EE201/MSE207 Lecture 10 Angular momentum
|
|
|
- Gerard Fletcher
- 5 years ago
- Views:
Transcription
1 EE20/MSE207 Lecture 0 Angular momentum (A lot of mathematics; I will try to explain as simple as possile) Classical mechanics r L = r p (angular momentum) Quantum mechanics The same, ut p x = iħ x p = m v L x = y p z z p y L y = z p x x p z L z = x p y y p x Therefore L x = iħ y z z y z x y p y = iħ y L y = iħ z x x z p z = iħ z L z = iħ x y y x To find quantized values of angular momentum, we can solve 3D differential equations to find eigenvalues and eigenstates. However, there is a simpler way, similar to the trick for oscillator.
2 Commutation relations Simple to show: (check yourself) L x, L y = iħ L z L y, L z = iħ L x L z, L x = iħ L y Most of the theory of angular momentum (and spin) can e derived from these commutation relations. Will not write hats elow for revity First oservation: L x, L y, L z are all incompatile (do not commute), so if L x has a definite value, then L y and L z do not have definite values (except when all are zero, σ Lx σ Ly ħ L 2 z ). However, L 2 = L 2 x + L 2 y + L 2 z commutes with each component. Simple to show: L 2, L x = 0 L 2, L y = 0 L 2, L = 0 L 2, L z = 0 Therefore, L 2 (square of total angular momentum) and one component can have definite values at the same time (usually z-component is chosen)
3 Eigenvalues of L 2 and L z We want to find eigenvalues of operators L 2 and L z (they are the quantized values for the angular momentum). Need to solve: L 2 f = λ f L z f = μ f (f is not very important) Trick of ladder operators Let us introduce operators Will show (next goal): L ± = L x ± il y λ = ħ 2 l l + μ = ħ m l = 0, 2,, 3 2, m = l, l +, l These operators are not Hermitian (unphysical), while L x, L y, L z are of course Hermitian [L z, L ± ] = L z, L x ± i L x, L y = iħl y ± i iħ L x = ±ħl ± Thus L z, L ± = ±ħ L ± [L 2, L ± ] = 0
4 Lemma If f is an eigenvector of oth L 2 (with eigenvalue λ) and L z (with eigenvalue μ), then L ± f are also eigenvectors, with the same λ, ut μ μ ± ħ. Proof L 2 L ± f = L ± L 2 f = λl ± f L z, L ± = ±ħ L ± [L 2, L ± ] = 0 L z L ± f = L z, L ± f + L ± L z f = ±ħl ± f + μl ± f = μ ± ħ L ± f Now the same logic as for oscillator: apply L + many times, the process should stop at some point (since L z 2 cannot exceed L 2 ), therefore L + f top = 0. L z f top = ħlf top (so far l is aritrary) f top (L + ) 2 f L + f f L 2 f top = λf top Let us show λ = ħ 2 l(l + ) (eigenvalues are related!) Use formula L 2 = L L + + L z 2 + ħl z (simple to derive explicitly), then λ f top = ħl 2 f top + ħ 2 lf top = ħ 2 l l + f top
5 Similarly, applying L many times, we should eventually get L f ottom = 0. Then similarly L z f ottom = ħl f ottom (with aritrary l ), and can show the relation etween eigenvalues: λ = ħ 2 l (l ) Comparing these two equations, we otain l = l. But they are related y integer numer (integer numer of applying L ± ). Therefore, l is integer or half-integer. Thus, result: Eigenvalues of L 2 (i.e., possile measured values) are λ = ħ 2 l(l + ), where l = 0,,, 3, 2,, 2 2 eigenvalues of L z (i.e., possile measured values) are μ = ħ m, where m = l, l +, l, l If we denote the corresponding eigenvectors y f l m, then L 2 f l m = ħ 2 l l + f l m L z f l m = ħm f l m Interesting that L z 2 ħ 2 m 2 ħ 2 l 2 is always smaller than L 2 ħ 2 l l +. So, z-component cannot e parallel to L. (If it would, then L x = L y = 0, impossile.)
6 Eigenvectors of L 2 and L z We found eigenvalues of L 2 and L z : What are the eigenvectors f l m? L 2 f l m = ħ 2 l l + f l m L z f l m = ħm f l m This is not simple. Discuss only the result (not the derivation). If the eigenvector f l m is represented y ψ-function in real space, ψ(r, θ, φ), then l is integer (not half-integer) and is the eigenfunction, L 2 ψ = ħ 2 l l + ψ L z ψ = ħm ψ ψ r, θ, φ = R r Y l m (θ, φ) Thus, azimuthal quantum numer l means that square of the total angular momentum is L 2 = ħ 2 l(l + ) (s, p, d, f oritals). Magnetic quantum numer m means that L z = ħm (degeneracy 2l + ). any However, half-integer l values really exist: spin (consider next). spherical harmonics
7 Spin Existence of half-integer values of l is experimental fact (Stern-Gerlach, 922: neutral silver atoms travel though inhomogeneous magnets field, split into two eams) Cannot explain as ψ(r, θ, φ) something else. It is an angular momentum, which does not correspond to motion in (x, y, z) intrinsic angular momentum (spin). The theory of spin is essentially the same as theory of angular momentum, just no ψ(r, θ, φ), only operator formalism. S x, S y S y, S z S z, S x Total spin: = iħ S z = iħ S x = iħ S y Useful relation: s = 0, 2,, 3 2, Denote eigenstates of S 2 and S z as s, m This is jargon, actually More correctly, z-component: m = s, s +, s total spin z-component S 2 = ħ s(s + ), S z = ħm S 2 s, m = ħ 2 s s + s, m S z s, m = ħm s, m S ± s, m = ħ s s + m(m ± ) s, m ± s = 0: pi-mesons, Higgs oson, s = 2: electron, proton, neutron, quarks, s = : photon, W, Z, s = 3 2: deltas, omega, s = 2: graviton, etc.
8 S 2 = ħ 2 2 The case s = /2 (spin one-half) (simplest and most important) 3 2 = 3 4 ħ2 S z : mħ = ± ħ 2 What is the wavefunction? It cannot depend on x, y, z. Let us introduce something that satisfies formalism (commutation relations). Spinor χ = a = a Spin operators S z = ħ 2 S x = ħ 2 S y = ħ 2 spin-up χ 0 0 = ħ 2 σ z 0 0 = ħ 2 σ x 0 i i 0 = ħ 2 σ y σ x, σ y, σ z are called Pauli matrices spin-down χ (χ is chosen to resemle ψ) Normalization: a = These operators satisfy desired commutation relations: S x, S y = iħ S z, S y, S z = iħ S x, S z, S x = iħ S y Also, as desired S 2 = S 2 x + S 2 y + S 2 z = ħ2 4 (σ x 2 + σ 2 y + σ 2 z ) = ħ2 0 = 3 4 ħ2
9 Eigenvectors and eigenvalues We know that S z has eigenvalues ±ħ/2; what are the eigenvectors? S z a ± ħ 2 = λ a S z = ħ 2 χ = 0 Check: S z 0 = ħ (spin-up) 0 = ħ 2 0 Similarly S z = ħ 2 χ = 0 (spin-down) However, z-direction is not different from other directions, therefore the same eigenvalues for S x and S y. S x a ± ħ 2 = λ a Check: S x / 2 / 2 = ħ 2 S x = ħ 2 χ = / 2 / 2 = 2 S x = ħ 2 χ = / 2 / 2 = / 2 / 2 = ħ 2 / 2 / 2 (similar for ħ/2)
10 not included into this course Similarly find eigenvectors for S y S y a = λ a S y = ħ 2 χ = / 2 i/ 2 = 2 i ± ħ 2 S y = ħ 2 χ = / 2 i/ 2 = 2 i Actually, S x and S y correspond to new ases of eigenstates, and everything can also e considered in these new ases a (z) a i 2 a + i 2 (y) a + 2 a 2 (x) (we will consider only the standard z-asis)
11 χ = a Measurement of spin (in standard z-representation) Measure S z : get S z = get S z = ħ 2 with proaility P z+ = a 2, after that χ = 0 ħ 2 with proaility P z = 2, after that χ = 0 Measure S x : get S x = S x = P x+ = 2 ħ 2 with P x = 2 ħ 2 with proaility a 2 2 = a + 2 after that χ = a 2 = 2 a 2 then χ = 2 Measure S y : get S y = ħ 2 with proaility P y+ = i a = a i 2 then χ = 2 2 i S y = ħ 2 with P y = +i a = a + i 2 then χ = 2 2 i
12 Experimental measurement of spin (Stern-Gerlach experiment, 922) (not included into this course) N inhomogeneous magnetic field produces force onto a magnetic moment neutral atoms S H = γb S angular momentum (or spin) magnetic field gyromagnetic ratio If B is inhomogeneous (not constant), then F = H = γ (B S) So, force depends on S. If B x, y, z = B 0 + αz k αx i then F = γα S z k S x i F z = γαs z spin-up is deflected down (γ < 0), spin-down is deflected up (for magnetic field B x not important + B y + B z x y not important (oscillates ecause of Larmor precession aout z-axis, so zero on average) z = 0), (particle should e neutral ecause otherwise charge will circle in magnetic field)
Lecture 11 Spin, orbital, and total angular momentum Mechanics. 1 Very brief background. 2 General properties of angular momentum operators
 Lecture Spin, orbital, and total angular momentum 70.00 Mechanics Very brief background MATH-GA In 9, a famous experiment conducted by Otto Stern and Walther Gerlach, involving particles subject to a nonuniform
Lecture Spin, orbital, and total angular momentum 70.00 Mechanics Very brief background MATH-GA In 9, a famous experiment conducted by Otto Stern and Walther Gerlach, involving particles subject to a nonuniform
Angular Momentum. Andreas Wacker Mathematical Physics Lund University
 Angular Momentum Andreas Wacker Mathematical Physics Lund University Commutation relations of (orbital) angular momentum Angular momentum in analogy with classical case L= r p satisfies commutation relations
Angular Momentum Andreas Wacker Mathematical Physics Lund University Commutation relations of (orbital) angular momentum Angular momentum in analogy with classical case L= r p satisfies commutation relations
Total Angular Momentum for Hydrogen
 Physics 4 Lecture 7 Total Angular Momentum for Hydrogen Lecture 7 Physics 4 Quantum Mechanics I Friday, April th, 008 We have the Hydrogen Hamiltonian for central potential φ(r), we can write: H r = p
Physics 4 Lecture 7 Total Angular Momentum for Hydrogen Lecture 7 Physics 4 Quantum Mechanics I Friday, April th, 008 We have the Hydrogen Hamiltonian for central potential φ(r), we can write: H r = p
Angular momentum and spin
 Luleå tekniska universitet Avdelningen för Fysik, 007 Hans Weber Angular momentum and spin Angular momentum is a measure of how much rotation there is in particle or in a rigid body. In quantum mechanics
Luleå tekniska universitet Avdelningen för Fysik, 007 Hans Weber Angular momentum and spin Angular momentum is a measure of how much rotation there is in particle or in a rigid body. In quantum mechanics
Spin Dynamics Basic Theory Operators. Richard Green SBD Research Group Department of Chemistry
 Spin Dynamics Basic Theory Operators Richard Green SBD Research Group Department of Chemistry Objective of this session Introduce you to operators used in quantum mechanics Achieve this by looking at:
Spin Dynamics Basic Theory Operators Richard Green SBD Research Group Department of Chemistry Objective of this session Introduce you to operators used in quantum mechanics Achieve this by looking at:
The experiment consists of studying the deflection of a beam of neutral ground state paramagnetic atoms (silver) in inhomogeneous magnetic field:
 SPIN 1/2 PARTICLE Stern-Gerlach experiment The experiment consists of studying the deflection of a beam of neutral ground state paramagnetic atoms (silver) in inhomogeneous magnetic field: A silver atom
SPIN 1/2 PARTICLE Stern-Gerlach experiment The experiment consists of studying the deflection of a beam of neutral ground state paramagnetic atoms (silver) in inhomogeneous magnetic field: A silver atom
Lecture #13 1. Incorporating a vector potential into the Hamiltonian 2. Spin postulates 3. Description of spin states 4. Identical particles in
 Lecture #3. Incorporating a vector potential into the Hamiltonian. Spin postulates 3. Description of spin states 4. Identical particles in classical and QM 5. Exchange degeneracy - the fundamental problem
Lecture #3. Incorporating a vector potential into the Hamiltonian. Spin postulates 3. Description of spin states 4. Identical particles in classical and QM 5. Exchange degeneracy - the fundamental problem
PHY4604 Introduction to Quantum Mechanics Fall 2004 Final Exam SOLUTIONS December 17, 2004, 7:30 a.m.- 9:30 a.m.
 PHY464 Introduction to Quantum Mechanics Fall 4 Final Eam SOLUTIONS December 7, 4, 7:3 a.m.- 9:3 a.m. No other materials allowed. If you can t do one part of a problem, solve subsequent parts in terms
PHY464 Introduction to Quantum Mechanics Fall 4 Final Eam SOLUTIONS December 7, 4, 7:3 a.m.- 9:3 a.m. No other materials allowed. If you can t do one part of a problem, solve subsequent parts in terms
The Postulates of Quantum Mechanics Common operators in QM: Potential Energy. Often depends on position operator: Kinetic Energy 1-D case: 3-D case
 The Postulates of Quantum Mechanics Common operators in QM: Potential Energy Often depends on position operator: Kinetic Energy 1-D case: 3-D case Time Total energy = Hamiltonian To find out about the
The Postulates of Quantum Mechanics Common operators in QM: Potential Energy Often depends on position operator: Kinetic Energy 1-D case: 3-D case Time Total energy = Hamiltonian To find out about the
3. Quantum Mechanics in 3D
 3. Quantum Mechanics in 3D 3.1 Introduction Last time, we derived the time dependent Schrödinger equation, starting from three basic postulates: 1) The time evolution of a state can be expressed as a unitary
3. Quantum Mechanics in 3D 3.1 Introduction Last time, we derived the time dependent Schrödinger equation, starting from three basic postulates: 1) The time evolution of a state can be expressed as a unitary
But what happens when free (i.e. unbound) charged particles experience a magnetic field which influences orbital motion? e.g. electrons in a metal.
 Lecture 5: continued But what happens when free (i.e. unbound charged particles experience a magnetic field which influences orbital motion? e.g. electrons in a metal. Ĥ = 1 2m (ˆp qa(x, t2 + qϕ(x, t,
Lecture 5: continued But what happens when free (i.e. unbound charged particles experience a magnetic field which influences orbital motion? e.g. electrons in a metal. Ĥ = 1 2m (ˆp qa(x, t2 + qϕ(x, t,
Angular momentum. Quantum mechanics. Orbital angular momentum
 Angular momentum 1 Orbital angular momentum Consider a particle described by the Cartesian coordinates (x, y, z r and their conjugate momenta (p x, p y, p z p. The classical definition of the orbital angular
Angular momentum 1 Orbital angular momentum Consider a particle described by the Cartesian coordinates (x, y, z r and their conjugate momenta (p x, p y, p z p. The classical definition of the orbital angular
Basic Physical Chemistry Lecture 2. Keisuke Goda Summer Semester 2015
 Basic Physical Chemistry Lecture 2 Keisuke Goda Summer Semester 2015 Lecture schedule Since we only have three lectures, let s focus on a few important topics of quantum chemistry and structural chemistry
Basic Physical Chemistry Lecture 2 Keisuke Goda Summer Semester 2015 Lecture schedule Since we only have three lectures, let s focus on a few important topics of quantum chemistry and structural chemistry
G : Quantum Mechanics II
 G5.666: Quantum Mechanics II Notes for Lecture 5 I. REPRESENTING STATES IN THE FULL HILBERT SPACE Given a representation of the states that span the spin Hilbert space, we now need to consider the problem
G5.666: Quantum Mechanics II Notes for Lecture 5 I. REPRESENTING STATES IN THE FULL HILBERT SPACE Given a representation of the states that span the spin Hilbert space, we now need to consider the problem
The Hydrogen Atom. Dr. Sabry El-Taher 1. e 4. U U r
 The Hydrogen Atom Atom is a 3D object, and the electron motion is three-dimensional. We ll start with the simplest case - The hydrogen atom. An electron and a proton (nucleus) are bound by the central-symmetric
The Hydrogen Atom Atom is a 3D object, and the electron motion is three-dimensional. We ll start with the simplest case - The hydrogen atom. An electron and a proton (nucleus) are bound by the central-symmetric
Angular Momentum Algebra
 Angular Momentum Algebra Chris Clark August 1, 2006 1 Input We will be going through the derivation of the angular momentum operator algebra. The only inputs to this mathematical formalism are the basic
Angular Momentum Algebra Chris Clark August 1, 2006 1 Input We will be going through the derivation of the angular momentum operator algebra. The only inputs to this mathematical formalism are the basic
Physics Capstone. Karsten Gimre May 19, 2009
 Physics Capstone Karsten Gimre May 19, 009 Abstract Quantum mechanics is, without a doubt, one of the major advancements of the 0th century. It can be incredibly difficult to understand the fundamental
Physics Capstone Karsten Gimre May 19, 009 Abstract Quantum mechanics is, without a doubt, one of the major advancements of the 0th century. It can be incredibly difficult to understand the fundamental
L z L L. Think of it as also affecting the angle
 Quantum Mechanics and Atomic Physics Lecture 19: Quantized Angular Momentum and Electron Spin http://www.physics.rutgers.edu/ugrad/361 h / d/361 Prof. Sean Oh Last time Raising/Lowering angular momentum
Quantum Mechanics and Atomic Physics Lecture 19: Quantized Angular Momentum and Electron Spin http://www.physics.rutgers.edu/ugrad/361 h / d/361 Prof. Sean Oh Last time Raising/Lowering angular momentum
One-electron Atom. (in spherical coordinates), where Y lm. are spherical harmonics, we arrive at the following Schrödinger equation:
 One-electron Atom The atomic orbitals of hydrogen-like atoms are solutions to the Schrödinger equation in a spherically symmetric potential. In this case, the potential term is the potential given by Coulomb's
One-electron Atom The atomic orbitals of hydrogen-like atoms are solutions to the Schrödinger equation in a spherically symmetric potential. In this case, the potential term is the potential given by Coulomb's
4/21/2010. Schrödinger Equation For Hydrogen Atom. Spherical Coordinates CHAPTER 8
 CHAPTER 8 Hydrogen Atom 8.1 Spherical Coordinates 8.2 Schrödinger's Equation in Spherical Coordinate 8.3 Separation of Variables 8.4 Three Quantum Numbers 8.5 Hydrogen Atom Wave Function 8.6 Electron Spin
CHAPTER 8 Hydrogen Atom 8.1 Spherical Coordinates 8.2 Schrödinger's Equation in Spherical Coordinate 8.3 Separation of Variables 8.4 Three Quantum Numbers 8.5 Hydrogen Atom Wave Function 8.6 Electron Spin
Lecture 18: 3D Review, Examples
 Lecture 18: 3D Review, Examples A real (2D) quantum dot http://pages.unibas.ch/physmeso/pictures/pictures.html Lecture 18, p 1 Lect. 16: Particle in a 3D Box (3) The energy eigenstates and energy values
Lecture 18: 3D Review, Examples A real (2D) quantum dot http://pages.unibas.ch/physmeso/pictures/pictures.html Lecture 18, p 1 Lect. 16: Particle in a 3D Box (3) The energy eigenstates and energy values
Rotations in Quantum Mechanics
 Rotations in Quantum Mechanics We have seen that physical transformations are represented in quantum mechanics by unitary operators acting on the Hilbert space. In this section, we ll think about the specific
Rotations in Quantum Mechanics We have seen that physical transformations are represented in quantum mechanics by unitary operators acting on the Hilbert space. In this section, we ll think about the specific
Properties of Elementary Particles
 and of Elementary s 01/11/2018 My Office Hours: Thursday 1:00-3:00 PM 212 Keen Building Outline 1 2 3 Consider the world at different scales... Cosmology - only gravity matters XXXXX Input: Mass distributions
and of Elementary s 01/11/2018 My Office Hours: Thursday 1:00-3:00 PM 212 Keen Building Outline 1 2 3 Consider the world at different scales... Cosmology - only gravity matters XXXXX Input: Mass distributions
How NMR Works Section I
 How NMR Works ------- Section I by Shaoxiong Wu 02-20-2010 During the NMR course I use classic energy levels and vector model to explain simple NMR pulse sequences and spectra. It is limited, however,
How NMR Works ------- Section I by Shaoxiong Wu 02-20-2010 During the NMR course I use classic energy levels and vector model to explain simple NMR pulse sequences and spectra. It is limited, however,
10 Lorentz Group and Special Relativity
 Physics 129 Lecture 16 Caltech, 02/27/18 Reference: Jones, Groups, Representations, and Physics, Chapter 10. 10 Lorentz Group and Special Relativity Special relativity says, physics laws should look the
Physics 129 Lecture 16 Caltech, 02/27/18 Reference: Jones, Groups, Representations, and Physics, Chapter 10. 10 Lorentz Group and Special Relativity Special relativity says, physics laws should look the
Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall Duration: 2h 30m
 Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. ------------------- Duration: 2h 30m Chapter 39 Quantum Mechanics of Atoms Units of Chapter 39 39-1 Quantum-Mechanical View of Atoms 39-2
Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. ------------------- Duration: 2h 30m Chapter 39 Quantum Mechanics of Atoms Units of Chapter 39 39-1 Quantum-Mechanical View of Atoms 39-2
Appendix: SU(2) spin angular momentum and single spin dynamics
 Phys 7 Topics in Particles & Fields Spring 03 Lecture v0 Appendix: SU spin angular momentum and single spin dynamics Jeffrey Yepez Department of Physics and Astronomy University of Hawai i at Manoa Watanabe
Phys 7 Topics in Particles & Fields Spring 03 Lecture v0 Appendix: SU spin angular momentum and single spin dynamics Jeffrey Yepez Department of Physics and Astronomy University of Hawai i at Manoa Watanabe
Lecture3 (and part of lecture 4).
 Lecture3 (and part of lecture 4). Angular momentum and spin. Stern-Gerlach experiment Spin Hamiltonian Evolution of spin with time Evolution of spin in precessing magnetic field. In classical mechanics
Lecture3 (and part of lecture 4). Angular momentum and spin. Stern-Gerlach experiment Spin Hamiltonian Evolution of spin with time Evolution of spin in precessing magnetic field. In classical mechanics
Atkins & de Paula: Atkins Physical Chemistry 9e Checklist of key ideas. Chapter 8: Quantum Theory: Techniques and Applications
 Atkins & de Paula: Atkins Physical Chemistry 9e Checklist of key ideas Chapter 8: Quantum Theory: Techniques and Applications TRANSLATIONAL MOTION wavefunction of free particle, ψ k = Ae ikx + Be ikx,
Atkins & de Paula: Atkins Physical Chemistry 9e Checklist of key ideas Chapter 8: Quantum Theory: Techniques and Applications TRANSLATIONAL MOTION wavefunction of free particle, ψ k = Ae ikx + Be ikx,
Stern-Gerlach experiment
 Lezione 2 Stern-Gerlach experiment Performed in Frankfurt, Germany in 1922 and named after Otto Stern and Walther Gerlach 1888 1969 1889-1979 At the time, Stern (34) and Gerlach (33) were assistants at
Lezione 2 Stern-Gerlach experiment Performed in Frankfurt, Germany in 1922 and named after Otto Stern and Walther Gerlach 1888 1969 1889-1979 At the time, Stern (34) and Gerlach (33) were assistants at
Quantum Physics II (8.05) Fall 2002 Outline
 Quantum Physics II (8.05) Fall 2002 Outline 1. General structure of quantum mechanics. 8.04 was based primarily on wave mechanics. We review that foundation with the intent to build a more formal basis
Quantum Physics II (8.05) Fall 2002 Outline 1. General structure of quantum mechanics. 8.04 was based primarily on wave mechanics. We review that foundation with the intent to build a more formal basis
20 The Hydrogen Atom. Ze2 r R (20.1) H( r, R) = h2 2m 2 r h2 2M 2 R
 20 The Hydrogen Atom 1. We want to solve the time independent Schrödinger Equation for the hydrogen atom. 2. There are two particles in the system, an electron and a nucleus, and so we can write the Hamiltonian
20 The Hydrogen Atom 1. We want to solve the time independent Schrödinger Equation for the hydrogen atom. 2. There are two particles in the system, an electron and a nucleus, and so we can write the Hamiltonian
(n, l, m l ) 3/2/2016. Quantum Numbers (QN) Plots of Energy Level. Roadmap for Exploring Hydrogen Atom
 PHYS 34 Modern Physics Atom III: Angular Momentum and Spin Roadmap for Exploring Hydrogen Atom Today Contents: a) Orbital Angular Momentum and Magnetic Dipole Moment b) Electric Dipole Moment c) Stern
PHYS 34 Modern Physics Atom III: Angular Momentum and Spin Roadmap for Exploring Hydrogen Atom Today Contents: a) Orbital Angular Momentum and Magnetic Dipole Moment b) Electric Dipole Moment c) Stern
Atomic Systems (PART I)
 Atomic Systems (PART I) Lecturer: Location: Recommended Text: Dr. D.J. Miller Room 535, Kelvin Building d.miller@physics.gla.ac.uk Joseph Black C407 (except 15/1/10 which is in Kelvin 312) Physics of Atoms
Atomic Systems (PART I) Lecturer: Location: Recommended Text: Dr. D.J. Miller Room 535, Kelvin Building d.miller@physics.gla.ac.uk Joseph Black C407 (except 15/1/10 which is in Kelvin 312) Physics of Atoms
The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of hydrogen atom.
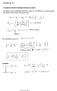 Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
charges q r p = q 2mc 2mc L (1.4) ptles µ e = g e
 APAS 5110. Atomic and Molecular Processes. Fall 2013. 1. Magnetic Moment Classically, the magnetic moment µ of a system of charges q at positions r moving with velocities v is µ = 1 qr v. (1.1) 2c charges
APAS 5110. Atomic and Molecular Processes. Fall 2013. 1. Magnetic Moment Classically, the magnetic moment µ of a system of charges q at positions r moving with velocities v is µ = 1 qr v. (1.1) 2c charges
Homework assignment 3: due Thursday, 10/26/2017
 Homework assignment 3: due Thursday, 10/6/017 Physics 6315: Quantum Mechanics 1, Fall 017 Problem 1 (0 points The spin Hilbert space is defined by three non-commuting observables, S x, S y, S z. These
Homework assignment 3: due Thursday, 10/6/017 Physics 6315: Quantum Mechanics 1, Fall 017 Problem 1 (0 points The spin Hilbert space is defined by three non-commuting observables, S x, S y, S z. These
Spin ½ (Pages 1-12 are needed)
 Prof. Dr. I. Nasser Phs- 55 (T-) October 3, 03 Spin3.doc Spin ½ (Pages - are needed) Recall that in the H-atom solution, we showed that the fact that the wavefunction ψ (r) is singlevalued requires that
Prof. Dr. I. Nasser Phs- 55 (T-) October 3, 03 Spin3.doc Spin ½ (Pages - are needed) Recall that in the H-atom solution, we showed that the fact that the wavefunction ψ (r) is singlevalued requires that
Lecture 4 Quantum mechanics in more than one-dimension
 Lecture 4 Quantum mechanics in more than one-dimension Background Previously, we have addressed quantum mechanics of 1d systems and explored bound and unbound (scattering) states. Although general concepts
Lecture 4 Quantum mechanics in more than one-dimension Background Previously, we have addressed quantum mechanics of 1d systems and explored bound and unbound (scattering) states. Although general concepts
Angular Momentum. Classically the orbital angular momentum with respect to a fixed origin is. L = r p. = yp z. L x. zp y L y. = zp x. xpz L z.
 Angular momentum is an important concept in quantum theory, necessary for analyzing motion in 3D as well as intrinsic properties such as spin Classically the orbital angular momentum with respect to a
Angular momentum is an important concept in quantum theory, necessary for analyzing motion in 3D as well as intrinsic properties such as spin Classically the orbital angular momentum with respect to a
LECTURES ON QUANTUM MECHANICS
 LECTURES ON QUANTUM MECHANICS GORDON BAYM Unitsersity of Illinois A II I' Advanced Bock Progrant A Member of the Perseus Books Group CONTENTS Preface v Chapter 1 Photon Polarization 1 Transformation of
LECTURES ON QUANTUM MECHANICS GORDON BAYM Unitsersity of Illinois A II I' Advanced Bock Progrant A Member of the Perseus Books Group CONTENTS Preface v Chapter 1 Photon Polarization 1 Transformation of
Quantum mechanics in one hour
 Chapter 2 Quantum mechanics in one hour 2.1 Introduction The purpose of this chapter is to refresh your knowledge of quantum mechanics and to establish notation. Depending on your background you might
Chapter 2 Quantum mechanics in one hour 2.1 Introduction The purpose of this chapter is to refresh your knowledge of quantum mechanics and to establish notation. Depending on your background you might
8.04 Spring 2013 March 12, 2013 Problem 1. (10 points) The Probability Current
 Prolem Set 5 Solutions 8.04 Spring 03 March, 03 Prolem. (0 points) The Proaility Current We wish to prove that dp a = J(a, t) J(, t). () dt Since P a (t) is the proaility of finding the particle in the
Prolem Set 5 Solutions 8.04 Spring 03 March, 03 Prolem. (0 points) The Proaility Current We wish to prove that dp a = J(a, t) J(, t). () dt Since P a (t) is the proaility of finding the particle in the
SU(N) representations
 Appendix C SU(N) representations The group SU(N) has N 2 1 generators t a which form the asis of a Lie algera defined y the commutator relations in (A.2). The group has rank N 1 so there are N 1 Casimir
Appendix C SU(N) representations The group SU(N) has N 2 1 generators t a which form the asis of a Lie algera defined y the commutator relations in (A.2). The group has rank N 1 so there are N 1 Casimir
Physics of Condensed Matter I
 Physics of Condensed Matter I 1100-4INZ`PC Faculty of Physics UW Jacek.Szczytko@fuw.edu.pl Summary of the lecture 1. Quantum mechanics 2. Optical transitions 3. Lasers 4. Optics 5. Molecules 6. Properties
Physics of Condensed Matter I 1100-4INZ`PC Faculty of Physics UW Jacek.Szczytko@fuw.edu.pl Summary of the lecture 1. Quantum mechanics 2. Optical transitions 3. Lasers 4. Optics 5. Molecules 6. Properties
conventions and notation
 Ph95a lecture notes, //0 The Bloch Equations A quick review of spin- conventions and notation The quantum state of a spin- particle is represented by a vector in a two-dimensional complex Hilbert space
Ph95a lecture notes, //0 The Bloch Equations A quick review of spin- conventions and notation The quantum state of a spin- particle is represented by a vector in a two-dimensional complex Hilbert space
5.61 Lecture #17 Rigid Rotor I
 561 Fall 2017 Lecture #17 Page 1 561 Lecture #17 Rigid Rotor I Read McQuarrie: Chapters E and 6 Rigid Rotors molecular rotation and the universal angular part of all central force problems another exactly
561 Fall 2017 Lecture #17 Page 1 561 Lecture #17 Rigid Rotor I Read McQuarrie: Chapters E and 6 Rigid Rotors molecular rotation and the universal angular part of all central force problems another exactly
1.6. Quantum mechanical description of the hydrogen atom
 29.6. Quantum mechanical description of the hydrogen atom.6.. Hamiltonian for the hydrogen atom Atomic units To avoid dealing with very small numbers, let us introduce the so called atomic units : Quantity
29.6. Quantum mechanical description of the hydrogen atom.6.. Hamiltonian for the hydrogen atom Atomic units To avoid dealing with very small numbers, let us introduce the so called atomic units : Quantity
Ket space as a vector space over the complex numbers
 Ket space as a vector space over the complex numbers kets ϕ> and complex numbers α with two operations Addition of two kets ϕ 1 >+ ϕ 2 > is also a ket ϕ 3 > Multiplication with complex numbers α ϕ 1 >
Ket space as a vector space over the complex numbers kets ϕ> and complex numbers α with two operations Addition of two kets ϕ 1 >+ ϕ 2 > is also a ket ϕ 3 > Multiplication with complex numbers α ϕ 1 >
Mathematical Tripos Part IB Michaelmas Term Example Sheet 1. Values of some physical constants are given on the supplementary sheet
 Mathematical Tripos Part IB Michaelmas Term 2015 Quantum Mechanics Dr. J.M. Evans Example Sheet 1 Values of some physical constants are given on the supplementary sheet 1. Whenasampleofpotassiumisilluminatedwithlightofwavelength3
Mathematical Tripos Part IB Michaelmas Term 2015 Quantum Mechanics Dr. J.M. Evans Example Sheet 1 Values of some physical constants are given on the supplementary sheet 1. Whenasampleofpotassiumisilluminatedwithlightofwavelength3
i = cos 2 0i + ei sin 2 1i
 Chapter 10 Spin 10.1 Spin 1 as a Qubit In this chapter we will explore quantum spin, which exhibits behavior that is intrinsically quantum mechanical. For our purposes the most important particles are
Chapter 10 Spin 10.1 Spin 1 as a Qubit In this chapter we will explore quantum spin, which exhibits behavior that is intrinsically quantum mechanical. For our purposes the most important particles are
Welcome back to PHYS 3305
 Welcome back to PHYS 3305 Otto Stern 1888-1969 Walther Gerlach 1889-1979 Today s Lecture: Angular Momentum Quantization Stern-Gerlach Experiment AnNouncements Reading Assignment for Nov 14th: Harris 8.2-8.5.
Welcome back to PHYS 3305 Otto Stern 1888-1969 Walther Gerlach 1889-1979 Today s Lecture: Angular Momentum Quantization Stern-Gerlach Experiment AnNouncements Reading Assignment for Nov 14th: Harris 8.2-8.5.
Introduction to Quantum Mechanics PVK - Solutions. Nicolas Lanzetti
 Introduction to Quantum Mechanics PVK - Solutions Nicolas Lanzetti lnicolas@student.ethz.ch 1 Contents 1 The Wave Function and the Schrödinger Equation 3 1.1 Quick Checks......................................
Introduction to Quantum Mechanics PVK - Solutions Nicolas Lanzetti lnicolas@student.ethz.ch 1 Contents 1 The Wave Function and the Schrödinger Equation 3 1.1 Quick Checks......................................
Welcome back to PHYS 3305
 Welcome back to PHYS 3305 Otto Stern 1888-1969 Walther Gerlach 1889-1979 Today s Lecture: Angular Momentum Quantization Stern-Gerlach Experiment Review: Orbital Dipole Moments The magnetic moment for an
Welcome back to PHYS 3305 Otto Stern 1888-1969 Walther Gerlach 1889-1979 Today s Lecture: Angular Momentum Quantization Stern-Gerlach Experiment Review: Orbital Dipole Moments The magnetic moment for an
Angular momentum & spin
 Angular momentum & spin January 8, 2002 1 Angular momentum Angular momentum appears as a very important aspect of almost any quantum mechanical system, so we need to briefly review some basic properties
Angular momentum & spin January 8, 2002 1 Angular momentum Angular momentum appears as a very important aspect of almost any quantum mechanical system, so we need to briefly review some basic properties
Physics 3220 Homework 13 Due Dec. 7, 2012
 This homework set concentrates on the spin ½ system and the closely related two-level system. All of quantum mechanics is right here, without a single differential equation. Have fun! 1. Spin 1/ system
This homework set concentrates on the spin ½ system and the closely related two-level system. All of quantum mechanics is right here, without a single differential equation. Have fun! 1. Spin 1/ system
ECE440 Nanoelectronics. Lecture 07 Atomic Orbitals
 ECE44 Nanoelectronics Lecture 7 Atomic Orbitals Atoms and atomic orbitals It is instructive to compare the simple model of a spherically symmetrical potential for r R V ( r) for r R and the simplest hydrogen
ECE44 Nanoelectronics Lecture 7 Atomic Orbitals Atoms and atomic orbitals It is instructive to compare the simple model of a spherically symmetrical potential for r R V ( r) for r R and the simplest hydrogen
Announcements. Lecture 20 Chapter. 7 QM in 3-dims & Hydrogen Atom. The Radial Part of Schrodinger Equation for Hydrogen Atom
 Announcements! HW7 : Chap.7 18, 20, 23, 32, 37, 38, 45, 47, 53, 57, 60! Physics Colloquium: Development in Electron Nuclear Dynamics Theory on Thursday @ 3:40pm! Quiz 2 (average: 9), Quiz 3: 4/19 *** Course
Announcements! HW7 : Chap.7 18, 20, 23, 32, 37, 38, 45, 47, 53, 57, 60! Physics Colloquium: Development in Electron Nuclear Dynamics Theory on Thursday @ 3:40pm! Quiz 2 (average: 9), Quiz 3: 4/19 *** Course
Quantum Mechanics II
 Quantum Mechanics II Prof. Boris Altshuler March 8, 011 1 Lecture 19 1.1 Second Quantization Recall our results from simple harmonic oscillator. We know the Hamiltonian very well so no need to repeat here.
Quantum Mechanics II Prof. Boris Altshuler March 8, 011 1 Lecture 19 1.1 Second Quantization Recall our results from simple harmonic oscillator. We know the Hamiltonian very well so no need to repeat here.
Review of the Formalism of Quantum Mechanics
 Review of the Formalism of Quantum Mechanics The postulates of quantum mechanics are often stated in textbooks. There are two main properties of physics upon which these postulates are based: 1)the probability
Review of the Formalism of Quantum Mechanics The postulates of quantum mechanics are often stated in textbooks. There are two main properties of physics upon which these postulates are based: 1)the probability
Solved radial equation: Last time For two simple cases: infinite and finite spherical wells Spherical analogs of 1D wells We introduced auxiliary func
 Quantum Mechanics and Atomic Physics Lecture 16: The Coulomb Potential http://www.physics.rutgers.edu/ugrad/361 h / d/361 Prof. Sean Oh Solved radial equation: Last time For two simple cases: infinite
Quantum Mechanics and Atomic Physics Lecture 16: The Coulomb Potential http://www.physics.rutgers.edu/ugrad/361 h / d/361 Prof. Sean Oh Solved radial equation: Last time For two simple cases: infinite
Quantum Physics 2: Homework #6
 Quantum Physics : Homework #6 [Total 10 points] Due: 014.1.1(Mon) 1:30pm Exercises: 014.11.5(Tue)/11.6(Wed) 6:30 pm; 56-105 Questions for problems: 민홍기 hmin@snu.ac.kr Questions for grading: 모도영 modori518@snu.ac.kr
Quantum Physics : Homework #6 [Total 10 points] Due: 014.1.1(Mon) 1:30pm Exercises: 014.11.5(Tue)/11.6(Wed) 6:30 pm; 56-105 Questions for problems: 민홍기 hmin@snu.ac.kr Questions for grading: 모도영 modori518@snu.ac.kr
Lecture 9. Angular momentum - 2
 Lecture 9 Angular momentum - 2 83 84 LECTURE 9. ANGULAR MOMENTUM - 2 9.1 Spectroscopic Notation We saw in the last section that the only allowed values of the angular momentum quantum number,, are =0,
Lecture 9 Angular momentum - 2 83 84 LECTURE 9. ANGULAR MOMENTUM - 2 9.1 Spectroscopic Notation We saw in the last section that the only allowed values of the angular momentum quantum number,, are =0,
PHY413 Quantum Mechanics B Duration: 2 hours 30 minutes
 BSc/MSci Examination by Course Unit Thursday nd May 4 : - :3 PHY43 Quantum Mechanics B Duration: hours 3 minutes YOU ARE NOT PERMITTED TO READ THE CONTENTS OF THIS QUESTION PAPER UNTIL INSTRUCTED TO DO
BSc/MSci Examination by Course Unit Thursday nd May 4 : - :3 PHY43 Quantum Mechanics B Duration: hours 3 minutes YOU ARE NOT PERMITTED TO READ THE CONTENTS OF THIS QUESTION PAPER UNTIL INSTRUCTED TO DO
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer. Lecture 20, March 8, 2006
 Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer Lecture 20, March 8, 2006 Solved Homework We determined that the two coefficients in our two-gaussian
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer Lecture 20, March 8, 2006 Solved Homework We determined that the two coefficients in our two-gaussian
P3317 HW from Lecture 15 and Recitation 8
 P3317 HW from Lecture 15 and Recitation 8 Due Oct 23, 218 Problem 1. Variational Energy of Helium Here we will estimate the ground state energy of Helium. Helium has two electrons circling around a nucleus
P3317 HW from Lecture 15 and Recitation 8 Due Oct 23, 218 Problem 1. Variational Energy of Helium Here we will estimate the ground state energy of Helium. Helium has two electrons circling around a nucleus
(a) Write down the total Hamiltonian of this system, including the spin degree of freedom of the electron, but neglecting spin-orbit interactions.
 1. Quantum Mechanics (Spring 2007) Consider a hydrogen atom in a weak uniform magnetic field B = Bê z. (a) Write down the total Hamiltonian of this system, including the spin degree of freedom of the electron,
1. Quantum Mechanics (Spring 2007) Consider a hydrogen atom in a weak uniform magnetic field B = Bê z. (a) Write down the total Hamiltonian of this system, including the spin degree of freedom of the electron,
3 Quantization of the Dirac equation
 3 Quantization of the Dirac equation 3.1 Identical particles As is well known, quantum mechanics implies that no measurement can be performed to distinguish particles in the same quantum state. Elementary
3 Quantization of the Dirac equation 3.1 Identical particles As is well known, quantum mechanics implies that no measurement can be performed to distinguish particles in the same quantum state. Elementary
PHY 407 QUANTUM MECHANICS Fall 05 Problem set 1 Due Sep
 Problem set 1 Due Sep 15 2005 1. Let V be the set of all complex valued functions of a real variable θ, that are periodic with period 2π. That is u(θ + 2π) = u(θ), for all u V. (1) (i) Show that this V
Problem set 1 Due Sep 15 2005 1. Let V be the set of all complex valued functions of a real variable θ, that are periodic with period 2π. That is u(θ + 2π) = u(θ), for all u V. (1) (i) Show that this V
3.024 Electrical, Optical, and Magnetic Properties of Materials Spring 2012 Recitation 3 Notes
 3.024 Electrical, Optical, and Magnetic Properties of Materials Spring 2012 Outline 1. Schr dinger: Eigenfunction Problems & Operator Properties 2. Piecewise Function/Continuity Review -Scattering from
3.024 Electrical, Optical, and Magnetic Properties of Materials Spring 2012 Outline 1. Schr dinger: Eigenfunction Problems & Operator Properties 2. Piecewise Function/Continuity Review -Scattering from
Clebsch-Gordan Coefficients
 Phy489 Lecture 7 Clebsch-Gordan Coefficients 2 j j j2 m m m 2 j= j j2 j + j j m > j m > = C jm > m = m + m 2 2 2 Two systems with spin j and j 2 and z components m and m 2 can combine to give a system
Phy489 Lecture 7 Clebsch-Gordan Coefficients 2 j j j2 m m m 2 j= j j2 j + j j m > j m > = C jm > m = m + m 2 2 2 Two systems with spin j and j 2 and z components m and m 2 can combine to give a system
Physics 342 Lecture 27. Spin. Lecture 27. Physics 342 Quantum Mechanics I
 Physics 342 Lecture 27 Spin Lecture 27 Physics 342 Quantum Mechanics I Monday, April 5th, 2010 There is an intrinsic characteristic of point particles that has an analogue in but no direct derivation from
Physics 342 Lecture 27 Spin Lecture 27 Physics 342 Quantum Mechanics I Monday, April 5th, 2010 There is an intrinsic characteristic of point particles that has an analogue in but no direct derivation from
Qualifying Exam. Aug Part II. Please use blank paper for your work do not write on problems sheets!
 Qualifying Exam Aug. 2015 Part II Please use blank paper for your work do not write on problems sheets! Solve only one problem from each of the four sections Mechanics, Quantum Mechanics, Statistical Physics
Qualifying Exam Aug. 2015 Part II Please use blank paper for your work do not write on problems sheets! Solve only one problem from each of the four sections Mechanics, Quantum Mechanics, Statistical Physics
1 Recall what is Spin
 C/CS/Phys C191 Spin measurement, initialization, manipulation by precession10/07/08 Fall 2008 Lecture 10 1 Recall what is Spin Elementary particles and composite particles carry an intrinsic angular momentum
C/CS/Phys C191 Spin measurement, initialization, manipulation by precession10/07/08 Fall 2008 Lecture 10 1 Recall what is Spin Elementary particles and composite particles carry an intrinsic angular momentum
Lecture #1. Review. Postulates of quantum mechanics (1-3) Postulate 1
 L1.P1 Lecture #1 Review Postulates of quantum mechanics (1-3) Postulate 1 The state of a system at any instant of time may be represented by a wave function which is continuous and differentiable. Specifically,
L1.P1 Lecture #1 Review Postulates of quantum mechanics (1-3) Postulate 1 The state of a system at any instant of time may be represented by a wave function which is continuous and differentiable. Specifically,
Sommerfeld (1920) noted energy levels of Li deduced from spectroscopy looked like H, with slight adjustment of principal quantum number:
 Spin. Historical Spectroscopy of Alkali atoms First expt. to suggest need for electron spin: observation of splitting of expected spectral lines for alkali atoms: i.e. expect one line based on analogy
Spin. Historical Spectroscopy of Alkali atoms First expt. to suggest need for electron spin: observation of splitting of expected spectral lines for alkali atoms: i.e. expect one line based on analogy
Quantum Physics 2006/07
 Quantum Physics 6/7 Lecture 7: More on the Dirac Equation In the last lecture we showed that the Dirac equation for a free particle i h t ψr, t = i hc α + β mc ψr, t has plane wave solutions ψr, t = exp
Quantum Physics 6/7 Lecture 7: More on the Dirac Equation In the last lecture we showed that the Dirac equation for a free particle i h t ψr, t = i hc α + β mc ψr, t has plane wave solutions ψr, t = exp
2.4. Quantum Mechanical description of hydrogen atom
 2.4. Quantum Mechanical description of hydrogen atom Atomic units Quantity Atomic unit SI Conversion Ang. mom. h [J s] h = 1, 05459 10 34 Js Mass m e [kg] m e = 9, 1094 10 31 kg Charge e [C] e = 1, 6022
2.4. Quantum Mechanical description of hydrogen atom Atomic units Quantity Atomic unit SI Conversion Ang. mom. h [J s] h = 1, 05459 10 34 Js Mass m e [kg] m e = 9, 1094 10 31 kg Charge e [C] e = 1, 6022
Reading: Mathchapters F and G, MQ - Ch. 7-8, Lecture notes on hydrogen atom.
 Chemistry 356 017: Problem set No. 6; Reading: Mathchapters F and G, MQ - Ch. 7-8, Lecture notes on hydrogen atom. The H atom involves spherical coordinates and angular momentum, which leads to the shapes
Chemistry 356 017: Problem set No. 6; Reading: Mathchapters F and G, MQ - Ch. 7-8, Lecture notes on hydrogen atom. The H atom involves spherical coordinates and angular momentum, which leads to the shapes
The 3 dimensional Schrödinger Equation
 Chapter 6 The 3 dimensional Schrödinger Equation 6.1 Angular Momentum To study how angular momentum is represented in quantum mechanics we start by reviewing the classical vector of orbital angular momentum
Chapter 6 The 3 dimensional Schrödinger Equation 6.1 Angular Momentum To study how angular momentum is represented in quantum mechanics we start by reviewing the classical vector of orbital angular momentum
PHY451, Spring /5
 PHY451, Spring 2011 Notes on Optical Pumping Procedure & Theory Procedure 1. Turn on the electronics and wait for the cell to warm up: ~ ½ hour. The oven should already e set to 50 C don t change this
PHY451, Spring 2011 Notes on Optical Pumping Procedure & Theory Procedure 1. Turn on the electronics and wait for the cell to warm up: ~ ½ hour. The oven should already e set to 50 C don t change this
Quantum Physics II (8.05) Fall 2002 Assignment 11
 Quantum Physics II (8.05) Fall 00 Assignment 11 Readings Most of the reading needed for this problem set was already given on Problem Set 9. The new readings are: Phase shifts are discussed in Cohen-Tannoudji
Quantum Physics II (8.05) Fall 00 Assignment 11 Readings Most of the reading needed for this problem set was already given on Problem Set 9. The new readings are: Phase shifts are discussed in Cohen-Tannoudji
Physics 215 Quantum Mechanics I Assignment 8
 Physics 15 Quantum Mechanics I Assignment 8 Logan A. Morrison March, 016 Problem 1 Let J be an angular momentum operator. Part (a) Using the usual angular momentum commutation relations, prove that J =
Physics 15 Quantum Mechanics I Assignment 8 Logan A. Morrison March, 016 Problem 1 Let J be an angular momentum operator. Part (a) Using the usual angular momentum commutation relations, prove that J =
Atomic Structure Ch , 9.6, 9.7
 Ch. 9.2-4, 9.6, 9.7 Magnetic moment of an orbiting electron: An electron orbiting a nucleus creates a current loop. A current loop behaves like a magnet with a magnetic moment µ:! µ =! µ B " L Bohr magneton:
Ch. 9.2-4, 9.6, 9.7 Magnetic moment of an orbiting electron: An electron orbiting a nucleus creates a current loop. A current loop behaves like a magnet with a magnetic moment µ:! µ =! µ B " L Bohr magneton:
Lecture #6 NMR in Hilbert Space
 Lecture #6 NMR in Hilbert Space Topics Review of spin operators Single spin in a magnetic field: longitudinal and transverse magnetiation Ensemble of spins in a magnetic field RF excitation Handouts and
Lecture #6 NMR in Hilbert Space Topics Review of spin operators Single spin in a magnetic field: longitudinal and transverse magnetiation Ensemble of spins in a magnetic field RF excitation Handouts and
Atoms, Molecules and Solids. From Last Time Superposition of quantum states Philosophy of quantum mechanics Interpretation of the wave function:
 Essay outline and Ref to main article due next Wed. HW 9: M Chap 5: Exercise 4 M Chap 7: Question A M Chap 8: Question A From Last Time Superposition of quantum states Philosophy of quantum mechanics Interpretation
Essay outline and Ref to main article due next Wed. HW 9: M Chap 5: Exercise 4 M Chap 7: Question A M Chap 8: Question A From Last Time Superposition of quantum states Philosophy of quantum mechanics Interpretation
The Stern-Gerlach experiment and spin
 The Stern-Gerlach experiment and spin Experiments in the early 1920s discovered a new aspect of nature, and at the same time found the simplest quantum system in existence. In the Stern-Gerlach experiment,
The Stern-Gerlach experiment and spin Experiments in the early 1920s discovered a new aspect of nature, and at the same time found the simplest quantum system in existence. In the Stern-Gerlach experiment,
Are these states normalized? A) Yes
 QMII-. Consider two kets and their corresponding column vectors: Ψ = φ = Are these two state orthogonal? Is ψ φ = 0? A) Yes ) No Answer: A Are these states normalized? A) Yes ) No Answer: (each state has
QMII-. Consider two kets and their corresponding column vectors: Ψ = φ = Are these two state orthogonal? Is ψ φ = 0? A) Yes ) No Answer: A Are these states normalized? A) Yes ) No Answer: (each state has
Chm 331 Fall 2015, Exercise Set 4 NMR Review Problems
 Chm 331 Fall 015, Exercise Set 4 NMR Review Problems Mr. Linck Version.0. Compiled December 1, 015 at 11:04:44 4.1 Diagonal Matrix Elements for the nmr H 0 Find the diagonal matrix elements for H 0 (the
Chm 331 Fall 015, Exercise Set 4 NMR Review Problems Mr. Linck Version.0. Compiled December 1, 015 at 11:04:44 4.1 Diagonal Matrix Elements for the nmr H 0 Find the diagonal matrix elements for H 0 (the
Chapter Electron Spin. * Fine structure:many spectral lines consist of two separate. lines that are very close to each other.
 Chapter 7 7. Electron Spin * Fine structure:many spectral lines consist of two separate lines that are very close to each other. ex. H atom, first line of Balmer series n = 3 n = => 656.3nm in reality,
Chapter 7 7. Electron Spin * Fine structure:many spectral lines consist of two separate lines that are very close to each other. ex. H atom, first line of Balmer series n = 3 n = => 656.3nm in reality,
ECE 487 Lecture 6 : Time-Dependent Quantum Mechanics I Class Outline:
 ECE 487 Lecture 6 : Time-Dependent Quantum Mechanics I Class Outline: Time-Dependent Schrödinger Equation Solutions to thetime-dependent Schrödinger Equation Expansion of Energy Eigenstates Things you
ECE 487 Lecture 6 : Time-Dependent Quantum Mechanics I Class Outline: Time-Dependent Schrödinger Equation Solutions to thetime-dependent Schrödinger Equation Expansion of Energy Eigenstates Things you
Physics 221A Fall 1996 Notes 13 Spins in Magnetic Fields
 Physics 221A Fall 1996 Notes 13 Spins in Magnetic Fields A nice illustration of rotation operator methods which is also important physically is the problem of spins in magnetic fields. The earliest experiments
Physics 221A Fall 1996 Notes 13 Spins in Magnetic Fields A nice illustration of rotation operator methods which is also important physically is the problem of spins in magnetic fields. The earliest experiments
The Hydrogen Atom. Thornton and Rex, Ch. 7
 The Hydrogen Atom Thornton and Rex, Ch. 7 Applying Schrodinger s Eqn to the Hydrogen Atom The potential: -1 e 2 V(r) = 4p e0 r Use spherical polar coordinates (with y(x,y,z) => y(r,q,f) ): 1 y 1 y ( r
The Hydrogen Atom Thornton and Rex, Ch. 7 Applying Schrodinger s Eqn to the Hydrogen Atom The potential: -1 e 2 V(r) = 4p e0 r Use spherical polar coordinates (with y(x,y,z) => y(r,q,f) ): 1 y 1 y ( r
PHYS 172H: Modern Mechanics Fall 2010
 PHYS 172H: Modern Mechanics Fall 2010 Lecture 20 Angular Momentum Quantization (mostly) Chapter 11.8 11.11 CLICKER POLL Not for Credit How many senses does the human body have? (Taste, smell...) A) 3 senses
PHYS 172H: Modern Mechanics Fall 2010 Lecture 20 Angular Momentum Quantization (mostly) Chapter 11.8 11.11 CLICKER POLL Not for Credit How many senses does the human body have? (Taste, smell...) A) 3 senses
量子力学 Quantum mechanics. School of Physics and Information Technology
 量子力学 Quantum mechanics School of Physics and Information Technology Shaanxi Normal University Chapter 9 Time-dependent perturation theory Chapter 9 Time-dependent perturation theory 9.1 Two-level systems
量子力学 Quantum mechanics School of Physics and Information Technology Shaanxi Normal University Chapter 9 Time-dependent perturation theory Chapter 9 Time-dependent perturation theory 9.1 Two-level systems
Degeneracy & in particular to Hydrogen atom
 Degeneracy & in particular to Hydrogen atom In quantum mechanics, an energy level is said to be degenerate if it corresponds to two or more different measurable states of a quantum system. Conversely,
Degeneracy & in particular to Hydrogen atom In quantum mechanics, an energy level is said to be degenerate if it corresponds to two or more different measurable states of a quantum system. Conversely,
Lecture 19: Building Atoms and Molecules
 Lecture 19: Building Atoms and Molecules +e r n = 3 n = 2 n = 1 +e +e r y even Lecture 19, p 1 Today Nuclear Magnetic Resonance Using RF photons to drive transitions between nuclear spin orientations in
Lecture 19: Building Atoms and Molecules +e r n = 3 n = 2 n = 1 +e +e r y even Lecture 19, p 1 Today Nuclear Magnetic Resonance Using RF photons to drive transitions between nuclear spin orientations in
Collection of formulae Quantum mechanics. Basic Formulas Division of Material Science Hans Weber. Operators
 Basic Formulas 17-1-1 Division of Material Science Hans Weer The de Broglie wave length λ = h p The Schrödinger equation Hψr,t = i h t ψr,t Stationary states Hψr,t = Eψr,t Collection of formulae Quantum
Basic Formulas 17-1-1 Division of Material Science Hans Weer The de Broglie wave length λ = h p The Schrödinger equation Hψr,t = i h t ψr,t Stationary states Hψr,t = Eψr,t Collection of formulae Quantum
Let us go back to what you knew in high school, or even earlier...
 Lecture I Quantum-Mechanical Way of Thinking To cultivate QM way of thinking, we will not start with the fascinating historical approach, but instead begin with one of the most important expt, that sends
Lecture I Quantum-Mechanical Way of Thinking To cultivate QM way of thinking, we will not start with the fascinating historical approach, but instead begin with one of the most important expt, that sends
1 Measurement and expectation values
 C/CS/Phys 191 Measurement and expectation values, Intro to Spin 2/15/05 Spring 2005 Lecture 9 1 Measurement and expectation values Last time we discussed how useful it is to work in the basis of energy
C/CS/Phys 191 Measurement and expectation values, Intro to Spin 2/15/05 Spring 2005 Lecture 9 1 Measurement and expectation values Last time we discussed how useful it is to work in the basis of energy
