CME 300 Properties of Materials. ANSWERS: Homework 4 October 12, 2011
|
|
|
- Brian Johns
- 6 years ago
- Views:
Transcription
1 CME 300 Properties of Materials ANSWERS: Homework 4 October 12, ) The Scherrer equation can be obtained by taking the derivative of the Bragg Equation. Perform this derivative. Begin by multiplying λ and d by an integer m such that md = t, the Scherrer Size. Differentiate keeping in mind that mλ is a constant. The smallest increment in t is d, so dt is equal to d. You must reuse Bragg s law to simplify your final expression to obtain the Scherrer Equation using dθ = B. mλ = 2t sinθ 0 = 2sinθdt + 2t cosθdθ 0 = 2d sinθ + 2t cosθb 0 = λ + t cosθb t = λ Bcosθ ~ 0.9λ Bcosθ 2) Debye-Waller thermal broadening of diffraction peaks is modeled with a Gaussian function, that predicts a diffracted peak shape of I(q) = exp q2 u 2 3 where q is the scattering vector q = 2π = 4π d Bragg λ sin( θ ) and u2 is the mean square free path for the atoms about the lattice site. Compare the Debye-Waller function with the Gaussian Distribution function and give a value for the standard deviation about the peak (peak width) due to thermal broadening. Do you think it is logical to use the Gaussian Distribution to describe a diffraction peak (explain your answer, you need to explain what a Gaussian Distribution is normally used to describe). ( )2 The Gaussian distribution is P(x) = P(0)exp x x 2σ 2 and describes a random distribution about a mean value <x> with the standard deviation of σ. This is the bell shaped curve for grade distribution; however, it is strictly inappropriate when there is a minimum or a maximum value (0 and 100 for grades). (A log- normal could be appropriate when there is a minimum value.) For the Debye- Waller Factor the mean is the atomic lattice position taken to be 0 and the angle converted to inverse distance or q replaces x in 3 the Gaussian distribution so the variance is σ 2 =. The Debye- Waller Factor is used as 2 u 2 a prefactor to the intensity for a diffraction peak. The larger the atomic motion, the larger u 2 and the smaller the peak intensity. If the diffraction peak were originally a Gaussian in ( ) 2 q, I( q) = I( 0)exp q q ( hkl ), the Debye- Waller Factor is centered on q = 0 not at q(hkl) so 2σ ( hkl ) mostly effects the intensity of the peak, not the shape. 1
2 3) Why can t diffraction be used to make a picture of a crystal lattice? (You answer should consider how a crystal would appear in inverse space). Diffraction gives statistical information about the crystalline structure. An image has details about a specific small group of atoms. In inverse space, lattice points are given by Miller indices for planes. Each point includes all of the planes of a given type. So the lattice is not a picture of a few atoms in their lattice positions but an average of all of the Miller indices in the entire crystal. The spatial connection between the Miller indices, that is their arrangement in 3d space is lost in inverse space. So we know that certain repeat distances occur but we do not have the ability to reconstruct the 3d image directly from the diffraction pattern. This is called the loss of phase information. 4) In class we discussed the phase diagram for two polymers that display partial miscibility. Sketch a plot of the free energy change on mixing versus composition at a temperature where there is a miscibility gap. Show in this plot the origin of the spinodal and binodal regimes. Sketch the morphology that result from spinodal and binodal decomposition. Show the spinodal and binodal curves in a UCST system and indicate the points that are determined from your free energy versus composition plot. The binodal line is associated with the circles and spinodal line with the squares. 2
3 Spinodal Binodal (Nucleation and Growth) χab (1/T) Problems from Hummel (Chapter 5) Amount of liquid = (34-28)/(34-23)=0.545; Amount of solid = (28-23)/(34-23)=
4 799 C α+l=>β; 586 C β => α+γ; 640 C γ+ε=>ξ; 640 C ξ=> ε+l; 526 C γ=>α+δ; 350 C δ=>α+ε; 415 C ε+l=>η; There are others but it gets confusing at the bottom right. 8.8% copper in silver and 8% silver in copper. Both occur at the eutectic temperature, 780 C. 4
5 At 25 C there is almost no solubility of copper in silver. At 400 C and 5% Cu a mixture of α and θ phases are present. There will be about (5-3)/(52-3)= 0.04 fraction of the θ phase and 96% of the α phase. This occurs at about 820 C. 5
6 No it does not exist since both the solidus and liquidus slope upwards in the phase diagram so when the nickel content in the solid increases the nickel content in the liquid will also increase. a) Eutectic temperature 780 C b) 28.1% Cu (Eutectic composition) c) about 812 C d) α phase about 5% Cu. 6
7 Problems from Callister p. 304 The chemical potential of two phases in equilibrium are the same. The chemical potential is the first derivative of free energy with respect to composition. a) ε+η C ε =87% Zn C η =97% Zn c) Liquid C L = 55% Ag e) β+γ C β =49% Zn C γ =58%Zn g) α C α = 63.8% Νi Not possible. At 8% Cu the β phase does not exist. 7
3.012 Fund of Mat Sci: Structure Lecture 18
 3.012 Fund of Mat Sci: Structure Lecture 18 X-RAYS AT WORK An X-ray diffraction image for the protein myoglobin. Source: Wikipedia. Model of helical domains in myoglobin. Image courtesy of Magnus Manske
3.012 Fund of Mat Sci: Structure Lecture 18 X-RAYS AT WORK An X-ray diffraction image for the protein myoglobin. Source: Wikipedia. Model of helical domains in myoglobin. Image courtesy of Magnus Manske
Introduction to crystallography The unitcell The resiprocal space and unitcell Braggs law Structure factor F hkl and atomic scattering factor f zθ
 Introduction to crystallography The unitcell The resiprocal space and unitcell Braggs law Structure factor F hkl and atomic scattering factor f zθ Introduction to crystallography We divide materials into
Introduction to crystallography The unitcell The resiprocal space and unitcell Braggs law Structure factor F hkl and atomic scattering factor f zθ Introduction to crystallography We divide materials into
FROM DIFFRACTION TO STRUCTURE
 3.012 Fund of Mat Sci: Structure Lecture 19 FROM DIFFRACTION TO STRUCTURE Images removed for copyright reasons. 3-fold symmetry in silicon along the [111] direction. Forward (left) and backward (right)
3.012 Fund of Mat Sci: Structure Lecture 19 FROM DIFFRACTION TO STRUCTURE Images removed for copyright reasons. 3-fold symmetry in silicon along the [111] direction. Forward (left) and backward (right)
Chapter 4 Polymer solutions
 Chapter 4 Polymer solutions 4.1 Introduction Solution: any phase containing more than one component.(gas, liquid or solid) Polymer solution is important: Classical analyses of polymers are conducted on
Chapter 4 Polymer solutions 4.1 Introduction Solution: any phase containing more than one component.(gas, liquid or solid) Polymer solution is important: Classical analyses of polymers are conducted on
X-ray, Neutron and e-beam scattering
 X-ray, Neutron and e-beam scattering Introduction Why scattering? Diffraction basics Neutrons and x-rays Techniques Direct and reciprocal space Single crystals Powders CaFe 2 As 2 an example What is the
X-ray, Neutron and e-beam scattering Introduction Why scattering? Diffraction basics Neutrons and x-rays Techniques Direct and reciprocal space Single crystals Powders CaFe 2 As 2 an example What is the
[ ( )] + exp 2α / ( k B T) [ ] + exp 3α / k B T
![[ ( )] + exp 2α / ( k B T) [ ] + exp 3α / k B T [ ( )] + exp 2α / ( k B T) [ ] + exp 3α / k B T](/thumbs/81/84416061.jpg) hermal Physics Homework #10 (optional) (19 points) 1 Your work on this homework set will not be accepted after 3 p.m. on December 4 (W) 1. (3 points) A common mistake people make when they try to calculate
hermal Physics Homework #10 (optional) (19 points) 1 Your work on this homework set will not be accepted after 3 p.m. on December 4 (W) 1. (3 points) A common mistake people make when they try to calculate
3.012 PS 6 THERMODYANMICS SOLUTIONS Issued: Fall 2005 Due:
 3.012 PS 6 THERMODYANMICS SOLUTIONS 3.012 Issued: 11.28.05 Fall 2005 Due: THERMODYNAMICS 1. Building a binary phase diagram. Given below are data for a binary system of two materials A and B. The two components
3.012 PS 6 THERMODYANMICS SOLUTIONS 3.012 Issued: 11.28.05 Fall 2005 Due: THERMODYNAMICS 1. Building a binary phase diagram. Given below are data for a binary system of two materials A and B. The two components
SOLID STATE 18. Reciprocal Space
 SOLID STATE 8 Reciprocal Space Wave vectors and the concept of K-space can simplify the explanation of several properties of the solid state. They will be introduced to provide more information on diffraction
SOLID STATE 8 Reciprocal Space Wave vectors and the concept of K-space can simplify the explanation of several properties of the solid state. They will be introduced to provide more information on diffraction
The structure of liquids and glasses. The lattice and unit cell in 1D. The structure of crystalline materials. Describing condensed phase structures
 Describing condensed phase structures Describing the structure of an isolated small molecule is easy to do Just specify the bond distances and angles How do we describe the structure of a condensed phase?
Describing condensed phase structures Describing the structure of an isolated small molecule is easy to do Just specify the bond distances and angles How do we describe the structure of a condensed phase?
Resolution: maximum limit of diffraction (asymmetric)
 Resolution: maximum limit of diffraction (asymmetric) crystal Y X-ray source 2θ X direct beam tan 2θ = Y X d = resolution 2d sinθ = λ detector 1 Unit Cell: two vectors in plane of image c* Observe: b*
Resolution: maximum limit of diffraction (asymmetric) crystal Y X-ray source 2θ X direct beam tan 2θ = Y X d = resolution 2d sinθ = λ detector 1 Unit Cell: two vectors in plane of image c* Observe: b*
Chapter 2. X-ray X. Diffraction and Reciprocal Lattice. Scattering from Lattices
 Chapter. X-ray X Diffraction and Reciprocal Lattice Diffraction of waves by crystals Reciprocal Lattice Diffraction of X-rays Powder diffraction Single crystal X-ray diffraction Scattering from Lattices
Chapter. X-ray X Diffraction and Reciprocal Lattice Diffraction of waves by crystals Reciprocal Lattice Diffraction of X-rays Powder diffraction Single crystal X-ray diffraction Scattering from Lattices
X-ray diffraction geometry
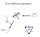 X-ray diffraction geometry Setting controls sample orientation in the diffraction plane. most important for single-crystal diffraction For any poly- (or nano-) crystalline specimen, we usually set: 1 X-ray
X-ray diffraction geometry Setting controls sample orientation in the diffraction plane. most important for single-crystal diffraction For any poly- (or nano-) crystalline specimen, we usually set: 1 X-ray
... 3, , = a (1) 3 3 a 2 = a (2) The reciprocal lattice vectors are defined by the condition a b = 2πδ ij, which gives
 PHZ646: Fall 013 Problem set # 4: Crystal Structure due Monday, 10/14 at the time of the class Instructor: D. L. Maslov maslov@phys.ufl.edu 39-0513 Rm. 114 Office hours: TR 3 pm-4 pm Please help your instructor
PHZ646: Fall 013 Problem set # 4: Crystal Structure due Monday, 10/14 at the time of the class Instructor: D. L. Maslov maslov@phys.ufl.edu 39-0513 Rm. 114 Office hours: TR 3 pm-4 pm Please help your instructor
de Broglie Waves h p de Broglie argued Light exhibits both wave and particle properties
 de Broglie argued de Broglie Waves Light exhibits both wave and particle properties Wave interference, diffraction Particle photoelectric effect, Compton effect Then matter (particles) should exhibit both
de Broglie argued de Broglie Waves Light exhibits both wave and particle properties Wave interference, diffraction Particle photoelectric effect, Compton effect Then matter (particles) should exhibit both
General theory of diffraction
 General theory of diffraction X-rays scatter off the charge density (r), neutrons scatter off the spin density. Coherent scattering (diffraction) creates the Fourier transform of (r) from real to reciprocal
General theory of diffraction X-rays scatter off the charge density (r), neutrons scatter off the spin density. Coherent scattering (diffraction) creates the Fourier transform of (r) from real to reciprocal
Small Angle X-ray Scattering (SAXS)
 Small Angle X-ray Scattering (SAXS) We have considered that Bragg's Law, d = λ/(2 sinθ), supports a minimum size of measurement of λ/2 in a diffraction experiment (limiting sphere of inverse space) but
Small Angle X-ray Scattering (SAXS) We have considered that Bragg's Law, d = λ/(2 sinθ), supports a minimum size of measurement of λ/2 in a diffraction experiment (limiting sphere of inverse space) but
Atomic and Nuclear Physics
 Atomic and Nuclear Physics Introductory experiments ualism of wave and particle L Physics Leaflets P6.1.5.1 iffraction of electrons in a polycrystalline lattice (ebye-scherrer diffraction) Objects of the
Atomic and Nuclear Physics Introductory experiments ualism of wave and particle L Physics Leaflets P6.1.5.1 iffraction of electrons in a polycrystalline lattice (ebye-scherrer diffraction) Objects of the
X-ray analysis. 1. Basic crystallography 2. Basic diffraction physics 3. Experimental methods
 X-ray analysis 1. Basic crystallography 2. Basic diffraction physics 3. Experimental methods Introduction Noble prizes associated with X-ray diffraction 1901 W. C. Roentgen (Physics) for the discovery
X-ray analysis 1. Basic crystallography 2. Basic diffraction physics 3. Experimental methods Introduction Noble prizes associated with X-ray diffraction 1901 W. C. Roentgen (Physics) for the discovery
Module 16. Diffusion in solids II. Lecture 16. Diffusion in solids II
 Module 16 Diffusion in solids II Lecture 16 Diffusion in solids II 1 NPTEL Phase II : IIT Kharagpur : Prof. R. N. Ghosh, Dept of Metallurgical and Materials Engineering Keywords: Micro mechanisms of diffusion,
Module 16 Diffusion in solids II Lecture 16 Diffusion in solids II 1 NPTEL Phase II : IIT Kharagpur : Prof. R. N. Ghosh, Dept of Metallurgical and Materials Engineering Keywords: Micro mechanisms of diffusion,
3.012 Structure An Introduction to X-ray Diffraction
 3.012 Structure An Introduction to X-ray Diffraction This handout summarizes some topics that are important for understanding x-ray diffraction. The following references provide a thorough explanation
3.012 Structure An Introduction to X-ray Diffraction This handout summarizes some topics that are important for understanding x-ray diffraction. The following references provide a thorough explanation
University of Ljubljana. Faculty of Mathematics and Physics. Department of Physics. High-entropy alloys. Author: Darja Gačnik
 University of Ljubljana Faculty of Mathematics and Physics Department of Physics Seminar I 1 st year of Master study programme High-entropy alloys Author: Darja Gačnik Mentor: prof. Janez Dolinšek Ljubljana,
University of Ljubljana Faculty of Mathematics and Physics Department of Physics Seminar I 1 st year of Master study programme High-entropy alloys Author: Darja Gačnik Mentor: prof. Janez Dolinšek Ljubljana,
Scattering and Diffraction
 Scattering and Diffraction Andreas Kreyssig, Alan Goldman, Rob McQueeney Ames Laboratory Iowa State University All rights reserved, 2018. Atomic scale structure - crystals Crystalline materials... atoms
Scattering and Diffraction Andreas Kreyssig, Alan Goldman, Rob McQueeney Ames Laboratory Iowa State University All rights reserved, 2018. Atomic scale structure - crystals Crystalline materials... atoms
V 11: Electron Diffraction
 Martin-Luther-University Halle-Wittenberg Institute of Physics Advanced Practical Lab Course V 11: Electron Diffraction An electron beam conditioned by an electron optical system is diffracted by a polycrystalline,
Martin-Luther-University Halle-Wittenberg Institute of Physics Advanced Practical Lab Course V 11: Electron Diffraction An electron beam conditioned by an electron optical system is diffracted by a polycrystalline,
Physical Chemistry I. Crystal Structure
 Physical Chemistry I Crystal Structure Crystal Structure Introduction Crystal Lattice Bravis Lattices Crytal Planes, Miller indices Distances between planes Diffraction patters Bragg s law X-ray radiation
Physical Chemistry I Crystal Structure Crystal Structure Introduction Crystal Lattice Bravis Lattices Crytal Planes, Miller indices Distances between planes Diffraction patters Bragg s law X-ray radiation
Setting The motor that rotates the sample about an axis normal to the diffraction plane is called (or ).
 X-Ray Diffraction X-ray diffraction geometry A simple X-ray diffraction (XRD) experiment might be set up as shown below. We need a parallel X-ray source, which is usually an X-ray tube in a fixed position
X-Ray Diffraction X-ray diffraction geometry A simple X-ray diffraction (XRD) experiment might be set up as shown below. We need a parallel X-ray source, which is usually an X-ray tube in a fixed position
Supplementary Information for Observation of dynamic atom-atom correlation in liquid helium in real space
 3 4 5 6 7 8 9 0 3 4 5 6 7 8 9 0 Supplementary Information for Observation of dynamic atom-atom correlation in liquid helium in real space Supplementary Note : Total PDF The total (snap-shot) PDF is obtained
3 4 5 6 7 8 9 0 3 4 5 6 7 8 9 0 Supplementary Information for Observation of dynamic atom-atom correlation in liquid helium in real space Supplementary Note : Total PDF The total (snap-shot) PDF is obtained
ENAS 606 : Polymer Physics
 ENAS 606 : Polymer Physics Professor Description Course Topics TA Prerequisite Class Office Hours Chinedum Osuji 302 Mason Lab, 432-4357, chinedum.osuji@yale.edu This course covers the static and dynamic
ENAS 606 : Polymer Physics Professor Description Course Topics TA Prerequisite Class Office Hours Chinedum Osuji 302 Mason Lab, 432-4357, chinedum.osuji@yale.edu This course covers the static and dynamic
3.012 Quiz Fall points total (50 in thermo + 50 in bonding)
 3.012 Quiz 3 3.012 12.20.05 Fall 2005 100 points total (50 in thermo + 50 in bonding) Give as much written explanation as possible of your reasoning, and write clearly and legibly. There is plenty of space
3.012 Quiz 3 3.012 12.20.05 Fall 2005 100 points total (50 in thermo + 50 in bonding) Give as much written explanation as possible of your reasoning, and write clearly and legibly. There is plenty of space
Scattering and Diffraction
 Scattering and Diffraction Adventures in k-space, part 1 Lenson Pellouchoud SLAC / SSL XSD summer school 7/16/018 lenson@slac.stanford.edu Outline Elastic Scattering eview / overview and terminology Form
Scattering and Diffraction Adventures in k-space, part 1 Lenson Pellouchoud SLAC / SSL XSD summer school 7/16/018 lenson@slac.stanford.edu Outline Elastic Scattering eview / overview and terminology Form
SOLID STATE 9. Determination of Crystal Structures
 SOLID STATE 9 Determination of Crystal Structures In the diffraction experiment, we measure intensities as a function of d hkl. Intensities are the sum of the x-rays scattered by all the atoms in a crystal.
SOLID STATE 9 Determination of Crystal Structures In the diffraction experiment, we measure intensities as a function of d hkl. Intensities are the sum of the x-rays scattered by all the atoms in a crystal.
Structural Characterization of Nanoparticles
 Structural Characterization of Nanoparticles Nicola Pinna Max Planck Institute of Colloids and Interfaces e-mail: pinna@mpikg-golm.mpg.de - http://www.pinna.cx Plan 1. Transmission Electron Microscopy
Structural Characterization of Nanoparticles Nicola Pinna Max Planck Institute of Colloids and Interfaces e-mail: pinna@mpikg-golm.mpg.de - http://www.pinna.cx Plan 1. Transmission Electron Microscopy
Handout 7 Reciprocal Space
 Handout 7 Reciprocal Space Useful concepts for the analysis of diffraction data http://homepages.utoledo.edu/clind/ Concepts versus reality Reflection from lattice planes is just a concept that helps us
Handout 7 Reciprocal Space Useful concepts for the analysis of diffraction data http://homepages.utoledo.edu/clind/ Concepts versus reality Reflection from lattice planes is just a concept that helps us
Thermodynamic of polymer blends Assoc.Prof.Dr. Jatyuphorn Wootthikanokkhan
 Thermodynamic of polymer blends Assoc.Prof.Dr. Jatyuphorn Wootthikanokkhan Division of Materials Technology, School of Energy, Environment and Materials, KMUTT, Thailand Classification of polymer blends
Thermodynamic of polymer blends Assoc.Prof.Dr. Jatyuphorn Wootthikanokkhan Division of Materials Technology, School of Energy, Environment and Materials, KMUTT, Thailand Classification of polymer blends
Errata for SOLIDIFICATION (First Edition, 2009)
 Errata for SOLIDIFICATION (First Edition, 29) J. A. Dantzig and M. Rappaz March 2, 217 Chapter 1: Overview Page 2, first line: the definition of r should have a square root. i.e., r = (ξ 2 + y 2 + z 2
Errata for SOLIDIFICATION (First Edition, 29) J. A. Dantzig and M. Rappaz March 2, 217 Chapter 1: Overview Page 2, first line: the definition of r should have a square root. i.e., r = (ξ 2 + y 2 + z 2
Scattering by two Electrons
 Scattering by two Electrons p = -r k in k in p r e 2 q k in /λ θ θ k out /λ S q = r k out p + q = r (k out - k in ) e 1 Phase difference of wave 2 with respect to wave 1: 2π λ (k out - k in ) r= 2π S r
Scattering by two Electrons p = -r k in k in p r e 2 q k in /λ θ θ k out /λ S q = r k out p + q = r (k out - k in ) e 1 Phase difference of wave 2 with respect to wave 1: 2π λ (k out - k in ) r= 2π S r
Crystal planes. Neutrons: magnetic moment - interacts with magnetic materials or nuclei of non-magnetic materials. (in Å)
 Crystallography: neutron, electron, and X-ray scattering from periodic lattice, scattering of waves by periodic structures, Miller indices, reciprocal space, Ewald construction. Diffraction: Specular,
Crystallography: neutron, electron, and X-ray scattering from periodic lattice, scattering of waves by periodic structures, Miller indices, reciprocal space, Ewald construction. Diffraction: Specular,
print first name print last name print student id grade
 print first name print last name print student id grade Experiment 2 X-ray fluorescence X-ray fluorescence (XRF) and X-ray diffraction (XRD) may be used to determine the constituent elements and the crystalline
print first name print last name print student id grade Experiment 2 X-ray fluorescence X-ray fluorescence (XRF) and X-ray diffraction (XRD) may be used to determine the constituent elements and the crystalline
Condensed Matter Physics Prof. G. Rangarajan Department of Physics Indian Institute of Technology, Madras
 (Refer Slide Time: 00:11) Condensed Matter Physics Prof. G. Rangarajan Department of Physics Indian Institute of Technology, Madras Diffraction Methods for Crystal Structure Worked Examples Next, we have
(Refer Slide Time: 00:11) Condensed Matter Physics Prof. G. Rangarajan Department of Physics Indian Institute of Technology, Madras Diffraction Methods for Crystal Structure Worked Examples Next, we have
Physical Chemistry of Polymers (4)
 Physical Chemistry of Polymers (4) Dr. Z. Maghsoud CONCENTRATED SOLUTIONS, PHASE SEPARATION BEHAVIOR, AND DIFFUSION A wide range of modern research as well as a variety of engineering applications exist
Physical Chemistry of Polymers (4) Dr. Z. Maghsoud CONCENTRATED SOLUTIONS, PHASE SEPARATION BEHAVIOR, AND DIFFUSION A wide range of modern research as well as a variety of engineering applications exist
(Section 5.3: Solving Trig Equations) Third factor. u 1 = 0. sin x = 1. x = π 2 + 2π n. ( n integer. Solution set:
 (Section 5.3: Solving Trig Equations) 5.32 Third factor u 1 = 0 u = 1 sin x = 1 x = π 2 + 2π n ( n integer ) Solution set: x x = π n, x = π 6 + 2π n, x = 5π 6 + 2π n, or x = π 2 + 2π n ( n integer ) When
(Section 5.3: Solving Trig Equations) 5.32 Third factor u 1 = 0 u = 1 sin x = 1 x = π 2 + 2π n ( n integer ) Solution set: x x = π n, x = π 6 + 2π n, x = 5π 6 + 2π n, or x = π 2 + 2π n ( n integer ) When
Roger Johnson Structure and Dynamics: X-ray Diffraction Lecture 6
 6.1. Summary In this Lecture we cover the theory of x-ray diffraction, which gives direct information about the atomic structure of crystals. In these experiments, the wavelength of the incident beam must
6.1. Summary In this Lecture we cover the theory of x-ray diffraction, which gives direct information about the atomic structure of crystals. In these experiments, the wavelength of the incident beam must
Introduction to X-ray and neutron scattering
 UNESCO/IUPAC Postgraduate Course in Polymer Science Lecture: Introduction to X-ray and neutron scattering Zhigunov Alexander Institute of Macromolecular Chemistry ASCR, Heyrovsky sq., Prague -16 06 http://www.imc.cas.cz/unesco/index.html
UNESCO/IUPAC Postgraduate Course in Polymer Science Lecture: Introduction to X-ray and neutron scattering Zhigunov Alexander Institute of Macromolecular Chemistry ASCR, Heyrovsky sq., Prague -16 06 http://www.imc.cas.cz/unesco/index.html
Crystal Structure and Electron Diffraction
 Crystal Structure and Electron Diffraction References: Kittel C.: Introduction to Solid State Physics, 8 th ed. Wiley 005 University of Michigan, PHY441-44 (Advanced Physics Laboratory Experiments, Electron
Crystal Structure and Electron Diffraction References: Kittel C.: Introduction to Solid State Physics, 8 th ed. Wiley 005 University of Michigan, PHY441-44 (Advanced Physics Laboratory Experiments, Electron
Lecture 20: Spinodals and Binodals; Continuous Phase Transitions; Introduction to Statistical Mechanics
 Lecture 20: 11.28.05 Spinodals and Binodals; Continuous Phase Transitions; Introduction to Statistical Mechanics Today: LAST TIME: DEFINING METASTABLE AND UNSTABLE REGIONS ON PHASE DIAGRAMS...2 Conditions
Lecture 20: 11.28.05 Spinodals and Binodals; Continuous Phase Transitions; Introduction to Statistical Mechanics Today: LAST TIME: DEFINING METASTABLE AND UNSTABLE REGIONS ON PHASE DIAGRAMS...2 Conditions
Structure of Surfaces
 Structure of Surfaces C Stepped surface Interference of two waves Bragg s law Path difference = AB+BC =2dsin ( =glancing angle) If, n =2dsin, constructive interference Ex) in a cubic lattice of unit cell
Structure of Surfaces C Stepped surface Interference of two waves Bragg s law Path difference = AB+BC =2dsin ( =glancing angle) If, n =2dsin, constructive interference Ex) in a cubic lattice of unit cell
Wave Properties of Particles Louis debroglie:
 Wave Properties of Particles Louis debroglie: If light is both a wave and a particle, why not electrons? In 194 Louis de Broglie suggested in his doctoral dissertation that there is a wave connected with
Wave Properties of Particles Louis debroglie: If light is both a wave and a particle, why not electrons? In 194 Louis de Broglie suggested in his doctoral dissertation that there is a wave connected with
Quiz 8 Introduction to Polymers
 100603 Quiz 8 Introduction to Polymers 1) a) Why is there no real termination for ionic polymerizations (i.e. no coupling or disproportionation)? b) Briefly outline how you would produce Kraton Rubber
100603 Quiz 8 Introduction to Polymers 1) a) Why is there no real termination for ionic polymerizations (i.e. no coupling or disproportionation)? b) Briefly outline how you would produce Kraton Rubber
Physics 541: Condensed Matter Physics
 Physics 541: Condensed Matter Physics Final Exam Monday, December 17, 2012 / 14:00 17:00 / CCIS 4-285 Student s Name: Instructions There are 24 questions. You should attempt all of them. Mark your response
Physics 541: Condensed Matter Physics Final Exam Monday, December 17, 2012 / 14:00 17:00 / CCIS 4-285 Student s Name: Instructions There are 24 questions. You should attempt all of them. Mark your response
Suppose that f is continuous on [a, b] and differentiable on (a, b). Then
![Suppose that f is continuous on [a, b] and differentiable on (a, b). Then Suppose that f is continuous on [a, b] and differentiable on (a, b). Then](/thumbs/74/71325034.jpg) Lectures 1/18 Derivatives and Graphs When we have a picture of the graph of a function f(x), we can make a picture of the derivative f (x) using the slopes of the tangents to the graph of f. In this section
Lectures 1/18 Derivatives and Graphs When we have a picture of the graph of a function f(x), we can make a picture of the derivative f (x) using the slopes of the tangents to the graph of f. In this section
Quantum Condensed Matter Physics Lecture 5
 Quantum Condensed Matter Physics Lecture 5 detector sample X-ray source monochromator David Ritchie http://www.sp.phy.cam.ac.uk/drp2/home QCMP Lent/Easter 2019 5.1 Quantum Condensed Matter Physics 1. Classical
Quantum Condensed Matter Physics Lecture 5 detector sample X-ray source monochromator David Ritchie http://www.sp.phy.cam.ac.uk/drp2/home QCMP Lent/Easter 2019 5.1 Quantum Condensed Matter Physics 1. Classical
Solutions for Problem Set #4 due October 10, 2003 Dustin Cartwright
 Solutions for Problem Set #4 due October 1, 3 Dustin Cartwright (B&N 4.3) Evaluate C f where f(z) 1/z as in Example, and C is given by z(t) sin t + i cos t, t π. Why is the result different from that of
Solutions for Problem Set #4 due October 1, 3 Dustin Cartwright (B&N 4.3) Evaluate C f where f(z) 1/z as in Example, and C is given by z(t) sin t + i cos t, t π. Why is the result different from that of
PART 1 Introduction to Theory of Solids
 Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:1 Trim:165 240MM TS: Integra, India PART 1 Introduction to Theory of Solids Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:2
Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:1 Trim:165 240MM TS: Integra, India PART 1 Introduction to Theory of Solids Elsevier UK Job code: MIOC Ch01-I044647 9-3-2007 3:03p.m. Page:2
Math 20C Homework 2 Partial Solutions
 Math 2C Homework 2 Partial Solutions Problem 1 (12.4.14). Calculate (j k) (j + k). Solution. The basic properties of the cross product are found in Theorem 2 of Section 12.4. From these properties, we
Math 2C Homework 2 Partial Solutions Problem 1 (12.4.14). Calculate (j k) (j + k). Solution. The basic properties of the cross product are found in Theorem 2 of Section 12.4. From these properties, we
10.1 Review of Parametric Equations
 10.1 Review of Parametric Equations Recall that often, instead of representing a curve using just x and y (called a Cartesian equation), it is more convenient to define x and y using parametric equations
10.1 Review of Parametric Equations Recall that often, instead of representing a curve using just x and y (called a Cartesian equation), it is more convenient to define x and y using parametric equations
The Solid State. Phase diagrams Crystals and symmetry Unit cells and packing Types of solid
 The Solid State Phase diagrams Crystals and symmetry Unit cells and packing Types of solid Learning objectives Apply phase diagrams to prediction of phase behaviour Describe distinguishing features of
The Solid State Phase diagrams Crystals and symmetry Unit cells and packing Types of solid Learning objectives Apply phase diagrams to prediction of phase behaviour Describe distinguishing features of
Crystal Structure Determination II
 Crystal Structure Determination II Dr. Falak Sher Pakistan Institute of Engineering and Applied Sciences 09/10/2010 Diffraction Intensities The integrated intensity, I (hkl) (peak area) of each powder
Crystal Structure Determination II Dr. Falak Sher Pakistan Institute of Engineering and Applied Sciences 09/10/2010 Diffraction Intensities The integrated intensity, I (hkl) (peak area) of each powder
Introduction to. Crystallography
 M. MORALES Introuction to Crystallography magali.morales@ensicaen.fr Classification of the matter in 3 states : Crystallise soli liqui or amorphous gaz soli Crystallise soli : unique arrangement of atoms
M. MORALES Introuction to Crystallography magali.morales@ensicaen.fr Classification of the matter in 3 states : Crystallise soli liqui or amorphous gaz soli Crystallise soli : unique arrangement of atoms
X-ray Diffraction. Diffraction. X-ray Generation. X-ray Generation. X-ray Generation. X-ray Spectrum from Tube
 X-ray Diffraction Mineral identification Mode analysis Structure Studies X-ray Generation X-ray tube (sealed) Pure metal target (Cu) Electrons remover inner-shell electrons from target. Other electrons
X-ray Diffraction Mineral identification Mode analysis Structure Studies X-ray Generation X-ray tube (sealed) Pure metal target (Cu) Electrons remover inner-shell electrons from target. Other electrons
Differential Equations: Homework 8
 Differential Equations: Homework 8 Alvin Lin January 08 - May 08 Section.6 Exercise Find a general solution to the differential equation using the method of variation of parameters. y + y = tan(t) r +
Differential Equations: Homework 8 Alvin Lin January 08 - May 08 Section.6 Exercise Find a general solution to the differential equation using the method of variation of parameters. y + y = tan(t) r +
FIRST PUBLIC EXAMINATION SUBJECT 3: PHYSICAL CHEMISTRY
 CCHE 4273 FIRST PUBLIC EXAMINATION Trinity Term 2005 Preliminary Examination in Chemistry SUBJECT 3: PHYSICAL CHEMISTRY Wednesday June 8 th 2005, 9:30am Time allowed: 2 ½ hours Candidates should answer
CCHE 4273 FIRST PUBLIC EXAMINATION Trinity Term 2005 Preliminary Examination in Chemistry SUBJECT 3: PHYSICAL CHEMISTRY Wednesday June 8 th 2005, 9:30am Time allowed: 2 ½ hours Candidates should answer
Structural characterization. Part 1
 Structural characterization Part 1 Experimental methods X-ray diffraction Electron diffraction Neutron diffraction Light diffraction EXAFS-Extended X- ray absorption fine structure XANES-X-ray absorption
Structural characterization Part 1 Experimental methods X-ray diffraction Electron diffraction Neutron diffraction Light diffraction EXAFS-Extended X- ray absorption fine structure XANES-X-ray absorption
AP Calculus (BC) Chapter 10 Test No Calculator Section. Name: Date: Period:
 AP Calculus (BC) Chapter 10 Test No Calculator Section Name: Date: Period: Part I. Multiple-Choice Questions (5 points each; please circle the correct answer.) 1. The graph in the xy-plane represented
AP Calculus (BC) Chapter 10 Test No Calculator Section Name: Date: Period: Part I. Multiple-Choice Questions (5 points each; please circle the correct answer.) 1. The graph in the xy-plane represented
Homework 3 Solutions Problem 1 (a) The technique is essentially that of Homework 2, problem 2. The situation is depicted in the figure:
 Homework 3 Solutions Problem (a) The technique is essentially that of Homework 2, problem 2. The situation is depicted in the figure: θ photon vdt A θ d Figure : The figure shows the system at time t.
Homework 3 Solutions Problem (a) The technique is essentially that of Homework 2, problem 2. The situation is depicted in the figure: θ photon vdt A θ d Figure : The figure shows the system at time t.
Short Sample Solutions to the Sample Exam for (3rd Year Course 6) Hilary Term 2011
 Short Sample Solutions to the Sample Exam for (3rd Year Course 6) Hilary Term 0. [4] A Lattice is an infinite set of points in space where the environment of any given point is identical to the enivironment
Short Sample Solutions to the Sample Exam for (3rd Year Course 6) Hilary Term 0. [4] A Lattice is an infinite set of points in space where the environment of any given point is identical to the enivironment
GEANT4 simulation of the 10 B-based Jalousie detector for neutron diffractometers
 GEANT4 simulation of the 10 B-based Jalousie detector for neutron diffractometers Irina Stefanescu 1, R. Hall-Wilton 1, G. Kemmerling 2, M. Klein 3, C.J. Schmidt 3,4, W. Schweika 1,2 1 European Spallation
GEANT4 simulation of the 10 B-based Jalousie detector for neutron diffractometers Irina Stefanescu 1, R. Hall-Wilton 1, G. Kemmerling 2, M. Klein 3, C.J. Schmidt 3,4, W. Schweika 1,2 1 European Spallation
Interpreting Phase Diagrams
 Interpreting Phase Diagrams Understanding chemical reactions requires that we know something about how materials behave as the temperature and pressure change. For a single component (like quartz or ice)
Interpreting Phase Diagrams Understanding chemical reactions requires that we know something about how materials behave as the temperature and pressure change. For a single component (like quartz or ice)
(Re-write, January 2011, from notes of S. C. Fain Jr., L. Sorensen, O. E. Vilches, J. Stoltenberg and D. B. Pengra, Version 1, preliminary)
 Electron Diffraction (Re-write, January 011, from notes of S. C. Fain Jr., L. Sorensen, O. E. Vilches, J. Stoltenberg and D. B. Pengra, Version 1, preliminary) References: Any introductory physics text
Electron Diffraction (Re-write, January 011, from notes of S. C. Fain Jr., L. Sorensen, O. E. Vilches, J. Stoltenberg and D. B. Pengra, Version 1, preliminary) References: Any introductory physics text
3.012 PS Issued: Fall 2003 Graded problems due:
 3.012 PS 4 3.012 Issued: 10.07.03 Fall 2003 Graded problems due: 10.15.03 Graded problems: 1. Planes and directions. Consider a 2-dimensional lattice defined by translations T 1 and T 2. a. Is the direction
3.012 PS 4 3.012 Issued: 10.07.03 Fall 2003 Graded problems due: 10.15.03 Graded problems: 1. Planes and directions. Consider a 2-dimensional lattice defined by translations T 1 and T 2. a. Is the direction
1 X-Ray Physics. 1.1 Introduction. 1.2 Creation of x-rays
 1 X-Ray Physics 1.1 Introduction One of the main methods to decode the structure of condensed matter on atomic scale is x-ray diffraction. While it was originally used to analyze crystalline structures
1 X-Ray Physics 1.1 Introduction One of the main methods to decode the structure of condensed matter on atomic scale is x-ray diffraction. While it was originally used to analyze crystalline structures
Analytical Methods for Materials
 Analytical Methods for Materials Lesson 15 Reciprocal Lattices and Their Roles in Diffraction Studies Suggested Reading Chs. 2 and 6 in Tilley, Crystals and Crystal Structures, Wiley (2006) Ch. 6 M. DeGraef
Analytical Methods for Materials Lesson 15 Reciprocal Lattices and Their Roles in Diffraction Studies Suggested Reading Chs. 2 and 6 in Tilley, Crystals and Crystal Structures, Wiley (2006) Ch. 6 M. DeGraef
1. (13%) Find the orthogonal trajectories of the family of curves y = tan 1 (kx), where k is an arbitrary constant. Solution: For the original curves:
 5 微甲 6- 班期末考解答和評分標準. (%) Find the orthogonal trajectories of the family of curves y = tan (kx), where k is an arbitrary constant. For the original curves: dy dx = tan y k = +k x x sin y cos y = +tan y
5 微甲 6- 班期末考解答和評分標準. (%) Find the orthogonal trajectories of the family of curves y = tan (kx), where k is an arbitrary constant. For the original curves: dy dx = tan y k = +k x x sin y cos y = +tan y
Chapter 12. Insert picture from First page of chapter. Intermolecular Forces and the Physical Properties of Liquids and Solids
 Chapter 12 Insert picture from First page of chapter Intermolecular Forces and the Physical Properties of Liquids and Solids Copyright McGraw-Hill 2009 1 12.1 Intermolecular Forces Intermolecular forces
Chapter 12 Insert picture from First page of chapter Intermolecular Forces and the Physical Properties of Liquids and Solids Copyright McGraw-Hill 2009 1 12.1 Intermolecular Forces Intermolecular forces
Intermolecular Forces and Liquids and Solids. Chapter 11. Copyright The McGraw Hill Companies, Inc. Permission required for
 Intermolecular Forces and Liquids and Solids Chapter 11 Copyright The McGraw Hill Companies, Inc. Permission required for 1 A phase is a homogeneous part of the system in contact with other parts of the
Intermolecular Forces and Liquids and Solids Chapter 11 Copyright The McGraw Hill Companies, Inc. Permission required for 1 A phase is a homogeneous part of the system in contact with other parts of the
X-ray Diffraction from Materials
 X-ray Diffraction from Materials 008 Spring Semester Lecturer; Yang Mo Koo Monday and Wednesday 4:45~6:00 8. X-ray Diffractometers Diffractometer: a measuring instrument for analyzing the structure of
X-ray Diffraction from Materials 008 Spring Semester Lecturer; Yang Mo Koo Monday and Wednesday 4:45~6:00 8. X-ray Diffractometers Diffractometer: a measuring instrument for analyzing the structure of
X-ray Data Collection. Bio5325 Spring 2006
 X-ray Data Collection Bio535 Spring 006 Obtaining I hkl and α (Ihkl) from Frame Images Braggs Law -predicts conditions for in-phase scattering by equivalent atoms lying in planes that transect a crystal.
X-ray Data Collection Bio535 Spring 006 Obtaining I hkl and α (Ihkl) from Frame Images Braggs Law -predicts conditions for in-phase scattering by equivalent atoms lying in planes that transect a crystal.
TEP Examination of the structure of NaCl monocrystals with different orientations
 Examination of the structure of NaCl TEP Related topics Characteristic X-radiation, energy levels, crystal structures, reciprocal lattices, Miller indices, atomic form factor, structure factor, and Bragg
Examination of the structure of NaCl TEP Related topics Characteristic X-radiation, energy levels, crystal structures, reciprocal lattices, Miller indices, atomic form factor, structure factor, and Bragg
High-Resolution. Transmission. Electron Microscopy
 Part 4 High-Resolution Transmission Electron Microscopy 186 Significance high-resolution transmission electron microscopy (HRTEM): resolve object details smaller than 1nm (10 9 m) image the interior of
Part 4 High-Resolution Transmission Electron Microscopy 186 Significance high-resolution transmission electron microscopy (HRTEM): resolve object details smaller than 1nm (10 9 m) image the interior of
- A general combined symmetry operation, can be symbolized by β t. (SEITZ operator)
 SPACE GROUP THEORY (cont) It is possible to represent combined rotational and translational symmetry operations in a single matrix, for example the C6z operation and translation by a in D 6h is represented
SPACE GROUP THEORY (cont) It is possible to represent combined rotational and translational symmetry operations in a single matrix, for example the C6z operation and translation by a in D 6h is represented
Oxide Structures & Networks
 Oxide Structures & Networks Unit Cell: Primitive Tetragonal (a = b c) 2TiO 2 per unit cell Motif: 2Ti at (0, 0, 0); ( 1 / 2, 1 / 2, 1 / 2 ) & 4O at ±(0.3, 0.3, 0); ±(0.8, 0.2, 1 / 2 ) Ti: 6 (octahedral
Oxide Structures & Networks Unit Cell: Primitive Tetragonal (a = b c) 2TiO 2 per unit cell Motif: 2Ti at (0, 0, 0); ( 1 / 2, 1 / 2, 1 / 2 ) & 4O at ±(0.3, 0.3, 0); ±(0.8, 0.2, 1 / 2 ) Ti: 6 (octahedral
X-ray diffraction is a non-invasive method for determining many types of
 Chapter X-ray Diffraction.1 Introduction X-ray diffraction is a non-invasive method for determining many types of structural features in both crystalline and amorphous materials. In the case of single
Chapter X-ray Diffraction.1 Introduction X-ray diffraction is a non-invasive method for determining many types of structural features in both crystalline and amorphous materials. In the case of single
8.04 Spring 2013 February 13, 2013 Problem 1. (15 points) Radiative collapse of a classical atom
 Problem Set 1 Solutions 8.04 Spring 01 February 1, 01 Problem 1. (15 points) Radiative collapse of a classical atom (a) (5 points) We begin by assuming that the orbit is circular. This seems like circular
Problem Set 1 Solutions 8.04 Spring 01 February 1, 01 Problem 1. (15 points) Radiative collapse of a classical atom (a) (5 points) We begin by assuming that the orbit is circular. This seems like circular
Structure Factors. How to get more than unit cell sizes from your diffraction data.
 Structure Factors How to get more than unit cell sizes from your diffraction data http://homepages.utoledo.edu/clind/ Yet again expanding convenient concepts First concept introduced: Reflection from lattice
Structure Factors How to get more than unit cell sizes from your diffraction data http://homepages.utoledo.edu/clind/ Yet again expanding convenient concepts First concept introduced: Reflection from lattice
Phys 412 Solid State Physics. Lecturer: Réka Albert
 Phys 412 Solid State Physics Lecturer: Réka Albert What is a solid? A material that keeps its shape Can be deformed by stress Returns to original shape if it is not strained too much Solid structure
Phys 412 Solid State Physics Lecturer: Réka Albert What is a solid? A material that keeps its shape Can be deformed by stress Returns to original shape if it is not strained too much Solid structure
A simple model for block-copolymer layer morphology formation
 A simple model for block-copolymer layer morphology formation A. J. Wagner and A. Croll, Department of Physics, North Dakota State University, Fargo, ND 58102 Prato Workshop, February 12, 2016 A big thanks
A simple model for block-copolymer layer morphology formation A. J. Wagner and A. Croll, Department of Physics, North Dakota State University, Fargo, ND 58102 Prato Workshop, February 12, 2016 A big thanks
Electron Diffraction
 Electron iffraction o moving electrons display wave nature? To answer this question you will direct a beam of electrons through a thin layer of carbon and analyze the resulting pattern. Theory Louis de
Electron iffraction o moving electrons display wave nature? To answer this question you will direct a beam of electrons through a thin layer of carbon and analyze the resulting pattern. Theory Louis de
When these terms occur in the text, they are hyperlinked to definitions in the glossary.
 Thermodynamics of Phase Diagrams Version 2.1 Abigail Callanan, MATTER Frank Noble, University of Liverpool Trevor Myers, UMIST/University of Manchester Assumed Pre-knowledge In order for the student to
Thermodynamics of Phase Diagrams Version 2.1 Abigail Callanan, MATTER Frank Noble, University of Liverpool Trevor Myers, UMIST/University of Manchester Assumed Pre-knowledge In order for the student to
XRD Intensity Calculations -Example FCC Cu (centric)
 Ihkl F hkl F hkl hkl f f Cu Cu ( e (1 e XRD Intensity Calculations -Example FCC Cu (centric) Consider Copper which is F 3 with a=3.615å; atoms in positions [0,0,0] m m [½,½,0][½,0,½][0,½,½] and l=1.54å
Ihkl F hkl F hkl hkl f f Cu Cu ( e (1 e XRD Intensity Calculations -Example FCC Cu (centric) Consider Copper which is F 3 with a=3.615å; atoms in positions [0,0,0] m m [½,½,0][½,0,½][0,½,½] and l=1.54å
Quiz 1 XRD ) Explain the error in the following statement: "a laser beam is a focused beam of monochromatic light".
 Quiz 1 XRD 092706 Diffraction involves constructive interference between waves that emanate from structurally organized matter such as from atoms in a crystal. X-ray diffraction uses a relationship of
Quiz 1 XRD 092706 Diffraction involves constructive interference between waves that emanate from structurally organized matter such as from atoms in a crystal. X-ray diffraction uses a relationship of
Rajesh Prasad Department of Applied Mechanics Indian Institute of Technology New Delhi
 TEQIP WORKSHOP ON HIGH RESOLUTION X-RAY AND ELECTRON DIFFRACTION, FEB 01, 2016, IIT-K. Introduction to x-ray diffraction Peak Positions and Intensities Rajesh Prasad Department of Applied Mechanics Indian
TEQIP WORKSHOP ON HIGH RESOLUTION X-RAY AND ELECTRON DIFFRACTION, FEB 01, 2016, IIT-K. Introduction to x-ray diffraction Peak Positions and Intensities Rajesh Prasad Department of Applied Mechanics Indian
Chap. 2. Polymers Introduction. - Polymers: synthetic materials <--> natural materials
 Chap. 2. Polymers 2.1. Introduction - Polymers: synthetic materials natural materials no gas phase, not simple liquid (much more viscous), not perfectly crystalline, etc 2.3. Polymer Chain Conformation
Chap. 2. Polymers 2.1. Introduction - Polymers: synthetic materials natural materials no gas phase, not simple liquid (much more viscous), not perfectly crystalline, etc 2.3. Polymer Chain Conformation
Crystallography Reading: Warren, Chapters 2.1, 2.2, 2.6, 8 Surface symmetry: Can be a clue to underlying structure. Examples:
 Crystallography Reading: Warren, Chapters 2.1, 2.2, 2.6, 8 Surface symmetry: Can be a clue to underlying structure. Examples: Snow (SnowCrystals.com) Bismuth (Bao, Kavanagh, APL 98 66103 (2005) Hexagonal,
Crystallography Reading: Warren, Chapters 2.1, 2.2, 2.6, 8 Surface symmetry: Can be a clue to underlying structure. Examples: Snow (SnowCrystals.com) Bismuth (Bao, Kavanagh, APL 98 66103 (2005) Hexagonal,
CHAPTER 3 THE STRUCTURE OF CRYSTALLINE SOLIDS PROBLEM SOLUTIONS
 CHAPTER THE STRUCTURE OF CRYSTALLINE SOLIDS PROBLEM SOLUTIONS Fundamental Concepts.1 What is the difference between atomic structure and crystal structure? Atomic structure relates to the number of protons
CHAPTER THE STRUCTURE OF CRYSTALLINE SOLIDS PROBLEM SOLUTIONS Fundamental Concepts.1 What is the difference between atomic structure and crystal structure? Atomic structure relates to the number of protons
S8P1C. STATES OF MATTER E.Q. WHAT ARE THE STATES OF MATTER? WHAT ARE THE CHARACTERISTICS OF EACH STATE OF MATTER?
 S8P1C. STATES OF MATTER E.Q. WHAT ARE THE STATES OF MATTER? WHAT ARE THE CHARACTERISTICS OF EACH STATE OF MATTER? ATOMIC MODEL PROJECT BE CREATIVE!!! MIXTURES VS. SUBSTANCES TASK DUE BY FRIDAY, NOVEMBER
S8P1C. STATES OF MATTER E.Q. WHAT ARE THE STATES OF MATTER? WHAT ARE THE CHARACTERISTICS OF EACH STATE OF MATTER? ATOMIC MODEL PROJECT BE CREATIVE!!! MIXTURES VS. SUBSTANCES TASK DUE BY FRIDAY, NOVEMBER
Phase Transitions in Multicomponent Systems
 Physics 127b: Statistical Mechanics Phase Transitions in Multicomponent Systems The ibbs Phase Rule Consider a system with n components (different types of molecules) with r s in equilibrium. The state
Physics 127b: Statistical Mechanics Phase Transitions in Multicomponent Systems The ibbs Phase Rule Consider a system with n components (different types of molecules) with r s in equilibrium. The state
Examples of the Accumulation Function (ANSWERS) dy dx. This new function now passes through (0,2). Make a sketch of your new shifted graph.
 Eamples of the Accumulation Function (ANSWERS) Eample. Find a function y=f() whose derivative is that f()=. dy d tan that satisfies the condition We can use the Fundamental Theorem to write a function
Eamples of the Accumulation Function (ANSWERS) Eample. Find a function y=f() whose derivative is that f()=. dy d tan that satisfies the condition We can use the Fundamental Theorem to write a function
Polymer Dynamics and Rheology
 Polymer Dynamics and Rheology 1 Polymer Dynamics and Rheology Brownian motion Harmonic Oscillator Damped harmonic oscillator Elastic dumbbell model Boltzmann superposition principle Rubber elasticity and
Polymer Dynamics and Rheology 1 Polymer Dynamics and Rheology Brownian motion Harmonic Oscillator Damped harmonic oscillator Elastic dumbbell model Boltzmann superposition principle Rubber elasticity and
Solid State Physics 460- Lecture 5 Diffraction and the Reciprocal Lattice Continued (Kittel Ch. 2)
 Solid State Physics 460- Lecture 5 Diffraction and the Reciprocal Lattice Continued (Kittel Ch. 2) Ewald Construction 2θ k out k in G Physics 460 F 2006 Lect 5 1 Recall from previous lectures Definition
Solid State Physics 460- Lecture 5 Diffraction and the Reciprocal Lattice Continued (Kittel Ch. 2) Ewald Construction 2θ k out k in G Physics 460 F 2006 Lect 5 1 Recall from previous lectures Definition
3.17 Strukturanalyse mit Röntgenstrahlen nach Debye- Scherrer
 3.17 Strukturanalyse mit Röntgenstrahlen nach Debye- Scherrer Ausarbeitung (engl.) Fortgeschrittenenpraktikum an der TU Darmstadt Versuch durchgeführt von: Mussie Beian, Jan Schupp, Florian Wetzel Versuchsdatum:
3.17 Strukturanalyse mit Röntgenstrahlen nach Debye- Scherrer Ausarbeitung (engl.) Fortgeschrittenenpraktikum an der TU Darmstadt Versuch durchgeführt von: Mussie Beian, Jan Schupp, Florian Wetzel Versuchsdatum:
Chapter 12 & 13 Test Review. Bond, Ionic Bond
 Chapter 12 & 13 Test Review A solid solute dissolved in a solid solvent is an Alloy What is happening in a solution at equilibrium? The Ionic rate of Bond dissolving is equal to the rate of crystallization.
Chapter 12 & 13 Test Review A solid solute dissolved in a solid solvent is an Alloy What is happening in a solution at equilibrium? The Ionic rate of Bond dissolving is equal to the rate of crystallization.
Phonons I - Crystal Vibrations (Kittel Ch. 4)
 Phonons I - Crystal Vibrations (Kittel Ch. 4) Displacements of Atoms Positions of atoms in their perfect lattice positions are given by: R 0 (n 1, n 2, n 3 ) = n 10 x + n 20 y + n 30 z For simplicity here
Phonons I - Crystal Vibrations (Kittel Ch. 4) Displacements of Atoms Positions of atoms in their perfect lattice positions are given by: R 0 (n 1, n 2, n 3 ) = n 10 x + n 20 y + n 30 z For simplicity here
