Positron Emission Tomography Compartmental Models: A Basis Pursuit Strategy for Kinetic Modelling
|
|
|
- Myrtle Richard
- 6 years ago
- Views:
Transcription
1 Positron Emission Tomography Compartmental Models: A Basis Pursuit Strategy for Kinetic Modelling Roger N. Gunn, Steve R. Gunn, Federico E. Turkheimer, John A. D. Aston, and Vincent J. Cunningham McConnell Brain Imaging Center, Montreal Neurological Institute, McGill University, Montreal, Canada Image, Speech and Intelligent Systems Group, Dept. of Electronics and Computer Science, University of Southampton, U.K. Statistical Research Division, US Census Bureau, Suitland, MD, U.S.A. Imaging Research Solutions Ltd., Cyclotron Building, Hammersmith Hospital, London, U.K. November 14, 2002 Abstract A kinetic modelling approach for the quantification of in vivo tracer studies with dynamic positron emission tomography (PET) is presented. The approach is based on a general compartmental description of the tracer s fate in vivo and determines a parsimonious model consistent with the measured data. The technique involves the determination of a sparse selection of kinetic basis functions from an overcomplete dictionary using the method of basis pursuit denoising. This enables the characterization of the systems impulse response function from which values of the systems macro parameters can be estimated. These parameter estimates can be obtained from a region of interest analysis or as parametric images from a voxel based analysis. In addition, model order estimates are returned which correspond to the number of compartments in the estimated compartmental model. Validation studies evaluate the methods performance against two pre-existing data led techniques, namely graphical analysis and spectral analysis. Application of this technique to measured PET data is demonstrated using [ 11 C]diprenorphine (opiate receptor) and [ 11 C]WAY (5-HT 1A receptor). Whilst, the method is presented in the context of PET neuroreceptor binding studies, it has general applicability to the quantification of PET/SPET radiotracer studies in neurology, oncology and cardiology. Keywords: PET, Tracer Kinetics, Compartmental Modelling, Parameter Estimation, Basis Pursuit Denoising, Sparse Basis Selection 1
2 PET Kinetic Modelling with Basis Pursuit 2 1 Introduction The development of Positron Emission Tomography (PET) over the last two decades has provided neuroscientists with a unique tool for investigating the neurochemistry of the human brain in vivo. The ever increasing library of radiolabelled tracers allows for imaging of a range of biochemical, physiological and pharmacological processes. Each radiotracer has its own distinct behavior in vivo and their characterization is an essential component for the development of new imaging techniques and their translation into clinical applications. Estimation of quantitative biological images from the rich 4D spatio-temporal data sets requires the application of appropriate tomographic reconstruction and tracer kinetic modelling techniques. The latter are used to estimate biological parameters by fitting a mathematical model to the time-activity curve (TAC) of a region of interest or voxel. Calculations at the voxel level produce parametric images but are associated with an increase in noise for the TACs. Analysis strategies must therefore be robust to noise, yet fast enough to be practical. There are a range of quantitative PET tracer kinetic modelling techniques which return biologically based parameter estimates. These techniques may be broadly divided into model-driven methods (Kety, 1951; Sokoloff et al., 1977; Phelps et al., 1979; Mintun et al., 1984; Huang and Phelps, 1986; Gunn et al., 2001) and data-driven methods (Gjedde, 1982; Patlak et al., 1983; Patlak and Blasberg, 1985; Logan et al., 1990, 1996; Cunningham and Jones, 1993). The clear distinction is that the datadriven methods require no a priori decision about the most appropriate model structure. Instead this information is obtained directly from the kinetic data. Model-driven methods use a particular compartmental structure to describe the behaviour of the tracer and allow for an estimation of either micro or macro system parameters. Well established compartmental models in PET, (see Figure 1), include those used for the quantification of blood flow (Kety, 1951), cerebral metabolic rate for glucose (Sokoloff et al., 1977) and for neuroreceptor ligand binding (Mintun et al., 1984). Further developments have produced a series of reference tissue models which avoid the need for blood sampling (Blomqvist et al., 1989; Cunningham et al., 1991; Hume et al., 1992; Lammertsma et al., 1996; Lammertsma and Hume, 1996; Watabe et al., 2000). Parameter estimates are obtained from a priori specified compartmental structures using one of a variety of least squares fitting procedures; linear least squares (Carson, 1986), non-linear least squares (Carson, 1986), generalized linear least squares (Feng et al., 1996), weighted integration (Carson et al., 1986) or basis function techniques (Koeppe et al., 1985; Cunningham and Jones, 1993; Gunn et al., 1997). Data-driven methods such as graphical analysis (Gjedde, 1982; Patlak et al., 1983; Patlak and Blasberg, 1985; Logan et al., 1990, 1996) or spectral analysis (Cunningham and Jones, 1993) derive macro system parameters from a less constrained description of the kinetic processes. The graphical methods (Patlak and Logan plots) employ a transformation of the data such that a linear regression of the transformed data yields the macro system parameter of interest and are attractive and elegant due to their simplicity. However, they require the determination of when the plot becomes linear, they may be biased by statistical noise (Slifstein and Laruelle, 2000) and they fail to return any information about the underlying compartmental structure. Appendix A gives a formal derivation of the Logan plot and shows that it is valid for an arbitrary number of compartments for both plasma and reference input models when the data are free from noise. Spectral analysis (Cunningham and Jones, 1993) characterizes the systems impulse response function (IRF) as a positive sum of exponentials and uses non-negative least squares to fit a set of these basis functions to the data. The macro system parameters of interest are then calculated as functions of the IRF (Cunningham and Jones, 1993; Gunn et al., 2001). Spectral analysis also returns information on the number of tissue compartments evident in the data and is defined as a transparent technique. Schmidt (1999) showed that for the majority of plasma input models the observation of all compartments led to only positive coefficients, and as such the spectral analysis (Cunningham and Jones, 1993) solution using non-negative least squares is valid.
3 PET Kinetic Modelling with Basis Pursuit 3 PET Kinetic Modelling with Basis Pursuit 3 Reversible Irreversible Plasma Reference Figure 1: A range of PET compartmental models commonly used to quantify PET radiotracers. These Figure include1: models A range for tracers of PETthat compartmental exhibit reversible models andcommonly irreversible used kinetics to quantify and models PETwhich radio-ustracers. These include either models a plasma for tracers or reference that exhibit tissuereversible input function. and irreversible kinetics and models which use either a plasma or reference tissue input function. However, it is straightforward to deduce that for reference tissue input models negative coefficients can be theencountered systems impulse and that response this approach function is(irf) strictly asnot a positive valid (see sumappendix of exponentials C in Gunn andetuses al. (2001)). nonnegative least squares to fit a set of these basis functions to the data. The macro system Recently, parameters we have ofpublished interest are general then theory calculated for both as functions plasma input of theand IRF reference (Cunningham tissue and inputjones models (Gunn 1993; et al., Gunn 2001). et al. This 2001). work Spectral showsanalysis that a general also returns PETinformation tracer compartmental on the number system of tissue may be characterized compartments in terms evident of its inimpulse the dataresponse and is defined function as a(irf), transparent and that technique. this is independent Schmidt (1999) of the administration showed that of for the the tracer majority and is therefore of plasmaapplicable input models to both thebolus observation and bolus-infusion of all compartments methodologies. led The to termonly tracer positive excludes coefficients, multipleand injection as suchprotocols the spectral involving analysis low(cunningham specific activity andinjections Jones 1993) which are classed solutionasusing non-tracer non-negative studies least and lead squares to non-linear is valid. However, compartmental it is straightforward systems. In brief, to deduce a general PETthat tracer forcompartmental reference tissuemodel inputleads models to anegative set of first coefficients order linear candifferential be encountered equations. and that Thisthis set of equations approach mayis bestrictly solved not to yield valid an(see expression Appendix forc the in total Gunntissue et al. radioactivity (2001)). concentration in terms of either a plasma input function or a suitable reference tissue input function. These results are derived from Recently, linear systems we have theory published which lead general to the theory deduction for both that plasma the IRF input is comprised and reference solely of tissue exponentials input and a models delta function (Gunn et (Gunn al. 2001). et al., 2001). This work This work shows forms that the a general foundation PET for tracer the presented compartmental method; Data-driven system may estimation be characterized of parametric in terms images of based its impulse on compartmental response function theory (IRF), (DEPICT). and that this is independent of the administration of the tracer and is therefore applicable to both bolus and DEPICT bolus-infusion allows formethodologies. the estimationthe of parametric term tracer images excludes or regional multipleparameter injection protocols values from involving dynamic PETlow data. specific DEPICT activity requires injections no a priori which description are classed as of the non-tracer tracers studies fate in vivo, and lead it derives to non-linear the model description compartmental from the systems. data and it Inreturns brief, athe general number PET of compartments tracer compartmental (model order). model leads This transparent to a set modelling of firsttechnique order linear has differential applicationequations. to a wide range This set of PET of equations radiotracers may but be solved the emphasis to yieldhere an is withexpression respect to for the the analysis totalof tissue radioligands radioactivity whichconcentration bind to specific in neuroreceptor terms of eithersites. a plasma input function or a suitable reference tissue input function. These results are derived from linear systems theory which lead to the deduction that the IRF is comprised solely of exponentials and a delta function (Gunn et al. 2001). This work forms the foundation for the presented method; 2 Theory This section introduces the theory behind DEPICT; Firstly, the general forms for plasma input and reference tissue input models are presented, secondly, key parameters for neuroreceptor binding studies are cast in this framework, and thirdly, the general parameter estimation approach is constructed using
4 PET Kinetic Modelling with Basis Pursuit 4 basis functions and solved via basis pursuit denoising. The method encompasses the majority of linear compartmental systems which are applicable to tracer studies with an arbitrary input function. It assumes that there is only one form (the parent compound) in which radioactivity enters the tissue from the arterial plasma. Specifically, those models which are defined by Definitions 1 and 2 in Gunn et al. (2001) are considered. These sets of models encompass all non-cyclic systems, the subset of cyclic systems in which the product of rate constants is the same regardless of direction for every cycle and requires the eigenvalues of the system to be distinct. 2.1 Plasma Input Models The general equation for a plasma input compartmental model is given by, n = V B C B (t) + (1 V B ) e θit C P (t). (1) where n is the total number of tissue compartments in the target tissue, is the convolution operator, V B is the fractional blood volume, C T,C P, and C B are the concentration time courses in the target tissue, plasma and whole blood respectively. The delivery of the tracer to the tissue is given by n K 1 =. Reversible Kinetics [ > 0] For compartmental models which exhibit reversible kinetics the volume of distribution, V D, which is equal to the integral of the impulse response function is given by, V D = n. (2) Irreversible Kinetics [ n > 0, θ n = 0] For compartmental models which exhibit irreversible kinetics the net irreversible uptake rate constant from plasma, K I, is given by, K I = φ n. (3) 2.2 Reference Tissue Input Models The general equation for a reference tissue input compartmental model is given by, m+n 1 = φ 0 C R (t) + e θit C R (t). (4) where m is the total number of tissue compartments in the reference tissue, and n is the total number of tissue compartments in the target tissue, C T and C R are the concentration time courses in the target and reference tissues respectively and R I (= φ 0 ) is the ratio of delivery of the tracer between
5 PET Kinetic Modelling with Basis Pursuit 5 the target and reference tissue. This definition excludes the presence of blood volume components to either of the tissues. Explicit formulation including a blood volume term has been given previously (Gunn et al., 2001). Reversible Target Tissue Kinetics [ > 0] For reference tissue models which exhibit reversible kinetics in both the target and reference tissues the volume of distribution ratio is given by the integral of the impulse response of the system, V D V D m+n 1 = φ 0 +. (5) Irreversible Target Tissue Kinetics [ m+n 1 > 0, θ m+n 1 = 0] For reference tissue models which exhibit irreversible kinetics in the target tissue and reversible kinetics in the reference tissue the normalised irreversible uptake rate constant from plasma is given by, K I V D = φ m+n 1. (6) 2.3 Parameters for Neuroreceptor Binding Studies For reversible neuroreceptor binding studies the binding potential may be calculated from the macro parameters, using either a plasma or reference tissue input function, under the assumption that the volume of distribution of the free and non-specific binding is the same in the target and reference tissues. Throughout this paper the term binding potential is used interchangeably to refer to BP (the binding potential as originally defined by Mintun et al. (1984)), BP.f 2 and BP.f 1 where f 2 and f 1 are the tissue and plasma free fractions respectively. For a reference tissue input analysis BP.f 2 is the only binding potential estimate possible, whilst with a plasma input analysis it is possible to obtain estimates for BP.f 1, BP.f 2 and if a separate measure of the plasma free fraction exists BP. The binding potential is a useful measure of receptor specific parameters which includes the both the maximum concentration of receptor sites and the affinity, BP = B max K DT racer (1 + i F i K Di ), (7) where B max is the maximum concentration of receptor sites, K DT racer is the equilibrium disassociation rate constant of the radioligand, F i and K Di are the free concentration and equilibrium disassociation constants of i competing ligands. To derive values for both the receptor concentration and affinity it is necessary to perform a multi-injection study with differing specific activities of the the radioligand. A summary of commonly used receptor binding parameters and their calculation from the general form of the IRF are given in Table 1, for further details see (Gunn et al., 2001). The relative merits of these different binding parameters are discussed elsewhere (Laruelle, 2000).
6 PET Kinetic Modelling with Basis Pursuit 6 Parameter Target Reference Input Calculation V D R C n P K I I C P φ n BP.f 1 R R C n P θ m i ϕ i m ϕ i ϑ i n BP.f 2 R R C P 1 ϑ i BP.f 2 R R C R φ m+n 1 K I V D I R C R φ m+n 1 Table 1: Calculation of commonly used binding parameters from the general impulse response function. φ and θ are the parameters for the target tissue, ϕ and ϑ are the parameters for the reference tissue and R and I denote reversible and irreversible tissue kinetics respectively. 2.4 Construction of the General Model in a Basis Function Framework Plasma input 1 and reference tissue input PET compartmental models are characterized by, [ ] q = φ 0 δ(t) + e t C I (t). (8) where C T is the tissue concentration time course and C I is the concentration time course of the input function (plasma or reference tissue). This can be expressed as an expansion on a basis, = N ψ i (t), (9) i=0 where ψ 0 (t) = C I (t), (10) ψ i (t) = 0 e (t τ) C I (τ)dτ. (11) A set of N values for may be pre-chosen from a physiologically plausible range θ min θ max. Here, the values are spaced in a logarithmic manner to elicit a suitable coverage of the kinetic spectrum. Other possibilities for the spacing of the basis exist such as an equi-angular scheme (Cunningham et al., 1998). For data that has not been corrected for the decay of the isotope θ min may be chosen as (or close to the decay constant (θ min = λ min 1 ) for the radioisotope and θ max may be chosen as a suitably large value (θ max = 6 min 1 ). For reversible systems, where the calculation of V D or BP.f 2 is the goal, the choice of a value for θ min which is slightly bigger than λ can suppress the calculation of infinite V D and BP.f 2 values from noisy data. PET measurements are acquired as a sequence of (F ) temporal frames. Thus, the continuous functions must be integrated over the individual frames and normalized to the frame length to correspond to the data sampling procedure. 1 Here, the blood volume term for plasma input models is treated as a plasma volume term (i.e. C B(t) = C P(t)) which enables us to express both the plasma and reference tissue input cases in the same framework. This is simply so that we may be concise in our notation. To use a whole blood volume term ψ 0 = C P is replaced by ψ 0 = C B in equation (10).
7 PET Kinetic Modelling with Basis Pursuit 7 The tissue observations, y, already exist in this form and correspond to, y = [y 1... y F ] T, y j = 1 t e j ts j e j t s j dt, and the matrix of kinetic basis functions (or dictionary), Ψ, are pre-calculated as, ψ 01 ψ ψ N1 Ψ =...., ψ 0F ψ 1F... ψ NF ψ 0j = ψ ij = 1 t e j ts j 1 t e j ts j e j t s j e j t t s j C I (t)dt, 0 e (t τ) C I (τ)dτdt, (12) (13) where t s j and te j are the sequences of start and end frame times (j = 1,..., F ). For all practical purposes (i.e. choosing a large enough value for N to obtain a good coverage of the kinetic spectrum), this leads to an overcomplete basis (N > F 1) which by definition is non-orthogonal. Examples bases for a plasma and reference tissue input are displayed in Figure 2. To determine the fit to the data it is necessary to solve the underdetermined system of equations, y = Ψφ. (14) To account for the temporally varying statistical uncertainty of the measurements it is more appropriate to consider the weighted least squares problem, W 1 2 y = W 1 2 Ψφ, (15) where W is the inverse of the covariance matrix. Given the statistical independence of the frames, W is diagonal. These diagonal elements can be approximated from the total image TAC and frame durations (Mazoyer et al., 1986). The weighted least squares solution is simply obtained by weighting both the data and the basis functions i.e. y is replaced by W 1 2 y and Ψ is replaced by W 1 2 Ψ. On solution of this equation the appropriate macro parameters (V D, K I, K 1, V D V D, K I V D, R I ) may be calculated from the φ s (see equations 2,3,5, and 6). If the data are not corrected for the decay of the isotope, then λ should first be subtracted from the θ values Solution of the General Model by Basis Pursuit Denoising Standard least squares techniques are not applicable because of the overcomplete basis which leads to an under-determined set of equations. This ill-posed problem requires an additional constraint to impose a unique solution on the estimation process. This constraint is chosen to be consistent with prior knowledge about the solution; namely that the solution will be sparse in the basis coefficients. This constraint has also been used in the wavelet community, for the estimation of sparse representations from over-complete dictionaries (Chen, 1995; Chen et al., 1999). The motivation for sparseness is consistent with the expectation that the data is accurately described by a few compartments (such
8 PET Kinetic Modelling with Basis Pursuit 8 (a) Plasma input basis (b) Normalised plasma input basis (c) Reference tissue input basis (d) Normalised reference tissue input basis Figure 2: Example Dictionaries (Ψ) as the models in Figure 1). To transform the problem so that a unique solution exists it is necessary to change the metric from ordinary least squares. The introduction of a regularizer or penalty function to the standard least squares metric offers a framework for this, 1 min W (y Ψφ) φ 2 + µ φ p, (16) 2 where the regularization parameter µ(> 0) controls the trade-off between approximation error and sparseness, Ψ is the overcomplete basis, φ are the basis coefficients to be determined and y is the observed data. The addition of an L p norm to the standard L 2 norm in equation 16 allows for a uniquely determined parameter estimation process. Some possible choices for L p are L 0 (atomic decomposition), L 1 (basis pursuit de-noising), and L 2 (ridge regression) (Chen, 1995). The three regularisers have different characteristics and implementations. Both atomic decomposition and basis pursuit denoising promote a sparse solution which is consistent with our expectation of a compartmental model consisting of just a few tissue compartments, whilst ridge regression will encourage all parameters to be non-zero. Atomic decomposition is computationally demanding due to its combinatorial nature. Both basis pursuit de-noising and ridge regression can be constructed in a quadratic programming framework. Given these factors, a good choice for the estimation process is basis pursuit denoising as this balances both a sparse solution with a computationally feasible parameter estimation approach.
9 PET Kinetic Modelling with Basis Pursuit 9 The basis pursuit denoising solution to the problem is presented here, 1 min W (y Ψφ) φ 2 + µ φ 1, (17) 2 With the introduction of slack variables basis pursuit de-noising can be written as a simple bound constrained quadratic program, (Chen, 1995; Chen et al., 1999), such that x i 0 and where, 1 min x 2 xt Hx + c T x, (18) [ Ψ H = T WΨ Ψ T ] WΨ Ψ T WΨ Ψ T, (19) WΨ [ Ψ c = µ1 T ] Wy Ψ T, (20) Wy [ ] φ + x =. (21) φ The basis coefficients are given by, φ = φ + φ. (22) The quadratic program can be solved readily using standard optimisers (Mészáros, 1998). Thus, given a suitable regularization parameter, the general compartmental model can be fitted to the data. A method which enables the selection of an appropriate regularization parameter is now considered. Selection of the Regularization Parameter (µ) Basis pursuit denoising requires the determination of the regularization parameter, µ, for the penalty term. Here, this is obtained by the method of leave one out cross-validation (LOOCV) (Shao, 1993; Hjorth, 1994). LOOCV is a resampling method which allows the generalisation error to be estimated. Fits are performed in which each data point is omitted in turn, and the generalisation error is then estimated by summing up the prediction error for each omitted data point. The regularization parameter, µ, is chosen to minimize this estimate (see Figure 3). To achieve a more robust estimate of the most appropriate µ value LOOCV is applied to a set of time activity curves and the mean µ value is selected. Model Order and Transparency The presented method is transparent because it returns information about the underlying compartmental structure. The number of non-zero coefficients returned corresponds to the model order which is related to the number of tissue compartments. The number of non-zero components is counted as the number of distinct peaks within the spectrum. The method will often include two peaks next to each other in order to approximate an exponential in between them. This occurrence is treated as a single non-zero coefficient. For plasma input models the model order equals the number of tissue compartments (ignoring the blood volume component) and for reference tissue models the number of non-zero coefficients corresponds to the total number of tissue compartments in the reference and target tissues.
10 PET Kinetic Modelling with Basis Pursuit 11 PET Kinetic Modelling with Basis Pursuit 10 µ Too Small µ Correct µ Too Large D 1 D 3 D 2 Figure 3: Effect of the regularization parameter (µ) and selection via leave one out cross validation: When Figure the 3: µ value Effectis of toothe small regularization the data ( ) are parameter overfitted(µ) and and whenselection the valuevia is too leave bigone the data out cross are underfitted. The best µ value is chosen using LOOCV. A data point is omitted ( ) and the data validation: When the value is too small the data ( ) are overfitted and when the value is are fit for a set of µ values. The error in the models prediction of the omitted data point is then calculated too big the in data a least are squares underfitted. sense (i.e. The D best µ value is chosen using LOOCV. A data point is i 2 ). This is repeated for the omission of each data point in turn omitted and ( ) the and resultant the data generalisation are fit forerrors a set are of µ summated. values. The A µ error is selected in the models by choosing prediction the value of of the µ omitted which minimises data point thisisgeneralisation then calculated error. inthe a least figuresquares shows how sense the(i.e. parsimonious Di 2 ). This model is repeated minimises for the omission of each datathe point generalisation in turn and error the(i.e. resultant D2 2 < Dgeneralisation 3 2 < D1). 2 errors are summated. A µ is selected by choosing the value of µ which minimises this generalisation error. The figure Whilst, shows the how model the order parsimonious dictates the model number minimises of compartments, the generalisation any decision error (i.e. about D2 2 < the D2 3 true < D2 model 1 ). configuration is limited by the problem of indistinguishability. Figures 4 and 5 depict the sets of models components whichis arecounted equivalent as the in terms number of model of distinct order for peaks plasma within input theand spectrum. reference tissue The method input models. will often If one include requires twoa compartmental peaks next todescription each otherofin theorder tracer, toand approximate the set is not ansingular, exponential then itin is between necessary them. to invoke This biological occurrence information is treated about as a the single system non-zero in order coefficient. to select amongst For plasma the possible input models. models the model order equals the number of tissue compartments (ignoring the blood volume component) and for reference tissue models the number of non-zero coefficients corresponds to Figure 4: Indistinguishability for plasma input models. (top) Model Order=1: One possible configuration, (middle) Model Order=2: Two possible configurations, (bottom) Model Order=3: Four the total number of tissue compartments in the reference and target tissues. possible configurations. Whilst, the model order dictates the number of compartments, any decision about the true model configuration is limited by the problem of indistinguishability. Figures 4 and 5 depict the sets of models which are equivalent in terms of model order for plasma input and reference Figure 5: Indistinguishability for reference tissue input models. (top) Model Order=2: One possible tissue configuration, input models. (middle) If one Model requires Order=3: a compartmental Four possible configurations, description (bottom) of the tracer, Model and Order=4: the set is not Eight singular, possible thenconfigurations. it is necessary There to invoke are twice biological as many possible information configurations about the thansystem are shown, in order as to select amongst the possible themodels. reference and target tissues may be reversed. 3 Methods 3 Methods For DEPICT and andspectral analysis the the tissue tissue and and plasma plasma data were data uncorrected were uncorrected for the for decay theofdecay isotope. of the isotope. Instead, Instead, a decay constant a decay was constant allowed was forallowed in the exponential for in the exponential coefficients (decay coefficients constant (decay for [ constant 11 C]: λ = for [ 11 C]: minλ 1 = ) For the min Logan 1 ). analysis For the the Logan tissue analysis and plasma the tissue data were and plasma pre-corrected data were for the pre-corrected decay of thefor isotope. the decay of the isotope.
11 PET Kinetic Modelling with Basis Pursuit 11 DEPICT The Basis Pursuit denoising approach was implemented with 30 basis functions (logarithmically spaced between and 6 min 1 ). The number of basis functions was chosen to be 30 based on a balance between precision and computation time (data not shown). The weighting matrix was determined from the true TAC activity and the frame duration as described previously (Gunn et al., 1997). The regularization parameter, µ, was determined by numerically minimising the LOOCV error across a discrete set of logarithmically spaced µ values ( µ ). For the 1D simulations, the parameter ˆµ was determined as the mean value from the 1000 realisations. For parametric imaging the ˆµ value was obtained from a series of LOOCV estimates (µ i ) obtained from random voxel locations contained within a brain mask. This process examined at least 20 voxels and continued until the relative precision of ˆµ was less than 50%, t 0.975,df σ(µ) N µ < 0.5, (23) where df is the degrees of freedom and is given by the length of µ 1. ˆµ was then estimated as the mean of the vector µ. DEPICT is available at rgunn. Spectral Analysis Spectral analysis was implemented using the non-negative least squares algorithm (Cunningham and Jones, 1993) with the identical basis and weighting matrix as were used for DEPICT. Logan Analysis The Logan analysis (Logan et al., 1990, 1996) was implemented as a linear regression of the transformed data after 45 minutes D Simulations Simulations were performed to assess the stability of the three methods to different noise levels. A measured input function from a PET scan was used in conjunction with a two tissue compartmental model to simulate noise free data representative of a target tissue (K 1 = 0.4 (ml plasma).(ml tissue) 1, k 2 = 0.2 min 1, k 3 = 0.4 min 1 and k 4 = 0.1 min 1 ) and a reference tissue (K 1 = 0.4 (ml plasma).(ml tissue) 1, k 2 = 0.4 min 1, k 5 = 1 min 1 and k 6 = 1 min 1 ). 90 minutes of data were simulated for a total of 24 temporal frames (3x10s, 3x20s, 3x60s, 5x120s, 5x300s, 5x600s). The noise free simulated data are shown in Figure 6. For a range of noise levels 1000 realisations of noisy target tissue TAC s were generated by adding normally distributed noise proportional to the activity/frame duration at each point. The noise variance was scaled so that a value of 1 corresponded to the largest noise free TAC value. These data were then analyzed in two different ways; 1) using the plasma input function the noisy target tissue TAC s were fitted with the basis pursuit method, the Logan plot and spectral analysis to derive V D estimates, 2) using the reference tissue input function the noisy target tissue TAC s were fitted with the basis pursuit method and the Logan plot to derive BP.f 2 estimates.
12 PET Kinetic Modelling with Basis Pursuit 12 (a) Plasma input function (b) Reference tissue TAC (c) Target tissue TAC Figure 6: 1D Simulation Data: Noise free time activity curves 3.2 4D Simulations Simulated dynamic PET data were generated using PETSim, a dynamic PET simulator (Ma et al., 1993; Ma and Evans, 1997) as described previously (Aston et al., 2000). In short, the simulator takes an MRI image volume segmented into 28 regions, each of which is assigned an activity value, and generates a simulated PET volume. The resolution and noise characteristics where chosen to correspond to the ECAT HR+ PET camera (CTI, Knoxville, TN) in 3-D mode with a resolution of 4 x 4 x 4.2 mm FWHM at the center of the field of view and images were reconstructed using a 6mm FWHM Hanning filter. A simulated [ 11 C]SCH data set was generated using a measured plasma input function and a two tissue compartmental model to simulate regional tissue kinetics using rate constants for different brain regions taken from human PET data, K 1 (0.08 to 0.13 ml plasma 1 min 1 ), k 2 (0.24 to 0.37 min 1 ), k 3 (0 to 0.14 min 1 ), k 4 (0.1 min 1 ) for 34 time frames (4x15s, 4x 30s, 7x60s, 5x120s,14x300s). The cerebellum was simulated with a one tissue compartment model. Two dynamic data sets were generated; a noise free data set and the other consistent with an injection of 370MBq of activity. The two data sets were analysed with DEPICT to estimate parametric images of V D and model order. 3.3 Measured Data Sets Two measured data sets, which were taken from ongoing clinical studies (MRC Cyclotron Unit, Hammersmith Hospital, London U.K.), were analysed with DEPICT. Both data sets were acquired on the ECAT EXACT3D (CTI, Knoxville, TN) (Spinks et al., 2000). The data were reconstructed with model based scatter correction and measured attenuation correction using the method of filtered back projection (ramp filter, 0.5 Ny). The reconstructed images had a resolution of 4.8 x 4.8 x 5.6 mm at the centre of the field of view : 11 C Diprenorphine (opiate receptor): 130 MBq of the radioligand was injected into a normal male volunteer and acquisition consisted of 32 temporal frames of data (1x50s, 3x10s, 7x30s, 12x120s, 6x300s, 3x600s). Continuous arterial sampling and metabolite analyses were performed during the scan which allowed for the generation of a metabolite corrected plasma input function as described previously (Jones et al., 1994). DEPICT was used to estimate parametric images of V D and model order using a plasma input analysis. 11 C WAY (5-HT 1A receptor): 243 MBq of the radioligand was injected into a normal male volunteer and acquisition consisted of 22 temporal frames of data (1x15s, 3x5s, 2x15s, 4x60s,
13 PET Kinetic Modelling with Basis Pursuit 13 7x300s, 5x600s). A region of interest was defined on the cerebellum for the extraction of a reference region TAC. DEPICT was used to estimate parametric images of BP.f 2 and model order using a reference tissue input analysis. 4 Results 4.1 1D Simulations A summary of the results from the 1D noise simulations are presented in Figure 7 for the plasma input model. DEPICT was able to obtain a good fit to the data. Both DEPICT and spectral analysis performed well in terms of parameter and model order estimation. Table 2 shows that DEPICT produces the lowest Mean Square Error (MSE) of the three methods. The reference tissue input (a) Example DEPICT fit (b) DEPICT spectrum from fit in (a) (c) DEPICT V D (d) Spectral analysis V D (e) Logan analysis V D (f) DEPICT Model Order (g) Spectral analysis Model Order Figure 7: 1D Simulation: Plasma Analysis. In subfigures (f) and (g) the colour bar indicates the fraction of fits which corresponded to a particular model order.
14 PET Kinetic Modelling with Basis Pursuit 14 simulations are summarised in Figure 8 and Table 3. Again DEPICT obtained a good fit to the data and a reasonable model order estimation. Table 3 shows that DEPICT produces the lowest Mean Square Error (MSE) of the two methods. (a) Example DEPICT fit (b) DEPICT spectrum from fit in (a) (c) DEPICT BP.f 2 (d) Logan analysis BP.f 2 (e) DEPICT Model Order Figure 8: 1D Simulation: Reference Tissue Analysis. In subfigure (e) the colour bar indicates the fraction of fits which corresponded to a particular model order D Simulations Parametric images estimated from the noisy 4D realisation using DEPICT were of good quality and are presented in Figure 9. DEPICT accurately estimates the number of tissue compartments used in the simulation, (see Figure 9b), with two tissue compartments estimated on average within the cortical regions and one tissue compartment on average within the cerebellum. The regularization parameter was calculated as ˆµ = (see Figure 10(a)). DEPICT was also applied to the noise free data set and this allowed for the calculation of the percentage error in the V D estimate. The error distribution is given in Figure 10(b).
15 PET Kinetic Modelling with Basis Pursuit 15 Noise Level DEPICT ˆµ MSE Logan analysis MSE Spectral analysis MSE Table 2: 1D Simulation: Summary Parameters for V D estimation with a plasma input function. Mean Squared Error (MSE), Regularization Parameter ˆµ. Noise Level DEPICT ˆµ MSE Logan analysis MSE Table 3: 1D Simulation: Summary Parameters for BP.f 2 estimation with a reference tissue input function. Mean Squared Error (MSE), Regularization Parameter ˆµ. (a) V D image (b) Model Order image Figure 9: 4D Simulation: Parametric images estimated using DEPICT.
16 PET Kinetic Modelling with Basis Pursuit 16 (a) Cost function for regularization parameter (µ) (b) % Error of V D estimate shown in Figure 9 Figure 10: 4D Simulation: Cost function and error in V D estimate. 4.3 Measured Data Both the parametric images of V D for [ 11 C]diprenorphine and BP.f 2 for [ 11 C]WAY were of good quality and reflected the known distribution of opiate and 5-HT 1A receptor sites respectively. The model order images for both radioligands showed less structure than the 4D simulation but both reflected the model order expected for [ 11 C]diprenorphine (Jones et al., 1994) and [ 11 C]WAY (Gunn et al., 1998; Farde et al., 1998; Parsey et al., 2000). These parametric images took 1 hour to compute with DEPICT on a desktop workstation (equivalent computation times for parametric images generated by the Logan analysis and Spectral analysis would be 5 mins and 30 mins respectively). The regularization parameters were calculated as ˆµ = for [ 11 C]diprenorphine and ˆµ = for [ 11 C]WAY (see Figure 12). 5 Discussion The current paper has introduced, DEPICT, a tracer kinetic modelling technique for the quantitative analysis of dynamic in vivo radiotracer studies which allows for the data-driven estimation of parametric images based on compartmental theory. DEPICT requires no a priori decision about the tracers fate in vivo, instead determining the most appropriate model from the information contained within the data. Although, the method is classed as data-driven it is founded on compartmental theory (Gunn et al., 2001) and this enables parameter estimates to be interpreted within a traditional compartmental framework. The system macro parameters are simply determined from the estimated IRF. The method can be applied to dynamic radiotracer studies involving either a bolus or bolus-infusion tracer administration scheme. DEPICT is general, and whilst the examples here are concerned with tracers exhibiting reversible kinetics, the method is equally applicable to systems with irreversible kinetics. In addition to parameter estimates, DEPICT returns model order estimates which correspond to the number of numerically identifiable compartments in the system. There may be additional compartments that are not supported by the statistical quality of the data, however this is not in any way a practical restriction. DEPICT uses a basis function approach (Koeppe et al., 1985; Cunningham and Jones, 1993; Gunn et al., 1997) to the parameter estimation problem. The constructed problem is ill-posed and its solution requires an appropriate constraint. Whilst, spectral analysis approaches this problem using the nonnegative least squares algorithm (Cunningham and Jones, 1993), DEPICT employs the method of basis
17 PET Kinetic Modelling with Basis Pursuit 17 (a) [ 11 C]Diprenorphine: V D image (b) [ 11 C]Diprenorphine: Model Order image (c) [ 11 C]WAY : BP.f 2 image (d) [ 11 C]WAY : Model Order image Figure 11: Measured data: Parametric images estimated using DEPICT.
18 PET Kinetic Modelling with Basis Pursuit 18 (a) [ 11 C]Diprenorphine (b) [ 11 C]WAY Figure 12: Measured data: Cost functions for regularization parameter (µ). pursuit denoising (Chen, 1995) which involves a 1-norm penalty function on the coefficients. Both methods lead to a sparse solution but DEPICT does not constrain the coefficients to be positive which makes it appropriate for application to the general reference tissue model. Basis pursuit denoising is a technique that extracts a subset of terms from an overcomplete dictionary and thus it is possible to provide a model which is interpretable, whilst retaining good approximating capability. The principle behind the approach is to trade off the error in approximation with the sparseness of the representation. The three data-driven methods investigated all performed well for low noise levels, as determined from the 1D simulations, with DEPICT returning the lowest mean squared error. The Logan analysis demonstrated a bias at higher noise levels which has been documented recently (Slifstein and Laruelle, 2000). To address the issue of noise induced bias in the Logan analysis two approaches have since been developed (Logan et al., 2001; Varga and Szabo, 2002). These modifications would have improved the performance of the Logan analysis at high noise levels but were not considered here as the aim was to introduce DEPICT and compare it against the data-driven methods in common use. DEPICT and spectral analysis allow for the estimation of the model order. The model order was well characterized by both methods for the 1D plasma input simulations and using DEPICT for the reference tissue input simulations. Spectral analysis is not valid for the general reference tissue model as it does not permit negative coefficients which can occur in the impulse response function. In summary, this paper has introduced a new method, DEPICT, which delivers parametric images or regional parameter estimates from dynamic radiotracer imaging studies without the need to specify a compartmental structure. DEPICT is applicable to both plasma and reference tissue input analyses. The results presented demonstrate that DEPICT is highly competitive with existing data-driven estimation methods. Furthermore, DEPICT is a transparent data-driven modelling approach as it returns, not only macro parameter values, but also information on the underlying model structure.
19 PET Kinetic Modelling with Basis Pursuit 19 A Logan Plot derivation Here, a formal derivation of the Logan plot is presented for both a plasma and reference tissue input function. The plasma input Logan plot corresponds to the original presentation by Logan et al. (1990). The reference tissue input analysis presented here differs from Logan et al. (1996) in that it proves that the plot is valid for an arbitrary number of compartments in the reference tissue as well as the target tissue. Both derivations presented exclude the presence of vascular contribution to the tissue signal. A.1 Plasma Input The Logan plot with a plasma input (Logan et al., 1990) is given by, 0 dt 0 V C P(t)dt D + c. (24) From Gunn et al. (2001) the general expression for the target tissue is given by, = n e θit C P (t), (25) and the volume of distribution by, V D = n. (26) Without loss of generality an ordering on the θ s is imposed such that θ 1 > θ 2 >... > θ n. Substituting equation (25) into the left hand side of equation (24) yields, 0 dt = = = = n 0 e θit C P (t)dt, (27) n n n (1 e θit ) C P (t), (28) C P (t) 0 C P(t)dt n n n e θit C P (t), (29) e t C P (t), (30) 0 = V C P(t) θ D i e θit C P (t) n e θit C P (t). (31) For suitably large t, 0 dt 0 V C P(t)dt D 1. (32) θ n
20 PET Kinetic Modelling with Basis Pursuit 20 A.2 Reference Tissue Input The Logan plot with a reference tissue input (Logan et al., 1996) is given by, 0 dt V D 0 C R(t)dt V D + c. (33) From Gunn et al. (2001) the general expression for the target tissue is given by, and the volume of distribution ratio by, m+n 1 = φ 0 δ(t) + e θit C R (t), (34) V D V D m+n 1 = φ 0 +. (35) Without loss of generality an ordering on the θ s is imposed such that θ 1 > θ 2 >... > θ m+n 1. Substituting equation (34) into the left hand side of equation (33) yields, 0 dt For suitably large t, 0 = (φ 0δ(t) + m+n 1 e θit ) C R (t)dt = φ 0 0 C R(t)dt + m+n 1 0 C R(t)dt = = ( φ 0 + ) m+n 1 C R (t) ( φ 0 + m+n 1 = V D 0 C R(t)dt V D ) 0 C R(t)dt m+n 1 m+n 1 m+n 1, (36) m+n 1 e t C R (t), (37) e θit C R (t), (38) e t C R (t) e t C R (t), (39) φ 0 δ(t) + m+n 1 e t C R (t). (40) 0 dt V D 0 C R(t)dt 1 V D. (41) θ m+n 1
21 PET Kinetic Modelling with Basis Pursuit 21 B Glossary Symbol Description Units Concentration time course of radioactivity in the target tissue kbq.ml 1 C R (t) Concentration time course of radioactivity in the reference tissue kbq.ml 1 C P (t) Concentration time course of radioactivity in plasma kbq.ml 1 C B (t) Concentration time course of radioactivity in whole blood kbq.ml 1 C I (t) Concentration time course of radioactivity of the input function kbq.ml 1 φ Parameter vector for coefficients of the general impulse response function θ Parameter vector for exponents of the general impulse response function t s j Start time for the jth temporal frame min t e j End time for the jth temporal frame min n Number of tissue compartments in the target tissue m Number of tissue compartments in the reference tissue q Number of exponential terms in the impulse response function Ψ Matrix of basis functions or Dictionary kbq.ml 1 y Measured PET tissue data kbq.ml 1 µ Regularization parameter V D Volume of distribution of the target tissue (ml plasma).(ml tissue) 1 V D Volume of distribution of the reference tissue (ml plasma).(ml tissue) 1 K I Irreversible uptake rate constant from plasma for the target tissue (ml plasma).min 1.(mL tissue) 1 BP Binding Potential (ml plasma).(ml tissue) 1 V B Fractional blood volume f 1 Plasma free fraction f 2 Tissue free fraction B max Maximum concentration of receptor sites nm K DT racer Equilibrium disassociation rate constant nm F i Free concentration of competing ligand nm K Di Equilibrium disassociation rate constant of competing ligand nm Convolution operator
22 REFERENCES 22 References J. A. D. Aston, R. N. Gunn, K. J. Worsley, Y. Ma, A. C. Evans, and A. Dagher. A statistical method for the analysis of positron emission tomography neuroreceptor ligand data. Neuroimage, 12(3): , G. Blomqvist, S. Pauli, L. Farde, L. Ericksson, A. Persson, and C. Halldin. Clinical Research and Clinical Diagnosis (C. Beckers, A. Goffinet, and A. Bol, Eds), chapter : Dynamic models of reversible ligand binding, pages Kluwer Academic Publishers, R. E. Carson. Positron emission tomography and autoradiography: Principles and applications for the brain and heart, chapter 8: Parameter estimation in positron emission tomography, pages Raven Press, New York, R. E. Carson, S. C. Huang, and M. E. Green. Weighted integration method for local cerebral blood flow measurements with positron emission tomography. J Cereb Blood Flow Metab, 6: , S.S. Chen. Basis Pursuit. Ph.D. Thesis, Dept. of Statistics, Stanford University, S.S. Chen, D.L. Donoho, and M.A. Saunders. Atomic decomposition by basis pursuit. SIAM Journal on Scientific Computing, 20(1):33 61, V. J. Cunningham, S. P. Hume, G. R. Price, R. G. Ahier, J. E. Cremer, and A. K. Jones. Compartmental analysis of diprenorphine binding to opiate receptors in the rat in vivo and its comparison with equilibrium data in vitro. J Cereb Blood Flow Metab, 11(1):1 9, V. J. Cunningham and T. Jones. Spectral analysis of dynamic pet studies. J Cereb Blood Flow Metab, 13(1):15 23., V.J. Cunningham, R.N. Gunn, H. Byrne, and J.C. Matthews. Quantitative functional brain imaging with positron emission tomography (Eds Carson RE, Daube-Witherspoon ME, Herscovitch P), chapter 49: Suppression of noise artefacts in spectral analysis of dynamic PET data, pages Academic Press, L. Farde, H. Ito, C.G. Swahn, V.W. Pike, and C. Halldin. Quantitative analyses of carbonyl-carbon- 11-WAY binding to central 5-hydroxytryptamine-1A receptors in man. J Nucl Med, 39(11): , D. Feng, S. C. Huang, Z. Wang, and D. Ho. An unbiased parametric imaging algorithm for uniformly sampled biomedical system parameter estimation. IEEE Trans Med Imag, 15(4): , A. Gjedde. Calculation of cerebral glucose phosphorylation from brain uptake of glucose analogs in vivo: a re-examination. Brain Res Rev, 4: , R. N. Gunn, S. R. Gunn, and V. J. Cunningham. Positron emission tomography compartmental models. J Cereb Blood Flow Metab, 21(6): , R. N. Gunn, A. A. Lammertsma, S. P. Hume, and V. J. Cunningham. Parametric imaging of ligandreceptor binding in PET using a simplified reference region model. NeuroImage, 6(4): , R. N. Gunn, P. A. Sargent, C. J. Bench, E. A. Rabiner, S. Osman, V. W. Pike, S. P. Hume, P. M. Grasby, and A. A. Lammertsma. Tracer kinetic modeling of the 5-HT 1A receptor ligand [carbonyl- 11C]WAY for PET. Neuroimage, 8(4):426 40, 1998.
Using mathematical models & approaches to quantify BRAIN (dynamic) Positron Emission Tomography (PET) data
 Using mathematical models & approaches to quantify BRAIN (dynamic) Positron Emission Tomography (PET) data Imaging Seminars Series Stony Brook University, Health Science Center Stony Brook, NY - January
Using mathematical models & approaches to quantify BRAIN (dynamic) Positron Emission Tomography (PET) data Imaging Seminars Series Stony Brook University, Health Science Center Stony Brook, NY - January
Basic Principles of Tracer Kinetic Modelling
 The Spectrum of Medical Imaging Basic Principles of Tracer Kinetic Modelling Adriaan A. Lammertsma Structure X-ray/CT/MRI Physiology US, SPECT, PET, MRI/S Metabolism PET, MRS Drug distribution PET Molecular
The Spectrum of Medical Imaging Basic Principles of Tracer Kinetic Modelling Adriaan A. Lammertsma Structure X-ray/CT/MRI Physiology US, SPECT, PET, MRI/S Metabolism PET, MRS Drug distribution PET Molecular
PET Tracer Kinetic Modeling In Drug
 PET Tracer Kinetic Modeling In Drug Discovery Research Applications Sandra M. Sanabria-Bohórquez Imaging Merck & Co., Inc. Positron Emission Tomography - PET PET is an advanced d imaging i technique permitting
PET Tracer Kinetic Modeling In Drug Discovery Research Applications Sandra M. Sanabria-Bohórquez Imaging Merck & Co., Inc. Positron Emission Tomography - PET PET is an advanced d imaging i technique permitting
Statistical estimation with Kronecker products in positron emission tomography
 Linear Algebra and its Applications 398 (2005) 25 36 www.elsevier.com/locate/laa Statistical estimation with Kronecker products in positron emission tomography John A.D. Aston a, Roger N. Gunn b,,1 a Statistical
Linear Algebra and its Applications 398 (2005) 25 36 www.elsevier.com/locate/laa Statistical estimation with Kronecker products in positron emission tomography John A.D. Aston a, Roger N. Gunn b,,1 a Statistical
Strategies to Improve Neuroreceptor Parameter Estimation by Linear Regression Analysis
 Journal of Cerebral Blood Flow & Metabolism 22:1271 1281 22 he International Society for Cerebral Blood Flow and Metabolism Published by Lippincott Williams & Wilkins, Inc., Philadelphia Strategies to
Journal of Cerebral Blood Flow & Metabolism 22:1271 1281 22 he International Society for Cerebral Blood Flow and Metabolism Published by Lippincott Williams & Wilkins, Inc., Philadelphia Strategies to
Journal of Biomedical Research, 2012, 26(2): Review. PET imaging for receptor occupancy: meditations on calculation and simplification
 Journal of Biomedical Research, 2012, 26(2): 69-76 JBR Review PET imaging for receptor occupancy: meditations on calculation and simplification Yumin Zhang *, Gerard B. Fox Translational Sciences, Global
Journal of Biomedical Research, 2012, 26(2): 69-76 JBR Review PET imaging for receptor occupancy: meditations on calculation and simplification Yumin Zhang *, Gerard B. Fox Translational Sciences, Global
Sequential Monte Carlo Methods for Bayesian Model Selection in Positron Emission Tomography
 Methods for Bayesian Model Selection in Positron Emission Tomography Yan Zhou John A.D. Aston and Adam M. Johansen 6th January 2014 Y. Zhou J. A. D. Aston and A. M. Johansen Outline Positron emission tomography
Methods for Bayesian Model Selection in Positron Emission Tomography Yan Zhou John A.D. Aston and Adam M. Johansen 6th January 2014 Y. Zhou J. A. D. Aston and A. M. Johansen Outline Positron emission tomography
Scan Time Optimization for Post-injection PET Scans
 Presented at 998 IEEE Nuc. Sci. Symp. Med. Im. Conf. Scan Time Optimization for Post-injection PET Scans Hakan Erdoğan Jeffrey A. Fessler 445 EECS Bldg., University of Michigan, Ann Arbor, MI 4809-222
Presented at 998 IEEE Nuc. Sci. Symp. Med. Im. Conf. Scan Time Optimization for Post-injection PET Scans Hakan Erdoğan Jeffrey A. Fessler 445 EECS Bldg., University of Michigan, Ann Arbor, MI 4809-222
Tomography is imaging by sections. 1
 Tomography is imaging by sections. 1 It is a technique used in clinical medicine and biomedical research to create images that show how certain tissues are performing their physiological functions. 1 Conversely,
Tomography is imaging by sections. 1 It is a technique used in clinical medicine and biomedical research to create images that show how certain tissues are performing their physiological functions. 1 Conversely,
King s Research Portal
 King s Research Portal DOI: 10.1038/s41598-017-18890-x Document Version Publisher's PDF, also known as Version of record Link to publication record in King's Research Portal Citation for published version
King s Research Portal DOI: 10.1038/s41598-017-18890-x Document Version Publisher's PDF, also known as Version of record Link to publication record in King's Research Portal Citation for published version
On the Undecidability Among Kinetic Models: From Model Selection to Model Averaging
 Journal of Cerebral Blood Flow & Metabolism 23:490 498 2003 The International Society for Cerebral Blood Flow and Metabolism Published by Lippincott Williams & Wilkins, Inc., Philadelphia On the Undecidability
Journal of Cerebral Blood Flow & Metabolism 23:490 498 2003 The International Society for Cerebral Blood Flow and Metabolism Published by Lippincott Williams & Wilkins, Inc., Philadelphia On the Undecidability
EL-GY 6813/BE-GY 6203 Medical Imaging, Fall 2016 Final Exam
 EL-GY 6813/BE-GY 6203 Medical Imaging, Fall 2016 Final Exam (closed book, 1 sheets of notes double sided allowed, no calculator or other electronic devices allowed) 1. Ultrasound Physics (15 pt) A) (9
EL-GY 6813/BE-GY 6203 Medical Imaging, Fall 2016 Final Exam (closed book, 1 sheets of notes double sided allowed, no calculator or other electronic devices allowed) 1. Ultrasound Physics (15 pt) A) (9
An Input Function Estimation Method for. FDG-PET Human Brain Studies 1
 An Input Function Estimation Method for FDG-PET Human Brain Studies 1 Hongbin Guo,2 Rosemary A Renaut 2 Kewei Chen 3 Abbreviated Title: Input Function Estimation for FDG-PET Abstract Introduction: A new
An Input Function Estimation Method for FDG-PET Human Brain Studies 1 Hongbin Guo,2 Rosemary A Renaut 2 Kewei Chen 3 Abbreviated Title: Input Function Estimation for FDG-PET Abstract Introduction: A new
1st Faculty of Medicine, Charles University in Prague Center for Advanced Preclinical Imaging (CAPI)
 Radioation Resolution and Sensitivity Nuclear Imaging PET + SPECT Radioactive Decay (EC,Ɣ), (β -,Ɣ), (I.T.,Ɣ) β + Projection imaging collimator needed one angular view Projection imaging coincidence imaging,
Radioation Resolution and Sensitivity Nuclear Imaging PET + SPECT Radioactive Decay (EC,Ɣ), (β -,Ɣ), (I.T.,Ɣ) β + Projection imaging collimator needed one angular view Projection imaging coincidence imaging,
GLOBAL KINETIC IMAGING USING DYNAMIC POSITRON EMISSION TOMOGRAPHY DATA by Cristina Negoita
 GLOBAL KINETIC IMAGING USING DYNAMIC POSITRON EMISSION TOMOGRAPHY DATA by Cristina Negoita A Dissertation Presented in Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy ARIZONA
GLOBAL KINETIC IMAGING USING DYNAMIC POSITRON EMISSION TOMOGRAPHY DATA by Cristina Negoita A Dissertation Presented in Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy ARIZONA
Spectral analysis: principle and clinical applications
 REVIEW Annals of Nuclear Medicine Vol. 17, No. 6, 427 434, 2003 Spectral analysis: principle and clinical applications Kenya MURASE Department of Medical Physics and Engineering, Division of Medical Technology
REVIEW Annals of Nuclear Medicine Vol. 17, No. 6, 427 434, 2003 Spectral analysis: principle and clinical applications Kenya MURASE Department of Medical Physics and Engineering, Division of Medical Technology
Parameter identification in medical imaging
 Trabalho apresentado no XXXVII CNMAC, S.J. dos Campos - SP, 017. Proceeding Series of the Brazilian Society of Computational and Applied Mathematics Parameter identification in medical imaging Louise Reips
Trabalho apresentado no XXXVII CNMAC, S.J. dos Campos - SP, 017. Proceeding Series of the Brazilian Society of Computational and Applied Mathematics Parameter identification in medical imaging Louise Reips
Faculty of Sciences. Analysis of Positron Emission Tomography data: A comparison between approaches for linearized time-activity curves.
 Faculty of Sciences Analysis of Positron Emission Tomography data: A comparison between approaches for linearized time-activity curves Bart de Laat Master dissertation submitted to obtain the degree of
Faculty of Sciences Analysis of Positron Emission Tomography data: A comparison between approaches for linearized time-activity curves Bart de Laat Master dissertation submitted to obtain the degree of
Graphical Evaluation of Blood-to-Brain Transfer Constants from Multiple-T ime Uptake Data. Generalizations
 Journal o/cerebral Blood Flow and Metabolism 5:584-590 1985 Raven Press, New York Graphical Evaluation of Blood-to-Brain Transfer Constants from Multiple-T ime Uptake Data. Generalizations Clifford S.
Journal o/cerebral Blood Flow and Metabolism 5:584-590 1985 Raven Press, New York Graphical Evaluation of Blood-to-Brain Transfer Constants from Multiple-T ime Uptake Data. Generalizations Clifford S.
Optimal design for inverse problems
 School of Mathematics and Statistical Sciences Research Institute University of Southampton, UK Joint work with Nicolai Bissantz, Holger Dette (both Ruhr-Universität Bochum) and Edmund Jones (University
School of Mathematics and Statistical Sciences Research Institute University of Southampton, UK Joint work with Nicolai Bissantz, Holger Dette (both Ruhr-Universität Bochum) and Edmund Jones (University
NIH Public Access Author Manuscript Phys Med Biol. Author manuscript; available in PMC 2013 September 21.
 NIH Public Access Author Manuscript Published in final edited form as: Phys Med Biol. 2012 September 21; 57(18): 5809 5821. doi:10.1088/0031-9155/57/18/5809. Kinetic Parameter Estimation Using a Closed-Form
NIH Public Access Author Manuscript Published in final edited form as: Phys Med Biol. 2012 September 21; 57(18): 5809 5821. doi:10.1088/0031-9155/57/18/5809. Kinetic Parameter Estimation Using a Closed-Form
Degrees of Freedom in Regression Ensembles
 Degrees of Freedom in Regression Ensembles Henry WJ Reeve Gavin Brown University of Manchester - School of Computer Science Kilburn Building, University of Manchester, Oxford Rd, Manchester M13 9PL Abstract.
Degrees of Freedom in Regression Ensembles Henry WJ Reeve Gavin Brown University of Manchester - School of Computer Science Kilburn Building, University of Manchester, Oxford Rd, Manchester M13 9PL Abstract.
The General Linear Model. Guillaume Flandin Wellcome Trust Centre for Neuroimaging University College London
 The General Linear Model Guillaume Flandin Wellcome Trust Centre for Neuroimaging University College London SPM Course Lausanne, April 2012 Image time-series Spatial filter Design matrix Statistical Parametric
The General Linear Model Guillaume Flandin Wellcome Trust Centre for Neuroimaging University College London SPM Course Lausanne, April 2012 Image time-series Spatial filter Design matrix Statistical Parametric
Introduction to SPECT & PET TBMI02 - Medical Image Analysis 2017
 Introduction to SPECT & PET TBMI02 - Medical Image Analysis 2017 Marcus Ressner, PhD, Medical Radiation Physicist, Linköping University Hospital Content What is Nuclear medicine? Basic principles of Functional
Introduction to SPECT & PET TBMI02 - Medical Image Analysis 2017 Marcus Ressner, PhD, Medical Radiation Physicist, Linköping University Hospital Content What is Nuclear medicine? Basic principles of Functional
Physics in Nuclear Medicine
 SIMON R. CHERRY, PH.D. Professor Department of Biomedical Engineering University of California-Davis Davis, California JAMES A. SORENSON, PH.D. Emeritus Professor of Medical Physics University of Wisconsin-Madison
SIMON R. CHERRY, PH.D. Professor Department of Biomedical Engineering University of California-Davis Davis, California JAMES A. SORENSON, PH.D. Emeritus Professor of Medical Physics University of Wisconsin-Madison
Noncompartmental vs. Compartmental Approaches to Pharmacokinetic Data Analysis Paolo Vicini, Ph.D. Pfizer Global Research and Development David M.
 Noncompartmental vs. Compartmental Approaches to Pharmacokinetic Data Analysis Paolo Vicini, Ph.D. Pfizer Global Research and Development David M. Foster., Ph.D. University of Washington October 28, 2010
Noncompartmental vs. Compartmental Approaches to Pharmacokinetic Data Analysis Paolo Vicini, Ph.D. Pfizer Global Research and Development David M. Foster., Ph.D. University of Washington October 28, 2010
Radionuclide Imaging MII Positron Emission Tomography (PET)
 Radionuclide Imaging MII 3073 Positron Emission Tomography (PET) Positron (β + ) emission Positron is an electron with positive charge. Positron-emitting radionuclides are most commonly produced in cyclotron
Radionuclide Imaging MII 3073 Positron Emission Tomography (PET) Positron (β + ) emission Positron is an electron with positive charge. Positron-emitting radionuclides are most commonly produced in cyclotron
Confidence Estimation Methods for Neural Networks: A Practical Comparison
 , 6-8 000, Confidence Estimation Methods for : A Practical Comparison G. Papadopoulos, P.J. Edwards, A.F. Murray Department of Electronics and Electrical Engineering, University of Edinburgh Abstract.
, 6-8 000, Confidence Estimation Methods for : A Practical Comparison G. Papadopoulos, P.J. Edwards, A.F. Murray Department of Electronics and Electrical Engineering, University of Edinburgh Abstract.
Wavelet Footprints: Theory, Algorithms, and Applications
 1306 IEEE TRANSACTIONS ON SIGNAL PROCESSING, VOL. 51, NO. 5, MAY 2003 Wavelet Footprints: Theory, Algorithms, and Applications Pier Luigi Dragotti, Member, IEEE, and Martin Vetterli, Fellow, IEEE Abstract
1306 IEEE TRANSACTIONS ON SIGNAL PROCESSING, VOL. 51, NO. 5, MAY 2003 Wavelet Footprints: Theory, Algorithms, and Applications Pier Luigi Dragotti, Member, IEEE, and Martin Vetterli, Fellow, IEEE Abstract
Sparse Linear Models (10/7/13)
 STA56: Probabilistic machine learning Sparse Linear Models (0/7/) Lecturer: Barbara Engelhardt Scribes: Jiaji Huang, Xin Jiang, Albert Oh Sparsity Sparsity has been a hot topic in statistics and machine
STA56: Probabilistic machine learning Sparse Linear Models (0/7/) Lecturer: Barbara Engelhardt Scribes: Jiaji Huang, Xin Jiang, Albert Oh Sparsity Sparsity has been a hot topic in statistics and machine
ISyE 691 Data mining and analytics
 ISyE 691 Data mining and analytics Regression Instructor: Prof. Kaibo Liu Department of Industrial and Systems Engineering UW-Madison Email: kliu8@wisc.edu Office: Room 3017 (Mechanical Engineering Building)
ISyE 691 Data mining and analytics Regression Instructor: Prof. Kaibo Liu Department of Industrial and Systems Engineering UW-Madison Email: kliu8@wisc.edu Office: Room 3017 (Mechanical Engineering Building)
TWO METHODS FOR ESTIMATING OVERCOMPLETE INDEPENDENT COMPONENT BASES. Mika Inki and Aapo Hyvärinen
 TWO METHODS FOR ESTIMATING OVERCOMPLETE INDEPENDENT COMPONENT BASES Mika Inki and Aapo Hyvärinen Neural Networks Research Centre Helsinki University of Technology P.O. Box 54, FIN-215 HUT, Finland ABSTRACT
TWO METHODS FOR ESTIMATING OVERCOMPLETE INDEPENDENT COMPONENT BASES Mika Inki and Aapo Hyvärinen Neural Networks Research Centre Helsinki University of Technology P.O. Box 54, FIN-215 HUT, Finland ABSTRACT
Chapter 2 PET Imaging Basics
 Chapter 2 PET Imaging Basics Timothy G. Turkington PET Radiotracers Positron emission tomography (PET) imaging is the injection (or inhalation) of a substance containing a positron emitter, the subsequent
Chapter 2 PET Imaging Basics Timothy G. Turkington PET Radiotracers Positron emission tomography (PET) imaging is the injection (or inhalation) of a substance containing a positron emitter, the subsequent
Nuclear Medicine Intro & Physics from Medical Imaging Signals and Systems, Chapter 7, by Prince and Links
 Nuclear Medicine Intro & Physics from Medical Imaging Signals and Systems, Chapter 7, by Prince and Links NM - introduction Relies on EMISSION of photons from body (versus transmission of photons through
Nuclear Medicine Intro & Physics from Medical Imaging Signals and Systems, Chapter 7, by Prince and Links NM - introduction Relies on EMISSION of photons from body (versus transmission of photons through
Bioimage Informatics. Lecture 23, Spring Emerging Applications: Molecular Imaging
 Bioimage Informatics Lecture 23, Spring 2012 Emerging Applications: Molecular Imaging Lecture 23 April 25, 2012 1 Outline Overview of molecular imaging Molecular imaging modalities Molecular imaging applications
Bioimage Informatics Lecture 23, Spring 2012 Emerging Applications: Molecular Imaging Lecture 23 April 25, 2012 1 Outline Overview of molecular imaging Molecular imaging modalities Molecular imaging applications
Objective Functions for Tomographic Reconstruction from. Randoms-Precorrected PET Scans. gram separately, this process doubles the storage space for
 Objective Functions for Tomographic Reconstruction from Randoms-Precorrected PET Scans Mehmet Yavuz and Jerey A. Fessler Dept. of EECS, University of Michigan Abstract In PET, usually the data are precorrected
Objective Functions for Tomographic Reconstruction from Randoms-Precorrected PET Scans Mehmet Yavuz and Jerey A. Fessler Dept. of EECS, University of Michigan Abstract In PET, usually the data are precorrected
On the Projection Matrices Influence in the Classification of Compressed Sensed ECG Signals
 On the Projection Matrices Influence in the Classification of Compressed Sensed ECG Signals Monica Fira, Liviu Goras Institute of Computer Science Romanian Academy Iasi, Romania Liviu Goras, Nicolae Cleju,
On the Projection Matrices Influence in the Classification of Compressed Sensed ECG Signals Monica Fira, Liviu Goras Institute of Computer Science Romanian Academy Iasi, Romania Liviu Goras, Nicolae Cleju,
Outline Chapter 14 Nuclear Medicine
 Outline Chapter 14 uclear Medicine Radiation Dosimetry I Text: H.E Johns and J.R. Cunningham, The physics of radiology, 4 th ed. http://www.utoledo.edu/med/depts/radther Introduction Detectors for nuclear
Outline Chapter 14 uclear Medicine Radiation Dosimetry I Text: H.E Johns and J.R. Cunningham, The physics of radiology, 4 th ed. http://www.utoledo.edu/med/depts/radther Introduction Detectors for nuclear
Evaluation of Compartmental and Spectral Analysis Models of [ F]FDG Kinetics for Heart and Brain Studies with PET
![Evaluation of Compartmental and Spectral Analysis Models of [ F]FDG Kinetics for Heart and Brain Studies with PET Evaluation of Compartmental and Spectral Analysis Models of [ F]FDG Kinetics for Heart and Brain Studies with PET](/thumbs/83/88147426.jpg) IEEE TRANSACTIONS ON BIOMEDICAL ENGINEERING, VOL. 45, NO. 12, DECEMBER 1998 1429 Evaluation of Compartmental and Spectral Analysis Models of [ F]FDG Kinetics for Heart and Brain Studies with PET Alessandra
IEEE TRANSACTIONS ON BIOMEDICAL ENGINEERING, VOL. 45, NO. 12, DECEMBER 1998 1429 Evaluation of Compartmental and Spectral Analysis Models of [ F]FDG Kinetics for Heart and Brain Studies with PET Alessandra
c 2011 International Press Vol. 18, No. 1, pp , March DENNIS TREDE
 METHODS AND APPLICATIONS OF ANALYSIS. c 2011 International Press Vol. 18, No. 1, pp. 105 110, March 2011 007 EXACT SUPPORT RECOVERY FOR LINEAR INVERSE PROBLEMS WITH SPARSITY CONSTRAINTS DENNIS TREDE Abstract.
METHODS AND APPLICATIONS OF ANALYSIS. c 2011 International Press Vol. 18, No. 1, pp. 105 110, March 2011 007 EXACT SUPPORT RECOVERY FOR LINEAR INVERSE PROBLEMS WITH SPARSITY CONSTRAINTS DENNIS TREDE Abstract.
Lossless Compression of List-Mode 3D PET Data 1
 Lossless Compression of List-Mode 3D PET Data 1 Evren Asma, David W. Shattuck and Richard M. Leahy Signal and Image Processing Institute, University of Southern California, Los Angeles, CA 989-56 Abstract
Lossless Compression of List-Mode 3D PET Data 1 Evren Asma, David W. Shattuck and Richard M. Leahy Signal and Image Processing Institute, University of Southern California, Los Angeles, CA 989-56 Abstract
MA 575 Linear Models: Cedric E. Ginestet, Boston University Regularization: Ridge Regression and Lasso Week 14, Lecture 2
 MA 575 Linear Models: Cedric E. Ginestet, Boston University Regularization: Ridge Regression and Lasso Week 14, Lecture 2 1 Ridge Regression Ridge regression and the Lasso are two forms of regularized
MA 575 Linear Models: Cedric E. Ginestet, Boston University Regularization: Ridge Regression and Lasso Week 14, Lecture 2 1 Ridge Regression Ridge regression and the Lasso are two forms of regularized
HST 583 FUNCTIONAL MAGNETIC RESONANCE IMAGING DATA ANALYSIS AND ACQUISITION A REVIEW OF STATISTICS FOR FMRI DATA ANALYSIS
 HST 583 FUNCTIONAL MAGNETIC RESONANCE IMAGING DATA ANALYSIS AND ACQUISITION A REVIEW OF STATISTICS FOR FMRI DATA ANALYSIS EMERY N. BROWN AND CHRIS LONG NEUROSCIENCE STATISTICS RESEARCH LABORATORY DEPARTMENT
HST 583 FUNCTIONAL MAGNETIC RESONANCE IMAGING DATA ANALYSIS AND ACQUISITION A REVIEW OF STATISTICS FOR FMRI DATA ANALYSIS EMERY N. BROWN AND CHRIS LONG NEUROSCIENCE STATISTICS RESEARCH LABORATORY DEPARTMENT
Longitudinal growth analysis of early childhood brain using deformation based morphometry
 Longitudinal growth analysis of early childhood brain using deformation based morphometry Junki Lee 1, Yasser Ad-Dab'bagh 2, Vladimir Fonov 1, Alan C. Evans 1 and the Brain Development Cooperative Group
Longitudinal growth analysis of early childhood brain using deformation based morphometry Junki Lee 1, Yasser Ad-Dab'bagh 2, Vladimir Fonov 1, Alan C. Evans 1 and the Brain Development Cooperative Group
Compressed sensing. Or: the equation Ax = b, revisited. Terence Tao. Mahler Lecture Series. University of California, Los Angeles
 Or: the equation Ax = b, revisited University of California, Los Angeles Mahler Lecture Series Acquiring signals Many types of real-world signals (e.g. sound, images, video) can be viewed as an n-dimensional
Or: the equation Ax = b, revisited University of California, Los Angeles Mahler Lecture Series Acquiring signals Many types of real-world signals (e.g. sound, images, video) can be viewed as an n-dimensional
A. I, II, and III B. I C. I and II D. II and III E. I and III
 BioE 1330 - Review Chapters 7, 8, and 9 (Nuclear Medicine) 9/27/2018 Instructions: On the Answer Sheet, enter your 2-digit ID number (with a leading 0 if needed) in the boxes of the ID section. Fill in
BioE 1330 - Review Chapters 7, 8, and 9 (Nuclear Medicine) 9/27/2018 Instructions: On the Answer Sheet, enter your 2-digit ID number (with a leading 0 if needed) in the boxes of the ID section. Fill in
Recap from last time
 Recap from last time Nuclear Decay Occurs. when a nucleus is unstable (lower open energy levels) An unstable nucleus metamorphoses ( decays ) into a more stable (more tightly bound) nucleus Difference
Recap from last time Nuclear Decay Occurs. when a nucleus is unstable (lower open energy levels) An unstable nucleus metamorphoses ( decays ) into a more stable (more tightly bound) nucleus Difference
AN NONNEGATIVELY CONSTRAINED ITERATIVE METHOD FOR POSITRON EMISSION TOMOGRAPHY. Johnathan M. Bardsley
 Volume X, No. 0X, 0X, X XX Web site: http://www.aimsciences.org AN NONNEGATIVELY CONSTRAINED ITERATIVE METHOD FOR POSITRON EMISSION TOMOGRAPHY Johnathan M. Bardsley Department of Mathematical Sciences
Volume X, No. 0X, 0X, X XX Web site: http://www.aimsciences.org AN NONNEGATIVELY CONSTRAINED ITERATIVE METHOD FOR POSITRON EMISSION TOMOGRAPHY Johnathan M. Bardsley Department of Mathematical Sciences
 www.aask24.com www.aask24.com www.aask24.com P=Positron E= Emission T=Tomography Positron emission or beta plus decay (+ ) is a particular type of radioactive decay, in which a proton inside a radionuclide
www.aask24.com www.aask24.com www.aask24.com P=Positron E= Emission T=Tomography Positron emission or beta plus decay (+ ) is a particular type of radioactive decay, in which a proton inside a radionuclide
Sparse Approximation and Variable Selection
 Sparse Approximation and Variable Selection Lorenzo Rosasco 9.520 Class 07 February 26, 2007 About this class Goal To introduce the problem of variable selection, discuss its connection to sparse approximation
Sparse Approximation and Variable Selection Lorenzo Rosasco 9.520 Class 07 February 26, 2007 About this class Goal To introduce the problem of variable selection, discuss its connection to sparse approximation
First Technical Course, European Centre for Soft Computing, Mieres, Spain. 4th July 2011
 First Technical Course, European Centre for Soft Computing, Mieres, Spain. 4th July 2011 Linear Given probabilities p(a), p(b), and the joint probability p(a, B), we can write the conditional probabilities
First Technical Course, European Centre for Soft Computing, Mieres, Spain. 4th July 2011 Linear Given probabilities p(a), p(b), and the joint probability p(a, B), we can write the conditional probabilities
ON THE CONVERGENCE OF THE GENERALIZED LINEAR LEAST SQUARES ALGORITHM
 Convergence of GLLS ON THE CONVERGENCE OF THE GENERALIZED LINEAR LEAST SQUARES ALGORITHM C. NEGOITA and R. A. RENAUT Department of Mathematics and Statistics, Arizona State University, Tempe, AZ 85287-84,
Convergence of GLLS ON THE CONVERGENCE OF THE GENERALIZED LINEAR LEAST SQUARES ALGORITHM C. NEGOITA and R. A. RENAUT Department of Mathematics and Statistics, Arizona State University, Tempe, AZ 85287-84,
Multiple Change Point Detection by Sparse Parameter Estimation
 Multiple Change Point Detection by Sparse Parameter Estimation Department of Econometrics Fac. of Economics and Management University of Defence Brno, Czech Republic Dept. of Appl. Math. and Comp. Sci.
Multiple Change Point Detection by Sparse Parameter Estimation Department of Econometrics Fac. of Economics and Management University of Defence Brno, Czech Republic Dept. of Appl. Math. and Comp. Sci.
Sparse Kernel Density Estimation Technique Based on Zero-Norm Constraint
 Sparse Kernel Density Estimation Technique Based on Zero-Norm Constraint Xia Hong 1, Sheng Chen 2, Chris J. Harris 2 1 School of Systems Engineering University of Reading, Reading RG6 6AY, UK E-mail: x.hong@reading.ac.uk
Sparse Kernel Density Estimation Technique Based on Zero-Norm Constraint Xia Hong 1, Sheng Chen 2, Chris J. Harris 2 1 School of Systems Engineering University of Reading, Reading RG6 6AY, UK E-mail: x.hong@reading.ac.uk
Thresholds for the Recovery of Sparse Solutions via L1 Minimization
 Thresholds for the Recovery of Sparse Solutions via L Minimization David L. Donoho Department of Statistics Stanford University 39 Serra Mall, Sequoia Hall Stanford, CA 9435-465 Email: donoho@stanford.edu
Thresholds for the Recovery of Sparse Solutions via L Minimization David L. Donoho Department of Statistics Stanford University 39 Serra Mall, Sequoia Hall Stanford, CA 9435-465 Email: donoho@stanford.edu
Noncompartmental vs. Compartmental Approaches to Pharmacokinetic Data Analysis Paolo Vicini, Ph.D. Pfizer Global Research and Development David M.
 Noncompartmental vs. Compartmental Approaches to Pharmacokinetic Data Analysis Paolo Vicini, Ph.D. Pfizer Global Research and Development David M. Foster., Ph.D. University of Washington October 18, 2012
Noncompartmental vs. Compartmental Approaches to Pharmacokinetic Data Analysis Paolo Vicini, Ph.D. Pfizer Global Research and Development David M. Foster., Ph.D. University of Washington October 18, 2012
A New Approach of Weighted Integration Technique Based on Accumulated Images Using Dynamic PET and H 50
 Journal of Cerebral Blood Flow and Metabolism 11:492-501 1991 The International Society of Cerebral Blood Flow and Metabolism Published by Raven Press, Ltd., New York A New Approach of Weighted Integration
Journal of Cerebral Blood Flow and Metabolism 11:492-501 1991 The International Society of Cerebral Blood Flow and Metabolism Published by Raven Press, Ltd., New York A New Approach of Weighted Integration
ADME studies with radiolabeled compounds. Biomedical Applications of Radioisotopes and Pharmacokinetics Unit
 ADME studies with radiolabeled compounds Miguel Ángel Morcillo Alonso Biomedical Applications of Radioisotopes and Pharmacokinetics Unit CIEMAT (Centro de Investigaciones Energéticas, Medioambientales
ADME studies with radiolabeled compounds Miguel Ángel Morcillo Alonso Biomedical Applications of Radioisotopes and Pharmacokinetics Unit CIEMAT (Centro de Investigaciones Energéticas, Medioambientales
Estimation of Input Function and Kinetic Parameters Using Simulated Annealing: Application in a Flow Model
 IEEE TRANSACTIONS ON NUCLEAR SCIENCE, VOL. 49, NO. 3, JUNE 2002 707 Estimation of Input Function and Kinetic Parameters Using Simulated Annealing: Application in a Flow Model Koon-Pong Wong, Member, IEEE,
IEEE TRANSACTIONS ON NUCLEAR SCIENCE, VOL. 49, NO. 3, JUNE 2002 707 Estimation of Input Function and Kinetic Parameters Using Simulated Annealing: Application in a Flow Model Koon-Pong Wong, Member, IEEE,
CT-PET calibration : physical principles and operating procedures F.Bonutti. Faustino Bonutti Ph.D. Medical Physics, Udine University Hospital.
 CT-PET calibration : physical principles and operating procedures Faustino Bonutti Ph.D. Medical Physics, Udine University Hospital Topics Introduction to PET physics F-18 production β + decay and annichilation
CT-PET calibration : physical principles and operating procedures Faustino Bonutti Ph.D. Medical Physics, Udine University Hospital Topics Introduction to PET physics F-18 production β + decay and annichilation
Lossless Compression of Dynamic PET Data
 IEEE TRANSACTIONS ON NUCLEAR SCIENCE, VOL. 50, NO. 1, FEBRUARY 2003 9 Lossless Compression of Dynamic PET Data Evren Asma, Student Member, IEEE, David W. Shattuck, Member, IEEE, and Richard M. Leahy, Senior
IEEE TRANSACTIONS ON NUCLEAR SCIENCE, VOL. 50, NO. 1, FEBRUARY 2003 9 Lossless Compression of Dynamic PET Data Evren Asma, Student Member, IEEE, David W. Shattuck, Member, IEEE, and Richard M. Leahy, Senior
Inference and estimation in probabilistic time series models
 1 Inference and estimation in probabilistic time series models David Barber, A Taylan Cemgil and Silvia Chiappa 11 Time series The term time series refers to data that can be represented as a sequence
1 Inference and estimation in probabilistic time series models David Barber, A Taylan Cemgil and Silvia Chiappa 11 Time series The term time series refers to data that can be represented as a sequence
CHAPTER 11. A Revision. 1. The Computers and Numbers therein
 CHAPTER A Revision. The Computers and Numbers therein Traditional computer science begins with a finite alphabet. By stringing elements of the alphabet one after another, one obtains strings. A set of
CHAPTER A Revision. The Computers and Numbers therein Traditional computer science begins with a finite alphabet. By stringing elements of the alphabet one after another, one obtains strings. A set of
NIH Public Access Author Manuscript Med Image Comput Comput Assist Interv. Author manuscript; available in PMC 2014 May 19.
 NIH Public Access Author Manuscript Published in final edited form as: Med Image Comput Comput Assist Interv. 2009 ; 12(0 1): 919 926. Bias of Least Squares Approaches for Diffusion Tensor Estimation from
NIH Public Access Author Manuscript Published in final edited form as: Med Image Comput Comput Assist Interv. 2009 ; 12(0 1): 919 926. Bias of Least Squares Approaches for Diffusion Tensor Estimation from
ON THE CONVERGENCE OF THE GENERALIZED LINEAR LEAST SQUARES ALGORITHM
 Convergence of GLLS 1 ON THE CONVERGENCE OF THE GENERALIZED LINEAR LEAST SQUARES ALGORITHM C. NEGOITA 1 and R. A. RENAUT 2 1 Department of Mathematics, Oregon Institute of Technology, Klamath Falls, OR
Convergence of GLLS 1 ON THE CONVERGENCE OF THE GENERALIZED LINEAR LEAST SQUARES ALGORITHM C. NEGOITA 1 and R. A. RENAUT 2 1 Department of Mathematics, Oregon Institute of Technology, Klamath Falls, OR
SIMPLE MODEL Direct Binding Analysis
 Neurochemistry, 56:120:575 Dr. Patrick J. McIlroy Supplementary Notes SIMPLE MODEL Direct Binding Analysis The interaction of a (radio)ligand, L, with its receptor, R, to form a non-covalent complex, RL,
Neurochemistry, 56:120:575 Dr. Patrick J. McIlroy Supplementary Notes SIMPLE MODEL Direct Binding Analysis The interaction of a (radio)ligand, L, with its receptor, R, to form a non-covalent complex, RL,
Deep Learning: Approximation of Functions by Composition
 Deep Learning: Approximation of Functions by Composition Zuowei Shen Department of Mathematics National University of Singapore Outline 1 A brief introduction of approximation theory 2 Deep learning: approximation
Deep Learning: Approximation of Functions by Composition Zuowei Shen Department of Mathematics National University of Singapore Outline 1 A brief introduction of approximation theory 2 Deep learning: approximation
Sparse Time-Frequency Transforms and Applications.
 Sparse Time-Frequency Transforms and Applications. Bruno Torrésani http://www.cmi.univ-mrs.fr/~torresan LATP, Université de Provence, Marseille DAFx, Montreal, September 2006 B. Torrésani (LATP Marseille)
Sparse Time-Frequency Transforms and Applications. Bruno Torrésani http://www.cmi.univ-mrs.fr/~torresan LATP, Université de Provence, Marseille DAFx, Montreal, September 2006 B. Torrésani (LATP Marseille)
Statistical Geometry Processing Winter Semester 2011/2012
 Statistical Geometry Processing Winter Semester 2011/2012 Linear Algebra, Function Spaces & Inverse Problems Vector and Function Spaces 3 Vectors vectors are arrows in space classically: 2 or 3 dim. Euclidian
Statistical Geometry Processing Winter Semester 2011/2012 Linear Algebra, Function Spaces & Inverse Problems Vector and Function Spaces 3 Vectors vectors are arrows in space classically: 2 or 3 dim. Euclidian
Micro Computed Tomography Based Quantification of Pore Size in Electron Beam Melted Titanium Biomaterials
 Micro Computed Tomography Based Quantification of Pore Size in Electron Beam Melted Titanium Biomaterials SJ Tredinnick* JG Chase ** *NZi3 National ICT Innovation Institute, University of Canterbury, Christchurch,
Micro Computed Tomography Based Quantification of Pore Size in Electron Beam Melted Titanium Biomaterials SJ Tredinnick* JG Chase ** *NZi3 National ICT Innovation Institute, University of Canterbury, Christchurch,
MS-C1620 Statistical inference
 MS-C1620 Statistical inference 10 Linear regression III Joni Virta Department of Mathematics and Systems Analysis School of Science Aalto University Academic year 2018 2019 Period III - IV 1 / 32 Contents
MS-C1620 Statistical inference 10 Linear regression III Joni Virta Department of Mathematics and Systems Analysis School of Science Aalto University Academic year 2018 2019 Period III - IV 1 / 32 Contents
On Algorithms for Solving Least Squares Problems under an L 1 Penalty or an L 1 Constraint
 On Algorithms for Solving Least Squares Problems under an L 1 Penalty or an L 1 Constraint B.A. Turlach School of Mathematics and Statistics (M19) The University of Western Australia 35 Stirling Highway,
On Algorithms for Solving Least Squares Problems under an L 1 Penalty or an L 1 Constraint B.A. Turlach School of Mathematics and Statistics (M19) The University of Western Australia 35 Stirling Highway,
Sparse regression. Optimization-Based Data Analysis. Carlos Fernandez-Granda
 Sparse regression Optimization-Based Data Analysis http://www.cims.nyu.edu/~cfgranda/pages/obda_spring16 Carlos Fernandez-Granda 3/28/2016 Regression Least-squares regression Example: Global warming Logistic
Sparse regression Optimization-Based Data Analysis http://www.cims.nyu.edu/~cfgranda/pages/obda_spring16 Carlos Fernandez-Granda 3/28/2016 Regression Least-squares regression Example: Global warming Logistic
L-statistics based Modification of Reconstruction Algorithms for Compressive Sensing in the Presence of Impulse Noise
 L-statistics based Modification of Reconstruction Algorithms for Compressive Sensing in the Presence of Impulse Noise Srdjan Stanković, Irena Orović and Moeness Amin 1 Abstract- A modification of standard
L-statistics based Modification of Reconstruction Algorithms for Compressive Sensing in the Presence of Impulse Noise Srdjan Stanković, Irena Orović and Moeness Amin 1 Abstract- A modification of standard
Year 12 Notes Radioactivity 1/5
 Year Notes Radioactivity /5 Radioactivity Stable and Unstable Nuclei Radioactivity is the spontaneous disintegration of certain nuclei, a random process in which particles and/or high-energy photons are
Year Notes Radioactivity /5 Radioactivity Stable and Unstable Nuclei Radioactivity is the spontaneous disintegration of certain nuclei, a random process in which particles and/or high-energy photons are
MINIMUM PEAK IMPULSE FIR FILTER DESIGN
 MINIMUM PEAK IMPULSE FIR FILTER DESIGN CHRISTINE S. LAW AND JON DATTORRO Abstract. Saturation pulses rf(t) are essential to many imaging applications. Criteria for desirable saturation profile are flat
MINIMUM PEAK IMPULSE FIR FILTER DESIGN CHRISTINE S. LAW AND JON DATTORRO Abstract. Saturation pulses rf(t) are essential to many imaging applications. Criteria for desirable saturation profile are flat
Model Selection Tutorial 2: Problems With Using AIC to Select a Subset of Exposures in a Regression Model
 Model Selection Tutorial 2: Problems With Using AIC to Select a Subset of Exposures in a Regression Model Centre for Molecular, Environmental, Genetic & Analytic (MEGA) Epidemiology School of Population
Model Selection Tutorial 2: Problems With Using AIC to Select a Subset of Exposures in a Regression Model Centre for Molecular, Environmental, Genetic & Analytic (MEGA) Epidemiology School of Population
A Generalized Uncertainty Principle and Sparse Representation in Pairs of Bases
 2558 IEEE TRANSACTIONS ON INFORMATION THEORY, VOL 48, NO 9, SEPTEMBER 2002 A Generalized Uncertainty Principle Sparse Representation in Pairs of Bases Michael Elad Alfred M Bruckstein Abstract An elementary
2558 IEEE TRANSACTIONS ON INFORMATION THEORY, VOL 48, NO 9, SEPTEMBER 2002 A Generalized Uncertainty Principle Sparse Representation in Pairs of Bases Michael Elad Alfred M Bruckstein Abstract An elementary
Low Energy Medical Isotope Production. Naomi Ratcliffe IIAA, University of Huddersfield UK
 Low Energy Medical Isotope Production Naomi Ratcliffe naomi.ratcliffe@hud.ac.uk IIAA, University of Huddersfield UK Overview: Nuclear Medicine Cover the use of radioactive isotopes for diagnostic and therapy
Low Energy Medical Isotope Production Naomi Ratcliffe naomi.ratcliffe@hud.ac.uk IIAA, University of Huddersfield UK Overview: Nuclear Medicine Cover the use of radioactive isotopes for diagnostic and therapy
Cheng Soon Ong & Christian Walder. Canberra February June 2018
 Cheng Soon Ong & Christian Walder Research Group and College of Engineering and Computer Science Canberra February June 2018 (Many figures from C. M. Bishop, "Pattern Recognition and ") 1of 254 Part V
Cheng Soon Ong & Christian Walder Research Group and College of Engineering and Computer Science Canberra February June 2018 (Many figures from C. M. Bishop, "Pattern Recognition and ") 1of 254 Part V
Recent developments on sparse representation
 Recent developments on sparse representation Zeng Tieyong Department of Mathematics, Hong Kong Baptist University Email: zeng@hkbu.edu.hk Hong Kong Baptist University Dec. 8, 2008 First Previous Next Last
Recent developments on sparse representation Zeng Tieyong Department of Mathematics, Hong Kong Baptist University Email: zeng@hkbu.edu.hk Hong Kong Baptist University Dec. 8, 2008 First Previous Next Last
a Short Introduction
 Collaborative Filtering in Recommender Systems: a Short Introduction Norm Matloff Dept. of Computer Science University of California, Davis matloff@cs.ucdavis.edu December 3, 2016 Abstract There is a strong
Collaborative Filtering in Recommender Systems: a Short Introduction Norm Matloff Dept. of Computer Science University of California, Davis matloff@cs.ucdavis.edu December 3, 2016 Abstract There is a strong
One Picture and a Thousand Words Using Matrix Approximtions October 2017 Oak Ridge National Lab Dianne P. O Leary c 2017
 One Picture and a Thousand Words Using Matrix Approximtions October 2017 Oak Ridge National Lab Dianne P. O Leary c 2017 1 One Picture and a Thousand Words Using Matrix Approximations Dianne P. O Leary
One Picture and a Thousand Words Using Matrix Approximtions October 2017 Oak Ridge National Lab Dianne P. O Leary c 2017 1 One Picture and a Thousand Words Using Matrix Approximations Dianne P. O Leary
Functional Latent Feature Models. With Single-Index Interaction
 Generalized With Single-Index Interaction Department of Statistics Center for Statistical Bioinformatics Institute for Applied Mathematics and Computational Science Texas A&M University Naisyin Wang and
Generalized With Single-Index Interaction Department of Statistics Center for Statistical Bioinformatics Institute for Applied Mathematics and Computational Science Texas A&M University Naisyin Wang and
The General Linear Model (GLM)
 he General Linear Model (GLM) Klaas Enno Stephan ranslational Neuromodeling Unit (NU) Institute for Biomedical Engineering University of Zurich & EH Zurich Wellcome rust Centre for Neuroimaging Institute
he General Linear Model (GLM) Klaas Enno Stephan ranslational Neuromodeling Unit (NU) Institute for Biomedical Engineering University of Zurich & EH Zurich Wellcome rust Centre for Neuroimaging Institute
The ASL signal. Parenchy mal signal. Venous signal. Arterial signal. Input Function (Label) Dispersion: (t e -kt ) Relaxation: (e -t/t1a )
 Lecture Goals Other non-bold techniques (T2 weighted, Mn contrast agents, SSFP, Dynamic Diffusion, ASL) Understand Basic Principles in Spin labeling : spin inversion, flow vs. perfusion ASL variations
Lecture Goals Other non-bold techniques (T2 weighted, Mn contrast agents, SSFP, Dynamic Diffusion, ASL) Understand Basic Principles in Spin labeling : spin inversion, flow vs. perfusion ASL variations
Quantitative Imaging with Y-90 Bremsstrahlung SPECT (Single Positron Emission Computed Tomography)
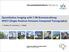 Quantitative Imaging with Y-90 Bremsstrahlung SPECT (Single Positron Emission Computed Tomography) S. Beykan, M. Lassmann, S. Schlögl Klinik und Poliklinik für Nuklearmedizin Direktor: Prof. Dr. A. Buck
Quantitative Imaging with Y-90 Bremsstrahlung SPECT (Single Positron Emission Computed Tomography) S. Beykan, M. Lassmann, S. Schlögl Klinik und Poliklinik für Nuklearmedizin Direktor: Prof. Dr. A. Buck
Compressed sensing for radio interferometry: spread spectrum imaging techniques
 Compressed sensing for radio interferometry: spread spectrum imaging techniques Y. Wiaux a,b, G. Puy a, Y. Boursier a and P. Vandergheynst a a Institute of Electrical Engineering, Ecole Polytechnique Fédérale
Compressed sensing for radio interferometry: spread spectrum imaging techniques Y. Wiaux a,b, G. Puy a, Y. Boursier a and P. Vandergheynst a a Institute of Electrical Engineering, Ecole Polytechnique Fédérale
Parametric [ 11 C]flumazenil images. Ursula MH Klumpers, Ronald Boellaard, Dick J Veltman, Reina W Kloet, Witte JG Hoogendijk, Adriaan A Lammertsma
![Parametric [ 11 C]flumazenil images. Ursula MH Klumpers, Ronald Boellaard, Dick J Veltman, Reina W Kloet, Witte JG Hoogendijk, Adriaan A Lammertsma Parametric [ 11 C]flumazenil images. Ursula MH Klumpers, Ronald Boellaard, Dick J Veltman, Reina W Kloet, Witte JG Hoogendijk, Adriaan A Lammertsma](/thumbs/81/84602235.jpg) 3 Parametric [ 11 C]flumazenil images Ursula MH Klumpers, Ronald Boellaard, Dick J Veltman, Reina W Kloet, Witte JG Hoogendijk, Adriaan A Lammertsma 50 Chapter 3 ABSTRACT Objective: This [ 11 C]flumazenil
3 Parametric [ 11 C]flumazenil images Ursula MH Klumpers, Ronald Boellaard, Dick J Veltman, Reina W Kloet, Witte JG Hoogendijk, Adriaan A Lammertsma 50 Chapter 3 ABSTRACT Objective: This [ 11 C]flumazenil
Radioisotopes in action. Diagnostic application of radioisotopes. Steps of diagnostic procedure. Information from various medical imaging techniques
 Radioisotopes in action Diagnostic application of radioisotopes Steps of diagnostic procedure - Radioactive material introduced into the patient - Distribution and alteration of activity is detected -
Radioisotopes in action Diagnostic application of radioisotopes Steps of diagnostic procedure - Radioactive material introduced into the patient - Distribution and alteration of activity is detected -
Mathematical Segmentation of Grey Matter, White Matter
 Tina Memo No. 2000-006 Short Version published in: British Journal of Radiology, 74, 234-242, 2001. Mathematical Segmentation of Grey Matter, White Matter and Cerebral Spinal Fluid from MR image Pairs.
Tina Memo No. 2000-006 Short Version published in: British Journal of Radiology, 74, 234-242, 2001. Mathematical Segmentation of Grey Matter, White Matter and Cerebral Spinal Fluid from MR image Pairs.
Dictionary Learning for L1-Exact Sparse Coding
 Dictionary Learning for L1-Exact Sparse Coding Mar D. Plumbley Department of Electronic Engineering, Queen Mary University of London, Mile End Road, London E1 4NS, United Kingdom. Email: mar.plumbley@elec.qmul.ac.u
Dictionary Learning for L1-Exact Sparse Coding Mar D. Plumbley Department of Electronic Engineering, Queen Mary University of London, Mile End Road, London E1 4NS, United Kingdom. Email: mar.plumbley@elec.qmul.ac.u
A Modern Look at Classical Multivariate Techniques
 A Modern Look at Classical Multivariate Techniques Yoonkyung Lee Department of Statistics The Ohio State University March 16-20, 2015 The 13th School of Probability and Statistics CIMAT, Guanajuato, Mexico
A Modern Look at Classical Multivariate Techniques Yoonkyung Lee Department of Statistics The Ohio State University March 16-20, 2015 The 13th School of Probability and Statistics CIMAT, Guanajuato, Mexico
Dynamic Contrast Enhance (DCE)-MRI
 Dynamic Contrast Enhance (DCE)-MRI contrast enhancement in ASL: labeling of blood (endogenous) for this technique: usage of a exogenous contras agent typically based on gadolinium molecules packed inside
Dynamic Contrast Enhance (DCE)-MRI contrast enhancement in ASL: labeling of blood (endogenous) for this technique: usage of a exogenous contras agent typically based on gadolinium molecules packed inside
Positron Emission Tomography
 Positron Emission Tomography Presenter: Difei Wang June,2018 Universität Bonn Contents 2 / 24 1 2 3 4 Positron emission Detected events Detectors and configuration Data acquisition Positron emission Positron
Positron Emission Tomography Presenter: Difei Wang June,2018 Universität Bonn Contents 2 / 24 1 2 3 4 Positron emission Detected events Detectors and configuration Data acquisition Positron emission Positron
Mayneord-Phillips Summer School St Edmund Hall, University of Oxford July Proton decays to n, e +, ν
 Positron Emission Tomography Physics & Instrumentation Dimitra G. Darambara, Ph.D Multimodality Molecular Imaging Joint Department of Physics RMH/ICR Outline Introduction PET Physics overview Types of
Positron Emission Tomography Physics & Instrumentation Dimitra G. Darambara, Ph.D Multimodality Molecular Imaging Joint Department of Physics RMH/ICR Outline Introduction PET Physics overview Types of
BLIND SOURCE SEPARATION TECHNIQUES ANOTHER WAY OF DOING OPERATIONAL MODAL ANALYSIS
 BLIND SOURCE SEPARATION TECHNIQUES ANOTHER WAY OF DOING OPERATIONAL MODAL ANALYSIS F. Poncelet, Aerospace and Mech. Eng. Dept., University of Liege, Belgium G. Kerschen, Aerospace and Mech. Eng. Dept.,
BLIND SOURCE SEPARATION TECHNIQUES ANOTHER WAY OF DOING OPERATIONAL MODAL ANALYSIS F. Poncelet, Aerospace and Mech. Eng. Dept., University of Liege, Belgium G. Kerschen, Aerospace and Mech. Eng. Dept.,
Machine Learning - MT & 5. Basis Expansion, Regularization, Validation
 Machine Learning - MT 2016 4 & 5. Basis Expansion, Regularization, Validation Varun Kanade University of Oxford October 19 & 24, 2016 Outline Basis function expansion to capture non-linear relationships
Machine Learning - MT 2016 4 & 5. Basis Expansion, Regularization, Validation Varun Kanade University of Oxford October 19 & 24, 2016 Outline Basis function expansion to capture non-linear relationships
WAVELET RECONSTRUCTION OF NONLINEAR DYNAMICS
 International Journal of Bifurcation and Chaos, Vol. 8, No. 11 (1998) 2191 2201 c World Scientific Publishing Company WAVELET RECONSTRUCTION OF NONLINEAR DYNAMICS DAVID ALLINGHAM, MATTHEW WEST and ALISTAIR
International Journal of Bifurcation and Chaos, Vol. 8, No. 11 (1998) 2191 2201 c World Scientific Publishing Company WAVELET RECONSTRUCTION OF NONLINEAR DYNAMICS DAVID ALLINGHAM, MATTHEW WEST and ALISTAIR
CONVOLUTIVE NON-NEGATIVE MATRIX FACTORISATION WITH SPARSENESS CONSTRAINT
 CONOLUTIE NON-NEGATIE MATRIX FACTORISATION WITH SPARSENESS CONSTRAINT Paul D. O Grady Barak A. Pearlmutter Hamilton Institute National University of Ireland, Maynooth Co. Kildare, Ireland. ABSTRACT Discovering
CONOLUTIE NON-NEGATIE MATRIX FACTORISATION WITH SPARSENESS CONSTRAINT Paul D. O Grady Barak A. Pearlmutter Hamilton Institute National University of Ireland, Maynooth Co. Kildare, Ireland. ABSTRACT Discovering
