Comparative Anatomy of the Absorption Roots of Terrestrial and Epiphytic Orchids
|
|
|
- Clarissa George
- 5 years ago
- Views:
Transcription
1 83 Vol.51, n. 1 : pp.83-93, January-February 2008 ISSN Printed in Brazil BRAZILIAN ARCHIVES OF BIOLOGY AND TECHNOLOGY AN INTERNATIONAL JOURNAL Comparative Anatomy of the Absorption Roots of Terrestrial and Epiphytic Orchids Ana Sílvia Franco Pinheiro Moreira* and Rosy Mary dos Santos Isaias Universidade Federal de Minas Gerais; Instituto de Ciências Biológicas; Departamento de Botânica; moreira_ana@yahoo.com.br; ; Belo Horizonte - MG - Brasil ABSTRACT The present study compared roots of terrestrial and epiphytic Orchidaceae, analyzing the anatomical characteristics from an ecological point of view. The material was collected at three different sites in Minas Gerais / Brazil and was fixed in FAA. Transverse sections were obtained by freehand sections or from material previously embedded in Paraplast or Historesin. The prominent characteristics of the epiphytic group were: significant smaller perimeter, epidermis with 3 or more cell layers, U-thickened exodermal cell walls, O-thickened endodermal cell walls, and a low ratio between the caliber and the number of protoxylem arches. The terrestrial group presented simple or multiseriate epidermis, and exodermis and endodermis with typical Casparian strips. The anatomical characteristics should have evolved with several adaptations to distinct environments during evolutionary process. Key words: Orchids, terrestrial, epiphytic, anatomy, adaptations INTRODUCTION The Orchidaceae constitute one of the largest families of flowering plants, having around 20,000 species. They are unique in forms, colors and flower structure. However, the vegetative organization is variable among the species (Dressler, 1993). In general, they are composed of adventitious roots, rhizome, stem, leaves, floral ramets, and flowers (Kramer, 1975). During evolutionary process, the orchids have become adapted to distinct environments, so they can be epiphytes, terrestrials, saprophytes or lithophytes (Black, 1973). These environmental variations contribute for structural alterations in vegetative organs (Pabst and Dungs, 1975). The epiphytic orchids constitute at about 75% of all the other Orchidaceae species (Atwood, 1986). They have aerial roots with the function of fixation on substrate, and water and minerals absorption. The terrestrial species absorb their nutrients direct from the soil, and may present three kinds of roots: (1) one adapted to absorption, and fixation; (2) a tuber adapted to nutritive substances storage, and (3) one specialized in storage, absorption, and fixation (Black, 1973). According to Dressler (1993), the tubers are able to survive during dry season, and nourish the plant when it is necessary. The Orchidaceae are subdivided into five subfamilies: Apostasioideae, Cypripedioideae, Epidendroideae, Spiranthoideae, and Orchidoideae (Dressler, 1993). The ten species presently studied belongs to the last three groups. Spiranthoideae and Orchidoideae are constituted mostly of terrestrial species, and so have adaptive characteristics to soil environment, while * Author for correspondence
2 84 Moreira, A. S. F. P. and Isaias, R. M. S. Epidendroideae species are mostly epiphytes (Dressler, 1993). Even though the orchids are considered a derived group, some primitive characteristics may be observed. The reduced size of their seeds is efficient for plant dispersion in environments of difficult colonization, and the flowers specialization to ornitophyly and entomophyly are characteristics to be considered (Robinson and Burns-Balogh, 1982). These authors suggested that the subfamilies Spiranthoideae, Apostasioideae, and Orchidoideae can be viewed as a succession of forms that have reverted to the terrestrial habits from epiphytic ancestry. This research compares terrestrial and epiphytic Orchidaceae absorption roots, analyzing their anatomical characteristics in relation to the environment. MATERIALS AND METHODS The terrestrial orchids: Habenaria petalodes Lindl., Prescottia montana Barb.Rodrig., Prescottia oligantha Lindl. and Sauroglossum nitidum (Vell.) Schltr., and the epiphytic orchids: Epidendrum saxatile Lindl., Encyclia patens Hook., Lanium avicula Lindl., Polystachya estrellensis Rchb. f., Sophronitis pumila (Hook) Van den Berg and M.W.Chase and Scaphyglottis modesta Schltr. were collected at Serra da Calçada, Brumadinho/MG, and Serra do Caraça, Catas Altas/MG. Twenty samples of the roots of each species were fixed in FAA 50 for 48 hours and then transferred to ethanol 50% (Johansen, 1940). The perimeter (p) of twenty roots of each species were calculated by the formula p = 2πr. The diameter of the roots at 2cm from the apex was obtained with a caliper Digimess and used to calculate de radius by the formula d = 2r. The perimeter:protoxylem arches ratio (p/px) was calculated to estimate the relative water and nutrients conduction from the root cortex to the other parts of the plant. Histological slides were prepared by freehand sections or from material previously embedded in Paraplast and/or Leica historesin. The freehand sections were obtained using a razor blade, and stained with 0.5% astra blue and safranin (2:8) (Kraus and Arduin, 1997). The slides were mounted in Haupt glycerinate gelatin (Kraus and Arduin, 1997). Root fragments were dehydrated in buthyl or ethyl series (Johansen, 1940) and embedded in Paraplast (Kraus and Arduin, 1997) or Leica Historesin. Transverse sections were cut on a Leica Jung Biocut microtome, with 5-6µm thickness for Historesin and 10-14µm for Paraplast embedded material. The sections were stained with 0.5% astra blue and safranin (2:8) or 0.5% toluidine blue (Kraus and Arduin, 1997), and mounted in Entellan. Root sections were analyzed in light microscope. Photomicrographs were obtained in an Olympus BH2-BHS photomicroscope. RESULTS Terrestrial orchids Most of the terrestrial orchids have tubers, while Habenaria petalodes has dimorphic roots; one for absorption and soil fixation, and other for nutrient storage. In this research just the absorption roots were analyzed and are described in sequence. Habenaria petalodes (fig. 1). Hairs unicellular. epidermis uniseriate (fig. 11). Exodermis uniseriate. Cortex 7-9 cells wide, parenchymatic, with mycorrhizae sparsely disposed inside cells; cells of 3-4 layers subtending epidermis with reduced intercellular spaces, small size, thin walled, with raphids. Cells of inner 4-5 layers circular, with triangular intercellular spaces, higher sizes. Endodermis uniseriate, with inconspicuous Casparian strips. Vascular cylinder 10-arch, inconspicuous xylem and phloem strands. Pith cells mostly circular, thin walled, reduced intercellular spaces. The perimeter:protoxylem arches ratio was Prescottia oligantha (fig. 2). Velamen 3-4 cell layers, with thick walls and supporting strips. Exodermis uniseriate. Cortex cells wide. Cells thin walled, variable in size. Cells 2-3 layers subtending exodermis and 3-4 layers off endodermis with raphids. Little storage starch grains. Endodermis uniseriate with conspicuous Casparian strips (fig. 15). Vascular cylinder 4- arch, xylem and phloem easily distinguishable. Pith cells mostly circular with no intercellular spaces. The perimeter:protoxylem arches ratio was 3.94.
3 Comparative Anatomy of the Absorption Roots of Terrestrial and Epiphytic Orchids 85 Prescottia montana (fig. 3). Velamen 2-3 cell layers, with thin walls and supporting strips. Exodermis uniseriate. Cortex cells wide, parenchymatic; cells of regular shapes and sizes, with triangular intercellular spaces. All cells thinwalled. Raphids in 1-2 cell layers subtending exodermis and nearby endodermis. Pelotons in parenchymatic cells next to the site of fixation to the substrate and storage starch grains. 2-6 layers nearby endodermis with raphids (fig. 14). Endodermis uniseriate, cells isodiametric, with conspicuous Casparian strips. Vascular cylinder 11-arch, xylem scarcely distinguished within phloem strands. PITH cells mostly circular with thick walls and reduced intercellular spaces. The perimeter:protoxylem arches ratio was Sauroglossum nitidum (fig. 4). Hairs unicellular. velamen 9-10 cell layers (fig. 12), with thick walls and supporting strips. Exodermis uniseriate, cells polygonal, with no apparent thickening. Cortex cells wide, parenchymatic; small cells of 1-3 cell layers subtending exodermis with some raphids; the next cell layers circular and of higher sizes; cells of inner layers reduced in size, with raphids and starch grains. Endodermis uniseriate, with evident Casparian strips. Vascular cylinder 17-arch of intercalated strands of xylem and phloem. Pith cells mostly circular, thick walled, with triangular intercellular spaces and raphids. The perimeter:protoxylem arches ratio was Epiphytic orchids Encyclia patens (fig. 5). Velamen cell layers with thin walls and inconspicuous supporting strips. Exodermis U-thickened with conspicuous passage cells. Cortex 8-9 cells wide, parenchymatic; cells isodiametric, mostly same size, but evidently smaller subjacent to the endodermal and exodermal layers. Cells phithickened (fig. 16). Cells of 3-4 cell layers subtending the exodermis with raphids. Endodermis O-thickened, clusters of passage cells opposite to xylem arches. Vascular cylinder 21- arch, intercalated strands of xylem and phloem surrounded by lignified cells. Pith cells mostly circular with thin walls and triangular intercellular spaces. The perimeter:protoxylem arches ratio was Epidendrum saxatile (fig. 6). Velamen 3-4 cell layers, with thick walls. Exodermis U-thickened (fig. 60), passage cells intermittent. Cortex cells wide, parenchymatic. Cells circular, thin walled, with raphids. Endodermis O-thickened, passage cells opposite to the protoxylem arches (fig. 17). Vascular cylinder 14-arch, with intercalated strands of xylem and phloem. PITH cells mostly circular with triangular intercellular spaces. The perimeter:protoxylem arches ratio was Sophronitis pumila (fig. 7). Velamen 4-5 cell layers, with thin walls and supporting strips. Exodermis uniseriate, U-thickened. Cortex 8-12 cell wide, parenchymatic; cells variable in size, with thin walls, raphids, little starch grains and mycorrhizae in cells closer to the substrate (fig. 18). Endodermis uniseriate, O-thickened. Clusters of passage cells opposite to xylem arches. Vascular cylinder 16-arch, intercalated strands of xylem and phloem surrounded by lignified cells. Pith cells circular, with thin walls and triangular intercellular spaces. The perimeter:protoxylem arches ratio was Polystachia estrellensis (fig. 8) roots were star shaped in transverse section. Velamen 3 cell layers with supporting strips. Exodermis uniseriate, cells domed, mostly isodiametric, U-thickened. Tilosomes disposed near passage cells (fig. 20). Cortex 8-16 cells wide, parenchymatic, cells thin walled with triangular intercellular spaces filled with mucilage-like substance. Hiphae might be immersed in this substance (fig. 19). Thick walled cell layers subtending exodermis, storage raphids (fig. 19), and starch grains. Endodermis uniseriate, O-thickened, with clusters of 3-4 grouped passage cells. Vascular cylinder 11-arch, intercalated strands of xylem and phloem surrounded by lignified cells. PITH of reduced dimension, cells circular, thin walls and triangular intercellular spaces. The perimeter:protoxylem arches ratio was Lanium avicula (fig. 9). Velamen biseriate with supporting strips (fig. 13). Exodermis U- thickened. Cortex 4-5 cells wide, parenchymatic; cell layers subtending exodermis and endodermis of smaller size. Intermediate cell layers circular, thin walled, with intercelular spaces. Endodermis uniseriate, O-thickened (fig. 21). Vascular cylinder 15-arch, with intercalated strands of xylem and phloem, surrounded by lignified cells. Pith cells mostly circular, with thick walls, and no intercellular spaces. The perimeter:protoxylem arches ratio was Scaphyglottis modesta (fig. 10). Velamen 4 cell layers, with hiphae of fungi, mostly concentrated near passage cells of exodermis. Exodermis
4 86 Moreira, A. S. F. P. and Isaias, R. M. S. uniseriate, O-thickened. Passage cells isolated. Cortex 6-8 cells wide, parenchymatic, with micorrhizae. Cells subtending exodermis and endodermis of reduced size. Cells of intermediate layers circular to oval, with thin walls. Endodermis uniseriate, O-thickened. Vascular cylinder 12- arch. Intercalated strands of xylem and phloem surrounded by lignified cells. Pith cells mostly circular, lignified, with no intercellular spaces. The perimeter:protoxylem arches ratio was parenchyma vascular cylinder epidermis mycorrizae Figures Absorption roots diagrams of terrestrial orchids. 1) Habenaria petalodes. 2) Prescottia oligantha. 3) Prescottia montana. 4) Sauroglossum nitidum. Arrows indicate raphids. (bars = 0.5mm in 1; and 1mm in 2-4)
5 Comparative Anatomy of the Absorption Roots of Terrestrial and Epiphytic Orchids parenchyma vascular cylinder velamen mycorrizae 10 Figures Absorption roots diagrams of epiphytic orchids. 5) Encyclia patens. 6) Epidendrum saxatile. 7) Sophronitis pumila. 8) Polystachia estrellensis. 9) Lanium avicula. 10) Scaphyglottis modesta. Arrows indicate idioblasts with raphids. (bars = 1mm in 5-8; and 0.4mm in 9-10)
6 88 Moreira, A. S. F. P. and Isaias, R. M. S Figures ) Uniseriate epidermis in Habenaria petalodes. 12) Velamen and exodermis in Sauroglossum nitidum. 13) Velamen and U-thickened exodermis in Lanium avicula. (bars = 100µm) * * 16 * 17 Figures ) Detail of raphid near endodermis and starch grains in the cortex of Prescottia montana. 15) Casparian strips (arrows) in the endodermis of Prescottia oligantha. 16) phi- thickened (*) parenchymatic cells in Encyclia patens, the arrows indicate passage cells in endodermis e exodermis. 17) O-thickened endodermis, passage cells (arrows) opposite to the protoxylem arches in Epidendrum saxatile. (bars = 100µm in 14, 16-17; and 50µm in 15)
7 Comparative Anatomy of the Absorption Roots of Terrestrial and Epiphytic Orchids Figures ) Sophronitis pumila with cortex mycorrhizae forming pelotons. 19) Raphids in thick walled idioblasts and fungi imerse in a mucilage-like substance in Polystachia estrellensis. 20) U-thickened exodermis and tilosome (arrows) in P. estrellensis. 21) Lanium avicula O-thickened endodermis. (bars = 100µm) Table 1 - Variation in root perimeter, number of protoxylem arches, and perimeter/protoxylem arches ratio in absorption roots of terrestrial and epiphytic orchids. Orchidacae species Perimeter* (mm) Protoxilem arches Perimeter/protoxylem arches ratio Habenaria petalodes 6.33 ± Prescottia montana ± 1, Prescottia oligantha ± Sauroglossum nitidum ± Encyclia patens 9.63 ± Epidendrum saxatile 7.39 ± Lanium avicula 2.62 ± Polystachia estrellensis 9.73 ± Sophronitis pumila 7.62 ± Scaphyglottis modesta 4.34 ± *Average (± standard deviation) in twenty roots of each species.
8 90 Moreira, A. S. F. P. and Isaias, R. M. S. DISCUSSION All the studied species have similar root structure, with parenchymatic pith involved by a variable number of xylem and phloem strands, endodermis with or without evident Casparian strips, parenchymatic cells, exodermis, and simple epidermis or velamen. However, distinct tissue distribution, cell shapes, and number of cell layers can be noted. The diameter of the roots of the terrestrial Orchidaceae is wider than that of the epiphytic ones. This characteristic can be associated with the storage of nutritive substances and water (Zots, 1999). Habenaria roots have the smallest diameter when compared to the other species, at least in terms of absorption roots. The function of storing substances in this species is concentrated in tubers. The star form of P. estrellensis roots may amplify the contact of root surface with the substrate and with the atmosphere, facilitating water, and possibly nutrients absorption, as well. The root dermal system may consist of a single or of many cell layers, morphologically and physiologically distinct (Fahn, 1990). The velamen, a multiple seriate dermal system, is formed by dead cells at maturity. The number of cell layers does not change with different environmental conditions, however, the rate of maturation, size of cells and hence thickness of the velamen can be markedly altered (Oliveira and Sajo, 1999; Dycus and Knudson, 1957). Many functions are attributed to this tissue, like amplifying access to mineral-rich solutions (Benzing et al., 1982), reducing transpiration, reflecting infrared radiation, offering substantial mechanical protection (Benzing et al., 1982; Pridgeon, 1986), and exchanging oxygen and carbon dioxide between the root and atmosphere (Dycus and Knudson, 1957; Sanford and Adanlawo, 1973). The significantly higher number of cell layers in S. nitidum velamen when compared to the other terrestrial orchids may be related to their bigger diameter, and can also amplify the mechanical protection. Benzing et al. (1982) believe that the orchids velamen act as a sponge, allowing the root to immobilize a temporary but rightly accessible reservoirs of moisture and minerals, however brief its contact with precipitation or canopy leachates. The presence of velamen is observed in almost all of the studied species, except in H. petalodes where there is just a simple epidermis. This feature has been described by Stern (1997) for H. distans and H. repens, even though it is not present in all Habenariinae species. According to Dressler (1993), the velamen is, generally, spongy, and with fibrous thickened cell walls. This thickness appears to confirm on a rather large scale the hypothesis that the functions of the velamen are water conservation (Sanford and Adanlawo, 1973), inhibition of cellular collapse during periods of desiccation (Pridgeon, 1986), and mechanical protection (Noel, 1974). The velamen fibrous thickened cell walls are observed in all of the studied species. The tilosomes observed in P. estrellensis are near passage cells and may be considered an important feature. This structure permits water absorption, possibly prevent the entry of pathogens (Holtzmeier et al., 1998), and reduce root transpiration (Benzing et al., 1982; Benzing et al. 1983). Benzing et al. (1982) call attention for the fact that the movements of substances beyond the velamen may be partially blocked by the presence of these fibrous bodies. At the bottom of them, however, the extent of solid wall between the velamen dead space and the root symplast is much thinner than at any other point along the exodermis-velamen interface (Benzing et al. 1982). Tilosomes are previously referred by Pridgeon et al. (1983) for the epiphytic group as an adaptative feature. The exodermis is the outer layer of cortex (Engard, 1944), and may be constituted, in transverse section, of long and short cells alternately disposed. The long cells develop thick secondary walls, and are empty and dead at maturity (Pridgeon, 1986). Wall thickening of exodermal cells is frequently caused by lignin and suberin deposition (Fahn, 1990). It provides mechanical protection, and the provision of a high-humidity envelope around the cortex (Benzing et al. 1983; Sanford and Adanlawo, 1973). The shorter cells, called passage cells, are thin walled and living at maturity, what makes them important to the nutrition, and hydration of the epiphytic orchids. The passage cells in the exodermis attract endomycorrhizal fungi and are thus capable of ion uptake, once the velamen and the other exodermal cells are dead (Peterson and Enstone, 1996; Senthilkumar et al. 2000a). The terrestrial orchids
9 Comparative Anatomy of the Absorption Roots of Terrestrial and Epiphytic Orchids 91 exodermis in the studied species exhibit just Casparian strips in their walls, what facilitates water and nutrients absorption, while the exodermis of epiphytic species exhibit U and O- thickness. The parenchymatic cortex in the studied terrestrial orchids is wider than the vascular cylinder, except for H. petalodes. The presence of tubers in this species can influence the structure of main roots, once just the tubers have cortical development. This result may be related to the highest water and nutritive storage in terrestrial orchids. In E. patens, there are branched phi-thickenings in cortical cell walls. These kind of cell wall thickenings have been already described by Haas et al. (1976) in Pelargonium roots. To Dickison (2000), these thickenings have cellulosic, lignin or condensate tannin origin, with the function of mechanical support, water regulation or ions transport through the apoplast. Raphids of calcium oxalate are observed in all the studied terrestrial and epiphytic orchids, except in L. avicula and S. modesta. The crystal idioblasts have different forms and characteristic wall thickenings. In P. montana, these cells are conspicuously cylindrical, while in the other studied species they present the same shape of the cortical cells. P. estrellensis idioblasts have cellulosic cell wall thickenings, noted by purplish color when toluidine blue is used (O Brien and McCully, 1981). The calcium oxalate crystals have many different functions, like storage forms of calcium and oxalic acid, plant defense, ionic balance, osmotic regulation, and calcium levels in the sieve elements regulation (Franceschi and Horner, 1980; Bonates, 1993; Paiva and Machado, 2005). Prychid and Rudall (1999) believe there is calcium oxalate resorption in times of calcium depletion. Many studies have showed symbiotic relation between fungi and orchids. According to Zots (1999), this kind of relation is common among Orchidaceae. To Senthilkumar and Krishnamurthy (1998), there are different kinds of micorritics associations if the site of fungi installation in root cortex is taken into consideration. The orchids have a special kind of endomycorrizae where the hyphae of fungi make characteristics pelotons inside cortical cells. The pelotons form intracellular bodies which are digested by their host cells, and are important for plant nutrition (Lesica and Antibus, 1990; Senthilkumar et al., 2000b). In this research, mycorrizae are observed in almost all studied species, except in P. oligantha and S. modesta. Senthilkumer et al. (2000a) affirm that mycorrizae cannot be detected in the stele or in endodermal cells, and that pelotons may be present just in cortical cells, what is coherent with our observations in S. pumila, P. montana, H. petalodes, and P. estrellensis. For these authors, the hiphae penetrate root cortex through exodermal passage cells (which can control the passage of these fungi). In S. modesta, this fact can be easily noted, and the presence of hiphae is evident in velamen cells concentrated, especially, near passage cells regions. The purple colored substance detected in the cortex cells of P. estrellensis may be of pectic nature, for toluidin blue stains alginates and pectic substances in purple (O Brien and McCully, 1981). All the epiphytic orchids studied show secondary thickening in endodermal cells. This thickening, as described by Fahn (1990) and Mauseth (1988), is deposited over the Casparian strips by additional deposition of suberin and lignin. The endodermal thickenings constitute an efficient apoplastic barrier to water and nutrients (Ma and Peterson 2003). So, just like in the exodermal layer, the presence of passage cells in endodermis is necessary. The vascular cylinder exhibits similar characteristics in all the studied species. Xylem and phloem strands are intercalated and involve a central parenchymatic pith. The xylem arches are located in the direction of passage cells in endodermis, which facilitates water and nutrient transport to the rest of the plant. All of the studied species were considered medulated monosteles. In general, the analysis of some of the structures discussed above is too interesting and may relate these plants to their habits. The presence of velamen in epiphytic orchids is fundamentally important to facilitate water and nutrients absorption. Lignified exodermis and endodermis comply intensive mechanical protection against water evaporation, and control the entrance of mycorrizae in cortical cells. The terrestrial habit implies less structural adaptations for water deficit. The perimeter:protoxylem arches ratio calls special attention. The higher number of protoxylem arches related to root size observed in epiphytic roots, may be a response to the necessity of efficient substances transport to the rest of the plant. This efficient transportation possibly compensates the absence of storage tissues. So, in
10 92 Moreira, A. S. F. P. and Isaias, R. M. S. terrestrial orchids transportation may be slower as the nutrients are available nearby. The presence of velamen, lignified exodermis and endodermis, and a higher number of protoxylem arches in relation to root size are important characteristics for an efficient colonization of the epiphytic environment. The morphological characteristics of terrestrial and epiphytic orchids feed constant discussions about the origin of orchids (Porembski and Barthlott, 1988; Robinson and Burns-Balogh, 1982). The evolution of this group of plants has been discussed for a long time, but even nowadays it is difficult to establish all the points of the process. The variety of morphological, anatomical and physiological traits in orchids maintains the uncertainty about the primitive characteristics or derivates features. ACKNOWLEDGEMENTS The authors would like to thank CAPES for financial support and the biologists Rubens Custódio da Motta and Pedro Lage Vianna for taxonomical contributions. RESUMO O presente trabalho comparou raízes de Orchidaceae epífitas e terrestres, analisando as características sob um ponto de vista ecológico. O material foi coletado em três diferentes regiões de Minas Gerais/Brasil e fixado em FAA. Cortes transversais foram obtidos à mão livre ou em material infiltrado em Paraplast ou Historresina. As principais características do grupo epifítico foram: perímetro significativamente menor, epiderme com 3 ou mais camadas celulares, exoderme com espessamento em U, endoderme com espessamento em O e uma baixa razão entre o calibre e o número de pólos de protoxilema. O grupo terrestre apresentou epiderme simples ou bisseriada e exoderme e endoderme com estrias de Caspary. As características anatômicas podem estar envolvidas com diferentes adaptações aos distintos ambientes durante o processo evolutivo. REFERENCES Atwood, J. T. (1986), Two new species in the Maxillaria rufescens complex from Central America. Lindleyana, 11, Benzing, D. H.; Friedman, W. E.; Peterson, G. and Renfrow, A. (1983), Shootlessness, velamentous roots, and the pre-eminence of Orchidaceae in the epiphytic biotipe. American Journal of Botany, 70(1), Benzing, D. H.; Ott, D. W. and Friedman, W. E. (1982), Roots of Sobralia macrantha (Orchidaceae): structure and function of the velamen-exodermis complex. American Journal of Botany, 69(4), Black, P. M. (1973), Orquídeas. Ed. Ao Livro Técnico S/A. Rio de Janeiro. Bonates, L. C. M. (1993), Estudos ecofisiológicos de Orchidaceae da Amazônia. II - Anatomia ecológica foliar de espécies com metabolismo CAM de uma campina da Amazônia central. Acta Amazonica, 23(4), Dickison, W. C. (2000), Integrative plant anatomy. Harcourt Academic Press, USA. Dressler, R. L. (1993), Phylogeny and classification of the Orchid family. Cambridge University Press. Deoscorides Press, Oregon. Dycus, A. M. and Knudson, L. (1957), The role of the velamen of the aerial roots of orchids. Botanical Gazette, 119, Engard, C. J. (1944), Morphological identity of the velamen and exodermis in orchids. Botanical Gazette, 105, Fahn, A. (1990), Plant anatomy. Pergamon Press, Oxford. Franceschi, V. R. and Horner, H. T. (1980), Calcium oxalate crystals in plants. Botanical Review, 46, Haas, D. L.; Carothers, Z. B. and Robbins, R. R. (1976), Observations on the phi-thickenings and Casparian strips in Pelargonium roots. American Journal of Botany, 63(6), Holtzmeier, M. A.; Stern, W. L. and Judd, W. S. (1998), Comparative anatomy and systematics of Senghas's cuschion of Maxillaria (Orchidaceae). Botanical Journal of the Linnean Society, 127, Johansen, D. A. (1940), Plant microtechnique. McGraw Hill Book Co, New York. Kramer, J. (1975), Orchids. H. N. Abrams Publishers, New York. Kraus, J. E. and Arduin, M. (1997), Manual básico de métodos em morfologia vegetal. Editora da Universidade Federal Rural do Rio de Janeiro, Seropédica. Lesica, P. and Antibus, R. K. (1990), The occurrence of mycorrhizae in vascular epiphytes of two Costa Rican rain forests. Biotropica, 22(3),
11 Comparative Anatomy of the Absorption Roots of Terrestrial and Epiphytic Orchids 93 Ma, F. and Peterson, C. A. (2003), Current insights into the development, structure and chemistry of the endodermis and exodermis of roots. Canadian Journal of Botany, 81, Mauseth, J. D. (1988), Plant anatomy. Benjamin/Cumming. Menlo Park. Noel, A. R. A. (1974), Aspects of cell wall structure and the development of the velamen in Ansellia gigantean Reichb. f. Annals of Botany, 38, O Brien, T. P. and McCully, M. E. (1981), The study of plant. Structure principles and selected methods. Termarcarphi Pty, Melbourne. Oliveira, V. C. and Sajo, M. G. (1999), Root anatomy of nine Orchidaceae species. Brazilian Archives of Biology and Technology, 42(4), Pabst, G. F. J. and Dungs, F. (1975), Orchidaceae Brasiliensis I. Hildesheim, Brucke-Verlag. Kurt Schmerson. Paiva, E. A. S. and Machado, S. R. (2005), Role of intermediary cells in Peltodon radicans (Lamiaceae) in the transfer of calcium and formation of calcium oxalate crystals. Brazilian Archives of Biology and Technology, 84(1), Peterson, C. A. and Enstone, D. E. (1996), Functions of passage cells in the endodermis and exodermis of root. Physiologia Plantarum, 97, Porembski, S. and Barthlott, W. (1988), Velamen radicum micromorphology and classification of Orchidaceae. Nordic Journal of Botany, 8, Pridgeon, A. M.; Stern, W. L. and Benzing, D. H. (1983), Tilosomes in roots of Orchidaceae: Morphology and systematic occurrence. American Journal of Botany, 70(9), Pridgeon, A. M. (1986), Anatomical adaptations in Orchidaceae. Lindleyana, 1(2), Prychid, C. J. and Rudall, P. J. (1999), Calcium oxalate crystals in monocotyledons: A review of their structure and systematics. Annals of Botany, 84, Robinson, H. and Burns-Balogh, P. (1982), Evidence for a primitively epiphytic habit in Orchidaceae. Sistematic Botany, 7(4), Sanford, W. W. and Adanlawo, I. (1973), Velamen and exodermis characters of West African epiphytic orchids in relation to taxonomic grouping and habit tolerance. Botanical Journal of the Linnean Society, 66, Senthilkumar, S.; Krishnamurthy, K. V.; Britto, S. J. and Arockiasamy, D. I. (2000a), Visualization of orchid mycorrhizal fungal structures with fluorescence dye using epifluorescence microscopy. Current Science, 79(11), Senthilkumar, S.; Britto, S. J.; Krishnamurthy, K. V. and Hariharam, C. (2000b). Biochemical analysis of mycorrhizal roots of Aerides maculosum. Phytomorphology, 50(3and4), Senthilkumar, S. and Krishnamurthy, K. V. (1998), A cytochemical study on the mycorrhizae of Spathoglottis plicata. Biologia Plantarum, 41(1), Stern, W. L. (1997), Vegetative anatomy of subtribe Habenariinae (Orchidaceae). Botanical Journal of the Linnean Society, 125, Zots, G. (1999), What are Backshoots good for? Seasonal changes in mineral, carbohydrate and water content of different organs of the epiphytic orchid, Dimerandra emarginata. Annals of Botany, 84, Received: August 18, 2005; Revised: May 31, 2006; Accepted: April 23, 2007.
! Xylem - Chief conducting tissue for water and minerals absorbed by the roots.
 + Complex Tissues! Complex tissues are made up of two or more cell types.! Xylem - Chief conducting tissue for water and minerals absorbed by the roots.! Vessels - Made of vessel elements.! Long tubes
+ Complex Tissues! Complex tissues are made up of two or more cell types.! Xylem - Chief conducting tissue for water and minerals absorbed by the roots.! Vessels - Made of vessel elements.! Long tubes
~*~ Roots ~*~ BI 103: Plant-Animal A&P
 ~*~ Roots ~*~ BI 103: Plant-Animal A&P Outline: 1. Quiz #1 2. Leaves review 3. Lecture 4. Outside: roots? Plant Anatomy: Vegetative Organs Leaves: Photosynthesis Gas exchange Light absorption Stem: Support
~*~ Roots ~*~ BI 103: Plant-Animal A&P Outline: 1. Quiz #1 2. Leaves review 3. Lecture 4. Outside: roots? Plant Anatomy: Vegetative Organs Leaves: Photosynthesis Gas exchange Light absorption Stem: Support
23 2 Roots Slide 2 of 36
 2 of 36 Types of Roots Types of Roots What are the two main types of roots? 3 of 36 Types of Roots The two main types of roots are: taproots, which are found mainly in dicots, and fibrous roots, which
2 of 36 Types of Roots Types of Roots What are the two main types of roots? 3 of 36 Types of Roots The two main types of roots are: taproots, which are found mainly in dicots, and fibrous roots, which
Dynamic Plant. Functions of Primary Systems. History of Plants. Plants invaded the land around 400 mya.
 Dynamic Plant Roots & Water Acquisition Roots 1) Anchor the plant 2) Absorb water 3) Absorb minerals 4) Store surplus sugars 5) Transport water, minerals and sugars and hormones 6) Produce some hormones
Dynamic Plant Roots & Water Acquisition Roots 1) Anchor the plant 2) Absorb water 3) Absorb minerals 4) Store surplus sugars 5) Transport water, minerals and sugars and hormones 6) Produce some hormones
Visit For All NCERT solutions, CBSE sample papers, Question papers, Notes for Class 6 to 12. Chapter-6 ANATOMY OF FLOWERING PLANTS
 Chapter-6 ANATOMY OF FLOWERING PLANTS POINTS TO REMEMBER Anatomy : Anatomy is the study of internal structure of organisms. Plant anatomy includes organisation and structure of tissues. Tissue : A group
Chapter-6 ANATOMY OF FLOWERING PLANTS POINTS TO REMEMBER Anatomy : Anatomy is the study of internal structure of organisms. Plant anatomy includes organisation and structure of tissues. Tissue : A group
Topic 2: Plant Structure & Growth Ch. 35 Angiosperms are the most complex plants. They are composed of cells, tissues, organs and organ systems.
 Topic 2: Plant Structure & Growth Ch. 35 Angiosperms are the most complex plants. They are composed of cells, tissues, organs and organ systems. Fig. 35.8 Plant Cells pp.798-802 Types of plant cells Include:
Topic 2: Plant Structure & Growth Ch. 35 Angiosperms are the most complex plants. They are composed of cells, tissues, organs and organ systems. Fig. 35.8 Plant Cells pp.798-802 Types of plant cells Include:
Biology Slide 1 of 36
 Biology 1 of 36 2 of 36 Types of Roots Types of Roots What are the two main types of roots? 3 of 36 Types of Roots The two main types of roots are: taproots, which are found mainly in dicots, and fibrous
Biology 1 of 36 2 of 36 Types of Roots Types of Roots What are the two main types of roots? 3 of 36 Types of Roots The two main types of roots are: taproots, which are found mainly in dicots, and fibrous
MORPHOLOGICAL AND STRUCTURAL FEATURES OF MYCORRIZAL ROOTS OF SPATHOGLOTTIS PLICATA AND DENDROBOIUM SPECIES
 MORPHOLOGICAL AND STRUCTURAL FEATURES OF MYCORRIZAL ROOTS OF SPATHOGLOTTIS PLICATA AND DENDROBOIUM SPECIES S. Senthilkumar 1, P. Saravanakumar 2, K. V. Krishnamurthy 3 and S. John Britto 1, 1 Department
MORPHOLOGICAL AND STRUCTURAL FEATURES OF MYCORRIZAL ROOTS OF SPATHOGLOTTIS PLICATA AND DENDROBOIUM SPECIES S. Senthilkumar 1, P. Saravanakumar 2, K. V. Krishnamurthy 3 and S. John Britto 1, 1 Department
ARE YOU familiar with the sayings Get to
 Root Anatomy ARE YOU familiar with the sayings Get to the root of the problem or the root of all evil? Both these sayings suggest that the root is an essential part of something. With plants, the essential
Root Anatomy ARE YOU familiar with the sayings Get to the root of the problem or the root of all evil? Both these sayings suggest that the root is an essential part of something. With plants, the essential
Plant Structure. Lab Exercise 24. Objectives. Introduction
 Lab Exercise Plant Structure Objectives - Be able to identify plant organs and give their functions. - Learn distinguishing characteristics between monocot and dicot plants. - Understand the anatomy of
Lab Exercise Plant Structure Objectives - Be able to identify plant organs and give their functions. - Learn distinguishing characteristics between monocot and dicot plants. - Understand the anatomy of
Organs and leaf structure
 Organs and leaf structure Different types of tissues are arranged together to form organs. Structure: 2 parts (Petiole and Leaf Blade) Thin flat blade, large surface area Leaves contain all 3 types of
Organs and leaf structure Different types of tissues are arranged together to form organs. Structure: 2 parts (Petiole and Leaf Blade) Thin flat blade, large surface area Leaves contain all 3 types of
CHAPTER TRANSPORT
 CHAPTER 2 2.4 TRANSPORT Uptake of CO2 FOCUS: Uptake and transport of water and mineral salts Transport of organic substances Physical forces drive the transport of materials in plants over a range of distances
CHAPTER 2 2.4 TRANSPORT Uptake of CO2 FOCUS: Uptake and transport of water and mineral salts Transport of organic substances Physical forces drive the transport of materials in plants over a range of distances
Plant Structure and Function
 Plant Structure and Function A Meridian Biology AP Study Guide by John Ho and Tim Qi Plant Terms Growth: Growth Types Type Location Description Primary Primary Vertical growth (up-down), dominant direction
Plant Structure and Function A Meridian Biology AP Study Guide by John Ho and Tim Qi Plant Terms Growth: Growth Types Type Location Description Primary Primary Vertical growth (up-down), dominant direction
Roots and Soil Chapter 5
 Roots and Soil Chapter 5 Plant Organs Plant organs are groups of several types of tissues that together perform a particular function. Vegetative organs roots, stems, leaves make and use food, absorb water
Roots and Soil Chapter 5 Plant Organs Plant organs are groups of several types of tissues that together perform a particular function. Vegetative organs roots, stems, leaves make and use food, absorb water
 Question 1: What are the factors affecting the rate of diffusion? Diffusion is the passive movement of substances from a region of higher concentration to a region of lower concentration. Diffusion of
Question 1: What are the factors affecting the rate of diffusion? Diffusion is the passive movement of substances from a region of higher concentration to a region of lower concentration. Diffusion of
Chapter 23 Notes Roots Stems Leaves
 Chapter 23 Notes Roots Stems Leaves I. Specialized tissue in plants - effective way to ensure the plant s survival A. Seed plant structure 1. Roots - a. Absorbs water and dissolves nutrients b. anchors
Chapter 23 Notes Roots Stems Leaves I. Specialized tissue in plants - effective way to ensure the plant s survival A. Seed plant structure 1. Roots - a. Absorbs water and dissolves nutrients b. anchors
Plant Anatomy: roots, stems and leaves
 Plant Anatomy: roots, stems and leaves The plant body has a hierarchy of organs, tissues and cells Plants, like animals, have organs composed of different tissues, which are composed of cells. Tissue is
Plant Anatomy: roots, stems and leaves The plant body has a hierarchy of organs, tissues and cells Plants, like animals, have organs composed of different tissues, which are composed of cells. Tissue is
Transport in Plants AP Biology
 Transport in Plants 2006-2007 Water & mineral absorption Water absorption from soil osmosis aquaporins Mineral absorption active transport proton pumps active transport of H + aquaporin root hair proton
Transport in Plants 2006-2007 Water & mineral absorption Water absorption from soil osmosis aquaporins Mineral absorption active transport proton pumps active transport of H + aquaporin root hair proton
NOTES: CH 35 - Plant Structure & Growth
 NOTES: CH 35 - Plant Structure & Growth In their evolutionary journey, plants adapted to the problems of a terrestrial existence as they moved from water to land ANGIOSPERMS (flowering plants) -most diverse
NOTES: CH 35 - Plant Structure & Growth In their evolutionary journey, plants adapted to the problems of a terrestrial existence as they moved from water to land ANGIOSPERMS (flowering plants) -most diverse
Anatomy of Flowering Plants. K C Meena PGT Biology
 Anatomy of Flowering Plants K C Meena PGT Biology Tissues A group of similar cells performing same function. Types of plant tissues - Meristematic tissues and permanent tissues. Meristematic tissues Have
Anatomy of Flowering Plants K C Meena PGT Biology Tissues A group of similar cells performing same function. Types of plant tissues - Meristematic tissues and permanent tissues. Meristematic tissues Have
Downloaded from
 POINTS TO REMEMBER : 6. Anatomy of Flowering Plants Study of internal structure of plant is called anatomy. In plants cells are the basic unit. Cells organized into tissues and tissues organized into organs.
POINTS TO REMEMBER : 6. Anatomy of Flowering Plants Study of internal structure of plant is called anatomy. In plants cells are the basic unit. Cells organized into tissues and tissues organized into organs.
Biology 2 Chapter 21 Review
 Biology 2 Chapter 21 Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following is not a tissue system of vascular plants? a. vascular
Biology 2 Chapter 21 Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following is not a tissue system of vascular plants? a. vascular
OCR (A) Biology A-level
 OCR (A) Biology A-level Topic 3.3: Transport in plants Notes Plants require a transport system to ensure that all the cells of a plant receive a sufficient amount of nutrients. This is achieved through
OCR (A) Biology A-level Topic 3.3: Transport in plants Notes Plants require a transport system to ensure that all the cells of a plant receive a sufficient amount of nutrients. This is achieved through
Bio Factsheet. Transport in Plants. Number 342
 Number 342 Transport in Plants This Factsheet: Explains why plants need a transport system Describes what plants transport Describes the tissues which carry out transport Outlines the position of the xylem
Number 342 Transport in Plants This Factsheet: Explains why plants need a transport system Describes what plants transport Describes the tissues which carry out transport Outlines the position of the xylem
ENDODERMIS & POLARITY
 https://en.wikipedia.org/wiki/casparian_strip ENDODERMIS & POLARITY Niloufar Pirayesh 13.01.2016 PCDU SEMINAR 2 What is Endodermis? It helps with Regulate the movement of water ions and hormones. (in and
https://en.wikipedia.org/wiki/casparian_strip ENDODERMIS & POLARITY Niloufar Pirayesh 13.01.2016 PCDU SEMINAR 2 What is Endodermis? It helps with Regulate the movement of water ions and hormones. (in and
Transport in Plants (Ch. 23.5)
 Transport in Plants (Ch. 23.5) Transport in plants H 2 O & minerals transport in xylem Transpiration Adhesion, cohesion & Evaporation Sugars transport in phloem bulk flow Gas exchange photosynthesis CO
Transport in Plants (Ch. 23.5) Transport in plants H 2 O & minerals transport in xylem Transpiration Adhesion, cohesion & Evaporation Sugars transport in phloem bulk flow Gas exchange photosynthesis CO
Vegetative anatomy and systematics of Triphorinae (Orchidaceae)
 Eastern Illinois University The Keep Faculty Research & Creative Activity Biological Sciences January 2009 Vegetative anatomy and systematics of Triphorinae (Orchidaceae) Barbara S. Carlsward Eastern Illinois
Eastern Illinois University The Keep Faculty Research & Creative Activity Biological Sciences January 2009 Vegetative anatomy and systematics of Triphorinae (Orchidaceae) Barbara S. Carlsward Eastern Illinois
Chapter 35~ Plant Structure and Growth
 Chapter 35~ Plant Structure and Growth Plant Organization Plant morphology is based on plant s evolutionary history Need to draw in nutrients from the ground and the air Plant Organs Root system = roots
Chapter 35~ Plant Structure and Growth Plant Organization Plant morphology is based on plant s evolutionary history Need to draw in nutrients from the ground and the air Plant Organs Root system = roots
Transport of substances in plants
 Transport of substances in plants We have already looked at why many organisms need transport systems with special reference to surface area and volume. The larger the volume : surface area ratio, the
Transport of substances in plants We have already looked at why many organisms need transport systems with special reference to surface area and volume. The larger the volume : surface area ratio, the
Plant Tissues and Organs. Topic 13 Plant Science Subtopics , ,
 Plant Tissues and Organs Topic 13 Plant Science Subtopics 13.1.2, 13.1.3, 13.1.4 Objectives: List and describe the major plant organs their structure and function List and describe the major types of plant
Plant Tissues and Organs Topic 13 Plant Science Subtopics 13.1.2, 13.1.3, 13.1.4 Objectives: List and describe the major plant organs their structure and function List and describe the major types of plant
Bring Your Text to Lab!!!
 Bring Your Text to Lab!!! Vascular Plant Anatomy: Flowering Plants Objectives: 1. To observe what the basic structure of vascular plants is, and how and where this form originates. 2. To begin to understand
Bring Your Text to Lab!!! Vascular Plant Anatomy: Flowering Plants Objectives: 1. To observe what the basic structure of vascular plants is, and how and where this form originates. 2. To begin to understand
Plant Structure. Objectives At the end of this sub section students should be able to:
 Name: 3.2 Organisation and the Vascular Structures 3.2.1 Flowering plant structure and root structure Objectives At the end of this sub section students should be able to: 1. Label a diagram of the external
Name: 3.2 Organisation and the Vascular Structures 3.2.1 Flowering plant structure and root structure Objectives At the end of this sub section students should be able to: 1. Label a diagram of the external
Vegetative anatomical adaptations of Epidendrum radicans (Epidendroideae, Orchidaceae) to epiphytic conditions of growth
 ISSN 2226-3063 e-issn 2227-9555 Modern Phytomorphology 11: 117 130, 2017 doi: 10.5281/zenodo.1078523 Research Article Vegetative anatomical adaptations of Epidendrum radicans (Epidendroideae, Orchidaceae)
ISSN 2226-3063 e-issn 2227-9555 Modern Phytomorphology 11: 117 130, 2017 doi: 10.5281/zenodo.1078523 Research Article Vegetative anatomical adaptations of Epidendrum radicans (Epidendroideae, Orchidaceae)
Chapter 36: Transport in Vascular Plants - Pathways for Survival
 Chapter 36: Transport in Vascular Plants - Pathways for Survival For vascular plants, the evolutionary journey onto land involved differentiation into roots and shoots Vascular tissue transports nutrients
Chapter 36: Transport in Vascular Plants - Pathways for Survival For vascular plants, the evolutionary journey onto land involved differentiation into roots and shoots Vascular tissue transports nutrients
The three principal organs of seed plants are roots, stems, and leaves.
 23 1 Specialized Tissues in Plants Seed Plant Structure The three principal organs of seed plants are roots, stems, and leaves. 1 of 34 23 1 Specialized Tissues in Plants Seed Plant Structure Roots: absorb
23 1 Specialized Tissues in Plants Seed Plant Structure The three principal organs of seed plants are roots, stems, and leaves. 1 of 34 23 1 Specialized Tissues in Plants Seed Plant Structure Roots: absorb
Exercise 12. Procedure. Aim: To study anatomy of stem and root of monocots and dicots.
 Aim: To study anatomy of stem and root of monocots and dicots. Principle: The study of internal morphology, i.e., cells of various tissues in an organ of a living body is called Anatomy. Tissue, which
Aim: To study anatomy of stem and root of monocots and dicots. Principle: The study of internal morphology, i.e., cells of various tissues in an organ of a living body is called Anatomy. Tissue, which
Lecture 4 Root Put line under your answer! There is only one correct answer in the multiple choice questions
 Lecture 4 Root Put line under your answer! There is only one correct answer in the multiple choice questions 1. The perception of gravity by a root is thought to take place in a) root hairs b) the region
Lecture 4 Root Put line under your answer! There is only one correct answer in the multiple choice questions 1. The perception of gravity by a root is thought to take place in a) root hairs b) the region
 Question 1: State the location and function of different types of meristem. Meristems are specialised regions of plant growth. The meristems mark the regions where active cell division and rapid division
Question 1: State the location and function of different types of meristem. Meristems are specialised regions of plant growth. The meristems mark the regions where active cell division and rapid division
(Photo Atlas: Figures 9.147, 9.148, 9.150, 9.1, 9.2, )
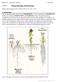 BIOL 221 Concepts of Botany Fall 2007 Topic 07: Primary Plant Body: The Root System (Photo Atlas: Figures 9.147, 9.148, 9.150, 9.1, 9.2, 9.5 9.23) A. Introduction The root has the primary functions of
BIOL 221 Concepts of Botany Fall 2007 Topic 07: Primary Plant Body: The Root System (Photo Atlas: Figures 9.147, 9.148, 9.150, 9.1, 9.2, 9.5 9.23) A. Introduction The root has the primary functions of
Transport in Vascular Plants
 Chapter 36 Transport in Vascular Plants PowerPoint Lectures for Biology, Seventh Edition Neil Campbell and Jane Reece Lectures by Chris Romero Vascular tissue Transports nutrients throughout a plant; such
Chapter 36 Transport in Vascular Plants PowerPoint Lectures for Biology, Seventh Edition Neil Campbell and Jane Reece Lectures by Chris Romero Vascular tissue Transports nutrients throughout a plant; such
ANATOMY OF PLANTS Introduction: The study of gross internal structure of plant organs by the technique of section cutting is called plant anatomy.
 ANATOMY OF PLANTS Introduction: The study of gross internal structure of plant organs by the technique of section cutting is called plant anatomy. (Pandey, 2002). Various plant organ viz. root, stem, leaves,
ANATOMY OF PLANTS Introduction: The study of gross internal structure of plant organs by the technique of section cutting is called plant anatomy. (Pandey, 2002). Various plant organ viz. root, stem, leaves,
2.1 PLANT TISSUE HALIMAHTUN SAEDIAH BT ABU BAKAR KOLEJ TEKNOLOGI TIMUR
 2.1 PLANT TISSUE HALIMAHTUN SAEDIAH BT ABU BAKAR KOLEJ TEKNOLOGI TIMUR GENERAL Plant cell are differentiated possessing structural adaptations that make specific functions possible. Modifications of cell
2.1 PLANT TISSUE HALIMAHTUN SAEDIAH BT ABU BAKAR KOLEJ TEKNOLOGI TIMUR GENERAL Plant cell are differentiated possessing structural adaptations that make specific functions possible. Modifications of cell
Plant Anatomy: roots, stems and leaves
 Plant Anatomy: roots, stems and leaves The plant body has a hierarchy of organs, tissues and cells Plants, like animals, have organs composed of different tissues, which are composed of cells. Tissue is
Plant Anatomy: roots, stems and leaves The plant body has a hierarchy of organs, tissues and cells Plants, like animals, have organs composed of different tissues, which are composed of cells. Tissue is
Today: Plant Structure Exam II is on F March 31
 Next few lectures are on plant form and function Today: Plant Structure Exam II is on F March 31 Outline Plant structure I. Plant Cells structure & different types II. Types of meristems Apical meristems:
Next few lectures are on plant form and function Today: Plant Structure Exam II is on F March 31 Outline Plant structure I. Plant Cells structure & different types II. Types of meristems Apical meristems:
Plants. Tissues, Organs, and Systems
 Plants Tissues, Organs, and Systems Meristematic cells Specialized cells that are responsible for producing specialized cells, they produce three types of tissue in the body of a plant. Meristematic Cells
Plants Tissues, Organs, and Systems Meristematic cells Specialized cells that are responsible for producing specialized cells, they produce three types of tissue in the body of a plant. Meristematic Cells
Non Permanent Tissues - Meristematic Tissue
 PLANT TISSUES Non Permanent Tissues - Meristematic Tissue Undifferentiated plant cells that are continually dividing by mitosis Large thin walled cells No vacuole Dense cytoplasm Large nucleus Found at
PLANT TISSUES Non Permanent Tissues - Meristematic Tissue Undifferentiated plant cells that are continually dividing by mitosis Large thin walled cells No vacuole Dense cytoplasm Large nucleus Found at
Topic 14. The Root System. II. Anatomy of an Actively Growing Root Tip
 Topic 14. The Root System Introduction. This is the first of two lab topics that focus on the three plant organs (root, stem, leaf). In these labs we want you to recognize how tissues are organized in
Topic 14. The Root System Introduction. This is the first of two lab topics that focus on the three plant organs (root, stem, leaf). In these labs we want you to recognize how tissues are organized in
IB Bio: Plant Biology. Topic 9
 IB Bio: Plant Biology Topic 9 9.1: Transport in xylem How and why does water move up a plant? How do plants conserve water? 9.2: Transport in phloem How and why and where does food move in a plant? 9.3:
IB Bio: Plant Biology Topic 9 9.1: Transport in xylem How and why does water move up a plant? How do plants conserve water? 9.2: Transport in phloem How and why and where does food move in a plant? 9.3:
13.4 Roots Figure 2 primary root: primary root secondary root: secondary root taproots fibrous taproots: roots. fibrous roots: adventitious roots
 10. Why is it not surprising that many hydrophytes have little or no tissue? 11. The leaves of many underwater plants are finely divided, dramatically increasing the surface area that is in contact with
10. Why is it not surprising that many hydrophytes have little or no tissue? 11. The leaves of many underwater plants are finely divided, dramatically increasing the surface area that is in contact with
Class XI Chapter 6 Anatomy of Flowering Plants Biology
 Class XI Chapter 6 Anatomy of Flowering Plants Biology Question 1: State the location and function of different types of meristem. Meristems are specialised regions of plant growth. The meristems mark
Class XI Chapter 6 Anatomy of Flowering Plants Biology Question 1: State the location and function of different types of meristem. Meristems are specialised regions of plant growth. The meristems mark
MONOCOT ROOT FIBROUS ROOT SYSTEM
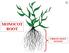 A MONOCOT FIBROUS SYSTEM ^ MONOCOT FIBROUS SYSTEM ADVENTITIOUS S A ADVENTITIOUS S ADVENTITIOUS FIBROUS- SYSTEM ADVENTITOUS NON-EMBRYONIC DERIVED FROM STEM OR LEAF FIBROUS- SYSTEM ADVENTITOUS D MONOCOT
A MONOCOT FIBROUS SYSTEM ^ MONOCOT FIBROUS SYSTEM ADVENTITIOUS S A ADVENTITIOUS S ADVENTITIOUS FIBROUS- SYSTEM ADVENTITOUS NON-EMBRYONIC DERIVED FROM STEM OR LEAF FIBROUS- SYSTEM ADVENTITOUS D MONOCOT
TARGET STUDY MATERIAL
 TARGET STUDY MATERIAL Plus-1 Botany VOL I TARGET EDUCATIONAL INSTITUTION Target Educational institution is the one and only Entrance coaching and CBSE 10 th coaching centre at Mukkam with advanced technologies
TARGET STUDY MATERIAL Plus-1 Botany VOL I TARGET EDUCATIONAL INSTITUTION Target Educational institution is the one and only Entrance coaching and CBSE 10 th coaching centre at Mukkam with advanced technologies
Chapter 6. Biology of Flowering Plants. Anatomy Seedlings, Meristems, Stems, and Roots
 BOT 3015L (Outlaw/Sherdan/Aghoram); Page 1 of 6 Chapter 6 Biology of Flowering Plants Anatomy Seedlings, Meristems, Stems, and Roots Objectives Seedling germination and anatomy. Understand meristem structure
BOT 3015L (Outlaw/Sherdan/Aghoram); Page 1 of 6 Chapter 6 Biology of Flowering Plants Anatomy Seedlings, Meristems, Stems, and Roots Objectives Seedling germination and anatomy. Understand meristem structure
SESSION 6: SUPPORT AND TRANSPORT SYSTEMS IN PLANTS PART 1
 SESSION 6: SUPPORT AND TRANSPORT SYSTEMS IN PLANTS PART 1 KEY CONCEPTS In this session we will focus on summarising what you need to know about: - Anatomy of dicotyledonous plants Root and stem: distribution
SESSION 6: SUPPORT AND TRANSPORT SYSTEMS IN PLANTS PART 1 KEY CONCEPTS In this session we will focus on summarising what you need to know about: - Anatomy of dicotyledonous plants Root and stem: distribution
Recap. Waxy layer which protects the plant & conserves water. Contains chloroplasts: Specialized for light absorption.
 Recap Contains chloroplasts: Specialized for light absorption Waxy layer which protects the plant & conserves water mesophyll Layer contains air spaces: Specialized for gas exchange Vascular Tissue Exchange
Recap Contains chloroplasts: Specialized for light absorption Waxy layer which protects the plant & conserves water mesophyll Layer contains air spaces: Specialized for gas exchange Vascular Tissue Exchange
BIOL 305L Laboratory One
 Please print Full name clearly: BIOL 305L Laboratory One General plant anatomy a great place to start! Introduction Botany is the science of plant life. Traditionally, the science included the study of
Please print Full name clearly: BIOL 305L Laboratory One General plant anatomy a great place to start! Introduction Botany is the science of plant life. Traditionally, the science included the study of
Plant Organs. Roots & Stems
 Plant Organs Roots & Stems I. Roots A. F(x)s = grow underground 1. Absorb water & nutrients from soil 2. Anchor plant in the soil 3. Make hormones important for growth & development I. Roots B. Structure
Plant Organs Roots & Stems I. Roots A. F(x)s = grow underground 1. Absorb water & nutrients from soil 2. Anchor plant in the soil 3. Make hormones important for growth & development I. Roots B. Structure
PLANT TISSUES 12 MARCH 2014
 PLANT TISSUES 12 MARCH 2014 Lesson Description In this lesson we: Identify the different types of plant tissue Be able to relate the different structures with the different functions Plant Tissue Summary
PLANT TISSUES 12 MARCH 2014 Lesson Description In this lesson we: Identify the different types of plant tissue Be able to relate the different structures with the different functions Plant Tissue Summary
Forms strands that conduct water, minerals, and organic compounds. Much of the inside of nonwoody parts of plants. Includes roots, stems, and leaves
 Biology II Vascular plants have 3 tissue systems: Dermal Protective outer layer of plant Vascular Forms strands that conduct water, minerals, and organic compounds Ground Much of the inside of nonwoody
Biology II Vascular plants have 3 tissue systems: Dermal Protective outer layer of plant Vascular Forms strands that conduct water, minerals, and organic compounds Ground Much of the inside of nonwoody
Sci30264 Plant Anatomy and Physiology 1/2012 Endodermis and passage cell Bualuang FAIYUE, PhD. Endodermis and passage cell
 1. Endodermis and its development Endodermis is the innermost layer of the root cortex (Esau, 1965; Brundrett et al., 1990; Fahn, 1990; Peterson and Enstone, 1996). It is characterised by Casparian strip
1. Endodermis and its development Endodermis is the innermost layer of the root cortex (Esau, 1965; Brundrett et al., 1990; Fahn, 1990; Peterson and Enstone, 1996). It is characterised by Casparian strip
Bio 10 Lecture Notes 7: Plant Diversity, Structure and Function SRJC
 Physiology study of the adaptations by which organisms function in their environ. 1.) Plants, Tissues and Function Plant types and their evolution Terrestrial plants evolved from aquatic green algae There
Physiology study of the adaptations by which organisms function in their environ. 1.) Plants, Tissues and Function Plant types and their evolution Terrestrial plants evolved from aquatic green algae There
Mycorrhiza Fungus + Plant Host (Root)
 Mycorrhiza Fungus + Plant Host (Root) Root Anatomy Mycorrhizal fungi Cryptomycota http://www.mykoweb.com/articles/index.html#apm1_4 Summary Mycorrhizal symbioses are mutualistic Fungal benefits carbohydrates
Mycorrhiza Fungus + Plant Host (Root) Root Anatomy Mycorrhizal fungi Cryptomycota http://www.mykoweb.com/articles/index.html#apm1_4 Summary Mycorrhizal symbioses are mutualistic Fungal benefits carbohydrates
CASE STUDY WATER ABSORPTION AND TRANSPORT IN PLANTS
 CASE STUDY WATER ABSORPTION AND TRANSPORT IN PLANTS Presentation of the problem: We need a pump to uplift water to a tank. The requirement of a pump is to pull water against the gravity. Look at the human
CASE STUDY WATER ABSORPTION AND TRANSPORT IN PLANTS Presentation of the problem: We need a pump to uplift water to a tank. The requirement of a pump is to pull water against the gravity. Look at the human
Plant Structure And Growth
 Plant Structure And Growth The Plant Body is Composed of Cells and Tissues Tissue systems (Like Organs) made up of tissues Made up of cells Plant Tissue Systems Ground Tissue System Ø photosynthesis Ø
Plant Structure And Growth The Plant Body is Composed of Cells and Tissues Tissue systems (Like Organs) made up of tissues Made up of cells Plant Tissue Systems Ground Tissue System Ø photosynthesis Ø
DEPARTMENT OF LIFE AND CONSUMER SCIENCES. Plant Structure BOT1501. Semester I: Assignment no. 2 Memorandum
 University Examinations DEPARTMENT OF LIFE AND CONSUMER SCIENCES Plant Structure BOT1501 Semester I: Assignment no. 2 Memorandum 2018 QUESTION 1 1.1 Primary growth is the production of new primary tissues
University Examinations DEPARTMENT OF LIFE AND CONSUMER SCIENCES Plant Structure BOT1501 Semester I: Assignment no. 2 Memorandum 2018 QUESTION 1 1.1 Primary growth is the production of new primary tissues
2/25/2013. o Plants take up water and minerals from below ground o Plants take up CO2 and light from above ground THREE BASIC PLANT ORGANS ROOTS
 o Plants take up water and minerals from below ground o Plants take up CO2 and light from above ground THREE BASIC PLANT ORGANS o Roots o Stems o Leaves ROOTS o Anchor plant o Absorb water and minerals
o Plants take up water and minerals from below ground o Plants take up CO2 and light from above ground THREE BASIC PLANT ORGANS o Roots o Stems o Leaves ROOTS o Anchor plant o Absorb water and minerals
Chapter 30: Plant Nutrition & Transport
 Chapter 30: Plant Nutrition & Transport Carnivorous Plants Capture animals to supplement their nutrient intake Venus flytrap lures insects with sugary bait; closes on victim Cobra lily lures insects down
Chapter 30: Plant Nutrition & Transport Carnivorous Plants Capture animals to supplement their nutrient intake Venus flytrap lures insects with sugary bait; closes on victim Cobra lily lures insects down
Plant Form and Function I
 An overwiev of a flowering plant Main root Root Roots of most plants are covered with root hairs. Their function is to increase root surface area for more efficient absorption of water and nutrients from
An overwiev of a flowering plant Main root Root Roots of most plants are covered with root hairs. Their function is to increase root surface area for more efficient absorption of water and nutrients from
Page 1. Gross Anatomy of a typical plant (Angiosperm = Flowering Plant): Gross Anatomy of a typical plant (Angiosperm = Flowering Plant):
 Chapter 43: Plant Form and Function Gross Anatomy of a typical plant (Angiosperm = Flowering Plant): Root System Anchor plant Absorb water / nutrients Store surplus sugars Transport materials from / to
Chapter 43: Plant Form and Function Gross Anatomy of a typical plant (Angiosperm = Flowering Plant): Root System Anchor plant Absorb water / nutrients Store surplus sugars Transport materials from / to
2014 Pearson Education, Inc. 1
 1 CO 2 O 2 Light Sugar O 2 and minerals CO 2 2 Buds 42 29 21 34 13 26 5 18 10 31 23 8 15 28 16 2 24 Shoot apical meristem 7 3 20 1 mm 32 11 19 12 6 4 1 25 17 14 9 40 27 22 3 Cell wall Apoplastic route
1 CO 2 O 2 Light Sugar O 2 and minerals CO 2 2 Buds 42 29 21 34 13 26 5 18 10 31 23 8 15 28 16 2 24 Shoot apical meristem 7 3 20 1 mm 32 11 19 12 6 4 1 25 17 14 9 40 27 22 3 Cell wall Apoplastic route
Plant Anatomy Lab 7 - Stems II
 Plant Anatomy Lab 7 - Stems II This exercise continues the previous lab in studying primary growth in the stem. We will be looking at stems from a number of different plant species, and emphasize (1) the
Plant Anatomy Lab 7 - Stems II This exercise continues the previous lab in studying primary growth in the stem. We will be looking at stems from a number of different plant species, and emphasize (1) the
Chapter. Transport in. Structure of. 1- Epidermis: 2- Cortex: All plants 2- a specialized. In higher moving by. hydra and. with cuticles) 1-2-
 Chapter 2 Transport in living organisms The concept of transport and the need for it: All plants need CO 2, water and mineral salts to perform photosynthesis In primitive plants such as algae these materials
Chapter 2 Transport in living organisms The concept of transport and the need for it: All plants need CO 2, water and mineral salts to perform photosynthesis In primitive plants such as algae these materials
Homework for Monday: Correct potometer questions Complete transport in plants worksheet
 Transport in plants Homework for Monday: Correct potometer questions Complete transport in plants worksheet Transpiration the loss of water from a plant through evaporation Did you know? A 15m maple tree
Transport in plants Homework for Monday: Correct potometer questions Complete transport in plants worksheet Transpiration the loss of water from a plant through evaporation Did you know? A 15m maple tree
Anatomy of dicotyledonous plants
 Anatomy of dicotyledonous plants Differences between Monocotyledons and Dicotyledons All plants are classified as producing seeds or not producing seeds. Those that produce seeds are divided into flowering
Anatomy of dicotyledonous plants Differences between Monocotyledons and Dicotyledons All plants are classified as producing seeds or not producing seeds. Those that produce seeds are divided into flowering
AP Biology. Basic anatomy. Chapter 35. Plant Anatomy. Shoots. Expanded anatomy. Roots. Modified shoots root shoot (stem) leaves
 Chapter 35. Basic anatomy root shoot (stem) leaves Plant Anatomy Expanded anatomy root root tip root hairs shoot (stem) nodes internodes apical buds axillary buds flowers leaves veins Shoots Shoots consist
Chapter 35. Basic anatomy root shoot (stem) leaves Plant Anatomy Expanded anatomy root root tip root hairs shoot (stem) nodes internodes apical buds axillary buds flowers leaves veins Shoots Shoots consist
Chapter 36. Transport in Vascular Plants
 Chapter 36 Transport in Vascular Plants Overview: Pathways for Survival For vascular plants The evolutionary journey onto land involved the differentiation of the plant body into roots and shoots Vascular
Chapter 36 Transport in Vascular Plants Overview: Pathways for Survival For vascular plants The evolutionary journey onto land involved the differentiation of the plant body into roots and shoots Vascular
Plant Structure and Growth
 Plant Structure and Growth A. Flowering Plant Parts: The flowering plants or are the most diverse group of plants. They are divided into 2 classes and. Examples of monocots: Examples of dicots: The morphology
Plant Structure and Growth A. Flowering Plant Parts: The flowering plants or are the most diverse group of plants. They are divided into 2 classes and. Examples of monocots: Examples of dicots: The morphology
Leaf Anatomy of Three Varians of Arundina graminifolia (D. Don.) Hochr
 Jurnal Natur Indonesia 11(2), April 2009: 78-82 78 ISSN 1410-9379, Jurnal Natur Keputusan Indonesia Akreditasi 11(2): No 78-82 65a/DIKTI/Kep./2008 Sulistiarini, et al. Leaf Anatomy of Three Varians of
Jurnal Natur Indonesia 11(2), April 2009: 78-82 78 ISSN 1410-9379, Jurnal Natur Keputusan Indonesia Akreditasi 11(2): No 78-82 65a/DIKTI/Kep./2008 Sulistiarini, et al. Leaf Anatomy of Three Varians of
13.2 The Vascular Plant Body (textbook p )
 13.2 The Vascular Plant Body (textbook p544 550) Learning Goal: Label and explain the anatomy of the Vascular Plant and it's Tissue Types Plants are classified into two main groups: and. Vascular plants
13.2 The Vascular Plant Body (textbook p544 550) Learning Goal: Label and explain the anatomy of the Vascular Plant and it's Tissue Types Plants are classified into two main groups: and. Vascular plants
2018 Version. Photosynthesis Junior Science
 2018 Version Photosynthesis Junior Science 1 Plants fill the role of Producers in a community Plants are special because they have leaves and are able to produce their own food by the process of photosynthesis
2018 Version Photosynthesis Junior Science 1 Plants fill the role of Producers in a community Plants are special because they have leaves and are able to produce their own food by the process of photosynthesis
1. Climatic Factors. Light Water Temperature Wind Humidity
 Plant Environment - Factors Affecting Plant Growth & Distribution 1. Climatic Factors Light Water Temperature Wind Humidity 1. Climatic factors (Light) Effect of light intensities, quality, and duration
Plant Environment - Factors Affecting Plant Growth & Distribution 1. Climatic Factors Light Water Temperature Wind Humidity 1. Climatic factors (Light) Effect of light intensities, quality, and duration
Chapter 36~ Transport in Plants
 Chapter 36~ Transport in Plants Structural Features Used for Resource Acquistion Roots and stems to do transport of resources Diffusion, active transport, and bulk flow Work in vascular plants to transport
Chapter 36~ Transport in Plants Structural Features Used for Resource Acquistion Roots and stems to do transport of resources Diffusion, active transport, and bulk flow Work in vascular plants to transport
Plant Structure and Function (Ch. 23)
 Plant Structure and Function (Ch. 23) Basic plant anatomy 1 root root tip root hairs Roots Roots anchor plant in soil, absorb minerals & water, & store food fibrous roots (1) mat of thin roots that spread
Plant Structure and Function (Ch. 23) Basic plant anatomy 1 root root tip root hairs Roots Roots anchor plant in soil, absorb minerals & water, & store food fibrous roots (1) mat of thin roots that spread
Plants. Plant Form and Function. Tissue Systems 6/4/2012. Chapter 17. Herbaceous (nonwoody) Woody. Flowering plants can be divided into two groups:
 Monocots Dicots 6/4/2012 Plants Plant Form and Function Chapter 17 Herbaceous (nonwoody) In temperate climates, aerial parts die back Woody In temperate climates, aerial parts persist The Plant Body Functions
Monocots Dicots 6/4/2012 Plants Plant Form and Function Chapter 17 Herbaceous (nonwoody) In temperate climates, aerial parts die back Woody In temperate climates, aerial parts persist The Plant Body Functions
(A) Buds (B) Lateral meristem (C) Apical meristem (D) Stem (E) Trichomes
 AP Biology - Problem Drill 17: Plant Structure Question No. 1 of 10 1. What are hair-like outgrowths that protect and absorb nutrients? Question #01 (A) Buds (B) Lateral meristem (C) Apical meristem (D)
AP Biology - Problem Drill 17: Plant Structure Question No. 1 of 10 1. What are hair-like outgrowths that protect and absorb nutrients? Question #01 (A) Buds (B) Lateral meristem (C) Apical meristem (D)
2014 Pearson Education, Inc. 1. Light. Sugar O 2 H 2 O. and minerals CO Pearson Education, Inc.
 1 CO 2 O 2 Light ugar O 2 and minerals CO 2 2 Buds 34 42 29 26 31 18 21 13 5 10 23 8 15 28 16 24 hoot apical meristem 2 7 3 20 32 11 19 12 6 4 1 25 17 14 9 40 27 22 1 mm 3 Cell wall Apoplastic route Cytosol
1 CO 2 O 2 Light ugar O 2 and minerals CO 2 2 Buds 34 42 29 26 31 18 21 13 5 10 23 8 15 28 16 24 hoot apical meristem 2 7 3 20 32 11 19 12 6 4 1 25 17 14 9 40 27 22 1 mm 3 Cell wall Apoplastic route Cytosol
CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-11 TRANSPORT IN PLANTS
 CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-11 TRANSPORT IN PLANTS Plant transport various substance like gases, minerals, water, hormones, photosynthetes and organic solutes to short distance
CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-11 TRANSPORT IN PLANTS Plant transport various substance like gases, minerals, water, hormones, photosynthetes and organic solutes to short distance
UNIT 6 - STRUCTURES OF FLOWERING PLANTS & THEIR FUNCTIONS
 6.1 Plant Tissues A tissue is a group of cells with common function, structures or both. In plants we can find 2 types of tissues: Meristem Permanent tissues Meristem is found in regions with continuous
6.1 Plant Tissues A tissue is a group of cells with common function, structures or both. In plants we can find 2 types of tissues: Meristem Permanent tissues Meristem is found in regions with continuous
The Petiolar Structure of Christella dentata (Forssk.) Brownsey & Jermy (Thelypteridaceae, Pteridophyta)
 Ethnobotanical Leaflets 12: 96-102. 2008. The Petiolar Structure of Christella dentata (Forssk.) Brownsey & Jermy (Thelypteridaceae, Pteridophyta) KAMINI SRIVASTAVA, M.Sc, D.Phil Department of Botany,
Ethnobotanical Leaflets 12: 96-102. 2008. The Petiolar Structure of Christella dentata (Forssk.) Brownsey & Jermy (Thelypteridaceae, Pteridophyta) KAMINI SRIVASTAVA, M.Sc, D.Phil Department of Botany,
Chapter 29. Table of Contents. Section 1 Plant Cells and Tissues. Section 2 Roots. Section 3 Stems. Section 4 Leaves. Plant Structure and Function
 Plant Structure and Function Table of Contents Section 1 Plant Cells and Tissues Section 2 Roots Section 3 Stems Section 4 Leaves Section 1 Plant Cells and Tissues Objectives Describe the three basic types
Plant Structure and Function Table of Contents Section 1 Plant Cells and Tissues Section 2 Roots Section 3 Stems Section 4 Leaves Section 1 Plant Cells and Tissues Objectives Describe the three basic types
-Each asexual organs. -Anchors the plant -Absorbs water and minerals -Stores sugars and starches
 Plants are made up of: -organs, tissues, and cells The three major plant organs are: -Roots, stems, and leaves -Each asexual organs Plants have a Root System beneath the ground that us a multicellular
Plants are made up of: -organs, tissues, and cells The three major plant organs are: -Roots, stems, and leaves -Each asexual organs Plants have a Root System beneath the ground that us a multicellular
II. SIMPLE TISSUES Bot 404--Fall A. Introduction to Tissues (DIAGRAM allow a full page)
 II. SIMPLE TISSUES Bot 404--Fall 2004 A. Introduction to Tissues (DIAGRAM allow a full page) B. Definitions Adaxial = facing the axil; upper surface of leaf Abaxial = facing away from the axil; lower surface
II. SIMPLE TISSUES Bot 404--Fall 2004 A. Introduction to Tissues (DIAGRAM allow a full page) B. Definitions Adaxial = facing the axil; upper surface of leaf Abaxial = facing away from the axil; lower surface
Primary Internal structure & Normal Secondary growth in Sunflower stem
 Primary Internal structure & Normal Secondary growth in Sunflower stem B. Sc. II - Botany Dr. (Miss) Kalpana R. Datar Assistant Professor DEPARTMENT OF BOTANY Willingdon College, Sangli. kalpana_datar@yahoo.com.
Primary Internal structure & Normal Secondary growth in Sunflower stem B. Sc. II - Botany Dr. (Miss) Kalpana R. Datar Assistant Professor DEPARTMENT OF BOTANY Willingdon College, Sangli. kalpana_datar@yahoo.com.
PLANT STRUCTURE: PARTS (ORGANS) Roots Leaves Stems
 PLANT STRUCTURE: PARTS (ORGANS) Roots Leaves Stems ROOTS El Hiquieron. Strangulating Plant Ficusjimenezii The trees you see growing on the wall are the Higueron. The Higueronsare plants that can grow in
PLANT STRUCTURE: PARTS (ORGANS) Roots Leaves Stems ROOTS El Hiquieron. Strangulating Plant Ficusjimenezii The trees you see growing on the wall are the Higueron. The Higueronsare plants that can grow in
23 1 Specialized Tissues in Plants Slide 1 of 34
 23 1 Specialized Tissues in Plants 1 of 34 Seed Plant Structure The three principal organs of seed plants are roots, stems, and leaves. These organs perform functions such as the transport of nutrients,
23 1 Specialized Tissues in Plants 1 of 34 Seed Plant Structure The three principal organs of seed plants are roots, stems, and leaves. These organs perform functions such as the transport of nutrients,
Biology 1030 Winter 2009
 Meeting Tissue Needs II Chapter 36 (738-755) Chapter 37 (756-770) Cellular Currency Plants harvest solar energy Photosynthesis Produces sugars Proteins, nucleic acids, lipids? H 2 O CO 2 Plants cells still
Meeting Tissue Needs II Chapter 36 (738-755) Chapter 37 (756-770) Cellular Currency Plants harvest solar energy Photosynthesis Produces sugars Proteins, nucleic acids, lipids? H 2 O CO 2 Plants cells still
Plant Anatomy AP Biology
 Plant Anatomy 2006-2007 Basic plant anatomy 1 root root tip root hairs Roots 1 Roots anchor plant in soil, absorb minerals & water, & store food fibrous roots (1) mat of thin roots that spread out monocots
Plant Anatomy 2006-2007 Basic plant anatomy 1 root root tip root hairs Roots 1 Roots anchor plant in soil, absorb minerals & water, & store food fibrous roots (1) mat of thin roots that spread out monocots
Chapter 29: Plant Tissues
 Chapter 29: Plant Tissues Shoots and Roots Shoots (Leaves and Stem) Produce food by photosynthesis Carry out reproductive functions Roots Anchor the plant Penetrate the soil and absorb water and dissolved
Chapter 29: Plant Tissues Shoots and Roots Shoots (Leaves and Stem) Produce food by photosynthesis Carry out reproductive functions Roots Anchor the plant Penetrate the soil and absorb water and dissolved
Topic 2: Plants Ch. 16,28
 Topic 2: Plants Ch. 16,28 Characteristics of Plants p. 316 1. Multicellular eukaryotic organisms 2. Composed of tissues, organs and organ systems. 3. Cell walls made of cellulose. 4. Store energy as starch.
Topic 2: Plants Ch. 16,28 Characteristics of Plants p. 316 1. Multicellular eukaryotic organisms 2. Composed of tissues, organs and organ systems. 3. Cell walls made of cellulose. 4. Store energy as starch.
Chapter 21: Plant Structure & Function
 Chapter 21: Plant Structure & Function Chapter 21: Plant Structure & Function All organisms must: Take in certain materials, e.g. O 2, food, drink Eliminate other materials, e.g. CO 2, waste products Chapter
Chapter 21: Plant Structure & Function Chapter 21: Plant Structure & Function All organisms must: Take in certain materials, e.g. O 2, food, drink Eliminate other materials, e.g. CO 2, waste products Chapter
