CHL 5225 H Crossover Trials. CHL 5225 H Crossover Trials
|
|
|
- Matilda Rich
- 6 years ago
- Views:
Transcription
1 CHL 55 H Crossover Trials The Two-sequence, Two-Treatment, Two-period Crossover Trial Definition A trial in which patients are randomly allocated to one of two sequences of treatments (either 1 then, or then 1) so that within-patient differences can be used to compare treatments (i.e. patients can be used as their own control) Patients must stop the first treatment and either start the second or enter a washout period at a predetermined point in time which is the same for all patients CHL 55 H Crossover Trials Sequence (order) 1 1 Period 1 Patient receives Treatment 1 Patient receives Treatment Patient receives Treatment Patient receives Treatment 1
2 CHL 55 H Crossover Trials Blocking Blocking may be employed to ensure roughly the same number of patients in each sequence Stratification Blocking within strata to balance for prognostic factors is usually not necessary since patients are their own control CHL 55 H Crossover Trials Appropriate Diseases and Conditions Chronic, relatively stable Manifest as patient symptoms or disability Cyclical conditions such as nausea/vomiting with chemotherapy Appropriate Treatments Transient, non-curative Provide symptom relief Short half-lives
3 CHL 55 H Crossover Trials Appropriate Measurements Subjective symptom scores, ratings of pain, etc. Objective strength Preference which treatment period did the patient prefer CHL 55 H Crossover Trials Blinding Whenever possible patients, clinicians and observers (research staff) should be Blinded (masked) to treatment sequence and, if possible, unaware of the time at which the crossover from one treatment to the other occurs Other Design Features Possible wash-out period between treatment periods Possible baseline measurements prior to both treatment periods
4 CHL 55 H Crossover Trials Advantages Treatments are compared within patients, thereby the influence of patient factors (age, sex, disease severity) are subtracted out ; that is, the between-patient variance is removed, leading to: - smaller variances - increase power - smaller required sample sizes CHL 55 H Crossover Trials Advantages N ( ) 4 z z 1 1 N = number of patients required for a parallel groups trial, n ( z z ) 1 1 (1 ) where n = number of patients required for a crossover trial and = is the correlation between measurements made on the same patient
5 CHL 55 H Crossover Trials Advantages n N 1 Typically is between 0.3 and 0.6 For = 0.5 n 1 N 4 CHL 55 H Crossover Trials Advantages Permits the use of preference data, which is particularly useful if a validated instrument for measuring outcome or disease status is not available
6 CHL 55 H Crossover Trials Disadvantages Short time frame does not permit the assessment of long term benefits and harms Crossover trial can be used because patients are more likely to consent Period effect disease not a stable as expected CHL 55 H Crossover Trials Disadvantages Bad press: 1977 report of the Biometric and Epidemiology Methodolgical Advisory Committee of the US FDA states that the two-period crossover design is not the design of choice in clinical trials where unequivocal evidence of effects is required.
7 CHL 55 H Crossover Trials Disadvantages Treatment by period interaction - sequence (order) effect - residual carryover - partially confounds treatment effect i.e. produces biased estimates of Treatment effect More suitable for an early phase III trial CHL 55 H Crossover Trials Parametric model for continuous outcome i = 1: treat. 1 treat. i = : treat. treat. 1 Let Y ijk be the observed outcome on the j th patient (j = 1,,... n i ) randomized to the i th sequence (i = 1, ) during the k th period (k = 1, ) Y ( k 1) ijk k v (, i k ) i ij ijk where overall mean th effect of k period, 0 k 1 v(i,k) effect of treatment v( ik, ) i* k(mod3), 1 0 i the carryover effect of treatment i from period1to period th th effect of patient in the order ij j i the within patient deviation for period k ijk
8 CHL 55 H Crossover Trials ijk ~ N(0, ) and ~ N(0, ), mutally independent ij Cov Y, Y ijk ijk ' { k : k k' : k' Corr Y, ij1yij Finally, let (period effect) (treatment effect) (residual carryover) (a.k.a. treatment by period interaction a.k.a. sequence effect) Means by Period and i = 1 k = 1 i = k = 1 i = k = i = 1 k = Period 1 Period
9 CHL 55 H Crossover Trials Y ik. 1 n n i i j 1 Sequence (order) Y ijk 1 (i = 1) 1 (i = ) Period k = 1 k = EY ( ) EY ( ) EY ( ) EY ( ) Treatment effect in Period 1: E( Y ) EY ( ) Treatment effect in Period : E( Y ) EY ( ) Therefore, Treatment X Period interaction exists 0 Effect of Sequence 1: { EY ( ) EY ( )} ( ) Effect of Sequence : { EY ( ) EY ( )} ( ) Sequence effect: ( ) 1 Therefore, Sequence effect exists CHL 55 H Crossover Trials Period k = 1 k = Sequence 1 (i = 1) EY ( 1.1) 1 1 EY ( ) (order) 1 (i = ) EY ( ) EY ( ) Estimator of Treatment effect in Sequence 1: Estimator of Treatment effect in Sequence : Y1. Y 1.1 Y.1 Y. Expected value of overall estimator of Treatment effect: E Y1. Y1.1 Y.1 Y. EY ( ) EY ( ) EY ( ) EY ( ) Overall estimator of Treatment effect is biased 0
10 sequence patient period treatment outcome sex m m m 1 60 m f f m m m m f f m m f f f f m m m m m m m m Sample means are rounded to whole numbers i = 1 k = 1 i = k = 1 i = k = i = 1 k = Period 1 Period ˆ ˆ ˆ
11 CHL 55 H Crossover Trials SAS code for the analysis of the X X crossover trial proc MIXED data=sasf.xoverexampledata; class patient; model outcome = sequence period treatment / solution; repeated / subject=patient type=cs r rcorr; title 'All Patients'; run; Because of the coding (1, ) we don t need to declare as class variable. Will give means minus 1 CHL 55 H Crossover Trials Alternative SAS code for the analysis of the X X crossover trial proc MIXED data=sasf.xoverexampledata; class patient sequence period treatment; model outcome = sequence period treatment; repeated / subject=patient type=cs r rcorr; estimate 'PHI' treatment -1 ; estimate 'PI' period -1 ; estimate 'LAMBDA/' sequence -1 ; title 'All Patients'; run;
12 Solution for Fixed Effects Standard Effect Estimate Error DF t Value Pr > t Intercept sequence ( ˆ ) period ( ˆ ) treatment ( ˆ ) Estimated R Correlation Matrix for patient 1 Row Col1 Col ˆ Covariance Parameter Estimates Cov Parm Subject Estimate CS patient Residual ˆ ˆ ˆ ˆ ˆ ˆ = /( ) =
13 CHL 55 H Crossover Trials Covariates examining subgroups data temp; set sasf.xoverexampledata; m1f0 = 1; if sex eq 'f' then m1f0 = 0; m0f1 = 1; if sex eq 'm' then m0f1 = 0; run; proc MIXED data=temp; class patient; model outcome = sequence period treatment m1f0 m1f0*treatment / solution; repeated / subject=patient type=cs r rcorr; title 'Effect in Female Patients'; run; proc MIXED data=temp; class patient; model outcome = sequence period treatment m0f1 m0f1*treatment / solution; repeated / subject=patient type=cs r rcorr; title 'Effect in Male Patients'; run; Standard Effect Estimate Error DF t Value Pr > t Intercept sequence period treatment m1f treatment*m1f effect in females Test for interaction i.e. effect in females = effect in males Standard Effect Estimate Error DF t Value Pr > t Intercept sequence period treatment m0f treatment*m0f effect in males
14 CHL 55 H Crossover Trials Test for treatment effect (i.e. H: 0) is valid 0 0 When might this be true? Placebo-controlled trial Differential conditioning (nausea example) Floor effect (caused by strong period effect) Unmasking (leading to an over estimate of treatment effect in nd period) Differential conditioning (nausea example) i = 1 k = 1 i = k = 1 i = k = i = 1 k = Period 1 Period Means by Period and Treatment
15 Floor effect (caused by strong period effect) i = 1 k = 1 1 i = k = 1 30 i = k = 1 Period 1 Period Means by Period and Treatment 5 i = 1 k = Unmasking i = 1 k = 1 i = k = 1 i = k = 30 0 i = 1 k = Period 1 Period Means by Period and Treatment
16 CHL 55 H Crossover Trials Gotta watch out for 0 0 ˆ (0 5)/.5 (favours treatment 1) This implies physical carryover, and the analysis must be restricted to Period 1 data only (a two-sample t-test or comparable non-parametric test) 100 / i = 1 k = 1 i = k = 1 i = k = i = 1 k = CHL 55 H Crossover Trials But even if 0 0, and therefore test of null hypothesis is valid, E( ˆ ) and is therefore biased Only way to avoid bias is to use first period data only, that is ˆ P.1 Y.1 Y1.1 V( ˆ ) {( n1n) ( nn 1 )} V( ˆ ) {( n n ) ( nn )}( ) P V( ˆ) V( ˆ ) 1 P.1
17 CHL 55 H Crossover Trials The test of hypothesis based on ˆ has more power (i.e. has a greater probability of rejecting the null hypothesis when it is false) than the test based on ˆ P.1 (1 ) Willan and Pater Biometrics 1986; 4: Willan Biometrics 1988; 44:11-18 (1 ) CHL 55 H Crossover Trials When does (1 ) hold????? Well ˆ ˆ P.1 (1 ˆ) holds test statistic( ˆ) > test statistic( ˆ P.1 ) ˆ where ˆ ˆ ˆ is the estimate of Willan (1988) suggests basing analysis on the test statistic with the largest absolute value i.e. declare significance if either one is significant Introduces multiplicity Therefore must adjust level of significance to maintain type I error probability
18 CHL 55 H Crossover Trials ˆ Nominal level for 0.05 Nominal level for CHL 55 H Crossover Trials proc MIXED data=sasf.xoverexampledata; class patient; model outcome = sequence period treatment / solution; repeated / subject=patient type=cs r rcorr; title 'All Patients'; run; proc glm data=sasf.xoverexampledata; where period eq 1; model outcome = treatment / solution; title 'Period 1 Data Only'; run;
19 Standard Effect Estimate Error DF t Value Pr > t Intercept sequence period treatment Estimated R Correlation Row Col1 Col Standard Parameter Estimate Error t Value Pr > t Intercept treatment CHL 55 H Crossover Trials Reject null hypothesis: if the maximum of -4.3 or exceeds cut-off point or equivalently if the smallest associated p-value is less than nominal value from the table for two-sided test, smallest associated p-value is min. of (0.001, 0.57) for one-sided test, smallest associated p-value is min. of ( 0.001, ) Use nominal level of to achieve a type I error probability of 0.05
20 CHL 55 H Crossover Trials Grizzle Procedure Grizzle JE. Biometrics 1965; 1: Test H : 0 using a two-sided level of 0.1 If H is rejected then use period 1 data only If H is not rejected then use data from both periods Not an issue of statistical inference (sample size being the biggest determinant of significance) The issue is the size of and whether it is likely to be non-zero if 0 Standard Effect Estimate Error DF t Value Pr > t Intercept sequence period treatment Since p-value > 0.1, Use data from both periods Estimated R Correlation Row Col1 Col Standard Parameter Estimate Error t Value Pr > t Intercept treatment
21 CHL 55 H Crossover Trials Missing Second Period Data Analyze complete data (i.e. patients with period 1 & data) as before Yields: ˆ C and V( ˆ C ) ( SE ) df C = m C, where m C = # patients with complete data Analyze incomplete (i.e. patients with period 1 data only) data as -sample t-test Yields: ˆ INC and V( ˆ INC ) ( SE ) df INC = m INC, where m INC = # patients with missing period data CHL 55 H Crossover Trials Missing Second Period Data ˆ ˆ ˆ ( ˆ C INC 1 1 V ) ( ˆ ) ( ˆ, where V( ˆ ) V C V INC) ˆ ˆ V( C) V( INC) t t t ˆ V ( ˆ) df df m m 4 n n 4 C INC C INC 1 1 Where n i = # of patients randomized to sequence i There fore m m n n C INC 1
22 CHL 55 H Crossover Trials Binary Outcome (1 = success; 0 = failure) (outcome period 1, outcome period ) Sequence (0,0) (0,1) (1,0) (1,1) Total 1 (1,) (,1) Total Analyze red table using methods appropriate for testing for association in a x table Fisher Exact test Chi-squared tests Use Fisher exact or continuity-corrected Chi-square for small cell frequencies Valid in the presence of period effect, but NOT in the presence of a physical residual carryover effect CHL 55 H Crossover Trials sequence y Frequency Row Pct 1 Total ƒƒƒƒƒƒƒƒƒˆƒƒƒƒƒƒƒƒˆƒƒƒƒƒƒƒƒˆ ƒƒƒƒƒƒƒƒƒˆƒƒƒƒƒƒƒƒˆƒƒƒƒƒƒƒƒˆ ƒƒƒƒƒƒƒƒƒˆƒƒƒƒƒƒƒƒˆƒƒƒƒƒƒƒƒˆ Total Statistics for Table of sequence by y Statistic DF Value Prob ƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒ Chi-Square Likelihood Ratio Chi-Square Fisher's Exact Test ƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒƒ Cell (1,1) Frequency (F) 41 Left-sided Pr <= F Right-sided Pr >= F Table Probability (P) Two-sided Pr <= P
23 CHL 55 H Crossover Trials Alternatively, could use PROC GENMOD in SAS Patient Sequence Period Treatment Outcome ect. proc genmod data=binaryxover; class patient; model outcome = sequence period treatment / dist=bin link=logit; repeated subject=patient / type=exch; run; CHL 55 H Crossover Trials Exchangeable Working Correlation Correlation GEE Fit Criteria QIC QICu Analysis Of GEE Parameter Estimates Empirical Standard Error Estimates Standard 95% Confidence Parameter Estimate Error Limits Z Pr > Z Intercept sequence period treatment (-3.49) = 1.18 Recall: square of normal = chi-squared(1)
24 CHL 55 H Crossover Trials Binary Outcome (outcome period 1, outcome period ) Sequence (0,0) (0,1) (1,0) (1,1) Total 1 (1,) n 11 n 1 n 13 n 14 n 1+ (,1) n 1 n n 3 n 4 n + Total n + n +3 n ++ ( ) n n n n n n1 nnn3 c1 n( n1n3 n13n n ) n n n n LR nij nijn n jn 1 i1 j log ( ) These are two-sided tests Reject at two-sided, level 0.05 if test statistic exceeds 3.84 Reject at one-sided, level 0.05 if test statistic exceeds.7 and observe treatment effect in appropriate direction CHL 55 H Crossover Trials x Cluster Crossover Trial Sequence Cluster Period 1 Period 1 ( 1 ) ( 1) 1 1 (n 11 ) (n 1 ) 1 (n 1 ) (n ) 3 1 (n 31 ) (n 3 ) 4 1 (n 41 ) (n 4 ) 5 1 (n 51 ) (n 5 ) 6 1 (n 61 ) (n 6 ) 7 (n 71 ) 1 (n 7 ) 8 (n 81 ) 1 (n 8 ) 9 (n 91 ) 1 (n 9 ) 10 (n 10,1 ) 1 (n 10, ) 11 (n 11,1 ) 1 (n 11, ) 1 (n 1,1 ) 1 (n 1, ) Assume that n ij = m, and that within a cluster the patients in Period 1 are different from the patients in Period
25 CHL 55 H Crossover Trials x Cluster Crossover Trial sample size for continuous outcome ( z z ) N 1 ( m 1) m 1 1 N = total number of patients required = between patient variance, within a cluster = smallest clinically important difference = ICC for patients from the same cluster in the same period = ICC for patients from the same cluster in different periods c N ( pm) N ( m) c = total number of clusters m = number of patients per period, per cluster p = number of periods CHL 55 H Crossover Trials x Cluster Crossover Trial sample size for binary outcome N ( z z ) m m 1 1 1(1 1) (1 ) 1 ( 1) 1 1 and are the probabilities of the outcome on Treatment 1 and Treatment, respectively, under the alternative hypothesis That is, 1 is the smallest clinically important difference Giraudeau et al. Statist Med 008; 7(7): Rietbergen C. J of Educ and Behav Statistics 011; 36(4):47-490
26 CHL 55 H Crossover Trials PADIT Cluster Crossover Design High risk patients undergoing arrhythmia device procedures Treatment 1 (Control): single dose of preoperative cefazolin Treatment (Intervention): single dose of preoperative cefazolin plus: single dose preoperative of vancomycin plus: intraoperative wound pocket wash plus: postoperative oral cephalexin or cephadroxil The primary outcome of the trial is admission to hospital for proven device or pocket infection Six-month periods Connolly J. et al Canadian J of Cardiology 013; 9(6): CHL 55 H Crossover Trials PADIT Cluster Crossover Design m = 100 = = = 0.05 = 0. 1 = 0.0 = =0.007 ( z z ) N m m 1 1 1(1 1) (1 ) 1 ( 1) 1 ( ) N (0.98) 0.013(0.987) ,661 c N ( m) (x100) 54, assuming x design
27 CHL 55 H Crossover Trials PADIT Cluster Crossover Design Sequence Period 1 Period 1 1 Clusters 1 to patients Clusters 1 to patients 1 Clusters 9 to patients 1 Clusters 9 to patients CHL 55 H Crossover Trials PADIT Cluster Crossover Design Consider 4-sequence 4-period design c N ( pm) (4x100) 7,
28 CHL 55 H Crossover Trials PADIT Cluster Crossover Design Sequence Period 1 Period Period 3 Period Clusters 1 to 7 Clusters 1 to 7 1 Clusters 1 to 7 Clusters 1 to Clusters 8 to 14 1 Clusters 8 to 14 Clusters 8 to 14 1 Clusters 8 to Clusters 15 to 1 Clusters 15 to 1 Clusters 15 to 1 1 Clusters 15 to Clusters to 8 1 Clusters to 8 1 Clusters to 8 Clusters to 8 CHL 55 H Crossover Trials Cluster Crossover Trial analysis for continuous outcome y x e ijk j ij i ij ijk i 1,... # clusters ( c) j 1,... #periods ( p) k 1,...# patients N(0, ); N(0, ); e N(0, ) i ij ijk x 1if cluster i receives treatment during peiod j ij 0 if cluster i receives treatment 1during peiod j
29 apentchl 55 H Crossover Trials Cluster Crossover Trial analysis for binary outcome logit( ) x ij j ij i ij i 1,... # clusters ( c) j 1,... #periods ( p) rob.ofoutcomij fromclustei during period jpr ij N N(0, ); (0, ); i eforatix 1if cluster i receives treatment during peiod j ij 0 if cluster i receives treatment 1during peiod j CHL 55 H Crossover Trials Assignment: First line of data: 1, , , 1, 1, 1, 1
Testing Independence
 Testing Independence Dipankar Bandyopadhyay Department of Biostatistics, Virginia Commonwealth University BIOS 625: Categorical Data & GLM 1/50 Testing Independence Previously, we looked at RR = OR = 1
Testing Independence Dipankar Bandyopadhyay Department of Biostatistics, Virginia Commonwealth University BIOS 625: Categorical Data & GLM 1/50 Testing Independence Previously, we looked at RR = OR = 1
Statistical Methods in Clinical Trials Categorical Data
 Statistical Methods in Clinical Trials Categorical Data Types of Data quantitative Continuous Blood pressure Time to event Categorical sex qualitative Discrete No of relapses Ordered Categorical Pain level
Statistical Methods in Clinical Trials Categorical Data Types of Data quantitative Continuous Blood pressure Time to event Categorical sex qualitative Discrete No of relapses Ordered Categorical Pain level
ESTIMATE PROP. IMPAIRED PRE- AND POST-INTERVENTION FOR THIN LIQUID SWALLOW TASKS. The SURVEYFREQ Procedure
 ESTIMATE PROP. IMPAIRED PRE- AND POST-INTERVENTION FOR THIN LIQUID SWALLOW TASKS 18:58 Sunday, July 26, 2015 1 The SURVEYFREQ Procedure Data Summary Number of Clusters 30 Number of Observations 360 time_cat
ESTIMATE PROP. IMPAIRED PRE- AND POST-INTERVENTION FOR THIN LIQUID SWALLOW TASKS 18:58 Sunday, July 26, 2015 1 The SURVEYFREQ Procedure Data Summary Number of Clusters 30 Number of Observations 360 time_cat
Longitudinal Modeling with Logistic Regression
 Newsom 1 Longitudinal Modeling with Logistic Regression Longitudinal designs involve repeated measurements of the same individuals over time There are two general classes of analyses that correspond to
Newsom 1 Longitudinal Modeling with Logistic Regression Longitudinal designs involve repeated measurements of the same individuals over time There are two general classes of analyses that correspond to
UNIVERSITY OF TORONTO. Faculty of Arts and Science APRIL 2010 EXAMINATIONS STA 303 H1S / STA 1002 HS. Duration - 3 hours. Aids Allowed: Calculator
 UNIVERSITY OF TORONTO Faculty of Arts and Science APRIL 2010 EXAMINATIONS STA 303 H1S / STA 1002 HS Duration - 3 hours Aids Allowed: Calculator LAST NAME: FIRST NAME: STUDENT NUMBER: There are 27 pages
UNIVERSITY OF TORONTO Faculty of Arts and Science APRIL 2010 EXAMINATIONS STA 303 H1S / STA 1002 HS Duration - 3 hours Aids Allowed: Calculator LAST NAME: FIRST NAME: STUDENT NUMBER: There are 27 pages
Contrasting Marginal and Mixed Effects Models Recall: two approaches to handling dependence in Generalized Linear Models:
 Contrasting Marginal and Mixed Effects Models Recall: two approaches to handling dependence in Generalized Linear Models: Marginal models: based on the consequences of dependence on estimating model parameters.
Contrasting Marginal and Mixed Effects Models Recall: two approaches to handling dependence in Generalized Linear Models: Marginal models: based on the consequences of dependence on estimating model parameters.
Models for binary data
 Faculty of Health Sciences Models for binary data Analysis of repeated measurements 2015 Julie Lyng Forman & Lene Theil Skovgaard Department of Biostatistics, University of Copenhagen 1 / 63 Program for
Faculty of Health Sciences Models for binary data Analysis of repeated measurements 2015 Julie Lyng Forman & Lene Theil Skovgaard Department of Biostatistics, University of Copenhagen 1 / 63 Program for
BIOSTATISTICAL METHODS
 BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH Cross-over Designs #: DESIGNING CLINICAL RESEARCH The subtraction of measurements from the same subject will mostly cancel or minimize effects
BIOSTATISTICAL METHODS FOR TRANSLATIONAL & CLINICAL RESEARCH Cross-over Designs #: DESIGNING CLINICAL RESEARCH The subtraction of measurements from the same subject will mostly cancel or minimize effects
Sample Size and Power Considerations for Longitudinal Studies
 Sample Size and Power Considerations for Longitudinal Studies Outline Quantities required to determine the sample size in longitudinal studies Review of type I error, type II error, and power For continuous
Sample Size and Power Considerations for Longitudinal Studies Outline Quantities required to determine the sample size in longitudinal studies Review of type I error, type II error, and power For continuous
Lecture 3.1 Basic Logistic LDA
 y Lecture.1 Basic Logistic LDA 0.2.4.6.8 1 Outline Quick Refresher on Ordinary Logistic Regression and Stata Women s employment example Cross-Over Trial LDA Example -100-50 0 50 100 -- Longitudinal Data
y Lecture.1 Basic Logistic LDA 0.2.4.6.8 1 Outline Quick Refresher on Ordinary Logistic Regression and Stata Women s employment example Cross-Over Trial LDA Example -100-50 0 50 100 -- Longitudinal Data
Clinical Trials. Olli Saarela. September 18, Dalla Lana School of Public Health University of Toronto.
 Introduction to Dalla Lana School of Public Health University of Toronto olli.saarela@utoronto.ca September 18, 2014 38-1 : a review 38-2 Evidence Ideal: to advance the knowledge-base of clinical medicine,
Introduction to Dalla Lana School of Public Health University of Toronto olli.saarela@utoronto.ca September 18, 2014 38-1 : a review 38-2 Evidence Ideal: to advance the knowledge-base of clinical medicine,
SAS Syntax and Output for Data Manipulation: CLDP 944 Example 3a page 1
 CLDP 944 Example 3a page 1 From Between-Person to Within-Person Models for Longitudinal Data The models for this example come from Hoffman (2015) chapter 3 example 3a. We will be examining the extent to
CLDP 944 Example 3a page 1 From Between-Person to Within-Person Models for Longitudinal Data The models for this example come from Hoffman (2015) chapter 3 example 3a. We will be examining the extent to
Logistic Regression. Interpretation of linear regression. Other types of outcomes. 0-1 response variable: Wound infection. Usual linear regression
 Logistic Regression Usual linear regression (repetition) y i = b 0 + b 1 x 1i + b 2 x 2i + e i, e i N(0,σ 2 ) or: y i N(b 0 + b 1 x 1i + b 2 x 2i,σ 2 ) Example (DGA, p. 336): E(PEmax) = 47.355 + 1.024
Logistic Regression Usual linear regression (repetition) y i = b 0 + b 1 x 1i + b 2 x 2i + e i, e i N(0,σ 2 ) or: y i N(b 0 + b 1 x 1i + b 2 x 2i,σ 2 ) Example (DGA, p. 336): E(PEmax) = 47.355 + 1.024
Logistic Regression Analyses in the Water Level Study
 Logistic Regression Analyses in the Water Level Study A. Introduction. 166 students participated in the Water level Study. 70 passed and 96 failed to correctly draw the water level in the glass. There
Logistic Regression Analyses in the Water Level Study A. Introduction. 166 students participated in the Water level Study. 70 passed and 96 failed to correctly draw the water level in the glass. There
Rerandomization to Balance Covariates
 Rerandomization to Balance Covariates Kari Lock Morgan Department of Statistics Penn State University Joint work with Don Rubin University of Minnesota Biostatistics 4/27/16 The Gold Standard Randomized
Rerandomization to Balance Covariates Kari Lock Morgan Department of Statistics Penn State University Joint work with Don Rubin University of Minnesota Biostatistics 4/27/16 The Gold Standard Randomized
You can specify the response in the form of a single variable or in the form of a ratio of two variables denoted events/trials.
 The GENMOD Procedure MODEL Statement MODEL response = < effects > < /options > ; MODEL events/trials = < effects > < /options > ; You can specify the response in the form of a single variable or in the
The GENMOD Procedure MODEL Statement MODEL response = < effects > < /options > ; MODEL events/trials = < effects > < /options > ; You can specify the response in the form of a single variable or in the
Review. Timothy Hanson. Department of Statistics, University of South Carolina. Stat 770: Categorical Data Analysis
 Review Timothy Hanson Department of Statistics, University of South Carolina Stat 770: Categorical Data Analysis 1 / 22 Chapter 1: background Nominal, ordinal, interval data. Distributions: Poisson, binomial,
Review Timothy Hanson Department of Statistics, University of South Carolina Stat 770: Categorical Data Analysis 1 / 22 Chapter 1: background Nominal, ordinal, interval data. Distributions: Poisson, binomial,
Non-Inferiority Tests for the Ratio of Two Proportions in a Cluster- Randomized Design
 Chapter 236 Non-Inferiority Tests for the Ratio of Two Proportions in a Cluster- Randomized Design Introduction This module provides power analysis and sample size calculation for non-inferiority tests
Chapter 236 Non-Inferiority Tests for the Ratio of Two Proportions in a Cluster- Randomized Design Introduction This module provides power analysis and sample size calculation for non-inferiority tests
2 >1. That is, a parallel study design will require
 Cross Over Design Cross over design is commonly used in various type of research for its unique feature of accounting for within subject variability. For studies with short length of treatment time, illness
Cross Over Design Cross over design is commonly used in various type of research for its unique feature of accounting for within subject variability. For studies with short length of treatment time, illness
Statistical tables are attached Two Hours UNIVERSITY OF MANCHESTER. May 2007 Final Draft
 Statistical tables are attached wo Hours UNIVERSIY OF MNHESER Ma 7 Final Draft Medical Statistics M377 Electronic calculators ma be used provided that the cannot store tet nswer LL si questions in SEION
Statistical tables are attached wo Hours UNIVERSIY OF MNHESER Ma 7 Final Draft Medical Statistics M377 Electronic calculators ma be used provided that the cannot store tet nswer LL si questions in SEION
SAS Syntax and Output for Data Manipulation:
 CLP 944 Example 5 page 1 Practice with Fixed and Random Effects of Time in Modeling Within-Person Change The models for this example come from Hoffman (2015) chapter 5. We will be examining the extent
CLP 944 Example 5 page 1 Practice with Fixed and Random Effects of Time in Modeling Within-Person Change The models for this example come from Hoffman (2015) chapter 5. We will be examining the extent
13.1 Categorical Data and the Multinomial Experiment
 Chapter 13 Categorical Data Analysis 13.1 Categorical Data and the Multinomial Experiment Recall Variable: (numerical) variable (i.e. # of students, temperature, height,). (non-numerical, categorical)
Chapter 13 Categorical Data Analysis 13.1 Categorical Data and the Multinomial Experiment Recall Variable: (numerical) variable (i.e. # of students, temperature, height,). (non-numerical, categorical)
MIXED MODELS FOR REPEATED (LONGITUDINAL) DATA PART 2 DAVID C. HOWELL 4/1/2010
 MIXED MODELS FOR REPEATED (LONGITUDINAL) DATA PART 2 DAVID C. HOWELL 4/1/2010 Part 1 of this document can be found at http://www.uvm.edu/~dhowell/methods/supplements/mixed Models for Repeated Measures1.pdf
MIXED MODELS FOR REPEATED (LONGITUDINAL) DATA PART 2 DAVID C. HOWELL 4/1/2010 Part 1 of this document can be found at http://www.uvm.edu/~dhowell/methods/supplements/mixed Models for Repeated Measures1.pdf
Introduction to mtm: An R Package for Marginalized Transition Models
 Introduction to mtm: An R Package for Marginalized Transition Models Bryan A. Comstock and Patrick J. Heagerty Department of Biostatistics University of Washington 1 Introduction Marginalized transition
Introduction to mtm: An R Package for Marginalized Transition Models Bryan A. Comstock and Patrick J. Heagerty Department of Biostatistics University of Washington 1 Introduction Marginalized transition
STA6938-Logistic Regression Model
 Dr. Ying Zhang STA6938-Logistic Regression Model Topic 2-Multiple Logistic Regression Model Outlines:. Model Fitting 2. Statistical Inference for Multiple Logistic Regression Model 3. Interpretation of
Dr. Ying Zhang STA6938-Logistic Regression Model Topic 2-Multiple Logistic Regression Model Outlines:. Model Fitting 2. Statistical Inference for Multiple Logistic Regression Model 3. Interpretation of
Robust covariance estimator for small-sample adjustment in the generalized estimating equations: A simulation study
 Science Journal of Applied Mathematics and Statistics 2014; 2(1): 20-25 Published online February 20, 2014 (http://www.sciencepublishinggroup.com/j/sjams) doi: 10.11648/j.sjams.20140201.13 Robust covariance
Science Journal of Applied Mathematics and Statistics 2014; 2(1): 20-25 Published online February 20, 2014 (http://www.sciencepublishinggroup.com/j/sjams) doi: 10.11648/j.sjams.20140201.13 Robust covariance
Designs for Clinical Trials
 Designs for Clinical Trials Chapter 5 Reading Instructions 5.: Introduction 5.: Parallel Group Designs (read) 5.3: Cluster Randomized Designs (less important) 5.4: Crossover Designs (read+copies) 5.5:
Designs for Clinical Trials Chapter 5 Reading Instructions 5.: Introduction 5.: Parallel Group Designs (read) 5.3: Cluster Randomized Designs (less important) 5.4: Crossover Designs (read+copies) 5.5:
over Time line for the means). Specifically, & covariances) just a fixed variance instead. PROC MIXED: to 1000 is default) list models with TYPE=VC */
 CLP 944 Example 4 page 1 Within-Personn Fluctuation in Symptom Severity over Time These data come from a study of weekly fluctuation in psoriasis severity. There was no intervention and no real reason
CLP 944 Example 4 page 1 Within-Personn Fluctuation in Symptom Severity over Time These data come from a study of weekly fluctuation in psoriasis severity. There was no intervention and no real reason
IP WEIGHTING AND MARGINAL STRUCTURAL MODELS (CHAPTER 12) BIOS IPW and MSM
 IP WEIGHTING AND MARGINAL STRUCTURAL MODELS (CHAPTER 12) BIOS 776 1 12 IPW and MSM IP weighting and marginal structural models ( 12) Outline 12.1 The causal question 12.2 Estimating IP weights via modeling
IP WEIGHTING AND MARGINAL STRUCTURAL MODELS (CHAPTER 12) BIOS 776 1 12 IPW and MSM IP weighting and marginal structural models ( 12) Outline 12.1 The causal question 12.2 Estimating IP weights via modeling
McGill University. Faculty of Science MATH 204 PRINCIPLES OF STATISTICS II. Final Examination
 McGill University Faculty of Science MATH 204 PRINCIPLES OF STATISTICS II Final Examination Date: 20th April 2009 Time: 9am-2pm Examiner: Dr David A Stephens Associate Examiner: Dr Russell Steele Please
McGill University Faculty of Science MATH 204 PRINCIPLES OF STATISTICS II Final Examination Date: 20th April 2009 Time: 9am-2pm Examiner: Dr David A Stephens Associate Examiner: Dr Russell Steele Please
Statistical Methods III Statistics 212. Problem Set 2 - Answer Key
 Statistical Methods III Statistics 212 Problem Set 2 - Answer Key 1. (Analysis to be turned in and discussed on Tuesday, April 24th) The data for this problem are taken from long-term followup of 1423
Statistical Methods III Statistics 212 Problem Set 2 - Answer Key 1. (Analysis to be turned in and discussed on Tuesday, April 24th) The data for this problem are taken from long-term followup of 1423
Epidemiology Wonders of Biostatistics Chapter 13 - Effect Measures. John Koval
 Epidemiology 9509 Wonders of Biostatistics Chapter 13 - Effect Measures John Koval Department of Epidemiology and Biostatistics University of Western Ontario What is being covered 1. risk factors 2. risk
Epidemiology 9509 Wonders of Biostatistics Chapter 13 - Effect Measures John Koval Department of Epidemiology and Biostatistics University of Western Ontario What is being covered 1. risk factors 2. risk
PubH 7470: STATISTICS FOR TRANSLATIONAL & CLINICAL RESEARCH
 PubH 7470: STATISTICS FOR TRANSLATIONAL & CLINICAL RESEARCH The First Step: SAMPLE SIZE DETERMINATION THE ULTIMATE GOAL The most important, ultimate step of any of clinical research is to do draw inferences;
PubH 7470: STATISTICS FOR TRANSLATIONAL & CLINICAL RESEARCH The First Step: SAMPLE SIZE DETERMINATION THE ULTIMATE GOAL The most important, ultimate step of any of clinical research is to do draw inferences;
McGill University. Faculty of Science MATH 204 PRINCIPLES OF STATISTICS II. Final Examination
 McGill University Faculty of Science MATH 204 PRINCIPLES OF STATISTICS II Final Examination Date: 23rd April 2007 Time: 2pm-5pm Examiner: Dr. David A. Stephens Associate Examiner: Dr. Russell Steele Please
McGill University Faculty of Science MATH 204 PRINCIPLES OF STATISTICS II Final Examination Date: 23rd April 2007 Time: 2pm-5pm Examiner: Dr. David A. Stephens Associate Examiner: Dr. Russell Steele Please
Introduction to Crossover Trials
 Introduction to Crossover Trials Stat 6500 Tutorial Project Isaac Blackhurst A crossover trial is a type of randomized control trial. It has advantages over other designed experiments because, under certain
Introduction to Crossover Trials Stat 6500 Tutorial Project Isaac Blackhurst A crossover trial is a type of randomized control trial. It has advantages over other designed experiments because, under certain
Tests for the Odds Ratio of Two Proportions in a 2x2 Cross-Over Design
 Chapter 170 Tests for the Odds Ratio of Two Proportions in a 2x2 Cross-Over Design Introduction Senn (2002) defines a cross-over design as one in which each subject receives all treatments and the objective
Chapter 170 Tests for the Odds Ratio of Two Proportions in a 2x2 Cross-Over Design Introduction Senn (2002) defines a cross-over design as one in which each subject receives all treatments and the objective
Statistics in medicine
 Statistics in medicine Lecture 3: Bivariate association : Categorical variables Proportion in one group One group is measured one time: z test Use the z distribution as an approximation to the binomial
Statistics in medicine Lecture 3: Bivariate association : Categorical variables Proportion in one group One group is measured one time: z test Use the z distribution as an approximation to the binomial
Analysis of variance, multivariate (MANOVA)
 Analysis of variance, multivariate (MANOVA) Abstract: A designed experiment is set up in which the system studied is under the control of an investigator. The individuals, the treatments, the variables
Analysis of variance, multivariate (MANOVA) Abstract: A designed experiment is set up in which the system studied is under the control of an investigator. The individuals, the treatments, the variables
COMPLEMENTARY LOG-LOG MODEL
 COMPLEMENTARY LOG-LOG MODEL Under the assumption of binary response, there are two alternatives to logit model: probit model and complementary-log-log model. They all follow the same form π ( x) =Φ ( α
COMPLEMENTARY LOG-LOG MODEL Under the assumption of binary response, there are two alternatives to logit model: probit model and complementary-log-log model. They all follow the same form π ( x) =Φ ( α
STA 4504/5503 Sample Exam 1 Spring 2011 Categorical Data Analysis. 1. Indicate whether each of the following is true (T) or false (F).
 STA 4504/5503 Sample Exam 1 Spring 2011 Categorical Data Analysis 1. Indicate whether each of the following is true (T) or false (F). (a) (b) (c) (d) (e) In 2 2 tables, statistical independence is equivalent
STA 4504/5503 Sample Exam 1 Spring 2011 Categorical Data Analysis 1. Indicate whether each of the following is true (T) or false (F). (a) (b) (c) (d) (e) In 2 2 tables, statistical independence is equivalent
Lecturer: Dr. Adote Anum, Dept. of Psychology Contact Information:
 Lecturer: Dr. Adote Anum, Dept. of Psychology Contact Information: aanum@ug.edu.gh College of Education School of Continuing and Distance Education 2014/2015 2016/2017 Session Overview In this Session
Lecturer: Dr. Adote Anum, Dept. of Psychology Contact Information: aanum@ug.edu.gh College of Education School of Continuing and Distance Education 2014/2015 2016/2017 Session Overview In this Session
E509A: Principle of Biostatistics. (Week 11(2): Introduction to non-parametric. methods ) GY Zou.
 E509A: Principle of Biostatistics (Week 11(2): Introduction to non-parametric methods ) GY Zou gzou@robarts.ca Sign test for two dependent samples Ex 12.1 subj 1 2 3 4 5 6 7 8 9 10 baseline 166 135 189
E509A: Principle of Biostatistics (Week 11(2): Introduction to non-parametric methods ) GY Zou gzou@robarts.ca Sign test for two dependent samples Ex 12.1 subj 1 2 3 4 5 6 7 8 9 10 baseline 166 135 189
Lecture 2: Poisson and logistic regression
 Dankmar Böhning Southampton Statistical Sciences Research Institute University of Southampton, UK S 3 RI, 11-12 December 2014 introduction to Poisson regression application to the BELCAP study introduction
Dankmar Böhning Southampton Statistical Sciences Research Institute University of Southampton, UK S 3 RI, 11-12 December 2014 introduction to Poisson regression application to the BELCAP study introduction
R Hints for Chapter 10
 R Hints for Chapter 10 The multiple logistic regression model assumes that the success probability p for a binomial random variable depends on independent variables or design variables x 1, x 2,, x k.
R Hints for Chapter 10 The multiple logistic regression model assumes that the success probability p for a binomial random variable depends on independent variables or design variables x 1, x 2,, x k.
STA 4504/5503 Sample Exam 1 Spring 2011 Categorical Data Analysis. 1. Indicate whether each of the following is true (T) or false (F).
 STA 4504/5503 Sample Exam 1 Spring 2011 Categorical Data Analysis 1. Indicate whether each of the following is true (T) or false (F). (a) T In 2 2 tables, statistical independence is equivalent to a population
STA 4504/5503 Sample Exam 1 Spring 2011 Categorical Data Analysis 1. Indicate whether each of the following is true (T) or false (F). (a) T In 2 2 tables, statistical independence is equivalent to a population
Answer to exercise: Blood pressure lowering drugs
 Answer to exercise: Blood pressure lowering drugs The data set bloodpressure.txt contains data from a cross-over trial, involving three different formulations of a drug for lowering of blood pressure:
Answer to exercise: Blood pressure lowering drugs The data set bloodpressure.txt contains data from a cross-over trial, involving three different formulations of a drug for lowering of blood pressure:
BIOS 2083 Linear Models c Abdus S. Wahed
 Chapter 5 206 Chapter 6 General Linear Model: Statistical Inference 6.1 Introduction So far we have discussed formulation of linear models (Chapter 1), estimability of parameters in a linear model (Chapter
Chapter 5 206 Chapter 6 General Linear Model: Statistical Inference 6.1 Introduction So far we have discussed formulation of linear models (Chapter 1), estimability of parameters in a linear model (Chapter
Review of Statistics 101
 Review of Statistics 101 We review some important themes from the course 1. Introduction Statistics- Set of methods for collecting/analyzing data (the art and science of learning from data). Provides methods
Review of Statistics 101 We review some important themes from the course 1. Introduction Statistics- Set of methods for collecting/analyzing data (the art and science of learning from data). Provides methods
Correlated data. Non-normal outcomes. Reminder on binary data. Non-normal data. Faculty of Health Sciences. Non-normal outcomes
 Faculty of Health Sciences Non-normal outcomes Correlated data Non-normal outcomes Lene Theil Skovgaard December 5, 2014 Generalized linear models Generalized linear mixed models Population average models
Faculty of Health Sciences Non-normal outcomes Correlated data Non-normal outcomes Lene Theil Skovgaard December 5, 2014 Generalized linear models Generalized linear mixed models Population average models
ST3241 Categorical Data Analysis I Multicategory Logit Models. Logit Models For Nominal Responses
 ST3241 Categorical Data Analysis I Multicategory Logit Models Logit Models For Nominal Responses 1 Models For Nominal Responses Y is nominal with J categories. Let {π 1,, π J } denote the response probabilities
ST3241 Categorical Data Analysis I Multicategory Logit Models Logit Models For Nominal Responses 1 Models For Nominal Responses Y is nominal with J categories. Let {π 1,, π J } denote the response probabilities
Chapter 1. Modeling Basics
 Chapter 1. Modeling Basics What is a model? Model equation and probability distribution Types of model effects Writing models in matrix form Summary 1 What is a statistical model? A model is a mathematical
Chapter 1. Modeling Basics What is a model? Model equation and probability distribution Types of model effects Writing models in matrix form Summary 1 What is a statistical model? A model is a mathematical
Chapter Seven: Multi-Sample Methods 1/52
 Chapter Seven: Multi-Sample Methods 1/52 7.1 Introduction 2/52 Introduction The independent samples t test and the independent samples Z test for a difference between proportions are designed to analyze
Chapter Seven: Multi-Sample Methods 1/52 7.1 Introduction 2/52 Introduction The independent samples t test and the independent samples Z test for a difference between proportions are designed to analyze
STA 303 H1S / 1002 HS Winter 2011 Test March 7, ab 1cde 2abcde 2fghij 3
 STA 303 H1S / 1002 HS Winter 2011 Test March 7, 2011 LAST NAME: FIRST NAME: STUDENT NUMBER: ENROLLED IN: (circle one) STA 303 STA 1002 INSTRUCTIONS: Time: 90 minutes Aids allowed: calculator. Some formulae
STA 303 H1S / 1002 HS Winter 2011 Test March 7, 2011 LAST NAME: FIRST NAME: STUDENT NUMBER: ENROLLED IN: (circle one) STA 303 STA 1002 INSTRUCTIONS: Time: 90 minutes Aids allowed: calculator. Some formulae
Count data page 1. Count data. 1. Estimating, testing proportions
 Count data page 1 Count data 1. Estimating, testing proportions 100 seeds, 45 germinate. We estimate probability p that a plant will germinate to be 0.45 for this population. Is a 50% germination rate
Count data page 1 Count data 1. Estimating, testing proportions 100 seeds, 45 germinate. We estimate probability p that a plant will germinate to be 0.45 for this population. Is a 50% germination rate
Session 9 Power and sample size
 Session 9 Power and sample size 9.1 Measure of the treatment difference 9.2 The power requirement 9.3 Application to a proportional odds analysis 9.4 Limitations and alternative approaches 9.5 Sample size
Session 9 Power and sample size 9.1 Measure of the treatment difference 9.2 The power requirement 9.3 Application to a proportional odds analysis 9.4 Limitations and alternative approaches 9.5 Sample size
Logistic Regression - problem 6.14
 Logistic Regression - problem 6.14 Let x 1, x 2,, x m be given values of an input variable x and let Y 1,, Y m be independent binomial random variables whose distributions depend on the corresponding values
Logistic Regression - problem 6.14 Let x 1, x 2,, x m be given values of an input variable x and let Y 1,, Y m be independent binomial random variables whose distributions depend on the corresponding values
WITHIN-PARTICIPANT EXPERIMENTAL DESIGNS
 1 WITHIN-PARTICIPANT EXPERIMENTAL DESIGNS I. Single-factor designs: the model is: yij i j ij ij where: yij score for person j under treatment level i (i = 1,..., I; j = 1,..., n) overall mean βi treatment
1 WITHIN-PARTICIPANT EXPERIMENTAL DESIGNS I. Single-factor designs: the model is: yij i j ij ij where: yij score for person j under treatment level i (i = 1,..., I; j = 1,..., n) overall mean βi treatment
Multinomial Logistic Regression Models
 Stat 544, Lecture 19 1 Multinomial Logistic Regression Models Polytomous responses. Logistic regression can be extended to handle responses that are polytomous, i.e. taking r>2 categories. (Note: The word
Stat 544, Lecture 19 1 Multinomial Logistic Regression Models Polytomous responses. Logistic regression can be extended to handle responses that are polytomous, i.e. taking r>2 categories. (Note: The word
Chapter 9. Multivariate and Within-cases Analysis. 9.1 Multivariate Analysis of Variance
 Chapter 9 Multivariate and Within-cases Analysis 9.1 Multivariate Analysis of Variance Multivariate means more than one response variable at once. Why do it? Primarily because if you do parallel analyses
Chapter 9 Multivariate and Within-cases Analysis 9.1 Multivariate Analysis of Variance Multivariate means more than one response variable at once. Why do it? Primarily because if you do parallel analyses
(c) Interpret the estimated effect of temperature on the odds of thermal distress.
 STA 4504/5503 Sample questions for exam 2 1. For the 23 space shuttle flights that occurred before the Challenger mission in 1986, Table 1 shows the temperature ( F) at the time of the flight and whether
STA 4504/5503 Sample questions for exam 2 1. For the 23 space shuttle flights that occurred before the Challenger mission in 1986, Table 1 shows the temperature ( F) at the time of the flight and whether
Lecture 14: Introduction to Poisson Regression
 Lecture 14: Introduction to Poisson Regression Ani Manichaikul amanicha@jhsph.edu 8 May 2007 1 / 52 Overview Modelling counts Contingency tables Poisson regression models 2 / 52 Modelling counts I Why
Lecture 14: Introduction to Poisson Regression Ani Manichaikul amanicha@jhsph.edu 8 May 2007 1 / 52 Overview Modelling counts Contingency tables Poisson regression models 2 / 52 Modelling counts I Why
Modelling counts. Lecture 14: Introduction to Poisson Regression. Overview
 Modelling counts I Lecture 14: Introduction to Poisson Regression Ani Manichaikul amanicha@jhsph.edu Why count data? Number of traffic accidents per day Mortality counts in a given neighborhood, per week
Modelling counts I Lecture 14: Introduction to Poisson Regression Ani Manichaikul amanicha@jhsph.edu Why count data? Number of traffic accidents per day Mortality counts in a given neighborhood, per week
A3. Statistical Inference Hypothesis Testing for General Population Parameters
 Appendix / A3. Statistical Inference / General Parameters- A3. Statistical Inference Hypothesis Testing for General Population Parameters POPULATION H 0 : θ = θ 0 θ is a generic parameter of interest (e.g.,
Appendix / A3. Statistical Inference / General Parameters- A3. Statistical Inference Hypothesis Testing for General Population Parameters POPULATION H 0 : θ = θ 0 θ is a generic parameter of interest (e.g.,
Analysis of Longitudinal Data. Patrick J. Heagerty PhD Department of Biostatistics University of Washington
 Analysis of Longitudinal Data Patrick J Heagerty PhD Department of Biostatistics University of Washington Auckland 8 Session One Outline Examples of longitudinal data Scientific motivation Opportunities
Analysis of Longitudinal Data Patrick J Heagerty PhD Department of Biostatistics University of Washington Auckland 8 Session One Outline Examples of longitudinal data Scientific motivation Opportunities
Binomial Model. Lecture 10: Introduction to Logistic Regression. Logistic Regression. Binomial Distribution. n independent trials
 Lecture : Introduction to Logistic Regression Ani Manichaikul amanicha@jhsph.edu 2 May 27 Binomial Model n independent trials (e.g., coin tosses) p = probability of success on each trial (e.g., p =! =
Lecture : Introduction to Logistic Regression Ani Manichaikul amanicha@jhsph.edu 2 May 27 Binomial Model n independent trials (e.g., coin tosses) p = probability of success on each trial (e.g., p =! =
Monitoring clinical trial outcomes with delayed response: incorporating pipeline data in group sequential designs. Christopher Jennison
 Monitoring clinical trial outcomes with delayed response: incorporating pipeline data in group sequential designs Christopher Jennison Department of Mathematical Sciences, University of Bath http://people.bath.ac.uk/mascj
Monitoring clinical trial outcomes with delayed response: incorporating pipeline data in group sequential designs Christopher Jennison Department of Mathematical Sciences, University of Bath http://people.bath.ac.uk/mascj
Lecture 8: Summary Measures
 Lecture 8: Summary Measures Dipankar Bandyopadhyay, Ph.D. BMTRY 711: Analysis of Categorical Data Spring 2011 Division of Biostatistics and Epidemiology Medical University of South Carolina Lecture 8:
Lecture 8: Summary Measures Dipankar Bandyopadhyay, Ph.D. BMTRY 711: Analysis of Categorical Data Spring 2011 Division of Biostatistics and Epidemiology Medical University of South Carolina Lecture 8:
Lecture 10: Introduction to Logistic Regression
 Lecture 10: Introduction to Logistic Regression Ani Manichaikul amanicha@jhsph.edu 2 May 2007 Logistic Regression Regression for a response variable that follows a binomial distribution Recall the binomial
Lecture 10: Introduction to Logistic Regression Ani Manichaikul amanicha@jhsph.edu 2 May 2007 Logistic Regression Regression for a response variable that follows a binomial distribution Recall the binomial
Ron Heck, Fall Week 3: Notes Building a Two-Level Model
 Ron Heck, Fall 2011 1 EDEP 768E: Seminar on Multilevel Modeling rev. 9/6/2011@11:27pm Week 3: Notes Building a Two-Level Model We will build a model to explain student math achievement using student-level
Ron Heck, Fall 2011 1 EDEP 768E: Seminar on Multilevel Modeling rev. 9/6/2011@11:27pm Week 3: Notes Building a Two-Level Model We will build a model to explain student math achievement using student-level
Faculty of Health Sciences. Correlated data. Count variables. Lene Theil Skovgaard & Julie Lyng Forman. December 6, 2016
 Faculty of Health Sciences Correlated data Count variables Lene Theil Skovgaard & Julie Lyng Forman December 6, 2016 1 / 76 Modeling count outcomes Outline The Poisson distribution for counts Poisson models,
Faculty of Health Sciences Correlated data Count variables Lene Theil Skovgaard & Julie Lyng Forman December 6, 2016 1 / 76 Modeling count outcomes Outline The Poisson distribution for counts Poisson models,
Group Sequential Designs: Theory, Computation and Optimisation
 Group Sequential Designs: Theory, Computation and Optimisation Christopher Jennison Department of Mathematical Sciences, University of Bath, UK http://people.bath.ac.uk/mascj 8th International Conference
Group Sequential Designs: Theory, Computation and Optimisation Christopher Jennison Department of Mathematical Sciences, University of Bath, UK http://people.bath.ac.uk/mascj 8th International Conference
AP Statistics Cumulative AP Exam Study Guide
 AP Statistics Cumulative AP Eam Study Guide Chapters & 3 - Graphs Statistics the science of collecting, analyzing, and drawing conclusions from data. Descriptive methods of organizing and summarizing statistics
AP Statistics Cumulative AP Eam Study Guide Chapters & 3 - Graphs Statistics the science of collecting, analyzing, and drawing conclusions from data. Descriptive methods of organizing and summarizing statistics
Non-parametric methods
 Eastern Mediterranean University Faculty of Medicine Biostatistics course Non-parametric methods March 4&7, 2016 Instructor: Dr. Nimet İlke Akçay (ilke.cetin@emu.edu.tr) Learning Objectives 1. Distinguish
Eastern Mediterranean University Faculty of Medicine Biostatistics course Non-parametric methods March 4&7, 2016 Instructor: Dr. Nimet İlke Akçay (ilke.cetin@emu.edu.tr) Learning Objectives 1. Distinguish
One-Way ANOVA. Some examples of when ANOVA would be appropriate include:
 One-Way ANOVA 1. Purpose Analysis of variance (ANOVA) is used when one wishes to determine whether two or more groups (e.g., classes A, B, and C) differ on some outcome of interest (e.g., an achievement
One-Way ANOVA 1. Purpose Analysis of variance (ANOVA) is used when one wishes to determine whether two or more groups (e.g., classes A, B, and C) differ on some outcome of interest (e.g., an achievement
GEE for Longitudinal Data - Chapter 8
 GEE for Longitudinal Data - Chapter 8 GEE: generalized estimating equations (Liang & Zeger, 1986; Zeger & Liang, 1986) extension of GLM to longitudinal data analysis using quasi-likelihood estimation method
GEE for Longitudinal Data - Chapter 8 GEE: generalized estimating equations (Liang & Zeger, 1986; Zeger & Liang, 1986) extension of GLM to longitudinal data analysis using quasi-likelihood estimation method
Epidemiology Principle of Biostatistics Chapter 14 - Dependent Samples and effect measures. John Koval
 Epidemiology 9509 Principle of Biostatistics Chapter 14 - Dependent Samples and effect measures John Koval Department of Epidemiology and Biostatistics University of Western Ontario What is being covered
Epidemiology 9509 Principle of Biostatistics Chapter 14 - Dependent Samples and effect measures John Koval Department of Epidemiology and Biostatistics University of Western Ontario What is being covered
REGRESSION ANALYSIS FOR TIME-TO-EVENT DATA THE PROPORTIONAL HAZARDS (COX) MODEL ST520
 REGRESSION ANALYSIS FOR TIME-TO-EVENT DATA THE PROPORTIONAL HAZARDS (COX) MODEL ST520 Department of Statistics North Carolina State University Presented by: Butch Tsiatis, Department of Statistics, NCSU
REGRESSION ANALYSIS FOR TIME-TO-EVENT DATA THE PROPORTIONAL HAZARDS (COX) MODEL ST520 Department of Statistics North Carolina State University Presented by: Butch Tsiatis, Department of Statistics, NCSU
STAT 526 Spring Final Exam. Thursday May 5, 2011
 STAT 526 Spring 2011 Final Exam Thursday May 5, 2011 Time: 2 hours Name (please print): Show all your work and calculations. Partial credit will be given for work that is partially correct. Points will
STAT 526 Spring 2011 Final Exam Thursday May 5, 2011 Time: 2 hours Name (please print): Show all your work and calculations. Partial credit will be given for work that is partially correct. Points will
Hypothesis Testing, Power, Sample Size and Confidence Intervals (Part 2)
 Hypothesis Testing, Power, Sample Size and Confidence Intervals (Part 2) B.H. Robbins Scholars Series June 23, 2010 1 / 29 Outline Z-test χ 2 -test Confidence Interval Sample size and power Relative effect
Hypothesis Testing, Power, Sample Size and Confidence Intervals (Part 2) B.H. Robbins Scholars Series June 23, 2010 1 / 29 Outline Z-test χ 2 -test Confidence Interval Sample size and power Relative effect
Estimating the Mean Response of Treatment Duration Regimes in an Observational Study. Anastasios A. Tsiatis.
 Estimating the Mean Response of Treatment Duration Regimes in an Observational Study Anastasios A. Tsiatis http://www.stat.ncsu.edu/ tsiatis/ Introduction to Dynamic Treatment Regimes 1 Outline Description
Estimating the Mean Response of Treatment Duration Regimes in an Observational Study Anastasios A. Tsiatis http://www.stat.ncsu.edu/ tsiatis/ Introduction to Dynamic Treatment Regimes 1 Outline Description
Binary Logistic Regression
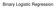 The coefficients of the multiple regression model are estimated using sample data with k independent variables Estimated (or predicted) value of Y Estimated intercept Estimated slope coefficients Ŷ = b
The coefficients of the multiple regression model are estimated using sample data with k independent variables Estimated (or predicted) value of Y Estimated intercept Estimated slope coefficients Ŷ = b
UNIVERSITY OF MASSACHUSETTS Department of Mathematics and Statistics Applied Statistics Friday, January 15, 2016
 UNIVERSITY OF MASSACHUSETTS Department of Mathematics and Statistics Applied Statistics Friday, January 15, 2016 Work all problems. 60 points are needed to pass at the Masters Level and 75 to pass at the
UNIVERSITY OF MASSACHUSETTS Department of Mathematics and Statistics Applied Statistics Friday, January 15, 2016 Work all problems. 60 points are needed to pass at the Masters Level and 75 to pass at the
Analysis of Longitudinal Data: Comparison Between PROC GLM and PROC MIXED. Maribeth Johnson Medical College of Georgia Augusta, GA
 Analysis of Longitudinal Data: Comparison Between PROC GLM and PROC MIXED Maribeth Johnson Medical College of Georgia Augusta, GA Overview Introduction to longitudinal data Describe the data for examples
Analysis of Longitudinal Data: Comparison Between PROC GLM and PROC MIXED Maribeth Johnson Medical College of Georgia Augusta, GA Overview Introduction to longitudinal data Describe the data for examples
Correlated data. Overview. Cross-over study. Repetition. Faculty of Health Sciences. Variance component models, II. More on variance component models
 Faculty of Health Sciences Overview Correlated data More on variance component models Variance component models, II Cross-over studies Non-normal data Comparing measurement devices Lene Theil Skovgaard
Faculty of Health Sciences Overview Correlated data More on variance component models Variance component models, II Cross-over studies Non-normal data Comparing measurement devices Lene Theil Skovgaard
Multilevel Methodology
 Multilevel Methodology Geert Molenberghs Interuniversity Institute for Biostatistics and statistical Bioinformatics Universiteit Hasselt, Belgium geert.molenberghs@uhasselt.be www.censtat.uhasselt.be Katholieke
Multilevel Methodology Geert Molenberghs Interuniversity Institute for Biostatistics and statistical Bioinformatics Universiteit Hasselt, Belgium geert.molenberghs@uhasselt.be www.censtat.uhasselt.be Katholieke
Model Based Statistics in Biology. Part V. The Generalized Linear Model. Chapter 18.1 Logistic Regression (Dose - Response)
 Model Based Statistics in Biology. Part V. The Generalized Linear Model. Logistic Regression ( - Response) ReCap. Part I (Chapters 1,2,3,4), Part II (Ch 5, 6, 7) ReCap Part III (Ch 9, 10, 11), Part IV
Model Based Statistics in Biology. Part V. The Generalized Linear Model. Logistic Regression ( - Response) ReCap. Part I (Chapters 1,2,3,4), Part II (Ch 5, 6, 7) ReCap Part III (Ch 9, 10, 11), Part IV
Propensity Score Methods for Causal Inference
 John Pura BIOS790 October 2, 2015 Causal inference Philosophical problem, statistical solution Important in various disciplines (e.g. Koch s postulates, Bradford Hill criteria, Granger causality) Good
John Pura BIOS790 October 2, 2015 Causal inference Philosophical problem, statistical solution Important in various disciplines (e.g. Koch s postulates, Bradford Hill criteria, Granger causality) Good
Lecture 7 Time-dependent Covariates in Cox Regression
 Lecture 7 Time-dependent Covariates in Cox Regression So far, we ve been considering the following Cox PH model: λ(t Z) = λ 0 (t) exp(β Z) = λ 0 (t) exp( β j Z j ) where β j is the parameter for the the
Lecture 7 Time-dependent Covariates in Cox Regression So far, we ve been considering the following Cox PH model: λ(t Z) = λ 0 (t) exp(β Z) = λ 0 (t) exp( β j Z j ) where β j is the parameter for the the
STAT 5200 Handout #23. Repeated Measures Example (Ch. 16)
 Motivating Example: Glucose STAT 500 Handout #3 Repeated Measures Example (Ch. 16) An experiment is conducted to evaluate the effects of three diets on the serum glucose levels of human subjects. Twelve
Motivating Example: Glucose STAT 500 Handout #3 Repeated Measures Example (Ch. 16) An experiment is conducted to evaluate the effects of three diets on the serum glucose levels of human subjects. Twelve
STAT 5200 Handout #26. Generalized Linear Mixed Models
 STAT 5200 Handout #26 Generalized Linear Mixed Models Up until now, we have assumed our error terms are normally distributed. What if normality is not realistic due to the nature of the data? (For example,
STAT 5200 Handout #26 Generalized Linear Mixed Models Up until now, we have assumed our error terms are normally distributed. What if normality is not realistic due to the nature of the data? (For example,
Mixed Models for Longitudinal Ordinal and Nominal Outcomes
 Mixed Models for Longitudinal Ordinal and Nominal Outcomes Don Hedeker Department of Public Health Sciences Biological Sciences Division University of Chicago hedeker@uchicago.edu Hedeker, D. (2008). Multilevel
Mixed Models for Longitudinal Ordinal and Nominal Outcomes Don Hedeker Department of Public Health Sciences Biological Sciences Division University of Chicago hedeker@uchicago.edu Hedeker, D. (2008). Multilevel
Study Design: Sample Size Calculation & Power Analysis
 Study Design: Sample Size Calculation & Power Analysis RCMAR/CHIME/EXPORT April 21, 2008 Honghu Liu, Ph.D. Contents Background Common Designs Examples Computer Software Summary & Discussion Background
Study Design: Sample Size Calculation & Power Analysis RCMAR/CHIME/EXPORT April 21, 2008 Honghu Liu, Ph.D. Contents Background Common Designs Examples Computer Software Summary & Discussion Background
Section 9c. Propensity scores. Controlling for bias & confounding in observational studies
 Section 9c Propensity scores Controlling for bias & confounding in observational studies 1 Logistic regression and propensity scores Consider comparing an outcome in two treatment groups: A vs B. In a
Section 9c Propensity scores Controlling for bias & confounding in observational studies 1 Logistic regression and propensity scores Consider comparing an outcome in two treatment groups: A vs B. In a
STAT 705 Generalized linear mixed models
 STAT 705 Generalized linear mixed models Timothy Hanson Department of Statistics, University of South Carolina Stat 705: Data Analysis II 1 / 24 Generalized Linear Mixed Models We have considered random
STAT 705 Generalized linear mixed models Timothy Hanson Department of Statistics, University of South Carolina Stat 705: Data Analysis II 1 / 24 Generalized Linear Mixed Models We have considered random
7/28/15. Review Homework. Overview. Lecture 6: Logistic Regression Analysis
 Lecture 6: Logistic Regression Analysis Christopher S. Hollenbeak, PhD Jane R. Schubart, PhD The Outcomes Research Toolbox Review Homework 2 Overview Logistic regression model conceptually Logistic regression
Lecture 6: Logistic Regression Analysis Christopher S. Hollenbeak, PhD Jane R. Schubart, PhD The Outcomes Research Toolbox Review Homework 2 Overview Logistic regression model conceptually Logistic regression
Final Exam. Name: Solution:
 Final Exam. Name: Instructions. Answer all questions on the exam. Open books, open notes, but no electronic devices. The first 13 problems are worth 5 points each. The rest are worth 1 point each. HW1.
Final Exam. Name: Instructions. Answer all questions on the exam. Open books, open notes, but no electronic devices. The first 13 problems are worth 5 points each. The rest are worth 1 point each. HW1.
Salt Lake Community College MATH 1040 Final Exam Fall Semester 2011 Form E
 Salt Lake Community College MATH 1040 Final Exam Fall Semester 011 Form E Name Instructor Time Limit: 10 minutes Any hand-held calculator may be used. Computers, cell phones, or other communication devices
Salt Lake Community College MATH 1040 Final Exam Fall Semester 011 Form E Name Instructor Time Limit: 10 minutes Any hand-held calculator may be used. Computers, cell phones, or other communication devices
Hierarchical Generalized Linear Models. ERSH 8990 REMS Seminar on HLM Last Lecture!
 Hierarchical Generalized Linear Models ERSH 8990 REMS Seminar on HLM Last Lecture! Hierarchical Generalized Linear Models Introduction to generalized models Models for binary outcomes Interpreting parameter
Hierarchical Generalized Linear Models ERSH 8990 REMS Seminar on HLM Last Lecture! Hierarchical Generalized Linear Models Introduction to generalized models Models for binary outcomes Interpreting parameter
Dose-response modeling with bivariate binary data under model uncertainty
 Dose-response modeling with bivariate binary data under model uncertainty Bernhard Klingenberg 1 1 Department of Mathematics and Statistics, Williams College, Williamstown, MA, 01267 and Institute of Statistics,
Dose-response modeling with bivariate binary data under model uncertainty Bernhard Klingenberg 1 1 Department of Mathematics and Statistics, Williams College, Williamstown, MA, 01267 and Institute of Statistics,
SAS Analysis Examples Replication C8. * SAS Analysis Examples Replication for ASDA 2nd Edition * Berglund April 2017 * Chapter 8 ;
 SAS Analysis Examples Replication C8 * SAS Analysis Examples Replication for ASDA 2nd Edition * Berglund April 2017 * Chapter 8 ; libname ncsr "P:\ASDA 2\Data sets\ncsr\" ; data c8_ncsr ; set ncsr.ncsr_sub_13nov2015
SAS Analysis Examples Replication C8 * SAS Analysis Examples Replication for ASDA 2nd Edition * Berglund April 2017 * Chapter 8 ; libname ncsr "P:\ASDA 2\Data sets\ncsr\" ; data c8_ncsr ; set ncsr.ncsr_sub_13nov2015
