EQUILIBRIUM ADSORPTION OF METHANE AND CARBON DIOXIDE ON 5A MOLECULAR SIEVE
|
|
|
- Grace Jones
- 6 years ago
- Views:
Transcription
1 Volume3 December 2007 EQUILIBRIUM ADSORPTION OF METHANE AND CARBON DIOXIDE ON 5A MOLECULAR SIEVE Dr. Abbas Khalaf Mohammed Shua'ab Chemical Engineering Department, Baghdad University, P. O. Box 47024, Baghdad, Iraq ABSTRACT Experimental and theoretical studies have been carried out to study the adsorption of methane and carbon dioxide on 5A molecular sieve. Adsorption equilibrium isotherms of methane and carbon dioxide are reported for the temperature range K and pressure up to 2.5MPa. Experimental data were obtained using a static system for gas solid adsorption. The Langmuir adsorption equilibrium equation gave good predictions. Adsorption of methane and carbon dioxide on 5A molecular sieve is purely physical since the isosteric heat of adsorption was found to be equal to and kj mole - for methane and carbon dioxide, respectively. الخالصة تم الحصول على توازن االمتزاز االيزوثرمي لغازز اليثاازن و زازز ثازيي اوسياثا البرناون نزساتداا الينزخا ألجزيئثاااو ياااو 5A و عناااا ت حاااز تااارا تتاااراو مااا 303 سلفااا إلاااى 333 سلفااا و ضاااغل يصااا إلاااى.2 مثبزنزسبزل. النتزئج أليدتبريو تتطزنق نشب حثا مع النيوذج التجريبي لاليبيزير. امتزاز ززز اليثاازن و زازز ثزيي اوسيثا البرنون على الينزخ ألجزيئثو يو 5A هو امتزاز فثزيزوي وذلا نيابإ إن تارا االمتازاز لغازز اليثازن تيزوي 2..0 سثلوحول/مول و لغزز ثزيي اوسيثا البرنون تيزوي. 3.2 سثلوحول/مول. KEYWORDS molecular sieve; adsorption; methane; carbon dioxide) 269
2 A. K. Mohammed Equilibrium Adsorption of Methane and Carbon Dioxide on 5A Molecular Sieve. INTRODUCTION The increasing importance of adsorption processes for the separation of gaseous mixtures is due to the high selectivity and adsorption capacity of solid adsorbents, the less extreme operation conditions needed, and the small energy consumption required. Adsorption is becoming a competitive operation that can advantageously substitute for other separation operations such as distillation or absorption. The advantages are especially attractive when the problem is the separation of light gases, since their separation by distillation or absorption requires expensive high pressure units. The adsorptive separation is achieved by one of the three mechanisms: steric, kinetic or equilibrium effect. The steric effect derives from the molecular sieving property of zeolites. In this case only small and properly shaped molecules can diffuse into the adsorbent, whereas other molecules are totally excluded. Kinetic separation is achieved by virtue of the difference in diffusion rates of different molecules into the adsorbent. By far, however, most processes operate through the equilibrium (or competitive) adsorption of the mixture, and hence are called equilibrium separation processes. Commercial separation processes using molecular sieve zeolites are mainly equilibrium processes (Kapoor & Yang, 989). Separation of bulk gas mixtures containing methane and carbon dioxide is an important problem in the chemical industry. Many natural gas reservoirs contain up to 50% CO 2 as impurity. The effluent gas from an oil well undergoing CO 2 flooding for enhanced oil recovery may contain 20 80% CO 2 and CH 4. It may be necessary to separate the CH 4 and the CO 2 from these gases in order to improve their fuel value and to recover and reuse the CO 2. Data on the adsorption of methane and carbon dioxide on molecular sieves are scarce in the literature. Some low pressure data and correlations have been reported for Linde 5A and 3X molecular sieve, where carbon dioxide was found to be preferentially adsorbed on the former molecular sieve (Tańczyk & Warmuziński, 998; Kumar, 989; Hyun & Danner, 982). Limitation are placed on the usefulness of these data for the design of commercial molecular sieve based separation systems, since a large part of these data was obtained at temperatures and pressures which are not ordinarily considered practical from a commercial stand point. 963
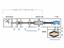
3 Volume3 December 2007 This paper reports a fundamental equilibrium adsorption data obtained for methane and carbon dioxide on a single sample of 5A molecular sieve (supplied by Rhone Poulenc industries). To simulate industrial conditions, the temperature ranged from ambient to 60 C and the pressure to 2.5 MPa. These data are of value in process and design studies of 5A molecular sieve based separation systems involving the separation of carbon dioxide from natural gas and oil well effluent. EXPERIMENTAL WORK A schematic diagram of the apparatus is shown in figure. All tubing was thick wall stainless steel, to minimize the dead volume. The tubing was connected by Crawford Swagelock compression fittings. Parker CPI series severe service union bonnet valves, with a hardened ball stem tip, were chosen because of their durability under repetitive use. A metal stem tip was selected instead of a soft stem tip to avert destruction of the valve seat in the event that particulate matter should escape from the adsorption chamber, enter the valve and score the valve seat. The adsorption chamber and reservoir were 20 cm 3 iron sample cylinders. Entrainment of adsorbent particles in the tubing and valving system was prevented by employing a dual screen method. This was accomplished by inserting Pyrex glass wool and a conical brass wire mesh screen, capable of containing particles. The gas pressure was measured via a Heise bourdon gauge with a 430 mm diameter dial and a pressure range from 0 to 689 kpa. The accuracy of the gauge was rated at 0.% of the full scale and the sensitivity at 0.02% of the full scale at all points. The pressure gauge enclosed a dead volume of approximately 30 cm 3. A water bath was employed to provide a constant temperature environment up to 353 K for both the adsorber and the reservoir. The bath was thermo stated and vigorously mixed by using a magnetic stirrer thermo stat hot plate. The temperature of the bath was continuously measured and recorded via a digital recorder and a thermocouple wire, calibrated with mercury thermometer. The whole equipment was evacuated by vacuum pump and absolute gauge pressure prior to each experiment. The feed gas supplied from cylinder was regulated by pressure regulator.\ 269
4 A. K. Mohammed Equilibrium Adsorption of Methane and Carbon Dioxide on 5A Molecular Sieve. C V PR V2 A 2 V V4 V5 B 9 V6 AG Vent Gas cylinder Heise pressure gauge Reservoir bomb Adsorber bomb Water bath Stir bar Magnetic stirrer thermostat hot plat Chromel alumel thermocouple Digital recorder Vacuum pump V PR AG Manual valve Pressure regulator Absolute gauge Fig () Schematic diagram of apparatus used for adsorption equilibrium measurement The volume of the reservoir and the adsorber (together with the Swagelock fittings) and the other sections of the system were measured manometrically using nitrogen gas. Section C was charged with nitrogen gas to a known pressure P = 2.5 MPa. Valve 5 was connected to a standard known volume previously evacuated cylinder vessel V = liter and opened until equilibrium was reached with a new pressure P 2, then the volume of section C was determined by the ideal gas equation of state: V C P V P P 2 () 2 The volume of section A and B were determined in the same manner. The granular adsorbent (0 g) was packed into the adsorption vessel and degassed for overnight under vacuum at a pressure of less than 3 Pa to clean the adsorbent. This 965
5 Volume3 December 2007 pretreatment enabled us to make adsorption measurements without changing the adsorbent. After the adsorbent had been regenerated, the vacuum pump was switched off. The water bath with electrical stirrer thermo stated hot plate was switched on until constant temperature was reached. The procedure for measuring equilibrium adsorption was as follow:. Section A and C were pressurized with the adsorbate, corresponding to a pressure P 3 then the number of moles of the adsorbate was estimated according to the relation: n P3 VC VA (2) RT Passage of gas to section B was allowed until equilibrium ( P 3 kpa/h). The equilibrium pressure was recorded as P Adsorber was isolated from the system. The number of moles of the adsorbate was estimated according to the relation: n P V V RT V 4 C A B 2 (3) The amount adsorbed of the adsorbate at partial pressure equal to P 4 is determined according to a material balance equation: n n2 q (4) W RESULTS & DISCUSSIONS The partial pressure and temperature ranges were chosen for each component in viewing to the expected values in the natural gas and oil well effluent properties and component vapor pressure, which almost must be greater than the component partial pressure under specified temperature, to avoid condensation of gases on the adsorbent. 2699
6 A. K. Mohammed Equilibrium Adsorption of Methane and Carbon Dioxide on 5A Molecular Sieve. The experimental results for single gas equilibrium were fitted with Langmuir and Freundlich equations. BET and other equations that represent multi layer adsorption were excluded because it is unlikely that multi layer adsorption could have occurred in this study were all temperatures above the critical (Yang & Saunders, 985). The Langmuir equation takes the form: q qm BP (5) BP The coefficients B and q m depend on temperature in accordance to the following equations: m a 2 q a T (6) 3 a T 4 B a e (7) The Freundlich equation takes the form: n q k F F P (8) The coefficients k F and n F depend on temperature in accordance to the following equations: F b 2 k b T (9) nf 3 4 b b T (0) The objective function (average relative error) used in the minimization routine was defined as: 967
7 Volume3 December N qi,exp qi,cal E () N i qi,exp where N is the number of experimental points and q i,exp and q i,cal are the experimental and calculated values of q i, respectively. The calculated constants for the two model equations along with the average relative error values were presented in table. The table shows that Langmuir equation correlates the experimental data with mean average relative error of.59% while 2.29% for Freundlich equation. Therefore, the best fit was achieved with Langmuir equation. The experimental equilibrium data was correlated by Langmuir equation and presented in figures 2 and 3. Table () Summary of single gas adsorption isotherms results Adsorbate.a.a 2.a 3.a 4 E % CO CH Adsorbate.b.b 2.b 3.b 4 E % CO CH All curves for any specified gas are similar in shape and are of the classic adsorption isotherm form. However, it was concluded that the strength of adsorption for CO 2 is greater than for CH 4, since it was recognized that CO 2 has isotherms with greater slopes at P 0, which indicates that the affinity for adsorption of CO 2 is greater than for CH 4. The results also indicate that there is a fair correlation for the amount of adsorption on an adsorbent correspond to increased adsorption with higher gas molecular weight or critical temperature. This result is in agreement with published works (Lewis et al., 950). Also, vapor pressure can be expected to be the predominant factor favoring adsorption. The much less volatile gas is more likely to condense on the surface since it has a much larger relative saturation value (P / P*) than the more volatile one. This result was verified by several authors (Hyun & Danner, 982). 269.
8 A. K. Mohammed Equilibrium Adsorption of Methane and Carbon Dioxide on 5A Molecular Sieve. Adsorption isotherms for the CH 4 and CO 2 gases on 5A molecular sieve at moderate temperature ranges are presented in figures 2 and 3. The marked decrease in adsorption with increasing temperature is obvious from these diagrams. The heat of adsorption ( H) was calculated by application of the Clausius Clapeyron equation (Smith, 988) at neighboring temperatures. When this equation is applied to the two phase system of gas and adsorbed component on the surface, we get: dp dt H T V V a (2) where V and V a are the volumes per mole of adsorbed component in the gas and on the surface, respectively. V a is very small and could be neglected. By assuming the ideal gas law for V we get: d ln P R d T H (3) Table 2 shows the results of this equation. Table (2) Heat of adsorption and condensation for gases Component H, (kj mole - ) H cond at BP (kj mole - ) CH CO Sublimes at 95 K In both cases the heats of adsorption are higher or expected to be higher than the latent heats of condensation but are low enough to be characterized as physical adsorption. CONCLUSIONS. Equilibrium isotherms for single component adsorption of methane and carbon dioxide on 5A molecular sieve can be correlated by Langmuir equation. 2. The strength of adsorption for carbon dioxide is greater than for methane on 5A molecular sieve. 969
9 Volume3 December The heat of adsorption for both methane and carbon dioxide on 5A molecular sieve always greater than the latent heat of condensation. NOMENCLATURE a,.., a 4 Langmuir isotherm coefficients - b,.., b 4 Freundlich isotherm coefficients - k F Constant in Freundlich equation Pa -nf n Number of moles mole n F Parameter in Freundlich equation - q Adsorbed phase concentration mole g - y Mole fraction in gas phase - B Adsorption coefficient in Langmuir equation Pa - E Average relative error - N Number of experimental points - P Pressure Pa P* Vapor pressure Pa R Ideal gas constant J mole - K - T Temperature K V Volume m 3 W Mass of adsorbent g θ Fraction of surface coverage - H Heat of adsorption J mole - 260
10 A. K. Mohammed Equilibrium Adsorption of Methane and Carbon Dioxide on 5A Molecular Sieve. 97
11 Volume3 December 2007 REFERENCES Hyun S.H. & Danner R.P., Equilibrium adsorption of ethane, ethylene, isobutane, carbon dioxide and their binary mixtures on 3 X molecular sieve, J. Chem. Eng. Data, 27, 96 (982). Kapoor A. & Yang R.T., Kinetic separation of methane-carbon dioxide mixture by adsorption on molecular sieve carbon, Chem. Eng. Sci., 44, 723 (989). Kumar R., Adsorption column blow down, Ind. Eng. Chem. Res., 28, 677 (989). Lewis W.K., Gilliland E.R., Chertow B. & Cadogan W.P., Adsorption equilibria, Ind. Eng. Chem., 42, 326 (950). Smith J.M., Chemical engineering kinetics, 3 rd ed., McGraw Hill, Singapore, 988. Tańczyk M. & Warmuziński K., Multi-component pressure swing adsorption. Part II. Experimental verification of the model, Chem. Eng. Proc., 37, 30 (998). Yang R.T. & Saunders J.T., Adsorption of gases on coals and heat-treated coals at elevated temperature and pressure, Fuel, 64, 66 (985). 26
High-Pressure Volumetric Analyzer
 High-Pressure Volumetric Analyzer High-Pressure Volumetric Analysis HPVA II Benefits Dual free-space measurement for accurate isotherm data Free space can be measured or entered Correction for non-ideality
High-Pressure Volumetric Analyzer High-Pressure Volumetric Analysis HPVA II Benefits Dual free-space measurement for accurate isotherm data Free space can be measured or entered Correction for non-ideality
Adsorption Processes. Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad
 Adsorption Processes Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad Contents Introduction Principles of adsorption Types of adsorption Definitions Brief history Adsorption isotherms Mechanism
Adsorption Processes Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad Contents Introduction Principles of adsorption Types of adsorption Definitions Brief history Adsorption isotherms Mechanism
12. Heat of melting and evaporation of water
 VS 12. Heat of melting and evaporation of water 12.1 Introduction The change of the physical state of a substance in general requires the absorption or release of heat. In this case, one speaks of a first
VS 12. Heat of melting and evaporation of water 12.1 Introduction The change of the physical state of a substance in general requires the absorption or release of heat. In this case, one speaks of a first
Investigation of Mixed Gas Sorption in Lab-Scale. Dr. Andreas Möller
 Investigation of Mixed Gas Sorption in Lab-Scale Dr. Andreas Möller 1 Technical Necessity: Application of porous Materials as Adsorbents Fine cleaning of Gases (i.e. purification of H 2, natural gas, bio
Investigation of Mixed Gas Sorption in Lab-Scale Dr. Andreas Möller 1 Technical Necessity: Application of porous Materials as Adsorbents Fine cleaning of Gases (i.e. purification of H 2, natural gas, bio
Kinetic, Thermodynamic and Regeneration Studies for CO 2 Adsorption onto Activated Carbon
 International Journal of Advanced Mechanical Engineering. ISSN 50-334 Volume 4, Number 1 (014), pp. 7-3 Research India Publications http://www.ripublication.com/ijame.htm Kinetic, Thermodynamic and Regeneration
International Journal of Advanced Mechanical Engineering. ISSN 50-334 Volume 4, Number 1 (014), pp. 7-3 Research India Publications http://www.ripublication.com/ijame.htm Kinetic, Thermodynamic and Regeneration
Vapor-hydrate phases equilibrium of (CH 4 +C 2 H 6 ) and (CH 4 +C 2 H 4 ) systems
 Pet.Sci.(2008)5:359-366 DOI 10.7/s12182-008-0042-0 359 Vapor-hydrate phases equilibrium of (CH 4 +C 2 H 6 ) and (CH 4 +C 2 H 4 ) systems Ma Qinglan, Chen Guangjin and Zhang Lingwei High Pressure Fluid
Pet.Sci.(2008)5:359-366 DOI 10.7/s12182-008-0042-0 359 Vapor-hydrate phases equilibrium of (CH 4 +C 2 H 6 ) and (CH 4 +C 2 H 4 ) systems Ma Qinglan, Chen Guangjin and Zhang Lingwei High Pressure Fluid
THE IRANIAN JAM PETROCHEMICAL S H 2 -PSA ENHANCEMENT USING A NEW STEPS SEQUENCE TABLE
 Petroleum & Coal ISSN 1337-707 Available online at www.vurup.sk/petroleum-coal Petroleum & Coal 56 (1) 13-18, 014 THE IRANIAN JAM PETROCHEMICAL S H -PSA ENHANCEMENT USING A NEW STEPS SEQUENCE TABLE Ehsan
Petroleum & Coal ISSN 1337-707 Available online at www.vurup.sk/petroleum-coal Petroleum & Coal 56 (1) 13-18, 014 THE IRANIAN JAM PETROCHEMICAL S H -PSA ENHANCEMENT USING A NEW STEPS SEQUENCE TABLE Ehsan
Proceedings of the 7th WSEAS International Conference on SYSTEM SCIENCE and SIMULATION in ENGINEERING (ICOSSSE '08)
 ADSORPTION EQUILIBRIA OF PROPANE ON ACTIVATED CARBON AND MOLECULAR SIEVES Z.YAAKOB 1, S.K.KAMARUDIN 1, I.KAMARUZAMAN 1, A.IBRAHIM 2 Department of Chemical & Process Engineering, Universiti Kebangsaan Malaysia
ADSORPTION EQUILIBRIA OF PROPANE ON ACTIVATED CARBON AND MOLECULAR SIEVES Z.YAAKOB 1, S.K.KAMARUDIN 1, I.KAMARUZAMAN 1, A.IBRAHIM 2 Department of Chemical & Process Engineering, Universiti Kebangsaan Malaysia
Adsorption (Ch 12) - mass transfer to an interface
 Adsorption (Ch 12) - mass transfer to an interface (Absorption - mass transfer to another phase) Gas or liquid adsorption (molecular) onto solid surface Porous solids provide high surface area per weight
Adsorption (Ch 12) - mass transfer to an interface (Absorption - mass transfer to another phase) Gas or liquid adsorption (molecular) onto solid surface Porous solids provide high surface area per weight
Chapter 11 Ideal gases
 OCR (A) specifications: 5.4.10c,d,e,i,j,k Chapter 11 Ideal gases Worksheet Worked examples Practical: Determining absolute zero of temperature from the pressure law End-of-chapter test Marking scheme:
OCR (A) specifications: 5.4.10c,d,e,i,j,k Chapter 11 Ideal gases Worksheet Worked examples Practical: Determining absolute zero of temperature from the pressure law End-of-chapter test Marking scheme:
SUPERCRITICAL CARBON DIOXIDE DESORPTION OF XYLENE FROM ZEOLITE
 SUPERCRITICAL CARBON DIOXIDE DESORPTION OF XYLENE FROM ZEOLITE Stéphane VITU and Danielle BARTH ( * ) Institut National Polytechnique de Lorraine Ecole Nationale Supérieure des Industries Chimiques Laboratoire
SUPERCRITICAL CARBON DIOXIDE DESORPTION OF XYLENE FROM ZEOLITE Stéphane VITU and Danielle BARTH ( * ) Institut National Polytechnique de Lorraine Ecole Nationale Supérieure des Industries Chimiques Laboratoire
Experiment and Simulation Study of Multi-component Gas Adsorption On MSC3A By Volumetric method
 Experiment and Simulation Study of Multi-component Gas Adsorption On MSC3A By Volumetric method Kazuyuki. Chihara, Shohei. Koide, Masashi Nomoto, Yuzo. Amari, Meiji University,Kawasaki,Japan Abstruct Equilibria
Experiment and Simulation Study of Multi-component Gas Adsorption On MSC3A By Volumetric method Kazuyuki. Chihara, Shohei. Koide, Masashi Nomoto, Yuzo. Amari, Meiji University,Kawasaki,Japan Abstruct Equilibria
Vacuum Pumps. Two general classes exist: Gas transfer physical removal of matter. Mechanical, diffusion, turbomolecular
 Vacuum Technology Vacuum Pumps Two general classes exist: Gas transfer physical removal of matter Mechanical, diffusion, turbomolecular Adsorption entrapment of matter Cryo, sublimation, ion Mechanical
Vacuum Technology Vacuum Pumps Two general classes exist: Gas transfer physical removal of matter Mechanical, diffusion, turbomolecular Adsorption entrapment of matter Cryo, sublimation, ion Mechanical
4.1. Physics Module Form 4 Chapter 4 - Heat GCKL UNDERSTANDING THERMAL EQUILIBRIUM. What is thermal equilibrium?
 Physics Module Form 4 Chapter 4 - Heat GCKL 2010 4.1 4 UNDERSTANDING THERMAL EQUILIBRIUM What is thermal equilibrium? 1. (, Temperature ) is a form of energy that flows from a hot body to a cold body.
Physics Module Form 4 Chapter 4 - Heat GCKL 2010 4.1 4 UNDERSTANDING THERMAL EQUILIBRIUM What is thermal equilibrium? 1. (, Temperature ) is a form of energy that flows from a hot body to a cold body.
ADSORPTION AND DESORPTION OF CO ON SOLID SORBENTS
 ADSORPTION AND DESORPTION OF CO ON SOLID SORBENTS Ranjani Siriwardane (rsiiw@netl.doe.gov; 34-85-4513) Ming Shen (mshen@netl.doe.gov; 34-85-411) Edward Fisher (efishe@netl.doe.gov; 34-85-411) James Poston
ADSORPTION AND DESORPTION OF CO ON SOLID SORBENTS Ranjani Siriwardane (rsiiw@netl.doe.gov; 34-85-4513) Ming Shen (mshen@netl.doe.gov; 34-85-411) Edward Fisher (efishe@netl.doe.gov; 34-85-411) James Poston
Preparation of biomass derived porous carbon: Application for methane energy storage
 Edith Cowan University Research Online ECU Publications Post 013 016 Preparation of biomass derived porous carbon: Application for methane energy storage Yong Sun Edith Cowan University, y.sun@ecu.edu.au
Edith Cowan University Research Online ECU Publications Post 013 016 Preparation of biomass derived porous carbon: Application for methane energy storage Yong Sun Edith Cowan University, y.sun@ecu.edu.au
Chapter 5: The First Law of Thermodynamics: Closed Systems
 Chapter 5: The First Law of Thermodynamics: Closed Systems The first law of thermodynamics can be simply stated as follows: during an interaction between a system and its surroundings, the amount of energy
Chapter 5: The First Law of Thermodynamics: Closed Systems The first law of thermodynamics can be simply stated as follows: during an interaction between a system and its surroundings, the amount of energy
ADSORPTION IN MICROPOROUS MATERIALS: ANALYTICAL EQUATIONS FOR TYPE I ISOTHERMS AT HIGH PRESSURE
 ADSORPTION IN MICROPOROUS MATERIALS: ANALYTICAL EQUATIONS FOR TYPE I ISOTHERMS AT HIGH PRESSURE A. L. MYERS Department of Chemical and Biomolecular Engineering University of Pennsylvania, Philadelphia
ADSORPTION IN MICROPOROUS MATERIALS: ANALYTICAL EQUATIONS FOR TYPE I ISOTHERMS AT HIGH PRESSURE A. L. MYERS Department of Chemical and Biomolecular Engineering University of Pennsylvania, Philadelphia
Experimental Methods and Analysis
 Chapter 3 28 Experimental Methods and Analysis 1. Equipment The fundamental basis of quantitative adsorption analysis is the measurement of excess adsorption isotherms. Each isotherm comprises a series
Chapter 3 28 Experimental Methods and Analysis 1. Equipment The fundamental basis of quantitative adsorption analysis is the measurement of excess adsorption isotherms. Each isotherm comprises a series
A 3 Vapor pressure of volatile liquids
 Versuchsanleitungen zum Praktikum Physikalische Chemie für Anfänger 1 A 3 Vapor pressure of volatile liquids Purpose The purpose of this experiment is to determine the vaporization enthalpy, entropy and
Versuchsanleitungen zum Praktikum Physikalische Chemie für Anfänger 1 A 3 Vapor pressure of volatile liquids Purpose The purpose of this experiment is to determine the vaporization enthalpy, entropy and
Kinetic Separation of Oxygen and Argon Using Molecular Sieve Carbon
 Adsorption 6, 15 22 (2000) c 2000 Kluwer Academic Publishers. Manufactured in The Netherlands. Kinetic Separation of Oxygen and Argon Using Molecular Sieve Carbon SALIL U. REGE AND RALPH T. YANG Department
Adsorption 6, 15 22 (2000) c 2000 Kluwer Academic Publishers. Manufactured in The Netherlands. Kinetic Separation of Oxygen and Argon Using Molecular Sieve Carbon SALIL U. REGE AND RALPH T. YANG Department
Separation Benzene and Toluene from BTX using Zeolite 13X
 Iraqi Journal of Chemical and Petroleum Engineering Iraqi Journal of Chemical and Petroleum Engineering Vol.9 No.3 (September 27) 7-24 ISSN: 997-4884 University of Baghdad College of Engineering Separation
Iraqi Journal of Chemical and Petroleum Engineering Iraqi Journal of Chemical and Petroleum Engineering Vol.9 No.3 (September 27) 7-24 ISSN: 997-4884 University of Baghdad College of Engineering Separation
Adsorption dynamics and effects of carbon to zeolite ratio of layered beds for multicomponent gas adsorption
 Korean J. Chem. Eng., 28(2),583-590 (2011) DOI: 10.1007/s11814-010-0399-9 INVITED REVIEW PAPER Adsorption dynamics and effects of carbon to zeolite ratio of layered beds for multicomponent gas adsorption
Korean J. Chem. Eng., 28(2),583-590 (2011) DOI: 10.1007/s11814-010-0399-9 INVITED REVIEW PAPER Adsorption dynamics and effects of carbon to zeolite ratio of layered beds for multicomponent gas adsorption
The Adsorption Isotherms of the Bleaching of Sunflower-Seed Oil
 Tr. J. of Chemistry 22 (1998), 143 148. c TÜBİTAK The Adsorption Isotherms of the Bleaching of Sunflower-Seed Oil Hüseyin TOPALLAR Trakya University, Faculty of Sciences and Letters Department of Chemistry,
Tr. J. of Chemistry 22 (1998), 143 148. c TÜBİTAK The Adsorption Isotherms of the Bleaching of Sunflower-Seed Oil Hüseyin TOPALLAR Trakya University, Faculty of Sciences and Letters Department of Chemistry,
AE 3051, Lab #16. Investigation of the Ideal Gas State Equation. By: George P. Burdell. Group E3
 AE 3051, Lab #16 Investigation of the Ideal Gas State Equation By: George P. Burdell Group E3 Summer Semester 000 Abstract The validity of the ideal gas equation of state was experimentally tested for
AE 3051, Lab #16 Investigation of the Ideal Gas State Equation By: George P. Burdell Group E3 Summer Semester 000 Abstract The validity of the ideal gas equation of state was experimentally tested for
Pressure Swing Adsorption: A Gas Separation & Purification Process
 Pressure Swing Adsorption: A Gas Separation & Purification Process Pressure swing adsorption is an adsorption-based process that has been used for various gas separation and purification purposes. Separation
Pressure Swing Adsorption: A Gas Separation & Purification Process Pressure swing adsorption is an adsorption-based process that has been used for various gas separation and purification purposes. Separation
PHYS102 Previous Exam Problems. Temperature, Heat & The First Law of Thermodynamics
 PHYS102 Previous Exam Problems CHAPTER 18 Temperature, Heat & The First Law of Thermodynamics Equilibrium & temperature scales Thermal expansion Exchange of heat First law of thermodynamics Heat conduction
PHYS102 Previous Exam Problems CHAPTER 18 Temperature, Heat & The First Law of Thermodynamics Equilibrium & temperature scales Thermal expansion Exchange of heat First law of thermodynamics Heat conduction
3.5. Kinetic Approach for Isotherms
 We have arrived isotherm equations which describe adsorption from the two dimensional equation of state via the Gibbs equation (with a saturation limit usually associated with monolayer coverage). The
We have arrived isotherm equations which describe adsorption from the two dimensional equation of state via the Gibbs equation (with a saturation limit usually associated with monolayer coverage). The
Silver-Decorated Hafnium Metal-Organic Framework for. Ethylene/Ethane Separation
 Supporting Information Silver-Decorated Hafnium Metal-Organic Framework for Ethylene/Ethane Separation Yuxiang Wang, Zhigang Hu, Youdong Cheng, and Dan Zhao * Department of Chemical & Biomolecular Engineering,
Supporting Information Silver-Decorated Hafnium Metal-Organic Framework for Ethylene/Ethane Separation Yuxiang Wang, Zhigang Hu, Youdong Cheng, and Dan Zhao * Department of Chemical & Biomolecular Engineering,
CO 2 capture by Adsorption Processes: From Materials to Process Development to Practical Implementation
 CO 2 capture by Adsorption Processes: From Materials to Process Development to Practical Implementation Paul A. Webley Dept. of Chemical Engineering Talloires, July 2010 Outline Adsorption for Post-Combustion
CO 2 capture by Adsorption Processes: From Materials to Process Development to Practical Implementation Paul A. Webley Dept. of Chemical Engineering Talloires, July 2010 Outline Adsorption for Post-Combustion
Adsorption Equilibrium and Kinetics of H 2 O on Zeolite 13X
 Korean J. Chem. Eng., 8(4), 55-530 (00) Adsorption Equilibrium and Kinetics of H O on Zeolite 3X Young Ki Ryu*, Seung Ju Lee, Jong Wha Kim and Chang-Ha Lee *External Relations Department, Procter & Gamble
Korean J. Chem. Eng., 8(4), 55-530 (00) Adsorption Equilibrium and Kinetics of H O on Zeolite 3X Young Ki Ryu*, Seung Ju Lee, Jong Wha Kim and Chang-Ha Lee *External Relations Department, Procter & Gamble
Thermochemistry/Calorimetry. Determination of the enthalpy of vaporization of liquids LEC 02. What you need: What you can learn about
 LEC 02 Thermochemistry/Calorimetry Determination of the enthalpy of vaporization of liquids What you can learn about Enthalpy of vaporisation Entropy of vaporisation Trouton s rule Calorimetry Heat capacity
LEC 02 Thermochemistry/Calorimetry Determination of the enthalpy of vaporization of liquids What you can learn about Enthalpy of vaporisation Entropy of vaporisation Trouton s rule Calorimetry Heat capacity
Lecture 7. Sorption-Separation Equipment
 Lecture 7. Sorption-Separation Equipment Adsorption - Stirred-tank, slurry operation - Cyclic fixed-bed batch operation - Thermal (temperature)-swing adsorption - Fluidizing bed for adsorption and moving
Lecture 7. Sorption-Separation Equipment Adsorption - Stirred-tank, slurry operation - Cyclic fixed-bed batch operation - Thermal (temperature)-swing adsorption - Fluidizing bed for adsorption and moving
The dynamic adsorption of Xe on a fixed bed adsorber at 77 K
 The dynamic adsorption of Xe on a fixed bed adsorber at 77 K Bin Long 1,2,1), Jun- Li Li 1, Qun- Shu Wang 2, Shu- Juan Feng 2, Guo- Qing Zhou 2, Tian- Cheng Feng 2, Yan- Jie Tian 2, Huai- Cheng Ma 2 1
The dynamic adsorption of Xe on a fixed bed adsorber at 77 K Bin Long 1,2,1), Jun- Li Li 1, Qun- Shu Wang 2, Shu- Juan Feng 2, Guo- Qing Zhou 2, Tian- Cheng Feng 2, Yan- Jie Tian 2, Huai- Cheng Ma 2 1
Copyright 2008, University of Chicago, Department of Physics. Experiment I. RATIO OF SPECIFIC HEATS OF GASES; γ C p
 Experiment I RATIO OF SPECIFIC HEATS OF GASES; γ C p / C v 1. Recommended Reading M. W. Zemansky, Heat and Thermodynamics, Fifth Edition, McGraw Hill, 1968, p. 122-132, 161-2. 2. Introduction You have
Experiment I RATIO OF SPECIFIC HEATS OF GASES; γ C p / C v 1. Recommended Reading M. W. Zemansky, Heat and Thermodynamics, Fifth Edition, McGraw Hill, 1968, p. 122-132, 161-2. 2. Introduction You have
CHEM 254 EXPERIMENT 2 Critical point determination for SF 6
 CHEM 254 EXPERIMENT 2 Critical point determination for SF 6 The equation of state of a gas defines the relationship between the pressure, temperature and volume of the gas. For ideal gases the equation
CHEM 254 EXPERIMENT 2 Critical point determination for SF 6 The equation of state of a gas defines the relationship between the pressure, temperature and volume of the gas. For ideal gases the equation
VAPOR-LIQUID EQUILIBRIA OF BINARY AND TERNARY SYSTEMS CONTAINING HYDROGEN AND LIGHT HYDROCARBONS'
 Nomenclature A = constant in Eq.(4) [ K] B - constant in Eq.(4) [-] C = coefficient in Eq.(8) [-] F = parameter in Eq.(8) [-] K = vapor-liquid equilibrium ratio [-] PiQ = vapor pressure of pure component
Nomenclature A = constant in Eq.(4) [ K] B - constant in Eq.(4) [-] C = coefficient in Eq.(8) [-] F = parameter in Eq.(8) [-] K = vapor-liquid equilibrium ratio [-] PiQ = vapor pressure of pure component
Solubility of carbon dioxide in aqueous solutions of 2-amino-2-ethyl-1,3-propanediol
 Fluid Phase Equilibria 202 (2002) 359 366 Solubility of carbon dioxide in aqueous solutions of 2-amino-2-ethyl-1,3-propanediol Jung-Yeon Park a, Sang Jun Yoon a, Huen Lee a,, Ji-Ho Yoon b, Jae-Goo Shim
Fluid Phase Equilibria 202 (2002) 359 366 Solubility of carbon dioxide in aqueous solutions of 2-amino-2-ethyl-1,3-propanediol Jung-Yeon Park a, Sang Jun Yoon a, Huen Lee a,, Ji-Ho Yoon b, Jae-Goo Shim
Evaluation of the Characteristic of Adsorption in a Double-Effect Adsorption Chiller with FAM-Z01
 Journal of Materials Science and Chemical Engineering, 2016, 4, 8-19 http://www.scirp.org/journal/msce ISSN Online: 2327-6053 ISSN Print: 2327-6045 Evaluation of the Characteristic of Adsorption in a Double-Effect
Journal of Materials Science and Chemical Engineering, 2016, 4, 8-19 http://www.scirp.org/journal/msce ISSN Online: 2327-6053 ISSN Print: 2327-6045 Evaluation of the Characteristic of Adsorption in a Double-Effect
11B, 11E Temperature and heat are related but not identical.
 Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
HEAT- I Part - A C D A B. Te m p. Heat input
 e m p HE- I Part -. solid material is supplied with heat at a constant rate. he temperature of the material is changing with heat input as shown in the graph. Study the graph carefully and answer the following
e m p HE- I Part -. solid material is supplied with heat at a constant rate. he temperature of the material is changing with heat input as shown in the graph. Study the graph carefully and answer the following
Comparison of distillation arrangement for the recovery process of dimethyl sulfoxide
 Korean J. Chem. Eng., 24(3), 438-444 (2007) SHORT COMMUNICATION Comparison of distillation arrangement for the recovery process of dimethyl sulfoxide Jungho Cho and Dong Min Kim* Department of Chemical
Korean J. Chem. Eng., 24(3), 438-444 (2007) SHORT COMMUNICATION Comparison of distillation arrangement for the recovery process of dimethyl sulfoxide Jungho Cho and Dong Min Kim* Department of Chemical
Gases. A gas. Difference between gas and vapor: Why Study Gases?
 Gases Chapter 5 Gases A gas Uniformly fills any container. Is easily compressed. Mixes completely with any other gas. Exerts pressure on its surroundings. Difference between gas and vapor: A gas is a substance
Gases Chapter 5 Gases A gas Uniformly fills any container. Is easily compressed. Mixes completely with any other gas. Exerts pressure on its surroundings. Difference between gas and vapor: A gas is a substance
Adsorption Equilibrium of Solvent Vapors on Activated Carbons
 Korean J. Chem. Eng., 18(4), 518-524 (2001) Adsorption Equilibrium of Solvent Vapors on Activated Carbons Dae Jung Kim*, Wang Geun Shim and Hee Moon *Neophotech Inc., 633-1 Goan-Ri, Backam-Myun, Yongin
Korean J. Chem. Eng., 18(4), 518-524 (2001) Adsorption Equilibrium of Solvent Vapors on Activated Carbons Dae Jung Kim*, Wang Geun Shim and Hee Moon *Neophotech Inc., 633-1 Goan-Ri, Backam-Myun, Yongin
Evaluation of pressure-temperature swing adsorption for sulfur hexafluoride (SF 6 ) recovery from SF 6 and N 2 gas mixture
 Korean J. Chem. Eng., 28(11), 2196-2201 (2011) DOI: 10.1007/s11814-011-0104-7 INVITED REVIEW PAPER Evaluation of pressure-temperature swing adsorption for sulfur hexafluoride (SF 6 ) recovery from SF 6
Korean J. Chem. Eng., 28(11), 2196-2201 (2011) DOI: 10.1007/s11814-011-0104-7 INVITED REVIEW PAPER Evaluation of pressure-temperature swing adsorption for sulfur hexafluoride (SF 6 ) recovery from SF 6
Tutorial 1 (not important for 2015)
 Tutorial 1 (not important for 2015) 1 st Law of thermodynamics and other basic concepts Do No. 5 (05-03-2015) 1. One mole of an ideal gas is allowed to expand against a piston which supports 41 atm pressures.
Tutorial 1 (not important for 2015) 1 st Law of thermodynamics and other basic concepts Do No. 5 (05-03-2015) 1. One mole of an ideal gas is allowed to expand against a piston which supports 41 atm pressures.
PROPULSION LAB MANUAL
 PROPULSION LAB MANUAL Measurement of Calorific Value of a Solid Fuel Sample using a Bomb Calorimeter DEPARTMENT OF AEROSPACE ENGINEERING Indian Institute of Technology Kharagpur CONTENTS 1. Introduction
PROPULSION LAB MANUAL Measurement of Calorific Value of a Solid Fuel Sample using a Bomb Calorimeter DEPARTMENT OF AEROSPACE ENGINEERING Indian Institute of Technology Kharagpur CONTENTS 1. Introduction
International Journal of Scientific Research and Modern Education (IJSRME) ISSN (Online): (www.rdmodernresearch.com) Volume I, Issue I,
 OXYGEN CONCENTRATORS A STUDY Mohammed Salique*, Nabila Rumane**, RohanBholla***, Siddharth Bhawnani**** & Anita Kumari***** Chemical Engineering Department, Thadomal Shahani Engineering College, Off Linking
OXYGEN CONCENTRATORS A STUDY Mohammed Salique*, Nabila Rumane**, RohanBholla***, Siddharth Bhawnani**** & Anita Kumari***** Chemical Engineering Department, Thadomal Shahani Engineering College, Off Linking
UBMCC11 - THERMODYNAMICS. B.E (Marine Engineering) B 16 BASIC CONCEPTS AND FIRST LAW PART- A
 UBMCC11 - THERMODYNAMICS B.E (Marine Engineering) B 16 UNIT I BASIC CONCEPTS AND FIRST LAW PART- A 1. What do you understand by pure substance? 2. Define thermodynamic system. 3. Name the different types
UBMCC11 - THERMODYNAMICS B.E (Marine Engineering) B 16 UNIT I BASIC CONCEPTS AND FIRST LAW PART- A 1. What do you understand by pure substance? 2. Define thermodynamic system. 3. Name the different types
Chapter 3 PROPERTIES OF PURE SUBSTANCES
 Thermodynamics: An Engineering Approach Seventh Edition Yunus A. Cengel, Michael A. Boles McGraw-Hill, 2011 Chapter 3 PROPERTIES OF PURE SUBSTANCES Copyright The McGraw-Hill Companies, Inc. Permission
Thermodynamics: An Engineering Approach Seventh Edition Yunus A. Cengel, Michael A. Boles McGraw-Hill, 2011 Chapter 3 PROPERTIES OF PURE SUBSTANCES Copyright The McGraw-Hill Companies, Inc. Permission
Properties of Gases. The perfect gas. States of gases Gas laws Kinetic model of gases (Ch th ed, th ed.) Real gases
 Properties of Gases Chapter 1 of Physical Chemistry - 6th Edition P.W. Atkins. Chapter 1 and a little bit of Chapter 24 of 7th Edition. Chapter 1 and a little bit of Chapter 21 of 8th edition. The perfect
Properties of Gases Chapter 1 of Physical Chemistry - 6th Edition P.W. Atkins. Chapter 1 and a little bit of Chapter 24 of 7th Edition. Chapter 1 and a little bit of Chapter 21 of 8th edition. The perfect
MANOMETRICAL STUDIES ON THE ADSORPTION OF SOME ORGANIC GASES/VAPORS ON THE SURFACE OF ACTIVATED CHARCOAL
 MANOMETRICAL STUDIES ON THE ADSORPTION OF SOME ORGANIC GASES/VAPORS ON THE SURFACE OF ACTIVATED CHARCOAL Ghayasuddin Khan KCP-I, Pakistan Atomic Energy Commission P.O. Box 1114, Islamabad, Pakistan Fahim
MANOMETRICAL STUDIES ON THE ADSORPTION OF SOME ORGANIC GASES/VAPORS ON THE SURFACE OF ACTIVATED CHARCOAL Ghayasuddin Khan KCP-I, Pakistan Atomic Energy Commission P.O. Box 1114, Islamabad, Pakistan Fahim
Experimental Vapor-Liquid Equilibria for the Carbon Dioxide + Octane, and Carbon Dioxide + Decane Systems from 313 to 373 K
 Experimental Vapor-Liquid Equilibria for the Carbon Dioxide + Octane, and Carbon Dioxide + Decane Systems from to K R. Jiménez-Gallegos, Luis A. Galicia-Luna* and O. Elizalde-Solis Instituto Politécnico
Experimental Vapor-Liquid Equilibria for the Carbon Dioxide + Octane, and Carbon Dioxide + Decane Systems from to K R. Jiménez-Gallegos, Luis A. Galicia-Luna* and O. Elizalde-Solis Instituto Politécnico
VACUUM PUMPING METHODS
 VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
Distillation of Liquids: Separation of 2-Propanol from Water by Fractional Distillation
 Distillation of Liquids: Separation of 2-Propanol from Water by Fractional Distillation Introduction: Distillation is the process of vaporizing a liquid, condensing the vapor, and collecting the condensate
Distillation of Liquids: Separation of 2-Propanol from Water by Fractional Distillation Introduction: Distillation is the process of vaporizing a liquid, condensing the vapor, and collecting the condensate
Atmospheric Analysis Gases. Sampling and analysis of gaseous compounds
 Atmospheric Analysis Gases Sampling and analysis of gaseous compounds Introduction - External environment (ambient air) ; global warming, acid rain, introduction of pollutants, etc - Internal environment
Atmospheric Analysis Gases Sampling and analysis of gaseous compounds Introduction - External environment (ambient air) ; global warming, acid rain, introduction of pollutants, etc - Internal environment
Correlation of High Pressure Density Behaviors for Fluid Mixtures made of Carbon Dioxide with Solvent at K
 The Open Thermodynamics Journal, 9, 3, -6 Open Access Correlation of High Pressure Density Behaviors for Fluid Mixtures made of Carbon Dioxide with Solvent at 33.5 K Masahiro Kato, *, Masaki Kokubo, Kensuke
The Open Thermodynamics Journal, 9, 3, -6 Open Access Correlation of High Pressure Density Behaviors for Fluid Mixtures made of Carbon Dioxide with Solvent at 33.5 K Masahiro Kato, *, Masaki Kokubo, Kensuke
I. CHEM. E. SYMPOSIUM SERIES No. 49
 FLAMMABILITY AND EXPLOSIBILITY OF AMMONIA G.F.P.Harris, P.E.MacDermott Research Dept. ICI Organics Division, Blackley, Manchester The flammability limits of oxygen/nitrogen/ammonia mixtures have been determined
FLAMMABILITY AND EXPLOSIBILITY OF AMMONIA G.F.P.Harris, P.E.MacDermott Research Dept. ICI Organics Division, Blackley, Manchester The flammability limits of oxygen/nitrogen/ammonia mixtures have been determined
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supporting Information Novel Nanoporous Ferrocenyl Framework for Clean Energy Application Qingquan
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supporting Information Novel Nanoporous Ferrocenyl Framework for Clean Energy Application Qingquan
Methods for High Pressure VLE Measurement
 Experimental Methods for High Pressure VLE Measurement by Rajpuri Sai Kiran Singh", Varun Bhalla*, S.NKaul**, VA.Shinde*** *UG Students; ** HOD., Dept. of Chemical Engineering; *** Assistant Professor,
Experimental Methods for High Pressure VLE Measurement by Rajpuri Sai Kiran Singh", Varun Bhalla*, S.NKaul**, VA.Shinde*** *UG Students; ** HOD., Dept. of Chemical Engineering; *** Assistant Professor,
Classification following properties of the system in Intensive and Extensive
 Unit I Classification following properties of the system in Intensive and Extensive Extensive : mass, weight, volume, potential energy, Kinetic energy, Internal energy, entropy, exergy, energy, magnetization
Unit I Classification following properties of the system in Intensive and Extensive Extensive : mass, weight, volume, potential energy, Kinetic energy, Internal energy, entropy, exergy, energy, magnetization
Determination of effective diffusion coefficient of methane adsorption on activated carbon
 Trade Science Inc. ISSN : 0974-7443 Volume 7 Issue 2 CTAIJ 7(2) 2012 [39-44] Determination of effective diffusion coefficient of methane adsorption on activated carbon Alireza Azimi*, Masoomeh Mirzaei
Trade Science Inc. ISSN : 0974-7443 Volume 7 Issue 2 CTAIJ 7(2) 2012 [39-44] Determination of effective diffusion coefficient of methane adsorption on activated carbon Alireza Azimi*, Masoomeh Mirzaei
Introduction: Introduction. material is transferred from one phase (gas, liquid, or solid) into another.
 Introduction: Virtually all commercial chemical processes involve operations in which material is transferred from one phase (gas, liquid, or solid) into another. rewing a cup of Coffee (Leaching) Removal
Introduction: Virtually all commercial chemical processes involve operations in which material is transferred from one phase (gas, liquid, or solid) into another. rewing a cup of Coffee (Leaching) Removal
Building multiple adsorption sites in porous polymer networks for carbon capture applications
 Electronic Supplementary Information Building multiple adsorption sites in porous polymer networks for carbon capture applications Weigang Lu, a Wolfgang M. Verdegaal, a Jiamei Yu, b Perla B. Balbuena,
Electronic Supplementary Information Building multiple adsorption sites in porous polymer networks for carbon capture applications Weigang Lu, a Wolfgang M. Verdegaal, a Jiamei Yu, b Perla B. Balbuena,
Chapter 11 Gases 1 Copyright McGraw-Hill 2009
 Chapter 11 Gases Copyright McGraw-Hill 2009 1 11.1 Properties of Gases The properties of a gas are almost independent of its identity. (Gas molecules behave as if no other molecules are present.) Compressible
Chapter 11 Gases Copyright McGraw-Hill 2009 1 11.1 Properties of Gases The properties of a gas are almost independent of its identity. (Gas molecules behave as if no other molecules are present.) Compressible
Modification In Charging Composition In Order To Arrive At Desired Circulation Composition In The Context Of Sorption Compressor Based J-T Cooler
 Modification In Charging Composition In Order To Arrive At Desired Circulation Composition In The Context Of Sorption Compressor Based J-T Cooler R. N. Mehta, S. L. Bapat, M. D. Atrey Department of Mechanical
Modification In Charging Composition In Order To Arrive At Desired Circulation Composition In The Context Of Sorption Compressor Based J-T Cooler R. N. Mehta, S. L. Bapat, M. D. Atrey Department of Mechanical
Scale-up problems are often perceived as difficult. Here the reaction calorimetry has proven to be
 APPLICATION OF REACTION CALORIMETRY FOR THE SOLUTION OF SCALE-UP PROBLEMS A paper from the RC User Forum Europe, Interlaken, 1995 Francis Stoessel, Ciba AG, Basel, Switzerland. Scale-up problems are often
APPLICATION OF REACTION CALORIMETRY FOR THE SOLUTION OF SCALE-UP PROBLEMS A paper from the RC User Forum Europe, Interlaken, 1995 Francis Stoessel, Ciba AG, Basel, Switzerland. Scale-up problems are often
Separation of CO 2 from CH 4 using Mixed-Ligand Metal-Organic Frameworks
 Langmuir Supporting Information Separation of CO 2 from CH 4 using Mixed-Ligand Metal-Organic Frameworks Youn-Sang Bae, Karen L. Mulfort, %, Houston Frost, Patrick Ryan, Sudeep Punnathanam, Linda J. Broadbelt,
Langmuir Supporting Information Separation of CO 2 from CH 4 using Mixed-Ligand Metal-Organic Frameworks Youn-Sang Bae, Karen L. Mulfort, %, Houston Frost, Patrick Ryan, Sudeep Punnathanam, Linda J. Broadbelt,
MODELLING OF EQUILIBRIUM SORPTION OF M-XYLENE ON DAY ZEOLITE AND SUPERCRITICAL DESORPTION
 MODELLING OF EQUILIBRIUM SORPTION OF M-XYLENE ON DAY ZEOLITE AND SUPERCRITICAL DESORPTION Taoufik EL BRIHI, Jean-Noël JAUBERT, Danielle BARTH (*) Laboratoire de Thermodynamique des Milieux Polyphasés ENSIC
MODELLING OF EQUILIBRIUM SORPTION OF M-XYLENE ON DAY ZEOLITE AND SUPERCRITICAL DESORPTION Taoufik EL BRIHI, Jean-Noël JAUBERT, Danielle BARTH (*) Laboratoire de Thermodynamique des Milieux Polyphasés ENSIC
Thermodynamics I. Properties of Pure Substances
 Thermodynamics I Properties of Pure Substances Dr.-Eng. Zayed Al-Hamamre 1 Content Pure substance Phases of a pure substance Phase-change processes of pure substances o Compressed liquid, Saturated liquid,
Thermodynamics I Properties of Pure Substances Dr.-Eng. Zayed Al-Hamamre 1 Content Pure substance Phases of a pure substance Phase-change processes of pure substances o Compressed liquid, Saturated liquid,
A comparative study on the recovery of 1,2-dichloroethane and the removal of benzene contained in the byproducts of VCM process
 Korean J. Chem. Eng., 27(3), 970-976 (2010) DOI: 10.1007/s11814-010-0206-7 RAPID COMMUNICATION A comparative study on the recovery of 1,2-dichloroethane and the removal of benzene contained in the byproducts
Korean J. Chem. Eng., 27(3), 970-976 (2010) DOI: 10.1007/s11814-010-0206-7 RAPID COMMUNICATION A comparative study on the recovery of 1,2-dichloroethane and the removal of benzene contained in the byproducts
PROPERTIES OF PURE SUBSTANCES. Chapter 3. Mehmet Kanoglu. Thermodynamics: An Engineering Approach, 6 th Edition. Yunus A. Cengel, Michael A.
 Thermodynamics: An Engineering Approach, 6 th Edition Yunus A. Cengel, Michael A. Boles McGraw-Hill, 2008 Chapter 3 PROPERTIES OF PURE SUBSTANCES Mehmet Kanoglu Copyright The McGraw-Hill Companies, Inc.
Thermodynamics: An Engineering Approach, 6 th Edition Yunus A. Cengel, Michael A. Boles McGraw-Hill, 2008 Chapter 3 PROPERTIES OF PURE SUBSTANCES Mehmet Kanoglu Copyright The McGraw-Hill Companies, Inc.
Temperature C. Heat Added (Joules)
 Now let s apply the heat stuff to real-world stuff like phase changes and the energy or cost it takes to carry it out. A heating curve...a plot of temperature of a substance vs heat added to a substance.
Now let s apply the heat stuff to real-world stuff like phase changes and the energy or cost it takes to carry it out. A heating curve...a plot of temperature of a substance vs heat added to a substance.
Thermodynamics INTRODUCTION AND BASIC CONCEPTS. Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
 Thermodynamics INTRODUCTION AND BASIC CONCEPTS Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. THERMODYNAMICS AND ENERGY Thermodynamics: The science of energy.
Thermodynamics INTRODUCTION AND BASIC CONCEPTS Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. THERMODYNAMICS AND ENERGY Thermodynamics: The science of energy.
Biogas Clean-up and Upgrading by Adsorption on Commercial Molecular Sieves
 A publication of CHEMICAL ENGINEERING TRANSACTIONS VOL. 29, 2012 Guest Editors: Petar Sabev Varbanov, Hon Loong Lam, Jiří Jaromír Klemeš Copyright 2012, AIDIC Servizi S.r.l., ISBN 978-88-95608-20-4; ISSN
A publication of CHEMICAL ENGINEERING TRANSACTIONS VOL. 29, 2012 Guest Editors: Petar Sabev Varbanov, Hon Loong Lam, Jiří Jaromír Klemeš Copyright 2012, AIDIC Servizi S.r.l., ISBN 978-88-95608-20-4; ISSN
DESORPTION OF TOLUENE FROM MODIFIED CLAYS USING SUPERCRITICAL CARBON DIOXIDE
 Brazilian Journal of Chemical Engineering ISSN 14-6632 Printed in Brazil Vol. 21, No. 4, pp. 641-646, October - December 24 DESORPTION OF TOLUENE FROM MODIFIED CLAYS USING SUPERCRITICAL CARBON DIOXIDE
Brazilian Journal of Chemical Engineering ISSN 14-6632 Printed in Brazil Vol. 21, No. 4, pp. 641-646, October - December 24 DESORPTION OF TOLUENE FROM MODIFIED CLAYS USING SUPERCRITICAL CARBON DIOXIDE
IMPLEMENTATION OF ETHANOL HEAT PIPE AT CETIAT
 IMPLEMENTATION OF ETHANOL HEAT PIPE AT CETIAT JO Favreau 1, E Georgin 1, B Savanier 1, A. Merlone 2 1 CETIAT, 96100 Villeurbanne, France 2 INRIM, Torino, Italy Abstract. CETIAT is a calibration laboratory
IMPLEMENTATION OF ETHANOL HEAT PIPE AT CETIAT JO Favreau 1, E Georgin 1, B Savanier 1, A. Merlone 2 1 CETIAT, 96100 Villeurbanne, France 2 INRIM, Torino, Italy Abstract. CETIAT is a calibration laboratory
Interactions and exchange of CO 2 and H 2 O in coals: an investigation by low-field NMR relaxation
 Interactions and exchange of CO 2 and H 2 O in coals: an investigation by low-field NMR relaxation Xiaoxiao Sun, Yanbin Yao *, Dameng Liu, Derek Elsworth, Zhejun Pan Supplementary Figures 1 Supplementary
Interactions and exchange of CO 2 and H 2 O in coals: an investigation by low-field NMR relaxation Xiaoxiao Sun, Yanbin Yao *, Dameng Liu, Derek Elsworth, Zhejun Pan Supplementary Figures 1 Supplementary
Recap: Introduction 12/1/2015. EVE 402 Air Pollution Generation and Control. Adsorption
 EVE 402 Air Pollution Generation and Control Chapter #6 Lectures Adsorption Recap: Solubility: the extent of absorption into the bulk liquid after the gas has diffused through the interface An internal
EVE 402 Air Pollution Generation and Control Chapter #6 Lectures Adsorption Recap: Solubility: the extent of absorption into the bulk liquid after the gas has diffused through the interface An internal
Possibilities and Limits for the Determination of. Adsorption Data Pure Gases and Gas Mixtures
 MOF-Workshop, Leipzig, March 2010 Possibilities and Limits for the Determination of Adsorption Data Pure Gases and Gas Mixtures Reiner Staudt Instutut für Nichtklassische Chemie e.v. Permoserstraße 15,
MOF-Workshop, Leipzig, March 2010 Possibilities and Limits for the Determination of Adsorption Data Pure Gases and Gas Mixtures Reiner Staudt Instutut für Nichtklassische Chemie e.v. Permoserstraße 15,
Engineering Thermodynamics. Chapter 1. Introductory Concepts and Definition
 1.1 Introduction Chapter 1 Introductory Concepts and Definition Thermodynamics may be defined as follows : Thermodynamics is an axiomatic science which deals with the relations among heat, work and properties
1.1 Introduction Chapter 1 Introductory Concepts and Definition Thermodynamics may be defined as follows : Thermodynamics is an axiomatic science which deals with the relations among heat, work and properties
SOLUBILITIES OF NON-STEROIDAL ANTI-INFLAMMATORY DRUGS IN SUPERCRITICAL CARBON DIOXIDE
 SOLUBILITIES OF NON-STEROIDAL ANTI-INFLAMMATORY DRUGS IN SUPERCRITICAL CARBON DIOXIDE Ming-Jer Lee*, Cheng-Chou Tsai, Ho-mu Lin Department of Chemical Engineering, National Taiwan University of Science
SOLUBILITIES OF NON-STEROIDAL ANTI-INFLAMMATORY DRUGS IN SUPERCRITICAL CARBON DIOXIDE Ming-Jer Lee*, Cheng-Chou Tsai, Ho-mu Lin Department of Chemical Engineering, National Taiwan University of Science
4.1. Physics Module Form 4 Chapter 4 - Heat GCKL UNDERSTANDING THERMAL EQUILIBRIUM. What is thermal equilibrium?
 4.1 4 UNDERSTANDING THERMAL EQUILIBRIUM What is thermal equilibrium? 1. ( Heat, Temperature ) is a form of energy that flows from a hot body to a cold body. 2. The SI unit for ( heat, temperature) is Joule,
4.1 4 UNDERSTANDING THERMAL EQUILIBRIUM What is thermal equilibrium? 1. ( Heat, Temperature ) is a form of energy that flows from a hot body to a cold body. 2. The SI unit for ( heat, temperature) is Joule,
Topic 19b. Thermal Properties of Matter
 Topic 19b The infra-red image of a head shows the distribution of heat. Different colours indicate different temperatures. Which do you think are the warmest regions? Thermal Properties of Matter contents
Topic 19b The infra-red image of a head shows the distribution of heat. Different colours indicate different temperatures. Which do you think are the warmest regions? Thermal Properties of Matter contents
Isopropyl Alcohol Vapour Removal from Diluted Gaseous Stream by Adsorption: Experimental Results and Dynamic Model
 451 A publication of CHEMICAL ENGINEERING TRANSACTIONS VOL. 47, 16 Guest Editors: Angelo Chianese, Luca Di Palma, Elisabetta Petrucci, Marco Stoller Copyright 16, AIDIC Servizi S.r.l., ISBN 978-88-9568-38-9;
451 A publication of CHEMICAL ENGINEERING TRANSACTIONS VOL. 47, 16 Guest Editors: Angelo Chianese, Luca Di Palma, Elisabetta Petrucci, Marco Stoller Copyright 16, AIDIC Servizi S.r.l., ISBN 978-88-9568-38-9;
Table of Contents. Experiment 1: Vapour Pressure of Water at High Temperature 2. Experiment 2: Heat Capacity of Gases 5
 1 Table of Contents EXPERIMENT PAGE Experiment 1: Vapour Pressure of Water at High Temperature 2 Experiment 2: Heat Capacity of Gases 5 Experiment 3: Joule-Thomson Effect 11 Experiment 4: Thermal and Electrical
1 Table of Contents EXPERIMENT PAGE Experiment 1: Vapour Pressure of Water at High Temperature 2 Experiment 2: Heat Capacity of Gases 5 Experiment 3: Joule-Thomson Effect 11 Experiment 4: Thermal and Electrical
Thermodynamic Properties of Low-GWP Refrigerant for Centrifugal Chiller
 Purdue University Purdue e-pubs International Refrigeration and Air Conditioning Conference School of Mechanical Engineering 2016 Thermodynamic Properties of Low-GWP Refrigerant for Centrifugal Chiller
Purdue University Purdue e-pubs International Refrigeration and Air Conditioning Conference School of Mechanical Engineering 2016 Thermodynamic Properties of Low-GWP Refrigerant for Centrifugal Chiller
Chapter 1 INTRODUCTION AND BASIC CONCEPTS
 Thermodynamics: An Engineering Approach Seventh Edition in SI Units Yunus A. Cengel, Michael A. Boles McGraw-Hill, 2011 Chapter 1 INTRODUCTION AND BASIC CONCEPTS Mehmet Kanoglu University of Gaziantep
Thermodynamics: An Engineering Approach Seventh Edition in SI Units Yunus A. Cengel, Michael A. Boles McGraw-Hill, 2011 Chapter 1 INTRODUCTION AND BASIC CONCEPTS Mehmet Kanoglu University of Gaziantep
The Vacuum Sorption Solution
 The Total Sorption Solution The Vacuum Sorption Solution The Vacuum Sorption Solution www.thesorptionsolution.com About the Technique DVS Vacuum - the only gravimetric system that supports static and dynamic
The Total Sorption Solution The Vacuum Sorption Solution The Vacuum Sorption Solution www.thesorptionsolution.com About the Technique DVS Vacuum - the only gravimetric system that supports static and dynamic
Hydrogenation of CO Over a Cobalt/Cerium Oxide Catalyst for Production of Lower Olefins
 Hydrogenation of CO Over a Cobalt/Cerium Oxide Catalyst for Production of Lower Olefins Proceedings of European Congress of Chemical Engineering (ECCE-6) Copenhagen, 16-2 September 27 Hydrogenation of
Hydrogenation of CO Over a Cobalt/Cerium Oxide Catalyst for Production of Lower Olefins Proceedings of European Congress of Chemical Engineering (ECCE-6) Copenhagen, 16-2 September 27 Hydrogenation of
Distillation. Boiling
 Distillation The most important technique for separating and purifying organic liquids is distillation 21. A gross oversimplification of the technique is this: the impure liquid in one vessel is vaporized,
Distillation The most important technique for separating and purifying organic liquids is distillation 21. A gross oversimplification of the technique is this: the impure liquid in one vessel is vaporized,
Heuristic design of pressure swing adsorption: a preliminary study
 Heuristic design of pressure swing adsorption: a preliminary study S. Jain a, A.S. Moharir a,p. Li b, *, G. Wozny b a Department of Chemical Engineering, Indian Institute of Technology, Bombay 400 076,
Heuristic design of pressure swing adsorption: a preliminary study S. Jain a, A.S. Moharir a,p. Li b, *, G. Wozny b a Department of Chemical Engineering, Indian Institute of Technology, Bombay 400 076,
First Law of Thermodynamics Closed Systems
 First Law of Thermodynamics Closed Systems Content The First Law of Thermodynamics Energy Balance Energy Change of a System Mechanisms of Energy Transfer First Law of Thermodynamics in Closed Systems Moving
First Law of Thermodynamics Closed Systems Content The First Law of Thermodynamics Energy Balance Energy Change of a System Mechanisms of Energy Transfer First Law of Thermodynamics in Closed Systems Moving
S.E. (Chemical Engineering) (Second Semester)EXAMINATION, 2012 THERMODYNAMICS-I (2008 PATTERN) Time : Three Hours Maximum Marks : 100
 Total No. of Questions 12] [Total No. of Printed Pages 7 Seat No. [4162]-189 S.E. (Chemical Engineering) (Second Semester)EXAMINATION, 2012 THERMODYNAMICS-I (2008 PATTERN) Time : Three Hours Maximum Marks
Total No. of Questions 12] [Total No. of Printed Pages 7 Seat No. [4162]-189 S.E. (Chemical Engineering) (Second Semester)EXAMINATION, 2012 THERMODYNAMICS-I (2008 PATTERN) Time : Three Hours Maximum Marks
Storage of Hydrogen, Methane and Carbon Dioxide in Highly Porous Covalent Organic Frameworks for Clean Energy Applications
 Storage of Hydrogen, Methane and Carbon Dioxide in Highly Porous Covalent Organic Frameworks for Clean Energy Applications (Supporting Information: 33 pages) Hiroyasu Furukawa and Omar M. Yaghi Center
Storage of Hydrogen, Methane and Carbon Dioxide in Highly Porous Covalent Organic Frameworks for Clean Energy Applications (Supporting Information: 33 pages) Hiroyasu Furukawa and Omar M. Yaghi Center
CHAPTER 8 ENTROPY. Blank
 CHAPER 8 ENROPY Blank SONNAG/BORGNAKKE SUDY PROBLEM 8-8. A heat engine efficiency from the inequality of Clausius Consider an actual heat engine with efficiency of η working between reservoirs at and L.
CHAPER 8 ENROPY Blank SONNAG/BORGNAKKE SUDY PROBLEM 8-8. A heat engine efficiency from the inequality of Clausius Consider an actual heat engine with efficiency of η working between reservoirs at and L.
Heat capacity of gases
 Heat capacity of gases LEP Related topics Equation of state for ideal gases, 1st law of thermodynamics, universal gas constant, degree of freedom, mole volumes, isobars, isotherms, isochors and adiabatic
Heat capacity of gases LEP Related topics Equation of state for ideal gases, 1st law of thermodynamics, universal gas constant, degree of freedom, mole volumes, isobars, isotherms, isochors and adiabatic
Gas Chromatography Separation of H 2 -D 2 -Ar Using Pd/K
 Gas Chromatography Separation of H 2 -D 2 -Ar Using Pd/K FTP/P1-06 X. J. Qian 1), D. L. Luo 1), C. Qin 1), G. Q. Huang 1), W. Yang 1) 1) China Academy of Engineering Physics, Mianyang, SiChuan, P.R.China
Gas Chromatography Separation of H 2 -D 2 -Ar Using Pd/K FTP/P1-06 X. J. Qian 1), D. L. Luo 1), C. Qin 1), G. Q. Huang 1), W. Yang 1) 1) China Academy of Engineering Physics, Mianyang, SiChuan, P.R.China
SEPARATION BY BARRIER
 SEPARATION BY BARRIER SEPARATION BY BARRIER Phase 1 Feed Barrier Phase 2 Separation by barrier uses a barrier which restricts and/or enhances the movement of certain chemical species with respect to other
SEPARATION BY BARRIER SEPARATION BY BARRIER Phase 1 Feed Barrier Phase 2 Separation by barrier uses a barrier which restricts and/or enhances the movement of certain chemical species with respect to other
A) 2.0 atm B) 2.2 atm C) 2.4 atm D) 2.9 atm E) 3.3 atm
 Name: Date: 1. On a cold day ( 3 C), the gauge pressure on a tire reads 2.0 atm. If the tire is heated to 27 C, what will be the absolute pressure of the air inside the tire? A) 2.0 atm B) 2.2 atm C) 2.4
Name: Date: 1. On a cold day ( 3 C), the gauge pressure on a tire reads 2.0 atm. If the tire is heated to 27 C, what will be the absolute pressure of the air inside the tire? A) 2.0 atm B) 2.2 atm C) 2.4
