DO NOT WRITE YOUR NAME UNTIL TOLD TO START! CHEM 8B Organic Chemistry II EXAM 2, Summer 2018 (300 points)
|
|
|
- Jayson Stuart Cross
- 5 years ago
- Views:
Transcription
1 UCSC, Binder Exam 2, SS 18 Full First and Last Name Underline the first initial of your last name D NT WRITE YUR NAME UNTIL TLD T START! CHEM 8B rganic Chemistry II EXAM 2, Summer 2018 (300 points) 1 (50) 2 (50) 3 (50) 4 (50) 5 (50) 6 (50) Total / 300 % In each of the following problems, use your knowledge of organic chemistry conventions to answer the questions in the proper manner. Be sure to read all questions & instructions carefully, paying special attention to directions to skip parts of problems. Choose which problems to skip rather than doing every problem, otherwise you may run out of time. Make sure it is crystal clear which problems should and should not be graded by circling and X ing out those problems, respectively. You will have the entire class period to complete this exam (2 hours), but hopefully you won t need it! You are welcome to use pre-built models. Keep your eyes on your own paper. Electronic devices of any kind are not allowed, including cell phones and calculators. Any student found using any of said devices, or found examining another student s exam or cheat sheet, will be promptly removed from the exam room and will receive a zero on this exam. Such an incident will be reported to the UCSC Judiciary Affairs Committee, which determines the consequences (it s not worth it). Functional Group Acid chloride Acid Anhydride Carboxylic Acid Esters Amides Suffix -oyl chloride -oic anhyride -oic acid -oate -amide
2 1. Nomenclature & Acid-Base Chemistry suffixes for acyl derivatives on the cover page (a) (15 points) Draw structures corresponding to any three of the following names. Skip one by drawing a large X over it, otherwise (i) - (iii) will be graded. (i) 2,4-Dimethylpentanenitrile (ii) Acetyl chloride (ethanoyl chloride) (iii) Propyl benzoate (iv) N-ethyl-N-methyl-cyclohexane carboxamide (b) (15 points) Draw the structure of 4-cyano-8-hydroxy-5,9-dioxo-nonanoic acid. (c) (10 points) Circle the more acidic compound in each pair and provide the approximate pka of each on the line provided: less than (<), greater than (>), or about equal to (~) one of the memorized pka standards). F vs. H vs. H Approximate pka (d) (10 points) Draw the enol tautomer of acetophenone. acetophenone (keto form) enol form 1
3 2. (50 points) Single Step Reactions Fill in the box with the reactant, reagent(s), or product. Choose any five (skip one by drawing an X over the box), otherwise (a) (e) are graded. (a) H 1. Br 2, PBr 3 2. H 2 (b) NaH, H 2 Br 2 (c) EtH, NaH (2 mol) (1 mol) H + H 2 (1 mol) (d) Br 2 HAc major (more stable) product (e) H H 3 + (f) 2
4 3. (50 points) Reaction Puzzles CHSE NE Puzzle 1 Fill in the missing reagents for reactions (a) through (f) on the lines below. a H b Br c H 2 C d N f HC e H (a) (b) (c) (d) (e) (f) Puzzle 2 Fill in the missing reagents and products in the boxes provided. Br H 2 NPh H + H 1. NaBH 4 2. H 2 Cr 3 H 2 S 4 CN NHPh LiAlH 4 2. H 2 SCl 2 NH 2 H 2 NPh 3
5 4. Basic Mechanisms Draw the arrow-pushing mechanism for both reactions below, including all arrows for proton transfers. Include all intermediates with proper charges circled for each step. FILL IN THE BLANK: Basic mechanisms should never have charges. (a) (20 points) Draw the mechanism for the basic hydrolysis of the following acid chloride. Cl NaH + HCl (b) (30 points) Draw the mechanism for the reduction of the lactone (cyclic ester) with lithium aluminum hydride, followed by quenching with acid. 1. LiAlH 4 2. H 3 + H H 4
6 5. (20 points) Acidic Mechanisms, CHSE NE - Draw the arrow-pushing mechanism for either reaction below, including all arrows for proton transfers. Include all intermediates with proper charges circled for each step. FILL IN THE BLANK: Acidic mechanisms should never have charges. (a) The reaction of nitriles with Grignard reagents yields a ketone product. This reaction goes through an imine ion intermediate, which is then hydrolyzed under acidic conditions. Draw the mechanism from the imine ion to the ketone product in the space below. No mechanism necessary for the Grignard addition step. CN CH 3 MgBr N H + H 2 + NH 4 + Imine Ion Ketone Product (b) Draw the mechanism for the acid-catalyzed reaction of acetone with methanol to give the acetal product below. excess CH 3 H H 3 C CH 3 + H 2 H + Acetal X UT THE PART T SKIP WE WILL NT CHSE THE BEST NE FR YU! 5
7 6. (50 points) Multi-Step Synthesis - Carry out any two of the syntheses below using the starting material provided and any other reagents or carbon sources needed. Draw the product after each synthetic step. No mechanisms. (a) H (b) H Ph H Ph Ph H Ph H (c) H N(CH 3 ) 2 (d) CN X UT THE TW PARTS T SKIP WE WILL NT CHSE THE BEST NES FR YU! 6
Full First and Last Name DO NOT WRITE YOUR NAME UNTIL TOLD TO START! CHEM 8B Organic Chemistry II EXAM 2, Winter 2017 (200 points)
 UCSC, Binder Exam 2, W17 Full First and Last Name D NT WRITE YUR NAME UNTIL TLD T START! CHEM 8B rganic Chemistry II EXAM 2, Winter 2017 (200 points) In each of the following problems, use your knowledge
UCSC, Binder Exam 2, W17 Full First and Last Name D NT WRITE YUR NAME UNTIL TLD T START! CHEM 8B rganic Chemistry II EXAM 2, Winter 2017 (200 points) In each of the following problems, use your knowledge
DO NOT WRITE YOUR NAME UNTIL TOLD TO START! CHEM 8B Organic Chemistry II EXAM 2, Summer 2018 (300 points)
 ' UCSC Binder KEY Exam SS 18 Full First and Last Name Underline the first initial of your last name 1 (50) non (50) single step 3 (50) Puzzle 4 (50) men 5 (50) mean D NT WRITE YUR NAME UNTIL TLD T START!
' UCSC Binder KEY Exam SS 18 Full First and Last Name Underline the first initial of your last name 1 (50) non (50) single step 3 (50) Puzzle 4 (50) men 5 (50) mean D NT WRITE YUR NAME UNTIL TLD T START!
UCSC, Binder. CHEM 8B Organic Chemistry II EXAM 2A, Winter 2018 (200 points)
 UCSC, Binder Name CEM 8B rganic Chemistry II EXAM 2A, Winter 2018 (200 points) Use your knowledge of organic chemistry conventions to answer the questions in the proper manner. Be sure to read all questions
UCSC, Binder Name CEM 8B rganic Chemistry II EXAM 2A, Winter 2018 (200 points) Use your knowledge of organic chemistry conventions to answer the questions in the proper manner. Be sure to read all questions
UCSC, Binder. Section TA. CHEM 108B Organic Chemistry II FINAL EXAM, Version B (400 points)
 UCSC, Binder Name Section TA SID CEM 108B rganic Chemistry II FINAL EXAM, Version B (400 points) In each of the following problems, use your knowledge of organic chemistry conventions to answer the questions
UCSC, Binder Name Section TA SID CEM 108B rganic Chemistry II FINAL EXAM, Version B (400 points) In each of the following problems, use your knowledge of organic chemistry conventions to answer the questions
UCSC, Binder. CHEM 8B Organic Chemistry II FINAL EXAM, Version A (300 points)
 UCSC, Binder TA Name SID CEM 8B rganic Chemistry II FINAL EXAM, Version A (300 points) In each of the following problems, use your knowledge of organic chemistry conventions to answer the questions in
UCSC, Binder TA Name SID CEM 8B rganic Chemistry II FINAL EXAM, Version A (300 points) In each of the following problems, use your knowledge of organic chemistry conventions to answer the questions in
UCSC, Binder (First and Last) CHEM 8A, Organic Chemistry I Summer 18 FINAL EXAM (400 points) 1 (60)
 UCSC, Binder Name (First and Last) CEM 8A, rganic Chemistry I Summer 18 FINAL EXAM (400 points) 1 (60) 2 (65) 3 (60) 4 (70) 5 (75) 6 (70) Total (400) % In each of the following problems, use your knowledge
UCSC, Binder Name (First and Last) CEM 8A, rganic Chemistry I Summer 18 FINAL EXAM (400 points) 1 (60) 2 (65) 3 (60) 4 (70) 5 (75) 6 (70) Total (400) % In each of the following problems, use your knowledge
UCSC, Binder. CHEM 8B Organic Chemistry II FINAL EXAM (300 points)
 UCSC, Binder Name CEM 8B rganic Chemistry II FINAL EXAM (300 points) In each of the following problems, use your knowledge of organic chemistry conventions to answer the questions in the proper manner.
UCSC, Binder Name CEM 8B rganic Chemistry II FINAL EXAM (300 points) In each of the following problems, use your knowledge of organic chemistry conventions to answer the questions in the proper manner.
CHEM 8A Summer Student ID # Organic Chemistry EXAM 2 (300 points)
 CEM 8A Summer 2017 UCSC, Binder Name Student ID # rganic Chemistry EXAM 2 (300 points) In each of the following problems, you will use your knowledge of organic chemistry conventions to answer the questions
CEM 8A Summer 2017 UCSC, Binder Name Student ID # rganic Chemistry EXAM 2 (300 points) In each of the following problems, you will use your knowledge of organic chemistry conventions to answer the questions
CHEM 8A Summer Student ID # Organic Chemistry FINAL EXAM (400 points)
 CEM 8A Summer 2017 UCSC, Binder Name Student ID # rganic Chemistry FINAL EXAM (400 points) In each of the following problems, you will use your knowledge of organic chemistry conventions to answer the
CEM 8A Summer 2017 UCSC, Binder Name Student ID # rganic Chemistry FINAL EXAM (400 points) In each of the following problems, you will use your knowledge of organic chemistry conventions to answer the
PLEASE DO NOT WRITE ON THE EXAM (EVEN YOUR NAME) UNTIL TOLD TO START! Student ID # CHEM 8A, Organic Chemistry EXAM 1 (300 points)
 PLEASE D T WRITE TE EXAM (EVE YUR AME) UTIL TLD T START! UCSC, Binder ame Student ID # CEM 8A, rganic Chemistry EXAM (300 points) In each of the following problems, use your knowledge of organic chemistry
PLEASE D T WRITE TE EXAM (EVE YUR AME) UTIL TLD T START! UCSC, Binder ame Student ID # CEM 8A, rganic Chemistry EXAM (300 points) In each of the following problems, use your knowledge of organic chemistry
CHEM Organic Chemistry with Biological Applications EXAM 1A (250 points)
 UCSC, Binder Name Student ID # Section Day/Time CEM 109 rganic Chemistry with Biological Applications EXAM 1A (250 points) D NT BEGIN TE EXAM R TURN TE PAGE UNTIL INSTRUCTED T D S. In the meantime, please
UCSC, Binder Name Student ID # Section Day/Time CEM 109 rganic Chemistry with Biological Applications EXAM 1A (250 points) D NT BEGIN TE EXAM R TURN TE PAGE UNTIL INSTRUCTED T D S. In the meantime, please
TA Section Day/Time. Organic Chemistry EXAM 1A (200 points)
 UCSC, Binder ame TA Section Day/Time rganic Chemistry EXAM 1A (200 points) D T WRITE TE EXAM R TUR TE PAGE UTIL ISTRUCTED T D S. In the meantime, please read the instructions below. Page 1 (40) Page 2
UCSC, Binder ame TA Section Day/Time rganic Chemistry EXAM 1A (200 points) D T WRITE TE EXAM R TUR TE PAGE UTIL ISTRUCTED T D S. In the meantime, please read the instructions below. Page 1 (40) Page 2
CHEM 343 Principles of Organic Chemistry II Summer Instructor: Paul J. Bracher. Quiz # 3. Monday, July 21 st, :30 a.m.
 CHEM 343 Principles of Organic Chemistry II Summer 2014 Quiz # 3 Problem Booklet Page 1 of 7 CHEM 343 Principles of Organic Chemistry II Summer 2014 Instructor: Paul J. Bracher Quiz # 3 Monday, July 21
CHEM 343 Principles of Organic Chemistry II Summer 2014 Quiz # 3 Problem Booklet Page 1 of 7 CHEM 343 Principles of Organic Chemistry II Summer 2014 Instructor: Paul J. Bracher Quiz # 3 Monday, July 21
Chemistry 234 Practice Exam 3. The Periodic Table
 ame: Last First MI Chemistry 234 Practice Exam 3 Instructions: The first 15 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron sheet.
ame: Last First MI Chemistry 234 Practice Exam 3 Instructions: The first 15 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron sheet.
LECTURE #22 Thurs., Nov.15, 2007
 Provide a rxn sequence to make these as the major products Answers: 1. i Pr-Cl, AlCl 3 2. conc. fuming? H 2 S 4 3. Cl 2, FeCl 3 or AlCl 3 4. dilute H 2 S 4 note: normally aqueous workup after step 1, but
Provide a rxn sequence to make these as the major products Answers: 1. i Pr-Cl, AlCl 3 2. conc. fuming? H 2 S 4 3. Cl 2, FeCl 3 or AlCl 3 4. dilute H 2 S 4 note: normally aqueous workup after step 1, but
CHEM 343 Principles of Organic Chemistry II Summer Instructor: Paul J. Bracher. Quiz # 3. Monday, July 21 st, :30 a.m.
 CHEM 343 Principles of Organic Chemistry II Summer 2014 Quiz # 3 Solutions Key Page 1 of 9 CHEM 343 Principles of Organic Chemistry II Summer 2014 Instructor: Paul J. Bracher Quiz # 3 Monday, July 21 st,
CHEM 343 Principles of Organic Chemistry II Summer 2014 Quiz # 3 Solutions Key Page 1 of 9 CHEM 343 Principles of Organic Chemistry II Summer 2014 Instructor: Paul J. Bracher Quiz # 3 Monday, July 21 st,
Student ID # Organic Chemistry EXAM 1 (300 points)
 UCSC, Binder Name Student ID # rganic Chemistry EXAM 1 (300 points) In each of the following problems, use your knowledge of organic chemistry conventions to answer the questions in the proper manner.
UCSC, Binder Name Student ID # rganic Chemistry EXAM 1 (300 points) In each of the following problems, use your knowledge of organic chemistry conventions to answer the questions in the proper manner.
UCSC, Binder. CHEM 8A, Organic Chemistry I Summer 18 EXAM 1 (300 points)
 UCSC, Binder Name CEM 8A, rganic Chemistry I Summer 18 EXAM 1 (300 points) 1 (40) 2 (50) 3 (65) 4 (45) 5 (45) 6 (50) Total (300) % In each of the following problems, use your knowledge of organic chemistry
UCSC, Binder Name CEM 8A, rganic Chemistry I Summer 18 EXAM 1 (300 points) 1 (40) 2 (50) 3 (65) 4 (45) 5 (45) 6 (50) Total (300) % In each of the following problems, use your knowledge of organic chemistry
ORGANIC - BROWN 8E CH CARBOXYLIC ACIDS.
 RGANIC - BRWN 8E CH. 17 - CARBXYLIC ACIDS!! www.clutchprep.com RGANIC - BRWN 8E CH. 17 - CARBXYLIC ACIDS CNCEPT: CARBXYLIC ACID NMENCLATURE IUPAC: Replace alkane -e with Substituents are located using
RGANIC - BRWN 8E CH. 17 - CARBXYLIC ACIDS!! www.clutchprep.com RGANIC - BRWN 8E CH. 17 - CARBXYLIC ACIDS CNCEPT: CARBXYLIC ACID NMENCLATURE IUPAC: Replace alkane -e with Substituents are located using
EXAM #3 EXTRA PROBLEMS
 Massachusetts Institute of Technology 5.13, Fall 2006 Dr. Kimberly L. Berkowski rganic chemistry II EXAM #3 EXTRA PRBLEMS What to expect on Exam #3: 1. ~1 Labeling experiment 2. ~2 chanisms 3. ~2 Syntheses
Massachusetts Institute of Technology 5.13, Fall 2006 Dr. Kimberly L. Berkowski rganic chemistry II EXAM #3 EXTRA PRBLEMS What to expect on Exam #3: 1. ~1 Labeling experiment 2. ~2 chanisms 3. ~2 Syntheses
Chemistry 234 Exam 3. The Periodic Table
 ame: Last First MI Chemistry 234 Exam 3 Fall 2017 Dr. J. sbourn Instructions: The first 24 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
ame: Last First MI Chemistry 234 Exam 3 Fall 2017 Dr. J. sbourn Instructions: The first 24 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
Name. Department of Chemistry SUNY/Oneonta. Chem Organic Chemistry II Examination #3 - March 31, 2003
 INSTRUTINS Name Department of hemistry SUNY/neonta hem 322 - rganic hemistry II Examination #3 - March 31, 2003 This examination has two parts. Part I is in multiple choice format and the answers should
INSTRUTINS Name Department of hemistry SUNY/neonta hem 322 - rganic hemistry II Examination #3 - March 31, 2003 This examination has two parts. Part I is in multiple choice format and the answers should
Name: Date: I. Multiple Choice (Circle the letter for the best answer)
 Organic Chemistry II Sample Exam 3 You should also be able to name compounds, draw structures from names, and complete reactions given the reactants and conditions of the reaction. Name: Date: I. Multiple
Organic Chemistry II Sample Exam 3 You should also be able to name compounds, draw structures from names, and complete reactions given the reactants and conditions of the reaction. Name: Date: I. Multiple
Organic Chemistry FINAL EXAM A (250 points)
 UCSC, Binder ame Student ID # Section Day/Time rganic Chemistry FIAL EXAM A (250 points) D T BEGI TE EXAM R TUR TE PAGE UTIL ISTRUCTED T D S. In the meantime, please read the instructions below. In each
UCSC, Binder ame Student ID # Section Day/Time rganic Chemistry FIAL EXAM A (250 points) D T BEGI TE EXAM R TUR TE PAGE UTIL ISTRUCTED T D S. In the meantime, please read the instructions below. In each
C h a p t e r T w e n t y : Carboxylic Acids & Their Derivatives
 C h a p t e r T w e n t y : Carboxylic Acids & Their Derivatives N C3 N Lysergic acid, the depressant used to make LSD, the famous acid of the 1960s LSD and the Search for God, a San Francisco-based band
C h a p t e r T w e n t y : Carboxylic Acids & Their Derivatives N C3 N Lysergic acid, the depressant used to make LSD, the famous acid of the 1960s LSD and the Search for God, a San Francisco-based band
Chapter 10: Carboxylic Acids and Their Derivatives
 Chapter 10: Carboxylic Acids and Their Derivatives The back of the white willow tree (Salix alba) is a source of salicylic acid which is used to make aspirin (acetylsalicylic acid) The functional group
Chapter 10: Carboxylic Acids and Their Derivatives The back of the white willow tree (Salix alba) is a source of salicylic acid which is used to make aspirin (acetylsalicylic acid) The functional group
21.1 Introduction Carboxylic Acids Nomenclature of Carboxylic Acids. Acids Structure and Properties of Carboxylic Acids.
 21.1 Introduction Carboxylic Acids Carboxylic acids are abundant in nature and in pharmaceuticals. 21.1 Introduction Carboxylic Acids The US produces over 2.5 million tons of acetic acid per year, which
21.1 Introduction Carboxylic Acids Carboxylic acids are abundant in nature and in pharmaceuticals. 21.1 Introduction Carboxylic Acids The US produces over 2.5 million tons of acetic acid per year, which
Loudon Chapter 20 & 21 Review: Carboxylic Acids & Derivatives CHEM 3331, Jacquie Richardson, Fall Page 1
 Loudon Chapter 20 & 21 eview: Carboxylic Acids & Derivatives CEM 3331, Jacquie ichardson, Fall 2010 - Page 1 These two chapters cover compounds which are all at the three bonds to more electronegative
Loudon Chapter 20 & 21 eview: Carboxylic Acids & Derivatives CEM 3331, Jacquie ichardson, Fall 2010 - Page 1 These two chapters cover compounds which are all at the three bonds to more electronegative
Chemistry 52 Exam #2. Name: 5 February This exam has six (6) questions, two cover pages, six exam pages, and three scratch pages.
 Chemistry 52 Exam #2 Name: 5 February 2003 This exam has six (6) questions, two cover pages, six exam pages, and three scratch pages. Please check before beginning to make sure no questions are missing.
Chemistry 52 Exam #2 Name: 5 February 2003 This exam has six (6) questions, two cover pages, six exam pages, and three scratch pages. Please check before beginning to make sure no questions are missing.
State University of New York at Stony Brook Department of Chemistry. CHE 322, Organic Chemistry II Exam II March 17, 2004 Form 1
 State University of New York at Stony Brook Department of Chemistry CHE 22, rganic Chemistry II Exam II March 17, 2004 Form 1 Please answer all questions specifically, concisely, and readably in the spaces
State University of New York at Stony Brook Department of Chemistry CHE 22, rganic Chemistry II Exam II March 17, 2004 Form 1 Please answer all questions specifically, concisely, and readably in the spaces
Carboxylic Acids O R C + H + O - Chemistry 618B
 arboxylic Acids R H R + H + - R - Nomenclature - IUPA IUPA names: drop the -e from the parent alkane and add the suffix -oic acid If the compound contains a carbon-carbon double bond, change the infix
arboxylic Acids R H R + H + - R - Nomenclature - IUPA IUPA names: drop the -e from the parent alkane and add the suffix -oic acid If the compound contains a carbon-carbon double bond, change the infix
CHEM Chapter 21. Carboxylic Acid Derivatives_Nucleophilic Acyl Substitution Reactions (homework) W
 Short Answer IUPAC Naming Instructions: Provide proper IUPAC names. 1. Name: 2. Name: 3. Name: Drawing Instructions: Draw structures corresponding to each of the given names. 4. Draw: acetic formic anhydride
Short Answer IUPAC Naming Instructions: Provide proper IUPAC names. 1. Name: 2. Name: 3. Name: Drawing Instructions: Draw structures corresponding to each of the given names. 4. Draw: acetic formic anhydride
The Claisen Condensation
 Lecture 22 The Claisen Condensation CH 3 CCH 2 CH 3 CH 2 CCH 2 CH 3 April 10, 2018 Hydrolysis of Amides Hydrolysis of amides is irreversible. In acid solution the amine product is protonated to give an
Lecture 22 The Claisen Condensation CH 3 CCH 2 CH 3 CH 2 CCH 2 CH 3 April 10, 2018 Hydrolysis of Amides Hydrolysis of amides is irreversible. In acid solution the amine product is protonated to give an
CHAPTER 22 HW: CO 2 H DERIVATIVES
 CHAPTER 22 HW: C 2 H DERIVATIVES MECLATURE 1. Give the name for each compound (IUPAC or common name). Use R/S naming where needed. Structure Br ame Structure ame 2. Give the name for each ester. Structure
CHAPTER 22 HW: C 2 H DERIVATIVES MECLATURE 1. Give the name for each compound (IUPAC or common name). Use R/S naming where needed. Structure Br ame Structure ame 2. Give the name for each ester. Structure
Ch 22 Carbonyl Alpha ( ) Substitution
 Ch 22 Carbonyl Alpha () Substitution The overall reaction replaces an H with an E + The acid-catalyzed reaction has an enol intermediate The base-catalyzed reaction has an enolate intermediate Keto-Enol
Ch 22 Carbonyl Alpha () Substitution The overall reaction replaces an H with an E + The acid-catalyzed reaction has an enol intermediate The base-catalyzed reaction has an enolate intermediate Keto-Enol
CHEM 347 Organic Chemistry II (for Majors) Instructor: Paul J. Bracher. Quiz # 4. Due in Monsanto Hall 103 by: Friday, April 4 th, 2014, 7:00 p.m.
 CHEM 347 Quiz # 4 Spring 2014 Page 1 of 9 CHEM 347 Organic Chemistry II (for Majors) Instructor: Paul J. Bracher Quiz # 4 Due in Monsanto Hall 103 by: Friday, April 4 th, 2014, 7:00 p.m. Student Name (Printed)
CHEM 347 Quiz # 4 Spring 2014 Page 1 of 9 CHEM 347 Organic Chemistry II (for Majors) Instructor: Paul J. Bracher Quiz # 4 Due in Monsanto Hall 103 by: Friday, April 4 th, 2014, 7:00 p.m. Student Name (Printed)
CHEM J-8 June Complete the following table. Make sure you give the name of the starting material where indicated. REAGENTS/ CONDITIONS
 CHEM1102 2014-J-8 June 2014 Complete the following table. Make sure you give the name of the starting material where indicated. STARTING MATERIAL REAGENTS/ CNDITINS STRUCTURAL FRMULA(S) F MAJR RGANIC PRDUCT(S)
CHEM1102 2014-J-8 June 2014 Complete the following table. Make sure you give the name of the starting material where indicated. STARTING MATERIAL REAGENTS/ CNDITINS STRUCTURAL FRMULA(S) F MAJR RGANIC PRDUCT(S)
CH 320/328 N Summer II 2018
 CH 320/328 N Summer II 2018 HW 1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. (5 pts each) 1. Which
CH 320/328 N Summer II 2018 HW 1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. (5 pts each) 1. Which
SYSTEMWIDE CHEM 2425 FINAL EXAM. Department Of Physical Sciences
 SYSTEMWIDE CHEM 2425 FINAL EXAM Department f Physical Sciences Morphine NAME: RGANIC CHEM 2425 FINAL EXAM DIRECTINS- A periodic table is attached at the end of this exam. Please answer all questions in
SYSTEMWIDE CHEM 2425 FINAL EXAM Department f Physical Sciences Morphine NAME: RGANIC CHEM 2425 FINAL EXAM DIRECTINS- A periodic table is attached at the end of this exam. Please answer all questions in
Note: You must have your answers written in pen if you want a regrade!!!!
 NAME (Print): SIGNATURE: Chemistry 310N Dr. Brent Iverson 2nd Midterm March 27, 2008 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit long,
NAME (Print): SIGNATURE: Chemistry 310N Dr. Brent Iverson 2nd Midterm March 27, 2008 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit long,
Organic Chemistry FINAL EXAM A (250 points)
 UCSC, Binder ame Student ID # Section Day/Time rganic Chemistry FIAL EXAM A (250 points) D T BEGI TE EXAM R TUR TE PAGE UTIL ISTRUCTED T D S. In the meantime, please read the instructions below. In each
UCSC, Binder ame Student ID # Section Day/Time rganic Chemistry FIAL EXAM A (250 points) D T BEGI TE EXAM R TUR TE PAGE UTIL ISTRUCTED T D S. In the meantime, please read the instructions below. In each
Physical Properties. Alcohols can be: CH CH 2 OH CH 2 CH 3 C OH CH 3. Secondary alcohol. Primary alcohol. Tertiary alcohol
 Chapter 10: Structure and Synthesis of Alcohols 100 Physical Properties Alcohols can be: CH 3 CH 3 CH CH 2 OH * Primary alcohol CH 3 OH CH * CH 2 CH 3 Secondary alcohol CH 3 CH 3 * C OH CH 3 Tertiary alcohol
Chapter 10: Structure and Synthesis of Alcohols 100 Physical Properties Alcohols can be: CH 3 CH 3 CH CH 2 OH * Primary alcohol CH 3 OH CH * CH 2 CH 3 Secondary alcohol CH 3 CH 3 * C OH CH 3 Tertiary alcohol
Chapter 20 Carboxylic Acid Derivatives. Nucleophilic Acyl Substitution
 ucleophilic Acyl Substitution hapter 20 arboxylic Acid Derivatives ucleophilic Acyl Substitution Y (1) need to have Y as a u Y u u + Y (2) could not happen with aldehydes or ketones as : and : are poor
ucleophilic Acyl Substitution hapter 20 arboxylic Acid Derivatives ucleophilic Acyl Substitution Y (1) need to have Y as a u Y u u + Y (2) could not happen with aldehydes or ketones as : and : are poor
i. NaCN ii. 70% H 2 SO 4, reflux
 CHE 325 CARBXYLIC ACID CHAP 18 AIGN 1. In which of the following sequences are the compounds listed in order of decreasing acidity? A. CH 3CH > H 2 > CH 3CH 2H > HC CH > NH 3 B. CH 3CH 2H > CH 3CH > H
CHE 325 CARBXYLIC ACID CHAP 18 AIGN 1. In which of the following sequences are the compounds listed in order of decreasing acidity? A. CH 3CH > H 2 > CH 3CH 2H > HC CH > NH 3 B. CH 3CH 2H > CH 3CH > H
CHEM 345 Problem Set 07 Key
 CHEM 345 Problem Set 07 Key 1.) Fill in the appropriate reaction arrow. The starting material is on the left, the product is on the right. Use. Simple Ring Size. 5 and 6 are favored. 3 is not. That s it.
CHEM 345 Problem Set 07 Key 1.) Fill in the appropriate reaction arrow. The starting material is on the left, the product is on the right. Use. Simple Ring Size. 5 and 6 are favored. 3 is not. That s it.
Chemistry 233 Exam 3. The Periodic Table
 Name: Last First MI Chemistry 233 Exam 3 Fall 2017 Dr. J. sbourn Instructions: The first 8 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
Name: Last First MI Chemistry 233 Exam 3 Fall 2017 Dr. J. sbourn Instructions: The first 8 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
CHEM1902/ N-8 November Consider the following reaction sequences beginning with the carboxylic acid, E.
 CHEM1902/4 2014-N-8 November 2014 Consider the following reaction sequences beginning with the carboxylic acid, E. 6 Name compounds E and G. E: G: Propose structures for compounds F, H and J. F H J Propose
CHEM1902/4 2014-N-8 November 2014 Consider the following reaction sequences beginning with the carboxylic acid, E. 6 Name compounds E and G. E: G: Propose structures for compounds F, H and J. F H J Propose
Hour Examination # 3
 CHEM 343 Principles of Organic Chemistry II Summer 2014 Exam # 3 Problem Booklet Page 1 of 8 CHEM 343 Principles of Organic Chemistry II Summer 2014 Instructor: Paul J. Bracher Hour Examination # 3 Tuesday,
CHEM 343 Principles of Organic Chemistry II Summer 2014 Exam # 3 Problem Booklet Page 1 of 8 CHEM 343 Principles of Organic Chemistry II Summer 2014 Instructor: Paul J. Bracher Hour Examination # 3 Tuesday,
1. What is the major organic product obtained from the following sequence of reactions?
 CH320 N N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. Carefully record your answers on the Scantron
CH320 N N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. Carefully record your answers on the Scantron
22.7 Reactions of Amines: A Review and a Preview
 22.7 Reactions of Amines: A Review and a Preview Preparation of Amines Two questions to answer: 1) How is the C N C N bond to be formed? 2) How do we obtain the correct oxidation state of nitrogen (and
22.7 Reactions of Amines: A Review and a Preview Preparation of Amines Two questions to answer: 1) How is the C N C N bond to be formed? 2) How do we obtain the correct oxidation state of nitrogen (and
COURSE OBJECTIVES / OUTCOMES / COMPETENCIES.
 COURSE OBJECTIVES / OUTCOMES / COMPETENCIES. By the end of the course, students should be able to do the following: See Test1-4 Objectives/Competencies as listed in the syllabus and on the main course
COURSE OBJECTIVES / OUTCOMES / COMPETENCIES. By the end of the course, students should be able to do the following: See Test1-4 Objectives/Competencies as listed in the syllabus and on the main course
Faculty of Science Mid-Term Examination II. Chem 222/234 Introductory Organic Chemistry I
 Student Name: Student ID #: Faculty of Science Mid-Term Examination II Chem 222/234 Introductory rganic Chemistry I Examiner: Professor James L. Gleason Tuesday, November 10, 2009 Associate Examiner: Professor
Student Name: Student ID #: Faculty of Science Mid-Term Examination II Chem 222/234 Introductory rganic Chemistry I Examiner: Professor James L. Gleason Tuesday, November 10, 2009 Associate Examiner: Professor
N_HW1 N_HW1. 1. What is the purpose of the H 2 O in this sequence?
 N_HW1 N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. 1. What is the purpose of the H 2 O in this
N_HW1 N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. 1. What is the purpose of the H 2 O in this
Chemistry 2050 Introduction to Organic Chemistry Fall Semester 2011 Dr. Rainer Glaser
 Chemistry 2050 Introduction to Organic Chemistry Fall Semester 2011 Dr. Rainer Glaser Examination #4 Practice Edition Carbonyl Compounds and Amines. Wednesday, November 16, 2011, 10 10:50 am Name: Question
Chemistry 2050 Introduction to Organic Chemistry Fall Semester 2011 Dr. Rainer Glaser Examination #4 Practice Edition Carbonyl Compounds and Amines. Wednesday, November 16, 2011, 10 10:50 am Name: Question
Alpha Substitution and Condensations of Enols and Enolate Ions. Alpha Substitution
 Alpha Substitution and ondensations of Enols and Enolate Ions hap 23 W: 27, 28, 30, 31, 37, 39, 42-44, 47, 51, 54-56 Alpha Substitution Replacement of a hydrogen on the carbon adjacent to the carbonyl,
Alpha Substitution and ondensations of Enols and Enolate Ions hap 23 W: 27, 28, 30, 31, 37, 39, 42-44, 47, 51, 54-56 Alpha Substitution Replacement of a hydrogen on the carbon adjacent to the carbonyl,
(1) Recall the different types of intermolecular interactions. (2) Look at the structure and determine the correct answer.
 MCAT rganic Chemistry - Problem Drill 11: Carboxylic Acids Question No. 1 of 10 Question 1. What kinds of interactions are holding together the carboxylic acid dimer shown? Question #01 3 C C 3 (A) Van
MCAT rganic Chemistry - Problem Drill 11: Carboxylic Acids Question No. 1 of 10 Question 1. What kinds of interactions are holding together the carboxylic acid dimer shown? Question #01 3 C C 3 (A) Van
Chemistry 2030 Introduction to Organic Chemistry Fall Semester 2012 Dr. Rainer Glaser
 Chemistry 2030 Introduction to Organic Chemistry Fall Semester 2012 Dr. Rainer Glaser Examination #4 Carbonyl Compounds and Amines. Thursday, November 15, 2012, 8:25 9:15 am Name: Question 1. Aldehydes
Chemistry 2030 Introduction to Organic Chemistry Fall Semester 2012 Dr. Rainer Glaser Examination #4 Carbonyl Compounds and Amines. Thursday, November 15, 2012, 8:25 9:15 am Name: Question 1. Aldehydes
Michael and Aldol CH391 December 4, 2002
 Michael and Aldol H391 December 4, 2002 RH 2 H pk a = 16-20 + H RHH + Enolate anions... So.a basic solution contains comparable amounts of the aldehyde and its enolate. Which gives rise to..the Aldol ondensation
Michael and Aldol H391 December 4, 2002 RH 2 H pk a = 16-20 + H RHH + Enolate anions... So.a basic solution contains comparable amounts of the aldehyde and its enolate. Which gives rise to..the Aldol ondensation
Carboxylic Acids and Nitriles
 Carboxylic Acids and Nitriles Why this Chapter? Carboxylic acids present in many industrial processes and most biological processes They are the starting materials from which other acyl derivatives are
Carboxylic Acids and Nitriles Why this Chapter? Carboxylic acids present in many industrial processes and most biological processes They are the starting materials from which other acyl derivatives are
Hour Examination # 2
 CHEM 343 Principles of Organic Chemistry II Summer 2014 Exam # 2 Solutions Key Page 1 of 7 CHEM 343 Principles of Organic Chemistry II Summer 2014 Instructor: Paul J. Bracher Hour Examination # 2 Tuesday,
CHEM 343 Principles of Organic Chemistry II Summer 2014 Exam # 2 Solutions Key Page 1 of 7 CHEM 343 Principles of Organic Chemistry II Summer 2014 Instructor: Paul J. Bracher Hour Examination # 2 Tuesday,
21.1 Introduction Carboxylic Acids
 21.1 Introduction Carboxylic Acids Carboxylic acids are abundant in nature and in pharmaceuticals. Klein, Organic Chemistry 1e 21-1 The US produces over 2.5 million tons of acetic acid per year, which
21.1 Introduction Carboxylic Acids Carboxylic acids are abundant in nature and in pharmaceuticals. Klein, Organic Chemistry 1e 21-1 The US produces over 2.5 million tons of acetic acid per year, which
Chem 22 Final Exam Practice
 Chem 22 Final Exam Practice Questions taken from regular tests given during the previous semesters. Only one answer is correct unless the question says otherwise. The questions are somewhat scrambled with
Chem 22 Final Exam Practice Questions taken from regular tests given during the previous semesters. Only one answer is correct unless the question says otherwise. The questions are somewhat scrambled with
Dr. Mohamed El-Newehy
 By Dr. Mohamed El-Newehy Chemistry Department, College of Science, King Saud University http://fac.ksu.edu.sa/melnewehy Carboxylic acids and Their Derivatives 1 Structure of Carboxylic Acids -The functional
By Dr. Mohamed El-Newehy Chemistry Department, College of Science, King Saud University http://fac.ksu.edu.sa/melnewehy Carboxylic acids and Their Derivatives 1 Structure of Carboxylic Acids -The functional
Final Exam. Chem 3B, Fall 2016 Monday, Dec 12, pm. Name Answer Key. Student ID. If you are making up an incomplete, list the semester here:
 Final Exam Chem 3B, Fall 2016 Monday, Dec 12, 2016 3-6 pm ame Answer Key Student ID If you are making up an incomplete, list the semester here: You have 180 minutes to complete this exam. Please provide
Final Exam Chem 3B, Fall 2016 Monday, Dec 12, 2016 3-6 pm ame Answer Key Student ID If you are making up an incomplete, list the semester here: You have 180 minutes to complete this exam. Please provide
Synthesis of Nitriles a. dehydration of 1 amides using POCl 3 : b. SN2 reaction of cyanide ion on halides:
 I. Nitriles Nitriles consist of the CN functional group, and are linear with sp hybridization on C and N. Nitriles are non-basic at nitrogen, since the lone pair exists in an sp orbital (50% s character
I. Nitriles Nitriles consist of the CN functional group, and are linear with sp hybridization on C and N. Nitriles are non-basic at nitrogen, since the lone pair exists in an sp orbital (50% s character
1/4/2011. Chapter 18 Aldehydes and Ketones Reaction at the -carbon of carbonyl compounds
 Chapter 18 Aldehydes and Ketones Reaction at the -carbon of carbonyl compounds The Acidity of the Hydrogens of Carbonyl Compounds: Enolate Anions Hydrogens on carbons to carbonyls are unusually acidic
Chapter 18 Aldehydes and Ketones Reaction at the -carbon of carbonyl compounds The Acidity of the Hydrogens of Carbonyl Compounds: Enolate Anions Hydrogens on carbons to carbonyls are unusually acidic
Sametz: CHEM 322 Spring 2012 Organic Chemistry Final
 Name: (Print your name clearly!) Sametz: CHEM 322 Spring 2012 rganic Chemistry Final All answers should be written CLEARLY in the space provided. (If it s not clear, it s wrong). FR QUESTINS 9-10: read
Name: (Print your name clearly!) Sametz: CHEM 322 Spring 2012 rganic Chemistry Final All answers should be written CLEARLY in the space provided. (If it s not clear, it s wrong). FR QUESTINS 9-10: read
Chapter 12: Carbonyl Compounds II
 Chapter 12: Carbonyl Compounds II Learning bjectives: 1. Recognize and assign names to aldehydes and ketones. 2. Write the mechanism for nucleophilic addition and nucleophilic addition-elimination reactions
Chapter 12: Carbonyl Compounds II Learning bjectives: 1. Recognize and assign names to aldehydes and ketones. 2. Write the mechanism for nucleophilic addition and nucleophilic addition-elimination reactions
Chapter 20: Aldehydes and Ketones
 hem A225 Notes Page 67 I. Introduction hapter 20: Aldehydes and Ketones Aldehydes and ketones contain a carbonyl group (=) with no other heteroatoms attached. An aldehyde has at least one hydrogen attached;
hem A225 Notes Page 67 I. Introduction hapter 20: Aldehydes and Ketones Aldehydes and ketones contain a carbonyl group (=) with no other heteroatoms attached. An aldehyde has at least one hydrogen attached;
Nomenclature of Aldehydes and Ketones
 Aldehydes and Ketones Nomenclature of Aldehydes and Ketones Common aldehydes H H Methanl (formaldehyde) H3C H CH3CH2 ethanl (acetaldehyde) H propanal (propionaldehyde) CH3CH2CH2 butanal (n-butyraldehyde)
Aldehydes and Ketones Nomenclature of Aldehydes and Ketones Common aldehydes H H Methanl (formaldehyde) H3C H CH3CH2 ethanl (acetaldehyde) H propanal (propionaldehyde) CH3CH2CH2 butanal (n-butyraldehyde)
c. Oxidizing agent shown here oxidizes 2º alcohols to ketones and 1º alcohols to carboxylic acids. 3º alcohols DO NOT REACT.
 Exam 1 (Ch 17 and Review of CEM 331) Answer Key: 1. ne-step Questions: You need to know reagents for reagent arrows and to be able to draw products. I know a lot of them seem to look alike its your job
Exam 1 (Ch 17 and Review of CEM 331) Answer Key: 1. ne-step Questions: You need to know reagents for reagent arrows and to be able to draw products. I know a lot of them seem to look alike its your job
The Organic Acids. Carboxylic Acids * *
 arboxylic Acids The rganic Acids Some Notation: Acids and their conjugate bases pka ~ 15 - weak acid arboxylic Acid pka ~ 4 moderate acid - arboxylate Anion pka ~ -7 very strong acid l - l arboxylic acids
arboxylic Acids The rganic Acids Some Notation: Acids and their conjugate bases pka ~ 15 - weak acid arboxylic Acid pka ~ 4 moderate acid - arboxylate Anion pka ~ -7 very strong acid l - l arboxylic acids
Chemistry 2050 Introduction to Organic Chemistry Fall Semester 2011 Dr. Rainer Glaser
 Chemistry 2050 Introduction to Organic Chemistry Fall Semester 2011 Dr. Rainer Glaser Examination #4 Make-Up Carbonyl Compounds and Amines. Wednesday, November 30, 2011, 10 10:50 am Name: Answer Key Question
Chemistry 2050 Introduction to Organic Chemistry Fall Semester 2011 Dr. Rainer Glaser Examination #4 Make-Up Carbonyl Compounds and Amines. Wednesday, November 30, 2011, 10 10:50 am Name: Answer Key Question
Definition: A carboxylic acid derivative undergoes hydrolysis (bond breaking with water) to form a carboxylic acid R C N + H 2 O + H O R
 arboxylic Acid Derivatives Addition/Elimination Definition: A carboxylic acid derivative undergoes hydrolysis (bond breaking with water) to form a carboxylic acid X = l, Br addition/elimination X acid
arboxylic Acid Derivatives Addition/Elimination Definition: A carboxylic acid derivative undergoes hydrolysis (bond breaking with water) to form a carboxylic acid X = l, Br addition/elimination X acid
DAMIETTA UNIVERSITY CHEM-103: BASIC ORGANIC CHEMISTRY LECTURE
 DAMIETTA UNIVERSITY CHEM-103: BASIC ORGANIC CHEMISTRY LECTURE 6 Dr Ali El-Agamey 1 Oxidation States Easy for inorganic salts: CrO 4 2- reduced to Cr 2 O 3. KMnO 4 reduced to MnO 2. Oxidation: Gain of O,
DAMIETTA UNIVERSITY CHEM-103: BASIC ORGANIC CHEMISTRY LECTURE 6 Dr Ali El-Agamey 1 Oxidation States Easy for inorganic salts: CrO 4 2- reduced to Cr 2 O 3. KMnO 4 reduced to MnO 2. Oxidation: Gain of O,
CARBOXYLIC ACIDS AND THEIR DERIVATIVES
 ARBXYLI AIDS AND TEIR DERIVATIVES A STUDENT SULD BE ABLE T: 1. Give the IUPA name given the structure, and draw the structure given the name, of carboxylic acids and their metal salts, acyl chlorides,
ARBXYLI AIDS AND TEIR DERIVATIVES A STUDENT SULD BE ABLE T: 1. Give the IUPA name given the structure, and draw the structure given the name, of carboxylic acids and their metal salts, acyl chlorides,
CARBOXYLIC ACIDS AND THEIR DERIVATIVES. 3. Predict the relative acidity and basicity of compounds and ions. Important criteria include:
 CARBXYLIC ACIDS AND TEIR DERIVATIVES A STUDENT SULD BE ABLE T: 1. Give the IUPAC name given the structure, and draw the structure given the name, of carboxylic acids and their metal salts, acyl chlorides,
CARBXYLIC ACIDS AND TEIR DERIVATIVES A STUDENT SULD BE ABLE T: 1. Give the IUPAC name given the structure, and draw the structure given the name, of carboxylic acids and their metal salts, acyl chlorides,
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group
 Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Hour Examination # 2
 CHEM 347 Hour Examination # 2 Spring 2014 Page 1 of 8 CHEM 347 rganic Chemistry II (for Majors) Instructor: Paul J. Bracher Hour Examination # 2 Wednesday, March 5 th, 2014 5:30 8:30 p.m. Student Name
CHEM 347 Hour Examination # 2 Spring 2014 Page 1 of 8 CHEM 347 rganic Chemistry II (for Majors) Instructor: Paul J. Bracher Hour Examination # 2 Wednesday, March 5 th, 2014 5:30 8:30 p.m. Student Name
CHEM 203. Final Exam December 15, 2010 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
Chapter 20 Carboxylic Acid Derivatives Nucleophilic Acyl Substitution
 Chapter 20 Carboxylic Acid Derivatives Nucleophilic Acyl Substitution Nomenclature: In carboxylic acid chlorides, anhydrides, esters and amides, the parent is the carboxylic acid. In each case be sure
Chapter 20 Carboxylic Acid Derivatives Nucleophilic Acyl Substitution Nomenclature: In carboxylic acid chlorides, anhydrides, esters and amides, the parent is the carboxylic acid. In each case be sure
Chapter 19 Substitutions at the Carbonyl Group
 Chapter 19 Substitutions at the Carbonyl Group In Chapter 18 Additions to the Carbonyl Groups In Chapter 19 Substitutions at the Carbonyl Group O O - - O - O R Y R C+ Y R Y Nu -Ȳ R N u + Y=goodleavinggroup
Chapter 19 Substitutions at the Carbonyl Group In Chapter 18 Additions to the Carbonyl Groups In Chapter 19 Substitutions at the Carbonyl Group O O - - O - O R Y R C+ Y R Y Nu -Ȳ R N u + Y=goodleavinggroup
ROADMAP FOR REACTIONS Chapter 6
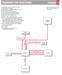 RADMAP FR REACTINS Chapter 6 (A) (B) (C) (D) (E) (F) (G) (H) (I) (J) (K) (L) (M) (N) () Regiochemistry: Markovnikov addition to a bond Stereochemistry: anti-addition Regiochemistry: non-markovnikov addition
RADMAP FR REACTINS Chapter 6 (A) (B) (C) (D) (E) (F) (G) (H) (I) (J) (K) (L) (M) (N) () Regiochemistry: Markovnikov addition to a bond Stereochemistry: anti-addition Regiochemistry: non-markovnikov addition
Exam 3 Professor R. Hoenigman
 I pledge to uphold the CU onor Code: CEM 3331-100 Spring 2008 Exam 3 Professor R. oenigman igh = 102 Low = 15 Average = 68 Signature ame (printed) Last four digits of your student ID number Recitation
I pledge to uphold the CU onor Code: CEM 3331-100 Spring 2008 Exam 3 Professor R. oenigman igh = 102 Low = 15 Average = 68 Signature ame (printed) Last four digits of your student ID number Recitation
ORGANIC - BRUICE 8E CH CARBONYL COMPOUNDS III: REACTIONS AT THE ALPHA-CARBON
 !! www.clutchprep.com CNCEPT: ALPHA CARBNS AND TAUTMERIZATIN We have discussed the high reactivity of the carbonyl carbon. However, carbonyls contain another highly reactive component. What is the acidity
!! www.clutchprep.com CNCEPT: ALPHA CARBNS AND TAUTMERIZATIN We have discussed the high reactivity of the carbonyl carbon. However, carbonyls contain another highly reactive component. What is the acidity
Chapter 19. Synthesis and Reactions of b-dicarbonyl Compounds: More Chemistry of Enolate Anions. ß-dicarbonyl compounds. Why are ß-dicarbonyls useful?
 Chapter 19 Synthesis and Reactions of b-dicarbonyl Compounds: More Chemistry of Enolate Anions ß-dicarbonyl compounds Two carbonyl groups separated by a carbon Three common types ß-diketone ß-ketoester
Chapter 19 Synthesis and Reactions of b-dicarbonyl Compounds: More Chemistry of Enolate Anions ß-dicarbonyl compounds Two carbonyl groups separated by a carbon Three common types ß-diketone ß-ketoester
UNIVERSITY OF NATAL DURBAN EXAMINATIONS : NOVEMBER 2001 ORGANIC CHEMISTRY FOR CHEMICAL ENGINEERS DSC 2OE2. Time : 2 Hours Total Marks : 100
 UNIVERSITY F NATAL DURBAN EXAMINATINS : NVEMBER 2001 RGANIC CHEMISTRY FR CHEMICAL ENGINEERS DSC 2E2 Time : 2 Hours Total Marks : 100 INTERNAL EXAMINERS : Professor H C Brookes Dr N Koorbanally EXTERNAL
UNIVERSITY F NATAL DURBAN EXAMINATINS : NVEMBER 2001 RGANIC CHEMISTRY FR CHEMICAL ENGINEERS DSC 2E2 Time : 2 Hours Total Marks : 100 INTERNAL EXAMINERS : Professor H C Brookes Dr N Koorbanally EXTERNAL
Carbonyl Chemistry. aldehydes ketones. carboxylic acid and derivatives. Wednesday, April 29, 2009
 Carbonyl Chemistry X aldehydes ketones carboxylic acid and derivatives Electrophiles (eg. + ) Nucleophiles (eg. C 3 MgBr) an enolate Base β β β α α α 1 2 3 4 Nuc- 1 2 Nuc 3 4 1,2-addition 1 2 3 4 Nuc-
Carbonyl Chemistry X aldehydes ketones carboxylic acid and derivatives Electrophiles (eg. + ) Nucleophiles (eg. C 3 MgBr) an enolate Base β β β α α α 1 2 3 4 Nuc- 1 2 Nuc 3 4 1,2-addition 1 2 3 4 Nuc-
CH310N Spring Anslyn. March 23, Exam 2
 C310N Spring 2010 Anslyn March 23, 2010 Exam 2 Please PRINT the first three letters of your last name in the boxes below. PRINT Full Name UT-EID 1) ( 8 pts ) 2) ( 5 pts ) 3) ( 5 pts ) 4) ( 22 pts ) 5)
C310N Spring 2010 Anslyn March 23, 2010 Exam 2 Please PRINT the first three letters of your last name in the boxes below. PRINT Full Name UT-EID 1) ( 8 pts ) 2) ( 5 pts ) 3) ( 5 pts ) 4) ( 22 pts ) 5)
Chem 263 March 28, 2006
 Chem 263 March 28, 2006 Properties of Carboxylic Acids Since carboxylic acids are structurally related to both ketones and aldehydes, we would expect to see some similar structural properties. The carbonyl
Chem 263 March 28, 2006 Properties of Carboxylic Acids Since carboxylic acids are structurally related to both ketones and aldehydes, we would expect to see some similar structural properties. The carbonyl
You must have your answers written in PERMANENT ink if you want a regrade!!!! This means no test written in pencil or ERASABLE INK will be regraded.
 NAME (Print): SIGNATURE: hemistry 310N Dr. Brent Iverson 2nd Midterm March 25, 2010 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit long, but
NAME (Print): SIGNATURE: hemistry 310N Dr. Brent Iverson 2nd Midterm March 25, 2010 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit long, but
Note: You must have your answers written in pen if you want a regrade!!!!
 AME (Print): SIGATURE: hemistry 310 Dr. Brent Iverson 2nd Midterm March 29, 2007 Please print the first three letters of your last name in the three boxes Please ote: This test may be a bit long, but there
AME (Print): SIGATURE: hemistry 310 Dr. Brent Iverson 2nd Midterm March 29, 2007 Please print the first three letters of your last name in the three boxes Please ote: This test may be a bit long, but there
Chemistry 233 Exam 3 (Green) The Periodic Table
 Name: Last First MI Chemistry Exam (Green) Summer 7 Dr. J. sbourn Instructions: The first questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
Name: Last First MI Chemistry Exam (Green) Summer 7 Dr. J. sbourn Instructions: The first questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
MCAT Organic Chemistry Problem Drill 10: Aldehydes and Ketones
 MCAT rganic Chemistry Problem Drill 10: Aldehydes and Ketones Question No. 1 of 10 Question 1. Which of the following is not a physical property of aldehydes and ketones? Question #01 (A) Hydrogen bonding
MCAT rganic Chemistry Problem Drill 10: Aldehydes and Ketones Question No. 1 of 10 Question 1. Which of the following is not a physical property of aldehydes and ketones? Question #01 (A) Hydrogen bonding
Chemistry 14D Winter 2017 Final Exam Part A Page 1
 Chemistry 14D Winter 2017 Final Exam Part A Page 1 Please use the backs of the exam pages for scratch space. Please do not use the exam margins for this purpose. All reactants and solvents written above
Chemistry 14D Winter 2017 Final Exam Part A Page 1 Please use the backs of the exam pages for scratch space. Please do not use the exam margins for this purpose. All reactants and solvents written above
Score: Homework Problem Set 9 Iverson CH320N Due Monday, April 17. NAME (Print): Chemistry 320N Dr. Brent Iverson 9th Homework April 10, 2017
 omework Problem Set 9 Iverson C0N Due Monday, April 7 NAME (Print): SIGNATURE: Chemistry 0N Dr. ent Iverson 9th omework April 0, 07 Please print the first three letters of your last name in the three boxes
omework Problem Set 9 Iverson C0N Due Monday, April 7 NAME (Print): SIGNATURE: Chemistry 0N Dr. ent Iverson 9th omework April 0, 07 Please print the first three letters of your last name in the three boxes
ALDEHYDES AND KETONES
 ALDEHYDES AND KETONES IN WEEK 1, A STUDENT SHOULD BE ABLE TO: 1. Give the IUPAC name given the structure, and draw the structure given the name, of aldehydes and ketones. Also, draw the structure given
ALDEHYDES AND KETONES IN WEEK 1, A STUDENT SHOULD BE ABLE TO: 1. Give the IUPAC name given the structure, and draw the structure given the name, of aldehydes and ketones. Also, draw the structure given
CARBOXYLIC ACIDS and their Derivatives Nucleophilic Acyl substitution - Review the nomenclature for these compounds in your textbook
 CARBXYLIC ACIDS and their Derivatives Nucleophilic Acyl substitution - Review the nomenclature for these compounds in your textbook R Z R Z R Z - the basicity of Z determines the relative stability of
CARBXYLIC ACIDS and their Derivatives Nucleophilic Acyl substitution - Review the nomenclature for these compounds in your textbook R Z R Z R Z - the basicity of Z determines the relative stability of
Sodium Borohydride Reduction of Benzoin
 Sodium Borohydride Reduction of Benzoin Introduction The most common and useful reducing agents for reducing aldehydes, ketones, and other functional groups are metal hydride reagents. The two most common
Sodium Borohydride Reduction of Benzoin Introduction The most common and useful reducing agents for reducing aldehydes, ketones, and other functional groups are metal hydride reagents. The two most common
Chem 2320 Final 210 points Dr. Luther Giddings
 Chem 2320 Final 210 points Dr. Luther Giddings Name Phone or E-Mail Instructions: This is a closed book, closed notebook test. You may not discuss this exam with anyone, either during or after the exam,
Chem 2320 Final 210 points Dr. Luther Giddings Name Phone or E-Mail Instructions: This is a closed book, closed notebook test. You may not discuss this exam with anyone, either during or after the exam,
