/ cm.33bar.33 5 ascal( N / m ).4696
2. Molecules in Motion
 2. Molecules in Motion Kinetic Theory of Gases (microscopic viewpoint) assumptions (1) particles of mass m and diameter d; ceaseless random motion (2) dilute gas: d λ, λ = mean free path = average distance
2. Molecules in Motion Kinetic Theory of Gases (microscopic viewpoint) assumptions (1) particles of mass m and diameter d; ceaseless random motion (2) dilute gas: d λ, λ = mean free path = average distance
Kinetic Molecular Theory. 1. What are the postulates of the kinetic molecular theory?
 Kinetic Molecular Theory 1. What are the postulates of the kinetic molecular theory? *These postulates are particularly problematic when it comes to extending this theory to real gases. Regardless of how
Kinetic Molecular Theory 1. What are the postulates of the kinetic molecular theory? *These postulates are particularly problematic when it comes to extending this theory to real gases. Regardless of how
2002NSC Human Physiology Semester Summary
 2002NSC Human Physiology Semester Summary Griffith University, Nathan Campus Semester 1, 2014 Topics include: - Diffusion, Membranes & Action Potentials - Fundamentals of the Nervous System - Neuroanatomy
2002NSC Human Physiology Semester Summary Griffith University, Nathan Campus Semester 1, 2014 Topics include: - Diffusion, Membranes & Action Potentials - Fundamentals of the Nervous System - Neuroanatomy
KINETIC MOLECULAR THEORY
 KINETIC MOLECULAR THEORY IMPORTANT CHARACTERISTICS OF GASES 1) Gases are highly compressible An external force compresses the gas sample and decreases its volume, removing the external force allows the
KINETIC MOLECULAR THEORY IMPORTANT CHARACTERISTICS OF GASES 1) Gases are highly compressible An external force compresses the gas sample and decreases its volume, removing the external force allows the
How DLS Works: Interference of Light
 Static light scattering vs. Dynamic light scattering Static light scattering measures time-average intensities (mean square fluctuations) molecular weight radius of gyration second virial coefficient Dynamic
Static light scattering vs. Dynamic light scattering Static light scattering measures time-average intensities (mean square fluctuations) molecular weight radius of gyration second virial coefficient Dynamic
The corresponding number of atoms per cubic meter is given by:
 Class 10: Drude Model: Source of shortcomings To understand the source of shortcomings in the Drude model, let us numerically estimate some of the important quantities predicted by the Drude model. These
Class 10: Drude Model: Source of shortcomings To understand the source of shortcomings in the Drude model, let us numerically estimate some of the important quantities predicted by the Drude model. These
Chapter 9. Solids and Fluids (c)
 Chapter 9 Solids and Fluids (c) EXAMPLE A small swimming pool has an area of 0 square meters. A wooden 4000-kg statue of density 500 kg/m 3 is then floated on top of the pool. How far does the water rise?
Chapter 9 Solids and Fluids (c) EXAMPLE A small swimming pool has an area of 0 square meters. A wooden 4000-kg statue of density 500 kg/m 3 is then floated on top of the pool. How far does the water rise?
Student Exploration: Temperature and Particle Motion
 Name: Date: Student Exploration: Temperature and Particle Motion Vocabulary: absolute zero, Kelvin scale, kinetic energy, molecule, temperature, Prior Knowledge Questions (Do these BEFORE using the Gizmo.)
Name: Date: Student Exploration: Temperature and Particle Motion Vocabulary: absolute zero, Kelvin scale, kinetic energy, molecule, temperature, Prior Knowledge Questions (Do these BEFORE using the Gizmo.)
ε tran ε tran = nrt = 2 3 N ε tran = 2 3 nn A ε tran nn A nr ε tran = 2 N A i.e. T = R ε tran = 2
 F1 (a) Since the ideal gas equation of state is PV = nrt, we can equate the right-hand sides of both these equations (i.e. with PV = 2 3 N ε tran )and write: nrt = 2 3 N ε tran = 2 3 nn A ε tran i.e. T
F1 (a) Since the ideal gas equation of state is PV = nrt, we can equate the right-hand sides of both these equations (i.e. with PV = 2 3 N ε tran )and write: nrt = 2 3 N ε tran = 2 3 nn A ε tran i.e. T
Theme Music: Robert Alda Luck be a lady Cartoon: Bill Amend FoxTrot. Foothold ideas: Inter-atomic interactions
 February 1, 2013 Prof. E. F. Redish Theme Music: Robert Alda Luck be a lady Cartoon: Bill Amend FoxTrot From Guys & Dolls original cast recording 1 Inter-atomic interactions The interaction between atoms
February 1, 2013 Prof. E. F. Redish Theme Music: Robert Alda Luck be a lady Cartoon: Bill Amend FoxTrot From Guys & Dolls original cast recording 1 Inter-atomic interactions The interaction between atoms
Hydrodynamics: Viscosity and Diffusion Hydrodynamics is the study of mechanics in a liquid, where the frictional drag of the liquid cannot be ignored
 Hydrodynamics: Viscosity and Diffusion Hydrodynamics is the study of mechanics in a liquid, where the frictional drag of the liquid cannot be ignored First let s just consider fluid flow, where the fluid
Hydrodynamics: Viscosity and Diffusion Hydrodynamics is the study of mechanics in a liquid, where the frictional drag of the liquid cannot be ignored First let s just consider fluid flow, where the fluid
Chapter 14 Kinetic Theory
 Chapter 14 Kinetic Theory Kinetic Theory of Gases A remarkable triumph of molecular theory was showing that the macroscopic properties of an ideal gas are related to the molecular properties. This is the
Chapter 14 Kinetic Theory Kinetic Theory of Gases A remarkable triumph of molecular theory was showing that the macroscopic properties of an ideal gas are related to the molecular properties. This is the
Biological and Medical Applications of Pressures and Fluids. Lecture 2.13 MH
 Biological and Medical Applications of Pressures and Fluids Foundation Physics Lecture 2.13 MH Pressures in the human body All pressures quoted are gauge pressure Bladder Pressure Cerebrospinal Pressure
Biological and Medical Applications of Pressures and Fluids Foundation Physics Lecture 2.13 MH Pressures in the human body All pressures quoted are gauge pressure Bladder Pressure Cerebrospinal Pressure
Lecture 3: From Random Walks to Continuum Diffusion
 Lecture 3: From Random Walks to Continuum Diffusion Martin Z. Bazant Department of Mathematics, MIT February 3, 6 Overview In the previous lecture (by Prof. Yip), we discussed how individual particles
Lecture 3: From Random Walks to Continuum Diffusion Martin Z. Bazant Department of Mathematics, MIT February 3, 6 Overview In the previous lecture (by Prof. Yip), we discussed how individual particles
Chemical Processes. Transport Processes in the Environment. Transport Processes. Advection. Advection. t 1 t 2
 hemical Processes by Stefan Trapp (taken from course 4) Transport Processes in the Environment a definition of our nomenklatura Transport Processes Only 3 types of transport processes exist ) by advection
hemical Processes by Stefan Trapp (taken from course 4) Transport Processes in the Environment a definition of our nomenklatura Transport Processes Only 3 types of transport processes exist ) by advection
Statistical Physics. Problem Set 4: Kinetic Theory
 Statistical Physics xford hysics Second year physics course Dr A. A. Schekochihin and Prof. A. Boothroyd (with thanks to Prof. S. J. Blundell) Problem Set 4: Kinetic Theory PROBLEM SET 4: Collisions and
Statistical Physics xford hysics Second year physics course Dr A. A. Schekochihin and Prof. A. Boothroyd (with thanks to Prof. S. J. Blundell) Problem Set 4: Kinetic Theory PROBLEM SET 4: Collisions and
Chapter 2. Electronics I - Semiconductors
 Chapter 2 Electronics I - Semiconductors Fall 2017 talarico@gonzaga.edu 1 Charged Particles The operation of all electronic devices is based on controlling the flow of charged particles There are two type
Chapter 2 Electronics I - Semiconductors Fall 2017 talarico@gonzaga.edu 1 Charged Particles The operation of all electronic devices is based on controlling the flow of charged particles There are two type
Physical Pharmacy. Diffusion
 Physical Pharmacy Diffusion Diffusion Diffusion is defined as a process of mass transfer of individual molecules of a substance brought about by random molecular motion and associated with a driving force
Physical Pharmacy Diffusion Diffusion Diffusion is defined as a process of mass transfer of individual molecules of a substance brought about by random molecular motion and associated with a driving force
BME Engineering Molecular Cell Biology. Basics of the Diffusion Theory. The Cytoskeleton (I)
 BME 42-620 Engineering Molecular Cell Biology Lecture 07: Basics of the Diffusion Theory The Cytoskeleton (I) BME42-620 Lecture 07, September 20, 2011 1 Outline Diffusion: microscopic theory Diffusion:
BME 42-620 Engineering Molecular Cell Biology Lecture 07: Basics of the Diffusion Theory The Cytoskeleton (I) BME42-620 Lecture 07, September 20, 2011 1 Outline Diffusion: microscopic theory Diffusion:
Fundamentals of Plasma Physics Transport in weakly ionized plasmas
 Fundamentals of Plasma Physics Transport in weakly ionized plasmas APPLAuSE Instituto Superior Técnico Instituto de Plasmas e Fusão Nuclear Luís L Alves (based on Vasco Guerra s original slides) 1 As perguntas
Fundamentals of Plasma Physics Transport in weakly ionized plasmas APPLAuSE Instituto Superior Técnico Instituto de Plasmas e Fusão Nuclear Luís L Alves (based on Vasco Guerra s original slides) 1 As perguntas
Matter, Atoms & Molecules
 Matter, Atoms & Molecules Matter is anything that has mass and takes up space. All matter is made of tiny particles called atoms, which are too small to see with the naked eye. Matter Matter is anything
Matter, Atoms & Molecules Matter is anything that has mass and takes up space. All matter is made of tiny particles called atoms, which are too small to see with the naked eye. Matter Matter is anything
Lecture Presentation. Chapter 10. Gases. James F. Kirby Quinnipiac University Hamden, CT Pearson Education, Inc.
 Lecture Presentation Chapter 10 James F. Kirby Quinnipiac University Hamden, CT Characteristics of Physical properties of gases are all similar. Composed mainly of nonmetallic elements with simple formulas
Lecture Presentation Chapter 10 James F. Kirby Quinnipiac University Hamden, CT Characteristics of Physical properties of gases are all similar. Composed mainly of nonmetallic elements with simple formulas
Speed Distribution at CONSTANT Temperature is given by the Maxwell Boltzmann Speed Distribution
 Temperature ~ Average KE of each particle Particles have different speeds Gas Particles are in constant RANDOM motion Average KE of each particle is: 3/2 kt Pressure is due to momentum transfer Speed Distribution
Temperature ~ Average KE of each particle Particles have different speeds Gas Particles are in constant RANDOM motion Average KE of each particle is: 3/2 kt Pressure is due to momentum transfer Speed Distribution
Diffusion of a density in a static fluid
 Diffusion of a density in a static fluid u(x, y, z, t), density (M/L 3 ) of a substance (dye). Diffusion: motion of particles from places where the density is higher to places where it is lower, due to
Diffusion of a density in a static fluid u(x, y, z, t), density (M/L 3 ) of a substance (dye). Diffusion: motion of particles from places where the density is higher to places where it is lower, due to
Introduction to electrophysiology 1. Dr. Tóth András
 Introduction to electrophysiology 1. Dr. Tóth András Today topics Transmembran transport Donnan equilibrium Resting potential Level of significance Entry level (even under 6) Student level (for most of
Introduction to electrophysiology 1. Dr. Tóth András Today topics Transmembran transport Donnan equilibrium Resting potential Level of significance Entry level (even under 6) Student level (for most of
Diffusion By Cindy Grigg
 Diffusion By Cindy Grigg 1 Why do we smell bread baking throughout the house? The answer is diffusion. A good way to describe diffusion is the moving of molecules from a place where they are concentrated
Diffusion By Cindy Grigg 1 Why do we smell bread baking throughout the house? The answer is diffusion. A good way to describe diffusion is the moving of molecules from a place where they are concentrated
Kinetics. Rate of change in response to thermodynamic forces
 Kinetics Rate of change in response to thermodynamic forces Deviation from local equilibrium continuous change T heat flow temperature changes µ atom flow composition changes Deviation from global equilibrium
Kinetics Rate of change in response to thermodynamic forces Deviation from local equilibrium continuous change T heat flow temperature changes µ atom flow composition changes Deviation from global equilibrium
Liquids and solids are essentially incompressible substances and the variation of their density with pressure is usually negligible.
 Properties of Fluids Intensive properties are those that are independent of the mass of a system i.e. temperature, pressure and density. Extensive properties are those whose values depend on the size of
Properties of Fluids Intensive properties are those that are independent of the mass of a system i.e. temperature, pressure and density. Extensive properties are those whose values depend on the size of
Diffusional Growth of Liquid Phase Hydrometeros.
 Diffusional Growth of Liquid Phase Hydrometeros. I. Diffusional Growth of Liquid Phase Hydrometeors A. Basic concepts of diffusional growth. 1. To understand the diffusional growth of a droplet, we must
Diffusional Growth of Liquid Phase Hydrometeros. I. Diffusional Growth of Liquid Phase Hydrometeors A. Basic concepts of diffusional growth. 1. To understand the diffusional growth of a droplet, we must
Convection. forced convection when the flow is caused by external means, such as by a fan, a pump, or atmospheric winds.
 Convection The convection heat transfer mode is comprised of two mechanisms. In addition to energy transfer due to random molecular motion (diffusion), energy is also transferred by the bulk, or macroscopic,
Convection The convection heat transfer mode is comprised of two mechanisms. In addition to energy transfer due to random molecular motion (diffusion), energy is also transferred by the bulk, or macroscopic,
Molecular Motion and Gas Laws
 Molecular Motion and Gas Laws What is the connection between the motion of molecules (F = ma and K = mv 2 /2) and the thermodynamics of gases (pv = nrt and U = 3nRT/2)? In this lab, you will discover how
Molecular Motion and Gas Laws What is the connection between the motion of molecules (F = ma and K = mv 2 /2) and the thermodynamics of gases (pv = nrt and U = 3nRT/2)? In this lab, you will discover how
KINETICE THEROY OF GASES
 INTRODUCTION: Kinetic theory of gases relates the macroscopic properties of gases (like pressure, temperature, volume... etc) to the microscopic properties of the gas molecules (like speed, momentum, kinetic
INTRODUCTION: Kinetic theory of gases relates the macroscopic properties of gases (like pressure, temperature, volume... etc) to the microscopic properties of the gas molecules (like speed, momentum, kinetic
Chapter 7-3 Cells and Their Environment
 Chapter 7-3 Cells and Their Environment 7-3 Passive Transport Passive transport-the movement of substances across the cell membrane without using NRG Concentration Gradient-difference in concentration
Chapter 7-3 Cells and Their Environment 7-3 Passive Transport Passive transport-the movement of substances across the cell membrane without using NRG Concentration Gradient-difference in concentration
Theme Music: Take the A Train Duke Ellington Cartoon: FoxTrot Bill Amend
 November 11, 2016 Prof. E. F. Redish Theme Music: Take the A Train Duke Ellington Cartoon: FoxTrot Bill Amend 1 Basic Principles of Motion Prof. Redish Description of motion v = Δ r vector displacement
November 11, 2016 Prof. E. F. Redish Theme Music: Take the A Train Duke Ellington Cartoon: FoxTrot Bill Amend 1 Basic Principles of Motion Prof. Redish Description of motion v = Δ r vector displacement
Summary of electrostatics
 Summary of electrostatics 1 In electrostatics we deal with the electric effects of charges at rest. Electric charge can be defined as is the intrinsic characteristic that is associated with fundamental
Summary of electrostatics 1 In electrostatics we deal with the electric effects of charges at rest. Electric charge can be defined as is the intrinsic characteristic that is associated with fundamental
Lecture 3. The Kinetic Molecular Theory of Gases
 Lecture 3. The Kinetic Molecular Theory of Gases THE IDEAL GAS LAW: A purely empirical law solely the consequence of experimental observations Explains the behavior of gases over a limited range of conditions
Lecture 3. The Kinetic Molecular Theory of Gases THE IDEAL GAS LAW: A purely empirical law solely the consequence of experimental observations Explains the behavior of gases over a limited range of conditions
Engage 1. Compare the total distance traveled between A and B, if both paths arrive at the factory.
 Unit 1: Phenomenon The Physics of Skydiving Lesson 2.f Displacement and Velocity Student Performance Objectives Students will define displacement. Students will define velocity. Students will differentiate
Unit 1: Phenomenon The Physics of Skydiving Lesson 2.f Displacement and Velocity Student Performance Objectives Students will define displacement. Students will define velocity. Students will differentiate
Special relativity and light RL 4.1, 4.9, 5.4, (6.7)
 Special relativity and light RL 4.1, 4.9, 5.4, (6.7) First: Bremsstrahlung recap Braking radiation, free-free emission Important in hot plasma (e.g. coronae) Most relevant: thermal Bremsstrahlung What
Special relativity and light RL 4.1, 4.9, 5.4, (6.7) First: Bremsstrahlung recap Braking radiation, free-free emission Important in hot plasma (e.g. coronae) Most relevant: thermal Bremsstrahlung What
Fluorescence Spectroscopy
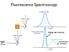 Fluorescence Spectroscopy Raleigh light scattering light all freqs Fluorescence emission Raleigh Scattering 10 nm Raleigh light scattering Fluorescence emission 400 nm Scattering - TWO particle 10 nm Particle
Fluorescence Spectroscopy Raleigh light scattering light all freqs Fluorescence emission Raleigh Scattering 10 nm Raleigh light scattering Fluorescence emission 400 nm Scattering - TWO particle 10 nm Particle
SC/BIOL Current Topics in Biophysics TERM TEST ONE
 Page 1 of 1 SC/BIOL 2090.02 Current Topics in Biophysics TERM TEST ONE Name: KEY Student ID: There are three questions. You must complete all three. Ensure that you show your work (that is, equations,
Page 1 of 1 SC/BIOL 2090.02 Current Topics in Biophysics TERM TEST ONE Name: KEY Student ID: There are three questions. You must complete all three. Ensure that you show your work (that is, equations,
Chapter 2. Motion along a Straight Line
 Chapter 2 Motion along a Straight Line 1 2.1 Motion Everything in the universe, from atoms to galaxies, is in motion. A first step to study motion is to consider simplified cases. In this chapter we study
Chapter 2 Motion along a Straight Line 1 2.1 Motion Everything in the universe, from atoms to galaxies, is in motion. A first step to study motion is to consider simplified cases. In this chapter we study
Chapter 17 Temperature & Kinetic Theory of Gases 1. Thermal Equilibrium and Temperature
 Chapter 17 Temperature & Kinetic Theory of Gases 1. Thermal Equilibrium and Temperature Any physical property that changes with temperature is called a thermometric property and can be used to measure
Chapter 17 Temperature & Kinetic Theory of Gases 1. Thermal Equilibrium and Temperature Any physical property that changes with temperature is called a thermometric property and can be used to measure
Chapter 2: Steady Heat Conduction
 2-1 General Relation for Fourier s Law of Heat Conduction 2-2 Heat Conduction Equation 2-3 Boundary Conditions and Initial Conditions 2-4 Variable Thermal Conductivity 2-5 Steady Heat Conduction in Plane
2-1 General Relation for Fourier s Law of Heat Conduction 2-2 Heat Conduction Equation 2-3 Boundary Conditions and Initial Conditions 2-4 Variable Thermal Conductivity 2-5 Steady Heat Conduction in Plane
