Role of the substrate in electronic structure, molecular orientation, and morphology of organic thin films: diindenoperylene on rutile TiO 2 (110)
|
|
|
- Helen Pitts
- 5 years ago
- Views:
Transcription
1 COMMUNICATION Physical Chemistry Chemical Physics Role of the substrate in electronic structure, molecular orientation, and morphology of organic thin films: diindenoperylene on rutile TiO 2 (110) Britt-Elfriede Schuster, a M. Benedetta Casu,* a Indro Biswas, a Alexander Hinderhofer, b Alexander Gerlach, b Frank Schreiber b and Thomas Chassé a Received 29th June 2009, Accepted 5th August 2009 First published as an Advance Article on the web 18th August 2009 DOI: /b912790a The results of our multitechnique investigation performed on diindenoperylene thin films deposited on rutile TiO 2 (110) show island growth, with crystallites nucleating preferentially along the [1 10] substrate crystallographic axis. The findings evidence that the films properties at the interface are common to those found for a number of organic molecules deposited on the same substrate, revealing that the structural and morphological properties of organic thin films on rutile TiO 2 (110) are completely driven by its surface morphology. In the past organic materials were considered very appealing not only because of a pure academic interest but also because of their promising characteristics towards electronic applications. 1 Nowadays we can affirm that these materials kept their promises and they are widely present on the market. Some of their properties demand deeper investigation and a better understanding, also in view of their use in an even larger number of applications, where low costs, chemical flexibility and energy saving technologies play a major role and organic molecules may achieve a different or a better performance than inorganic semiconductors. 2 Organic thin films properties like morphology, structure, and electronic structure are strongly influenced by preparation conditions, the substrate, and post-growth treatment. 3 5 Among small molecules, the most investigated ones, because of their importance as a model system and as an active layer in devices, are certainly the acenes, the phthalocyanines, and the perylene-based molecules. 6,7 Diindenoperylene (DIP, C 32 H 16, Fig. 1) is a perylene-based molecule that shows a high hole mobility already in thin films, 8 good film forming properties and thermal stability Thus, DIP is a molecule that can be used as a model system but at the same time it is very promising from a technological point of view. Titanium dioxide is an optically clear insulating oxide, and it is the most investigated single crystalline system among all metal oxides. 14 The rutile TiO 2 (110) surface is the most stable surface configuration. The application potential is enormous, ranging from medicine to electronics. 14,15 Besides the interest in the investigation of a new organic/oxide system, it is clear how the coupling of two such excellent materials may also lead to further steps in device engineering. a Institute of Physical and Theoretical Chemistry, University of Tu bingen, Auf der Morgenstelle 8, Tu bingen, Germany. benedetta.casu@uni-tuebingen.de; Fax: +49 (0) ; Tel: +49 (0) b Institute for Applied Physics, University of Tu bingen, Auf der Morgenstelle 10, Tu bingen, Germany Here, we report the results of a multitechnique investigation performed on DIP thin films deposited on rutile TiO 2 (110). We have found evidence for island growth, with DIP crystallites nucleating preferentially along the [1 10] crystallographic axis of the substrate. In thicker films, the molecules assume an upright standing configuration. Sample preparation and photoemission experiments (X-ray photoelectron spectroscopy, XPS) were carried out in an ultrahigh vacuum (UHV) system consisting of two preparation chambers (base pressure better than 10 9 mbar), and an analyzing chamber (base pressure of around mbar) equipped with a low energy electron diffraction (LEED) system, a SPECS Phoibos 100 analyzer, and a monochromatized X-ray source (Focus 500, XR50M X-ray source, Al Ka ev). A clean carbon-free rutile TiO 2 (110) single crystal (PI-KEM Ltd, United Kingdom) was prepared by a very gentle and brief Ar + ion bombardment (500 V), followed by annealing in UHV at approximately 830 K, which gave a sharp LEED pattern of an (1 1) reconstruction. 16 After several cycles of sputtering and annealing the crystal showed a pale blue color, a convenient indicator for the concentration of bulk oxygen vacancies and the associated color centers. The Ti 2p core level photoemission spectrum showed only a very weak low binding energy shoulder usually attributed to the presence of surface non-stoichiometry due to oxygen vacancies (3 5% in our clean substrates). The root mean square (rms) roughness of the bare substrate after preparation was 11 nm. Thin films of DIP were prepared in situ by organic molecular beam deposition (OMBD) using strictly controlled evaporation conditions (evaporation rate = 3 A min 1, T sub =201C). The nominal thicknesses were measured with a quartz crystal microbalance and were cross-checked by using the attenuation of the XPS substrate signal after DIP deposition. Survey XPS spectra were recorded using a pass energy of 30 ev, detailed spectra were performed with a pass energy of 10 ev resulting in an experimental resolution of 450 mev. All spectra were calibrated to the Ti 2p 3/2 emission of the substrate. Atomic force microscopy (AFM) measurements were performed under ambient conditions in tapping mode with a Nanoscope IIIa (Digital Instruments) scanning probe microscope. Near edge X-ray absorption spectroscopy (NEXAFS) measurements were performed at the beamline UE52-PGM at BESSY (Berlin). This beamline is characterized by a plane grating monochromator. The photon energy ranges from 100 to 1500 ev, with an energy resolving power of E/DE = at 401 ev (c ff = 10, 10 mm exit slit). The main chamber (base pressure mbar) is equipped with a standard twin 9000 Phys. Chem. Chem. Phys., 2009, 11, This journal is c the Owner Societies 2009
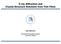
2 Fig. 1 (a) Thickness-dependent C 1s core level photoemission spectra of DIP deposited on rutile (110) are shown. (b): Ti 2p core level photoemission spectra of rutile TiO 2 (110) as prepared (1) and after deposition of a DIP film (nominal thickness: 91 A ) (2). C1s NEXAFS spectra obtained from 3 (c), 7 (d) and 24 Å (e) of DIP thin films deposited on TiO 2 (110) at RT. The spectra were taken in grazing incidence for p- (black curve) and s- (grey curve) polarisation. The geometry of the NEXAFS experiment and the molecular structure are also shown. anode X-ray source, a SCIENTA R4000 electron energy analyzer, and a home-made partial electron yield detector. The X-ray reflectivity measurements were made ex situ with a two-circle instrument (XRD, 3003 TT, Seifert Analytical X-ray). Using a X-ray tube with a copper anode and a Ni/C multilayer mirror in combination with a germanium channel-cut crystal gives a monochromatic and parallel beam with l = 1.54 A (Cu K a1 ). The chosen slits in front of the scintillation counter resulted in an angular resolution of B0.011, and a dynamic range of more than seven orders of magnitude. All films were carefully checked for radiation damage during data acquisition. No degradation of the samples was observed on the time scale of all performed experiments. XPS is a very powerful technique to gain information about electronic structure and chemical bonding. In Fig. 1a the thickness-dependent C 1s core level photoemission spectra of DIP deposited on TiO 2 (110) are shown. A strong peak dominates the spectrum at ev in the thicker films, and a smaller shake-up satellite at ev is clearly visible. The peaks stem from the C 1s core levels. Considering the DIP molecular structure, at first two contributions from different carbon sites can be expected, namely, the carbon atom sites that belong to the aromatic rings surrounded by adjacent carbon atoms, and the remaining carbon sites (C H bonds). In principle, because of the molecular symmetry (the free diindenoperylene molecule belongs to the D 2h point group), the C 1s core level features are expected to be due to at least nine contributions, one for each different non-equivalent carbon site. However, due to their similar binding energy and the finite resolution of our experiment their separation and energy determination by curve fitting would be quite speculative. The shake-up satellite at ev (HOMO LUMO shake up satellite) is already clearly visible for the first layer deposited on top of the TiO 2 surface. Its relative intensity does not change relevantly with thickness indicating that the satellite is due to intramolecular screening effects of the core hole. 17 Comparing its separation from the main C 1s peak, we observe that it is lower than the DIP optical gap. 13 This suggests that the HOMO LUMO shake up excitations contribute to screen the core hole via a charge distribution within the molecule, as seen in polyacenes, due to the delocalization of the charge in the aromatic system. 18 In addition, a 0.2 ev energy shift of the C 1s peak towards higher binding energies with increasing film thickness is observable. This relatively small shift confirms a poor screening effect of the C 1s core hole depending on the substrate and is in agreement with the insulating nature of the substrate. 17 Another observed effect is the decrease of the line width with increasing film thickness. There are several contributions that affect the line shapes in XPS spectra of large p-conjugated organic molecules, like intrinsic lifetime broadening, experimental This journal is c the Owner Societies 2009 Phys.Chem.Chem.Phys., 2009, 11,
3 contributions, and vibronic and inhomogeneous broadening. 19 In this case, most of these contributions (e.g. lifetime, experimental set up) are constant for each presented curve, thus the difference in line width indicates a different strength of the interaction among the molecules in the few layers up to 20 A (e.g. different molecular packing and/or orientation). In particular, the larger full width at half maximum (FWHM) of the C 1s peak for thinner films (FWHM = 1.1 ev) indicates a stronger inhomogeneity of the first layers on top of the substrate with respect to the successive layers (FWHM = 0.9 ev). Thickness dependent XPS investigations offer also the opportunity to identify the growth mode. 20 Thus, we have simultaneously monitored the C 1s and the Ti 2p core levels with XPS during growth. In Fig. 1b, the Ti 2p core level signals are shown, upon deposition of a nominally 91 A thick film. Under the present experimental conditions (kinetic energy of 1000 ev) the inelastic mean free path (l) ofti2p electrons is about 23 A. 21 Although the information depth (3l =69A ) is lower than the film thickness (91 A ) the XPS substrate signal is still clearly visible. This means that, while we can exclude the occurrence of a perfect layer-by-layer growth, we expect a growth mode characterized by the presence of islands, i.e. Stranski Krastanov growth mode (layers plus islands), or Volmer Weber growth mode (island formation). By NEXAFS it is possible to gain information about the unoccupied states, the environment of specific chemical elements, bonding properties, and local charge distributions. NEXAFS can be described by transition matrix elements containing the dipole selection rules, thus the intensity of the NEXAFS resonances will depend on the mutual orientation of the molecular orbital involved on the given transition and the incident radiation. This aspect of the NEXAFS spectroscopy can be used to calculate the molecular orientation (angle between the molecular axis and the substrate) directly from the spectra. 22 Fig. 1c e show C 1s NEXAFS spectra for different film thicknesses as indicated. The spectra were taken in p-polarization (black curves) and s-polarization (red curves) (see the experimental geometry in Fig. 1). Two main groups of resonances dominate the spectra. We can identify the p*-region up to about 290 ev (dominant features 1 and 2), and the s*-region above 290 ev. Let us focus on feature 2, which is particularly sharp, in order to discuss the obtained spectra. In Fig. 1c, where the NEXAFS spectra of a 3 A thick film are shown, we observe that the intensity of feature 2 is rather insensitive to the change of polarization of the incident radiation. This is also true for the NEXAFS spectra of a 7 A thick film (Fig. 1d). On the contrary, a strong dichroism is clearly observable for a 24 A thick film (Fig. 1e). This is also confirmed when calculating the molecular orientation: 23 we find a tilt angle of 741 for the latter film, while it is around 501 for the former. In other words, we observe an increase of the molecular orientation with the tendency toward upright standing molecules in thicker films. The structure in the monolayer regime is more complicated. The calculated molecular orientation suggests three possible textures (note that standard NEXAFS is averaged on the area seen by the beam, in our experiment around mm 2 ): (i) 501 is the real molecular arrangement. (ii) The first layer is a disordered film. (iii) The film is characterized by small domains with differently distributed orientation. The XPS results have already shown that the first layers on top of the substrate have a poorer degree of order than the following layers. Matching XPS and NEXAFS results, we can therefore exclude that the molecules really accommodate themselves with their axis having a 501 angle with respect to the substrate. Furthermore, the comparison of the NEXAFS spectra of the first layer and the thicker films excludes the occurrence of a chemical bond. As a matter of fact, in that case, the monolayer spectra would exhibit a change in the intensity of the feature in the p* region. Such a decrease in intensity of the p*-resonances is not observed for a pure van-der-waals bond between substrate and adsorbate, because this does not involve p* orbitals like, for example, the LUMO or the LUMO+1, thus there is no effect on the transitions from C 1s levels to those same orbitals. In our case, there is neither an indication of such a broadening nor an intensity reduction for resonance 2 when comparing monolayer and multilayer spectra. Consequently, we may exclude a chemical bond as a cause for the difference in structure of the first layer. This result also implies that the coupling between the p system of the DIP molecules and the TiO 2 (110) substrate is weak. This observation is in good agreement with the passivated nature of the substrate (the preparation was performed to minimize the presence of oxygen vacancies), and the fact that the rutile TiO 2 (110) surface is less reactive in comparison with the other TiO 2 crystallographic faces. To further characterise the structure, X-ray reflectivity measurements of the DIP thin films were performed (Fig. 2). Similarly to DIP growth on other rough substrates 9 two Bragg reflections at q z = 0.37 A 1 and q z = 1.47 A 1 indicate the polycrystalline structure of the thin films. The Bragg reflections can be assigned to the s-phase at q z = 0.37 A 1 1 (standing molecules) and to the l-phase at q z = 1.47 A (lying molecules), respectively. 4 Rocking scans on both Bragg reflections were measured to probe the thin films for the orientation distribution of the crystallites of each phase. The rocking curve belonging to the l-phase exhibits a large mosaicity with an angular distribution width of 6.21 (FWHM). In comparison, the angular distribution width of the s-phase Fig. 2 X-Ray reflectivity data from a nominally 56 Å thick DIP film showing two Bragg reflections, which can be assigned to the s-phase (standing molecules) and l-phase (lying molecules) of DIP. The inset shows the narrow rocking width of the s-phase Bragg reflection, indicating the low mosaicity of this phase Phys. Chem. Chem. Phys., 2009, 11, This journal is c the Owner Societies 2009
4 is very small: (inset of Fig. 2), which indicates low mosaicity. These observations are consistent with the NEXAFS measurements, where a molecular tilt angle of B501 is found for thin films measured and highly oriented DIP molecules are found in higher thicknesses. The TiO 2 (110)-(1 1) surface is characterized by parallel rows of Ti atoms running along the [001] direction, alternated with rows of bridging oxygen atoms. 14 The oxygen atoms protrude above the surface plane and they dominate the surface topography. Density functional theory calculations performed on benzene, pyridine and derivatives have shown that there is the possibility for those molecules to adsorb both in upright standing and in flat lying configuration on rutile TiO 2 (110). 14,23 The two configurations may occur simultaneously. In particular, the most stable configuration for pyridine is the upright standing one with the carbon atoms interacting with the bridging oxygen atoms via a hydrogen bond, while flat lying molecules are found in the [001] direction. Also in this case, there is not a chemical bond; the major role in the adsorption is played by van-der-waals interactions, and the molecules are rather mobile. We may reckon that an analogous situation occurs when depositing DIP on TiO 2 (110): the bridging oxygen atoms force a part of the molecules impinging the surface into the upright standing position, while a part stays flat on the substrate and it is free to move probably along the [001] direction, as discussed for pyridine and acenes. 14,24 Also for DIP, the upright arrangement of the molecular axis is expected to be the most energetically favourable, since the molecules in the single crystal are known to assume this configuration with respect to the cleavage plane. 25 This also explains the reason why the degree of order is higher in the thicker films: the influence of the substrate becomes weaker and the molecules minimize their energy according to the 3D fashion. Atomic force microscopy (AFM) images of DIP deposited on TiO 2 (110) clearly reveal the island formation we expected from the XPS results (Fig. 3a). We observe that the islands are separated by regions where DIP forms layers suggesting Stranski Krastanov growth mode (see, as an example, the profile analysis in Fig. 3b). In addition, we observe that the molecules assemble themselves in parallel crystallites, with lateral dimensions up to 400 nm 200 nm that adopt the [1 10] crystallographic axis as a preferential orientation. This effect has been also found for para-sexiphenyl deposited on this same substrate. 26 However, the reason of the crystallite orientation remained unresolved. Thus, in order to find a possible explanation, we have also investigated the bare substrate morphology after preparation by AFM. We observe in the AFM micrographs the presence of steps along the [1 10] direction (Fig. 3c). As a consequence, the molecules are forced to nucleate islands along that direction, which also determines the elongation of the crystallite and their preferential orientation. We have investigated DIP molecules deposited on rutile TiO 2 (110) single crystals. We have found that the films, under Fig. 3 (a) 5 mm 5 mm AFM images of nominally 56 Å thick DIP film deposited on TiO 2 (110). The corresponding substrate azimuthal directions are also given. (b) 3 mm 3 mm AFM images of nominally 56 Å thick DIP film: profile analysis of the same sample. The profiles were extracted as indicated by the lines. (c) 5 mm 5 mm AFM images of TiO 2 (110) after preparation. The corresponding substrate azimuthal directions are also given. This journal is c the Owner Societies 2009 Phys.Chem.Chem.Phys., 2009, 11,
5 these preparation conditions, follow a Stranski Krastanov or Volmer Weber growth mode. The molecules are physisorbed on the substrate. The bridging oxygen atoms influence the degree of order, presumably via a hydrogen bond, in the first layer on top of the substrate, and various arrangements of the molecular axes are found also in the few successive layers. The thicker films are characterized by a high orientational order with upright standing molecules. In addition, due to the substrate morphology after preparation, DIP forms crystallites preferentially oriented along the [1 10] crystallographic axis of the substrate. Our results show the important role played by the substrate in electronic structure, molecular orientation, and morphology in the system DIP/TiO 2 (110) and they confirm it represents an interesting candidate for use in organic electronics. Comparing these results with previous works, 14,15,24,26 we conclude that the structural and morphological properties of the interface of organic thin films deposited on rutile TiO 2 (110) are driven by the substrate surface topography, with its rows of bridging oxygen atoms, while the molecular properties are less relevant. The authors thank BESSY for providing beamtime, BESSY staff, in particular the Users support, and Matthias Mast. We also thank David Batchelor, and Stefan Pohl, for support at the beamline. Francesco Allegretti, University of Graz, is gratefully acknowledged for helpful discussions on rutile TiO 2 (110) surface preparation. We are grateful to Wolgang Neu for his kind technical support. Financial support by BMBF under contract 05 ES3XBA/5 and by Ministerium fu r Wissenschaft, Forschung und Kunst Baden-Wu rttemberg, under contract AZ /2 is gratefully acknowledged. References 1 D. Braga and G. Horowitz, Adv. Mater., 2009, 21, S. R. Forrest, Org. Electron., 2003, 4, M. B. Casu, X. Yu, S. Schmitt, C. Heske and E. Umbach, J. Chem. Phys., 2008, 129, S. Kowarik, A. Gerlach, S. Sellner, F. Schreiber, L. Cavalcanti and O. Konovalov, Phys. Rev. Lett., 2006, 96, S. Kera, M. B. Casu, K. R. Bauchspieß, D. Batchelor, Th. Schmidt and E. Umbach, Surf. Sci., 2006, 600, S. Kowarik, A. Gerlach and F. Schreiber, J. Phys.: Condens. Matter, 2008, 20, and references therein. 7 G. Witte and C. Woll, J. Mater. Res., 2004, 19, 1889 and references therein. 8 N. Karl, Synth. Met., 2003, , A. C. Dürr, N. Koch, M. Kelsch, A. Ru hm, J. Ghijsen, R. L. Johnson, J.-J. Pireaux, J. Schwartz, F. Schreiber, H. Dosch and A. Kahn, Phys. Rev. B, 2003, 68, S. Sellner, A. Gerlach, F. Schreiber, M. Kelsch, N. Kasper, H. Dosch, S. Meyer, J. Pflaum, M. Fischer and B. Gompf, Adv. Mater., 2004, 16, M. B. Casu, I. Biswas, M. Nagel, P. Nagel, S. Schuppler and T. Chassé, Phys. Rev. B: Condens. Matter Mater. Phys., 2008, 78, M. B. Casu, I. Biswas, B.-E. Schuster, M. Nagel, P. Nagel, S. Schuppler and T. Chassé, Appl. Phys. Lett., 2008, 93, A. C. Du rr, N. Koch, M. Kelsch, A. Ru hm, J. Ghijsen, R. L. Johnson, J.-J. Pireaux, J. Schwartz, F. Schreiber, H. Dosch and A. Kahn, Phys. Rev. B: Condens. Matter Mater. Phys., 2003, 68, U. Diebold, Surf. Sci. Rep., 2003, 48, 53 and references therein. 15 F. H. Jones, Surf. Sci. Rep., 2001, 42, 75 and references therein. 16 F. Allegretti, S. O Brien, M. Polcik, D. I. Sayago and D. P. Woodruff, Phys. Rev. Lett., 2005, 95, A. Schöll, Y. Zou, Th. Schmidt, R. Fink and E. Umbach, J. Phys. Chem. B, 2004, 108, M. L. Rocco, M. Haeming, D. R. Batchelor, R. Fink and E. Umbach, J. Chem. Phys., 2008, 129, A. Schöll, Y. Zou, M. Jung, Th. Schmidt, R. Fink and E. Umbach, J. Chem. Phys., 2004, 121, J. A. Venables and J. Liu, J. Electron Spectrosc. Relat. Phenom., 2005, 143, S. Tanuma, C. J. Powell and D. R. Penn, Surf. Interface Anal., 1988, 11, J. Stöhr and D. Outka, Phys. Rev. B: Condens.Matter Mater. Phys., 1987, 36, M. B. Casu, P. Cosseddu, D. Batchelor, A. Bonfiglio and E. Umbach, J. Chem. Phys., 2008, 128, S. Reiß, H. Krumm, A. Niklewski, V. Staemmler and Ch. Wo ll, J. Chem. Phys., 2002, 116, M. A. Heinrich, J. Pflaum, A. K. Tripathi, W. Frey, M. L. Steigerwald and T. Siegrist, J. Phys. Chem. C, 2007, 111, G. Koller, S. Berkebile, J. R. Krenn, G. Tzvetkov, G. Hlawacek, O. Lengyel, F. P. Netzer, C. Teichert, R. Resel and M. G. Ramsey, Adv. Mater., 2004, 16, Phys. Chem. Chem. Phys., 2009, 11, This journal is c the Owner Societies 2009
structure and paramagnetic character R. Kakavandi, S-A. Savu, A. Caneschi, T. Chassé, M. B. Casu Electronic Supporting Information
 At the interface between organic radicals and TiO 2 (110) single crystals: electronic structure and paramagnetic character R. Kakavandi, S-A. Savu, A. Caneschi, T. Chassé, M. B. Casu Electronic Supporting
At the interface between organic radicals and TiO 2 (110) single crystals: electronic structure and paramagnetic character R. Kakavandi, S-A. Savu, A. Caneschi, T. Chassé, M. B. Casu Electronic Supporting
Spin-resolved photoelectron spectroscopy
 Spin-resolved photoelectron spectroscopy Application Notes Spin-resolved photoelectron spectroscopy experiments were performed in an experimental station consisting of an analysis and a preparation chamber.
Spin-resolved photoelectron spectroscopy Application Notes Spin-resolved photoelectron spectroscopy experiments were performed in an experimental station consisting of an analysis and a preparation chamber.
Photon Interaction. Spectroscopy
 Photon Interaction Incident photon interacts with electrons Core and Valence Cross Sections Photon is Adsorbed Elastic Scattered Inelastic Scattered Electron is Emitted Excitated Dexcitated Stöhr, NEXAPS
Photon Interaction Incident photon interacts with electrons Core and Valence Cross Sections Photon is Adsorbed Elastic Scattered Inelastic Scattered Electron is Emitted Excitated Dexcitated Stöhr, NEXAPS
A Momentum Space View of the Surface Chemical Bond - Supplementary Information
 A Momentum Space View of the Surface Chemical Bond - Supplementary Information Stephen Berkebile, a Thomas Ules, a Peter Puschnig, b Lorenz Romaner, b Georg Koller, a Alexander J. Fleming, a Konstantin
A Momentum Space View of the Surface Chemical Bond - Supplementary Information Stephen Berkebile, a Thomas Ules, a Peter Puschnig, b Lorenz Romaner, b Georg Koller, a Alexander J. Fleming, a Konstantin
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Conductance Measurements The conductance measurements were performed at the University of Aarhus. The Ag/Si surface was prepared using well-established procedures [1, 2]. After
SUPPLEMENTARY INFORMATION Conductance Measurements The conductance measurements were performed at the University of Aarhus. The Ag/Si surface was prepared using well-established procedures [1, 2]. After
In order to determine the energy level alignment of the interface between cobalt and
 SUPPLEMENTARY INFORMATION Energy level alignment of the CuPc/Co interface In order to determine the energy level alignment of the interface between cobalt and CuPc, we have performed one-photon photoemission
SUPPLEMENTARY INFORMATION Energy level alignment of the CuPc/Co interface In order to determine the energy level alignment of the interface between cobalt and CuPc, we have performed one-photon photoemission
Studying Metal to Insulator Transitions in Solids using Synchrotron Radiation-based Spectroscopies.
 PY482 Lecture. February 28 th, 2013 Studying Metal to Insulator Transitions in Solids using Synchrotron Radiation-based Spectroscopies. Kevin E. Smith Department of Physics Department of Chemistry Division
PY482 Lecture. February 28 th, 2013 Studying Metal to Insulator Transitions in Solids using Synchrotron Radiation-based Spectroscopies. Kevin E. Smith Department of Physics Department of Chemistry Division
X-ray Photoelectron Spectroscopy (XPS)
 X-ray Photoelectron Spectroscopy (XPS) As part of the course Characterization of Catalysts and Surfaces Prof. Dr. Markus Ammann Paul Scherrer Institut markus.ammann@psi.ch Resource for further reading:
X-ray Photoelectron Spectroscopy (XPS) As part of the course Characterization of Catalysts and Surfaces Prof. Dr. Markus Ammann Paul Scherrer Institut markus.ammann@psi.ch Resource for further reading:
Supporting Information. for. Advanced Materials, adma Wiley-VCH 2005
 Supporting Information for Advanced Materials, adma.200501482 Wiley-VCH 2005 69451 Weinheim, Germany Supplementary Material Characterization of SAMs The HBCS monolayers were characterized by XPS, NEXAFS
Supporting Information for Advanced Materials, adma.200501482 Wiley-VCH 2005 69451 Weinheim, Germany Supplementary Material Characterization of SAMs The HBCS monolayers were characterized by XPS, NEXAFS
Probing Matter: Diffraction, Spectroscopy and Photoemission
 Probing Matter: Diffraction, Spectroscopy and Photoemission Anders Nilsson Stanford Synchrotron Radiation Laboratory Why X-rays? VUV? What can we hope to learn? 1 Photon Interaction Incident photon interacts
Probing Matter: Diffraction, Spectroscopy and Photoemission Anders Nilsson Stanford Synchrotron Radiation Laboratory Why X-rays? VUV? What can we hope to learn? 1 Photon Interaction Incident photon interacts
Supporting Information
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Supporting Information A direct Fe-O coordination at FePc/MoO x interface investigated
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Supporting Information A direct Fe-O coordination at FePc/MoO x interface investigated
Lecture 5. X-ray Photoemission Spectroscopy (XPS)
 Lecture 5 X-ray Photoemission Spectroscopy (XPS) 5. Photoemission Spectroscopy (XPS) 5. Principles 5.2 Interpretation 5.3 Instrumentation 5.4 XPS vs UV Photoelectron Spectroscopy (UPS) 5.5 Auger Electron
Lecture 5 X-ray Photoemission Spectroscopy (XPS) 5. Photoemission Spectroscopy (XPS) 5. Principles 5.2 Interpretation 5.3 Instrumentation 5.4 XPS vs UV Photoelectron Spectroscopy (UPS) 5.5 Auger Electron
SUPPLEMENTARY INFORMATION
 DOI: 1.138/NMAT3449 Topological crystalline insulator states in Pb 1 x Sn x Se Content S1 Crystal growth, structural and chemical characterization. S2 Angle-resolved photoemission measurements at various
DOI: 1.138/NMAT3449 Topological crystalline insulator states in Pb 1 x Sn x Se Content S1 Crystal growth, structural and chemical characterization. S2 Angle-resolved photoemission measurements at various
Energy Spectroscopy. Ex.: Fe/MgO
 Energy Spectroscopy Spectroscopy gives access to the electronic properties (and thus chemistry, magnetism,..) of the investigated system with thickness dependence Ex.: Fe/MgO Fe O Mg Control of the oxidation
Energy Spectroscopy Spectroscopy gives access to the electronic properties (and thus chemistry, magnetism,..) of the investigated system with thickness dependence Ex.: Fe/MgO Fe O Mg Control of the oxidation
Electronic Supporting Information for
 Electronic Supplementary Material (ESI) for Materials Horizons. This journal is The Royal Society of Chemistry 2015 Electronic Supporting Information for Probing the Energy Levels in Hole-doped Molecular
Electronic Supplementary Material (ESI) for Materials Horizons. This journal is The Royal Society of Chemistry 2015 Electronic Supporting Information for Probing the Energy Levels in Hole-doped Molecular
Name: (a) What core levels are responsible for the three photoelectron peaks in Fig. 1?
 Physics 243A--Surface Physics of Materials: Spectroscopy Final Examination December 16, 2014 (3 problems, 100 points total, open book, open notes and handouts) Name: [1] (50 points), including Figures
Physics 243A--Surface Physics of Materials: Spectroscopy Final Examination December 16, 2014 (3 problems, 100 points total, open book, open notes and handouts) Name: [1] (50 points), including Figures
Organic semiconductor heterointerfaces containing bathocuproine
 JOURNAL OF APPLIED PHYSICS VOLUME 86, NUMBER 8 15 OCTOBER 1999 Organic semiconductor heterointerfaces containing bathocuproine I. G. Hill a) and A. Kahn Department of Electrical Engineering, Princeton
JOURNAL OF APPLIED PHYSICS VOLUME 86, NUMBER 8 15 OCTOBER 1999 Organic semiconductor heterointerfaces containing bathocuproine I. G. Hill a) and A. Kahn Department of Electrical Engineering, Princeton
On surface synthesis of a 2D boroxine framework: a route to a novel 2D material?
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 On surface synthesis of a 2D boroxine framework: a route to a novel 2D material? Matus Stredansky,
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 On surface synthesis of a 2D boroxine framework: a route to a novel 2D material? Matus Stredansky,
Initial Stages of Growth of Organic Semiconductors on Graphene
 Initial Stages of Growth of Organic Semiconductors on Graphene Presented by: Manisha Chhikara Supervisor: Prof. Dr. Gvido Bratina University of Nova Gorica Outline Introduction to Graphene Fabrication
Initial Stages of Growth of Organic Semiconductors on Graphene Presented by: Manisha Chhikara Supervisor: Prof. Dr. Gvido Bratina University of Nova Gorica Outline Introduction to Graphene Fabrication
Microstructure evolution during BaTiO 3 formation by solid-state reactions on rutile single crystal surfaces
 Journal of the European Ceramic Society 25 (2005) 2201 2206 Microstructure evolution during BaTiO 3 formation by solid-state reactions on rutile single crystal surfaces Andreas Graff a,, Stephan Senz a,
Journal of the European Ceramic Society 25 (2005) 2201 2206 Microstructure evolution during BaTiO 3 formation by solid-state reactions on rutile single crystal surfaces Andreas Graff a,, Stephan Senz a,
X-ray diffraction and Crystal Structure Solutions from Thin Films
 X-ray diffraction and Crystal Structure Solutions from Thin Films Ingo Salzmann Humboldt-Universität zu Berlin Institut für Physik Overview Experimental technique X-ray diffraction The principal phenomenon
X-ray diffraction and Crystal Structure Solutions from Thin Films Ingo Salzmann Humboldt-Universität zu Berlin Institut für Physik Overview Experimental technique X-ray diffraction The principal phenomenon
Electron Rutherford Backscattering, a versatile tool for the study of thin films
 Electron Rutherford Backscattering, a versatile tool for the study of thin films Maarten Vos Research School of Physics and Engineering Australian National University Canberra Australia Acknowledgements:
Electron Rutherford Backscattering, a versatile tool for the study of thin films Maarten Vos Research School of Physics and Engineering Australian National University Canberra Australia Acknowledgements:
MODERN TECHNIQUES OF SURFACE SCIENCE
 MODERN TECHNIQUES OF SURFACE SCIENCE Second edition D. P. WOODRUFF & T. A. DELCHAR Department ofphysics, University of Warwick CAMBRIDGE UNIVERSITY PRESS Contents Preface to first edition Preface to second
MODERN TECHNIQUES OF SURFACE SCIENCE Second edition D. P. WOODRUFF & T. A. DELCHAR Department ofphysics, University of Warwick CAMBRIDGE UNIVERSITY PRESS Contents Preface to first edition Preface to second
Energy level alignment and two-dimensional structure of pentacene on Au 111 surfaces
 JOURNAL OF APPLIED PHYSICS VOLUME 91, NUMBER 5 1 MARCH 2002 Energy level alignment and two-dimensional structure of pentacene on Au 111 surfaces P. G. Schroeder, C. B. France, J. B. Park, and B. A. Parkinson
JOURNAL OF APPLIED PHYSICS VOLUME 91, NUMBER 5 1 MARCH 2002 Energy level alignment and two-dimensional structure of pentacene on Au 111 surfaces P. G. Schroeder, C. B. France, J. B. Park, and B. A. Parkinson
Imaging Methods: Scanning Force Microscopy (SFM / AFM)
 Imaging Methods: Scanning Force Microscopy (SFM / AFM) The atomic force microscope (AFM) probes the surface of a sample with a sharp tip, a couple of microns long and often less than 100 Å in diameter.
Imaging Methods: Scanning Force Microscopy (SFM / AFM) The atomic force microscope (AFM) probes the surface of a sample with a sharp tip, a couple of microns long and often less than 100 Å in diameter.
Reduced preferential sputtering of TiO 2 (and Ta 2 O 5 ) thin films through argon cluster ion bombardment.
 NATIOMEM Reduced preferential sputtering of TiO 2 (and Ta 2 O 5 ) thin films through argon cluster ion bombardment. R. Grilli *, P. Mack, M.A. Baker * * University of Surrey, UK ThermoFisher Scientific
NATIOMEM Reduced preferential sputtering of TiO 2 (and Ta 2 O 5 ) thin films through argon cluster ion bombardment. R. Grilli *, P. Mack, M.A. Baker * * University of Surrey, UK ThermoFisher Scientific
Supplementary Figures
 Supplementary Figures Supplementary Figure 1 Measured versus calculated optical transitions in the CPX. The UV/Vis/NIR spectrum obtained experimentally for the 1:1 blend of 4T and F4TCNQ (red curve) is
Supplementary Figures Supplementary Figure 1 Measured versus calculated optical transitions in the CPX. The UV/Vis/NIR spectrum obtained experimentally for the 1:1 blend of 4T and F4TCNQ (red curve) is
Special Properties of Au Nanoparticles
 Special Properties of Au Nanoparticles Maryam Ebrahimi Chem 7500/750 March 28 th, 2007 1 Outline Introduction The importance of unexpected electronic, geometric, and chemical properties of nanoparticles
Special Properties of Au Nanoparticles Maryam Ebrahimi Chem 7500/750 March 28 th, 2007 1 Outline Introduction The importance of unexpected electronic, geometric, and chemical properties of nanoparticles
Temperature-dependent templated growth of porphine thin films on the (111) facets of copper and silver Supporting Information
 Temperature-dependent templated growth of porphine thin films on the (111) facets of copper and silver Supporting Information Katharina Diller, Florian Klappenberger, Francesco Allegretti, Anthoula C.
Temperature-dependent templated growth of porphine thin films on the (111) facets of copper and silver Supporting Information Katharina Diller, Florian Klappenberger, Francesco Allegretti, Anthoula C.
X-Ray Photoelectron Spectroscopy (XPS)-2
 X-Ray Photoelectron Spectroscopy (XPS)-2 Louis Scudiero http://www.wsu.edu/~scudiero; 5-2669 Fulmer 261A Electron Spectroscopy for Chemical Analysis (ESCA) The 3 step model: 1.Optical excitation 2.Transport
X-Ray Photoelectron Spectroscopy (XPS)-2 Louis Scudiero http://www.wsu.edu/~scudiero; 5-2669 Fulmer 261A Electron Spectroscopy for Chemical Analysis (ESCA) The 3 step model: 1.Optical excitation 2.Transport
Supporting Information. Effect of Backbone Chemistry on the Structure of Polyurea Films Deposited by Molecular Layer Deposition
 Supporting Information Effect of Backbone Chemistry on the Structure of Polyurea Films Deposited by Molecular Layer Deposition David S. Bergsman, Richard G. Closser, Christopher J. Tassone, Bruce M. Clemens
Supporting Information Effect of Backbone Chemistry on the Structure of Polyurea Films Deposited by Molecular Layer Deposition David S. Bergsman, Richard G. Closser, Christopher J. Tassone, Bruce M. Clemens
Transport gap of organic semiconductors in organic modified Schottky contacts
 Applied Surface Science 212 213 (2003) 423 427 Transport gap of organic semiconductors in organic modified Schottky contacts Dietrich R.T. Zahn *, Thorsten U. Kampen, Henry Méndez Institut für Physik,
Applied Surface Science 212 213 (2003) 423 427 Transport gap of organic semiconductors in organic modified Schottky contacts Dietrich R.T. Zahn *, Thorsten U. Kampen, Henry Méndez Institut für Physik,
Introduction to X-ray Photoelectron Spectroscopy (XPS) XPS which makes use of the photoelectric effect, was developed in the mid-1960
 Introduction to X-ray Photoelectron Spectroscopy (XPS) X-ray Photoelectron Spectroscopy (XPS), also known as Electron Spectroscopy for Chemical Analysis (ESCA) is a widely used technique to investigate
Introduction to X-ray Photoelectron Spectroscopy (XPS) X-ray Photoelectron Spectroscopy (XPS), also known as Electron Spectroscopy for Chemical Analysis (ESCA) is a widely used technique to investigate
X-Ray Photoelectron Spectroscopy (XPS)-2
 X-Ray Photoelectron Spectroscopy (XPS)-2 Louis Scudiero http://www.wsu.edu/~pchemlab ; 5-2669 Fulmer 261A Electron Spectroscopy for Chemical Analysis (ESCA) The 3 step model: 1.Optical excitation 2.Transport
X-Ray Photoelectron Spectroscopy (XPS)-2 Louis Scudiero http://www.wsu.edu/~pchemlab ; 5-2669 Fulmer 261A Electron Spectroscopy for Chemical Analysis (ESCA) The 3 step model: 1.Optical excitation 2.Transport
Photoelectron Spectroscopy
 Stefan Hüfner Photoelectron Spectroscopy Principles and Applications Third Revised and Enlarged Edition With 461 Figures and 28 Tables JSJ Springer ... 1. Introduction and Basic Principles 1 1.1 Historical
Stefan Hüfner Photoelectron Spectroscopy Principles and Applications Third Revised and Enlarged Edition With 461 Figures and 28 Tables JSJ Springer ... 1. Introduction and Basic Principles 1 1.1 Historical
M2 TP. Low-Energy Electron Diffraction (LEED)
 M2 TP Low-Energy Electron Diffraction (LEED) Guide for report preparation I. Introduction: Elastic scattering or diffraction of electrons is the standard technique in surface science for obtaining structural
M2 TP Low-Energy Electron Diffraction (LEED) Guide for report preparation I. Introduction: Elastic scattering or diffraction of electrons is the standard technique in surface science for obtaining structural
Spectro-microscopic photoemission evidence of surface dissociation and charge uncompensated areas in Pb(Zr,Ti)O 3 (001) layers
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Electronic Supplementary Information - Phys. Chem. Chem. Phys. Spectro-microscopic
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Electronic Supplementary Information - Phys. Chem. Chem. Phys. Spectro-microscopic
Photoelectron Interference Pattern (PEIP): A Two-particle Bragg-reflection Demonstration
 Photoelectron Interference Pattern (PEIP): A Two-particle Bragg-reflection Demonstration Application No. : 2990 Beamlime: BL25SU Project Leader: Martin Månsson 0017349 Team Members: Dr. Oscar Tjernberg
Photoelectron Interference Pattern (PEIP): A Two-particle Bragg-reflection Demonstration Application No. : 2990 Beamlime: BL25SU Project Leader: Martin Månsson 0017349 Team Members: Dr. Oscar Tjernberg
Electron Spectroscopy
 Electron Spectroscopy Photoelectron spectroscopy is based upon a single photon in/electron out process. The energy of a photon is given by the Einstein relation : E = h ν where h - Planck constant ( 6.62
Electron Spectroscopy Photoelectron spectroscopy is based upon a single photon in/electron out process. The energy of a photon is given by the Einstein relation : E = h ν where h - Planck constant ( 6.62
Molecular Ordering at the Interface Between Liquid Water and Rutile TiO 2 (110)
 Molecular Ordering at the Interface Between Liquid Water and Rutile TiO 2 (110) B E A T R I C E B O N A N N I D i p a r t i m e n t o d i F i s i c a, U n i v e r s i t a di R o m a T o r V e r g a t a
Molecular Ordering at the Interface Between Liquid Water and Rutile TiO 2 (110) B E A T R I C E B O N A N N I D i p a r t i m e n t o d i F i s i c a, U n i v e r s i t a di R o m a T o r V e r g a t a
Surface Analysis - The Principal Techniques
 Surface Analysis - The Principal Techniques Edited by John C. Vickerman Surface Analysis Research Centre, Department of Chemistry UMIST, Manchester, UK JOHN WILEY & SONS Chichester New York Weinheim Brisbane
Surface Analysis - The Principal Techniques Edited by John C. Vickerman Surface Analysis Research Centre, Department of Chemistry UMIST, Manchester, UK JOHN WILEY & SONS Chichester New York Weinheim Brisbane
Surface Sensitivity & Surface Specificity
 Surface Sensitivity & Surface Specificity The problems of sensitivity and detection limits are common to all forms of spectroscopy. In its simplest form, the question of sensitivity boils down to whether
Surface Sensitivity & Surface Specificity The problems of sensitivity and detection limits are common to all forms of spectroscopy. In its simplest form, the question of sensitivity boils down to whether
Advanced Lab Course. X-Ray Photoelectron Spectroscopy 1 INTRODUCTION 1 2 BASICS 1 3 EXPERIMENT Qualitative analysis Chemical Shifts 7
 Advanced Lab Course X-Ray Photoelectron Spectroscopy M210 As of: 2015-04-01 Aim: Chemical analysis of surfaces. Content 1 INTRODUCTION 1 2 BASICS 1 3 EXPERIMENT 3 3.1 Qualitative analysis 6 3.2 Chemical
Advanced Lab Course X-Ray Photoelectron Spectroscopy M210 As of: 2015-04-01 Aim: Chemical analysis of surfaces. Content 1 INTRODUCTION 1 2 BASICS 1 3 EXPERIMENT 3 3.1 Qualitative analysis 6 3.2 Chemical
MSE 321 Structural Characterization
 Auger Spectroscopy Auger Electron Spectroscopy (AES) Scanning Auger Microscopy (SAM) Incident Electron Ejected Electron Auger Electron Initial State Intermediate State Final State Physical Electronics
Auger Spectroscopy Auger Electron Spectroscopy (AES) Scanning Auger Microscopy (SAM) Incident Electron Ejected Electron Auger Electron Initial State Intermediate State Final State Physical Electronics
Modern Methods in Heterogeneous Catalysis Research
 Modern Methods in Heterogeneous Catalysis Research Axel Knop-Gericke, January 09, 2004 In situ X-ray photoelectron spectroscopy (XPS) In situ near edge X-ray absorption fine structure (NEXAFS) in the soft
Modern Methods in Heterogeneous Catalysis Research Axel Knop-Gericke, January 09, 2004 In situ X-ray photoelectron spectroscopy (XPS) In situ near edge X-ray absorption fine structure (NEXAFS) in the soft
Supporting Information
 Temperature Effect on Transport, Charging and Binding of Low-Energy Electrons Interacting with Amorphous Solid Water Films Roey Sagi, Michelle Akerman, Sujith Ramakrishnan and Micha Asscher * Institute
Temperature Effect on Transport, Charging and Binding of Low-Energy Electrons Interacting with Amorphous Solid Water Films Roey Sagi, Michelle Akerman, Sujith Ramakrishnan and Micha Asscher * Institute
Sfb 658 Colloquium 11 May Part II. Introduction to Two-Photon-Photoemission (2PPE) Spectroscopy. Martin Wolf
 Sfb 658 Colloquium 11 May 2006 Part II Introduction to Two-Photon-Photoemission (2PPE) Spectroscopy Martin Wolf Motivation: Electron transfer across interfaces key step for interfacial and surface dynamics
Sfb 658 Colloquium 11 May 2006 Part II Introduction to Two-Photon-Photoemission (2PPE) Spectroscopy Martin Wolf Motivation: Electron transfer across interfaces key step for interfacial and surface dynamics
XPS o ESCA UPS. Photoemission Spectroscopies. Threshold Spectroscopies (NEXAFS, APS etc ) The physics of photoemission.
 XPS o ESCA Photoemission Spectroscopies UPS Threshold Spectroscopies (NEXAFS, APS etc ) The physics of photoemission. How are photoemission spectra recorded: sources and analyzers Semi-quantitative analysis.
XPS o ESCA Photoemission Spectroscopies UPS Threshold Spectroscopies (NEXAFS, APS etc ) The physics of photoemission. How are photoemission spectra recorded: sources and analyzers Semi-quantitative analysis.
The,electronic structure of adsorbed aromatic molecules: Perylene and PTCDA on Si( 111) and Ag( 111)
 Journal of Molecular Structure, 293 (1993) 239-244 Elsevier Science Publishers B.V., Amsterdam 239 The,electronic structure of adsorbed aromatic molecules: Perylene and PTCDA on Si( 111) and Ag( 111) M.
Journal of Molecular Structure, 293 (1993) 239-244 Elsevier Science Publishers B.V., Amsterdam 239 The,electronic structure of adsorbed aromatic molecules: Perylene and PTCDA on Si( 111) and Ag( 111) M.
X-Ray Photoelectron Spectroscopy (XPS)
 X-Ray Photoelectron Spectroscopy (XPS) Louis Scudiero http://www.wsu.edu/~scudiero; 5-2669 Fulmer 261A Electron Spectroscopy for Chemical Analysis (ESCA) The basic principle of the photoelectric effect
X-Ray Photoelectron Spectroscopy (XPS) Louis Scudiero http://www.wsu.edu/~scudiero; 5-2669 Fulmer 261A Electron Spectroscopy for Chemical Analysis (ESCA) The basic principle of the photoelectric effect
Supporting Data. The University of Texas at Dallas, 800 West Campbell Road, Richardson, Texas 75080, United
 Supporting Data MoS 2 Functionalization for Ultra-thin Atomic Layer Deposited Dielectrics Angelica Azcatl, 1 Stephen McDonnell, 1 Santosh KC, 1 Xing Peng, 1 Hong Dong, 1 Xiaoye Qin, 1 Rafik Addou, 1 Greg
Supporting Data MoS 2 Functionalization for Ultra-thin Atomic Layer Deposited Dielectrics Angelica Azcatl, 1 Stephen McDonnell, 1 Santosh KC, 1 Xing Peng, 1 Hong Dong, 1 Xiaoye Qin, 1 Rafik Addou, 1 Greg
Crystalline Surfaces for Laser Metrology
 Crystalline Surfaces for Laser Metrology A.V. Latyshev, Institute of Semiconductor Physics SB RAS, Novosibirsk, Russia Abstract: The number of methodological recommendations has been pronounced to describe
Crystalline Surfaces for Laser Metrology A.V. Latyshev, Institute of Semiconductor Physics SB RAS, Novosibirsk, Russia Abstract: The number of methodological recommendations has been pronounced to describe
Secondary Ion Mass Spectrometry (SIMS)
 CHEM53200: Lecture 10 Secondary Ion Mass Spectrometry (SIMS) Major reference: Surface Analysis Edited by J. C. Vickerman (1997). 1 Primary particles may be: Secondary particles can be e s, neutral species
CHEM53200: Lecture 10 Secondary Ion Mass Spectrometry (SIMS) Major reference: Surface Analysis Edited by J. C. Vickerman (1997). 1 Primary particles may be: Secondary particles can be e s, neutral species
Ted Madey s Scientific Career at NBS/NIST: Aspects of Auger Electron Spectroscopy (AES), X-ray Photoelectron Spectroscopy (XPS), and Vacuum Science
 Ted Madey s Scientific Career at NBS/NIST: Aspects of Auger Electron Spectroscopy (AES), X-ray Photoelectron Spectroscopy (XPS), and Vacuum Science Cedric J. Powell 1. Ted s 25-year career at NBS/NIST:
Ted Madey s Scientific Career at NBS/NIST: Aspects of Auger Electron Spectroscopy (AES), X-ray Photoelectron Spectroscopy (XPS), and Vacuum Science Cedric J. Powell 1. Ted s 25-year career at NBS/NIST:
5) Surface photoelectron spectroscopy. For MChem, Spring, Dr. Qiao Chen (room 3R506) University of Sussex.
 For MChem, Spring, 2009 5) Surface photoelectron spectroscopy Dr. Qiao Chen (room 3R506) http://www.sussex.ac.uk/users/qc25/ University of Sussex Today s topics 1. Element analysis with XPS Binding energy,
For MChem, Spring, 2009 5) Surface photoelectron spectroscopy Dr. Qiao Chen (room 3R506) http://www.sussex.ac.uk/users/qc25/ University of Sussex Today s topics 1. Element analysis with XPS Binding energy,
1 Introduction COPYRIGHTED MATERIAL. 1.1 HowdoweDefinetheSurface?
 1 Introduction JOHN C. VICKERMAN Manchester Interdisciplinary Biocentre, School of Chemical Engineering and Analytical Science, The University of Manchester, Manchester, UK The surface behaviour of materials
1 Introduction JOHN C. VICKERMAN Manchester Interdisciplinary Biocentre, School of Chemical Engineering and Analytical Science, The University of Manchester, Manchester, UK The surface behaviour of materials
Evidence for partial dissociation of water on flat MgO(1 0 0) surfaces
 6 February 2002 Chemical Physics Letters 352 (2002) 318 322 www.elsevier.com/locate/cplett Evidence for partial dissociation of water on flat MgO(1 0 0) surfaces Y.D. Kim a, R.M. Lynden-Bell b, *, A. Alavi
6 February 2002 Chemical Physics Letters 352 (2002) 318 322 www.elsevier.com/locate/cplett Evidence for partial dissociation of water on flat MgO(1 0 0) surfaces Y.D. Kim a, R.M. Lynden-Bell b, *, A. Alavi
Pre-nucleation dynamics of organic molecule self-assembly investigated by PEEM
 PCCP Dynamic Article Links View Online Cite this: DOI: 10.1039/c0cp01516d www.rsc.org/pccp PAPER Pre-nucleation dynamics of organic molecule self-assembly investigated by PEEM Alexander J. Fleming,* Stephen
PCCP Dynamic Article Links View Online Cite this: DOI: 10.1039/c0cp01516d www.rsc.org/pccp PAPER Pre-nucleation dynamics of organic molecule self-assembly investigated by PEEM Alexander J. Fleming,* Stephen
X- ray Photoelectron Spectroscopy and its application in phase- switching device study
 X- ray Photoelectron Spectroscopy and its application in phase- switching device study Xinyuan Wang A53073806 I. Background X- ray photoelectron spectroscopy is of great importance in modern chemical and
X- ray Photoelectron Spectroscopy and its application in phase- switching device study Xinyuan Wang A53073806 I. Background X- ray photoelectron spectroscopy is of great importance in modern chemical and
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/29891 holds various files of this Leiden University dissertation Author: Roobol, Sander Bas Title: The structure of a working catalyst : from flat surfaces
Cover Page The handle http://hdl.handle.net/1887/29891 holds various files of this Leiden University dissertation Author: Roobol, Sander Bas Title: The structure of a working catalyst : from flat surfaces
Supplementary Figure 1 Experimental setup for crystal growth. Schematic drawing of the experimental setup for C 8 -BTBT crystal growth.
 Supplementary Figure 1 Experimental setup for crystal growth. Schematic drawing of the experimental setup for C 8 -BTBT crystal growth. Supplementary Figure 2 AFM study of the C 8 -BTBT crystal growth
Supplementary Figure 1 Experimental setup for crystal growth. Schematic drawing of the experimental setup for C 8 -BTBT crystal growth. Supplementary Figure 2 AFM study of the C 8 -BTBT crystal growth
Nanostructure Fabrication Using Selective Growth on Nanosize Patterns Drawn by a Scanning Probe Microscope
 Nanostructure Fabrication Using Selective Growth on Nanosize Patterns Drawn by a Scanning Probe Microscope Kentaro Sasaki, Keiji Ueno and Atsushi Koma Department of Chemistry, The University of Tokyo,
Nanostructure Fabrication Using Selective Growth on Nanosize Patterns Drawn by a Scanning Probe Microscope Kentaro Sasaki, Keiji Ueno and Atsushi Koma Department of Chemistry, The University of Tokyo,
ELECTRON CURRENT IMAGE DIFFRACTION FROM CRYSTAL SURFACES AT LOW ENERGIES
 C. BRENT BARGERON, A. NORMAN JETTE, and BERRY H. NALL ELECTRON CURRENT IMAGE DIFFRACTION FROM CRYSTAL SURFACES AT LOW ENERGIES Low-energy electron diffraction patterns in current images of crystal surfaces
C. BRENT BARGERON, A. NORMAN JETTE, and BERRY H. NALL ELECTRON CURRENT IMAGE DIFFRACTION FROM CRYSTAL SURFACES AT LOW ENERGIES Low-energy electron diffraction patterns in current images of crystal surfaces
Photoelectron spectroscopy Instrumentation. Nanomaterials characterization 2
 Photoelectron spectroscopy Instrumentation Nanomaterials characterization 2 RNDr. Věra V Vodičkov ková,, PhD. Photoelectron Spectroscopy general scheme Impact of X-ray emitted from source to the sample
Photoelectron spectroscopy Instrumentation Nanomaterials characterization 2 RNDr. Věra V Vodičkov ková,, PhD. Photoelectron Spectroscopy general scheme Impact of X-ray emitted from source to the sample
Energy-level alignment at interfaces between metals and the organic semiconductor 4,4 -N,N -dicarbazolyl-biphenyl
 JOURNAL OF APPLIED PHYSICS VOLUME 84, NUMBER 6 15 SEPTEMBER 1998 Energy-level alignment at interfaces between metals and the organic semiconductor 4,4 -N,N -dicarbazolyl-biphenyl I. G. Hill, a) A. Rajagopal,
JOURNAL OF APPLIED PHYSICS VOLUME 84, NUMBER 6 15 SEPTEMBER 1998 Energy-level alignment at interfaces between metals and the organic semiconductor 4,4 -N,N -dicarbazolyl-biphenyl I. G. Hill, a) A. Rajagopal,
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Towards wafer-size graphene layers by atmospheric pressure graphitization of silicon carbide Supporting online material Konstantin V. Emtsev 1, Aaron Bostwick 2, Karsten Horn
SUPPLEMENTARY INFORMATION Towards wafer-size graphene layers by atmospheric pressure graphitization of silicon carbide Supporting online material Konstantin V. Emtsev 1, Aaron Bostwick 2, Karsten Horn
Surface Transfer Doping of Diamond by Organic Molecules
 Surface Transfer Doping of Diamond by Organic Molecules Qi Dongchen Department of Physics National University of Singapore Supervisor: Prof. Andrew T. S. Wee Dr. Gao Xingyu Scope of presentation Overview
Surface Transfer Doping of Diamond by Organic Molecules Qi Dongchen Department of Physics National University of Singapore Supervisor: Prof. Andrew T. S. Wee Dr. Gao Xingyu Scope of presentation Overview
Stability of Organic Cations in Solution-Processed CH3NH3PbI3 Perovskites: Formation of Modified Surface Layers
 Stability of Organic Cations in Solution-Processed CH3NH3PbI3 Perovskites: Formation of Modified Surface Layers A. Calloni,*, A. Abate, G. Bussetti, G. Berti, R. Yivlialin, F. Ciccacci, and L. Duò Dipartimento
Stability of Organic Cations in Solution-Processed CH3NH3PbI3 Perovskites: Formation of Modified Surface Layers A. Calloni,*, A. Abate, G. Bussetti, G. Berti, R. Yivlialin, F. Ciccacci, and L. Duò Dipartimento
X-Ray Photoelectron Spectroscopy (XPS) Prof. Paul K. Chu
 X-Ray Photoelectron Spectroscopy (XPS) Prof. Paul K. Chu X-ray Photoelectron Spectroscopy Introduction Qualitative analysis Quantitative analysis Charging compensation Small area analysis and XPS imaging
X-Ray Photoelectron Spectroscopy (XPS) Prof. Paul K. Chu X-ray Photoelectron Spectroscopy Introduction Qualitative analysis Quantitative analysis Charging compensation Small area analysis and XPS imaging
Scanning Tunneling Microscopy Studies of the Ge(111) Surface
 VC Scanning Tunneling Microscopy Studies of the Ge(111) Surface Anna Rosen University of California, Berkeley Advisor: Dr. Shirley Chiang University of California, Davis August 24, 2007 Abstract: This
VC Scanning Tunneling Microscopy Studies of the Ge(111) Surface Anna Rosen University of California, Berkeley Advisor: Dr. Shirley Chiang University of California, Davis August 24, 2007 Abstract: This
The Benefit of Wide Energy Range Spectrum Acquisition During Sputter Depth Profile Measurements
 The Benefit of Wide Energy Range Spectrum Acquisition During Sputter Depth Profile Measurements Uwe Scheithauer, 82008 Unterhaching, Germany E-Mail: scht.uhg@googlemail.com Internet: orcid.org/0000-0002-4776-0678;
The Benefit of Wide Energy Range Spectrum Acquisition During Sputter Depth Profile Measurements Uwe Scheithauer, 82008 Unterhaching, Germany E-Mail: scht.uhg@googlemail.com Internet: orcid.org/0000-0002-4776-0678;
Structure analysis: Electron diffraction LEED TEM RHEED
 Structure analysis: Electron diffraction LEED: Low Energy Electron Diffraction SPA-LEED: Spot Profile Analysis Low Energy Electron diffraction RHEED: Reflection High Energy Electron Diffraction TEM: Transmission
Structure analysis: Electron diffraction LEED: Low Energy Electron Diffraction SPA-LEED: Spot Profile Analysis Low Energy Electron diffraction RHEED: Reflection High Energy Electron Diffraction TEM: Transmission
X-ray Standing-Wave Investigation of (1X2)Rb/ Cu(110)
 University of Rhode Island DigitalCommons@URI Physics Faculty Publications Physics 2000 X-ray Standing-Wave Investigation of (1X2)Rb/ Cu(110) David R. Heskett University of Rhode Island, dheskett@uri.edu
University of Rhode Island DigitalCommons@URI Physics Faculty Publications Physics 2000 X-ray Standing-Wave Investigation of (1X2)Rb/ Cu(110) David R. Heskett University of Rhode Island, dheskett@uri.edu
Surface Characte i r i zat on LEED Photoemission Phot Linear optics
 Surface Characterization i LEED Photoemission Linear optics Surface characterization with electrons MPS M.P. Seah, WA W.A. Dench, Surf. Interf. Anal. 1 (1979) 2 LEED low energy electron diffraction De
Surface Characterization i LEED Photoemission Linear optics Surface characterization with electrons MPS M.P. Seah, WA W.A. Dench, Surf. Interf. Anal. 1 (1979) 2 LEED low energy electron diffraction De
Spin pumping in Ferromagnet-Topological Insulator-Ferromagnet Heterostructures Supplementary Information.
 Spin pumping in Ferromagnet-Topological Insulator-Ferromagnet Heterostructures Supplementary Information. A.A. Baker,, 2 A.I. Figueroa, 2 L.J. Collins-McIntyre, G. van der Laan, 2 and T., a) Hesjedal )
Spin pumping in Ferromagnet-Topological Insulator-Ferromagnet Heterostructures Supplementary Information. A.A. Baker,, 2 A.I. Figueroa, 2 L.J. Collins-McIntyre, G. van der Laan, 2 and T., a) Hesjedal )
X-Ray Photoelectron Spectroscopy (XPS)
 X-Ray Photoelectron Spectroscopy (XPS) Louis Scudiero http://www.wsu.edu/~scudiero; 5-2669 Electron Spectroscopy for Chemical Analysis (ESCA) The basic principle of the photoelectric effect was enunciated
X-Ray Photoelectron Spectroscopy (XPS) Louis Scudiero http://www.wsu.edu/~scudiero; 5-2669 Electron Spectroscopy for Chemical Analysis (ESCA) The basic principle of the photoelectric effect was enunciated
Quantum Condensed Matter Physics Lecture 12
 Quantum Condensed Matter Physics Lecture 12 David Ritchie QCMP Lent/Easter 2016 http://www.sp.phy.cam.ac.uk/drp2/home 12.1 QCMP Course Contents 1. Classical models for electrons in solids 2. Sommerfeld
Quantum Condensed Matter Physics Lecture 12 David Ritchie QCMP Lent/Easter 2016 http://www.sp.phy.cam.ac.uk/drp2/home 12.1 QCMP Course Contents 1. Classical models for electrons in solids 2. Sommerfeld
Lecture 23 X-Ray & UV Techniques
 Lecture 23 X-Ray & UV Techniques Schroder: Chapter 11.3 1/50 Announcements Homework 6/6: Will be online on later today. Due Wednesday June 6th at 10:00am. I will return it at the final exam (14 th June).
Lecture 23 X-Ray & UV Techniques Schroder: Chapter 11.3 1/50 Announcements Homework 6/6: Will be online on later today. Due Wednesday June 6th at 10:00am. I will return it at the final exam (14 th June).
Molecular Dynamics on the Angstrom Scale
 Probing Interface Reactions by STM: Molecular Dynamics on the Angstrom Scale Zhisheng Li Prof. Richard Osgood Laboratory for Light-Surface Interactions, Columbia University Outline Motivation: Why do we
Probing Interface Reactions by STM: Molecular Dynamics on the Angstrom Scale Zhisheng Li Prof. Richard Osgood Laboratory for Light-Surface Interactions, Columbia University Outline Motivation: Why do we
Substrate-mediated band-dispersion of adsorbate molecular states - Supplementary Information
 Substrate-mediated band-dispersion of adsorbate molecular states - Supplementary Information M. Wießner, 1, 2 J. Ziroff, 1, 2 F. Forster, 1, 2 M. Arita, 3 K. Shimada, 3 P. Puschnig, 4 A. Schöll*, 1, 2,
Substrate-mediated band-dispersion of adsorbate molecular states - Supplementary Information M. Wießner, 1, 2 J. Ziroff, 1, 2 F. Forster, 1, 2 M. Arita, 3 K. Shimada, 3 P. Puschnig, 4 A. Schöll*, 1, 2,
Metal Organic interfaces
 ORGANIC ELECTRONICS Principles, devices and applications Metal Organic interfaces D. Natali Milano, 23-27 Novembre 2015 Outline general concepts energetics Interfaces: tailoring injection mechanisms Thermal
ORGANIC ELECTRONICS Principles, devices and applications Metal Organic interfaces D. Natali Milano, 23-27 Novembre 2015 Outline general concepts energetics Interfaces: tailoring injection mechanisms Thermal
Electronic Supplementary Information. Molecular Antenna Tailored Organic Thin-film Transistor for. Sensing Application
 Electronic Supplementary Material (ESI) for Materials Horizons. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Molecular Antenna Tailored Organic Thin-film Transistor
Electronic Supplementary Material (ESI) for Materials Horizons. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Molecular Antenna Tailored Organic Thin-film Transistor
Vibrational Spectroscopies. C-874 University of Delaware
 Vibrational Spectroscopies C-874 University of Delaware Vibrational Spectroscopies..everything that living things do can be understood in terms of the jigglings and wigglings of atoms.. R. P. Feymann Vibrational
Vibrational Spectroscopies C-874 University of Delaware Vibrational Spectroscopies..everything that living things do can be understood in terms of the jigglings and wigglings of atoms.. R. P. Feymann Vibrational
Characterisation of vibrational modes of adsorbed species
 17.7.5 Characterisation of vibrational modes of adsorbed species Infrared spectroscopy (IR) See Ch.10. Infrared vibrational spectra originate in transitions between discrete vibrational energy levels of
17.7.5 Characterisation of vibrational modes of adsorbed species Infrared spectroscopy (IR) See Ch.10. Infrared vibrational spectra originate in transitions between discrete vibrational energy levels of
Ma5: Auger- and Electron Energy Loss Spectroscopy
 Ma5: Auger- and Electron Energy Loss Spectroscopy 1 Introduction Electron spectroscopies, namely Auger electron- and electron energy loss spectroscopy are utilized to determine the KLL spectrum and the
Ma5: Auger- and Electron Energy Loss Spectroscopy 1 Introduction Electron spectroscopies, namely Auger electron- and electron energy loss spectroscopy are utilized to determine the KLL spectrum and the
Covalent assembly of a two-dimensional molecular sponge on a Cu(111) surface: Confined electronic surface states in open and closed pores
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting information: Covalent assembly of a two-dimensional molecular sponge on a Cu(111) surface:
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting information: Covalent assembly of a two-dimensional molecular sponge on a Cu(111) surface:
The Use of Synchrotron Radiation in Modern Research
 The Use of Synchrotron Radiation in Modern Research Physics Chemistry Structural Biology Materials Science Geochemical and Environmental Science Atoms, molecules, liquids, solids. Electronic and geometric
The Use of Synchrotron Radiation in Modern Research Physics Chemistry Structural Biology Materials Science Geochemical and Environmental Science Atoms, molecules, liquids, solids. Electronic and geometric
Supporting Information
 Supporting Information Non-Fullerene/Fullerene Acceptor Blend with Tunable Energy State for High- Performance Ternary Organic Solar Cells Min Kim 1, Jaewon Lee 1, Dong Hun Sin 1, Hansol Lee 1, Han Young
Supporting Information Non-Fullerene/Fullerene Acceptor Blend with Tunable Energy State for High- Performance Ternary Organic Solar Cells Min Kim 1, Jaewon Lee 1, Dong Hun Sin 1, Hansol Lee 1, Han Young
PHI 5000 Versaprobe-II Focus X-ray Photo-electron Spectroscopy
 PHI 5000 Versaprobe-II Focus X-ray Photo-electron Spectroscopy The very basic theory of XPS XPS theroy Surface Analysis Ultra High Vacuum (UHV) XPS Theory XPS = X-ray Photo-electron Spectroscopy X-ray
PHI 5000 Versaprobe-II Focus X-ray Photo-electron Spectroscopy The very basic theory of XPS XPS theroy Surface Analysis Ultra High Vacuum (UHV) XPS Theory XPS = X-ray Photo-electron Spectroscopy X-ray
Ag-Diffusion in the Organic Semiconductor Diindenoperylene
 Defect and Diffusion Forum Online: 2005-04-30 ISSN: 1662-9507, Vols. 237-240, pp 993-997 doi:10.4028/www.scientific.net/ddf.237-240.993 2005 Trans Tech Publications, Switzerland Ag-Diffusion in the Organic
Defect and Diffusion Forum Online: 2005-04-30 ISSN: 1662-9507, Vols. 237-240, pp 993-997 doi:10.4028/www.scientific.net/ddf.237-240.993 2005 Trans Tech Publications, Switzerland Ag-Diffusion in the Organic
Spectroscopy of Nanostructures. Angle-resolved Photoemission (ARPES, UPS)
 Spectroscopy of Nanostructures Angle-resolved Photoemission (ARPES, UPS) Measures all quantum numbers of an electron in a solid. E, k x,y, z, point group, spin E kin, ϑ,ϕ, hν, polarization, spin Electron
Spectroscopy of Nanostructures Angle-resolved Photoemission (ARPES, UPS) Measures all quantum numbers of an electron in a solid. E, k x,y, z, point group, spin E kin, ϑ,ϕ, hν, polarization, spin Electron
The Chemical Control of Superconductivity in Bi 2 Sr 2 (Ca 1 x Y x )Cu 2 O 8+±
 CHINESE JOURNAL OF PHYSICS VOL. 38, NO. 2-II APRIL 2000 The Chemical Control of Superconductivity in Bi 2 Sr 2 (Ca 1 x Y x )Cu 2 O 8+± R. S. Liu 1, I. J. Hsu 1, J. M. Chen 2, and R. G. Liu 2 1 Department
CHINESE JOURNAL OF PHYSICS VOL. 38, NO. 2-II APRIL 2000 The Chemical Control of Superconductivity in Bi 2 Sr 2 (Ca 1 x Y x )Cu 2 O 8+± R. S. Liu 1, I. J. Hsu 1, J. M. Chen 2, and R. G. Liu 2 1 Department
PEEM and XPEEM: methodology and applications for dynamic processes
 PEEM and XPEEM: methodology and applications for dynamic processes PEEM methods and General considerations Chemical imaging Magnetic imaging XMCD/XMLD Examples Dynamic studies PEEM and XPEEM methods 1
PEEM and XPEEM: methodology and applications for dynamic processes PEEM methods and General considerations Chemical imaging Magnetic imaging XMCD/XMLD Examples Dynamic studies PEEM and XPEEM methods 1
Fundamentals of Nanoscale Film Analysis
 Fundamentals of Nanoscale Film Analysis Terry L. Alford Arizona State University Tempe, AZ, USA Leonard C. Feldman Vanderbilt University Nashville, TN, USA James W. Mayer Arizona State University Tempe,
Fundamentals of Nanoscale Film Analysis Terry L. Alford Arizona State University Tempe, AZ, USA Leonard C. Feldman Vanderbilt University Nashville, TN, USA James W. Mayer Arizona State University Tempe,
The Diffusion of H in Mg and the Nucleation and Growth of MgH2 in Thin Films*
 - Zeitschrift für Physikalische Chemie, Bd. 181, S. 393-397 (1993) by R. Oldenbourg Verlag, München 1993 0942-9352/93 $ 3.00 + 0.00 The Diffusion of H in Mg and the Nucleation and Growth of MgH2 in Thin
- Zeitschrift für Physikalische Chemie, Bd. 181, S. 393-397 (1993) by R. Oldenbourg Verlag, München 1993 0942-9352/93 $ 3.00 + 0.00 The Diffusion of H in Mg and the Nucleation and Growth of MgH2 in Thin
ORGANIC SEMICONDUCTOR 3,4,9,10-Perylenetetracarboxylic dianhydride (PTCDA)
 ORGANIC SEMICONDUCTOR 3,4,9,10-Perylenetetracarboxylic dianhydride (PTCDA) Suvranta Tripathy Department of Physics University of Cincinnati Cincinnati, Ohio 45221 March 8, 2002 Abstract In the last decade
ORGANIC SEMICONDUCTOR 3,4,9,10-Perylenetetracarboxylic dianhydride (PTCDA) Suvranta Tripathy Department of Physics University of Cincinnati Cincinnati, Ohio 45221 March 8, 2002 Abstract In the last decade
Chemical Vapor Deposition Graphene Grown on Peeled- Off Epitaxial Cu(111) Foil: A Simple Approach to Improved Properties
 Supplementary information Chemical Vapor Deposition Graphene Grown on Peeled- Off Epitaxial Cu(111) Foil: A Simple Approach to Improved Properties Hak Ki Yu 1,2, Kannan Balasubramanian 3, Kisoo Kim 4,
Supplementary information Chemical Vapor Deposition Graphene Grown on Peeled- Off Epitaxial Cu(111) Foil: A Simple Approach to Improved Properties Hak Ki Yu 1,2, Kannan Balasubramanian 3, Kisoo Kim 4,
Effect of Spiral Microwave Antenna Configuration on the Production of Nano-crystalline Film by Chemical Sputtering in ECR Plasma
 THE HARRIS SCIENCE REVIEW OF DOSHISHA UNIVERSITY, VOL. 56, No. 1 April 2015 Effect of Spiral Microwave Antenna Configuration on the Production of Nano-crystalline Film by Chemical Sputtering in ECR Plasma
THE HARRIS SCIENCE REVIEW OF DOSHISHA UNIVERSITY, VOL. 56, No. 1 April 2015 Effect of Spiral Microwave Antenna Configuration on the Production of Nano-crystalline Film by Chemical Sputtering in ECR Plasma
Set-up for ultrafast time-resolved x-ray diffraction using a femtosecond laser-plasma kev x-ray-source
 Set-up for ultrafast time-resolved x-ray diffraction using a femtosecond laser-plasma kev x-ray-source C. Blome, K. Sokolowski-Tinten *, C. Dietrich, A. Tarasevitch, D. von der Linde Inst. for Laser- and
Set-up for ultrafast time-resolved x-ray diffraction using a femtosecond laser-plasma kev x-ray-source C. Blome, K. Sokolowski-Tinten *, C. Dietrich, A. Tarasevitch, D. von der Linde Inst. for Laser- and
Photovoltaics. Lecture 7 Organic Thin Film Solar Cells Photonics - Spring 2017 dr inż. Aleksander Urbaniak
 Photovoltaics Lecture 7 Organic Thin Film Solar Cells Photonics - Spring 2017 dr inż. Aleksander Urbaniak Barcelona, Spain Perpignan train station, France source: pinterest Why organic solar cells? 1.
Photovoltaics Lecture 7 Organic Thin Film Solar Cells Photonics - Spring 2017 dr inż. Aleksander Urbaniak Barcelona, Spain Perpignan train station, France source: pinterest Why organic solar cells? 1.
