Understanding the drivers for the 20th century change of hydrogen peroxide in Antarctic ice cores
|
|
|
- Tobias Lee
- 5 years ago
- Views:
Transcription
1 GEOPHYSICAL RESEARCH LETTERS, VOL. 38,, doi: /2010gl045992, 2011 Understanding the drivers for the 20th century change of hydrogen peroxide in Antarctic ice cores Jean François Lamarque, 1 J. R. McConnell, 2 D. T. Shindell, 3 J. J. Orlando, 1 and G. S. Tyndall 1 Received 27 October 2010; revised 5 January 2011; accepted 14 January 2011; published 25 February [1] Observations and model simulations of an Antarctic icecore record of hydrogen peroxide during the last 150 years are analyzed. The observations indicate a relative increase in hydrogen peroxide by approximately 50% since 1900, with most of the change since the early 1970s. Using two model simulations spanning 1850 to present, we show that the modeled relative change in annual mean surface hydrogen peroxide parallels the equivalent signal from the ice core record. In addition, we show that this relative change can be explained by the relative changes in tropospheric ozone concentration and mostly in ozone photolysis rates (J(O 1 D)). The simulated signal is therefore intimately related to the changes in stratospheric ozone associated with increases in chlorofluorocarbons; this is further demonstrated using total ozone column observations and the associated observed change in ice core hydrogen peroxide. Citation: Lamarque, J. F., J. R. McConnell, D. T. Shindell, J. J. Orlando, and G. S. Tyndall (2011), Understanding the drivers for the 20th century change of hydrogen peroxide in Antarctic ice cores, Geophys. Res. Lett., 38,, doi: /2010gl Introduction [2] The main production path for hydrogen peroxide (H 2 O 2 ) in the remote atmosphere is the HO 2 self reaction [Finlayson Pitts and Pitts, 2000]. Once formed, hydrogen peroxide can be removed from the atmosphere by wet removal (uptake in cloud droplets), dry deposition, photolysis and reaction with OH [Sigg et al., 1992]. As a consequence, the lifetime of hydrogen peroxide ranges from a few hours to days in warm sunlit conditions [Jaegle et al., 2001] to several days during dark winter months [e.g., Snow et al., 2003]. Its presence in polar ice has been detected for over 25 years [Neftel et al., 1984]. [3] Hydrogen peroxide is reversibly deposited to snow, meaning that it continues to exchange between snow and air as environmental conditions change during the early stages of burial [Sigg et al., 1992; McConnell et al., 1998; Frey et al., 2006]. In general, high net snowfall rates and low air temperatures lead to higher preserved concentrations in 1 NCAR Earth System Laboratory, National Center for Atmospheric Research, Boulder, Colorado, USA. 2 Desert Research Institute, Nevada System of Higher Education, Reno, Nevada, USA. 3 NASA Goddard Institute for Space Studies, New York, New York, USA. Copyright 2011 by the American Geophysical Union /11/2010GL ice. In addition to atmospheric concentrations above the deposition site, hydrogen peroxide concentrations measured in ice cores, therefore, are affected by (1) changes in snow temperature because of temperature dependent air snow partitioning of hydrogen peroxide, (2) the seasonal timing of net snow accumulation at the site, and (3) the rate of burial (i.e., net annual snowfall), with the latter being the most important factor [McConnell et al., 1998]. More precisely, while photochemical reactions may modify the composition of snow and interstitial air near the surface, the post depositional impacts on the snow concentrations are minimized at sites with annual accumulation rates higher than the e folding depth of light penetration ( 0.3 m); in that case, surface snow is quickly buried and so removed from solar radiation and chemical exchanges between the snow and overlying atmosphere are limited. Any temporal changes in postdepositional perturbations also are minimized in ice core records collected from sites with relatively constant accumulation rates [McConnell et al., 1998; Frey et al., 2006]. [4] It is also important to recognize that air snow interactions driven by snowpack photochemistry can significantly perturb the air composition near the surface (<30 m) in the Polar regions, as comprehensively reviewed by Dominé and Shepson [2002] and Grannas et al. [2007]. However, Frey et al. [2006] indicate that H 2 O 2 concentrations in fresh snow reflect ratios in clouds, indicating that the influence of boundary layer chemistry is probably small. [5] In the Antarctic troposphere, where NO and NO 2 concentrations are low (especially above the shallow boundarylayer but also more generally in the West Antarctic Ice Sheet [Frey et al., 2005], where the cores discussed here originate), the chemical reaction relevant to the production of hydrogen peroxide (H 2 O 2 )is[finlayson Pitts and Pitts, 2000] HO 2 þ HO 2! H 2 O 2 þ O 2 indicating that hydrogen peroxide formation depends on the square of the HO 2 concentration. At steady state, ignoring the contribution of RO 2, which is significantly smaller than HO 2 in the Southern polar regions [Chen et al., 2001; Mauldin et al., 2004], the concentration of HO 2 can be expressed as [Finlayson Pitts and Pitts, 2000] q ffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffiffi ½HO 2 Š / J O 1 D ½ O3 Š where J(O 1 D) is the rate at which ozone is photolyzed and forms an excited oxygen atom (O 1 D). Note that, in particular, the potential role for CO in increasing the HO 2 /OH ratio is explicitly used in the derivation of equation (1). Such squareroot relationships between HO 2 and J(O 1 D) have also been ð1þ 1of5
2 observationally deduced [Bloss et al., 2007]. Based on this analysis, we can therefore expect that, at steady state, ½H 2 O 2 Š / J O 1 D ½ O3 Š ð2þ indicating that hydrogen peroxide is mostly dependent on ozone, since J(O 1 D) is approximately a simple function (power law) of total ozone column [Madronich and Granier, 1992]. The overall sensitivity of tropospheric hydrogen peroxide to stratospheric ozone has been discussed in previous studies of global chemistry, such as Thompson [1991], Thompson et al. [1993] and Fuglestved et al. [1994]; in addition, its role in the modulation of Arctic [e.g., Anklin and Bales, 1997] and Antarctic hydrogen peroxide has been postulated [e.g., Frey et al., 2005]. Here we extend these analyses beyond the Antarctic ozone hole period and compare the modeled change to ice core observations. [6] In this paper, we use continuous measurements in three recently collected, closely spaced ice cores from the West Antarctic Ice Sheet (WAIS) deep ice core site spanning several centuries to study the change in hydrogen peroxide, with a focus on the change since This is a relatively cold region (mean annual air temperature is 31 C) with high snowfall rates (>0.6 m of snow year 1 ) where detailed chemical measurements in these ice cores show that no significant long term changes in snowfall have occurred during the past 200 years. In addition, we use results from simulations spanning by two chemistry climate models [Lamarque et al., 2010] to identify the specific role of parameters driving the observed and simulated change in hydrogen peroxide. The paper is organized as follows: in section 2, we describe the ice core data. Then, in section 3, the model and model simulations are described. The comparison with observations and analysis of the model simulations are done in section 4. Discussion and conclusions are in section Data Description [7] We have constructed a composite record of annual hydrogen peroxide concentration (Figure 1) by combining measurements from three ice cores recently collected near the WAIS Divide ice core site in the dry snow zone of West Antarctica (79.5 S, E). These include the ITASE00_1 core collected in late 1999 [Frey et al., 2006] and the WDC05A and WDC05Q cores collected in 2005 [Banta et al., 2008]. The WDC cores are located within 1 km of each other near the main deep borehole, while the ITASE00_1 core is located 20 km to the northwest. Measurements from the selected ice cores are very consistent with the highaccumulation sites of Frey et al. [2006]. Additional details can be found in the auxiliary material. 1 [8] Mean net snowfall rates from 1900 through 1999 C.E. were 200, 200, and 227 kg m 2 a 1 for WDC05A, WDC06A, and ITASE00_1, respectively. In agreement with accumulation rate dependent post depositional loss of hydrogen peroxide from the snow [McConnell et al., 1998], concentrations at the higher snow accumulation ITASE00_1 site are consistently 10% higher than in the lower accumulation WDC cores. Annual concentrations of hydrogen 1 Auxiliary materials are available in the HTML. doi: / 2010GL peroxide also vary from core to core because of small differences in the amount and seasonal timing of net snow accumulation that primarily results from wind redistribution ( 15% of the annual average net snowfall at WAIS Divide [Banta et al., 2008]). Despite these short spatial scale differences in snow accumulation, correlations between the individual annual hydrogen peroxide records reported here ranged between and for the 1900 through 1999 period common to all three cores, significant above the 99.9% confidence level [Banta et al., 2008]. To reduce any impact of short scale depositional differences, we combined the three individual records to obtain a composite, more regionally representative hydrogen peroxide record. Analysis of snowfall in these three ice core records indicates that there has been no significant long term change in snowfall during the past 100 years at WAIS Divide [Banta et al., 2008] so the observed increase of both the individual and regional peroxide concentration profiles between the pre 1960s and the mid 1990s is likely not the result of changes in depositional processes. 3. Model Descriptions and Simulation Setup [9] Model descriptions are limited to the specifics relevant to this study. In particular, the anthropogenic and biomass burning emissions are from Lamarque et al. [2010]. In addition, note that neither model has a representation of the specific polar boundary layer chemistry related with air snow exchanges (as discussed in the Introduction). The reader is referred to the auxiliary material for additional information CAM chem [10] We use the Community Atmosphere Model (CAM) version 3.5 [Gent et al., 2009] modified to include interactive chemistry to calculate distributions of gases and aerosols. The model configuration used in this study includes a horizontal resolution of 1.9 (latitude) by 2.5 (longitude) and 26 hybrid levels, from the surface to 40 km with a timestep of 30 minutes; the transient simulation was performed continuously between 1850 and The chemical mechanism used in this study is formulated to provide an accurate representation of both tropospheric and stratospheric chemistry [Lamarque et al., 2008; Lamarque and Solomon, 2010]. As the model top is 40 km, only wavelengths greater than 200 nm are considered in photolysis calculations. A flux lookup table (LUT) approach is used [Kinnison et al., 2007], based on the 4 stream version of the Stratosphere, Troposphere, Ultraviolet (STUV) radiative transfer model (S. Madronich, personal communication, 2006). The transmission function is interpolated from the LUT as a function of altitude, overhead ozone column, surface albedo, and zenith angle. The temperature and pressure dependences of the molecular cross sections and quantum yields for each photolytic process are also represented by a LUT in this wavelength region. In all cases, the photolysis computation is performed every timestep (30 minutes) and takes into account the model distribution of ozone and clouds. [11] In addition, the monthly mean time varying observed sea surface temperatures (SSTs) and sea ice distributions are taken from a 20th century CCSM 3 simulation[meehl et al., 2007]; as this simulation only extended from 1870 to present, the sea surface temperature and ice extent between 1850 and 1870 are assumed to be the same as of5
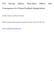
3 Figure 1. Time evolution of the relative change since 1900 in the observed annual mean H 2 O 2 ice core concentration (black line, smoothed with a 3 year running average; grey shading indicates one standard deviation) and in the modeled annual mean H 2 O 2 surface concentration (zonally averaged over 80 S 90 S) over Antarctica from CAM chem (red line) and G PUCCINI (blue squares) G PUCCINI [12] Simulations were also performed with the Goddard Institute for Space Studies (GISS) model for Physical Understanding of Composition Climate INteractions and Impacts (G PUCCINI) [Shindell et al., 2006]. The chemistry model is fully embedded in the GISS modele climate model [Schmidt et al., 2006]. The model was run at 2 latitude by 2.5 longitude Cartesian horizontal resolution. This configuration had 40 vertical hybrid sigma layers from the surface to 0.01 hpa ( 80 km). Time slice simulations were performed every 20 years during the period, and every 10 years from Values during the period were interpolated to give decadal means. Simulations were carried out for 8 years, with the average of the last 5 used for analysis. The GCM was driven by observed decadal mean sea surface temperatures and sea ice distribution [Rayner et al., 2003]. Photolysis rates are calculated online every timestep (20 minutes) using the Fast J2 scheme [Bian and Prather, 2002]; in particular, this scheme takes into account the model distribution of clouds, aerosols and ozone Surface Concentrations Over Antarctica [13] As presented by Lamarque et al. [2010, see Figure 6], both models have a very realistic representation of surface ozone over the observational record ( ); the only bias is for G PUCCINI, with an underestimate of surface ozone (by about 4 ppbv) in the 2000 simulation. On the other hand CAM chem seems to slightly overestimate ozone in the mid 1990s. In addition, analysis of the reconstructed CO change by Assonov et al. [2007] indicates an increase of approximately 40% between 1900 and 2000; this is quite well matched by the model results (Figure S2), especially for CAM chem. Additional discussions on CO are given by Lamarque et al. [2010]. 4. Analysis [14] We discuss in this section results from the simulations described in section 3. Additional information on the simulations and associated results are given by Lamarque et al. [2010]. [15] As mentioned in section 2, hydrogen peroxide in the ice core is a record of its concentration when deposited at the surface through wet and dry deposition. We make here the assumption that the model representation of hydrogen peroxide in ice cores is proportional to its surface gas phase concentration over Antarctica. Indeed, observations in the Northern Hemisphere high latitudes [Snow et al., 2007] indicate a fairly small vertical gradient within the first 5 6 kilometers (also true in our model simulations, not shown). Modeled surface (i.e., lowest model layer) hydrogen peroxide is therefore a reasonable proxy for model tropospheric values at the level where its uptake by water (in any phase and subsequent snow/ice fall) or dry deposition occurs. Note that neither model has a representation of the snow and ice surface processes that can affect hydrogen peroxide concentrations [e.g., Hutterli et al., 2001] in the deposited snow. As a consequence, it might be difficult to directly compare observed concentrations of hydrogen peroxide with modeled surface values. Instead, since hydrogen peroxide concentration over the Antarctic region is expected to be proportional to ozone (local and total column, equation (2)), we can expect observations and model simulations to exhibit similar relative changes. [16] Therefore, we present in the subsequent analysis the various data (ice core and modeled surface hydrogen peroxide) relative to their values (Figure 1); note that the results shown in Figure 1 are from the full model simulations, not the application of equation (2). This period is chosen as no information on biomass burning emissions (the only emissions that are significantly changing in the Southern Hemisphere) before 1900 is available [Lamarque Figure 2. (top left) Time evolution of the relative change since 1900 in modeled annual mean surface O 3 (blue line) and the inverse of the total ozone column (1/TOC, green line) for the CAM chem simulation. (top right) Time evolution of the relative change since 1900 in CAM chem modeled annual mean H 2 O 2 (blue line) and in the right hand side of equation (2) (green line). (bottom) Same as Figure 2 (top) but for the G PUCCINI simulation. 3of5
4 Figure 3. Time evolution of the relative change since the 1960s in the observed annual mean H 2 O 2 ice core concentration (black line, smoothed with a 3 year running average; grey shading indicates one standard deviation); red line is the observed inverse of total ozone column squared. See text for details. et al., 2010], meaning that the model simulations of H 2 O 2 prior to 1900 could be affected by this simplification. Furthermore, this averaging period exhibits fairly constant values of ice core hydrogen peroxide. Using this relative measure, we find that both model simulations (surface hydrogen peroxide concentration, annually averaged over Antarctica to match the observational record) provide a reasonable representation of the long term change in hydrogen peroxide (Figure 1) since In particular, we find that observations and simulations indicate a rapid rise starting in the 1970s. [17] Following equation (2), we construct the timeseries of the relative change in surface ozone and in J(O 1 D) from the simulations. As model results are not available for this latter quantity, we use instead total ozone column (TOC) inverse squared [Madronich and Granier, 1992]). The combination (as in equation (2)) of those relative changes for each model indicates a very good agreement with their respective change in simulated H 2 O 2 (Figure 2); this comparison indicates therefore that equation (2) is a realistic representation of the H 2 O 2 changes simulated by our full chemistry models. Note that the agreement of the G PUC- CINI results is optimal with TOC 2.5, still within the range of Madronich and Granier. It is not clear at this point why this exponent is different between models (a possible explanation is related to differences in the representation of photolysis rates, especially in low overhead ozone column), but this does not strongly affect our conclusion that the relative changes in hydrogen peroxide between 1900 and present are very closely following [O 3 ]*J(O1D), and are therefore mainly a function of ozone. More precisely, the relative change in surface ozone provides an explanation for the small relative change in hydrogen peroxide prior to the 1960s or 1970s. Over the most recent decades, it is however the decrease in total ozone column that provides the rapid increasing contribution to surface hydrogen peroxide in the model, as indicated in Figure 2 by the green lines in the left column. Indeed, the role of tropospheric ozone (a small steady increase over the 20th century but little change since 1980s in CAM chem but a large decrease over that latter period in G PUCCINI, as mentioned in section 3) is then overwhelmed by the J(O 1 D) signal. This analysis is therefore a complement to the results of Frey et al. [2006] where the variability in ice core hydrogen peroxide in the pre 1960 period was found to be related to internal modes such as the El Niño Southern Oscillation. [18] It is clear that other chemical compounds have significantly increased between 1900 and present. In particular, and of strong importance to the OH and HO 2 budget, is CO. However, while both models indicate a relative rise in CO of approximately 1.5 over that period (see discussion in section 3.3), its rate of increase (Figure S2) is much too rapid in the early part of the 20th century to be correlated with the H 2 O 2 change and therefore cannot by itself explain this trend. [19] To further analyze the role of long term changes in total ozone column on hydrogen peroxide, we show (Figure 3; note that because of the limited ozone record, scaling is performed with respect to the decadal average instead of 1900 as in Figures 1 and 2) the ice core record of hydrogen peroxide (same as in Figure 1) in conjunction with the observed record of inverse total ozone column (using an average of all stations located poleward of 70 S) squared, as discussed above. Similar to the model results, we find that the long term change of both signals over that period is quite similar in amplitude, as expected from equation (2) since surface ozone over South Pole has not changed much over that period [see Lamarque et al., 2010, Figure 6; Helmig et al., 2007, Figure 3]. It is, however, clear that the agreement between the two curves of Figure 3 is not perfect; this is possibly due to limitations in the data coverage (time and space) or modulation by missing mechanisms in our analysis; in particular, the proportionality factors implicit in equation (2) could exhibit variability and trends ignored in the present study. 5. Discussion and Conclusions [20] We have shown that model simulations of the 20th century, driven by transient emissions and observed changes in long lived gases, are able to reproduce the observed relative increase in hydrogen peroxide over Antarctica, as recorded in three closely spaced ice cores from West Antarctica. In particular, hydrogen peroxide in ice cores has increased rapidly since the 1970s. Based on gas phase chemistry of the remote atmosphere, we showed that, in our model simulations, this relative change is mostly driven by the combined changes in tropospheric and stratospheric ozone (through its influence on the ozone photolysis rates); however, over Antarctica, it is clear that most of the signal is associated with changes in stratospheric ozone, as was postulated by Frey et al. [2005]. This was further confirmed by the analysis of the change since the 1960s in ice core hydrogen peroxide and in the observed total ozone column, as surface observations at the South Pole show little to no trends [Helmig et al., 2007]. However, while the results of our study provide an explanation for long term changes, the extent of the role of boundary layer snow chemistry in inducing variability in ice core hydrogen period remains to be quantified. It would therefore be interesting to include such chemistry in global models, and to improve the representation of depositional processes. Similarly, to further verify the validity of the correlations demonstrated here, frequent joint measurements of hydrogen peroxide, surface ozone column and total ozone column would be useful. [21] This analysis highlights the potential for using icecores to understand long term changes in ozone (tropospheric 4of5
5 and stratospheric) in the remote Southern Hemisphere, as ice core records extend beyond the period analyzed here. In particular, it might provide additional information on volcanic perturbations of stratospheric ozone since, in pre CFC era, large volcanic eruptions likely led to an increase in stratospheric ozone [Tie and Brasseur, 1995], which could therefore be identified via the observation of a significant decrease in hydrogen peroxide and correlated with other volcanic signatures such as sulfur isotopes [Baroni et al., 2007]. [22] Acknowledgments. The total ozone column data were obtained from the World Ozone and Ultraviolet Radiation Data Centre (WOUDC) operated by Environment Canada, Toronto, Ontario, Canada under the auspices of the World Meteorological Organization. The authors would like to thank S. Madronich and D. Kinnison for their constructive feedback on a previous version of this paper. Development of the ice hydrogen peroxide records was funded by NSF grants OPP and OPP , and we gratefully acknowledge the U.S. ITASE and WAIS Divide projects teams for assistance in obtaining the ice cores. The National Center for Atmospheric Research is operated by the University Corporation for Atmospheric Research under sponsorship of the National Science Foundation. References Anklin, M., and R. C. Bales (1997), Recent increase in H 2 O 2 concentration at Summit, Greenland, J. Geophys. Res., 102(D15), 19,099 19,104, doi: /97jd Assonov, S. S., C. A. M. Brenninkmeijer, P. Jöckel, R. Mulvaney, S. Bernard, and J. Chappellaz (2007), Evidence for a CO increase in the SH during the 20th century based on firn air samples from Berkner Island, Antarctica, Atmos. Chem. Phys., 7, , doi: /acp Banta, J. R., J. R. McConnell, M. Frey, and K. Taylor (2008), Spatial and temporal variability in snow accumulation at WAIS Divide over recent centuries, J. Geophys. Res., 113, D23102, doi: /2008jd Baroni, M., M. H. Thiemens, R. J. Delmas, and J. Savarino (2007), Massindependent sulfur isotopic compositions in stratospheric volcanic eruptions, Science, 315, 84 87, doi: /science Bian, H., and M. Prather (2002), Fast j2: Accurate simulations of photolysis in global climate models, J. Atmos. Chem., 41, , doi: / A: Bloss, W. J., et al. (2007), Observations of OH and HO 2 radicals in coastal Antarctica, Atmos. Chem. Phys., 7, , doi: /acp Chen, G., et al. (2001), An investigation of South Pole HO x chemistry: Comparison of model results with ISCAT observations, Geophys. Res. Lett., 28, , doi: /2001gl Dominé, F., and P. B. Shepson (2002), Air snow interactions and atmospheric chemistry, Science, 297, , doi: /science Finlayson Pitts, B. J., and J. N. Pitts Jr. (2000), Chemistry of the Upper and Lower Atmosphere: Theory, Experiments, and Applications, 1st ed., 969 pp., Academic, New York. Frey, M. M., R. W. Stewart, J. R. McConnell, and R. C. Bales (2005), Atmospheric hydroperoxides in West Antarctica: Links to stratospheric ozone and atmospheric oxidation capacity, J. Geophys. Res., 110, D23301, doi: /2005jd Frey, M. M., R. C. Bales, and J. R. McConnell (2006), Climate sensitivity of the century scale hydrogen peroxide (H 2 O 2 ) record preserved in 23 ice cores from West Antarctica, J. Geophys. Res., 111, D21301, doi: /2005jd Fuglestved, J. S., J. E. Jonson, and I. S. A. Isaksen (1994), Effects of reductions in stratospheric ozone on tropospheric chemistry through changes in photolysis rates, Tellus,Ser.B, 46, , doi: /j t x-i1. Gent, P. R., S. G. Yeager, R. B. Neale, S. Levis, and D. A. Bailey (2009), Improvements in a half degree atmosphere/land version of the CCSM, Clim. Dyn., 79, 25 58, doi: /s Grannas, A. M., et al. (2007), An overview of snow photochemistry: Evidence, mechanisms and impacts, Atmos. Chem. Phys., 7, , doi: /acp Helmig, D., S. J. Oltmans, D. Carlson, J. F.Lamarque,A.Jones, C. Labuschagne, K. Anlauf, and K. Hayden (2007), A review of surface ozone in the polar regions, Atmos. Environ., 41, , doi: /j.atmosenv Hutterli, M. A., et al. (2001), Impact of temperature driven cycling of hydrogen peroxide (H 2 O 2 ) between air and snow on the planetary boundary layer, J. Geophys. Res., 106(D14), 15,395 15,404, doi: / 2001JD Jaegle, L. H., D. J. Jacob, W. H. Brune, and P. O. Wennberg (2001), Chemistry of HO x radicals in the upper troposphere, Atmos. Environ., 35, , doi: /s (00) Kinnison, D. E., et al. (2007), Sensitivity of chemical tracers to meteorological parameters in the MOZART 3 chemical transport model, J. Geophys. Res., 112, D20302, doi: /2006jd Lamarque, J. F., and S. Solomon (2010), Impact of changes in climate and halocarbons on recent lower stratosphere ozone and temperature trends, J. Clim., 23, , doi: /2010jcli Lamarque, J. F., D. E. Kinnison, P. G. Hess, and F. Vitt (2008), Simulated lower stratospheric trends between 1970 and 2005: Identifying the role of climate and composition changes, J. Geophys. Res., 113, D12301, doi: /2007jd Lamarque, J. F., et al. (2010), Historical ( ) gridded anthropogenic and biomass burning emissions of reactive gases and aerosols: methodology and application, Atmos. Chem. Phys., 10, , doi: /acp Madronich, S., and C. Granier (1992), Impact of recent total ozone changes on tropospheric ozone photodissociation, hydroxyl radicals, and methane trends, Geophys. Res. Lett., 19, , doi: /92gl Mauldin, R. L., III, et al. (2004), Measurements of OH, HO 2 +RO 2,H 2 SO 4, and MSA at the South Pole during ISCAT 2000, Atmos. Environ., 38, , doi: /j.atmosenv McConnell, J. R., R. C. Bales, R. W. Stewart, A. M. Thompson, M. R. Albert, and R. Ramos (1998), Physically based modeling of atmosphere tosnow to firn transfer of hydrogen peroxide at South Pole, J. Geophys. Res., 103(D9), 10,561 10,570, doi: /98jd Meehl, G. A., et al. (2007), Global climate projections, in Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, edited by S. Solomon et al., pp , Cambridge Univ. Press, Cambridge, U. K. Neftel, A., P. Jacob, and D. Klockow (1984), Measurements of hydrogen peroxide in polar ice samples, Nature, 311, 43 45, doi: /311043a0. Rayner, N. A., et al. (2003), Global analyses of sea surface temperature, sea ice, and night marine air temperature since the late nineteenth century, J. Geophys. Res., 108(D14), 4407, doi: /2002jd Schmidt, G. A., et al. (2006), Present day atmospheric simulations using GISS modele: Comparison to in situ, satellite and reanalysis data, J. Clim., 19, , doi: /jcli Shindell, D. T., et al. (2006), Simulations of preindustrial, present day, and 2100 conditions in the NASA GISS composition and climate model G PUCCINI, Atmos. Chem. Phys., 6, , doi: /acp Sigg, A., T. Staffelbach, and A. Neftel (1992), Gas phase measurements of hydrogen peroxide in Greenland and their meaning for the interpretation of H 2 O 2 records in ice cores, J. Atmos. Chem., 14, , doi: /bf Snow, J. A., et al. (2003), Winter spring evolution and variability of HO x reservoir species, hydrogen peroxide, and methyl hydroperoxide, in the northern middle to high latitudes, J. Geophys. Res., 108(D4), 8362, doi: /2002jd Snow, J. A., et al. (2007), Hydrogen peroxide, methyl hydroperoxide, and formaldehyde over North America and the North Atlantic, J. Geophys. Res., 112, D12S07, doi: /2006jd Thompson, A. M. (1991), New ozone hole phenomenon, Nature, 352, , doi: /352282a0. Thompson, A. M., J. A. Chappellaz, and I. Y. Fung (1993), The atmospheric CH 4 increase since the Last Glacial Maximum: (2) Interactions with oxidants, Tellus,Ser.B, 45, , doi: /j t x. Tie, X., and G. Brasseur (1995), The response of stratospheric ozone to volcanic eruptions: Sensitivity to atmospheric chlorine loading, Geophys. Res. Lett., 22(22), , doi: /95gl J. F. Lamarque, J. J. Orlando, and G. S. Tyndall, NCAR Earth System Laboratory, National Center for Atmospheric Research, 1850 Table Mesa Dr., Boulder, CO 80305, USA. (lamar@ucar.edu) J. R. McConnell, Desert Research Institute, Nevada System of Higher Education, 2215 Raggio Pkwy., Reno, NV 89512, USA. D. T. Shindell, NASA Goddard Institute for Space Studies, 2880 Broadway, New York, NY 10025, USA. 5of5
PROBLEMS Sources of CO Sources of tropospheric ozone
 220 PROBLEMS 11. 1 Sources of CO The two principal sources of CO to the atmosphere are oxidation of CH 4 and combustion. Mean rate constants for oxidation of CH 4 and CO by OH in the troposphere are k
220 PROBLEMS 11. 1 Sources of CO The two principal sources of CO to the atmosphere are oxidation of CH 4 and combustion. Mean rate constants for oxidation of CH 4 and CO by OH in the troposphere are k
Effect of zonal asymmetries in stratospheric ozone on simulated Southern Hemisphere climate trends
 Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 36, L18701, doi:10.1029/2009gl040419, 2009 Effect of zonal asymmetries in stratospheric ozone on simulated Southern Hemisphere climate trends
Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 36, L18701, doi:10.1029/2009gl040419, 2009 Effect of zonal asymmetries in stratospheric ozone on simulated Southern Hemisphere climate trends
High initial time sensitivity of medium range forecasting observed for a stratospheric sudden warming
 GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2010gl044119, 2010 High initial time sensitivity of medium range forecasting observed for a stratospheric sudden warming Yuhji Kuroda 1 Received 27 May
GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2010gl044119, 2010 High initial time sensitivity of medium range forecasting observed for a stratospheric sudden warming Yuhji Kuroda 1 Received 27 May
The ozone hole indirect effect: Cloud-radiative anomalies accompanying the poleward shift of the eddy-driven jet in the Southern Hemisphere
 GEOPHYSICAL RESEARCH LETTERS, VOL. 4, 388 392, doi:1.12/grl.575, 213 The ozone hole indirect effect: Cloud-radiative anomalies accompanying the poleward shift of the eddy-driven jet in the Southern Hemisphere
GEOPHYSICAL RESEARCH LETTERS, VOL. 4, 388 392, doi:1.12/grl.575, 213 The ozone hole indirect effect: Cloud-radiative anomalies accompanying the poleward shift of the eddy-driven jet in the Southern Hemisphere
Temperature responses to spectral solar variability on decadal time scales
 Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2009gl041898, 2010 Temperature responses to spectral solar variability on decadal time scales Robert F. Cahalan, 1 Guoyong
Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2009gl041898, 2010 Temperature responses to spectral solar variability on decadal time scales Robert F. Cahalan, 1 Guoyong
Science Results Based on Aura OMI-MLS Measurements of Tropospheric Ozone and Other Trace Gases
 Science Results Based on Aura OMI-MLS Measurements of Tropospheric Ozone and Other Trace Gases J. R. Ziemke Main Contributors: P. K. Bhartia, S. Chandra, B. N. Duncan, L. Froidevaux, J. Joiner, J. Kar,
Science Results Based on Aura OMI-MLS Measurements of Tropospheric Ozone and Other Trace Gases J. R. Ziemke Main Contributors: P. K. Bhartia, S. Chandra, B. N. Duncan, L. Froidevaux, J. Joiner, J. Kar,
CONTENTS 1 MEASURES OF ATMOSPHERIC COMPOSITION
 i CONTENTS 1 MEASURES OF ATMOSPHERIC COMPOSITION 1 1.1 MIXING RATIO 1 1.2 NUMBER DENSITY 2 1.3 PARTIAL PRESSURE 6 PROBLEMS 10 1.1 Fog formation 10 1.2 Phase partitioning of water in cloud 10 1.3 The ozone
i CONTENTS 1 MEASURES OF ATMOSPHERIC COMPOSITION 1 1.1 MIXING RATIO 1 1.2 NUMBER DENSITY 2 1.3 PARTIAL PRESSURE 6 PROBLEMS 10 1.1 Fog formation 10 1.2 Phase partitioning of water in cloud 10 1.3 The ozone
Global Warming and Climate Change Part I: Ozone Depletion
 GCOE-ARS : November 18, 2010 Global Warming and Climate Change Part I: Ozone Depletion YODEN Shigeo Department of Geophysics, Kyoto University 1. Stratospheric Ozone and History of the Earth 2. Observations
GCOE-ARS : November 18, 2010 Global Warming and Climate Change Part I: Ozone Depletion YODEN Shigeo Department of Geophysics, Kyoto University 1. Stratospheric Ozone and History of the Earth 2. Observations
Attribution of anthropogenic influence on seasonal sea level pressure
 Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 36, L23709, doi:10.1029/2009gl041269, 2009 Attribution of anthropogenic influence on seasonal sea level pressure N. P. Gillett 1 and P. A.
Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 36, L23709, doi:10.1029/2009gl041269, 2009 Attribution of anthropogenic influence on seasonal sea level pressure N. P. Gillett 1 and P. A.
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1153966/dc1 Supporting Online Material for The Sensitivity of Polar Ozone Depletion to Proposed Geoengineering Schemes Simone Tilmes,* Rolf Müller, Ross Salawitch *To
www.sciencemag.org/cgi/content/full/1153966/dc1 Supporting Online Material for The Sensitivity of Polar Ozone Depletion to Proposed Geoengineering Schemes Simone Tilmes,* Rolf Müller, Ross Salawitch *To
Is Antarctic climate most sensitive to ozone depletion in the middle or lower stratosphere?
 Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 34, L22812, doi:10.1029/2007gl031238, 2007 Is Antarctic climate most sensitive to ozone depletion in the middle or lower stratosphere? S.
Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 34, L22812, doi:10.1029/2007gl031238, 2007 Is Antarctic climate most sensitive to ozone depletion in the middle or lower stratosphere? S.
1. The frequency of an electromagnetic wave is proportional to its wavelength. a. directly *b. inversely
 CHAPTER 3 SOLAR AND TERRESTRIAL RADIATION MULTIPLE CHOICE QUESTIONS 1. The frequency of an electromagnetic wave is proportional to its wavelength. a. directly *b. inversely 2. is the distance between successive
CHAPTER 3 SOLAR AND TERRESTRIAL RADIATION MULTIPLE CHOICE QUESTIONS 1. The frequency of an electromagnetic wave is proportional to its wavelength. a. directly *b. inversely 2. is the distance between successive
3. Carbon Dioxide (CO 2 )
 3. Carbon Dioxide (CO 2 ) Basic information on CO 2 with regard to environmental issues Carbon dioxide (CO 2 ) is a significant greenhouse gas that has strong absorption bands in the infrared region and
3. Carbon Dioxide (CO 2 ) Basic information on CO 2 with regard to environmental issues Carbon dioxide (CO 2 ) is a significant greenhouse gas that has strong absorption bands in the infrared region and
Tananyag fejlesztés idegen nyelven
 Tananyag fejlesztés idegen nyelven Prevention of the atmosphere KÖRNYEZETGAZDÁLKODÁSI AGRÁRMÉRNÖKI MSC (MSc IN AGRO-ENVIRONMENTAL STUDIES) Fundamentals to atmospheric chemical reactions. The stratospheric
Tananyag fejlesztés idegen nyelven Prevention of the atmosphere KÖRNYEZETGAZDÁLKODÁSI AGRÁRMÉRNÖKI MSC (MSc IN AGRO-ENVIRONMENTAL STUDIES) Fundamentals to atmospheric chemical reactions. The stratospheric
CLIMATE AND CLIMATE CHANGE MIDTERM EXAM ATM S 211 FEB 9TH 2012 V1
 CLIMATE AND CLIMATE CHANGE MIDTERM EXAM ATM S 211 FEB 9TH 2012 V1 Name: Student ID: Please answer the following questions on your Scantron Multiple Choice [1 point each] (1) The gases that contribute to
CLIMATE AND CLIMATE CHANGE MIDTERM EXAM ATM S 211 FEB 9TH 2012 V1 Name: Student ID: Please answer the following questions on your Scantron Multiple Choice [1 point each] (1) The gases that contribute to
2. Fargo, North Dakota receives more snow than Charleston, South Carolina.
 2015 National Tournament Division B Meteorology Section 1: Weather versus Climate Chose the answer that best answers the question 1. The sky is partly cloudy this morning in Lincoln, Nebraska. 2. Fargo,
2015 National Tournament Division B Meteorology Section 1: Weather versus Climate Chose the answer that best answers the question 1. The sky is partly cloudy this morning in Lincoln, Nebraska. 2. Fargo,
Introduction to Climate ~ Part I ~
 2015/11/16 TCC Seminar JMA Introduction to Climate ~ Part I ~ Shuhei MAEDA (MRI/JMA) Climate Research Department Meteorological Research Institute (MRI/JMA) 1 Outline of the lecture 1. Climate System (
2015/11/16 TCC Seminar JMA Introduction to Climate ~ Part I ~ Shuhei MAEDA (MRI/JMA) Climate Research Department Meteorological Research Institute (MRI/JMA) 1 Outline of the lecture 1. Climate System (
SUPPLEMENTARY INFORMATION. Antarctic-wide array of high-resolution ice core records reveals pervasive lead pollution began in 1889 and persists today
 SUPPLEMENTARY INFORMATION Antarctic-wide array of high-resolution ice core records reveals pervasive lead pollution began in 1889 and persists today J.R. McConnell 1, O.J. Maselli 1, M. Sigl 1, P. Vallelonga
SUPPLEMENTARY INFORMATION Antarctic-wide array of high-resolution ice core records reveals pervasive lead pollution began in 1889 and persists today J.R. McConnell 1, O.J. Maselli 1, M. Sigl 1, P. Vallelonga
Assessment Schedule 2017 Earth and Space Science: Demonstrate understanding of processes in the atmosphere system (91414)
 NCEA Level 3 Earth and Space Science (91414) 2017 page 1 of 6 Assessment Schedule 2017 Earth and Space Science: Demonstrate understanding of processes in the atmosphere system (91414) Evidence Statement
NCEA Level 3 Earth and Space Science (91414) 2017 page 1 of 6 Assessment Schedule 2017 Earth and Space Science: Demonstrate understanding of processes in the atmosphere system (91414) Evidence Statement
Environmental Science Chapter 13 Atmosphere and Climate Change Review
 Environmental Science Chapter 13 Atmosphere and Climate Change Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Climate in a region is a. the long-term,
Environmental Science Chapter 13 Atmosphere and Climate Change Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Climate in a region is a. the long-term,
CAM-Chem Chemical Forecasts
 CAM-Chem Chemical Forecasts D. Kinnison, J-F Lamarque, F. Vitt, S. Tilmes, C. Homeyer, L. Pan, S. Honomichi, J. Luo, E. Apel, R. Hornbrook, & A. Weinheimer (NCAR) A. Saiz-Lopez & R. Fernandez (CISC, Spain)
CAM-Chem Chemical Forecasts D. Kinnison, J-F Lamarque, F. Vitt, S. Tilmes, C. Homeyer, L. Pan, S. Honomichi, J. Luo, E. Apel, R. Hornbrook, & A. Weinheimer (NCAR) A. Saiz-Lopez & R. Fernandez (CISC, Spain)
Recent Climate History - The Instrumental Era.
 2002 Recent Climate History - The Instrumental Era. Figure 1. Reconstructed surface temperature record. Strong warming in the first and late part of the century. El Ninos and major volcanic eruptions are
2002 Recent Climate History - The Instrumental Era. Figure 1. Reconstructed surface temperature record. Strong warming in the first and late part of the century. El Ninos and major volcanic eruptions are
2. Sketch a plot of R vs. z. Comment on the shape. Explain physically why R(z) has a maximum in the atmospheric column.
 190 PROBLEMS 10. 1 Shape of the ozone layer Consider a beam of solar radiation of wavelength λ propagating downward in the vertical direction with an actinic flux I at the top of the atmosphere. Assume
190 PROBLEMS 10. 1 Shape of the ozone layer Consider a beam of solar radiation of wavelength λ propagating downward in the vertical direction with an actinic flux I at the top of the atmosphere. Assume
Climate Change 2007: The Physical Science Basis
 Climate Change 2007: The Physical Science Basis Working Group I Contribution to the IPCC Fourth Assessment Report Presented by R.K. Pachauri, IPCC Chair and Bubu Jallow, WG 1 Vice Chair Nairobi, 6 February
Climate Change 2007: The Physical Science Basis Working Group I Contribution to the IPCC Fourth Assessment Report Presented by R.K. Pachauri, IPCC Chair and Bubu Jallow, WG 1 Vice Chair Nairobi, 6 February
Tropospheric OH chemistry
 Tropospheric OH chemistry CO Oxidation mechanism: CO + OH CO 2 + H, H + O 2 + M HO 2 + M, HO 2 + NO OH + NO 2 NO 2 + hν (+O 2 ) NO + O 3 Initiation step Propagation Net: CO + 2 O 2 CO 2 + O 3 HO 2 + HO
Tropospheric OH chemistry CO Oxidation mechanism: CO + OH CO 2 + H, H + O 2 + M HO 2 + M, HO 2 + NO OH + NO 2 NO 2 + hν (+O 2 ) NO + O 3 Initiation step Propagation Net: CO + 2 O 2 CO 2 + O 3 HO 2 + HO
Measurements of Ozone. Why is Ozone Important?
 Anthropogenic Climate Changes CO 2 CFC CH 4 Human production of freons (CFCs) Ozone Hole Depletion Human production of CO2 and CH4 Global Warming Human change of land use Deforestation (from Earth s Climate:
Anthropogenic Climate Changes CO 2 CFC CH 4 Human production of freons (CFCs) Ozone Hole Depletion Human production of CO2 and CH4 Global Warming Human change of land use Deforestation (from Earth s Climate:
Summary. Introduction The solar variability Some words about the Earth's atmosphere Simulations and results
 Summary Introduction The solar variability Some words about the Earth's atmosphere Simulations and results Introduction Understanding and quantifying the natural variability of climate on decadal and centennial
Summary Introduction The solar variability Some words about the Earth's atmosphere Simulations and results Introduction Understanding and quantifying the natural variability of climate on decadal and centennial
8.2 Tropospheric ozone
 8.2 Tropospheric ozone Prev Chapter 8. Ozone Next 8.2 Tropospheric ozone Tropospheric ozone is only about 10% of the total amount of ozone contained in a vertical column in the atmosphere. However, this
8.2 Tropospheric ozone Prev Chapter 8. Ozone Next 8.2 Tropospheric ozone Tropospheric ozone is only about 10% of the total amount of ozone contained in a vertical column in the atmosphere. However, this
Future climate change in the Southern Hemisphere: Competing effects of ozone and greenhouse gases
 GEOPHYSICAL RESEARCH LETTERS, VOL. 38,, doi:10.1029/2010gl045384, 2011 Future climate change in the Southern Hemisphere: Competing effects of ozone and greenhouse gases J. M. Arblaster, 1,2,3 G. A. Meehl,
GEOPHYSICAL RESEARCH LETTERS, VOL. 38,, doi:10.1029/2010gl045384, 2011 Future climate change in the Southern Hemisphere: Competing effects of ozone and greenhouse gases J. M. Arblaster, 1,2,3 G. A. Meehl,
First observations of atmospheric Hydrogen Peroxide (H 2. ) and Methylhydroperoxide (CH 3
 First observations of atmospheric Hydrogen Peroxide (H 2 ) and Methylhydroperoxide (CH 3 OOH) in West Antarctica: comparison of experiment and model results Markus M. Frey a, Richard W. Stewart b, Joseph
First observations of atmospheric Hydrogen Peroxide (H 2 ) and Methylhydroperoxide (CH 3 OOH) in West Antarctica: comparison of experiment and model results Markus M. Frey a, Richard W. Stewart b, Joseph
Twenty-five winters of unexpected Eurasian cooling unlikely due to Arctic sea-ice loss
 SUPPLEMENTARY INFORMATION DOI: 10.1038/NGEO2820 Twenty-five winters of unexpected Eurasian cooling unlikely due to Arctic sea-ice loss Kelly E. McCusker 1,2, John C. Fyfe 2 & Michael Sigmond 2 1 School
SUPPLEMENTARY INFORMATION DOI: 10.1038/NGEO2820 Twenty-five winters of unexpected Eurasian cooling unlikely due to Arctic sea-ice loss Kelly E. McCusker 1,2, John C. Fyfe 2 & Michael Sigmond 2 1 School
Supplementary Figure 1 Trends of annual mean maximum ocean mixed layer depth. Trends from uninitialized simulations (a) and assimilation simulation
 Supplementary Figure 1 Trends of annual mean maximum ocean mixed layer depth. Trends from uninitialized simulations (a) and assimilation simulation (b) from 1970-1995 (units: m yr -1 ). The dots show grids
Supplementary Figure 1 Trends of annual mean maximum ocean mixed layer depth. Trends from uninitialized simulations (a) and assimilation simulation (b) from 1970-1995 (units: m yr -1 ). The dots show grids
The ocean s overall role in climate
 The ocean s overall role in climate - moderates climate in time (diurnally, annually) - redistributes heat spatially in the largescale ocean circulation - lower albedo (sea ice higher albedo) - dry atmosphere
The ocean s overall role in climate - moderates climate in time (diurnally, annually) - redistributes heat spatially in the largescale ocean circulation - lower albedo (sea ice higher albedo) - dry atmosphere
Observed Trends in Wind Speed over the Southern Ocean
 GEOPHYSICAL RESEARCH LETTERS, VOL. 39,, doi:10.1029/2012gl051734, 2012 Observed s in over the Southern Ocean L. B. Hande, 1 S. T. Siems, 1 and M. J. Manton 1 Received 19 March 2012; revised 8 May 2012;
GEOPHYSICAL RESEARCH LETTERS, VOL. 39,, doi:10.1029/2012gl051734, 2012 Observed s in over the Southern Ocean L. B. Hande, 1 S. T. Siems, 1 and M. J. Manton 1 Received 19 March 2012; revised 8 May 2012;
Solar Influence on climate: Particle precipitation effects on the southern hemisphere tropical/subtropical lower stratosphere temperature
 Solar Influence on climate: Particle precipitation effects on the southern hemisphere tropical/subtropical lower stratosphere temperature Luis Eduardo Vieira and Ligia Alves da Silva SOHO observations
Solar Influence on climate: Particle precipitation effects on the southern hemisphere tropical/subtropical lower stratosphere temperature Luis Eduardo Vieira and Ligia Alves da Silva SOHO observations
Eurasian Snow Cover Variability and Links with Stratosphere-Troposphere Coupling and Their Potential Use in Seasonal to Decadal Climate Predictions
 US National Oceanic and Atmospheric Administration Climate Test Bed Joint Seminar Series NCEP, Camp Springs, Maryland, 22 June 2011 Eurasian Snow Cover Variability and Links with Stratosphere-Troposphere
US National Oceanic and Atmospheric Administration Climate Test Bed Joint Seminar Series NCEP, Camp Springs, Maryland, 22 June 2011 Eurasian Snow Cover Variability and Links with Stratosphere-Troposphere
Supplement of Nitrate deposition and preservation in the snowpack along a traverse from coast to the ice sheet summit (Dome A) in East Antarctica
 Supplement of The Cryosphere, 12, 1177 1194, 2018 https://doi.org/10.5194/tc-12-1177-2018-supplement Author(s) 2018. This work is distributed under the Creative Commons Attribution 4.0 License. Supplement
Supplement of The Cryosphere, 12, 1177 1194, 2018 https://doi.org/10.5194/tc-12-1177-2018-supplement Author(s) 2018. This work is distributed under the Creative Commons Attribution 4.0 License. Supplement
Trends of Tropospheric Ozone over China Based on Satellite Data ( )
 ADVANCES IN CLIMATE CHANGE RESEARCH 2(1): 43 48, 2011 www.climatechange.cn DOI: 10.3724/SP.J.1248.2011.00043 ARTICLE Trends of Tropospheric Ozone over China Based on Satellite Data (1979 2005) Xiaobin
ADVANCES IN CLIMATE CHANGE RESEARCH 2(1): 43 48, 2011 www.climatechange.cn DOI: 10.3724/SP.J.1248.2011.00043 ARTICLE Trends of Tropospheric Ozone over China Based on Satellite Data (1979 2005) Xiaobin
Data and formulas at the end. Exam would be Weds. May 8, 2008
 ATMS 321: Science of Climate Practice Mid Term Exam - Spring 2008 page 1 Atmospheric Sciences 321 Science of Climate Practice Mid-Term Examination: Would be Closed Book Data and formulas at the end. Exam
ATMS 321: Science of Climate Practice Mid Term Exam - Spring 2008 page 1 Atmospheric Sciences 321 Science of Climate Practice Mid-Term Examination: Would be Closed Book Data and formulas at the end. Exam
What is the IPCC? Intergovernmental Panel on Climate Change
 IPCC WG1 FAQ What is the IPCC? Intergovernmental Panel on Climate Change The IPCC is a scientific intergovernmental body set up by the World Meteorological Organization (WMO) and by the United Nations
IPCC WG1 FAQ What is the IPCC? Intergovernmental Panel on Climate Change The IPCC is a scientific intergovernmental body set up by the World Meteorological Organization (WMO) and by the United Nations
The North Atlantic Oscillation: Climatic Significance and Environmental Impact
 1 The North Atlantic Oscillation: Climatic Significance and Environmental Impact James W. Hurrell National Center for Atmospheric Research Climate and Global Dynamics Division, Climate Analysis Section
1 The North Atlantic Oscillation: Climatic Significance and Environmental Impact James W. Hurrell National Center for Atmospheric Research Climate and Global Dynamics Division, Climate Analysis Section
Chapter outline. Reference 12/13/2016
 Chapter 2. observation CC EST 5103 Climate Change Science Rezaul Karim Environmental Science & Technology Jessore University of science & Technology Chapter outline Temperature in the instrumental record
Chapter 2. observation CC EST 5103 Climate Change Science Rezaul Karim Environmental Science & Technology Jessore University of science & Technology Chapter outline Temperature in the instrumental record
Externally forced and internal variability in multi-decadal climate evolution
 Externally forced and internal variability in multi-decadal climate evolution During the last 150 years, the increasing atmospheric concentration of anthropogenic greenhouse gases has been the main driver
Externally forced and internal variability in multi-decadal climate evolution During the last 150 years, the increasing atmospheric concentration of anthropogenic greenhouse gases has been the main driver
Arctic sea ice response to atmospheric forcings with varying levels of anthropogenic warming and climate variability
 GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2010gl044988, 2010 Arctic sea ice response to atmospheric forcings with varying levels of anthropogenic warming and climate variability Jinlun Zhang,
GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2010gl044988, 2010 Arctic sea ice response to atmospheric forcings with varying levels of anthropogenic warming and climate variability Jinlun Zhang,
Whole Atmosphere Simulation of Anthropogenic Climate Change
 Whole Atmosphere Simulation of Anthropogenic Climate Change Stan Solomon, Hanli Liu, Dan Marsh, Joe McInerney, Liying Qian, and Francis Vitt High Altitude Observatory National Center for Atmospheric Research
Whole Atmosphere Simulation of Anthropogenic Climate Change Stan Solomon, Hanli Liu, Dan Marsh, Joe McInerney, Liying Qian, and Francis Vitt High Altitude Observatory National Center for Atmospheric Research
Decreasing trend of tropical cyclone frequency in 228-year high-resolution AGCM simulations
 GEOPHYSICAL RESEARCH LETTERS, VOL. 39,, doi:10.1029/2012gl053360, 2012 Decreasing trend of tropical cyclone frequency in 228-year high-resolution AGCM simulations Masato Sugi 1,2 and Jun Yoshimura 2 Received
GEOPHYSICAL RESEARCH LETTERS, VOL. 39,, doi:10.1029/2012gl053360, 2012 Decreasing trend of tropical cyclone frequency in 228-year high-resolution AGCM simulations Masato Sugi 1,2 and Jun Yoshimura 2 Received
ATMS 321: Sci. of Climate Final Examination Study Guide Page 1 of 4
 ATMS 321: Sci. of Climate Final Examination Study Guide Page 1 of 4 Atmospheric Sciences 321: Final Examination Study Guide The final examination will consist of similar questions Science of Climate Multiple
ATMS 321: Sci. of Climate Final Examination Study Guide Page 1 of 4 Atmospheric Sciences 321: Final Examination Study Guide The final examination will consist of similar questions Science of Climate Multiple
On the remarkable Arctic winter in 2008/2009
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 114,, doi:10.1029/2009jd012273, 2009 On the remarkable Arctic winter in 2008/2009 K. Labitzke 1 and M. Kunze 1 Received 17 April 2009; revised 11 June 2009; accepted
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 114,, doi:10.1029/2009jd012273, 2009 On the remarkable Arctic winter in 2008/2009 K. Labitzke 1 and M. Kunze 1 Received 17 April 2009; revised 11 June 2009; accepted
ABSTRACT 2 DATA 1 INTRODUCTION
 16B.7 MODEL STUDY OF INTERMEDIATE-SCALE TROPICAL INERTIA GRAVITY WAVES AND COMPARISON TO TWP-ICE CAM- PAIGN OBSERVATIONS. S. Evan 1, M. J. Alexander 2 and J. Dudhia 3. 1 University of Colorado, Boulder,
16B.7 MODEL STUDY OF INTERMEDIATE-SCALE TROPICAL INERTIA GRAVITY WAVES AND COMPARISON TO TWP-ICE CAM- PAIGN OBSERVATIONS. S. Evan 1, M. J. Alexander 2 and J. Dudhia 3. 1 University of Colorado, Boulder,
Modeled response of Greenland snowmelt to the presence of biomass burning based absorbing aerosols
 Modeled response of Greenland snowmelt to the presence of biomass burning based absorbing aerosols Jamie Ward University of Michigan Climate and Space Science 1 Introduction Black carbon (BC): aerosol
Modeled response of Greenland snowmelt to the presence of biomass burning based absorbing aerosols Jamie Ward University of Michigan Climate and Space Science 1 Introduction Black carbon (BC): aerosol
Consequences for Climate Feedback Interpretations
 CO 2 Forcing Induces Semi-direct Effects with Consequences for Climate Feedback Interpretations Timothy Andrews and Piers M. Forster School of Earth and Environment, University of Leeds, Leeds, LS2 9JT,
CO 2 Forcing Induces Semi-direct Effects with Consequences for Climate Feedback Interpretations Timothy Andrews and Piers M. Forster School of Earth and Environment, University of Leeds, Leeds, LS2 9JT,
Baseline Ozone in Western North America: Measurements and Models. David Parrish
 Baseline Ozone in Western North America: Measurements and Models David Parrish CIRES University of Colorado NOAA/ESRL Chemical Sciences Division Boulder, Colorado USA Consultant with David.D.Parrish, LLC
Baseline Ozone in Western North America: Measurements and Models David Parrish CIRES University of Colorado NOAA/ESRL Chemical Sciences Division Boulder, Colorado USA Consultant with David.D.Parrish, LLC
Impacts of Climate Change on Autumn North Atlantic Wave Climate
 Impacts of Climate Change on Autumn North Atlantic Wave Climate Will Perrie, Lanli Guo, Zhenxia Long, Bash Toulany Fisheries and Oceans Canada, Bedford Institute of Oceanography, Dartmouth, NS Abstract
Impacts of Climate Change on Autumn North Atlantic Wave Climate Will Perrie, Lanli Guo, Zhenxia Long, Bash Toulany Fisheries and Oceans Canada, Bedford Institute of Oceanography, Dartmouth, NS Abstract
Comparison of Global Mean Temperature Series
 ADVANCES IN CLIMATE CHANGE RESEARCH 2(4): 187 192, 2011 www.climatechange.cn DOI: 10.3724/SP.J.1248.2011.00187 REVIEW Comparison of Global Mean Temperature Series Xinyu Wen 1,2, Guoli Tang 3, Shaowu Wang
ADVANCES IN CLIMATE CHANGE RESEARCH 2(4): 187 192, 2011 www.climatechange.cn DOI: 10.3724/SP.J.1248.2011.00187 REVIEW Comparison of Global Mean Temperature Series Xinyu Wen 1,2, Guoli Tang 3, Shaowu Wang
Chemistry 471/671. Atmospheric Chemistry III: Stratospheric Ozone Depletion
 Chemistry 471/671 Atmospheric Chemistry III: Stratospheric Ozone Depletion 2 The Chapman Mechanism O 2 + hn 2 O( 1 D) O( 1 D) + O 2 + M O 3 + M Exothermic O( 1 D) + O 3 2 O 2 O 3 + hn O( 1 D) + O 2 ( 1
Chemistry 471/671 Atmospheric Chemistry III: Stratospheric Ozone Depletion 2 The Chapman Mechanism O 2 + hn 2 O( 1 D) O( 1 D) + O 2 + M O 3 + M Exothermic O( 1 D) + O 3 2 O 2 O 3 + hn O( 1 D) + O 2 ( 1
Historical trends in the jet streams
 GEOPHYSICAL RESEARCH LETTERS, VOL. 35, L08803, doi:10.1029/2008gl033614, 2008 Historical trends in the jet streams Cristina L. Archer 1 and Ken Caldeira 1 Received 12 February 2008; revised 10 March 2008;
GEOPHYSICAL RESEARCH LETTERS, VOL. 35, L08803, doi:10.1029/2008gl033614, 2008 Historical trends in the jet streams Cristina L. Archer 1 and Ken Caldeira 1 Received 12 February 2008; revised 10 March 2008;
The aerosol- and water vapor-related variability of precipitation in the West Africa Monsoon
 The aerosol- and water vapor-related variability of precipitation in the West Africa Monsoon Jingfeng Huang *, C. Zhang and J. M. Prospero Rosenstiel School of Marine and Atmospheric Science, University
The aerosol- and water vapor-related variability of precipitation in the West Africa Monsoon Jingfeng Huang *, C. Zhang and J. M. Prospero Rosenstiel School of Marine and Atmospheric Science, University
Tropospheric ozone decrease due to the Mount Pinatubo eruption: Reduced stratospheric influx
 GEOPHYSICAL RESEARCH LETTERS, VOL. 40, 5553 5558, doi:10.1002/2013gl056563, 2013 Tropospheric ozone decrease due to the Mount Pinatubo eruption: Reduced stratospheric influx Qi Tang, 1,2 Peter G. Hess,
GEOPHYSICAL RESEARCH LETTERS, VOL. 40, 5553 5558, doi:10.1002/2013gl056563, 2013 Tropospheric ozone decrease due to the Mount Pinatubo eruption: Reduced stratospheric influx Qi Tang, 1,2 Peter G. Hess,
Budget of tropospheric ozone during TOPSE from two chemical transport models
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. D8, 8372, doi:10.1029/2002jd002665, 2003 Budget of tropospheric ozone during TOPSE from two chemical transport models L. K. Emmons, 1 P. Hess, 1 A. Klonecki,
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. D8, 8372, doi:10.1029/2002jd002665, 2003 Budget of tropospheric ozone during TOPSE from two chemical transport models L. K. Emmons, 1 P. Hess, 1 A. Klonecki,
On the importance of aqueous-phase chemistry on the oxidative capacity of the troposphere: A 3-dimensional global modeling study
 C O M E C A P 2 0 1 4 e - b o o k o f p r o c e e d i n g s v o l. 2 P a g e 282 On the importance of aqueous-phase chemistry on the oxidative capacity of the troposphere: A 3-dimensional global modeling
C O M E C A P 2 0 1 4 e - b o o k o f p r o c e e d i n g s v o l. 2 P a g e 282 On the importance of aqueous-phase chemistry on the oxidative capacity of the troposphere: A 3-dimensional global modeling
P4.2 THE THREE DIMENSIONAL STRUCTURE AND TIME EVOLUTION OF THE DECADAL VARIABILITY REVEALED IN ECMWF REANALYSES
 P4.2 THE THREE DIMENSIONAL STRUCTURE AND TIME EVOLUTION OF THE DECADAL VARIABILITY REVEALED IN ECMWF REANALYSES Taehyoun Shim 1, Gyu-Ho Lim* 1 and Dong-In Lee 2 1 School of Earth and Environmental Sciences,
P4.2 THE THREE DIMENSIONAL STRUCTURE AND TIME EVOLUTION OF THE DECADAL VARIABILITY REVEALED IN ECMWF REANALYSES Taehyoun Shim 1, Gyu-Ho Lim* 1 and Dong-In Lee 2 1 School of Earth and Environmental Sciences,
ttp://news.discovery.com/earth/iceland-volcano-aurora.html
 ttp://news.discovery.com/earth/iceland-volcano-aurora.html Outline Role of volcanism on the climate system Distribution of Arctic volcanoes Types of eruptions Frequency of Arctic eruptions Influence on
ttp://news.discovery.com/earth/iceland-volcano-aurora.html Outline Role of volcanism on the climate system Distribution of Arctic volcanoes Types of eruptions Frequency of Arctic eruptions Influence on
Lower Stratospheric Cooling. and. Abrupt Change in Arctic Sea Ice
 Lower Stratospheric Cooling and Abrupt Change in Arctic Sea Ice Carl Drews March 16, 2007 ATOC 7500-002 Human Influences on Weather and Climate University of Colorado at Boulder Dr. Roger Pielke Sr, Instructor
Lower Stratospheric Cooling and Abrupt Change in Arctic Sea Ice Carl Drews March 16, 2007 ATOC 7500-002 Human Influences on Weather and Climate University of Colorado at Boulder Dr. Roger Pielke Sr, Instructor
Stratosphere Troposphere Coupling in a Relatively Simple AGCM: Impact of the Seasonal Cycle
 1 NOVEMBER 2006 N O T E S A N D C O R R E S P O N D E N C E 5721 Stratosphere Troposphere Coupling in a Relatively Simple AGCM: Impact of the Seasonal Cycle PAUL J. KUSHNER Department of Physics, University
1 NOVEMBER 2006 N O T E S A N D C O R R E S P O N D E N C E 5721 Stratosphere Troposphere Coupling in a Relatively Simple AGCM: Impact of the Seasonal Cycle PAUL J. KUSHNER Department of Physics, University
The Atmospheric Circulation
 The Atmospheric Circulation Vertical structure of the Atmosphere http://www.uwsp.edu/geo/faculty/ritter/geog101/textbook/atmosphere/atmospheric_structure.html The global heat engine [courtesy Kevin Trenberth,
The Atmospheric Circulation Vertical structure of the Atmosphere http://www.uwsp.edu/geo/faculty/ritter/geog101/textbook/atmosphere/atmospheric_structure.html The global heat engine [courtesy Kevin Trenberth,
The Climatology of Clouds using surface observations. S.G. Warren and C.J. Hahn Encyclopedia of Atmospheric Sciences.
 The Climatology of Clouds using surface observations S.G. Warren and C.J. Hahn Encyclopedia of Atmospheric Sciences Gill-Ran Jeong Cloud Climatology The time-averaged geographical distribution of cloud
The Climatology of Clouds using surface observations S.G. Warren and C.J. Hahn Encyclopedia of Atmospheric Sciences Gill-Ran Jeong Cloud Climatology The time-averaged geographical distribution of cloud
Investigating snow accumulation variability on the Antarctic Peninsula using Ground Penetrating Radar. - A tool for interpreting ice core records
 Investigating snow accumulation variability on the Antarctic Peninsula using Ground - A tool for interpreting ice core records Elizabeth R. Thomas June 2008 Scientific Report in support of Loan 824 Identifying
Investigating snow accumulation variability on the Antarctic Peninsula using Ground - A tool for interpreting ice core records Elizabeth R. Thomas June 2008 Scientific Report in support of Loan 824 Identifying
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION DOI: 10.1038/NGEO1854 Anthropogenic aerosol forcing of Atlantic tropical storms N. J. Dunstone 1, D. S. Smith 1, B. B. B. Booth 1, L. Hermanson 1, R. Eade 1 Supplementary information
SUPPLEMENTARY INFORMATION DOI: 10.1038/NGEO1854 Anthropogenic aerosol forcing of Atlantic tropical storms N. J. Dunstone 1, D. S. Smith 1, B. B. B. Booth 1, L. Hermanson 1, R. Eade 1 Supplementary information
Arctic Oxidation Chemistry
 19 July 2016 Connaught Summer Institute 1 Arctic Oxidation Chemistry Connaught Summer Institute 2016 William (Bill) Simpson Geophysical Institute and Department of Chemistry, University of Alaska Fairbanks
19 July 2016 Connaught Summer Institute 1 Arctic Oxidation Chemistry Connaught Summer Institute 2016 William (Bill) Simpson Geophysical Institute and Department of Chemistry, University of Alaska Fairbanks
The atmospheric response to solar irradiance variations: Simulations with HAMMONIA
 The atmospheric response to solar irradiance variations: Simulations with HAMMONIA Hauke Schmidt, Marco A. Giorgetta Max Planck Institute for Meteorology, Hamburg, Germany Guy P. Brasseur National Center
The atmospheric response to solar irradiance variations: Simulations with HAMMONIA Hauke Schmidt, Marco A. Giorgetta Max Planck Institute for Meteorology, Hamburg, Germany Guy P. Brasseur National Center
ATMS 321: Natural Climate Variability Chapter 11
 ATMS 321: Natural Climate Variability Chapter 11 Solar Variability: Total solar irradiance variability is relatively small about a tenth of a percent. Ultraviolet variability is larger, and so could affect
ATMS 321: Natural Climate Variability Chapter 11 Solar Variability: Total solar irradiance variability is relatively small about a tenth of a percent. Ultraviolet variability is larger, and so could affect
Arctic Climate Change. Glen Lesins Department of Physics and Atmospheric Science Dalhousie University Create Summer School, Alliston, July 2013
 Arctic Climate Change Glen Lesins Department of Physics and Atmospheric Science Dalhousie University Create Summer School, Alliston, July 2013 When was this published? Observational Evidence for Arctic
Arctic Climate Change Glen Lesins Department of Physics and Atmospheric Science Dalhousie University Create Summer School, Alliston, July 2013 When was this published? Observational Evidence for Arctic
Influence of eddy driven jet latitude on North Atlantic jet persistence and blocking frequency in CMIP3 integrations
 GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2010gl045700, 2010 Influence of eddy driven jet latitude on North Atlantic jet persistence and blocking frequency in CMIP3 integrations Elizabeth A.
GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2010gl045700, 2010 Influence of eddy driven jet latitude on North Atlantic jet persistence and blocking frequency in CMIP3 integrations Elizabeth A.
Equilibrium Climate Sensitivity: is it accurate to use a slab ocean model? Gokhan Danabasoglu and Peter R. Gent
 Equilibrium Climate Sensitivity: is it accurate to use a slab ocean model? by Gokhan Danabasoglu and Peter R. Gent National Center for Atmospheric Research Boulder, Colorado 80307 Abstract The equilibrium
Equilibrium Climate Sensitivity: is it accurate to use a slab ocean model? by Gokhan Danabasoglu and Peter R. Gent National Center for Atmospheric Research Boulder, Colorado 80307 Abstract The equilibrium
Global long lived chemical modes excited in a 3 D chemistry transport model: Stratospheric N 2 O, NO y,o 3 and CH 4 chemistry
 Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2009gl042243, 2010 Global long lived chemical modes excited in a 3 D chemistry transport model: Stratospheric N 2 O, NO y,o
Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 37,, doi:10.1029/2009gl042243, 2010 Global long lived chemical modes excited in a 3 D chemistry transport model: Stratospheric N 2 O, NO y,o
Persistent shift of the Arctic polar vortex towards the Eurasian continent in recent decades
 SUPPLEMENTARY INFORMATION DOI: 10.1038/NCLIMATE3136 Persistent shift of the Arctic polar vortex towards the Eurasian continent in recent decades Jiankai Zhang 1, Wenshou Tian 1 *, Martyn P. Chipperfield
SUPPLEMENTARY INFORMATION DOI: 10.1038/NCLIMATE3136 Persistent shift of the Arctic polar vortex towards the Eurasian continent in recent decades Jiankai Zhang 1, Wenshou Tian 1 *, Martyn P. Chipperfield
NATS 101 Section 13: Lecture 31. Air Pollution Part II
 NATS 101 Section 13: Lecture 31 Air Pollution Part II Last time we talked mainly about two types of smog:. 1. London-type smog 2. L.A.-type smog or photochemical smog What are the necessary ingredients
NATS 101 Section 13: Lecture 31 Air Pollution Part II Last time we talked mainly about two types of smog:. 1. London-type smog 2. L.A.-type smog or photochemical smog What are the necessary ingredients
Seasonal cycles of O 3, CO, and convective outflow at the tropical tropopause
 GEOPHYSICAL RESEARCH LETTERS, VOL. 33, L16802, doi:10.1029/2006gl026602, 2006 Seasonal cycles of O 3, CO, and convective outflow at the tropical tropopause Ian Folkins, 1 P. Bernath, 2 C. Boone, 2 G. Lesins,
GEOPHYSICAL RESEARCH LETTERS, VOL. 33, L16802, doi:10.1029/2006gl026602, 2006 Seasonal cycles of O 3, CO, and convective outflow at the tropical tropopause Ian Folkins, 1 P. Bernath, 2 C. Boone, 2 G. Lesins,
Tropical stratospheric zonal winds in ECMWF ERA-40 reanalysis, rocketsonde data, and rawinsonde data
 GEOPHYSICAL RESEARCH LETTERS, VOL. 32, L09806, doi:10.1029/2004gl022328, 2005 Tropical stratospheric zonal winds in ECMWF ERA-40 reanalysis, rocketsonde data, and rawinsonde data Mark P. Baldwin Northwest
GEOPHYSICAL RESEARCH LETTERS, VOL. 32, L09806, doi:10.1029/2004gl022328, 2005 Tropical stratospheric zonal winds in ECMWF ERA-40 reanalysis, rocketsonde data, and rawinsonde data Mark P. Baldwin Northwest
SUPPLEMENTARY INFORMATION
 Effect of remote sea surface temperature change on tropical cyclone potential intensity Gabriel A. Vecchi Geophysical Fluid Dynamics Laboratory NOAA Brian J. Soden Rosenstiel School for Marine and Atmospheric
Effect of remote sea surface temperature change on tropical cyclone potential intensity Gabriel A. Vecchi Geophysical Fluid Dynamics Laboratory NOAA Brian J. Soden Rosenstiel School for Marine and Atmospheric
ATOC 3500/CHEM 3151 Air Pollution Chemistry Lecture 1
 ATOC 3500/CHEM 3151 Air Pollution Chemistry Lecture 1 Note Page numbers refer to Daniel Jacob s online textbook: http://acmg.seas.harvard.edu/publications/ jacobbook/index.html Atmos = vapor + sphaira
ATOC 3500/CHEM 3151 Air Pollution Chemistry Lecture 1 Note Page numbers refer to Daniel Jacob s online textbook: http://acmg.seas.harvard.edu/publications/ jacobbook/index.html Atmos = vapor + sphaira
Linkages between Arctic sea ice loss and midlatitude
 Linkages between Arctic sea ice loss and midlatitude weather patterns Response of the wintertime atmospheric circulation to current and projected Arctic sea ice decline Gudrun Magnusdottir and Yannick
Linkages between Arctic sea ice loss and midlatitude weather patterns Response of the wintertime atmospheric circulation to current and projected Arctic sea ice decline Gudrun Magnusdottir and Yannick
10. Stratospheric chemistry. Daniel J. Jacob, Atmospheric Chemistry, Harvard University, Spring 2017
 10. Stratospheric chemistry Daniel J. Jacob, Atmospheric Chemistry, Harvard University, Spring 2017 The ozone layer Dobson unit: physical thickness (0.01 mm) of ozone layer if compressed to 1 atm, 0 o
10. Stratospheric chemistry Daniel J. Jacob, Atmospheric Chemistry, Harvard University, Spring 2017 The ozone layer Dobson unit: physical thickness (0.01 mm) of ozone layer if compressed to 1 atm, 0 o
warmest (coldest) temperatures at summer heat dispersed upward by vertical motion Prof. Jin-Yi Yu ESS200A heated by solar radiation at the base
 Pole Eq Lecture 3: ATMOSPHERE (Outline) JS JP Hadley Cell Ferrel Cell Polar Cell (driven by eddies) L H L H Basic Structures and Dynamics General Circulation in the Troposphere General Circulation in the
Pole Eq Lecture 3: ATMOSPHERE (Outline) JS JP Hadley Cell Ferrel Cell Polar Cell (driven by eddies) L H L H Basic Structures and Dynamics General Circulation in the Troposphere General Circulation in the
Trends in Climate Teleconnections and Effects on the Midwest
 Trends in Climate Teleconnections and Effects on the Midwest Don Wuebbles Zachary Zobel Department of Atmospheric Sciences University of Illinois, Urbana November 11, 2015 Date Name of Meeting 1 Arctic
Trends in Climate Teleconnections and Effects on the Midwest Don Wuebbles Zachary Zobel Department of Atmospheric Sciences University of Illinois, Urbana November 11, 2015 Date Name of Meeting 1 Arctic
Surface ozone variability and the jet position: Implications for projecting future air quality
 GEOPHYSICAL RESEARCH LETTERS, VOL. 40, 2839 2844, doi:10.1002/grl.50411, 2013 Surface ozone variability and the jet position: Implications for projecting future air quality Elizabeth A. Barnes 1 and Arlene
GEOPHYSICAL RESEARCH LETTERS, VOL. 40, 2839 2844, doi:10.1002/grl.50411, 2013 Surface ozone variability and the jet position: Implications for projecting future air quality Elizabeth A. Barnes 1 and Arlene
Centennial-scale Climate Change from Decadally-paced Explosive Volcanism
 Centennial-scale Climate Change from Decadally-paced Explosive Volcanism Yafang Zhong and Gifford Miller INSTAAR, University of Colorado at Boulder, USA Bette Otto-Bliesner, Caspar Ammann, Marika Holland,
Centennial-scale Climate Change from Decadally-paced Explosive Volcanism Yafang Zhong and Gifford Miller INSTAAR, University of Colorado at Boulder, USA Bette Otto-Bliesner, Caspar Ammann, Marika Holland,
The Formation of Precipitation Anomaly Patterns during the Developing and Decaying Phases of ENSO
 ATMOSPHERIC AND OCEANIC SCIENCE LETTERS, 2010, VOL. 3, NO. 1, 25 30 The Formation of Precipitation Anomaly Patterns during the Developing and Decaying Phases of ENSO HU Kai-Ming and HUANG Gang State Key
ATMOSPHERIC AND OCEANIC SCIENCE LETTERS, 2010, VOL. 3, NO. 1, 25 30 The Formation of Precipitation Anomaly Patterns during the Developing and Decaying Phases of ENSO HU Kai-Ming and HUANG Gang State Key
WACCM-X Simulations of Climate Change in the Upper Atmosphere Stan Solomon, Hanli Liu, Dan Marsh, Joe McInerney, Liying Qian, and Francis Vitt
 WACCM-X Simulations of Climate Change in the Upper Atmosphere Stan Solomon, Hanli Liu, Dan Marsh, Joe McInerney, Liying Qian, and Francis Vitt High Altitude Observatory National Center for Atmospheric
WACCM-X Simulations of Climate Change in the Upper Atmosphere Stan Solomon, Hanli Liu, Dan Marsh, Joe McInerney, Liying Qian, and Francis Vitt High Altitude Observatory National Center for Atmospheric
The Atmosphere. Topic 3: Global Cycles and Physical Systems. Topic 3: Global Cycles and Physical Systems. Topic 3: Global Cycles and Physical Systems
 The Atmosphere 1 How big is the atmosphere? Why is it cold in Geneva? Why do mountaineers need oxygen on Everest? 2 A relatively thin layer of gas over the Earths surface Earth s radius ~ 6400km Atmospheric
The Atmosphere 1 How big is the atmosphere? Why is it cold in Geneva? Why do mountaineers need oxygen on Everest? 2 A relatively thin layer of gas over the Earths surface Earth s radius ~ 6400km Atmospheric
Paleoceanography Spring 2008
 MIT OpenCourseWare http://ocw.mit.edu 12.740 Paleoceanography Spring 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. PALEOCEANOGRAPHY 12.740 SPRING
MIT OpenCourseWare http://ocw.mit.edu 12.740 Paleoceanography Spring 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. PALEOCEANOGRAPHY 12.740 SPRING
Challenges for Climate Science in the Arctic. Ralf Döscher Rossby Centre, SMHI, Sweden
 Challenges for Climate Science in the Arctic Ralf Döscher Rossby Centre, SMHI, Sweden The Arctic is changing 1) Why is Arctic sea ice disappearing so rapidly? 2) What are the local and remote consequences?
Challenges for Climate Science in the Arctic Ralf Döscher Rossby Centre, SMHI, Sweden The Arctic is changing 1) Why is Arctic sea ice disappearing so rapidly? 2) What are the local and remote consequences?
Simulation of Polar Ozone Depletion: An Update
 Simulation of Polar Ozone Depletion: An Update Image taken from www.zmescience.com D. Kinnison (NCAR), S. Solomon (MIT), and J. Bandoro (MIT) February 17, 2015 WACCM Working Group Meeting, Boulder Co.
Simulation of Polar Ozone Depletion: An Update Image taken from www.zmescience.com D. Kinnison (NCAR), S. Solomon (MIT), and J. Bandoro (MIT) February 17, 2015 WACCM Working Group Meeting, Boulder Co.
Climate Variability and Change Past, Present and Future An Overview
 Climate Variability and Change Past, Present and Future An Overview Dr Jim Salinger National Institute of Water and Atmospheric Research Auckland, New Zealand INTERNATIONAL WORKSHOP ON REDUCING VULNERABILITY
Climate Variability and Change Past, Present and Future An Overview Dr Jim Salinger National Institute of Water and Atmospheric Research Auckland, New Zealand INTERNATIONAL WORKSHOP ON REDUCING VULNERABILITY
Projections of future climate change
 Projections of future climate change Matthew Collins 1,2 and Catherine A. Senior 2 1 Centre for Global Atmospheric Modelling, Department of Meteorology, University of Reading 2 Met Office Hadley Centre,
Projections of future climate change Matthew Collins 1,2 and Catherine A. Senior 2 1 Centre for Global Atmospheric Modelling, Department of Meteorology, University of Reading 2 Met Office Hadley Centre,
Short-Term Climate Variability (Ch.15) Volcanos and Climate Other Causes of Holocene Climate Change
 Short-Term Climate Variability (Ch.15) Volcanos and Climate Other Causes of Holocene Climate Change Volcanos and Climate We learned in Chapter 12 that the volanos play an important role in Earth s climate
Short-Term Climate Variability (Ch.15) Volcanos and Climate Other Causes of Holocene Climate Change Volcanos and Climate We learned in Chapter 12 that the volanos play an important role in Earth s climate
Historical Changes in Climate
 Historical Changes in Climate Medieval Warm Period (MWP) Little Ice Age (LIA) Lamb, 1969 Hunters in the snow by Pieter Bruegel, 1565 Retreat of the Rhone Glacier shown by comparing the drawing from 1750
Historical Changes in Climate Medieval Warm Period (MWP) Little Ice Age (LIA) Lamb, 1969 Hunters in the snow by Pieter Bruegel, 1565 Retreat of the Rhone Glacier shown by comparing the drawing from 1750
Ozone production in the upper troposphere and the influence of aircraft during SONEX: approach of NO x -saturated conditions
 Ozone production in the upper troposphere and the influence of aircraft during SONEX: approach of NO x -saturated conditions The Harvard community has made this article openly available. Please share how
Ozone production in the upper troposphere and the influence of aircraft during SONEX: approach of NO x -saturated conditions The Harvard community has made this article openly available. Please share how
Terrestrial Climate Change Variables
 Terrestrial Climate Change Variables Content Terrestrial Climate Change Variables Surface Air Temperature Land Surface Temperature Sea Level Ice Level Aerosol Particles (acid rain) Terrestrial Climate
Terrestrial Climate Change Variables Content Terrestrial Climate Change Variables Surface Air Temperature Land Surface Temperature Sea Level Ice Level Aerosol Particles (acid rain) Terrestrial Climate
5. In which diagram is the observer experiencing the greatest intensity of insolation? A) B)
 1. Which factor has the greatest influence on the number of daylight hours that a particular Earth surface location receives? A) longitude B) latitude C) diameter of Earth D) distance from the Sun 2. In
1. Which factor has the greatest influence on the number of daylight hours that a particular Earth surface location receives? A) longitude B) latitude C) diameter of Earth D) distance from the Sun 2. In
