1. weak acid in water 2. weak base in water 3. strong acid in water 4. strong base in water 5. buffer
|
|
|
- Ann Berry
- 5 years ago
- Views:
Transcription
1 A 15-mL sample of 0.1 M HA (a weak acid) is titrated with 0.3 M NaH. alculate the ph with 5 ml of NaH added past the equivalence point. 1 This is a problem. 1. weak acid in water 2. weak base in water 3. strong acid in water 4. strong base in water 5. buffer
2 A 15-mL sample of 0.1 M HA (a weak acid) is titrated with 0.3 M NaH. alculate the ph with 5 ml of NaH added past the equivalence point. 2 This is a problem. 10% 1. weak acid in water 15% 1% 63% 10% 2. weak base in water 3. strong acid in water 4. strong base in water 5. buffer
3 Using the periodic table information below, pick the correct Lewis Structure(s) for
4 Using the periodic table information below, pick the correct Lewis Structure(s) for % % 6% 2% 4%
5 5 For 2 with the Lewis structure which of the following are true: A. Geometry is AX 2 and linear B. Geometry is AX 4 and bent U. The hybridization of and are 2sp V. The hybridization of and are 2sp 2 W. The hybridization of is 2sp and of is 2sp 2 X. The hybridization of is 2sp and of is none 1. A and U 2. A and V 3. A and W 4. A and X 5. B and U 6. B and V 7. B and W 8. B and X
6 6 For 2 with the Lewis structure which of the following are true: A. Geometry is AX 2 and linear B. Geometry is AX 4 and bent U. The hybridization of and are 2sp V. The hybridization of and are 2sp 2 W. The hybridization of is 2sp and of is 2sp 2 X. The hybridization of is 2sp and of is none 7% 1. A and U 9% 71% 12% 0% 0% 0% 1% 2. A and V 3. A and W 4. A and X 5. B and U 6. B and V 7. B and W 8. B and X
7 For 2 with the Lewis structure and the information that the χ of is 2.55 and χ of is 3.44, which of the following is true: is a polar molecule: electronegativity difference of and is > is a nonpolar molecule: electronegativity difference of and is > is a nonpolar molecule. It has polar bonds but no net dipole is a polar molecule because it has polar bonds and is linear.
8 For 2 with the Lewis structure and the information that the χ of is 2.55 and χ of is 3.44, which of the following is true: is a polar molecule: electronegativity difference of and is > is a nonpolar molecule: electronegativity difference of and is > is a nonpolar molecule. It has polar bonds but no net dipole is a polar molecule because it has polar bonds and is linear % 5% 89% 0%
9 9 2 H 3 + oa H 3 oa biofuels 2 What redox process is happening to carbon of the 2? 1. Both carbons are being reduced from +4 to The blue is being reduced from +4 to -2 and the red is being reduced from +4 to Both carbons are being oxidized from +4 to No redox change is happening.
10 10 2 H 3 + oa H 3 oa biofuels 2 What redox process is happening to carbon of the 2? 11% 1. Both carbons are being reduced from +4 to +2 83% 4% 1% 2. The blue is being reduced from +4 to -2 and the red is being reduced from +4 to Both carbons are being oxidized from +4 to No redox change is happening.
11 At physiological ph (7.4), how much folic acid is protonated? 11 pk a for folic acid is More folic acid is protonated than deprotonated. 2. More folic acid is deprotonated than protonated. 3. The amounts of deprotonated and protonated are about equal. 4. Not enough information is provided.
12 At physiological ph (7.4), how much folic acid is protonated? 12 pk a for folic acid is % 75% 2% 1% 1. More folic acid is protonated than deprotonated. 2. More folic acid is deprotonated than protonated. 3. The amounts of deprotonated and protonated are about equal. 4. Not enough information is provided.
13 Tie-breaker Question ml of 0.10 M HH with 0.15 M NaH (K a = 1.77 x 10-4 for HH) ph = -log [ ] = (to how many sig figs?)
14 Tie-breaker Question ml of 0.10 M HH with 0.15 M NaH (K a = 1.77 x 10-4 for HH) ph = -log [ ] = (to how many sig figs?) % 25% 25% 25%
15 Tiebreaker question 2: Which is true for this structure? Select the most complete true answer Formula type is AX4. 2. Formula type is AX4E. 3. SN is SN is Geometry is pyramidal. 6. Geometry is see-saw. 7. 1, 3, and 5 are all true. 8. 2, 4, and 6 are all true. 9. 2, 3, and 6 are all true. 10
16 Tiebreaker question 2: Which is true for this structure? Select the most complete true answer % 1. Formula type is AX4. 11% 11% 11% 11% 11% 11% 11% 11% 2. Formula type is AX4E. 3. SN is SN is Geometry is pyramidal. 6. Geometry is see-saw. 7. 1, 3, and 5 are all true. 8. 2, 4, and 6 are all true. 9. 2, 3, and 6 are all true. 10
17 Indicate which of the following statements about catalysts are true. A) In an equilibrium process, a catalyst increases the rate of the forward reaction, but leaves the rate of the reverse reaction unchanged. B) A catalyst is not consumed in the course of a reaction. ) A catalyst must be carefully chosen to shift the equilibrium toward products nly statement A is true. 2. nly statement B is true. 3. nly statement is true. 4. A and B are true. 5. B and are true. 6. A and are true. 7. All of the statements are true. 8. None of the statements are true. 10
18 Indicate which of the following statements about catalysts are true. A) In an equilibrium process, a catalyst increases the rate of the forward reaction, but leaves the rate of the reverse reaction unchanged. B) A catalyst is not consumed in the course of a reaction. ) A catalyst must be carefully chosen to shift the equilibrium toward products E % 1.60E-02 7% 16% 1.60E E E-02 nly statement A is true. 2. nly statement B is true. 3. nly statement is true. 4. A and B are true. 5. B and are true. 6. A and are true. 7. All of the statements are true. 8. None of the statements are true. 10
19 MIT penourseware Principles of hemical Science Fall 2014 For information about citing these materials or our Terms of Use, visit:
5.111 Principles of Chemical Science
 MIT penourseware http://ocw.mit.edu 5.111 Principles of hemical Science all 2008 or information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.111 Lecture Summary
MIT penourseware http://ocw.mit.edu 5.111 Principles of hemical Science all 2008 or information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.111 Lecture Summary
AP Chemistry - Problem Drill 15: Lewis Structures and VSEPR Theory
 AP Chemistry - Problem Drill 15: Lewis Structures and VSEPR Theory No. 1 of 10 1. Which shape would have sp 3 hybridization? (A) Linear (B) Bent (C) Tetrahedron (D) Trigonal planar (E) Octahedron C. Correct.
AP Chemistry - Problem Drill 15: Lewis Structures and VSEPR Theory No. 1 of 10 1. Which shape would have sp 3 hybridization? (A) Linear (B) Bent (C) Tetrahedron (D) Trigonal planar (E) Octahedron C. Correct.
Chapters 8 and 9. Octet Rule Breakers Shapes
 Chapters 8 and 9 Octet Rule Breakers Shapes Bond Energies Bond Energy (review): The energy needed to break one mole of covalent bonds in the gas phase Breaking bonds consumes energy; forming bonds releases
Chapters 8 and 9 Octet Rule Breakers Shapes Bond Energies Bond Energy (review): The energy needed to break one mole of covalent bonds in the gas phase Breaking bonds consumes energy; forming bonds releases
5.111 Lecture Summary #35 Wednesday, December 10, 2014
 5111 Lecture Summary #35 Wednesday, December 10, 2014 Topic: Applying Chemistry Knowledge: A Semester in Review A look back at the course objectives: My goal is for 5111 students to have a working knowledge
5111 Lecture Summary #35 Wednesday, December 10, 2014 Topic: Applying Chemistry Knowledge: A Semester in Review A look back at the course objectives: My goal is for 5111 students to have a working knowledge
Chemical Bonding II: Molecular Geometry and Hybridization of Atomic Orbitals
 Chemical Bonding II: Molecular Geometry and Hybridization of Atomic Orbitals 1 Chemical Bonding II Molecular Geometry (10.1) Dipole Moments (10.2) Valence Bond Theory (10.3) Hybridization of Atomic Orbitals
Chemical Bonding II: Molecular Geometry and Hybridization of Atomic Orbitals 1 Chemical Bonding II Molecular Geometry (10.1) Dipole Moments (10.2) Valence Bond Theory (10.3) Hybridization of Atomic Orbitals
Electron Geometry Hybrid Orbitals
 Molecular Shape and Hybridized Orbitals CH2000: Introduction to General Chemistry, Plymouth State University Introduction: In chemistry, the three dimensional shape of a molecule is as important as the
Molecular Shape and Hybridized Orbitals CH2000: Introduction to General Chemistry, Plymouth State University Introduction: In chemistry, the three dimensional shape of a molecule is as important as the
Form J. Test #4 Last Name First Name Zumdahl, Chapters 8 and 9 November 23, 2004
 Form J Chemistry 1441-023 Name (please print) Test #4 Last Name First Name Zumdahl, Chapters 8 and 9 November 23, 2004 Instructions: 1. This exam consists of 27 questions. 2. No scratch paper is allowed.
Form J Chemistry 1441-023 Name (please print) Test #4 Last Name First Name Zumdahl, Chapters 8 and 9 November 23, 2004 Instructions: 1. This exam consists of 27 questions. 2. No scratch paper is allowed.
Lecture outline: Section 9. theory 2. Valence bond theory 3. Molecular orbital theory. S. Ensign, Chem. 1210
 Lecture outline: Section 9 Molecular l geometry and bonding theories 1. Valence shell electron pair repulsion theory 2. Valence bond theory 3. Molecular orbital theory 1 Ionic bonding Covalent bonding
Lecture outline: Section 9 Molecular l geometry and bonding theories 1. Valence shell electron pair repulsion theory 2. Valence bond theory 3. Molecular orbital theory 1 Ionic bonding Covalent bonding
Molecular Polarity Guided Inquiry (Textbook 10.4) Essential Questions How do the bonds between atoms, as well as electron location, determine
 Polarity Guided Inquiry (Textbook 10.4) Essential Questions How do the bonds between atoms, as well as electron location, determine interactions between molecules? How do electronegativity and molecular
Polarity Guided Inquiry (Textbook 10.4) Essential Questions How do the bonds between atoms, as well as electron location, determine interactions between molecules? How do electronegativity and molecular
Electron Geometry Hybrid Orbitals
 Molecular Shape and Hybridized Orbitals CH2000: Introduction to General Chemistry, Plymouth State University, Fall 2014 Introduction: In chemistry, the three dimensional shape of a molecule is as important
Molecular Shape and Hybridized Orbitals CH2000: Introduction to General Chemistry, Plymouth State University, Fall 2014 Introduction: In chemistry, the three dimensional shape of a molecule is as important
Chapter 13: Phenomena
 Chapter 13: Phenomena Phenomena: Scientists measured the bond angles of some common molecules. In the pictures below each line represents a bond that contains 2 electrons. If multiple lines are drawn together
Chapter 13: Phenomena Phenomena: Scientists measured the bond angles of some common molecules. In the pictures below each line represents a bond that contains 2 electrons. If multiple lines are drawn together
Chemistry: The Central Science Twelfth Edition, AP* Edition 2012
 A Correlation of The Central Science Twelfth Edition, AP* Edition 2012 to the AP* Chemistry Topics I. Structure of Matter A. Atomic theory and atomic structure 1. Evidence for the atomic theory SECTIONS:
A Correlation of The Central Science Twelfth Edition, AP* Edition 2012 to the AP* Chemistry Topics I. Structure of Matter A. Atomic theory and atomic structure 1. Evidence for the atomic theory SECTIONS:
5.111 Principles of Chemical Science
 MIT OpenCourseWare http://ocw.mit.edu 5.111 Principles of Chemical Science Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 24.1 5.111 Lecture
MIT OpenCourseWare http://ocw.mit.edu 5.111 Principles of Chemical Science Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 24.1 5.111 Lecture
Warm Up 9: Definitions LT I can explain how covalent molecules create attractive forces between molecules.
 Warm Up 9: Definitions 11-3-17 LT I can explain how covalent molecules create attractive forces between molecules. Q1. Draw the Lewis dot structure, label oxidation number and structural formula for: O
Warm Up 9: Definitions 11-3-17 LT I can explain how covalent molecules create attractive forces between molecules. Q1. Draw the Lewis dot structure, label oxidation number and structural formula for: O
Sample Free-Response Questions
 Exam Information Sample Free-Response Questions Question 1 is a long constructed-response question that should require about 20 minutes to answer. Questions 2, 3, and 4 are short constructed-response questions
Exam Information Sample Free-Response Questions Question 1 is a long constructed-response question that should require about 20 minutes to answer. Questions 2, 3, and 4 are short constructed-response questions
Chemistry 1A, Spring 2007 Midterm Exam 2 March 5, 2007 (90 min, closed book)
 Chemistry 1A, Spring 2007 Midterm Exam 2 March 5, 2007 (90 min, closed book) Name: KEY SID: TA Name: 1.) Write your name on every page of this exam. 2.) This exam has 40 multiple choice questions. Fill
Chemistry 1A, Spring 2007 Midterm Exam 2 March 5, 2007 (90 min, closed book) Name: KEY SID: TA Name: 1.) Write your name on every page of this exam. 2.) This exam has 40 multiple choice questions. Fill
Water. Water participates in H-bonding with biomolecules.
 Water Most biochemical reactions occur in an aqueous environment. Water is highly polar because of its bent geometry. Water is highly cohesive because of intermolecular hydrogen bonding. Water participates
Water Most biochemical reactions occur in an aqueous environment. Water is highly polar because of its bent geometry. Water is highly cohesive because of intermolecular hydrogen bonding. Water participates
Lewis Structures and Molecular Shapes
 Lewis Structures and Molecular Shapes Drawing Lewis Structures Determine from formula if ionic or covalent Count the electrons If ionic : add valence # to charge if (-), subtract if (+) - = 7+1 electrons;
Lewis Structures and Molecular Shapes Drawing Lewis Structures Determine from formula if ionic or covalent Count the electrons If ionic : add valence # to charge if (-), subtract if (+) - = 7+1 electrons;
VSEPR. Valence Shell Electron Pair Repulsion Theory
 VSEPR Valence Shell Electron Pair Repulsion Theory Vocabulary: domain = any electron pair or bond (single, double or triple) is considered one domain. bonding pair = shared pair = any electron pair that
VSEPR Valence Shell Electron Pair Repulsion Theory Vocabulary: domain = any electron pair or bond (single, double or triple) is considered one domain. bonding pair = shared pair = any electron pair that
Chapter 6 PRETEST: Chemical Bonding
 Chapter 6 PRETEST: Chemical In the space provided, write the letter of the term or phrase that best completes each statement or best answers each question. 1.The charge on an ion is a. always positive.
Chapter 6 PRETEST: Chemical In the space provided, write the letter of the term or phrase that best completes each statement or best answers each question. 1.The charge on an ion is a. always positive.
Chemistry: Molecules, Matter, and Change, Fourth Edition Loretta Jones and Peter Atkins Correlated with AP Chemistry, May 2002, May 2003
 Chemistry: Molecules, Matter, and Change, Fourth Edition Loretta Jones and Peter Atkins Correlated with AP Chemistry, May 2002, May 2003 ST=Student textbook I. Structure of Matter A. Atomic theory and
Chemistry: Molecules, Matter, and Change, Fourth Edition Loretta Jones and Peter Atkins Correlated with AP Chemistry, May 2002, May 2003 ST=Student textbook I. Structure of Matter A. Atomic theory and
Chemical Bonding II: Molecular Geometry and Hybridization of Atomic Orbitals Chapter 1
 Chemical Bonding II: Molecular Geometry and Hybridization of Atomic Orbitals Chapter 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. How to get the book of
Chemical Bonding II: Molecular Geometry and Hybridization of Atomic Orbitals Chapter 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. How to get the book of
5.111 Principles of Chemical Science
 MIT penourseware http://ocw.mit.edu 5.111 Principles of hemical Science Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.111 Lecture Summary
MIT penourseware http://ocw.mit.edu 5.111 Principles of hemical Science Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.111 Lecture Summary
Adapted from CHM 130 Maricopa County, AZ Molecular Geometry and Lewis Dot Formulas Introduction
 Adapted from CHM 130 Maricopa County, AZ Molecular Geometry and Lewis Dot Formulas Introduction A chemical bond is an intramolecular (within the molecule) force holding two or more atoms together. Covalent
Adapted from CHM 130 Maricopa County, AZ Molecular Geometry and Lewis Dot Formulas Introduction A chemical bond is an intramolecular (within the molecule) force holding two or more atoms together. Covalent
AP CHEMISTRY CHAPTERS 5 & 6 Problem Set #4. (Questions 1-13) Choose the letter that best answers the question or completes the statement.
 NAME: AP CHEMISTRY CHAPTERS 5 & 6 Problem Set #4 (Questions 1-13) Choose the letter that best answers the question or completes the statement. (Questions 1-2) Consider atoms of the following elements.
NAME: AP CHEMISTRY CHAPTERS 5 & 6 Problem Set #4 (Questions 1-13) Choose the letter that best answers the question or completes the statement. (Questions 1-2) Consider atoms of the following elements.
Organic Chemistry. Review Information for Unit 1. VSEPR Hybrid Orbitals Polar Molecules
 rganic hemistry Review Information for Unit 1 VSEPR ybrid rbitals Polar Molecules VSEPR The valence shell electron pair repulsion model (VSEPR) can be used to predict the geometry around a particular atom
rganic hemistry Review Information for Unit 1 VSEPR ybrid rbitals Polar Molecules VSEPR The valence shell electron pair repulsion model (VSEPR) can be used to predict the geometry around a particular atom
Shapes of Molecules and Hybridization
 Shapes of Molecules and Hybridization A. Molecular Geometry Lewis structures provide us with the number and types of bonds around a central atom, as well as any NB electron pairs. They do not tell us the
Shapes of Molecules and Hybridization A. Molecular Geometry Lewis structures provide us with the number and types of bonds around a central atom, as well as any NB electron pairs. They do not tell us the
QUESTION (2012:1) The 3-dimensional diagrams of two molecules are shown below.
 QUESTION (2012:1) (c) The 3-dimensional diagrams of two molecules are shown below. Circle the word that describes the polarity of each of the molecules CBr 4 and CH 3 Br. CBr 4 Polar Non-polar CH 3 Br
QUESTION (2012:1) (c) The 3-dimensional diagrams of two molecules are shown below. Circle the word that describes the polarity of each of the molecules CBr 4 and CH 3 Br. CBr 4 Polar Non-polar CH 3 Br
CHEMISTRY - ZUMDAHL 2E CH.4 - MOLECULAR STRUCTURE AND ORBITALS.
 !! www.clutchprep.com CONCEPT: ELECTRONIC GEOMETRY When drawing a compound you have to take into account two different systems of geometrical shape. The simpler system known as electronic geometry or shape
!! www.clutchprep.com CONCEPT: ELECTRONIC GEOMETRY When drawing a compound you have to take into account two different systems of geometrical shape. The simpler system known as electronic geometry or shape
CHAPTER 8 BONDING: GENERAL CONCEPTS Ionic solids are held together by strong electrostatic forces that are omnidirectional.
 CAPTER 8 BDIG: GEERAL CCEPTS 1 CAPTER 8 BDIG: GEERAL CCEPTS Questions 15. a. This diagram represents a polar covalent bond as in. In a polar covalent bond, there is an electron rich region (indicated by
CAPTER 8 BDIG: GEERAL CCEPTS 1 CAPTER 8 BDIG: GEERAL CCEPTS Questions 15. a. This diagram represents a polar covalent bond as in. In a polar covalent bond, there is an electron rich region (indicated by
CHEM PRACTICE EXAM IV CLASS - SPRING 2017 ANSWER KEY
 CHEM 1031 - PRACTICE EXAM IV CLASS - SPRING 2017 ANSWER KEY 1. When Group 1A (except for H) and Group 17 (7A) elements react with each other, they are most likely to form: A. Covalent or ionic bonds B.
CHEM 1031 - PRACTICE EXAM IV CLASS - SPRING 2017 ANSWER KEY 1. When Group 1A (except for H) and Group 17 (7A) elements react with each other, they are most likely to form: A. Covalent or ionic bonds B.
Page 1 of 16. Final Exam 5.111
 Final Exam 5.111 Page 1 of 16 Write your name and your TA's name below. Do not open the exam until the start of the exam is announced. 1) Read each part of a problem thoroughly. Many of a problem s latter
Final Exam 5.111 Page 1 of 16 Write your name and your TA's name below. Do not open the exam until the start of the exam is announced. 1) Read each part of a problem thoroughly. Many of a problem s latter
Unit 6: Molecular Geometry
 Unit 6: Molecular Geometry Molecular Geometry [6-5] the polarity of each bond, along with the geometry of the molecule determines Molecular Polarity. To predict the geometries of more complicated molecules,
Unit 6: Molecular Geometry Molecular Geometry [6-5] the polarity of each bond, along with the geometry of the molecule determines Molecular Polarity. To predict the geometries of more complicated molecules,
Lewis Formulas CHAPTER 7.
 ATER 7. Lewis ormulas 7-1. See the text. Because silicon is below carbon in the periodic table, we can determine the Lewis formula of Si 4 in the same manner as for 4. 7-2. Because phosphorus is the unique
ATER 7. Lewis ormulas 7-1. See the text. Because silicon is below carbon in the periodic table, we can determine the Lewis formula of Si 4 in the same manner as for 4. 7-2. Because phosphorus is the unique
6.2 Electron Movements in Brønsted Acid Base Reactions. Copyright 2018 by Nelson Education Limited 1
 6.2 Electron Movements in Brønsted Acid Base Reactions Copyright 2018 by Nelson Education Limited 1 Recall: Brønsted Acid Base Reactions often simply termed acid base reactions Recall: H + does not actually
6.2 Electron Movements in Brønsted Acid Base Reactions Copyright 2018 by Nelson Education Limited 1 Recall: Brønsted Acid Base Reactions often simply termed acid base reactions Recall: H + does not actually
Chem 121 Exam 4 Practice Exam
 Chem 121 Exam 4 Practice Exam 1. What is the correct electron configuration for bromine? b. 1s 2 2s 2 2p 6 3s 2 3p 6 3d 9 4s 2 4p 6 c. 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 5 d. 1s 2 2s 2 2p 6 3s 2 3p
Chem 121 Exam 4 Practice Exam 1. What is the correct electron configuration for bromine? b. 1s 2 2s 2 2p 6 3s 2 3p 6 3d 9 4s 2 4p 6 c. 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 5 d. 1s 2 2s 2 2p 6 3s 2 3p
Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks =
 Name:. orrect Questions = Wrong Questions =.. Unattempt Questions = Marks = 1. (11%) A(g) + 3B(g) r 2(g) Use the tabulated data to answer the questions about this reaction, which is carried out in a 1.0
Name:. orrect Questions = Wrong Questions =.. Unattempt Questions = Marks = 1. (11%) A(g) + 3B(g) r 2(g) Use the tabulated data to answer the questions about this reaction, which is carried out in a 1.0
Molecular Geometry and Chemical Bonding Theory
 Molecular Geometry and Chemical Bonding Theory The Valence -Shell Electron -Pair Repulsion (VSEPR) Model predicts the shapes of the molecules and ions by assuming that the valence shell electron pairs
Molecular Geometry and Chemical Bonding Theory The Valence -Shell Electron -Pair Repulsion (VSEPR) Model predicts the shapes of the molecules and ions by assuming that the valence shell electron pairs
AP Chemistry- Practice Bonding Questions for Exam
 AP Chemistry- Practice Bonding Questions for Exam Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following is a correct Lewis structure for
AP Chemistry- Practice Bonding Questions for Exam Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following is a correct Lewis structure for
Assignment 09 A. 2- The image below depicts a seesaw structure. Which of the following has such a structure?
 Assignment 09 A 1- Give the total number of electron domains, the number of bonding and nonbonding domains, and the molecular geometry, respectively, for the central atom of P 3. a) four electron domains,
Assignment 09 A 1- Give the total number of electron domains, the number of bonding and nonbonding domains, and the molecular geometry, respectively, for the central atom of P 3. a) four electron domains,
CHEM 1A03 TUTORIAL #4 Oct 1 Oct. 5, 2012 Solutions Chapter 10. The following problems (1-8) are based on material from Chapter 10 and class notes.
 HEM 1A03 TUTRIAL #4 ct 1 ct. 5, 2012 olutions hapter 10 The following problems (1-8) are based on material from hapter 10 and class notes. 1. Arrange the following in terms of increasing electronegativity:
HEM 1A03 TUTRIAL #4 ct 1 ct. 5, 2012 olutions hapter 10 The following problems (1-8) are based on material from hapter 10 and class notes. 1. Arrange the following in terms of increasing electronegativity:
CHAPTER 8 BONDING: GENERAL CONCEPTS
 Advanced Chemistry Name Hour Advanced Chemistry Approximate Timeline Students are expected to keep up with class work when absent. CHAPTER 8 BONDING: GENERAL CONCEPTS Day Plans for the day Assignment(s)
Advanced Chemistry Name Hour Advanced Chemistry Approximate Timeline Students are expected to keep up with class work when absent. CHAPTER 8 BONDING: GENERAL CONCEPTS Day Plans for the day Assignment(s)
Version 188 Exam 2 mccord (51600) 1
 Version 188 Exam 2 mccord (51600) 1 This print-out should have 35 questions. Multiple-choice questions may continue on the next column or page find all choices before answering. l I l l 001 3.0 points
Version 188 Exam 2 mccord (51600) 1 This print-out should have 35 questions. Multiple-choice questions may continue on the next column or page find all choices before answering. l I l l 001 3.0 points
Third Hour Exam 5.111
 Page 1 of 10 pages Third Hour Exam 5.111 Write your name below. This is a closed book exam. Solve all 6 problems. Read all problems thoroughly and read all parts of a problem. Many of the latter parts
Page 1 of 10 pages Third Hour Exam 5.111 Write your name below. This is a closed book exam. Solve all 6 problems. Read all problems thoroughly and read all parts of a problem. Many of the latter parts
Chem 1210 Final Spring points Dr. Luther Giddings
 Chem 1210 Final Spring 2002 150 points Dr. Luther Giddings Name Instructions: This is a closed book, closed notebook test. You may not discuss this exam with anyone, either during or after the exam, until
Chem 1210 Final Spring 2002 150 points Dr. Luther Giddings Name Instructions: This is a closed book, closed notebook test. You may not discuss this exam with anyone, either during or after the exam, until
A Simple Model for Chemical Bonds
 A Simple Model for hemical Bonds Multiple hoice 1. Modern organic chemistry a. is the study of carbon-containing compounds. b. is the study of compounds from living organisms. c. deals exclusively with
A Simple Model for hemical Bonds Multiple hoice 1. Modern organic chemistry a. is the study of carbon-containing compounds. b. is the study of compounds from living organisms. c. deals exclusively with
MAJOR FIELD TEST IN CHEMISTRY SAMPLE QUESTIONS
 MAJOR FIELD TEST IN CHEMISTRY SAMPLE QUESTIONS The following questions illustrate the range of the test in terms of the abilities measured, the disciplines covered, and the difficulty of the questions
MAJOR FIELD TEST IN CHEMISTRY SAMPLE QUESTIONS The following questions illustrate the range of the test in terms of the abilities measured, the disciplines covered, and the difficulty of the questions
B L U E V A L L E Y D I S T R I C T C U R R I C U L U M Science AP Chemistry
 B L U E V A L L E Y D I S T R I C T C U R R I C U L U M Science AP Chemistry ORGANIZING THEME/TOPIC UNIT 1: ATOMIC STRUCTURE Atomic Theory Electron configuration Periodic Trends Big Idea 1: The chemical
B L U E V A L L E Y D I S T R I C T C U R R I C U L U M Science AP Chemistry ORGANIZING THEME/TOPIC UNIT 1: ATOMIC STRUCTURE Atomic Theory Electron configuration Periodic Trends Big Idea 1: The chemical
Chemistry 1A, Spring 2006 Midterm Exam II, Version 1 March 6, 2006 (90 min, closed book)
 Chemistry 1A, Spring 2006 Midterm Exam II, Version 1 March 6, 2006 (90 min, closed book) Name: SID: Identification Sticker TA Name: Write your name on every page of this exam. This exam has 38 multiple
Chemistry 1A, Spring 2006 Midterm Exam II, Version 1 March 6, 2006 (90 min, closed book) Name: SID: Identification Sticker TA Name: Write your name on every page of this exam. This exam has 38 multiple
Relevant Equations and Constants particle mole. = x10 1mL = 1cm 1 amu = x 10 kg
 CHEM 116 Study Guide for General Chemistry Atom First by McMurry & Fay ( nd edition) Chapter 0 Chemical Tools: Experimentation and Measurement pp 1 Appendix A: Mathematical Operations Recommended Problems:
CHEM 116 Study Guide for General Chemistry Atom First by McMurry & Fay ( nd edition) Chapter 0 Chemical Tools: Experimentation and Measurement pp 1 Appendix A: Mathematical Operations Recommended Problems:
CHEMISTRY 102B Hour Exam III. Dr. D. DeCoste T.A. Show all of your work and provide complete answers to questions 16 and (45 pts.
 CHEMISTRY 102B Hour Exam III April 28, 2016 Dr. D. DeCoste Name Signature T.A. This exam contains 17 questions on 8 numbered pages. Check now to make sure you have a complete exam. You have one hour and
CHEMISTRY 102B Hour Exam III April 28, 2016 Dr. D. DeCoste Name Signature T.A. This exam contains 17 questions on 8 numbered pages. Check now to make sure you have a complete exam. You have one hour and
; (c) [Li] [: O :] [Li]. 5a. The electrostatic potential map that corresponds to IF is the one with the most red in it. ... C C H
![; (c) [Li] [: O :] [Li]. 5a. The electrostatic potential map that corresponds to IF is the one with the most red in it. ... C C H ; (c) [Li] [: O :] [Li]. 5a. The electrostatic potential map that corresponds to IF is the one with the most red in it. ... C C H](/thumbs/87/96516624.jpg) hapter 10 Answers ractice Examples 1a Mg 1b n, Ge, [: Br :], K, : e: + 2 : : +, [Tl ] +, 2 : : [] 2a (a) [a] [ ] [a] ; (b) [Mg] [: :] [Mg] [: :] [Mg] 2+ 3 2+ 3 2+ 2+ 2b (a) [: I :] [a] [: I :] 2+ 2 ; (b)
hapter 10 Answers ractice Examples 1a Mg 1b n, Ge, [: Br :], K, : e: + 2 : : +, [Tl ] +, 2 : : [] 2a (a) [a] [ ] [a] ; (b) [Mg] [: :] [Mg] [: :] [Mg] 2+ 3 2+ 3 2+ 2+ 2b (a) [: I :] [a] [: I :] 2+ 2 ; (b)
Nature of Molecules. Chapter 2. All matter: composed of atoms
 Nature of Molecules Chapter 2 Atomic Structure All matter: composed of atoms Understanding structure of atoms critical to understanding nature of biological molecules 2 1 Atomic Structure Atoms composed
Nature of Molecules Chapter 2 Atomic Structure All matter: composed of atoms Understanding structure of atoms critical to understanding nature of biological molecules 2 1 Atomic Structure Atoms composed
You May Remove this page from the front of the exam. Avogadro s number: NA = 6.022x 1023 mol-1
 You May Remove this page from the front of the exam Avogadro s number: NA = 6.022x 1023 mol-1 JBA 2018 Chemistry Exam 1 Name: Score: /100 = /80 1. Which of the following is a member of the group of elements
You May Remove this page from the front of the exam Avogadro s number: NA = 6.022x 1023 mol-1 JBA 2018 Chemistry Exam 1 Name: Score: /100 = /80 1. Which of the following is a member of the group of elements
H O H. Chapter 3: Outline-2. Chapter 3: Outline-1
 Chapter 3: utline-1 Molecular Nature of Water Noncovalent Bonding Ionic interactions van der Waals Forces Thermal Properties of Water Solvent Properties of Water ydrogen Bonds ydrophilic, hydrophobic,
Chapter 3: utline-1 Molecular Nature of Water Noncovalent Bonding Ionic interactions van der Waals Forces Thermal Properties of Water Solvent Properties of Water ydrogen Bonds ydrophilic, hydrophobic,
Midterm Exam III. CHEM 181: Introduction to Chemical Principles November 25, 2014 Answer Key
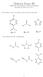 Midterm Exam III EM 181: Introduction to hemical Principles ovember 25, 2014 Answer Key 1. For reference, here are the pk a values for three weak acids: 3 pk a = 7.2 pk a = 4.8 pk a = 15 ow consider this
Midterm Exam III EM 181: Introduction to hemical Principles ovember 25, 2014 Answer Key 1. For reference, here are the pk a values for three weak acids: 3 pk a = 7.2 pk a = 4.8 pk a = 15 ow consider this
Chemistry 201 FINAL EXAM Name Illinois Wesleyan University December 13, 2005 Fall 2005
 Chemistry 201 FINAL EXAM Name Illinois Wesleyan University December 13, 2005 Fall 2005 There are 200 points on this exam, and you have exactly two hours to complete it. 1. (12 points) a. Give the name
Chemistry 201 FINAL EXAM Name Illinois Wesleyan University December 13, 2005 Fall 2005 There are 200 points on this exam, and you have exactly two hours to complete it. 1. (12 points) a. Give the name
Bonding/Lewis Dots Lecture Page 1 of 12 Date. Bonding. What is Coulomb's Law? Energy Profile: Covalent Bonds. Electronegativity and Linus Pauling
 Bonding/Lewis Dots Lecture Page 1 of 12 Date Bonding What is Coulomb's Law? Energy Profile: Covalent Bonds Electronegativity and Linus Pauling 2.1 H 1.0 Li 0.9 Na 0.8 K 0.8 Rb 0.7 Cs 0.7 Fr 1.5 Be 1.2
Bonding/Lewis Dots Lecture Page 1 of 12 Date Bonding What is Coulomb's Law? Energy Profile: Covalent Bonds Electronegativity and Linus Pauling 2.1 H 1.0 Li 0.9 Na 0.8 K 0.8 Rb 0.7 Cs 0.7 Fr 1.5 Be 1.2
Chapter 9. Molecular Geometry and Bonding Theories
 Sample Exercise 9.1 (p. 347) Use the VSEPR model to predict the molecular geometries of a) O 3 b) SnCl 3 - Practice Exercise 1 (9.1) Consider the AB 3 molecules and ions: PCl 3, SO 3, AlCl 3, SO 3 2-,
Sample Exercise 9.1 (p. 347) Use the VSEPR model to predict the molecular geometries of a) O 3 b) SnCl 3 - Practice Exercise 1 (9.1) Consider the AB 3 molecules and ions: PCl 3, SO 3, AlCl 3, SO 3 2-,
What Do Molecules Look Like?
 What Do Molecules Look Like? The Lewis Dot Structure approach provides some insight into molecular structure in terms of bonding, but what about 3D geometry? Recall that we have two types of electron pairs:
What Do Molecules Look Like? The Lewis Dot Structure approach provides some insight into molecular structure in terms of bonding, but what about 3D geometry? Recall that we have two types of electron pairs:
Exam 2 Review Problems
 09-107 onors Chemistry Carnegie Mellon University Exam 2 Review Problems Exam 2 will be held on Tuesday, ctober 30, in Porter all A18C. We will be there from 2:45 to 4:45 pm. Disclaimer: This review sheet
09-107 onors Chemistry Carnegie Mellon University Exam 2 Review Problems Exam 2 will be held on Tuesday, ctober 30, in Porter all A18C. We will be there from 2:45 to 4:45 pm. Disclaimer: This review sheet
Chemistry 6 12 Section 03
 Chemistry 6 12 Section 03 1 Knowledge of the nature of matter 1. Differentiate between pure substances, homogeneous mixtures, and heterogeneous mixtures. 2. Determine the effects of changes in temperature,
Chemistry 6 12 Section 03 1 Knowledge of the nature of matter 1. Differentiate between pure substances, homogeneous mixtures, and heterogeneous mixtures. 2. Determine the effects of changes in temperature,
Amino acids have the general structure: All amino acids have at least two acidic protons. One is the proton
 Worksheet rganic Acids and Bases II Amino Acids Acids are proton donors in aqueous solutions: A + 2 3 + + A - acid base conjugate conjugate base acid The strength of an acid is measured by the [ 3 + ]
Worksheet rganic Acids and Bases II Amino Acids Acids are proton donors in aqueous solutions: A + 2 3 + + A - acid base conjugate conjugate base acid The strength of an acid is measured by the [ 3 + ]
CHEM 110 Exam 2 - Practice Test 1 - Solutions
 CHEM 110 Exam 2 - Practice Test 1 - Solutions 1D 1 has a triple bond. 2 has a double bond. 3 and 4 have single bonds. The stronger the bond, the shorter the length. 2A A 1:1 ratio means there must be the
CHEM 110 Exam 2 - Practice Test 1 - Solutions 1D 1 has a triple bond. 2 has a double bond. 3 and 4 have single bonds. The stronger the bond, the shorter the length. 2A A 1:1 ratio means there must be the
B. (i), (iii), and (v) C. (iv) D. (i), (ii), (iii), and (v) E. (i), (iii), (iv), and (v) Answer: B. SO 3, and NO 3 - both have 24 VE and have Lewis
 SCCH 161 Homework 3 1. Give the number of lone pairs around the central atom and the molecular geometry of CBr 4. Answer: Carbon has 4 valence electrons and bonds to four bromine atoms (each has 7 VE s).
SCCH 161 Homework 3 1. Give the number of lone pairs around the central atom and the molecular geometry of CBr 4. Answer: Carbon has 4 valence electrons and bonds to four bromine atoms (each has 7 VE s).
Lewis Dot Structures for Methane, CH 4 The central C atom is bonded by single bonds (-) to 4 individual H atoms
 Chapter 10 (Hill/Petrucci/McCreary/Perry Bonding Theory and Molecular Structure This chapter deals with two additional approaches chemists use to describe chemical bonding: valence-shell electron pair
Chapter 10 (Hill/Petrucci/McCreary/Perry Bonding Theory and Molecular Structure This chapter deals with two additional approaches chemists use to describe chemical bonding: valence-shell electron pair
AP CHEMISTRY Pacing Guide
 AP CHEMISTRY Pacing Guide Course Description: AP CHEMISTRY (one term guide) EOC AP/IB VOCATS Teacher-made final exam Day Date SCS Objectives Essential Questions Content Tasks/Strategies 1 2 3 Objective
AP CHEMISTRY Pacing Guide Course Description: AP CHEMISTRY (one term guide) EOC AP/IB VOCATS Teacher-made final exam Day Date SCS Objectives Essential Questions Content Tasks/Strategies 1 2 3 Objective
CHEM J-3 June 2012
 CEM1611 2012-J-3 June 2012 In a standard acid-base titration, 25.00 ml of 0.1043 M a solution was found to react exactly with 28.45 ml of an Cl solution of unknown concentration. What is the p of the unknown
CEM1611 2012-J-3 June 2012 In a standard acid-base titration, 25.00 ml of 0.1043 M a solution was found to react exactly with 28.45 ml of an Cl solution of unknown concentration. What is the p of the unknown
Carbon-based molecules are held together by covalent bonds between atoms
 hapter 1: hemical bonding and structure in organic compounds arbon-based molecules are held together by covalent bonds between atoms omposition: Mainly nonmetals; especially,, O, N, S, P and the halogens
hapter 1: hemical bonding and structure in organic compounds arbon-based molecules are held together by covalent bonds between atoms omposition: Mainly nonmetals; especially,, O, N, S, P and the halogens
Homework 08 VSEPR. The active ingredient in some oral anesthetics used in sore throat sprays. What is the molar mass of phenol?
 HW08 VSEPR This is a preview of the published version of the quiz Started: Oct 21 at 11:14am Quiz Instruc ons Homework 08 VSEPR Question 1 Consider the structural formula of phenol. The active ingredient
HW08 VSEPR This is a preview of the published version of the quiz Started: Oct 21 at 11:14am Quiz Instruc ons Homework 08 VSEPR Question 1 Consider the structural formula of phenol. The active ingredient
CHEMISTRY. Section II (Total time 95 minutes) Part A Time 55 minutes YOU MAY USE YOUR CALCULATOR FOR PART A.
 CHEMISTRY Section II (Total time 95 minutes) Part A Time 55 minutes YOU MAY USE YOUR CALCULATOR FOR PART A. CLEARLY SHOW THE METHOD USED AND THE STEPS INVOLVED IN ARRIVING AT YOUR ANSWERS. It is to your
CHEMISTRY Section II (Total time 95 minutes) Part A Time 55 minutes YOU MAY USE YOUR CALCULATOR FOR PART A. CLEARLY SHOW THE METHOD USED AND THE STEPS INVOLVED IN ARRIVING AT YOUR ANSWERS. It is to your
Name. CHM 115 EXAM #2 Practice KEY. a. N Cl b. N F c. F F d. I I e. N Br. a. K b. Be c. O d. Al e. S
 Name CHM 115 EXAM #2 Practice KEY Circle the correct answer. (numbers 1-8, 2.5 points each) 1. Which of the following bonds should be the most polar? a. N Cl b. N F c. F F d. I I e. N Br 2. Choose the
Name CHM 115 EXAM #2 Practice KEY Circle the correct answer. (numbers 1-8, 2.5 points each) 1. Which of the following bonds should be the most polar? a. N Cl b. N F c. F F d. I I e. N Br 2. Choose the
1. The total number of protons, neutrons, and electrons in 98 Mo 7+ are:
 hemistry 11 SP 2013_ANSWERS Examination #1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
hemistry 11 SP 2013_ANSWERS Examination #1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Illinois JETS Academic Challenge Chemistry Test (District) Useful Information. PV = nrt R = L atm mol -1 K -1.
 Useful Information PV = nrt R = 0.08206 L atm mol -1 K -1 Q = m x T x s 1 atm = 760 torr Avogadro's number = 6.022 x 10 23 K = C + 273 ph = -log [H + ] 1 1 E = (-R H ) - n 2 n 2 f i R H = 2.18 x 10-18
Useful Information PV = nrt R = 0.08206 L atm mol -1 K -1 Q = m x T x s 1 atm = 760 torr Avogadro's number = 6.022 x 10 23 K = C + 273 ph = -log [H + ] 1 1 E = (-R H ) - n 2 n 2 f i R H = 2.18 x 10-18
5.111 Lecture Summary #12 Friday, October 3, 2014
 5.111 Lecture Summary #12 riday, October 3, 2014 Readings for today: Section 3.1 The Basic VSEPR Model, Section 3.2 Molecules with Lone Pairs on the Central Atom. (Same sections in 5 th and 4 th ed.) Read
5.111 Lecture Summary #12 riday, October 3, 2014 Readings for today: Section 3.1 The Basic VSEPR Model, Section 3.2 Molecules with Lone Pairs on the Central Atom. (Same sections in 5 th and 4 th ed.) Read
Name AP Chemistry January 25, 2013
 Name AP Chemistry January 25, 2013 AP Chemistry Midterm Exam Part I: 75 Questions, 80 minutes, Multiple Choice, No Calculator Allowed Bubble the correct answer on your scantron for each of the following.
Name AP Chemistry January 25, 2013 AP Chemistry Midterm Exam Part I: 75 Questions, 80 minutes, Multiple Choice, No Calculator Allowed Bubble the correct answer on your scantron for each of the following.
C PM RESURRECTION
 Announcements Final Exam TIME: October 8, 7:30-9:30AM VENUE: CTC 105 65-Multiple Choice Questions 3 Questions Each Chapter 2-5 7 Questions Each Chapter 6-8 30 Questions From Chapter 9-11 Saturday Review
Announcements Final Exam TIME: October 8, 7:30-9:30AM VENUE: CTC 105 65-Multiple Choice Questions 3 Questions Each Chapter 2-5 7 Questions Each Chapter 6-8 30 Questions From Chapter 9-11 Saturday Review
Organic Chemistry CHM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Structure & Shape of Organic Molecules - Chapter 1 (Wade)
 rganic hemistry M 314 Dr. Laurie S. Starkey, al Poly Pomona Structure & Shape of rganic Molecules - hapter 1 (Wade) rganic (living things, chemistry of carbon) Examples of rganic ompounds: Inorganic (rocks,
rganic hemistry M 314 Dr. Laurie S. Starkey, al Poly Pomona Structure & Shape of rganic Molecules - hapter 1 (Wade) rganic (living things, chemistry of carbon) Examples of rganic ompounds: Inorganic (rocks,
Orbital Shapes Carbon: Electron configuration Carbon: Full. Short form. Orbital energy diagram. Orbital energy levels diagram
 rganic hemistry involves mostly NPS and the halogens. rganic compounds use valence shell electrons to bond. Usually only in the s and p orbitals. rbital Shapes arbon: z y z y z y z y z y x x x x x 1s n=1
rganic hemistry involves mostly NPS and the halogens. rganic compounds use valence shell electrons to bond. Usually only in the s and p orbitals. rbital Shapes arbon: z y z y z y z y z y x x x x x 1s n=1
Today: QUIZ 3 Next Week in Lab: Covering Lectures Next Meeting
 Today: Ionic Bonding vs. Covalent Bonding Bond Polarities: Nonpolar Covalent vs. Polar Covalent vs. Ionic Electronegativity Differences Dipole Moments Molecular Polarities: Molecular Symmetry QUIZ 3 Next
Today: Ionic Bonding vs. Covalent Bonding Bond Polarities: Nonpolar Covalent vs. Polar Covalent vs. Ionic Electronegativity Differences Dipole Moments Molecular Polarities: Molecular Symmetry QUIZ 3 Next
Unit 2 Exam (Fall 2017) Seat # Name Lab Section: M Tu W Th
 Unit 2 Exam (Fall 2017) Seat # Name Lab Section: M Tu W Th MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. (3.5 pts ea) 1) In a covalent compound
Unit 2 Exam (Fall 2017) Seat # Name Lab Section: M Tu W Th MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. (3.5 pts ea) 1) In a covalent compound
NChO 2006 ANNOTATED ANSWERS
 NChO 2006 ANNOTATED ANSWERS 1. D HCl H 2 and CH 4 are nonpolar and will dissolve very little in polar water. CO is polar and will dissolve OK, but HCl is very polar and dissolves amazingly well in water.
NChO 2006 ANNOTATED ANSWERS 1. D HCl H 2 and CH 4 are nonpolar and will dissolve very little in polar water. CO is polar and will dissolve OK, but HCl is very polar and dissolves amazingly well in water.
Chemical Bonding II: Molecular Geometry and Hybridization of Atomic Orbitals
 Chemical Bonding II: and ybridization of Atomic rbitals Chapter 10 Valence shell electron pair repulsion (VSEPR) model: Predict the geometry of the molecule from the electrostatic repulsions between the
Chemical Bonding II: and ybridization of Atomic rbitals Chapter 10 Valence shell electron pair repulsion (VSEPR) model: Predict the geometry of the molecule from the electrostatic repulsions between the
SUPeR Chemistry CH 222 Practice Exam
 SUPeR Chemistry CH 222 Practice Exam This exam has been designed to help you practice working multiple choice problems over the material that will be covered on the first CH 222 midterm. The actual exams
SUPeR Chemistry CH 222 Practice Exam This exam has been designed to help you practice working multiple choice problems over the material that will be covered on the first CH 222 midterm. The actual exams
Chapter 10 Shapes of Molecules. Dr. Sapna Gupta
 Chapter 10 Shapes of Molecules Dr. Sapna Gupta Shapes of Molecules - Importance All molecules have a 3D orientations; even the diatomic ones because atoms have a volume. In case of tri atomic or polyatomic
Chapter 10 Shapes of Molecules Dr. Sapna Gupta Shapes of Molecules - Importance All molecules have a 3D orientations; even the diatomic ones because atoms have a volume. In case of tri atomic or polyatomic
What is the correct name and bonding of BF 3? What is the correct name and bonding of BF 3?
 What is the correct name and bonding of BF 3? 1. boron trifluoride, covalent compound 2. boron trifluoride, ionic compound 3. boron fluoride, covalent compound 4. boron fluoride, ionic compound What is
What is the correct name and bonding of BF 3? 1. boron trifluoride, covalent compound 2. boron trifluoride, ionic compound 3. boron fluoride, covalent compound 4. boron fluoride, ionic compound What is
Lewis Theory of Shapes and Polarities of Molecules
 Lewis Theory of Shapes and Polarities of Molecules Sulfanilamide Lewis Structures and the Real 3D-Shape of Molecules Molecular Shape or Geometry The way in which atoms of a molecule are arranged in space
Lewis Theory of Shapes and Polarities of Molecules Sulfanilamide Lewis Structures and the Real 3D-Shape of Molecules Molecular Shape or Geometry The way in which atoms of a molecule are arranged in space
NAME: SPRING 2015 MIDTERM
 page 1 pts NAME: SPRING 2015 MIDTERM hemistry 231 Professor: Dr. Gergens take-home portion (DUE at the beginning of the period, 4/6) Do your best on this take-home portion of your mid-term. I may grade
page 1 pts NAME: SPRING 2015 MIDTERM hemistry 231 Professor: Dr. Gergens take-home portion (DUE at the beginning of the period, 4/6) Do your best on this take-home portion of your mid-term. I may grade
Lecture Presentation. Chapter 10 Chemical Bonding II: Molecular Shapes, Valence Bond Theory, and Molecular Orbital Theory
 Lecture Presentation Chapter 10 Chemical Bonding II: Molecular Shapes, Valence Bond Theory, and Molecular Orbital Theory Predicting Molecular Geometry 1. Draw the Lewis structure. 2. Determine the number
Lecture Presentation Chapter 10 Chemical Bonding II: Molecular Shapes, Valence Bond Theory, and Molecular Orbital Theory Predicting Molecular Geometry 1. Draw the Lewis structure. 2. Determine the number
What is this? Electrons: charge, mass? Atom. Negative charge(-), mass = 0. The basic unit of matter. Made of subatomic particles:
 Chemical Bonds What is this? Atom The basic unit of matter. Electrons: charge, mass? Negative charge(-), mass = 0 Made of subatomic particles: Protons: charge, mass? Positive charge (+), mass = 1 Neutrons:
Chemical Bonds What is this? Atom The basic unit of matter. Electrons: charge, mass? Negative charge(-), mass = 0 Made of subatomic particles: Protons: charge, mass? Positive charge (+), mass = 1 Neutrons:
Chemistry 1A, Fall 2003 Midterm 2 Oct 14, 2003 (90 min, closed book)
 Name: SID: TA Name: Chemistry 1A, Fall 2003 Midterm 2 Oct 14, 2003 (90 min, closed book) This exam has 45 multiple choice questions. Fill in the Scantron form AND circle your answer on the exam. Each question
Name: SID: TA Name: Chemistry 1A, Fall 2003 Midterm 2 Oct 14, 2003 (90 min, closed book) This exam has 45 multiple choice questions. Fill in the Scantron form AND circle your answer on the exam. Each question
Big Idea 1: Structure of Matter Learning Objective Check List
 Big Idea 1: Structure of Matter Learning Objective Check List Structure of Matter Mole Concept: Empirical Formula, Percent Composition, Stoichiometry Learning objective 1.1 The student can justify the
Big Idea 1: Structure of Matter Learning Objective Check List Structure of Matter Mole Concept: Empirical Formula, Percent Composition, Stoichiometry Learning objective 1.1 The student can justify the
Classes of Organic Compounds
 Unit 1 Functional Groups Depicting Structures of rganic ompounds Lewis Structures ondensed structural formulas Line angle drawings 3-dimensional structures Resonance Structures Acid-Base Reactions urved
Unit 1 Functional Groups Depicting Structures of rganic ompounds Lewis Structures ondensed structural formulas Line angle drawings 3-dimensional structures Resonance Structures Acid-Base Reactions urved
Chapter 10 Molecular Geometry and Chemical Bonding Theory. Copyright Cengage Learning. All rights reserved. 10 1
 Chapter 10 Molecular Geometry and Chemical Bonding Theory Copyright Cengage Learning. All rights reserved. 10 1 Molecular geometry is the general shape of a molecule, as determined by the relative positions
Chapter 10 Molecular Geometry and Chemical Bonding Theory Copyright Cengage Learning. All rights reserved. 10 1 Molecular geometry is the general shape of a molecule, as determined by the relative positions
CHEM 130 Final Review
 CHEM 130 Final Review Chapter Chp 1 Chemistry Define Introduction Scientific approach Describe Theory, natural law Distinguish between theory & law Chp 2 Scientific notation Conversions between standard
CHEM 130 Final Review Chapter Chp 1 Chemistry Define Introduction Scientific approach Describe Theory, natural law Distinguish between theory & law Chp 2 Scientific notation Conversions between standard
Subtopic 4.2 MOLECULAR SHAPE AND POLARITY
 Subtopic 4.2 MOLECULAR SHAPE AND POLARITY 1 LEARNING OUTCOMES (covalent bonding) 1. Draw the Lewis structure of covalent molecules (octet rule such as NH 3, CCl 4, H 2 O, CO 2, N 2 O 4, and exception to
Subtopic 4.2 MOLECULAR SHAPE AND POLARITY 1 LEARNING OUTCOMES (covalent bonding) 1. Draw the Lewis structure of covalent molecules (octet rule such as NH 3, CCl 4, H 2 O, CO 2, N 2 O 4, and exception to
Solutions of sodium hydroxide and hydrochloric acid are placed together H + + OH - H 2 O
 AP Chemistry Exam Review Reactions Metal and oxygen gas Metal oxide Hydrogen gas is burned Water Solid calcium is placed in oxygen gas Calcium oxide Alkali metals and water Metal Hydroxide (maybe split
AP Chemistry Exam Review Reactions Metal and oxygen gas Metal oxide Hydrogen gas is burned Water Solid calcium is placed in oxygen gas Calcium oxide Alkali metals and water Metal Hydroxide (maybe split
Question 1. Electron Configurations Noble Gases and The Rule of Eight. Chapter 1. What is the electronic configuration of carbon?
 hapter ~. nm Electronic Structure and Bonding Anders Jöns Ångström (84-874) Å = picometers =. nanometers = -4 microns = -8 centimeters Acids and Bases nm = Å An atom vs. a nucleus ~, x larger ucleus =
hapter ~. nm Electronic Structure and Bonding Anders Jöns Ångström (84-874) Å = picometers =. nanometers = -4 microns = -8 centimeters Acids and Bases nm = Å An atom vs. a nucleus ~, x larger ucleus =
Chapter 13: Phenomena
 Chapter 13: Phenomena Phenomena: Scientists measured the bond angles of some common molecules. In the pictures below each line represents a bond that contains 2 electrons. If multiple lines are drawn together
Chapter 13: Phenomena Phenomena: Scientists measured the bond angles of some common molecules. In the pictures below each line represents a bond that contains 2 electrons. If multiple lines are drawn together
Chapter 9. Molecular Geometry and Bonding Theories
 9.1 Molecular Shapes Read Sec. 9.1 and 9.2, then complete the Sample and Practice Exercises in these sections. Sample Exercise 9.1 (p. 347) Use the VSEPR model to predict the molecular geometries of a)
9.1 Molecular Shapes Read Sec. 9.1 and 9.2, then complete the Sample and Practice Exercises in these sections. Sample Exercise 9.1 (p. 347) Use the VSEPR model to predict the molecular geometries of a)
