3. High Temperature Gases FPK1 2009/MZ 1/23
|
|
|
- Milton Gilmore
- 6 years ago
- Views:
Transcription
1 3. High Temerature Gases FP 9/MZ /3
2 Terms and Concets Dissociation Diatomic element gases Bond energy, dissociation energy (enthali) Flood s dissociation diagram Vaorization, eaoration, boiling, sublimation Heat of eaoration, enthaly of eaoration FP 9/MZ /3
3 Bond dissociation enthaly Only a limited number of enthalies of formation hae been measured, and there are many reactions for which H o f data is not aailable for one or more reagent. When this haens, H o rxn for the reaction can not be redicted. The enthaly of reaction can be estimated using bonddissociation enthalies. By definition, the bond-dissociation enthaly for an X-Y bond is the enthaly of the gas-hase reaction in which this bond is broken to gie isolated X and Y atoms. XY(g) X(g) + Y(g) FP 9/MZ 3/3
4 Bond dissociation enthaly The bond-dissociation enthaly for a C-H bond can be calculated by combining H o f data to gie a net equation in which the only thing that haens is the breaking of C-H bonds in the gas hase. CH 4 (g) C(s) + H (g) Ho = mol x 74.8 kj/mol CH 4 C(s) C(g) Ho = mol x kj/mol C H (g) 4 H(g) Ho = 4 mol x 7.65 kj/mol H CH 4 (g) C(g) + 4 H(g) Ho = 66.9 kj If it takes 66 kj/mol to break the four moles of C-H bonds in a mole of CH4, the aerage bond-dissociation enthaly for a single C-H bond is about 45 kj/mol. Bond-dissociation enthalies are always ositie numbers because it takes energy to break a bond. When a table of bond energies is used to estimate the enthaly associated with the formation of a bond, the sign becomes negatie because energy is released when bonds are formed. FP 9/MZ 4/3
5 Reference: Huheey, s. A- to A-34; T.L. Cottrell, "The Strengths of Chemical Bonds," nd ed., Butterworths, London, 958; B. deb. Darwent, "National Standard Reference Data Series," National Bureau of Standards, No. 3, Washington, DC, 97; S.W. Benson, J. Chem. Educ., 4, 5 (965). FP 9/MZ 5/3
6 Element gases Many elements form two-atomic molecules; these dissociate: O O Na Na (g) Equilibrium: (I) (II) (III) P i ni n tot Na Na n Na tot n Na n Na, degree of dissociation FP 9/MZ 6/3
7 Degree of dissociation s The degree of dissociation in gases is denoted by the symbol α where α refers to the ercentage of gas molecules which dissociate. Various relationshis between and α exist deending on the stoichiometry of the equation. FP 9/MZ 7/3
8 Degree of dissociation s : examle N O 4 (g) NO (g) When assuming mol N O 4 that dissociates Left N O 4 will be (-α), NO formed will be α Thus n NO4 =(- α); n NO = α n tot = n NO4 +n NO =- α + α =+ α =( NO ) / NO4 FP 9/MZ 8/3
9 FP 9/MZ 9/3 Degree of dissociation s And rearranging gies tot tot tot O N NO O N x x NO tot tot tot tot tot
10 Degree of dissociation s 4 tot We aroximate: and further: ln H RT S 98 5 cal mol S R We can generalize: f H 98, T tot Degree of dissociation can be estimated if we know H 98 and secify T and tot (see Flood s diagram). FP 9/MZ /3
11 FP 9/MZ /3
12 FP 9/MZ /3
13 Boiling oint The boiling oint of a liquid is the temerature at which the aor ressure of the liquid equals the enironmental ressure surrounding the liquid A liquid in a acuum enironment has a lower boiling oint than when the liquid is at atmosheric ressure. A liquid in a high ressure enironment has a higher boiling oint than when the liquid is at atmosheric ressure. In other words, the boiling oint of liquids aries with and deends uon the surrounding enironmental ressure. FP 9/MZ 3/3
14 Heat of aorisation The enthaly of aorization, (symbol H), also known as the heat of aorization or heat of eaoration, is the energy required to transform a gien quantity of a substance into a gas. It is often measured at the normal boiling oint of a substance; although tabulated alues are usually corrected to 98, The heat of aorization is temerature-deendent, though a constant heat of aorization can only be assumed for small temerature ranges The heat of aorization diminishes with increasing temerature and it anishes comletely at the critical temerature (Tr=) because aboe the critical temerature the liquid and aor hases don't coexist anymore. FP 9/MZ 4/3
15 FP 9/MZ 5/3 Determining T b Vaorization equilibrium: NaCl (s,l) NaCl (g) At boiling oint (T B ), NaCl = atm, thus: R S RT H RT G NaCl e e e ln S H T S T H R S RT H R S RT H B B B B NaCl Clausius Claeyron
16 For ideal gases: S 5 cal mol We get: T B 5 mol cal H Boiling oint (ery) roughly linearly deendant on H. FP 9/MZ 6/3
17 H FP 9/MZ 7/3
18 H Hydrogen bonding gies larges H Polar substances hae higher H than similar non olar substances Similar bonding in similar comounds gies similar H NH 3 lowest H amonh hydrogen bounded molecules F, Cl, Br, I regular increase in H Methane, ethane, roane regular increase in H FP 9/MZ 8/3
19 FP 9/MZ 9/3 Determining T b and H: Neat aroach H can be determined by measuring x at different temeratures. Tb = boiling tem at atm (bar) ln ln ln H T T R T T R H R S RT H
20 Ex. A liquid has aour ressure of 54mmHg at 5 C and 648mmHg at 45 C. What is H and T b? FP 9/MZ /3
21 Heat of sublimation The heat of sublimation, or enthaly of sublimation, is defined as the heat required to sublime one mole of the substance at a gien combination of temerature and ressure, usually standard temerature and ressure (STP). The enthaly of sublimation is the heat of sublimation for aorizing one mole of the solid substance under three secific conditions: () the ressure remains constant, () the only ossible work that occurs is exansion against the atmoshere (so-called PdV work) and (3) the temerature remains constant during the rocess. A heat of sublimation for a substance is only alid for conersion of the ure solid to the ure gaseous state of the substance. FP 9/MZ /3
22 FP 9/MZ /3
23 Terms and Concets Dissociation Diatomic element gases Bond energy, dissociation energy (enthali) Flood s dissociation diagram Vaorization, eaoration, boiling, sublimation Heat of eaoration, enthaly of eaoration FP 9/MZ 3/3
dn i where we have used the Gibbs equation for the Gibbs energy and the definition of chemical potential
 Chem 467 Sulement to Lectures 33 Phase Equilibrium Chemical Potential Revisited We introduced the chemical otential as the conjugate variable to amount. Briefly reviewing, the total Gibbs energy of a system
Chem 467 Sulement to Lectures 33 Phase Equilibrium Chemical Potential Revisited We introduced the chemical otential as the conjugate variable to amount. Briefly reviewing, the total Gibbs energy of a system
Thermodynamics in combustion
 Thermodynamics in combustion 2nd ste in toolbox Thermodynamics deals with a equilibrium state and how chemical comosition can be calculated for a system with known atomic or molecular comosition if 2 indeendent
Thermodynamics in combustion 2nd ste in toolbox Thermodynamics deals with a equilibrium state and how chemical comosition can be calculated for a system with known atomic or molecular comosition if 2 indeendent
Chemistry 531 Spring 2009 Problem Set 6 Solutions
 Chemistry 531 Sring 2009 Problem Set 6 Solutions 1. In a thermochemical study of N 2, the following heat caacity data were found: t 0 C,m d 27.2Jmol 1 K 1 f t b f C,m d 23.4Jmol 1 K 1 C,m d 11.4Jmol 1
Chemistry 531 Sring 2009 Problem Set 6 Solutions 1. In a thermochemical study of N 2, the following heat caacity data were found: t 0 C,m d 27.2Jmol 1 K 1 f t b f C,m d 23.4Jmol 1 K 1 C,m d 11.4Jmol 1
I affirm that I have never given nor received aid on this examination. I understand that cheating in the exam will result in a grade F for the class.
 Chem340 Physical Chemistry for Biochemists Exam Mar 16, 011 Your Name _ I affirm that I have never given nor received aid on this examination. I understand that cheating in the exam will result in a grade
Chem340 Physical Chemistry for Biochemists Exam Mar 16, 011 Your Name _ I affirm that I have never given nor received aid on this examination. I understand that cheating in the exam will result in a grade
Principles of Food and Bioprocess Engineering (FS 231) Solutions to Example Problems on Evaporation
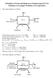 Princiles of Food and Biorocess Engineering (FS 231) Solutions to Examle Problems on Eaoration 1. The system diagram is as follows: Solids balance equation: 0.1 (m f) = 0.5 (1,000) Thus, m f = 5,000 kg/hr
Princiles of Food and Biorocess Engineering (FS 231) Solutions to Examle Problems on Eaoration 1. The system diagram is as follows: Solids balance equation: 0.1 (m f) = 0.5 (1,000) Thus, m f = 5,000 kg/hr
FUGACITY. It is simply a measure of molar Gibbs energy of a real gas.
 FUGACITY It is simly a measure of molar Gibbs energy of a real gas. Modifying the simle equation for the chemical otential of an ideal gas by introducing the concet of a fugacity (f). The fugacity is an
FUGACITY It is simly a measure of molar Gibbs energy of a real gas. Modifying the simle equation for the chemical otential of an ideal gas by introducing the concet of a fugacity (f). The fugacity is an
Equilibrium point of any reaction is characterized by a single number: K eq is the equilibrium constant for the reaction
 Lecture 19 Equilibrium Constant Equilibrium oint of any reaction is characterized by a single number: K eq is the equilibrium constant for the reaction In general: ja + kb R + qs K eq [ R] [ S] [ A] [
Lecture 19 Equilibrium Constant Equilibrium oint of any reaction is characterized by a single number: K eq is the equilibrium constant for the reaction In general: ja + kb R + qs K eq [ R] [ S] [ A] [
Introductory Inorganic Chemistry
 Introductory Inorganic Chemistry What is Inorganic Chemistry? As: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 3 Classes of Inorganic Substances Elements Ionic Compounds Covalent Compounds Atomic/Molecular
Introductory Inorganic Chemistry What is Inorganic Chemistry? As: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 3 Classes of Inorganic Substances Elements Ionic Compounds Covalent Compounds Atomic/Molecular
The Second Law: The Machinery
 The Second Law: The Machinery Chater 5 of Atkins: The Second Law: The Concets Sections 3.7-3.9 8th Ed, 3.3 9th Ed; 3.4 10 Ed.; 3E 11th Ed. Combining First and Second Laws Proerties of the Internal Energy
The Second Law: The Machinery Chater 5 of Atkins: The Second Law: The Concets Sections 3.7-3.9 8th Ed, 3.3 9th Ed; 3.4 10 Ed.; 3E 11th Ed. Combining First and Second Laws Proerties of the Internal Energy
Name: Date: Grade. Work Session # 12: Intermolecular Forces
 Name: Date: Grade Work Session # 12: Intermolecular Forces All questions below must be answered during the lab. Show all work and express your answers with appropriate units and the correct number of significant
Name: Date: Grade Work Session # 12: Intermolecular Forces All questions below must be answered during the lab. Show all work and express your answers with appropriate units and the correct number of significant
= = 10.1 mol. Molar Enthalpies of Vaporization (at Boiling Point) Molar Enthalpy of Vaporization (kj/mol)
 Ch 11 (Sections 11.1 11.5) Liquid Phase Volume and Density - Liquid and solid are condensed phases and their volumes are not simple to calculate. - This is different from gases, which have volumes that
Ch 11 (Sections 11.1 11.5) Liquid Phase Volume and Density - Liquid and solid are condensed phases and their volumes are not simple to calculate. - This is different from gases, which have volumes that
Phase transition. Asaf Pe er Background
 Phase transition Asaf Pe er 1 November 18, 2013 1. Background A hase is a region of sace, throughout which all hysical roerties (density, magnetization, etc.) of a material (or thermodynamic system) are
Phase transition Asaf Pe er 1 November 18, 2013 1. Background A hase is a region of sace, throughout which all hysical roerties (density, magnetization, etc.) of a material (or thermodynamic system) are
THE FIRST LAW OF THERMODYNAMICS
 THE FIRST LA OF THERMODYNAMIS 9 9 (a) IDENTIFY and SET UP: The ressure is constant and the volume increases (b) = d Figure 9 Since is constant, = d = ( ) The -diagram is sketched in Figure 9 The roblem
THE FIRST LA OF THERMODYNAMIS 9 9 (a) IDENTIFY and SET UP: The ressure is constant and the volume increases (b) = d Figure 9 Since is constant, = d = ( ) The -diagram is sketched in Figure 9 The roblem
Homework 01. Phase Changes and Solutions
 HW01 - Phase Changes and Solu!ons! This is a preview of the published version of the quiz Started: Jan 16 at 1:pm Quiz Instruc!ons Homework 01 Phase Changes and Solutions Question 1 Given that you have
HW01 - Phase Changes and Solu!ons! This is a preview of the published version of the quiz Started: Jan 16 at 1:pm Quiz Instruc!ons Homework 01 Phase Changes and Solutions Question 1 Given that you have
AT 25 C! CH10090 Thermodynamics (part 2) Enthalpy changes during reactions. Let s remember what we did in CH10089
 CH10090 hermodynamics (art ) Let s remember what we did in CH10089 Enthaly changes during reactions o o o H98 ( reaction) = νi Hf, 98( roducts) νi Hf, 98( reactants) ν i reresents the stoichiometric coefficient.
CH10090 hermodynamics (art ) Let s remember what we did in CH10089 Enthaly changes during reactions o o o H98 ( reaction) = νi Hf, 98( roducts) νi Hf, 98( reactants) ν i reresents the stoichiometric coefficient.
Prediction of Vapour Pressures of Dioxin Congeners. R & D Center, Baoshan Iron & Steel Co., Ltd., Fujin Rd., Baoshan District, Shanghai , China
 Prediction of Vaour Pressures of Dioxin Congeners Xian-Wei Li, 1 Etsuro Shibata, 2 Eiki Kasai, 2 Takashi Nakamura 2 1 R & D Center, Baoshan Iron & Steel Co., Ltd., Fujin Rd., Baoshan District, Shanghai
Prediction of Vaour Pressures of Dioxin Congeners Xian-Wei Li, 1 Etsuro Shibata, 2 Eiki Kasai, 2 Takashi Nakamura 2 1 R & D Center, Baoshan Iron & Steel Co., Ltd., Fujin Rd., Baoshan District, Shanghai
Conservation of Energy Thermodynamic Energy Equation
 Conseration of Energy Thermodynamic Energy Equation The reious two sections dealt with conseration of momentum (equations of motion) and the conseration of mass (continuity equation). This section addresses
Conseration of Energy Thermodynamic Energy Equation The reious two sections dealt with conseration of momentum (equations of motion) and the conseration of mass (continuity equation). This section addresses
HEAT, WORK, AND THE FIRST LAW OF THERMODYNAMICS
 HET, ORK, ND THE FIRST L OF THERMODYNMIS 8 EXERISES Section 8. The First Law of Thermodynamics 5. INTERPRET e identify the system as the water in the insulated container. The roblem involves calculating
HET, ORK, ND THE FIRST L OF THERMODYNMIS 8 EXERISES Section 8. The First Law of Thermodynamics 5. INTERPRET e identify the system as the water in the insulated container. The roblem involves calculating
Lecture 2. Review of Basic Concepts
 Lecture 2 Review of Basic Concepts Thermochemistry Enthalpy H heat content H Changes with all physical and chemical changes H Standard enthalpy (25 C, 1 atm) (H=O for all elements in their standard forms
Lecture 2 Review of Basic Concepts Thermochemistry Enthalpy H heat content H Changes with all physical and chemical changes H Standard enthalpy (25 C, 1 atm) (H=O for all elements in their standard forms
Adsorption of Atoms and Molecules. Physisorption Chemisorption Surface Bonding Kinetics of Adsorption/Diffusion/Desorption (Scattering Dynamics)
 Adsortion of Atoms and Molecules Physisortion Chemisortion Surface Bonding Kinetics of Adsortion/Diffusion/Desortion (Scattering Dynamics) Outcomes of Collision Process Rebound (elastically or inelastically)
Adsortion of Atoms and Molecules Physisortion Chemisortion Surface Bonding Kinetics of Adsortion/Diffusion/Desortion (Scattering Dynamics) Outcomes of Collision Process Rebound (elastically or inelastically)
Lecture 8, the outline
 Lecture, the outline loose end: Debye theory of solids more remarks on the first order hase transition. Bose Einstein condensation as a first order hase transition 4He as Bose Einstein liquid Lecturer:
Lecture, the outline loose end: Debye theory of solids more remarks on the first order hase transition. Bose Einstein condensation as a first order hase transition 4He as Bose Einstein liquid Lecturer:
Solids, Liquids and Gases
 WHY? Why is water usually a liquid and not a gas? Why does liquid water boil at such a high temperature for such a small molecule? Why does ice float on water? Why do snowflakes have 6 sides? Why is I
WHY? Why is water usually a liquid and not a gas? Why does liquid water boil at such a high temperature for such a small molecule? Why does ice float on water? Why do snowflakes have 6 sides? Why is I
Chem 112 Exam 1 Version A Spring /16/ :00am/Odago, M. O.
 Chem 112 Exam 1 Version A Spring 2011 02/16/2011 10:00am/Odago, M. O. 1. The pressure of a certain gas is measured to be 25.1 mmhg. What is this pressure expressed in units of pascals? (1 atm=1.0125 x10
Chem 112 Exam 1 Version A Spring 2011 02/16/2011 10:00am/Odago, M. O. 1. The pressure of a certain gas is measured to be 25.1 mmhg. What is this pressure expressed in units of pascals? (1 atm=1.0125 x10
PHYS1001 PHYSICS 1 REGULAR Module 2 Thermal Physics Chapter 17 First Law of Thermodynamics
 PHYS1001 PHYSICS 1 REGULAR Module Thermal Physics Chater 17 First Law of Thermodynamics References: 17.1 to 17.9 Examles: 17.1 to 17.7 Checklist Thermodynamic system collection of objects and fields. If
PHYS1001 PHYSICS 1 REGULAR Module Thermal Physics Chater 17 First Law of Thermodynamics References: 17.1 to 17.9 Examles: 17.1 to 17.7 Checklist Thermodynamic system collection of objects and fields. If
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following is most likely not a gas at room temperature? 1) A) H2 B) Cl2
Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following is most likely not a gas at room temperature? 1) A) H2 B) Cl2
ALE 27. Hess s Law. (Reference: Chapter 6 - Silberberg 5 th edition)
 Answer Key ALE 27. Hess s Law (Reference: Chapter 6 - Silberberg 5 th edition) Important!! For answers that involve a calculation you must show your work neatly using dimensional analysis with correct
Answer Key ALE 27. Hess s Law (Reference: Chapter 6 - Silberberg 5 th edition) Important!! For answers that involve a calculation you must show your work neatly using dimensional analysis with correct
Thermodynamics. Standard enthalpy change, H
 Standard enthalpy change, H Thermodynamics Enthalpy change, H, is defined as the heat energy change measured under conditions of constant pressure. The value of the enthalpy change for a particular reaction
Standard enthalpy change, H Thermodynamics Enthalpy change, H, is defined as the heat energy change measured under conditions of constant pressure. The value of the enthalpy change for a particular reaction
JJMIE Jordan Journal of Mechanical and Industrial Engineering
 JJMIE Jordan Journal of Mechanical and Industrial Engineering Volume, Number, Jun. 8 ISSN 995-6665 Pages 7-75 Efficiency of Atkinson Engine at Maximum Power Density using emerature Deendent Secific Heats
JJMIE Jordan Journal of Mechanical and Industrial Engineering Volume, Number, Jun. 8 ISSN 995-6665 Pages 7-75 Efficiency of Atkinson Engine at Maximum Power Density using emerature Deendent Secific Heats
5.2 Energy. N Goalby chemrevise.org Lattice Enthalpy. Definitions of enthalpy changes
 5.2 Energy 5.2.1 Lattice Enthalpy Definitions of enthalpy changes Enthalpy change of formation The standard enthalpy change of formation of a compound is the energy transferred when 1 mole of the compound
5.2 Energy 5.2.1 Lattice Enthalpy Definitions of enthalpy changes Enthalpy change of formation The standard enthalpy change of formation of a compound is the energy transferred when 1 mole of the compound
_ + Units of Energy. Energy in Thermochemistry. Thermochemistry. Energy flow between system and surroundings. 100º C heat 50º C
 Units of Energy Like we saw with pressure, many different units are used throughout the world for energy. SI unit for energy 1kg m 1J = 2 s 2 Joule (J) calorie (cal) erg (erg) electron volts (ev) British
Units of Energy Like we saw with pressure, many different units are used throughout the world for energy. SI unit for energy 1kg m 1J = 2 s 2 Joule (J) calorie (cal) erg (erg) electron volts (ev) British
2011, Robert Ayton. All rights reserved.
 Liquids, Solids, and Intermolecular Forces Outline 1. Phase Diagrams and Triple Point Diagrams 2. Intermolecular Forces Review 1. Phase Diagrams and Triple Point Diagrams Phase Diagram of Water Triple
Liquids, Solids, and Intermolecular Forces Outline 1. Phase Diagrams and Triple Point Diagrams 2. Intermolecular Forces Review 1. Phase Diagrams and Triple Point Diagrams Phase Diagram of Water Triple
HOMEWORK 11-1 (pp )
 CHAPTER 11 HOMEWORK 11-1 (pp. 333 335) VOCABULARY Define. 1. Gay-Lussac s law of combining volumes of gases 2. Avogadro s law Answer each question. 3. Write and explain the equation that expresses the
CHAPTER 11 HOMEWORK 11-1 (pp. 333 335) VOCABULARY Define. 1. Gay-Lussac s law of combining volumes of gases 2. Avogadro s law Answer each question. 3. Write and explain the equation that expresses the
Ch 10 Practice Problems
 Ch 10 Practice Problems 1. Which of the following result(s) in an increase in the entropy of the system? I. (See diagram.) II. Br 2(g) Br 2(l) III. NaBr(s) Na + (aq) + Br (aq) IV. O 2(298 K) O 2(373 K)
Ch 10 Practice Problems 1. Which of the following result(s) in an increase in the entropy of the system? I. (See diagram.) II. Br 2(g) Br 2(l) III. NaBr(s) Na + (aq) + Br (aq) IV. O 2(298 K) O 2(373 K)
I. Multiple Choice Questions (Type-I)
 I. Multiple Choice Questions (Type-I) 1. Thermodynamics is not concerned about. (i) energy changes involved in a chemical reaction. the extent to which a chemical reaction proceeds. the rate at which a
I. Multiple Choice Questions (Type-I) 1. Thermodynamics is not concerned about. (i) energy changes involved in a chemical reaction. the extent to which a chemical reaction proceeds. the rate at which a
FACULTY OF SCIENCE MID-TERM EXAMINATION CHEMISTRY 120 GENERAL CHEMISTRY MIDTERM 1. Examiners: Prof. B. Siwick Prof. I. Butler Dr. A.
 FACULTY OF SCIENCE MID-TERM EXAMINATION CHEMISTRY 120 GENERAL CHEMISTRY MIDTERM 1 Examiners: Prof. B. Siwick Prof. I. Butler Dr. A. Fenster Name: INSTRUCTIONS 1. Enter your student number and name on the
FACULTY OF SCIENCE MID-TERM EXAMINATION CHEMISTRY 120 GENERAL CHEMISTRY MIDTERM 1 Examiners: Prof. B. Siwick Prof. I. Butler Dr. A. Fenster Name: INSTRUCTIONS 1. Enter your student number and name on the
Enthalpy Chapter 5.3-4,7
 Enthalpy Chapter 5.3-4,7 heat transfer in (endothermic), +q heat transfer out (exothermic), -q SYSTEM E = q + w w transfer in (+w) w transfer out (-w) Internal Energy at Constant Volume E = E K + E P ΔE
Enthalpy Chapter 5.3-4,7 heat transfer in (endothermic), +q heat transfer out (exothermic), -q SYSTEM E = q + w w transfer in (+w) w transfer out (-w) Internal Energy at Constant Volume E = E K + E P ΔE
Chap 10 Part 4Ta.notebook December 08, 2017
 Chapter 10 Section 1 Intermolecular Forces the forces between molecules or between ions and molecules in the liquid or solid state Stronger Intermolecular forces cause higher melting points and boiling
Chapter 10 Section 1 Intermolecular Forces the forces between molecules or between ions and molecules in the liquid or solid state Stronger Intermolecular forces cause higher melting points and boiling
Chem 112 Dr. Kevin Moore
 Chem 112 Dr. Kevin Moore Gas Liquid Solid Polar Covalent Bond Partial Separation of Charge Electronegativity: H 2.1 Cl 3.0 H Cl δ + δ - Dipole Moment measure of the net polarity in a molecule Q Q magnitude
Chem 112 Dr. Kevin Moore Gas Liquid Solid Polar Covalent Bond Partial Separation of Charge Electronegativity: H 2.1 Cl 3.0 H Cl δ + δ - Dipole Moment measure of the net polarity in a molecule Q Q magnitude
1.8 Thermodynamics. N Goalby chemrevise.org. Definitions of enthalpy changes
 1.8 Thermodynamics Definitions of enthalpy changes Enthalpy change of formation The standard enthalpy change of formation of a compound is the energy transferred when 1 mole of the compound is formed from
1.8 Thermodynamics Definitions of enthalpy changes Enthalpy change of formation The standard enthalpy change of formation of a compound is the energy transferred when 1 mole of the compound is formed from
Chapter 5 Thermochemistry
 Chapter 5 Thermochemistry Learning Outcomes: Interconvert energy units Distinguish between the system and the surroundings in thermodynamics Calculate internal energy from heat and work and state sign
Chapter 5 Thermochemistry Learning Outcomes: Interconvert energy units Distinguish between the system and the surroundings in thermodynamics Calculate internal energy from heat and work and state sign
CHEMISTRY 110 EXAM 3 NOVEMER 12, 2012 FORM A
 CHEMISTRY 110 EXAM 3 NOVEMER 12, 2012 FORM A 1. Consider a balloon filled with 5 L of an ideal gas at 20 C. If the temperature of the balloon is increased by 70 C and the external pressure acting on the
CHEMISTRY 110 EXAM 3 NOVEMER 12, 2012 FORM A 1. Consider a balloon filled with 5 L of an ideal gas at 20 C. If the temperature of the balloon is increased by 70 C and the external pressure acting on the
Upon successful completion of this unit, the students should be able to:
 Unit 9. Liquids and Solids - ANSWERS Upon successful completion of this unit, the students should be able to: 9.1 List the various intermolecular attractions in liquids and solids (dipole-dipole, London
Unit 9. Liquids and Solids - ANSWERS Upon successful completion of this unit, the students should be able to: 9.1 List the various intermolecular attractions in liquids and solids (dipole-dipole, London
Honors text: Ch 10 & 12 Unit 06 Notes: Balancing Chemical Equations
 Notes: Balancing Chemical Equations Effects of chemical reactions: Chemical reactions rearrange atoms in the reactants to form new products. The identities and properties of the products are completely
Notes: Balancing Chemical Equations Effects of chemical reactions: Chemical reactions rearrange atoms in the reactants to form new products. The identities and properties of the products are completely
Thermochemistry Chapter 4
 Thermochemistry Chapter 4 Thermochemistry is the study of energy changes that occur during chemical reactions Focus is on heat and matter transfer between the system and the surroundings Energy The ability
Thermochemistry Chapter 4 Thermochemistry is the study of energy changes that occur during chemical reactions Focus is on heat and matter transfer between the system and the surroundings Energy The ability
Property of liquid and Phase Diagram for EN 2017
 Property of Liquid and Phase Diagram by Assist.Prof.Dr.Choosak Poonsawat choosak@kku.ac.th FB: เคม อ ช ศ กด You should know - Intermolecular forces - Properties of Liquids - Phase Diagram Properties of
Property of Liquid and Phase Diagram by Assist.Prof.Dr.Choosak Poonsawat choosak@kku.ac.th FB: เคม อ ช ศ กด You should know - Intermolecular forces - Properties of Liquids - Phase Diagram Properties of
Chapter 19 Chemical Thermodynamics
 Chapter 19. Chemical Thermodynamics Sample Exercise 19.2 (p. 819) Elemental mercury is a silver liquid at room temperature. Its normal freezing point is -38.9 o C, and its molar enthalpy of fusion is H
Chapter 19. Chemical Thermodynamics Sample Exercise 19.2 (p. 819) Elemental mercury is a silver liquid at room temperature. Its normal freezing point is -38.9 o C, and its molar enthalpy of fusion is H
Useful Information to be provided on the exam: 1 atm = 760 mm Hg = 760 torr = lb/in 2 = 101,325 Pa = kpa. q = m C T. w = -P V.
 Chem 101A Study Questions, Chapters 5 & 6 Name: Review Tues 10/25/16 Due 10/27/16 (Exam 3 date) This is a homework assignment. Please show your work for full credit. If you do work on separate paper, attach
Chem 101A Study Questions, Chapters 5 & 6 Name: Review Tues 10/25/16 Due 10/27/16 (Exam 3 date) This is a homework assignment. Please show your work for full credit. If you do work on separate paper, attach
Thermochemistry: Energy Flow and Chemical Reactions
 Thermochemistry: Energy Flow and Chemical Reactions Outline thermodynamics internal energy definition, first law enthalpy definition, energy diagrams, calorimetry, theoretical calculation (heats of formation
Thermochemistry: Energy Flow and Chemical Reactions Outline thermodynamics internal energy definition, first law enthalpy definition, energy diagrams, calorimetry, theoretical calculation (heats of formation
δq T = nr ln(v B/V A )
 hysical Chemistry 007 Homework assignment, solutions roblem 1: An ideal gas undergoes the following reversible, cyclic rocess It first exands isothermally from state A to state B It is then comressed adiabatically
hysical Chemistry 007 Homework assignment, solutions roblem 1: An ideal gas undergoes the following reversible, cyclic rocess It first exands isothermally from state A to state B It is then comressed adiabatically
Chapter 16. Thermodynamics. Thermochemistry Review. Calculating H o rxn. Predicting sign for H o rxn. Creative Commons License
 Chapter 16 Thermodynamics GCC CHM152 Creative Commons License Images and tables in this file have been used from the following sources: OpenStax: Creative Commons Attribution License 4.0. ChemWiki (CC
Chapter 16 Thermodynamics GCC CHM152 Creative Commons License Images and tables in this file have been used from the following sources: OpenStax: Creative Commons Attribution License 4.0. ChemWiki (CC
Chem Midterm 3 April 23, 2009
 Chem. 101 - Midterm 3 April 3, 009 Name All work must be shown on the exam for partial credit. Points will be taken off for incorrect or no units and for the incorrect number of significant figures. Only
Chem. 101 - Midterm 3 April 3, 009 Name All work must be shown on the exam for partial credit. Points will be taken off for incorrect or no units and for the incorrect number of significant figures. Only
7/19/2011. Models of Solution. State of Equilibrium. State of Equilibrium Chemical Reaction
 Models of Solution Chemistry- I State of Equilibrium A covered cup of coffee will not be colder than or warmer than the room temperature Heat is defined as a form of energy that flows from a high temperature
Models of Solution Chemistry- I State of Equilibrium A covered cup of coffee will not be colder than or warmer than the room temperature Heat is defined as a form of energy that flows from a high temperature
Exam 2. CHEM Spring Name: Class: Date:
 CHEM-112-01 Spring 2012 Name: Class: Date: 1. Record your name and ID number on the scantron form. 2. Record the test ID letter in the top right box of the scantron form. 3. Record all of your answers
CHEM-112-01 Spring 2012 Name: Class: Date: 1. Record your name and ID number on the scantron form. 2. Record the test ID letter in the top right box of the scantron form. 3. Record all of your answers
Energy, Heat and Chemical Change
 Energy, Heat and Chemical Change Chemistry 35 Fall 2000 Thermochemistry A part of Thermodynamics dealing with energy changes associated with physical and chemical reactions Why do we care? -will a reaction
Energy, Heat and Chemical Change Chemistry 35 Fall 2000 Thermochemistry A part of Thermodynamics dealing with energy changes associated with physical and chemical reactions Why do we care? -will a reaction
Chem 11 UNIT 3: STOICHIOMETRY Name:
 Chem 11 UNIT 3: STOICHIOMETRY Name: Ms. Pirvu Period: Writing & Balancing Equations Chemical reactions can be described by chemical equations. Recall Law of Conservation of Mass mass cannot be nor. This
Chem 11 UNIT 3: STOICHIOMETRY Name: Ms. Pirvu Period: Writing & Balancing Equations Chemical reactions can be described by chemical equations. Recall Law of Conservation of Mass mass cannot be nor. This
Chapter 12 INTERMOLECULAR FORCES. Covalent Radius and van der Waals Radius. Intraand. Intermolecular Forces. ½ the distance of non-bonded
 Chapter 2 INTERMOLECULAR FORCES Intraand Intermolecular Forces Covalent Radius and van der Waals Radius ½ the distance of bonded ½ the distance of non-bonded Dipole Dipole Interactions Covalent and van
Chapter 2 INTERMOLECULAR FORCES Intraand Intermolecular Forces Covalent Radius and van der Waals Radius ½ the distance of bonded ½ the distance of non-bonded Dipole Dipole Interactions Covalent and van
Calculators are permitted. Computers, PDAs, and other electronic devices with a keyboard are not. Cell phones may not be used as calculators.
 Chem. 30 Sring 008 Exam 3 VERSION A Professors Williams/Whetten his test is closed note/book One 8.5 x handwritten crib sheet (one sided) is ermitted. Please turn off your cell hone. Use a # encil. Calculators
Chem. 30 Sring 008 Exam 3 VERSION A Professors Williams/Whetten his test is closed note/book One 8.5 x handwritten crib sheet (one sided) is ermitted. Please turn off your cell hone. Use a # encil. Calculators
MCGILL UNIVERSITY FACULTY OF SCIENCE MIDTERM EXAMINATION CHEM 120 MONDAY MARCH 16, :30PM 8:30PM VERSION NUMBER: 1
 MCGILL UNIVERSITY FACULTY OF SCIENCE MIDTERM EXAMINATION CHEM 120 MONDAY MARCH 16, 2009 6:30PM 8:30PM VERSION NUMBER: 1 Instructions: BEFORE YOU BEGIN: Enter your student number and name on the computer
MCGILL UNIVERSITY FACULTY OF SCIENCE MIDTERM EXAMINATION CHEM 120 MONDAY MARCH 16, 2009 6:30PM 8:30PM VERSION NUMBER: 1 Instructions: BEFORE YOU BEGIN: Enter your student number and name on the computer
Chapter 11 Review Packet
 Chapter 11 Review Packet Name Multiple Choice Portion: 1. Which of the following terms is not a quantitative description of a solution? a. molarity b. molality c. mole fraction d. supersaturation 2. Which
Chapter 11 Review Packet Name Multiple Choice Portion: 1. Which of the following terms is not a quantitative description of a solution? a. molarity b. molality c. mole fraction d. supersaturation 2. Which
Unit Five: Intermolecular Forces MC Question Practice April 14, 2017
 Unit Five: Intermolecular Forces Name MC Question Practice April 14, 2017 1. Which of the following should have the highest surface tension at a given temperature? 2. The triple point of compound X occurs
Unit Five: Intermolecular Forces Name MC Question Practice April 14, 2017 1. Which of the following should have the highest surface tension at a given temperature? 2. The triple point of compound X occurs
Contents. Content Guidance. Questions & Answers. Getting the most from this book... 4 About this book... 5
 Contents Getting the most from this book... 4 About this book.... 5 Content Guidance Atomic structure......................................... 6 Amount of substance....................................
Contents Getting the most from this book... 4 About this book.... 5 Content Guidance Atomic structure......................................... 6 Amount of substance....................................
12A Entropy. Entropy change ( S) N Goalby chemrevise.org 1. System and Surroundings
 12A Entropy Entropy change ( S) A SPONTANEOUS PROCESS (e.g. diffusion) will proceed on its own without any external influence. A problem with H A reaction that is exothermic will result in products that
12A Entropy Entropy change ( S) A SPONTANEOUS PROCESS (e.g. diffusion) will proceed on its own without any external influence. A problem with H A reaction that is exothermic will result in products that
Georgia Institute of Technology. CHEM 1310: Exam II. October 21, 2009
 Georgia Institute of Technology CHEM 1310: Exam II October 21, 2009 Select the best answer for each of the following problems. Each problem is worth 5 points with no partial credit. 1. A solution is prepared
Georgia Institute of Technology CHEM 1310: Exam II October 21, 2009 Select the best answer for each of the following problems. Each problem is worth 5 points with no partial credit. 1. A solution is prepared
SUPeR Chemistry CH 222 Practice Exam
 SUPeR Chemistry CH 222 Practice Exam This exam has been designed to help you practice working multiple choice problems over the material that will be covered on the first CH 222 midterm. The actual exams
SUPeR Chemistry CH 222 Practice Exam This exam has been designed to help you practice working multiple choice problems over the material that will be covered on the first CH 222 midterm. The actual exams
Thermodynamics Spontaneity. 150/151 Thermochemistry Review. Spontaneity. Ch. 16: Thermodynamics 12/14/2017
 Ch. 16: Thermodynamics Geysers are a dramatic display of thermodynamic principles in nature. As water inside the earth heats up, it rises to the surface through small channels. Pressure builds up until
Ch. 16: Thermodynamics Geysers are a dramatic display of thermodynamic principles in nature. As water inside the earth heats up, it rises to the surface through small channels. Pressure builds up until
Chapter 11 Intermolecular Forces, Liquids, and Solids
 Chapter 11 Intermolecular Forces, Liquids, and Solids Dissolution of an ionic compound States of Matter The fundamental difference between states of matter is the distance between particles. States of
Chapter 11 Intermolecular Forces, Liquids, and Solids Dissolution of an ionic compound States of Matter The fundamental difference between states of matter is the distance between particles. States of
 Thermodynamics- 1) Hess's law states that 1) The standard enthalpy of an overall reaction is the sum of the enthalpy changes in individual reaction. ) Enthalpy of formation of compound is same as the enthalpy
Thermodynamics- 1) Hess's law states that 1) The standard enthalpy of an overall reaction is the sum of the enthalpy changes in individual reaction. ) Enthalpy of formation of compound is same as the enthalpy
STATES OF MATTER UNIT 5
 32 32 CHEMISTRY UNIT 5 After studying this unit you will be able to exlain the existence of different states of matter in terms of balance between intermolecular forces and thermal energy of articles;
32 32 CHEMISTRY UNIT 5 After studying this unit you will be able to exlain the existence of different states of matter in terms of balance between intermolecular forces and thermal energy of articles;
9/2/10 TYPES OF INTERMOLECULAR INTERACTIONS
 Tro Chpt. 11 Liquids, solids and intermolecular forces Solids, liquids and gases - A Molecular Comparison Intermolecular forces Intermolecular forces in action: surface tension, viscosity and capillary
Tro Chpt. 11 Liquids, solids and intermolecular forces Solids, liquids and gases - A Molecular Comparison Intermolecular forces Intermolecular forces in action: surface tension, viscosity and capillary
b. Free energy changes provide a good indication of which reactions are favorable and fast, as well as those that are unfavorable and slow.
 Chem 130 Name Exam 3, Ch 7, 19, 14 November 9, 2018 100 Points Please follow the instructions for each section of the exam. Show your work on all mathematical problems. Provide answers with the correct
Chem 130 Name Exam 3, Ch 7, 19, 14 November 9, 2018 100 Points Please follow the instructions for each section of the exam. Show your work on all mathematical problems. Provide answers with the correct
Chemistry 123: Physical and Organic Chemistry Topic 2: Thermochemistry S H 2 = S H 2 R ln P H2 P NH
 N (g) + 3 H (g) NH 3 (g) S N = S H = S NH 3 = S N R ln P N S H R ln P H S NH 3 R ln P NH3 ΔS rxn = (S Rln P NH 3 NH3 ) (S N Rln P N ) 3 (S H Rln P H ) ΔS rxn = S S NH 3 N 3S H + Rln P P 3 N H ΔS rxn =
N (g) + 3 H (g) NH 3 (g) S N = S H = S NH 3 = S N R ln P N S H R ln P H S NH 3 R ln P NH3 ΔS rxn = (S Rln P NH 3 NH3 ) (S N Rln P N ) 3 (S H Rln P H ) ΔS rxn = S S NH 3 N 3S H + Rln P P 3 N H ΔS rxn =
LECTURE 4 Variation of enthalpy with temperature
 LECTURE 4 Variation of enthalpy with temperature So far, we can only work at 25 C. Like c v we define a constant pressure heat capacity, c p, as the amount of heat energy needed to raise the temperature
LECTURE 4 Variation of enthalpy with temperature So far, we can only work at 25 C. Like c v we define a constant pressure heat capacity, c p, as the amount of heat energy needed to raise the temperature
Chapter 4: Properties of Pure Substances. Pure Substance. Phases of a Pure Substance. Phase-Change Processes of Pure Substances
 Chapter 4: roperties o ure Substances ure Substance A substance that has a ixed chemical composition throughout is called a pure substance such as water, air, and nitrogen A pure substance does not hae
Chapter 4: roperties o ure Substances ure Substance A substance that has a ixed chemical composition throughout is called a pure substance such as water, air, and nitrogen A pure substance does not hae
Thermochemistry is the study of the relationships between chemical reactions and energy changes involving heat.
 CHEM134- F18 Dr. Al- Qaisi Chapter 06: Thermodynamics Thermochemistry is the study of the relationships between chemical reactions and energy changes involving heat. Energy is anything that has the capacity
CHEM134- F18 Dr. Al- Qaisi Chapter 06: Thermodynamics Thermochemistry is the study of the relationships between chemical reactions and energy changes involving heat. Energy is anything that has the capacity
Thermodynamics. Chem 36 Spring The study of energy changes which accompany physical and chemical processes
 Thermodynamics Chem 36 Spring 2002 Thermodynamics The study of energy changes which accompany physical and chemical processes Why do we care? -will a reaction proceed spontaneously? -if so, to what extent?
Thermodynamics Chem 36 Spring 2002 Thermodynamics The study of energy changes which accompany physical and chemical processes Why do we care? -will a reaction proceed spontaneously? -if so, to what extent?
4. What is the number of unpaired electrons in Ni? Chemistry 12 Final Exam Form A May 4, 2001
 Chemistry 12 Final Exam Form A May 4, 2001 In all questions involving gases, assume that the ideal-gas laws hold, unless the question specifically refers to the non-ideal behavior. 1. Which of the following
Chemistry 12 Final Exam Form A May 4, 2001 In all questions involving gases, assume that the ideal-gas laws hold, unless the question specifically refers to the non-ideal behavior. 1. Which of the following
Unit 7 Thermochemistry Chemistry 020, R. R. Martin
 Unit 7 Thermochemistry Chemistry 020, R. R. Martin 1. Thermochemistry Heat is a form of energy - which may take many forms: - Kinetic energy due to motion, ½ mv 2 - Potential energy due to position - Electrical
Unit 7 Thermochemistry Chemistry 020, R. R. Martin 1. Thermochemistry Heat is a form of energy - which may take many forms: - Kinetic energy due to motion, ½ mv 2 - Potential energy due to position - Electrical
6.02 x 1023 CHAPTER 10. Mole. Avogadro s Number. Chemical Quantities The Mole: A Measurement of Matter Matter is measured in one of three ways:
 Chapter 10 Notes CHAPTER 10 10.1 The Mole: A Measurement of Matter Matter is measured in one of three ways: Chemical Quantities Mole SI unit that measures the amount of a substance A mole of a substance
Chapter 10 Notes CHAPTER 10 10.1 The Mole: A Measurement of Matter Matter is measured in one of three ways: Chemical Quantities Mole SI unit that measures the amount of a substance A mole of a substance
Thermodynamics Born- Haber cycles
 Thermodynamics Born- Haber cycles Born Haber cycles Is when Hess s Law is extended towards ionic compounds. We usually calculate Enthalpy of formation for substances using bond energy data. But, we cannot
Thermodynamics Born- Haber cycles Born Haber cycles Is when Hess s Law is extended towards ionic compounds. We usually calculate Enthalpy of formation for substances using bond energy data. But, we cannot
1. Increasing the pressure above a liquid will cause the boiling point of the liquid to:
 JASPERSE CHEM 210 PRACTICE TEST 1 VERSION 2 Ch. 11 Liquids, Solids, and Materials Ch. 10 Gases Ch. 15 The Chemistry of Solutes and Solutions Ch. 13 Chemical Kinetics 1 Constants and/or Formulas Formulas
JASPERSE CHEM 210 PRACTICE TEST 1 VERSION 2 Ch. 11 Liquids, Solids, and Materials Ch. 10 Gases Ch. 15 The Chemistry of Solutes and Solutions Ch. 13 Chemical Kinetics 1 Constants and/or Formulas Formulas
Internal Energy in terms of Properties
 Lecture #3 Internal Energy in terms of roerties Internal energy is a state function. A change in the state of the system causes a change in its roerties. So, we exress the change in internal energy in
Lecture #3 Internal Energy in terms of roerties Internal energy is a state function. A change in the state of the system causes a change in its roerties. So, we exress the change in internal energy in
CHEM 1105 S10 March 11 & 14, 2014
 CHEM 1105 S10 March 11 & 14, 2014 Today s topics: Thermochemistry (Chapter 6) Basic definitions Calorimetry Enthalpy Thermochemical equations Calculating heats of reaction Hess s Law Energy and Heat Some
CHEM 1105 S10 March 11 & 14, 2014 Today s topics: Thermochemistry (Chapter 6) Basic definitions Calorimetry Enthalpy Thermochemical equations Calculating heats of reaction Hess s Law Energy and Heat Some
TERMS AND DEFINITIONS IN THERMOCHEMISTRY
 (v.2.0 16/3/90 8hrs.) TERMS AND DEFINITIONS IN THERMOCHEMISTRY (v.2.1 12/4/90 2hrs.) 1) ENTHALPY CHANGE ( H) This is the same as the heat change involved in a process (provided the initial and final states
(v.2.0 16/3/90 8hrs.) TERMS AND DEFINITIONS IN THERMOCHEMISTRY (v.2.1 12/4/90 2hrs.) 1) ENTHALPY CHANGE ( H) This is the same as the heat change involved in a process (provided the initial and final states
ENTHALPY CHANGE CHAPTER 4
 ENTHALPY CHANGE CHAPTER 4 ENTHALPY Is the total energy of a system. E k = Kinetic energy. Vibrational Rotational Translational E due to motion H = E k + E p E P = Potential energy Attractive force b/w
ENTHALPY CHANGE CHAPTER 4 ENTHALPY Is the total energy of a system. E k = Kinetic energy. Vibrational Rotational Translational E due to motion H = E k + E p E P = Potential energy Attractive force b/w
CHEMISTRY The Molecular Nature of Matter and Change
 CHEMISTRY The Molecular Nature of Matter and Change Third Edition Chapter 12 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Chapter 11 INTERMOLECULAR FORCES
CHEMISTRY The Molecular Nature of Matter and Change Third Edition Chapter 12 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Chapter 11 INTERMOLECULAR FORCES
Name AP CHEM / / Collected AP Exam Essay Answers for Chapter 16
 Name AP CHEM / / Collected AP Exam Essay Answers for Chapter 16 1980 - #7 (a) State the physical significance of entropy. Entropy (S) is a measure of randomness or disorder in a system. (b) From each of
Name AP CHEM / / Collected AP Exam Essay Answers for Chapter 16 1980 - #7 (a) State the physical significance of entropy. Entropy (S) is a measure of randomness or disorder in a system. (b) From each of
Free-energy change ( G) and entropy change ( S)
 Free-energy change ( G) and entropy change ( S) A SPONTANEOUS PROCESS (e.g. diffusion) will proceed on its own without any external influence. A problem with H A reaction that is exothermic will result
Free-energy change ( G) and entropy change ( S) A SPONTANEOUS PROCESS (e.g. diffusion) will proceed on its own without any external influence. A problem with H A reaction that is exothermic will result
Solutions: Formation and Properties
 New Jersey Center for Teaching and Learning Slide 1 / 48 Progressive Science Initiative This material is made freely available at www.njctl.org and is intended for the non-commercial use of students and
New Jersey Center for Teaching and Learning Slide 1 / 48 Progressive Science Initiative This material is made freely available at www.njctl.org and is intended for the non-commercial use of students and
Unit 12. Thermochemistry
 Unit 12 Thermochemistry A reaction is spontaneous if it will occur without a continuous input of energy However, it may require an initial input of energy to get it started (activation energy) For Thermochemistry
Unit 12 Thermochemistry A reaction is spontaneous if it will occur without a continuous input of energy However, it may require an initial input of energy to get it started (activation energy) For Thermochemistry
Melting. Freezing. Triple Point. Sublimation. Deposition. Temperature. 2. How many protons and electrons do the following atoms have?
 CST Review Part 2 1. In the phase diagram, correctly label the x-axis and the triple point write the names of all six phases transitions in the arrows provided. Melting Liquid Freezing Pressure (ATM) Solid
CST Review Part 2 1. In the phase diagram, correctly label the x-axis and the triple point write the names of all six phases transitions in the arrows provided. Melting Liquid Freezing Pressure (ATM) Solid
1. Rank the following elements in order of increasing atomic radius: P, Al, Cl, F, S
 Useful constants and other information: R = 0.0821 LCatm/KCmole R = 8.314 J/KCmole h = 6.626 x 10-34 JCs 1 atm = 760 torr Specific heat of H 2 O(l) = 4.184 J/gC C 1 cal = 4.184 J c = 3 x 10 8 m/s PART
Useful constants and other information: R = 0.0821 LCatm/KCmole R = 8.314 J/KCmole h = 6.626 x 10-34 JCs 1 atm = 760 torr Specific heat of H 2 O(l) = 4.184 J/gC C 1 cal = 4.184 J c = 3 x 10 8 m/s PART
Chemistry B11 Chapter 6 Gases, Liquids, and Solids
 Chapter 6 Gases, Liquids, and Solids States of matter: the physical state of matter depends on a balance between the kinetic energy of particles, which tends to keep them apart, and the attractive forces
Chapter 6 Gases, Liquids, and Solids States of matter: the physical state of matter depends on a balance between the kinetic energy of particles, which tends to keep them apart, and the attractive forces
Let a system undergo changes from a state 1 to state 2.
 ME 410 Day 11 Topics First Law of Thermodynamics Applied to Combustion Constant olume Combustion Constant ressure Combustion Enthalpy of Formation Heating alues of Fuels 1. The First Law of Thermodynamics
ME 410 Day 11 Topics First Law of Thermodynamics Applied to Combustion Constant olume Combustion Constant ressure Combustion Enthalpy of Formation Heating alues of Fuels 1. The First Law of Thermodynamics
Chapter 17.3 Entropy and Spontaneity Objectives Define entropy and examine its statistical nature Predict the sign of entropy changes for phase
 Chapter 17.3 Entropy and Spontaneity Objectives Define entropy and examine its statistical nature Predict the sign of entropy changes for phase changes Apply the second law of thermodynamics to chemical
Chapter 17.3 Entropy and Spontaneity Objectives Define entropy and examine its statistical nature Predict the sign of entropy changes for phase changes Apply the second law of thermodynamics to chemical
AP CHEMISTRY 2009 SCORING GUIDELINES
 2009 SCORING GUIDELINES Question 1 (10 points) Answer the following questions that relate to the chemistry of halogen oxoacids. (a) Use the information in the table below to answer part (a)(i). Acid HOCl
2009 SCORING GUIDELINES Question 1 (10 points) Answer the following questions that relate to the chemistry of halogen oxoacids. (a) Use the information in the table below to answer part (a)(i). Acid HOCl
CHAPTER 11. The Mole. Mole. One mole of = 6.02 x 10 = 6.02 x 10 CaCl = 6.02 x x 10. Representative Particle. molecules, or formula units
 CHAPTER 11 The Mole 11.1 The Mole: Measurement of Matter Matter is measured in one of three ways: (How many?) Mole SI unit that measures the amount of a substance 6.02 x 10 particles of that substance.
CHAPTER 11 The Mole 11.1 The Mole: Measurement of Matter Matter is measured in one of three ways: (How many?) Mole SI unit that measures the amount of a substance 6.02 x 10 particles of that substance.
Name: Class: Date: SHORT ANSWER Answer the following questions in the space provided.
 CHAPTER 10 REVIEW States of Matter SECTION 1 SHORT ANSWER Answer the following questions in the space provided. 1. Identify whether the descriptions below describe an ideal gas or a real gas. a. The gas
CHAPTER 10 REVIEW States of Matter SECTION 1 SHORT ANSWER Answer the following questions in the space provided. 1. Identify whether the descriptions below describe an ideal gas or a real gas. a. The gas
General Chemistry I. Dr. PHAN TẠI HUÂN Faculty of Food Science and Technology Nong Lam University. Module 4: Chemical Thermodynamics
 General Chemistry I Dr. PHAN TẠI HUÂN Faculty of Food Science and Technology Nong Lam University Module 4: Chemical Thermodynamics Zeroth Law of Thermodynamics. First Law of Thermodynamics (state quantities:
General Chemistry I Dr. PHAN TẠI HUÂN Faculty of Food Science and Technology Nong Lam University Module 4: Chemical Thermodynamics Zeroth Law of Thermodynamics. First Law of Thermodynamics (state quantities:
PLEASE DO NOT MARK ON THE EXAM. ALL ANSWERS SHOULD BE INDICATED ON THE ANSWER SHEET. c) SeF 4
 Chem 130 EXAM 4 Fall 99 PLEASE DO NOT MARK ON THE EXAM. ALL ANSWERS SHOULD BE INDICATED ON THE ANSWER SHEET QUESTIONS 1-5 MAY HAVE MORE THAN ONE POSSIBLE ANSWER CIRCLE ALL CORRECT RESPONSES TO EACH QUESTION
Chem 130 EXAM 4 Fall 99 PLEASE DO NOT MARK ON THE EXAM. ALL ANSWERS SHOULD BE INDICATED ON THE ANSWER SHEET QUESTIONS 1-5 MAY HAVE MORE THAN ONE POSSIBLE ANSWER CIRCLE ALL CORRECT RESPONSES TO EACH QUESTION
not to be republished NCERT STATES OF MATTER UNIT 5 INTRODUCTION
 32 CHEMISRY UNI 5 SAES OF MAER After studying this unit you will be able to exlain the existence of different states of matter in terms of balance between intermolecular forces and thermal energy of articles;
32 CHEMISRY UNI 5 SAES OF MAER After studying this unit you will be able to exlain the existence of different states of matter in terms of balance between intermolecular forces and thermal energy of articles;
