and conformational flexibility of (bio)molecules
|
|
|
- Tamsyn Hubbard
- 5 years ago
- Views:
Transcription
1 Raman optical activity (ROA) and conformational flexibility of (bio)molecules in solution Vladimír r Baumruk Charles University in Prague Faculty of Mathematics and Physics Institute of Physics seminář Katedry fyzikální a makromolekulární chemie, PřF UK, 14. března 27
2 Vibrational Spectroscopy infrared spectroscopy (IR) Raman spectroscopy (RS) Vibrational Optical Activity (VOA) Vibrational Circular Dichroism (VCD) Raman Optical Activity (ROA)
3 Raman Optical Activity - principles 2 1 L-Alanyl-L-Alanine ROA I R -I L (x1-5 ) D-Alanyl-D-Alanine Raman I R +I L (x1-8 ) L-alanyl-L-alanine D-alanyl-D-alanine wavenumbers (cm -1 ) The sensitivity of optical activity to mirror-symmetry properties of chiral molecules is the source of its remarkable ability to specify absolute stereochemical properties of chiral molecules in solution.
4 Raman optical activity (ROA) method of vibrational optical activity (VOA); ; complementary to vibrational circular dichroism (VCD) in a similar way like Raman spectroscopy is complementary to IR spectroscopy, difference technique difference in response of a chiral molecule with respect to right and left circularly polarized light, combines the stereochemical sensitivity of natural optical activity with the rich structural content and conformational sensitivity of vibrational spectroscopy, the time scale of the Raman scattering event (~ 1-14 s), is much shorter than that of the fastest conformational fluctuations in biomolecules the ROA spectrum is a superposition of snapshot spectra from all the distinct chiral conformers present in the sample enhanced sensitivity to the dynamic aspects of biomolecular structure.
5 Vibrational optical activity The sign and magnitude of VCD are conveniently expressed as the dimensionless anisotropy ratio defined as a ratio of the experimental VCD band absorbance to the experimental IR band absorbance 4Im g Δ εε= ε ε ε ε L R + 4R μ. m L R g = = D 2 ROA intensity is usually expressed as dimensionless ratio known as the circular intensity difference Δ I I I + I R L R L Anisotropy ratio Δε/ε and circular intensity difference Δ are proportional to d / /λ (d is a typical internuclear distance in a molecule and λ is the wavelength of the light) in the near UV region Δε /ε is typically 1-3 (ECD), in the IR region Δε /ε is typically (VCD), in the visible region Δ is typically (ROA). μ
6 Raman optical activity experimental geometry Raman scattering and therefore ROA is associated with a two- photon process in comparison to VCD, ROA is inherently more complex,, both theoretically and experimentally. there are many distinct forms of ROA from which to chose in setting up an experiment the scattering geometry (scattering angle), modulation scheme. Scattering angle ξ can generally have any value between º and 18º but ROA is usually measured in backscattering geometry (ξ( =18º ), right-angle scattering geometry (ξ( =9º), forward scattering geometry (ξ =º).
7 Raman optical activity modulation schemes incident circular polarization (ICP) scattered circular polarization (SCP) in-phase dual circular polarization (DCPI) out-of of-phase dual circular polarization (DCPII) If one seeks the optimum ROA intensity for a given investment in incident laser intensity, the backscattering ICP and DCPI ROA have been found to be superior to other approaches. ChiralRAMAN TM commercial ROA spectrometer from BioTools (SCP)
8 Prague ROA Spectrometer Block diagram of the spectrometer ICP modulation scheme backscattering geometry Optical diagram spectrograph HoloSpec (optical speed f/1.4) back-illuminated CCD detector 134x1 pixels precise alignment of optical elements
9 Prague ROA spectrometer stable enables long time accumulation times (~1h) spectra accessible down to 1 cm -1 back-illuminated CCD detector (134x1 pixels) spectrograph HoloSpec f/1.4 EOM
10 Spectral data treatment 1 st step - solvent subtraction A B final spectrum ("solvent" subtracted) I R +I L (x1-9 ) 2 raw sample spectrum "solvent" spectrum sample spectrum after solvent subtraction 1 "solvent" spectrum I R +I L (x1-9 ) 1 2 nd step - baseline correction 2 sample spectrum after solvent subtraction sample spectrum after baseline correction I R -I L (x1-6 ) FT filtered spectrum raw spectrum wavenumber (cm -1 ) wavenumber (cm -1 ) I R +I L (x1-9 ) st step - solvent subtraction raw sample spectrum "solvent" spectrum sample spectrum after solvent subtraction A B final spectrum (after baseline correction) after "solvent" subtraction I R +I L (x1-9 ) nd step - baseline correction sample spectrum after solvent subtraction sample spectrum after baseline correction I R -I L (x1-6 ) "solvent" spectrum raw spectrum wavenumber (cm -1 ) wavenumber (cm -1 )
11 Raman optical activity basic theory Raman scattering and ROA two-photon phenomena tensor quantities ROA depends on the interaction of molecules with radiation beyond the electric- dipole approximation. In the case of ROA it is both magnetic-dipole and electric- quadrupole interaction (whereas for VCD it is only the magnetic-dipole interaction). General ROA theory is complicated, there are two useful approximations: far from resonance (FFR) strong resonance with a single electronic state (SES) Raman and ROA intensities can be expressed as a linear combination on of invariants of polarizability tensor α αβ (Raman) a ROA tensors (magnetic( dipole G αβ and electric quadrupole A αβγ ) Raman scattering in the general case 3 invariants, in the FFR only 2 ROA in the general case 1 invariants, in the FFR the number of invariants reduces to 3 or to even 2 in case of backscattering geometry
12 Raman optical activity basic theory In the far from resonance approximation: 2 ω polarizability tensor α = Re n μα j j μ n αβ ω β electric dipole-magnetic dipole optical activity tensor electric dipole-electric electric quadrupole optical activity tensor j n jn 2 ω2 jn G 2 ω = Im n μ αβ α ω ω j jm β n j n 2 2 jn A 2 ω = Re αβγ n μα ω j j Θ βγ n j n isotropic invariants of the polarizability tensor and electric dipole-magnetic dipole optical activity tensor jn 2 ω2 jn α= 1α G 1G 3 αα = 3 αα anisotropic invariants of the polarizability-polarizability tensor polarizability-magnetic dipole OA tensor polarizability-electric quadrupole OA tensor ( ) αα 2 αβ αβ ββ 2 G 1 = 3 G ααg 2 αβ αβ ββ βα = α α α α β α α 2 β A 1 = ωα ε 2 αβ αγδ A γδβ
13 Raman optical activity circular intensity difference for unpolarized ICP ROA in backscattering geometry ( G ) + β ( A) ( ) ( ) 3 β ΔI 18 u R L c 18 Iu Iu u R L I 2 u ( 18 ) Iu + I u 245 α2 + 7β ( α) Δ = = circular intensity difference for DCP I ROA in backscattering geometry ( G ) + β ( A) 48 ( ) ( ) 3 β ΔI 18 R L I I I c Δ 18 = R L = I R L I ( 18 I ) I + I R L 26 βα ( ) ROA Raman ROA Raman In the FFR approximation, backscattering ICP and DCP I modulation schemes yield the same expression for ROA intensity, as has been demonstrated experimentally, but the corresponding Raman intensity is greater for the ICP experiment (polarized) than for the DCP I one (depolarized).
14 Raman optical activity ICP DCPI * * Comparison of ICP (left( left) ) anda DCP I (right)) ROA anda Raman spectra of (-)-transtrans pinane in backscattering geometry.
15 Raman optical activity basic applications probe of absolute configuration, determination of stereo-conformational features of chiral molecules (in solution!!!) diagnostic tool to monitor optical purity (direct measurement of enantiomeric access) or identify chiral compounds from a chromatographic column structure elucidation of biologically significant molecules in solution including peptides, proteins, nucleic acids, carbohydrates, viruses, natural products, pharmaceutical molecules )
16 Simulations of ROA spectra (1S)-(-)-α-pinen pinene exp exp ROA and Raman spectra of (1S)-(-)-α-pinen pinene: (a) simple (so called polar model) and (b) conventional model of ROA intensity calculation, (c) experimental ental ROA spectrum, (d) simulated and (e) experimental parent Raman spectrum. Bouř et al., CCCC 62 (1997) 1384
17 Interpretation of ROA spectra Several levels of interpretation: Empirical identification of characteristic markers
18 Proteins X-Ray PDB: 69,2% α-helix 1,7% 3 1 -helix protein backbone vibrations (C α -C, C α -C β, C α -N stretching) ~ cm -1 extended amide III region (N-H bending, C α -N stretching + C α -H bending) 43,5% β-sheet 1,7% α-helix 1.3% 3 1 -helix ~ cm -1 amide I (C=O stretching) ~ cm -1 28,7% α-helix 1,9% 3 1 -helix 6,2% β-sheet Blanch et al., Vib. Spectrosc. 35 (24) 87
19 Interpretation of ROA spectra Several levels of interpretation: Empirical identification of characteristic markers Statistical methods e.g. principal component analysis
20 Proteins Principal Component Analysis Blanch et al., Vib. Spectrosc. 35 (24) 87
21 Interpretation of ROA spectra Several levels of interpretation: Empirical identification of characteristic markers Statistical methods e.g. principal component analysis Large biomolecules GAP Ab initio methods (HF, DFT) Small model systems
22 Ab initio calculations Major limitations: size of studied systems interaction with solvent (zwittterionic molecules) system flexibility (real system x model)
23 Ab initio calculations Overview Conformer selection Geometry Optimisation in internal coordinates (standard procedure) in normal modes 1 (useful for flexible molecules or clusters with water; normal modes with energies lower than a treshold can be constrained) methods involving electron correlation (DFT, MP2) Frequencies same level as above (COSMO/B3LYP/6-31++G**) Intensities derivatives of polarizability and optical activity tensors) computationally very demanding (vacuum/hf/6-31g) Comparison with Experiment 1 Bouř et al., J. Chem. Phys. 117 (22) 4126
24 Solvent simulations Implicit solvent models Onsager dipole model (the simplest) spherical cavity in dielectrics COSMO (COnductor like Screening MOdel) more flexible and more realistic COSMO cavity around proline molecule; colors correspond to charge induced on the surface by the molecule Explicit solvent models solvation with explicit molecules of water 4 conformations of proline calculated with explicit water molecules
25 L-Alanine the simplest amino acid ideal benchmark system for studying: conformational behavior interaction with solvent
26 L-Alanine potential energy surface 1D scan 2D scan B3LYP/6-31G** B3LYP/6-311G** B3LYP/6-31++G** 3 B3P86/6-31G** B3P86/6-311G** B3P86/6-31++G** ϕ, ψ B3LYP/COSMO/6-31++G** E (kcal/mol) ϕ (NH 3 + ) ψ (COO - ) χ (CH 3 ) χ (CH 3 ) E (kcal/mol) ϕ, χ -3-6 ϕ (NH 3 + ) χ (CH 3 ) E (kcal/mol) ψ (COO - ) ψ, χ -3 ψ (COO - ) ψ (COO - ) χ (CH 3 ) Dependencies of molecular energy (E, left) and dihedral angles (right) on the angles ϕ, ψ, and χ.. The remaining coordinates in the one- dimensional scans were allowed to fully relax ϕ (NH 3 + ) ϕ (NH 3 + ) Kapitán et al., J. Phys. Chem. 11 (26) 4689
27 Mode number: I R + I L (I R - I L ) 1 4 I R + I L (I R - I L ) 1 4 I R + I L , ,13 14, ,19 22, , Wavenumber to 9 Wavenumber ψ (COO - ) -9 to to Wavenumber χ (CH 3 ) Wavenumber ϕ (NH 3 + ) NH 3 + COO - (I R - I L ) to Wavenumber CH Kapitán et al., J. Phys. Chem. 11 (26) 4689
28 L-Alanine Raman and ROA spectra can be reliably interpreted if the movement of flexible molecular parts is considered (I R - I L ) 1-5 (I L + I R ) Experiment , , D-Ala Wavenumber , ,21 22, ,21 L-Ala Raman ROA (I R - I L ) 1 4 I R + I L Calculation 2, Wavenumber Wavenumber , , ,26 12, , , Kapitán et al., J. Phys. Chem. 11 (26) 4689
29 L-Proline proline exhibits two major conformations with very similar energies (ΔE <.3 kcal/mol) Conformation A Conformation B conformational space of proline was investigated in detail by rotating the COO - group and ring puckering checked influence of water molecules
30 L-Proline ring puckering ring puckering phase 1 ring puckering amplitude 1 1 Altona et al., JACS 94 (1972) 825 Dependent rotation of COO - group:
31 L-Proline rotation of COO - group E (kcal/mol) kt (298K) conf. A conf. B ψ (COO - ) 3 27 Phase conf. B conf. A ψ (COO - )
32 L-Proline different approaches Experim ent in H 2 O A,6B 12,13 14,15 18, ,29 3,31 32, B A , B W avenum ber , A , Puckering average W avenum ber W avenum ber 3, Exp Experiment Ring puckering COO - average W avenum ber (cm -1 ) W avenum ber Rot. COO W a te r c lu s te rs 1 4 W ater 8 not subtracted S o lu te o n ly W avenum ber (cm -1 ) Water clusters
33 Model dipeptides There is a significant difference between Ala-Pro x Pro-Ala Gly-Pro x Pro-Gly ROA (Raman) can directly reflect flexibility of the molecule
34 Model dipeptides rigidity flexibility Gly-Pro Pro-Gly ψ ϕ ψ ϕ (I R + I L ) 1-9 (I R - I L ) Wavenumber (I R + I L ) 1-9 (I R - I L ) Wavenumber
35 Poly(L-Proline Proline) in polyproline II (PPII) conformation (φ=-78, ψ=149 ) left-handed 3 1 -helix Schematic representation of the tensor transfer technique. Atomic property tensors in the tripeptides were obtained ab initio and transferred by propagation of these smaller fragments along the longer polypeptide chain.
36 Poly(L-Proline Proline) ring conformation generated 2-mer sequences with different A/B conformational content averaging over 5 generated sequences A B I R +I L I R -I L x1 3 1 Raman 5 ROA
37 Poly(L-Proline Proline) ring conformation generated 2-mer sequences with different A/B conformational content averaging over 5 generated sequences A B I R +I L I R -I L x1 3 1 Raman 5 ROA
38 Poly(L-Proline Proline) ring conformation generated 2-mer sequences with different A/B conformational content averaging over 5 generated sequences A B I R +I L I R -I L x1 3 1 Raman 5 ROA
39 Poly(L-Proline Proline) ring conformation generated 2-mer sequences with different A/B conformational content averaging over 5 generated sequences A B I R +I L I R -I L x1 3 1 Raman 5 ROA
40 Poly(L-Proline Proline) ring conformation generated 2-mer sequences with different A/B conformational content averaging over 5 generated sequences A B I R +I L I R -I L x1 3 1 Raman 5 ROA
41 Poly(L-Proline Proline) ring conformation generated 2-mer sequences with different A/B conformational content averaging over 5 generated sequences A B I R +I L I R -I L x1 3 1 Raman 5 ROA
42 Poly(L-Proline Proline) ring conformation generated 2-mer sequences with different A/B conformational content averaging over 5 generated sequences A B I R +I L I R -I L x1 3 1 Raman 5 ROA
43 Poly(L-Proline Proline) ring conformation generated 2-mer sequences with different A/B conformational content averaging over 5 generated sequences A B I R +I L I R -I L x1 3 1 Raman 5 ROA
44 Poly(L-Proline Proline) ring conformation generated 2-mer sequences with different A/B conformational content averaging over 5 generated sequences A B I R +I L I R -I L x1 3 1 Raman 5 ROA
45 Poly(L-Proline Proline) ring conformation generated 2-mer sequences with different A/B conformational content averaging over 5 generated sequences A B I R +I L I R -I L x1 3 1 Raman 5 ROA
46 Poly(L-Proline Proline) ring conformation generated 2-mer sequences with different A/B conformational content averaging over 5 generated sequences A B I R +I L I R -I L x1 3 1 Raman 5 ROA
47 Poly(L-Proline Proline) grand finale Comparison of experimental data with simulated 2-mer A:B=1:1 ( ) I R +I L Calc 5 I R -I L Exp (++-) Kapitán et al., JACS 128 (26) 2438
48 Hinge peptide and its analogs HINGE peptide is a fragment / from the core of human immunoglobulin IgG1. It acts in this parent molecule as a swivel point crosslinking two rather heavy peptide chains. Being a parallel dimer of the octapeptide H-Thr-Cys-Pro-Pro-Cys-Pro-Ala-Pro-OH (C 2 symmetry). The sequence is rich in proline residues and is expected to be quite rigid also due to a presence of two disulphide bridges. The peptide offers several advantages for use as a universal carrier of various active sequences (immunologically neutral, possesses six independent terminal groups: -NH and -OH on Thr residues and C-terminal carboxyl). hinge peptide (double thread) single thread Met analog S-S bridged tetrapeptide (H-Gly-Cys-OH) 2 Model of an entire human IgG1 molecule. 1 1 Padlan E.A, Mol. Immunol. 31 (1994) 169.
49 poly(l-pro) (H-Thr-Cys-Pro-Pro-Cys-Pro-Ala-Pro-OH) 2 H-Thr-Met-Pro-Pro-Met-Pro-Ala-Pro-OH (H-Gly-Cys-OH) wavenumber (cm -1 ) I R -I L (x1-6 ) I R -I L (x1-6 ) I R -I L (x1-6 ) I R -I L (x1-6 ) (H-Thr-Cys-Pro-Pro-Cys-Pro-Ala-Pro-OH) 2 H-Thr-Met-Pro-Pro-Met-Pro-Ala-Pro-OH poly(l-pro) (H-Gly-Cys-OH) 2 Amide III wavenumber (cm -1 ) I R +I L (x1-9 ) I R +I L (x1-9 ) I R +I L (x1-9 ) I R +I L (x1-9 ) ν(s-s) ν(c-s) δ(ch2 ) 1453 δ(ch 2 ) ν(s-s) 657 ν(c-s) Amide I δ(ch2 ) 1454 Amide I Amide I 1685
50 Summary successful reconstruction of ROA spectrometer spectra of selected biomolecular systems were successfully measured and interpreted new approaches to simulation of Raman and ROA spectra were developed it is essential to take into account dynamic factors for successful simulation conformational flexibility of molecules affect significantly Raman and ROA spectra and can offer good estimate of system rigidity or flexibility previously neglected low energy vibrations (below 6 cm -1 ) are very sensitive to conformational changes and can give new insight into molecular flexibility.
51 Acknowledgement Josef Kapitán Petr Bouř Jiří Bok Petr Praus Petr Maloň Jaroslav Šebestík Hana Hulačová GAČR, GAUK, MŠMT (výzkumný záměr)
52
Raman Optical Activity Comes of Age
 Raman Optical Activity Comes of Age Laurence D. Barron Department of Chemistry, University of Glasgow, Glasgow G12 8QQ, UK E-mail: laurence.barron@glasgow.ac.uk Raman optical activity (ROA) provides vibrational
Raman Optical Activity Comes of Age Laurence D. Barron Department of Chemistry, University of Glasgow, Glasgow G12 8QQ, UK E-mail: laurence.barron@glasgow.ac.uk Raman optical activity (ROA) provides vibrational
Chem 524 Lecture Notes CD (Section 18) update 2011
 Chem 5 Lecture Notes CD (Section 8) update For HTML of 5 notes, click here XV. Circular Dichroism A. Differential absorption of left and right circular polarized light by molecular transition. Measure
Chem 5 Lecture Notes CD (Section 8) update For HTML of 5 notes, click here XV. Circular Dichroism A. Differential absorption of left and right circular polarized light by molecular transition. Measure
INTRODUCTION TO MODERN VIBRATIONAL SPECTROSCOPY
 INTRODUCTION TO MODERN VIBRATIONAL SPECTROSCOPY MAX DIEM Department of Chemistry City University of New York Hunter College A Wiley-Interscience Publication JOHN WILEY & SONS New York Chichester Brisbane
INTRODUCTION TO MODERN VIBRATIONAL SPECTROSCOPY MAX DIEM Department of Chemistry City University of New York Hunter College A Wiley-Interscience Publication JOHN WILEY & SONS New York Chichester Brisbane
MEASUREMENT AND CALCULATION OF THE RAMAN OPTICAL ACTIVITY OF α-pinene AND trans-pinane
 1384 Bour, Baumruk, Hanzlikova: MEASUREMENT AND CALCULATION OF THE RAMAN OPTICAL ACTIVITY OF α-pinene AND trans-pinane Petr BOUR a, Vladimir BAUMRUK b1 and Jana HANZLIKOVA b2 a Institute of Organic Chemistry
1384 Bour, Baumruk, Hanzlikova: MEASUREMENT AND CALCULATION OF THE RAMAN OPTICAL ACTIVITY OF α-pinene AND trans-pinane Petr BOUR a, Vladimir BAUMRUK b1 and Jana HANZLIKOVA b2 a Institute of Organic Chemistry
Infrared Spectroscopy: Identification of Unknown Substances
 Infrared Spectroscopy: Identification of Unknown Substances Suppose a white powder is one of the four following molecules. How can they be differentiated? H N N H H H H Na H H H H H A technique that is
Infrared Spectroscopy: Identification of Unknown Substances Suppose a white powder is one of the four following molecules. How can they be differentiated? H N N H H H H Na H H H H H A technique that is
Skoog Chapter 6 Introduction to Spectrometric Methods
 Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
Layout. Introduction -! Why, o why? -! What & how?
 Gas Phase IR spectroscopy: towards large biomolecules & far-ir wavelengths Anouk M. Rijs Layout Introduction -! Why, o why? -! What & how? Experimental methods -! Cooling methods -! Why cooling -! Supersonic
Gas Phase IR spectroscopy: towards large biomolecules & far-ir wavelengths Anouk M. Rijs Layout Introduction -! Why, o why? -! What & how? Experimental methods -! Cooling methods -! Why cooling -! Supersonic
Infrared spectra of small biomolecules from first-principle molecular dynamics simulations and effective normal mode analysis
 Infrared spectra of small biomolecules from first-principle molecular dynamics simulations and effective normal mode analysis R. Vuilleumier, M.-P. Gaigeot and D. Borgis Département de chimie, Ecole Normale
Infrared spectra of small biomolecules from first-principle molecular dynamics simulations and effective normal mode analysis R. Vuilleumier, M.-P. Gaigeot and D. Borgis Département de chimie, Ecole Normale
Chiral Sum Frequency Generation for In Situ Probing Proton Exchange in Antiparallel β-sheets at Interfaces
 Supporting Information for Chiral Sum Freuency Generation for In Situ Probing Proton Exchange in Antiparallel β-sheets at Interfaces Li Fu, Deuan Xiao, Zhuguang Wang, Victor S. Batista *, and Elsa C. Y.
Supporting Information for Chiral Sum Freuency Generation for In Situ Probing Proton Exchange in Antiparallel β-sheets at Interfaces Li Fu, Deuan Xiao, Zhuguang Wang, Victor S. Batista *, and Elsa C. Y.
Introduction to Comparative Protein Modeling. Chapter 4 Part I
 Introduction to Comparative Protein Modeling Chapter 4 Part I 1 Information on Proteins Each modeling study depends on the quality of the known experimental data. Basis of the model Search in the literature
Introduction to Comparative Protein Modeling Chapter 4 Part I 1 Information on Proteins Each modeling study depends on the quality of the known experimental data. Basis of the model Search in the literature
7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text , , 12.10)
 2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
4. Circular Dichroism - Spectroscopy
 4. Circular Dichroism - Spectroscopy The optical rotatory dispersion (ORD) and the circular dichroism (CD) are special variations of absorption spectroscopy in the UV and VIS region of the spectrum. The
4. Circular Dichroism - Spectroscopy The optical rotatory dispersion (ORD) and the circular dichroism (CD) are special variations of absorption spectroscopy in the UV and VIS region of the spectrum. The
What happens when light falls on a material? Transmission Reflection Absorption Luminescence. Elastic Scattering Inelastic Scattering
 Raman Spectroscopy What happens when light falls on a material? Transmission Reflection Absorption Luminescence Elastic Scattering Inelastic Scattering Raman, Fluorescence and IR Scattering Absorption
Raman Spectroscopy What happens when light falls on a material? Transmission Reflection Absorption Luminescence Elastic Scattering Inelastic Scattering Raman, Fluorescence and IR Scattering Absorption
Chapter 4: Amino Acids
 Chapter 4: Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. lipid polysaccharide enzyme 1940s 1980s. Lipids membrane 1960s. Polysaccharide Are energy metabolites and many of
Chapter 4: Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. lipid polysaccharide enzyme 1940s 1980s. Lipids membrane 1960s. Polysaccharide Are energy metabolites and many of
Parag Mukhopadhyay. Department of Chemistry Duke University. Date: Approved: Prof. David N. Beratan, Supervisor. Prof. Weitao Yang. Prof.
 Theoretical Simulations of Optical Rotation and Raman Optical Activity of Molecules in Solution by Parag Mukhopadhyay Department of Chemistry Duke University Date: Approved: Prof. David N. Beratan, Supervisor
Theoretical Simulations of Optical Rotation and Raman Optical Activity of Molecules in Solution by Parag Mukhopadhyay Department of Chemistry Duke University Date: Approved: Prof. David N. Beratan, Supervisor
Infrared Spectroscopy
 Infrared Spectroscopy Introduction Spectroscopy is an analytical technique which helps determine structure. It destroys little or no sample. The amount of light absorbed by the sample is measured as wavelength
Infrared Spectroscopy Introduction Spectroscopy is an analytical technique which helps determine structure. It destroys little or no sample. The amount of light absorbed by the sample is measured as wavelength
Chem 524 Lecture Notes Raman (Section 17) 2013
 Chem 524 Lecture Notes Raman (Section 17) 2013 For HTML of 2005 notes, click here XIII. Molecular Light Scattering and Raman Spectroscopy (Read Ch. 16) A. Elastic Scattering o = s - basis for Dynamic Light
Chem 524 Lecture Notes Raman (Section 17) 2013 For HTML of 2005 notes, click here XIII. Molecular Light Scattering and Raman Spectroscopy (Read Ch. 16) A. Elastic Scattering o = s - basis for Dynamic Light
Spectroscopy in Inorganic Chemistry. Vibration and Rotation Spectroscopy
 Spectroscopy in Inorganic Chemistry Symmetry requirement for coupling combination bands and Fermi resonance 2 3 V 3 1505 cm -1 (R, IR) E' stretches v 1 888 cm -1 (R) A 1 ' stretch V 2 718 cm -1 (IR) A
Spectroscopy in Inorganic Chemistry Symmetry requirement for coupling combination bands and Fermi resonance 2 3 V 3 1505 cm -1 (R, IR) E' stretches v 1 888 cm -1 (R) A 1 ' stretch V 2 718 cm -1 (IR) A
Introduction. The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants
 Introduction The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants Spectroscopy and the Electromagnetic Spectrum Unlike mass spectrometry,
Introduction The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants Spectroscopy and the Electromagnetic Spectrum Unlike mass spectrometry,
Chemistry 524--Final Exam--Keiderling May 4, :30 -?? pm SES
 Chemistry 524--Final Exam--Keiderling May 4, 2011 3:30 -?? pm -- 4286 SES Please answer all questions in the answer book provided. Calculators, rulers, pens and pencils are permitted. No open books or
Chemistry 524--Final Exam--Keiderling May 4, 2011 3:30 -?? pm -- 4286 SES Please answer all questions in the answer book provided. Calculators, rulers, pens and pencils are permitted. No open books or
Vladimír Baumruk Charles University in Prague Faculty of Mathematics and Physics Institute of Physics
 Spectral Techniques for Rapid Quantification of Protein Structure in Solution Vladimír Baumruk Charles University in Prague Faculty of Mathematics and Physics Institute of Physics primary structure Protein
Spectral Techniques for Rapid Quantification of Protein Structure in Solution Vladimír Baumruk Charles University in Prague Faculty of Mathematics and Physics Institute of Physics primary structure Protein
requency generation spectroscopy Rahul N
 requency generation spectroscopy Rahul N 2-11-2013 Sum frequency generation spectroscopy Sum frequency generation spectroscopy (SFG) is a technique used to analyze surfaces and interfaces. SFG was first
requency generation spectroscopy Rahul N 2-11-2013 Sum frequency generation spectroscopy Sum frequency generation spectroscopy (SFG) is a technique used to analyze surfaces and interfaces. SFG was first
Molecular Mechanics. I. Quantum mechanical treatment of molecular systems
 Molecular Mechanics I. Quantum mechanical treatment of molecular systems The first principle approach for describing the properties of molecules, including proteins, involves quantum mechanics. For example,
Molecular Mechanics I. Quantum mechanical treatment of molecular systems The first principle approach for describing the properties of molecules, including proteins, involves quantum mechanics. For example,
/2Mα 2 α + V n (R)] χ (R) = E υ χ υ (R)
![/2Mα 2 α + V n (R)] χ (R) = E υ χ υ (R) /2Mα 2 α + V n (R)] χ (R) = E υ χ υ (R)](/thumbs/75/72875547.jpg) Spectroscopy: Engel Chapter 18 XIV 67 Vibrational Spectroscopy (Typically IR and Raman) Born-Oppenheimer approx. separate electron-nuclear Assume elect-nuclear motion separate, full wave fct. ψ (r,r) =
Spectroscopy: Engel Chapter 18 XIV 67 Vibrational Spectroscopy (Typically IR and Raman) Born-Oppenheimer approx. separate electron-nuclear Assume elect-nuclear motion separate, full wave fct. ψ (r,r) =
Vibrational Spectral Simulation for Peptides of Mixed Secondary Structure: Method Comparisons with the Trpzip Model Hairpin
 J. Phys. Chem. B 2005, 109, 23687-23697 23687 Vibrational Spectral Simulation for Peptides of Mixed Secondary Structure: Method Comparisons with the Trpzip Model Hairpin Petr Bouř*, and Timothy A. Keiderling*,
J. Phys. Chem. B 2005, 109, 23687-23697 23687 Vibrational Spectral Simulation for Peptides of Mixed Secondary Structure: Method Comparisons with the Trpzip Model Hairpin Petr Bouř*, and Timothy A. Keiderling*,
Fluorescence 2009 update
 XV 74 Fluorescence 2009 update Jablonski diagram Where does the energy go? Can be viewed like multistep kinetic pathway 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet
XV 74 Fluorescence 2009 update Jablonski diagram Where does the energy go? Can be viewed like multistep kinetic pathway 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet
Computational and spectroscopic investigation of 7-azaindole: Solvation and intermolecular interactions
 Computational and spectroscopic investigation of 7-azaindole: Solvation and intermolecular interactions Michael Kamrath, Krista Cruse, Nathan Erickson, Molly Beernink Abstract We report results of an experimental
Computational and spectroscopic investigation of 7-azaindole: Solvation and intermolecular interactions Michael Kamrath, Krista Cruse, Nathan Erickson, Molly Beernink Abstract We report results of an experimental
24/ Rayleigh and Raman scattering. Stokes and anti-stokes lines. Rotational Raman spectroscopy. Polarizability ellipsoid. Selection rules.
 Subject Chemistry Paper No and Title Module No and Title Module Tag 8/ Physical Spectroscopy 24/ Rayleigh and Raman scattering. Stokes and anti-stokes lines. Rotational Raman spectroscopy. Polarizability
Subject Chemistry Paper No and Title Module No and Title Module Tag 8/ Physical Spectroscopy 24/ Rayleigh and Raman scattering. Stokes and anti-stokes lines. Rotational Raman spectroscopy. Polarizability
Molecular Modeling lecture 2
 Molecular Modeling 2018 -- lecture 2 Topics 1. Secondary structure 3. Sequence similarity and homology 2. Secondary structure prediction 4. Where do protein structures come from? X-ray crystallography
Molecular Modeling 2018 -- lecture 2 Topics 1. Secondary structure 3. Sequence similarity and homology 2. Secondary structure prediction 4. Where do protein structures come from? X-ray crystallography
Vibrational states of molecules. Diatomic molecules Polyatomic molecules
 Vibrational states of molecules Diatomic molecules Polyatomic molecules Diatomic molecules V v 1 v 0 Re Q Birge-Sponer plot The solution of the Schrödinger equation can be solved analytically for the
Vibrational states of molecules Diatomic molecules Polyatomic molecules Diatomic molecules V v 1 v 0 Re Q Birge-Sponer plot The solution of the Schrödinger equation can be solved analytically for the
Spectroscopy: Tinoco Chapter 10 (but vibration, Ch.9)
 Spectroscopy: Tinoco Chapter 10 (but vibration, Ch.9) XIV 67 Vibrational Spectroscopy (Typical for IR and Raman) Born-Oppenheimer separate electron-nuclear motion ψ (rr) = χ υ (R) φ el (r,r) -- product
Spectroscopy: Tinoco Chapter 10 (but vibration, Ch.9) XIV 67 Vibrational Spectroscopy (Typical for IR and Raman) Born-Oppenheimer separate electron-nuclear motion ψ (rr) = χ υ (R) φ el (r,r) -- product
ADVANCED VIBRATIONAL SPECTROSCOPIC STUDIES OF BIOLOGICAL MOLECULES
 ADVANCED VIBRATIONAL SPECTROSCOPIC STUDIES OF BIOLOGICAL MOLECULES A thesis submitted to the University of Manchester for the degree of Doctor in Philosophy in the Faculty of Life Sciences 2012 Saeideh
ADVANCED VIBRATIONAL SPECTROSCOPIC STUDIES OF BIOLOGICAL MOLECULES A thesis submitted to the University of Manchester for the degree of Doctor in Philosophy in the Faculty of Life Sciences 2012 Saeideh
IR Spectrography - Absorption. Raman Spectrography - Scattering. n 0 n M - Raman n 0 - Rayleigh
 RAMAN SPECTROSCOPY Scattering Mid-IR and NIR require absorption of radiation from a ground level to an excited state, requires matching of radiation from source with difference in energy states. Raman
RAMAN SPECTROSCOPY Scattering Mid-IR and NIR require absorption of radiation from a ground level to an excited state, requires matching of radiation from source with difference in energy states. Raman
Sequential resonance assignments in (small) proteins: homonuclear method 2º structure determination
 Lecture 9 M230 Feigon Sequential resonance assignments in (small) proteins: homonuclear method 2º structure determination Reading resources v Roberts NMR of Macromolecules, Chap 4 by Christina Redfield
Lecture 9 M230 Feigon Sequential resonance assignments in (small) proteins: homonuclear method 2º structure determination Reading resources v Roberts NMR of Macromolecules, Chap 4 by Christina Redfield
Visible and IR Absorption Spectroscopy. Andrew Rouff and Kyle Chau
 Visible and IR Absorption Spectroscopy Andrew Rouff and Kyle Chau The Basics wavelength= (λ) original intensity= Ι o sample slab thickness= dl Final intensity= I f ε = molar extinction coefficient -di=
Visible and IR Absorption Spectroscopy Andrew Rouff and Kyle Chau The Basics wavelength= (λ) original intensity= Ι o sample slab thickness= dl Final intensity= I f ε = molar extinction coefficient -di=
Application of IR Raman Spectroscopy
 Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Solutions and Non-Covalent Binding Forces
 Chapter 3 Solutions and Non-Covalent Binding Forces 3.1 Solvent and solution properties Molecules stick together using the following forces: dipole-dipole, dipole-induced dipole, hydrogen bond, van der
Chapter 3 Solutions and Non-Covalent Binding Forces 3.1 Solvent and solution properties Molecules stick together using the following forces: dipole-dipole, dipole-induced dipole, hydrogen bond, van der
ion mobility spectrometry IR spectroscopy
 Debasmita Gho 29.10.2016 Introducti on Owing to its accuracy, sensitivity, and speed, mass spectrometry (MS) coupled to fragmentation techniques is the method of choice for determining the primary structure
Debasmita Gho 29.10.2016 Introducti on Owing to its accuracy, sensitivity, and speed, mass spectrometry (MS) coupled to fragmentation techniques is the method of choice for determining the primary structure
Vibrational Spectroscopy
 Vibrational Spectroscopy In this part of the course we will look at the kind of spectroscopy which uses light to excite the motion of atoms. The forces required to move atoms are smaller than those required
Vibrational Spectroscopy In this part of the course we will look at the kind of spectroscopy which uses light to excite the motion of atoms. The forces required to move atoms are smaller than those required
Specialized Raman Techniques. Strictly speaking the radiation-induced dipole moment should be expressed as
 Nonlinear effects Specialized Raman Techniques Strictly speaking the radiation-induced dipole moment should be expressed as M = E + ½E 2 + (1/6)E 3 +... Where and are the first and second hyperpolarizabilities.
Nonlinear effects Specialized Raman Techniques Strictly speaking the radiation-induced dipole moment should be expressed as M = E + ½E 2 + (1/6)E 3 +... Where and are the first and second hyperpolarizabilities.
Radiant energy is proportional to its frequency (cycles/s = Hz) as a wave (Amplitude is its height) Different types are classified by frequency or
 CHEM 241 UNIT 5: PART B INFRA-RED RED SPECTROSCOPY 1 Spectroscopy of the Electromagnetic Spectrum Radiant energy is proportional to its frequency (cycles/s = Hz) as a wave (Amplitude is its height) Different
CHEM 241 UNIT 5: PART B INFRA-RED RED SPECTROSCOPY 1 Spectroscopy of the Electromagnetic Spectrum Radiant energy is proportional to its frequency (cycles/s = Hz) as a wave (Amplitude is its height) Different
Exam I Answer Key: Summer 2006, Semester C
 1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
What is spectroscopy?
 Absorption Spectrum What is spectroscopy? Studying the properties of matter through its interaction with different frequency components of the electromagnetic spectrum. With light, you aren t looking directly
Absorption Spectrum What is spectroscopy? Studying the properties of matter through its interaction with different frequency components of the electromagnetic spectrum. With light, you aren t looking directly
Welcome to Organic Chemistry II
 Welcome to Organic Chemistry II Erika Bryant, Ph.D. erika.bryant@hccs.edu Class Syllabus 3 CHAPTER 12: STRUCTURE DETERMINATION 4 What is this solution Soda Tea Coffee??? 5 What is this solution Soda Tea
Welcome to Organic Chemistry II Erika Bryant, Ph.D. erika.bryant@hccs.edu Class Syllabus 3 CHAPTER 12: STRUCTURE DETERMINATION 4 What is this solution Soda Tea Coffee??? 5 What is this solution Soda Tea
Circular Dichroism & Optical Rotatory Dispersion. Proteins (KCsa) Polysaccharides (agarose) DNA CHEM 305. Many biomolecules are α-helical!
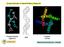 Circular Dichroism & Optical Rotatory Dispersion Polysaccharides (agarose) DNA Proteins (KCsa) Many biomolecules are α-helical! How can we measure the amount and changes in amount of helical structure
Circular Dichroism & Optical Rotatory Dispersion Polysaccharides (agarose) DNA Proteins (KCsa) Many biomolecules are α-helical! How can we measure the amount and changes in amount of helical structure
V( x) = V( 0) + dv. V( x) = 1 2
 Spectroscopy 1: rotational and vibrational spectra The vibrations of diatomic molecules Molecular vibrations Consider a typical potential energy curve for a diatomic molecule. In regions close to R e (at
Spectroscopy 1: rotational and vibrational spectra The vibrations of diatomic molecules Molecular vibrations Consider a typical potential energy curve for a diatomic molecule. In regions close to R e (at
Chemistry 431. NC State University. Lecture 17. Vibrational Spectroscopy
 Chemistry 43 Lecture 7 Vibrational Spectroscopy NC State University The Dipole Moment Expansion The permanent dipole moment of a molecule oscillates about an equilibrium value as the molecule vibrates.
Chemistry 43 Lecture 7 Vibrational Spectroscopy NC State University The Dipole Moment Expansion The permanent dipole moment of a molecule oscillates about an equilibrium value as the molecule vibrates.
Timescales of Protein Dynamics
 Timescales of Protein Dynamics From Henzler-Wildman and Kern, Nature 2007 Summary of 1D Experiment time domain data Fourier Transform (FT) frequency domain data or Transverse Relaxation Ensemble of Nuclear
Timescales of Protein Dynamics From Henzler-Wildman and Kern, Nature 2007 Summary of 1D Experiment time domain data Fourier Transform (FT) frequency domain data or Transverse Relaxation Ensemble of Nuclear
Physiochemical Properties of Residues
 Physiochemical Properties of Residues Various Sources C N Cα R Slide 1 Conformational Propensities Conformational Propensity is the frequency in which a residue adopts a given conformation (in a polypeptide)
Physiochemical Properties of Residues Various Sources C N Cα R Slide 1 Conformational Propensities Conformational Propensity is the frequency in which a residue adopts a given conformation (in a polypeptide)
PAPER No. : 8 (PHYSICAL SPECTROSCOPY) MODULE No. : 5 (TRANSITION PROBABILITIES AND TRANSITION DIPOLE MOMENT. OVERVIEW OF SELECTION RULES)
 Subject Chemistry Paper No and Title Module No and Title Module Tag 8 and Physical Spectroscopy 5 and Transition probabilities and transition dipole moment, Overview of selection rules CHE_P8_M5 TABLE
Subject Chemistry Paper No and Title Module No and Title Module Tag 8 and Physical Spectroscopy 5 and Transition probabilities and transition dipole moment, Overview of selection rules CHE_P8_M5 TABLE
Figure 1. Molecules geometries of 5021 and Each neutral group in CHARMM topology was grouped in dash circle.
 Project I Chemistry 8021, Spring 2005/2/23 This document was turned in by a student as a homework paper. 1. Methods First, the cartesian coordinates of 5021 and 8021 molecules (Fig. 1) are generated, in
Project I Chemistry 8021, Spring 2005/2/23 This document was turned in by a student as a homework paper. 1. Methods First, the cartesian coordinates of 5021 and 8021 molecules (Fig. 1) are generated, in
Contents. xiii. Preface v
 Contents Preface Chapter 1 Biological Macromolecules 1.1 General PrincipIes 1.1.1 Macrornolecules 1.2 1.1.2 Configuration and Conformation Molecular lnteractions in Macromolecular Structures 1.2.1 Weak
Contents Preface Chapter 1 Biological Macromolecules 1.1 General PrincipIes 1.1.1 Macrornolecules 1.2 1.1.2 Configuration and Conformation Molecular lnteractions in Macromolecular Structures 1.2.1 Weak
CHEM Atomic and Molecular Spectroscopy
 CHEM 21112 Atomic and Molecular Spectroscopy References: 1. Fundamentals of Molecular Spectroscopy by C.N. Banwell 2. Physical Chemistry by P.W. Atkins Dr. Sujeewa De Silva Sub topics Light and matter
CHEM 21112 Atomic and Molecular Spectroscopy References: 1. Fundamentals of Molecular Spectroscopy by C.N. Banwell 2. Physical Chemistry by P.W. Atkins Dr. Sujeewa De Silva Sub topics Light and matter
Chapter 12 Mass Spectrometry and Infrared Spectroscopy
 Organic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 12 Mass Spectrometry and Infrared Spectroscopy Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice
Organic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 12 Mass Spectrometry and Infrared Spectroscopy Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice
6.2 Polyatomic Molecules
 6.2 Polyatomic Molecules 6.2.1 Group Vibrations An N-atom molecule has 3N - 5 normal modes of vibrations if it is linear and 3N 6 if it is non-linear. Lissajous motion A polyatomic molecule undergoes a
6.2 Polyatomic Molecules 6.2.1 Group Vibrations An N-atom molecule has 3N - 5 normal modes of vibrations if it is linear and 3N 6 if it is non-linear. Lissajous motion A polyatomic molecule undergoes a
What makes a good graphene-binding peptide? Adsorption of amino acids and peptides at aqueous graphene interfaces: Electronic Supplementary
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 21 What makes a good graphene-binding peptide? Adsorption of amino acids and
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 21 What makes a good graphene-binding peptide? Adsorption of amino acids and
J.Phys. & Theo.Chem.I.A.U. Iran M.Monajjemi et al. Vol.4, No.1, Spring 2007
 Journal of Physical & Theoretical Chemistry Islamic Azad University of Iran 4 (1) (27) Science and Research Campus ISSN: 1735-2126 AB Initio Calculations and IR Studies of Tautometric forms of Uracil and
Journal of Physical & Theoretical Chemistry Islamic Azad University of Iran 4 (1) (27) Science and Research Campus ISSN: 1735-2126 AB Initio Calculations and IR Studies of Tautometric forms of Uracil and
APPLICATIONS OF RAMAN AND AND BIOSYSTEMS APPLICATION: WESLEY THOMPSON JULY 17 TH, 2008
 APPLICATIONS OF RAMAN AND MINIATURIZATION IN INDUSTRIAL AND BIOSYSTEMS APPLICATION: BRIAN MARQUARDT CPAC SUMMER INSTITUTE WESLEY THOMPSON JULY 17 TH, 2008 Applied Optical Sensing Lab Raman Sampling Applications
APPLICATIONS OF RAMAN AND MINIATURIZATION IN INDUSTRIAL AND BIOSYSTEMS APPLICATION: BRIAN MARQUARDT CPAC SUMMER INSTITUTE WESLEY THOMPSON JULY 17 TH, 2008 Applied Optical Sensing Lab Raman Sampling Applications
Supporting Information
 Supporting Information Micelle-Triggered b-hairpin to a-helix Transition in a 14-Residue Peptide from a Choline-Binding Repeat of the Pneumococcal Autolysin LytA HØctor Zamora-Carreras, [a] Beatriz Maestro,
Supporting Information Micelle-Triggered b-hairpin to a-helix Transition in a 14-Residue Peptide from a Choline-Binding Repeat of the Pneumococcal Autolysin LytA HØctor Zamora-Carreras, [a] Beatriz Maestro,
Modern Optical Spectroscopy
 Modern Optical Spectroscopy With Exercises and Examples from Biophysics and Biochemistry von William W Parson 1. Auflage Springer-Verlag Berlin Heidelberg 2006 Verlag C.H. Beck im Internet: www.beck.de
Modern Optical Spectroscopy With Exercises and Examples from Biophysics and Biochemistry von William W Parson 1. Auflage Springer-Verlag Berlin Heidelberg 2006 Verlag C.H. Beck im Internet: www.beck.de
Photoelectron Spectroscopy of the Hydroxymethoxide Anion, H 2 C(OH)O
 Supplementary Material for: Photoelectron Spectroscopy of the Hydroxymethoxide Anion, H 2 C(OH)O Allan M. Oliveira, Julia H. Lehman, Anne B. McCoy 2 and W. Carl Lineberger JILA and Department of Chemistry
Supplementary Material for: Photoelectron Spectroscopy of the Hydroxymethoxide Anion, H 2 C(OH)O Allan M. Oliveira, Julia H. Lehman, Anne B. McCoy 2 and W. Carl Lineberger JILA and Department of Chemistry
6 NMR Interactions: Zeeman and CSA
 6 NMR Interactions: Zeeman and CSA 6.1 Zeeman Interaction Up to this point, we have mentioned a number of NMR interactions - Zeeman, quadrupolar, dipolar - but we have not looked at the nature of these
6 NMR Interactions: Zeeman and CSA 6.1 Zeeman Interaction Up to this point, we have mentioned a number of NMR interactions - Zeeman, quadrupolar, dipolar - but we have not looked at the nature of these
Probing and Driving Molecular Dynamics with Femtosecond Pulses
 Miroslav Kloz Probing and Driving Molecular Dynamics with Femtosecond Pulses (wavelengths above 200 nm, energies below mj) Why femtosecond lasers in biology? Scales of size and time are closely rerated!
Miroslav Kloz Probing and Driving Molecular Dynamics with Femtosecond Pulses (wavelengths above 200 nm, energies below mj) Why femtosecond lasers in biology? Scales of size and time are closely rerated!
Lecture 6: Physical Methods II. UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy
 Lecture 6: Physical Methods II UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy Physical Methods used in bioinorganic chemistry X ray crystallography X ray absorption (XAS)
Lecture 6: Physical Methods II UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy Physical Methods used in bioinorganic chemistry X ray crystallography X ray absorption (XAS)
Application of Raman Spectroscopy for Noninvasive Detection of Target Compounds. Kyung-Min Lee
 Application of Raman Spectroscopy for Noninvasive Detection of Target Compounds Kyung-Min Lee Office of the Texas State Chemist, Texas AgriLife Research January 24, 2012 OTSC Seminar OFFICE OF THE TEXAS
Application of Raman Spectroscopy for Noninvasive Detection of Target Compounds Kyung-Min Lee Office of the Texas State Chemist, Texas AgriLife Research January 24, 2012 OTSC Seminar OFFICE OF THE TEXAS
CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray
 CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray crystallography An example of the use of CD Modeling
CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray crystallography An example of the use of CD Modeling
2. Infrared spectroscopy
 2. Infrared spectroscopy 2-1Theoretical principles An important tool of the organic chemist is Infrared Spectroscopy, or IR. IR spectra are acquired on a special instrument, called an IR spectrometer.
2. Infrared spectroscopy 2-1Theoretical principles An important tool of the organic chemist is Infrared Spectroscopy, or IR. IR spectra are acquired on a special instrument, called an IR spectrometer.
Peptides And Proteins
 Kevin Burgess, May 3, 2017 1 Peptides And Proteins from chapter(s) in the recommended text A. Introduction B. omenclature And Conventions by amide bonds. on the left, right. 2 -terminal C-terminal triglycine
Kevin Burgess, May 3, 2017 1 Peptides And Proteins from chapter(s) in the recommended text A. Introduction B. omenclature And Conventions by amide bonds. on the left, right. 2 -terminal C-terminal triglycine
Other Methods for Generating Ions 1. MALDI matrix assisted laser desorption ionization MS 2. Spray ionization techniques 3. Fast atom bombardment 4.
 Other Methods for Generating Ions 1. MALDI matrix assisted laser desorption ionization MS 2. Spray ionization techniques 3. Fast atom bombardment 4. Field Desorption 5. MS MS techniques Matrix assisted
Other Methods for Generating Ions 1. MALDI matrix assisted laser desorption ionization MS 2. Spray ionization techniques 3. Fast atom bombardment 4. Field Desorption 5. MS MS techniques Matrix assisted
Protein Structure Basics
 Protein Structure Basics Presented by Alison Fraser, Christine Lee, Pradhuman Jhala, Corban Rivera Importance of Proteins Muscle structure depends on protein-protein interactions Transport across membranes
Protein Structure Basics Presented by Alison Fraser, Christine Lee, Pradhuman Jhala, Corban Rivera Importance of Proteins Muscle structure depends on protein-protein interactions Transport across membranes
Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis)
 Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis) 2 2. Circular dichroism (optical activity) CD / ORD 3 3. Fluorescence spectroscopy and energy transfer Electromagnetic Spectrum Electronic Molecular
Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis) 2 2. Circular dichroism (optical activity) CD / ORD 3 3. Fluorescence spectroscopy and energy transfer Electromagnetic Spectrum Electronic Molecular
Lecture 2 and 3: Review of forces (ctd.) and elementary statistical mechanics. Contributions to protein stability
 Lecture 2 and 3: Review of forces (ctd.) and elementary statistical mechanics. Contributions to protein stability Part I. Review of forces Covalent bonds Non-covalent Interactions: Van der Waals Interactions
Lecture 2 and 3: Review of forces (ctd.) and elementary statistical mechanics. Contributions to protein stability Part I. Review of forces Covalent bonds Non-covalent Interactions: Van der Waals Interactions
Physical principles of IR and Raman. Infrared Spectroscopy
 Physical principles of IR and Raman IR results from the absorption of energy by vibrating chemical bonds. Raman scattering results from the same types of transitions, but the selection rules are different
Physical principles of IR and Raman IR results from the absorption of energy by vibrating chemical bonds. Raman scattering results from the same types of transitions, but the selection rules are different
Spectroscopic Selection Rules
 E 0 v = 0 v = 1 v = 2 v = 4 v = 3 For a vibrational fundamental (Δv = ±1), the transition will have nonzero intensity in either the infrared or Raman spectrum if the appropriate transition moment is nonzero.
E 0 v = 0 v = 1 v = 2 v = 4 v = 3 For a vibrational fundamental (Δv = ±1), the transition will have nonzero intensity in either the infrared or Raman spectrum if the appropriate transition moment is nonzero.
A prevalent intraresidue hydrogen bond stabilizes proteins
 Supplementary Information A prevalent intraresidue hydrogen bond stabilizes proteins Robert W. Newberry 1 & Ronald T. Raines 1,2 * 1 Department of Chemistry and 2 Department of Biochemistry, University
Supplementary Information A prevalent intraresidue hydrogen bond stabilizes proteins Robert W. Newberry 1 & Ronald T. Raines 1,2 * 1 Department of Chemistry and 2 Department of Biochemistry, University
Lecture 11. IR Theory. Next Class: Lecture Problem 4 due Thin-Layer Chromatography
 Lecture 11 IR Theory Next Class: Lecture Problem 4 due Thin-Layer Chromatography This Week In Lab: Ch 6: Procedures 2 & 3 Procedure 4 (outside of lab) Next Week in Lab: Ch 7: PreLab Due Quiz 4 Ch 5 Final
Lecture 11 IR Theory Next Class: Lecture Problem 4 due Thin-Layer Chromatography This Week In Lab: Ch 6: Procedures 2 & 3 Procedure 4 (outside of lab) Next Week in Lab: Ch 7: PreLab Due Quiz 4 Ch 5 Final
Supporting Information. Probing the structure of water oxidizing anodic. Iridium oxide catalyst using Raman spectroscopy
 Supporting Information Probing the structure of water oxidizing anodic Iridium oxide catalyst using Raman spectroscopy Zoran Pavlovic, Chinmoy Ranjan, Qiang Gao, Maurice van Gastel, and Robert Schlögl
Supporting Information Probing the structure of water oxidizing anodic Iridium oxide catalyst using Raman spectroscopy Zoran Pavlovic, Chinmoy Ranjan, Qiang Gao, Maurice van Gastel, and Robert Schlögl
QUESTION 1 Which two functional groups react to form the peptide link found in proteins?
 QUESTION 1 Which two functional groups react to form the peptide link found in proteins? NH and NH and NH2 and and NH2 A 2 B 2 C D OH COOH OH COOH QUESTION 2 The elements present in proteins are A B C
QUESTION 1 Which two functional groups react to form the peptide link found in proteins? NH and NH and NH2 and and NH2 A 2 B 2 C D OH COOH OH COOH QUESTION 2 The elements present in proteins are A B C
BSc and MSc Degree Examinations
 Examination Candidate Number: Desk Number: BSc and MSc Degree Examinations 2018-9 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes Marking
Examination Candidate Number: Desk Number: BSc and MSc Degree Examinations 2018-9 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes Marking
5.3 Rotational Raman Spectroscopy General Introduction
 5.3 Rotational Raman Spectroscopy 5.3.1 General Introduction When EM radiation falls on atoms or molecules, it may be absorbed or scattered. If λis unchanged, the process is referred as Rayleigh scattering.
5.3 Rotational Raman Spectroscopy 5.3.1 General Introduction When EM radiation falls on atoms or molecules, it may be absorbed or scattered. If λis unchanged, the process is referred as Rayleigh scattering.
Chapter 17: Fundamentals of Spectrophotometry
 Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
Applications of Terahertz Radiation (T-ray) Yao-Chang Lee, National Synchrotron Research Radiation Center
 Applications of Terahertz Radiation (T-ray) Yao-Chang Lee, yclee@nsrrc.org.tw National Synchrotron Research Radiation Center Outline Terahertz radiation (THz) or T-ray The Interaction between T-ray and
Applications of Terahertz Radiation (T-ray) Yao-Chang Lee, yclee@nsrrc.org.tw National Synchrotron Research Radiation Center Outline Terahertz radiation (THz) or T-ray The Interaction between T-ray and
Rotational Raman Spectroscopy
 Rotational Raman Spectroscopy If EM radiation falls upon an atom or molecule, it may be absorbed if the energy of the radiation corresponds to the separation of two energy levels of the atoms or molecules.
Rotational Raman Spectroscopy If EM radiation falls upon an atom or molecule, it may be absorbed if the energy of the radiation corresponds to the separation of two energy levels of the atoms or molecules.
CHEM6416 Theory of Molecular Spectroscopy 2013Jan Spectroscopy frequency dependence of the interaction of light with matter
 CHEM6416 Theory of Molecular Spectroscopy 2013Jan22 1 1. Spectroscopy frequency dependence of the interaction of light with matter 1.1. Absorption (excitation), emission, diffraction, scattering, refraction
CHEM6416 Theory of Molecular Spectroscopy 2013Jan22 1 1. Spectroscopy frequency dependence of the interaction of light with matter 1.1. Absorption (excitation), emission, diffraction, scattering, refraction
Molecular Symmetry. Symmetry is relevant to: spectroscopy, chirality, polarity, Group Theory, Molecular Orbitals
 Molecular Symmetry Symmetry is relevant to: spectroscopy, chirality, polarity, Group Theory, Molecular Orbitals - A molecule has a symmetry element if it is unchanged by a particular symmetry operation
Molecular Symmetry Symmetry is relevant to: spectroscopy, chirality, polarity, Group Theory, Molecular Orbitals - A molecule has a symmetry element if it is unchanged by a particular symmetry operation
Reflection = EM strikes a boundary between two media differing in η and bounces back
 Reflection = EM strikes a boundary between two media differing in η and bounces back Incident ray θ 1 θ 2 Reflected ray Medium 1 (air) η = 1.00 Medium 2 (glass) η = 1.50 Specular reflection = situation
Reflection = EM strikes a boundary between two media differing in η and bounces back Incident ray θ 1 θ 2 Reflected ray Medium 1 (air) η = 1.00 Medium 2 (glass) η = 1.50 Specular reflection = situation
Laser induced Temperature jump
 Laser induced Temperature jump IR pulse heats the solvent ( Raman shifted YAG to 1.9 μ for D 2 O) Probe heated spot with tunable IR laser (Pb-salt diode, FTIR experiments proposed) Fast MCT needed for
Laser induced Temperature jump IR pulse heats the solvent ( Raman shifted YAG to 1.9 μ for D 2 O) Probe heated spot with tunable IR laser (Pb-salt diode, FTIR experiments proposed) Fast MCT needed for
Computational and Spectroscopic Investigation of Solution Phase Excited State Dynamics in 7 azaindole
 Computational and Spectroscopic Investigation of Solution Phase Excited State Dynamics in 7 azaindole Nathan Erickson, Molly Beernink, and Nathaniel Swenson Midwest Undergraduate Computational Chemistry
Computational and Spectroscopic Investigation of Solution Phase Excited State Dynamics in 7 azaindole Nathan Erickson, Molly Beernink, and Nathaniel Swenson Midwest Undergraduate Computational Chemistry
Session #1: Theoretical background and computer simulations of molecular vibrations.
 Raman Spectroscopy Session #1: Theoretical background and computer simulations of molecular vibrations. Goals: Understand the origin of the Raman effect. Understand the vibrational normal modes of molecules.
Raman Spectroscopy Session #1: Theoretical background and computer simulations of molecular vibrations. Goals: Understand the origin of the Raman effect. Understand the vibrational normal modes of molecules.
Principles of Physical Biochemistry
 Principles of Physical Biochemistry Kensal E. van Hold e W. Curtis Johnso n P. Shing Ho Preface x i PART 1 MACROMOLECULAR STRUCTURE AND DYNAMICS 1 1 Biological Macromolecules 2 1.1 General Principles
Principles of Physical Biochemistry Kensal E. van Hold e W. Curtis Johnso n P. Shing Ho Preface x i PART 1 MACROMOLECULAR STRUCTURE AND DYNAMICS 1 1 Biological Macromolecules 2 1.1 General Principles
ZAHID IQBAL WARRAICH
 Q1 Chromatography is an important analytical technique in chemistry. There is a number of techniques under the general heading of chromatography. (a) Paper and gas chromatography rely on partition to separate
Q1 Chromatography is an important analytical technique in chemistry. There is a number of techniques under the general heading of chromatography. (a) Paper and gas chromatography rely on partition to separate
MOLECULAR LIGHT SCATTERING AND OPTICAL ACTIVITY
 MOLECULAR LIGHT SCATTERING AND OPTICAL ACTIVITY Second edition, revised and enlarged LAURENCE D. BARRON, F.R.S.E. Gardiner Professor of Chemistry, University of Glasgow 122. CAMBRIDGE UNIVERSITY PRESS
MOLECULAR LIGHT SCATTERING AND OPTICAL ACTIVITY Second edition, revised and enlarged LAURENCE D. BARRON, F.R.S.E. Gardiner Professor of Chemistry, University of Glasgow 122. CAMBRIDGE UNIVERSITY PRESS
Structure Determination. How to determine what compound that you have? One way to determine compound is to get an elemental analysis
 Structure Determination How to determine what compound that you have? ne way to determine compound is to get an elemental analysis -basically burn the compound to determine %C, %H, %, etc. from these percentages
Structure Determination How to determine what compound that you have? ne way to determine compound is to get an elemental analysis -basically burn the compound to determine %C, %H, %, etc. from these percentages
Investigating excited state dynamics in 7 azaindole. Nathan Erickson, Molly Beernink, and Nathaniel Swenson
 Investigating excited state dynamics in 7 azaindole Nathan Erickson, Molly Beernink, and Nathaniel Swenson 1 Background I 7AI Dimer Previous studies have shown that 7 azaindole (7AI) readily forms H bonded
Investigating excited state dynamics in 7 azaindole Nathan Erickson, Molly Beernink, and Nathaniel Swenson 1 Background I 7AI Dimer Previous studies have shown that 7 azaindole (7AI) readily forms H bonded
This is a simple input file for the calculation of NMR chemical shieldings for a given molecule using the B3LYP functional and def2-tzvpp basis set:
 Computing NMR parameters using ORCA This practical comes with a short lecture on the basics of the computation of NMR parameters using standard electronic structure theory methods. By now you should have
Computing NMR parameters using ORCA This practical comes with a short lecture on the basics of the computation of NMR parameters using standard electronic structure theory methods. By now you should have
XV 74. Flouorescence-Polarization-Circular-Dichroism- Jablonski diagram Where does the energy go?
 XV 74 Flouorescence-Polarization-Circular-Dichroism- Jablonski diagram Where does the energy go? 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet S = 0 could be any of them
XV 74 Flouorescence-Polarization-Circular-Dichroism- Jablonski diagram Where does the energy go? 1) Excite system through A Absorbance S 0 S n Excite from ground excited singlet S = 0 could be any of them
Vibrational Spectroscopies. C-874 University of Delaware
 Vibrational Spectroscopies C-874 University of Delaware Vibrational Spectroscopies..everything that living things do can be understood in terms of the jigglings and wigglings of atoms.. R. P. Feymann Vibrational
Vibrational Spectroscopies C-874 University of Delaware Vibrational Spectroscopies..everything that living things do can be understood in terms of the jigglings and wigglings of atoms.. R. P. Feymann Vibrational
Calculate a rate given a species concentration change.
 Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
BioMolecular Optical Spectroscopy:
 BioMolecular Optical Spectroscopy: Part 1: Infrared and Raman Vibrational Spectra Background Special Lectures for Chem 344 Fall, 2007 im Keiderling University of Illinois at Chicago tak@uic.edu Vibrational
BioMolecular Optical Spectroscopy: Part 1: Infrared and Raman Vibrational Spectra Background Special Lectures for Chem 344 Fall, 2007 im Keiderling University of Illinois at Chicago tak@uic.edu Vibrational
The Physical Basis of the NMR Experiment
 The Physical Basis of the NMR Experiment 1 Interaction of Materials with Magnetic Fields F F S N S N Paramagnetism Diamagnetism 2 Microscopic View: Single Spins an electron has mass and charge in addition
The Physical Basis of the NMR Experiment 1 Interaction of Materials with Magnetic Fields F F S N S N Paramagnetism Diamagnetism 2 Microscopic View: Single Spins an electron has mass and charge in addition
