Assembly Dynamics of FtsZ Rings in Bacillus subtilis and Escherichia coli and Effects of FtsZ-Regulating Proteins
|
|
|
- Harry Ross
- 5 years ago
- Views:
Transcription
1 JOURNAL OF BACTERIOLOGY, Sept. 2004, p Vol. 186, No /04/$ DOI: /JB Copyright 2004, American Society for Microbiology. All Rights Reserved. Assembly Dynamics of FtsZ Rings in Bacillus subtilis and Escherichia coli and Effects of FtsZ-Regulating Proteins David E. Anderson, 1 Frederico J. Gueiros-Filho, 2 and Harold P. Erickson 1 * Department of Cell Biology, Duke University Medical Center, Durham, North Carolina, 1 and Department of Molecular and Cellular Biology, Harvard University, Cambridge, Massachusetts 2 Received 3 March 2004/Accepted 26 May 2004 FtsZ is the major cytoskeletal component of the bacterial cell division machinery. It forms a ring-shaped structure (the Z ring) that constricts as the bacterium divides. Previous in vivo experiments with green fluorescent protein-labeled FtsZ and fluorescence recovery after photobleaching have shown that the Escherichia coli Z ring is extremely dynamic, continually remodeling itself with a half time of 30 s, similar to microtubules in the mitotic spindle. In the present work, under different experimental conditions, we have found that the half time for fluorescence recovery of E. coli Z rings is even shorter ( 9 s). As before, the turnover appears to be coupled to GTP hydrolysis, since the mutant FtsZ84 protein, with reduced GTPase in vitro, showed an 3-fold longer half time. We have also extended the studies to Bacillus subtilis and found that this species exhibits equally rapid dynamics of the Z ring (half time, 8 s). Interestingly, null mutations of the FtsZ-regulating proteins ZapA, EzrA, and MinCD had only modest effects on the assembly dynamics. This suggests that these proteins do not directly regulate FtsZ subunit exchange in and out of polymers. In B. subtilis, only 30 to 35% of the FtsZ protein was in the Z ring, from which we conclude that a Z ring only 2 or 3 protofilaments thick can function for cell division. FtsZ is a structural homolog of tubulin found in virtually all prokaryotes, where it is the major cytoskeletal protein in cell division. It forms a ring structure (the Z ring) at the midpoint of the cell and, for both Escherichia coli and Bacillus subtilis, is required for the recruitment of all of the subsequent proteins involved in the synthesis of the new septum (7). FtsZ has many features in common with eukaryotic tubulin, including a very similar tertiary structure, GTPase activity, and the ability to assemble into protofilaments in vitro (6, 14, 19, 20, 30). It is generally assumed that the Z ring in vivo consists of some arrangement of polymerized FtsZ protofilaments. Proper localization of the Z ring to mid-cell is accomplished by at least two mechanisms, (i) nucleoid occlusion and (ii) inhibition of polar FtsZ assembly by the Min system (MinCD and DivIVA in B. subtilis and MinCDE in E. coli) (7, 11, 18). In addition to the Min proteins, other potential regulators of FtsZ polymerization have been identified. In B. subtilis, lack of the negative regulator EzrA lowers the FtsZ concentration required for Z-ring formation in vivo and gives rise to extraneous, mislocalized FtsZ structures (13). In addition, a positive regulator, ZapA, was found to promote FtsZ polymerization and bundling in vitro, and deletion of its gene led to death of cells with abnormally low FtsZ levels (10). Recent results have shown that the Z ring is a surprisingly dynamic structure (28), with subunits turning over at a rate similar to that of microtubules in mitotic spindles of eukaryotic cells (2, 25). However, it was unclear whether this dynamic behavior is unique to E. coli or occurs in other species. Another question is whether the rapid dynamics are intrinsic to * Corresponding author. Mailing address: Box 3709, Duke University Medical Center, Durham, NC Phone: (919) Fax: (919) h.erickson@cellbio.duke.edu. Present address: Departamento de Bioquimica, Instituto de Quimica, Universidade de Sao Paulo, Sao Paulo, SP , Brazil. FtsZ assembly or are regulated by other proteins, in particular, the proteins described above. We have therefore extended our fluorescence recovery after photobleaching (FRAP) studies to examine Z-ring dynamics in another species, B. subtilis, and have investigated the effect of known FtsZ-regulating proteins. MATERIALS AND METHODS Bacterial strains and plasmids. The E. coli and B. subtilis strains used in this study are listed in Table 1. The B. subtilis strains were all derivatives of the wild-type PY79 strain and contained an integrated chromosomal copy of ftsz-gfp (in addition to the genomic ftsz gene) under the control of the isopropyl- -Dthiogalactopyranoside (IPTG)-inducible P spac promoter. Mutants were constructed by transforming P spac -ftsz-gfp strain JDB401 with genomic DNA from strains PL990 ( mincd::spc) (12a), PL867 ( ezra::spc) (13), FG345 ( zapayshb::tet) (10), and FG196 ( mind::kan). The E. coli strains contained either plasmid pjsb150 (ftsz-gfp) or pjsb151 (ftsz84-gfp). Plasmids pjsb150 and pjsb151 are derivatives of pjsb2, an expression plasmid based on pbad18 but containing an altered multicloning site and a less efficient Shine-Dalgarno sequence and with the ampicillin resistance replaced by chloramphenicol resistance (27). Media and growth conditions. For the B. subtilis strains, overnight cultures were diluted 100- or 200-fold in Luria-Bertani (LB) medium or LB plus 5 g of chloramphenicol per ml. They were allowed to recover for 1 h at 37 C with shaking and then induced with 50 M IPTG until mid-log phase (2 to 3 h) prior to viewing in the microscope. E. coli BW27783(pJSB150) was grown similarly, but in LB plus 34 g of chloramphenicol per ml, and was induced with to % arabinose. Strain WM1032(pJSB150) was diluted and grown in LB plus 30 g of kanamycin per ml plus 34 g of chloramphenicol per ml at room temperature ( 24 C) until early log phase (optical density, 0.05 to 0.1) and then induced with 0.001% arabinose for 2 to 3 h before microscopy. To avoid salt rescue of the ftsz84 phenotype, strain JFL101(pJSB151) was grown in MOPS medium (28) containing 0.5% glycerol and 34 g of chloramphenicol per ml at 30 C until early log phase (optical density, 0.05 to 0.1) and then induced with 0.001% arabinose for 2 to 3 h before microscopy. For all strains, the final 30 min or more of shaking prior to microscopy was done at room temperature. Also, during FRAP experiments requiring multiple slides, cultures were rediluted with continued shaking at room temperature in order to maintain a log-phase culture. To check that the green fluorescent protein (GFP) remained attached to the FtsZ protein, we performed Western blot assays with a polyclonal anti-gfp antibody (Clontech). The B. subtilis fusion protein showed no visible degrada- 5775
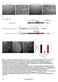
2 5776 ANDERSON ET AL. J. BACTERIOL. TABLE 1. Bacterial strains used in this study Strain Genotype Reference Source B. subtilis JDB401 amye::p spac -ftsz-gfp cat 1 J. Dworkin FG487 zapa-yshb::tet amye::p spac -ftsz-gfp cat This study F. Gueiros-Filho FG489 ezra::spc amye::p spac -ftsz-gfp cat This study F. Gueiros-Filho FG522 mind::kan amye::p spac -ftsz-gfp cat This study F. Gueiros-Filho FG524 mincd::spc amye::p spac -ftsz-gfp cat This study F. Gueiros-Filho E. coli BW27783 laci q rrnb3 lacz4787 hsdr514 (arabad)567 (rhabad)568 (arafgh) 12 J. Keasling ( araep P CP8 -arae) JFL101 F ilv deo ara(am) lac-125(am) galu42(am) tyrt(supf tsa81 ) reca ftsz84(ts) 16 J. Lutkenhaus WM1032 MG1655 lac mincde::kan 29 W. Margolin tion, and in E. coli 5% of the GFP was cleaved off. This is an important control for our measurement of the relative amounts of FtsZ in the ring and cytoplasm. Microscopy and FRAP. Slides were prepared by (i) melting a solution of 1% agarose in MOPS medium 0.5% glycerol, (ii) pipetting 50 l onto a siliconized glass slide with two pieces of laboratory tape as spacers, and (iii) dropping a plain glass slide on top to create a thin agarose pad. After separating the two slides, 5 l of culture was pipetted onto the agarose pad on the plain glass slide and then covered with a number 1 coverslip that was fixed to the slide with 2 drops of melted Valap (1:1:1 petrolatum-lanolin-paraffin). Cells were imaged and FRAP experiments were performed on a Nikon TE300 inverted fluorescence microscope equipped with a laser source with Metamorph software as previously described (17, 28). Unless otherwise noted, all images and FRAP measurements were made at room temperature (24 to 26 C). Exposures were 350 to 400 ms, and laser bleach pulses were 35 to 45 ms. Time series were typically taken over 1 to 5 min with exposures every 2 to 10 s, depending on the speed of recovery. Measurements of integrated fluorescence intensity in the bleached and other regions at each time point were made in Metamorph and exported to Excel For each measured region, background intensity was subtracted and a correction factor was applied for overall photobleaching of the sample during observation (determined from intensity over time of a control cell in the same field). Recovery half times were determined by performing a least-squares fit of the intensity of the bleached region over time to the single-exponential equation F F(t) [F F 0 ] e kt, where F is the fluorescence of the bleached region after maximal recovery, F(t) isthefluorescence at time t, and F 0 is the initial fluorescence just after bleaching (t 0). Fitting was performed with the Solver function of Excel and allowing F, F 0, and the half-time, (t 1/2 t 1/2 ln 2/k), to vary. RESULTS Assembly dynamics of B. subtilis and E. coli Z rings are equally rapid. In order to test whether the rapid subunit exchange observed in E. coli Z rings also occurs in other bacterial species, we performed FRAP experiments with B. subtilis strain JDB401, which expresses an FtsZ-GFP fusion at relatively low levels, along with the normal level of endogenous FtsZ (13). Figure 1A shows selected time points of a typical FRAP image series in which we bleached one-half of the Z ring, and Fig. 1B shows the corresponding time course of fluorescence recovery in the bleached region. Fitting the corrected fluorescence intensity to the equation given in Materials and Methods gave a half time of recovery of 7.0 s for the series shown. This shows that Z rings of other bacterial species do in fact exhibit extremely rapid dynamics. The average half time of recovery for JDB401 cells was 8 3s(n 19). Because the 8-s half time observed for B. subtilis was much shorter than the 30-s half time previously reported for E. coli, we also performed FRAP with E. coli BW27783(pJSB150) cells under the same conditions (agarose pad immobilization, LB medium, 25 C). E. coli BW27783 cells were chosen because of their more uniform level of expression from pbad within a population of E. coli cells (12). A typical FRAP time series for these cells is shown in Fig. 2. Surprisingly, the E. coli Z rings exhibited the same rapid turnover as those of B. subtilis, with an average half time of 9 3s(n 10). This exchange is about threefold faster than that observed previously for E. coli (28). The different exchange rates in the two studies could not be wholly attributed to any one factor, but the method of immobilization (agarose pad in the present study, polylysine-coated coverslips in the previous one) had the greatest effect (data not shown). The exact method of data analysis may also be a factor. A contrast-enhanced movie showing disassembly and reassembly of Z rings in dividing E. coli BW27783(pJSB150) cells at 37 C can be accessed at /Erickson/. The overall time scale is 10 min, with frames taken at 10-s intervals. An interesting feature of this movie is that individual FtsZ-GFP foci can be seen translocating across the cells, into and out of the constricting and reforming Z rings. We also found, on the basis of integrated fluorescence intensities in unbleached cells, that the proportion of FtsZ protein in the Z ring is the same, approximately 30 to 35%, in both E. coli and B. subtilis cells, and we did not observe any obvious correlation between the degree of constriction of rings and their rate of recovery (data not shown). Because of the size of the focused laser beam and the small volume of bacterial cells, we unavoidably bleached a significant portion of the FtsZ-GFP pool with the laser photobleaching pulse. Thus, on the basis of this study, we are not able to draw any conclusions about percent recovery and mobile or immobile fractions. The graphs of fluorescence recovery over time in Fig. 1 to 4 are not normalized. Effect of FtsZ-regulating proteins on Z-ring turnover. To determine whether the rapid dynamics observed are due to the action of known FtsZ-interacting and -regulating proteins, we performed FRAP with strains containing null mutations of several genes. We will subsequently refer to E. coli BW27783 and B. subtilis JDB401 as the wild-type strains since they served as our control strains for comparison with the mutants. The first mutant that we tested was a B. subtilis strain, FG487, that lacks the protein ZapA, a positive FtsZ assembly factor (10). As previously reported, the FtsZ structures in this mutant appeared normal. We had expected that the absence of the stabilizing factor might increase assembly dynamics, but the average recovery half time was similar to that of the wild
3 VOL. 186, 2004 IN VIVO DYNAMICS OF B. SUBTILIS AND E. COLI FtsZ RINGS 5777 factor of 2. The final two B. subtilis mutant strains examined were FG522, which lacks MinD, and FG524, which lacks both MinC and MinD. As expected, these cells showed extra, mislocalized Z rings and some double rings in FG524 (Fig. 4). Similar to the results obtained with the zapa and ezra mutant strains, neither FG522 nor FG524 showed a large difference in Z-ring stability, with average recovery half times of 12 6(n 8) and 10 2(n 5) s. In general for all of these strains, we chose single, medial rings for the FRAP measurements. In the case of the ezra and mincd strains, we also examined several mislocalized rings, and these showed dynamic behavior similar to that of the medial rings (data not shown). FIG. 1. FRAP of an FtsZ ring in wild-type B. subtilis cells. (A) Time-lapse series of fluorescence images showing the time course of recovery. The arrow shows the half-ring to be bleached. (B) Intensity of the photobleached region over time. The data points have been corrected for background and photobleaching during the observation period. The points represent the fluorescence data, and the solid line is the predicted recovery curve given the first-order rate constant, k, determined by least-squares fitting of the data to the equation given in Materials and Methods. The recovery half time for this series was 7.0 s. type, at 10 4s(n 12). The next strain we examined was FG489, shown in Fig. 3, which lacks the negative regulator of ring assembly, EzrA (13). As previously reported, cells often showed extraneous, mislocalized rings and also residual Z-ring material at previous division sites. In this case, the absence of the destabilizing factor did give a longer recovery half time of 14 5s(n 13), which was statistically significant (Table 2). However, the absolute difference is not very large, less than a FIG. 2. FRAP of an FtsZ ring in wild-type E. coli BW27783 (pjsb150) cells. (A) Time-lapse series of fluorescence images. (B) Intensity of the photobleached region over time. The half time of recovery for the series shown was 8.9 s.
4 5778 ANDERSON ET AL. J. BACTERIOL. type. Table 2 summarizes the average recovery times for all of the strains and their statistical significances. Finally, we attempted to measure FRAP of the cytoplasmic pool of FtsZ to gain information about its polymerization state. The recovery was too fast for accurate measurement. When a spot was bleached in the cytoplasm on one side of the bacterium, that side showed a uniform decrease in fluorescence in 0.5 s, and the fluorescence was equilibrated on the two sides in 1 s (data not shown). DISCUSSION The rapid assembly dynamics of the Z ring were virtually identical for E. coli and B. subtilis, suggesting that rapid exchange of subunits is a conserved property of Z rings in most or all bacterial species. This continuous turnover of subunits may therefore play a role in the mechanism of Z-ring function. It is, however, important to remember that the mutant FtsZ84 protein appears to divide normally at 30 C, despite a threefold reduction in dynamics. This suggests that the dynamics of FtsZ have evolved to exceed what may be essential for division and that some other factor is rate limiting for division and cell cycle timing. An important observation is that the fraction of FtsZ incorporated into the ring, 30%, is the same for both species. On the basis of an estimation of 15,000 FtsZ molecules per E. coli cell (15), if one-third of that protein is in the ring, the E. coli Z ring would be an average of 6 protofilaments wide (28). In a cell ready for division (about 1.4 times as long as an average cell) it could be just over 8 protofilaments thick. However, in B. subtilis, it has been estimated that there are only 5,000 FtsZ molecules per cell (8). Thus, in B. subtilis we estimate the Z ring to be only 2 protofilaments wide on average, or about 3 protofilaments at the time of constriction. E. coli strain MC1061 was likewise reported to have only 5,000 molecules of FtsZ when growing in minimal medium (24). Assuming that it also has only 30% of its FtsZ in the Z ring, this E. coli B/r K strain can achieve cell division with a ring only 2 to 3 protofilaments thick. It has been hypothesized that Z-ring dynamics are the result of a balance of stabilizing and destabilizing factors (10, 22), and in this study we tested several potential modulators of this FIG. 3. FRAP of FtsZ rings in B. subtilis cells lacking ezra. (A) Time-lapse series of fluorescence images. (B) Intensity of the photobleached region over time. Recovery half time, 13.0 s. We also investigated Z-ring stability in two mutant E. coli strains. The first, JFL101(pJSB151), carries the ftsz84 mutation (G105S), which was shown previously to reduce the turnover of the Z ring by a factor of about 10, consistent with its reduced GTPase activity in vitro (28). Under our present experimental conditions this strain gave an approximately threeto fourfold decrease in the turnover of Z rings (t 1/ s; n 9). The second E. coli strain, WM1032(pJSB150), lacks the MinC, MinD, and MinE proteins. Similar to B. subtilis, most of these cells had extraneous, polar Z-ring structures. In this case, however, the absence of the MinCDE system had a more pronounced effect on Z-ring turnover, with a recovery half time of 19 3s(n 3), approximately twice that of the wild TABLE 2. Summary of B. subtilis and E. coli FRAP measurements Strain Recovery half time SD (s) B. subtilis JDB FG487 ( zapa) FG489 ( ezra) a FG522 ( mind) FG524 ( mincd) b E. coli BW JFL101 ftsz c WM1032 ( mincde) d a Significantly different from wild type (P 0.001). b Significantly different from wild type (P 0.05). c Significantly different from wild type (P ). d Significantly different from wild type (P ).
5 VOL. 186, 2004 IN VIVO DYNAMICS OF B. SUBTILIS AND E. COLI FtsZ RINGS 5779 subtilis also produced only a small increase in the turnover half time, suggesting that direct inhibition of FtsZ assembly in the Z ring is not a primary function of B. subtilis MinC. In E. coli, however, Z rings of the mincde strain were stabilized approximately twofold, almost as much as those formed of FtsZ84 with its decreased GTPase activity. This suggests that in E. coli, MinC(DE) might directly interact with already formed Z rings to inhibit or actively disassemble FtsZ polymers. The recovery times showed a large variation for each strain examined (Fig. 5), with half times ranging from 4 s to more than 20 s even in the wild-type strains. The few cells with long recovery times ( 20 s) were considered possibly unhealthy and FIG. 4. FRAP of FtsZ rings in B. subtilis cells lacking mincd. (A) Time-lapse series of fluorescence images. (B) Intensity of the photobleached region over time. Recovery half time, 10.7 s. behavior. It is not very surprising that assembly dynamics are not altered by a lack of ZapA, given that the null strain has almost no phenotype in terms of septation. It also has only a small effect on the distribution of FtsZ between the ring and the cytoplasm (wild type, 30% 4%; zapa mutant, 25% 4%; F. Gueiros-Filho, unpublished observations). However, we have not tested the effect of zapa deletion in the absence of EzrA or with lowered levels of FtsZ. EzrA, which has a more noticeable phenotype when deleted, did affect the dynamics more significantly and in the direction expected. However, recovery half times from this strain were also more variable and the small absolute difference from the wild type is of questionable significance. Removing the Min proteins from B. FIG. 5. Histograms of Z-ring fluorescence recovery times for wildtype (wt) versus mutant cells of B. subtilis (Bs) (A) and E. coli (Ec) (B).
6 5780 ANDERSON ET AL. J. BACTERIOL. were excluded from the wild-type average recovery times. Given this large variability in the populations of wild-type cells, we suggest that the slightly altered dynamics in the mutant B. subtilis strains may not represent an important functional difference. The simplest interpretation is that the dynamic behavior of the Z ring is an intrinsic property of FtsZ polymers and is only slightly altered by known regulatory proteins. It is notable, however, that E. coli strains with min deleted showed a more pronounced increase in recovery half times. This may represent a direct effect on Z-ring stability, but the stabilization was still only a factor of 2. We tried to address the question of whether cytoplasmic FtsZ is monomeric or assembled into protofilaments. In vitro studies with E. coli FtsZ indicate a critical concentration of 1 M under physiological solution conditions (9), so we expect most of the cytoplasmic FtsZ ( 8 M for E. coli, assuming that 70% of the total is in the cytoplasm) to be assembled into protofilaments. When we did FRAP with cytoplasmic FtsZ in strain BW27783, we found that it was completely re-equilibrated across the bleached half of the cell in 0.5 s and across both sides of the cell in 1 s. This seems somewhat slower than the 0.5-s time for equilibration of monomeric GFP (4), but our instrument did not have a time resolution that could accurately measure a diffusion coefficient. If the equilibration time is slower than for GFP it would suggest that cytoplasmic FtsZ is assembled into protofilaments, but with the present data we cannot make a clear distinction between monomers and short polymers of FtsZ. We attempted to use fluorescence correlation spectroscopy to determine a diffusion coefficient for the cytoplasmic form(s) of FtsZ, but we found that the cellular FtsZ-GFP pool was photobleached too rapidly to obtain useful measurements. The 8- to 9-s half time we observed for turnover of the FtsZ ring in B. subtilis and E. coli is similar to the fastest turnover in the eukaryotic cytoskeleton, specifically, the mitotic apparatus of cultured mammalian cells (26). Turnover of tubulin is generated by dynamic instability, which involves extended cycles of assembly and disassembly at the microtubule ends (3, 5). The assembly dynamics of FtsZ are much less well understood. Recent work in our laboratory suggests that FtsZ protofilaments assemble cooperatively by addition of subunits to the ends, with an assembly time similar to the in vivo turnover rate (Y. Chen et al., submitted for publication). Novel experiments are needed to confirm this and establish a detailed assembly mechanism. Finally, the rapid dynamics has implications for the structure of the Z ring. We have estimated that the Z ring is approximately 6 to 8 or 2 to 3 protofilaments wide for E. coli and B. subtilis, but we have not resolved whether there are a few very long protofilaments or many short ones. We will postulate that assembly occurs by addition of subunits to the ends at a rate of 5 M 1 s 1. This is the diffusion-limited rate for proteinprotein association (21) and is in agreement with our recent in vitro kinetic studies (Chen et al., submitted). If the concentration of free subunits is 1 M, which is the critical concentration determined in vitro (9), then protofilaments would grow at a rate of five subunits per s. This would mean that protofilaments could grow only 45 subunits long during the 9-s turnover half time, or 90 subunits long during the entire recovery time. A protofilament 45 subunits long would be about 200 nm long, much shorter than the 3,000-nm circumference of the bacterium. This estimate of protofilament length is similar to the average protofilament length of 23 subunits for E. coli FtsZ measured in vitro (23). Thus, both in vitro studies and the rapid assembly dynamics observed in vivo suggest that the Z ring is an assembly of many short protofilaments. ACKNOWLEDGMENTS This work was supported by NIH grants GM to H. P. Erickson and GM to R. Losick. F. J. Gueiros-Filho was a Helen Hay Whitney Foundation postdoctoral fellow during part of this work. We especially thank the laboratory of E. D. Salmon, University of North Carolina, for the use of the FRAP microscope and R. Losick, Harvard University, for support of F.J.G.-F. and comments on the manuscript. We also thank J. Lutkenhaus, J. D. Keasling, P. A. Levin, and W. Margolin for providing bacterial strains and J. Stricker for construction of pjsb150 and pjsb151. REFERENCES 1. Ben-Yehuda, S., and R. Losick Asymmetric cell division in B. subtilis involves a spiral-like intermediate of the cytokinetic protein FtsZ. Cell 109: Cassimeris, L. U., R. A. Walker, N. K. Pryer, and E. D. Salmon Dynamic instability of microtubules. Bioessays 7: Desai, A., and T. J. Mitchison Microtubule polymerization dynamics. Annu. Rev. Cell Dev. Biol. 13: Elowitz, M. B., M. G. Surette, P. E. Wolf, J. B. Stock, and S. Leibler Protein mobility in the cytoplasm of Escherichia coli. J. Bacteriol. 181: Erickson, H. P., and E. T. O Brien Microtubule dynamic instability and GTP hydrolysis. Annu. Rev. Biophys. Struct. Biol. 21: Erickson, H. P., D. W. Taylor, K. A. Taylor, and D. Bramhill Bacterial cell division protein FtsZ assembles into protofilament sheets and minirings, structural homologs of tubulin polymers. Proc. Natl. Acad. Sci. USA 93: Errington, J., R. A. Daniel, and D. J. Scheffers Cytokinesis in bacteria. Microbiol. Mol. Biol. Rev. 67: Feucht, A., I. Lucet, M. D. Yudkin, and J. Errington Cytological and biochemical characterization of the FtsA cell division protein of Bacillus subtilis. Mol. Microbiol. 40: Gonzalez, J. M., M. Jimenez, M. Velez, J. Mingorance, J. M. Andreu, M. Vicente, and G. Rivas Essential cell division protein FtsZ assembles into one monomer-thick ribbons under conditions resembling the crowded intracellular environment. J. Biol. Chem. 278: Gueiros-Filho, F. J., and R. Losick A widely conserved bacterial cell division protein that promotes assembly of the tubulin-like protein FtsZ. Genes Dev. 16: Harry, E. J Bacterial cell division: regulating Z-ring formation. Mol. Microbiol. 40: Khlebnikov, A., K. A. Datsenko, T. Skaug, B. L. Wanner, and J. D. Keasling Homogeneous expression of the P(BAD) promoter in Escherichia coli by constitutive expression of the low-affinity high-capacity AraE transporter. Microbiology 147: a.Levin, P. A., J. J. Shim, and A. D. Grossman Effect of mincd on FtsZ ring position and polar septation in Bacillus subtilis J. Bacteriol. 180: Levin, P. A., I. G. Kurtser, and A. D. Grossman Identification and characterization of a negative regulator of FtsZ ring formation in Bacillus subtilis. Proc. Natl. Acad. Sci. USA 96: Lowe, J., and L. A. Amos Crystal structure of the bacterial cell-division protein FtsZ. Nature 391: Lu, C., J. Stricker, and H. P. Erickson FtsZ from Escherichia coli, Azotobacter vinelandii, and Thermotoga maritima quantitation, GTP hydrolysis, and assembly. Cell Motil. Cytoskelet. 40: Lutkenhaus, J. F., H. Wolf-Watz, and W. D. Donachie Organization of genes in the ftsa-enva region of the Escherichia coli genetic map and identification of a new fts locus (ftsz). J. Bacteriol. 142: Maddox, P. S., K. S. Bloom, and E. D. Salmon The polarity and dynamics of microtubule assembly in the budding yeast Saccharomyces cerevisiae. Nat. Cell Biol. 2: Margolin, W Spatial regulation of cytokinesis in bacteria. Curr. Opin. Microbiol. 4: Mukherjee, A., K. Dai, and J. Lutkenhaus Escherichia coli cell division protein FtsZ is a guanine nucleotide binding protein. Proc. Natl. Acad. Sci. USA 90: Nogales, E., S. G. Wolf, and K. H. Downing Structure of the alpha beta tubulin dimer by electron crystallography. Nature 391: Northrup, S. H., and H. P. Erickson Kinetics of protein-protein
7 VOL. 186, 2004 IN VIVO DYNAMICS OF B. SUBTILIS AND E. COLI FtsZ RINGS 5781 association explained by Brownian dynamics computer simulation. Proc. Natl. Acad. Sci. USA 89: Romberg, L., and P. A. Levin Assembly dynamics of the bacterial cell division protein FtsZ: poised at the edge of stability. Annu. Rev. Microbiol. 57: Romberg, L., M. Simon, and H. P. Erickson Polymerization of Ftsz, a bacterial homolog of tubulin. is assembly cooperative? J. Biol. Chem. 276: Rueda, S., M. Vicente, and J. Mingorance Concentration and assembly of the division ring proteins FtsZ, FtsA, and ZipA during the Escherichia coli cell cycle. J. Bacteriol. 185: Salmon, E. D., and P. Wadsworth Fluorescence studies of tubulin and microtubule dynamics in living cells, p In D. L. Taylor, A. S. Waggoner, R. F. Murphy, F. Lanni, and R. R. Birge (ed.), Applications of fluorescence in the biomedical sciences. Alan R. Liss, Inc., New York. 26. Saxton, W. M., D. L. Stemple, R. J. Leslie, E. D. Salmon, M. Zavortink, and J. R. McIntosh Tubulin dynamics in cultured mammalian cells. J. Cell Biol. 99: Stricker, J., and H. P. Erickson In vivo characterization of Escherichia coli ftsz mutants: effects on Z-ring structure and function. J. Bacteriol. 185: Stricker, J., P. Maddox, E. D. Salmon, and H. P. Erickson Rapid assembly dynamics of the Escherichia coli FtsZ-ring demonstrated by fluorescence recovery after photobleaching. Proc. Natl. Acad. Sci. USA 99: Sun, Q., and W. Margolin Influence of the nucleoid on placement of FtsZ and MinE rings in Escherichia coli. J. Bacteriol. 183: Wang, X., and J. Lutkenhaus The FtsZ protein of Bacillus subtilis is localized at the division site and has GTPase activity that is dependent upon FtsZ concentration. Mol. Microbiol. 9:
Mechanisms for Precise Positional Information in Bacteria: The Min system in E. coli and B. subtilis
 Mechanisms for Precise Positional Information in Bacteria: The Min system in E. coli and B. subtilis Martin Howard Imperial College London Bacterial Organization Many processes where bacterial cell needs
Mechanisms for Precise Positional Information in Bacteria: The Min system in E. coli and B. subtilis Martin Howard Imperial College London Bacterial Organization Many processes where bacterial cell needs
In Escherichia coli, two systems are known to regulate the
 Dynamic structures in Escherichia coli: Spontaneous formation of MinE rings and MinD polar zones Kerwyn Casey Huang*, Yigal Meir, and Ned S. Wingreen *Department of Physics, Massachusetts Institute of
Dynamic structures in Escherichia coli: Spontaneous formation of MinE rings and MinD polar zones Kerwyn Casey Huang*, Yigal Meir, and Ned S. Wingreen *Department of Physics, Massachusetts Institute of
Cytokinesis in Bacteria
 MICROBIOLOGY AND MOLECULAR BIOLOGY REVIEWS, Mar. 2003, p. 52 65 Vol. 67, No. 1 1092-2172/03/$08.00 0 DOI: 10.1128/MMBR.67.1.52 65.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved.
MICROBIOLOGY AND MOLECULAR BIOLOGY REVIEWS, Mar. 2003, p. 52 65 Vol. 67, No. 1 1092-2172/03/$08.00 0 DOI: 10.1128/MMBR.67.1.52 65.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved.
Changes in Min oscillation pattern before and after cell birth
 JB Accepts, published online ahead of print on June 00 J. Bacteriol. doi:0./jb.00-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved. Juarez &
JB Accepts, published online ahead of print on June 00 J. Bacteriol. doi:0./jb.00-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved. Juarez &
Changes in the Min Oscillation Pattern before and after Cell Birth #
 JOURNAL OF BACTERIOLOGY, Aug. 2010, p. 4134 4142 Vol. 192, No. 16 0021-9193/10/$12.00 doi:10.1128/jb.00364-10 Copyright 2010, American Society for Microbiology. All Rights Reserved. Changes in the Min
JOURNAL OF BACTERIOLOGY, Aug. 2010, p. 4134 4142 Vol. 192, No. 16 0021-9193/10/$12.00 doi:10.1128/jb.00364-10 Copyright 2010, American Society for Microbiology. All Rights Reserved. Changes in the Min
Stochastic simulations
 Stochastic simulations Application to molecular networks Literature overview Noise in genetic networks Origins How to measure and distinguish between the two types of noise (intrinsic vs extrinsic)? What
Stochastic simulations Application to molecular networks Literature overview Noise in genetic networks Origins How to measure and distinguish between the two types of noise (intrinsic vs extrinsic)? What
arxiv:q-bio/ v1 [q-bio.sc] 29 Nov 2005
![arxiv:q-bio/ v1 [q-bio.sc] 29 Nov 2005 arxiv:q-bio/ v1 [q-bio.sc] 29 Nov 2005](/thumbs/92/110105519.jpg) arxiv:q-bio/0511049v1 [q-bio.sc] 29 Nov 2005 A stochastic model of Min oscillations in Escherichia coli and Min protein segregation during cell division Filipe Tostevin and Martin Howard Department of
arxiv:q-bio/0511049v1 [q-bio.sc] 29 Nov 2005 A stochastic model of Min oscillations in Escherichia coli and Min protein segregation during cell division Filipe Tostevin and Martin Howard Department of
The polymerization mechanism of the bacterial cell division protein FtsZ
 FEBS Letters 506 (2001) 6^10 FEBS 25254 Minireview The polymerization mechanism of the bacterial cell division protein FtsZ Dirk-Jan Sche ers 1, Arnold J.M. Driessen* Department of Microbiology, Groningen
FEBS Letters 506 (2001) 6^10 FEBS 25254 Minireview The polymerization mechanism of the bacterial cell division protein FtsZ Dirk-Jan Sche ers 1, Arnold J.M. Driessen* Department of Microbiology, Groningen
the noisy gene Biology of the Universidad Autónoma de Madrid Jan 2008 Juan F. Poyatos Spanish National Biotechnology Centre (CNB)
 Biology of the the noisy gene Universidad Autónoma de Madrid Jan 2008 Juan F. Poyatos Spanish National Biotechnology Centre (CNB) day III: noisy bacteria - Regulation of noise (B. subtilis) - Intrinsic/Extrinsic
Biology of the the noisy gene Universidad Autónoma de Madrid Jan 2008 Juan F. Poyatos Spanish National Biotechnology Centre (CNB) day III: noisy bacteria - Regulation of noise (B. subtilis) - Intrinsic/Extrinsic
CHAPTER 13 PROKARYOTE GENES: E. COLI LAC OPERON
 PROKARYOTE GENES: E. COLI LAC OPERON CHAPTER 13 CHAPTER 13 PROKARYOTE GENES: E. COLI LAC OPERON Figure 1. Electron micrograph of growing E. coli. Some show the constriction at the location where daughter
PROKARYOTE GENES: E. COLI LAC OPERON CHAPTER 13 CHAPTER 13 PROKARYOTE GENES: E. COLI LAC OPERON Figure 1. Electron micrograph of growing E. coli. Some show the constriction at the location where daughter
Plasmid Segregation: Is a Total Understanding Within Reach?
 Plasmid Segregation: Is a Total Understanding Within Reach? The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation Published
Plasmid Segregation: Is a Total Understanding Within Reach? The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation Published
A redundant function for the N-terminal tail of Ndc80 in kinetochore-microtubule interaction in Saccharomyces cerevisiae.
 Genetics: Published Articles Ahead of Print, published on July 30, 2012 as 10.1534/genetics.112.143818 A redundant function for the N-terminal tail of Ndc80 in kinetochore-microtubule interaction in Saccharomyces
Genetics: Published Articles Ahead of Print, published on July 30, 2012 as 10.1534/genetics.112.143818 A redundant function for the N-terminal tail of Ndc80 in kinetochore-microtubule interaction in Saccharomyces
7.06 Problem Set #4, Spring 2005
 7.06 Problem Set #4, Spring 2005 1. You re doing a mutant hunt in S. cerevisiae (budding yeast), looking for temperaturesensitive mutants that are defective in the cell cycle. You discover a mutant strain
7.06 Problem Set #4, Spring 2005 1. You re doing a mutant hunt in S. cerevisiae (budding yeast), looking for temperaturesensitive mutants that are defective in the cell cycle. You discover a mutant strain
Mechanistic basis of equal plasmid spacing in E. coli: a general mechanism for sizing and spacing
 Mechanistic basis of equal plasmid spacing in E. coli: a general mechanism for sizing and spacing CANES, Kings College London November 11 th, 2015 Martin Howard Computational & Systems Biology John Innes
Mechanistic basis of equal plasmid spacing in E. coli: a general mechanism for sizing and spacing CANES, Kings College London November 11 th, 2015 Martin Howard Computational & Systems Biology John Innes
monomer polymer polymeric network cell
 3.1 Motivation 3.2 Polymerization The structural stability of the cell is provided by the cytoskeleton. Assembling and disassembling dynamically, the cytoskeleton enables cell movement through a highly
3.1 Motivation 3.2 Polymerization The structural stability of the cell is provided by the cytoskeleton. Assembling and disassembling dynamically, the cytoskeleton enables cell movement through a highly
arxiv:q-bio.sc/ v1 13 Apr 2005
 Min-oscillations in Escherichia coli induced by interactions of membrane-bound proteins arxiv:q-bio.sc/0504017 v1 13 Apr 2005 Giovanni Meacci and Karsten Kruse Max-Planck Institut für Physik komplexer
Min-oscillations in Escherichia coli induced by interactions of membrane-bound proteins arxiv:q-bio.sc/0504017 v1 13 Apr 2005 Giovanni Meacci and Karsten Kruse Max-Planck Institut für Physik komplexer
The mechanisms that drive bacterial cell division have been
 Defining the rate-limiting processes of bacterial cytokinesis Carla Coltharp a, Jackson Buss a,1, Trevor M. Plumer a, and Jie Xiao a,2 a Department of Biophysics and Biophysical Chemistry, Johns Hopkins
Defining the rate-limiting processes of bacterial cytokinesis Carla Coltharp a, Jackson Buss a,1, Trevor M. Plumer a, and Jie Xiao a,2 a Department of Biophysics and Biophysical Chemistry, Johns Hopkins
Diffusion of Green Fluorescent Protein in Three Cell Environments in Escherichia Coli
 JOURNAL OF BACTERIOLOGY, May 2006, p. 3442 3448 Vol. 188, No. 10 0021-9193/06/$08.00 0 doi:10.1128/jb.188.10.3442 3448.2006 Copyright 2006, American Society for Microbiology. All Rights Reserved. Diffusion
JOURNAL OF BACTERIOLOGY, May 2006, p. 3442 3448 Vol. 188, No. 10 0021-9193/06/$08.00 0 doi:10.1128/jb.188.10.3442 3448.2006 Copyright 2006, American Society for Microbiology. All Rights Reserved. Diffusion
Supporting Information
 Supporting Information Espinar et al. 1.173/pnas.12169111 SI Materials and Methods Growth Conditions for Microscopy. For the experiments presented in the main text, Bacillus subtilis cells were grown at
Supporting Information Espinar et al. 1.173/pnas.12169111 SI Materials and Methods Growth Conditions for Microscopy. For the experiments presented in the main text, Bacillus subtilis cells were grown at
Evidence for cyclic-di-gmp-mediated signaling pathway in Bacillus subtilis by Chen Y. et al.
 Supplemental materials for Evidence for cyclic-di-gmp-mediated signaling pathway in Bacillus subtilis by Chen Y. et al. 1. Table S1. Strains used in this study 2. Table S2. Plasmids used in this study
Supplemental materials for Evidence for cyclic-di-gmp-mediated signaling pathway in Bacillus subtilis by Chen Y. et al. 1. Table S1. Strains used in this study 2. Table S2. Plasmids used in this study
JBC Papers in Press. Published on February 1, 2012 as Manuscript M interactions between FtsZ filaments are regulated by its C-terminus
 JBC Papers in Press. Published on February 1, 2012 as Manuscript M111.330324 The latest version Lateral is http://www.jbc.org/cgi/doi/10.1074/jbc.m111.330324 interactions between FtsZ filaments are regulated
JBC Papers in Press. Published on February 1, 2012 as Manuscript M111.330324 The latest version Lateral is http://www.jbc.org/cgi/doi/10.1074/jbc.m111.330324 interactions between FtsZ filaments are regulated
ACCEPTED. Why spherical E. coli dies: The inside story. Kevin D. Young. Department of Microbiology and Immunology,
 JB Accepts, published online ahead of print on 28 December 2007 J. Bacteriol. doi:10.1128/jb.01975-07 Copyright 2007, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JB Accepts, published online ahead of print on 28 December 2007 J. Bacteriol. doi:10.1128/jb.01975-07 Copyright 2007, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
Review. Diverse Paths to Midcell: Assembly of the Bacterial Cell Division Machinery. Nathan W. Goehring and Jon Beckwith*
 , Vol. 15, R514 R526, July 12, 2005, 2005 Elsevier Ltd. All rights reserved. DOI 10.1016/j.cub.2005.06.038 Diverse Paths to Midcell: Assembly of the Bacterial Cell Division Machinery Review Nathan W. Goehring
, Vol. 15, R514 R526, July 12, 2005, 2005 Elsevier Ltd. All rights reserved. DOI 10.1016/j.cub.2005.06.038 Diverse Paths to Midcell: Assembly of the Bacterial Cell Division Machinery Review Nathan W. Goehring
Introduction. Gene expression is the combined process of :
 1 To know and explain: Regulation of Bacterial Gene Expression Constitutive ( house keeping) vs. Controllable genes OPERON structure and its role in gene regulation Regulation of Eukaryotic Gene Expression
1 To know and explain: Regulation of Bacterial Gene Expression Constitutive ( house keeping) vs. Controllable genes OPERON structure and its role in gene regulation Regulation of Eukaryotic Gene Expression
doi: /j.jmb J. Mol. Biol. (2009) 390, Available online at COMMUNICATION
 doi:10.1016/j.jmb.2009.05.018 J. Mol. Biol. (2009) 390, 17 25 Available online at www.sciencedirect.com COMMUNICATION Structural and Functional Model for Ionic (K + /Na + ) and ph Dependence of GTPase
doi:10.1016/j.jmb.2009.05.018 J. Mol. Biol. (2009) 390, 17 25 Available online at www.sciencedirect.com COMMUNICATION Structural and Functional Model for Ionic (K + /Na + ) and ph Dependence of GTPase
Helical Macrofiber Formation in Bacillus subtilis: Inhibition by Penicillin G
 JOURNAL OF BACTERIOLOGY, June 1984, p. 1182-1187 0021-9193/84/061182-06$02.00/0 Copyright C 1984, American Society for Microbiology Vol. 158, No. 3 Helical Macrofiber Formation in Bacillus subtilis: Inhibition
JOURNAL OF BACTERIOLOGY, June 1984, p. 1182-1187 0021-9193/84/061182-06$02.00/0 Copyright C 1984, American Society for Microbiology Vol. 158, No. 3 Helical Macrofiber Formation in Bacillus subtilis: Inhibition
Gene expression in prokaryotic and eukaryotic cells, Plasmids: types, maintenance and functions. Mitesh Shrestha
 Gene expression in prokaryotic and eukaryotic cells, Plasmids: types, maintenance and functions. Mitesh Shrestha Plasmids 1. Extrachromosomal DNA, usually circular-parasite 2. Usually encode ancillary
Gene expression in prokaryotic and eukaryotic cells, Plasmids: types, maintenance and functions. Mitesh Shrestha Plasmids 1. Extrachromosomal DNA, usually circular-parasite 2. Usually encode ancillary
Growth Rate-Dependent Regulation of Medial FtsZ Ring Formation
 Growth Rate-Dependent Regulation of Medial FtsZ Ring Formation Richard B. Weart and Petra Anne Levin J. Bacteriol. 2003, 185(9):2826. DOI: 10.1128/JB.185.9.2826-2834.2003. REFERENCES CONTENT ALERTS Updated
Growth Rate-Dependent Regulation of Medial FtsZ Ring Formation Richard B. Weart and Petra Anne Levin J. Bacteriol. 2003, 185(9):2826. DOI: 10.1128/JB.185.9.2826-2834.2003. REFERENCES CONTENT ALERTS Updated
Supporting Information
 Supporting Information López et al. 10.1073/pnas.0810940106 1. Ivey DM, et al. (1993) Cloning and characterization of a putative Ca2 /H antiporter gene from Escherichia coli upon functional complementation
Supporting Information López et al. 10.1073/pnas.0810940106 1. Ivey DM, et al. (1993) Cloning and characterization of a putative Ca2 /H antiporter gene from Escherichia coli upon functional complementation
Chromosome Chr Duplica Duplic t a ion Pixley
 Chromosome Duplication Pixley Figure 4-6 Molecular Biology of the Cell ( Garland Science 2008) Figure 4-72 Molecular Biology of the Cell ( Garland Science 2008) Interphase During mitosis (cell division),
Chromosome Duplication Pixley Figure 4-6 Molecular Biology of the Cell ( Garland Science 2008) Figure 4-72 Molecular Biology of the Cell ( Garland Science 2008) Interphase During mitosis (cell division),
Cell Structure and Function. The Development of Cell Theory
 Cell Structure and Function The Development of Cell Theory Hooke's original microscope, that he used at Oxford University, in 1665. Robert Hooke first examined a thin piece of cork (see below), and coined
Cell Structure and Function The Development of Cell Theory Hooke's original microscope, that he used at Oxford University, in 1665. Robert Hooke first examined a thin piece of cork (see below), and coined
Cellular organization by self organization: mechanisms and
 Cellular organization by self organization: mechanisms and models for Min protein dynamics Martin Howard 1 and Karsten Kruse 2 1 Department of Mathematics, Imperial College London, South Kensington Campus,
Cellular organization by self organization: mechanisms and models for Min protein dynamics Martin Howard 1 and Karsten Kruse 2 1 Department of Mathematics, Imperial College London, South Kensington Campus,
Dynamic Filaments of the Bacterial Cytoskeleton
 I ANRV277-BI75-21 ARI 24 February 2006 2:50 R E V I E W S First published online as a Review in Advance on March 29, 2006 E N C A D V A N Annu. Rev. Biochem. 2006. 75:467 92 The Annual Review of Biochemistry
I ANRV277-BI75-21 ARI 24 February 2006 2:50 R E V I E W S First published online as a Review in Advance on March 29, 2006 E N C A D V A N Annu. Rev. Biochem. 2006. 75:467 92 The Annual Review of Biochemistry
Bi 1x Spring 2014: LacI Titration
 Bi 1x Spring 2014: LacI Titration 1 Overview In this experiment, you will measure the effect of various mutated LacI repressor ribosome binding sites in an E. coli cell by measuring the expression of a
Bi 1x Spring 2014: LacI Titration 1 Overview In this experiment, you will measure the effect of various mutated LacI repressor ribosome binding sites in an E. coli cell by measuring the expression of a
Bio Microbiology - Spring 2014 Learning Guide 04.
 Bio 230 - Microbiology - Spring 2014 Learning Guide 04 http://pessimistcomic.blogspot.com/ Cell division is a part of a replication cycle that takes place throughout the life of the bacterium A septum
Bio 230 - Microbiology - Spring 2014 Learning Guide 04 http://pessimistcomic.blogspot.com/ Cell division is a part of a replication cycle that takes place throughout the life of the bacterium A septum
The Diffusion Coefficient for PGK Folding in Eukaryotic Cells
 Biophysical Journal, Volume 99 Supporting Material The Diffusion Coefficient for PGK Folding in Eukaryotic Cells Apratim Dhar, Simon Ebbinghaus, Zhen Shen, Tripta Mishra, and Martin Gruebele 1 Supplementary
Biophysical Journal, Volume 99 Supporting Material The Diffusion Coefficient for PGK Folding in Eukaryotic Cells Apratim Dhar, Simon Ebbinghaus, Zhen Shen, Tripta Mishra, and Martin Gruebele 1 Supplementary
Bacterial ancestry of actin and tubulin Fusinita van den Ent, Linda Amos and Jan Löwe*
 634 Bacterial ancestry of actin and tubulin Fusinita van den Ent, Linda Amos and Jan Löwe* The structural and functional resemblance between the bacterial cell-division protein FtsZ and eukaryotic tubulin
634 Bacterial ancestry of actin and tubulin Fusinita van den Ent, Linda Amos and Jan Löwe* The structural and functional resemblance between the bacterial cell-division protein FtsZ and eukaryotic tubulin
Neurite formation & neuronal polarization. The cytoskeletal components of neurons have characteristic distributions and associations
 Mechanisms of neuronal migration & Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 The cytoskeletal
Mechanisms of neuronal migration & Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 The cytoskeletal
Bi 8 Lecture 11. Quantitative aspects of transcription factor binding and gene regulatory circuit design. Ellen Rothenberg 9 February 2016
 Bi 8 Lecture 11 Quantitative aspects of transcription factor binding and gene regulatory circuit design Ellen Rothenberg 9 February 2016 Major take-home messages from λ phage system that apply to many
Bi 8 Lecture 11 Quantitative aspects of transcription factor binding and gene regulatory circuit design Ellen Rothenberg 9 February 2016 Major take-home messages from λ phage system that apply to many
Activity and Polymerization of FtsZ, the Prokaryotic Ortholog of Tubulin.
 (K + / Na + ) and ph dependence of FtsZ activity 1 JOURNAL OF MOLECULAR BIOLOGY 39, 17-25. (29). Structural and Functional Model for Ionic (K + /Na + ) and ph Dependence of GTPase Activity and Polymerization
(K + / Na + ) and ph dependence of FtsZ activity 1 JOURNAL OF MOLECULAR BIOLOGY 39, 17-25. (29). Structural and Functional Model for Ionic (K + /Na + ) and ph Dependence of GTPase Activity and Polymerization
SUPPLEMENTARY INFORMATION
 Supplementary Results and Discussion General properties of L-form cells Phase contrast microscopy of L-form strain Bs115 sup21 revealed that cells were very heterogeneous in size (Fig. 1h) ranging from
Supplementary Results and Discussion General properties of L-form cells Phase contrast microscopy of L-form strain Bs115 sup21 revealed that cells were very heterogeneous in size (Fig. 1h) ranging from
Supplementary Information for. Single-cell dynamics of the chromosome replication and cell division cycles in mycobacteria
 Supplementary Information for Single-cell dynamics of the chromosome replication and cell division cycles in mycobacteria Isabella Santi 1 *, Neeraj Dhar 1, Djenet Bousbaine 1, Yuichi Wakamoto, John D.
Supplementary Information for Single-cell dynamics of the chromosome replication and cell division cycles in mycobacteria Isabella Santi 1 *, Neeraj Dhar 1, Djenet Bousbaine 1, Yuichi Wakamoto, John D.
Analysis of the Interaction of FtsZ with Itself, GTP, and FtsA
 JOURNAL OF BACTERIOLOGY, Sept. 1997, p. 5551 5559 Vol. 179, No. 17 0021-9193/97/$04.00 0 Copyright 1997, American Society for Microbiology Analysis of the Interaction of FtsZ with Itself, GTP, and FtsA
JOURNAL OF BACTERIOLOGY, Sept. 1997, p. 5551 5559 Vol. 179, No. 17 0021-9193/97/$04.00 0 Copyright 1997, American Society for Microbiology Analysis of the Interaction of FtsZ with Itself, GTP, and FtsA
This is a repository copy of Microbiology: Mind the gaps in cellular evolution.
 This is a repository copy of Microbiology: Mind the gaps in cellular evolution. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/114978/ Version: Accepted Version Article:
This is a repository copy of Microbiology: Mind the gaps in cellular evolution. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/114978/ Version: Accepted Version Article:
Initiation of DNA Replication Lecture 3! Linda Bloom! Office: ARB R3-165! phone: !
 Initiation of DNA Replication Lecture 3! Linda Bloom! Office: ARB R3-165! email: lbloom@ufl.edu! phone: 392-8708! 1! Lecture 3 Outline! Students will learn! Basic techniques for visualizing replicating
Initiation of DNA Replication Lecture 3! Linda Bloom! Office: ARB R3-165! email: lbloom@ufl.edu! phone: 392-8708! 1! Lecture 3 Outline! Students will learn! Basic techniques for visualizing replicating
Describe the process of cell division in prokaryotic cells. The Cell Cycle
 The Cell Cycle Objective # 1 In this topic we will examine the cell cycle, the series of changes that a cell goes through from one division to the next. We will pay particular attention to how the genetic
The Cell Cycle Objective # 1 In this topic we will examine the cell cycle, the series of changes that a cell goes through from one division to the next. We will pay particular attention to how the genetic
NOTE: Questions are written on both sides of the sheets of paper making up this exam booklet!
 Biology 1010 Section A Midterm 1 January 30, 2008 (print): ANSWER KEY Name (signature): Student I.D. #: Time: 50 minutes Read the following instructions: 1. Do not open the examination until you are instructed
Biology 1010 Section A Midterm 1 January 30, 2008 (print): ANSWER KEY Name (signature): Student I.D. #: Time: 50 minutes Read the following instructions: 1. Do not open the examination until you are instructed
BE/APh161 Physical Biology of the Cell. Rob Phillips Applied Physics and Bioengineering California Institute of Technology
 BE/APh161 Physical Biology of the Cell Rob Phillips Applied Physics and Bioengineering California Institute of Technology Cells Decide: Where to Go The Hunters of the Immune Response (Berman et al.) There
BE/APh161 Physical Biology of the Cell Rob Phillips Applied Physics and Bioengineering California Institute of Technology Cells Decide: Where to Go The Hunters of the Immune Response (Berman et al.) There
Dynamic proteins are vital components of bacterial organization
 Stochastic model for Soj relocation dynamics in Bacillus subtilis Konstantin Doubrovinski* and Martin Howard* *Department of Mathematics, Imperial College London, South Kensington Campus, London SW7 2AZ,
Stochastic model for Soj relocation dynamics in Bacillus subtilis Konstantin Doubrovinski* and Martin Howard* *Department of Mathematics, Imperial College London, South Kensington Campus, London SW7 2AZ,
Gene regulation I Biochemistry 302. Bob Kelm February 25, 2005
 Gene regulation I Biochemistry 302 Bob Kelm February 25, 2005 Principles of gene regulation (cellular versus molecular level) Extracellular signals Chemical (e.g. hormones, growth factors) Environmental
Gene regulation I Biochemistry 302 Bob Kelm February 25, 2005 Principles of gene regulation (cellular versus molecular level) Extracellular signals Chemical (e.g. hormones, growth factors) Environmental
Regulation of Gene Expression in Bacteria and Their Viruses
 11 Regulation of Gene Expression in Bacteria and Their Viruses WORKING WITH THE FIGURES 1. Compare the structure of IPTG shown in Figure 11-7 with the structure of galactose shown in Figure 11-5. Why is
11 Regulation of Gene Expression in Bacteria and Their Viruses WORKING WITH THE FIGURES 1. Compare the structure of IPTG shown in Figure 11-7 with the structure of galactose shown in Figure 11-5. Why is
Name 8 Cell Cycle and Meiosis Test Date Study Guide You must know: The structure of the replicated chromosome. The stages of mitosis.
 Name 8 Cell Cycle and Meiosis Test Date Study Guide You must know: The structure of the replicated chromosome. The stages of mitosis. The role of kinases and cyclin in the regulation of the cell cycle.
Name 8 Cell Cycle and Meiosis Test Date Study Guide You must know: The structure of the replicated chromosome. The stages of mitosis. The role of kinases and cyclin in the regulation of the cell cycle.
A synthetic oscillatory network of transcriptional regulators
 A synthetic oscillatory network of transcriptional regulators Michael B. Elowitz & Stanislas Leibler, Nature, 403, 2000 igem Team Heidelberg 2008 Journal Club Andreas Kühne Introduction Networks of interacting
A synthetic oscillatory network of transcriptional regulators Michael B. Elowitz & Stanislas Leibler, Nature, 403, 2000 igem Team Heidelberg 2008 Journal Club Andreas Kühne Introduction Networks of interacting
Molecules in Time and Space
 Molecules in Time and Space Molecules in Time and Space Bacterial Shape, Division and Phylogeny Edited by Miguel Vicente CSIC National Biotechnology Centre Javier Tamames BioAlma, S.L. Alfonso Valencia
Molecules in Time and Space Molecules in Time and Space Bacterial Shape, Division and Phylogeny Edited by Miguel Vicente CSIC National Biotechnology Centre Javier Tamames BioAlma, S.L. Alfonso Valencia
Bio Microbiology - Spring 2012 Learning Guide 04.
 Bio 230 - Microbiology - Spring 2012 Learning Guide 04 http://pessimistcomic.blogspot.com/ A septum assembles at the center of the cell. This molecular "purse string" is linked to the inner surface of
Bio 230 - Microbiology - Spring 2012 Learning Guide 04 http://pessimistcomic.blogspot.com/ A septum assembles at the center of the cell. This molecular "purse string" is linked to the inner surface of
Initiation of translation in eukaryotic cells:connecting the head and tail
 Initiation of translation in eukaryotic cells:connecting the head and tail GCCRCCAUGG 1: Multiple initiation factors with distinct biochemical roles (linking, tethering, recruiting, and scanning) 2: 5
Initiation of translation in eukaryotic cells:connecting the head and tail GCCRCCAUGG 1: Multiple initiation factors with distinct biochemical roles (linking, tethering, recruiting, and scanning) 2: 5
Translation - Prokaryotes
 1 Translation - Prokaryotes Shine-Dalgarno (SD) Sequence rrna 3 -GAUACCAUCCUCCUUA-5 mrna...ggagg..(5-7bp)...aug Influences: Secondary structure!! SD and AUG in unstructured region Start AUG 91% GUG 8 UUG
1 Translation - Prokaryotes Shine-Dalgarno (SD) Sequence rrna 3 -GAUACCAUCCUCCUUA-5 mrna...ggagg..(5-7bp)...aug Influences: Secondary structure!! SD and AUG in unstructured region Start AUG 91% GUG 8 UUG
Lecture 4: Transcription networks basic concepts
 Lecture 4: Transcription networks basic concepts - Activators and repressors - Input functions; Logic input functions; Multidimensional input functions - Dynamics and response time 2.1 Introduction The
Lecture 4: Transcription networks basic concepts - Activators and repressors - Input functions; Logic input functions; Multidimensional input functions - Dynamics and response time 2.1 Introduction The
ROLE OF FtsA IN CELL DIVISION IN Neisseria gonorrhoeae
 ROLE OF FtsA IN CELL DIVISION IN Neisseria gonorrhoeae A Thesis Submitted to the College of Graduate Studies and Research In Partial Fulfillment of the Requirements For the Degree of Doctor of Philosophy
ROLE OF FtsA IN CELL DIVISION IN Neisseria gonorrhoeae A Thesis Submitted to the College of Graduate Studies and Research In Partial Fulfillment of the Requirements For the Degree of Doctor of Philosophy
DYNAMIC INSTABILITY - A COMMON DENOMINATOR IN PROKARYOTIC AND EUKARYOTIC DNA SEGREGATION AND CELL DIVISION
 CELLULAR & MOLECULAR BIOLOGY LETTERS http://www.cmbl.org.pl Received: 16 March 2012 Volume 17 (2012) pp 542-548 Final form accepted: 10 August 2012 DOI: 10.2478/s11658-012-0026-3 Published online: 15 August
CELLULAR & MOLECULAR BIOLOGY LETTERS http://www.cmbl.org.pl Received: 16 March 2012 Volume 17 (2012) pp 542-548 Final form accepted: 10 August 2012 DOI: 10.2478/s11658-012-0026-3 Published online: 15 August
Chapter 16 Lecture. Concepts Of Genetics. Tenth Edition. Regulation of Gene Expression in Prokaryotes
 Chapter 16 Lecture Concepts Of Genetics Tenth Edition Regulation of Gene Expression in Prokaryotes Chapter Contents 16.1 Prokaryotes Regulate Gene Expression in Response to Environmental Conditions 16.2
Chapter 16 Lecture Concepts Of Genetics Tenth Edition Regulation of Gene Expression in Prokaryotes Chapter Contents 16.1 Prokaryotes Regulate Gene Expression in Response to Environmental Conditions 16.2
SUPPLEMENTARY FIGURE 1. Force dependence of the unbinding rate: (a) Force-dependence
 (b) BSA-coated beads double exponential low force exponential high force exponential 1 unbinding time tb [sec] (a) unbinding time tb [sec] SUPPLEMENTARY FIGURES BSA-coated beads without BSA.2.1 5 1 load
(b) BSA-coated beads double exponential low force exponential high force exponential 1 unbinding time tb [sec] (a) unbinding time tb [sec] SUPPLEMENTARY FIGURES BSA-coated beads without BSA.2.1 5 1 load
Cell Division and Reproduction
 Cell Division and Reproduction What do you think this picture shows? If you guessed that it s a picture of two cells, you are right. In fact, the picture shows human cancer cells, and they are nearing
Cell Division and Reproduction What do you think this picture shows? If you guessed that it s a picture of two cells, you are right. In fact, the picture shows human cancer cells, and they are nearing
BME 5742 Biosystems Modeling and Control
 BME 5742 Biosystems Modeling and Control Lecture 24 Unregulated Gene Expression Model Dr. Zvi Roth (FAU) 1 The genetic material inside a cell, encoded in its DNA, governs the response of a cell to various
BME 5742 Biosystems Modeling and Control Lecture 24 Unregulated Gene Expression Model Dr. Zvi Roth (FAU) 1 The genetic material inside a cell, encoded in its DNA, governs the response of a cell to various
Bacterial Chemotaxis
 Bacterial Chemotaxis Bacteria can be attracted/repelled by chemicals Mechanism? Chemoreceptors in bacteria. attractant Adler, 1969 Science READ! This is sensing, not metabolism Based on genetic approach!!!
Bacterial Chemotaxis Bacteria can be attracted/repelled by chemicals Mechanism? Chemoreceptors in bacteria. attractant Adler, 1969 Science READ! This is sensing, not metabolism Based on genetic approach!!!
Introduction to Molecular and Cell Biology
 Introduction to Molecular and Cell Biology Molecular biology seeks to understand the physical and chemical basis of life. and helps us answer the following? What is the molecular basis of disease? What
Introduction to Molecular and Cell Biology Molecular biology seeks to understand the physical and chemical basis of life. and helps us answer the following? What is the molecular basis of disease? What
Fitness constraints on horizontal gene transfer
 Fitness constraints on horizontal gene transfer Dan I Andersson University of Uppsala, Department of Medical Biochemistry and Microbiology, Uppsala, Sweden GMM 3, 30 Aug--2 Sep, Oslo, Norway Acknowledgements:
Fitness constraints on horizontal gene transfer Dan I Andersson University of Uppsala, Department of Medical Biochemistry and Microbiology, Uppsala, Sweden GMM 3, 30 Aug--2 Sep, Oslo, Norway Acknowledgements:
Neurite formation & neuronal polarization
 Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 An immature neuron in cell culture first sprouts
Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 An immature neuron in cell culture first sprouts
5.1 Cell Division and the Cell Cycle
 5.1 Cell Division and the Cell Cycle Lesson Objectives Contrast cell division in prokaryotes and eukaryotes. Identify the phases of the eukaryotic cell cycle. Explain how the cell cycle is controlled.
5.1 Cell Division and the Cell Cycle Lesson Objectives Contrast cell division in prokaryotes and eukaryotes. Identify the phases of the eukaryotic cell cycle. Explain how the cell cycle is controlled.
Dynamic optimisation identifies optimal programs for pathway regulation in prokaryotes. - Supplementary Information -
 Dynamic optimisation identifies optimal programs for pathway regulation in prokaryotes - Supplementary Information - Martin Bartl a, Martin Kötzing a,b, Stefan Schuster c, Pu Li a, Christoph Kaleta b a
Dynamic optimisation identifies optimal programs for pathway regulation in prokaryotes - Supplementary Information - Martin Bartl a, Martin Kötzing a,b, Stefan Schuster c, Pu Li a, Christoph Kaleta b a
Stochastic simulations!
 Stochastic simulations! Application to biomolecular networks! Literature overview Noise in genetic networks! Origins! How to measure the noise and distinguish between the two sources of noise (intrinsic
Stochastic simulations! Application to biomolecular networks! Literature overview Noise in genetic networks! Origins! How to measure the noise and distinguish between the two sources of noise (intrinsic
Cytokinesis in fission yeast: Modeling the assembly of the contractile ring
 Cytokinesis in fission yeast: Modeling the assembly of the contractile ring Nikola Ojkic, Dimitrios Vavylonis Department of Physics, Lehigh University Damien Laporte, Jian-Qiu Wu Department of Molecular
Cytokinesis in fission yeast: Modeling the assembly of the contractile ring Nikola Ojkic, Dimitrios Vavylonis Department of Physics, Lehigh University Damien Laporte, Jian-Qiu Wu Department of Molecular
2012 Univ Aguilera Lecture. Introduction to Molecular and Cell Biology
 2012 Univ. 1301 Aguilera Lecture Introduction to Molecular and Cell Biology Molecular biology seeks to understand the physical and chemical basis of life. and helps us answer the following? What is the
2012 Univ. 1301 Aguilera Lecture Introduction to Molecular and Cell Biology Molecular biology seeks to understand the physical and chemical basis of life. and helps us answer the following? What is the
Interactions between proteins, DNA and RNA. The energy, length and time coordinate system to find your way in the cell
 Interactions between proteins, DNA and RNA The energy, length and time coordinate system to find your way in the cell Scanning/atomic force microscope (SFM/AFM) mirror laser beam photo diode fluid cell
Interactions between proteins, DNA and RNA The energy, length and time coordinate system to find your way in the cell Scanning/atomic force microscope (SFM/AFM) mirror laser beam photo diode fluid cell
2. Which of the following are NOT prokaryotes? A) eubacteria B) archaea C) viruses D) ancient bacteria
 1. Which of the following statements is FALSE? A) Errors in chromosome separation are rarely a problem for an organism. B) Errors in chromosome separation can result in a miscarriage. C) Errors in chromosome
1. Which of the following statements is FALSE? A) Errors in chromosome separation are rarely a problem for an organism. B) Errors in chromosome separation can result in a miscarriage. C) Errors in chromosome
7.32/7.81J/8.591J. Rm Rm (under construction) Alexander van Oudenaarden Jialing Li. Bernardo Pando. Rm.
 Introducing... 7.32/7.81J/8.591J Systems Biology modeling biological networks Lectures: Recitations: ti TR 1:00-2:30 PM W 4:00-5:00 PM Rm. 6-120 Rm. 26-204 (under construction) Alexander van Oudenaarden
Introducing... 7.32/7.81J/8.591J Systems Biology modeling biological networks Lectures: Recitations: ti TR 1:00-2:30 PM W 4:00-5:00 PM Rm. 6-120 Rm. 26-204 (under construction) Alexander van Oudenaarden
GENETICS - CLUTCH CH.11 TRANSLATION.
 !! www.clutchprep.com CONCEPT: GENETIC CODE Nucleotides and amino acids are translated in a 1 to 1 method The triplet code states that three nucleotides codes for one amino acid - A codon is a term for
!! www.clutchprep.com CONCEPT: GENETIC CODE Nucleotides and amino acids are translated in a 1 to 1 method The triplet code states that three nucleotides codes for one amino acid - A codon is a term for
REVIEW SESSION. Wednesday, September 15 5:30 PM SHANTZ 242 E
 REVIEW SESSION Wednesday, September 15 5:30 PM SHANTZ 242 E Gene Regulation Gene Regulation Gene expression can be turned on, turned off, turned up or turned down! For example, as test time approaches,
REVIEW SESSION Wednesday, September 15 5:30 PM SHANTZ 242 E Gene Regulation Gene Regulation Gene expression can be turned on, turned off, turned up or turned down! For example, as test time approaches,
downstream (0.8 kb) homologous sequences to the genomic locus of DIC. A DIC mutant strain (ro- 6
 A B C D ts Figure S1 Generation of DIC- mcherry expressing N.crassa strain. A. N. crassa colony morphology. When a cot1 (top, left panel) strain is grown at permissive temperature (25 C), it exhibits straight
A B C D ts Figure S1 Generation of DIC- mcherry expressing N.crassa strain. A. N. crassa colony morphology. When a cot1 (top, left panel) strain is grown at permissive temperature (25 C), it exhibits straight
On the stall force for growing microtubules
 Eur Biophys J (2000) 29: 2±6 Ó Springer-Verlag 2000 ARTICLE G. Sander van Doorn á Catalin Tanase á Bela M. Mulder Marileen Dogterom On the stall force for growing microtubules Received: 27 September 1999
Eur Biophys J (2000) 29: 2±6 Ó Springer-Verlag 2000 ARTICLE G. Sander van Doorn á Catalin Tanase á Bela M. Mulder Marileen Dogterom On the stall force for growing microtubules Received: 27 September 1999
16 The Cell Cycle. Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization
 The Cell Cycle 16 The Cell Cycle Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization Introduction Self-reproduction is perhaps
The Cell Cycle 16 The Cell Cycle Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization Introduction Self-reproduction is perhaps
15.2 Prokaryotic Transcription *
 OpenStax-CNX module: m52697 1 15.2 Prokaryotic Transcription * Shannon McDermott Based on Prokaryotic Transcription by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons
OpenStax-CNX module: m52697 1 15.2 Prokaryotic Transcription * Shannon McDermott Based on Prokaryotic Transcription by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons
High Specificity and Reversibility
 Lecture #8 The Cell as a Machine High Specificity and Reversibility In considering the problem of transcription factor binding in the nucleus and the great specificity that is called for to transcribe
Lecture #8 The Cell as a Machine High Specificity and Reversibility In considering the problem of transcription factor binding in the nucleus and the great specificity that is called for to transcribe
Molecular Biology (9)
 Molecular Biology (9) Translation Mamoun Ahram, PhD Second semester, 2017-2018 1 Resources This lecture Cooper, Ch. 8 (297-319) 2 General information Protein synthesis involves interactions between three
Molecular Biology (9) Translation Mamoun Ahram, PhD Second semester, 2017-2018 1 Resources This lecture Cooper, Ch. 8 (297-319) 2 General information Protein synthesis involves interactions between three
Lecture 10: Cyclins, cyclin kinases and cell division
 Chem*3560 Lecture 10: Cyclins, cyclin kinases and cell division The eukaryotic cell cycle Actively growing mammalian cells divide roughly every 24 hours, and follow a precise sequence of events know as
Chem*3560 Lecture 10: Cyclins, cyclin kinases and cell division The eukaryotic cell cycle Actively growing mammalian cells divide roughly every 24 hours, and follow a precise sequence of events know as
Division site selection protein DivIVA of Bacillus subtilis has a second distinct function in chromosome segregation during sporulation
 Division site selection protein DivIVA of Bacillus subtilis has a second distinct function in chromosome segregation during sporulation Helena B. Thomaides, 1 Marcelle Freeman, 2 Meriem El Karoui, 3 and
Division site selection protein DivIVA of Bacillus subtilis has a second distinct function in chromosome segregation during sporulation Helena B. Thomaides, 1 Marcelle Freeman, 2 Meriem El Karoui, 3 and
Lecture 6: The feed-forward loop (FFL) network motif
 Lecture 6: The feed-forward loop (FFL) network motif Chapter 4 of Alon x 4. Introduction x z y z y Feed-forward loop (FFL) a= 3-node feedback loop (3Loop) a=3 Fig 4.a The feed-forward loop (FFL) and the
Lecture 6: The feed-forward loop (FFL) network motif Chapter 4 of Alon x 4. Introduction x z y z y Feed-forward loop (FFL) a= 3-node feedback loop (3Loop) a=3 Fig 4.a The feed-forward loop (FFL) and the
Simulating ribosome biogenesis in replicating whole cells
 Simulating ribosome biogenesis in replicating whole cells Tyler Earnest / PI: Zan Luthey-Schulten (UIUC) NCSA Blue Waters Symposium for Petascale Science and Beyond: June 15, 2016 Multi-scale modeling
Simulating ribosome biogenesis in replicating whole cells Tyler Earnest / PI: Zan Luthey-Schulten (UIUC) NCSA Blue Waters Symposium for Petascale Science and Beyond: June 15, 2016 Multi-scale modeling
Development Team. Regulation of gene expression in Prokaryotes: Lac Operon. Molecular Cell Biology. Department of Zoology, University of Delhi
 Paper Module : 15 : 23 Development Team Principal Investigator : Prof. Neeta Sehgal Department of Zoology, University of Delhi Co-Principal Investigator : Prof. D.K. Singh Department of Zoology, University
Paper Module : 15 : 23 Development Team Principal Investigator : Prof. Neeta Sehgal Department of Zoology, University of Delhi Co-Principal Investigator : Prof. D.K. Singh Department of Zoology, University
Activation of a receptor. Assembly of the complex
 Activation of a receptor ligand inactive, monomeric active, dimeric When activated by growth factor binding, the growth factor receptor tyrosine kinase phosphorylates the neighboring receptor. Assembly
Activation of a receptor ligand inactive, monomeric active, dimeric When activated by growth factor binding, the growth factor receptor tyrosine kinase phosphorylates the neighboring receptor. Assembly
This is DUE: Come prepared to share your findings with your group.
 Biology 160 NAME: Reading Guide 11: Population Dynamics, Humans, Part I This is DUE: Come prepared to share your findings with your group. *As before, please turn in only the Critical Thinking questions
Biology 160 NAME: Reading Guide 11: Population Dynamics, Humans, Part I This is DUE: Come prepared to share your findings with your group. *As before, please turn in only the Critical Thinking questions
Chapter 6- An Introduction to Metabolism*
 Chapter 6- An Introduction to Metabolism* *Lecture notes are to be used as a study guide only and do not represent the comprehensive information you will need to know for the exams. The Energy of Life
Chapter 6- An Introduction to Metabolism* *Lecture notes are to be used as a study guide only and do not represent the comprehensive information you will need to know for the exams. The Energy of Life
Basic Structure of a Cell
 Basic Structure of a Cell Prokaryotic Cells No nucleus Archaea & Eubacteria One circular chromosome Extremely small Eukaryotic Cells Has a nucleus!!! Membrane-bound organelles Plants, Animals, Fungi, &
Basic Structure of a Cell Prokaryotic Cells No nucleus Archaea & Eubacteria One circular chromosome Extremely small Eukaryotic Cells Has a nucleus!!! Membrane-bound organelles Plants, Animals, Fungi, &
Copyright Mark Brandt, Ph.D A third method, cryogenic electron microscopy has seen increasing use over the past few years.
 Structure Determination and Sequence Analysis The vast majority of the experimentally determined three-dimensional protein structures have been solved by one of two methods: X-ray diffraction and Nuclear
Structure Determination and Sequence Analysis The vast majority of the experimentally determined three-dimensional protein structures have been solved by one of two methods: X-ray diffraction and Nuclear
Use of time-lapse microscopy to visualize rapid movement of the replication origin region of the chromosome during the cell cycle in Bacillus subtilis
 Molecular Microbiology (1998) 28(5), 883 892 Use of time-lapse microscopy to visualize rapid movement of the replication origin region of the chromosome during the cell cycle in Bacillus subtilis Chris
Molecular Microbiology (1998) 28(5), 883 892 Use of time-lapse microscopy to visualize rapid movement of the replication origin region of the chromosome during the cell cycle in Bacillus subtilis Chris
Mukogawa Women s University Nishinomiya, Hyogo , Japan 2 Hyogo Nutrition Vocational College, Nishinomiya, Hyogo , Japan
 J. Biol. Macromol., 4(1) 13-22 (2004) Article Stimulating Effect of High Concentration of Calcium Ion on the Polymerization of the Tubulin-Colchicine Complex. Relationship between Magnesium and Calcium
J. Biol. Macromol., 4(1) 13-22 (2004) Article Stimulating Effect of High Concentration of Calcium Ion on the Polymerization of the Tubulin-Colchicine Complex. Relationship between Magnesium and Calcium
ydci GTC TGT TTG AAC GCG GGC GAC TGG GCG CGC AAT TAA CGG TGT GTA GGC TGG AGC TGC TTC
 Table S1. DNA primers used in this study. Name ydci P1ydcIkd3 Sequence GTC TGT TTG AAC GCG GGC GAC TGG GCG CGC AAT TAA CGG TGT GTA GGC TGG AGC TGC TTC Kd3ydcIp2 lacz fusion YdcIendP1 YdcItrgP2 GAC AGC
Table S1. DNA primers used in this study. Name ydci P1ydcIkd3 Sequence GTC TGT TTG AAC GCG GGC GAC TGG GCG CGC AAT TAA CGG TGT GTA GGC TGG AGC TGC TTC Kd3ydcIp2 lacz fusion YdcIendP1 YdcItrgP2 GAC AGC
Transmembrane Domains (TMDs) of ABC transporters
 Transmembrane Domains (TMDs) of ABC transporters Most ABC transporters contain heterodimeric TMDs (e.g. HisMQ, MalFG) TMDs show only limited sequence homology (high diversity) High degree of conservation
Transmembrane Domains (TMDs) of ABC transporters Most ABC transporters contain heterodimeric TMDs (e.g. HisMQ, MalFG) TMDs show only limited sequence homology (high diversity) High degree of conservation
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins
 13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
10 CELL DIVISION AND MITOSIS
 10 CELL DIVISION AND MITOSIS Chapter Outline Why It Matters 10.1 THE CYCLE OF CELL GROWTH AND DIVISION: OVERVIEW The products of mitosis are genetic duplicates of the dividing cell Chromosomes are the
10 CELL DIVISION AND MITOSIS Chapter Outline Why It Matters 10.1 THE CYCLE OF CELL GROWTH AND DIVISION: OVERVIEW The products of mitosis are genetic duplicates of the dividing cell Chromosomes are the
