:Philips Research RfP~rts
|
|
|
- Ferdinand Ward
- 6 years ago
- Views:
Transcription
1 VOL: 24 No. 1 FEBRUARY 1969 :Philips Research RfPrts EDITED BY THE RESEARCH LABORATORY OF N.V. PHILlPS' GLOEILAMPENFABRIEKEN, EINDHOVEN, NETHERLANDS R 683 Philips Res. Repts 24, 1-14, 1969 SEMICONDUCTING PROPERTIES OF LANTHANUM-COBALT OXI0E by G. H. JONKER *) Abstract The semiconducting properties of p- and /I-doped LaCo03 samples have been reinvestigated. For the analysis of the measurements use is made of curves representing the Seebeck coefficient as a function of the resistivity of these samples. The temperature coefficient of the conductivity is explained as being due to the variation of both the number and the mobility of the charge carriers. The activation energy of the mobility is probably caused by "spin-state trapping". 1.: Introduction The compound lanthanum-cobalt oxide LaCo03 has been the subject of many crystallographic 1.2), magnetic 3-8) and semiconductor 3.5.9) investigations. At room temperature it has a rhombohedrally distorted perovskite structure (space group R3c) with the following lattice parameters: a = A, Cl( = 90 41', V = A3, with 8 molecules La.co03 per unit cell. At 937 C a first-order transition to a pseudocubic structure occurs 2). The ground state of Co3+ is the diamagnetic low-spin state. The energy of this state differs very little from that of the paramagnetic high-spin state 8) so that at room temperature a mixture of these two states is present. It has been found that mixed crystals of LaCo0 3 with SrCo0 3 show a weak ferromagnetism 3). At room temperature pure LaCo0 3 has a resistivity of about 3 Q cm, which points to a low value of the forbidden energy gap Eg. The possibility of investigating the normal semiconducting properties of LaCo0 3 is severely restricted by the fact that above room temperature an anomalously fast increase of the conductivity occurs 5). This has been explained as being due to the influence of the high concentrations of charge carriers formed at this temperature on the value of Eg. This value goes rapidly to zero, leading to metallic or pseudo metallic conduction near 1000 "K, At low temperature another limit caused by impurity conduction is to be expected. From conductivity and thermoelectric measurements on Th (n)- and "') Now at Technical University Twente, Enschede, The Netherlands.,.
2 2 G. H. JONKER Sr (p)-doped LaCo03 it has been -concluded that at room temperature Eg is 0 35 ev and that the ratio between the hole and electron mobilities #+/IL is 10 to 15 9 ). These conclusions were based on the assumptions that at room temperature (a) the donor and acceptor centres were completely ionized, (b) the conduction was of the small-polaron type, so that the densities of states in the two bands could be taken equal to the atomic density and (c) the transport coefficients could be neglected. The presence of small polarons was clearly affirmed by optical investigations 10). Since new experiments have shown that there is no hopping of charge carriers in simple oxides like NiO, CoO and Fe203 and that a better description of the semiconducting properties can be given using a band picture with high densities of states 11) it seemed to us of interest to reconsider the properties of LaCo03. In this paper we report on the semiconducting properties of a large series of LaCo0 3 samples doped with foreign ions which replace either the La or the Co ion. Further it was of interest to know whether the electrical properties are influenced by the rhombohedral deformation of the crystal structure. Therefore, we also prepared a number of samples from mixed crystals of LaCo03 and YCo03 having an orthorhombic distortion ofthe lattice 8). For the combined analysis ofresistivity and Seebeckeffect measurements a new method is used which is especially applicable in the case of semiconductors with a smaii forbidden energy gap 12). 2. Experimental part 2.1. Preparation Lanthanum-cobalt oxide was prepared using normal ceramic techniques consisting of milling weighed amounts of La203 and CoC0 3, prefiring the mixture at 900 C for 15 hours, milling the product and firing isostatically pressed rods for 10 to 20 hours in air or in oxygen at temperatures varying from 1200 to 1400 C. The samples were cooled either slowly in the furnace or quenched by shifting them rapidly to the cold end of the furnace. The La203 was 99 9 % quality, the main impurities being other rare-earth elements; CoC0 3 of 99 8 % purity was used, the main impurities being 0 1 % Ni and 0'1 % Na. After it was found that these impurities formed acceptor centres we used especially purified,coc03 *) containing less than 10 ppm Na and Ni. Solid solutions of (1 - x) LaCo03 - x YCo03 (x = 0-0'10) were prepared. by the same method. X-ray analysis showed that samples with x < 0 06 were rhombohedral and with x > 0 06 orthorhombic in structure. The composition La. 9YO'l Co03 was chosen for further experiments. It is known that small amounts of Sr and Th substituted for La act as P: *) Thanks are due to Dr W. Kwestroo of this Iaboratory, who purified the CoC0 3
3 SEMICONDUCTING "' PROPERTIES OF LANTHANUM-COBALT OXIDE 3 and n-type dopes, respectively 9). In addition we investigated a number of small ions (Mg, Ti, Mn, Ni, Cu, Zn, Zr, Ce) as substitutes for Co. For most of these dopes series of samples with varying amounts were prepared by substituting the appropriate oxides or carbonates in the raw material. The densities of the samples varied between 95 and 97 % of the theoretical value of Chemical analysis As the ratio between the metal oxides was fixed by the weights of the oxides or carbonates in the initial mixtures, only the oxygen content of the samples needed examination by chemical analysis. The standard method of wet chemical analysis described in ref. 13 was applied. This method determines the amount of "chemically active oxygen", which is to in pure LaCoÛ3 As the normal -: accuracy of this method of ± 0 5 % is relative to to, the accuracy with respect to the total amount of oxygen ions in the compound is ± 0 1 %. This seems to be a good result but it corresponds at the same time to an inaccuracy.of ± 1% in the mean valency of Co, which is much too high for semiconductor investigations. Fortunately, doped samples have a natural tendency to be constant in composition so that most of the care of preparation was coneentrated on the undoped or slightly doped samples. Inhomogeneous deviations in oxygen content appear more clearly in the semiconductor properties than in chemical analysis Physical measurements Both for the resistivity and the thermoelectric measurements rods of 15 to 25 mm length were used, the thickness being adjusted in dependence of the resistance value. For resistivity measurements at lower temperatures four indiummercury electrodes were applied. These were replaced by silver electrodes for high temperatures. For the thermoelectric measurements use was again made of indium-mercury electrodes, care being taken to obtain a complete liquidlayer contact between the end faces of the samples and the silver or copper blocks ofthe apparatus. For thermoelectric measurements near room temperature one metal block was kept at a temperature of about 14 C by fast-running tap water, the other at 34 5 C by the vapour of boiling diethylether. The lowtemperature measurements were performed in an apparatus described in ref. 11. In fig. 1 the room-temperature resistivities of a few series of LaCo03 doped with different types of foreign ions are shown as a function of the dope coneentration. Roughly, for each type of dope a linear relation between resistivity and concentration is present. For Mn and Ti deviations occur both at low and at high concentrations. In table I the dope elements are divided into two groups, one leading to x-type, the other to p-typé conduction, respectively. For a number of extrinsic p- and n-type doped LaCo03 samples the resis-
4 4 G.H.JONKER 8. Cl.. 'mlr-.0 '_ -+ :;:;. I. m t-----"--j-----7''r----rf- - Dope concentrapon Fig. 1. Resistivity of LaCo03 samples at room temperature as a function of the dope concentration; 000 Sr;... Ti; x x x Mn. TABLE I type n p site La I Co I La Co Th Mn Ti Sn Zr Ce Sr Mg Ni Cu Zn tivity (! and the Seebeck coefficient a have been measured as a function of temperature. The results are shown in figs 2 and 3. In these plots the ordinates for log (! and a have been chosen in such a way that (kie) In 10 = 198 {LV;oK for a corresponds 'to one decade in (!.. Further, the measurements of a and (! at fixed' temperatures for a.iarge series of samples of LaCo03 and of Lao.gYo'lCo03 are presented in combined a-log (! plots in figs 4, 5 and 6.: Both a and (! depend on the actual chargecarrier concentrations, so that in these combined plots irregularities that are mainly due to slight chemical variations in the samples disappear. These
5 SEMICONDUCTING PROPERTIES OF LANTHANUM-COBALT OXIDE 'i Ë 105 s- o -2: )' S.!::S._ Cl.... c:.!!!,. :;:; la'... lij. 0 11) lij ct: 1.Q lij 10 3,,- // I' 1 / 10 2 I I / 10' ---' I, lj -'C lj lij 1 1Or O o la IT Fig. 2. Resistivity e and Seebeck coefficient IX as a function of the reciprocal temperature for LaCo0 3 samples doped with 0, 0 2 (curve b), 0 4 and 1'0% Sr (curve a: 0'2%, after ref. 9). chemical variations include errors, impurities and also deviations from the ideal oxygen content, but not inhomogeneities which remain as a disturbing factor. Indeed, most of the points form together smooth symmetrical curves that can be,considered as characteristics of the compounds at 298 and 200 "K, respectively.
6 6 G.H.JONKER J 'loog!t Fig. 3. Resistivity and Seebeck coefficient as a function of the reciprocal temperature for LaCo03 samples doped with 0'2, 0'5 and 1'0% Mn. 3. Analysis 3.1. The infiuence of foreign ions It is not difficult to understand that foreign ions having fixed valencies like Sr2+ and Th 4 +, substituted for La3+ are compensated byco+" or holes and by Co2+ or electrons respectively. The same holds for Mg2+ and Zr4+ ions, substituted for Co3+. The other dopes used, belonging to the first series of transition elements, need a more careful inspection. It is a peculiar property that elements with a lower atomic number act as donors (Ti, Mn) and those with a higher atomic number as acceptors (Ni, Cu, Zn). This is just the reverse ofwhat is known of common broad-band semiconductors in which the chemical bonding is preponderantly covalent. For compounds with a more ionic character, obviously a different mechanism determines the character of the dopes. Here the ionization potentials of the ions are of more importance than the atomic numbers The temperature dependence of «and e.. Ithas been mentioned in the introduction that the temperature range available for the analysis of the normal conduction properties is rather small. From the
7 SEMICONDUCfING PROPERTIES-OF LANTHANUM-COBALT OXIDE l::l... c:.!!! 600 QJ -g 4-00 c'l o VB+ / / - Á i\ /' / \./.. 0 7'... C /1 <, /11 ;!" +_... \ '" 1 B:S::::_ r-, x... ẋ" 7 9- '1, ",E la 2 10-' 100 la' -----<_ Resistivity P(.11cm) Fig. 4. IX -log (! plot for LaCo0 3 at 298 "K; 000 undoped samples, H after ref. 5; i: intrinsic value; p-type:., Sr n-type: x x x Ti - M.g Mn III NI measurements it can be concluded that room temperature is near the upper limit and that a temperature of about 170 C may be considered as the lower limit. In this range the slope of the IX vs lit curves of doped samples is due to variation of the concentration of one type of charge carriers only, according to IX = (kie) {In (Nln) + A}. Here N represents the density of states in the conduction band, and A the transport coefficient. On the other hand, the slope of the log e vs 1IT curves is due to the variation of the number and of the mobility of the charge carriers, according to e- 1 = ne ft. In the case of NiO of CoO it has been found that the two curves are parallel. This points to the absence of an activation energy of the mobility. In our case we see that nonparallel curves of IX and log e occur both for p- and for n-type conduction (figs 2 and 3). From the difference in slope of the IX and the log e curves a small activation energy q; = 0 05 ev is found for the p-type mobility leaving 0, = 0 07 ev for the acceptor distance EA' Such low values are un-
8 8 G. H. JONKER o !7 _,/' 0 /- -\ t> 0 / NV A C V"", -: -. V r-, I x / J - 1'", E, la la Resistivity p(.[1cm) Fig. 5. IX-log (! plot for Lao'9 YO.IC003 at 298 ok; 000 undoped samples; p-type:... Sr; /I-type: x x x Mn. certain of course, as also impurity conduction would lead o a deviation in slope 11). The presence of impurity conduction is obvious below 150 "K in the Cl: - T curves of fig. 2. Further both the curves of Cl: and of log e point to a beginning of saturation of the charge-carrier concentration near room temperature. As the anomalous intrinsic conduction starts at room temperature as well, the real value of the saturated extrinsic conductivity cannot- be determined. We may assume, however, that the value of the conductivity at room temperature corresponds quite well to the value of the saturated conductivity. This leads to a value of the hole mobility f-t+ of 0 3 cm2fv s. The n-type samples show amore pronounced difference in slope between the Cl: and the log e curves. Here we calculate activation energies q: for the electron mobility f-t- varying between 0'12 and 0 13 ev and very small dissociation energies varying between 0 for Mn-doped and 0,02-0,05 ev for Ti-doped samples, respectively. As saturated conduction isreally present in these samples at room temperature, the electron mobility f-t- can be calculated again assuming complete equality between donor and electron concentration. This gives a value 0,,-
9 SEMICONDUCTING PROPERTIES OF LANTHANUM-COBALT OXIDE 9 o "'-n // V i\.// \ / / I -: \ 1/ \ /-,B;/ A' C' / k' -, // -. <, VB+/B'+ B-"'-.., -,<, B'- <, "+<,, '" / x <, To x I + / / ' ID' Resistivity p(l1cm) Fig. 6. IX - log e plot for LaCo0 3 at 200 "K (the curve of 298 "K is given for comparison); i: intrinsicvalue; 000 undoped samples, H after ref. 5; p-type:... Sr; 1I-type: x x x Ti, Mn. of 0 03 cm 2 fy S for IL, in agreement with ref. 9. The combination leads to a value of b = fh+ffh- = 10. As this value too' is inaccurate we shall consider in the following sections besides this value also a higher value, e.g. ') = Analysis of the 0: - log ç plots In ref. 11 it has been shown that a semiconductor can be characterized at a certain temperature by a pear-shaped 0: vs log e curve, representing samples with p-type, n-type and mixed conduction. The three 0: - log e plots presented in figs 4, 5 and 6 indeed show for the greater part the expected symmetric shape. This was not so in the beginning of our experiments, when we found deviations near the extreme values of 0:,' especially near the positive maximum, where too low values of 0: were measured. The origin of this effect was probably a slight surface oxidation during cooling of the samples in the furnace. This causes parallel conduction in a thin layer, which.affects both the apparent 0: and e. The «-Iog e combination of such a layer can be represented on the 0: - log e curve by a point that is shifted towards the p side with respect to the point representing the bulk properties of the sample. In parts of the 0: - log e curve that are straight, these incorrect samples stiii give points lying nearly on the
10 10. G. H. JQNKER ideal curve. This holds for the extrinsic regions and fortunately also for the region around the maximum of log ç. The 'deviations occur in parts of high curvature, near the extreme values of IX. Samples of stoichiometrie composition show the highest sensitivity to oxidation or reduction so that for LaCo03 the deviations are maximal near the positive maximum of IX. Further it is clear that at lower temperatures stronger deviations occur and therefore the analysis of the low-temperature curve is less certain. Another uncertainty about the correctness of the analysis is caused by im- purity conduction. The presence of this effect is obvious below 150 "K (fig. 2) but a rest may be present at 200 "K. This effect is stronger in doped.samples, increasing with the dope concentration. Therefore, especially the extrinsic n part of the curve of 200 "K is uncertain. This effect too has only little influence on the properties of samples with maximum resistivity. Accepting now the IX - log {}curves of figs 4, 5 and 6, the analysis goes along lines reported previously 12). The IX -log {}curves are described by k ( {}2 )1/2 k [{}max { ( {}2 )1/2}] k IX = ± -E In - 1 ± ln(bc), e {}max e {} {}max e with 1 (}max = HVb + Vb) {}I, #+ N+ e A + b=- and c=, #_ N_ e A - where Eg is the forbidden energy gap, A+ and A_ are the transport coefficients and N+ and N_ are the densities of states in the p- and n-type conduction bands. The value of E can be derived in different ways, for instance from the extreme values of IX according to k (IX+ + IX_)cxtreme = - [E - 1-In {2(E - I))]. e For LaCo03 at room temperature (25 C) (fig. 4) the value of E is 15,4, corresponding to Eg + (A+ + A_) k T = ev. The complete curve, calculated with E = 15 4 fits very well to the experimental points, especially in the region of lower dope concentrations. This curve gives (}mnx = 9 0.Q cm.. For LaO.9YO'lC003 at 25 C(fig. 5) E = 14 3 corresponding to Eg + (A+ + + A_) k T = ev. Here too {}mnx = 9 0.Q cm. This shows that there is no fundamental difference between the rhombohèdral and the orthorhombic compound. Therefore the analysis has been continued for pure LaCo03 only.
11 TABLE II Results derived directly from ct - log e plots of LaCo I I Tl = 298 ok T I 2 = K I E = Eglk T + A+ + A_ a = Egolk T + A+ + A_ Ego = 0 32 ev -PIk Eg + (A+ + A_) k T b or 0'39 ev Ego + (A+ + A_ - Plk)kT 0'37 ev A+ + A_ - PIk = 2'7 c f3max 9 0 ncm 1' ncm Ego + q; + q_ = 0 50 ev Ego = 0 32 ev (point A) d (N+ e A + e Jl.+) ncm 3' ncm I q; = 0 05 ev I (point B+) I (Ne e A - ejl._)-l (point B_) I 5' ncm I 6' ncm I q: = 0 13 ev I f. I asymmetry of IX 165 (J.VrK 230 (J.VrK 1 I -I be = N+ e A + Jl q : -q+ = 0 08 ev g N_ ea- p-: I (/.I!:ti s:: I ig I I:::
12 12 G.H.JONKER Fór this?ase a - log e plots at two temperatures are available. In table 11 the data that can be directly calculated, using the methods previously described 12), are given in columns 3 and 4. Further, column 5 gives data derived by combining the data obtained at 200 and 28? "K and column 6 data derived from column 5. For these calculations the following considerations have been used:. ".. (1) Generally a forbidden energy gap is weakly temperature-dependent according to Eg = Ego _:_f3 T.. " (2) In the product N /.l = N /-to e- q / k T the temperature variation of N /.lo has been neglected with respect to the exponential function. This is of course only permissible for q» k T. (3) For the temperature dependence of N e A be = + 1""+ N_ e A - /-tit is assumed that c is constant. The activation energy of b then is equal to the difference of q; and q_. In table III further results are shown using a value 'of 0 03 cm 2 fv s for /-t'- and b= 10 and 16, respectively, and further a few rasonable values for the sum of the transport constants. TABLE III Further results calculated for LaCo assumptions at 298 C quantity T= 298 K T= K a b = 10 2 em.x 5 2 ncm 250 ncm b = 16 et = Vb + lïvb 4 25 ncm 180 ncm f.t- = 0 03 cm 2 jv s 1 b 3' cmb= 10 N 3 t = b = 16 et e (p,+ + f.t-) 2' cm- 3 f.t- = 0'03 cm 2 jv s N_ e A - 4' cm- 3 c b= 10 N+ e A + 1' cm- 3 b= 16 N+ e A ,1 cm-3 ct A+ + A_ - {Jjk=2'7 A+ +A_ =4 =5 = ev 0 30 ev Eg 0 26 ev 0 28 ev 0 23 ev 0 26 ev, In fig. 4 the points. representing,. n-type samples'doped with high amounts of, -, '.-. Mn or Ti (> 3 %) show a strong deviation from the calculated curve. The resistivity passes through a minimum at a dope concentration of 8 %. This effect has been explained in ref. 8 where the complete solid-solution system of
13 SEMICONDUCTING PROPERTIES OF LANTHANUM-COBALT OXIDE 13 Laéo03 with LaMn03 has been described. At a ratio of 1 : 1 an ordered compound La2Co2+Mn4+06 is formed. With Ti a comparable compound La2Co2+Ti4+06 is formed. Both these compounds have a high resistivity, pointing to a much larger forbidden energy gap than has been found' for LaCo03. Both these compounds are p';type semiconductors if a small excess Co is present (Co3+). This shows that somewhere between this composition and pure LaCo03 the resistivity must show a minimum and that a change in.sign of oe has to occur. One would expect a quasi-metallic behaviour at this \ transition point where the Fermi level passes through the n-conduction level. The fact that this does not occur 'may be due to the formation of clusters of Co2+Mn4+ or C02+Ti4+, with short-range order. 4. Discussion The present results confirm in general the data given by Gerthsen and Härdtl 9). Only quantitative differences occur as a result of an improved method of analysis. Both in the rhombohedral and in the orthorhombic crystal structure LaCo03' proves to be a semiconductor with a narrow forbidden energy gap Eg of about 0 30 ev, separating two transport bands with high densities of states. The mobilities of the charge carriers are low, as is generally found in this type of semiconductors: at room temperature t-t+ 0 3 cm 2 JV s, and t-t cm2jv s. For t-t+ a weak thermal activation energy of 0 05 ev has been found. This value is rather uncertain as some influence of impurity conduction cannot be excluded with certainty. The conduction electrons, however, show clearly a high activation energy q.: of 0 13 ev. This property, which distinguishes LaCo03 from other transition-metal oxides, needs. further examination. In our opinion this property is connected to the variation in spin state of the different valency states of Co. This idea has been applied earlier in the discussion of the dipole centres in CoO doped with Li 11). In pure LaCo03 the trivalent Co ions have the diamagnetic low-spin state as a ground state. The energy difference between the ground state and the paramagnetic highspin state is only 0 01 ev 8). Thus, at higher temperatures mixtures of the two states occur which are estimated to contain at200 ok at 298 ok 25 % Co3+ h.s., 32 % Co3+ h.s. In oxides Co2+ ions are only known in the high-spin state; C04+ ions are less well known, but are most probably in the low-spin state. The exchange of electrons between two neighbour ions goes easily if the exchange occurs between equal types of energy levels (te or t 20 ) and if the new states of the ions after the exchange are equilibrium state. This condition isautomatically present for two neighbour-ions having the same spin state, but not if one of the ions is in the high-spin. state and the other in the low-spin state. Then at first sight,
14 14. G. H. JONKER one would expect an easy exchange o'f holes between C04+ l.s. and Co3+ l.s. ions and also that of electrons between C0 2 + h.s. and Co3+ h.s. ions, respectively. However; as has been discussed by Goodenough 4) and Blasse 7), such neighbour pairs do not occur, as in the perovskite structure the crystal-field splitting of the d levels at a certain site is very sensitive to the occupation of the neighbour sites. In a configuration Co4+-O-Co3+ the polarization of the oxygen ion increases the crystal field at the Co4+ site and decreases that of the Co3+ site. Therefore the Co4+ l.s. state is stabilized, but the 6 surrounding Co3+ ions obtain the high-spin state. This means at the same time a localization.of the hole. This "spin-state localization" has to be added to the normal polarization localization considered in Holstein's small-polaron theory. The opposite effect occurs at the site of a Co2+ ion where the high-spin state is stabilized and at the same time the low-spin states of the 6 surrounding Co3+ ions. The extra electrons of C0 2 + h.s. and the holes of Co4+ l.s. ions are situated on f 2g levels. The f 2y wave functions are extended in directions pointing to the next-nearest Co3+ neighbours. The majority of these Co3+ ions are in the low-spin state and this may be the origin 'of the rather high activation energy of 1"- and a rather low one of /1-+, respectively. The "spin-state localization" will have the same sensitivity to lattice vibrations as the dielectric-polarization trapping normally considered in smallpolaron theories. Therefore, it may be that the sum of these two effects is strong enough to cause a thermal activation energy in the electron and hole mobilities of LaCo0 3, which effect has not been found in the d.c. properties of other transition-metal compounds (compare De Wit 14)). Eindhoven, June 1968 REFERENCES 1) H. L. Yakel, Acta cryst. 8, 394, ) P. M. Raccah and J. B. Goodenough, Phys, Rev. 155,932, ) G. H. Jonker and J. H. van Santen, Physica 19, 120, ) J. B. Goodenough, J. Phys. Chem. Solids 6, 287, ) R. R. Heikes, R. C. Miller and R. Mázelsky, Physics 30, 1600, ) C. S. Naiman, R. Gilmore and B. D. BartoIo, J. appl. Phys. 36, 1074, )'G. Blasse, J. appl. Phys. 36, 879, ) G. H. Jonker, J. appl, Phys. 37, 1424, ) P. Gerthsen and K. H. Härdtl, Z. Naturf. 17a, 514, ) H. G. Reik, E. Kauer and P. Gerthsen, Phys. Letters 8, 29, H. G. Reik and R. Milhlstroh, Solid State Comm. 5, 105, R. Müh lst roh and H. R. Reik, Phys. Rev. 162, 703, ) A. J. Bosman and C. Crevecoeur, Phys. Rev. 144, 763, ) G. H. Jonker, Philips Res. Repts 23, 131, ) G. W. van. Oosterhout and J. Visser, Anal. chim. Acta 33,330, ) H. J. de Wit, Philips Res. Repts 23, 449, 1968.
THE APPLICATION OF COMBINED CONDUCTIVITY AND SEEBECK-EFFECT PLOTS FOR THE ANALYSIS OF SEMICONDUCTOR PROPERTIES
 R657. Philips Res. Repts 23, 131-138, 1968 THE APPLICATION OF COMBINED CONDUCTIITY AND SEEBECK-EFFECT PLOTS FOR THE ANALYSIS OF SEMICONDUCTOR PROPERTIES by G. H. JONKER Abstract A semiconductor can be
R657. Philips Res. Repts 23, 131-138, 1968 THE APPLICATION OF COMBINED CONDUCTIITY AND SEEBECK-EFFECT PLOTS FOR THE ANALYSIS OF SEMICONDUCTOR PROPERTIES by G. H. JONKER Abstract A semiconductor can be
ON THE DOUBLE-EXCHANGE INTERACTIONS IN La 1 _xba x Mn03
 R 712 Philips Res. Repts 25,8-16,1970 ON THE DOUBLE-EXCHANGE INTERACTIONS IN La 1 _xba x Mn03 Abstract 1. Introduetion by F. K. LOTGERING Magnetic and electrical measurements on Lal_",Ba",Mn03 and Lal_",Ba",Mnl_",Ti",03
R 712 Philips Res. Repts 25,8-16,1970 ON THE DOUBLE-EXCHANGE INTERACTIONS IN La 1 _xba x Mn03 Abstract 1. Introduetion by F. K. LOTGERING Magnetic and electrical measurements on Lal_",Ba",Mn03 and Lal_",Ba",Mnl_",Ti",03
Lecture 1. OUTLINE Basic Semiconductor Physics. Reading: Chapter 2.1. Semiconductors Intrinsic (undoped) silicon Doping Carrier concentrations
 Lecture 1 OUTLINE Basic Semiconductor Physics Semiconductors Intrinsic (undoped) silicon Doping Carrier concentrations Reading: Chapter 2.1 EE105 Fall 2007 Lecture 1, Slide 1 What is a Semiconductor? Low
Lecture 1 OUTLINE Basic Semiconductor Physics Semiconductors Intrinsic (undoped) silicon Doping Carrier concentrations Reading: Chapter 2.1 EE105 Fall 2007 Lecture 1, Slide 1 What is a Semiconductor? Low
ELEMENTARY BAND THEORY
 ELEMENTARY BAND THEORY PHYSICIST Solid state band Valence band, VB Conduction band, CB Fermi energy, E F Bloch orbital, delocalized n-doping p-doping Band gap, E g Direct band gap Indirect band gap Phonon
ELEMENTARY BAND THEORY PHYSICIST Solid state band Valence band, VB Conduction band, CB Fermi energy, E F Bloch orbital, delocalized n-doping p-doping Band gap, E g Direct band gap Indirect band gap Phonon
Lecture 7: Extrinsic semiconductors - Fermi level
 Lecture 7: Extrinsic semiconductors - Fermi level Contents 1 Dopant materials 1 2 E F in extrinsic semiconductors 5 3 Temperature dependence of carrier concentration 6 3.1 Low temperature regime (T < T
Lecture 7: Extrinsic semiconductors - Fermi level Contents 1 Dopant materials 1 2 E F in extrinsic semiconductors 5 3 Temperature dependence of carrier concentration 6 3.1 Low temperature regime (T < T
Energy bands in solids. Some pictures are taken from Ashcroft and Mermin from Kittel from Mizutani and from several sources on the web.
 Energy bands in solids Some pictures are taken from Ashcroft and Mermin from Kittel from Mizutani and from several sources on the web. we are starting to remind p E = = mv 1 2 = k mv = 2 2 k 2m 2 Some
Energy bands in solids Some pictures are taken from Ashcroft and Mermin from Kittel from Mizutani and from several sources on the web. we are starting to remind p E = = mv 1 2 = k mv = 2 2 k 2m 2 Some
EE 346: Semiconductor Devices. 02/08/2017 Tewodros A. Zewde 1
 EE 346: Semiconductor Devices 02/08/2017 Tewodros A. Zewde 1 DOPANT ATOMS AND ENERGY LEVELS Without help the total number of carriers (electrons and holes) is limited to 2ni. For most materials, this is
EE 346: Semiconductor Devices 02/08/2017 Tewodros A. Zewde 1 DOPANT ATOMS AND ENERGY LEVELS Without help the total number of carriers (electrons and holes) is limited to 2ni. For most materials, this is
ESE 372 / Spring 2013 / Lecture 5 Metal Oxide Semiconductor Field Effect Transistor
 Metal Oxide Semiconductor Field Effect Transistor V G V G 1 Metal Oxide Semiconductor Field Effect Transistor We will need to understand how this current flows through Si What is electric current? 2 Back
Metal Oxide Semiconductor Field Effect Transistor V G V G 1 Metal Oxide Semiconductor Field Effect Transistor We will need to understand how this current flows through Si What is electric current? 2 Back
PN Junction
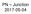 P Junction 2017-05-04 Definition Power Electronics = semiconductor switches are used Analogue amplifier = high power loss 250 200 u x 150 100 u Udc i 50 0 0 50 100 150 200 250 300 350 400 i,u dc i,u u
P Junction 2017-05-04 Definition Power Electronics = semiconductor switches are used Analogue amplifier = high power loss 250 200 u x 150 100 u Udc i 50 0 0 50 100 150 200 250 300 350 400 i,u dc i,u u
Introduction to Engineering Materials ENGR2000. Dr.Coates
 Introduction to Engineering Materials ENGR2000 Chapter 18: Electrical Properties Dr.Coates 18.2 Ohm s Law V = IR where R is the resistance of the material, V is the voltage and I is the current. l R A
Introduction to Engineering Materials ENGR2000 Chapter 18: Electrical Properties Dr.Coates 18.2 Ohm s Law V = IR where R is the resistance of the material, V is the voltage and I is the current. l R A
Semiconductors 1. Explain different types of semiconductors in detail with necessary bond diagrams. Intrinsic semiconductors:
 Semiconductors 1. Explain different types of semiconductors in detail with necessary bond diagrams. There are two types of semi conductors. 1. Intrinsic semiconductors 2. Extrinsic semiconductors Intrinsic
Semiconductors 1. Explain different types of semiconductors in detail with necessary bond diagrams. There are two types of semi conductors. 1. Intrinsic semiconductors 2. Extrinsic semiconductors Intrinsic
Semiconductor physics I. The Crystal Structure of Solids
 Lecture 3 Semiconductor physics I The Crystal Structure of Solids 1 Semiconductor materials Types of solids Space lattices Atomic Bonding Imperfection and doping in SOLIDS 2 Semiconductor Semiconductors
Lecture 3 Semiconductor physics I The Crystal Structure of Solids 1 Semiconductor materials Types of solids Space lattices Atomic Bonding Imperfection and doping in SOLIDS 2 Semiconductor Semiconductors
Electrons, Holes, and Defect ionization
 Electrons, Holes, and Defect ionization The process of forming intrinsic electron-hole pairs is excitation a cross the band gap ( formation energy ). intrinsic electronic reaction : null e + h When electrons
Electrons, Holes, and Defect ionization The process of forming intrinsic electron-hole pairs is excitation a cross the band gap ( formation energy ). intrinsic electronic reaction : null e + h When electrons
ADVANCED UNDERGRADUATE LABORATORY EXPERIMENT 20. Semiconductor Resistance, Band Gap, and Hall Effect
 ADVANCED UNDERGRADUATE LABORATORY EXPERIMENT 20 Semiconductor Resistance, Band Gap, and Hall Effect Revised: November 1996 by David Bailey March 1990 by John Pitre & Taek-Soon Yoon Introduction Solid materials
ADVANCED UNDERGRADUATE LABORATORY EXPERIMENT 20 Semiconductor Resistance, Band Gap, and Hall Effect Revised: November 1996 by David Bailey March 1990 by John Pitre & Taek-Soon Yoon Introduction Solid materials
Mat E 272 Lecture 25: Electrical properties of materials
 Mat E 272 Lecture 25: Electrical properties of materials December 6, 2001 Introduction: Calcium and copper are both metals; Ca has a valence of +2 (2 electrons per atom) while Cu has a valence of +1 (1
Mat E 272 Lecture 25: Electrical properties of materials December 6, 2001 Introduction: Calcium and copper are both metals; Ca has a valence of +2 (2 electrons per atom) while Cu has a valence of +1 (1
A semiconductor is an almost insulating material, in which by contamination (doping) positive or negative charge carriers can be introduced.
 Semiconductor A semiconductor is an almost insulating material, in which by contamination (doping) positive or negative charge carriers can be introduced. Page 2 Semiconductor materials Page 3 Energy levels
Semiconductor A semiconductor is an almost insulating material, in which by contamination (doping) positive or negative charge carriers can be introduced. Page 2 Semiconductor materials Page 3 Energy levels
EE 346: Semiconductor Devices
 EE 346: Semiconductor Devices Lecture - 6 02/06/2017 Tewodros A. Zewde 1 DENSTY OF STATES FUNCTON Since current is due to the flow of charge, an important step in the process is to determine the number
EE 346: Semiconductor Devices Lecture - 6 02/06/2017 Tewodros A. Zewde 1 DENSTY OF STATES FUNCTON Since current is due to the flow of charge, an important step in the process is to determine the number
Ch. 2: Energy Bands And Charge Carriers In Semiconductors
 Ch. 2: Energy Bands And Charge Carriers In Semiconductors Discrete energy levels arise from balance of attraction force between electrons and nucleus and repulsion force between electrons each electron
Ch. 2: Energy Bands And Charge Carriers In Semiconductors Discrete energy levels arise from balance of attraction force between electrons and nucleus and repulsion force between electrons each electron
Semiconductors. SEM and EDAX images of an integrated circuit. SEM EDAX: Si EDAX: Al. Institut für Werkstoffe der ElektrotechnikIWE
 SEM and EDAX images of an integrated circuit SEM EDAX: Si EDAX: Al source: [Cal 99 / 605] M&D-.PPT, slide: 1, 12.02.02 Classification semiconductors electronic semiconductors mixed conductors ionic conductors
SEM and EDAX images of an integrated circuit SEM EDAX: Si EDAX: Al source: [Cal 99 / 605] M&D-.PPT, slide: 1, 12.02.02 Classification semiconductors electronic semiconductors mixed conductors ionic conductors
THE ELECTRICAL CONDUCTIVITY OF SILICON BETWEEN 500 C AND 1200 C
 R949 Philips Res. Repts 31,278-283,1976 THE ELECTRICAL CONDUCTIVITY OF SILICON BETWEEN 500 C AND 1200 C M. C. H. M. WOUTERS*) **), H. M. EIJKMAN*) and L. J. van RUYVEN*) Abstract The electrical conductivity
R949 Philips Res. Repts 31,278-283,1976 THE ELECTRICAL CONDUCTIVITY OF SILICON BETWEEN 500 C AND 1200 C M. C. H. M. WOUTERS*) **), H. M. EIJKMAN*) and L. J. van RUYVEN*) Abstract The electrical conductivity
The Electromagnetic Properties of Materials
 The lectromagnetic Properties of Materials lectrical conduction Metals Semiconductors Insulators (dielectrics) Superconductors Magnetic materials Ferromagnetic materials Others Photonic Materials (optical)
The lectromagnetic Properties of Materials lectrical conduction Metals Semiconductors Insulators (dielectrics) Superconductors Magnetic materials Ferromagnetic materials Others Photonic Materials (optical)
First-Hand Investigation: Modeling of Semiconductors
 perform an investigation to model the behaviour of semiconductors, including the creation of a hole or positive charge on the atom that has lost the electron and the movement of electrons and holes in
perform an investigation to model the behaviour of semiconductors, including the creation of a hole or positive charge on the atom that has lost the electron and the movement of electrons and holes in
Note that it is traditional to draw the diagram for semiconductors rotated 90 degrees, i.e. the version on the right above.
 5 Semiconductors The nearly free electron model applies equally in the case where the Fermi level lies within a small band gap (semiconductors), as it does when the Fermi level lies within a band (metal)
5 Semiconductors The nearly free electron model applies equally in the case where the Fermi level lies within a small band gap (semiconductors), as it does when the Fermi level lies within a band (metal)
The Semiconductor in Equilibrium
 Lecture 6 Semiconductor physics IV The Semiconductor in Equilibrium Equilibrium, or thermal equilibrium No external forces such as voltages, electric fields. Magnetic fields, or temperature gradients are
Lecture 6 Semiconductor physics IV The Semiconductor in Equilibrium Equilibrium, or thermal equilibrium No external forces such as voltages, electric fields. Magnetic fields, or temperature gradients are
THE IONIZATION ENERGY OF NITROGEN DONORS IN 6H AND 15R SiC
 VOL. 25 No. 1 FEBRUARY 1970 Philips Research Reports EDITED BY THE RESEARCH LABORATORY OF N.V. PHILIPS' GLOEILAMPENFABRIEKEN. EINDHOVEN. NETHERLANDS R711 Philips Res. Repts 25, 1-7, 1970 THE IONIZATION
VOL. 25 No. 1 FEBRUARY 1970 Philips Research Reports EDITED BY THE RESEARCH LABORATORY OF N.V. PHILIPS' GLOEILAMPENFABRIEKEN. EINDHOVEN. NETHERLANDS R711 Philips Res. Repts 25, 1-7, 1970 THE IONIZATION
Introduction into defect studies. in ceramic materials(iii) Structure, Defects and Defect Chemistry. Z. Wang. January 18, 2002
 Introduction into defect studies in ceramic materials(iii) Structure, Defects and Defect Chemistry Z. Wang January 18, 2002 1. Mass, Charge and Site Balance The Schottky reactions for NaCl and MgO, respectively,
Introduction into defect studies in ceramic materials(iii) Structure, Defects and Defect Chemistry Z. Wang January 18, 2002 1. Mass, Charge and Site Balance The Schottky reactions for NaCl and MgO, respectively,
OBSERVATION OF Se 77 SUPERHYPERFINE STRUCTURE ON THE ELECTRON-PARAMAGNETIC RESONANCE OF Fe3+ (3d S ) IN CUBIC ZnSe
 R540 Philips Res. Repts 20, 206-212, 1965 OBSERVATION OF Se 77 SUPERHYPERFINE STRUCTURE ON THE ELECTRON-PARAMAGNETIC RESONANCE OF Fe3+ (3d S ) IN CUBIC ZnSe by J. DIELEMAN Abstract The electron-paramagnetic-resonance
R540 Philips Res. Repts 20, 206-212, 1965 OBSERVATION OF Se 77 SUPERHYPERFINE STRUCTURE ON THE ELECTRON-PARAMAGNETIC RESONANCE OF Fe3+ (3d S ) IN CUBIC ZnSe by J. DIELEMAN Abstract The electron-paramagnetic-resonance
Materials and Devices in Electrical Engineering
 Examination WS 02/03 Materials and Devices in Electrical Engineering Monday 17 th of March, 9:00 11:00, International Department, SR. 203 Notice 1. It is allowed to use any kind of aids (books, scripts,
Examination WS 02/03 Materials and Devices in Electrical Engineering Monday 17 th of March, 9:00 11:00, International Department, SR. 203 Notice 1. It is allowed to use any kind of aids (books, scripts,
Density of states for electrons and holes. Distribution function. Conduction and valence bands
 Intrinsic Semiconductors In the field of semiconductors electrons and holes are usually referred to as free carriers, or simply carriers, because it is these particles which are responsible for carrying
Intrinsic Semiconductors In the field of semiconductors electrons and holes are usually referred to as free carriers, or simply carriers, because it is these particles which are responsible for carrying
CHEM1902/ N-2 November 2014
 CHEM1902/4 2014-N-2 November 2014 The cubic form of boron nitride (borazon) is the second-hardest material after diamond and it crystallizes with the structure shown below. The large spheres represent
CHEM1902/4 2014-N-2 November 2014 The cubic form of boron nitride (borazon) is the second-hardest material after diamond and it crystallizes with the structure shown below. The large spheres represent
Semiconductor Physics
 Semiconductor Physics Motivation Is it possible that there might be current flowing in a conductor (or a semiconductor) even when there is no potential difference supplied across its ends? Look at the
Semiconductor Physics Motivation Is it possible that there might be current flowing in a conductor (or a semiconductor) even when there is no potential difference supplied across its ends? Look at the
Electrical Properties
 Electrical Properties Electrical Conduction R Ohm s law V = IR I l Area, A V where I is current (Ampere), V is voltage (Volts) and R is the resistance (Ohms or ) of the conductor Resistivity Resistivity,
Electrical Properties Electrical Conduction R Ohm s law V = IR I l Area, A V where I is current (Ampere), V is voltage (Volts) and R is the resistance (Ohms or ) of the conductor Resistivity Resistivity,
R measurements (resistivity, magnetoresistance, Hall). Makariy A. Tanatar
 R measurements (resistivity, magnetoresistance, Hall). 590B Makariy A. Tanatar April 18, 2014 Resistivity Typical resistivity temperature dependence: metals, semiconductors Magnetic scattering Resistivities
R measurements (resistivity, magnetoresistance, Hall). 590B Makariy A. Tanatar April 18, 2014 Resistivity Typical resistivity temperature dependence: metals, semiconductors Magnetic scattering Resistivities
Classification of Solids, Fermi Level and Conductivity in Metals Dr. Anurag Srivastava
 Classification of Solids, Fermi Level and Conductivity in Metals Dr. Anurag Srivastava Web address: http://tiiciiitm.com/profanurag Email: profanurag@gmail.com Visit me: Room-110, Block-E, IIITM Campus
Classification of Solids, Fermi Level and Conductivity in Metals Dr. Anurag Srivastava Web address: http://tiiciiitm.com/profanurag Email: profanurag@gmail.com Visit me: Room-110, Block-E, IIITM Campus
MAGNETIC PROPERTIES OF OXIDIC SPINELS CONTAINING TETRAVALENT MANGANESE
 R490 Philips Res. Repts 18, 400-404, 196: MAGNETIC PROPERTIES OF OXIDIC SPINELS CONTAINING TETRAVALENT MANGANESE Abstract by G. BLASSE Measurements of the magnetic susceptibility as a function of the temperature
R490 Philips Res. Repts 18, 400-404, 196: MAGNETIC PROPERTIES OF OXIDIC SPINELS CONTAINING TETRAVALENT MANGANESE Abstract by G. BLASSE Measurements of the magnetic susceptibility as a function of the temperature
Transport and Magnetic Properties of La 0.85 Ca Mn 1-x Al x O 3 Manganites
 Asian Journal of Chemistry Vol. 21, No. 1 (29), S86-9 Transport and Magnetic Properties of La.85 Ca. 15 Mn 1-x Al x O 3 Manganites G ANJUM and S MOLLAH Department of Physics, Aligarh Muslim University,
Asian Journal of Chemistry Vol. 21, No. 1 (29), S86-9 Transport and Magnetic Properties of La.85 Ca. 15 Mn 1-x Al x O 3 Manganites G ANJUM and S MOLLAH Department of Physics, Aligarh Muslim University,
Chapter 1 Overview of Semiconductor Materials and Physics
 Chapter 1 Overview of Semiconductor Materials and Physics Professor Paul K. Chu Conductivity / Resistivity of Insulators, Semiconductors, and Conductors Semiconductor Elements Period II III IV V VI 2 B
Chapter 1 Overview of Semiconductor Materials and Physics Professor Paul K. Chu Conductivity / Resistivity of Insulators, Semiconductors, and Conductors Semiconductor Elements Period II III IV V VI 2 B
CH676 Physical Chemistry: Principles and Applications. CH676 Physical Chemistry: Principles and Applications
 CH676 Physical Chemistry: Principles and Applications Band Theory Fermi-Dirac Function f(e) = 1/[1 + e (E-E F)/kT ] Where the Fermi Energy, E F, is defined as the energy where f(e) = 1/2. That is to say
CH676 Physical Chemistry: Principles and Applications Band Theory Fermi-Dirac Function f(e) = 1/[1 + e (E-E F)/kT ] Where the Fermi Energy, E F, is defined as the energy where f(e) = 1/2. That is to say
SEMICONDUCTOR DIODE. Unbiased (non-polarized) PN junction
 SEMICONDUCTOR DIODE Semiconductor diode is an electronic element made of different types of extrinsic semiconductor: N-type semiconductor doped by donor impurities and P-type semiconductor doped by acceptor
SEMICONDUCTOR DIODE Semiconductor diode is an electronic element made of different types of extrinsic semiconductor: N-type semiconductor doped by donor impurities and P-type semiconductor doped by acceptor
4. Interpenetrating simple cubic
 2 1. The correct structure t of CsClCl crystal is 1. Simple cubic 2. Body centered cubic 3. Face centered cubic 4. Interpenetrating simple cubic If corner as well as the particle at the center are same
2 1. The correct structure t of CsClCl crystal is 1. Simple cubic 2. Body centered cubic 3. Face centered cubic 4. Interpenetrating simple cubic If corner as well as the particle at the center are same
EXTRINSIC SEMICONDUCTOR
 EXTRINSIC SEMICONDUCTOR In an extrinsic semiconducting material, the charge carriers originate from impurity atoms added to the original material is called impurity [or] extrinsic semiconductor. This Semiconductor
EXTRINSIC SEMICONDUCTOR In an extrinsic semiconducting material, the charge carriers originate from impurity atoms added to the original material is called impurity [or] extrinsic semiconductor. This Semiconductor
ELECTRONIC I Lecture 1 Introduction to semiconductor. By Asst. Prof Dr. Jassim K. Hmood
 ELECTRONIC I Lecture 1 Introduction to semiconductor By Asst. Prof Dr. Jassim K. Hmood SOLID-STATE ELECTRONIC MATERIALS Electronic materials generally can be divided into three categories: insulators,
ELECTRONIC I Lecture 1 Introduction to semiconductor By Asst. Prof Dr. Jassim K. Hmood SOLID-STATE ELECTRONIC MATERIALS Electronic materials generally can be divided into three categories: insulators,
Materials and Devices in Electrical Engineering
 Examination WS 01/02 Materials and Devices in Electrical Engineering Monday 11 th of March, 9:00 to 11:00, SR 203, International Department building It is allowed to use any kind of media (books, scripts,
Examination WS 01/02 Materials and Devices in Electrical Engineering Monday 11 th of March, 9:00 to 11:00, SR 203, International Department building It is allowed to use any kind of media (books, scripts,
 lectures accompanying the book: Solid State Physics: An Introduction, by Philip ofmann (2nd edition 2015, ISBN-10: 3527412824, ISBN-13: 978-3527412822, Wiley-VC Berlin. www.philiphofmann.net 1 Bonds between
lectures accompanying the book: Solid State Physics: An Introduction, by Philip ofmann (2nd edition 2015, ISBN-10: 3527412824, ISBN-13: 978-3527412822, Wiley-VC Berlin. www.philiphofmann.net 1 Bonds between
Crystal Properties. MS415 Lec. 2. High performance, high current. ZnO. GaN
 Crystal Properties Crystal Lattices: Periodic arrangement of atoms Repeated unit cells (solid-state) Stuffing atoms into unit cells Determine mechanical & electrical properties High performance, high current
Crystal Properties Crystal Lattices: Periodic arrangement of atoms Repeated unit cells (solid-state) Stuffing atoms into unit cells Determine mechanical & electrical properties High performance, high current
Electro - Principles I
 Electro - Principles I Page 10-1 Atomic Theory It is necessary to know what goes on at the atomic level of a semiconductor so the characteristics of the semiconductor can be understood. In many cases a
Electro - Principles I Page 10-1 Atomic Theory It is necessary to know what goes on at the atomic level of a semiconductor so the characteristics of the semiconductor can be understood. In many cases a
Self-compensating incorporation of Mn in Ga 1 x Mn x As
 Self-compensating incorporation of Mn in Ga 1 x Mn x As arxiv:cond-mat/0201131v1 [cond-mat.mtrl-sci] 9 Jan 2002 J. Mašek and F. Máca Institute of Physics, Academy of Sciences of the CR CZ-182 21 Praha
Self-compensating incorporation of Mn in Ga 1 x Mn x As arxiv:cond-mat/0201131v1 [cond-mat.mtrl-sci] 9 Jan 2002 J. Mašek and F. Máca Institute of Physics, Academy of Sciences of the CR CZ-182 21 Praha
EXTRINSIC SEMICONDUCTOR
 EXTRINSIC SEMICONDUCTOR EXTRINSIC SEMICONDUCTOR A semiconductor in which the impurity atoms are added by doping process is called Extrinsic semiconductor. The addition of impurities increases the carrier
EXTRINSIC SEMICONDUCTOR EXTRINSIC SEMICONDUCTOR A semiconductor in which the impurity atoms are added by doping process is called Extrinsic semiconductor. The addition of impurities increases the carrier
Introduction to Semiconductor Physics. Prof.P. Ravindran, Department of Physics, Central University of Tamil Nadu, India
 Introduction to Semiconductor Physics 1 Prof.P. Ravindran, Department of Physics, Central University of Tamil Nadu, India http://folk.uio.no/ravi/cmp2013 Review of Semiconductor Physics Semiconductor fundamentals
Introduction to Semiconductor Physics 1 Prof.P. Ravindran, Department of Physics, Central University of Tamil Nadu, India http://folk.uio.no/ravi/cmp2013 Review of Semiconductor Physics Semiconductor fundamentals
CLASS 12th. Semiconductors
 CLASS 12th Semiconductors 01. Distinction Between Metals, Insulators and Semi-Conductors Metals are good conductors of electricity, insulators do not conduct electricity, while the semiconductors have
CLASS 12th Semiconductors 01. Distinction Between Metals, Insulators and Semi-Conductors Metals are good conductors of electricity, insulators do not conduct electricity, while the semiconductors have
Review of Semiconductor Fundamentals
 ECE 541/ME 541 Microelectronic Fabrication Techniques Review of Semiconductor Fundamentals Zheng Yang (ERF 3017, email: yangzhen@uic.edu) Page 1 Semiconductor A semiconductor is an almost insulating material,
ECE 541/ME 541 Microelectronic Fabrication Techniques Review of Semiconductor Fundamentals Zheng Yang (ERF 3017, email: yangzhen@uic.edu) Page 1 Semiconductor A semiconductor is an almost insulating material,
V = IR or R = V I. R = ρ l A
 Metals and Semiconductors Ram Seshadri MRL 2031, x6129, seshadri@mrl.ucsb.edu Electrical resistance and Ohm s Law: If an electric current I (units of A, Ampère) flows through a conductor with resistance
Metals and Semiconductors Ram Seshadri MRL 2031, x6129, seshadri@mrl.ucsb.edu Electrical resistance and Ohm s Law: If an electric current I (units of A, Ampère) flows through a conductor with resistance
Final Exam, MENA3000 / MENA4000 Functional Materials, 6 th June 2016
 Final Exam, MENA3000 / MENA4000 Functional Materials, 6 th June 2016 Task 1 Crystal structure, chemical bonding and non-stoichiometry (25 %) For one of the modifications of molybdenum disulphide, MoS 2,
Final Exam, MENA3000 / MENA4000 Functional Materials, 6 th June 2016 Task 1 Crystal structure, chemical bonding and non-stoichiometry (25 %) For one of the modifications of molybdenum disulphide, MoS 2,
Magnetic Oxides. Gerald F. Dionne. Department of Materials Science and Engineering Massachusetts Institute of Technology
 Magnetic Oxides Gerald F. Dionne Department of Materials Science and Engineering Massachusetts Institute of Technology Spins in Solids Summer School University of Virginia Charlottesville, VA 21 June 2006
Magnetic Oxides Gerald F. Dionne Department of Materials Science and Engineering Massachusetts Institute of Technology Spins in Solids Summer School University of Virginia Charlottesville, VA 21 June 2006
What so special about LaAlO3/SrTiO3 interface? Magnetism, Superconductivity and their coexistence at the interface
 What so special about LaAlO3/SrTiO3 interface? Magnetism, Superconductivity and their coexistence at the interface Pramod Verma Indian Institute of Science, Bangalore 560012 July 24, 2014 Pramod Verma
What so special about LaAlO3/SrTiO3 interface? Magnetism, Superconductivity and their coexistence at the interface Pramod Verma Indian Institute of Science, Bangalore 560012 July 24, 2014 Pramod Verma
SEMICONDUCTOR PHYSICS
 SEMICONDUCTOR PHYSICS by Dibyendu Chowdhury Semiconductors The materials whose electrical conductivity lies between those of conductors and insulators, are known as semiconductors. Silicon Germanium Cadmium
SEMICONDUCTOR PHYSICS by Dibyendu Chowdhury Semiconductors The materials whose electrical conductivity lies between those of conductors and insulators, are known as semiconductors. Silicon Germanium Cadmium
CHAPTER 5 CATALYTIC OXIDATION OF CARBON MONOXIDE
 CHAPTER 5 CATALYTIC OXIDATION OF CARBON MONOXIDE CATALYTIC OXIDATION OF CARBON MONOXIDE The transition metal oxidic perovskites provide an excellent base for correlating catalytic and solid state properties,
CHAPTER 5 CATALYTIC OXIDATION OF CARBON MONOXIDE CATALYTIC OXIDATION OF CARBON MONOXIDE The transition metal oxidic perovskites provide an excellent base for correlating catalytic and solid state properties,
Phase Diagram of Spin States and Magnetic Interactions in Isotope Substituted (Pr,Eu) 0.7 Ca 0.3 CoO 3
 Solid State Phenomena Vols. 168-169 (2011) pp 465-468 Online available since 2010/Dec/30 at www.scientific.net (2011) Trans Tech Publications, Switzerland doi:10.4028/www.scientific.net/ssp.168-169.465
Solid State Phenomena Vols. 168-169 (2011) pp 465-468 Online available since 2010/Dec/30 at www.scientific.net (2011) Trans Tech Publications, Switzerland doi:10.4028/www.scientific.net/ssp.168-169.465
Diamond. Covalent Insulators and Semiconductors. Silicon, Germanium, Gray Tin. Chem 462 September 24, 2004
 Covalent Insulators and Chem 462 September 24, 2004 Diamond Pure sp 3 carbon All bonds staggered- ideal d(c-c) - 1.54 Å, like ethane Silicon, Germanium, Gray Tin Diamond structure Si and Ge: semiconductors
Covalent Insulators and Chem 462 September 24, 2004 Diamond Pure sp 3 carbon All bonds staggered- ideal d(c-c) - 1.54 Å, like ethane Silicon, Germanium, Gray Tin Diamond structure Si and Ge: semiconductors
Electronic Processes on Semiconductor Surfaces during Chemisorption
 Electronic Processes on Semiconductor Surfaces during Chemisorption T. Wolkenstein Translatedfrom Russian by E. M. Yankovskii Translation edited in part by Roy Morrison CONSULTANTS BUREAU NEW YORK AND
Electronic Processes on Semiconductor Surfaces during Chemisorption T. Wolkenstein Translatedfrom Russian by E. M. Yankovskii Translation edited in part by Roy Morrison CONSULTANTS BUREAU NEW YORK AND
Chapter 6 ELECTRICAL CONDUCTIVITY ANALYSIS
 Chapter 6 ELECTRICAL CONDUCTIVITY ANALYSIS CHAPTER-6 6.1 Introduction The suitability and potentiality of a material for device applications can be determined from the frequency and temperature response
Chapter 6 ELECTRICAL CONDUCTIVITY ANALYSIS CHAPTER-6 6.1 Introduction The suitability and potentiality of a material for device applications can be determined from the frequency and temperature response
PH575 Spring Lecture #19 Semiconductors: electrical & optical properties: Kittel Ch. 8 pp ; Ch. 20
 PH575 Spring 2014 Lecture #19 Semiconductors: electrical & optical properties: Kittel Ch. 8 pp. 205-214; Ch. 20 Simplified diagram of the filling of electronic band structure in various types of material,
PH575 Spring 2014 Lecture #19 Semiconductors: electrical & optical properties: Kittel Ch. 8 pp. 205-214; Ch. 20 Simplified diagram of the filling of electronic band structure in various types of material,
VERY SHORT ANSWER TYPE QUESTIONS (1 Mark)
 UNIT I 10 Chemistry-XII THE SOLID STATE VERY SHORT ANSWER TYPE QUESTIONS (1 Mark) Q. 1. What do you mean by paramagnetic substance? Ans. Weakly attracted by magnetic eld and these substances are made of
UNIT I 10 Chemistry-XII THE SOLID STATE VERY SHORT ANSWER TYPE QUESTIONS (1 Mark) Q. 1. What do you mean by paramagnetic substance? Ans. Weakly attracted by magnetic eld and these substances are made of
Minimal Update of Solid State Physics
 Minimal Update of Solid State Physics It is expected that participants are acquainted with basics of solid state physics. Therefore here we will refresh only those aspects, which are absolutely necessary
Minimal Update of Solid State Physics It is expected that participants are acquainted with basics of solid state physics. Therefore here we will refresh only those aspects, which are absolutely necessary
Lecture 2. Semiconductor Physics. Sunday 4/10/2015 Semiconductor Physics 1-1
 Lecture 2 Semiconductor Physics Sunday 4/10/2015 Semiconductor Physics 1-1 Outline Intrinsic bond model: electrons and holes Charge carrier generation and recombination Intrinsic semiconductor Doping:
Lecture 2 Semiconductor Physics Sunday 4/10/2015 Semiconductor Physics 1-1 Outline Intrinsic bond model: electrons and holes Charge carrier generation and recombination Intrinsic semiconductor Doping:
Carriers Concentration in Semiconductors - V. Prof.P. Ravindran, Department of Physics, Central University of Tamil Nadu, India
 Carriers Concentration in Semiconductors - V 1 Prof.P. Ravindran, Department of Physics, Central University of Tamil Nadu, India http://folk.uio.no/ravi/semi2013 Motion and Recombination of Electrons and
Carriers Concentration in Semiconductors - V 1 Prof.P. Ravindran, Department of Physics, Central University of Tamil Nadu, India http://folk.uio.no/ravi/semi2013 Motion and Recombination of Electrons and
Atomic Structure & Interatomic Bonding
 Atomic Structure & Interatomic Bonding Chapter Outline Review of Atomic Structure Atomic Bonding Atomic Structure Atoms are the smallest structural units of all solids, liquids & gases. Atom: The smallest
Atomic Structure & Interatomic Bonding Chapter Outline Review of Atomic Structure Atomic Bonding Atomic Structure Atoms are the smallest structural units of all solids, liquids & gases. Atom: The smallest
KJM5120 and KJM9120 Defects and Reactions
 KJM510 and KJM910 Defects and Reactions Truls Norby Ch. Defect reactions Department of Chemistry University of slo Centre for Materials Science and Nanotechnology (SMN) = v / 1 ) + + e ( g ) FERMI slo
KJM510 and KJM910 Defects and Reactions Truls Norby Ch. Defect reactions Department of Chemistry University of slo Centre for Materials Science and Nanotechnology (SMN) = v / 1 ) + + e ( g ) FERMI slo
EECS130 Integrated Circuit Devices
 EECS130 Integrated Circuit Devices Professor Ali Javey 8/30/2007 Semiconductor Fundamentals Lecture 2 Read: Chapters 1 and 2 Last Lecture: Energy Band Diagram Conduction band E c E g Band gap E v Valence
EECS130 Integrated Circuit Devices Professor Ali Javey 8/30/2007 Semiconductor Fundamentals Lecture 2 Read: Chapters 1 and 2 Last Lecture: Energy Band Diagram Conduction band E c E g Band gap E v Valence
HALL. Semiconductor Resistance, Band Gap, and Hall Effect
 ADVANCED UNDERGRADUATE LABORATORY HALL Semiconductor Resistance, Band Gap, and Hall Effect Revisions: September 2016, January 2018: Young-June Kim November 2011, January 2016: David Bailey October 2010:
ADVANCED UNDERGRADUATE LABORATORY HALL Semiconductor Resistance, Band Gap, and Hall Effect Revisions: September 2016, January 2018: Young-June Kim November 2011, January 2016: David Bailey October 2010:
BOUND EXCITON LUMINESCENCE IN PHOSPHORUS DOPED Cd1- xmn xte CRYSTALS
 Vol. 94 (1998) ACTA PHYSICA POLONICA A Νo. 3 Proc. of the XXVII Intern. School on Physics of Semiconducting Compounds, Jaszowiec 1998 BOUND EXCITON LUMINESCENCE IN PHOSPHORUS DOPED Cd1- xmn xte CRYSTALS
Vol. 94 (1998) ACTA PHYSICA POLONICA A Νo. 3 Proc. of the XXVII Intern. School on Physics of Semiconducting Compounds, Jaszowiec 1998 BOUND EXCITON LUMINESCENCE IN PHOSPHORUS DOPED Cd1- xmn xte CRYSTALS
Chapter 4: Bonding in Solids and Electronic Properties. Free electron theory
 Chapter 4: Bonding in Solids and Electronic Properties Free electron theory Consider free electrons in a metal an electron gas. regards a metal as a box in which electrons are free to move. assumes nuclei
Chapter 4: Bonding in Solids and Electronic Properties Free electron theory Consider free electrons in a metal an electron gas. regards a metal as a box in which electrons are free to move. assumes nuclei
Semiconductors. Semiconductors also can collect and generate photons, so they are important in optoelectronic or photonic applications.
 Semiconductors Semiconducting materials have electrical properties that fall between true conductors, (like metals) which are always highly conducting and insulators (like glass or plastic or common ceramics)
Semiconductors Semiconducting materials have electrical properties that fall between true conductors, (like metals) which are always highly conducting and insulators (like glass or plastic or common ceramics)
Micron School of Materials Science and Engineering. Problem Set 9 Solutions
 Problem Set 9 Solutions 1. Mobility in extrinsic semiconductors is affected by phonon scattering and impurity scattering. Thoroughly explain the mobility plots for the following figures from your textbook
Problem Set 9 Solutions 1. Mobility in extrinsic semiconductors is affected by phonon scattering and impurity scattering. Thoroughly explain the mobility plots for the following figures from your textbook
CHAPTER 18: Electrical properties
 CHAPTER 18: Electrical properties ISSUES TO ADDRESS... How are electrical conductance and resistance characterized? What are the physical phenomena that distinguish conductors, semiconductors, and insulators?
CHAPTER 18: Electrical properties ISSUES TO ADDRESS... How are electrical conductance and resistance characterized? What are the physical phenomena that distinguish conductors, semiconductors, and insulators?
V, I, R measurements: how to generate and measure quantities and then how to get data (resistivity, magnetoresistance, Hall). Makariy A.
 V, I, R measurements: how to generate and measure quantities and then how to get data (resistivity, magnetoresistance, Hall). 590B Makariy A. Tanatar November 12, 2008 Resistivity Typical resistivity temperature
V, I, R measurements: how to generate and measure quantities and then how to get data (resistivity, magnetoresistance, Hall). 590B Makariy A. Tanatar November 12, 2008 Resistivity Typical resistivity temperature
LN 3 IDLE MIND SOLUTIONS
 IDLE MIND SOLUTIONS 1. Let us first look in most general terms at the optical properties of solids with band gaps (E g ) of less than 4 ev, semiconductors by definition. The band gap energy (E g ) can
IDLE MIND SOLUTIONS 1. Let us first look in most general terms at the optical properties of solids with band gaps (E g ) of less than 4 ev, semiconductors by definition. The band gap energy (E g ) can
EE301 Electronics I , Fall
 EE301 Electronics I 2018-2019, Fall 1. Introduction to Microelectronics (1 Week/3 Hrs.) Introduction, Historical Background, Basic Consepts 2. Rewiev of Semiconductors (1 Week/3 Hrs.) Semiconductor materials
EE301 Electronics I 2018-2019, Fall 1. Introduction to Microelectronics (1 Week/3 Hrs.) Introduction, Historical Background, Basic Consepts 2. Rewiev of Semiconductors (1 Week/3 Hrs.) Semiconductor materials
High T C copper oxide superconductors and CMR:
 High T C copper oxide superconductors and CMR: Ram Seshadri (seshadri@mrl.ucsb.edu) The Ruddlesden-Popper phases: Ruddlesden-Popper phases are intergrowths of perovskite slabs with rock salt slabs. First
High T C copper oxide superconductors and CMR: Ram Seshadri (seshadri@mrl.ucsb.edu) The Ruddlesden-Popper phases: Ruddlesden-Popper phases are intergrowths of perovskite slabs with rock salt slabs. First
Lecture 12 Multiplet splitting
 Lecture 12 Multiplet splitting Multiplet splitting Atomic various L and S terms Both valence and core levels Rare earths Transition metals Paramagnetic free molecules Consider 3s level emission from Mn2+
Lecture 12 Multiplet splitting Multiplet splitting Atomic various L and S terms Both valence and core levels Rare earths Transition metals Paramagnetic free molecules Consider 3s level emission from Mn2+
Engineering 2000 Chapter 8 Semiconductors. ENG2000: R.I. Hornsey Semi: 1
 Engineering 2000 Chapter 8 Semiconductors ENG2000: R.I. Hornsey Semi: 1 Overview We need to know the electrical properties of Si To do this, we must also draw on some of the physical properties and we
Engineering 2000 Chapter 8 Semiconductors ENG2000: R.I. Hornsey Semi: 1 Overview We need to know the electrical properties of Si To do this, we must also draw on some of the physical properties and we
Due to the quantum nature of electrons, one energy state can be occupied only by one electron.
 In crystalline solids, not all values of the electron energy are possible. The allowed intervals of energy are called allowed bands (shown as blue and chess-board blue). The forbidden intervals are called
In crystalline solids, not all values of the electron energy are possible. The allowed intervals of energy are called allowed bands (shown as blue and chess-board blue). The forbidden intervals are called
4.1 Atomic structure and the periodic table. GCSE Chemistry
 4.1 Atomic structure and the periodic table GCSE Chemistry All substances are made of atoms this is cannot be chemically broken down it is the smallest part of an element. Elements are made of only one
4.1 Atomic structure and the periodic table GCSE Chemistry All substances are made of atoms this is cannot be chemically broken down it is the smallest part of an element. Elements are made of only one
* motif: a single or repeated design or color
 Chapter 2. Structure A. Electronic structure vs. Geometric structure B. Clean surface vs. Adsorbate covered surface (substrate + overlayer) C. Adsorbate structure - how are the adsorbed molecules bound
Chapter 2. Structure A. Electronic structure vs. Geometric structure B. Clean surface vs. Adsorbate covered surface (substrate + overlayer) C. Adsorbate structure - how are the adsorbed molecules bound
Understanding Solid State Physics Additional Questions. Sharon Ann Holgate
 Understanding Solid State Physics Additional Questions Sharon Ann Holgate Questions for Chapter 2 2.1(a) What structure does the compound caesium iodide crystallise in? 2.1(b) What does the term "coordination
Understanding Solid State Physics Additional Questions Sharon Ann Holgate Questions for Chapter 2 2.1(a) What structure does the compound caesium iodide crystallise in? 2.1(b) What does the term "coordination
3. Consider a semiconductor. The concentration of electrons, n, in the conduction band is given by
 Colloqium problems to chapter 13 1. What is meant by an intrinsic semiconductor? n = p All the electrons are originating from thermal excitation from the valence band for an intrinsic semiconductor. Then
Colloqium problems to chapter 13 1. What is meant by an intrinsic semiconductor? n = p All the electrons are originating from thermal excitation from the valence band for an intrinsic semiconductor. Then
Advantages / Disadvantages of semiconductor detectors
 Advantages / Disadvantages of semiconductor detectors Semiconductor detectors have a high density (compared to gas detector) large energy loss in a short distance diffusion effect is smaller than in gas
Advantages / Disadvantages of semiconductor detectors Semiconductor detectors have a high density (compared to gas detector) large energy loss in a short distance diffusion effect is smaller than in gas
2.1 Experimental and theoretical studies
 Chapter 2 NiO As stated before, the first-row transition-metal oxides are among the most interesting series of materials, exhibiting wide variations in physical properties related to electronic structure.
Chapter 2 NiO As stated before, the first-row transition-metal oxides are among the most interesting series of materials, exhibiting wide variations in physical properties related to electronic structure.
The German University in Cairo. Faculty of Information Engineering & Technology Semiconductors (Elct 503) Electronics Department Fall 2014
 The German University in Cairo th Electronics 5 Semester Faculty of Information Engineering & Technology Semiconductors (Elct 503) Electronics Department Fall 2014 Problem Set 3 1- a) Find the resistivity
The German University in Cairo th Electronics 5 Semester Faculty of Information Engineering & Technology Semiconductors (Elct 503) Electronics Department Fall 2014 Problem Set 3 1- a) Find the resistivity
Transport properties of composition tuned - and -Eu 8 Ga 16 x Ge 30+x
 Transport properties of composition tuned - and -Eu 8 Ga 16 x Ge 30+x A. Bentien, V. Pacheco,* S. Paschen, Yu. Grin, and F. Steglich Max Planck Institute for Chemical Physics of Solids, Nöthnitzer Str.
Transport properties of composition tuned - and -Eu 8 Ga 16 x Ge 30+x A. Bentien, V. Pacheco,* S. Paschen, Yu. Grin, and F. Steglich Max Planck Institute for Chemical Physics of Solids, Nöthnitzer Str.
ECE 340 Lecture 6 : Intrinsic and Extrinsic Material I Class Outline:
 ECE 340 Lecture 6 : Intrinsic and Extrinsic Material I Class Outline: Effective Mass Intrinsic Material Extrinsic Material Things you should know when you leave Key Questions What is the physical meaning
ECE 340 Lecture 6 : Intrinsic and Extrinsic Material I Class Outline: Effective Mass Intrinsic Material Extrinsic Material Things you should know when you leave Key Questions What is the physical meaning
SYED AMMAL ENGINEERING COLLEGE: RAMANATHAPURAM Dr.E.M.Abdullah Campus DEPARTMENT OF PHYSICS Question Bank Engineering physics II PH6251 (R-2013)
 SYED AMMAL ENGINEERING COLLEGE: RAMANATHAPURAM Dr.E.M.Abdullah Campus DEPARTMENT OF PHYSICS Question Bank Engineering physics II PH6251 (R-2013) PART A UNIT-I Conducting Materials 1. What are the classifications
SYED AMMAL ENGINEERING COLLEGE: RAMANATHAPURAM Dr.E.M.Abdullah Campus DEPARTMENT OF PHYSICS Question Bank Engineering physics II PH6251 (R-2013) PART A UNIT-I Conducting Materials 1. What are the classifications
Colossal magnetoresistance:
 Colossal magnetoresistance: Ram Seshadri (seshadri@mrl.ucsb.edu) The simplest example of magnetoresistance is transverse magnetoresistance associated with the Hall effect: H + + + + + + + + + + E y - -
Colossal magnetoresistance: Ram Seshadri (seshadri@mrl.ucsb.edu) The simplest example of magnetoresistance is transverse magnetoresistance associated with the Hall effect: H + + + + + + + + + + E y - -
1. Following Dalton s Atomic Theory, 2. In 1869 Russian chemist published a method. of organizing the elements. Mendeleev showed that
 20 CHEMISTRY 11 D. Organizing the Elements The Periodic Table 1. Following Dalton s Atomic Theory, By 1817, chemists had discovered 52 elements and by 1863 that number had risen to 62. 2. In 1869 Russian
20 CHEMISTRY 11 D. Organizing the Elements The Periodic Table 1. Following Dalton s Atomic Theory, By 1817, chemists had discovered 52 elements and by 1863 that number had risen to 62. 2. In 1869 Russian
Processing of Semiconducting Materials Prof. Pallab Banerji Department of Material Science Indian Institute of Technology, Kharagpur
 Processing of Semiconducting Materials Prof. Pallab Banerji Department of Material Science Indian Institute of Technology, Kharagpur Lecture - 4 Doping in Semiconductors Good morning. Let us start with
Processing of Semiconducting Materials Prof. Pallab Banerji Department of Material Science Indian Institute of Technology, Kharagpur Lecture - 4 Doping in Semiconductors Good morning. Let us start with
Lecture 2 Electrons and Holes in Semiconductors
 EE 471: Transport Phenomena in Solid State Devices Spring 2018 Lecture 2 Electrons and Holes in Semiconductors Bryan Ackland Department of Electrical and Computer Engineering Stevens Institute of Technology
EE 471: Transport Phenomena in Solid State Devices Spring 2018 Lecture 2 Electrons and Holes in Semiconductors Bryan Ackland Department of Electrical and Computer Engineering Stevens Institute of Technology
Molecules and Condensed Matter
 Chapter 42 Molecules and Condensed Matter PowerPoint Lectures for University Physics, Thirteenth Edition Hugh D. Young and Roger A. Freedman Lectures by Wayne Anderson Goals for Chapter 42 To understand
Chapter 42 Molecules and Condensed Matter PowerPoint Lectures for University Physics, Thirteenth Edition Hugh D. Young and Roger A. Freedman Lectures by Wayne Anderson Goals for Chapter 42 To understand
materials and their properties
 materials and their properties macroscopic properties phase state strength / stiffness electrical conductivity chemical properties color / transparence spectroscopical properties surface properties density
materials and their properties macroscopic properties phase state strength / stiffness electrical conductivity chemical properties color / transparence spectroscopical properties surface properties density
Chemistry Institute B6, SUPRATECS, University of Liège, Sart-Tilman, B-4000 Liège, Belgium b
 Synthesis and characterization of Bi 2 Sr 2 CaCu 2 O 8 ceramics prepared in presence of sodium S. Rahier a*, S. Stassen a, R. Cloots a and M. Ausloos b a Chemistry Institute B6, SUPRATECS, University of
Synthesis and characterization of Bi 2 Sr 2 CaCu 2 O 8 ceramics prepared in presence of sodium S. Rahier a*, S. Stassen a, R. Cloots a and M. Ausloos b a Chemistry Institute B6, SUPRATECS, University of
Electrical Resistance
 Electrical Resistance I + V _ W Material with resistivity ρ t L Resistance R V I = L ρ Wt (Unit: ohms) where ρ is the electrical resistivity 1 Adding parts/billion to parts/thousand of dopants to pure
Electrical Resistance I + V _ W Material with resistivity ρ t L Resistance R V I = L ρ Wt (Unit: ohms) where ρ is the electrical resistivity 1 Adding parts/billion to parts/thousand of dopants to pure
