Lecture 36. Igneous geochemistry
|
|
|
- Hugh Gray
- 6 years ago
- Views:
Transcription
1 Lecture 36 Igneous geochemistry Reading - White Chapter 7 Today 1. Overview 2. solid-melt distribution coefficients Igneous geochemistry The chemistry of igneous systems provides clues to a number of important whole-earth processes, including the processes and timing of planetary differentiation, the production and destruction of the lithosphere, and the relationships between magmatic styles, composition and plate-tectonic environment. The planet s most volcanically active zones are indicated schematically in the figure. 1
2 Igneous geochemistry Magmatism occurs in extrusive (volcanic) and intrusive (plutonic) forms. Estimates of the volumes of magmatic rock produced each year in the four types of plate-tectonic environment are listed in the table below, broken into categories of extrusive (volcanic) and intrusive (plutonic). ( not shown) * * * * * * * * * * Almost entirely basalt * * mixture of basaltic, intermediate and silicic Igneous geochemistry We ve already seen that the Earth s crustal rocks are bimodal in density, and by inference, composition. Here s a composition histogram. Mafic Silicic 2
3 Igneous geochemistry The magmas that ultimately produce the crust are commonly referred to as mafic (rich in Mg and Fe, poor in Si) and silicic (the opposite). But, the bimodality is far from perfect. A range of compositions and processes form more of a continuum of igneous rock compositions. Some magmas represent melts of the mantle whereas others represent melts of the crust, particularly on the continents. Other magmas are mixtures thereof. Mafic Silicic Early Igneous geochemistry History By the early 1900s, a great deal was already known about the chemical compositions of igneous rocks. However, an understanding of why certain compositions occurred in certain places had to wait until the advent of plate tectonics theory in the 1960s. The first large compendium of major-element analyses of igneous rocks from around the world was published in Using 5159 samples, it demonstrated that most igneous rocks are mixtures of just 10 major elements (O, Si, Al, Mg, Fe, Ca, Ti, Na, K, P), plus minor (usually <1%) amounts of Mn and H 2 O. 3
4 Major Elements Chemical compositions of igneous rocks are usually reported as weight % (wt%) of each element as an oxide. There are typically 9 major and 2 minor element oxides listed in a rock analysis. This is true when Fe is reported as total FeO or total Fe 2 O 3. Sometimes both oxidation states [Fe 2+ (FeO) and Fe 3+ (Fe 2 O 3 )] are analyzed and reported separately, as in the olivine thermometry homework problem we had earlier this semester. Be sure to check this aspect when scrutinizing rock analysis data. Igneous geochemistry The early data base was strongly biased toward rocks from easily-accessed locations, nearly all on land, so some details about global variations in composition have changed. M SiO 2 content in igneous rocks is bimodal, as is CaO and K 2 O M Each of the other major element oxides has one dominant range of composition 4
5 Igneous geochemistry The major elements are found in different proportions in the main minerals of igneous rocks, and these minerals vary in proportion and composition with rock type. Silicic Mafic intermediate ultramafic Igneous geochemistry TAS diagrams Some of you have already taken igneous petrology (the study of the distributions of minerals in igneous rocks and their relationship to the conditions of melting and crystallization). Many others of you will be taking such a course, so we won t focus here on the major element or mineral compositions of igneous rocks. These figures summarize one of the common chemical classifications of the main volcanic and plutonic rock types in terms of SiO 2 and total alkalis (TAS = Na 2 O + K 2 O) concentrations in wt%. 5
6 Mafic Silicic Igneous geochemistry One potential compositional evolution path of basaltic magmas as a function of Temperature, SiO 2, and MgO is: Harker diagram Pseudo Harker diagram Note reverses axis order inflections in the trends indicate the appearance or disappearance of minerals. 6
7 Igneous geochemistry We will instead focus mainly on evidence provided by the trace elements, which are covered in less detail in petrology courses. Unlike major element analyses, trace element data are usually are reported as an element s relative concentration by mass (e.g., ppm, ppb). By definition, trace elements are present at concentrations less than about 0.1 wt%. Igneous geochemistry As we saw last week, trace elements provide key insights into the composition of the mantle. We will look in the next few days at how they are used as tracers of the composition of mantle and crustal source rocks that melt to produce magmas. Trace elements also yield important information about the processes and conditions of melting and crystallization. Trace elements usually do not form the major rock-forming phases. Instead, they partition themselves among the different major phases as contaminants, according to M ionic radius M ionic charge M electronegativity M lattice energy of substitution site 7
8 "Goldschmidt's Rules" These outline the conditions for trace element partitioning between igneous phases. Ions will substitute readily for each other in a mineral lattice if Size: Their ionic radii differ by <15%. 2. Charge: They have the same charge or ±1 unit of charge difference (substitution with greater charge differences may occur but to a significantly lesser degree). Of two ions with the charge and radius to occupy a lattice site The ion with the higher ionic potential (z/r) is favored because it will make stronger bonds. A fourth rule was added more recently by Ringwood: 4. The ion with the most similar electronegativity to that of the major element being replaced will be favored because it destabilizes the crystal lattice the least. Trace element partitioning example This diagram shows contours of the clinopyroxene-melt distribution (partition) coefficient for various ions as a function of charge and radius (i.e., primarily rules #1 and #2) Rb K Ba 120 Sr Pb 100 Ca La Y RE E Th Mn Lu U 80 V Li Fe Co Sc Hf Mg 0.2 Ni 60 Cr Ga Zr Ta, N b >1 Ti 0.01 P Be Ionic Charge Figure Ionic radius (picometers) vs. ionic charge contoured for clinopyroxene/liquid partition coefficients. Cations normally present in clinopyroxene are Ca, Mg, and Fe, shown by symbols. Elements whose charge and ionic radius most closely match that of the major elements have the highest partition coefficients. modified from White, Geochemistry 8
9 Trace element partitioning Size is fairly intuitive control, since the substituting ion needs to fit into a mineral lattice: Too big or too small a won't be energetically stable. Charge is also intuitive, since charge must be balance within a lattice and if a charge imbalance is generated by a substitution, a second substitution must occur to correct for this. Electronegativity is harder to visualize, but the disruption of replacing a greedy element with a giving element or vice versa is too much for a lattice to take. example: we rarely see substitutions of like-charge and like-size pairs like Na + - Cu + and Ca +2 and Cd +2 due to electronegativity differences. Trace element distribution We can determine whether a particular substitution is favored or not by using the solid-melt distribution (partition) coefficient. Recall the equation for the simple case of melt + one solid: A k d = [conc. of A] solid /[conc. of A] melt For a multi-phase system (one melt + more than one solid; e.g., several minerals), we use the bulk distribution coefficient: Bulk A k d = A K d = D A = k A = [modal conc. of A] solids /[conc. of A] melt 9
10 Trace element distribution K d and D A values tell us about the tendency of an element to be proportioned between coexisting melt and solids. D A > 1 The element is compatible (a.k.a. captured ). D A = 1 The element is neutral (a.k.a. camouflaged ). D A < 1 The element is incompatible (a.k.a. released ) Trace element distribution A rigorous definition of bulk k d ( D ) for a multiphase solid (a, b, c, j) + one melt system is: D = ak d a + bk d b + gk d c + k d j = x j k d j where a, b, g, and are the proportions of the minerals that comprise the solid, and k da, k db, k dc, k d j are the mineral-melt partition coefficients for each of the different minerals, a, b, c, j that are in equilibrium with the melt. X j is the mole fraction of mineral a to j present. 10
11 Trace element distribution in addition to ionic charge, radius, and electronegativity, the temperature and pressure of the system play a key role in trace element substitution, because as we ve seen throughout the course, T and P determine which solids will be present in a given system. Also, structural controls on crystal lattice energy are such that each type of lattice site has a specific energy of ion substitution. For a given crystal lattice, that energy is a function of T and P. Trace element distribution To summarize, the energy of substitution is minimized (made most favorable) for substitution by the right ion; that is, the one with the best combination of size, charge, electronegativity. as a function of T and P A substitution into a phase becomes less favorable (i.e., k d goes down) the more any of these values vary from the ideal. The incompatible elements have k d < 1 in all the common mantle minerals (and D < 1 in mantle rocks) because their substitution energies are high in all of these minerals. 11
12 Trace element distribution We can understand quite a lot about k d differences by observing how k d varies in a given mineral as a function of ionic size in an iso-charged series (same valence or oxidation number). This helps us understand the structural controls on the distribution of ions between a mineral and a melt. Here s an example for olivine and melt. These types of diagrams are known as Onuma diagrams, after their originator. Examples for plagioclase and clinopyroxene are given in the next three slides. Mafic Silicic Plagioclase has one k d peak at 1.18 Å, which is the radius for the site into which ion substitution occurs (ignoring Al, which is part of the Al-Si structural framework). However, the actual value of the partition coefficient depends on the overall melt composition. 12
13 Trace element distribution Lava composition and temperature determine the An number of the plagioclase feldspar. Remember... An 100 = 100% anorthite (CaAl 2 Si 2 O 8 ) An 0 = 100% albite (NaAlSi 3 O 8 ) An 50 = an equal mixture of the two Notice that K d Sr varies from 5 in basalt to 50 in rhyolite. Trace element distribution The double peak in the augite curve is from substitution into two structural sites with radii of 0.79 Å and 1.01 Å. 13
14 Trace element distribution The very latest models for partition coefficients incorporate lattice strain and electrostatic factors to derive best-fit parameterizations. Notice that the plagioclase peak is still at 1.18 Å. Also note that only the peak at 1.10 Å is shown in this clinopyroxene diagram (i.e., the 0.79 Å peak is still there, but the figure s scale stops at 0.8 Å). If you need a k d value for a given element but can t find a published value, you can estimate it from such model curves. Trace element distribution Compilations of measured values for mineral-melt distribution coefficients for many of the geochemically important elements are given in the next two slides (as well as in your reading). There are two tables: one for mafic lavas and one for silicic lavas. As we saw in the Onuma diagrams for plagioclase, the k d values for a given mineral are often quite different in mafic vs. silicic melts. These two tables are relatively complete, but the data are only for magmas at atmospheric pressure and the data are quite old. Slightly different values apply at higher pressures, but we ll assume for the purposes of this course that we can use low- and moderately high-pressure values interchangeably. Note: when melt solidifies rapidly, it forms a glassy "matrix" of an igneous rock. Slow cooling magmas tend to have little to none of this. 14
15 Trace element distribution coefficients Mafic Trace element distribution coefficients Silicic 15
16 Trace element distribution coefficients This table from White s book is compiled from newer data, but includes fewer elements Trace element distribution coefficients You can find a great deal of data at the GERM (geochemical earth reference model) website ( Select databases, and then choose partition coefficients. There are multiple ways to search (by element, rock type, mineral, etc., but this periodic chart search is the easiest to execute. This link will be useful for your final exam. 16
17 Trace element distribution As mentioned above, the bulk K d, D, is a weighted average of all the individual mineral-melt K d s for the system. Let's calculate bulk K d values for Rb and Co for melting within the shallow mantle of a basalt composed of 45% olivine, 35% opx and 20% cpx using the data from the Henderson tables as an example. We will use K d = ak d a + bk d b + gk d c K ol d K opx d K cpx d Bulk K d or D Rb ~ * * *0.04 = Co 3.8 ~ * * *1.2 = 3.0 Rb behaves incompatibly, Co behaves compatibly in this system. 17
Chapter 9: Trace Elements
 Lecture 13 Introduction to Trace Elements Wednesday, March 9, 2005 Chapter 9: Trace Elements Note magnitude of major element changes Figure 8-2. Harker variation diagram for 310 analyzed volcanic rocks
Lecture 13 Introduction to Trace Elements Wednesday, March 9, 2005 Chapter 9: Trace Elements Note magnitude of major element changes Figure 8-2. Harker variation diagram for 310 analyzed volcanic rocks
Trace Elements. Today s lecture
 Trace Elements 300 Ni 200 ppm 100 0 300 Zr 200 100 0 40 50 60 70 80 SiO 2 wt. % Updates: M&M due date: Tuesday Today s lecture Topics: Trace element compositions Trace element behavior Partitioning Spider(
Trace Elements 300 Ni 200 ppm 100 0 300 Zr 200 100 0 40 50 60 70 80 SiO 2 wt. % Updates: M&M due date: Tuesday Today s lecture Topics: Trace element compositions Trace element behavior Partitioning Spider(
Chapter 9: Trace Elements
 Chapter 9: Trace Elements Note magnitude of major element changes Figure 8.2. Harker variation diagram for 310 analyzed volcanic rocks from Crater Lake (Mt. Mazama), Oregon Cascades. Data compiled by Rick
Chapter 9: Trace Elements Note magnitude of major element changes Figure 8.2. Harker variation diagram for 310 analyzed volcanic rocks from Crater Lake (Mt. Mazama), Oregon Cascades. Data compiled by Rick
Worked Example of Batch Melting: Rb and Sr
 Worked Example of Batch Melting: Rb and Sr Basalt with the mode: Table 9.2. Conversion from mode to weight percent Mineral Mode Density Wt prop Wt% ol 15 3.6 54 0.18 cpx 33 3.4 112.2 0.37 plag 51 2.7 137.7
Worked Example of Batch Melting: Rb and Sr Basalt with the mode: Table 9.2. Conversion from mode to weight percent Mineral Mode Density Wt prop Wt% ol 15 3.6 54 0.18 cpx 33 3.4 112.2 0.37 plag 51 2.7 137.7
Earth and Planetary Materials
 Earth and Planetary Materials Spring 2013 Lecture 4 2013.01.16 Example Beryl Be 3 Al 2 (SiO 3 ) 6 Goshenite Aquamarine Emerald Heliodor Red beryl Morganite pure Fe 2+ & Fe 3+ Cr 3+ Fe 3+ Mn 3+ Mn 2+ Rules
Earth and Planetary Materials Spring 2013 Lecture 4 2013.01.16 Example Beryl Be 3 Al 2 (SiO 3 ) 6 Goshenite Aquamarine Emerald Heliodor Red beryl Morganite pure Fe 2+ & Fe 3+ Cr 3+ Fe 3+ Mn 3+ Mn 2+ Rules
LAB 9: ULTRAMAFIC ROCKS, CUMULATES AND MELT SOURCES
 Geology 316 (Petrology) (03/26/2012) Name LAB 9: ULTRAMAFIC ROCKS, CUMULATES AND MELT SOURCES INTRODUCTION Ultramafic rocks are igneous rocks containing less than 10% felsic minerals (quartz + feldspars
Geology 316 (Petrology) (03/26/2012) Name LAB 9: ULTRAMAFIC ROCKS, CUMULATES AND MELT SOURCES INTRODUCTION Ultramafic rocks are igneous rocks containing less than 10% felsic minerals (quartz + feldspars
How many molecules? Pyrite FeS 2. Would there be any other elements in there???
 How many molecules? Pyrite FeS 2 Would there be any other elements in there??? Goldschmidt s rules of Substitution 1. The ions of one element can extensively replace those of another in ionic crystals
How many molecules? Pyrite FeS 2 Would there be any other elements in there??? Goldschmidt s rules of Substitution 1. The ions of one element can extensively replace those of another in ionic crystals
Effect of tectonic setting on chemistry of mantle-derived melts
 Effect of tectonic setting on chemistry of mantle-derived melts Lherzolite Basalt Factors controlling magma composition Composition of the source Partial melting process Fractional crystallization Crustal
Effect of tectonic setting on chemistry of mantle-derived melts Lherzolite Basalt Factors controlling magma composition Composition of the source Partial melting process Fractional crystallization Crustal
12 Chemistry (Mg,Fe) 2 SiO 4 Olivine is forms what is called an isomorphous solid solution series that ranges between two end members: Forsterite Mg
 11 Olivine Structure Olivine is a common green or brown rock forming minerals which consists of a solid-solution series between Forsterite (Fo) and Fayalite (Fa). It is an orthorhombic orthosilicate with
11 Olivine Structure Olivine is a common green or brown rock forming minerals which consists of a solid-solution series between Forsterite (Fo) and Fayalite (Fa). It is an orthorhombic orthosilicate with
DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER
 Geol 2312 Igneous and Metamorphic Petrology Spring 2009 Name DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER Objective: This exercise is intended to improve understanding
Geol 2312 Igneous and Metamorphic Petrology Spring 2009 Name DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER Objective: This exercise is intended to improve understanding
EPSC 233. Compositional variation in minerals. Recommended reading: PERKINS, p. 286, 41 (Box 2-4).
 EPSC 233 Compositional variation in minerals Recommended reading: PERKINS, p. 286, 41 (Box 2-4). Some minerals are nearly pure elements. These are grouped under the category of native elements. This includes
EPSC 233 Compositional variation in minerals Recommended reading: PERKINS, p. 286, 41 (Box 2-4). Some minerals are nearly pure elements. These are grouped under the category of native elements. This includes
Chapter 4 Up from the Inferno: Magma and Igneous Rocks
 Chapter 4 Up from the Inferno: Magma and Igneous Rocks Up from the Inferno: Magma and Igneous Rocks Updated by: Rick Oches, Professor of Geology & Environmental Sciences Bentley University Waltham, Massachusetts
Chapter 4 Up from the Inferno: Magma and Igneous Rocks Up from the Inferno: Magma and Igneous Rocks Updated by: Rick Oches, Professor of Geology & Environmental Sciences Bentley University Waltham, Massachusetts
Plate tectonics, rock cycle
 Dikes, Antarctica Rock Cycle Plate tectonics, rock cycle The Rock Cycle A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one
Dikes, Antarctica Rock Cycle Plate tectonics, rock cycle The Rock Cycle A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one
GEOL 2312 Igneous and Metamorphic Petrology Spring 2016 Score / 58. Midterm 1 Chapters 1-10
 GEOL 2312 Igneous and Metamorphic Petrology Name KEY Spring 2016 Score / 58 Midterm 1 Chapters 1-10 1) Name two things that petrologists want to know about magmas (1 pt) Formation, source, composition,
GEOL 2312 Igneous and Metamorphic Petrology Name KEY Spring 2016 Score / 58 Midterm 1 Chapters 1-10 1) Name two things that petrologists want to know about magmas (1 pt) Formation, source, composition,
Earth Science 11: Earth Materials: Rock Cycle
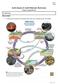 Name: Date: Earth Science 11: Earth Materials: Rock Cycle Chapter 2, pages 44 to 46 2.1: Rock Cycle What is a Rock? A solid mass of mineral or mineral-like matter that occurs naturally as part of our planet
Name: Date: Earth Science 11: Earth Materials: Rock Cycle Chapter 2, pages 44 to 46 2.1: Rock Cycle What is a Rock? A solid mass of mineral or mineral-like matter that occurs naturally as part of our planet
Harker diagram for Crater Lake. Closure problem! What does this mean?
 Announcements Reading: p.155-160 Important not to fall behind in reading or assignments! Turn in field trip notes at the end of next week Gimme your pictures Homework 3 due May 8 Harker diagram for Crater
Announcements Reading: p.155-160 Important not to fall behind in reading or assignments! Turn in field trip notes at the end of next week Gimme your pictures Homework 3 due May 8 Harker diagram for Crater
Imagine the first rock and the cycles that it has been through.
 A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one type of rocky material gets transformed into another The Rock Cycle Representation
A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one type of rocky material gets transformed into another The Rock Cycle Representation
A Rock is a solid aggregate of minerals.
 Quartz A Rock is a solid aggregate of minerals. Orthoclase Feldspar Plagioclase Feldspar Biotite Four different minerals are obvious in this piece of Granite. The average automobile contains: Minerals
Quartz A Rock is a solid aggregate of minerals. Orthoclase Feldspar Plagioclase Feldspar Biotite Four different minerals are obvious in this piece of Granite. The average automobile contains: Minerals
Chapter 4 Rocks & Igneous Rocks
 Chapter 4 Rocks & Igneous Rocks Rock Definition A naturally occurring consolidated mixture of one or more minerals e.g, marble, granite, sandstone, limestone Rock Definition Must naturally occur in nature,
Chapter 4 Rocks & Igneous Rocks Rock Definition A naturally occurring consolidated mixture of one or more minerals e.g, marble, granite, sandstone, limestone Rock Definition Must naturally occur in nature,
amphibole PART 3 Pyroxene: augite CHAIN SILICATES
 amphibole PART 3 Pyroxene: augite CHAIN SILICATES CHAIN SILICATES = INOSILICATES inos = chains Basic structural group: Si 2 O 6 (each tetrahedra shared two corners) Simple or double chains linked by cations
amphibole PART 3 Pyroxene: augite CHAIN SILICATES CHAIN SILICATES = INOSILICATES inos = chains Basic structural group: Si 2 O 6 (each tetrahedra shared two corners) Simple or double chains linked by cations
Lecture 38. Igneous geochemistry. Read White Chapter 7 if you haven t already
 Lecture 38 Igneous geochemistry Read White Chapter 7 if you haven t already Today. Magma mixing/afc 2. Spot light on using the Rare Earth Elements (REE) to constrain mantle sources and conditions of petrogenesis
Lecture 38 Igneous geochemistry Read White Chapter 7 if you haven t already Today. Magma mixing/afc 2. Spot light on using the Rare Earth Elements (REE) to constrain mantle sources and conditions of petrogenesis
Student Name: College: Grade:
 Student Name: College: Grade: Physical Geology Laboratory IGNEOUS MINERALS AND ROCKS IDENTIFICATION - INTRODUCTION & PURPOSE: In this lab you will learn to identify igneous rocks in hand samples from their
Student Name: College: Grade: Physical Geology Laboratory IGNEOUS MINERALS AND ROCKS IDENTIFICATION - INTRODUCTION & PURPOSE: In this lab you will learn to identify igneous rocks in hand samples from their
Rocks: Materials of the Solid Earth
 1 Rocks: Materials of the Solid Earth Presentation modified from: Instructor Resource Center on CD-ROM, Foundations of Earth Science,, 4 th Edition, Lutgens/Tarbuck, Rock Cycle Igneous Rocks Today 2 Rock
1 Rocks: Materials of the Solid Earth Presentation modified from: Instructor Resource Center on CD-ROM, Foundations of Earth Science,, 4 th Edition, Lutgens/Tarbuck, Rock Cycle Igneous Rocks Today 2 Rock
TWO COMPONENT (BINARY) PHASE DIAGRAMS. Experimental Determination of 2-Component Phase Diagrams
 Page 1 of 12 EENS 211 Earth Materials Tulane University Prof. Stephen A. Nelson TWO COMPONENT (BINARY) PHASE DIAGRAMS This document last updated on 08-Oct-2003 Experimental Determination of 2-Component
Page 1 of 12 EENS 211 Earth Materials Tulane University Prof. Stephen A. Nelson TWO COMPONENT (BINARY) PHASE DIAGRAMS This document last updated on 08-Oct-2003 Experimental Determination of 2-Component
PHY120AExam questions 0.5 points each; 19 True/False, 31 Multiple Choice
 1 PHY120AExam 1 2018 50 questions 0.5 points each; 19 True/False, 31 Multiple Choice True/False Indicate whether the statement is true or false. F 1. The distribution of volcanoes is random.. F 2. The
1 PHY120AExam 1 2018 50 questions 0.5 points each; 19 True/False, 31 Multiple Choice True/False Indicate whether the statement is true or false. F 1. The distribution of volcanoes is random.. F 2. The
Classification of Igneous Rocks
 Classification of Igneous Rocks Textures: Glassy- no crystals formed Aphanitic- crystals too small to see by eye Phaneritic- can see the constituent minerals Fine grained- < 1 mm diameter Medium grained-
Classification of Igneous Rocks Textures: Glassy- no crystals formed Aphanitic- crystals too small to see by eye Phaneritic- can see the constituent minerals Fine grained- < 1 mm diameter Medium grained-
Composition of the Earth and its reservoirs: Geochemical observables
 Composition of the Earth and its reservoirs: Geochemical observables Cin-Ty A. Lee Rice University MYRES-I 2004 The Earth is dynamic and heterogeneous Atmosphere Midocean Ridge Plume Ocean Crust Oceanic
Composition of the Earth and its reservoirs: Geochemical observables Cin-Ty A. Lee Rice University MYRES-I 2004 The Earth is dynamic and heterogeneous Atmosphere Midocean Ridge Plume Ocean Crust Oceanic
Chapter 4 8/27/2013. Igneous Rocks. and Intrusive Igneous Activity. Introduction. The Properties and Behavior of Magma and Lava
 Introduction Chapter 4 Igneous rocks form by the cooling of magma (or lava). Large parts of the continents and all the oceanic crust are composed of. and Intrusive Igneous Activity The Properties and Behavior
Introduction Chapter 4 Igneous rocks form by the cooling of magma (or lava). Large parts of the continents and all the oceanic crust are composed of. and Intrusive Igneous Activity The Properties and Behavior
The Nature of Igneous Rocks
 The Nature of Igneous Rocks Form from Magma Hot, partially molten mixture of solid liquid and gas Mineral crystals form in the magma making a crystal slush Gases - H 2 O, CO 2, etc. - are dissolved in
The Nature of Igneous Rocks Form from Magma Hot, partially molten mixture of solid liquid and gas Mineral crystals form in the magma making a crystal slush Gases - H 2 O, CO 2, etc. - are dissolved in
Thursday, October 4 th
 Thursday, October 4 th Objective: We will use and define the different ways to classify igneous rocks. Warm-up: 1. Which type of lava is most viscous? 2. Which type of lava has the least amount of silicate?
Thursday, October 4 th Objective: We will use and define the different ways to classify igneous rocks. Warm-up: 1. Which type of lava is most viscous? 2. Which type of lava has the least amount of silicate?
Lecture 25 Subduction Related Magmatism
 Lecture 25 Subduction Related Magmatism Monday, May 2 nd 2005 Subduction Related Magmatism Activity along arcuate volcanic chains along subduction zones Distinctly different from the mainly basaltic provinces
Lecture 25 Subduction Related Magmatism Monday, May 2 nd 2005 Subduction Related Magmatism Activity along arcuate volcanic chains along subduction zones Distinctly different from the mainly basaltic provinces
Engineering Geology ECIV 2204
 Engineering Geology ECIV 2204 Instructor : Dr. Jehad Hamad 2017-2016 Chapter (3) Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth
Engineering Geology ECIV 2204 Instructor : Dr. Jehad Hamad 2017-2016 Chapter (3) Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth
WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY
 WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY Brandon E. Schwab Department of Geology Humboldt State University
WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY Brandon E. Schwab Department of Geology Humboldt State University
Igneous and Metamorphic Rock Forming Minerals. Department of Geology Mr. Victor Tibane SGM 210_2013
 Igneous and Metamorphic Rock Forming Minerals Department of Geology Mr. Victor Tibane 1 SGM 210_2013 Grotzinger Jordan Understanding Earth Sixth Edition Chapter 4: IGNEOUS ROCKS Solids from Melts 2011
Igneous and Metamorphic Rock Forming Minerals Department of Geology Mr. Victor Tibane 1 SGM 210_2013 Grotzinger Jordan Understanding Earth Sixth Edition Chapter 4: IGNEOUS ROCKS Solids from Melts 2011
GLY 155 Introduction to Physical Geology, W. Altermann. Grotzinger Jordan. Understanding Earth. Sixth Edition
 Grotzinger Jordan Understanding Earth Sixth Edition Chapter 4: IGNEOUS ROCKS Solids from Melts 2011 by W. H. Freeman and Company Chapter 4: Igneous Rocks: Solids from Melts 1 About Igneous Rocks Igneous
Grotzinger Jordan Understanding Earth Sixth Edition Chapter 4: IGNEOUS ROCKS Solids from Melts 2011 by W. H. Freeman and Company Chapter 4: Igneous Rocks: Solids from Melts 1 About Igneous Rocks Igneous
Block: Igneous Rocks. From this list, select the terms which answer the following questions.
 Geology 12 Name: Mix and Match: Igneous Rocks Refer to the following list. Block: porphyritic volatiles mafic glassy magma mixing concordant discontinuous reaction series igneous vesicular partial melting
Geology 12 Name: Mix and Match: Igneous Rocks Refer to the following list. Block: porphyritic volatiles mafic glassy magma mixing concordant discontinuous reaction series igneous vesicular partial melting
Earth Materials I Crystal Structures
 Earth Materials I Crystal Structures Isotopes same atomic number, different numbers of neutrons, different atomic mass. Ta ble 1-1. Su mmar y of quantu m num bers Name Symbol Values Principal n 1, 2,
Earth Materials I Crystal Structures Isotopes same atomic number, different numbers of neutrons, different atomic mass. Ta ble 1-1. Su mmar y of quantu m num bers Name Symbol Values Principal n 1, 2,
Lecture 6 - Igneous Rocks and Volcanoes
 Lecture 6 - Igneous Rocks and Volcanoes Learning objectives Understand and be able to predict where and why magma will be forming at different tectonic settings Understand the factors controlling magma
Lecture 6 - Igneous Rocks and Volcanoes Learning objectives Understand and be able to predict where and why magma will be forming at different tectonic settings Understand the factors controlling magma
Chapter 3: Elements and Compounds. 3.1 Elements
 Chapter 3: Elements and Compounds 3.1 Elements An element is a fundamental substance that cannot be broken down by chemical or physical methods to simpler substances. The 118 known elements are nature
Chapter 3: Elements and Compounds 3.1 Elements An element is a fundamental substance that cannot be broken down by chemical or physical methods to simpler substances. The 118 known elements are nature
Atomic Structure & Interatomic Bonding
 Atomic Structure & Interatomic Bonding Chapter Outline Review of Atomic Structure Atomic Bonding Atomic Structure Atoms are the smallest structural units of all solids, liquids & gases. Atom: The smallest
Atomic Structure & Interatomic Bonding Chapter Outline Review of Atomic Structure Atomic Bonding Atomic Structure Atoms are the smallest structural units of all solids, liquids & gases. Atom: The smallest
GEOLOGY. Subject : GEOLOGY (For under graduate student.) Paper No. : Paper 02 Introduction to Geology 02
 GEOLOGY Subject : GEOLOGY (For under graduate student.) Paper No. : Paper 02 Introduction to Geology 02 Topic No. & Title : 37 Magma Bowen Series (Part 01) Academic Script What is Igneous Petrology? Igneous
GEOLOGY Subject : GEOLOGY (For under graduate student.) Paper No. : Paper 02 Introduction to Geology 02 Topic No. & Title : 37 Magma Bowen Series (Part 01) Academic Script What is Igneous Petrology? Igneous
Lecture 3 Rocks and the Rock Cycle Dr. Shwan Omar
 Rocks A naturally occurring aggregate of one or more minerals (e.g., granite), or a body of non-crystalline material (e.g., obsidian glass), or of solid organic material (e.g., coal). Rock Cycle A sequence
Rocks A naturally occurring aggregate of one or more minerals (e.g., granite), or a body of non-crystalline material (e.g., obsidian glass), or of solid organic material (e.g., coal). Rock Cycle A sequence
Trace Elements - Definitions
 Trace Elements - Definitions Elements that are not stoichiometric constituents in phases in the system of interest For example, IG/MET systems would have different trace elements than aqueous systems Do
Trace Elements - Definitions Elements that are not stoichiometric constituents in phases in the system of interest For example, IG/MET systems would have different trace elements than aqueous systems Do
Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100
 Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100 Ray Rector - Instructor Major Concepts 1) Igneous rocks form directly from the crystallization of a magma or lava 2)
Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100 Ray Rector - Instructor Major Concepts 1) Igneous rocks form directly from the crystallization of a magma or lava 2)
Geos 306, Mineralogy Final Exam, Dec 12, pts
 Name: Geos 306, Mineralogy Final Exam, Dec 12, 2014 200 pts 1. (9 pts) What are the 4 most abundant elements found in the Earth and what are their atomic abundances? Create a reasonable hypothetical charge-balanced
Name: Geos 306, Mineralogy Final Exam, Dec 12, 2014 200 pts 1. (9 pts) What are the 4 most abundant elements found in the Earth and what are their atomic abundances? Create a reasonable hypothetical charge-balanced
Pyroxenes (Mg, Fe 2+ ) 2 Si 2 O 6 (monoclinic) and. MgSiO 3 FeSiO 3 (orthorhombic) Structure (Figure 2 of handout)
 Pyroxenes (Mg, Fe 2+ ) 2 Si 2 O 6 (monoclinic) and 20 MgSiO 3 FeSiO 3 (orthorhombic) Structure (Figure 2 of handout) Chain silicate eg Diopside Mg and Fe ions link SiO 3 chains The chain runs up and down
Pyroxenes (Mg, Fe 2+ ) 2 Si 2 O 6 (monoclinic) and 20 MgSiO 3 FeSiO 3 (orthorhombic) Structure (Figure 2 of handout) Chain silicate eg Diopside Mg and Fe ions link SiO 3 chains The chain runs up and down
Lecture 31. Planetary Accretion the raw materials and the final compositions
 Lecture 31 Planetary Accretion the raw materials and the final compositions Reading this week: White Ch 11 (sections 11.1-11.4) Today 1. Boundary conditions for Planetary Accretion Growth and Differentiation
Lecture 31 Planetary Accretion the raw materials and the final compositions Reading this week: White Ch 11 (sections 11.1-11.4) Today 1. Boundary conditions for Planetary Accretion Growth and Differentiation
Lecture 37. Igneous geochemistry. Crystallization Let's look at how we use differences in element distribution to understand crystallization process.
 Reading White Chapter 7 Lecture 37 Igneous geochemistry Today: Using trace elements to study 1. crystallization 2. melting Crystallization Let's look at how we use differences in element distribution to
Reading White Chapter 7 Lecture 37 Igneous geochemistry Today: Using trace elements to study 1. crystallization 2. melting Crystallization Let's look at how we use differences in element distribution to
PLATE TECTONICS, VOLCANISM AND IGNEOUS ROCKS
 PLATE TECTONICS, VOLCANISM AND IGNEOUS ROCKS PLATE TECTONICS TO IGNEOUS ROCKS Internal Heat Seafloor Spreading/Plate Tectonics Volcanism Plate Boundary Intra-plate (hot spot) Divergent Convergent Igneous
PLATE TECTONICS, VOLCANISM AND IGNEOUS ROCKS PLATE TECTONICS TO IGNEOUS ROCKS Internal Heat Seafloor Spreading/Plate Tectonics Volcanism Plate Boundary Intra-plate (hot spot) Divergent Convergent Igneous
General Introduction. The Earth as an evolving geologic body
 General Introduction The Earth as an evolving geologic body Unique/important attributes of Planet Earth 1. Rocky planet w/ strong magnetic field Mercury has a weak field, Mars has a dead field 1 Unique/important
General Introduction The Earth as an evolving geologic body Unique/important attributes of Planet Earth 1. Rocky planet w/ strong magnetic field Mercury has a weak field, Mars has a dead field 1 Unique/important
Chemical bonds. In some minerals, other (less important) bond types include:
 Chemical bonds Chemical bond: force of attraction between two or more atoms/ions Types of bonds in crystals: Ionic bond: electrostatic attraction between two oppositely charged ions. This type of bond
Chemical bonds Chemical bond: force of attraction between two or more atoms/ions Types of bonds in crystals: Ionic bond: electrostatic attraction between two oppositely charged ions. This type of bond
The Periodic Table. Periodic Properties. Can you explain this graph? Valence Electrons. Valence Electrons. Paramagnetism
 Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Magma Formation and Behavior
 Magma Formation and Behavior Introduction: The study of body waves as they pass through Earth's interior provides strong evidence that the Earth's mantle is composed almost entirely of solid ultramafic
Magma Formation and Behavior Introduction: The study of body waves as they pass through Earth's interior provides strong evidence that the Earth's mantle is composed almost entirely of solid ultramafic
Earth Science 11: Minerals
 lname: Date: Earth Science 11: Minerals Purpose: Text Pages: I can identify and classify minerals using their physical and chemical properties 90-111 *This is recommended reading! Matter and Atoms (5.1)
lname: Date: Earth Science 11: Minerals Purpose: Text Pages: I can identify and classify minerals using their physical and chemical properties 90-111 *This is recommended reading! Matter and Atoms (5.1)
GY 111: Physical Geology
 UNIVERSITY OF SOUTH ALABAMA GY 111: Physical Geology Lecture 9: Extrusive Igneous Rocks Instructor: Dr. Douglas W. Haywick Last Time 1) The chemical composition of the crust 2) Crystallization of molten
UNIVERSITY OF SOUTH ALABAMA GY 111: Physical Geology Lecture 9: Extrusive Igneous Rocks Instructor: Dr. Douglas W. Haywick Last Time 1) The chemical composition of the crust 2) Crystallization of molten
Engineering Geology. Igneous rocks. Hussien Al - deeky
 Igneous rocks Hussien Al - deeky 1 The Geology Definition of Rocks In Geology Rock is defined as the solid material forming the outer rocky shell or crust of the earth. There are three major groups of
Igneous rocks Hussien Al - deeky 1 The Geology Definition of Rocks In Geology Rock is defined as the solid material forming the outer rocky shell or crust of the earth. There are three major groups of
N = N 0 e -λt D* = N 0 -N D* = N 0 (1-e -λt ) or N(e λt -1) where N is number of parent atoms at time t, N 0
 N = N 0 e -λt D* = N 0 -N D* = N 0 (1-e -λt ) or N(e λt -1) where N is number of parent atoms at time t, N 0 is initial number of parents, D* is number of radiogenic daughter atoms, and λ is the decay
N = N 0 e -λt D* = N 0 -N D* = N 0 (1-e -λt ) or N(e λt -1) where N is number of parent atoms at time t, N 0 is initial number of parents, D* is number of radiogenic daughter atoms, and λ is the decay
Overview of the KAHT system. Ian E.M. Smith, School of Environment, University of Auckland
 Overview of the KAHT system Ian E.M. Smith, School of Environment, University of Auckland Tonga-Kermadec-New Zealand Arc Developed on the Pacific - Australian convergent margin Mainly intraoceanic except
Overview of the KAHT system Ian E.M. Smith, School of Environment, University of Auckland Tonga-Kermadec-New Zealand Arc Developed on the Pacific - Australian convergent margin Mainly intraoceanic except
REMINDER. MOVIE: Rocks that Originate Underground 5:41 to 12:40
 REMINDER 2 chapters covered next week Sedimentary Rocks Soils and Weathering (first half) Learn vocabulary for both! Turn to Neighbor: Without using your book or notes, try to remember which te mineral
REMINDER 2 chapters covered next week Sedimentary Rocks Soils and Weathering (first half) Learn vocabulary for both! Turn to Neighbor: Without using your book or notes, try to remember which te mineral
Differentiation of Magmas By Fractional Crystallization Modified from Karl Wirth, rev. July 2011
 M&M s Magma Chamber 1 Differentiation of Magmas By Fractional Crystallization Modified from Karl Wirth, rev. July 2011 Objective The objective of this exercise is to gain first-hand knowledge of the process
M&M s Magma Chamber 1 Differentiation of Magmas By Fractional Crystallization Modified from Karl Wirth, rev. July 2011 Objective The objective of this exercise is to gain first-hand knowledge of the process
Topic 3: Periodicity OBJECTIVES FOR TODAY: Fall in love with the Periodic Table, Interpret trends in atomic radii, ionic radii, ionization energies &
 Topic 3: Periodicity OBJECTIVES FOR TODAY: Fall in love with the Periodic Table, Interpret trends in atomic radii, ionic radii, ionization energies & electronegativity The Periodic Table What is the periodic
Topic 3: Periodicity OBJECTIVES FOR TODAY: Fall in love with the Periodic Table, Interpret trends in atomic radii, ionic radii, ionization energies & electronegativity The Periodic Table What is the periodic
Funsheet 8.0 [SCIENCE 10 REVIEW] Gu 2015
![Funsheet 8.0 [SCIENCE 10 REVIEW] Gu 2015 Funsheet 8.0 [SCIENCE 10 REVIEW] Gu 2015](/thumbs/78/77112923.jpg) Funsheet 8.0 [SCIENCE 10 REVIEW] Gu 2015 1. Fill in the following tables. Symbol # # protons electrons # neutrons Atomic number Mass Number Atomic Mass Charge 56 54 83 18 16 32 35 47 1 19 40 1+ 92 241
Funsheet 8.0 [SCIENCE 10 REVIEW] Gu 2015 1. Fill in the following tables. Symbol # # protons electrons # neutrons Atomic number Mass Number Atomic Mass Charge 56 54 83 18 16 32 35 47 1 19 40 1+ 92 241
IGNEOUS ROCKS. SECTION 5.1 What are igneous rocks?
 Date Period Name IGNEOUS ROCKS SECTION.1 What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. basaltic
Date Period Name IGNEOUS ROCKS SECTION.1 What are igneous rocks? In your textbook, read about the nature of igneous rocks. Use each of the terms below just once to complete the following statements. basaltic
Differentiation 2: mantle, crust OUTLINE
 Differentiation 2: mantle, crust OUTLINE Reading this week: Should have been White Ch 10 and 11!! 7- Nov Differentiation of the Earth, Core formation W 10.6.6, 11.4 9- Nov Moon, crust, mantle, atmosphere
Differentiation 2: mantle, crust OUTLINE Reading this week: Should have been White Ch 10 and 11!! 7- Nov Differentiation of the Earth, Core formation W 10.6.6, 11.4 9- Nov Moon, crust, mantle, atmosphere
A Rock is A group of minerals that have been put together in several different ways.
 A Rock is A group of minerals that have been put together in several different ways. Depending on how they are put together, rocks are classified as: 1. Sedimentary 2. Igneous 3. Metamorphic Sedimentary
A Rock is A group of minerals that have been put together in several different ways. Depending on how they are put together, rocks are classified as: 1. Sedimentary 2. Igneous 3. Metamorphic Sedimentary
GEOL 2312 Igneous and Metamorphic Petrology Spring 2009 Sc ore / 40
 GEOL 2312 Igneous and Metamorphic Petrology Name Spring 2009 Sc ore / 40 QUIZ 3 1) Name two geologic features that provide physical evidence for the mineralogy of the earth s mantle (2 pts) Ophiolites,
GEOL 2312 Igneous and Metamorphic Petrology Name Spring 2009 Sc ore / 40 QUIZ 3 1) Name two geologic features that provide physical evidence for the mineralogy of the earth s mantle (2 pts) Ophiolites,
At the beginning. Matter + antimatter. Matter has the advantage. baryons quarks, leptons, electrons, photons (no protons or neutrons)
 At the beginning Matter + antimatter Matter has the advantage baryons quarks, leptons, electrons, photons (no protons or neutrons) Hadrons protons, neutrons Hydrogen, helium (:0 H:He) Origin of the Universe
At the beginning Matter + antimatter Matter has the advantage baryons quarks, leptons, electrons, photons (no protons or neutrons) Hadrons protons, neutrons Hydrogen, helium (:0 H:He) Origin of the Universe
Igneous Rocks. Igneous Rocks - 1. Environment of Formation - Magma - Plutonic - rock that formed within the Earth. Intrusive - Earth s crust.
 Name: Date: Period: Minerals and Rocks The Physical Setting: Earth Science CLASS NOTES - Methods to classify igneous rocks: 1. Environment of Formation - Magma - Plutonic - rock that formed within the
Name: Date: Period: Minerals and Rocks The Physical Setting: Earth Science CLASS NOTES - Methods to classify igneous rocks: 1. Environment of Formation - Magma - Plutonic - rock that formed within the
Earth Science 232 Petrography
 Earth Science 232 Petrography Course notes by Shaun Frape and Alec Blyth Winter 2002 1 Petrology - Introduction Some Definitions Petra Greek for rock Logos Greek for disclosure or explanation Petrology
Earth Science 232 Petrography Course notes by Shaun Frape and Alec Blyth Winter 2002 1 Petrology - Introduction Some Definitions Petra Greek for rock Logos Greek for disclosure or explanation Petrology
1 - C Systems. The system H 2 O. Heat an ice at 1 atm from-5 to 120 o C. Heat vs. Temperature
 1 - C Systems The system H 2 O Heat an ice at 1 atm from-5 to 120 o C Heat vs. Temperature Fig. 6.7. After Bridgman (1911) Proc. Amer. Acad. Arts and Sci., 5, 441-513; (1936) J. Chem. Phys., 3, 597-605;
1 - C Systems The system H 2 O Heat an ice at 1 atm from-5 to 120 o C Heat vs. Temperature Fig. 6.7. After Bridgman (1911) Proc. Amer. Acad. Arts and Sci., 5, 441-513; (1936) J. Chem. Phys., 3, 597-605;
All chemical bonding is based on the following relationships of electrostatics: 2. Each period on the periodic table
 UNIT VIII ATOMS AND THE PERIODIC TABLE 25 E. Chemical Bonding 1. An ELECTROSTATIC FORCE is All chemical bonding is based on the following relationships of electrostatics: The greater the distance between
UNIT VIII ATOMS AND THE PERIODIC TABLE 25 E. Chemical Bonding 1. An ELECTROSTATIC FORCE is All chemical bonding is based on the following relationships of electrostatics: The greater the distance between
GY303 Igneous & Metamorphic Petrology. Lecture 7: Magma Sources and Tectonic Environments
 GY303 Igneous & Metamorphic Petrology Lecture 7: Magma Sources and Tectonic Environments Factors controlling Magma production Source rock composition Amount of fluids, especially H 2 O Pressure (Depth)
GY303 Igneous & Metamorphic Petrology Lecture 7: Magma Sources and Tectonic Environments Factors controlling Magma production Source rock composition Amount of fluids, especially H 2 O Pressure (Depth)
INTRODUCTION ROCK COLOR
 LAST NAME (ALL IN CAPS): FIRST NAME: 6. IGNEOUS ROCKS Instructions: Some rocks that you would be working with may have sharp edges and corners, therefore, be careful when working with them! When you are
LAST NAME (ALL IN CAPS): FIRST NAME: 6. IGNEOUS ROCKS Instructions: Some rocks that you would be working with may have sharp edges and corners, therefore, be careful when working with them! When you are
Igneous Rocks. Igneous Rocks. Genetic Classification of
 Igneous Rocks Fig. 5.1 Genetic Classification of Igneous Rocks Intrusive: crystallized from slowly cooling magma intruded within the Earth s crust; e.g. granite, gabbro 1 Fig. 5.2 Genetic Classification
Igneous Rocks Fig. 5.1 Genetic Classification of Igneous Rocks Intrusive: crystallized from slowly cooling magma intruded within the Earth s crust; e.g. granite, gabbro 1 Fig. 5.2 Genetic Classification
Standard 2, Objective 1: Evaluate the source of Earth s internal heat and the evidence of Earth s internal structure.
 Standard 2: Students will understand Earth s internal structure and the dynamic nature of the tectonic plates that form its surface. Standard 2, Objective 1: Evaluate the source of Earth s internal heat
Standard 2: Students will understand Earth s internal structure and the dynamic nature of the tectonic plates that form its surface. Standard 2, Objective 1: Evaluate the source of Earth s internal heat
9/4/2015. Feldspars White, pink, variable Clays White perfect Quartz Colourless, white, red, None
 ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks Chapter 3.0: Weathering & soils Chapter 4.0: Geological
ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks Chapter 3.0: Weathering & soils Chapter 4.0: Geological
Geochemical and mineralogical technics to investigate the lithosphere and the asthenosphere. 07/11/2017 GEO-DEEP 9300 Claire Aupart
 Geochemical and mineralogical technics to investigate the lithosphere and the asthenosphere 07/11/2017 GEO-DEEP 9300 Claire Aupart Introduction Introduction Getting samples Cores: Maximum depth reach in
Geochemical and mineralogical technics to investigate the lithosphere and the asthenosphere 07/11/2017 GEO-DEEP 9300 Claire Aupart Introduction Introduction Getting samples Cores: Maximum depth reach in
The mantle metasomatism: diversity and impact What the mantle xenoliths tell us?
 The mantle metasomatism: diversity and impact What the mantle xenoliths tell us? Mantle metasomatism Physical and chemical processes that are implemented during the flow of magmas and / or fluids within
The mantle metasomatism: diversity and impact What the mantle xenoliths tell us? Mantle metasomatism Physical and chemical processes that are implemented during the flow of magmas and / or fluids within
1 The Earth as a Planet
 General Astronomy (29:61) Fall 2012 Lecture 27 Notes, November 5, 2012 1 The Earth as a Planet As we start studying the planets, we begin with Earth. To begin with, it gives us a different perspective
General Astronomy (29:61) Fall 2012 Lecture 27 Notes, November 5, 2012 1 The Earth as a Planet As we start studying the planets, we begin with Earth. To begin with, it gives us a different perspective
High-T heating stage: application for igneous petrogenesis and mantle processes - melt inclusions as key tools -
 High-T heating stage: application for igneous petrogenesis and mantle processes - melt inclusions as key tools - SZABÓ, Csaba Lithosphere Fluid Research Lab (LRG), Department of Petrology and Geochemistry,
High-T heating stage: application for igneous petrogenesis and mantle processes - melt inclusions as key tools - SZABÓ, Csaba Lithosphere Fluid Research Lab (LRG), Department of Petrology and Geochemistry,
Introduction. Volcano a vent where molten rock comes out of Earth
 Introduction Volcano a vent where molten rock comes out of Earth Example: Kilauea Volcano, Hawaii Hot (~1,200 o C) lava pools around the volcanic vent. Hot, syrupy lava runs downhill as a lava flow. The
Introduction Volcano a vent where molten rock comes out of Earth Example: Kilauea Volcano, Hawaii Hot (~1,200 o C) lava pools around the volcanic vent. Hot, syrupy lava runs downhill as a lava flow. The
Silicates. The most common group of minerals forming the silicate Earth
 Silicates The most common group of minerals forming the silicate Earth 25% of all minerals (~1000) 40% of rock forming minerals 90% of earth s crust i.e those minerals you are likely to find ~100 of earth
Silicates The most common group of minerals forming the silicate Earth 25% of all minerals (~1000) 40% of rock forming minerals 90% of earth s crust i.e those minerals you are likely to find ~100 of earth
CHAPTER 4. Crystal Structure
 CHAPTER 4 Crystal Structure We can assume minerals to be made of orderly packing of atoms or rather ions or molecules. Many mineral properties like symmetry, density etc are dependent on how the atoms
CHAPTER 4 Crystal Structure We can assume minerals to be made of orderly packing of atoms or rather ions or molecules. Many mineral properties like symmetry, density etc are dependent on how the atoms
Chapter 3: Igneous Rocks 3.2 IGNEOUS ROCK ORIGIN
 Chapter 3: Igneous Rocks Adapted by Lyndsay R. Hauber & Michael B. Cuggy (2018) University of Saskatchewan from Deline B, Harris R & Tefend K. (2015) "Laboratory Manual for Introductory Geology". First
Chapter 3: Igneous Rocks Adapted by Lyndsay R. Hauber & Michael B. Cuggy (2018) University of Saskatchewan from Deline B, Harris R & Tefend K. (2015) "Laboratory Manual for Introductory Geology". First
Wed. Oct. 04, Makeup lecture time? Will Friday noon work for everyone? No class Oct. 16, 18, 20?
 Wed. Oct. 04, 2017 Reading: For Friday: Bugiolacchi et al. 2008 Laurence et al. 1998" Makeup lecture time? Will Friday noon work for everyone? No class Oct. 16, 18, 20? Today: Finish Lunar overview (from
Wed. Oct. 04, 2017 Reading: For Friday: Bugiolacchi et al. 2008 Laurence et al. 1998" Makeup lecture time? Will Friday noon work for everyone? No class Oct. 16, 18, 20? Today: Finish Lunar overview (from
CERAMIC GLAZING as an IGNEOUS PROCESS
 GEOL 640: Geology through Global Arts and Artifacts CERAMIC GLAZING as an IGNEOUS PROCESS GLAZE COMPONENTS A glaze is a waterproof silica glass on the surface of a ceramic pot, and was first produced by
GEOL 640: Geology through Global Arts and Artifacts CERAMIC GLAZING as an IGNEOUS PROCESS GLAZE COMPONENTS A glaze is a waterproof silica glass on the surface of a ceramic pot, and was first produced by
1 Electrons and Chemical Bonding
 CHAPTER 13 1 Electrons and Chemical Bonding SECTION Chemical Bonding BEFORE YOU READ After you read this section, you should be able to answer these questions: What is chemical bonding? What are valence
CHAPTER 13 1 Electrons and Chemical Bonding SECTION Chemical Bonding BEFORE YOU READ After you read this section, you should be able to answer these questions: What is chemical bonding? What are valence
6. IGNEOUS ROCKS AND VOLCANIC HAZARDS
 LAST NAME (ALL IN CAPS): FIRST NAME: 6. IGNEOUS ROCKS AND VOLCANIC HAZARDS Instructions: Refer to Laboratory 5 in your lab book on pages 129-152 to answer the questions in this work sheet. Your work will
LAST NAME (ALL IN CAPS): FIRST NAME: 6. IGNEOUS ROCKS AND VOLCANIC HAZARDS Instructions: Refer to Laboratory 5 in your lab book on pages 129-152 to answer the questions in this work sheet. Your work will
Lecture 8: Igneous Petrogenesis. Igneous rock classification Phase relations Mantle melting Trace element geochemistry
 Lecture 8: Igneous Petrogenesis Igneous rock classification Phase relations Mantle melting Trace element geochemistry >70% of Earth s annual volcanic budget is erupted in the oceans. Igneous Rock Classification:
Lecture 8: Igneous Petrogenesis Igneous rock classification Phase relations Mantle melting Trace element geochemistry >70% of Earth s annual volcanic budget is erupted in the oceans. Igneous Rock Classification:
The Geochemistry of Basalts in northeast of Darood Neyshaboor, NE Iran
 The Geochemistry of asalts in northeast of Darood Neyshaboor, NE Iran Mehdi rbabi Tehran 1, H. Mehdizadeh Shahri 2, M. E. Fazel Valipour 3 1) Islamic zad University of Shahrood, Department of Geology 2)
The Geochemistry of asalts in northeast of Darood Neyshaboor, NE Iran Mehdi rbabi Tehran 1, H. Mehdizadeh Shahri 2, M. E. Fazel Valipour 3 1) Islamic zad University of Shahrood, Department of Geology 2)
Differentiation 1: core formation OUTLINE
 Differentiation 1: core formation Reading this week: White Ch 12 OUTLINE Today 1.Finish some slides 2.Layers 3.Core formation 1 Goldschmidt Classification/Geochemical Periodic Chart Elements can be assigned
Differentiation 1: core formation Reading this week: White Ch 12 OUTLINE Today 1.Finish some slides 2.Layers 3.Core formation 1 Goldschmidt Classification/Geochemical Periodic Chart Elements can be assigned
Silicate Structures. Silicate Minerals: Pauling s s Rules and. Elemental Abundance in Crust. Elemental Abundance in Crust: Pauling s s Rules
 Silicate Minerals: Pauling s s Rules and Silicate Structures February 6, 2007 Elemental Abundance in Crust Fe Ion O 2- Si 4+ Al 3+, 3+ Ca Na + K + Mg mol % 2.6 1.4 mol% x charge 4.8 3.8 2.6 1.4 3.8 Sum
Silicate Minerals: Pauling s s Rules and Silicate Structures February 6, 2007 Elemental Abundance in Crust Fe Ion O 2- Si 4+ Al 3+, 3+ Ca Na + K + Mg mol % 2.6 1.4 mol% x charge 4.8 3.8 2.6 1.4 3.8 Sum
23/9/2013 ENGINEERING GEOLOGY. Chapter 2: Rock classification:
 ENGINEERING GEOLOGY Chapter 2: Rock classification: ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks
ENGINEERING GEOLOGY Chapter 2: Rock classification: ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks
Magma Formation and Behavior
 Magma Formation and Behavior Questions What causes mantle rock to melt, resulting in magma formation? Why is magma formation restricted to specific plate tectonic settings? Why are mafic (basaltic) magmas
Magma Formation and Behavior Questions What causes mantle rock to melt, resulting in magma formation? Why is magma formation restricted to specific plate tectonic settings? Why are mafic (basaltic) magmas
Lab 3 - Identification of Igneous Rocks
 Lab 3 - Identification of Igneous Rocks Page - 1 Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly
Lab 3 - Identification of Igneous Rocks Page - 1 Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly
Interpreting Phase Diagrams
 Interpreting Phase Diagrams Understanding chemical reactions requires that we know something about how materials behave as the temperature and pressure change. For a single component (like quartz or ice)
Interpreting Phase Diagrams Understanding chemical reactions requires that we know something about how materials behave as the temperature and pressure change. For a single component (like quartz or ice)
How 2 nd half labs will work
 How 2 nd half labs will work Continue to use your mineral identification skills Learn to describe, classify, interpret rock hand samples: Igneous sedimentary metamorphic volcanic plutonic (1 week) (1 wk)
How 2 nd half labs will work Continue to use your mineral identification skills Learn to describe, classify, interpret rock hand samples: Igneous sedimentary metamorphic volcanic plutonic (1 week) (1 wk)
Igneous Rocks. Definition of Igneous Rocks. Igneous rocks form from cooling and crystallization of molten rock- magma
 Igneous Rocks Definition of Igneous Rocks Igneous rocks form from cooling and crystallization of molten rock- magma Magma molten rock within the Earth Lava molten rock on the Earth s s surface Igneous
Igneous Rocks Definition of Igneous Rocks Igneous rocks form from cooling and crystallization of molten rock- magma Magma molten rock within the Earth Lava molten rock on the Earth s s surface Igneous
100% ionic compounds do not exist but predominantly ionic compounds are formed when metals combine with non-metals.
 2.21 Ionic Bonding 100% ionic compounds do not exist but predominantly ionic compounds are formed when metals combine with non-metals. Forming ions Metal atoms lose electrons to form +ve ions. Non-metal
2.21 Ionic Bonding 100% ionic compounds do not exist but predominantly ionic compounds are formed when metals combine with non-metals. Forming ions Metal atoms lose electrons to form +ve ions. Non-metal
SAMPLE PROBLEMS! 1. From which of the following is it easiest to remove an electron? a. Mg b. Na c. K d. Ca
 SAMPLE PROBLEMS! 1. From which of the following is it easiest to remove an electron? a. Mg b. Na c. K d. Ca 2. Which of the following influenced your answer to number one the most? a. effective nuclear
SAMPLE PROBLEMS! 1. From which of the following is it easiest to remove an electron? a. Mg b. Na c. K d. Ca 2. Which of the following influenced your answer to number one the most? a. effective nuclear
