Membrane Preparation and Characterization
|
|
|
- Natalie Casey
- 5 years ago
- Views:
Transcription
1 Membrane Preparation and Characterization Prepare New Polymeric Membranes for Energy Applications A membrane can be considered as a selective barrier that allows transport of some compounds while rejecting others. Depending on the application different types of membranes are needed. Given a specific application it is possible to target the desired membrane structure and morphology. Polymeric membranes are desirable in a lot of applications due to e.g. the high mechanical stability, good selectivity and easy preparation on industrial scale. In the figure above is shown three examples of generic structures of some polymer chains. By carefully choosing the monomer units (the building blocks) and the reaction method it is possible to design different polymer structures and morphologies. One application for the polymeric membranes could be for example the flow batteries where a low crossover of the electrolytes (ions) is needed to get a high efficiency. Preparation of polymeric structures for membranes could be a project where you will get to work with organic synthesis (polymerizations), characterization techniques (such NMR, IR, GPC, contact angle, impedance spectroscopy) but also testing the membrane for relevant transport properties (could be selectivity, permeability, charge-discharge). Surface or pore modifications of support materials can be used to optimize the properties of the exiting support, stabilize a mechanical unstable material and/or get enhanced properties by (hopefully) getting the best from both the used materials. This method can be used to prepare membranes for applications within gas separation where the trade-off between diffusivity, permeability and selectivity is essential. Again synthesis and characterization will be part of the project as well as characterization the produced membrane with respect to the mentioned properties. For further information: Anders Bentien, Jacopo Catalano or Mette Kristensen Great bachelor and masters projects for students from Chemistry, Chemical Engineering and inano
2 Electrokinetic Energy Conversion Efficiency of Nano-porous, Ion-conductive Membranes This project applies membranes for energy conversion, i.e. for transforming energy from one form to another. Kinetic energy, in form of a pressure difference, is converted to electrical energy and vice versa. The main focus of this project is to measure the direct efficiency of this conversion for various membranes. Applying, improving and designing specialized equipment. The use of various sensors (pressure, mass, temperature, potential, current etc.) Developing LabVIEW based programs for collecting data. Hydrophobic surface modification of pores For further information contact: David Østedgaard-Munck or Jacopo Catalano Great bachelor and masters projects for students from Chemistry, Engineering and Physics Electrokinetic Hydrogen Compression In this project the feasibility of using Electrokinetic phenomena for gas compression is investigated. This is a new pathway for refrigerators or, more generally, gas/vapor compression systems. Bake-in of a H 2 compression prototype (available at the Chem. Eng. laboratories). Acquire experience in chemical engineering processes, system design and control Exploring disruptive processes and collecting data never reported in the literature. For further information contact: Anders Bentien, David Østedgaard-Munck or Jacopo Catalano Great masters projects for students from Engineering and Physics
3 Continuum/CFD Modeling of Pore Nanostructures Charged ion-exchange membranes, or other films containing pores or channels of nanoscopic dimensions carrying immobile wall charge, can be used for energy conversion (for instance reverse electrodialysis, electrokinetic energy conversion) where the flow of fluid down a pressure or a salinity gradient or through such materials creates an electric current and voltage. Considering ions as point charges, the mathematical theory combines the Navier-Stokes, Nernst-Planck and Poisson equations. In this project you will acquire experience in the description of electro-migration phenomena which characterize not only membrane but in general transport in the exciting new classes of materials such as carbon nanotubes. Modelling of transport properties of nanocapillaries and pore network Simulations of electro-migration of ions Material/polymer optimization This project requires a good mathematical and physical background and it is part of a collaboration between Aarhus University and Wetsus, European Centre of Excellence for Sustainable Water Technology. For further information: Jacopo Catalano Great masters projects for students from Engineering and Physics Design and Manufacturing of Electrochemical Cells This project aims to investigate the transport properties of ions typically used in novel energy storage systems (for instance flow batteries) in polymeric materials. The knowledge of the diffusion coefficients is of fundamental importance on designing the batteries separators for efficient batteries and to improve their cyclability. In particular you will design an electrochemical cell for measuring the ion diffusion coefficient upon a chemical potential driving force and you are expected to integrate UV/VIS spectroscopy probes. You will acquire knowledge on process design, optimization, data acquisition and analysis. Design of set-ups for measuring diffusion of ions Design of Osmometers Data acquisition and analysis (python, matlab, Labivew) For further information: Jacopo Catalano, Emil Drazevic or Mette Kristensen Great bachelor and masters projects for students from Chemistry, Engineering, inano and Physics
4 Solar Batteries Optimization of Lab-scale Production of Thin Film Semiconductor Photoelectrodes We have recently shown that certain electrolytes in redox flow batteries can be directly solar charged (with a bias potential) by putting them into contact with thin-film photoelectrodes and shining light on them. However, the efficiency depends very much on the production method. You will produce the photoelectrodes under various conditions and characterize them in terms of how efficiently they can charge a selected redox flow battery using a solar simulator. XRD, impedance spectroscopy & SEM measurements can also be included in the characterization. Possible photoelectrodes are: Ta 3 N 5, WSe 2, Fe 2 O 3 (hematite) For further information: Kristina Wedege Great bachelor and masters projects for students from Chemistry and inano Novel Membranes for Gas Separation and Carbon Capture Glassy or rubbery polymer filled with inorganics for CO 2 capture Pore-filling modifications Effect of post-treatment of membranes Gas permeability measurements Structural and microscopic characterization For further information contact: Jacopo Catalano Great bachelor and masters projects for students from Chemistry
5 Flow Batteries Using Electrochemistry to Discover Alternative Electrolytes for Redox Flow Batteries in Basic Environment It is important to find new and better electrolytes for use in redox flow batteries to improve the technology to a commercial level. Electrolytes that works at ph > 12 are good because crossover can be limited, kinetics improved and the electrolytes can possibly be coupled with solar charging. Electrolytes in basic environment can be both organic or inorganic, e.g. anthraquinones, iron-cyanide complexes or cobalt/iron complexes with organic ligands: You will determine standard redox potentials, stability and kinetic parameters by using electrochemical methods (e.g. cyclic voltammetry, rotating disk electrode experiments) and will eventually test the electrolytes in a lab scale battery. For further information contact: Kristina Wedege, Amirreza Khataee or Anders Bentien Great bachelor (masters) projects for students from Chemistry Synthesis, Purification and Characterization of Organic Quinone based Derivatives for use in Redox Flow Batteries Batteries for a future renewable based utility grid must be based on earth abundant raw materials and must be at considerably lower costs. Some Anthraquinones have shown a huge potential in aqueous based redox flow batteries. Some of the important parameters for batteries, e.g., solubility, stability and redox potential are to a large extent determined by the side groups on the Quinones. The goal is to synthesize organic species which are not commercially available but are promising as organic redox species. To achieve our goal of finding the best possible not from the shelf candidate, we follow two different methodologies. (i) The first approach is to modify quinone structures in order to achieve better solubility and stability. In this case the quinone moiety is substituted with hydrophilic groups to increase solubility. (ii) The second methodology concerns modifying and testing core structures where instead of the oxygen (Quinone derivatives) the nitrogen is the containing core structures like viologen, NAD and FAD analogues. You will synthesize and purify selected anthraquinone molecules from unsubstituted anthraquinone and determine the standard potential by electrochemical measurements. For further information contact: Emil Drazevic or Anders Bentien Great masters projects for students from Chemistry
6 Redox Buffers - a Pathway to All Quinone Flow Battery The open circuit voltage of currently feasible all quinone battery is only 0.7 V. The electrode potential of quinones strongly depends on the ph. If the difference in ph on catholyte and anolyte side is 2 ph units, additional 0.12 V can be extracted from the system. For instance the potential of catechol is V at ph=6. On the negative side anthraflavic acid has potential of 0.48 V at ph=8. This amounts to 0.92 V of cell potential if this ph difference can be maintained. Good s biological buffers (MES, MOPS, MOPSO) utilize morpholino and sulfonic groups on a single organic molecule. Sulfonic groups act as a strong proton donor while morpholino groups acts as proton acceptors. Inspired by the Goods buffers we intend to attach both morpholino and acidic groups and make quinones-buffers that are incredibly soluble in water and can keep the cell voltage constant during charging/discharging. Some of the possible chemistries are find below/above. TAPS derivative of anthraflavic acid You will do some organic synthesis, basic electrochemical characterization and thereafter some battery tests. For further information contact: Emil Drazevic or Amirreza Khataee MOPS derivative of catehol Great masters projects for students from Chemistry Optimization Studies for a kw-scale Vanadium Redox Flow Battery and Photovoltaic System The development of the electric system, due to the strong increase of the distributed and renewable generation, is posing new challenges to the management and control of the power system. Concerning the photovoltaic power generation, one of the major issues is related to its production profile, hardly predictable and manageable, that could cause overloading or overvoltage. This project is related to the development and optimization of control strategies of a kw-scale Vanadium Redox Flow Battery System that is charged with solar power and discharges energy to the local power grid. kw-scale Redox Flow Battery stack Schematics of the hybrid energy system You will work with a state-of-the-art Vanadium Redox Flow Battery stack. Your task will be to develop control strategies for the energy storage system with respect to e.g. power generation and energy generation. Of utmost importance to the project is to consider the interaction between the energy storage and the solar energy system. The behavior of the solar energy system will be mimicked using an industrial power supply. For further information contact: Luis Carlos Pérez Matínez Great masters projects for students from Engineering
7 Solid State Batteries New materials are experimented in rechargeable aqueous based solid state batteries. Commercial Ni-Cd and Ni-MH batteries outperform in many different aspects, however the former are toxic and the latter appear too expensive for large scale energy storage. Therefore, we have started to look into new anodes for Ni based batteries. Currently we use Ni(OH) 2 as the cathode material, and a redox active polymer as anode material. We have achieved 1000 cycles with 60% loss in capacity. Most of the capacity loss (50%) results after 1 st charging. It is hypothesized that our polymer has very low polydisperse index i.e. broad molecular weight distribution, which is why smaller molecules escape across the microporous separator and causes irreversible capacity loss. As with any battery technology in its beginnings, many obstacles are on the way. The process of making the batteries is shown below. It is a fun way of doing science! Some of the possible projects are described below, but we can always sit down and hear your own ideas for the projects! Optimization of Existing Custom made Electrodes The goal of this project is to make thicker polymer and nickel hydroxide electrodes that can be charged and discharged at a rate of ¼ of the rated capacity. This is a standard charging/discharging rate in large scale energy storage systems. The thicker electrodes will reduce the cost of the battery but that also means electrical resistance will increase and lower the charge/discharge rate. One of the strategies to overcome this problem is to implement carbon fibers into existing electrodes. However, the process of making the electrodes has many other variables, e.g. composition, pressure and pressing temperature of electrodes, electrolyte composition, etc., so there are many other possible ways for achieving the goal. All Organic Solid State Batteries The goal of this project is to make an all organic solid state battery with a voltage of at least 1 V in aqueous media. Interesting thing here, besides the low cost potential, is that all organic solid state batteries are environmentally friendly in a sense that they can be burned or biodegraded after use, thus we could avoid the need for specific recycling process. It is anticipated that one side of the battery utilize the existing redox polymer anode while the other side of the battery would utilize water insoluble redox polymer with sufficiently positive formal potential. The idea is to use TEMPO radical, which is insoluble in water, as the cathode material. The problem is once TEMPO is charged it becomes cation which is then soluble in water. In this sense the main research challenge is to make it insoluble and one of the strategies is attaching the TEMPO radical on a polymer backbone. There are existing papers in the literature with recipes for TEMPO radical polymerization, since the rumor is TEMPO is used as cathode in Li ion polymer solid state battery.
8 Novel Redox Polymer Anodes The goal of the project is to find a way to synthesize other low cost redox polymers for their use as an anode in rechargeable solid state batteries. This project will be the most research intensive. It will probably include a lot of initial literature reading and teamwork, also with our polymer guys at Hangøvej 2. The unknown state of this project at this point will include lot of risk. But, there could also be some low hanging fruits here. For further information contact: Casper Clausen, Emil Drazevic or Anders Bentien Great bachelor and masters projects for students from Chemistry and Chemical Engineering Projects with Companies Grundfos and Kamstrup have a range of master projects. If you are interested please contact Anders Bentien Further Information To get further information regarding the different projects feel free to contact us by or come by Department of Engineering on Hangøvej 2. Anders Bentien (Associate Professor and group leader) Jacopo Catalano (Assistant Professor) Emil Drazevic (Post Doc) Luis Carlos Pérez Matínez (Post Doc) Amirreza Khataee (PhD student) Casper Clausen (PhD student) David N. Østedgaard-Munck (PhD student) Kristina Wedege (PhD student) Mette B. Kristensen (PhD student) bentien@eng.au.dk jcatalano@eng.au.dk edrazevic@eng.au.dk luis.perez@eng.au.dk khataee@eng.au.dk casperc@eng.au.dk dnmunck@eng.au.dk wedege@eng.au.dk mettebk@eng.au.dk
Batteries (Electrochemical Power Sources)
 Batteries (Electrochemical Power Sources) 1. Primary (single-discharge) batteries. => finite quantity of the reactants 2. Secondary or rechargeable batteries => regeneration of the original reactants by
Batteries (Electrochemical Power Sources) 1. Primary (single-discharge) batteries. => finite quantity of the reactants 2. Secondary or rechargeable batteries => regeneration of the original reactants by
Optimization of MnO2 Electrodeposits using Graphenated Carbon Nanotube Electrodes for Supercapacitors
 Optimization of MnO2 Electrodeposits using Graphenated Carbon Nanotube Electrodes for Supercapacitors Waleed Nusrat, 100425398 PHY 3090U Material Science Thursday April 9 th 2015 Researchers optimize the
Optimization of MnO2 Electrodeposits using Graphenated Carbon Nanotube Electrodes for Supercapacitors Waleed Nusrat, 100425398 PHY 3090U Material Science Thursday April 9 th 2015 Researchers optimize the
e - Galvanic Cell 1. Voltage Sources 1.1 Polymer Electrolyte Membrane (PEM) Fuel Cell
 Galvanic cells convert different forms of energy (chemical fuel, sunlight, mechanical pressure, etc.) into electrical energy and heat. In this lecture, we are interested in some examples of galvanic cells.
Galvanic cells convert different forms of energy (chemical fuel, sunlight, mechanical pressure, etc.) into electrical energy and heat. In this lecture, we are interested in some examples of galvanic cells.
Prof. Mario L. Ferrari
 Sustainable Energy Mod.1: Fuel Cells & Distributed Generation Systems Dr. Ing. Mario L. Ferrari Thermochemical Power Group (TPG) - DiMSET University of Genoa, Italy Lesson II Lesson II: fuel cells (electrochemistry)
Sustainable Energy Mod.1: Fuel Cells & Distributed Generation Systems Dr. Ing. Mario L. Ferrari Thermochemical Power Group (TPG) - DiMSET University of Genoa, Italy Lesson II Lesson II: fuel cells (electrochemistry)
NANOCOMPOSITE CATION EXCHANGE MEMBRANE WITH FOULING RESISTANCE AND ENHANCED SALINITY GRADIENT POWER GENERATION FOR REVERSE ELECTRODIALYSIS
 NANOCOMPOSITE CATION EXCHANGE MEMBRANE WITH FOULING RESISTANCE AND ENHANCED SALINITY GRADIENT POWER GENERATION FOR REVERSE ELECTRODIALYSIS X I N T O N G, B O P E N G Z H A N G, A N D Y O N G S H E N G
NANOCOMPOSITE CATION EXCHANGE MEMBRANE WITH FOULING RESISTANCE AND ENHANCED SALINITY GRADIENT POWER GENERATION FOR REVERSE ELECTRODIALYSIS X I N T O N G, B O P E N G Z H A N G, A N D Y O N G S H E N G
Supplementary Figure 1 Supplementary Figure 2
 Supplementary Figure 1 XRD pattern of pure 3D PGC framework. The pure 3D PGC was obtained by immersing NaCl Na 2 S@GC in water to remove the NaCl and Na 2 S. The broad reflection peak in the range of 15
Supplementary Figure 1 XRD pattern of pure 3D PGC framework. The pure 3D PGC was obtained by immersing NaCl Na 2 S@GC in water to remove the NaCl and Na 2 S. The broad reflection peak in the range of 15
Advanced Analytical Chemistry Lecture 12. Chem 4631
 Advanced Analytical Chemistry Lecture 12 Chem 4631 What is a fuel cell? An electro-chemical energy conversion device A factory that takes fuel as input and produces electricity as output. O 2 (g) H 2 (g)
Advanced Analytical Chemistry Lecture 12 Chem 4631 What is a fuel cell? An electro-chemical energy conversion device A factory that takes fuel as input and produces electricity as output. O 2 (g) H 2 (g)
Chapter - 8. Summary and Conclusion
 Chapter - 8 Summary and Conclusion The present research explains the synthesis process of two transition metal oxide semiconductors SnO 2 and V 2 O 5 thin films with different morphologies and studies
Chapter - 8 Summary and Conclusion The present research explains the synthesis process of two transition metal oxide semiconductors SnO 2 and V 2 O 5 thin films with different morphologies and studies
Allen J. Bard, Netzahualcóyotl Arroyo-Currás (Netz Arroyo), Jinho Chang, Brent Bennett. Department of Chemistry and Center for Electrochemistry
 REDOX Allen J. Bard, Netzahualcóyotl Arroyo-Currás (Netz Arroyo), Jinho Chang, Brent Bennett Department of Chemistry and Center for Electrochemistry The University of Texas at Austin 1 What is a redox
REDOX Allen J. Bard, Netzahualcóyotl Arroyo-Currás (Netz Arroyo), Jinho Chang, Brent Bennett Department of Chemistry and Center for Electrochemistry The University of Texas at Austin 1 What is a redox
State of São Paulo. Brasil. Localization. Santo André
 Brasil State of São Paulo Localization Santo André The Group www.crespilho.com Frank N. Crespilho Pablo R. S. Abdias Group Leader Synthesis of nanostructured materials using polyelectrolytes Andressa R.
Brasil State of São Paulo Localization Santo André The Group www.crespilho.com Frank N. Crespilho Pablo R. S. Abdias Group Leader Synthesis of nanostructured materials using polyelectrolytes Andressa R.
Small-scale demo, large-scale promise of novel bromine battery 27 June 2014, by Nancy W. Stauffer
 Small-scale demo, large-scale promise of novel bromine battery 27 June 2014, by Nancy W. Stauffer Figure 1 The availability of low-cost, high-capacity energy storage technology could profoundly change
Small-scale demo, large-scale promise of novel bromine battery 27 June 2014, by Nancy W. Stauffer Figure 1 The availability of low-cost, high-capacity energy storage technology could profoundly change
Structure. relevant. Example. What atomic. How can the. Research
 Structure property studies; intermetallics and functional oxides In this project you will combine methods like X ray or neutron diffraction that enables determination of the atomic arrangement of crystalline
Structure property studies; intermetallics and functional oxides In this project you will combine methods like X ray or neutron diffraction that enables determination of the atomic arrangement of crystalline
Chemistry: The Central Science. Chapter 20: Electrochemistry
 Chemistry: The Central Science Chapter 20: Electrochemistry Redox reaction power batteries Electrochemistry is the study of the relationships between electricity and chemical reactions o It includes the
Chemistry: The Central Science Chapter 20: Electrochemistry Redox reaction power batteries Electrochemistry is the study of the relationships between electricity and chemical reactions o It includes the
Electrolytes for Fuel Cells
 Electrolytes for Fuel Cells Tom Zawodzinski Materials Science and Technology Division Los Alamos National Laboratory Air-Breather Fuel Cell Stack Systems Laptop Demo DCH/Enable Prototype Small Battery
Electrolytes for Fuel Cells Tom Zawodzinski Materials Science and Technology Division Los Alamos National Laboratory Air-Breather Fuel Cell Stack Systems Laptop Demo DCH/Enable Prototype Small Battery
Materials and Structural Design for Advanced Energy Storage Devices
 Materials and Structural Design for Advanced Energy Storage Devices Imran Shakir Sustainable Energy Technologies Center (SET) King Saud University Saudi Arabia Specific Power (W/kg) Introduction and Motivation
Materials and Structural Design for Advanced Energy Storage Devices Imran Shakir Sustainable Energy Technologies Center (SET) King Saud University Saudi Arabia Specific Power (W/kg) Introduction and Motivation
Chapter 18. Electrochemistry
 Chapter 18 Electrochemistry Section 17.1 Spontaneous Processes and Entropy Section 17.1 http://www.bozemanscience.com/ap-chemistry/ Spontaneous Processes and Entropy Section 17.1 Spontaneous Processes
Chapter 18 Electrochemistry Section 17.1 Spontaneous Processes and Entropy Section 17.1 http://www.bozemanscience.com/ap-chemistry/ Spontaneous Processes and Entropy Section 17.1 Spontaneous Processes
Electrochemical Cell - Basics
 Electrochemical Cell - Basics The electrochemical cell e - (a) Load (b) Load e - M + M + Negative electrode Positive electrode Negative electrode Positive electrode Cathode Anode Anode Cathode Anode Anode
Electrochemical Cell - Basics The electrochemical cell e - (a) Load (b) Load e - M + M + Negative electrode Positive electrode Negative electrode Positive electrode Cathode Anode Anode Cathode Anode Anode
Activity. Modeling the Fuel Cell Reaction. Overview. Advance Preparation. Background Information
 4 Activity 1-2 class sessions Modeling the uel Cell Reaction 2011 Regents of the University of California Overview n order to understand the chemistry of fuel cells, students are introduced to oxidation-reduction
4 Activity 1-2 class sessions Modeling the uel Cell Reaction 2011 Regents of the University of California Overview n order to understand the chemistry of fuel cells, students are introduced to oxidation-reduction
Electrophoretic Deposition. - process in which particles, suspended in a liquid medium, migrate in an electric field and deposit on an electrode
 Electrophoretic Deposition - process in which particles, suspended in a liquid medium, migrate in an electric field and deposit on an electrode no redox differs from electrolytic in several ways deposit
Electrophoretic Deposition - process in which particles, suspended in a liquid medium, migrate in an electric field and deposit on an electrode no redox differs from electrolytic in several ways deposit
Bio-electrochemistry Prof. Mainak Das Department of Biological Sciences & Bioengineering & Design Programme Indian Institute of Technology, Kanpur
 Bio-electrochemistry Prof. Mainak Das Department of Biological Sciences & Bioengineering & Design Programme Indian Institute of Technology, Kanpur Lecture 04 Galvanic Cells-I Welcome back to the lecture
Bio-electrochemistry Prof. Mainak Das Department of Biological Sciences & Bioengineering & Design Programme Indian Institute of Technology, Kanpur Lecture 04 Galvanic Cells-I Welcome back to the lecture
This material is based upon work supported by the National Science Foundation under Grant Number DUE
 This material is based upon work supported by the National Science Foundation under Grant Number DUE-1140469. Any opinions, findings, and conclusions or recommendations expressed in this material are those
This material is based upon work supported by the National Science Foundation under Grant Number DUE-1140469. Any opinions, findings, and conclusions or recommendations expressed in this material are those
Modeling as a tool for understanding the MEA. Henrik Ekström Utö Summer School, June 22 nd 2010
 Modeling as a tool for understanding the MEA Henrik Ekström Utö Summer School, June 22 nd 2010 COMSOL Multiphysics and Electrochemistry Modeling The software is based on the finite element method A number
Modeling as a tool for understanding the MEA Henrik Ekström Utö Summer School, June 22 nd 2010 COMSOL Multiphysics and Electrochemistry Modeling The software is based on the finite element method A number
Chapter 7 Electrochemistry
 Chapter 7 Electrochemistry Outside class reading Levine: pp. 417 14.4 Galvanic cells: pp. 423 14.5 types of reversible electrodes 7.6.1 Basic concepts of electrochemical apparatus (1) Electrochemical apparatus
Chapter 7 Electrochemistry Outside class reading Levine: pp. 417 14.4 Galvanic cells: pp. 423 14.5 types of reversible electrodes 7.6.1 Basic concepts of electrochemical apparatus (1) Electrochemical apparatus
Lattice Boltzmann simulation of ion and electron transport in lithium ion battery porous electrode during discharge process
 Available online at www.sciencedirect.com ScienceDirect Energy Procedia 88 (2016 ) 642 646 CUE2015-Applied Energy Symposium and Summit 2015: Low carbon cities and urban energy systems Lattice Boltzmann
Available online at www.sciencedirect.com ScienceDirect Energy Procedia 88 (2016 ) 642 646 CUE2015-Applied Energy Symposium and Summit 2015: Low carbon cities and urban energy systems Lattice Boltzmann
Direct Energy Conversion: Fuel Cells
 Direct Energy Conversion: Fuel Cells References and Sources: Direct Energy Conversion by Stanley W. Angrist, Allyn and Beacon, 1982. Fuel Cell Systems, Explained by James Larminie and Andrew Dicks, Wiley,
Direct Energy Conversion: Fuel Cells References and Sources: Direct Energy Conversion by Stanley W. Angrist, Allyn and Beacon, 1982. Fuel Cell Systems, Explained by James Larminie and Andrew Dicks, Wiley,
Evaluation of design options for tubular redox flow batteries
 Dept. Mechanical Engineering and Production Heinrich-Blasius-Institute for Physical Technologies Evaluation of design options for tubular redox flow batteries Thorsten Struckmann, Max Nix, Simon Ressel
Dept. Mechanical Engineering and Production Heinrich-Blasius-Institute for Physical Technologies Evaluation of design options for tubular redox flow batteries Thorsten Struckmann, Max Nix, Simon Ressel
Monolithic Cells for Solar Fuels
 Electronic Supplementary Material (ESI) for Chemical Society Reviews. This journal is The Royal Society of Chemistry 2014 Monolithic Cells for Solar Fuels Jan Rongé, Tom Bosserez, David Martel, Carlo Nervi,
Electronic Supplementary Material (ESI) for Chemical Society Reviews. This journal is The Royal Society of Chemistry 2014 Monolithic Cells for Solar Fuels Jan Rongé, Tom Bosserez, David Martel, Carlo Nervi,
IV. Transport Phenomena. Lecture 23: Ion Concentration Polarization
 IV. Transport Phenomena Lecture 23: Ion Concentration Polarization MIT Student (and MZB) Ion concentration polarization in electrolytes refers to the additional voltage drop (or internal resistance ) across
IV. Transport Phenomena Lecture 23: Ion Concentration Polarization MIT Student (and MZB) Ion concentration polarization in electrolytes refers to the additional voltage drop (or internal resistance ) across
Principles and Applications of Electrochemistry
 Principles and Applications of Electrochemistry Fourth edition D. R. CROW Professor of Electrochemistry and Dean of Research University of Wolverhampton BLACKIE ACADEMIC & PROFESSIONAL An Imprint of Chapman
Principles and Applications of Electrochemistry Fourth edition D. R. CROW Professor of Electrochemistry and Dean of Research University of Wolverhampton BLACKIE ACADEMIC & PROFESSIONAL An Imprint of Chapman
12.05 Galvanic Cells. Zn(s) + 2 Ag + (aq) Zn 2+ (aq) + 2 Ag(s) Ni(s) + Pb 2+ (aq) «Ni 2+ (aq) + Pb(s)
 12.05 Galvanic Cells 1. In an operating voltaic cell, reduction occurs A) at the anode B) at the cathode C) in the salt bridge D) in the wire 2. Which process occurs in an operating voltaic cell? A) Electrical
12.05 Galvanic Cells 1. In an operating voltaic cell, reduction occurs A) at the anode B) at the cathode C) in the salt bridge D) in the wire 2. Which process occurs in an operating voltaic cell? A) Electrical
Supporting Information
 Supporting Information A Low-Potential Pyridinium Anolyte for Aqueous Redox Flow Batteries Christo S. Sevov, a,b Koen. endriks, a,b and Melanie S. Sanford* a,b a. Department of Chemistry, University of
Supporting Information A Low-Potential Pyridinium Anolyte for Aqueous Redox Flow Batteries Christo S. Sevov, a,b Koen. endriks, a,b and Melanie S. Sanford* a,b a. Department of Chemistry, University of
ORGANIC ELECTRODE MATERIALS AND THEIR APPLICATIONS IN RECHARGEABLE BATTERIES
 ORGANIC ELECTRODE MATERIALS AND THEIR APPLICATIONS IN RECHARGEABLE BATTERIES Assoc. Prof. Burak ESAT Fatih University, Department Of Chemistry Istanbul-Turkey besat@fatih.edu.tr COST-EXIL October 2015
ORGANIC ELECTRODE MATERIALS AND THEIR APPLICATIONS IN RECHARGEABLE BATTERIES Assoc. Prof. Burak ESAT Fatih University, Department Of Chemistry Istanbul-Turkey besat@fatih.edu.tr COST-EXIL October 2015
Chapter 8: Energy from Electron Transfer
 Chapter 8: Energy from Electron Transfer In his 2006 State of the Union address, President George W. Bush proclaimed... we are addicted to oil Are we doomed as a country to go on to the bitter end in terms
Chapter 8: Energy from Electron Transfer In his 2006 State of the Union address, President George W. Bush proclaimed... we are addicted to oil Are we doomed as a country to go on to the bitter end in terms
Principles of Electrochemistry Second Edition
 Principles of Electrochemistry Second Edition Jiri Koryta Institute of Physiology, Czechoslovak Academy of Sciences, Prague HKJin Dvorak Department of Physical Chemistry, Faculty of Science, Charles University,
Principles of Electrochemistry Second Edition Jiri Koryta Institute of Physiology, Czechoslovak Academy of Sciences, Prague HKJin Dvorak Department of Physical Chemistry, Faculty of Science, Charles University,
Lecture Presentation. Chapter 18. Electrochemistry. Sherril Soman Grand Valley State University Pearson Education, Inc.
 Lecture Presentation Chapter 18 Electrochemistry Sherril Soman Grand Valley State University Harnessing the Power in Nature The goal of scientific research is to understand nature. Once we understand the
Lecture Presentation Chapter 18 Electrochemistry Sherril Soman Grand Valley State University Harnessing the Power in Nature The goal of scientific research is to understand nature. Once we understand the
ELECTROCHEMICAL COMPRESSION OF PRODUCT HYDROGEN FROM PEM ELECTROLYZER STACK
 ELECTROCHEMICAL COMPRESSION OF PRODUCT HYDROGEN FROM PEM ELECTROLYZER STACK N.V. Dale 1,*, C. Y. Biaku 1, M. D. Mann 1, H. Salehfar 2, A. J. Peters 2 Abstract The low volumetric energy density of hydrogen
ELECTROCHEMICAL COMPRESSION OF PRODUCT HYDROGEN FROM PEM ELECTROLYZER STACK N.V. Dale 1,*, C. Y. Biaku 1, M. D. Mann 1, H. Salehfar 2, A. J. Peters 2 Abstract The low volumetric energy density of hydrogen
Modeling the next battery generation: Lithium-sulfur and lithium-air cells
 Modeling the next battery generation: Lithium-sulfur and lithium-air cells D. N. Fronczek, T. Danner, B. Horstmann, Wolfgang G. Bessler German Aerospace Center (DLR) University Stuttgart (ITW) Helmholtz
Modeling the next battery generation: Lithium-sulfur and lithium-air cells D. N. Fronczek, T. Danner, B. Horstmann, Wolfgang G. Bessler German Aerospace Center (DLR) University Stuttgart (ITW) Helmholtz
i i ne. (1) i The potential difference, which is always defined to be the potential of the electrode minus the potential of the electrolyte, is ln( a
 We re going to calculate the open circuit voltage of two types of electrochemical system: polymer electrolyte membrane (PEM) fuel cells and lead-acid batteries. To do this, we re going to make use of two
We re going to calculate the open circuit voltage of two types of electrochemical system: polymer electrolyte membrane (PEM) fuel cells and lead-acid batteries. To do this, we re going to make use of two
Half-Cell, Steady-State Flow-Battery Experiments. Robert M. Darling and Mike L. Perry
 Half-Cell, Steady-State Flow-Battery Experiments Robert M. Darling and Mike L. Perry United Technologies Research Center, East Hartford, Connecticut, 06108, USA An experimental approach designed to separately
Half-Cell, Steady-State Flow-Battery Experiments Robert M. Darling and Mike L. Perry United Technologies Research Center, East Hartford, Connecticut, 06108, USA An experimental approach designed to separately
Porous Electrodes with Lower Impedance for Vanadium Redox Flow Batteries
 Korean Chem. Eng. Res., 53(5), 638-645 (2015) http://dx.doi.org/10.9713/kcer.2015.53.5.638 PISSN 0304-128X, EISSN 2233-9558 Porous Electrodes with Lower Impedance for Vanadium Redox Flow Batteries Su Mi
Korean Chem. Eng. Res., 53(5), 638-645 (2015) http://dx.doi.org/10.9713/kcer.2015.53.5.638 PISSN 0304-128X, EISSN 2233-9558 Porous Electrodes with Lower Impedance for Vanadium Redox Flow Batteries Su Mi
ELECTROCHEMISTRY OXIDATION-REDUCTION
 ELECTROCHEMISTRY Electrochemistry involves the relationship between electrical energy and chemical energy. OXIDATION-REDUCTION REACTIONS SPONTANEOUS REACTIONS Can extract electrical energy from these.
ELECTROCHEMISTRY Electrochemistry involves the relationship between electrical energy and chemical energy. OXIDATION-REDUCTION REACTIONS SPONTANEOUS REACTIONS Can extract electrical energy from these.
SYNTHESIS AND CHARACTERIZATION OF TITANIUM METAL CARBON NANO TUBES
 International Journal of Nanotechnology and Application (IJNA) ISSN(P): 2277-4777; ISSN(E): 2278-9391 Vol. 4, Issue 2, Apr 2014, 15-20 TJPRC Pvt. Ltd. SYNTHESIS AND CHARACTERIZATION OF TITANIUM METAL CARBON
International Journal of Nanotechnology and Application (IJNA) ISSN(P): 2277-4777; ISSN(E): 2278-9391 Vol. 4, Issue 2, Apr 2014, 15-20 TJPRC Pvt. Ltd. SYNTHESIS AND CHARACTERIZATION OF TITANIUM METAL CARBON
Chemistry 112 Name Exam III Form A Section November 13,
 Chemistry 112 Name Exam III Form A Section November 13, 2012 email IMPORTANT: On the scantron (answer sheet), you MUST clearly fill your name, your student number, section number, and test form (white
Chemistry 112 Name Exam III Form A Section November 13, 2012 email IMPORTANT: On the scantron (answer sheet), you MUST clearly fill your name, your student number, section number, and test form (white
Basic overall reaction for hydrogen powering
 Fuel Cell Basics Basic overall reaction for hydrogen powering 2H 2 + O 2 2H 2 O Hydrogen produces electrons, protons, heat and water PEMFC Anode reaction: H 2 2H + + 2e Cathode reaction: (½)O 2 + 2H +
Fuel Cell Basics Basic overall reaction for hydrogen powering 2H 2 + O 2 2H 2 O Hydrogen produces electrons, protons, heat and water PEMFC Anode reaction: H 2 2H + + 2e Cathode reaction: (½)O 2 + 2H +
CHEMISTRY 13 Electrochemistry Supplementary Problems
 1. When the redox equation CHEMISTRY 13 Electrochemistry Supplementary Problems MnO 4 (aq) + H + (aq) + H 3 AsO 3 (aq) Mn 2+ (aq) + H 3 AsO 4 (aq) + H 2 O(l) is properly balanced, the coefficients will
1. When the redox equation CHEMISTRY 13 Electrochemistry Supplementary Problems MnO 4 (aq) + H + (aq) + H 3 AsO 3 (aq) Mn 2+ (aq) + H 3 AsO 4 (aq) + H 2 O(l) is properly balanced, the coefficients will
The goal of this project is to enhance the power density and lowtemperature efficiency of solid oxide fuel cells (SOFC) manufactured by atomic layer
 Stanford University Michael Shandalov1, Shriram Ramanathan2, Changhyun Ko2 and Paul McIntyre1 1Department of Materials Science and Engineering, Stanford University 2Division of Engineering and Applied
Stanford University Michael Shandalov1, Shriram Ramanathan2, Changhyun Ko2 and Paul McIntyre1 1Department of Materials Science and Engineering, Stanford University 2Division of Engineering and Applied
Electrochemistry and battery technology Contents
 Electrochemistry and battery technology Contents Introduction Redox overview voltaic cells, electrolytic cells, fuel cells, Primary and secondary batteries. Other batteries; Construction, working and applications
Electrochemistry and battery technology Contents Introduction Redox overview voltaic cells, electrolytic cells, fuel cells, Primary and secondary batteries. Other batteries; Construction, working and applications
Chemistry 112 Name Exam III Form A Section April 2,
 Chemistry 112 Name Exam III Form A Section April 2, 2013 email IMPORTANT: On the scantron (answer sheet), you MUST clearly fill your name, your student number, section number, and test form (white cover
Chemistry 112 Name Exam III Form A Section April 2, 2013 email IMPORTANT: On the scantron (answer sheet), you MUST clearly fill your name, your student number, section number, and test form (white cover
Electronic Supplementary Material (ESI) for Chemical Communications This journal is The Royal Society of Chemistry 2011
 Supplementary Information for Selective adsorption toward toxic metal ions results in selective response: electrochemical studies on polypyrrole/reduced graphene oxide nanocomposite Experimental Section
Supplementary Information for Selective adsorption toward toxic metal ions results in selective response: electrochemical studies on polypyrrole/reduced graphene oxide nanocomposite Experimental Section
CHAPTER 4 CHEMICAL MODIFICATION OF ACTIVATED CARBON CLOTH FOR POTENTIAL USE AS ELECTRODES IN CAPACITIVE DEIONIZATION PROCESS
 CHAPTER 4 CHEMICAL MODIFICATION OF ACTIVATED CARBON CLOTH FOR POTENTIAL USE AS ELECTRODES IN CAPACITIVE DEIONIZATION PROCESS 4.1 INTRODUCTION Capacitive deionization (CDI) is one of the promising energy
CHAPTER 4 CHEMICAL MODIFICATION OF ACTIVATED CARBON CLOTH FOR POTENTIAL USE AS ELECTRODES IN CAPACITIVE DEIONIZATION PROCESS 4.1 INTRODUCTION Capacitive deionization (CDI) is one of the promising energy
electrodeposition is a special case of electrolysis where the result is deposition of solid material on an electrode surface.
 Electrochemical Methods Electrochemical Deposition is known as electrodeposition - see CHEM* 1050 - electrolysis electrodeposition is a special case of electrolysis where the result is deposition of solid
Electrochemical Methods Electrochemical Deposition is known as electrodeposition - see CHEM* 1050 - electrolysis electrodeposition is a special case of electrolysis where the result is deposition of solid
Introduction Fuel Cells Repetition
 Introduction Fuel Cells Repetition Fuel cell applications PEMFC PowerCell AB, (S1-S3) PEMFC,1-100 kw Toyota Mirai a Fuel Cell Car A look inside The hydrogen tank 1. Inside Layer of polymer closest to the
Introduction Fuel Cells Repetition Fuel cell applications PEMFC PowerCell AB, (S1-S3) PEMFC,1-100 kw Toyota Mirai a Fuel Cell Car A look inside The hydrogen tank 1. Inside Layer of polymer closest to the
Cyclic Voltammetry. Objective: To learn the basics of cyclic voltammetry with a well-behaved echem system
 Cyclic Voltammetry Objective: To learn the basics of cyclic voltammetry with a well-behaved echem system Introduction Cyclic voltammetry (CV) is a popular electroanalytical technique for its relative simplicity
Cyclic Voltammetry Objective: To learn the basics of cyclic voltammetry with a well-behaved echem system Introduction Cyclic voltammetry (CV) is a popular electroanalytical technique for its relative simplicity
Chemistry 1011 TOPIC TEXT REFERENCE. Electrochemistry. Masterton and Hurley Chapter 18. Chemistry 1011 Slot 5 1
 Chemistry 1011 TOPIC Electrochemistry TEXT REFERENCE Masterton and Hurley Chapter 18 Chemistry 1011 Slot 5 1 18.5 Electrolytic Cells YOU ARE EXPECTED TO BE ABLE TO: Construct a labelled diagram to show
Chemistry 1011 TOPIC Electrochemistry TEXT REFERENCE Masterton and Hurley Chapter 18 Chemistry 1011 Slot 5 1 18.5 Electrolytic Cells YOU ARE EXPECTED TO BE ABLE TO: Construct a labelled diagram to show
Lecture 12: Electroanalytical Chemistry (I)
 Lecture 12: Electroanalytical Chemistry (I) 1 Electrochemistry Electrochemical processes are oxidation-reduction reactions in which: Chemical energy of a spontaneous reaction is converted to electricity
Lecture 12: Electroanalytical Chemistry (I) 1 Electrochemistry Electrochemical processes are oxidation-reduction reactions in which: Chemical energy of a spontaneous reaction is converted to electricity
Capacity fade studies of Lithium Ion cells
 Capacity fade studies of Lithium Ion cells by Branko N. Popov, P.Ramadass, Bala S. Haran, Ralph E. White Center for Electrochemical Engineering, Department of Chemical Engineering, University of South
Capacity fade studies of Lithium Ion cells by Branko N. Popov, P.Ramadass, Bala S. Haran, Ralph E. White Center for Electrochemical Engineering, Department of Chemical Engineering, University of South
Current based methods
 Current based methods Amperometric and voltammetric sensors More significant influence on analytical parameters (sensitivity, selectivity, interferences elimination) kind of method, potential range, electrode
Current based methods Amperometric and voltammetric sensors More significant influence on analytical parameters (sensitivity, selectivity, interferences elimination) kind of method, potential range, electrode
Oxidation-Reduction Review. Electrochemistry. Oxidation-Reduction Reactions. Oxidation-Reduction Reactions. Sample Problem.
 1 Electrochemistry Oxidation-Reduction Review Topics Covered Oxidation-reduction reactions Balancing oxidationreduction equations Voltaic cells Cell EMF Spontaneity of redox reactions Batteries Electrolysis
1 Electrochemistry Oxidation-Reduction Review Topics Covered Oxidation-reduction reactions Balancing oxidationreduction equations Voltaic cells Cell EMF Spontaneity of redox reactions Batteries Electrolysis
Introduction. can be rewritten as follows: Oxidation reaction. H2 2H + +2e. Reduction reaction: F2+2e 2F. Overall Reaction H2+F2 2H + +2F
 Electrochemistry is the study of chemical processes that cause electrons to move. This movement of electrons is called electricity, which can be generated by movements of electrons from one element to
Electrochemistry is the study of chemical processes that cause electrons to move. This movement of electrons is called electricity, which can be generated by movements of electrons from one element to
Electrodeposited nickel hydroxide on nickel foam with ultrahigh. capacitance
 Electrodeposited nickel hydroxide on nickel foam with ultrahigh capacitance Guang-Wu Yang, Cai-Ling Xu* and Hu-Lin Li* College of Chemistry and Chemical Engineering, Lanzhou University, 73 (PR China) 1.
Electrodeposited nickel hydroxide on nickel foam with ultrahigh capacitance Guang-Wu Yang, Cai-Ling Xu* and Hu-Lin Li* College of Chemistry and Chemical Engineering, Lanzhou University, 73 (PR China) 1.
Solutions for Assignment-8
 Solutions for Assignment-8 Q1. The process of adding impurities to a pure semiconductor is called: [1] (a) Mixing (b) Doping (c) Diffusing (d) None of the above In semiconductor production, doping intentionally
Solutions for Assignment-8 Q1. The process of adding impurities to a pure semiconductor is called: [1] (a) Mixing (b) Doping (c) Diffusing (d) None of the above In semiconductor production, doping intentionally
Lithium Batteries. Rechargeable batteries
 Lithium Batteries One of the main attractions of lithium as an anode material is its position as the most electronegative metal in the electrochemical series combined with its low density, thus offering
Lithium Batteries One of the main attractions of lithium as an anode material is its position as the most electronegative metal in the electrochemical series combined with its low density, thus offering
Electron Transfer Reactions
 ELECTROCHEMISTRY 1 Electron Transfer Reactions 2 Electron transfer reactions are oxidation- reduction or redox reactions. Results in the generation of an electric current (electricity) or be caused by
ELECTROCHEMISTRY 1 Electron Transfer Reactions 2 Electron transfer reactions are oxidation- reduction or redox reactions. Results in the generation of an electric current (electricity) or be caused by
Saltwater as the energy source for low-cost, safe rechargeable. batteries
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Saltwater as the energy source for
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Saltwater as the energy source for
Hybrid CFD and equivalent-circuit impedance modeling of solid oxide electrochemical cells
 Risø campus Hybrid CFD and equivalent-circuit impedance modeling of solid oxide electrochemical cells Valerio Novaresio, Christopher Graves, Henrik Lund Frandsen, Massimo Santarelli Valerio Novaresio 11/12/2013
Risø campus Hybrid CFD and equivalent-circuit impedance modeling of solid oxide electrochemical cells Valerio Novaresio, Christopher Graves, Henrik Lund Frandsen, Massimo Santarelli Valerio Novaresio 11/12/2013
Electroanalytical and Electrosynthetic Studies Carbonyl Derivatives
 84 85 CHARTER II AIM AND SCOPE The growing global concern in the chemical and allied industrial sector is maneuvering industrial waste management and pollution control. It is the commitment for the researchers
84 85 CHARTER II AIM AND SCOPE The growing global concern in the chemical and allied industrial sector is maneuvering industrial waste management and pollution control. It is the commitment for the researchers
Multiscale Materials Modeling At the University of Tennessee
 Multiscale Materials Modeling At the University of Tennessee David Keffer Dept. of Materials Science & Engineering The University of Tennessee Knoxville, TN 37996-2100 dkeffer@utk.edu http://clausius.engr.utk.edu/
Multiscale Materials Modeling At the University of Tennessee David Keffer Dept. of Materials Science & Engineering The University of Tennessee Knoxville, TN 37996-2100 dkeffer@utk.edu http://clausius.engr.utk.edu/
Nickel Sulfides Freestanding Holey Films as Air-Breathing Electrodes for. Flexible Zn-Air Batteries
 Nickel Sulfides Freestanding Holey Films as Air-Breathing Electrodes for Flexible Zn-Air Batteries Kyle Marcus, 1,# Kun Liang, 1,# Wenhan Niu, 1,# Yang Yang 1,* 1 NanoScience Technology Center, Department
Nickel Sulfides Freestanding Holey Films as Air-Breathing Electrodes for Flexible Zn-Air Batteries Kyle Marcus, 1,# Kun Liang, 1,# Wenhan Niu, 1,# Yang Yang 1,* 1 NanoScience Technology Center, Department
Modeling the Behaviour of a Polymer Electrolyte Membrane within a Fuel Cell Using COMSOL
 Modeling the Behaviour of a Polymer Electrolyte Membrane within a Fuel Cell Using COMSOL S. Beharry 1 1 University of the West Indies, St. Augustine, Trinidad and Tobago Abstract: In recent years, scientists
Modeling the Behaviour of a Polymer Electrolyte Membrane within a Fuel Cell Using COMSOL S. Beharry 1 1 University of the West Indies, St. Augustine, Trinidad and Tobago Abstract: In recent years, scientists
REFRACTORY METAL OXIDES: FABRICATION OF NANOSTRUCTURES, PROPERTIES AND APPLICATIONS
 REFRACTORY METAL OXIDES: FABRICATION OF NANOSTRUCTURES, PROPERTIES AND APPLICATIONS S.K. Lazarouk, D.A. Sasinovich BELARUSIAN STATE UNIVERSITY OF INFORMATICS AND RADIOELECTRONICS Outline: -- experimental
REFRACTORY METAL OXIDES: FABRICATION OF NANOSTRUCTURES, PROPERTIES AND APPLICATIONS S.K. Lazarouk, D.A. Sasinovich BELARUSIAN STATE UNIVERSITY OF INFORMATICS AND RADIOELECTRONICS Outline: -- experimental
PGM-free OER Catalysts for Proton Exchange Membrane Electrolyzer
 PGM-free OER Catalysts for Proton Exchange Membrane Electrolyzer Di-Jia Liu, Argonne National Laboratory November 14, 2017 HydroGEN Kick-Off Meeting, National Renewable Energy Laboratory HydroGEN Kick-Off
PGM-free OER Catalysts for Proton Exchange Membrane Electrolyzer Di-Jia Liu, Argonne National Laboratory November 14, 2017 HydroGEN Kick-Off Meeting, National Renewable Energy Laboratory HydroGEN Kick-Off
An Introduction to Ionic Liquids. Michael Freemantle. RSC Publishing
 An to Ionic Liquids Michael Freemantle RSC Publishing Chapter 1 1 1.1 Definition of Ionic Liquids 1 1.2 Synonyms 1 1.3 Attraction of Ionic Liquids 2 1.4 Cations and Anions 3 1.5 Shorthand Notation for
An to Ionic Liquids Michael Freemantle RSC Publishing Chapter 1 1 1.1 Definition of Ionic Liquids 1 1.2 Synonyms 1 1.3 Attraction of Ionic Liquids 2 1.4 Cations and Anions 3 1.5 Shorthand Notation for
Development of Bifunctional Electrodes for Closed-loop Fuel Cell Applications. Pfaffenwaldring 6, Stuttgart, Germany
 Development of Bifunctional Electrodes for Closed-loop Fuel Cell Applications S. Altmann a,b, T. Kaz b, K. A. Friedrich a,b a Institute of Thermodynamics and Thermal Engineering, University Stuttgart,
Development of Bifunctional Electrodes for Closed-loop Fuel Cell Applications S. Altmann a,b, T. Kaz b, K. A. Friedrich a,b a Institute of Thermodynamics and Thermal Engineering, University Stuttgart,
June 16 th, Charles Monroe, Levi Thompson, Alice Sleightholme, and Aaron Shinkle University of Michigan Department of Chemical Engineering
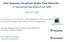 Non Aqueous Vanadium Redox Flow Batteries June 16 th, 2010 Charles Monroe, Levi Thompson, Alice Sleightholme, and Aaron Shinkle University of Michigan Department of Chemical Engineering Christian Doetsch,
Non Aqueous Vanadium Redox Flow Batteries June 16 th, 2010 Charles Monroe, Levi Thompson, Alice Sleightholme, and Aaron Shinkle University of Michigan Department of Chemical Engineering Christian Doetsch,
Cyclic Voltammetry. Fundamentals of cyclic voltammetry
 Cyclic Voltammetry Cyclic voltammetry is often the first experiment performed in an electrochemical study of a compound, biological material, or an electrode surface. The effectiveness of cv results from
Cyclic Voltammetry Cyclic voltammetry is often the first experiment performed in an electrochemical study of a compound, biological material, or an electrode surface. The effectiveness of cv results from
SEPARATION BY BARRIER
 SEPARATION BY BARRIER SEPARATION BY BARRIER Phase 1 Feed Barrier Phase 2 Separation by barrier uses a barrier which restricts and/or enhances the movement of certain chemical species with respect to other
SEPARATION BY BARRIER SEPARATION BY BARRIER Phase 1 Feed Barrier Phase 2 Separation by barrier uses a barrier which restricts and/or enhances the movement of certain chemical species with respect to other
Dry Cell: a galvanic cell with the electrolyte contained in a paste thickened by starch. anode and an inert graphite cathode.
 1 BATTERIES Text Pages: 764-766, 787,788 Battery: a set of galvanic cells connected in series - The negative electrode of one cell is connected to the positive electrode of the next cell - The total voltage
1 BATTERIES Text Pages: 764-766, 787,788 Battery: a set of galvanic cells connected in series - The negative electrode of one cell is connected to the positive electrode of the next cell - The total voltage
Redox Titration. Properties of Umass Boston
 Redox Titration Redox Titration Ce 4+ + Fe 2+ Ce 3+ + Fe 3+ Redox titration is based on the redox reaction (oxidation-reduction) between analyte and titrant. Position of the end point Determine the end
Redox Titration Redox Titration Ce 4+ + Fe 2+ Ce 3+ + Fe 3+ Redox titration is based on the redox reaction (oxidation-reduction) between analyte and titrant. Position of the end point Determine the end
Additional higher level topics
 Option C: Energy 15/25 hours 150 Essential idea: Chemical energy from redox reactions can be used as a portable source of electrical energy. C.6 Electrochemistry, rechargeable batteries and fuel cells
Option C: Energy 15/25 hours 150 Essential idea: Chemical energy from redox reactions can be used as a portable source of electrical energy. C.6 Electrochemistry, rechargeable batteries and fuel cells
18.3 Electrolysis. Dr. Fred Omega Garces. Chemistry 201. Driving a non-spontaneous Oxidation-Reduction Reaction. Miramar College.
 18.3 Electrolysis Driving a non-spontaneous Oxidation-Reduction Reaction Dr. Fred Omega Garces Chemistry 201 Miramar College 1 Electrolysis Voltaic Vs. Electrolytic Cells Voltaic Cell Energy is released
18.3 Electrolysis Driving a non-spontaneous Oxidation-Reduction Reaction Dr. Fred Omega Garces Chemistry 201 Miramar College 1 Electrolysis Voltaic Vs. Electrolytic Cells Voltaic Cell Energy is released
UNIT IV NON-CONVENTIONAL ENERGY SOURCES
 POLLACHI INSTITUTE OF ENGINEERING AND TECHNOLOGY POOSARIPATTI, POLLACHI 642 205 DEPARTMENT OF CHEMISTRY ENGINEERING CHEMISTRY I UNIT IV NON-CONVENTIONAL ENERGY SOURCES PART-A 1. Define nuclear fission?
POLLACHI INSTITUTE OF ENGINEERING AND TECHNOLOGY POOSARIPATTI, POLLACHI 642 205 DEPARTMENT OF CHEMISTRY ENGINEERING CHEMISTRY I UNIT IV NON-CONVENTIONAL ENERGY SOURCES PART-A 1. Define nuclear fission?
Review. Chapter 17 Electrochemistry. Outline. Voltaic Cells. Electrochemistry. Mnemonic
 Review William L Masterton Cecile N. Hurley Edward J. Neth cengage.com/chemistry/masterton Chapter 17 Electrochemistry Oxidation Loss of electrons Occurs at electrode called the anode Reduction Gain of
Review William L Masterton Cecile N. Hurley Edward J. Neth cengage.com/chemistry/masterton Chapter 17 Electrochemistry Oxidation Loss of electrons Occurs at electrode called the anode Reduction Gain of
Carbon nanotubes and conducting polymer composites
 University of Wollongong Thesis Collections University of Wollongong Thesis Collection University of Wollongong Year 4 Carbon nanotubes and conducting polymer composites May Tahhan University of Wollongong
University of Wollongong Thesis Collections University of Wollongong Thesis Collection University of Wollongong Year 4 Carbon nanotubes and conducting polymer composites May Tahhan University of Wollongong
Nanostructured materials for solar energy
 Nanostructured materials for solar energy Water Splitting & Dye Solar Cells Journée Scientifique des Comices «Energie Solaire» du WARE 23 avril 2012 à Jambes Prof. Rudi Cloots, C. Henrist, Contributors:
Nanostructured materials for solar energy Water Splitting & Dye Solar Cells Journée Scientifique des Comices «Energie Solaire» du WARE 23 avril 2012 à Jambes Prof. Rudi Cloots, C. Henrist, Contributors:
Performance Analysis of a Two phase Non-isothermal PEM Fuel Cell
 Performance Analysis of a Two phase Non-isothermal PEM Fuel Cell A. H. Sadoughi 1 and A. Asnaghi 2 and M. J. Kermani 3 1, 2 Ms Student of Mechanical Engineering, Sharif University of Technology Tehran,
Performance Analysis of a Two phase Non-isothermal PEM Fuel Cell A. H. Sadoughi 1 and A. Asnaghi 2 and M. J. Kermani 3 1, 2 Ms Student of Mechanical Engineering, Sharif University of Technology Tehran,
Solar desalination coupled with water remediation and molecular hydrogen production: A novel solar water-energy nexus
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 17 Supporting Information Solar desalination coupled with water remediation and
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 17 Supporting Information Solar desalination coupled with water remediation and
Lecture 30 Chapter 19, Sections 3-4 Galvanic Cells Electrochemical Potential
 Lecture 30 Chapter 19, Sections 3-4 Galvanic Cells Electrochemical Potential Galvanic Cells Defined Standard Hydrogen Electrode Standard Reduction Potentials Redox Balancing One More Example OK, then here
Lecture 30 Chapter 19, Sections 3-4 Galvanic Cells Electrochemical Potential Galvanic Cells Defined Standard Hydrogen Electrode Standard Reduction Potentials Redox Balancing One More Example OK, then here
9.1 Introduction to Oxidation and Reduction
 9.1 Introduction to Oxidation and Reduction 9.1.1 - Define oxidation and reduction in terms of electron loss and gain Oxidation The loss of electrons from a substance. This may happen through the gain
9.1 Introduction to Oxidation and Reduction 9.1.1 - Define oxidation and reduction in terms of electron loss and gain Oxidation The loss of electrons from a substance. This may happen through the gain
MODELING THE BEHAVIOR OF A POLYMER ELECTROLYTE MEMBRANE WITHIN A FUEL CELL USING COMSOL
 MODELING THE BEHAVIOR OF A POLYMER ELECTROLYTE MEMBRANE WITHIN A FUEL CELL USING COMSOL PRESENTER: SUPERVISOR: STEFAN BEHARRY DR. DAVINDER PAL SHARMA (PHYSICS DEPARTMENT, UWI) Excerpt from the Proceedings
MODELING THE BEHAVIOR OF A POLYMER ELECTROLYTE MEMBRANE WITHIN A FUEL CELL USING COMSOL PRESENTER: SUPERVISOR: STEFAN BEHARRY DR. DAVINDER PAL SHARMA (PHYSICS DEPARTMENT, UWI) Excerpt from the Proceedings
Instrumental Chemical Analysis. Dr. Abdul Muttaleb Jaber Professor Faculty of Pharmacy Philadelphia University Fall 2012/2013
 0510212 Instrumental Chemical Analysis Dr. Abdul Muttaleb Jaber Professor Faculty of Pharmacy Philadelphia University Fall 2012/2013 Chapter 1 Electroanalytical Methods Electroanalytical Chemistry Electroanalytical
0510212 Instrumental Chemical Analysis Dr. Abdul Muttaleb Jaber Professor Faculty of Pharmacy Philadelphia University Fall 2012/2013 Chapter 1 Electroanalytical Methods Electroanalytical Chemistry Electroanalytical
Electrochemistry. Review oxidation reactions and how to assign oxidation numbers (Ch 4 Chemical Reactions).
 Electrochemistry Oxidation-Reduction: Review oxidation reactions and how to assign oxidation numbers (Ch 4 Chemical Reactions). Half Reactions Method for Balancing Redox Equations: Acidic solutions: 1.
Electrochemistry Oxidation-Reduction: Review oxidation reactions and how to assign oxidation numbers (Ch 4 Chemical Reactions). Half Reactions Method for Balancing Redox Equations: Acidic solutions: 1.
Name: Regents Chemistry Date:
 Name: Date: 1. The reaction CuO + CO CO 2 + Cu is an example of (A) reduction, only (B) oxidation, only (C) both oxidation and reduction (D) neither oxidation nor reduction 6. In which compound does chlorine
Name: Date: 1. The reaction CuO + CO CO 2 + Cu is an example of (A) reduction, only (B) oxidation, only (C) both oxidation and reduction (D) neither oxidation nor reduction 6. In which compound does chlorine
Electrochemistry. Part One: Introduction to Electrolysis and the Electrolysis of Molten Salts
 Part One: Introduction to Electrolysis and the Electrolysis of Molten Salts What do I need to know about electrochemistry? Electrochemistry Learning Outcomes: Candidates should be able to: a) Describe
Part One: Introduction to Electrolysis and the Electrolysis of Molten Salts What do I need to know about electrochemistry? Electrochemistry Learning Outcomes: Candidates should be able to: a) Describe
Electrolytic membranes : Ion conduction in nanometric channels
 Electrolytic membranes : Ion conduction in nanometric channels Philippe Knauth Aix Marseille Université - CNRS Marseille, France Philippe Knauth 1 utline of the Presentation 1. Introduction 2. Synthesis
Electrolytic membranes : Ion conduction in nanometric channels Philippe Knauth Aix Marseille Université - CNRS Marseille, France Philippe Knauth 1 utline of the Presentation 1. Introduction 2. Synthesis
Chemistry PhD Qualifying Exam Paper 1 Syllabus
 Chemistry PhD Qualifying Exam Paper 1 Syllabus Preface This document comprises all topics relevant for Paper 1 of the Ph.D. Qualifying Exam in Chemistry at Eastern Mediterranean University, in accordance
Chemistry PhD Qualifying Exam Paper 1 Syllabus Preface This document comprises all topics relevant for Paper 1 of the Ph.D. Qualifying Exam in Chemistry at Eastern Mediterranean University, in accordance
An Ideal Electrode Material, 3D Surface-Microporous Graphene for Supercapacitors with Ultrahigh Areal Capacitance
 Supporting Information An Ideal Electrode Material, 3D Surface-Microporous Graphene for Supercapacitors with Ultrahigh Areal Capacitance Liang Chang, 1 Dario J. Stacchiola 2 and Yun Hang Hu 1, * 1. Department
Supporting Information An Ideal Electrode Material, 3D Surface-Microporous Graphene for Supercapacitors with Ultrahigh Areal Capacitance Liang Chang, 1 Dario J. Stacchiola 2 and Yun Hang Hu 1, * 1. Department
8. Draw Lewis structures and determine molecular geometry based on VSEPR Theory
 Chemistry Grade 12 Outcomes 1 Quantum Chemistry and Atomic Structure Unit I 1. Perform calculations on wavelength, frequency and energy. 2. Have an understanding of the electromagnetic spectrum. 3. Relate
Chemistry Grade 12 Outcomes 1 Quantum Chemistry and Atomic Structure Unit I 1. Perform calculations on wavelength, frequency and energy. 2. Have an understanding of the electromagnetic spectrum. 3. Relate
Fernando O. Raineri. Office Hours: MWF 9:30-10:30 AM Room 519 Tue. 3:00-5:00 CLC (lobby).
 Fernando O. Raineri Office Hours: MWF 9:30-10:30 AM Room 519 Tue. 3:00-5:00 CLC (lobby). P1) What is the reduction potential of the hydrogen electrode g bar H O aq Pt(s) H,1 2 3 when the aqueous solution
Fernando O. Raineri Office Hours: MWF 9:30-10:30 AM Room 519 Tue. 3:00-5:00 CLC (lobby). P1) What is the reduction potential of the hydrogen electrode g bar H O aq Pt(s) H,1 2 3 when the aqueous solution
Uncovering the role of flow rate in redox-active polymer flow batteries: simulation of. reaction distributions with simultaneous mixing in tanks
 Uncovering the role of flow rate in redox-active polymer flow batteries: simulation of reaction distributions with simultaneous mixing in tanks V. Pavan Nemani a,d and Kyle C. Smith a,b,c,d * a Department
Uncovering the role of flow rate in redox-active polymer flow batteries: simulation of reaction distributions with simultaneous mixing in tanks V. Pavan Nemani a,d and Kyle C. Smith a,b,c,d * a Department
CARBON. Electrochemical ond Physicochemicol Properties KIM KINOSHITA. Lawrence Berkeley Laboratory Berkeley, California
 CARBON Electrochemical ond Physicochemicol Properties KIM KINOSHITA Lawrence Berkeley Laboratory Berkeley, California A Wiley-Interscience Publicotion JOHN WILEY & SONS New York / Chichester / Brisbane
CARBON Electrochemical ond Physicochemicol Properties KIM KINOSHITA Lawrence Berkeley Laboratory Berkeley, California A Wiley-Interscience Publicotion JOHN WILEY & SONS New York / Chichester / Brisbane
