Slow cooling of the lowermost oceanic crust at the fastspreading
|
|
|
- Jessie Warren
- 6 years ago
- Views:
Transcription
1 GSA Data Repository Slow cooling of the lowermost oceanic crust at the fastspreading East Pacific Rise Kathrin Faak and Kathryn M. Gillis Model parameters and input conditions for the diffusion models Ca-in-olivine The Ca-in-olivine geospeedometer is based on the diffusive exchange of Ca between olivine and clinopyroxene during cooling, and cooling rates are determined from the measured Ca-concentration profiles in olivine. A detailed description of the Ca-in-olivine geospeedometer may be found in Coogan et al. (2002 and 2007), here we briefly summarize the modeling approach and the input parameters used, and review the inherent uncertainties. The method is based on Dodson s concept of closure temperature Tc (Dodson, 1973), which was extended by Dodson (1986) to the calculation of a closure profile (Tc-profile) to give: Tc x E R ln RTc2 D 0 G x Esa 2, Eq. (DR1) at which R=ideal gas constant, s=cooling rate around the closure temperature Tc, and a=radius of the grain. The parameters E and D 0 refer to activation energy and the pre-exponential factor in the diffusion equation. Here, we used the diffusion data of Coogan et al. (2005) for diffusion along the c-axis and fo 2 =10-12 bar (E= J/mol and D 0 =1*10-10 ). The closure function G(x) depends on the geometry of the cooling object and the position x within it. Here, we used the geometry of a sphere:
2 n 1 sin(n x)ln n z -1 G(x) , Eq. (DR2) n x n 1 with z= As Tc appears on both sides of Eq. (DR1), the equation has to be solved iteratively. The measured Ca-concentration profiles in olivine can be translated into closure temperatures using the thermometer of Köhler and Brey (1990): Tc x 42.5 P 5792 ln C 273, Ol Ca x Cpx 1.25 C Ca Eq. (DR3) olivine, at which P=pressure (here, we assumed P=2kbar), Ol C Ca = measured concentration of Ca in Cpx C Ca =measured concentration of Ca in clinopyroxene (adjacent to the olivine grain). The cooling rate s in Eq. (DR1) is iteratively changed until the best visual fit between the closure profile calculated from Eq. (DR1) and the closure profile calculated from Eq. (DR3) is attained. Uncertainties inherent in the approach Coogan et al. (2005) show that quantifiable systematic inaccuracies in cooling rates obtained from the Ca-in-olivine geospeedometer principally result from uncertainties in the Cpx partition coefficient for Ca between olivine and clinopyroxene ( K / ), and in the diffusion coefficient (D 0 and E) of Ca in olivine. They performed Monte Carlo simulations to evaluate the potential inaccuracies arising from uncertainties in these parameters. Cooling rates for a given Cpx measured Ca profile were calculated 2000 times allowing K /, E and D 0 to vary within given Ol Ca Ol Ca
3 bounds. These bounds were taken from the expeental uncertainties in these parameters (Köhler and Brey, 1990 for Ol Cpx K / Ca and Coogan et al., 2005 for D 0 and E). The predicted uncertainty (1) in the cooling rate based on these simulations is a function of cooling rate, being 0.3 log units at a cooling rate of Cyr -1 to 0.2 log units at a cooling rate of 0.1 Cyr -1. We note that Shejwalkar and Coogan (2013) show that the Ca-content in olivine in equilibrium with clinopyroxene additionally depends on the forsterite content (Fo) of the olivine. However, their expeental calibration was carried out in a temperature range of C, and cannot easily be extrapolated to the range of closure temperatures determined in olivine in the samples investigated here (Tc core = C using the calibration of Köhler and Brey, 1990; Table DR2). Equation (13) in Shejwalkar and Coogan (2013) combines their results with the previous expeents by Köhler and Brey (1990) and was used to quantify the effect of the compositional dependence on the closure temperatures and cooling rates of the samples investigated here. It is found that Tc core is increased by 30 C (for Fo=0.79; lowest Fo in investigated sample suite) and by 40 C (for Fo=0.84; highest Fo in investigated sample suite) when the compositional effect is accounted for. The resulting effect on the cooling rate depends on the length of the observed profile, but is below 0.5 orders of magnitude even for the longest profiles investigated here (and below a factor of two for the shortest profiles). Fitting full profiles instead of using the highest Ca concentration measured in one olivine grain VanTongeren et al. (2008) used the Ca-in-olivine geospeedometer to obtain cooling rates from the lower oceanic crust exposed in the Wadi Khafifah section of the Oman ophiolite. Instead of measuring and fitting full profiles (-core-) as recommended by Coogan et al. (2002), these authors only measured between 2-7 (on average 3) analyses per olivine grain and
4 then used the highest measured Ca values from each crystal core for the calculation of closure temperature and cooling rate. We caution that this approach will overestimate the cooling rate and result in inaccurate results. Figure DR2 shows cooling rates obtained from a synthetic Caconcentration profile in olivine. Fitting the full profile results in a cooling rate of Cyr -1. Cases (i), (ii) and (iii) simulate data point affected by alteration (see Fig. DR 2 for details) Following the approach taken by VanTongeren et al. (2008) and using only the data points with highest Ca-concentrations overestimates the cooling rate of this crystal by up to 1.5 orders of magnitude (we note that given the limited number of analyses per grain carried out by VanTongeren et al., 2008, it would not be possible to identify these measurements as outliers from the actual profile shape). To illustrate this point using natural samples, we show full concentration profiles of Ca in olivines from the Wadi Abyad in the Oman ophiolite from Coogan et al. (2002). Fitting these complete profiles with the approach outlined above results in cooling rates between Cyr -1 to Cyr -1, at which cooling rates are decreasing with decreasing sample depth above the Moho (Fig. DR3). Using the approach by VanTongeren et al. (2008) overestimates cooling rates by up to 2 orders of magnitude and shows no systematics of cooling rate with sample depth. Mg-in-plagioclase The Mg-in-plagioclase geospeedometer is based on the diffusive exchange of Mg between plagioclase and clinopyroxene during cooling (Faak et al., 2014). Partition and diffusion coefficients for Mg in plagioclase depend on the composition of the plagioclase (i.e., the anorthite-content), which has to be accounted for in the diffusion equation (Costa et al., 2003). Thus, it is not possible to use Dodson s analytical equation for the Mg-in-plagioclase system. Faak et al. (2014) presented a Mg-in-plagioclase geospeedometer based on a finite difference
5 modeling approach using the diffusion equation of Costa et al. (2003) and applied this method to samples from the oceanic crust (Faak et al., 2014, 2015). A detailed description of the used diffusion model may be found in Faak et al. (2014), here we briefly summarize the modeling approach and the input parameters used. Diffusion equation and diffusion coefficient The diffusion of Mg in plagioclase is coupled with the anorthite content, X An, in plagioclase, which needs to be accounted for in the diffusion equation (Eq. (7) in Costa et al., 2003) and the resulting diffusion equation to describe the flux of Mg has been presented by Costa et al. (2003): C Mg t 2 C Mg D Mg x 2 C Mg x D Mg x A RT D Mg C Mg x X An x C Mg D Mg x X An x D MgC Mg 2 X An x 2 (Eq. DR4) where C Mg =concentration of Mg in plagioclase, t=time, D Mg =diffusion coefficient of Mg in plagioclase, x=distance and A=factor to describe the dependence of the partition coefficient on X An. We used the diffusion coefficient of Mg in plagioclase from Faak et al. (2013) that was determined expeentally in the compositional range of the lower oceanic crust: Pl D Mg m 2 s m 2 s -1 Jmol-1 exp RT a SiO (Eq. DR5) Initial and boundary conditions
6 The partition coefficient, K /, has been determined expeentally by Faak et al. Pl Cpx Mg (2013) and is given by the relationship: Pl lnk /Cpx Mg 9219 K 1 T Jmol1 RT X An ln a SiO2 (Eq. DR6) This can be re-arranged to give the interface plagioclase composition as a function of the measured clinopyroxene composition, temperature and the silica activity in the system: Pl C Mg C Cpx Mg exp 9219 K 1 T Jmol1 RT X An ln a SiO2 (Eq. DR7) An initial profile is calculated based on Eq. (DR7) at temperatures around 1200 C (the exact starting temperature T start depends on the grain size of the plagioclase and is given in Table DR1). Silica activity Solving equations (DR5-DR7) requires knowledge of the silica activity, a SiO2, as a function of temperature. Here, we assume a SiO2 is constrained by the assemblage olivine+orthopyroxene as these are commonly observed phases in the samples studied. Faak et al. (2014 and 2015) show that in this case, the temperature dependent silica activity can be approximated with a polynomial of the form: a SiO T T (Eq. DR8) Uncertainties inherent in the approach As discussed previously for the Ca-in-olivine geospeedometer, the main source of inaccuracy for cooling rates obtained from Mg-in-plagioclase geospeedometer results from
7 uncertainties in the partition coefficient Pl Cpx K / Mg and the diffusion coefficient D Pl Mg. Faat et al. (2014) estimated the effect of uncertainties in these parameters on the cooling rate by performing Monte Carlo simulations. Randomly distributed probability distributions of the parameters were used in simulations, at which the parameters can vary within the bounds of the uncertainties in their calibration (Faak et al., 2013). The predicted uncertainty based on these simulations is 0.3 log units for a cooling rate of 0.1 Cyr -1 and 0.3 log units for a cooling rate of Cyr -1. Faak et al. (2014) also discuss how the uncertainty of the cooling rate is affected if the chosen diffusion coefficient has a significant dependence on X An or not (Costa et al., 2003; Faak et al., 2013; Van Orman et al., 2014). They show that the cooling rate obtained using the diffusion coefficient of Faak et al. (2013) is by a factor of 3 faster for a normally zoned plagioclase and by a factor of 4 slower for an inversely zoned plagioclase, if the diffusion coefficient depends on X An (Van Orman et al., 2014). Additionally, Faak et al. (2015) use the scatter in the data for one sample (i.e., the difference in the obtained cooling rates from multiple Mg-profiles in up to 5 plagioclase crystals in the same sample) as a measure for the precision of the cooling rate estimate for a single sample. Following this approach, they find the precision on the obtained cooling rate to be better than half an order of magnitude. References Coogan, L. A., Jenkin, G. R. T., and Wilson, R. N., 2002, Constraining the cooling rate of the lower oceanic crust: a new approach applied to the Oman ophiolite: Earth and Planetary Science Letters, v. 199, p
8 Coogan, L. A., Hain, A., Stahl, S., and Chakraborty, S., 2005, Expeental determination of the diffusion coefficient for calcium in olivine between 900 C and 1500 C: Geochimica et Cosmochimica Acta, v. 69, p Coogan, L. A., Jenkin, G. R. T., and Wilson, R. N., 2007, Contrasting cooling rates in the oceanic lithosphere at fast- and slow-spreading mid-ocean ridges derived from geospeedometry: Journal of Petrology, v. 48, p Costa, F., Chakraborty, S., and Dohmen, R., 2003, Diffusion coupling between major and trace elements and a model for the calculation of magma chamber residence times using plagioclase: Geochimica et Cosmochimica Acta, v. 67, p Dalton, J. A., and Lane, S. J., 1996, Electron microprobe analysis of Ca in olivine close to grain boundaries: The problem of secondary X-ray fluorescence: American Mineralogist, v. 81, pp Dodson, M. H., 1973, Closure temperature in cooling geochronological and petrological systems: Contributions to. Mineralogy and Petrology, v. 40, pp Dodson, M. H., 1986, Closure profiles in cooling systems: Material Science Forum, v. 7, p Faak, K., Chakraborty, S., Coogan, and L. A., 2013, Mg in plagioclase: Expeental calibration of a new geothermometer and diffusion coefficients: Geochimica et Cosmochimica Acta, v. 123, p Faak., K., Coogan, L. A., and Chakraborty, S., 2014, A new Mg-in-plagioclase geospeedometer for the determination of cooling rates of mafic rocks: Geochimica et Cosmochimica Acta, v. 140, p
9 Faak., K., Coogan, L. A., and Chakraborty, S., 2015, Near conductive cooling rates in the upperplutonic section of crust formed at the East Pacific Rise: Earth and Planetary Science Letters, v. 423, p Köhler, T. P., and Brey, G. P., 1990, Calcium exchange between olivine and clinopyroxene calibrated as a geothermometer for natural peridotites from 2 to 60 kb with applications: Geochimica et Cosmochimica Acta, v. 54, p Shejwalkar, A., and Coogan, L. A., 2013, Expeental calibration of the roles of temperature and composition in the Ca-in-olivine geothermometer at 0.1 MPa: Lithos, v. 177, pp Van Orman J. A., Cherniak D. J. and Kita N. T., 2014, Magnesium diffusion in plagioclase: dependence on composition, and implications for thermal resetting of the 26Al 26Mg early solar system chronometer. Earth and Planetary Science Letters, v. 385, pp VanTongeren, J.A., Kelemen, P.B., and Hanghoj, K., 2008, Cooling rates in the lower crust of the Oman ophiolite: Ca in olivine, revisited: Earth and Planetary Science Letters, v. 267, p
10 Shallow gabbros (Faak et. al, 2015) increasing depth below DGB in m [mbdgb] (a) (17mbDGB) (b) (282 mbdgb) (c) (522 mbdgb) MgO [wt%] 0.15 dt/dt = Cyr 0.10 dt/dt = 0.3 Cyr -1 dt/dt = Cyr core 1000 Distance [µm] core 1000 Distance [µm] core 400 Distance [µm] 600 Deeper plutonics (>2000 mbdgb), Exp 345 Site U1415 (d) P6R2-14 (e) J11R1-26 (f) P11R1-38 MgO [wt%] dt/dt = Cyr-1 dt/dt = Cyr-1 dt/dt = Cyr core 50 Distance [µm] 100 core 70 Distance [µm] 140 core 200 Distance [µm] 400 Figure DR1. Direct comparison of 3 different Mg-profiles in plagioclase for samples from shallow gabbros from increasing depth (in meters below DGB) at the Hess Deep Rift (upper panel, data from Faak et al., 2015), and 3 different Mgprofiles in plagioclase for samples from the deeper plutonics drilled at IODP Site U1415 (lower panel).
11 (iii) Cyr Tc [ C] (ii) Cyr (i) Cyr dt/dt = Cyr Distance [µm] Figure DR2. Cooling rates obtained from fitting a synthetic profile of Ca in olivine (solid line). Cases (i), (ii) and (iii) simulate the effect of secondary fluorescence on measurements of Ca in olivine at a distance of 60 µm, 40 µm and 20 µm away from a CaCO3 vein with 40 wt% Ca (Dalton and Lane, 1996). We note that this is just one possibility of alteration that can be assed quantitatively. Higher values of measured Ca-concentrations in olivine may also result from other forms of alteration or inclusions. Grey dashed lines show profiles from fitting just the highest Ca concentrations and light grey numbers give the obtained cooling rates.
12 Figure DR3. Cooling rates obtained from fitting full concentration profiles of Ca in olivine in three samples from different depths in the Wadi Abyad (lower dt/dt) (Coogan et al., 2002; sample depth is given in m above the Moho) in comparison with cooling rates obtained from using only the highest Ca-concentrations measured near the core of the grain (upper dt/dt). Open symbols were excluded from the modeling approach (see Coogan et al., 2002 for details).
13 TABLE DR1. Summary of the cooling rates that were obtained from the Mg-in-plagioclase geospeedometer. Sample Rock type 1 Depth 2 Crystal Profile Length dt/dt log dt/dt T-interval 3 [m] [µm] [ C/yr] [ C] J10R1-49 troctolite Plag1 P J10R1-49 troctolite Plag2 P J11R1-26 troctolite Plag1 P J11R1-26 troctolite Plag2 P J11R1-26 troctolite Plag3 P J25G1-32 troctolite Plag2 P J25G1-32 troctolite Plag2 P P3R1-31 Ol gabbro Plag2 P P4G1-5 Ol gabbro Plag4 P P4G1-5 Ol gabbro Plag6 P P4G1-10 Ol gabbro Plag1 P P6R2-14 Ol gabbro Plag3 P P11R1-38 Ol gabbro Plag1 P P11R1-38 Ol gabbro Plag2 P P11R1-38 Ol gabbro Plag4 P Ol = olivine 2 Depth is given in m below seafloor. 3 T-interval is the temperature range over which the diffusion process was modeled (see Faak et al., 2014 for details).
14 TABLE DR2. Summary of the cooling rates that were obtained from the Ca-in-Olivine geospeedometer. Sample Rock type 1 Depth 2 Crystal Profile 3 Length dt/dt log dt/dt 4 Tc core [m] [µm] [ C/yr] [ C] J3R1-66 Ol gabbro Ol1 P1 f J3R1-66 Ol gabbro Ol1 P2 h J3R1-66 Ol gabbro Ol2 P1 f J3R1-66 Ol gabbro Ol2 P2 f J3R1-66 Ol gabbro Ol3 P1 f J5R1-131 Ol gabbro Ol2 P1 h J5R1-131 Ol gabbro Ol3 P1 h J5R1-131 Ol gabbro Ol3 P2 h J8R2-79 J8R2-79 J8R2-79 J9R1-13 J9R1-13 Cpx oik ol gabbro Cpx oik ol gabbro Cpx oik ol gabbro Cpx oik ol gabbro Cpx oik ol gabbro Ol1 P1 f Ol2 P1 f Ol3 P1 f Ol1 P1 f Ol2 P2 h J10R1-49 troctolite Ol1 P1 h J10R1-49 troctolite Ol2 P1 f J10R1-49 troctolite Ol2 P2 f J10R1-49 troctolite Ol2 P3 h J10R1-49 troctolite Ol3 P1 h J10R1-49 troctolite Ol3 P2 h J10R1-49 troctolite Ol4 P1 h J25G1-32 troctolite Ol1 P1 f J25G1-32 troctolite Ol2 P1 h J25G1-32 troctolite Ol3 P1 f J25G1-32 troctolite Ol3 P2 h P3R1-31 Ol gabbro Ol1 P1 h P3R1-31 Ol gabbro Ol1 P2 h P3R1-31 Ol gabbro Ol2 P1 f P4G1-5 Ol gabbro Ol1 P1 h P4G1-5 Ol gabbro Ol1 P2 h P4G1-5 Ol gabbro Ol2 P1 h
15 P4G1-5 Ol gabbro Ol2 P2 h P4G1-5 Ol gabbro Ol3 P1 h P11R1-38 Ol gabbro Ol1 P1 f P11R1-38 Ol gabbro Ol1 P2 h P11R1-38 Ol gabbro Ol2 P1 f Ol = olivine; Cpx oik ol gabbro = clinopyroxene oikocryst-bearing olivine gabbro 2 Depth is given in m below seafloor. 3 The letters f and h in the profile name refer to full profiles (core--core) or half profiles (core). 4 Tc core refers to the obtained closure temperature at the core of the profile.
Constraining the cooling rate of the lower oceanic crust: a new approach applied to the Oman ophiolite
 Earth and Planetary Science Letters 199 (2002) 127^146 www.elsevier.com/locate/epsl Constraining the cooling rate of the lower oceanic crust: a new approach applied to the Oman ophiolite Laurence A. Coogan
Earth and Planetary Science Letters 199 (2002) 127^146 www.elsevier.com/locate/epsl Constraining the cooling rate of the lower oceanic crust: a new approach applied to the Oman ophiolite Laurence A. Coogan
12 Chemistry (Mg,Fe) 2 SiO 4 Olivine is forms what is called an isomorphous solid solution series that ranges between two end members: Forsterite Mg
 11 Olivine Structure Olivine is a common green or brown rock forming minerals which consists of a solid-solution series between Forsterite (Fo) and Fayalite (Fa). It is an orthorhombic orthosilicate with
11 Olivine Structure Olivine is a common green or brown rock forming minerals which consists of a solid-solution series between Forsterite (Fo) and Fayalite (Fa). It is an orthorhombic orthosilicate with
Effect of tectonic setting on chemistry of mantle-derived melts
 Effect of tectonic setting on chemistry of mantle-derived melts Lherzolite Basalt Factors controlling magma composition Composition of the source Partial melting process Fractional crystallization Crustal
Effect of tectonic setting on chemistry of mantle-derived melts Lherzolite Basalt Factors controlling magma composition Composition of the source Partial melting process Fractional crystallization Crustal
What do variable magnetic fabrics in gabbros of the Oman ophiolite reveal about lower oceanic crustal magmatism at fast spreading ridges?
 GSA Data Repository 1998 1 2 3 4 5 6 7 8 9 11 12 13 14 15 16 17 18 19 21 22 23 24 25 26 27 28 29 3 31 32 33 34 35 36 37 38 39 4 41 42 43 44 45 46 47 48 49 5 51 52 What do variable magnetic fabrics in of
GSA Data Repository 1998 1 2 3 4 5 6 7 8 9 11 12 13 14 15 16 17 18 19 21 22 23 24 25 26 27 28 29 3 31 32 33 34 35 36 37 38 39 4 41 42 43 44 45 46 47 48 49 5 51 52 What do variable magnetic fabrics in of
GEOL 2312 Igneous and Metamorphic Petrology Spring 2016 Score / 58. Midterm 1 Chapters 1-10
 GEOL 2312 Igneous and Metamorphic Petrology Name KEY Spring 2016 Score / 58 Midterm 1 Chapters 1-10 1) Name two things that petrologists want to know about magmas (1 pt) Formation, source, composition,
GEOL 2312 Igneous and Metamorphic Petrology Name KEY Spring 2016 Score / 58 Midterm 1 Chapters 1-10 1) Name two things that petrologists want to know about magmas (1 pt) Formation, source, composition,
Notes for Use of the Cpx-Plag-Ol Thermobar Workbook Last Updated:
 Notes for Use of the Cpx-Plag-Ol Thermobar Workbook Last Updated: 7-22-05 Cpx-Plag-Ol Thermobar is an Excel workbook that can be used to calculate crystallization pressures and temperatures for clinopyroxene-
Notes for Use of the Cpx-Plag-Ol Thermobar Workbook Last Updated: 7-22-05 Cpx-Plag-Ol Thermobar is an Excel workbook that can be used to calculate crystallization pressures and temperatures for clinopyroxene-
LAB 9: ULTRAMAFIC ROCKS, CUMULATES AND MELT SOURCES
 Geology 316 (Petrology) (03/26/2012) Name LAB 9: ULTRAMAFIC ROCKS, CUMULATES AND MELT SOURCES INTRODUCTION Ultramafic rocks are igneous rocks containing less than 10% felsic minerals (quartz + feldspars
Geology 316 (Petrology) (03/26/2012) Name LAB 9: ULTRAMAFIC ROCKS, CUMULATES AND MELT SOURCES INTRODUCTION Ultramafic rocks are igneous rocks containing less than 10% felsic minerals (quartz + feldspars
GLY 155 Introduction to Physical Geology, W. Altermann. Grotzinger Jordan. Understanding Earth. Sixth Edition
 Grotzinger Jordan Understanding Earth Sixth Edition Chapter 4: IGNEOUS ROCKS Solids from Melts 2011 by W. H. Freeman and Company Chapter 4: Igneous Rocks: Solids from Melts 1 About Igneous Rocks Igneous
Grotzinger Jordan Understanding Earth Sixth Edition Chapter 4: IGNEOUS ROCKS Solids from Melts 2011 by W. H. Freeman and Company Chapter 4: Igneous Rocks: Solids from Melts 1 About Igneous Rocks Igneous
GSA DATA REPOSITORY
 GSA DATA REPOSITORY 2013019 Supplemental information for The Solidus of Alkaline Carbonatite in the Deep Mantle Konstantin D. Litasov, Anton Shatskiy, Eiji Ohtani, and Gregory M. Yaxley EXPERIMENTAL METHODS
GSA DATA REPOSITORY 2013019 Supplemental information for The Solidus of Alkaline Carbonatite in the Deep Mantle Konstantin D. Litasov, Anton Shatskiy, Eiji Ohtani, and Gregory M. Yaxley EXPERIMENTAL METHODS
Chapter 4 Rocks & Igneous Rocks
 Chapter 4 Rocks & Igneous Rocks Rock Definition A naturally occurring consolidated mixture of one or more minerals e.g, marble, granite, sandstone, limestone Rock Definition Must naturally occur in nature,
Chapter 4 Rocks & Igneous Rocks Rock Definition A naturally occurring consolidated mixture of one or more minerals e.g, marble, granite, sandstone, limestone Rock Definition Must naturally occur in nature,
Igneous and Metamorphic Rock Forming Minerals. Department of Geology Mr. Victor Tibane SGM 210_2013
 Igneous and Metamorphic Rock Forming Minerals Department of Geology Mr. Victor Tibane 1 SGM 210_2013 Grotzinger Jordan Understanding Earth Sixth Edition Chapter 4: IGNEOUS ROCKS Solids from Melts 2011
Igneous and Metamorphic Rock Forming Minerals Department of Geology Mr. Victor Tibane 1 SGM 210_2013 Grotzinger Jordan Understanding Earth Sixth Edition Chapter 4: IGNEOUS ROCKS Solids from Melts 2011
Clockwise rotation of the entire Oman ophiolite occurred in a suprasubduction zone setting Antony Morris et al.
 GSA Data Repository 216351 Clockwise rotation of the entire Oman ophiolite occurred in a suprasubduction zone setting Antony Morris et al. 1 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 2 21 22 23 24 25
GSA Data Repository 216351 Clockwise rotation of the entire Oman ophiolite occurred in a suprasubduction zone setting Antony Morris et al. 1 2 3 4 5 6 7 8 9 1 11 12 13 14 15 16 17 18 19 2 21 22 23 24 25
GSA Data Repository
 GSA Data Repository 2015244 1. Method of Statistical Analysis Appendix DR1 One has to be careful and use only samples with complete Sm-Eu-Gd concentration data to study Eu/Eu* in the crust. This is because
GSA Data Repository 2015244 1. Method of Statistical Analysis Appendix DR1 One has to be careful and use only samples with complete Sm-Eu-Gd concentration data to study Eu/Eu* in the crust. This is because
CLOSURE TEMPERATURES OF ACCESSORY MINERALS
 DR2005002 Flowers et al. CLOSURE TEMPERATURES OF ACCESSORY MINERALS The range of Pb diffusivity in accessory minerals provides the opportunity to reconstruct detailed thermal histories using the U-Pb isotopic
DR2005002 Flowers et al. CLOSURE TEMPERATURES OF ACCESSORY MINERALS The range of Pb diffusivity in accessory minerals provides the opportunity to reconstruct detailed thermal histories using the U-Pb isotopic
L.O: THE CRUST USE REFERENCE TABLE PAGE 10
 USE REFERENCE TABLE PAGE 10 1. The oceanic crust is thought to be composed mainly of A) granite B) sandstone C) basalt D) rhyolite 2. To get sample material from the mantle, drilling will be done through
USE REFERENCE TABLE PAGE 10 1. The oceanic crust is thought to be composed mainly of A) granite B) sandstone C) basalt D) rhyolite 2. To get sample material from the mantle, drilling will be done through
Geochemical and mineralogical technics to investigate the lithosphere and the asthenosphere. 07/11/2017 GEO-DEEP 9300 Claire Aupart
 Geochemical and mineralogical technics to investigate the lithosphere and the asthenosphere 07/11/2017 GEO-DEEP 9300 Claire Aupart Introduction Introduction Getting samples Cores: Maximum depth reach in
Geochemical and mineralogical technics to investigate the lithosphere and the asthenosphere 07/11/2017 GEO-DEEP 9300 Claire Aupart Introduction Introduction Getting samples Cores: Maximum depth reach in
OCEAN/ESS 410. Lab 8. Igneous rocks
 Lab 8. Igneous rocks Today s exercise is an introduction to rock identification and the crustal structure of the seafloor. All rocks are composed of two or more minerals, and can be classified based on
Lab 8. Igneous rocks Today s exercise is an introduction to rock identification and the crustal structure of the seafloor. All rocks are composed of two or more minerals, and can be classified based on
Alkaline Seafloor Hydrothermal Systems: Experimental Simulation of CO 2 -Peridotite-Seawater Reactions
 Alkaline Seafloor Hydrothermal Systems: Experimental Simulation of CO 2 -Peridotite-Seawater Reactions Thomas M. Carpenter John P. Kaszuba Melissa Fittipaldo Michael Rearick Los Alamos National Laboratory
Alkaline Seafloor Hydrothermal Systems: Experimental Simulation of CO 2 -Peridotite-Seawater Reactions Thomas M. Carpenter John P. Kaszuba Melissa Fittipaldo Michael Rearick Los Alamos National Laboratory
Kent and Cooper, 2018, How well do zircons record the thermal evolution of magmatic systems?: Geology,
 GSA Data Repository 2018024 Kent and Cooper, 2018, How well do zircons record the thermal evolution of magmatic systems?: Geology, https://doi.org/10.1130/g39690.1. Supplemental Text Modelling methodology
GSA Data Repository 2018024 Kent and Cooper, 2018, How well do zircons record the thermal evolution of magmatic systems?: Geology, https://doi.org/10.1130/g39690.1. Supplemental Text Modelling methodology
GEOL 2312 Igneous and Metamorphic Petrology Spring 2009 Sc ore / 40
 GEOL 2312 Igneous and Metamorphic Petrology Name Spring 2009 Sc ore / 40 QUIZ 3 1) Name two geologic features that provide physical evidence for the mineralogy of the earth s mantle (2 pts) Ophiolites,
GEOL 2312 Igneous and Metamorphic Petrology Name Spring 2009 Sc ore / 40 QUIZ 3 1) Name two geologic features that provide physical evidence for the mineralogy of the earth s mantle (2 pts) Ophiolites,
G 3. AN ELECTRONIC JOURNAL OF THE EARTH SCIENCES Published by AGU and the Geochemical Society
 Geosystems G 3 AN ELECTRONIC JOURNAL OF THE EARTH SCIENCES Published by AGU and the Geochemical Society Article Volume 3, Number 11 13 November 22 864, doi:1.129/21gc23 ISSN: 152527 Petrology and geochemistry
Geosystems G 3 AN ELECTRONIC JOURNAL OF THE EARTH SCIENCES Published by AGU and the Geochemical Society Article Volume 3, Number 11 13 November 22 864, doi:1.129/21gc23 ISSN: 152527 Petrology and geochemistry
WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY
 WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY Brandon E. Schwab Department of Geology Humboldt State University
WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY Brandon E. Schwab Department of Geology Humboldt State University
Igneous petrology EOSC 321
 Igneous petrology EOSC 321 Laboratory 2: Determination of plagioclase composition. Mafic and intermediate plutonic rocks Learning Goals. After this Lab, you should be able: Determine plagioclase composition
Igneous petrology EOSC 321 Laboratory 2: Determination of plagioclase composition. Mafic and intermediate plutonic rocks Learning Goals. After this Lab, you should be able: Determine plagioclase composition
Igneous Rocks. Igneous Rocks. Genetic Classification of
 Igneous Rocks Fig. 5.1 Genetic Classification of Igneous Rocks Intrusive: crystallized from slowly cooling magma intruded within the Earth s crust; e.g. granite, gabbro 1 Fig. 5.2 Genetic Classification
Igneous Rocks Fig. 5.1 Genetic Classification of Igneous Rocks Intrusive: crystallized from slowly cooling magma intruded within the Earth s crust; e.g. granite, gabbro 1 Fig. 5.2 Genetic Classification
Chapter 4 8/27/2013. Igneous Rocks. and Intrusive Igneous Activity. Introduction. The Properties and Behavior of Magma and Lava
 Introduction Chapter 4 Igneous rocks form by the cooling of magma (or lava). Large parts of the continents and all the oceanic crust are composed of. and Intrusive Igneous Activity The Properties and Behavior
Introduction Chapter 4 Igneous rocks form by the cooling of magma (or lava). Large parts of the continents and all the oceanic crust are composed of. and Intrusive Igneous Activity The Properties and Behavior
Earthquakes. Earthquakes are caused by a sudden release of energy
 Earthquakes Earthquakes are caused by a sudden release of energy The amount of energy released determines the magnitude of the earthquake Seismic waves carry the energy away from its origin Fig. 18.1 Origin
Earthquakes Earthquakes are caused by a sudden release of energy The amount of energy released determines the magnitude of the earthquake Seismic waves carry the energy away from its origin Fig. 18.1 Origin
Plate tectonics, rock cycle
 Dikes, Antarctica Rock Cycle Plate tectonics, rock cycle The Rock Cycle A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one
Dikes, Antarctica Rock Cycle Plate tectonics, rock cycle The Rock Cycle A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one
N = N 0 e -λt D* = N 0 -N D* = N 0 (1-e -λt ) or N(e λt -1) where N is number of parent atoms at time t, N 0
 N = N 0 e -λt D* = N 0 -N D* = N 0 (1-e -λt ) or N(e λt -1) where N is number of parent atoms at time t, N 0 is initial number of parents, D* is number of radiogenic daughter atoms, and λ is the decay
N = N 0 e -λt D* = N 0 -N D* = N 0 (1-e -λt ) or N(e λt -1) where N is number of parent atoms at time t, N 0 is initial number of parents, D* is number of radiogenic daughter atoms, and λ is the decay
PLATE TECTONICS, VOLCANISM AND IGNEOUS ROCKS
 PLATE TECTONICS, VOLCANISM AND IGNEOUS ROCKS PLATE TECTONICS TO IGNEOUS ROCKS Internal Heat Seafloor Spreading/Plate Tectonics Volcanism Plate Boundary Intra-plate (hot spot) Divergent Convergent Igneous
PLATE TECTONICS, VOLCANISM AND IGNEOUS ROCKS PLATE TECTONICS TO IGNEOUS ROCKS Internal Heat Seafloor Spreading/Plate Tectonics Volcanism Plate Boundary Intra-plate (hot spot) Divergent Convergent Igneous
American Journal of Science
 [American Journal of Science, Vol. 306, June, 2006, P.389 427, DOI 10.2475/06.2006.01] American Journal of Science JUNE 2006 CHEMICAL AND THERMAL CONSTRAINTS ON FOCUSSED FLUID FLOW IN THE LOWER OCEANIC
[American Journal of Science, Vol. 306, June, 2006, P.389 427, DOI 10.2475/06.2006.01] American Journal of Science JUNE 2006 CHEMICAL AND THERMAL CONSTRAINTS ON FOCUSSED FLUID FLOW IN THE LOWER OCEANIC
DIFFUSION OF NEODYMIUM IN ORTHOPYROXENE: EXPERIMENT, THEORY AND APPLICATIONS. Jennifer L. Sano. for the Degree of MASTER OF SCIENCE
 DIFFUSION OF NEODYMIUM IN ORTHOPYROXENE: EXPERIMENT, THEORY AND APPLICATIONS by Jennifer L. Sano A Thesis Submitted to the Faculty of the DEPARTMENT OF GEOSCIENCES In Partial Fulfillment of the Requirements
DIFFUSION OF NEODYMIUM IN ORTHOPYROXENE: EXPERIMENT, THEORY AND APPLICATIONS by Jennifer L. Sano A Thesis Submitted to the Faculty of the DEPARTMENT OF GEOSCIENCES In Partial Fulfillment of the Requirements
Time to Solidify an Ocean of Magma
 1 of 6 March 25, 2009 Time to Solidify an Ocean of Magma --- A small mineral grain places limits on how long it took the lunar magma ocean to solidify. Written by G. Jeffrey Taylor Hawai i Institute of
1 of 6 March 25, 2009 Time to Solidify an Ocean of Magma --- A small mineral grain places limits on how long it took the lunar magma ocean to solidify. Written by G. Jeffrey Taylor Hawai i Institute of
MACRORYTHMIC GABBRO TO GRANITE CYCLES OF CLAM COVE VINALHAVEN INTRUSION, MAINE
 MACRORYTHMIC GABBRO TO GRANITE CYCLES OF CLAM COVE VINALHAVEN INTRUSION, MAINE NICK CUBA Amherst College Sponsor: Peter Crowley INTRODUCTION The rocks of the layered gabbro-diorite unit of the Silurian
MACRORYTHMIC GABBRO TO GRANITE CYCLES OF CLAM COVE VINALHAVEN INTRUSION, MAINE NICK CUBA Amherst College Sponsor: Peter Crowley INTRODUCTION The rocks of the layered gabbro-diorite unit of the Silurian
Occurrence of mafic-ultramafic rocks
 Occurrence of mafic-ultramafic rocks Mantle-derived magmas Oceanic Lithospheric mantle Continental lithospheric mantle Ultramafic xenoliths Oman ophiolite harzburgite upper mantle both from: http://www.bris.ac.uk/depts/geol/vft/oman.html
Occurrence of mafic-ultramafic rocks Mantle-derived magmas Oceanic Lithospheric mantle Continental lithospheric mantle Ultramafic xenoliths Oman ophiolite harzburgite upper mantle both from: http://www.bris.ac.uk/depts/geol/vft/oman.html
Thursday, October 4 th
 Thursday, October 4 th Objective: We will use and define the different ways to classify igneous rocks. Warm-up: 1. Which type of lava is most viscous? 2. Which type of lava has the least amount of silicate?
Thursday, October 4 th Objective: We will use and define the different ways to classify igneous rocks. Warm-up: 1. Which type of lava is most viscous? 2. Which type of lava has the least amount of silicate?
Solubility, mixtures, non-ideality OUTLINE
 Solubility, mixtures, non-ideality Equilibrium? OUTLINE Class exercise next class bring laptop, or use class tablet Enthalpy, Entropy The Gibbs Function ΔG and K Mixtures Chemical Potential 1 Enthalpy
Solubility, mixtures, non-ideality Equilibrium? OUTLINE Class exercise next class bring laptop, or use class tablet Enthalpy, Entropy The Gibbs Function ΔG and K Mixtures Chemical Potential 1 Enthalpy
Data Repository Item
 Data Repository Item 2009003 An abrupt transition from magma-starved to magma-rich rifting in the eastern Black Sea Donna J. Shillington, Caroline L. Scott, Timothy A. Minshull, Rosemary A. Edwards, Peter
Data Repository Item 2009003 An abrupt transition from magma-starved to magma-rich rifting in the eastern Black Sea Donna J. Shillington, Caroline L. Scott, Timothy A. Minshull, Rosemary A. Edwards, Peter
G 3. AN ELECTRONIC JOURNAL OF THE EARTH SCIENCES Published by AGU and the Geochemical Society
 Geosystems G 3 AN ELECTRONIC JOURNAL OF THE EARTH SCIENCES Published by AGU and the Geochemical Society Article Volume 9, Number 7 29 July 2008 Q07025, doi: ISSN: 1525-2027 Click Here for Full Article
Geosystems G 3 AN ELECTRONIC JOURNAL OF THE EARTH SCIENCES Published by AGU and the Geochemical Society Article Volume 9, Number 7 29 July 2008 Q07025, doi: ISSN: 1525-2027 Click Here for Full Article
Pyroxenes (Mg, Fe 2+ ) 2 Si 2 O 6 (monoclinic) and. MgSiO 3 FeSiO 3 (orthorhombic) Structure (Figure 2 of handout)
 Pyroxenes (Mg, Fe 2+ ) 2 Si 2 O 6 (monoclinic) and 20 MgSiO 3 FeSiO 3 (orthorhombic) Structure (Figure 2 of handout) Chain silicate eg Diopside Mg and Fe ions link SiO 3 chains The chain runs up and down
Pyroxenes (Mg, Fe 2+ ) 2 Si 2 O 6 (monoclinic) and 20 MgSiO 3 FeSiO 3 (orthorhombic) Structure (Figure 2 of handout) Chain silicate eg Diopside Mg and Fe ions link SiO 3 chains The chain runs up and down
The Lead 206/207 Dating Method
 The Lead 206/207 Dating Method 1 U Pb Zircon Ages, Chemical Geology, Volume 211 (2004) Pages 87 109 2 Lead Isotope Planetary Profiling, Chemical Geology, Volume 233 (2006) Pages 1 45 3 U Pb Step-Leaching
The Lead 206/207 Dating Method 1 U Pb Zircon Ages, Chemical Geology, Volume 211 (2004) Pages 87 109 2 Lead Isotope Planetary Profiling, Chemical Geology, Volume 233 (2006) Pages 1 45 3 U Pb Step-Leaching
Trace Elements. Today s lecture
 Trace Elements 300 Ni 200 ppm 100 0 300 Zr 200 100 0 40 50 60 70 80 SiO 2 wt. % Updates: M&M due date: Tuesday Today s lecture Topics: Trace element compositions Trace element behavior Partitioning Spider(
Trace Elements 300 Ni 200 ppm 100 0 300 Zr 200 100 0 40 50 60 70 80 SiO 2 wt. % Updates: M&M due date: Tuesday Today s lecture Topics: Trace element compositions Trace element behavior Partitioning Spider(
Sphene (Titanite) Plane polarized light. Honey brown/orange Wedge-shaped crystals
 Sphene (Titanite) Plane polarized light Honey brown/orange Wedge-shaped crystals Sphene (Titanite) Crossed nicols High-order, washedout interference colors (light orange) #1 Rule for (Heavy) Radiogenic
Sphene (Titanite) Plane polarized light Honey brown/orange Wedge-shaped crystals Sphene (Titanite) Crossed nicols High-order, washedout interference colors (light orange) #1 Rule for (Heavy) Radiogenic
DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER
 Geol 2312 Igneous and Metamorphic Petrology Spring 2009 Name DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER Objective: This exercise is intended to improve understanding
Geol 2312 Igneous and Metamorphic Petrology Spring 2009 Name DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER Objective: This exercise is intended to improve understanding
Chapter 9: Trace Elements
 Chapter 9: Trace Elements Note magnitude of major element changes Figure 8.2. Harker variation diagram for 310 analyzed volcanic rocks from Crater Lake (Mt. Mazama), Oregon Cascades. Data compiled by Rick
Chapter 9: Trace Elements Note magnitude of major element changes Figure 8.2. Harker variation diagram for 310 analyzed volcanic rocks from Crater Lake (Mt. Mazama), Oregon Cascades. Data compiled by Rick
1 - C Systems. The system H 2 O. Heat an ice at 1 atm from-5 to 120 o C. Heat vs. Temperature
 1 - C Systems The system H 2 O Heat an ice at 1 atm from-5 to 120 o C Heat vs. Temperature Fig. 6.7. After Bridgman (1911) Proc. Amer. Acad. Arts and Sci., 5, 441-513; (1936) J. Chem. Phys., 3, 597-605;
1 - C Systems The system H 2 O Heat an ice at 1 atm from-5 to 120 o C Heat vs. Temperature Fig. 6.7. After Bridgman (1911) Proc. Amer. Acad. Arts and Sci., 5, 441-513; (1936) J. Chem. Phys., 3, 597-605;
Electronic Appendix A: Supplementary material to accompany the manuscript, Fe 3+ / Fe in Mariana Arc basalts and primary fo 2.
 Electronic Appendix A: Supplementary material to accompany the manuscript, Fe 3+ / Fe in Mariana Arc basalts and primary fo 2. Screening for olivine interference in Fe-µ-XANES spectra When collecting Fe-µ-XANES
Electronic Appendix A: Supplementary material to accompany the manuscript, Fe 3+ / Fe in Mariana Arc basalts and primary fo 2. Screening for olivine interference in Fe-µ-XANES spectra When collecting Fe-µ-XANES
GEOL 3313 Petrology of Igneous and Metamorphic Rocks Study Guide for Final Examination Glen Mattioli
 GEOL 3313 Petrology of Igneous and Metamorphic Rocks Study Guide for Final Examination Glen Mattioli Chapter 5: Crystal-Melt phase diagrams Effect of water pressure on feldspar stability Hypersolvus vs.
GEOL 3313 Petrology of Igneous and Metamorphic Rocks Study Guide for Final Examination Glen Mattioli Chapter 5: Crystal-Melt phase diagrams Effect of water pressure on feldspar stability Hypersolvus vs.
VOLCANIC STRATIGRAPHY AND PETROLOGY OF THE NORTHERN SNAEFELLSNES RIFT, SOUTHERN LAXÁRDALSFJÖLL, ICELAND
 VOLCANIC STRATIGRAPHY AND PETROLOGY OF THE NORTHERN SNAEFELLSNES RIFT, SOUTHERN LAXÁRDALSFJÖLL, ICELAND LEBN SCHUYLER Whitman College Sponsor: John Winter INTRODUCTION Iceland is exposed above sea level
VOLCANIC STRATIGRAPHY AND PETROLOGY OF THE NORTHERN SNAEFELLSNES RIFT, SOUTHERN LAXÁRDALSFJÖLL, ICELAND LEBN SCHUYLER Whitman College Sponsor: John Winter INTRODUCTION Iceland is exposed above sea level
EMMR25 Mineralogy: Ol + opx + chlorite + cpx + amphibole + serpentine + opaque
 GSA Data Repository 2017365 Marshall et al., 2017, The role of serpentinite derived fluids in metasomatism of the Colorado Plateau (USA) lithospheric mantle: Geology, https://doi.org/10.1130/g39444.1 Appendix
GSA Data Repository 2017365 Marshall et al., 2017, The role of serpentinite derived fluids in metasomatism of the Colorado Plateau (USA) lithospheric mantle: Geology, https://doi.org/10.1130/g39444.1 Appendix
Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100
 Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100 Ray Rector - Instructor Major Concepts 1) Igneous rocks form directly from the crystallization of a magma or lava 2)
Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100 Ray Rector - Instructor Major Concepts 1) Igneous rocks form directly from the crystallization of a magma or lava 2)
Partial melting of mantle peridotite
 Partial melting of mantle peridotite 1100 1200 1300 1400 1500 (TºC) Depth (km) 50 100 150 Plag lherzolite (ol-opx-cpx-pl) Spinel lherzolite (Ol-opx-cpx-sp) Garnet lherzolite (Ol-opx-cpx-gar) Graphite Diamond
Partial melting of mantle peridotite 1100 1200 1300 1400 1500 (TºC) Depth (km) 50 100 150 Plag lherzolite (ol-opx-cpx-pl) Spinel lherzolite (Ol-opx-cpx-sp) Garnet lherzolite (Ol-opx-cpx-gar) Graphite Diamond
A Rock is a solid aggregate of minerals.
 Quartz A Rock is a solid aggregate of minerals. Orthoclase Feldspar Plagioclase Feldspar Biotite Four different minerals are obvious in this piece of Granite. The average automobile contains: Minerals
Quartz A Rock is a solid aggregate of minerals. Orthoclase Feldspar Plagioclase Feldspar Biotite Four different minerals are obvious in this piece of Granite. The average automobile contains: Minerals
C = 3: Ternary Systems: Example 1: Ternary Eutectic
 Phase Equilibrium C = 3: Ternary Systems: Example 1: Ternary Eutectic Adding components, becomes increasingly difficult to depict 1-C: P - T diagrams easy 2-C: isobaric T-X, isothermal P-X 3-C:?? Still
Phase Equilibrium C = 3: Ternary Systems: Example 1: Ternary Eutectic Adding components, becomes increasingly difficult to depict 1-C: P - T diagrams easy 2-C: isobaric T-X, isothermal P-X 3-C:?? Still
Rocks: Materials of the Solid Earth
 1 Rocks: Materials of the Solid Earth Presentation modified from: Instructor Resource Center on CD-ROM, Foundations of Earth Science,, 4 th Edition, Lutgens/Tarbuck, Rock Cycle Igneous Rocks Today 2 Rock
1 Rocks: Materials of the Solid Earth Presentation modified from: Instructor Resource Center on CD-ROM, Foundations of Earth Science,, 4 th Edition, Lutgens/Tarbuck, Rock Cycle Igneous Rocks Today 2 Rock
Metcalf and Buck. GSA Data Repository
 GSA Data Repository 2015035 Metcalf and Buck Figure DR1. Secondary ionization mass-spectrometry U-Pb zircon geochronology plots for data collected on two samples of Wilson Ridge plutonic rocks. Data presented
GSA Data Repository 2015035 Metcalf and Buck Figure DR1. Secondary ionization mass-spectrometry U-Pb zircon geochronology plots for data collected on two samples of Wilson Ridge plutonic rocks. Data presented
Suzanne Picazo, Mathilde Cannat, Adelie Delacour, Javier Escartın, Stephane Roumejon, and Serguei Silantyev
 Correction Volume 14, Number 8 16 August 2013 doi: ISSN: 1525-2027 Correction to Deformation associated with the denudation of mantle-derived rocks at the Mid- Atlantic Ridge 13 15 N: The role of magmatic
Correction Volume 14, Number 8 16 August 2013 doi: ISSN: 1525-2027 Correction to Deformation associated with the denudation of mantle-derived rocks at the Mid- Atlantic Ridge 13 15 N: The role of magmatic
Essentials of Geology, 11e
 Essentials of Geology, 11e Igneous Rocks and Intrusive Activity Chapter 3 Instructor Jennifer Barson Spokane Falls Community College Geology 101 Stanley Hatfield Southwestern Illinois College Characteristics
Essentials of Geology, 11e Igneous Rocks and Intrusive Activity Chapter 3 Instructor Jennifer Barson Spokane Falls Community College Geology 101 Stanley Hatfield Southwestern Illinois College Characteristics
Lecture 36. Igneous geochemistry
 Lecture 36 Igneous geochemistry Reading - White Chapter 7 Today 1. Overview 2. solid-melt distribution coefficients Igneous geochemistry The chemistry of igneous systems provides clues to a number of important
Lecture 36 Igneous geochemistry Reading - White Chapter 7 Today 1. Overview 2. solid-melt distribution coefficients Igneous geochemistry The chemistry of igneous systems provides clues to a number of important
PETROGENESIS OF A SERIES OF MAFIC SHEETS WITHIN THE VINALHAVEN PLUTON, VINALHAVEN ISLAND, MAINE
 PETROGENESIS OF A SERIES OF MAFIC SHEETS WITHIN THE VINALHAVEN PLUTON, VINALHAVEN ISLAND, MAINE DANIEL HAWKINS Western Kentucky University Research Advisor: Andrew Wulff INTRODUCTION Round Point, in the
PETROGENESIS OF A SERIES OF MAFIC SHEETS WITHIN THE VINALHAVEN PLUTON, VINALHAVEN ISLAND, MAINE DANIEL HAWKINS Western Kentucky University Research Advisor: Andrew Wulff INTRODUCTION Round Point, in the
How 2 nd half labs will work
 How 2 nd half labs will work Continue to use your mineral identification skills Learn to describe, classify, interpret rock hand samples: Igneous sedimentary metamorphic volcanic plutonic (1 week) (1 wk)
How 2 nd half labs will work Continue to use your mineral identification skills Learn to describe, classify, interpret rock hand samples: Igneous sedimentary metamorphic volcanic plutonic (1 week) (1 wk)
Evolution of Mafic Lavas from the Main Ethiopian Rift. Research Scholar, Erica Campbell Research Mentor, Dr. Tanya Furman
 Evolution of Mafic Lavas from the Main Ethiopian Rift Research Scholar, Erica Campbell Research Mentor, Dr. Tanya Furman Abstract Understanding continental rifting processes means understanding plate tectonics,
Evolution of Mafic Lavas from the Main Ethiopian Rift Research Scholar, Erica Campbell Research Mentor, Dr. Tanya Furman Abstract Understanding continental rifting processes means understanding plate tectonics,
Experimental investigation of reaction-driven stress development during mineral carbonation:
 US National Academies Webinar on Subsurface Geological Capture and Storage of CO 2 15 November 2017 Experimental investigation of reaction-driven stress development during mineral carbonation: Implications
US National Academies Webinar on Subsurface Geological Capture and Storage of CO 2 15 November 2017 Experimental investigation of reaction-driven stress development during mineral carbonation: Implications
doi: /nature09369
 doi:10.1038/nature09369 Supplementary Figure S1 Scanning electron microscope images of experimental charges with vapour and vapour phase quench. Experimental runs are in the order of added water concentration
doi:10.1038/nature09369 Supplementary Figure S1 Scanning electron microscope images of experimental charges with vapour and vapour phase quench. Experimental runs are in the order of added water concentration
SUPPLEMENTARY INFORMATION
 GSA Data Repository 080 Schorn et al., 08, Thermal buffering in the orogenic crust: Geology, https://doi.org/0.30/g4046.. SUPPLEMENTARY INFORMATION 3 PHASE DIAGRAM MODELING 4 5 6 7 8 9 0 3 4 Phase diagrams
GSA Data Repository 080 Schorn et al., 08, Thermal buffering in the orogenic crust: Geology, https://doi.org/0.30/g4046.. SUPPLEMENTARY INFORMATION 3 PHASE DIAGRAM MODELING 4 5 6 7 8 9 0 3 4 Phase diagrams
Chapter 9: Trace Elements
 Lecture 13 Introduction to Trace Elements Wednesday, March 9, 2005 Chapter 9: Trace Elements Note magnitude of major element changes Figure 8-2. Harker variation diagram for 310 analyzed volcanic rocks
Lecture 13 Introduction to Trace Elements Wednesday, March 9, 2005 Chapter 9: Trace Elements Note magnitude of major element changes Figure 8-2. Harker variation diagram for 310 analyzed volcanic rocks
Tectonic-Igneous Associations
 Tectonic-Igneous Associations Associations on a larger scale than the petrogenetic provinces An attempt to address global patterns of igneous activity by grouping provinces based upon similarities in occurrence
Tectonic-Igneous Associations Associations on a larger scale than the petrogenetic provinces An attempt to address global patterns of igneous activity by grouping provinces based upon similarities in occurrence
Common non-silicate planetary minerals
 Common non-silicate planetary minerals Many of the non-silicate minerals are simple oxides. Corundum Al2O3 Al2+3 O3-2 Rutile Ti2O3 Ti2+3 O3-2 Ilmenite FeTiO3 Fe+3Ti+3O3-2 Hematite Fe2O3 Fe2+3 O3-2 Families
Common non-silicate planetary minerals Many of the non-silicate minerals are simple oxides. Corundum Al2O3 Al2+3 O3-2 Rutile Ti2O3 Ti2+3 O3-2 Ilmenite FeTiO3 Fe+3Ti+3O3-2 Hematite Fe2O3 Fe2+3 O3-2 Families
GY303 Igneous & Metamorphic Petrology. Lecture 7: Magma Sources and Tectonic Environments
 GY303 Igneous & Metamorphic Petrology Lecture 7: Magma Sources and Tectonic Environments Factors controlling Magma production Source rock composition Amount of fluids, especially H 2 O Pressure (Depth)
GY303 Igneous & Metamorphic Petrology Lecture 7: Magma Sources and Tectonic Environments Factors controlling Magma production Source rock composition Amount of fluids, especially H 2 O Pressure (Depth)
Block: Igneous Rocks. From this list, select the terms which answer the following questions.
 Geology 12 Name: Mix and Match: Igneous Rocks Refer to the following list. Block: porphyritic volatiles mafic glassy magma mixing concordant discontinuous reaction series igneous vesicular partial melting
Geology 12 Name: Mix and Match: Igneous Rocks Refer to the following list. Block: porphyritic volatiles mafic glassy magma mixing concordant discontinuous reaction series igneous vesicular partial melting
SEA-FLOOR SPREADING. In the 1950 s and early 1960 s detailed study of the oceans revealed the following surprising information:-
 SEA-FLOOR SPREADING In the 1950 s and early 1960 s detailed study of the oceans revealed the following surprising information:- Detailed bathymetric (depth) studies showed that there was an extensive submarine
SEA-FLOOR SPREADING In the 1950 s and early 1960 s detailed study of the oceans revealed the following surprising information:- Detailed bathymetric (depth) studies showed that there was an extensive submarine
Q. WANG, Q-K. XIA, S. Y. O REILLY, W. L. GRIFFIN, E. E. BEYER AND H. K. BRUECKNER
 Pressure- and stress-induced fabric transition in olivine from peridotites in the Western Gneiss Region (Norway): implications for mantle seismic anisotropy Q. WANG, Q-K. XIA, S. Y. O REILLY, W. L. GRIFFIN,
Pressure- and stress-induced fabric transition in olivine from peridotites in the Western Gneiss Region (Norway): implications for mantle seismic anisotropy Q. WANG, Q-K. XIA, S. Y. O REILLY, W. L. GRIFFIN,
Igneous Rock. Magma Chamber Large pool of magma in the lithosphere
 Igneous Rock Magma Molten rock under the surface Temperature = 600 o 1400 o C Magma Chamber Large pool of magma in the lithosphere Magma chamber - most all magma consists of silicon and oxygen (silicate)
Igneous Rock Magma Molten rock under the surface Temperature = 600 o 1400 o C Magma Chamber Large pool of magma in the lithosphere Magma chamber - most all magma consists of silicon and oxygen (silicate)
Engineering Geology ECIV 2204
 Engineering Geology ECIV 2204 Instructor : Dr. Jehad Hamad 2017-2016 Chapter (3) Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth
Engineering Geology ECIV 2204 Instructor : Dr. Jehad Hamad 2017-2016 Chapter (3) Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth
9/4/2015. Feldspars White, pink, variable Clays White perfect Quartz Colourless, white, red, None
 ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks Chapter 3.0: Weathering & soils Chapter 4.0: Geological
ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks Chapter 3.0: Weathering & soils Chapter 4.0: Geological
Worked Example of Batch Melting: Rb and Sr
 Worked Example of Batch Melting: Rb and Sr Basalt with the mode: Table 9.2. Conversion from mode to weight percent Mineral Mode Density Wt prop Wt% ol 15 3.6 54 0.18 cpx 33 3.4 112.2 0.37 plag 51 2.7 137.7
Worked Example of Batch Melting: Rb and Sr Basalt with the mode: Table 9.2. Conversion from mode to weight percent Mineral Mode Density Wt prop Wt% ol 15 3.6 54 0.18 cpx 33 3.4 112.2 0.37 plag 51 2.7 137.7
Ancient depletion and mantle heterogeneity: Revisiting the Jurassic-Permian paradox of Alpine peridotites
 GSA Data Repository 2015092 Ancient depletion and mantle heterogeneity: Revisiting the Jurassic-Permian paradox of Alpine peridotites McCarthy Anders 1 & Müntener Othmar 1 1 Institute of Earth Sciences
GSA Data Repository 2015092 Ancient depletion and mantle heterogeneity: Revisiting the Jurassic-Permian paradox of Alpine peridotites McCarthy Anders 1 & Müntener Othmar 1 1 Institute of Earth Sciences
Structure of the Earth
 Structure of the Earth Compositional (Chemical) Layers Crust: Low density Moho: Density boundary between crust and mantle Mantle: Higher density High in Magnesium (Mg) and Iron (Fe) Core: High in Nickel
Structure of the Earth Compositional (Chemical) Layers Crust: Low density Moho: Density boundary between crust and mantle Mantle: Higher density High in Magnesium (Mg) and Iron (Fe) Core: High in Nickel
PETROGENESIS OF EARLY SKAGI-SNAEFELLSNES RIFT BASALTS AT GRUNNAVIK, ICELAND
 PETROGENESIS OF EARLY SKAGI-SNAEFELLSNES RIFT BASALTS AT GRUNNAVIK, ICELAND SARA JOHNSON Beloit College Sponsor: Jim Rougvie INTRODUCTION The majority of field research in Iceland has been centered on
PETROGENESIS OF EARLY SKAGI-SNAEFELLSNES RIFT BASALTS AT GRUNNAVIK, ICELAND SARA JOHNSON Beloit College Sponsor: Jim Rougvie INTRODUCTION The majority of field research in Iceland has been centered on
Cosmochemical application of High Precision Multi-collector SIMS
 Cosmochemical application of High Precision Multi-collector SIMS Oxygen Three Isotopes in Chondrules Early Solar System Chronology of Refractory Inclusions Noriko Kita Wisc-SIMS Laboratory, University
Cosmochemical application of High Precision Multi-collector SIMS Oxygen Three Isotopes in Chondrules Early Solar System Chronology of Refractory Inclusions Noriko Kita Wisc-SIMS Laboratory, University
Geotherms. Reading: Fowler Ch 7. Equilibrium geotherms One layer model
 Geotherms Reading: Fowler Ch 7 Equilibrium geotherms One layer model (a) Standard model: k = 2.5 W m -1 C -1 A = 1.25 x 10-6 W m -3 Q moho = 21 x 10-3 W m -2 shallow T-gradient: 30 C km -1 deep T-gradient:
Geotherms Reading: Fowler Ch 7 Equilibrium geotherms One layer model (a) Standard model: k = 2.5 W m -1 C -1 A = 1.25 x 10-6 W m -3 Q moho = 21 x 10-3 W m -2 shallow T-gradient: 30 C km -1 deep T-gradient:
Supplementary Information
 Supplementary Information Supplementary Tables Major element compositions of host and daughter crystals: Table 1 reports composition for host-olivine. No daughter olivine has been found. The number for
Supplementary Information Supplementary Tables Major element compositions of host and daughter crystals: Table 1 reports composition for host-olivine. No daughter olivine has been found. The number for
1. In the diagram below, letters A and B represent locations near the edge of a continent.
 1. In the diagram below, letters A and B represent locations near the edge of a continent. A geologist who compares nonsedimentary rock samples from locations A and B would probably find that the samples
1. In the diagram below, letters A and B represent locations near the edge of a continent. A geologist who compares nonsedimentary rock samples from locations A and B would probably find that the samples
1. List the 3 main layers of Earth from the most dense to the least dense.
 1. List the 3 main layers of Earth from the most dense to the least dense. 2. List the 6 layers of earth based on their physical properties from the least dense to the most dense. 3. The thinnest layer
1. List the 3 main layers of Earth from the most dense to the least dense. 2. List the 6 layers of earth based on their physical properties from the least dense to the most dense. 3. The thinnest layer
G 3. AN ELECTRONIC JOURNAL OF THE EARTH SCIENCES Published by AGU and the Geochemical Society
 Geosystems G 3 AN ELECTRONIC JOURNAL OF THE EARTH SCIENCES Published by AGU and the Geochemical Society Article Volume 9, Number 10 16 October 2008 Q10007, doi:10.1029/2008gc002012 ISSN: 1525-2027 Stacked
Geosystems G 3 AN ELECTRONIC JOURNAL OF THE EARTH SCIENCES Published by AGU and the Geochemical Society Article Volume 9, Number 10 16 October 2008 Q10007, doi:10.1029/2008gc002012 ISSN: 1525-2027 Stacked
Metamorphic Petrology GLY 712 Geothermo-barometry
 Metamorphic Petrology GLY 712 Geothermo-barometry What is thermobarometry? Thermobarometry is concerned with estimating or inferring the temperatures and pressures at which a rock formed and/or subsequently
Metamorphic Petrology GLY 712 Geothermo-barometry What is thermobarometry? Thermobarometry is concerned with estimating or inferring the temperatures and pressures at which a rock formed and/or subsequently
A MICROANALYTICAL APPROACH TO UNDERSTANDING THE ORIGIN OF CUMULATE XENOLITHS FROM MAUNA KEA, HAWAII
 A MICROANALYTICAL APPROACH TO UNDERSTANDING THE ORIGIN OF CUMULATE XENOLITHS FROM MAUNA KEA, HAWAII Megan Pickard, Michael J. Dorais, Eric Christiansen, R. V. Fodor Limited Access to Subsurface Processes
A MICROANALYTICAL APPROACH TO UNDERSTANDING THE ORIGIN OF CUMULATE XENOLITHS FROM MAUNA KEA, HAWAII Megan Pickard, Michael J. Dorais, Eric Christiansen, R. V. Fodor Limited Access to Subsurface Processes
Name Class Date STUDY GUIDE FOR CONTENT MASTERY
 Igneous Rocks What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. extrusive igneous rock intrusive
Igneous Rocks What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. extrusive igneous rock intrusive
LAB 6: COMMON MINERALS IN IGNEOUS ROCKS
 GEOLOGY 17.01: Mineralogy LAB 6: COMMON MINERALS IN IGNEOUS ROCKS Part 2: Minerals in Gabbroic Rocks Learning Objectives: Students will be able to identify the most common silicate minerals in gabbroic
GEOLOGY 17.01: Mineralogy LAB 6: COMMON MINERALS IN IGNEOUS ROCKS Part 2: Minerals in Gabbroic Rocks Learning Objectives: Students will be able to identify the most common silicate minerals in gabbroic
23/9/2013 ENGINEERING GEOLOGY. Chapter 2: Rock classification:
 ENGINEERING GEOLOGY Chapter 2: Rock classification: ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks
ENGINEERING GEOLOGY Chapter 2: Rock classification: ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks
Earth and Planetary Science Letters
 Earth and Planetary Science Letters 308 (2011) 131 140 Contents lists available at ScienceDirect Earth and Planetary Science Letters journal homepage: www.elsevier.com/locate/epsl High-temperature inter-mineral
Earth and Planetary Science Letters 308 (2011) 131 140 Contents lists available at ScienceDirect Earth and Planetary Science Letters journal homepage: www.elsevier.com/locate/epsl High-temperature inter-mineral
How partial melts of mafic lower crust affect ascending magmas at oceanic ridges
 Contrib Mineral Petrol (2008) 156:49 71 DOI 10.1007/s00410-007-0273-x ORIGINAL PAPER How partial melts of mafic lower crust affect ascending magmas at oceanic ridges Astri J. S. Kvassnes Æ Timothy L. Grove
Contrib Mineral Petrol (2008) 156:49 71 DOI 10.1007/s00410-007-0273-x ORIGINAL PAPER How partial melts of mafic lower crust affect ascending magmas at oceanic ridges Astri J. S. Kvassnes Æ Timothy L. Grove
Lecture 3 Rocks and the Rock Cycle Dr. Shwan Omar
 Rocks A naturally occurring aggregate of one or more minerals (e.g., granite), or a body of non-crystalline material (e.g., obsidian glass), or of solid organic material (e.g., coal). Rock Cycle A sequence
Rocks A naturally occurring aggregate of one or more minerals (e.g., granite), or a body of non-crystalline material (e.g., obsidian glass), or of solid organic material (e.g., coal). Rock Cycle A sequence
THE MONTE MAGGIORE PERIDOTITE (CORSICA)
 MONTE MAGGIORE CAPO CORSO CORSICA Giovanni B. Piccardo THE MONTE MAGGIORE PERIDOTITE (CORSICA) FIELD RELATIONSHIPS MORB Gabbro Spinel (ex-garnet) pyroxenites L ESCURSIONE A MONTE MAGGIORE The Monte Maggiore
MONTE MAGGIORE CAPO CORSO CORSICA Giovanni B. Piccardo THE MONTE MAGGIORE PERIDOTITE (CORSICA) FIELD RELATIONSHIPS MORB Gabbro Spinel (ex-garnet) pyroxenites L ESCURSIONE A MONTE MAGGIORE The Monte Maggiore
Figure S1. CRISM maps of modeled mineralogy projected over CTX imagery (same
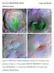 GSA DATA REPOSITORY 2015222 Cannon and Mustard Additional examples Figure S1. CRISM maps of modeled mineralogy projected over CTX imagery (same parameters as Figure 1). A: Taytay Crater (CRISM ID: HRL00005B77).
GSA DATA REPOSITORY 2015222 Cannon and Mustard Additional examples Figure S1. CRISM maps of modeled mineralogy projected over CTX imagery (same parameters as Figure 1). A: Taytay Crater (CRISM ID: HRL00005B77).
CO 2 quantification in magmatic systems
 CO 2 quantification in magmatic systems Innovative coupling of X-ray micro-tomography, in-situ microanalysis and thermodynamic modelling By Laura Créon Créon et al. (2018) Problematics 7 years work to
CO 2 quantification in magmatic systems Innovative coupling of X-ray micro-tomography, in-situ microanalysis and thermodynamic modelling By Laura Créon Créon et al. (2018) Problematics 7 years work to
Earth Science 232 Petrography
 Earth Science 232 Petrography Course notes by Shaun Frape and Alec Blyth Winter 2002 1 Petrology - Introduction Some Definitions Petra Greek for rock Logos Greek for disclosure or explanation Petrology
Earth Science 232 Petrography Course notes by Shaun Frape and Alec Blyth Winter 2002 1 Petrology - Introduction Some Definitions Petra Greek for rock Logos Greek for disclosure or explanation Petrology
Hirsch - Binary phase diagrams problems KEY
 Hirsch - Binary phase diagrams problems KEY Name Question 1. 1A. Using the phase rule, determine the degrees of freedom: a. at point e F = 2-3 + 1 = 0 b. within the field A + B F = 2-2 + 1 = 1 c. within
Hirsch - Binary phase diagrams problems KEY Name Question 1. 1A. Using the phase rule, determine the degrees of freedom: a. at point e F = 2-3 + 1 = 0 b. within the field A + B F = 2-2 + 1 = 1 c. within
Chapter 4 Up from the Inferno: Magma and Igneous Rocks
 Chapter 4 Up from the Inferno: Magma and Igneous Rocks Up from the Inferno: Magma and Igneous Rocks Updated by: Rick Oches, Professor of Geology & Environmental Sciences Bentley University Waltham, Massachusetts
Chapter 4 Up from the Inferno: Magma and Igneous Rocks Up from the Inferno: Magma and Igneous Rocks Updated by: Rick Oches, Professor of Geology & Environmental Sciences Bentley University Waltham, Massachusetts
Fluids, melts, and supercriticality in the MSH system and element transport in subduction zones
 cosmic rays Fluids, s, and supercriticality in the MSH system and element transport in subduction zones 10 Be volcanic front N, O 10 Be ocean water + CO 2 tracing petrologic and geotectonic processes (trace)
cosmic rays Fluids, s, and supercriticality in the MSH system and element transport in subduction zones 10 Be volcanic front N, O 10 Be ocean water + CO 2 tracing petrologic and geotectonic processes (trace)
GSA Data Repository
 GSA Data Repository 218145 Parolari et al., 218, A balancing act of crust creation and destruction along the western Mexican convergent margin: Geology, https://doi.org/1.113/g39972.1. 218145_Tables DR1-DR4.xls
GSA Data Repository 218145 Parolari et al., 218, A balancing act of crust creation and destruction along the western Mexican convergent margin: Geology, https://doi.org/1.113/g39972.1. 218145_Tables DR1-DR4.xls
