Answers to Problems. Chapter 2 2. (J' ~ 5. (a) n ~ Chapter 3. n* > n ~ n* n* < n ~ n* < n ~ (J'* > n ~ er* > n ~
|
|
|
- Matilda Skinner
- 5 years ago
- Views:
Transcription
1 Answers to Problems Chapter 1 3. Gamma rays > X-rays > UV > Visible > IR > Radio waves 6. (a) (i) 4 cm- 1 (ii) cm- 1 (iii) cm- 1 (b) x 1 8 MHz to x 1 8 MHz 7. (a) 7.15 kcal/mole (b) x 1 15 cps to.7495 x 1 15 cps 8. (a) kcals mole-1 or kilo Joule mole-1 (b) 4 cm- 1 to cm- 1 (c) x 1 13 cps (Hz) or xl 7 MHz Chapter 2 2. (J' ~ 5. (a) n ~ er* > n ~ (J'* > n ~ n* > n ~ n* n* < n ~ n* < n ~ er* 8. A, 1,4-Pentadiene; B, cis-1,3-pentadiene and C, trans-1,3-pentadiene 9. (i) 249 nm (ii) 342 nm (iii) 226 nm (iv) 418 nm (v) 28 nm (vi) 239 nm 12. (i) 254 nm (ii) 269 nm (iii) 285 nm 15. a < d < b < c 17. (a) 259nm 19. (d) (b) 315 nm (c) 254 nm 2. A is 1,3-cyclohexadiene and B is 1,4-cyclohexadiene. 21. (b) nm and 257 nm 24. (a) 2-propanol, ethanol, heptene, water and dioxane 26. Structure (a) is correct. 28. p-benzoquinone 29. (i) n-n* (ii) n-n* 3. (i) 229 nm (ii) 323 nm Chapter 3 (iii) 351 nm (iv) 281 nm I. (b) Rotational < Vibrational < Electronic 4. (i) 9 (ii) 21 (iii) 7 (iv) 12 (v) 1 5. (i) Esters (ii) Acid halides
2 Answers to Problems (iii) Amides (iv) Anhydrides 9. (a) p-aminoacetophenone < acetophenone < p-nitroacetophenone (b) cyclohexanone < cyclopentanone < cyclobutanone 12. (A) CH3COCH3 (B) CH2=CH-CH2H 14. (b) > (a) 15. (i) b < a < c (ii) (b) < (c) < (a) 16. CH3CH2CONH2 19. trichloroacetic acid < chloroacetic acid < acetic acid < ethanol 2. (i) 33 cm- 1 (Ya-H hydrogen bonded) (ii) 35 cm- 1 (Yc-H aromatic ring) (iii) 299 cm- 1 (Yc-H methyl group) (iv) 17 cm- 1 (Yc=O hydrogen-bonded ester) (v) 154 and 159 cm- 1 (Yc-a. aromatic ring) 21. C 6H 5CH2H 22. CH3COCH3 24. C 6H 5CHO < CH3COCH3 < CH3CHO < CH 3COCJ 26. (i) Inactive (ii) Active (iii) Inactive (iv) Active (v) Inactive (vi) Inactive 28. Butanone 3. m-cresol 31. Benzaldehyde Chapter A The energy of radiation ( kj) is lesser than the dissociation energy of H 2, hence it cannot dissociate. 9. CS 2 has a center of symmetry, whereas N 2 has no center of symmetry and thus the structures must be S-C-S and N-N-, respectively. 1. The compound has trans and planar structure: , 3,11, 454 and 758 cm (a), (b), (c) and (e) will exhibit both the vibrational and rotational Raman spectra; (d) will not exhibit rotational Raman spectrum. 18. H2 > HD > D2. 2. (a), (b) and (d) will exhibit rotational Raman spectra; (c) will not exhibit rotational Roman spectrum. 21. (i) Active (ii) Inactive (iii) Inactive (iv) Active (v) Active 22. The molecule has a trans, planar geometry: f Y-Z-Y I X
3 318 + ORGAN/C SPECTROSCOPY , 733, 743 and 7839 A 24. H3C-C =:C-CH3 Chapter 5 1. For 14N, 2H, 35Cl and 31 P NMR spectroscopy is possible. 3. (a) 2(5 : 3) (b) 3(3 : 2 : I) (c) 3(3 : 2 : 2) 6. (a) 3; one singlet, one triplet and one quartet (b) 3; one singlet, one triplet and one quintet (c) I; singlet ( d) I; singlet 1. Chemical shift positions are and 8 1.5; lax= 1 Hz. 11. (a) ClCHzCC1zCH3 (b) C 6H5-(CH3h (c) C 6H 5 -CH 2CH (CH3)z 12. (a) 1; 3 (3 : 2 : 1) (b) 3 (5 : 2 : 3); 2 (2 : 3) (c) 4 (3 : 2 : 2 : 3); 2 (3 : 2) 14. C 6H 5C(CH3)zCH 2Cl 18. BrCH 2CH 2CH 2Br 21. (i) (ii) (iii) (CH3)zCHOH 25. (a) (CH3)3COH ; 6.51" 27. C 6 H 5CH 2 CH CICH 2CH 2COOH 29. p-nitrophenol 31. C 6H 5CH 2CH 2H Chapter 6 l. (a) 2, (b) 5, (c) 2, (d) l. 4. (a) Two peaks; one singlet and one doublet (b) Three peaks; one singlet, one doublet and one quartet (c) One peak; one triplet (d) Three peaks; one doublet, one triplet and one quartet 5. (a) (CH 3)zCHC =CH (b) (CH 3CH 2)zCHCH 3 (c) CICH 2CHCICCI 3 8. (CH3)zCHCOCH3 9. (a) Six peaks; two singlets, three doublets, and one quartet; the singlet due to 1. (a) ketonic carbon will have the highest and the quartet the lowest 8 value. (b) Four peaks, one singlet, two triplets and one quartet; the singlet will have the highest and the quartet the lowest 8 value. (c) Four peaks; one singlet, one triplet and two quartets; the singlet will have the highest and the quartet the lowest 8 value. (d) Three peaks; one quartet and two triplets; the triplet due to C-3 will have (c) 14. (a) (b) (c) (d) the highest and the quartet the lowest 8 value. (CH3CH 2)JCH (b) CH3CH 2CHOHCH 3 CH 3C ==CCH 2CH 3 5; one singlet, three triplets and one quartet 9; four singlets, four doublets and one quartet 2; one doublet and one triplet 4; one singlet and three triplets
4 Answers to Problems ~ H, Cl Chapter 7 I. (a), (c) 3. (a) 12, (b) 14, (c) 1, (d) 7 5. (a) 3; I : 1 : 1 (b) 5; 1 : 4: 6: 4: I (c) 3; I : 2: I (d) 6; 1 : 5 : 1: 1: 5 : I 6. (a) 7 (b) 8. A, CH(COOH)z; B, CH 2COOH. 9. (a) 35 (b) 8 1. (a) 7; 1 : 6 : 15 : 2 : 15 : 6 :.1 (b) 4; I : 3 : 3 : 1 (c) 7; 1 : 6: 15 : 2: 15 : 6: I (d) 3; 1 : I : I T 13. (c) 14. C 6 H(; 15. Yes; 2 lines in intensity ratio I : /2 Chapter 8 2. (i) HC =C-Br (ii) HCOOCH 3 t C 2H 5 H, CH 3CH=OH, CH 2=H, C2 H ~. 12. CH n-heptane Chapter 9 CH3\ ~ I. CH-CH 2-C-CH3 CH 3d II 2. H 3C-C-CH 2 -CH 2 -COOH 3. H 3 C - ~ - C ( C H 3 h 4. N-C-CH 2-C-O-CH 2CH 3 II HC;;::O, OH and CH 3
5 32. ORGANIC SPECTROSCOPY CHOHCOOH 6. I CHOHCOOH 7. CH 3CH 2C ==CH 8. CH 2 = CH-CH 2H (allyl alcohol or 2-propen-1-ol) CH3CH 2CH 2C ==CH, the isomer may be CH 3CH 2C ==C-CH 3 (other structures are also possible) r CH 2CH 2CCH3 II (CH3hCH-O-CH (CH3h 13. CH 3 -CH-COOH I Br 14. CH 3CH 2CO-O-COCH 2CH (CH3h CHCONH H3C-O-CH 2-C ==N II 17. CH 3CH2CCH3 II 18. (CH 3)3C-OCH3 19. ~ C H, O H 2. CH3COCH 2CH3 11-@ CH3CHzQ-C C-üCH2CH3
6 Index a, ß-unsaturated carboxylic acids, UV, 34 a,ß-unsaturated esters, UV, 34 y-gauche steric compression, 21 Absorption bands, 12: designation of, 13 formation of, 12 Absorption 1aws, 7 Absorption of energy, NMR, 136 Absorption spectra, 5 Acetylenic protons, 147 Acid anhydrides, IR 78 Acid halides, IR 78 Alcohols, IR 69 Aldehydes, IR 72 Aldehydic protons, 148 Alkanes, IR 67 Alkenes, IR 68 Alkyl substituents, 21 Alkynes, IR 69 Allowed transition, 14 Allylic coupling, 167 Amides, IR, 79 Amines and their salts, IR 8 Aminoacidsand their salts, IR, 81 Analysis of NMR spectra, 168 Anisotropie effect, 146 applications of, 92 Aromatic compounds, 4, 41, 42 non-benzenoid, 42 polynuclear, 4 Aromatic coupling, 167 Aromatic hydrocarbons, IR 69 Aromatic protons, 149 Auxochrome, 16 8-Bands, 13 Base peak, 255 Bathochramie shift, 16 Bending vibrations, 56, 57 Benzene and its derivatives, UV 34 Blue shift, 16 13C chemical shifts, 22, 25, 211 a-, ß- and y-effects, 23 13C NMR spectroscopy, 195 applications of, 214 common modes of recording, 196 samp1e handling, 196 theory, 195 use of shift reagents, 214 Carbony1 carbons, 21 Carboxylate axions, IR 75 Carboxylic acid, IR 75 Chemical ionization (Cl) method, 251 Chemical shift, 142, 143, , 146, 152, 17, 199 Chromophore, 15 Combination bands, 61 Comparison between NMR and ESR., 243 Conjugated systems, UV, 17 Continuous wave NMR spectroscopy, 184 Coupled vibrations, 62 Coupling constant (1), 164, 165, 166 Cycloalkanes, IR 68 Daughter ions, 253 Deformations, 56 Deshielding, 142, 148, 149, 15 Detection of isotopes, 256 Deuteration, 178 Deuterium exchange, 178 Deuterium labelling, 178 Dicarbonyl compounds, UV, 32 Difference bands, 61 Double irradiation in ESR, 239 Double irradiation, 177 Double resonance in ESR, 239, 24 Double resonance, 177 DSS as reference, 143
7 322 +Index E-Bands, 13 ELDOR, 24 Electromagnetic radiations, I, 2 Electromagnetic spectrum, 4, 5 Electron impact (EI) method, 25 Electron paramagnetic resonance (EPR), 224 Electron spin resonance (ESR) spectroscopy, 224 instrumentation, 227 interpretation, 237 multiple! structures, 232 theory, 224 Electronic spectroscopy, 7, 15 Electronic transitions, 9 cr ~ n ~ n ~ n ~ cr*, lo cr*, lo n*, 11 n*, 11 Emission spectra, 5 ENDOR, 239 Epoxides, IR, 71 Equivalent and non-equivalent protons, 153 ESR spectra, interpetation of, 237 Esters, IR, 76 Ethers, IR, 71 Exocyclic double bonds, 21 Fermi resonance, 63 Fine structure, 232 Fingerprint region, 92 First order spectra, 169 Flipping of nucleus, 137 Forbidden transition, 14 Fragment ions, 253 Fragmentalion modes, 262 general, 262 of alcohols, 272 of aldehydes and ketones, 276 of alkanes, 265 of alkenes, 267 of alkynes, 268 of amines, 282 of aromatic hydrocarbons, 269 of carboxylic acids, esters and amides, 278 of ethers, acetals and ketals 275 of halogen compounds 27 of heterocychic compounds, 286 of nitriles 283 of nitro compounds, 284 of phenols, 274 of sulphur compounds, 285 of various classes of compounds, 265 Fragmentalion processes, 262 examples of, 262 factors governing, 262 representation of, 262 Frequency, I Fundamental vibrations, nurober of, 58 g factor, 226, 227, 229 Gated decoupling, 198 Geminal coupling, 165 Halogen compounds, IR, 69 Heteroannular dienes, 21 Heteroaromatic compounds, UV, 42, 43 Homoallylic coupling, 167 Homoannular dienes, 2 Hydrogen bonding, IR, 64 Hydrogen bonding, NMR, 151 Hyperchromic effect, 16 Hyperfine structure, 233 Hypsochromic shift, 16 Imides, IR, 79 Index of hydrogen deficiency, 258 Inductive effect, IR, 66 Instrumentation of IR spectroscopy, 52 PMR spectroscopy, 139 Raman, 119 Interpretation of NMR spectra, 168 lonization processes, 25 IR absorption frequencies, table of, 83 IR inactive vibrations, 57, 6 IR spectra, interpretation of, 94 IR spectroscopy, 52, 55, 92 Isotope peak, 256 K-Bands, 13 Ketones, IR, 72 Lactans, IR, 79 Lactones, IR, 76 Long range coupling, 167 Longitudinal relaxation, 139 Magnetic equivalence, 172
8 Index+ 323 Mass spectroscopy (MS), 25 applications, 26 instrumentation, 253 Mass spectrum, 255 Mesomerie effect, IR, 66 Metastahle ions or peaks, 26 Molar absorptivity, 7 Molecular ion peak, confirmation of, 257 Molecular ion, 252 Multiplicity, 159 Multiply charged ions, 259 Nitriles, IR, 83 Nitrites, IR, 82 Nitro compounds, IR, 82 Nitrogen rule, 258 NMR scale, 144 NMR spectra, 168, 17 ana1ysis of, 168 NMR versus ESR, 243 Non-benzenoid aromatic compounds, UV, 42 Nuclear Overhauser effect (NOE), 181 Number of PMR signals, 153 Off-resonance decoupling, 197 Olefinic protons, 148 Overtone bonds, 6 Parent ion, 252 Peak area and proton counting, 156 Phenols, IR, 69 Phosphorus compounds, IR, 83 Photon, energy for, 2 Polarization of Raman lines, 118 Poly-ynes, UV, 26 Polyenes, UV, 26 Precessional frequency, 13 7 Proton exchange reaction, 17 4 Proton nuclear magnetic resonance (PMR or 1H NMR) spectroscopy, 133 applications of, 181 continuous wave (CW), 184 Fourier transform (FT), 184 instrumentation, 14 sample handling, 14 s1gnals, 153 theory, 133 Proton-noise decoupling, 196 Protons on hetero atoms, 17 4 R-Bands, 13 Raman effect, I 7 theories of, 19 Raman spectra, pure rotational, 115, 116 vibrational, 114 vibration-rotational, 117 Raman spectroscopy, 17, 19, 12 applications, 121 effect, 17,18,19,111,112 instrumentation, 119 origin of, 17 samp1e handling, 121 theories of, 19 Raman tube, 12 Raman versus fluorescence spectra, 124 Raman versus IR spectra, 124, 125 Red shift, 16 Relaxation processes, 139 Resonance effect, IR, 66 Ring residues, 21 Rocking, 57 Rule of mutual exclusion, 118 Sampie handling, ESR, 231 IR, 54 PMR, 141 Raman, 121 UV, 9 Satellite peak, 256 Saturation, NMR, 138 Scissoring, 56 Second order spectra, 17 Shielding, 142, 147, 149, 151 Shift reagents, 179 Solvent effer, UV, 18 Spectroscopy (spectromctry), dcfinition, 1 Spectroscopy, infrared (IR), 52, 92 Spin lattice relaxation, 139 Spin number /, 133 Spin systems, nomenclature of, 17 Spin-spin coupling, 157, 172 Spin-spin coupling, 13 C, 212 Spin-spin decoupling, 177 Spin-spin relaxation, 139 Spin-spin splitting, 157, 158, 173 Stretching vibrations, 56, 58, 6, 61 Sulphur compounds, IR, 83
9 324 +Index Theory of ESR spectroscopy, 224 applications of, 24 Theory (origin) of IR spectroscopy, 55 TMS as reference, 143 Transition probability, 14 Transverse relaxation, I39 Twisting, 57 UV and visible spectroscop.y 7, 8, 9 Van der Waals deshielding,!51 Vicinal coupling, 166 Wagging, 57 Wavelength, I Wavenumber, I Woodward-Fieser rules for a,ß-unsaturated carbonyl compounds, 27 for dienes and trienes, 2 exceptions to, 25 Vibrational frequencies, calculation of, 61 Zero-point energy, 113
ORGANIC - CLUTCH CH ANALYTICAL TECHNIQUES: IR, NMR, MASS SPECT
 !! www.clutchprep.com CONCEPT: PURPOSE OF ANALYTICAL TECHNIQUES Classical Methods (Wet Chemistry): Chemists needed to run dozens of chemical reactions to determine the type of molecules in a compound.
!! www.clutchprep.com CONCEPT: PURPOSE OF ANALYTICAL TECHNIQUES Classical Methods (Wet Chemistry): Chemists needed to run dozens of chemical reactions to determine the type of molecules in a compound.
ORGANIC - CLUTCH CH ANALYTICAL TECHNIQUES: IR, NMR, MASS SPECT
 !! www.clutchprep.com CONCEPT: PURPOSE OF ANALYTICAL TECHNIQUES Classical Methods (Wet Chemistry): Chemists needed to run dozens of chemical reactions to determine the type of molecules in a compound.
!! www.clutchprep.com CONCEPT: PURPOSE OF ANALYTICAL TECHNIQUES Classical Methods (Wet Chemistry): Chemists needed to run dozens of chemical reactions to determine the type of molecules in a compound.
ORGANIC - BRUICE 8E CH MASS SPECT AND INFRARED SPECTROSCOPY
 !! www.clutchprep.com CONCEPT: PURPOSE OF ANALYTICAL TECHNIQUES Classical Methods (Wet Chemistry): Chemists needed to run dozens of chemical reactions to determine the type of molecules in a compound.
!! www.clutchprep.com CONCEPT: PURPOSE OF ANALYTICAL TECHNIQUES Classical Methods (Wet Chemistry): Chemists needed to run dozens of chemical reactions to determine the type of molecules in a compound.
ORGANIC - EGE 5E CH UV AND INFRARED MASS SPECTROMETRY
 !! www.clutchprep.com CONCEPT: IR SPECTROSCOPY- FREQUENCIES There are specific absorption frequencies in the functional group region that we should be familiar with EXAMPLE: What are the major IR absorptions
!! www.clutchprep.com CONCEPT: IR SPECTROSCOPY- FREQUENCIES There are specific absorption frequencies in the functional group region that we should be familiar with EXAMPLE: What are the major IR absorptions
Lecture 11. IR Theory. Next Class: Lecture Problem 4 due Thin-Layer Chromatography
 Lecture 11 IR Theory Next Class: Lecture Problem 4 due Thin-Layer Chromatography This Week In Lab: Ch 6: Procedures 2 & 3 Procedure 4 (outside of lab) Next Week in Lab: Ch 7: PreLab Due Quiz 4 Ch 5 Final
Lecture 11 IR Theory Next Class: Lecture Problem 4 due Thin-Layer Chromatography This Week In Lab: Ch 6: Procedures 2 & 3 Procedure 4 (outside of lab) Next Week in Lab: Ch 7: PreLab Due Quiz 4 Ch 5 Final
Table 8.2 Detailed Table of Characteristic Infrared Absorption Frequencies
 Table 8.2 Detailed Table of Characteristic Infrared Absorption Frequencies The hydrogen stretch region (3600 2500 cm 1 ). Absorption in this region is associated with the stretching vibration of hydrogen
Table 8.2 Detailed Table of Characteristic Infrared Absorption Frequencies The hydrogen stretch region (3600 2500 cm 1 ). Absorption in this region is associated with the stretching vibration of hydrogen
Infrared Spectroscopy
 Infrared Spectroscopy Introduction Spectroscopy is an analytical technique which helps determine structure. It destroys little or no sample. The amount of light absorbed by the sample is measured as wavelength
Infrared Spectroscopy Introduction Spectroscopy is an analytical technique which helps determine structure. It destroys little or no sample. The amount of light absorbed by the sample is measured as wavelength
General Infrared Absorption Ranges of Various Functional Groups
 General Infrared Absorption Ranges of Various Functional Groups Frequency Range Bond Type of Compound cm -1 Intensity C Alkanes 2850-2970 Strong 1340-1470 Strong C Alkenes 3010-3095 Medium 675-995 Strong
General Infrared Absorption Ranges of Various Functional Groups Frequency Range Bond Type of Compound cm -1 Intensity C Alkanes 2850-2970 Strong 1340-1470 Strong C Alkenes 3010-3095 Medium 675-995 Strong
SPECTROSCOPY MEASURES THE INTERACTION BETWEEN LIGHT AND MATTER
 SPECTROSCOPY MEASURES THE INTERACTION BETWEEN LIGHT AND MATTER c = c: speed of light 3.00 x 10 8 m/s (lamda): wavelength (m) (nu): frequency (Hz) Increasing E (J) Increasing (Hz) E = h h - Planck s constant
SPECTROSCOPY MEASURES THE INTERACTION BETWEEN LIGHT AND MATTER c = c: speed of light 3.00 x 10 8 m/s (lamda): wavelength (m) (nu): frequency (Hz) Increasing E (J) Increasing (Hz) E = h h - Planck s constant
STRUCTURE ELUCIDATION BY INTEGRATED SPECTROSCOPIC METHODS
 Miscellaneous Methods UNIT 14 STRUCTURE ELUCIDATION BY INTEGRATED SPECTROSCOPIC METHODS Structure 14.1 Introduction Objectives 14.2 Molecular Formula and Index of Hydrogen Deficiency 14.3 Structural Information
Miscellaneous Methods UNIT 14 STRUCTURE ELUCIDATION BY INTEGRATED SPECTROSCOPIC METHODS Structure 14.1 Introduction Objectives 14.2 Molecular Formula and Index of Hydrogen Deficiency 14.3 Structural Information
ORGANIC - BROWN 8E CH INFRARED SPECTROSCOPY.
 !! www.clutchprep.com CONCEPT: PURPOSE OF ANALYTICAL TECHNIQUES Classical Methods (Wet Chemistry): Chemists needed to run dozens of chemical reactions to determine the type of molecules in a compound.
!! www.clutchprep.com CONCEPT: PURPOSE OF ANALYTICAL TECHNIQUES Classical Methods (Wet Chemistry): Chemists needed to run dozens of chemical reactions to determine the type of molecules in a compound.
Objective 4. Determine (characterize) the structure of a compound using IR, NMR, MS.
 Objective 4. Determine (characterize) the structure of a compound using IR, NMR, MS. Skills: Draw structure IR: match bond type to IR peak NMR: ID number of non-equivalent H s, relate peak splitting to
Objective 4. Determine (characterize) the structure of a compound using IR, NMR, MS. Skills: Draw structure IR: match bond type to IR peak NMR: ID number of non-equivalent H s, relate peak splitting to
12. Structure Determination: Mass Spectrometry and Infrared Spectroscopy
 12. Structure Determination: Mass Spectrometry and Infrared Spectroscopy Determining the Structure of an Organic Compound The analysis of the outcome of a reaction requires that we know the full structure
12. Structure Determination: Mass Spectrometry and Infrared Spectroscopy Determining the Structure of an Organic Compound The analysis of the outcome of a reaction requires that we know the full structure
Introduction. The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants
 Introduction The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants Spectroscopy and the Electromagnetic Spectrum Unlike mass spectrometry,
Introduction The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants Spectroscopy and the Electromagnetic Spectrum Unlike mass spectrometry,
Infrared Spectroscopy An Instrumental Method for Detecting Functional Groups
 Infrared Spectroscopy An Instrumental Method for Detecting Functional Groups 1 The Electromagnetic Spectrum Infrared Spectroscopy I. Physics Review Frequency, υ (nu), is the number of wave cycles that
Infrared Spectroscopy An Instrumental Method for Detecting Functional Groups 1 The Electromagnetic Spectrum Infrared Spectroscopy I. Physics Review Frequency, υ (nu), is the number of wave cycles that
7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text , , 12.10)
 2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
E35 SPECTROSCOPIC TECHNIQUES IN ORGANIC CHEMISTRY
 E35 SPECTRSCPIC TECNIQUES IN RGANIC CEMISTRY Introductory Comments. These notes are designed to introduce you to the basic spectroscopic techniques which are used for the determination of the structure
E35 SPECTRSCPIC TECNIQUES IN RGANIC CEMISTRY Introductory Comments. These notes are designed to introduce you to the basic spectroscopic techniques which are used for the determination of the structure
ORGANIC SPECTROSCOPY NOTES
 - 1 - ORGANIC SPECTROSCOPY NOTES Basics of Spectroscopy UV/vis, IR and NMR are all types of Absorption Spectroscopy, where EM radiation corresponding to exactly the energy of specific excitations in molecules
- 1 - ORGANIC SPECTROSCOPY NOTES Basics of Spectroscopy UV/vis, IR and NMR are all types of Absorption Spectroscopy, where EM radiation corresponding to exactly the energy of specific excitations in molecules
CH 3. mirror plane. CH c d
 CAPTER 20 Practice Exercises 20.1 The index of hydrogen deficiency is two. The structural possibilities include two double bonds, a double do 20.3 (a) As this is an alkane, it contains only C and and has
CAPTER 20 Practice Exercises 20.1 The index of hydrogen deficiency is two. The structural possibilities include two double bonds, a double do 20.3 (a) As this is an alkane, it contains only C and and has
PAPER No.12 :Organic Spectroscopy MODULE No.29: Combined problem on UV, IR, 1 H NMR, 13 C NMR and Mass - Part I
 Subject Chemistry Paper No and Title Module No and Title Module Tag 12: rganic Spectroscopy 29: Combined problem on UV, IR, 1 H NMR, 13 C NMR and Mass - Part I CHE_P12_M29 TABLE F CNTENTS 1. Learning utcomes
Subject Chemistry Paper No and Title Module No and Title Module Tag 12: rganic Spectroscopy 29: Combined problem on UV, IR, 1 H NMR, 13 C NMR and Mass - Part I CHE_P12_M29 TABLE F CNTENTS 1. Learning utcomes
NUCLEAR MAGNETIC RESONANCE AND INTRODUCTION TO MASS SPECTROMETRY
 NUCLEAR MAGNETIC RESONANCE AND INTRODUCTION TO MASS SPECTROMETRY A STUDENT SHOULD BE ABLE TO: 1. Identify and explain the processes involved in proton ( 1 H) and carbon-13 ( 13 C) nuclear magnetic resonance
NUCLEAR MAGNETIC RESONANCE AND INTRODUCTION TO MASS SPECTROMETRY A STUDENT SHOULD BE ABLE TO: 1. Identify and explain the processes involved in proton ( 1 H) and carbon-13 ( 13 C) nuclear magnetic resonance
NMR = Nuclear Magnetic Resonance
 NMR = Nuclear Magnetic Resonance NMR spectroscopy is the most powerful technique available to organic chemists for determining molecular structures. Looks at nuclei with odd mass numbers or odd number
NMR = Nuclear Magnetic Resonance NMR spectroscopy is the most powerful technique available to organic chemists for determining molecular structures. Looks at nuclei with odd mass numbers or odd number
Infra-red Spectroscopy
 Molecular vibrations are associated with the absorption of energy (infrared activity) by the molecule as sets of atoms (molecular moieties) vibrate about the mean center of their chemical bonds. Infra-red
Molecular vibrations are associated with the absorption of energy (infrared activity) by the molecule as sets of atoms (molecular moieties) vibrate about the mean center of their chemical bonds. Infra-red
William H. Brown & Christopher S. Foote
 Requests for permission to make copies of any part of the work should be mailed to:permissions Department, Harcourt Brace & Company, 6277 Sea Harbor Drive, Orlando, Florida 32887-6777 William H. Brown
Requests for permission to make copies of any part of the work should be mailed to:permissions Department, Harcourt Brace & Company, 6277 Sea Harbor Drive, Orlando, Florida 32887-6777 William H. Brown
Chapter 12 Mass Spectrometry and Infrared Spectroscopy
 Organic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 12 Mass Spectrometry and Infrared Spectroscopy Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice
Organic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 12 Mass Spectrometry and Infrared Spectroscopy Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice
13.24: Mass Spectrometry: molecular weight of the sample
 hapter 13: Spectroscopy Methods of structure determination Nuclear Magnetic Resonances (NMR) Spectroscopy (Sections 13.3-13.19) Infrared (IR) Spectroscopy (Sections 13.20-13.22) Ultraviolet-visible (UV-Vis)
hapter 13: Spectroscopy Methods of structure determination Nuclear Magnetic Resonances (NMR) Spectroscopy (Sections 13.3-13.19) Infrared (IR) Spectroscopy (Sections 13.20-13.22) Ultraviolet-visible (UV-Vis)
Using NMR and IR Spectroscopy to Determine Structures Dr. Carl Hoeger, UCSD
 Using NMR and IR Spectroscopy to Determine Structures Dr. Carl Hoeger, UCSD The following guidelines should be helpful in assigning a structure from NMR (both PMR and CMR) and IR data. At the end of this
Using NMR and IR Spectroscopy to Determine Structures Dr. Carl Hoeger, UCSD The following guidelines should be helpful in assigning a structure from NMR (both PMR and CMR) and IR data. At the end of this
UV Spectroscopy: Empirical Approach to Molecular Structures. Dr. Mishu Singh Department of Chemistry M. P.Govt P. G.
 UV Spectroscopy: Empirical Approach to Molecular Structures Dr. Mishu Singh Department of Chemistry M. P.Govt P. G.College, Hardoi WHAT IS SPECTROSCOPY? Atoms and molecules interact with electromagnetic
UV Spectroscopy: Empirical Approach to Molecular Structures Dr. Mishu Singh Department of Chemistry M. P.Govt P. G.College, Hardoi WHAT IS SPECTROSCOPY? Atoms and molecules interact with electromagnetic
Learning Guide for Chapter 3 - Infrared Spectroscopy
 Learning Guide for hapter 3 - Infrared Spectroscopy I. Introduction to spectroscopy - p 1 II. Molecular vibrations - p 3 III. Identifying functional groups - p 6 IV. Interpreting an IR spectrum - p 12
Learning Guide for hapter 3 - Infrared Spectroscopy I. Introduction to spectroscopy - p 1 II. Molecular vibrations - p 3 III. Identifying functional groups - p 6 IV. Interpreting an IR spectrum - p 12
OAT Organic Chemistry - Problem Drill 19: NMR Spectroscopy and Mass Spectrometry
 OAT Organic Chemistry - Problem Drill 19: NMR Spectroscopy and Mass Spectrometry Question No. 1 of 10 Question 1. Which statement concerning NMR spectroscopy is incorrect? Question #01 (A) Only nuclei
OAT Organic Chemistry - Problem Drill 19: NMR Spectroscopy and Mass Spectrometry Question No. 1 of 10 Question 1. Which statement concerning NMR spectroscopy is incorrect? Question #01 (A) Only nuclei
Ultraviolet Spectroscopy. CH- 521 Course on Interpreta2ve Molecular Spectroscopy; Course Instructor: Krishna P. Kaliappan
 Ultraviolet Spectroscopy CH- 521 Course on Interpreta2ve Molecular Spectroscopy; Course Instructor: Krishna P. Kaliappan Ultraviolet Spectroscopy UV light can be absorbed by molecules to excite higher
Ultraviolet Spectroscopy CH- 521 Course on Interpreta2ve Molecular Spectroscopy; Course Instructor: Krishna P. Kaliappan Ultraviolet Spectroscopy UV light can be absorbed by molecules to excite higher
Radiant energy is proportional to its frequency (cycles/s = Hz) as a wave (Amplitude is its height) Different types are classified by frequency or
 CHEM 241 UNIT 5: PART B INFRA-RED RED SPECTROSCOPY 1 Spectroscopy of the Electromagnetic Spectrum Radiant energy is proportional to its frequency (cycles/s = Hz) as a wave (Amplitude is its height) Different
CHEM 241 UNIT 5: PART B INFRA-RED RED SPECTROSCOPY 1 Spectroscopy of the Electromagnetic Spectrum Radiant energy is proportional to its frequency (cycles/s = Hz) as a wave (Amplitude is its height) Different
CHEM Chapter 13. Nuclear Magnetic Spectroscopy (Homework) W
 CHEM 2423. Chapter 13. Nuclear Magnetic Spectroscopy (Homework) W Short Answer 1. For a nucleus to exhibit the nuclear magnetic resonance phenomenon, it must be magnetic. Magnetic nuclei include: a. all
CHEM 2423. Chapter 13. Nuclear Magnetic Spectroscopy (Homework) W Short Answer 1. For a nucleus to exhibit the nuclear magnetic resonance phenomenon, it must be magnetic. Magnetic nuclei include: a. all
Infrared Spectroscopy
 Infrared Spectroscopy IR Spectroscopy Used to identify organic compounds IR spectroscopy provides a 100% identification if the spectrum is matched. If not, IR at least provides information about the types
Infrared Spectroscopy IR Spectroscopy Used to identify organic compounds IR spectroscopy provides a 100% identification if the spectrum is matched. If not, IR at least provides information about the types
ORGAN IC STRUCTURAL SPECTROSCOPY
 ORGAN IC STRUCTURAL SPECTROSCOPY JOSEPH B. LAMBERT Northwestern University HERBERT F. SHURVELL Queen's University DAVID A. LIGHTNER University of Nevada at Reno R. GRAHAM COOKS Purdue University Prentice
ORGAN IC STRUCTURAL SPECTROSCOPY JOSEPH B. LAMBERT Northwestern University HERBERT F. SHURVELL Queen's University DAVID A. LIGHTNER University of Nevada at Reno R. GRAHAM COOKS Purdue University Prentice
Paper 12: Organic Spectroscopy
 Subject hemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy 34: ombined problem on UV, IR, 1 H NMR, 13 NMR and Mass- Part 6 HE_P12_M34 TABLE OF ONTENTS 1. Learning
Subject hemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy 34: ombined problem on UV, IR, 1 H NMR, 13 NMR and Mass- Part 6 HE_P12_M34 TABLE OF ONTENTS 1. Learning
Experiment 11: NUCLEAR MAGNETIC RESONANCE SPECTROSCOPY
 Experiment 11: NUCLEAR MAGNETIC RESONANCE SPECTROSCOPY Purpose: This is an exercise to introduce the use of nuclear magnetic resonance spectroscopy, in conjunction with infrared spectroscopy, to determine
Experiment 11: NUCLEAR MAGNETIC RESONANCE SPECTROSCOPY Purpose: This is an exercise to introduce the use of nuclear magnetic resonance spectroscopy, in conjunction with infrared spectroscopy, to determine
CH 320/328 N Summer II 2018
 CH 320/328 N Summer II 2018 HW 1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. (5 pts each) 1. Which
CH 320/328 N Summer II 2018 HW 1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. (5 pts each) 1. Which
ULTRAVIOLET SPECTROSCOPY or ELECTRONIC SPECTROSCOPY
 ULTRAVILET SPECTRSCPY or ELECTRNIC SPECTRSCPY S. SANKARARAMAN Department of Chemistry Indian Institute of Technology Madras Chennai 600036, INDIA Sanka@iitm.ac.in Absorption of electromagnetic radiation
ULTRAVILET SPECTRSCPY or ELECTRNIC SPECTRSCPY S. SANKARARAMAN Department of Chemistry Indian Institute of Technology Madras Chennai 600036, INDIA Sanka@iitm.ac.in Absorption of electromagnetic radiation
1.1. IR is part of electromagnetic spectrum between visible and microwave
 CH2SWK 44/6416 IR Spectroscopy 2013Feb5 1 1. Theory and properties 1.1. IR is part of electromagnetic spectrum between visible and microwave 1.2. 4000 to 400 cm -1 (wave numbers) most interesting to organic
CH2SWK 44/6416 IR Spectroscopy 2013Feb5 1 1. Theory and properties 1.1. IR is part of electromagnetic spectrum between visible and microwave 1.2. 4000 to 400 cm -1 (wave numbers) most interesting to organic
Structure Determination
 There are more than 5 million organic compounds, the great majority of which are colourless liquids or white solids. Identifying or at least characterising determining some of its properties and features
There are more than 5 million organic compounds, the great majority of which are colourless liquids or white solids. Identifying or at least characterising determining some of its properties and features
Chapter 25: The Chemistry of Life: Organic and Biological Chemistry
 Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Chapter 13. R.F.----µ-wave----I.R. (Heat)------Visible------U.V X-Ray------γ-Ray SPECTROSCOPY. Definition: Types to Be Covered:
 hamras Glendale ommunity ollege rganic hemistry 105 Exam 4 Materials hapter 13 SPETRSPY Definition: Types to Be overed: A) Infrared Spectroscopy (IR) B) Nuclear Magnetic Resonance Spectroscopy (NMR) )
hamras Glendale ommunity ollege rganic hemistry 105 Exam 4 Materials hapter 13 SPETRSPY Definition: Types to Be overed: A) Infrared Spectroscopy (IR) B) Nuclear Magnetic Resonance Spectroscopy (NMR) )
More information can be found in Chapter 12 in your textbook for CHEM 3750/ 3770 and on pages in your laboratory manual.
 CHEM 3780 rganic Chemistry II Infrared Spectroscopy and Mass Spectrometry Review More information can be found in Chapter 12 in your textbook for CHEM 3750/ 3770 and on pages 13-28 in your laboratory manual.
CHEM 3780 rganic Chemistry II Infrared Spectroscopy and Mass Spectrometry Review More information can be found in Chapter 12 in your textbook for CHEM 3750/ 3770 and on pages 13-28 in your laboratory manual.
Chapter 9. Nuclear Magnetic Resonance. Ch. 9-1
 Chapter 9 Nuclear Magnetic Resonance Ch. 9-1 1. Introduction Classic methods for organic structure determination Boiling point Refractive index Solubility tests Functional group tests Derivative preparation
Chapter 9 Nuclear Magnetic Resonance Ch. 9-1 1. Introduction Classic methods for organic structure determination Boiling point Refractive index Solubility tests Functional group tests Derivative preparation
C h a p t e r F o u r t e e n: Structure Determination: Mass Spectrometry and Infrared Spectroscopy
 C h a p t e r F o u r t e e n: Structure Determination: Mass Spectrometry and Infrared Spectroscopy Cl OH Cl An electron ionization mass spectrum of 2,5-dichlorophenol CHM 323: Summary of Important Concepts
C h a p t e r F o u r t e e n: Structure Determination: Mass Spectrometry and Infrared Spectroscopy Cl OH Cl An electron ionization mass spectrum of 2,5-dichlorophenol CHM 323: Summary of Important Concepts
FAMILIES of ORGANIC COMPOUNDS
 1 SCH4U October 2016 Organic Chemistry Chemistry of compounds that contain carbon (except: CO, CO 2, HCN, CO 3 - ) Carbon is covalently bonded to another carbon, hydrogen and possibly to oxygen, a halogen
1 SCH4U October 2016 Organic Chemistry Chemistry of compounds that contain carbon (except: CO, CO 2, HCN, CO 3 - ) Carbon is covalently bonded to another carbon, hydrogen and possibly to oxygen, a halogen
Learning Guide for Chapter 3 - Infrared Spectroscopy
 Learning Guide for hapter 3 - Infrared Spectroscopy I. Introduction to spectroscopy - p 1 II. Molecular vibrations - p 3 III. Identifying functional groups - p 6 IV. Interpreting an IR spectrum - p 12
Learning Guide for hapter 3 - Infrared Spectroscopy I. Introduction to spectroscopy - p 1 II. Molecular vibrations - p 3 III. Identifying functional groups - p 6 IV. Interpreting an IR spectrum - p 12
(DCHE 21) M.Sc. (Final) DEGREE EXAMINATION, DECEMBER Second Year. Chemistry. Paper V ANALYTICAL CHEMISTRY. PART A (4 10 = 40 marks)
 (DCHE 21) M.Sc. (Final) DEGREE EXAMINATION, DECEMBER 2010. Second Year Chemistry Paper V ANALYTICAL CHEMISTRY Time : Three hours Maximum : 100 marks PART A (4 10 = 40 marks) Answer any FOUR questions.
(DCHE 21) M.Sc. (Final) DEGREE EXAMINATION, DECEMBER 2010. Second Year Chemistry Paper V ANALYTICAL CHEMISTRY Time : Three hours Maximum : 100 marks PART A (4 10 = 40 marks) Answer any FOUR questions.
Magnetic Nuclei other than 1 H
 Magnetic Nuclei other than 1 H 2 H (Deuterium): I = 1 H,D-Exchange might be used to simplify 1 H-NMR spectra since H-D couplings are generally small; - - - -O- - - -D 2 -O- triplet of triplets slightly
Magnetic Nuclei other than 1 H 2 H (Deuterium): I = 1 H,D-Exchange might be used to simplify 1 H-NMR spectra since H-D couplings are generally small; - - - -O- - - -D 2 -O- triplet of triplets slightly
Chapter 14 Spectroscopy
 hapter 14 Spectroscopy There are four major analytical techniques used for identifying the structure of organic molecules 1. Nuclear Magnetic Resonance or NMR is the single most important technique for
hapter 14 Spectroscopy There are four major analytical techniques used for identifying the structure of organic molecules 1. Nuclear Magnetic Resonance or NMR is the single most important technique for
HWeb27 ( ; )
 HWeb27 (9.1-9.2; 9.12-9.18) 28.1. Which of the following cannot be determined about a compound by mass spectrometry? [a]. boiling point [b]. molecular formula [c]. presence of heavy isotopes (e.g., 2 H,
HWeb27 (9.1-9.2; 9.12-9.18) 28.1. Which of the following cannot be determined about a compound by mass spectrometry? [a]. boiling point [b]. molecular formula [c]. presence of heavy isotopes (e.g., 2 H,
Can you differentiate A from B using 1 H NMR in each pair?
 Can you differentiate A from B using 1 H NMR in each pair? To be NMR active any nucleus must have a spin quantum number, different from zero (I 0) As in 1 H, the spin quantum number (I) of 13 C is 1/2
Can you differentiate A from B using 1 H NMR in each pair? To be NMR active any nucleus must have a spin quantum number, different from zero (I 0) As in 1 H, the spin quantum number (I) of 13 C is 1/2
Chapter 13: Nuclear Magnetic Resonance (NMR) Spectroscopy direct observation of the H s and C s of a molecules
 hapter 13: Nuclear Magnetic Resonance (NMR) Spectroscopy direct observation of the s and s of a molecules Nuclei are positively charged and spin on an axis; they create a tiny magnetic field + + Not all
hapter 13: Nuclear Magnetic Resonance (NMR) Spectroscopy direct observation of the s and s of a molecules Nuclei are positively charged and spin on an axis; they create a tiny magnetic field + + Not all
Chapter 15 Lecture Outline
 Organic Chemistry, First Edition Janice Gorzynski Smith University of Hawaii Chapter 5 Lecture Outline Introduction to NMR Two common types of NMR spectroscopy are used to characterize organic structure:
Organic Chemistry, First Edition Janice Gorzynski Smith University of Hawaii Chapter 5 Lecture Outline Introduction to NMR Two common types of NMR spectroscopy are used to characterize organic structure:
Nuclear Magnetic Resonance H-NMR Part 1 Introduction to NMR, Instrumentation, Sample Prep, Chemical Shift. Dr. Sapna Gupta
 Nuclear Magnetic Resonance H-NMR Part 1 Introduction to NMR, Instrumentation, Sample Prep, Chemical Shift Dr. Sapna Gupta Introduction NMR is the most powerful tool available for organic structure determination.
Nuclear Magnetic Resonance H-NMR Part 1 Introduction to NMR, Instrumentation, Sample Prep, Chemical Shift Dr. Sapna Gupta Introduction NMR is the most powerful tool available for organic structure determination.
Paper 12: Organic Spectroscopy
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy 31: Combined problem on UV, IR, 1 H NMR, 13 C NMR and Mass - Part III CHE_P12_M31 TABLE OF CONTENTS 1.
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy 31: Combined problem on UV, IR, 1 H NMR, 13 C NMR and Mass - Part III CHE_P12_M31 TABLE OF CONTENTS 1.
Molecular Spectroscopy. H 2 O e -
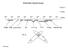 Molecular Spectroscopy ν (cm -1 ) λ (cm) 10 6 10 8 10 10 10 12 10 14 10 16 10 18 10 20 10 22 ν (Hz) NMR ESR microwave IR UV/Vis VUV X-Ray Gamma Ray H 2 e - UV/Vis Spectroscopy absorption technique X hν
Molecular Spectroscopy ν (cm -1 ) λ (cm) 10 6 10 8 10 10 10 12 10 14 10 16 10 18 10 20 10 22 ν (Hz) NMR ESR microwave IR UV/Vis VUV X-Ray Gamma Ray H 2 e - UV/Vis Spectroscopy absorption technique X hν
MOLECULAR REPRESENTATIONS AND INFRARED SPECTROSCOPY
 MOLEULAR REPRESENTATIONS AND INFRARED SPETROSOPY A STUDENT SOULD BE ABLE TO: 1. Given a Lewis (dash or dot), condensed, bond-line, or wedge formula of a compound draw the other representations. 2. Give
MOLEULAR REPRESENTATIONS AND INFRARED SPETROSOPY A STUDENT SOULD BE ABLE TO: 1. Given a Lewis (dash or dot), condensed, bond-line, or wedge formula of a compound draw the other representations. 2. Give
AQA A2 CHEMISTRY TOPIC 4.10 ORGANIC SYNTHESIS AND ANALYSIS TOPIC 4.11 STRUCTURE DETERMINATION BOOKLET OF PAST EXAMINATION QUESTIONS
 AQA A2 CHEMISTRY TOPIC 4.10 ORGANIC SYNTHESIS AND ANALYSIS TOPIC 4.11 STRUCTURE DETERMINATION BOOKLET OF PAST EXAMINATION QUESTIONS 1 1. Consider the following reaction sequence. CH 3 CH 3 CH 3 Step 1
AQA A2 CHEMISTRY TOPIC 4.10 ORGANIC SYNTHESIS AND ANALYSIS TOPIC 4.11 STRUCTURE DETERMINATION BOOKLET OF PAST EXAMINATION QUESTIONS 1 1. Consider the following reaction sequence. CH 3 CH 3 CH 3 Step 1
Lecture 13 Organic Chemistry 1
 EM 232 rganic hemistry I at hicago Lecture 13 rganic hemistry 1 Professor Duncan Wardrop February 23, 2010 1 EM 232 rganic hemistry I at hicago Spectroscopy & Spectrometry hapter 13 2 EM 232 rganic hemistry
EM 232 rganic hemistry I at hicago Lecture 13 rganic hemistry 1 Professor Duncan Wardrop February 23, 2010 1 EM 232 rganic hemistry I at hicago Spectroscopy & Spectrometry hapter 13 2 EM 232 rganic hemistry
22 and Applications of 13 C NMR
 Subject Chemistry Paper No and Title Module No and Title Module Tag 12 and rganic Spectroscopy 22 and Applications of 13 C NMR CHE_P12_M22 TABLE F CNTENTS 1. Learning utcomes 2. Introduction 3. Structural
Subject Chemistry Paper No and Title Module No and Title Module Tag 12 and rganic Spectroscopy 22 and Applications of 13 C NMR CHE_P12_M22 TABLE F CNTENTS 1. Learning utcomes 2. Introduction 3. Structural
Chem 213 Final 2012 Detailed Solution Key for Structures A H
 Chem 213 Final 2012 Detailed Solution Key for Structures A H COMPOUND A on Exam Version A (B on Exam Version B) C 8 H 6 Cl 2 O 2 DBE = 5 (aromatic + 1) IR: 1808 cm 1 suggests an acid chloride since we
Chem 213 Final 2012 Detailed Solution Key for Structures A H COMPOUND A on Exam Version A (B on Exam Version B) C 8 H 6 Cl 2 O 2 DBE = 5 (aromatic + 1) IR: 1808 cm 1 suggests an acid chloride since we
Chapter 13 Structure t Determination: Nuclear Magnetic Resonance Spectroscopy
 John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 13 Structure t Determination: ti Nuclear Magnetic Resonance Spectroscopy Revisions by Dr. Daniel Holmes MSU Paul D. Adams University of Arkansas
John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 13 Structure t Determination: ti Nuclear Magnetic Resonance Spectroscopy Revisions by Dr. Daniel Holmes MSU Paul D. Adams University of Arkansas
Infrared Spectroscopy
 x-rays ultraviolet (UV) visible Infrared (I) microwaves radiowaves near I middle I far I λ (cm) 8 x 10-5 2.5 x 10-4 2.5 x 10-3 2.5 x 10-2 µ 0.8 2.5 25 250 ν (cm -1 ) 13,000 4,000 400 40 ν (cm -1 1 ) =
x-rays ultraviolet (UV) visible Infrared (I) microwaves radiowaves near I middle I far I λ (cm) 8 x 10-5 2.5 x 10-4 2.5 x 10-3 2.5 x 10-2 µ 0.8 2.5 25 250 ν (cm -1 ) 13,000 4,000 400 40 ν (cm -1 1 ) =
Answers to Assignment #5
 Answers to Assignment #5 A. 9 8 l 2 5 DBE (benzene + 1 DBE) ( 9 2(9)+2-9 8+1+1 = 10 ˆ 5 DBE) nmr pattern of two doublets of equal integration at δ7.4 and 7.9 ppm means the group (the δ7.9 shift) IR band
Answers to Assignment #5 A. 9 8 l 2 5 DBE (benzene + 1 DBE) ( 9 2(9)+2-9 8+1+1 = 10 ˆ 5 DBE) nmr pattern of two doublets of equal integration at δ7.4 and 7.9 ppm means the group (the δ7.9 shift) IR band
IR, MS, UV, NMR SPECTROSCOPY
 CHEMISTRY 318 IR, MS, UV, NMR SPECTROSCOPY PROBLEM SET All Sections CHEMISTRY 318 IR, MS, UV, NMR SPECTROSCOPY PROBLEM SET General Instructions for the 318 Spectroscopy Problem Set Consult the Lab Manual,
CHEMISTRY 318 IR, MS, UV, NMR SPECTROSCOPY PROBLEM SET All Sections CHEMISTRY 318 IR, MS, UV, NMR SPECTROSCOPY PROBLEM SET General Instructions for the 318 Spectroscopy Problem Set Consult the Lab Manual,
Spectroscopy. Empirical Formula: Chemical Formula: Index of Hydrogen Deficiency (IHD)
 Spectroscopy Empirical Formula: Chemical Formula: Index of Hydrogen Deficiency (IHD) A)From a structure: B)From a molecular formula, C c H h N n O o X x, Formula for saturated hydrocarbons: Subtract the
Spectroscopy Empirical Formula: Chemical Formula: Index of Hydrogen Deficiency (IHD) A)From a structure: B)From a molecular formula, C c H h N n O o X x, Formula for saturated hydrocarbons: Subtract the
Terms used in UV / Visible Spectroscopy
 Terms used in UV / Visible Spectroscopy Chromophore The part of a molecule responsible for imparting color, are called as chromospheres. OR The functional groups containing multiple bonds capable of absorbing
Terms used in UV / Visible Spectroscopy Chromophore The part of a molecule responsible for imparting color, are called as chromospheres. OR The functional groups containing multiple bonds capable of absorbing
UV / Visible Spectroscopy. Click icon to add picture
 UV / Visible Spectroscopy Click icon to add picture Spectroscopy It is the branch of science that deals with the study of interaction of matter with light. OR It is the branch of science that deals with
UV / Visible Spectroscopy Click icon to add picture Spectroscopy It is the branch of science that deals with the study of interaction of matter with light. OR It is the branch of science that deals with
Welcome to Organic Chemistry II
 Welcome to Organic Chemistry II Erika Bryant, Ph.D. erika.bryant@hccs.edu Class Syllabus 3 CHAPTER 12: STRUCTURE DETERMINATION 4 What is this solution Soda Tea Coffee??? 5 What is this solution Soda Tea
Welcome to Organic Chemistry II Erika Bryant, Ph.D. erika.bryant@hccs.edu Class Syllabus 3 CHAPTER 12: STRUCTURE DETERMINATION 4 What is this solution Soda Tea Coffee??? 5 What is this solution Soda Tea
To Do s. Read Chapter 3. Complete the end-of-chapter problems, 3-1, 3-3, 3-4, 3-6 and 3-7. Answer Keys are available in CHB204H
 Read Chapter 3. To Do s Complete the end-of-chapter problems, 3-1, 3-3, 3-4, 3-6 and 3-7 Answer Keys are available in CB204 NMR Chemical Shifts Further Discussion A set of spectral data is reported when
Read Chapter 3. To Do s Complete the end-of-chapter problems, 3-1, 3-3, 3-4, 3-6 and 3-7 Answer Keys are available in CB204 NMR Chemical Shifts Further Discussion A set of spectral data is reported when
Carbon 13 NMR NUCLEAR MAGNETIC RESONANCE SPECTROSCOPY
 NUCLEAR MAGNETIC RESONANCE SPECTROSCOPY PRINCIPLE AND APPLICATION IN STRUCTURE ELUCIDATION Carbon 13 NMR Professor S. SANKARARAMAN Department of Chemistry Indian Institute of Technology Madras Chennai
NUCLEAR MAGNETIC RESONANCE SPECTROSCOPY PRINCIPLE AND APPLICATION IN STRUCTURE ELUCIDATION Carbon 13 NMR Professor S. SANKARARAMAN Department of Chemistry Indian Institute of Technology Madras Chennai
NUCLEAR MAGNETIC RESONANCE SPECTROSCOPY
 NMR Spectroscopy 1 NULEAR MAGNETI RESONANE SPETROSOPY Involves interaction of materials with the low-energy radiowave region of the electromagnetic spectrum Origin of Spectra Theory All nuclei possess
NMR Spectroscopy 1 NULEAR MAGNETI RESONANE SPETROSOPY Involves interaction of materials with the low-energy radiowave region of the electromagnetic spectrum Origin of Spectra Theory All nuclei possess
KOT 222 Organic Chemistry II
 KOT 222 Organic Chemistry II Course Objectives: 1) To introduce the chemistry of alcohols and ethers. 2) To study the chemistry of functional groups. 3) To learn the chemistry of aromatic compounds and
KOT 222 Organic Chemistry II Course Objectives: 1) To introduce the chemistry of alcohols and ethers. 2) To study the chemistry of functional groups. 3) To learn the chemistry of aromatic compounds and
Look for absorption bands in decreasing order of importance:
 1. Match the following to their IR spectra (30 points) Look for absorption bands in decreasing order of importance: a e a 2941 1716 d f b 3333 c b 1466 1.the - absorption(s) between 3100 and 2850 cm-1.
1. Match the following to their IR spectra (30 points) Look for absorption bands in decreasing order of importance: a e a 2941 1716 d f b 3333 c b 1466 1.the - absorption(s) between 3100 and 2850 cm-1.
Chemistry 343- Spring 2008
 Chemistry 343- Spring 2008 27 Chapter 2- Representative Carbon Compounds: Functional Groups, Intermolecular Forces and IR Spectroscopy A. ydrocarbons: Compounds composed of only C and Four Basic Types:
Chemistry 343- Spring 2008 27 Chapter 2- Representative Carbon Compounds: Functional Groups, Intermolecular Forces and IR Spectroscopy A. ydrocarbons: Compounds composed of only C and Four Basic Types:
16.1 Introduction to NMR Spectroscopy. Spectroscopy. Spectroscopy. Spectroscopy. Spectroscopy. Spectroscopy 4/11/2013
 What is spectroscopy? NUCLEAR MAGNETIC RESONANCE (NMR) spectroscopy may be the most powerful method of gaining structural information about organic compounds. NMR involves an interaction between electromagnetic
What is spectroscopy? NUCLEAR MAGNETIC RESONANCE (NMR) spectroscopy may be the most powerful method of gaining structural information about organic compounds. NMR involves an interaction between electromagnetic
Principles of Molecular Spectroscopy: Electromagnetic Radiation and Molecular structure. Nuclear Magnetic Resonance (NMR)
 Principles of Molecular Spectroscopy: Electromagnetic Radiation and Molecular structure Nuclear Magnetic Resonance (NMR) !E = h" Electromagnetic radiation is absorbed when the energy of photon corresponds
Principles of Molecular Spectroscopy: Electromagnetic Radiation and Molecular structure Nuclear Magnetic Resonance (NMR) !E = h" Electromagnetic radiation is absorbed when the energy of photon corresponds
Topic 10.1: Fundamentals of Organic Chemistry Notes
 Topic 10.1: Fundamentals of Organic Chemistry Notes Terminology Hydrocarbon: compounds that contain mostly hydrogen and carbon Homologous Series: compounds with the same general formula Molecular Formula:
Topic 10.1: Fundamentals of Organic Chemistry Notes Terminology Hydrocarbon: compounds that contain mostly hydrogen and carbon Homologous Series: compounds with the same general formula Molecular Formula:
Can First Year Organic Students Solve Complicated Organic Structures?
 an First Year rganic Students Solve omplicated rganic Structures? Tools of the trade (for problems in this discussion): 1. Mass Spec information limited data (M+, M+1, M+2, M+3, etc.) 2. I information
an First Year rganic Students Solve omplicated rganic Structures? Tools of the trade (for problems in this discussion): 1. Mass Spec information limited data (M+, M+1, M+2, M+3, etc.) 2. I information
CM Chemical Spectroscopy and Applications. Final Examination Solution Manual AY2013/2014
 NANYANG TECHNOLOGICAL UNIVERSITY DIVISION OF CHEMISTRY AND BIOLOGICAL CHEMISTRY SCHOOL OF PHYSICAL & MATHEMATICAL SCIENCES CM 3011 - Chemical Spectroscopy and Applications Final Examination Solution Manual
NANYANG TECHNOLOGICAL UNIVERSITY DIVISION OF CHEMISTRY AND BIOLOGICAL CHEMISTRY SCHOOL OF PHYSICAL & MATHEMATICAL SCIENCES CM 3011 - Chemical Spectroscopy and Applications Final Examination Solution Manual
C h a p t e r S i x t e e n: Nuclear Magnetic Resonance Spectroscopy. An 1 H NMR FID of ethanol
 0.2 0.4 0.6 0.8 1.0 1.2 1.4 1.6 1.8 2.0 2.2 2.4 2.6 2.8 3.0 3.2 3.4 3.6 C h a p t e r S i x t e e n: Nuclear Magnetic Resonance Spectroscopy An 1 NMR FID of ethanol Note: Problems with italicized numbers
0.2 0.4 0.6 0.8 1.0 1.2 1.4 1.6 1.8 2.0 2.2 2.4 2.6 2.8 3.0 3.2 3.4 3.6 C h a p t e r S i x t e e n: Nuclear Magnetic Resonance Spectroscopy An 1 NMR FID of ethanol Note: Problems with italicized numbers
EXPT. 9 DETERMINATION OF THE STRUCTURE OF AN ORGANIC COMPOUND USING UV, IR, NMR AND MASS SPECTRA
 EXPT. 9 DETERMINATION OF THE STRUCTURE OF AN ORGANIC COMPOUND USING UV, IR, NMR AND MASS SPECTRA Structure 9.1 Introduction Objectives 9.2 Principle 9.3 Requirements 9.4 Strategy for the Structure Elucidation
EXPT. 9 DETERMINATION OF THE STRUCTURE OF AN ORGANIC COMPOUND USING UV, IR, NMR AND MASS SPECTRA Structure 9.1 Introduction Objectives 9.2 Principle 9.3 Requirements 9.4 Strategy for the Structure Elucidation
JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II. 5 Credit Hours. Prepared by: Richard A. Pierce
 JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
The Use of NMR Spectroscopy
 Spektroskopi Molekul Organik (SMO): Nuclear Magnetic Resonance (NMR) Spectroscopy All is adopted from McMurry s Organic Chemistry The Use of NMR Spectroscopy Used to determine relative location of atoms
Spektroskopi Molekul Organik (SMO): Nuclear Magnetic Resonance (NMR) Spectroscopy All is adopted from McMurry s Organic Chemistry The Use of NMR Spectroscopy Used to determine relative location of atoms
Basic One- and Two-Dimensional NMR Spectroscopy
 Horst Friebolin Basic One- and Two-Dimensional NMR Spectroscopy Third Revised Edition Translated by Jack K. Becconsall WILEY-VCH Weinheim New York Chichester Brisbane Singapore Toronto Contents XV 1 The
Horst Friebolin Basic One- and Two-Dimensional NMR Spectroscopy Third Revised Edition Translated by Jack K. Becconsall WILEY-VCH Weinheim New York Chichester Brisbane Singapore Toronto Contents XV 1 The
Chapter 14. Nuclear Magnetic Resonance Spectroscopy
 Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 14 Nuclear Magnetic Resonance Spectroscopy Prepared by Rabi Ann Musah State University of New York at Albany Copyright
Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 14 Nuclear Magnetic Resonance Spectroscopy Prepared by Rabi Ann Musah State University of New York at Albany Copyright
Calculate a rate given a species concentration change.
 Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Chem 1075 Chapter 19 Organic Chemistry Lecture Outline
 Chem 1075 Chapter 19 Organic Chemistry Lecture Outline Slide 2 Introduction Organic chemistry is the study of and its compounds. The major sources of carbon are the fossil fuels: petroleum, natural gas,
Chem 1075 Chapter 19 Organic Chemistry Lecture Outline Slide 2 Introduction Organic chemistry is the study of and its compounds. The major sources of carbon are the fossil fuels: petroleum, natural gas,
1. Predict the structure of the molecules given by the following spectral data: a Mass spectrum:m + = 116
 Additional Problems for practice.. Predict the structure of the molecules given by the following spectral data: a Mass spectrum:m + = IR: weak absorption at 9 cm - medium absorption at cm - NMR 7 3 3 C
Additional Problems for practice.. Predict the structure of the molecules given by the following spectral data: a Mass spectrum:m + = IR: weak absorption at 9 cm - medium absorption at cm - NMR 7 3 3 C
CHEMISTRY 213 XMAS 04 1 ANSWER ON THE GREEN COMPUTER ANSWER SHEET PROVIDED USING A PENCIL CHOICES MAY BE USED MORE THAN ONCE
 CHEMISTRY 213 XMAS 04 1 ANSWER ON THE GREEN COMPUTER ANSWER SHEET PROVIDED USING A PENCIL CHOICES MAY BE USED MORE THAN ONCE Using the molecules: A: CH 3 CH CHCO 2 CH 3 B: CH 3 CO 2 CH CHCH 3 C: CH 3 CH
CHEMISTRY 213 XMAS 04 1 ANSWER ON THE GREEN COMPUTER ANSWER SHEET PROVIDED USING A PENCIL CHOICES MAY BE USED MORE THAN ONCE Using the molecules: A: CH 3 CH CHCO 2 CH 3 B: CH 3 CO 2 CH CHCH 3 C: CH 3 CH
Introduc)on to Func)onal Groups in Organic Molecules
 Introduc)on to Func)onal Groups in rganic Molecules CH 3 H 3 C N C C N C C N N CH CH 3 Caffeine Func)onal Group Func%onal group - collec)on of atoms at a site that have a characteris)c behavior in all
Introduc)on to Func)onal Groups in rganic Molecules CH 3 H 3 C N C C N C C N N CH CH 3 Caffeine Func)onal Group Func%onal group - collec)on of atoms at a site that have a characteris)c behavior in all
Structure Determination. How to determine what compound that you have? One way to determine compound is to get an elemental analysis
 Structure Determination How to determine what compound that you have? ne way to determine compound is to get an elemental analysis -basically burn the compound to determine %C, %H, %, etc. from these percentages
Structure Determination How to determine what compound that you have? ne way to determine compound is to get an elemental analysis -basically burn the compound to determine %C, %H, %, etc. from these percentages
Tuesday, January 13, NMR Spectroscopy
 NMR Spectroscopy NMR Phenomenon Nuclear Magnetic Resonance µ A spinning charged particle generates a magnetic field. A nucleus with a spin angular momentum will generate a magnetic moment (μ). If these
NMR Spectroscopy NMR Phenomenon Nuclear Magnetic Resonance µ A spinning charged particle generates a magnetic field. A nucleus with a spin angular momentum will generate a magnetic moment (μ). If these
Vibrations. Matti Hotokka
 Vibrations Matti Hotokka Identify the stuff I ve seen this spectrum before. I know what the stuff is Identify the stuff Let s check the bands Film: Polymer Aromatic C-H Aliphatic C-H Group for monosubstituted
Vibrations Matti Hotokka Identify the stuff I ve seen this spectrum before. I know what the stuff is Identify the stuff Let s check the bands Film: Polymer Aromatic C-H Aliphatic C-H Group for monosubstituted
SECOND YEAR ORGANIC CHEMISTRY - REVISION COURSE Lecture 2 MOLECULAR STRUCTURE 2: SPECTROSCOPIC ANALYSIS
 Prof Ben Davis SECOND YEAR ORGANIC CEMISTRY - REVISION COURSE Lecture 2 MOLECULAR STRUCTURE 2: SPECTROSCOPIC ANALYSIS Books: Williams and Fleming, " Spectroscopic Methods in Organic Chemistry", arwood
Prof Ben Davis SECOND YEAR ORGANIC CEMISTRY - REVISION COURSE Lecture 2 MOLECULAR STRUCTURE 2: SPECTROSCOPIC ANALYSIS Books: Williams and Fleming, " Spectroscopic Methods in Organic Chemistry", arwood
Chapter 1 Reactions of Organic Compounds. Reactions Involving Hydrocarbons
 Chapter 1 Reactions of Organic Compounds Reactions Involving Hydrocarbons Reactions of Alkanes Single bonds (C-C) are strong and very hard to break, therefore these compounds are relatively unreactive
Chapter 1 Reactions of Organic Compounds Reactions Involving Hydrocarbons Reactions of Alkanes Single bonds (C-C) are strong and very hard to break, therefore these compounds are relatively unreactive
Chapter 12 Structure Determination: Mass Spectrometry and Infrared Spectroscopy
 Chapter 12 Structure Determination: Mass Spectrometry and Infrared Spectroscopy Figure 12.1 - The electron-ionization, magneticsector mass spectrometer Representing the Mass Spectrum Base Peak Parent
Chapter 12 Structure Determination: Mass Spectrometry and Infrared Spectroscopy Figure 12.1 - The electron-ionization, magneticsector mass spectrometer Representing the Mass Spectrum Base Peak Parent
(2) Read each statement carefully and pick the one that is incorrect in its information.
 Organic Chemistry - Problem Drill 17: IR and Mass Spectra No. 1 of 10 1. Which statement about infrared spectroscopy is incorrect? (A) IR spectroscopy is a method of structure determination based on the
Organic Chemistry - Problem Drill 17: IR and Mass Spectra No. 1 of 10 1. Which statement about infrared spectroscopy is incorrect? (A) IR spectroscopy is a method of structure determination based on the
