molecules ISSN
|
|
|
- Isabella Potter
- 5 years ago
- Views:
Transcription
1 Molecules 001, 6, molecules ISSN Facile and Fast Pinacol Rearrangement by All 3 in the Solid State Parviz Rashidi-Ranjbar* and Ebrahim Kianmehr Department of hemistry Tehran University, Box , Tehran, Iran. Tel. (+98) , Fax (+98) * Author to whom correspondence should be addressed; Ranjbar@khayam.ut.ac.ir Received: 1 February 000; in revised form 9 April 001 / Accepted: 10 April 001 / Published: 30 April 001 Abstract: A facile and efficient synthetic procedure for effecting the pinacol rearrangement catalyzed by All 3 in the absence of solvent is developed. The rearrangement product is obtained at room temperature in a few minutes and in almost quantitative yield. Benzylic pinacols rearrange under these conditions, while aliphatic pinacols do not react. Keywords: Pinacol rearrangement, Solid state reactions, All 3, Lewis acid Introduction rganic reactions are found to occur efficiently in the solid state and many examples have been reported so far [1-5]. The pinacol rearrangement is usually carried out under drastic conditions, like heating in H S 4. Toda found that the reaction proceeds faster and more selectively in the solid state by using organic acids like l 3 H and p-ts [6]. It is possible to use a strong Lewis acid for the pinacol rearrangement and to the best of our knowledge, there has been no report on pinacol rearrangement by using a Lewis acid like All 3. This reagent has previously been used for Friedel- rafts alkylation and acylation in the solid state [7] and also in Beckman rearrangements [8] under similar conditions. The novel utility of aluminium chloride as an efficient reagent for pinacol rearrangement is reported in this paper.
2 Molecules 001, Results and Discussion Benzylic pinacols react with All 3 to give the corresponding rearrangement product in almost quantitative yield after only a few minutes. The reaction is easily carried out in an agate mortar and the reaction conditions and the subsequent workup are simple and quick. The pinacol compound is mixed with All 3 in a ratio of 1:3 respectively and is then thoroughly ground at room temperature. A fast reaction happens, Hl evolves and a colored mass is immediately obtained. Workup of the reaction mixture by adding dilute H S 4 and extracting with chloroform affords the rearrangement product in good yield. By adopting the above method, benzylic pinacol compounds have been rearranged to the corresponding pinacolones and it is found that the method is much better than all the methods known so far for its simplicity and high conversion. The list of substrates that have been rearranged by this method is given in Table 1. For anthrapinacol, entry 7, the only product detected was bianthryl which shows that this procedure can be used in systems that aromatize by dehydration. For tetralone pinacol, entry 5, the product of rearrangement was obtained in fairly good yield compared to the procedures described previously [9]. In series of compounds -6 it was found that the selectivity is affected by All 3 and for entry 6 the rearranged product, 8, undergoes keto-enol tautomerization to give 10- phenyl-9-phenanthrol, 6 (Scheme 1). All Scheme 1 Entry 8 shows that for rearrangement under these conditions, the existence of an aryl group adjacent to carbon which bears the group is necessary. The reaction is expected to proceed via the formation of an aluminium adduct 9 which is formed by reaction of aluminium chloride with the group followed by liberation of Hl gas. In the second step the rearrangement takes place by the migration of alkyl, aryl or hydride groups (Scheme ). - All + Ar All 3 R 4 Ar -Hl R 4 Product R R 3 R R 3 Scheme 9
3 Molecules 001, Table 1. Rearrangements of pinacols to the corresponding products. All the reactions were carried out at room temperature. Entry Pinacol product Time (min) Yield X X 4 94 X= no reaction
4 Molecules 001, Acknowledgments Parviz Rashidi Ranjbar is grateful to the Research ouncil of Tehran University for financial support. Professor Tony Durst is highly acknowledged for providing us access to NMR and HRMS facilities. Experimental General Solvents and reagents for the preparation of starting materials were used as received from Aldrich. 1 H- and 13 -NMR spectra were recorded on Varian Unity Plus instruments (500 and 00 MHz). FT-IR spectra was obtained using a Shimadzu 4300 spectrophotometer. General synthetic procedure A mixture of the appropriate pinacol compound and anhydrous aluminium chloride (Aldrich, %) was thoroughly ground in an agate mortar for a few minutes under an efficient hood. Hydrogen chloride is evolved violently. The reaction mixture was worked up by adding H S 4 (N) and extracting with chloroform. The organic layer was dried over Na S 4 and the solvent was removed in vacuo to give the products. Pinacol starting materials were prepared by the methods of Toda [10] and Khurana [11]. Spectral Data ompound 1, 1 H-NMR (500 MHz, Dl 3 ) δ: 6.61(dd, J=7.8 and 1.3 Hz, 1H), 7.03(d, J=7.6Hz, H), 7.06(dt, J=7.7 and 1.Hz, 1H), 7.16 (dt, J=7.5 and 1.1 Hz, H), 7.34(dd, J=8.4 and 1.4Hz, 1H), 7.37(dt, J=7.5 and 1.0Hz, H), 7.43(dt, J=7.6 and 1.0Hz, 1H), 7.79(d, J=7.6Hz, H), 7.8(m, 1H), 7.98(dd, J=7.7 and 1.5Hz, 1H), 8.08(dd, J=8. and 1.0Hz, 1H), 8.18(dd, J=8.1 and 0.3 Hz, 1H); 13 -NMR (15 MHz, Dl 3 ) δ: 68.68, 10.51, 13.17, 14.06, 14.70, , , , 18.58, 18.78, 18.59, , , , , , 139.3, , , 197.1, HRMS calcd for 6 H , found ompound, 1 H-NMR (00 MHz, Dl 3 ) δ: 4.30(s, H), (m, 8H), (m, H); 13 - NMR (50 MHz, Dl 3 ) δ: 45., 16.67, 18.38, 18.44, 19.9, 13.96, , , , HRMS calcd for 14 H , found ompound 3, 1 H-NMR (00 MHz, Dl 3 ) δ: 6.05(s, 1H), (m, 13H), (d, J=7.5Hz, H); 13 -NMR (50 MHz, Dl 3 ) δ: 59.39, 17.14, 18.61, 18.71, 18.96, 19.13, , , , , HRMS calcd for 0 H , found
5 Molecules 001, ompound 4, 1 H-NMR (500 MHz, Dl 3 ) δ: 3.75 (s, 3H), 5.97(s, 1H), 6.84 (m, H), (m, 5H), (m, H), (m, H), (m, 1H), (m, H); 13 -NMR (15 MHz, Dl 3 ) δ: 55.17, 58.55, 16.99, 18.53, 18.6, 18.89, 19.00, 130.1, , 13.90, , , , , HRMS calcd for 1 H , found ompound 5, 1 H-NMR (500 MHz, Dl 3 ) δ: (m, 6H),.15-.1(m, 1H),.7-.33(m, 1H), (m, 4H), (m, 1H), (m, 4H), (m, 1H), (m, H); 13 - NMR (15 MHz, Dl 3 ) δ: 19.17,.33, 9.44, 9.85, 3.31, 36.5, 5.8, 15.91, 16.16, 16.94, 17.70, 17.78, 18.45, 19.51, , , , , 14.47, 14.86, FTIR (1680 cm -1, ), HRMS calcd for 0 H , found ompound 6, 1 H-NMR (500 MHz, Dl 3 ) δ: 5.45(s, ), (m, H), (m, 4H), (m, 4H), 8.38(ddd, J=8., 1.5 and 0.5Hz, 1H), 8.66(ddd, J=8.7, 1. and 0.5Hz, 1H), 8.71(ddd, J=8.7, 1. and 0.5Hz, 1H); 13 -NMR (15 MHz, Dl 3 ) δ: 117.5, 1.51, 1.57, 13.07, 13.99, 14.98, 15.36, 16.41, 16.63, 16.81, 17., 17.85, 18.63, 19.80, , , 13.47, , HRMS calcd for 0 H , found ompound 7, 1 H-NMR (500 MHz, Dl 3 ) δ: (m, 8H), 7.41(dd, J=6.3 and 1.Hz, H), 7.43(dd, 6.3 and 1.Hz, H), 8.13(d, 8.5Hz, 4H), 8.66(s, H); 13 -NMR (15 MHz, Dl 3 ) δ: 15.4, 15.75, 16.78, 17.16, 18.47, , , , HRMS calcd for 8 H , found References: 1. Toda, F. and Imai, N. Ylide reactions in the solid state: a simple procedure for the synthesis of cyclopropanes. oxiranes and aziridines J. hem. Soc. Perkin Trans. 1, 1994, Toda, F.; Tanaka, K. and Hamai, K. Aldol condensation in the absence of solvent: Acceleration of the reaction and enhancement of the stereoselectivity J. hem. Soc. Perkin Trans 1, 1990, Toda, F. Hiroshi, A. Enantioselective Wittig-Horner reaction in the solid state. J. rg. hem.,1990, 55, Toda, F.; Kiyoshige, K.; Ygi, M. NaBH 4 reduction of ketones in the solid state. Angew.hem.Int. Ed. Engl. 1989, 8, Tanaka, K.; Kishigami, S.; Toda, F. Reformatsky and Luche reaction in the absence of solvent J. rg. hem. 1991, 56, a) Toda, F. and Shigemasa, T. Pinacol rearrangement in the solid state. J. hem. Soc. Perkin Trans.1, 1989, b) March, J. Advanced rganic hemistry, 4th Ed., John Wiley & Sons, New York, 199, p Ghiaci, M. and Asghari, Friedel rafts alkylation and acylation in the absence of solvent. Synth. ommun. 1998, 8, Ghiaci, M. and Imanzadeh, H. A facile Beckman rearrangement of oximes with All 3 in the solid state. ibid. 1998, 8,
6 Molecules 001, Yax, E. and urisson, G. Syntheses de produits polycyliques par transpositions. Bull. Soc. hem. Fr. 1967, 11, Toda, F.; Tanaka, K. and Kishigama, S. A new method for coupling aromatic aldehydes and ketones to produce glycols using Zn-Znl in aqueous solution and in the solid state. J. rg. hem. 1990, 55, Khurana, J. M. and Sehgal, A. Rapid pinacolization of carbonyl compounds with aluminium-k. J. hem. Soc., hem. ommun. 1994, 571. Sample Availability: Available from author. 001 by MDPI ( Reproduction is permitted for noncommercial purposes
Unexpected dimerization reaction of 5-methyl-6,7-methylendioxy-1- tetralone in the presence of TMSCl-ethylene glycol
 Issue in Honor of Prof. Joan Bosch ARKIVC 2007 (iv) 82-87 Unexpected dimerization reaction of 5-methyl-6,7-methylendioxy-- tetralone in the presence of TMSCl-ethylene glycol Gema Esteban and Joaquín Plumet
Issue in Honor of Prof. Joan Bosch ARKIVC 2007 (iv) 82-87 Unexpected dimerization reaction of 5-methyl-6,7-methylendioxy-- tetralone in the presence of TMSCl-ethylene glycol Gema Esteban and Joaquín Plumet
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group
 Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group
 Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
A Facile Procedure for the Generation of Dichlorocarbene from the Reaction of Carbon Tetrachloride and Magnesium using Ultrasonic Irradiation
 Molecules 2003, 8, 608-613 molecules ISSN 1420-3049 http://www.mdpi.org A Facile Procedure for the Generation of Dichlorocarbene from the Reaction of Carbon Tetrachloride and Magnesium using Ultrasonic
Molecules 2003, 8, 608-613 molecules ISSN 1420-3049 http://www.mdpi.org A Facile Procedure for the Generation of Dichlorocarbene from the Reaction of Carbon Tetrachloride and Magnesium using Ultrasonic
Microwave assisted solvent free oxidation of hydrobenzoins, benzoins and alcohols with NBS - Al 2 O 3
 General Papers ARKIV 2008 (xiv) 211-215 Microwave assisted solvent free oxidation of hydrobenzoins, benzoins and alcohols with NBS - Al 2 3 Jitender M. Khurana* and Reema Arora Department of hemistry,
General Papers ARKIV 2008 (xiv) 211-215 Microwave assisted solvent free oxidation of hydrobenzoins, benzoins and alcohols with NBS - Al 2 3 Jitender M. Khurana* and Reema Arora Department of hemistry,
2.222 Practice Problems 2003
 2.222 Practice Problems 2003 Set #1 1. Provide the missing starting compound(s), reagent/solvent, or product to correctly complete each of the following. Most people in the class have not done this type
2.222 Practice Problems 2003 Set #1 1. Provide the missing starting compound(s), reagent/solvent, or product to correctly complete each of the following. Most people in the class have not done this type
ζ ε δ γ β α α β γ δ ε ζ
 hem 263 Nov 17, 2016 eactions at the α-arbon The alpha carbon is the carbon adjacent to the carbonyl carbon. Beta is the next one, followed by gamma, delta, epsilon, and so on. 2 ε 2 δ 2 γ 2 2 β α The
hem 263 Nov 17, 2016 eactions at the α-arbon The alpha carbon is the carbon adjacent to the carbonyl carbon. Beta is the next one, followed by gamma, delta, epsilon, and so on. 2 ε 2 δ 2 γ 2 2 β α The
CHM 320 Laboratory Projects Spring, 2009
 M 320 Laboratory Projects Spring, 2009 I. Enantioselective Reduction of Benzofuran-2-yl Methyl Ketone using Enzymes from arrots. Typically, the reduction of an unsymmetrical, achiral ketone with a hydride
M 320 Laboratory Projects Spring, 2009 I. Enantioselective Reduction of Benzofuran-2-yl Methyl Ketone using Enzymes from arrots. Typically, the reduction of an unsymmetrical, achiral ketone with a hydride
CHEM 203. Final Exam December 15, 2010 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
The First Asymmetric Total Syntheses and. Determination of Absolute Configurations of. Xestodecalactones B and C
 Supporting Information The First Asymmetric Total Syntheses and Determination of Absolute Configurations of Xestodecalactones B and C Qiren Liang, Jiyong Zhang, Weiguo Quan, Yongquan Sun, Xuegong She*,,
Supporting Information The First Asymmetric Total Syntheses and Determination of Absolute Configurations of Xestodecalactones B and C Qiren Liang, Jiyong Zhang, Weiguo Quan, Yongquan Sun, Xuegong She*,,
Chapter 16. Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group. Physical Properties of Aldehydes and Ketones. Synthesis of Aldehydes
 Nomenclature of Aldehydes and Ketones Chapter 16 Aldehydes and Ketones I. Aldehydes replace the -e of the parent alkane with -al The functional group needs no number Nucleophilic Addition to the Carbonyl
Nomenclature of Aldehydes and Ketones Chapter 16 Aldehydes and Ketones I. Aldehydes replace the -e of the parent alkane with -al The functional group needs no number Nucleophilic Addition to the Carbonyl
An efficient condensation of 4-oxo-4H-benzopyran-3-carbaldehydes with 3-methyl-1-phenyl-1H-pyrazol-5 (4H)-one
 General Papers ARKIVC 2005 (xiv) 82-86 An efficient condensation of 4-oxo-4H-benzopyran-3-carbaldehydes with 3-methyl-1-phenyl-1H-pyrazol-5 (4H)-one Balaji R. Madje, Santosh S. Shindalkar, Madhav. Ware,
General Papers ARKIVC 2005 (xiv) 82-86 An efficient condensation of 4-oxo-4H-benzopyran-3-carbaldehydes with 3-methyl-1-phenyl-1H-pyrazol-5 (4H)-one Balaji R. Madje, Santosh S. Shindalkar, Madhav. Ware,
Lecture Notes Chemistry Mukund P. Sibi Lecture 36 Synthesis of Amines
 Lecture otes hemistry 42-2008 Mukund P. Sibi Synthesis of Amines Amines can be prepared from a variety of starting materials. All of these methods involve functional group transformations. The main methods
Lecture otes hemistry 42-2008 Mukund P. Sibi Synthesis of Amines Amines can be prepared from a variety of starting materials. All of these methods involve functional group transformations. The main methods
Name. Department of Chemistry SUNY/Oneonta. Chem Organic Chemistry II Examination #3 - March 31, 2003
 INSTRUTINS Name Department of hemistry SUNY/neonta hem 322 - rganic hemistry II Examination #3 - March 31, 2003 This examination has two parts. Part I is in multiple choice format and the answers should
INSTRUTINS Name Department of hemistry SUNY/neonta hem 322 - rganic hemistry II Examination #3 - March 31, 2003 This examination has two parts. Part I is in multiple choice format and the answers should
2.222 Introductory Organic Chemistry II Midterm Exam
 NAME: STUDENT NUMBE: Page 1 of 7 University of Manitoba Department of hemistry 2.222 Introductory rganic hemistry II Midterm Exam Wednesday February 20, 2002 Put all answers in the spaces provided. If
NAME: STUDENT NUMBE: Page 1 of 7 University of Manitoba Department of hemistry 2.222 Introductory rganic hemistry II Midterm Exam Wednesday February 20, 2002 Put all answers in the spaces provided. If
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Rh 2 (Ac) 4 -Catalyzed 2,3-Migration of -rrocenecarboxyl -Diazocarbonyl
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Rh 2 (Ac) 4 -Catalyzed 2,3-Migration of -rrocenecarboxyl -Diazocarbonyl
Silver-catalyzed decarboxylative acylfluorination of styrenes in aqueous media
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Silver-catalyzed decarboxylative acylfluorination of styrenes in aqueous
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Silver-catalyzed decarboxylative acylfluorination of styrenes in aqueous
INTRODUCTORY ORGANIC CHEMISTRY II --- PROBLEM SET #2
 CHEM 222 Section 01 Fall 2007 Dr. C. Rogers Page 1 of 5 INTRDUCTRY RGANIC CHEMISTRY II --- PRBLEM SET #2 DISTRIBUTED: Thurs.Nov.15 th. CMPLETIN DEADLINE: Thurs.Nov.29 th @ 10:15am. INSTRUCTINS: Work through
CHEM 222 Section 01 Fall 2007 Dr. C. Rogers Page 1 of 5 INTRDUCTRY RGANIC CHEMISTRY II --- PRBLEM SET #2 DISTRIBUTED: Thurs.Nov.15 th. CMPLETIN DEADLINE: Thurs.Nov.29 th @ 10:15am. INSTRUCTINS: Work through
Supporting Information
 Meyer, Ferreira, and Stoltz: Diazoacetoacetic acid Supporting Information S1 2-Diazoacetoacetic Acid, an Efficient and Convenient Reagent for the Synthesis of Substituted -Diazo- -ketoesters Michael E.
Meyer, Ferreira, and Stoltz: Diazoacetoacetic acid Supporting Information S1 2-Diazoacetoacetic Acid, an Efficient and Convenient Reagent for the Synthesis of Substituted -Diazo- -ketoesters Michael E.
CHM 292 Final Exam Answer Key
 CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
235 Organic II. Final Exam Review REACTIONS OF CONJUGATED DIENES 1,2 VS 1,4 ADDITION REACTIONS OF CONJUGATED DIENES
 b. the ompound 7 i 1 Spectral Data: singlet, 196.5 ppm singlet, 14.1 ppm singlet, 14.4 ppm doublet, 19.1 ppm doublet, 18.5 ppm 1 MR Mass Spectrum Absorbance Intensity Infrared Spectrum 65 91 9. Structure:
b. the ompound 7 i 1 Spectral Data: singlet, 196.5 ppm singlet, 14.1 ppm singlet, 14.4 ppm doublet, 19.1 ppm doublet, 18.5 ppm 1 MR Mass Spectrum Absorbance Intensity Infrared Spectrum 65 91 9. Structure:
Synthetic Studies on Norissolide; Enantioselective Synthesis of the Norrisane Side Chain
 rganic Lett. (Supporting Information) 1 Synthetic Studies on Norissolide; Enantioselective Synthesis of the Norrisane Side Chain Charles Kim, Richard Hoang and Emmanuel A. Theodorakis* Department of Chemistry
rganic Lett. (Supporting Information) 1 Synthetic Studies on Norissolide; Enantioselective Synthesis of the Norrisane Side Chain Charles Kim, Richard Hoang and Emmanuel A. Theodorakis* Department of Chemistry
Supporting Information
 Supporting Information Efficient Short Step Synthesis of Corey s Tamiflu Intermediate Nsiama Tienabe Kipassa, Hiroaki kamura, * Kengo Kina, Tetsuo Iwagawa, and Toshiyuki Hamada Department of Chemistry
Supporting Information Efficient Short Step Synthesis of Corey s Tamiflu Intermediate Nsiama Tienabe Kipassa, Hiroaki kamura, * Kengo Kina, Tetsuo Iwagawa, and Toshiyuki Hamada Department of Chemistry
Chapter 12 Alcohols from Carbonyl Compounds: Oxidation-Reduction and Organometallic Compounds
 Chapter 12 Alcohols from Carbonyl Compounds: Oxidation-Reduction and Organometallic Compounds Introduction Several functional groups contain the carbonyl group Carbonyl groups can be converted into alcohols
Chapter 12 Alcohols from Carbonyl Compounds: Oxidation-Reduction and Organometallic Compounds Introduction Several functional groups contain the carbonyl group Carbonyl groups can be converted into alcohols
Supporting Information
 Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Concise Stereoselective Synthesis of ( )-Podophyllotoxin by Intermolecular Fe III -catalyzed Friedel-Crafts Alkylation Daniel Stadler, Thorsten
Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Concise Stereoselective Synthesis of ( )-Podophyllotoxin by Intermolecular Fe III -catalyzed Friedel-Crafts Alkylation Daniel Stadler, Thorsten
Cape Cod Community College
 Cape Cod Community College Departmental Syllabus Prepared by the Department of Natural Sciences & Applied Technology Date of Departmental Approval: February 3, 2014 Date Approved by Curriculum and Programs:
Cape Cod Community College Departmental Syllabus Prepared by the Department of Natural Sciences & Applied Technology Date of Departmental Approval: February 3, 2014 Date Approved by Curriculum and Programs:
molecules ISSN by MDPI
 Molecules 2000, 5, 967-973 molecules ISS 1420-3049 2000 by MDPI http://www.mdpi.org Reactions with Hydrazonoyl Halides. 31. Synthesis of Some ew Pyrrolidino[3,4-c]pyrazolines, Pyrazoles, and Pyrazolo[3,4-d]pyridazines
Molecules 2000, 5, 967-973 molecules ISS 1420-3049 2000 by MDPI http://www.mdpi.org Reactions with Hydrazonoyl Halides. 31. Synthesis of Some ew Pyrrolidino[3,4-c]pyrazolines, Pyrazoles, and Pyrazolo[3,4-d]pyridazines
An improved preparation of isatins from indoles
 An improved preparation of isatins from indoles Jiro Tatsugi,* Tong Zhiwei, and Yasuji Izawa Department of Applied Chemistry, Faculty of Engineering, Aichi Institute of Technology, Yachigusa, Yakusa-cho,
An improved preparation of isatins from indoles Jiro Tatsugi,* Tong Zhiwei, and Yasuji Izawa Department of Applied Chemistry, Faculty of Engineering, Aichi Institute of Technology, Yachigusa, Yakusa-cho,
For more info visit Aldehyde and Ketones. Preparation of Aldehydes. a. Oxidation of primary alcohols
 Aldehyde and Ketones Preparation of Aldehydes a. Oxidation of primary alcohols Preparation of Ketones: a) Oxidation of Secondary alcohols: d) With Organometallics Reactions of Aldehydes and Ketones: a)
Aldehyde and Ketones Preparation of Aldehydes a. Oxidation of primary alcohols Preparation of Ketones: a) Oxidation of Secondary alcohols: d) With Organometallics Reactions of Aldehydes and Ketones: a)
Supporting Information for Synthesis of C(3) Benzofuran Derived Bis-Aryl Quaternary Centers: Approaches to Diazonamide A
 Fuerst et al. Synthesis of C(3) Benzofuran Derived Bis-Aryl Quaternary Centers: Approaches to Diazonamide A S1 Supporting Information for Synthesis of C(3) Benzofuran Derived Bis-Aryl Quaternary Centers:
Fuerst et al. Synthesis of C(3) Benzofuran Derived Bis-Aryl Quaternary Centers: Approaches to Diazonamide A S1 Supporting Information for Synthesis of C(3) Benzofuran Derived Bis-Aryl Quaternary Centers:
Polar Reactions. Indian Institute of Technology Madras CH 3 OH 2 -H 2 O CH 2. HCl. H 3 C CH 3 neopentyl alcohol CH 3 CH 3. 1,2-shift -H + H 3 C
 Polar Reactions Important points to be discussed in this module: 1. Chemistry of Carbocations with respect to: a. 1,2-shifts of carbocations b. Pinacol-pinacolone rearrangement c. Semipinacolone rearrangement
Polar Reactions Important points to be discussed in this module: 1. Chemistry of Carbocations with respect to: a. 1,2-shifts of carbocations b. Pinacol-pinacolone rearrangement c. Semipinacolone rearrangement
Supporting Information:
 Enantioselective Synthesis of (-)-Codeine and (-)-Morphine Barry M. Trost* and Weiping Tang Department of Chemistry, Stanford University, Stanford, CA 94305-5080 1. Aldehyde 7. Supporting Information:
Enantioselective Synthesis of (-)-Codeine and (-)-Morphine Barry M. Trost* and Weiping Tang Department of Chemistry, Stanford University, Stanford, CA 94305-5080 1. Aldehyde 7. Supporting Information:
Department of Chemistry and Biochemistry, California State University Northridge, Northridge, CA Experimental Procedures
 Supporting Information Low Temperature n-butyllithium-induced [3,3]-Sigmatropic Rearrangement/Electrophile Trapping Reactions of Allyl-1,1- Dichlorovinyl Ethers. Synthesis of - - and -lactones. Aaron Christopher
Supporting Information Low Temperature n-butyllithium-induced [3,3]-Sigmatropic Rearrangement/Electrophile Trapping Reactions of Allyl-1,1- Dichlorovinyl Ethers. Synthesis of - - and -lactones. Aaron Christopher
Chemistry 263 Laboratory Experiment 5: Dibenzalacetone 5/10/18. Introduction
 Chemistry 263 Laboratory Experiment 5: Dibenzalacetone 5/10/18 Introduction We have been examining the electrophilic nature of the carbonyl of late. In particular, we have been trying to determine how
Chemistry 263 Laboratory Experiment 5: Dibenzalacetone 5/10/18 Introduction We have been examining the electrophilic nature of the carbonyl of late. In particular, we have been trying to determine how
Aldehydes and Ketones : Aldol Reactions
 Aldehydes and Ketones : Aldol Reactions The Acidity of the a Hydrogens of Carbonyl Compounds: Enolate Anions Hydrogens on carbons a to carbonyls are unusually acidic The resulting anion is stabilized by
Aldehydes and Ketones : Aldol Reactions The Acidity of the a Hydrogens of Carbonyl Compounds: Enolate Anions Hydrogens on carbons a to carbonyls are unusually acidic The resulting anion is stabilized by
molecules ISSN
 Molecules 2003, 8, 467-471 Second Eurasian Meeting on eterocyclic Chemistry eterocycles in rganic and Combinatorial Chemistry molecules ISS 1420-3049 http://www.mdpi.org Solid-Phase Synthesis of Methyl
Molecules 2003, 8, 467-471 Second Eurasian Meeting on eterocyclic Chemistry eterocycles in rganic and Combinatorial Chemistry molecules ISS 1420-3049 http://www.mdpi.org Solid-Phase Synthesis of Methyl
Chem 263 Nov 19, Cl 2
 Chem 263 Nov 19, 2013 eactions of Enolates: X X alogenation X C 2 Alkylation C Aldol eaction X C Acylation Example: halogenation LDA 2 Chloroacetone is used in tear gas. chloroacetone In this reaction,
Chem 263 Nov 19, 2013 eactions of Enolates: X X alogenation X C 2 Alkylation C Aldol eaction X C Acylation Example: halogenation LDA 2 Chloroacetone is used in tear gas. chloroacetone In this reaction,
Synthetic possibilities Chem 315 Beauchamp 1
 Synthetic possibilities hem Beauchamp Propose reasonable syntheses f the following target molecules (TM-#). You can use the given starting materials and any typical ganic reagents studied in our course
Synthetic possibilities hem Beauchamp Propose reasonable syntheses f the following target molecules (TM-#). You can use the given starting materials and any typical ganic reagents studied in our course
CHAPTER 19: CARBONYL COMPOUNDS III
 CHAPTER 19: CARBONYL COMPOUNDS III A hydrogen bonded to a carbon adjacent to a carbonyl carbon is sufficiently acidic to be removed by a strong base. The carbon adjacent to a carbonyl carbon is called
CHAPTER 19: CARBONYL COMPOUNDS III A hydrogen bonded to a carbon adjacent to a carbonyl carbon is sufficiently acidic to be removed by a strong base. The carbon adjacent to a carbonyl carbon is called
Claisen-Schmidt condensation under solventfree
 Indian Journal of Chemistry Vol. 49B, March 2010, pp. 382-385 Note aisen-schmidt condensation under solventfree conditions K Mogilaiah*, T Kumara Swamy, A Vinay Chandra, N Srivani & K Vidya Department
Indian Journal of Chemistry Vol. 49B, March 2010, pp. 382-385 Note aisen-schmidt condensation under solventfree conditions K Mogilaiah*, T Kumara Swamy, A Vinay Chandra, N Srivani & K Vidya Department
Aziridine in Polymers: A Strategy to Functionalize Polymers by Ring- Opening Reaction of Aziridine
 Electronic Supplementary Material (ESI) for Polymer Chemistry. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information (ESI) Aziridine in Polymers: A Strategy to Functionalize
Electronic Supplementary Material (ESI) for Polymer Chemistry. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information (ESI) Aziridine in Polymers: A Strategy to Functionalize
Accessory Information
 Accessory Information Synthesis of 5-phenyl 2-Functionalized Pyrroles by amino Heck and tandem amino Heck Carbonylation reactions Shazia Zaman, *A,B Mitsuru Kitamura B, C and Andrew D. Abell A *A Department
Accessory Information Synthesis of 5-phenyl 2-Functionalized Pyrroles by amino Heck and tandem amino Heck Carbonylation reactions Shazia Zaman, *A,B Mitsuru Kitamura B, C and Andrew D. Abell A *A Department
Regioselective Ethanolamination and Ketalization of 3-Ph-2,4- diacetyl(diethoxycarbonyl)-5-hydroxy-5-methylcyclohexanones
 Molecules 003, 8, 51-55 molecules ISSN 140-3049 http://www.mdpi.org Regioselective Ethanolamination and Ketalization of 3--,4- diacetyl(diethoxycarbonyl)-5-hydroxy-5-methylcyclohexanones Adele P. Kriven'ko,
Molecules 003, 8, 51-55 molecules ISSN 140-3049 http://www.mdpi.org Regioselective Ethanolamination and Ketalization of 3--,4- diacetyl(diethoxycarbonyl)-5-hydroxy-5-methylcyclohexanones Adele P. Kriven'ko,
Experiment 2 Solvent-free Aldol Condensation between 3,4-dimethoxybenzaldehyde and 1-indanone
 Experiment 2 Solvent-free Aldol Condensation between 3,4-dimethoxybenzaldehyde and 1-indanone Chemical Concepts Carbonyl chemistry, base catalyzed aldol reaction, melting point, recrystallization Green
Experiment 2 Solvent-free Aldol Condensation between 3,4-dimethoxybenzaldehyde and 1-indanone Chemical Concepts Carbonyl chemistry, base catalyzed aldol reaction, melting point, recrystallization Green
September [KV 804] Sub. Code: 3804
![September [KV 804] Sub. Code: 3804 September [KV 804] Sub. Code: 3804](/thumbs/89/97855699.jpg) September 2009 [KV 804] Sub. Code: 3804 (Regulations 2008-2009) (Candidates admitted from 2008-2009 onwards) Paper IV PHARMACEUTICAL ORGANIC CHEMISTRY Time : Three hours Maximum : 70 marks Answer All questions
September 2009 [KV 804] Sub. Code: 3804 (Regulations 2008-2009) (Candidates admitted from 2008-2009 onwards) Paper IV PHARMACEUTICAL ORGANIC CHEMISTRY Time : Three hours Maximum : 70 marks Answer All questions
molecules ISSN
 Molecules 2000, 5, 127-131 molecules ISSN 1420-3049 http://www.mdpi.org Unexpected Thorpe Reaction of an α-alkoxynitrile Lajos Kovács Department of Medicinal Chemistry, University of Szeged, Dóm tér 8.,
Molecules 2000, 5, 127-131 molecules ISSN 1420-3049 http://www.mdpi.org Unexpected Thorpe Reaction of an α-alkoxynitrile Lajos Kovács Department of Medicinal Chemistry, University of Szeged, Dóm tér 8.,
acetaldehyde (ethanal)
 hem 263 Nov 2, 2010 Preparation of Ketones and Aldehydes from Alkenes zonolysis 1. 3 2. Zn acetone 1. 3 2. Zn acetone acetaldehyde (ethanal) Mechanism: 3 3 3 + - oncerted reaction 3 3 3 + ozonide (explosive)
hem 263 Nov 2, 2010 Preparation of Ketones and Aldehydes from Alkenes zonolysis 1. 3 2. Zn acetone 1. 3 2. Zn acetone acetaldehyde (ethanal) Mechanism: 3 3 3 + - oncerted reaction 3 3 3 + ozonide (explosive)
Tautomerism and Keto Enol Equilibrium
 Tautomerism and Keto Enol Equilibrium Enols & enolates are important nucleophiles in organic & biochemistry. Keto-Enol Equilibrium: Tautomerisation can be catalyzed by either acids or bases. Relative stability
Tautomerism and Keto Enol Equilibrium Enols & enolates are important nucleophiles in organic & biochemistry. Keto-Enol Equilibrium: Tautomerisation can be catalyzed by either acids or bases. Relative stability
Tsuji Trost N-Allylation with Allylic Acetates by Using a Cellulose Palladium Catalyst
 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln U.S. Environmental Protection Agency Papers U.S. Environmental Protection Agency 2012 Tsuji Trost N-Allylation with Allylic
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln U.S. Environmental Protection Agency Papers U.S. Environmental Protection Agency 2012 Tsuji Trost N-Allylation with Allylic
INSTRUCTIONS FOR. LITERATURE PROBLEM REPORT CHEM 30121, Fall 2018
 NSTRUCTNS FR LTERATURE PRBLEM REPRT CEM 30121, Fall 2018 Provide a reaction scheme for a fluorescent dye that features at least one reaction that you have performed and/or was covered during the lectures
NSTRUCTNS FR LTERATURE PRBLEM REPRT CEM 30121, Fall 2018 Provide a reaction scheme for a fluorescent dye that features at least one reaction that you have performed and/or was covered during the lectures
Cole Curtis, Chemistry 213. Synthetic #1 FFR. Synthesis and Characterization of 4-methoxychalcone
 1 Cole Curtis, Chemistry 213 Synthetic #1 FFR Synthesis and Characterization of 4-methoxychalcone Introduction Recrystallization is a very effective technique commonly used by chemists to purify solids
1 Cole Curtis, Chemistry 213 Synthetic #1 FFR Synthesis and Characterization of 4-methoxychalcone Introduction Recrystallization is a very effective technique commonly used by chemists to purify solids
CHE1502. Tutorial letter 203/1/2016. General Chemistry 1B. Semester 1. Department of Chemistry
 E1502/203/1/2016 Tutorial letter 203/1/2016 General hemistry 1B E1502 Semester 1 Department of hemistry This tutorial letter contains the answers to the questions in assignment 3. FIRST SEMESTER: KEY T
E1502/203/1/2016 Tutorial letter 203/1/2016 General hemistry 1B E1502 Semester 1 Department of hemistry This tutorial letter contains the answers to the questions in assignment 3. FIRST SEMESTER: KEY T
12/27/2010. Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
Module No and Title. PAPER No: 5 ; TITLE : Organic Chemistry-II MODULE No: 25 ; TITLE: S E 1 reactions
 Subject Chemistry Paper No and Title Module No and Title Module Tag 5; Organic Chemistry-II 25; S E 1 reactions CHE_P5_M25 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. S E 1 reactions 3.1
Subject Chemistry Paper No and Title Module No and Title Module Tag 5; Organic Chemistry-II 25; S E 1 reactions CHE_P5_M25 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. S E 1 reactions 3.1
Molecular Rearrangements
 Ø Migration of one the molecule group from one atom to another within Ø Generally the migrating group never leaves the molecule Ø There are five types of skeletal rearrangements- 1. Electron deficient
Ø Migration of one the molecule group from one atom to another within Ø Generally the migrating group never leaves the molecule Ø There are five types of skeletal rearrangements- 1. Electron deficient
CARBONYL COMPOUNDS: OXIDATION-REDUCTION REACTION
 CARBONYL COMPOUNDS: OXIDATION-REDUCTION REACTION Introduction Several functional groups contain the carbonyl group Carbonyl groups can be converted into alcohols by various reactions Structure of the Carbonyl
CARBONYL COMPOUNDS: OXIDATION-REDUCTION REACTION Introduction Several functional groups contain the carbonyl group Carbonyl groups can be converted into alcohols by various reactions Structure of the Carbonyl
Chap 11. Carbonyl Alpha-Substitution Reactions and Condensation Reactions
 Chap 11. Carbonyl Alpha-Substitution eactions and Condensation eactions Four fundamental reactions of carbonyl compounds 1) Nucleophilic addition (aldehydes and ketones) ) Nucleophilic acyl substitution
Chap 11. Carbonyl Alpha-Substitution eactions and Condensation eactions Four fundamental reactions of carbonyl compounds 1) Nucleophilic addition (aldehydes and ketones) ) Nucleophilic acyl substitution
Aldehydes & Ketones LEVEL I. Phenol (enol form) Phenol is aromatic, so equiulibrium is shifted to the right hand side. b) O
 Subjective Problems Aldehydes & Ketones LEVEL I 1. a) 2,4cyclohexadiene-1-one (keto form) Phenol (enol form) Phenol is aromatic, so equiulibrium is shifted to the right hand side. b) Base This ketone is
Subjective Problems Aldehydes & Ketones LEVEL I 1. a) 2,4cyclohexadiene-1-one (keto form) Phenol (enol form) Phenol is aromatic, so equiulibrium is shifted to the right hand side. b) Base This ketone is
Alpha Substitution and Condensations of Enols and Enolate Ions. Alpha Substitution
 Alpha Substitution and ondensations of Enols and Enolate Ions hap 23 W: 27, 28, 30, 31, 37, 39, 42-44, 47, 51, 54-56 Alpha Substitution Replacement of a hydrogen on the carbon adjacent to the carbonyl,
Alpha Substitution and ondensations of Enols and Enolate Ions hap 23 W: 27, 28, 30, 31, 37, 39, 42-44, 47, 51, 54-56 Alpha Substitution Replacement of a hydrogen on the carbon adjacent to the carbonyl,
Electronic Supplementary Material (ESI) for Chemical Communications This journal is The Royal Society of Chemistry 2012
 Ring Expansion of Alkynyl Cyclopropanes to Highly substituted Cyclobutenes via a N-Sulfonyl-1,2,3-Triazole Intermediate Renhe Liu, Min Zhang, Gabrielle Winston-Mcerson, and Weiping Tang* School of armacy,
Ring Expansion of Alkynyl Cyclopropanes to Highly substituted Cyclobutenes via a N-Sulfonyl-1,2,3-Triazole Intermediate Renhe Liu, Min Zhang, Gabrielle Winston-Mcerson, and Weiping Tang* School of armacy,
Chemistry 283g Experiment 4
 Chemistry 283g xperiment 4 XPRIMNT 4: lectrophilic Aromatic Substitution: A Friedel-Craft Acylation Reaction Relevant sections in the text: Fox & Whitesell, 3 rd d. Chapter 11, especially pg. 524-526,
Chemistry 283g xperiment 4 XPRIMNT 4: lectrophilic Aromatic Substitution: A Friedel-Craft Acylation Reaction Relevant sections in the text: Fox & Whitesell, 3 rd d. Chapter 11, especially pg. 524-526,
Aldehydes and Ketones
 Aldehydes and Ketones Preparation of Aldehydes xidation of Primary Alcohols --- 2 P 1o alcohol ydroboration of a Terminal Alkyne, followed by Tautomerization --- 1. B 3, TF 2. 2 2, K 2 terminal alkyne
Aldehydes and Ketones Preparation of Aldehydes xidation of Primary Alcohols --- 2 P 1o alcohol ydroboration of a Terminal Alkyne, followed by Tautomerization --- 1. B 3, TF 2. 2 2, K 2 terminal alkyne
Rhodamine B pentyl ester and its methyl, ethyl, propyl, and butyl homologues
 Rhodamine B pentyl ester and its methyl, ethyl, propyl, and butyl homologues Author Cosgrove, Kelly Published 2006 Journal Title Molbank Version Published DOI https://doi.org/10.3390/m515 Copyright Statement
Rhodamine B pentyl ester and its methyl, ethyl, propyl, and butyl homologues Author Cosgrove, Kelly Published 2006 Journal Title Molbank Version Published DOI https://doi.org/10.3390/m515 Copyright Statement
Lecture Notes Chem 51C S. King Chapter 24 Carbonyl Condensation Reactions
 Lecture Notes Chem 51C S. King Chapter 24 Carbonyl Condensation Reactions I. Reaction of Enols & Enolates with ther Carbonyls Enols and enolates are electron rich nucleophiles that react with a number
Lecture Notes Chem 51C S. King Chapter 24 Carbonyl Condensation Reactions I. Reaction of Enols & Enolates with ther Carbonyls Enols and enolates are electron rich nucleophiles that react with a number
Asymmetric 1,4-Reductions of Hindered β-substituted Cycloalkenones Using Catalytic CuH
 Asymmetric 1,4-Reductions of Hindered β-substituted Cycloalkenones Using Catalytic CuH Bruce H. Lipshutz,* Jeff M. Servesko, Tue B. Petersen, Patrick P. Papa, Andrew Lover Department of Chemistry & Biochemistry
Asymmetric 1,4-Reductions of Hindered β-substituted Cycloalkenones Using Catalytic CuH Bruce H. Lipshutz,* Jeff M. Servesko, Tue B. Petersen, Patrick P. Papa, Andrew Lover Department of Chemistry & Biochemistry
Table of Contents 1. General procedure for the chiral phosphoric acid catalyzed asymmetric reductive amination using benzothiazoline
 Enantioselective Organocatalytic Reductive Amination of Aliphatic Ketones by Benzothiazoline as Hydrogen Donor Kodai Saito, Takahiko Akiyama* Department of Chemistry, Faculty of Science, Gakushuin University,
Enantioselective Organocatalytic Reductive Amination of Aliphatic Ketones by Benzothiazoline as Hydrogen Donor Kodai Saito, Takahiko Akiyama* Department of Chemistry, Faculty of Science, Gakushuin University,
Answers To Chapter 7 Problems.
 Answers To Chapter Problems.. Most of the Chapter problems appear as end-of-chapter problems in later chapters.. The first reaction is an ene reaction. When light shines on in the presence of light and
Answers To Chapter Problems.. Most of the Chapter problems appear as end-of-chapter problems in later chapters.. The first reaction is an ene reaction. When light shines on in the presence of light and
Name: Student No: Page 1 of 10
 Name: Student No: Page 1 of 10 CEM 2220 rganic Chemistry II: Reactivity and Synthesis FINAL EXAM Winter Session 08R Paper 102 University centre, Room 210 224 Tuesday April 15, 2008 9:00 am 12:00 pm Students
Name: Student No: Page 1 of 10 CEM 2220 rganic Chemistry II: Reactivity and Synthesis FINAL EXAM Winter Session 08R Paper 102 University centre, Room 210 224 Tuesday April 15, 2008 9:00 am 12:00 pm Students
Exam 1 (Monday, July 6, 2015)
 Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
molecules ISSN by MDPI
 Molecules 2007, 12, 1674-1678 Communication molecules ISSN 1420-3049 2007 by MDPI www.mdpi.org/molecules Aqueous (IV)-Catalyzed Diels-Alder Reaction Kyle E. itz Applied Nanoworks, Inc. 9 University Place,
Molecules 2007, 12, 1674-1678 Communication molecules ISSN 1420-3049 2007 by MDPI www.mdpi.org/molecules Aqueous (IV)-Catalyzed Diels-Alder Reaction Kyle E. itz Applied Nanoworks, Inc. 9 University Place,
CHEM 347 Organic Chemistry II (for Majors) Instructor: Paul J. Bracher. Quiz # 4. Due in Monsanto Hall 103 by: Friday, April 4 th, 2014, 7:00 p.m.
 CHEM 347 Quiz # 4 Spring 2014 Page 1 of 9 CHEM 347 Organic Chemistry II (for Majors) Instructor: Paul J. Bracher Quiz # 4 Due in Monsanto Hall 103 by: Friday, April 4 th, 2014, 7:00 p.m. Student Name (Printed)
CHEM 347 Quiz # 4 Spring 2014 Page 1 of 9 CHEM 347 Organic Chemistry II (for Majors) Instructor: Paul J. Bracher Quiz # 4 Due in Monsanto Hall 103 by: Friday, April 4 th, 2014, 7:00 p.m. Student Name (Printed)
Chemistry Organic Chemistry II: Reactivity and Synthesis FINAL EXAM Winter 2003
 Name: Student No: Page 1 of 13 Chemistry 2.222 rganic Chemistry II: Reactivity and Synthesis FINAL EXAM Winter 2003 Paper Number 546 Thursday April 24, 2003 6:00 9:00 pm Frank Kennedy own Gym Students
Name: Student No: Page 1 of 13 Chemistry 2.222 rganic Chemistry II: Reactivity and Synthesis FINAL EXAM Winter 2003 Paper Number 546 Thursday April 24, 2003 6:00 9:00 pm Frank Kennedy own Gym Students
CHAPTER 24 HW: CARBONYL CONDENSATIONS
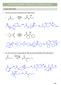 CAPTER 24 W: CARBNYL CNDENSATINS ALDL REACTIN 1. Draw the curved arrow mechanism for each aldol reaction. Na 2 product Na Et prod. 2. Give the curved arrow mechanism for this aldol reaction (and dehydration
CAPTER 24 W: CARBNYL CNDENSATINS ALDL REACTIN 1. Draw the curved arrow mechanism for each aldol reaction. Na 2 product Na Et prod. 2. Give the curved arrow mechanism for this aldol reaction (and dehydration
Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 1 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen
Chapter 15 1 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen
Chemistry 3720 Old Exams. Practice Exams & Keys
 Chemistry 3720 ld Exams Practice Exams & Keys 2015-17 Spring 2017 Page File 3 Spring 2017 Exam 1 10 Spring 2017 Exam 1 Key 16 Spring 2017 Exam 2 23 Spring 2017 Exam 2 Key 29 Spring 2017 Exam 3 36 Spring
Chemistry 3720 ld Exams Practice Exams & Keys 2015-17 Spring 2017 Page File 3 Spring 2017 Exam 1 10 Spring 2017 Exam 1 Key 16 Spring 2017 Exam 2 23 Spring 2017 Exam 2 Key 29 Spring 2017 Exam 3 36 Spring
18.8 Oxidation. Oxidation by silver ion requires an alkaline medium
 18.8 Oxidation Oxidation by silver ion requires an alkaline medium Test for detecting aldehydes Tollens reagent to prevent precipitation of the insoluble silver oxide, a complexing agent is added: ammonia
18.8 Oxidation Oxidation by silver ion requires an alkaline medium Test for detecting aldehydes Tollens reagent to prevent precipitation of the insoluble silver oxide, a complexing agent is added: ammonia
Synthesis and Structure of the Cadmium (II) Complex: [Cd(C 5 H 5 N) 2 (S 2 CO-n-C 4 H 9 ) 2 ]
![Synthesis and Structure of the Cadmium (II) Complex: [Cd(C 5 H 5 N) 2 (S 2 CO-n-C 4 H 9 ) 2 ] Synthesis and Structure of the Cadmium (II) Complex: [Cd(C 5 H 5 N) 2 (S 2 CO-n-C 4 H 9 ) 2 ]](/thumbs/77/75259656.jpg) Molecules 2002, 7, 549-553 molecules ISSN 1420-3049 http://www.mdpi.org Synthesis and Structure of the Cadmium (II) Complex: [Cd(C 5 H 5 N) 2 (S 2 CO-n-C 4 H 9 ) 2 ] Xu Hong Jiang 1,2, Wei Guang Zhang
Molecules 2002, 7, 549-553 molecules ISSN 1420-3049 http://www.mdpi.org Synthesis and Structure of the Cadmium (II) Complex: [Cd(C 5 H 5 N) 2 (S 2 CO-n-C 4 H 9 ) 2 ] Xu Hong Jiang 1,2, Wei Guang Zhang
*Corresponding author. Tel.: , ; fax: ; Materials and Method 2. Preparation of GO nanosheets 3
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Synthesis of 2,3-dihydroquinazolinones and quinazolin-4(3h)-one catalyzed by Graphene Oxide
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Synthesis of 2,3-dihydroquinazolinones and quinazolin-4(3h)-one catalyzed by Graphene Oxide
The Synthesis of Triphenylmethano. will synthesize Triphenylmethanol, a white crystalline aromatic
 HEM 333L rganic hemistry Laboratory Revision 2.0 The Synthesis of Triphenylmethano ol In this laboratory exercise we will synthesize Triphenylmethanol, a white crystalline aromatic compound. Triphenylmethanol
HEM 333L rganic hemistry Laboratory Revision 2.0 The Synthesis of Triphenylmethano ol In this laboratory exercise we will synthesize Triphenylmethanol, a white crystalline aromatic compound. Triphenylmethanol
Advanced Organic Chemistry
 D. A. Evans, G. Lalic Question of the day: Chemistry 530A TBS Me 2 C Me toluene, 130 C 70% TBS C 2 Me H H Advanced rganic Chemistry Me Lecture 16 Cycloaddition Reactions Diels _ Alder Reaction Photochemical
D. A. Evans, G. Lalic Question of the day: Chemistry 530A TBS Me 2 C Me toluene, 130 C 70% TBS C 2 Me H H Advanced rganic Chemistry Me Lecture 16 Cycloaddition Reactions Diels _ Alder Reaction Photochemical
Spring Term 2012 Dr. Williams (309 Zurn, ex 2386)
 Chemistry 242 Organic Chemistry II Spring Term 2012 Dr. Williams (309 Zurn, ex 2386) Web Page: http://math.mercyhurst.edu/~jwilliams/ jwilliams@mercyhurst.edu (or just visit Department web site and look
Chemistry 242 Organic Chemistry II Spring Term 2012 Dr. Williams (309 Zurn, ex 2386) Web Page: http://math.mercyhurst.edu/~jwilliams/ jwilliams@mercyhurst.edu (or just visit Department web site and look
Chem 316/422 Beauchamp 1 Match the step number in the synthesis with the letter of the reagents listed just below.
 hem 316/422 Beauchamp 1 Match the step number in the synthesis with the letter of the reagents listed just below. TP l Et step 8 4 5 eagents used in synthesis A B D E F a DMF (solvent) 1. (i-pr) 2 Li (LDA)/TF
hem 316/422 Beauchamp 1 Match the step number in the synthesis with the letter of the reagents listed just below. TP l Et step 8 4 5 eagents used in synthesis A B D E F a DMF (solvent) 1. (i-pr) 2 Li (LDA)/TF
Multistep Synthesis of 5-isopropyl-1,3-cyclohexanedione
 Multistep Synthesis of 5-isopropyl-1,3-cyclohexanedione The purpose of this experiment was to synthesize 5-isopropyl-1,3-cyclohexanedione from commercially available compounds. To do this, acetone and
Multistep Synthesis of 5-isopropyl-1,3-cyclohexanedione The purpose of this experiment was to synthesize 5-isopropyl-1,3-cyclohexanedione from commercially available compounds. To do this, acetone and
CARBOXYLIC ACIDS AND THEIR DERIVATIVES
 ARBXYLI AIDS AND TEIR DERIVATIVES A STUDENT SULD BE ABLE T: 1. Give the IUPA name given the structure, and draw the structure given the name, of carboxylic acids and their metal salts, acyl chlorides,
ARBXYLI AIDS AND TEIR DERIVATIVES A STUDENT SULD BE ABLE T: 1. Give the IUPA name given the structure, and draw the structure given the name, of carboxylic acids and their metal salts, acyl chlorides,
X-ray single-crystal structure of 2d. 31 P NMR study of the reaction mixture. S11-S14. 1 H and 13 C NMR spectra of all new compounds.
 Supporting Information for Cleavage of a Carbon-carbon Triple Bond via Gold-catalyzed Cascade Cyclization/xidative Cleavage Reactions of (Z)-Enynols with Molecular xygen Yuanhong Liu*, Feijie Song and
Supporting Information for Cleavage of a Carbon-carbon Triple Bond via Gold-catalyzed Cascade Cyclization/xidative Cleavage Reactions of (Z)-Enynols with Molecular xygen Yuanhong Liu*, Feijie Song and
Chapter 19. Organic Chemistry. Carbonyl Compounds III. Reactions at the a-carbon. 4 th Edition Paula Yurkanis Bruice
 Organic Chemistry 4 th Edition Paula Yurkanis Bruice Chapter 19 Carbonyl Compounds III Reactions at the a-carbon Disampaikan oleh: Dr. Sri Handayani 2013 Irene Lee Case Western Reserve University Cleveland,
Organic Chemistry 4 th Edition Paula Yurkanis Bruice Chapter 19 Carbonyl Compounds III Reactions at the a-carbon Disampaikan oleh: Dr. Sri Handayani 2013 Irene Lee Case Western Reserve University Cleveland,
Name: Student Number:
 Page 1 of 5 Name: Student Number: l University of Manitoba - Department of Chemistry CEM 2220 - Introductory rganic Chemistry II - Term Test 1 Thursday, February 14, 2008 This is a 2-hour test, marked
Page 1 of 5 Name: Student Number: l University of Manitoba - Department of Chemistry CEM 2220 - Introductory rganic Chemistry II - Term Test 1 Thursday, February 14, 2008 This is a 2-hour test, marked
EXPERIMENTS 5-8: BACKGROUND OXIDATION REDUCTION REACTIONS IN ORGANIC CHEMISTRY: THE INTERCONVERSION OF
 EXPERIMENTS 5-8: BACKGRUND XIDATIN REDUCTIN REACTINS IN RGANIC CEMISTRY: TE INTERCNVERSIN F 4-tert-BUTYLCYCLEXANL AND 4-tert-BUTYLCYCLEXANNE 1 UTLINE AND GALS F TE PRJECT: This is a 4 week project that
EXPERIMENTS 5-8: BACKGRUND XIDATIN REDUCTIN REACTINS IN RGANIC CEMISTRY: TE INTERCNVERSIN F 4-tert-BUTYLCYCLEXANL AND 4-tert-BUTYLCYCLEXANNE 1 UTLINE AND GALS F TE PRJECT: This is a 4 week project that
Chem 342 Organic Chemistry II Final Exam 13 May 2009
 hem 342 rganic hemistry II Final Exam 13 May 2009 KEY Please read through each question carefully and answer in the spaces provided. A good strategy is to go through the test and answer all the questions
hem 342 rganic hemistry II Final Exam 13 May 2009 KEY Please read through each question carefully and answer in the spaces provided. A good strategy is to go through the test and answer all the questions
Look for absorption bands in decreasing order of importance:
 1. Match the following to their IR spectra (30 points) Look for absorption bands in decreasing order of importance: a e a 2941 1716 d f b 3333 c b 1466 1.the - absorption(s) between 3100 and 2850 cm-1.
1. Match the following to their IR spectra (30 points) Look for absorption bands in decreasing order of importance: a e a 2941 1716 d f b 3333 c b 1466 1.the - absorption(s) between 3100 and 2850 cm-1.
ANSWER KEY Page 1 of 7
 ANSWER KEY Page 1 of 7 University of Manitoba Department of Chemistry 2.222 Introductory rganic Chemistry II Term Test 1 Thursday February 5, 2003 This is a 2-hour test, marked out of 50 points total.
ANSWER KEY Page 1 of 7 University of Manitoba Department of Chemistry 2.222 Introductory rganic Chemistry II Term Test 1 Thursday February 5, 2003 This is a 2-hour test, marked out of 50 points total.
ALDEHYDES AND KETONES
 ALDEHYDES AND KETONES IN WEEK 1, A STUDENT SHOULD BE ABLE TO: 1. Give the IUPAC name given the structure, and draw the structure given the name, of aldehydes and ketones. Also, draw the structure given
ALDEHYDES AND KETONES IN WEEK 1, A STUDENT SHOULD BE ABLE TO: 1. Give the IUPAC name given the structure, and draw the structure given the name, of aldehydes and ketones. Also, draw the structure given
CHEM 234: Organic Chemistry II Reaction Sheets
 EM234:rganichemistry eactionsheets ucleophilic addition at carbonyl groups: Grignards and reducing agents u: u u u: u u = or = or l u u u ucleophilic addition at carbonyl groups: oxygen and nitrogen nucleophiles:
EM234:rganichemistry eactionsheets ucleophilic addition at carbonyl groups: Grignards and reducing agents u: u u u: u u = or = or l u u u ucleophilic addition at carbonyl groups: oxygen and nitrogen nucleophiles:
Supporting Information
 Supporting Information Lewis acid-catalyzed intramolecular condensation of ynol ether-acetals. Synthesis of alkoxycycloalkene carboxylates Vincent Tran and Thomas G. Minehan * Department of Chemistry and
Supporting Information Lewis acid-catalyzed intramolecular condensation of ynol ether-acetals. Synthesis of alkoxycycloalkene carboxylates Vincent Tran and Thomas G. Minehan * Department of Chemistry and
Chemistry Organic Chemistry II: Reactivity and Synthesis ANSWERS FOR FINAL EXAM Winter 2004
 ASWER KEY Page 1 of 18 Chemistry 2.222 rganic Chemistry II: Reactivity and Synthesis ASWERS FR FIAL EXAM Winter 2004 Paper umber 631 Thursday April 22, 2004 1:30 4:30 pm University Centre Rm 210-224 Students
ASWER KEY Page 1 of 18 Chemistry 2.222 rganic Chemistry II: Reactivity and Synthesis ASWERS FR FIAL EXAM Winter 2004 Paper umber 631 Thursday April 22, 2004 1:30 4:30 pm University Centre Rm 210-224 Students
JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II. 5 Credit Hours. Prepared by: Richard A. Pierce
 JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
A New Model for Asymmetric Amplification in Amino Acid Catalysis - Supporting information
 A New Model for Asymmetric Amplification in Amino Acid Catalysis - Supporting information Martin Klussmann, Hiroshi Iwamura, Suju P. Mathew, David H. Wells, Urvish Pandya, Alan Armstrong and Donna G. Blackmond
A New Model for Asymmetric Amplification in Amino Acid Catalysis - Supporting information Martin Klussmann, Hiroshi Iwamura, Suju P. Mathew, David H. Wells, Urvish Pandya, Alan Armstrong and Donna G. Blackmond
Supporting Information
 Supporting Information Rhodium-Catalyzed Annulation Reactions of 2-Cyanophenylboronic Acid with Alkynes and Strained Alkenes Tomoya Miura and Masahiro Murakami* Department of Synthetic Chemistry and Biological
Supporting Information Rhodium-Catalyzed Annulation Reactions of 2-Cyanophenylboronic Acid with Alkynes and Strained Alkenes Tomoya Miura and Masahiro Murakami* Department of Synthetic Chemistry and Biological
Aromatic Compounds II
 2302272 Org Chem II Part I Lecture 2 Aromatic Compounds II Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 17 in Organic Chemistry, 8 th Edition, L.
2302272 Org Chem II Part I Lecture 2 Aromatic Compounds II Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 17 in Organic Chemistry, 8 th Edition, L.
Chemistry 2050 Introduction to Organic Chemistry Fall Semester 2011 Dr. Rainer Glaser
 Chemistry 2050 Introduction to Organic Chemistry Fall Semester 2011 Dr. Rainer Glaser Examination #3 Alcohols, Ethers, Aldehydes & Ketones. Wednesday, October 26, 2011, 10 10:50 am Name: Question 1. Names,
Chemistry 2050 Introduction to Organic Chemistry Fall Semester 2011 Dr. Rainer Glaser Examination #3 Alcohols, Ethers, Aldehydes & Ketones. Wednesday, October 26, 2011, 10 10:50 am Name: Question 1. Names,
