molecules ISSN
|
|
|
- Joanna McDaniel
- 5 years ago
- Views:
Transcription
1 Molecules 2000, 5, molecules ISSN Unexpected Thorpe Reaction of an α-alkoxynitrile Lajos Kovács Department of Medicinal Chemistry, University of Szeged, Dóm tér 8., H-6720 Szeged, Hungary Tel.: , Fax: , URL: Received: 19 January 2000 / Accepted: 14 February 2000 / Published: 17 February 2000 Abstract: α-alkoxynitrile 1 in the presence of tris(methylthio)methyllithium 2 at 78ºC gave the dimer 5 instead of the expected C 1 -elongated product 3. The formation of compound 5 is explained in terms of anion formation and self-condensation, a variant of the Thorpe reaction. Scrutinizing the 1 H NMR spectra revealed that the enamine tautomer 5b is predominant over the imine 5a in the solvents investigated. Keywords: Thorpe reaction, self-condensation. Introduction Nitrile 1 (Scheme 1) was obtained en route to acyclic analogues of the C-nucleoside tiazofurin [1]. This compound was deemed to be a good starting material for the synthesis of α-keto ester 4 via intermediate 3. This assumption was based on analogy with the Grignard reaction of nitriles giving rise to ketones [2] or the Blaise reaction (Reformatsky reaction of nitriles affording β-keto esters) [3]. For the required carboxylate anion synthon different tris(alkylthio or arylthio)methyllithiums, derived from tris(alkyl or arylthio)methanes [4], were used successfully in the C 1 elongation of ketones [5] and lactones [6]. Most often [tris(methylthio)methyl]lithium (2) was applied which is stable up to 40ºC [7]. The obtained trithioorthoesters can be transformed into the corresponding esters using the common mercury(ii) salt-assisted demercaptalisation procedure. In principle, alkyl chloroformates could also be employed as C 1 synthons. However, in the single reported Reformatsky-type reaction of ethyl chloroformate with ethyl acetoacetate in the presence of a zinc-copper couple leading to diethyl 2000 by MDPI ( Reproduction is permitted for noncommercial purposes.
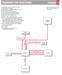
2 Molecules 2000, hydroxy-2-methylsuccinate, the yield was rather low (20%) [8]. In a recent application, ethyl chloroformate was allowed to react with methyl phenylglyoxylate in a titanium(iv)-promoted reaction to afford ethyl methyl 2-hydroxy-2-phenylmalonate in 60% yield [9]. Results and Discussion Nitrile 1 was allowed to react with compound 2 at 78ºC for 4 h. Contrary to our expectations, compound 3 was not obtained. The isolated substance exhibited surprisingly complicated 1 H NMR spectra in different solvents, according to the microanalysis it contained no sulfur and its elemental composition was identical with that of nitrile 1. Its low-voltage (15 ev) EI mass spectrum showed a molecular ion at m/z 622 (the molecular mass of the starting nitrile is 311) and a fragmentation pattern compatible only with the dimeric structures 5a and 5b. Scrutiny of the 1 H NMR spectrum revealed that the substance exists as an enamine (5b) rather than an imine (5a). The IR spectrum also confirmed the presence of a nitrile group (ν 2180 cm -1 ) (CH 3 S) 3 C: Li (2) THF, 4 h, -78 C 2. aq. work-up 40 % NH C(SCH 3 ) 3 Bn 3 Bn Hg 2+ 5a RH CR NH 2 Bn Bn 4 5b Scheme 1.
3 Molecules 2000, The literature search for precedents (Beilstein s CrossFire 4.0, release BS9902PRPR using Beilstein Commander 4.0) showed that α-alkoxynitriles react normally with Grignard reagents [10] while organolithium [11] and organosodium compounds [12,13] provoke self-condensation. This can be explained by the strong basicity of organolithiums and the result is, just as in the above instance, the special case of the well-known Thorpe reaction [14] (Scheme 2). R R'Li -R'H Li C N R R R Li N R aq. work-up R R R R NH 2 NH Scheme 2. It is noteworthy that even acetonitrile tends to react abnormally with Grignard reagents, e.g. with phenylmagnesium bromide, only 42% of the expected acetophenone has been obtained along with 32% of benzene [15]. Nitriles lacking α-hydrogens react normally and afford ketones. Experimental General To tris(methylthio)methane [4] (0.927 g, 6.0 mmol) dissolved in anhydrous THF (2.0 ml) was added butyllithium (1.26 M solution, 5.20 ml, 6.60 ml) at -78ºC under nitrogen. Nitrile 1 [1] (1.40 g, 4.5 mmol) dissolved in anhydrous THF (10.0 ml) was added dropwise to this solution after 20 min and kept at 78ºC for 4 h. The reaction mixture was quenched at 60ºC with satd. NH 4 Cl solution. After allowing to warm up to room temperature, the mixture was diluted and extracted with chloroform (25 ml). The aqueous phase was extracted with chloroform (2 50 ml). The combined organic phases were dried (MgS 4 ) and evaporated in vacuo. Chromatographic purification using 20% (v/v) ethyl acetate in hexanes afforded 3-amino-2,4-bis{2-(benzyloxy)-1-[(benzyloxy)methyl]-ethoxy}-2- butene-nitrile (5, 0.56 g, 40%) as a yellow oil.
4 Molecules 2000, Spectral Data IR (KBr, ν, cm -1 ): 3450m, 3330m, 3020m, 2910s, 2860s, 2180m, 1680m, 1493m, 1450m, 1090m. 1 H NMR (CDCl 3, 200 MHz, δ, ppm): 3.55 (d, J 6 Hz, 4 H, 2 x CH 2 ); 3.65 (d, J 6 Hz, 4 H, 2 x CH 2 ); 3.71 (m, 1 H, CH); 3.94 (m, 1 H, CH); 4.37 (s, 2 H, 4-CH 2 ); 4.49 (s, 4 H, 2 x PhCH 2 ); 4.52 (s, 4 H, 2 x PhCH 2 ); 5.45 (br s, exchangeable with D 2, 2 H, NH 2 ); 7.30 (m, 20 H, 4 x C 6 H 5 ). 1 H NMR (DMS-d 6, 200 MHz, δ, ppm): 3.52 (d, J 5 Hz, 4 H, 2 x CH 2 ); 3.65 (d, J 5 Hz, 4 H, 2 x CH 2 ); 3.72 (m, 1 H, CH); 3.88 (m, 1 H, H); 4.20 (s, 2 H, 4-CH 2 ); 4.45 (s, 8 H, 4 x PhCH 2 ); 6.00 (br s, exchangeable with D 2, 2 H, NH 2 ); 7.30 (m, 20 H, 4 x C 6 H 5 ). EI-MS (15 ev, m/z, %): 622 (5, M + ); 531 (1, M-91); 418 (4); 387 (4); 367 [4, M-CH(CH 2 Bn) 2 ]; 331 (6); 292 (9); 278 (24); 220 (14); 181 (75); 102 (88); 91 (100, C 7 H 7 + ). Anal. calcd. for C 38 H 42 N 2 6 ( ): C, 73.29; H, 6.80; N, 4.50; found: C, 73.49; H, 6.71; N, References and Notes 1. Kovács, L.; Herczegh, P.; Batta, G.; Farkas, I. Two acyclic analogues of 2-β-D-ribofuranosylthiazole-4-carboxamide (tiazofurin). Heterocycles 1987, 26, March, J. Advanced rganic Chemistry, 4th ed.; John Wiley and Sons: New York, 1992; pp Gaudemar, M. The Reformatsky reaction in the course of the last thirty years. rganometal. Chem. Rev. A 1972, 8, Holmberg, B. n the esters of orthotrithioformic acid. Ber. Dtsch. Chem. Ges. 1907, 40, Dailey,. D.; Fuchs, P. L. Synthesis of a model for the BCE ring system of bruceantin. A caveat on the cyclohexene trans diaxial diol conversion. J. rg. Chem. 1980, 45, Hengeveld, J. E.; Grief, V.; Tadanier, J.; Lee, C.-M.; Riley, D.; Lartey, P. A. A general synthesis of methyl aldulosonates using tris(methylthio)methyl lithium as the ester anion equivalent. Tetrahedron Lett. 1984, 25, Damon, R. E.; Schlessinger, R. H. An efficient and stereospecific total synthesis of DL-protolichesterinic acid. Tetrahedron Lett. 1976, Antia, M. B.; Kaushal, R. Reformatsky condensation of ketonic esters with halo esters. Agra Univ. J. Res., Sci. 1954, 3, (Chem. Abstr.1955, 49, 6121h) 9. Clerici, A.; Clerici, L.; Porta,. Acylation of a transient Ti(IV)-enolate by acyl halides and anhydrides. Facile synthesis of α-hydroxy-β-ketones. Tetrahedron 1996, 52, Lingo, S. P.; Henze, H. R. Substituted ethers derived from ethylene chlorohydrin. J. Am. Chem. Soc. 1939, 61, Gauthier, R.; Axiotis, G. P.; Chastrette, M. Double addition of organometallic compounds to β-
5 Molecules 2000, oxygenated nitriles R. Preparation of primary amines of RCR 1 R 2 NH 2 type. J. rganomet. Chem. 1977, 140, Allen, C. F. H.; van Allan, J. A. Some macrocyclic oxalactones and related substances. J. rg. Chem. 1949, 14, Guibert, S.; Cariou, M.; Simonet, J. Research on novel organic solvents with potential use in lithium batteries 2. The properties of certain aliphatic nitriles substituted by donor groups. Bull. Soc. Chim. Fr. 1988, March, J. Advanced rganic Chemistry, 4th ed.; John Wiley and Sons: New York, 1992; pp Jones, R. L.; Pearson, D. E.; Gordon, M. Studies on the reaction of phenylmagnesium bromides with acetonitrile. J. rg. Chem. 1972, 37, Samples Availability: not available by MDPI ( Reproduction is permitted for noncommercial purposes.
molecules ISSN
 Molecules 001, 6, 44-447 molecules ISSN 140-3049 http://www.mdpi.org Facile and Fast Pinacol Rearrangement by All 3 in the Solid State Parviz Rashidi-Ranjbar* and Ebrahim Kianmehr Department of hemistry
Molecules 001, 6, 44-447 molecules ISSN 140-3049 http://www.mdpi.org Facile and Fast Pinacol Rearrangement by All 3 in the Solid State Parviz Rashidi-Ranjbar* and Ebrahim Kianmehr Department of hemistry
Supporting Information
 Supporting Information Efficient Short Step Synthesis of Corey s Tamiflu Intermediate Nsiama Tienabe Kipassa, Hiroaki kamura, * Kengo Kina, Tetsuo Iwagawa, and Toshiyuki Hamada Department of Chemistry
Supporting Information Efficient Short Step Synthesis of Corey s Tamiflu Intermediate Nsiama Tienabe Kipassa, Hiroaki kamura, * Kengo Kina, Tetsuo Iwagawa, and Toshiyuki Hamada Department of Chemistry
Supplementary Information
 Supplementary Information C aryl -C alkyl bond formation from Cu(ClO 4 ) 2 -mediated oxidative cross coupling reaction between arenes and alkyllithium reagents through structurally well-defined Ar-Cu(III)
Supplementary Information C aryl -C alkyl bond formation from Cu(ClO 4 ) 2 -mediated oxidative cross coupling reaction between arenes and alkyllithium reagents through structurally well-defined Ar-Cu(III)
Supplementary Material for: Unexpected Decarbonylation during an Acid- Mediated Cyclization to Access the Carbocyclic Core of Zoanthenol.
 Tetrahedron Letters 1 Pergamon TETRAHEDRN LETTERS Supplementary Material for: Unexpected Decarbonylation during an Acid- Mediated Cyclization to Access the Carbocyclic Core of Zoanthenol. Jennifer L. Stockdill,
Tetrahedron Letters 1 Pergamon TETRAHEDRN LETTERS Supplementary Material for: Unexpected Decarbonylation during an Acid- Mediated Cyclization to Access the Carbocyclic Core of Zoanthenol. Jennifer L. Stockdill,
N_HW1 N_HW1. 1. What is the purpose of the H 2 O in this sequence?
 N_HW1 N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. 1. What is the purpose of the H 2 O in this
N_HW1 N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. 1. What is the purpose of the H 2 O in this
CH 320/328 N Summer II 2018
 CH 320/328 N Summer II 2018 HW 1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. (5 pts each) 1. Which
CH 320/328 N Summer II 2018 HW 1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. (5 pts each) 1. Which
Synthetic Studies on Norissolide; Enantioselective Synthesis of the Norrisane Side Chain
 rganic Lett. (Supporting Information) 1 Synthetic Studies on Norissolide; Enantioselective Synthesis of the Norrisane Side Chain Charles Kim, Richard Hoang and Emmanuel A. Theodorakis* Department of Chemistry
rganic Lett. (Supporting Information) 1 Synthetic Studies on Norissolide; Enantioselective Synthesis of the Norrisane Side Chain Charles Kim, Richard Hoang and Emmanuel A. Theodorakis* Department of Chemistry
Supporting Information for Synthesis of C(3) Benzofuran Derived Bis-Aryl Quaternary Centers: Approaches to Diazonamide A
 Fuerst et al. Synthesis of C(3) Benzofuran Derived Bis-Aryl Quaternary Centers: Approaches to Diazonamide A S1 Supporting Information for Synthesis of C(3) Benzofuran Derived Bis-Aryl Quaternary Centers:
Fuerst et al. Synthesis of C(3) Benzofuran Derived Bis-Aryl Quaternary Centers: Approaches to Diazonamide A S1 Supporting Information for Synthesis of C(3) Benzofuran Derived Bis-Aryl Quaternary Centers:
Department of Chemistry and Biochemistry, California State University Northridge, Northridge, CA Experimental Procedures
 Supporting Information Low Temperature n-butyllithium-induced [3,3]-Sigmatropic Rearrangement/Electrophile Trapping Reactions of Allyl-1,1- Dichlorovinyl Ethers. Synthesis of - - and -lactones. Aaron Christopher
Supporting Information Low Temperature n-butyllithium-induced [3,3]-Sigmatropic Rearrangement/Electrophile Trapping Reactions of Allyl-1,1- Dichlorovinyl Ethers. Synthesis of - - and -lactones. Aaron Christopher
18.8 Oxidation. Oxidation by silver ion requires an alkaline medium
 18.8 Oxidation Oxidation by silver ion requires an alkaline medium Test for detecting aldehydes Tollens reagent to prevent precipitation of the insoluble silver oxide, a complexing agent is added: ammonia
18.8 Oxidation Oxidation by silver ion requires an alkaline medium Test for detecting aldehydes Tollens reagent to prevent precipitation of the insoluble silver oxide, a complexing agent is added: ammonia
Synthesis of fluorophosphonylated acyclic nucleotide analogues via Copper (I)- catalyzed Huisgen 1-3 dipolar cycloaddition
 Synthesis of fluorophosphonylated acyclic nucleotide analogues via Copper (I)- catalyzed Huisgen 1-3 dipolar cycloaddition Sonia Amel Diab, Antje Hienzch, Cyril Lebargy, Stéphante Guillarme, Emmanuel fund
Synthesis of fluorophosphonylated acyclic nucleotide analogues via Copper (I)- catalyzed Huisgen 1-3 dipolar cycloaddition Sonia Amel Diab, Antje Hienzch, Cyril Lebargy, Stéphante Guillarme, Emmanuel fund
Accessory Information
 Accessory Information Synthesis of 5-phenyl 2-Functionalized Pyrroles by amino Heck and tandem amino Heck Carbonylation reactions Shazia Zaman, *A,B Mitsuru Kitamura B, C and Andrew D. Abell A *A Department
Accessory Information Synthesis of 5-phenyl 2-Functionalized Pyrroles by amino Heck and tandem amino Heck Carbonylation reactions Shazia Zaman, *A,B Mitsuru Kitamura B, C and Andrew D. Abell A *A Department
Regioselective Ethanolamination and Ketalization of 3-Ph-2,4- diacetyl(diethoxycarbonyl)-5-hydroxy-5-methylcyclohexanones
 Molecules 003, 8, 51-55 molecules ISSN 140-3049 http://www.mdpi.org Regioselective Ethanolamination and Ketalization of 3--,4- diacetyl(diethoxycarbonyl)-5-hydroxy-5-methylcyclohexanones Adele P. Kriven'ko,
Molecules 003, 8, 51-55 molecules ISSN 140-3049 http://www.mdpi.org Regioselective Ethanolamination and Ketalization of 3--,4- diacetyl(diethoxycarbonyl)-5-hydroxy-5-methylcyclohexanones Adele P. Kriven'ko,
SUPPORTING INFORMATION
 SUPPRTING INFRMATIN A Direct, ne-step Synthesis of Condensed Heterocycles: A Palladium-Catalyzed Coupling Approach Farnaz Jafarpour and Mark Lautens* Davenport Chemical Research Laboratories, Chemistry
SUPPRTING INFRMATIN A Direct, ne-step Synthesis of Condensed Heterocycles: A Palladium-Catalyzed Coupling Approach Farnaz Jafarpour and Mark Lautens* Davenport Chemical Research Laboratories, Chemistry
Unexpected dimerization reaction of 5-methyl-6,7-methylendioxy-1- tetralone in the presence of TMSCl-ethylene glycol
 Issue in Honor of Prof. Joan Bosch ARKIVC 2007 (iv) 82-87 Unexpected dimerization reaction of 5-methyl-6,7-methylendioxy-- tetralone in the presence of TMSCl-ethylene glycol Gema Esteban and Joaquín Plumet
Issue in Honor of Prof. Joan Bosch ARKIVC 2007 (iv) 82-87 Unexpected dimerization reaction of 5-methyl-6,7-methylendioxy-- tetralone in the presence of TMSCl-ethylene glycol Gema Esteban and Joaquín Plumet
The First Asymmetric Total Syntheses and. Determination of Absolute Configurations of. Xestodecalactones B and C
 Supporting Information The First Asymmetric Total Syntheses and Determination of Absolute Configurations of Xestodecalactones B and C Qiren Liang, Jiyong Zhang, Weiguo Quan, Yongquan Sun, Xuegong She*,,
Supporting Information The First Asymmetric Total Syntheses and Determination of Absolute Configurations of Xestodecalactones B and C Qiren Liang, Jiyong Zhang, Weiguo Quan, Yongquan Sun, Xuegong She*,,
Supplementary Note 1 : Chemical synthesis of (E/Z)-4,8-dimethylnona-2,7-dien-4-ol (4)
 Supplementary Note 1 : Chemical synthesis of (E/Z)-4,8-dimethylnona-2,7-dien-4-ol (4) A solution of propenyl magnesium bromide in THF (17.5 mmol) under nitrogen atmosphere was cooled in an ice bath and
Supplementary Note 1 : Chemical synthesis of (E/Z)-4,8-dimethylnona-2,7-dien-4-ol (4) A solution of propenyl magnesium bromide in THF (17.5 mmol) under nitrogen atmosphere was cooled in an ice bath and
SUPPORTING INFORMATION
 SUPPORTING INFORMATION Synthesis of Functionalized Thia Analogues of Phlorins and Covalently Linked Phlorin-Porphyrin Dyads Iti Gupta a, Roland Fröhlich b and Mangalampalli Ravikanth *a a Department of
SUPPORTING INFORMATION Synthesis of Functionalized Thia Analogues of Phlorins and Covalently Linked Phlorin-Porphyrin Dyads Iti Gupta a, Roland Fröhlich b and Mangalampalli Ravikanth *a a Department of
Supporting Information
 Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Concise Stereoselective Synthesis of ( )-Podophyllotoxin by Intermolecular Fe III -catalyzed Friedel-Crafts Alkylation Daniel Stadler, Thorsten
Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Concise Stereoselective Synthesis of ( )-Podophyllotoxin by Intermolecular Fe III -catalyzed Friedel-Crafts Alkylation Daniel Stadler, Thorsten
molecules ISSN
 Molecules 2003, 8, 467-471 Second Eurasian Meeting on eterocyclic Chemistry eterocycles in rganic and Combinatorial Chemistry molecules ISS 1420-3049 http://www.mdpi.org Solid-Phase Synthesis of Methyl
Molecules 2003, 8, 467-471 Second Eurasian Meeting on eterocyclic Chemistry eterocycles in rganic and Combinatorial Chemistry molecules ISS 1420-3049 http://www.mdpi.org Solid-Phase Synthesis of Methyl
Ring-Opening / Fragmentation of Dihydropyrones for the Synthesis of Homopropargyl Alcohols
 Ring-pening / Fragmentation of Dihydropyrones for the Synthesis of Homopropargyl Alcohols Jumreang Tummatorn, and Gregory B. Dudley, * Department of Chemistry and Biochemistry, Florida State University,
Ring-pening / Fragmentation of Dihydropyrones for the Synthesis of Homopropargyl Alcohols Jumreang Tummatorn, and Gregory B. Dudley, * Department of Chemistry and Biochemistry, Florida State University,
1. What is the major organic product obtained from the following sequence of reactions?
 CH320 N N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. Carefully record your answers on the Scantron
CH320 N N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. Carefully record your answers on the Scantron
hydroxyanthraquinones related to proisocrinins
 Supporting Information for Regiodefined synthesis of brominated hydroxyanthraquinones related to proisocrinins Joyeeta Roy, Tanushree Mal, Supriti Jana and Dipakranjan Mal* Address: Department of Chemistry,
Supporting Information for Regiodefined synthesis of brominated hydroxyanthraquinones related to proisocrinins Joyeeta Roy, Tanushree Mal, Supriti Jana and Dipakranjan Mal* Address: Department of Chemistry,
Photooxidations of 2-(γ,ε-dihydroxyalkyl) furans in Water: Synthesis of DE-Bicycles of the Pectenotoxins
 S1 Photooxidations of 2-(γ,ε-dihydroxyalkyl) furans in Water: Synthesis of DE-Bicycles of the Pectenotoxins Antonia Kouridaki, Tamsyn Montagnon, Maria Tofi and Georgios Vassilikogiannakis* Department of
S1 Photooxidations of 2-(γ,ε-dihydroxyalkyl) furans in Water: Synthesis of DE-Bicycles of the Pectenotoxins Antonia Kouridaki, Tamsyn Montagnon, Maria Tofi and Georgios Vassilikogiannakis* Department of
Efficient Mono- and Bis-Functionalization of 3,6-Dichloropyridazine using (tmp) 2 Zn 2MgCl 2 2LiCl ** Stefan H. Wunderlich and Paul Knochel*
 Efficient Mono- and Bis-Functionalization of 3,6-Dichloropyridazine using (tmp) 2 Zn 2Mg 2 2Li ** Stefan H. Wunderlich and Paul Knochel* Ludwig Maximilians-Universität München, Department Chemie & Biochemie
Efficient Mono- and Bis-Functionalization of 3,6-Dichloropyridazine using (tmp) 2 Zn 2Mg 2 2Li ** Stefan H. Wunderlich and Paul Knochel* Ludwig Maximilians-Universität München, Department Chemie & Biochemie
SUPPLEMENTARY INFORMATION
 Synthetic chemistry ML5 and ML4 were identified as K P.(TREK-) activators using a combination of fluorescence-based thallium flux and automated patch-clamp assays. ML5, ML4, and ML5a were synthesized using
Synthetic chemistry ML5 and ML4 were identified as K P.(TREK-) activators using a combination of fluorescence-based thallium flux and automated patch-clamp assays. ML5, ML4, and ML5a were synthesized using
Multistep Synthesis of 5-isopropyl-1,3-cyclohexanedione
 Multistep Synthesis of 5-isopropyl-1,3-cyclohexanedione The purpose of this experiment was to synthesize 5-isopropyl-1,3-cyclohexanedione from commercially available compounds. To do this, acetone and
Multistep Synthesis of 5-isopropyl-1,3-cyclohexanedione The purpose of this experiment was to synthesize 5-isopropyl-1,3-cyclohexanedione from commercially available compounds. To do this, acetone and
Organolithium Compounds *
 OpenStax-CNX module: m32444 1 Organolithium Compounds * Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 One of the major uses of lithium
OpenStax-CNX module: m32444 1 Organolithium Compounds * Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 One of the major uses of lithium
Formal Total Synthesis of Optically Active Ingenol via Ring-Closing Olefin Metathesis
 Formal Total Synthesis of Optically Active Ingenol via Ring-Closing Olefin Metathesis Kazushi Watanabe, Yuto Suzuki, Kenta Aoki, Akira Sakakura, Kiyotake Suenaga, and Hideo Kigoshi* Department of Chemistry,
Formal Total Synthesis of Optically Active Ingenol via Ring-Closing Olefin Metathesis Kazushi Watanabe, Yuto Suzuki, Kenta Aoki, Akira Sakakura, Kiyotake Suenaga, and Hideo Kigoshi* Department of Chemistry,
Synthesis of Trifluoromethylated Naphthoquinones via Copper-Catalyzed. Cascade Trifluoromethylation/Cyclization of. 2-(3-Arylpropioloyl)benzaldehydes
 Supporting Information to Synthesis of Trifluoromethylated Naphthoquinones via Copper-Catalyzed Cascade Trifluoromethylation/Cyclization of 2-(3-Arylpropioloyl)benzaldehydes Yan Zhang*, Dongmei Guo, Shangyi
Supporting Information to Synthesis of Trifluoromethylated Naphthoquinones via Copper-Catalyzed Cascade Trifluoromethylation/Cyclization of 2-(3-Arylpropioloyl)benzaldehydes Yan Zhang*, Dongmei Guo, Shangyi
Supporting Information
 Supporting Information Total Synthesis of (±)-Grandilodine B Chunyu Wang, Zhonglei Wang, Xiaoni Xie, Xiaotong Yao, Guang Li, and Liansuo Zu* School of Pharmaceutical Sciences, Tsinghua University, Beijing,
Supporting Information Total Synthesis of (±)-Grandilodine B Chunyu Wang, Zhonglei Wang, Xiaoni Xie, Xiaotong Yao, Guang Li, and Liansuo Zu* School of Pharmaceutical Sciences, Tsinghua University, Beijing,
Supporting Information for:
 Supporting Information for: Photoenolization of 2-(2-Methyl Benzoyl) Benzoic Acid, Methyl Ester: The Effect of The Lifetime of the E Photoenol on the Photochemistry Armands Konosonoks, P. John Wright,
Supporting Information for: Photoenolization of 2-(2-Methyl Benzoyl) Benzoic Acid, Methyl Ester: The Effect of The Lifetime of the E Photoenol on the Photochemistry Armands Konosonoks, P. John Wright,
Supporting Information. Sandmeyer Cyanation of Arenediazonium Tetrafluoroborate Using Acetonitrile as Cyanide Source
 Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2015 Supporting Information Sandmeyer Cyanation of Arenediazonium Tetrafluoroborate Using
Electronic Supplementary Material (ESI) for Organic Chemistry Frontiers. This journal is the Partner Organisations 2015 Supporting Information Sandmeyer Cyanation of Arenediazonium Tetrafluoroborate Using
Atovaquone: An Antipneumocystic Agent
 Atovaquone: An Antipneumocystic Agent Atovaquone is a pharmaceutical compound marketed in the United States under different combinations to prevent and treat pneumocystosis and malaria. In a report from
Atovaquone: An Antipneumocystic Agent Atovaquone is a pharmaceutical compound marketed in the United States under different combinations to prevent and treat pneumocystosis and malaria. In a report from
1/4/2011. Chapter 18 Aldehydes and Ketones Reaction at the -carbon of carbonyl compounds
 Chapter 18 Aldehydes and Ketones Reaction at the -carbon of carbonyl compounds The Acidity of the Hydrogens of Carbonyl Compounds: Enolate Anions Hydrogens on carbons to carbonyls are unusually acidic
Chapter 18 Aldehydes and Ketones Reaction at the -carbon of carbonyl compounds The Acidity of the Hydrogens of Carbonyl Compounds: Enolate Anions Hydrogens on carbons to carbonyls are unusually acidic
Supporting Information
 Supporting Information Macrocyclic Diorganotin Complexes of -Amino Acid Dithiocarbamates as Hosts for Ion-Pair Recognition Jorge Cruz-Huerta, a Manuel Carillo-Morales, a Ericka Santacruz-Juárez, a Irán
Supporting Information Macrocyclic Diorganotin Complexes of -Amino Acid Dithiocarbamates as Hosts for Ion-Pair Recognition Jorge Cruz-Huerta, a Manuel Carillo-Morales, a Ericka Santacruz-Juárez, a Irán
Supporting Information
 Electronic upplementary Material (EI) for rganic Chemistry rontiers. This journal is the Partner rganisations 0 upporting Information Convenient ynthesis of Pentafluoroethyl Thioethers via Catalytic andmeyer
Electronic upplementary Material (EI) for rganic Chemistry rontiers. This journal is the Partner rganisations 0 upporting Information Convenient ynthesis of Pentafluoroethyl Thioethers via Catalytic andmeyer
Supporting Information
 1 Supporting Information Gold-catalyzed Synthesis of 3-arylindoles via Annulation of Nitrosoarenes and Alkynes Siva Murru, August A. Gallo and Radhey S. Srivastava* Department of Chemistry, University
1 Supporting Information Gold-catalyzed Synthesis of 3-arylindoles via Annulation of Nitrosoarenes and Alkynes Siva Murru, August A. Gallo and Radhey S. Srivastava* Department of Chemistry, University
Highly stereocontrolled synthesis of trans-enediynes via
 Supporting Information for Highly stereocontrolled synthesis of trans-enediynes via carbocupration of fluoroalkylated diynes Tsutomu Konno*, Misato Kishi, and Takashi Ishihara Address: Department of Chemistry
Supporting Information for Highly stereocontrolled synthesis of trans-enediynes via carbocupration of fluoroalkylated diynes Tsutomu Konno*, Misato Kishi, and Takashi Ishihara Address: Department of Chemistry
Supporting Information: Regioselective esterification of vicinal diols on monosaccharide derivatives via
 Supporting Information: Regioselective esterification of vicinal diols on monosaccharide derivatives via Mitsunobu reactions. Guijun Wang,*Jean Rene Ella-Menye, Michael St. Martin, Hao Yang, Kristopher
Supporting Information: Regioselective esterification of vicinal diols on monosaccharide derivatives via Mitsunobu reactions. Guijun Wang,*Jean Rene Ella-Menye, Michael St. Martin, Hao Yang, Kristopher
Supporting Information. for. Angew. Chem. Int. Ed. Z Wiley-VCH 2003
 Supporting Information for Angew. Chem. Int. Ed. Z50567 Wiley-VCH 2003 69451 Weinheim, Germany Metallacarborane-Based Nanostructures: A Carbon-Wired Planar Octagon** Haijun Yao, Michal Sabat, and Russell
Supporting Information for Angew. Chem. Int. Ed. Z50567 Wiley-VCH 2003 69451 Weinheim, Germany Metallacarborane-Based Nanostructures: A Carbon-Wired Planar Octagon** Haijun Yao, Michal Sabat, and Russell
Aziridine in Polymers: A Strategy to Functionalize Polymers by Ring- Opening Reaction of Aziridine
 Electronic Supplementary Material (ESI) for Polymer Chemistry. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information (ESI) Aziridine in Polymers: A Strategy to Functionalize
Electronic Supplementary Material (ESI) for Polymer Chemistry. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information (ESI) Aziridine in Polymers: A Strategy to Functionalize
Supporting Information. for. Angew. Chem. Int. Ed. Z Wiley-VCH 2003
 Supporting Information for Angew. Chem. Int. Ed. Z53001 Wiley-VCH 2003 69451 Weinheim, Germany 1 Ordered Self-Assembly and Electronic Behavior of C 60 -Anthrylphenylacetylene Hybrid ** Seok Ho Kang 1,
Supporting Information for Angew. Chem. Int. Ed. Z53001 Wiley-VCH 2003 69451 Weinheim, Germany 1 Ordered Self-Assembly and Electronic Behavior of C 60 -Anthrylphenylacetylene Hybrid ** Seok Ho Kang 1,
Facile Multistep Synthesis of Isotruxene and Isotruxenone
 Facile Multistep Synthesis of Isotruxene and Isotruxenone Jye-Shane Yang*, Hsin-Hau Huang, and Shih-Hsun Lin Department of Chemistry, National Taiwan University, Taipei, Taiwan 10617 jsyang@ntu.edu.tw
Facile Multistep Synthesis of Isotruxene and Isotruxenone Jye-Shane Yang*, Hsin-Hau Huang, and Shih-Hsun Lin Department of Chemistry, National Taiwan University, Taipei, Taiwan 10617 jsyang@ntu.edu.tw
A Facile Procedure for the Generation of Dichlorocarbene from the Reaction of Carbon Tetrachloride and Magnesium using Ultrasonic Irradiation
 Molecules 2003, 8, 608-613 molecules ISSN 1420-3049 http://www.mdpi.org A Facile Procedure for the Generation of Dichlorocarbene from the Reaction of Carbon Tetrachloride and Magnesium using Ultrasonic
Molecules 2003, 8, 608-613 molecules ISSN 1420-3049 http://www.mdpi.org A Facile Procedure for the Generation of Dichlorocarbene from the Reaction of Carbon Tetrachloride and Magnesium using Ultrasonic
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Supporting Information TEMPO-catalyzed Synthesis of 5-Substituted Isoxazoles from Propargylic
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Supporting Information TEMPO-catalyzed Synthesis of 5-Substituted Isoxazoles from Propargylic
DO NOT WRITE YOUR NAME UNTIL TOLD TO START! CHEM 8B Organic Chemistry II EXAM 2, Summer 2018 (300 points)
 UCSC, Binder Exam 2, SS 18 Full First and Last Name Underline the first initial of your last name D NT WRITE YUR NAME UNTIL TLD T START! CHEM 8B rganic Chemistry II EXAM 2, Summer 2018 (300 points) 1 (50)
UCSC, Binder Exam 2, SS 18 Full First and Last Name Underline the first initial of your last name D NT WRITE YUR NAME UNTIL TLD T START! CHEM 8B rganic Chemistry II EXAM 2, Summer 2018 (300 points) 1 (50)
SUPPORTING INFORMATION
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 SUPPORTING INFORMATION Activation of 1, 3-dioxolane by protic ionic liquid in aqueous media:
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 SUPPORTING INFORMATION Activation of 1, 3-dioxolane by protic ionic liquid in aqueous media:
Suggested solutions for Chapter 28
 s for Chapter 28 28 PBLEM 1 ow would you make these four compounds? Give your disconnections, explain why you chose them and then give reagents for the. 2 2 Me S Exercises in basic one- group C X disconnections.
s for Chapter 28 28 PBLEM 1 ow would you make these four compounds? Give your disconnections, explain why you chose them and then give reagents for the. 2 2 Me S Exercises in basic one- group C X disconnections.
Efficient Syntheses of the Keto-carotenoids Canthaxanthin, Astaxanthin, and Astacene
 Efficient Syntheses of the Keto-carotenoids Canthaxanthin, Astaxanthin, and Astacene Seyoung Choi and Sangho Koo* Department of Chemistry, Myong Ji University, Yongin, Kyunggi-Do, 449-728, Korea. E-mail:
Efficient Syntheses of the Keto-carotenoids Canthaxanthin, Astaxanthin, and Astacene Seyoung Choi and Sangho Koo* Department of Chemistry, Myong Ji University, Yongin, Kyunggi-Do, 449-728, Korea. E-mail:
Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group
 Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Triazabicyclodecene: an Effective Isotope. Exchange Catalyst in CDCl 3
 Triazabicyclodecene: an Effective Isotope Exchange Catalyst in CDCl 3 Supporting Information Cyrille Sabot, Kanduluru Ananda Kumar, Cyril Antheaume, Charles Mioskowski*, Laboratoire de Synthèse Bio-rganique,
Triazabicyclodecene: an Effective Isotope Exchange Catalyst in CDCl 3 Supporting Information Cyrille Sabot, Kanduluru Ananda Kumar, Cyril Antheaume, Charles Mioskowski*, Laboratoire de Synthèse Bio-rganique,
Rhodamine B pentyl ester and its methyl, ethyl, propyl, and butyl homologues
 Rhodamine B pentyl ester and its methyl, ethyl, propyl, and butyl homologues Author Cosgrove, Kelly Published 2006 Journal Title Molbank Version Published DOI https://doi.org/10.3390/m515 Copyright Statement
Rhodamine B pentyl ester and its methyl, ethyl, propyl, and butyl homologues Author Cosgrove, Kelly Published 2006 Journal Title Molbank Version Published DOI https://doi.org/10.3390/m515 Copyright Statement
Drastically Decreased Reactivity of Thiols and Disulfides Complexed by Cucurbit[6]uril
![Drastically Decreased Reactivity of Thiols and Disulfides Complexed by Cucurbit[6]uril Drastically Decreased Reactivity of Thiols and Disulfides Complexed by Cucurbit[6]uril](/thumbs/94/119194061.jpg) SUPPORTING INFORMATION Drastically Decreased Reactivity of Thiols and Disulfides Complexed by Cucurbit[6]uril Lidia Strimbu Berbeci, Wei Wang and Angel E. Kaifer* Center for Supramolecular Science and
SUPPORTING INFORMATION Drastically Decreased Reactivity of Thiols and Disulfides Complexed by Cucurbit[6]uril Lidia Strimbu Berbeci, Wei Wang and Angel E. Kaifer* Center for Supramolecular Science and
Straightforward Synthesis of Enantiopure (R)- and (S)-trifluoroalaninol
 S1 Supplementary Material (ESI) for Organic & Biomolecular Chemistry This journal is (c) The Royal Society of Chemistry 2010 Straightforward Synthesis of Enantiopure (R)- and (S)-trifluoroalaninol Julien
S1 Supplementary Material (ESI) for Organic & Biomolecular Chemistry This journal is (c) The Royal Society of Chemistry 2010 Straightforward Synthesis of Enantiopure (R)- and (S)-trifluoroalaninol Julien
Supramolecular complexes of bambusuril with dialkyl phosphates
 Supramolecular complexes of bambusuril with dialkyl phosphates Tomas Fiala and Vladimir Sindelar RECETX, Masaryk University, Kamenice 5, 62500 Brno, Czech Republic Contents Synthesis... S2 Tripropargyl
Supramolecular complexes of bambusuril with dialkyl phosphates Tomas Fiala and Vladimir Sindelar RECETX, Masaryk University, Kamenice 5, 62500 Brno, Czech Republic Contents Synthesis... S2 Tripropargyl
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Rh 2 (Ac) 4 -Catalyzed 2,3-Migration of -rrocenecarboxyl -Diazocarbonyl
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Rh 2 (Ac) 4 -Catalyzed 2,3-Migration of -rrocenecarboxyl -Diazocarbonyl
Highly Luminescent -Conjugated Dithienometalloles: Photophysical Properties and Application to Organic Light-Emitting Diodes
 Electronic Supplementary Information (ESI) Highly Luminescent -Conjugated Dithienometalloles: Photophysical Properties and Application to Organic Light-Emitting Diodes Ryosuke Kondo, a Takuma Yasuda,*
Electronic Supplementary Information (ESI) Highly Luminescent -Conjugated Dithienometalloles: Photophysical Properties and Application to Organic Light-Emitting Diodes Ryosuke Kondo, a Takuma Yasuda,*
An Efficient Total Synthesis and Absolute Configuration. Determination of Varitriol
 An Efficient Total Synthesis and Absolute Configuration Determination of Varitriol Ryan T. Clemens and Michael P. Jennings * Department of Chemistry, University of Alabama, 500 Campus Dr. Tuscaloosa, AL
An Efficient Total Synthesis and Absolute Configuration Determination of Varitriol Ryan T. Clemens and Michael P. Jennings * Department of Chemistry, University of Alabama, 500 Campus Dr. Tuscaloosa, AL
Supporting Information
 Supporting Information Lewis acid-catalyzed intramolecular condensation of ynol ether-acetals. Synthesis of alkoxycycloalkene carboxylates Vincent Tran and Thomas G. Minehan * Department of Chemistry and
Supporting Information Lewis acid-catalyzed intramolecular condensation of ynol ether-acetals. Synthesis of alkoxycycloalkene carboxylates Vincent Tran and Thomas G. Minehan * Department of Chemistry and
Supporting Information. Table of Contents. 1. General Notes Experimental Details 3-12
 Supporting Information Table of Contents page 1. General Notes 2 2. Experimental Details 3-12 3. NMR Support for Timing of Claisen/Diels-Alder/Claisen 13 4. 1 H and 13 C NMR 14-37 General Notes All reagents
Supporting Information Table of Contents page 1. General Notes 2 2. Experimental Details 3-12 3. NMR Support for Timing of Claisen/Diels-Alder/Claisen 13 4. 1 H and 13 C NMR 14-37 General Notes All reagents
12.5 Organometallic Compounds
 12.5 rganometallic Compounds Compounds that contain carbon-metal bond are called organometallic compounds. C M C δ δ + M C M Primarily ionic Primarily covalent (M = Na + or K + )(M = Mg or Li) (M = Pb,
12.5 rganometallic Compounds Compounds that contain carbon-metal bond are called organometallic compounds. C M C δ δ + M C M Primarily ionic Primarily covalent (M = Na + or K + )(M = Mg or Li) (M = Pb,
Diastereoselectivity in the Staudinger reaction of. pentafluorosulfanylaldimines and ketimines
 Supporting Information for Diastereoselectivity in the Staudinger reaction of pentafluorosulfanylaldimines and ketimines Alexander Penger, Cortney. von ahmann, Alexander S. Filatov and John T. Welch* Address:
Supporting Information for Diastereoselectivity in the Staudinger reaction of pentafluorosulfanylaldimines and ketimines Alexander Penger, Cortney. von ahmann, Alexander S. Filatov and John T. Welch* Address:
Supporting Information
 Supporting Information Wiley-VCH 2012 69451 Weinheim, Germany Concise Syntheses of Insect Pheromones Using Z-Selective Cross Metathesis** Myles B. Herbert, Vanessa M. Marx, Richard L. Pederson, and Robert
Supporting Information Wiley-VCH 2012 69451 Weinheim, Germany Concise Syntheses of Insect Pheromones Using Z-Selective Cross Metathesis** Myles B. Herbert, Vanessa M. Marx, Richard L. Pederson, and Robert
Electronic Supplementary Information
 Electronic Supplementary Information Effect of polymer chain conformation on field-effect transistor performance: synthesis and properties of two arylene imide based D-A copolymers Dugang Chen, a Yan Zhao,
Electronic Supplementary Information Effect of polymer chain conformation on field-effect transistor performance: synthesis and properties of two arylene imide based D-A copolymers Dugang Chen, a Yan Zhao,
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group
 Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Supporting Information
 Supporting Information SmI 2 -Mediated Carbon-Carbon Bond Fragmentation in α-aminomethyl Malonates Qiongfeng Xu,, Bin Cheng, $, Xinshan Ye,*, and Hongbin Zhai*,,,$ The State Key Laboratory of Natural and
Supporting Information SmI 2 -Mediated Carbon-Carbon Bond Fragmentation in α-aminomethyl Malonates Qiongfeng Xu,, Bin Cheng, $, Xinshan Ye,*, and Hongbin Zhai*,,,$ The State Key Laboratory of Natural and
Chapter 19. Synthesis and Reactions of b-dicarbonyl Compounds: More Chemistry of Enolate Anions. ß-dicarbonyl compounds. Why are ß-dicarbonyls useful?
 Chapter 19 Synthesis and Reactions of b-dicarbonyl Compounds: More Chemistry of Enolate Anions ß-dicarbonyl compounds Two carbonyl groups separated by a carbon Three common types ß-diketone ß-ketoester
Chapter 19 Synthesis and Reactions of b-dicarbonyl Compounds: More Chemistry of Enolate Anions ß-dicarbonyl compounds Two carbonyl groups separated by a carbon Three common types ß-diketone ß-ketoester
ROADMAP FOR REACTIONS Chapter 6
 RADMAP FR REACTINS Chapter 6 (A) (B) (C) (D) (E) (F) (G) (H) (I) (J) (K) (L) (M) (N) () Regiochemistry: Markovnikov addition to a bond Stereochemistry: anti-addition Regiochemistry: non-markovnikov addition
RADMAP FR REACTINS Chapter 6 (A) (B) (C) (D) (E) (F) (G) (H) (I) (J) (K) (L) (M) (N) () Regiochemistry: Markovnikov addition to a bond Stereochemistry: anti-addition Regiochemistry: non-markovnikov addition
Preparation of the reagent TMPMgCl LiCl [1] (1):
![Preparation of the reagent TMPMgCl LiCl [1] (1): Preparation of the reagent TMPMgCl LiCl [1] (1):](/thumbs/80/82269925.jpg) Regio- and Chemoselective Magnesiation of Protected Uracils and Thiouracils using TMPMgCl LiCl and TMP 2 Mg 2LiCl upporting nformation Marc Mosrin, adège Boudet and Paul Knochel Ludwig-Maximilians-Universität
Regio- and Chemoselective Magnesiation of Protected Uracils and Thiouracils using TMPMgCl LiCl and TMP 2 Mg 2LiCl upporting nformation Marc Mosrin, adège Boudet and Paul Knochel Ludwig-Maximilians-Universität
CARBOXYLIC ACIDS AND THEIR DERIVATIVES. 3. Predict the relative acidity and basicity of compounds and ions. Important criteria include:
 CARBXYLIC ACIDS AND TEIR DERIVATIVES A STUDENT SULD BE ABLE T: 1. Give the IUPAC name given the structure, and draw the structure given the name, of carboxylic acids and their metal salts, acyl chlorides,
CARBXYLIC ACIDS AND TEIR DERIVATIVES A STUDENT SULD BE ABLE T: 1. Give the IUPAC name given the structure, and draw the structure given the name, of carboxylic acids and their metal salts, acyl chlorides,
CHEM 203. Final Exam December 15, 2010 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
Electronic Supplementary Information. Highly Efficient Deep-Blue Emitting Organic Light Emitting Diode Based on the
 Electronic Supplementary Information Highly Efficient Deep-Blue Emitting rganic Light Emitting Diode Based on the Multifunctional Fluorescent Molecule Comprising Covalently Bonded Carbazole and Anthracene
Electronic Supplementary Information Highly Efficient Deep-Blue Emitting rganic Light Emitting Diode Based on the Multifunctional Fluorescent Molecule Comprising Covalently Bonded Carbazole and Anthracene
Serendipitous synthesis of 1,4-benzothiazin derivatives using 2-[(2-aminophenyl)disulfanyl]aniline
![Serendipitous synthesis of 1,4-benzothiazin derivatives using 2-[(2-aminophenyl)disulfanyl]aniline Serendipitous synthesis of 1,4-benzothiazin derivatives using 2-[(2-aminophenyl)disulfanyl]aniline](/thumbs/94/118068899.jpg) erendipitous synthesis of 1,4-benzothiazin derivatives using 2-[(2-aminophenyl)disulfanyl]aniline Mohammad Reza Islami a *, Fouziyeh Mollazehi a, Alireza Badiei b, and assan heibani a a Department of Chemistry,
erendipitous synthesis of 1,4-benzothiazin derivatives using 2-[(2-aminophenyl)disulfanyl]aniline Mohammad Reza Islami a *, Fouziyeh Mollazehi a, Alireza Badiei b, and assan heibani a a Department of Chemistry,
Issue in Honor of Academician Michael G. Voronkov ARKIVOC 2001 (ix) 7-11
 Preparation of trialkylgermyl and -stannyl derivatives of methyl isothiocyanate by in situ trapping of anionic intermediates with chloro(trialkyl)germane and stannanes Lambert Brandsma a and Nina A. Nedolya
Preparation of trialkylgermyl and -stannyl derivatives of methyl isothiocyanate by in situ trapping of anionic intermediates with chloro(trialkyl)germane and stannanes Lambert Brandsma a and Nina A. Nedolya
SUPPORTING INFORMATION
 UPPRTING INFRMATIN Application of a Rhodium-Catalyzed Addition/Cyclization equence Toward the ynthesis of Polycyclic eteroaromatics Nai-Wen Tseng and Mark Lautens* Davenport Laboratories, Chemistry Department,
UPPRTING INFRMATIN Application of a Rhodium-Catalyzed Addition/Cyclization equence Toward the ynthesis of Polycyclic eteroaromatics Nai-Wen Tseng and Mark Lautens* Davenport Laboratories, Chemistry Department,
Supporting Information
 Supporting Information Wiley-VCH 2006 69451 Weinheim, Germany A Highly Enantioselective Brønsted Acid Catalyst for the Strecker Reaction Magnus Rueping, * Erli Sugiono and Cengiz Azap General: Unless otherwise
Supporting Information Wiley-VCH 2006 69451 Weinheim, Germany A Highly Enantioselective Brønsted Acid Catalyst for the Strecker Reaction Magnus Rueping, * Erli Sugiono and Cengiz Azap General: Unless otherwise
Metal-free general procedure for oxidation of secondary amines to nitrones
 S1 Supporting information Metal-free general procedure for oxidation of secondary amines to nitrones Carolina Gella, Èric Ferrer, Ramon Alibés, Félix Busqué,* Pedro de March, Marta Figueredo,* and Josep
S1 Supporting information Metal-free general procedure for oxidation of secondary amines to nitrones Carolina Gella, Èric Ferrer, Ramon Alibés, Félix Busqué,* Pedro de March, Marta Figueredo,* and Josep
Supporting Information. A rapid and efficient synthetic route to terminal. arylacetylenes by tetrabutylammonium hydroxide- and
 Supporting Information for A rapid and efficient synthetic route to terminal arylacetylenes by tetrabutylammonium hydroxide- and methanol-catalyzed cleavage of 4-aryl-2-methyl-3- butyn-2-ols Jie Li and
Supporting Information for A rapid and efficient synthetic route to terminal arylacetylenes by tetrabutylammonium hydroxide- and methanol-catalyzed cleavage of 4-aryl-2-methyl-3- butyn-2-ols Jie Li and
Significant improvement of dye-sensitized solar cell. performance by a slim phenothiazine based dyes
 Significant improvement of dye-sensitized solar cell performance by a slim phenothiazine based dyes Yong Hua, a Shuai Chang, b Dandan Huang, c Xuan Zhou, a Xunjin Zhu, *a,d Jianzhang Zhao, c Tao Chen,
Significant improvement of dye-sensitized solar cell performance by a slim phenothiazine based dyes Yong Hua, a Shuai Chang, b Dandan Huang, c Xuan Zhou, a Xunjin Zhu, *a,d Jianzhang Zhao, c Tao Chen,
An unusual dianion equivalent from acylsilanes for the synthesis of substituted β-keto esters
 S1 An unusual dianion equivalent from acylsilanes for the synthesis of substituted β-keto esters Chris V. Galliford and Karl A. Scheidt* Department of Chemistry, Northwestern University, 2145 Sheridan
S1 An unusual dianion equivalent from acylsilanes for the synthesis of substituted β-keto esters Chris V. Galliford and Karl A. Scheidt* Department of Chemistry, Northwestern University, 2145 Sheridan
A Mild, Catalytic and Highly Selective Method for the Oxidation of α,β- Enones to 1,4-Enediones. Jin-Quan Yu, a and E. J.
 A Mild, Catalytic and Highly Selective Method for the Oxidation of α,β- Enones to 1,4-Enediones Jin-Quan Yu, a and E. J. Corey b * a Department of Chemistry, Cambridge University, Cambridge CB2 1EW, United
A Mild, Catalytic and Highly Selective Method for the Oxidation of α,β- Enones to 1,4-Enediones Jin-Quan Yu, a and E. J. Corey b * a Department of Chemistry, Cambridge University, Cambridge CB2 1EW, United
Supporting Information. (1S,8aS)-octahydroindolizidin-1-ol.
 SI-1 Supporting Information Non-Racemic Bicyclic Lactam Lactones Via Regio- and cis-diastereocontrolled C H insertion. Asymmetric Synthesis of (8S,8aS)-octahydroindolizidin-8-ol and (1S,8aS)-octahydroindolizidin-1-ol.
SI-1 Supporting Information Non-Racemic Bicyclic Lactam Lactones Via Regio- and cis-diastereocontrolled C H insertion. Asymmetric Synthesis of (8S,8aS)-octahydroindolizidin-8-ol and (1S,8aS)-octahydroindolizidin-1-ol.
A fluorinated dendritic TsDPEN-Ru(II) catalyst for asymmetric transfer hydrogenation of prochiral ketones in aqueous media
 Supplementary Information A fluorinated dendritic TsDPEN-Ru(II) catalyst for asymmetric transfer hydrogenation of prochiral ketones in aqueous media Weiwei Wang and Quanrui Wang* Department of Chemistry,
Supplementary Information A fluorinated dendritic TsDPEN-Ru(II) catalyst for asymmetric transfer hydrogenation of prochiral ketones in aqueous media Weiwei Wang and Quanrui Wang* Department of Chemistry,
Supporting Information
 Supporting Information Efficient copper-catalyzed coupling of aryl chlorides, bromides and iodides with aqueous ammonia Hanhui Xu and Christian Wolf* Department of Chemistry, Georgetown University, Washington
Supporting Information Efficient copper-catalyzed coupling of aryl chlorides, bromides and iodides with aqueous ammonia Hanhui Xu and Christian Wolf* Department of Chemistry, Georgetown University, Washington
Influence of anellation in N-heterocyclic carbenes: Detection of novel quinoxalineanellated NHC by trapping as transition metal complexes
 Influence of anellation in N-heterocyclic carbenes: Detection of novel quinoxalineanellated NHC by trapping as transition metal complexes Shanmuganathan Saravanakumar, a Markus K. Kindermann, a Joachim
Influence of anellation in N-heterocyclic carbenes: Detection of novel quinoxalineanellated NHC by trapping as transition metal complexes Shanmuganathan Saravanakumar, a Markus K. Kindermann, a Joachim
Enantioselectivity switch in copper-catalyzed conjugate addition. reaction under influence of a chiral N-heterocyclic carbene-silver complex
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Supplementary Information Enantioselectivity switch in copper-catalyzed conjugate addition
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Supplementary Information Enantioselectivity switch in copper-catalyzed conjugate addition
Full First and Last Name DO NOT WRITE YOUR NAME UNTIL TOLD TO START! CHEM 8B Organic Chemistry II EXAM 2, Winter 2017 (200 points)
 UCSC, Binder Exam 2, W17 Full First and Last Name D NT WRITE YUR NAME UNTIL TLD T START! CHEM 8B rganic Chemistry II EXAM 2, Winter 2017 (200 points) In each of the following problems, use your knowledge
UCSC, Binder Exam 2, W17 Full First and Last Name D NT WRITE YUR NAME UNTIL TLD T START! CHEM 8B rganic Chemistry II EXAM 2, Winter 2017 (200 points) In each of the following problems, use your knowledge
Preparation and ring-opening reactions of N- diphenylphosphinyl vinyl aziridines
 Supporting Information for Preparation and ring-opening reactions of N- diphenylphosphinyl vinyl aziridines Ashley N. Jarvis 1, Andrew B. McLaren 1, Helen M. I. Osborn 1 and Joseph Sweeney* 1,2 Address:
Supporting Information for Preparation and ring-opening reactions of N- diphenylphosphinyl vinyl aziridines Ashley N. Jarvis 1, Andrew B. McLaren 1, Helen M. I. Osborn 1 and Joseph Sweeney* 1,2 Address:
Supporting Information
 Intramolecular hydrogen-bonding activation in cysteines. New effective radical scavenging Luisa Haya, a Iñaki Osante, b Ana M. Mainar, a Carlos Cativiela, b Jose S. Urieta*,a a Group of Applied Thermodynamics
Intramolecular hydrogen-bonding activation in cysteines. New effective radical scavenging Luisa Haya, a Iñaki Osante, b Ana M. Mainar, a Carlos Cativiela, b Jose S. Urieta*,a a Group of Applied Thermodynamics
Supporting Information
 An Improved ynthesis of the Pyridine-Thiazole Cores of Thiopeptide Antibiotics Virender. Aulakh, Marco A. Ciufolini* Department of Chemistry, University of British Columbia 2036 Main Mall, Vancouver, BC
An Improved ynthesis of the Pyridine-Thiazole Cores of Thiopeptide Antibiotics Virender. Aulakh, Marco A. Ciufolini* Department of Chemistry, University of British Columbia 2036 Main Mall, Vancouver, BC
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2017 Supporting Information Lithium Triethylborohydride-Promoted Generation of α,α-difluoroenolates
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2017 Supporting Information Lithium Triethylborohydride-Promoted Generation of α,α-difluoroenolates
Supporting Information For:
 Supporting Information For: Peptidic α-ketocarboxylic Acids and Sulfonamides as Inhibitors of Protein Tyrosine Phosphatases Yen Ting Chen, Jian Xie, and Christopher T. Seto* Department of Chemistry, Brown
Supporting Information For: Peptidic α-ketocarboxylic Acids and Sulfonamides as Inhibitors of Protein Tyrosine Phosphatases Yen Ting Chen, Jian Xie, and Christopher T. Seto* Department of Chemistry, Brown
dichloropyrimidine (1.5 g, 10.1 mmol) in THF (10 ml) added at -116 C under nitrogen atmosphere.
 Supporting Information Experimental The presence of atropisomerism arising from diastereoisomerism is indicated in the 13 C spectra of the relevant compounds with the second isomer being indicated with
Supporting Information Experimental The presence of atropisomerism arising from diastereoisomerism is indicated in the 13 C spectra of the relevant compounds with the second isomer being indicated with
Department of Chemistry, Colorado State University, Fort Collins, Colorado University of Colorado Cancer Center, Aurora, Colorado 80045
 Improved Biomimetic Total Synthesis of d,l-stephacidin A Thomas J. Greshock 1 and Robert M. Williams 1,2 * 1 Department of Chemistry, Colorado State University, Fort Collins, Colorado 80523 2 University
Improved Biomimetic Total Synthesis of d,l-stephacidin A Thomas J. Greshock 1 and Robert M. Williams 1,2 * 1 Department of Chemistry, Colorado State University, Fort Collins, Colorado 80523 2 University
Supporting Information:
 Enantioselective Synthesis of (-)-Codeine and (-)-Morphine Barry M. Trost* and Weiping Tang Department of Chemistry, Stanford University, Stanford, CA 94305-5080 1. Aldehyde 7. Supporting Information:
Enantioselective Synthesis of (-)-Codeine and (-)-Morphine Barry M. Trost* and Weiping Tang Department of Chemistry, Stanford University, Stanford, CA 94305-5080 1. Aldehyde 7. Supporting Information:
Supporting Information for
 Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2017 Supporting Information for
Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2017 Supporting Information for
Supporting Text Synthesis of (2 S ,3 S )-2,3-bis(3-bromophenoxy)butane (3). Synthesis of (2 S ,3 S
 Supporting Text Synthesis of (2S,3S)-2,3-bis(3-bromophenoxy)butane (3). Under N 2 atmosphere and at room temperature, a mixture of 3-bromophenol (0.746 g, 4.3 mmol) and Cs 2 C 3 (2.81 g, 8.6 mmol) in DMS
Supporting Text Synthesis of (2S,3S)-2,3-bis(3-bromophenoxy)butane (3). Under N 2 atmosphere and at room temperature, a mixture of 3-bromophenol (0.746 g, 4.3 mmol) and Cs 2 C 3 (2.81 g, 8.6 mmol) in DMS
Addition of Organochromium Reagents to Heteroaryl Aldehydes. Synthesis of Heteroaryl Substituted bis-allyl Ethers and Homoallyl Ethers
 Molecules 2004, 9, 22-28 molecules ISSN 1420-3049 http://www.mdpi.org Addition of rganochromium Reagents to Heteroaryl Aldehydes. Synthesis of Heteroaryl Substituted bis-allyl Ethers and Homoallyl Ethers
Molecules 2004, 9, 22-28 molecules ISSN 1420-3049 http://www.mdpi.org Addition of rganochromium Reagents to Heteroaryl Aldehydes. Synthesis of Heteroaryl Substituted bis-allyl Ethers and Homoallyl Ethers
Simplified platensimycin analogues as antibacterial agents
 Simplified platensimycin analogues as antibacterial agents Dragan Krsta, a Caron Ka, a Ian T. Crosby, a Ben Capuano a and David T. Manallack a * a Medicinal Chemistry and Drug Action, Monash Institute
Simplified platensimycin analogues as antibacterial agents Dragan Krsta, a Caron Ka, a Ian T. Crosby, a Ben Capuano a and David T. Manallack a * a Medicinal Chemistry and Drug Action, Monash Institute
