CC: Cause and Effect Cause and effect relationships may be used to predict phenomena in natural or designed systems. (MS-PS1-4)
|
|
|
- Donald Francis
- 5 years ago
- Views:
Transcription
1 SCUSD 5E Lesson Plan Lesson Series Title: STATES OF MATTER Unit: STRUCTURE AND PROPERTIES OF MATTER Teacher: Maria (Mafe) Aguilar Number of Days: 6 Subject/Grade Level: 8 th NOTE: This lesson plan is going to be presented at the California Science Teachers Association Conference in October 2017 in Sacramento, CA. Teachers will engage in 1.5 hour workshop to develop the particle arrangement of solids, liquids and gases based on the evidence collected during the How Squishable? lab activity. This simulation activity is another way of visualizing the particles of matter. Previous simulation techniques taught to my students used UC Davis-CSTEM QAnimate program. Content Standards and Understandings (NGSS/CCSS): MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state of a pure substance when thermal energy is added or removed. [Clarification Statement: Emphasis is on qualitative molecular-level models of solids, liquids, and gases to show that adding or removing thermal energy increases or decreases kinetic energy of the particles until a change of state occurs. Examples of models could include drawings and diagrams. Examples of particles could include molecules or inert atoms. Examples of pure substances could include water, carbon dioxide, and helium.] DCI: PS1.A: Structure and Properties of Matter Each pure substance has characteristic physical and chemical properties (for any bulk quantity under given conditions) that can be used to identify it. (MS-PS1-2),(MS-PS1-3) Gases and liquids are made of molecules or inert atoms that are moving about relative to each other. (MS-PS1-4) In a liquid, the molecules are constantly in contact with others; in a gas, they are widely spaced except when they happen to collide. In a solid, atoms are closely spaced and may vibrate in position but do not change relative locations. (MS-PS1-4) Solids may be formed from molecules, or they may be extended structures with repeating subunits (e.g., crystals). (MS-PS1-1) CC: Cause and Effect Cause and effect relationships may be used to predict phenomena in natural or designed systems. (MS-PS1-4) S&E Practices: Developing and Using Models Modeling in 6 8 builds on K 5 and progresses to developing, using and revising models to describe, test, and predict more abstract phenomena and design systems. Develop a model to predict and/or describe phenomena. (MS-PS1-1),(MS-PS1-4) Develop a model to describe unobservable mechanisms. (MS-PS1-5) Common Core State Standards Connections: ELA/Literacy - RST Follow precisely a multistep procedure when carrying out experiments, taking measurements, or performing technical tasks. (MS-PS1-6) RST Integrate quantitative or technical information expressed in words in a text with a version of that information expressed visually (e.g., in a flowchart, diagram, model, graph, or table). (MS-PS1-1),(MS- PS1-2),(MS-PS1-4),(MS-PS1-5) Driving Questions: (This questions will be formulated by the students after the engage activity. Is silly putty or oobleck solid? Differentiation strategies to meet diverse learner needs: Students will work in groups throughout the entire unit lesson. Students will use reading, writing and speaking literacy skills during metacognition to develop the ideas of the Particles of Matter model. Students will engage in hands on and visualization activities to represent unseen objects or particles of matter. Students will also engage in arguing using evidence in developing the particle model of the states of matter.
2 ENGAGEMENT (Day 1) Describe how the teacher will capture students interest. What kind of questions should the students ask themselves after the engagement? Students: 1. Students will make a silly putty or oobleck using simple household materials When done, ask students to make NOTICES.and WONDERINGS by filling out their interactive notebooks. Teacher Notes: When asking the students to engage in a phenomenon, use the questions to redirect phenomena protocol in order to generate related notices and wonderings. What Do You See? Protocol Review the image. Record your answers to the following questions: What do you notice about the image? What phenomenon is the image displaying? What in the image do you find surprising? What questions can you ask about the phenomenon? I Notice I Wonder. When asking the students to formulate questions as wonderings, ask them to complete the sentence stem I wonder in order to generate intuitive questions that are not usually answerable by yes or no. Rather, questions generated using this sentence frame, are how and why questions. From the list of questions, focus on one to investigate, one that is related to the focus DCI: structure and properties of solids, liquids and gases. EXPLORATION (Days 2-3) Describe what hands-on/minds-on activities students will be doing. List big idea conceptual questions the teacher will use to encourage and/or focus students exploration Students: 1. Driving Question: Is oobleck or silly putty solid or liquid? (Write in INB.) Before formulating a claim, uncover students ideas about solids based on what they know about particles of matter. 2. Uncovering Student Ideas: Is it Solid? By Paige Keeley 924/Centricity/ModuleInstance/22013/Is%20It%20a% 20Solid.pdf Consider these questions in formulating your claim: a. Why do solids appear the way they do? b. How do the particle arrangement of matter relate to their properties? c. Solids, liquids, and gases are all made of particles. Are these particles close together? far apart? NOTE: Do not discuss their responses. 3. Formulate a claim Example: An oobleck is a fluid because it doesn t maintain a specific shape. Teacher: Your task is to develop a model that explains the properties of the states of matter and use these ideas to support your claim. Introduce the activity How Squishable? (CSUS SASP 2011) 1. Purpose and expectations 2. Types of data to be obtained 3. Safety precautions Is it solid? 20Cards_SandC.pdf Teacher must have had a separate lesson on how students engage in Claim-Evidence-Reasoning CaseOfMissingMeatballs.pdf
3 4. Perform How Squishable? Follow the procedures as described in the handout. Essential Question: How do the particles of matter relate to their properties? Conceptual questions to be used to encourage and focus students exploration are found in the handout. howsquishable.pdf EXPLANATION (Days 3-4) Student explanations should precede introduction of terms or explanations by the teacher. What questions or techniques will the teacher use to help students connect their exploration to the concept under examination? List higher order thinking questions which teachers will use to solicit student explanations and help them to justify their explanations. Activate prior knowledge: This stage is necessary to develop the reasoning behind the evidence collected in the activity. What do you know about particles from our previous explorations? 1. Particles make up matter. 2. Particles have some stickiness between each other. 3. Particles move. 4. Particles are very tiny. Teacher s notes: You do not need to front load the vocabulary. Particles need not be called atoms or molecules. You can distinguish the times of particles later if your unit plan is in the direction of types of particles in relation to interactions of matter. Stickiness maybe referred to as attraction. Energy of particle motion may be discussed later during state changes. To begin exploring the relationship between the particles of the states of matter and their properties, students will draw how the particles are arranged in each state. If you can zoom into each of the samples of matter, how are their particles arranged? Solids Liquids Gases Teacher guides students to summarize the pieces of evidence from each part of the activity and develop the ideas that describe the properties of solids, liquids and gases. These ideas will comprise the particle model of the states of matter. Evidence: Part 1: Blocked and Unblocked Empty Syringe It was hard to pull the plunger when the nozzle is blocked. The plunger returns to zero volume reading when released. Part 2: Air in Syringe, Nozzle is blocked Plunger can be pushed to a certain volume. Volume decreases as the plunger is pushed down the syringe. Plunger goes back up to original volume when the pushing force is removed. Reasoning: Big Ideas (Describe the behavior of the particles based on the evidence collected.) Part 1: Empty Syringe Few air particles are trapped in the nozzle of the syringe to push the plunger when the nozzle is blocked. If the nozzle is unblocked, more air particles from the surroundings can freely enter the syringe. Since gas particles are moving, they tend to push each other and the surface of the plunger. Part 2: Air in the Syringe, Nozzle is blocked Air particles do not have stickiness. There are wide spaces between air particles. Air particles are compressible. Air particles constantly move.
4 Part 3: Liquid in Syringe, Nozzle is blocked Plunger does not move. Volume stays the same. Liquid flows. Part 4: Solid in Syringe, Nozzle is not blocked Plunger can be pushed only until the surface of the solid substance in the syringe. The solid material kept its shape. Part 3: Liquid in Syringe Liquid particles are as close to each other like the solid particles. Liquid particles have some stickiness.. Part 4: Solid in Syringe, nozzle is not blocked Solid particles are as close to each other like liquid particles. Solids particles have definite arrangement.. Based on these pieces of evidence and reasoning, revisit your drawings before performing How Squishable syringe activity. Re-draw the arrangement of the particles of each state of matter. Solids Liquids Gases ELABORATION Describe how students will develop a more sophisticated understanding of the concept. What vocabulary will be introduced and how will it connect to students observations? How is this knowledge applied in our daily lives? Based on the evidence collected, ask the student to relate their ideas about the behavior of the particles to their observed properties of solids, liquids and gases. 1. Air particles make up gases. They have very little to no stickiness or attraction between each other allowing them to move as far away as possible from each other and take up the space of their container. Since they are not attracted to each other, gases do not have definite volume and shape. 2. Liquid particles have some attraction between each other allowing them to flow together and take up the shape of their container. They have not definite shape. Liquids have definite volume since the particles are as close to each other as possible. 3. Solid particles have very strong attraction between each other. They form definite particle arrangement that allow solids to have definite shape and volume. Revisiting Uncovering Student Ideas: Is it Solid? Discuss students ideas why each item is a solid or not using the ideas developed about particles and states of matter. Use this NSTA article to guide your discussion with your students. Simulate the behavior of the particles of matter through Alice programming. (Days 5-6) Criteria for success: 1. There should be 3 scenes: solids, liquids and gases 2. Each scene should demonstrate the particle arrangement in each state. 3. Each scene should describe the properties of solids, liquids and gases. 4. Extra: each scene should be able to demonstrate the movement of the particles using event listeners.
5 EVALUATION How will students demonstrate that they have achieved the lesson objective? This should be embedded throughout the lesson as well as at the end of the lesson Reading in science: What is an oobleck/silly putty? Based on the evidence collected from the activity and the ideas developed on the particle model of the states of matter, and the ideas from the reading, revisit your claim. Write a complete explanation using the C-E-R template whether an oobleck or silly putty is a solid or not. Materials Required for This Lesson/Activity Quantity Description Potential Supplier (item #) Estimated Price 9x3=27 60-mL syringes (used for oral feeding for newly born animals or in the hospitals) Any farm / feed store; hospital supplier Amazon.com yringe&tag=googhydr- 20&index=aps&hvadid= &hvpos=1t1&hvnetw =g&hvrand= &hvpone=1264&hvptw o=37&hvqmt=e&hvdev=c&hvdvcmdl=&hvlocint=&hvlocphy = &hvtargid=kwd &ref=pd_sl_6vggbjeuhg_e_p20 S5.99/pack of 5 9 x 2 Plastic cups Any grocery stores Hefty Deluxe Clear Plastic Party Cups (9 Ounce, 40 Count) $3.86 per pack 9 1-inch wooden cubes (to fill the 60- ml syringes for solid in syringe) 9 Jumbo sidewalk chalk (used in place of wooden cubes) Amazon.com Chica- Jo/dp/B00J1KQYEQ/ref=sr_1_1?ie=UTF8&qid= &sr=8-1&keywords=i-inch+cubes Jo-Ann s 20pk/ html#q=chalk&start=1 $ 10.99/ pack of 10 $3.99/bucket of 20
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 2, LESSON 1 HEAT, TEMPERATURE, AND CONDUCTION MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state
The Next Generation Science Standards (NGSS) CHAPTER 2, LESSON 1 HEAT, TEMPERATURE, AND CONDUCTION MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 3, LESSON 1: WHAT IS DENSITY? MS-PS1-1. Develop models to describe the atomic composition of simple molecules and extended structures. DISCIPLINARY
The Next Generation Science Standards (NGSS) CHAPTER 3, LESSON 1: WHAT IS DENSITY? MS-PS1-1. Develop models to describe the atomic composition of simple molecules and extended structures. DISCIPLINARY
B L U E V A L L E Y D I S T R I C T CURRI C U L U M Science 8 th grade
 B L U E V A L L E Y D I S T R I C T CURRI C U L U M Science 8 th grade ORGANIZING THEME/TOPIC UNIT 1: ENERGY Definitions of Energy Potential and Kinetic Energy Conservation of Energy Energy Transfer MS-PS3-1:
B L U E V A L L E Y D I S T R I C T CURRI C U L U M Science 8 th grade ORGANIZING THEME/TOPIC UNIT 1: ENERGY Definitions of Energy Potential and Kinetic Energy Conservation of Energy Energy Transfer MS-PS3-1:
6th Grade: Great Salt Lake is Salty
 Curriculum written by Megan Black in partnership with The Great Salt Lake Institute at Westminster College. 6th Grade: Great Salt Lake is Salty Lesson Description: In this lesson students will compare
Curriculum written by Megan Black in partnership with The Great Salt Lake Institute at Westminster College. 6th Grade: Great Salt Lake is Salty Lesson Description: In this lesson students will compare
Performance Expectation MS-PS1-1: Develop models to describe the atomic composition of simple molecules and extended structures.
 CHEMISTRY OF MATERIALS OVERVIEW- NGSS Performance Expectation MS-PS1-1: Develop models to describe the atomic composition of simple molecules and extended structures. NGPEP11 DCI: Substances are made from
CHEMISTRY OF MATERIALS OVERVIEW- NGSS Performance Expectation MS-PS1-1: Develop models to describe the atomic composition of simple molecules and extended structures. NGPEP11 DCI: Substances are made from
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 1, LESSON 1 MOLECULES MATTER MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state of a pure substance
The Next Generation Science Standards (NGSS) CHAPTER 1, LESSON 1 MOLECULES MATTER MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state of a pure substance
Reviewing the Alignment of IPS with NGSS
 Reviewing the Alignment of IPS with NGSS Harold A. Pratt & Robert D. Stair Introductory Physical Science (IPS) was developed long before the release of the Next Generation Science Standards (NGSS); nevertheless,
Reviewing the Alignment of IPS with NGSS Harold A. Pratt & Robert D. Stair Introductory Physical Science (IPS) was developed long before the release of the Next Generation Science Standards (NGSS); nevertheless,
Name Date CUMULATIVE TEST FOR LESSON CLUSTERS 1-4
 Cumulative Test 1 Name Date CUMULATIVE TEST FOR LESSON CLUSTERS 1-4 1. Why can you change ice into water but not into glass? 2. Why can't you see air? 3. Describe the ways in which ice, liquid water, and
Cumulative Test 1 Name Date CUMULATIVE TEST FOR LESSON CLUSTERS 1-4 1. Why can you change ice into water but not into glass? 2. Why can't you see air? 3. Describe the ways in which ice, liquid water, and
What Is The Matter? Matter Concept Map
 What Is The Matter? Matter Concept Map Phases of Matter Solid Liquid Gas Use the selections below to complete the concept map for the phases of matter. Write the letter of each characteristic in the appropriate
What Is The Matter? Matter Concept Map Phases of Matter Solid Liquid Gas Use the selections below to complete the concept map for the phases of matter. Write the letter of each characteristic in the appropriate
DEMONSTRATION 4.1 MOLECULES HITTING EACH OTHER
 DEMONSTRATION 4.1 MOLECULES HITTING EACH OTHER Directions for doing the demonstration are in the Science Book Teacher's Guide. 1. Students should include these ideas: The air moving out of the hair dryer
DEMONSTRATION 4.1 MOLECULES HITTING EACH OTHER Directions for doing the demonstration are in the Science Book Teacher's Guide. 1. Students should include these ideas: The air moving out of the hair dryer
An Introduction to The Next Generation Science Standards. NSTA National Conference San Antonio, Texas April 11-14, 2013
 An Introduction to The Next Generation Science Standards NSTA National Conference San Antonio, Texas April 11-14, 2013 Science and Engineering Practices in the NGSS Colorado Science Education Network Denver,
An Introduction to The Next Generation Science Standards NSTA National Conference San Antonio, Texas April 11-14, 2013 Science and Engineering Practices in the NGSS Colorado Science Education Network Denver,
UC Irvine FOCUS! 5 E Lesson Plan Title: Marble Isotope Lab Grade Level and Course: 8 th Grade Physical Science and 9-12 High School Chemistry
 UC Irvine FOCUS! 5 E Lesson Plan Title: Marble Isotope Lab Grade Level and Course: 8 th Grade Physical Science and 9-12 High School Chemistry Materials: Red, blue and yellow marbles (25 each) Instructional
UC Irvine FOCUS! 5 E Lesson Plan Title: Marble Isotope Lab Grade Level and Course: 8 th Grade Physical Science and 9-12 High School Chemistry Materials: Red, blue and yellow marbles (25 each) Instructional
Grade Six: Earthquakes/Volcanoes Lesson 6.2: Fault Formations
 Lesson Concept Link Time Grade Six: Earthquakes/Volcanoes Lesson 6.2: Fault Formations Forces in the Earth (tension, compression, shearing) cause stress at plate boundaries. Lesson 6.2 builds on the earthquake
Lesson Concept Link Time Grade Six: Earthquakes/Volcanoes Lesson 6.2: Fault Formations Forces in the Earth (tension, compression, shearing) cause stress at plate boundaries. Lesson 6.2 builds on the earthquake
All instruction should be three-dimensional. Page 1 of 12
 High School Conceptual Progressions Model Course 1 - Bundle 2 Electrical Forces and Matter or Interactions Between Particles This is the second bundle of the High School Conceptual Progressions Model Course
High School Conceptual Progressions Model Course 1 - Bundle 2 Electrical Forces and Matter or Interactions Between Particles This is the second bundle of the High School Conceptual Progressions Model Course
States of Matter: A Solid Lesson where Liquids Can be a Gas!
 TEACHER GUIDE STATES OF MATTER 60 Minute Physical Science Lesson Science- to- Go! Program Grades: 1-3 States of Matter: A Solid Lesson where Liquids Can be a Gas! Description Your classroom will be converted
TEACHER GUIDE STATES OF MATTER 60 Minute Physical Science Lesson Science- to- Go! Program Grades: 1-3 States of Matter: A Solid Lesson where Liquids Can be a Gas! Description Your classroom will be converted
Investigation 4: Fizz Quiz
 5 th Science Notebook Mixtures and Solutions Investigation 4 Investigation 4: Fizz Quiz Name: Big Question: How can matter be changed? Explain. 1 Alignment with New York State Science Standards & Performance
5 th Science Notebook Mixtures and Solutions Investigation 4 Investigation 4: Fizz Quiz Name: Big Question: How can matter be changed? Explain. 1 Alignment with New York State Science Standards & Performance
UC Irvine FOCUS! 5 E Lesson Plan
 UC Irvine FOCUS! 5 E Lesson Plan Title: Stomp Rockets Grade Level and Course: Pre-Algebra, Geometry, Grade 8 Physical Science, Grades 9-12 Physics (extension) - Trigonometry Materials: 1 stomp rocket per
UC Irvine FOCUS! 5 E Lesson Plan Title: Stomp Rockets Grade Level and Course: Pre-Algebra, Geometry, Grade 8 Physical Science, Grades 9-12 Physics (extension) - Trigonometry Materials: 1 stomp rocket per
TEACHER NOTES: ICE CUBE POSTER
 TEACHER NOTES: NATIONAL CURRICULUM LINKS THE PARTICULATE NATURE OF MATTER the properties of the different states of matter (solid, liquid and gas) in terms of the particle model, including gas pressure
TEACHER NOTES: NATIONAL CURRICULUM LINKS THE PARTICULATE NATURE OF MATTER the properties of the different states of matter (solid, liquid and gas) in terms of the particle model, including gas pressure
Physical Science DCI Progression Chart
 DCI Progression Chart PS1: Matter and Its Interactions Grade Bands PS1.A Structure & Properties of Matter Grades K-2 Grades 3-5 Grades 6-8 Grades 9-12 Second Grade * Different kinds of matter exist and
DCI Progression Chart PS1: Matter and Its Interactions Grade Bands PS1.A Structure & Properties of Matter Grades K-2 Grades 3-5 Grades 6-8 Grades 9-12 Second Grade * Different kinds of matter exist and
Chapter 6 Kinetic Particle Theory
 Chapter 6 Kinetic Particle Theory To understand that matter is made up of small particles which are in constant and random motion. To describe simple model of solids, liquids and gases, in terms of the
Chapter 6 Kinetic Particle Theory To understand that matter is made up of small particles which are in constant and random motion. To describe simple model of solids, liquids and gases, in terms of the
5.PS1.A: Structure and Properties of Matter
 Disciplinary Core Idea 5.PS1.A: Structure and Properties of Matter Matter of any type can be subdivided into particles that are too small to see, but even then the matter still exists and can be detected
Disciplinary Core Idea 5.PS1.A: Structure and Properties of Matter Matter of any type can be subdivided into particles that are too small to see, but even then the matter still exists and can be detected
MS.PS3.A: Definitions of Energy. MS.PS3.A: Definitions of Energy. MS.PS3.A: Definitions of Energy. Disciplinary Core Idea. Disciplinary Core Idea
 MS.PS3.A: Definitions of Energy Temperature is not a measure of energy; the relationship between the temperature and the total energy of a system depends on the types, states, and amounts of matter present.
MS.PS3.A: Definitions of Energy Temperature is not a measure of energy; the relationship between the temperature and the total energy of a system depends on the types, states, and amounts of matter present.
Middle School Physical Science
 Middle School Physical Science Students in middle school continue to develop understanding of four core ideas in the physical sciences. The middle school performance expectations in the Physical Sciences
Middle School Physical Science Students in middle school continue to develop understanding of four core ideas in the physical sciences. The middle school performance expectations in the Physical Sciences
Middle School Physical Science
 Middle School Physical Science Students in middle school continue to develop understanding of four core ideas in the physical sciences. The middle school performance expectations in the Physical Sciences
Middle School Physical Science Students in middle school continue to develop understanding of four core ideas in the physical sciences. The middle school performance expectations in the Physical Sciences
INTRODUCTION TO LESSON CLUSTER 8 Explaining Evaporation and Boiling
 INTRODUCTION TO LESSON CLUSTER 8 Explaining Evaporation and Boiling A. Lesson Cluster Goals and Lesson Objectives Goals: Students should be able to explain evaporation and boiling, both in macroscopic
INTRODUCTION TO LESSON CLUSTER 8 Explaining Evaporation and Boiling A. Lesson Cluster Goals and Lesson Objectives Goals: Students should be able to explain evaporation and boiling, both in macroscopic
Matter. Gas. Solid Liquid. Both shape and volume are not fixed. It has a fixed shape and a fixed volume.
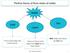 Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Hot and Cold Balloons
 Hot and Cold Balloons Moira filled a balloon with air. She tightly tied the balloon so no air could get in or out of the balloon. She kept the balloon in a warm room. An hour later she put the balloon
Hot and Cold Balloons Moira filled a balloon with air. She tightly tied the balloon so no air could get in or out of the balloon. She kept the balloon in a warm room. An hour later she put the balloon
Investigation 2: The Moon
 Science Notebook Sun, Moon, and Stars Investigation 2: The Moon Big Question: How, and why, does the Moon s appearance change? 1 Alignment with New York State Science Standards & Performance Indicators
Science Notebook Sun, Moon, and Stars Investigation 2: The Moon Big Question: How, and why, does the Moon s appearance change? 1 Alignment with New York State Science Standards & Performance Indicators
MIDDLE SCHOOL CYCLING OF EARTH S MATERIALS
 3D Science Performance Assessment Tasks MIDDLE SCHOOL CYCLING OF EARTH S MATERIALS MS: ES: Earth s Materials and Energy Performance Task Task Title Cycling of Earth's Materials Standards Bundle Information
3D Science Performance Assessment Tasks MIDDLE SCHOOL CYCLING OF EARTH S MATERIALS MS: ES: Earth s Materials and Energy Performance Task Task Title Cycling of Earth's Materials Standards Bundle Information
The 5E Model of Teaching Grade 8
 The 5E Model of Teaching Grade 8 Students Role and Actions in the 5E Model 5E s Consistent with Model Inconsistent with Model Engage Explore Explain Elaborate Evaluate Asks question such as why did this
The 5E Model of Teaching Grade 8 Students Role and Actions in the 5E Model 5E s Consistent with Model Inconsistent with Model Engage Explore Explain Elaborate Evaluate Asks question such as why did this
All instruction should be three-dimensional. NGSS Example Bundles. 1 of 10
 5 th Grade Thematic Model - Bundle 3 Stability and Change in Earth s Systems This is the third bundle of the Fifth Grade Thematic Model. Each bundle has connections to the other bundles in the course,
5 th Grade Thematic Model - Bundle 3 Stability and Change in Earth s Systems This is the third bundle of the Fifth Grade Thematic Model. Each bundle has connections to the other bundles in the course,
Kinetic Theory of Matter
 1 Temperature and Thermal Energy Kinetic Theory of Matter The motion of the particles in matter is described by kinetic theory of matter. Matter is composed of particles that are atoms, molecules, or ions
1 Temperature and Thermal Energy Kinetic Theory of Matter The motion of the particles in matter is described by kinetic theory of matter. Matter is composed of particles that are atoms, molecules, or ions
Name Date Block LESSON CLUSTER 6: Expansion and Contraction
 LESSON CLUSTER 6: Expansion and Contraction Did you know that when you say that something is hot or cold, you are actually saying something about the molecules of that substance? Words like hot and cold
LESSON CLUSTER 6: Expansion and Contraction Did you know that when you say that something is hot or cold, you are actually saying something about the molecules of that substance? Words like hot and cold
Connections to NGSS, CCSS-Mathematics, and CCSS-ELA/Literacy are at the end of this document.
 Conservation of Energy at the Skate Park Student Prior Knowledge: The equations for KE and PE and relationships of these with speed and height. Energy cannot be created or destroyed, but it can be transported
Conservation of Energy at the Skate Park Student Prior Knowledge: The equations for KE and PE and relationships of these with speed and height. Energy cannot be created or destroyed, but it can be transported
Learning Goals and Assessments in IQWST
 Learning Goals and Assessments in IQWST Joseph Krajcik Michigan State University Workshop on Developing Assessments to Meet the Goals of the 2012 Framework for K-12 Science Education September 13, 2012
Learning Goals and Assessments in IQWST Joseph Krajcik Michigan State University Workshop on Developing Assessments to Meet the Goals of the 2012 Framework for K-12 Science Education September 13, 2012
Bay Area Scientists in Schools Presentation Plan
 Bay Area Scientists in Schools Presentation Plan Lesson Name Presenter(s) Grade Level 3rd Melting, Freezing, and More!: Phase Transitions Steven Scroggins, Ailey Crow, Tom Holcombe, and Terence Choy California
Bay Area Scientists in Schools Presentation Plan Lesson Name Presenter(s) Grade Level 3rd Melting, Freezing, and More!: Phase Transitions Steven Scroggins, Ailey Crow, Tom Holcombe, and Terence Choy California
The performance expectation above was developed using the following elements from A Framework for K-12 Science Education: Disciplinary Core Ideas
 HS-PS2-1 HS-PS2-1. Analyze data to support the claim that Newton s second law of motion describes the mathematical relationship among the net force on a macroscopic object, its mass, and its acceleration.
HS-PS2-1 HS-PS2-1. Analyze data to support the claim that Newton s second law of motion describes the mathematical relationship among the net force on a macroscopic object, its mass, and its acceleration.
Alignment Guide PHYSICAL GLENCOE
 Alignment Guide PHYSICAL GLENCOE Glencoe Science Your Partner in Understanding and Implementing NGSS* Ease the Transition to Next Generation Science Standards Meeting NGSS Glencoe Science helps ease the
Alignment Guide PHYSICAL GLENCOE Glencoe Science Your Partner in Understanding and Implementing NGSS* Ease the Transition to Next Generation Science Standards Meeting NGSS Glencoe Science helps ease the
Thanks. You Might Also Like. I look forward helping you focus your instruction and save time prepping.
 Thanks Connect Thank you for downloading my product. I truly appreciate your support and look forward to hearing your feedback. You can connect with me and find many free activities and strategies over
Thanks Connect Thank you for downloading my product. I truly appreciate your support and look forward to hearing your feedback. You can connect with me and find many free activities and strategies over
Dublin City Schools Science Graded Course of Study Physical Science
 I. Content Standard: Students demonstrate an understanding of the composition of physical systems and the concepts and principles that describe and predict physical interactions and events in the natural
I. Content Standard: Students demonstrate an understanding of the composition of physical systems and the concepts and principles that describe and predict physical interactions and events in the natural
GRADE EIGHT CURRICULUM. Unit 1: The Makeup and Interactions of Matter
 Chariho Regional School District - Science Curriculum September, 2016 GRADE EIGHT CURRICULUM Unit 1: The Makeup and Interactions of Matter OVERVIEW Summary The performance expectations for this unit help
Chariho Regional School District - Science Curriculum September, 2016 GRADE EIGHT CURRICULUM Unit 1: The Makeup and Interactions of Matter OVERVIEW Summary The performance expectations for this unit help
Energy Changes in Chemical Reactions
 Energy Changes in Chemical Reactions Author(s): Ashley Colvin, Yunus Kinkhabwala, Prof. Song Lin, Jonathan Neff, & Greg Sauer Date Created: October 2016 Subject: Chemistry Grade Level: Middle School Standards:
Energy Changes in Chemical Reactions Author(s): Ashley Colvin, Yunus Kinkhabwala, Prof. Song Lin, Jonathan Neff, & Greg Sauer Date Created: October 2016 Subject: Chemistry Grade Level: Middle School Standards:
The performance expectation above was developed using the following elements from A Framework for K-12 Science Education: Disciplinary Core Ideas
 HS-PS3-1 HS-PS3-1. Create a computational model to calculate the change in the energy of one component in a system when the change in energy of the other component(s) and energy flows in and out of the
HS-PS3-1 HS-PS3-1. Create a computational model to calculate the change in the energy of one component in a system when the change in energy of the other component(s) and energy flows in and out of the
MS.PS1.A: Structure and Properties of Matter
 MS.PS1.A: Structure and Properties of Matter Each pure substance has characteristic physical and chemical properties (for any bulk quantity under given conditions) that can be used to identify it. (MS
MS.PS1.A: Structure and Properties of Matter Each pure substance has characteristic physical and chemical properties (for any bulk quantity under given conditions) that can be used to identify it. (MS
Unit 5: Types of Interactions. CONTENT AREA: General Physical Science GRADES: 6 UNIT: 5 of 7 Pacing: Approx. 1 Month (January)
 Unit 5: Types of Interactions CONTENT AREA: General Physical Science GRADES: 6 UNIT: 5 of 7 Pacing: Approx. 1 Month (January) Science and Engineering Practices Using Mathematics and Computational Thinking
Unit 5: Types of Interactions CONTENT AREA: General Physical Science GRADES: 6 UNIT: 5 of 7 Pacing: Approx. 1 Month (January) Science and Engineering Practices Using Mathematics and Computational Thinking
Lesson Plan: Stearic Acid
 Lesson Plan: Stearic Acid Created by: In this lesson, students investigate how stearic acid undergoes a 2014 AACT Middle School phase change from solid to liquid and back from liquid to solid. Content
Lesson Plan: Stearic Acid Created by: In this lesson, students investigate how stearic acid undergoes a 2014 AACT Middle School phase change from solid to liquid and back from liquid to solid. Content
Fairfield Public Schools Science Curriculum Physics
 Fairfield Public Schools Science Curriculum Physics BOE Approved 5/8/2018 1 Physics: Description Physics is the study of natural phenomena and interactions and between matter and energy using mathematical
Fairfield Public Schools Science Curriculum Physics BOE Approved 5/8/2018 1 Physics: Description Physics is the study of natural phenomena and interactions and between matter and energy using mathematical
Photosynthesis: How do plants get engery? Teacher Version
 Photosynthesis: How do plants get engery? Teacher Version In this lab, students explore the process of photosynthesis in spinach leaves. As oxygen is produced, the density of the leaves change and they
Photosynthesis: How do plants get engery? Teacher Version In this lab, students explore the process of photosynthesis in spinach leaves. As oxygen is produced, the density of the leaves change and they
Physical Science and Nature of Science Assessment Probes
 Physical Science and Nature of Science Assessment Probes Concept Matrix...6 Pennies...7 2 Is It a Solid?...25 3 Thermometer...33. 4 Floating Balloon... 39 5 Hot and Cold Balloons...45 6 Mirror on the Wall...5
Physical Science and Nature of Science Assessment Probes Concept Matrix...6 Pennies...7 2 Is It a Solid?...25 3 Thermometer...33. 4 Floating Balloon... 39 5 Hot and Cold Balloons...45 6 Mirror on the Wall...5
1) Of solids, liquids, and gases, the common state of matter is the liquid state.
 I. Properties of LIQUIDS: 1) Of solids, liquids, and gases, the common state of matter is the liquid state. a) can exist only within a relatively narrow range of temperature and pressure. 2) The kinetic-molecular
I. Properties of LIQUIDS: 1) Of solids, liquids, and gases, the common state of matter is the liquid state. a) can exist only within a relatively narrow range of temperature and pressure. 2) The kinetic-molecular
Lesson 1 Solids, Liquids, and Gases
 Lesson 1 Student Labs and Activities Page Launch Lab 8 Content Vocabulary 9 Lesson Outline 10 MiniLab 12 Content Practice A 13 Content Practice B 14 School to Home 15 Key Concept Builders 16 Enrichment
Lesson 1 Student Labs and Activities Page Launch Lab 8 Content Vocabulary 9 Lesson Outline 10 MiniLab 12 Content Practice A 13 Content Practice B 14 School to Home 15 Key Concept Builders 16 Enrichment
2. What is meant by Chemical State?. 3. Changing states of matter is about changing,,, and other.
 Name: Date: Period: Matter Mania! Online Computer Activity (3 pages) Part I: Go to http://www.chem4kids.com/ and answer the following questions in complete sentences. a. Click on MATTER (written in yellow)
Name: Date: Period: Matter Mania! Online Computer Activity (3 pages) Part I: Go to http://www.chem4kids.com/ and answer the following questions in complete sentences. a. Click on MATTER (written in yellow)
States of Matter. What physical changes and energy changes occur as matter goes from one state to another?
 Name States of Matter Date What physical changes and energy changes occur as matter goes from one state to another? Before You Read Before you read the chapter, think about what you know about states of
Name States of Matter Date What physical changes and energy changes occur as matter goes from one state to another? Before You Read Before you read the chapter, think about what you know about states of
Next Generation Science Standards for California Public Schools, Kindergarten through Grade Twelve
 California Department of Education Clarification statements were created by the writers of NGSS to supply examples or additional clarification to the performance expectations and assessment boundary statements.
California Department of Education Clarification statements were created by the writers of NGSS to supply examples or additional clarification to the performance expectations and assessment boundary statements.
4.PS4.B: Electromagnetic Radiation
 DCI: Waves and Their Applications in Technologies for Information 4.PS4.A: Wave Properties Waves, which are regular patterns of motion, can be made in water by disturbing the surface. When waves move across
DCI: Waves and Their Applications in Technologies for Information 4.PS4.A: Wave Properties Waves, which are regular patterns of motion, can be made in water by disturbing the surface. When waves move across
Copyright 2008 NSTA. All rights reserved. For more information, go to Pennies
 Pennies A shiny new penny is made up of atoms. Put an X next to all the things on the list that describe the atoms that make up the shiny new penny. hard soft I N G O D L I B E R T Y W E T R U S T 2 0
Pennies A shiny new penny is made up of atoms. Put an X next to all the things on the list that describe the atoms that make up the shiny new penny. hard soft I N G O D L I B E R T Y W E T R U S T 2 0
Objective Students will gain an understanding of how the properties of a solid material can affect how it interacts with water.
 OOBLECK! (1 Hour) Addresses NGSS Level of Difficulty: 4 Grade Range: K-2 OVERVIEW Students will examine the behavior of different types of solids when they are dissolved in water and explain those behaviors
OOBLECK! (1 Hour) Addresses NGSS Level of Difficulty: 4 Grade Range: K-2 OVERVIEW Students will examine the behavior of different types of solids when they are dissolved in water and explain those behaviors
Copyright 2009 NSTA. All rights reserved. For more information, go to Nails in a Jar
 Nails in a Jar Jake put a handful of wet, iron nails in a glass jar. He tightly closed the lid and set the jar aside. After a few weeks, he noticed that the nails inside the jar were rusty. Which sentence
Nails in a Jar Jake put a handful of wet, iron nails in a glass jar. He tightly closed the lid and set the jar aside. After a few weeks, he noticed that the nails inside the jar were rusty. Which sentence
heat By cillian bryan and scott doyle
 heat By cillian bryan and scott doyle What is heat Heat energy is the result of the movement of tiny particles called atoms molecules or ions in solids, liquids and gases. Heat energy can be transferred
heat By cillian bryan and scott doyle What is heat Heat energy is the result of the movement of tiny particles called atoms molecules or ions in solids, liquids and gases. Heat energy can be transferred
UC Irvine FOCUS! 5 E Lesson Plan Title: Acid/Base-pH Lab Grade Level and Course: 8 th grade Physical Science, Grades 9-12 Chemistry Materials:
 UC Irvine FOCUS! 5 E Lesson Plan Title: Acid/Base-pH Lab Grade Level and Course: 8 th grade Physical Science, Grades 9-12 Chemistry Materials: Detergent-quart, shampoo- quart Lemon-juice-quart Vinegar
UC Irvine FOCUS! 5 E Lesson Plan Title: Acid/Base-pH Lab Grade Level and Course: 8 th grade Physical Science, Grades 9-12 Chemistry Materials: Detergent-quart, shampoo- quart Lemon-juice-quart Vinegar
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED)
 CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
Chapter 2, Lesson 5: Changing State Melting
 Chapter 2, Lesson 5: Changing State Melting Key Concepts Melting is a process that causes a substance to change from a solid to a liquid. Melting occurs when the molecules of a solid speed up enough that
Chapter 2, Lesson 5: Changing State Melting Key Concepts Melting is a process that causes a substance to change from a solid to a liquid. Melting occurs when the molecules of a solid speed up enough that
Content and Vocabulary How They Support Each Other
 Content and Vocabulary How They Support Each Other 13 th Annual Academic Success Institute March 14, 2015 Sandi Yellenberg sandra_yellenberg@sccoe.org Thermal Expansion An Interactive Lecture Created by
Content and Vocabulary How They Support Each Other 13 th Annual Academic Success Institute March 14, 2015 Sandi Yellenberg sandra_yellenberg@sccoe.org Thermal Expansion An Interactive Lecture Created by
The complete lesson plan for this topic is included below.
 Home Connection Parent Information: Magnets provide a simple way to explore force with children. The power of a magnet is somewhat like magic to them and requires exploration to understand. When forces
Home Connection Parent Information: Magnets provide a simple way to explore force with children. The power of a magnet is somewhat like magic to them and requires exploration to understand. When forces
Properties of Matter
 Grade 7 Science, Quarter 2, Unit 2.1 Properties of Matter Overview Number of instructional days: 15 (1 day = 50 minutes) Content to be learned Identify different substances using data about characteristic
Grade 7 Science, Quarter 2, Unit 2.1 Properties of Matter Overview Number of instructional days: 15 (1 day = 50 minutes) Content to be learned Identify different substances using data about characteristic
Bay Area Scientists in Schools Presentation Plan
 Bay Area Scientists in Schools Presentation Plan Lesson Name Presenter(s) Chemical Reactions Mercedes Taylor, Nick Settineri, Jessica Ziegler, Tyler Hurlburt, Parker Deal Grade Level 5 Standards Connection(s)
Bay Area Scientists in Schools Presentation Plan Lesson Name Presenter(s) Chemical Reactions Mercedes Taylor, Nick Settineri, Jessica Ziegler, Tyler Hurlburt, Parker Deal Grade Level 5 Standards Connection(s)
2275 Speedway, Mail Code C9000 Austin, TX (512) Planet Fun
 Lesson Plan for Grades: Middle School Length of Lesson: 70 min Authored by: UT Environmental Science Institute Date created: 12/03/2016 Subject area/course: Mathematics, Astronomy, and Space Materials:
Lesson Plan for Grades: Middle School Length of Lesson: 70 min Authored by: UT Environmental Science Institute Date created: 12/03/2016 Subject area/course: Mathematics, Astronomy, and Space Materials:
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 6, LESSON 1: WHAT IS A CHEMICAL REACTION? MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact
The Next Generation Science Standards (NGSS) CHAPTER 6, LESSON 1: WHAT IS A CHEMICAL REACTION? MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact
Concepts Experimenting with Mixtures, chemical means. Lesson 6. SUBCONCEPT 5 Elements can be combined
 Quarter 1 PS21.A Structure and Properties of Matter Substances are made from different types of atoms, which combine with one another in various ways. Atoms form molecules that range in size from two to
Quarter 1 PS21.A Structure and Properties of Matter Substances are made from different types of atoms, which combine with one another in various ways. Atoms form molecules that range in size from two to
Gases: Properties and Behaviour
 SECTION 11.1 Gases: Properties and Behaviour Key Terms kinetic molecular theory of gases ideal gas On Earth, matter typically exists in three physical states: solid, liquid, and gas. All three states of
SECTION 11.1 Gases: Properties and Behaviour Key Terms kinetic molecular theory of gases ideal gas On Earth, matter typically exists in three physical states: solid, liquid, and gas. All three states of
Science & Literacy Activity GRADES 6-8
 Science & Literacy Activity GRADES 6-8 OVERVIEW This activity, which is aligned to the Common Core State Standards (CCSS) for English Language Arts, introduces students to scientific knowledge and language
Science & Literacy Activity GRADES 6-8 OVERVIEW This activity, which is aligned to the Common Core State Standards (CCSS) for English Language Arts, introduces students to scientific knowledge and language
What is so different about NGSS? Chemistry PD. Joe Krajcik. CREATE for STEM. Michigan State University. Atlanta, GA
 What is so different about NGSS? Chemistry PD Joe Krajcik CREATE for STEM Michigan State University Atlanta, GA Institute for Collaborative Research in Education, Assessment, and Teaching Environments
What is so different about NGSS? Chemistry PD Joe Krajcik CREATE for STEM Michigan State University Atlanta, GA Institute for Collaborative Research in Education, Assessment, and Teaching Environments
Properties of Matter
 Grade 7 Science, Quarter 1, Unit 1.1 Properties of Matter Overview Number of instructional days: 15 (1 day = 50 minutes) Content to be learned Identify different substances using data about characteristic
Grade 7 Science, Quarter 1, Unit 1.1 Properties of Matter Overview Number of instructional days: 15 (1 day = 50 minutes) Content to be learned Identify different substances using data about characteristic
Subject: Regents Chemistry Grade: 9-12 Unit #: 1 Title: Movie Special Effects
 UNIT OVERVIEW S Chemistry Standards: 3.2l, 3.3a, 3.1cc, 3.1kk, 4.2a, 4.2b, 4.2c, 3.1s, 3.1w, 3.1v, 3.2c, 3.1k, 3.1ff, 3.1gg STAGE ONE: Identify Desired Results Long-Term Transfer Goal At the end of this
UNIT OVERVIEW S Chemistry Standards: 3.2l, 3.3a, 3.1cc, 3.1kk, 4.2a, 4.2b, 4.2c, 3.1s, 3.1w, 3.1v, 3.2c, 3.1k, 3.1ff, 3.1gg STAGE ONE: Identify Desired Results Long-Term Transfer Goal At the end of this
INTRODUCTION TO LESSON CLUSTER 7
 INTRODUCTION TO LESSON CLUSTER 7 EXPLAINING MELTING AND SOLIDIFYING A. Lesson Cluster Goals and Lesson Objectives Goals Students should be able to explain melting and solidifying, by reference to the molecular
INTRODUCTION TO LESSON CLUSTER 7 EXPLAINING MELTING AND SOLIDIFYING A. Lesson Cluster Goals and Lesson Objectives Goals Students should be able to explain melting and solidifying, by reference to the molecular
B3 Relating Launch Speed and Range
 Key Question: What function relates the range and launch speed of a projectile? In this investigation, students identify the function relating projectile range to launch speed. In doing so, students are
Key Question: What function relates the range and launch speed of a projectile? In this investigation, students identify the function relating projectile range to launch speed. In doing so, students are
Investigation 3: The Stars
 Science Notebook Sun, Moon, and Stars Investigation 3: The Stars Big Question: How do stars compare and contrast from the Sun? 1 Alignment with New York State Science Standards & Performance Indicators
Science Notebook Sun, Moon, and Stars Investigation 3: The Stars Big Question: How do stars compare and contrast from the Sun? 1 Alignment with New York State Science Standards & Performance Indicators
ENGAGE. Daily Routines Common Core. Essential Question
 LESSON 7. Time to the Hour and Half Hour FOCUS COHERENCE RIGOR LESSON AT A GLANCE F C R Focus: Common Core State Standards Learning Objective.MD.C.7 Tell and write time from analog and digital clocks to
LESSON 7. Time to the Hour and Half Hour FOCUS COHERENCE RIGOR LESSON AT A GLANCE F C R Focus: Common Core State Standards Learning Objective.MD.C.7 Tell and write time from analog and digital clocks to
Research & reference tools & materials. Tools & materials for building out one selfwatering container per student (see
 Grade Level: Seventh Learners at the concrete operations stage of development with emerging formal operations are suited for this activity. STEM Science, Technology, Engineering, & Mathematics Common Core
Grade Level: Seventh Learners at the concrete operations stage of development with emerging formal operations are suited for this activity. STEM Science, Technology, Engineering, & Mathematics Common Core
U.S. Standards Alignment Guide
 U.S. Standards Alignment Guide Module 1: The Skate Blades Learning Objectives: Students will be able to (SWBAT) define and identify independent variables, dependent variables, and controls in an experiment
U.S. Standards Alignment Guide Module 1: The Skate Blades Learning Objectives: Students will be able to (SWBAT) define and identify independent variables, dependent variables, and controls in an experiment
ACTIVITY 3: Magnetic and Static Electric Interactions and Energy
 UNIT PEF Developing Ideas ACTIVITY 3: Magnetic and Static Electric Interactions and Energy Purpose In the first module of this course (or in the previous activity) you examined magnetic and static electric
UNIT PEF Developing Ideas ACTIVITY 3: Magnetic and Static Electric Interactions and Energy Purpose In the first module of this course (or in the previous activity) you examined magnetic and static electric
How Does the Sun s Energy Cause Rain?
 1.2 Investigate 3.3 Read How Does the Sun s Energy Cause Rain? In the water-cycle simulation, you observed water change from a liquid to a gas, and then back to a liquid falling to the bottom of the container.
1.2 Investigate 3.3 Read How Does the Sun s Energy Cause Rain? In the water-cycle simulation, you observed water change from a liquid to a gas, and then back to a liquid falling to the bottom of the container.
Simulation: Density FOR THE TEACHER
 Simulation: Density FOR THE TEACHER Summary In this simulation, students will investigate the effect of changing variables on both the volume and the density of a solid, a liquid and a gas sample. Students
Simulation: Density FOR THE TEACHER Summary In this simulation, students will investigate the effect of changing variables on both the volume and the density of a solid, a liquid and a gas sample. Students
SOLIDS, LIQUIDS, AND GASES
 CHAPTER 2 SOLIDS, LIQUIDS, AND GASES SECTION 2 1 States of Matter (pages 56-60) This section explains how shape, volume, and the motion of particles are useful in describing solids, liquids, and gases.
CHAPTER 2 SOLIDS, LIQUIDS, AND GASES SECTION 2 1 States of Matter (pages 56-60) This section explains how shape, volume, and the motion of particles are useful in describing solids, liquids, and gases.
The University of Texas at Austin. Build an Atom
 UTeach Outreach The University of Texas at Austin Build an Atom Content Standards Addressed in Lesson: TEKS8.5A describe the structure of atoms, including the masses, electrical charges, and locations,
UTeach Outreach The University of Texas at Austin Build an Atom Content Standards Addressed in Lesson: TEKS8.5A describe the structure of atoms, including the masses, electrical charges, and locations,
The performance expectation above was developed using the following elements from A Framework for K-12 Science Education: Disciplinary Core Ideas
 HS-PS1-1 HS-PS1-1. Use the periodic table as a model to predict the relative properties of elements based on the patterns of electrons in the outermost energy level of atoms. [Clarification Statement:
HS-PS1-1 HS-PS1-1. Use the periodic table as a model to predict the relative properties of elements based on the patterns of electrons in the outermost energy level of atoms. [Clarification Statement:
Investigation 1: Separating Mixtures
 5 th Science Notebook Mixtures and Solutions Investigation 1 Investigation 1: Separating Mixtures Name: Big Question: How are mixtures and solutions similar and different and how can they be separated?
5 th Science Notebook Mixtures and Solutions Investigation 1 Investigation 1: Separating Mixtures Name: Big Question: How are mixtures and solutions similar and different and how can they be separated?
Bay Area Scientists in School Presentation Plan
 Bay Area Scientists in School Presentation Plan Lesson Name It s just a phase! Presenter(s) Kevin Metcalf, David Ojala, Melanie Drake, Carly Anderson, Hilda Buss, Lin Louie, Chris Jakobson California Standards
Bay Area Scientists in School Presentation Plan Lesson Name It s just a phase! Presenter(s) Kevin Metcalf, David Ojala, Melanie Drake, Carly Anderson, Hilda Buss, Lin Louie, Chris Jakobson California Standards
Lesson 4. Stream Table Lab. Summary. Suggested Timeline. Objective. Materials. Teacher Background Knowledge
 Rivers Instructional Case: A series of student-centered science lessons Suggested Timeline 50 minutes Materials 1 Stream table for each station 1 Calculator for each station Approximately 3 lb of sand
Rivers Instructional Case: A series of student-centered science lessons Suggested Timeline 50 minutes Materials 1 Stream table for each station 1 Calculator for each station Approximately 3 lb of sand
Chapter 1, Lesson 3: The Ups and Downs of Thermometers
 Chapter 1, Lesson 3: The Ups and Downs of Thermometers Key Concepts The way a thermometer works is an example of heating and cooling a liquid. When heated, the molecules of the liquid in the thermometer
Chapter 1, Lesson 3: The Ups and Downs of Thermometers Key Concepts The way a thermometer works is an example of heating and cooling a liquid. When heated, the molecules of the liquid in the thermometer
Investigation 1: The Sun
 Science Notebook Sun, Moon, and Stars Investigation 1: The Sun Big Question: Does the Sun move, and why is its position in the sky important? 1 Alignment with New York State Science Standards & Performance
Science Notebook Sun, Moon, and Stars Investigation 1: The Sun Big Question: Does the Sun move, and why is its position in the sky important? 1 Alignment with New York State Science Standards & Performance
KINETIC PARTICLE THEORY
 KINETIC PARTICLE THEORY IMPORTANT DEFINITIONS: The mixing process in gases or solutions due to the random motion of particles is called Diffusion. The process by which a liquid changes into a vapour at
KINETIC PARTICLE THEORY IMPORTANT DEFINITIONS: The mixing process in gases or solutions due to the random motion of particles is called Diffusion. The process by which a liquid changes into a vapour at
Amarillo ISD Science Curriculum
 Amarillo Independent School District follows the Texas Essential Knowledge and Skills (TEKS). All of AISD curriculum and documents and resources are aligned to the TEKS. The State of Texas State Board
Amarillo Independent School District follows the Texas Essential Knowledge and Skills (TEKS). All of AISD curriculum and documents and resources are aligned to the TEKS. The State of Texas State Board
Atoms and molecules are in motion and have energy
 Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
MEASURING THE RATE OF PHOTOSYNTHESIS
 MEASURING THE RATE OF PHOTOSYNTHESIS QUESTION: What factors affect the rate of photosynthesis in plants? BACKGROUND KNOWLEDGE Leaf disks float, normally. When the air spaces in the leaf are filled with
MEASURING THE RATE OF PHOTOSYNTHESIS QUESTION: What factors affect the rate of photosynthesis in plants? BACKGROUND KNOWLEDGE Leaf disks float, normally. When the air spaces in the leaf are filled with
Pine Hill Public Schools Curriculum
 Content Area: Pine Hill Public Schools Curriculum Science Course Title/ Grade Level: Honors Physics / Gr. 11 & 12 Unit 1: Unit 2: Unit 3: Unit 4: Unit 4: Introduction, Measurement, Estimating Duration:
Content Area: Pine Hill Public Schools Curriculum Science Course Title/ Grade Level: Honors Physics / Gr. 11 & 12 Unit 1: Unit 2: Unit 3: Unit 4: Unit 4: Introduction, Measurement, Estimating Duration:
Tackling Potential and Kinetic Energy
 Tackling Potential and Kinetic Energy Overview In this lesson, students explore concepts of energy and relate them to tackling in football. Using manipulatives, such as marbles or ball, students will investigate
Tackling Potential and Kinetic Energy Overview In this lesson, students explore concepts of energy and relate them to tackling in football. Using manipulatives, such as marbles or ball, students will investigate
Most substances can be in three states: solid, liquid, and gas.
 States of Matter Most substances can be in three states: solid, liquid, and gas. Solid Particles Have Fixed Positions The particles in a solid are very close together and have an orderly, fixed arrangement.
States of Matter Most substances can be in three states: solid, liquid, and gas. Solid Particles Have Fixed Positions The particles in a solid are very close together and have an orderly, fixed arrangement.
4.4 Electricity & Magnetism
 4.4 Electricity & Magnetism Enduring Understanding: Electrical energy can be transferred and transformed. Magnetic energy can be transferred and transformed. Essential Question: How is energy transferred
4.4 Electricity & Magnetism Enduring Understanding: Electrical energy can be transferred and transformed. Magnetic energy can be transferred and transformed. Essential Question: How is energy transferred
Leonie Boshoff-Mostert Edited by Anne Starace
 GASES, LIQUIDS AND SOLIDS Density Leonie Boshoff-Mostert Edited by Anne Starace Abstract Matter is sorted into three groups: solids, liquids and gases. Solids, liquids and gases each have characteristic
GASES, LIQUIDS AND SOLIDS Density Leonie Boshoff-Mostert Edited by Anne Starace Abstract Matter is sorted into three groups: solids, liquids and gases. Solids, liquids and gases each have characteristic
