MECHANISMS. Croomine. Key reaction is the vinylogous Mannich reaction. (CH 2 ) 4 Br H N P. CO 2 Me. Iminium ion formation via decarboxylation
|
|
|
- Wilfrid Sullivan
- 5 years ago
- Views:
Transcription
1 MECAM Croomine Key reaction is the vinylogous Mannich reaction T C 2 Me T C 2 Me (C 2 ) 4 C 2 Me minium ion formation via decarboxylation C 2 Cl 3 Cl ndanomycin The Julia lefination Classical Julia Ar = h ' 2 Ar " 73TL4833; (M.Julia) C a / g ' 2 Ar Li + " sterics normally control " ' trans predominates mproved Julia Ar = ' 2 Li " 2 91TL1175 (. Julia) Allylic bromination n 2' driving force = formation Gareth owlands (g.rowlands@sussex.ac.uk) Ar402, etrosynthesis
2 MECAM F-ing Altohyrtin A oxymercuration with oxidative removal g(ac) 2 gl n 2 gl n gln mercury activates alkene to nucleophilic attack hydride source initiates radical decomposition ab 4 g g regioselective epoxide opening Bn Ti(ir) 4 δ+ Ti (ir) 3 h n 2 attack with inversion activation of the 3 position via chelation (ir) 3 Ti Ch Ch crotyl group prefers to be far from methyl substituents own Crotylation B vs B favoured interaction between B-substituents Gareth owlands (g.rowlands@sussex.ac.uk) Ar402, etrosynthesis
3 MECAM Epothilone A uzkui coupling " reductive elimination d(0) - oxidative addition " d L 2 d L 2 '-BY 2 a' " BY 2 d L 2 ' a enolate geometry controls syn or anti Z-enolate formed on steric grounds apamycin Evan's asymmetric aldol ' ' " B Bu B Bu " " Bu Bu ' h h h aldehyde approach controlled by geometry auxiliary alignment due to dipoles? ozaki-takai-iyama-kishi reaction Me + " TB CrCl 2, icl 2 (cat.) Me TB ' " catalytic reduces nickel i() + 2 Cr() i(0) X i()x Cr() transmetalltion Cr() 2 Cr() oxidative addition ' ' Gareth owlands (g.rowlands@sussex.ac.uk) Ar402, etrosynthesis
4 MECAM V Epoxydictymene Favourski rearrangement only one isomer; base equilibration Me C 2 Me ame icholas reaction 1. Co 2 (C) 8, Et 2 (C) 3 Co Co(C)3 TM Et cobalt complex can be used as alkyne protecting group here serves 3 purposes facilitates icholas cation formation, allowing neutral nucleophile to attack bends 'alkyne' so that reactive centres are in proximity facilitates the auson-khand reaction Et TM 1. TMTf, Et 2 reaction sets up taxing 8-ring sets up auson-khand precursor (C) 3 Co Co(C)3 (C) 3 Co TM Co(C) 3 Et lost in preference to allyl ether possibly because it is less sterically hindered so readily coordinates the Lewis acid possibly because the tertiary alkoxy group is more electron rich and encourages oxonoim ion formation no conclusion as mechanism very variable Gareth owlands (g.rowlands@sussex.ac.uk) Ar402, etrosynthesis
5 MECAM V Epoxydictymene auson-khand eaction L Co 2 (C) 8 (C) 3 Co (C) 3 Co L - C (C) 3 Co (C) 2 Co L insertion controls regiochemistry with substituents staying apart + C (C) 4 Co L Co(C) 3 reductive elimination (C) 3 Co + C (C) 3 Co L + C alkyl migration (or C insertion) (C) 3 Co (C) 3 Co L - Co 2 (C) 7 decomplexation L tandard Conditions: C under atmosphere of C luggish and very messy (low yield) Modern Conditions: Acclererated by -oxides (faster, milder, better yields) Also catalytic variants ecently asymmetric version eason for -oxide Acceleration M C Me 3 M Me 3 M + C 2 + Me 3 removes C from metal thus frees up coordination centre auson-khand eaction: Comp. rg. yn. Vol 5, Ch 9.1, p Accelerating Methods: ynthesis 1989, 472; JMC 1988, 354, 233; Tet. Lett. 1990, Catalytic: JAC 1994, Asymmetric: Angew. Chem. nt. Ed. 1998, 911. Gareth owlands (g.rowlands@sussex.ac.uk) Ar402, etrosynthesis
6 transmetallation or ET MECAM V Erythronolide B β-elimination Zn Zn sufficiently good leaving group Claisen rearrangement boat-like transition state TM TM LDA, TMCl C 2 Altohyrtin A Julia Methylenation 2 h ' Mg BuLi 2 h Mg ' ' "one-pot" bisalkylation TB tbume 2 i ' TB ' TB Gareth owlands (g.rowlands@sussex.ac.uk) Ar402, etrosynthesis
7 MECAM V kadiac acid spiroketalisation most thermodynamically stable complimentary anomeric effects Me Ac ' ' spiroketalisation ' Me ()h 2 C most thermodynamically stable complimentary anomeric effects Me Bn Ar LDA, tbuk Me Me Bn Ar deprotects BDA & forms oxonium ion TFA Bn Ar Bn Ar spiroketalisation h 2 BuLi h 2 cleaves protecting group and forms oxonium species spontaneous antiperiplanar elimination CA, Me most thermodynamically stable complimentary anomeric effects Gareth owlands (g.rowlands@sussex.ac.uk) Ar402, etrosynthesis
8 MECAM V ( )-tenine ossible explanation for the selectivity of the MDA conformation fixed by chelation and sterics Al Al MB(C 2 ) 3 endo selective h MB(C 2 ) 3 auxiliary blocks one face h MB(C 2 ) 4 * Curtius rearrangement (h) 2 3 Me 3 (h) 2 migration with retension Gareth owlands (g.rowlands@sussex.ac.uk) Ar402, etrosynthesis
Chiral Catalyst II. Palladium Catalysed Allylic Displacement ( -allyl complexes) 1. L n Pd(0) 2. Nuc
 Chiral Catalyst II ast lecture we looked at asymmetric catalysis for oxidation and reduction Many other organic transformations, this has led to much investigation Today we will look at some others...
Chiral Catalyst II ast lecture we looked at asymmetric catalysis for oxidation and reduction Many other organic transformations, this has led to much investigation Today we will look at some others...
[3,3]-sigmatropic Processes. [2,3]-sigmatropic Processes. Ene Reactions. Generalized Sigmatropic Processes X,Y=C, N, O, S X,Y=C, N, O, S
![[3,3]-sigmatropic Processes. [2,3]-sigmatropic Processes. Ene Reactions. Generalized Sigmatropic Processes X,Y=C, N, O, S X,Y=C, N, O, S [3,3]-sigmatropic Processes. [2,3]-sigmatropic Processes. Ene Reactions. Generalized Sigmatropic Processes X,Y=C, N, O, S X,Y=C, N, O, S](/thumbs/94/119475672.jpg) Generalized igmatropic Processes [3,3]-sigmatropic Processes 1 3,=C,,, 1 3 3,=C,,, 3 [2,3]-sigmatropic Processes 1 3,=C,,, 1 3 Ene eactions 1 3 1 3 Cope earrangement [3,3]- igmatropic earrangements Transition
Generalized igmatropic Processes [3,3]-sigmatropic Processes 1 3,=C,,, 1 3 3,=C,,, 3 [2,3]-sigmatropic Processes 1 3,=C,,, 1 3 Ene eactions 1 3 1 3 Cope earrangement [3,3]- igmatropic earrangements Transition
Chiral Auxiliaries. attach auxiliary Substrate Substrate Auxiliary
 Chiral Auxiliaries Previously on Advanced ynthesis... Discussed the need for stereoselective synthesis Looked at the use of resolution, the chiral pool and substrate control t there are some potential
Chiral Auxiliaries Previously on Advanced ynthesis... Discussed the need for stereoselective synthesis Looked at the use of resolution, the chiral pool and substrate control t there are some potential
Carbenes and Carbene Complexes I Introduction
 Carbenes and Carbene Complexes I Introduction A very interesting (honest) class of radical-like molecules Steadily becoming more important as they find far more synthetic applications We will primarily
Carbenes and Carbene Complexes I Introduction A very interesting (honest) class of radical-like molecules Steadily becoming more important as they find far more synthetic applications We will primarily
Guideline 5: Tactical Bonds
 Guideline 5: Tactical Bonds f the molecule contains more than one functional group and hence there is a choice of which bonds to disconnect, how do you decide? Practice, but here are a few rough guidelines
Guideline 5: Tactical Bonds f the molecule contains more than one functional group and hence there is a choice of which bonds to disconnect, how do you decide? Practice, but here are a few rough guidelines
1. Radical Substitution on Alkanes. 2. Radical Substitution with Alkenes. 3. Electrophilic Addition
 1. Radical Substitution on Alkanes Only Cl and Br are useful at the laboratory level. Alkane reactivity: tertiary > secondary > primary > methyl Numbers below products give their relative yield. Relative
1. Radical Substitution on Alkanes Only Cl and Br are useful at the laboratory level. Alkane reactivity: tertiary > secondary > primary > methyl Numbers below products give their relative yield. Relative
Total synthesis of Spongistatin
 Literature Semminar 1. Introduction: Total synthesis of Spongistatin Chen Zhihua (M2) Isolation: Pettit et al. J. rg. Chem. 1993, 58, 1302. Kitagawa et al. Tetrahedron Lett. 1993, 34, 1993. Fusetani et
Literature Semminar 1. Introduction: Total synthesis of Spongistatin Chen Zhihua (M2) Isolation: Pettit et al. J. rg. Chem. 1993, 58, 1302. Kitagawa et al. Tetrahedron Lett. 1993, 34, 1993. Fusetani et
HYDROGENATION. Concerned with two forms of hydrogenation: heterogeneous (catalyst insoluble) and homogeneous (catalyst soluble)
 YDGEATI Concerned with two forms of hydrogenation: heterogeneous (catalyst insoluble) and homogeneous (catalyst soluble) eterogeneous Catalysis Catalyst insoluble in reaction medium eactions take place
YDGEATI Concerned with two forms of hydrogenation: heterogeneous (catalyst insoluble) and homogeneous (catalyst soluble) eterogeneous Catalysis Catalyst insoluble in reaction medium eactions take place
Additions to Metal-Alkene and -Alkyne Complexes
 Additions to tal-alkene and -Alkyne Complexes ecal that alkenes, alkynes and other π-systems can be excellent ligands for transition metals. As a consequence of this binding, the nature of the π-system
Additions to tal-alkene and -Alkyne Complexes ecal that alkenes, alkynes and other π-systems can be excellent ligands for transition metals. As a consequence of this binding, the nature of the π-system
Suggested solutions for Chapter 27
 uggested solutions for Chapter 27 27 PRBLEM 1 uggest a mechanism for this reaction, commenting on the selectivity and the stereochemistry. Me 2 1. t-buk 2. Raney Ni Et The opportunity to explore the consequences
uggested solutions for Chapter 27 27 PRBLEM 1 uggest a mechanism for this reaction, commenting on the selectivity and the stereochemistry. Me 2 1. t-buk 2. Raney Ni Et The opportunity to explore the consequences
Organocopper Reagents
 rganocopper eagents General Information!!! why organocopper reagents? - Efficient method of C-C bond formation - Cu less electropositive than Li or Mg, so -Cu bond less polarized - consequences: 1. how
rganocopper eagents General Information!!! why organocopper reagents? - Efficient method of C-C bond formation - Cu less electropositive than Li or Mg, so -Cu bond less polarized - consequences: 1. how
Advanced Organic Chemistry
 D. A. Evans, G. Lalic Question of the day: Chemistry 530A TBS Me 2 C Me toluene, 130 C 70% TBS C 2 Me H H Advanced rganic Chemistry Me Lecture 16 Cycloaddition Reactions Diels _ Alder Reaction Photochemical
D. A. Evans, G. Lalic Question of the day: Chemistry 530A TBS Me 2 C Me toluene, 130 C 70% TBS C 2 Me H H Advanced rganic Chemistry Me Lecture 16 Cycloaddition Reactions Diels _ Alder Reaction Photochemical
Stereoselective reactions of enolates
 1 Stereoselective reactions of enolates Chiral auxiliaries are frequently used to allow diastereoselective enolate reactions Possibly the most extensively studied are the Evan s oxazolidinones These are
1 Stereoselective reactions of enolates Chiral auxiliaries are frequently used to allow diastereoselective enolate reactions Possibly the most extensively studied are the Evan s oxazolidinones These are
Chiral Catalysis. Chiral Catalyst. Substrate. Chiral Catalyst
 Chiral Catalysis Chiral (stoichiometric) reagents are a very important class of compound but... eed a stoichiometric quantity of the chiral component Unless it is cheap or recoverable this is not very
Chiral Catalysis Chiral (stoichiometric) reagents are a very important class of compound but... eed a stoichiometric quantity of the chiral component Unless it is cheap or recoverable this is not very
Stereoselective reactions of the carbonyl group
 1 Stereoselective reactions of the carbonyl group We have seen many examples of substrate control in nucleophilic addition to the carbonyl group (Felkin-Ahn & chelation control) If molecule does not contain
1 Stereoselective reactions of the carbonyl group We have seen many examples of substrate control in nucleophilic addition to the carbonyl group (Felkin-Ahn & chelation control) If molecule does not contain
Chapter 4. Reactions of alkenes. Addition reactions Carbocations Selectivity of reactions
 Chapter 4 Reactions of alkenes Addition reactions Carbocations Selectivity of reactions Prob 47 p192. Give the reagents that would be required (including catalyst). Ch 4 #2 Electrophilic addition Ch 4
Chapter 4 Reactions of alkenes Addition reactions Carbocations Selectivity of reactions Prob 47 p192. Give the reagents that would be required (including catalyst). Ch 4 #2 Electrophilic addition Ch 4
Preparation of alkenes
 Lecture 11 אלקנים הכנה ותגובות של אלקנים: הידרוגנציה, סיפוח הידרוהלוגנים )כלל מארקובניקוב(, סיפוח הלוגנים והסטראוכימיה של תוצרי הסיפוח, הידרובורציה, אפוקסידציה, אוזונוליזה. 1 Preparation of alkenes 1.
Lecture 11 אלקנים הכנה ותגובות של אלקנים: הידרוגנציה, סיפוח הידרוהלוגנים )כלל מארקובניקוב(, סיפוח הלוגנים והסטראוכימיה של תוצרי הסיפוח, הידרובורציה, אפוקסידציה, אוזונוליזה. 1 Preparation of alkenes 1.
Chem 251 Fall Learning Objectives
 Learning Objectives Chapter 8 (last semester) 1. Write an electron-pushing mechanism for an SN2 reaction between an alkyl halide and a nucleophile. 2. Describe the rate law and relative rate of reaction
Learning Objectives Chapter 8 (last semester) 1. Write an electron-pushing mechanism for an SN2 reaction between an alkyl halide and a nucleophile. 2. Describe the rate law and relative rate of reaction
Highlights of Schmidt Reaction in the Last Ten Years
 ighlights of Schmidt eaction in the Last Ten Years Dendrobates histrionicus Jack Liu ov. 18, 2003 Introduction Classical Schmidt reaction of aldehydes and carboxylic acids Classical Schmidt reaction of
ighlights of Schmidt eaction in the Last Ten Years Dendrobates histrionicus Jack Liu ov. 18, 2003 Introduction Classical Schmidt reaction of aldehydes and carboxylic acids Classical Schmidt reaction of
Suggested solutions for Chapter 40
 s for Chapter 40 40 PBLEM 1 Suggest mechanisms for these reactions, explaining the role of palladium in the first step. Ac Et Et BS () 4 2 1. 2. K 2 C 3 evision of enol ethers and bromination, the Wittig
s for Chapter 40 40 PBLEM 1 Suggest mechanisms for these reactions, explaining the role of palladium in the first step. Ac Et Et BS () 4 2 1. 2. K 2 C 3 evision of enol ethers and bromination, the Wittig
Shi Asymmetric Epoxidation
 Shi Asymmetric Epoxidation Chiral dioxirane strategy: R 3 + 1 xone, ph 10.5, K 2 C 3, H 2, C R 3 formed in situ catalyst (10-20 mol%) is prepared from D-fructose, and its enantiomer from L-sorbose oxone,
Shi Asymmetric Epoxidation Chiral dioxirane strategy: R 3 + 1 xone, ph 10.5, K 2 C 3, H 2, C R 3 formed in situ catalyst (10-20 mol%) is prepared from D-fructose, and its enantiomer from L-sorbose oxone,
π-alkyne metal complex and vinylidene metal complex in organic synthesis
 Literature Seminar 080220 Kenzo YAMATSUGU (D1) π-alkyne metal complex and vinylidene metal complex in organic synthesis 0. Introduction ' ' = π-alkyne metal complex vinylidene metal complex ecently, electrophilic
Literature Seminar 080220 Kenzo YAMATSUGU (D1) π-alkyne metal complex and vinylidene metal complex in organic synthesis 0. Introduction ' ' = π-alkyne metal complex vinylidene metal complex ecently, electrophilic
CHEM 330. Final Exam December 5, 2014 ANSWERS. This a closed-notes, closed-book exam. The use of molecular models is allowed
 CEM 330 Final Exam December 5, 2014 Your name: ASWERS This a closed-notes, closed-book exam The use of molecular models is allowed This exam consists of 12 pages Time: 2h 30 min 1. / 30 2. / 30 3. / 30
CEM 330 Final Exam December 5, 2014 Your name: ASWERS This a closed-notes, closed-book exam The use of molecular models is allowed This exam consists of 12 pages Time: 2h 30 min 1. / 30 2. / 30 3. / 30
Tips for taking exams in 852
 Comprehensive Tactical Methods in rganic Synthesis W. D. Wulff 1) Know the relative reactivity of carbonyl compounds Tips for taking exams in 852 Cl > > ' > > ' N2 eg: 'Mg Et ' 1equiv. 1equiv. ' ' Et 50%
Comprehensive Tactical Methods in rganic Synthesis W. D. Wulff 1) Know the relative reactivity of carbonyl compounds Tips for taking exams in 852 Cl > > ' > > ' N2 eg: 'Mg Et ' 1equiv. 1equiv. ' ' Et 50%
REACTION AND SYNTHESIS REVIEW
 REACTION AND SYNTHESIS REVIEW A STUDENT SHOULD BE ABLE TO PREDICT PRODUCTS, IDENTIFY REACTANTS, GIVE REACTION CONDITIONS, PROPOSE SYNTHESES, AND PROPOSE MECHANISMS (AS LISTED BELOW). REVIEW THE MECHANISM
REACTION AND SYNTHESIS REVIEW A STUDENT SHOULD BE ABLE TO PREDICT PRODUCTS, IDENTIFY REACTANTS, GIVE REACTION CONDITIONS, PROPOSE SYNTHESES, AND PROPOSE MECHANISMS (AS LISTED BELOW). REVIEW THE MECHANISM
Chapter 8 Alkenes and Alkynes II: Addition Reactions. Alkenes are electron rich. Additions to Alkenes
 Additions to Alkenes Chapter 8 Alkenes and Alkynes II: Addition Reactions Generally the reaction is exothermic because one p and one s bond are converted to two s bonds Alkenes are electron rich The carbocation
Additions to Alkenes Chapter 8 Alkenes and Alkynes II: Addition Reactions Generally the reaction is exothermic because one p and one s bond are converted to two s bonds Alkenes are electron rich The carbocation
ASPECTS OF ORGANIC SYNTHESIS STRATEGY / RETROSYNTHESIS OR
 ASPECTS F GAIC SYTESIS STATEGY / ETSYTESIS P P' C C ( )- -kainic acid P P' PhS Cl S C Me P 97SL75 P' P C tbu P P' 9JC6968 TBS EtS C tbu EtS TBS C C tbu 98P&A59 Course outline: rganic Synthesis what is
ASPECTS F GAIC SYTESIS STATEGY / ETSYTESIS P P' C C ( )- -kainic acid P P' PhS Cl S C Me P 97SL75 P' P C tbu P P' 9JC6968 TBS EtS C tbu EtS TBS C C tbu 98P&A59 Course outline: rganic Synthesis what is
Reactivity in Organic Chemistry. Mid term test October 31 st 2011, 9:30-12:30. Problem 1 (25p)
 eactivity in rganic Chemistry Mid term test ctober 31 st 2011, 9:30-12:30 Problem 1 (25p) - (+)- Limonone can be epoxidized (using peracetic acid) to give an inseparable mixture of two diastereomeric limonene
eactivity in rganic Chemistry Mid term test ctober 31 st 2011, 9:30-12:30 Problem 1 (25p) - (+)- Limonone can be epoxidized (using peracetic acid) to give an inseparable mixture of two diastereomeric limonene
Organometallic Study Meeting Chapter 17. Catalytic Carbonylation
 rganometallic Study Meeting Chapter 17. Catalytic Carbonylation 17.1 verview C or 3 3 C 3 C C 3 horrcat. Ar-X or alkene ' d cat. 2011/10/6 K.isaki or ' or N n 2 1 alkene, 2 Coorhcat. d cat. alkene C carbon
rganometallic Study Meeting Chapter 17. Catalytic Carbonylation 17.1 verview C or 3 3 C 3 C C 3 horrcat. Ar-X or alkene ' d cat. 2011/10/6 K.isaki or ' or N n 2 1 alkene, 2 Coorhcat. d cat. alkene C carbon
CuI CuI eage lic R tal ome rgan gbr ommon
 Common rganometallic eagents Li Et 2 Li Mg Et 2 Li alkyllithium rignard Mg Mg Li Zn TF ZnCl 2 TF dialkylzinc Zn 2 2 Zn Li CuI TF ganocuprate CuI 2 2 CuI common electrophile pairings ' Cl ' '' ' ' ' ' '
Common rganometallic eagents Li Et 2 Li Mg Et 2 Li alkyllithium rignard Mg Mg Li Zn TF ZnCl 2 TF dialkylzinc Zn 2 2 Zn Li CuI TF ganocuprate CuI 2 2 CuI common electrophile pairings ' Cl ' '' ' ' ' ' '
Asymmetric Catalysis by Lewis Acids and Amines
 Asymmetric Catalysis by Lewis Acids and Amines Asymmetric Lewis acid catalysis - Chiral (bisooxazoline) copper (II) complexes - Monodentate Lewis acids: the formyl -bond Amine catalysed reactions Asymmetric
Asymmetric Catalysis by Lewis Acids and Amines Asymmetric Lewis acid catalysis - Chiral (bisooxazoline) copper (II) complexes - Monodentate Lewis acids: the formyl -bond Amine catalysed reactions Asymmetric
Name: Student Number:
 Page 1 of 5 Name: Student Number: l University of Manitoba - Department of Chemistry CEM 2220 - Introductory rganic Chemistry II - Term Test 1 Thursday, February 14, 2008 This is a 2-hour test, marked
Page 1 of 5 Name: Student Number: l University of Manitoba - Department of Chemistry CEM 2220 - Introductory rganic Chemistry II - Term Test 1 Thursday, February 14, 2008 This is a 2-hour test, marked
Answers To Chapter 7 Problems.
 Answers To Chapter Problems.. Most of the Chapter problems appear as end-of-chapter problems in later chapters.. The first reaction is an ene reaction. When light shines on in the presence of light and
Answers To Chapter Problems.. Most of the Chapter problems appear as end-of-chapter problems in later chapters.. The first reaction is an ene reaction. When light shines on in the presence of light and
2. Reactions at Non-Anomeric Hydroxyl Groups
 2. Reactions at Non-Anomeric Hydroxyl Groups 2.1 Ether-Type Protecting Groups u Alkyl and aryl ethers are relatively stable to acids and bases due to the high C bond energy (358 KJ/mol) u most useful ether-type
2. Reactions at Non-Anomeric Hydroxyl Groups 2.1 Ether-Type Protecting Groups u Alkyl and aryl ethers are relatively stable to acids and bases due to the high C bond energy (358 KJ/mol) u most useful ether-type
Stereoselective reactions of enolates: auxiliaries
 1 Stereoselective reactions of enolates: auxiliaries Chiral auxiliaries are frequently used to allow diastereoselective enolate reactions Possibly the most extensively studied are the Evan s oxazolidinones
1 Stereoselective reactions of enolates: auxiliaries Chiral auxiliaries are frequently used to allow diastereoselective enolate reactions Possibly the most extensively studied are the Evan s oxazolidinones
Chapter 8 Alkenes and Alkynes II: Addition Reactions
 Chapter 8 Alkenes and Alkynes II: Addition Reactions Introduction: Additions to Alkenes Generally the reaction is exothermic because one π and one σ bond are converted to two σ bonds The π electrons of
Chapter 8 Alkenes and Alkynes II: Addition Reactions Introduction: Additions to Alkenes Generally the reaction is exothermic because one π and one σ bond are converted to two σ bonds The π electrons of
Alcohols, Ethers and Epoxides. Chapter Organic Chemistry, 8th Edition John McMurry
 Alcohols, Ethers and Epoxides Chapter 17-18 Organic Chemistry, 8th Edition John McMurry 1 Introduction Structure and Bonding Alcohols contain a hydroxy group (OH) bonded to an sp 3 hybridized carbon. 2
Alcohols, Ethers and Epoxides Chapter 17-18 Organic Chemistry, 8th Edition John McMurry 1 Introduction Structure and Bonding Alcohols contain a hydroxy group (OH) bonded to an sp 3 hybridized carbon. 2
Suggested solutions for Chapter 19
 s for Chapter 19 19 PRBLEM 1 Predict the orientation of addition to these alkenes. Simple examples of addition with regioselectivity. The first and last alkenes have different numbers of substituents at
s for Chapter 19 19 PRBLEM 1 Predict the orientation of addition to these alkenes. Simple examples of addition with regioselectivity. The first and last alkenes have different numbers of substituents at
CHEM 251 (4 credits): Description
 CHEM 251 (4 credits): Intermediate Reactions of Nucleophiles and Electrophiles (Reactivity 2) Description: An understanding of chemical reactivity, initiated in Reactivity 1, is further developed based
CHEM 251 (4 credits): Intermediate Reactions of Nucleophiles and Electrophiles (Reactivity 2) Description: An understanding of chemical reactivity, initiated in Reactivity 1, is further developed based
Exam 1 (Monday, July 6, 2015)
 Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
Stereoselective Organic Synthesis
 Stereoselective rganic Synthesis Prabhat Arya Professor and Leader, Chemical Biology Program Dean, Academic Affairs, Institute of Life Sciences (An Associate Institute of University of yderabad Supported
Stereoselective rganic Synthesis Prabhat Arya Professor and Leader, Chemical Biology Program Dean, Academic Affairs, Institute of Life Sciences (An Associate Institute of University of yderabad Supported
Radical Reactions. Radical Stability!!! bond dissociation energies X Y X + Y. bond BDE (kcal/mol) bond BDE (kcal/mol) CH 3 CH 3 CH 2 95 O H R 2 C H
 adical eactions adical Stability!!! bond dissociation energies X Y X Y bond BDE (kcal/mol) bond BDE (kcal/mol) C 3 104 108 C 3 C 2 98 110 95 2 C 102 (-) 93 (C-) 92 C 3 C 3 36 89 85 C 3 C 3 80 adical eactions
adical eactions adical Stability!!! bond dissociation energies X Y X Y bond BDE (kcal/mol) bond BDE (kcal/mol) C 3 104 108 C 3 C 2 98 110 95 2 C 102 (-) 93 (C-) 92 C 3 C 3 36 89 85 C 3 C 3 80 adical eactions
Denmark s Base Catalyzed Aldol/Allylation
 Denmark s Base Catalyzed Aldol/Allylation Evans Group Seminar ovember 1th, 003 Jimmy Wu Lead eferences: Denmark, S. E. Acc. Chem. es., 000, 33, 43 Denmark, S. E. Chem. Comm. 003, 167 Denmark, S. E. Chem.
Denmark s Base Catalyzed Aldol/Allylation Evans Group Seminar ovember 1th, 003 Jimmy Wu Lead eferences: Denmark, S. E. Acc. Chem. es., 000, 33, 43 Denmark, S. E. Chem. Comm. 003, 167 Denmark, S. E. Chem.
Only five of the molecules below may be prepared as the sole product of allylic halogenation of the respective alkene. Circle those five.
 Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 6 Answers Question 1. nly five of the molecules below may be prepared as the sole product of allylic halogenation of the respective alkene. Circle those
Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 6 Answers Question 1. nly five of the molecules below may be prepared as the sole product of allylic halogenation of the respective alkene. Circle those
Synthesis of Amines Amine Alkylation by SN2 reaction II. Reductive Amination
 Synthesis of Amines I. Amine Alkylation by SN2 reaction Amines can be alkylated in SN2 fashion by alkyl halides; primary halides are best for this purpose. This is not a practical reaction for formation
Synthesis of Amines I. Amine Alkylation by SN2 reaction Amines can be alkylated in SN2 fashion by alkyl halides; primary halides are best for this purpose. This is not a practical reaction for formation
Organometallic Chemistry and Homogeneous Catalysis
 rganometallic hemistry and omogeneous atalysis Dr. Alexey Zazybin Lecture N8 Kashiwa ampus, December 11, 2009 Types of reactions in the coordination sphere of T 3. Reductive elimination X-L n -Y L n +
rganometallic hemistry and omogeneous atalysis Dr. Alexey Zazybin Lecture N8 Kashiwa ampus, December 11, 2009 Types of reactions in the coordination sphere of T 3. Reductive elimination X-L n -Y L n +
Chem 315/316 Reactions. Bromo organic compounds C1 & C2 carbon skeletons C3 carbon skeletons C4 carbon skeletons. Alcohols. Chem 316 / Beauchamp 1
 hem 316 / Beauchamp 1 hem 315/316 eactions Name available sources of carbon 4 3 NaN methyl 3 2 ethyl 3 2 2 n-propyl 3 3 isopropyl 2 3 2 3 3 2 2 3 3 3 n-butyl sec butyl t-butyl phenyl available until topic
hem 316 / Beauchamp 1 hem 315/316 eactions Name available sources of carbon 4 3 NaN methyl 3 2 ethyl 3 2 2 n-propyl 3 3 isopropyl 2 3 2 3 3 2 2 3 3 3 n-butyl sec butyl t-butyl phenyl available until topic
Inorganic Chemistry Year 3
 Inorganic Chemistry Year 3 Transition Metal Catalysis Eighteen Electron Rule 1.Get the number of the group that the metal is in (this will be the number of d electrons) 2.Add to this the charge 1.Negative
Inorganic Chemistry Year 3 Transition Metal Catalysis Eighteen Electron Rule 1.Get the number of the group that the metal is in (this will be the number of d electrons) 2.Add to this the charge 1.Negative
ORGANOMETALLICS IN SYNTHESIS: CHROMIUM, IRON & COBALT REAGENTS
 - 1 - ORGANOMETALLICS IN SYNTHESIS: CHROMIUM, IRON & COBALT REAGENTS Introduction to Metal-Carbon Bonding Organometallic chemistry involves the interaction of an organic compound with a transition metal
- 1 - ORGANOMETALLICS IN SYNTHESIS: CHROMIUM, IRON & COBALT REAGENTS Introduction to Metal-Carbon Bonding Organometallic chemistry involves the interaction of an organic compound with a transition metal
Problem session (3) Daiki Kuwana. Please fill in the blank and explain reaction mechanisms and stereoselectivities.
 Problem session (3) Daiki Kuwana Please fill in the blank and explain reaction mechanisms and stereoselectivities. 1. 1-1 1. (Ac) 2 (10 mol%), DPEphos (20 mol%) Et 3, toluene, 90 C 2. s 4 (14 mol%), M;
Problem session (3) Daiki Kuwana Please fill in the blank and explain reaction mechanisms and stereoselectivities. 1. 1-1 1. (Ac) 2 (10 mol%), DPEphos (20 mol%) Et 3, toluene, 90 C 2. s 4 (14 mol%), M;
S N 1 Displacement Reactions
 S N 1 Displacement Reactions Tertiary alkyl halides cannot undergo S N 2 reactions because of the severe steric hindrance blocking a backside approach of the nucleophile. They can, however, react via an
S N 1 Displacement Reactions Tertiary alkyl halides cannot undergo S N 2 reactions because of the severe steric hindrance blocking a backside approach of the nucleophile. They can, however, react via an
Alcohol Synthesis. Dr. Sapna Gupta
 Alcohol Synthesis Dr. Sapna Gupta Synthesis of Alcohols Alcohols can be synthesized from several functional groups. Nucleophilic substitution of O - on alkyl halide ydration of alkenes water in acid solution
Alcohol Synthesis Dr. Sapna Gupta Synthesis of Alcohols Alcohols can be synthesized from several functional groups. Nucleophilic substitution of O - on alkyl halide ydration of alkenes water in acid solution
LECTURE #13 Tues., Oct.18, Midterm exam: Tues.Oct.25 during class Ch.1, , 7.10, 2, Sections
 CEM 221 section 01 LECTURE #13 Tues., Oct.18, 2005 Midterm exam: Tues.Oct.25 during class Ch.1, 7.2-7.5, 7.10, 2, 3.1-3.5 ASSGNED READNGS: TODAY S CLASS: Sections 4.1 4.6 NEXT CLASS: rest of Ch.4 http://artsandscience.concordia.ca/facstaff/p-r/rogers
CEM 221 section 01 LECTURE #13 Tues., Oct.18, 2005 Midterm exam: Tues.Oct.25 during class Ch.1, 7.2-7.5, 7.10, 2, 3.1-3.5 ASSGNED READNGS: TODAY S CLASS: Sections 4.1 4.6 NEXT CLASS: rest of Ch.4 http://artsandscience.concordia.ca/facstaff/p-r/rogers
mcpba e.g. mcpba (major) Section 7: Oxidation of C=X bonds
 Section 7: xidation of C=X bonds Functional Group Interconversions - Lecture 6 7.1 Epoxidation of Alkenes Epoxides are VEY useful in synthesis - the strain of the three membered ring makes these cyclic
Section 7: xidation of C=X bonds Functional Group Interconversions - Lecture 6 7.1 Epoxidation of Alkenes Epoxides are VEY useful in synthesis - the strain of the three membered ring makes these cyclic
Basic Organic Chemistry Course code : CHEM (Pre-requisites : CHEM 11122)
 Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Renaud Group Exercise Set
 Renaud Group Exercise Set Prepared by ick Tappin 08/07/16 Spectroscopy 1. Deduce the structures for compounds A, B, and C C 6 10 3 A IR: 1745 and 1720 cm -1 13 C-MR: δ 208, 172, 51, 37, 32, and 27 ab 4
Renaud Group Exercise Set Prepared by ick Tappin 08/07/16 Spectroscopy 1. Deduce the structures for compounds A, B, and C C 6 10 3 A IR: 1745 and 1720 cm -1 13 C-MR: δ 208, 172, 51, 37, 32, and 27 ab 4
Double and Triple Bonds. The addition of an electrophile and a
 Chapter 11 Additions to Carbon-Carbon Double and Triple Bonds The addition of an electrophile and a nucleophile to a C-C C double or triple bonds 11.1 The General Mechanism Pi electrons of the double bond
Chapter 11 Additions to Carbon-Carbon Double and Triple Bonds The addition of an electrophile and a nucleophile to a C-C C double or triple bonds 11.1 The General Mechanism Pi electrons of the double bond
Aldol Reactions pka of a-h ~ 20
 Enolate Anions Chapter 17 Hydrogen on a carbons a to a carbonyl is unusually acidic The resulting anion is stabilized by resonance to the carbonyl Aldehydes and Ketones II Aldol Reactions pka of a-h ~
Enolate Anions Chapter 17 Hydrogen on a carbons a to a carbonyl is unusually acidic The resulting anion is stabilized by resonance to the carbonyl Aldehydes and Ketones II Aldol Reactions pka of a-h ~
Lecture 6: Transition-Metal Catalysed C-C Bond Formation
 Lecture 6: Transition-Metal Catalysed C-C Bond Formation (a) Asymmetric allylic substitution 1 u - d u (b) Asymmetric eck reaction 2 3 Ar- d (0) Ar 2 3 (c) Asymmetric olefin metathesis alladium π-allyl
Lecture 6: Transition-Metal Catalysed C-C Bond Formation (a) Asymmetric allylic substitution 1 u - d u (b) Asymmetric eck reaction 2 3 Ar- d (0) Ar 2 3 (c) Asymmetric olefin metathesis alladium π-allyl
Suggested solutions for Chapter 34
 s for Chapter 34 34 PRBLEM 1 Predict the structure of the product of this Diels- Alder reaction. C 2 +? 3 Si Can you deal with a moderately complicated Diels- Alder? The diene is electron- rich and will
s for Chapter 34 34 PRBLEM 1 Predict the structure of the product of this Diels- Alder reaction. C 2 +? 3 Si Can you deal with a moderately complicated Diels- Alder? The diene is electron- rich and will
Electrophilic Carbenes
 Electrophilic Carbenes The reaction of so-called stabilized diazo compounds with late transition metals produces a metal carbene intermediate that is electrophilic. The most common catalysts are Cu(I)
Electrophilic Carbenes The reaction of so-called stabilized diazo compounds with late transition metals produces a metal carbene intermediate that is electrophilic. The most common catalysts are Cu(I)
2/26/18. Practice Questions. Practice Questions B F. How many steps are there in this reaction?
 Practice Questions Practice Questions D B F C E A G How many steps are there in this reaction? 1 Practice Questions D B F C E A G What is the highest-energy transitions state? Practice Questions D B F
Practice Questions Practice Questions D B F C E A G How many steps are there in this reaction? 1 Practice Questions D B F C E A G What is the highest-energy transitions state? Practice Questions D B F
Synthetic possibilities Chem 315 Beauchamp 1
 Synthetic possibilities hem Beauchamp Propose reasonable syntheses f the following target molecules (TM-#). You can use the given starting materials and any typical ganic reagents studied in our course
Synthetic possibilities hem Beauchamp Propose reasonable syntheses f the following target molecules (TM-#). You can use the given starting materials and any typical ganic reagents studied in our course
ROADMAP FOR REACTIONS Chapter 6
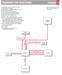 RADMAP FR REACTINS Chapter 6 (A) (B) (C) (D) (E) (F) (G) (H) (I) (J) (K) (L) (M) (N) () Regiochemistry: Markovnikov addition to a bond Stereochemistry: anti-addition Regiochemistry: non-markovnikov addition
RADMAP FR REACTINS Chapter 6 (A) (B) (C) (D) (E) (F) (G) (H) (I) (J) (K) (L) (M) (N) () Regiochemistry: Markovnikov addition to a bond Stereochemistry: anti-addition Regiochemistry: non-markovnikov addition
VI. Metal alkyls from oxidative addition / insertion
 V. Metal alkyls from oxidative addition / insertion A. Carbonylation - C insertion very facile, metal acyls easily cleaved, all substrates which undergo oxidative addition can in principle be carbonylated.
V. Metal alkyls from oxidative addition / insertion A. Carbonylation - C insertion very facile, metal acyls easily cleaved, all substrates which undergo oxidative addition can in principle be carbonylated.
Background Information
 ackground nformation ntroduction to Condensation eactions Condensation reactions occur between the α-carbon of one carbonyl-containing functional group and the carbonyl carbon of a second carbonyl-containing
ackground nformation ntroduction to Condensation eactions Condensation reactions occur between the α-carbon of one carbonyl-containing functional group and the carbonyl carbon of a second carbonyl-containing
Molecular Rearrangements
 Ø Migration of one the molecule group from one atom to another within Ø Generally the migrating group never leaves the molecule Ø There are five types of skeletal rearrangements- 1. Electron deficient
Ø Migration of one the molecule group from one atom to another within Ø Generally the migrating group never leaves the molecule Ø There are five types of skeletal rearrangements- 1. Electron deficient
A Simple Introduction of the Mizoroki-Heck Reaction
 A Simple Introduction of the Mizoroki-Heck Reaction Reporter: Supervisor: Zhe Niu Prof. Yang Prof. Chen Prof. Tang 2016/2/3 Content Introduction Intermolecular Mizoroki-Heck Reaction Intramolecular Mizoroki-Heck
A Simple Introduction of the Mizoroki-Heck Reaction Reporter: Supervisor: Zhe Niu Prof. Yang Prof. Chen Prof. Tang 2016/2/3 Content Introduction Intermolecular Mizoroki-Heck Reaction Intramolecular Mizoroki-Heck
1. Addition of HBr to alkenes
 eactions of Alkenes I eading: Wade chapter 8, sections 8-1- 8-8 tudy Problems: 8-47, 8-48, 8-55, 8-66, 8-67, 8-70 Key Concepts and kills: Predict the products of additions to alkenes, including regiochemistry
eactions of Alkenes I eading: Wade chapter 8, sections 8-1- 8-8 tudy Problems: 8-47, 8-48, 8-55, 8-66, 8-67, 8-70 Key Concepts and kills: Predict the products of additions to alkenes, including regiochemistry
Suggested solutions for Chapter 28
 s for Chapter 28 28 PBLEM 1 ow would you make these four compounds? Give your disconnections, explain why you chose them and then give reagents for the. 2 2 Me S Exercises in basic one- group C X disconnections.
s for Chapter 28 28 PBLEM 1 ow would you make these four compounds? Give your disconnections, explain why you chose them and then give reagents for the. 2 2 Me S Exercises in basic one- group C X disconnections.
Enols and Enolates. A type of reaction with carbonyl compounds is an α-substitution (an electrophile adds to the α carbon of a carbonyl)
 Enols and Enolates A type of reaction with carbonyl compounds is an α-substitution (an electrophile adds to the α carbon of a carbonyl) E+ E In the preceding chapters, we primarily studied nucleophiles
Enols and Enolates A type of reaction with carbonyl compounds is an α-substitution (an electrophile adds to the α carbon of a carbonyl) E+ E In the preceding chapters, we primarily studied nucleophiles
UNIVERSITY OF MANITOBA DEPARTMENT OF CHEMISTRY
 PAGE 1 of 11 UNVESTY F MANTBA DEPATMENT F CEMSTY 2.339 STUCTUAL TANSFMATNS N GANC CEMSTY FNAL EXAMNATN - PAPE # 432 Dr. Phil ultin Friday December 17, 2004. NAME: STUDENT NUMBE: 1. (20 Marks) For each
PAGE 1 of 11 UNVESTY F MANTBA DEPATMENT F CEMSTY 2.339 STUCTUAL TANSFMATNS N GANC CEMSTY FNAL EXAMNATN - PAPE # 432 Dr. Phil ultin Friday December 17, 2004. NAME: STUDENT NUMBE: 1. (20 Marks) For each
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320
 Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
Insertion Reactions. 1) 1,1 insertion in which the metal and the X ligand end up bound to the same (1,1) atom
 Insertion Reactions xidative addition and substitution allow us to assemble 1e and 2e ligands on the metal, respectively. With insertion, and its reverse reaction, elimination, we can now combine and transform
Insertion Reactions xidative addition and substitution allow us to assemble 1e and 2e ligands on the metal, respectively. With insertion, and its reverse reaction, elimination, we can now combine and transform
Organic Chemistry Lecture 2 - Hydrocarbons, Alcohols, Substitutions
 ALKANES Water-insoluble, low density C-C single bonds Higher MW -> higher BP, higher MP Branching -> lower BP, higher MP Forms cycloalkanes which can have ring strain Cyclohexane: chair vs. boat configuration
ALKANES Water-insoluble, low density C-C single bonds Higher MW -> higher BP, higher MP Branching -> lower BP, higher MP Forms cycloalkanes which can have ring strain Cyclohexane: chair vs. boat configuration
Part C- section 1 p-bonds as nucleophiles
 Part C- section 1 p-bonds as nucleophiles Chemistry of Alkenes (Ch 8, 9, 10) - the double bond prevents free rotation - isomerism cis and trans - nomenclature E and Z (3 or 4 different substituents around
Part C- section 1 p-bonds as nucleophiles Chemistry of Alkenes (Ch 8, 9, 10) - the double bond prevents free rotation - isomerism cis and trans - nomenclature E and Z (3 or 4 different substituents around
ANSWER GUIDE APRIL/MAY 2006 EXAMINATIONS CHEMISTRY 249H
 AWER GUIDE APRIL/MAY 2006 EXAMIATI CEMITRY 249 1. (a) PDC / C 2 2 (b) t-bume 2 i (1 equiv) / imidazole (1 equiv) i TBDM protection of the less sterically hindered alcohol (c) BuLi (1 equiv) then (d) 2
AWER GUIDE APRIL/MAY 2006 EXAMIATI CEMITRY 249 1. (a) PDC / C 2 2 (b) t-bume 2 i (1 equiv) / imidazole (1 equiv) i TBDM protection of the less sterically hindered alcohol (c) BuLi (1 equiv) then (d) 2
Nucleophilic attack on ligand
 Nucleophilic attack on ligand Nucleophile "substitutes" metal hapticity usually decreases xidation state mostly unchanged Competition: nucleophilic attack on metal usually leads to ligand substitution
Nucleophilic attack on ligand Nucleophile "substitutes" metal hapticity usually decreases xidation state mostly unchanged Competition: nucleophilic attack on metal usually leads to ligand substitution
Points: 1 / 15 / Total: 100 points /
 Chemistry 6372 Fall 2002 omework #6 Answer Key Name Points: 1 / 15 / 2 / 10 / 3 / 10 / 4 / 10 / 5 / 10 / 6 / 10 / 7 / 35 / Total: 100 points / Average: 73 1) Answer the following questions from our book:
Chemistry 6372 Fall 2002 omework #6 Answer Key Name Points: 1 / 15 / 2 / 10 / 3 / 10 / 4 / 10 / 5 / 10 / 6 / 10 / 7 / 35 / Total: 100 points / Average: 73 1) Answer the following questions from our book:
Electrophile = electron loving = any general electron pair acceptor = Lewis acid, (often an acidic proton)
 314 Arrow Pushing practice/eauchamp 1 Electrophile = electron loving = any general electron pair acceptor = Lewis acid, (often an acidic proton) ucleophile = nucleus/positive loving = any general electron
314 Arrow Pushing practice/eauchamp 1 Electrophile = electron loving = any general electron pair acceptor = Lewis acid, (often an acidic proton) ucleophile = nucleus/positive loving = any general electron
TMSCl imidazole DMF. Ph Ph OTMS. Michael reaction. Michael reaction Ph R 3. epoxidation O R
 eaction using diarylprolinol silyl ether derivatives as catalyst 1) C Et K C 3, ) MgBr, TF TMS hexane, 0 o C TBS p- C 6 4, T C Et 85%, 99% ee Angew. Chem., nt. Ed., 44, 41 (005). rg. Synth., 017, 94, 5.
eaction using diarylprolinol silyl ether derivatives as catalyst 1) C Et K C 3, ) MgBr, TF TMS hexane, 0 o C TBS p- C 6 4, T C Et 85%, 99% ee Angew. Chem., nt. Ed., 44, 41 (005). rg. Synth., 017, 94, 5.
Catalytic Asymmetric Pauson-Khand Reaction. Won-jin Chung 02/25/2003
 Catalytic Asymmetric Pauson-Khand eaction U. Khand; G.. Knox; P. L. Pauson; W. E. Watts J. Chem. Soc. Chem. Commun. 1971, 36 Won-jin Chung 02/25/2003 The General Pattern of the Pauson-Khand eaction Co
Catalytic Asymmetric Pauson-Khand eaction U. Khand; G.. Knox; P. L. Pauson; W. E. Watts J. Chem. Soc. Chem. Commun. 1971, 36 Won-jin Chung 02/25/2003 The General Pattern of the Pauson-Khand eaction Co
Answers To Chapter 4 Problems.
 Answers To Chapter Problems.. (a) An eight-electron [+] cycloaddition. It proceeds photochemically. (b) A four-electron conrotatory electrocyclic ring opening. It proceeds thermally. (c) A six-electron
Answers To Chapter Problems.. (a) An eight-electron [+] cycloaddition. It proceeds photochemically. (b) A four-electron conrotatory electrocyclic ring opening. It proceeds thermally. (c) A six-electron
Reaction chemistry of complexes Three general forms: 1. Reactions involving the gain and loss of ligands a. Ligand Dissoc. and Assoc. (Bala) b.
 eaction chemistry of complexes Three general forms: 1. eactions involving the gain and loss of ligands a. Ligand Dissoc. and Assoc. (Bala) b. Oxidative Addition c. eductive Elimination d. Nucleophillic
eaction chemistry of complexes Three general forms: 1. eactions involving the gain and loss of ligands a. Ligand Dissoc. and Assoc. (Bala) b. Oxidative Addition c. eductive Elimination d. Nucleophillic
A. Loupy, B.Tchoubar. Salt Effects in Organic and Organometallic Chemistry
 A. Loupy, B.Tchoubar Salt Effects in Organic and Organometallic Chemistry 1 Introduction - Classification of Specific Salt Effects 1 1.1 Specific Salt Effects Involving the Salt's Lewis Acid or Base Character
A. Loupy, B.Tchoubar Salt Effects in Organic and Organometallic Chemistry 1 Introduction - Classification of Specific Salt Effects 1 1.1 Specific Salt Effects Involving the Salt's Lewis Acid or Base Character
Use of Cp 2 TiCl in Synthesis
 Use of 2 TiCl in Synthesis eagent Control of adical eactions Jeff Kallemeyn May 21, 2002 eactions of 2 TiCl 1. Pinacol Coupling H H H 2. Epoxide pening H H E H Chemoselectivity Activated aldehydes (aromatic,
Use of 2 TiCl in Synthesis eagent Control of adical eactions Jeff Kallemeyn May 21, 2002 eactions of 2 TiCl 1. Pinacol Coupling H H H 2. Epoxide pening H H E H Chemoselectivity Activated aldehydes (aromatic,
Pericyclic Reactions 6 Lectures Year 3 Handout 2 Michaelmas 2017
 Pericyclic eactions 6 Lectures Year 3 andout 2 Michaelmas 27 π6 a σ2 s Prof Martin Smith CL 3.87 martin.smith@chem.ox.ac.uk http://msmith.chem.ox.ac.uk/ Cycloadditions: oxyallyl cation P46 ω s σ2 s odd
Pericyclic eactions 6 Lectures Year 3 andout 2 Michaelmas 27 π6 a σ2 s Prof Martin Smith CL 3.87 martin.smith@chem.ox.ac.uk http://msmith.chem.ox.ac.uk/ Cycloadditions: oxyallyl cation P46 ω s σ2 s odd
Topic 9. Aldehydes & Ketones
 Chemistry 2213a Fall 2012 Western University Topic 9. Aldehydes & Ketones A. Structure and Nomenclature The carbonyl group is present in aldehydes and ketones and is the most important group in bio-organic
Chemistry 2213a Fall 2012 Western University Topic 9. Aldehydes & Ketones A. Structure and Nomenclature The carbonyl group is present in aldehydes and ketones and is the most important group in bio-organic
Chapter 8 Reactions of Alkenes
 Chapter 8 Reactions of Alkenes Electrophilic Additions o Regio vs stereoselectivity Regio where do the pieces add? Markovnikov s rule hydrogen will go to the side of the double bond with most hydrogens.
Chapter 8 Reactions of Alkenes Electrophilic Additions o Regio vs stereoselectivity Regio where do the pieces add? Markovnikov s rule hydrogen will go to the side of the double bond with most hydrogens.
Chapter 3. Alkenes And Alkynes
 Chapter 3 Alkenes And Alkynes Alkenes ydrocarbons containing double bonds C C double bond the functional group center of reactivity Molecular Formula of Alkene Acyclic alkene: C n 2n Cyclic alkene: C n
Chapter 3 Alkenes And Alkynes Alkenes ydrocarbons containing double bonds C C double bond the functional group center of reactivity Molecular Formula of Alkene Acyclic alkene: C n 2n Cyclic alkene: C n
Chapter 11 - Alcohols and Ethers 1
 Andrew Rosen Chapter 11 - Alcohols and Ethers 1 11.1 - Structure and Nomenclature - The common naming calls alcohols as alkyl alcohols (eg: methyl alcohol) - The common names of ethers have the groups
Andrew Rosen Chapter 11 - Alcohols and Ethers 1 11.1 - Structure and Nomenclature - The common naming calls alcohols as alkyl alcohols (eg: methyl alcohol) - The common names of ethers have the groups
Lecture 1 ADVANCED SYNTHESIS Stereochemistry Introduction
 ecture 1 ADVACED YTEI tereochemistry Introduction ne of the most important issues in modern organic synthesis ost natural compounds are enantiomerically pure Frequently different enantiomers have different
ecture 1 ADVACED YTEI tereochemistry Introduction ne of the most important issues in modern organic synthesis ost natural compounds are enantiomerically pure Frequently different enantiomers have different
A Modular Approach to Polyketide Building Blocks: Cycloadditions of Nitrile Oxides and Homoallylic Alcohols
 A Modular Approach to Polyketide Building Blocks: Cycloadditions of itrile xides and Homoallylic Alcohols rganic Letters, 2005, ASAP ina Lohse-Fraefel and Erick M. Carreira * H H H + ' 1. t-bucl, -78 C
A Modular Approach to Polyketide Building Blocks: Cycloadditions of itrile xides and Homoallylic Alcohols rganic Letters, 2005, ASAP ina Lohse-Fraefel and Erick M. Carreira * H H H + ' 1. t-bucl, -78 C
A Tandem Semipinacol Rearrangement/Alkylation of a-epoxy Alcohols: An Efficient and Stereoselective Approach to Multifunctional 1,3-Diols
 A Tandem Semipinacol Rearrangement/Alkylation of a-epoxy Alcohols: An Efficient and Stereoselective Approach to Multifunctional 1,3-Diols B() 2 H H B() 2 H H Hu, X.-D.; Fan, C.-A.; Zhang, F.-M.; Tu, Y.
A Tandem Semipinacol Rearrangement/Alkylation of a-epoxy Alcohols: An Efficient and Stereoselective Approach to Multifunctional 1,3-Diols B() 2 H H B() 2 H H Hu, X.-D.; Fan, C.-A.; Zhang, F.-M.; Tu, Y.
[3,3]-Sigmatropic rearrangements
![[3,3]-Sigmatropic rearrangements [3,3]-Sigmatropic rearrangements](/thumbs/75/71504906.jpg) 1 [3,3]-Sigmatropic rearrangements heat R 1 R 3 R 1 R 3 R 1 R 3 A class of pericyclic reactions whose stereochemical outcome is governed by the geometric requirements of the cyclic transition state Reactions
1 [3,3]-Sigmatropic rearrangements heat R 1 R 3 R 1 R 3 R 1 R 3 A class of pericyclic reactions whose stereochemical outcome is governed by the geometric requirements of the cyclic transition state Reactions
Oxidative Addition/Reductive Elimination 1. Oxidative Addition
 Oxidative Addition Oxidative Addition/Reductive Elimination 1 An oxidative addition reaction is one in which (usually) a neutral ligand adds to a metal center and in doing so oxidizes the metal, typically
Oxidative Addition Oxidative Addition/Reductive Elimination 1 An oxidative addition reaction is one in which (usually) a neutral ligand adds to a metal center and in doing so oxidizes the metal, typically
Scope: 1. S N 2' Displacements. 2. Michael additions. Not Covered:
 Is There a "Crams's ule" for lefins? Part A C=C cleophile Additions cleophilic lefin Additions?! Any reaction which proceeds through electron donation (pair or radical) into the π* orbital of the olefin
Is There a "Crams's ule" for lefins? Part A C=C cleophile Additions cleophilic lefin Additions?! Any reaction which proceeds through electron donation (pair or radical) into the π* orbital of the olefin
Available chemicals from the catalog (the starting sources of carbon compounds will continually decrease as we learn new reactions.
 ucleophilic ubstitution & Elimination Chemistry Beauchamp 1 Available chemicals from the catalog (the starting sources of carbon compounds will continually decrease as we learn new reactions. ources of
ucleophilic ubstitution & Elimination Chemistry Beauchamp 1 Available chemicals from the catalog (the starting sources of carbon compounds will continually decrease as we learn new reactions. ources of
2.339 Practice Problems
 2.339 Practice Problems Basics of Structure and Mechanism 1. What is the expected site of protonation in an enol ether (C=C)? Justify your choice using resonance structures. 2. ank the following molecules
2.339 Practice Problems Basics of Structure and Mechanism 1. What is the expected site of protonation in an enol ether (C=C)? Justify your choice using resonance structures. 2. ank the following molecules
Lewis Base Catalysis: the Aldol Reaction (Scott Denmark) Tom Blaisdell Friday, January 17 th 2014 Topic Talk
 Lewis Base Catalysis: the Aldol eaction (Scott Denmark) Tom Blaisdell Friday, January 17 th 2014 Topic Talk Scott E. Denmark 1975 - S.B. in Chemistry MIT (ichard. olm and Daniel S. Kemp) 1980 - D.Sc in
Lewis Base Catalysis: the Aldol eaction (Scott Denmark) Tom Blaisdell Friday, January 17 th 2014 Topic Talk Scott E. Denmark 1975 - S.B. in Chemistry MIT (ichard. olm and Daniel S. Kemp) 1980 - D.Sc in
