Week 4. Even harder stuff!
|
|
|
- Ernest Miles
- 5 years ago
- Views:
Transcription
1 Week 4 Even harder stuff!
2 Focus: SN1 and SN2 Two organic reactions Learn about two basic pathways for how these reactions happen Focus on stereochemistry
3 Focus: SN1 and SN2 You need a couple of things for a reaction Nucleophile: atom or molecule that is electron rich, looks for a position charge. It s a Lewis Base: uses its e- to form a bond Electrophile: atom or molecule that is electron poor, looking for e- charge, Lewis Acid: accepts e- to form a bond. Leaving Group: atom that leaves a molecule, taking a pair of electrons with it R-L Nuc R-Nuc L R = any type of molecule
4 Example H Nucleophile H C Cl + I product H Electrophile Carbon is e-poor because of the polarization of the Cabon-Chloride bond (chlorine is more electronegative, so it s greedy and taking a lot of the electron density, leaving carbon more positive than normal) In order for the nucleophile to come in and form a bond with the central carbon, Chlorine needs to leave! (remember the octet rule, and carbon forms only 4 full bonds) Leaving groups are recognizable as they are usually halide groups! Notice the iodine? Yes, it is a halide, but it s a good nucleophile and will stick to the carbon!
5 Unimolecular nucleophilic substitution reactions (SN1) acetate ion tert-butylchloride H 3 C C Cl Nucleophile O O C O H 3 C C OC Cl Electrophile Leaving Group For this reaction, you have a Leaving Group, nucleophile and electrophile! Reaction depends only on the concentration of t-butyl chloride, it s independent of the acetate ion. First order rate law: rate = k [ t-bucl ] k=constant, [ ] = concentration of molecule The first step to this reaction involved t-bucl this is called the slow step of a reaction or the rate-determining step or RDS As was determined by the 1 st order rate law only t-bucl is involved in the first step HENCE why this is called a UNIMOLECULAR reaction
6 Step 1: Bromine Leaves Step 2: Nucleophile comes in to attack the positive C Here the extra proton is just coming off to form an OH group If you were curious, it s called a hydrolysis reaction because water comes in as a nucleophile!
7 Step 1 Focus Step 1: Leaving Group comes off It comes off primarily because it s more stable in solution than it is stuck to a molecule As it leaves, it takes a pair of electrons By taking a pair of electrons it leaves a positively charged carbon called a carbocation Carbo = carbon Cation = positively charged ion
8 Step 1 Focus Transition State * H 3 C C Cl As Chlorine leaves, it takes the electrons in that bond, so it becomes more negatively charged, while carbon becomes positively charged
9 SN1 mechanism H 3 C C Cl slow H 3 C C Cl Step 1 Arrows show movement of e- This is the carbocation It s a reactive intermediate But it s stabilized by the electron donating groups, the methyls H 3 C C O O C fast O H 3 C C OC Step 2
10 Energy Diagram
11 If the Leaving Group is At a chiral center? Then, we care about the stereochemistry of the SN1 reaction! When the carbocation is formed, the Nuc can attack from the front or back! So you can get both types of enantiomers in an SN1 rxn.
12 Here s another way to see this Racemic means both (S) and (R) enantiomers
13 SN1 Summary First step is the rate determining step Leaving Group comes off the electrophile Forms a carbocation You can usually tell because it is stabilized by electron donating groups like methyls! Then, the nucleophile attacks It can attack frontside and backside!
14 Bimolecular nucleophilic substitution reactions (SN2) H 2 C Cl OH H 2 C OH Cl For this reaction, you have a Leaving Group, nucleophile and electrophile! Reaction depends on the concentrations of both starting materials! Second order rate law: rate = k [ EtCl ] [ OH ] Both chloroethane and Hydroxide must be present in the first/slowest step of this reaction HENCE why it s called Bimolecular!!
15 SN2 Transition State This carbon in the transition state does have 5 bonds but it s ok, because they are just partial bonds between OH and Br, NOT full bonds. All at the same, partial bond is forming the C-OH bond while partial bond is breaking between C-Cl
16 Energy Diagram
17 SN2 stereochemistry IF Leaving Group is at a chiral center Nucleophile can only do a backside attack because L.G. hasn t left yet. No frontside attack! Forms only one enantiomer!
OChem 1 Mechanism Flashcards. Dr. Peter Norris, 2018
 OChem 1 Mechanism Flashcards Dr. Peter Norris, 2018 Mechanism Basics Chemical change involves bonds forming and breaking; a mechanism describes those changes using curved arrows to describe the electrons
OChem 1 Mechanism Flashcards Dr. Peter Norris, 2018 Mechanism Basics Chemical change involves bonds forming and breaking; a mechanism describes those changes using curved arrows to describe the electrons
OChem 1 Mechanism Flashcards. Dr. Peter Norris, 2015
 OChem 1 Mechanism Flashcards Dr. Peter Norris, 2015 Mechanism Basics Chemical change involves bonds forming and breaking; a mechanism describes those changes using curved arrows to describe the electrons
OChem 1 Mechanism Flashcards Dr. Peter Norris, 2015 Mechanism Basics Chemical change involves bonds forming and breaking; a mechanism describes those changes using curved arrows to describe the electrons
Glendale Community College Chemistry 105 Exam. 3 Lecture Notes Chapters 6 & 7
 Sevada Chamras, Ph.D. Glendale Community College Chemistry 105 Exam. 3 Lecture Notes Chapters 6 & 7 Description: Examples: 3 Major Types of Organic Halides: 1. Alkyl Halides: Chapter 6 (Part 1/2) : Alkyl
Sevada Chamras, Ph.D. Glendale Community College Chemistry 105 Exam. 3 Lecture Notes Chapters 6 & 7 Description: Examples: 3 Major Types of Organic Halides: 1. Alkyl Halides: Chapter 6 (Part 1/2) : Alkyl
Chapter 5. Nucleophilic aliphatic substitution mechanism. by G.DEEPA
 Chapter 5 Nucleophilic aliphatic substitution mechanism by G.DEEPA 1 Introduction The polarity of a carbon halogen bond leads to the carbon having a partial positive charge In alkyl halides this polarity
Chapter 5 Nucleophilic aliphatic substitution mechanism by G.DEEPA 1 Introduction The polarity of a carbon halogen bond leads to the carbon having a partial positive charge In alkyl halides this polarity
Organic Chemistry CHM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl Halides: Substitution Reactions - Chapter 6 (Wade)
 rganic Chemistry CM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl alides: Substitution Reactions - Chapter 6 (Wade) Chapter utline I. Intro to RX (6-1 - 6-7) II. Substitution Reactions A) S N 2 (6-8,
rganic Chemistry CM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl alides: Substitution Reactions - Chapter 6 (Wade) Chapter utline I. Intro to RX (6-1 - 6-7) II. Substitution Reactions A) S N 2 (6-8,
8.8 Unimolecular Nucleophilic Substitution S N 1
 8.8 Unimolecular Nucleophilic Substitution S N 1 A question. Tertiary alkyl halides are very unreactive in substitutions that proceed by the S N 2 mechanism. Do they undergo nucleophilic substitution at
8.8 Unimolecular Nucleophilic Substitution S N 1 A question. Tertiary alkyl halides are very unreactive in substitutions that proceed by the S N 2 mechanism. Do they undergo nucleophilic substitution at
Chapter 9. Nucleophilic Substitution and ß-Elimination
 Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides"
 Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides" t Introduction" The polarity of a carbon-halogen bond leads to the carbon having a partial positive charge"
Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides" t Introduction" The polarity of a carbon-halogen bond leads to the carbon having a partial positive charge"
Physical Properties: Structure:
 Nomenclature: Functional group suffix = -ol Functional group prefix = hydroxy- Primary, secondary or tertiary? Alcohols are described as primary (1 o ), secondary (2 o ) or tertiary (3 o ) depending on
Nomenclature: Functional group suffix = -ol Functional group prefix = hydroxy- Primary, secondary or tertiary? Alcohols are described as primary (1 o ), secondary (2 o ) or tertiary (3 o ) depending on
6-2 This exercise is worked out on page 220 as "Working with Concepts".
 Copyright 2009 James K Whitesell 6-1 Although we can approach this exercise from a chemical perspective, one can also teach a non-chemist how to derive the answer once the name of the starting material
Copyright 2009 James K Whitesell 6-1 Although we can approach this exercise from a chemical perspective, one can also teach a non-chemist how to derive the answer once the name of the starting material
Reactions of Alkyl Halides with Nucleophiles and Bases a substitution reaction
 Reactions of Alkyl Halides with Nucleophiles and Bases a substitution reaction Nucleophilic substitution and base induced elimination are among most widely occurring and versatile reaction types in organic
Reactions of Alkyl Halides with Nucleophiles and Bases a substitution reaction Nucleophilic substitution and base induced elimination are among most widely occurring and versatile reaction types in organic
Acid-Base -Bronsted-Lowry model: -Lewis model: -The more equilibrium lies to the right = More [H 3 O + ] = Higher K a = Lower pk a = Stronger acid
![Acid-Base -Bronsted-Lowry model: -Lewis model: -The more equilibrium lies to the right = More [H 3 O + ] = Higher K a = Lower pk a = Stronger acid Acid-Base -Bronsted-Lowry model: -Lewis model: -The more equilibrium lies to the right = More [H 3 O + ] = Higher K a = Lower pk a = Stronger acid](/thumbs/95/123929992.jpg) Revision Hybridisation -The valence electrons of a Carbon atom sit in 1s 2 2s 2 2p 2 orbitals that are different in energy. It has 2 x 2s electrons + 2 x 2p electrons are available to form 4 covalent bonds.
Revision Hybridisation -The valence electrons of a Carbon atom sit in 1s 2 2s 2 2p 2 orbitals that are different in energy. It has 2 x 2s electrons + 2 x 2p electrons are available to form 4 covalent bonds.
S N 1 Displacement Reactions
 S N 1 Displacement Reactions Tertiary alkyl halides cannot undergo S N 2 reactions because of the severe steric hindrance blocking a backside approach of the nucleophile. They can, however, react via an
S N 1 Displacement Reactions Tertiary alkyl halides cannot undergo S N 2 reactions because of the severe steric hindrance blocking a backside approach of the nucleophile. They can, however, react via an
BSc. II 3 rd Semester. Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1
 BSc. II 3 rd Semester Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1 Introduction to Alkyl Halides Alkyl halides are organic molecules containing a halogen atom bonded to an
BSc. II 3 rd Semester Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1 Introduction to Alkyl Halides Alkyl halides are organic molecules containing a halogen atom bonded to an
Organic Reactions Susbstitution S N. Dr. Sapna Gupta
 Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
CHAPTER 7. Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination
 CHAPTER 7 Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 7-1 Solvolysis of Tertiary and Secondary Haloalkanes The rate of S N 2 reactions decrease dramatically
CHAPTER 7 Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 7-1 Solvolysis of Tertiary and Secondary Haloalkanes The rate of S N 2 reactions decrease dramatically
Problem Set 8: Substitution Reactions-ANSWER KEY. (b) nucleophile NH 3 H C. (d) H 3 N
 Problem Set 8: Substitution Reactions-ANSWER KEY hemistry 260 rganic hemistry 1. The answers are (1), (3) and (5). Nucleophiles generally have lone pair and/or negative charge. 2. (a) neither 4 (c) nucleophile
Problem Set 8: Substitution Reactions-ANSWER KEY hemistry 260 rganic hemistry 1. The answers are (1), (3) and (5). Nucleophiles generally have lone pair and/or negative charge. 2. (a) neither 4 (c) nucleophile
CH 3 CHCH 3 CH 3 CHCH 3 Isopropyl cation. Oxomium ion intermediate. intermediate (an electrophile)
 Understanding (as opposed to memorizing) mechanisms is critical to mastering organic chemistry. Although the mechanisms you encounter throughout the course may seem entirely different, they are actually
Understanding (as opposed to memorizing) mechanisms is critical to mastering organic chemistry. Although the mechanisms you encounter throughout the course may seem entirely different, they are actually
4. Alkenes are nucleophiles and like to attack electrophiles, like protons, with positive charges:
 Mechanism Steps: Answer Key 1. Alcohols are nucleophilic and like to grab protons,, to form water molecules: 2. 3. 4. Alkenes are nucleophiles and like to attack electrophiles, like protons, with positive
Mechanism Steps: Answer Key 1. Alcohols are nucleophilic and like to grab protons,, to form water molecules: 2. 3. 4. Alkenes are nucleophiles and like to attack electrophiles, like protons, with positive
Lecture 18 Organic Chemistry 1
 CEM 232 rganic Chemistry I at Chicago Lecture 18 rganic Chemistry 1 Professor Duncan Wardrop March 9, 2010 1 Nucleophilicity nucleophilicity: measures the strength of the nucleophile ; more nucleophilic
CEM 232 rganic Chemistry I at Chicago Lecture 18 rganic Chemistry 1 Professor Duncan Wardrop March 9, 2010 1 Nucleophilicity nucleophilicity: measures the strength of the nucleophile ; more nucleophilic
Basic Organic Chemistry Course code : CHEM (Pre-requisites : CHEM 11122)
 Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Chapter 7 Substitution Reactions 7.1 Introduction to Substitution Reactions Substitution Reactions: two reactants exchange parts to give new products
 hapter 7 Substitution eactions 7.1 Introduction to Substitution eactions Substitution eactions: two reactants exchange parts to give new products A-B + -D A-D + B- 3 2 + Br 3 2 Br + Elimination eaction:
hapter 7 Substitution eactions 7.1 Introduction to Substitution eactions Substitution eactions: two reactants exchange parts to give new products A-B + -D A-D + B- 3 2 + Br 3 2 Br + Elimination eaction:
Understanding the basics. Mechanisms 5/24/11
 Understanding the basics Mechanisms Some Basic rgo I Reactions Mechanisms are the most mind-boggling part of organic chemistry. Students, generally speaking, have spent their time memorizing their way
Understanding the basics Mechanisms Some Basic rgo I Reactions Mechanisms are the most mind-boggling part of organic chemistry. Students, generally speaking, have spent their time memorizing their way
PAPER No. : 5; Organic Chemistry-II MODULE No. : 13; Mixed S N 1 and S N 2 Reactions
 Subject Chemistry Paper No and Title Module No and Title Module Tag 5; Organic Chemistry-II 13; Mixed S N 1 and S N 2 Reactions CHE_P5_M13 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. Nature
Subject Chemistry Paper No and Title Module No and Title Module Tag 5; Organic Chemistry-II 13; Mixed S N 1 and S N 2 Reactions CHE_P5_M13 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. Nature
Halo Alkanes and Halo Arenes
 alo Alkanes and alo Arenes Short Answer Questions: **1. Write the isomers of the compound having formula C 4 9 Br? Sol. There are five isomers of C 4 9 Br. These are: 2-bromobutane is expected to exhibit
alo Alkanes and alo Arenes Short Answer Questions: **1. Write the isomers of the compound having formula C 4 9 Br? Sol. There are five isomers of C 4 9 Br. These are: 2-bromobutane is expected to exhibit
Elimination Reactions Heating an alkyl halide with a strong base causes elimination of a. molecule of HX
 Elimination eactions eating an alkyl halide with a strong base causes elimination of a molecule of X 1. Potassium hydroxide dissolved in ethanol and the sodium salts of alcohols (such as sodium ethoxide)
Elimination eactions eating an alkyl halide with a strong base causes elimination of a molecule of X 1. Potassium hydroxide dissolved in ethanol and the sodium salts of alcohols (such as sodium ethoxide)
ORGANIC - EGE 5E CH. 7 - NUCLEOPHILIC SUBSTITUTION AND ELIMINATION REACTIONS
 !! www.clutchprep.com CONCEPT: INTRODUCTION TO SUBSTITUTION Previously, we discussed the various ways that acids could react with bases: Recall that in these mechanisms, electrons always travel from density
!! www.clutchprep.com CONCEPT: INTRODUCTION TO SUBSTITUTION Previously, we discussed the various ways that acids could react with bases: Recall that in these mechanisms, electrons always travel from density
Nucleophile. Reaction Intermediate. Introduction to Reaction mechanisms. Definitions 2/25/2012
 Introduction to Reaction mechanisms Definition A reaction mechanism is the step by step sequence of elementary reactions by which overall chemical change occurs. It is also a detailed description of the
Introduction to Reaction mechanisms Definition A reaction mechanism is the step by step sequence of elementary reactions by which overall chemical change occurs. It is also a detailed description of the
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2
 C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
Chapter 17. Reactions of Aromatic Compounds
 Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Although benzene s pi electrons are in a stable aromatic system, they are available to attack a strong electrophile to give
Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Although benzene s pi electrons are in a stable aromatic system, they are available to attack a strong electrophile to give
Chemistry 123: Physical and Organic Chemistry Topic 1: Organic Chemistry
 Concept Check: Topic 1: Conformation Winter 2009 Page 112 Concept Check: Topic 1: Conformation Winter 2009 Page 113 1 STEREOCHEMISTRY Winter 2009 Page 114 We have already covered two kinds of isomerism:
Concept Check: Topic 1: Conformation Winter 2009 Page 112 Concept Check: Topic 1: Conformation Winter 2009 Page 113 1 STEREOCHEMISTRY Winter 2009 Page 114 We have already covered two kinds of isomerism:
Organic Chemistry. Chapter 10
 Organic Chemistry Chapter 10 10.1 Homologous Series Overview We Are Here Organic Chemistry Organic chemistry is the chemistry of carbon containing compounds. From the very simple: methane To the very complex:
Organic Chemistry Chapter 10 10.1 Homologous Series Overview We Are Here Organic Chemistry Organic chemistry is the chemistry of carbon containing compounds. From the very simple: methane To the very complex:
Lab Activity 9: Introduction to Organic Chemical Reactivity, Lab 5 Prelab, Reflux
 Lab Activity 9: Introduction to Organic Chemical Reactivity, Lab 5 Prelab, Reflux Objectives 1. Identify structural features (pi bonds, bond polarity, lone pairs) of a compound 2. Determine whether a structural
Lab Activity 9: Introduction to Organic Chemical Reactivity, Lab 5 Prelab, Reflux Objectives 1. Identify structural features (pi bonds, bond polarity, lone pairs) of a compound 2. Determine whether a structural
Organic Chemistry Practice Problems: Solutions
 rganic Chemistry Practice Problems: Solutions 1. 2. a. B, A b. D, B c. A, D d. D, A a. Resonance b. Electronegativity of fluorine atoms F F c. Neither is very acidic, but the oxygen will help stabilise
rganic Chemistry Practice Problems: Solutions 1. 2. a. B, A b. D, B c. A, D d. D, A a. Resonance b. Electronegativity of fluorine atoms F F c. Neither is very acidic, but the oxygen will help stabilise
Chapter 10 Radical Reactions"
 Chapter 10 Radical Reactions Radicals are intermediates with an unpaired electron H. Cl. Hydrogen radical t Often called free radicals What are radicals? Chlorine radical t Formed by homolytic bond cleavage
Chapter 10 Radical Reactions Radicals are intermediates with an unpaired electron H. Cl. Hydrogen radical t Often called free radicals What are radicals? Chlorine radical t Formed by homolytic bond cleavage
Organic Chemistry. Alkenes (2)
 For updated version, please click on http://ocw.ump.edu.my Organic Chemistry Alkenes (2) by Dr. Seema Zareen & Dr. Izan Izwan Misnon Faculty Industrial Science & Technology seema@ump.edu.my & iezwan@ump.edu.my
For updated version, please click on http://ocw.ump.edu.my Organic Chemistry Alkenes (2) by Dr. Seema Zareen & Dr. Izan Izwan Misnon Faculty Industrial Science & Technology seema@ump.edu.my & iezwan@ump.edu.my
Chapter 10 Lecture Outline
 Organic Chemistry, Fifth Edition Janice Gorzynski Smith University of Hawai i Chapter 10 Lecture Outline Modified by Dr. Juliet Hahn Copyright 2017 McGraw-Hill Education. All rights reserved. No reproduction
Organic Chemistry, Fifth Edition Janice Gorzynski Smith University of Hawai i Chapter 10 Lecture Outline Modified by Dr. Juliet Hahn Copyright 2017 McGraw-Hill Education. All rights reserved. No reproduction
C h a p t e r S e v e n : Substitution Reactions S N 2 O H H H O H H. Br -
 C h a p t e r S e v e n : Substitution Reactions Br Br S N 2 CM 321: Summary of Important Concepts YConcepts for Chapter 7: Substitution Reactions I. Nomenclature of alkyl halides, R X A. Common name:
C h a p t e r S e v e n : Substitution Reactions Br Br S N 2 CM 321: Summary of Important Concepts YConcepts for Chapter 7: Substitution Reactions I. Nomenclature of alkyl halides, R X A. Common name:
Chapter 11, Part 1: Polar substitution reactions involving alkyl halides
 hapter 11, Part 1: Polar substitution reactions involving alkyl halides Overview: The nature of alkyl halides and other groups with electrophilic sp 3 hybridized leads them to react with nucleophiles and
hapter 11, Part 1: Polar substitution reactions involving alkyl halides Overview: The nature of alkyl halides and other groups with electrophilic sp 3 hybridized leads them to react with nucleophiles and
Learning Guide for Chapter 17 - Dienes
 Learning Guide for Chapter 17 - Dienes I. Isolated, conjugated, and cumulated dienes II. Reactions involving allylic cations or radicals III. Diels-Alder Reactions IV. Aromaticity I. Isolated, Conjugated,
Learning Guide for Chapter 17 - Dienes I. Isolated, conjugated, and cumulated dienes II. Reactions involving allylic cations or radicals III. Diels-Alder Reactions IV. Aromaticity I. Isolated, Conjugated,
1-What is substitution reaction? 2-What are can Nucleophilic Substitution Reaction? 3- SN1 reaction. 4-SN2 reaction 5- mechanisms of SN1&SN2
 1-What is substitution reaction? 2-What are can Nucleophilic Substitution eaction? 3- SN1 reaction. 4-SN2 reaction 5- mechanisms of SN1&SN2 1- SUBSTITUTION EACTIONS 1-Substitution eaction In this type
1-What is substitution reaction? 2-What are can Nucleophilic Substitution eaction? 3- SN1 reaction. 4-SN2 reaction 5- mechanisms of SN1&SN2 1- SUBSTITUTION EACTIONS 1-Substitution eaction In this type
CHEM Lecture 7
 CEM 494 Special Topics in Chemistry Illinois at Chicago CEM 494 - Lecture 7 Prof. Duncan Wardrop ctober 22, 2012 CEM 494 Special Topics in Chemistry Illinois at Chicago Preparation of Alkenes Elimination
CEM 494 Special Topics in Chemistry Illinois at Chicago CEM 494 - Lecture 7 Prof. Duncan Wardrop ctober 22, 2012 CEM 494 Special Topics in Chemistry Illinois at Chicago Preparation of Alkenes Elimination
Nucleophiles: nucleus liking species. Nucleophilic substitution
 1 Nucleophiles: nucleus liking species Electron rich Possesses negative charge or non bonded valence electrons Large abundance of nucleophiles in the environment (water itself is a nucleophile) Nucleophilic
1 Nucleophiles: nucleus liking species Electron rich Possesses negative charge or non bonded valence electrons Large abundance of nucleophiles in the environment (water itself is a nucleophile) Nucleophilic
Essential Organic Chemistry. Chapter 9
 Essential Organic Chemistry Paula Yurkanis Bruice Chapter 9 Substitution and Elimination Reactions of Alkyl Halides 9.1 How Alkyl Halides React Substitution Reactions One group takes the place of another.
Essential Organic Chemistry Paula Yurkanis Bruice Chapter 9 Substitution and Elimination Reactions of Alkyl Halides 9.1 How Alkyl Halides React Substitution Reactions One group takes the place of another.
Chemistry of the Halogenoalkanes SN1 and SN2 Reaction Mechanisms
 1 The primary halogenoalkane bromomethane reacts via an SN2 mechanism. Name: lass: Date: / / The tertiary halogenoalkane 2-bromo-2-methylpropane reacts via an SN1 mechanism. hemistry of the alogenoalkanes
1 The primary halogenoalkane bromomethane reacts via an SN2 mechanism. Name: lass: Date: / / The tertiary halogenoalkane 2-bromo-2-methylpropane reacts via an SN1 mechanism. hemistry of the alogenoalkanes
Chemistry 210 Organic Chemistry I Fall Semester 2000 Dr. Rainer Glaser
 hemistry 210 rganic hemistry I Fall Semester 2000 Dr. Rainer Glaser Examination #2 Alkyl alides: Their Synthesis by alogenation of Alkanes and Their Nucleophilic Substitution Reactions. Friday, ctober
hemistry 210 rganic hemistry I Fall Semester 2000 Dr. Rainer Glaser Examination #2 Alkyl alides: Their Synthesis by alogenation of Alkanes and Their Nucleophilic Substitution Reactions. Friday, ctober
Carbonyl Chemistry: Fundamentals
 Carbonyl Chemistry: Fundamentals What is a carbonyl group and what are its properties? A carbonyl is any compound that has a carbon double-bonded to an oxygen Carbonyls can behave as a nucleophile OR an
Carbonyl Chemistry: Fundamentals What is a carbonyl group and what are its properties? A carbonyl is any compound that has a carbon double-bonded to an oxygen Carbonyls can behave as a nucleophile OR an
Study of Chemical Reactions
 Study of Chemical Reactions Introduction to Mechanisms There are four different types of organic reactions: Additions Eliminations Substitutions Rearrangements 149 Addition Reactions Occur when 2 reactants
Study of Chemical Reactions Introduction to Mechanisms There are four different types of organic reactions: Additions Eliminations Substitutions Rearrangements 149 Addition Reactions Occur when 2 reactants
Chemistry 2000 Lecture 18: Reactions of organic compounds
 hemistry 2000 Lecture 18: Reactions of organic compounds Marc R. Roussel March 6, 2018 Marc R. Roussel Reactions of organic compounds March 6, 2018 1 / 27 Reactions of organic compounds Organic chemists
hemistry 2000 Lecture 18: Reactions of organic compounds Marc R. Roussel March 6, 2018 Marc R. Roussel Reactions of organic compounds March 6, 2018 1 / 27 Reactions of organic compounds Organic chemists
Some Arrow-Pushing Guidelines (Section 1.14) 1. Arrows follow electron movement.
 Chem 350 Jasperse Ch. 1 Notes 1 Note: The headers and associated chapters don t actually jive with the textbook we are using this summer. But otherwise this highlights a lot of the chemistry from Organic
Chem 350 Jasperse Ch. 1 Notes 1 Note: The headers and associated chapters don t actually jive with the textbook we are using this summer. But otherwise this highlights a lot of the chemistry from Organic
Facebook: UCI ORganic Chemistry Peer Tutoring King 51 Fall 2017
 1 Organic Chemistry 51A Professor King Final Exam Review Session December 7 th, 2017 Will Cabanela (rcabanel@uci.edu) Amanda Pinski (apinski@uci.edu) Zachary Valley (zvalley@uci.edu) http://sites.uci.edu/ochemtutors
1 Organic Chemistry 51A Professor King Final Exam Review Session December 7 th, 2017 Will Cabanela (rcabanel@uci.edu) Amanda Pinski (apinski@uci.edu) Zachary Valley (zvalley@uci.edu) http://sites.uci.edu/ochemtutors
CHEM 243 ORGANIC CHEMISTRY I Fall 2018 Exam II Information and Study Guide
 CHEM 243 ORGANIC CHEMISTRY I Fall 2018 Exam II Information and Study Guide page 1 CHEM 243 Exam II is scheduled for Monday, November 5 in DMF 477 & 481 (lab rooms). You must take the exam during your normal
CHEM 243 ORGANIC CHEMISTRY I Fall 2018 Exam II Information and Study Guide page 1 CHEM 243 Exam II is scheduled for Monday, November 5 in DMF 477 & 481 (lab rooms). You must take the exam during your normal
Substitution and Elimination reactions
 PART 3 Substitution and Elimination reactions Chapter 8. Substitution reactions of RX 9. Elimination reactions of RX 10. Substit n/elimin n of other comp ds 11. Organometallic comp ds 12. Radical reactions
PART 3 Substitution and Elimination reactions Chapter 8. Substitution reactions of RX 9. Elimination reactions of RX 10. Substit n/elimin n of other comp ds 11. Organometallic comp ds 12. Radical reactions
There are two main electronic effects that substituents can exert:
 Substituent Effects There are two main electronic effects that substituents can exert: RESONANCE effects are those that occur through the π system and can be represented by resonance structures. These
Substituent Effects There are two main electronic effects that substituents can exert: RESONANCE effects are those that occur through the π system and can be represented by resonance structures. These
2/28/2011. Chapter 6 An Overview of Organic Reactions. Organic Chemical Reactions. 6.1 Kinds of Organic Reactions
 John E. McMurry http://www.cengage.com/chemistry/mcmurry Chapter 6 An Overview of Organic Reactions CHP 6 Problems: 6.1-13, 17-36. Richard Morrison University of Georgia, Athens Organic Chemical Reactions
John E. McMurry http://www.cengage.com/chemistry/mcmurry Chapter 6 An Overview of Organic Reactions CHP 6 Problems: 6.1-13, 17-36. Richard Morrison University of Georgia, Athens Organic Chemical Reactions
Homework - Review of Chem 2310
 omework - Review of Chem 2310 Chapter 1 - Atoms and Molecules Name 1. What is organic chemistry? 2. Why is there an entire one year course devoted to the study of organic compounds? 3. Give 4 examples
omework - Review of Chem 2310 Chapter 1 - Atoms and Molecules Name 1. What is organic chemistry? 2. Why is there an entire one year course devoted to the study of organic compounds? 3. Give 4 examples
Chemistry 35 Exam 2 Answers - April 9, 2007
 Chemistry 35 Exam 2 Answers - April 9, 2007 Problem 1. (14 points) Provide the products for the reactions shown below. If stereochemistry is important, be sure to indicate stereochemistry clearly. If no
Chemistry 35 Exam 2 Answers - April 9, 2007 Problem 1. (14 points) Provide the products for the reactions shown below. If stereochemistry is important, be sure to indicate stereochemistry clearly. If no
Methyl > primary > secondary >> tertiary
 Lecture 1 Notes September 6, 2012 Welcome to CHM 427. For some of you, this will be the last organic chemistry class that you will ever take. So I see it as my personal mission to make sure that you know
Lecture 1 Notes September 6, 2012 Welcome to CHM 427. For some of you, this will be the last organic chemistry class that you will ever take. So I see it as my personal mission to make sure that you know
Reactions SN2 and SN1
 Reactions SN2 and SN1 Reactivity: Functional groups can be interconverted using a great variety of reagents. Millions of organic molecules have been synthesized via a series of functional-group interconversions.
Reactions SN2 and SN1 Reactivity: Functional groups can be interconverted using a great variety of reagents. Millions of organic molecules have been synthesized via a series of functional-group interconversions.
An Overview of Organic Reactions. Reaction types: Classification by outcome Most reactions produce changes in the functional group of the reactants:
 An Overview of Organic Reactions Reaction types: Classification by outcome Most reactions produce changes in the functional group of the reactants: 1. Addition (forward) Gain of atoms across a bond Example:
An Overview of Organic Reactions Reaction types: Classification by outcome Most reactions produce changes in the functional group of the reactants: 1. Addition (forward) Gain of atoms across a bond Example:
How alkyl halides react
 Chapter 10 1 How alkyl halides react δ+ δ- RCH 2 -X X= halogen X = higher EN C = lower EN This polar carbon-halogen bond causes alkyl halide to undergo S N and elimination reaction. 2 The mechanism of
Chapter 10 1 How alkyl halides react δ+ δ- RCH 2 -X X= halogen X = higher EN C = lower EN This polar carbon-halogen bond causes alkyl halide to undergo S N and elimination reaction. 2 The mechanism of
PRACTICE PROBLEMS UNIT 8
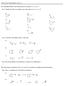 PRACTICE PROBLEMS UNIT 8 8A. Identify halides and carbocations as being 1 o, 2 o, or 3 o 8A.1 assify the following halides and carbocations as 1 o, 2 o, or 3 o. 8A.2 Circle the most stable cation in each
PRACTICE PROBLEMS UNIT 8 8A. Identify halides and carbocations as being 1 o, 2 o, or 3 o 8A.1 assify the following halides and carbocations as 1 o, 2 o, or 3 o. 8A.2 Circle the most stable cation in each
Alkyl Halides. Alkyl halides are a class of compounds where a halogen atom or atoms are bound to an sp 3 orbital of an alkyl group.
 Alkyl Halides Alkyl halides are a class of compounds where a halogen atom or atoms are bound to an sp 3 orbital of an alkyl group. CHCl 3 (Chloroform: organic solvent) CF 2 Cl 2 (Freon-12: refrigerant
Alkyl Halides Alkyl halides are a class of compounds where a halogen atom or atoms are bound to an sp 3 orbital of an alkyl group. CHCl 3 (Chloroform: organic solvent) CF 2 Cl 2 (Freon-12: refrigerant
Preparation of Alkyl Halides, R-X. Reaction of alkanes with Cl 2 & Br 2 (F 2 is too reactive, I 2 is unreactive): R + X X 2.
 Preparation of Alkyl alides, R-X Reaction of alkanes with Cl 2 & Br 2 (F 2 is too reactive, I 2 is unreactive): UV R + X 2 R X or heat + X This mechanism involves a free radical chain reaction. A chain
Preparation of Alkyl alides, R-X Reaction of alkanes with Cl 2 & Br 2 (F 2 is too reactive, I 2 is unreactive): UV R + X 2 R X or heat + X This mechanism involves a free radical chain reaction. A chain
REACTIONS OF HALOALKANES - SUBSTITUTION AND ELIMINATION
 REACTIONS OF HALOALKANES - SUBSTITUTION AND ELIMINATION Haloalkanes (also known as halogenoalkanes and alkyl halides) are organic compounds where one of the hydrogens of an alkane or cycloalkane has been
REACTIONS OF HALOALKANES - SUBSTITUTION AND ELIMINATION Haloalkanes (also known as halogenoalkanes and alkyl halides) are organic compounds where one of the hydrogens of an alkane or cycloalkane has been
Chapter 8. Substitution reactions of Alkyl Halides
 Chapter 8. Substitution reactions of Alkyl Halides There are two types of possible reaction in organic compounds in which sp 3 carbon is bonded to an electronegative atom or group (ex, halides) 1. Substitution
Chapter 8. Substitution reactions of Alkyl Halides There are two types of possible reaction in organic compounds in which sp 3 carbon is bonded to an electronegative atom or group (ex, halides) 1. Substitution
7. Haloalkanes (text )
 2009, Department of hemistry, The University of Western Ontario 7.1 7. aloalkanes (text 7.1 7.10) A. Structure and Nomenclature Like hydrogen, the halogens have a valence of one. Thus, a halogen atom can
2009, Department of hemistry, The University of Western Ontario 7.1 7. aloalkanes (text 7.1 7.10) A. Structure and Nomenclature Like hydrogen, the halogens have a valence of one. Thus, a halogen atom can
Writing a Correct Mechanism
 Chapter 2 1) Balancing Equations Writing a Correct Mechanism 2) Using Arrows to show Electron Movement 3) Mechanisms in Acidic and Basic Media 4) Electron rich Species: Nucleophile or Base? 5) Trimolecular
Chapter 2 1) Balancing Equations Writing a Correct Mechanism 2) Using Arrows to show Electron Movement 3) Mechanisms in Acidic and Basic Media 4) Electron rich Species: Nucleophile or Base? 5) Trimolecular
Real life example 1 Let s look at this series of chloroalcohols, and how fast the chloride gets displaced by an external nucleophile.
 Class 2 Carbocations Last time we talked about neighboring group participation in substitution reactions. I want to start today by talking about a few more real life examples. Real life example 1 Let s
Class 2 Carbocations Last time we talked about neighboring group participation in substitution reactions. I want to start today by talking about a few more real life examples. Real life example 1 Let s
Walden discovered a series of reactions that could interconvert (-)-malic acid and (+)-malic acid.
 Chapter 11: Reactions of alkyl halides: nucleophilic substitutions and eliminations Alkyl halides are polarized in the C-X bond, making carbon δ+ (electrophilic). A nucleophilecan attack this carbon, displacing
Chapter 11: Reactions of alkyl halides: nucleophilic substitutions and eliminations Alkyl halides are polarized in the C-X bond, making carbon δ+ (electrophilic). A nucleophilecan attack this carbon, displacing
Organic Chemistry HL IB CHEMISTRY HL
 Organic Chemistry HL IB CHEMISTRY HL Understandings: Nucleophilic Substitution Reactions: SN1 represents a nucleophilic unimolecular substitution reaction and SN2 represents a nucleophilic bimolecular
Organic Chemistry HL IB CHEMISTRY HL Understandings: Nucleophilic Substitution Reactions: SN1 represents a nucleophilic unimolecular substitution reaction and SN2 represents a nucleophilic bimolecular
12AL Experiment 11 (3 days): Nucleophilic Substitution Reactions
 12AL Experiment 11 (3 days): Nucleophilic Substitution Reactions Instructor note: Day 1 (half of the class); Day 2 (other half); Day 3 (everyone to finish up any separation & purification steps etc). Initial
12AL Experiment 11 (3 days): Nucleophilic Substitution Reactions Instructor note: Day 1 (half of the class); Day 2 (other half); Day 3 (everyone to finish up any separation & purification steps etc). Initial
Double and Triple Bonds. The addition of an electrophile and a
 Chapter 11 Additions to Carbon-Carbon Double and Triple Bonds The addition of an electrophile and a nucleophile to a C-C C double or triple bonds 11.1 The General Mechanism Pi electrons of the double bond
Chapter 11 Additions to Carbon-Carbon Double and Triple Bonds The addition of an electrophile and a nucleophile to a C-C C double or triple bonds 11.1 The General Mechanism Pi electrons of the double bond
Nucleophilic Substitution and Elimination
 Nucleophilic Substitution and Elimination Alkyl halides react with a nucleophile in one of two ways. Either they eliminate an X to form an alkene, or they undergo a substitution with the nucleophile, Nu,
Nucleophilic Substitution and Elimination Alkyl halides react with a nucleophile in one of two ways. Either they eliminate an X to form an alkene, or they undergo a substitution with the nucleophile, Nu,
Lab 11 Guide: Nucleophilic Substitution (Nov 10 16)
 Lab 11 Guide: Nucleophilic Substitution (Nov 10 16) Nucleophilic Substitution of Alkyl Halides, Exp. 17A, B, and C, pages 187-192 in Taber This week you will be doing examining real life S N 1 and S N
Lab 11 Guide: Nucleophilic Substitution (Nov 10 16) Nucleophilic Substitution of Alkyl Halides, Exp. 17A, B, and C, pages 187-192 in Taber This week you will be doing examining real life S N 1 and S N
Chapter 8: Nucleophilic Substitution 8.1: Functional Group Transformation By Nucleophilic Substitution
 hapter 8: Nucleophilic Substitution 8.1: Functional Group Transformation By Nucleophilic Substitution Nu: = l,, I Nu - Nucleophiles are Lewis bases (electron-pair donor) Nucleophiles are often negatively
hapter 8: Nucleophilic Substitution 8.1: Functional Group Transformation By Nucleophilic Substitution Nu: = l,, I Nu - Nucleophiles are Lewis bases (electron-pair donor) Nucleophiles are often negatively
PAPER No. 05: TITLE: ORGANIC CHEMISTRY-II MODULE No. 12: TITLE: S N 1 Reactions
 Subject hemistry Paper o and Title Module o and Title Module Tag 05, ORGAI EMISTRY-II 12, S 1 Reactions E_P5_M12 EMISTRY PAPER o. 05: TITLE: ORGAI EMISTRY-II TABLE OF OTETS 1. Learning Outcomes 2. Introduction
Subject hemistry Paper o and Title Module o and Title Module Tag 05, ORGAI EMISTRY-II 12, S 1 Reactions E_P5_M12 EMISTRY PAPER o. 05: TITLE: ORGAI EMISTRY-II TABLE OF OTETS 1. Learning Outcomes 2. Introduction
+ + CH 11: Substitution and Elimination Substitution reactions
 C 11: Substitution and Elimination Substitution reactions Things to sort out: Nucleophile Electrophile -- > substrate Leaving Group S N 2 S N 1 E 1 E 2 Analysis Scheme Kinetics Reaction profile Substrates
C 11: Substitution and Elimination Substitution reactions Things to sort out: Nucleophile Electrophile -- > substrate Leaving Group S N 2 S N 1 E 1 E 2 Analysis Scheme Kinetics Reaction profile Substrates
10. Organohalides. Based on McMurry s Organic Chemistry, 7 th edition
 10. Organohalides Based on McMurry s Organic Chemistry, 7 th edition What Is an Alkyl Halide An organic compound containing at least one carbonhalogen bond (C-X) X (F, Cl, Br, I) replaces H Can contain
10. Organohalides Based on McMurry s Organic Chemistry, 7 th edition What Is an Alkyl Halide An organic compound containing at least one carbonhalogen bond (C-X) X (F, Cl, Br, I) replaces H Can contain
Chapter 3 An Introduction to Organic Reactions: Acids and Bases
 There are 4 types of Organic Reactions Chapter 3 An Introduction to Organic Reactions: SUBSTITUTION: ADDITION: X Y + A X A + Y Example Example A B + X Y A B X Y ELIMINATION There are 4 Types of Organic
There are 4 types of Organic Reactions Chapter 3 An Introduction to Organic Reactions: SUBSTITUTION: ADDITION: X Y + A X A + Y Example Example A B + X Y A B X Y ELIMINATION There are 4 Types of Organic
7: Reactions of Haloalkanes, Alcohols, and Amines. Nucleophilic Substitution
 7: Reactions of Haloalkanes, Alcohols, and Amines. Nucleophilic Substitution Preview 7-4 7.1 Nucleophilic Substitution Reactions of Haloalkanes 7-5 Nucleophilic Substitution Mechanisms (7.1A) 7-5 The SN1
7: Reactions of Haloalkanes, Alcohols, and Amines. Nucleophilic Substitution Preview 7-4 7.1 Nucleophilic Substitution Reactions of Haloalkanes 7-5 Nucleophilic Substitution Mechanisms (7.1A) 7-5 The SN1
3-chloro-1-propene 1-chloropropane 2-chloropropene
 ANSWERS #1. (from 50 minute exam #3, Fall 2000) 5. (6 points) For each group of 3 compounds, identify the compound that expresses the indicated property the MOST and the compound that expresses it the
ANSWERS #1. (from 50 minute exam #3, Fall 2000) 5. (6 points) For each group of 3 compounds, identify the compound that expresses the indicated property the MOST and the compound that expresses it the
Chapter 2 Acids and Bases. Arrhenius Acid and Base Theory. Brønsted-Lowry Acid and Base Theory
 hapter 2 Acids and Bases A significant amount of chemistry can be described using different theories of acids and bases. We ll consider three different acid-base theories (listed in order of increasing
hapter 2 Acids and Bases A significant amount of chemistry can be described using different theories of acids and bases. We ll consider three different acid-base theories (listed in order of increasing
Chapter 11: Nucleophilic Substitution and Elimination Walden Inversion
 hapter 11: Nucleophilic Substitution and Elimination Walden Inversion (S)-(-) Malic acid [a] D = -2.3 Ag 2, 2 Pl 5 l Ag 2, 2 ()-2-hlorosuccinic acid l (-)-2-hlorosuccinic acid Pl 5 ()-() Malic acid [a]
hapter 11: Nucleophilic Substitution and Elimination Walden Inversion (S)-(-) Malic acid [a] D = -2.3 Ag 2, 2 Pl 5 l Ag 2, 2 ()-2-hlorosuccinic acid l (-)-2-hlorosuccinic acid Pl 5 ()-() Malic acid [a]
Examples of Substituted Benzenes
 Organic Chemistry 5 th Edition Paula Yurkanis Bruice Examples of Substituted Benzenes Chapter 15 Reactions of Substituted Benzenes Irene Lee Case Western Reserve University Cleveland, OH 2007, Prentice
Organic Chemistry 5 th Edition Paula Yurkanis Bruice Examples of Substituted Benzenes Chapter 15 Reactions of Substituted Benzenes Irene Lee Case Western Reserve University Cleveland, OH 2007, Prentice
Acids and Bases. Acids and Bases
 BrØnsted-Lowry A BrØnsted-Lowry acid is a proton donor. A BrØnsted-Lowry base is a proton acceptor. H + = proton BrØnsted-Lowry Some molecules contain both hydrogen atoms and lone pairs and thus, can act
BrØnsted-Lowry A BrØnsted-Lowry acid is a proton donor. A BrØnsted-Lowry base is a proton acceptor. H + = proton BrØnsted-Lowry Some molecules contain both hydrogen atoms and lone pairs and thus, can act
CHE1502. Tutorial letter 201/1/2016. General Chemistry 1B. Semester 1. Department of Chemistry CHE1502/201/1/2016
 CE1502/201/1/2016 Tutorial letter 201/1/2016 General Chemistry 1B CE1502 Semester 1 Department of Chemistry This tutorial letter contains the answers to the questions in assignment 1. FIRST SEMESTER: KEY
CE1502/201/1/2016 Tutorial letter 201/1/2016 General Chemistry 1B CE1502 Semester 1 Department of Chemistry This tutorial letter contains the answers to the questions in assignment 1. FIRST SEMESTER: KEY
LECTURE #14 Thurs., Oct.20, Midterm exam: Tues.Oct.25 during class Ch.1, , 7.10, 2, Sections
 CHEM 221 section 01 LECTURE #14 Thurs., Oct.20, 2005 Midterm exam: Tues.Oct.25 during class Ch.1, 7.2-7.5, 7.10, 2, 3.1-3.5 ASSIGNED READINGS: TODAY S CLASS: NEXT LECTURE: Sections 4.7-4.10 finish Ch.4,
CHEM 221 section 01 LECTURE #14 Thurs., Oct.20, 2005 Midterm exam: Tues.Oct.25 during class Ch.1, 7.2-7.5, 7.10, 2, 3.1-3.5 ASSIGNED READINGS: TODAY S CLASS: NEXT LECTURE: Sections 4.7-4.10 finish Ch.4,
The Study of Chemical Reactions. Mechanism: The complete, step by step description of exactly which bonds are broken, formed, and in which order.
 The Study of Chemical Reactions Mechanism: The complete, step by step description of exactly which bonds are broken, formed, and in which order. Thermodynamics: The study of the energy changes that accompany
The Study of Chemical Reactions Mechanism: The complete, step by step description of exactly which bonds are broken, formed, and in which order. Thermodynamics: The study of the energy changes that accompany
Learning Guide for Chapter 7 - Organic Reactions I
 Learning Guide for Chapter 7 - rganic Reactions I I. Introduction to Reactions II. Principles of Kinetics III. Principles of Thermodynamics IV. cleophiles and Electrophiles V. Acids and Bases What a chemical
Learning Guide for Chapter 7 - rganic Reactions I I. Introduction to Reactions II. Principles of Kinetics III. Principles of Thermodynamics IV. cleophiles and Electrophiles V. Acids and Bases What a chemical
CHEM Exam 2 October 25, 2007
 CEM 3311-200 Exam 2 October 25, 2007 By printing my name below, I pledge that "On my honor, as a University of Colorado at Boulder student, I have neither given nor received unauthorized assistance on
CEM 3311-200 Exam 2 October 25, 2007 By printing my name below, I pledge that "On my honor, as a University of Colorado at Boulder student, I have neither given nor received unauthorized assistance on
Organic Chemistry Review: Topic 10 & Topic 20
 Organic Structure Alkanes C C σ bond Mechanism Substitution (Incoming atom or group will displace an existing atom or group in a molecule) Examples Occurs with exposure to ultraviolet light or sunlight,
Organic Structure Alkanes C C σ bond Mechanism Substitution (Incoming atom or group will displace an existing atom or group in a molecule) Examples Occurs with exposure to ultraviolet light or sunlight,
Química Orgânica I. Organic Reactions
 Química Orgânica I 2008/09 w3.ualg.pt\~abrigas QOI 0809 A6 1 Organic Reactions Addition two molecules combine Elimination one molecule splits Substitution parts from two molecules exchange Rearrangement
Química Orgânica I 2008/09 w3.ualg.pt\~abrigas QOI 0809 A6 1 Organic Reactions Addition two molecules combine Elimination one molecule splits Substitution parts from two molecules exchange Rearrangement
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions
 Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
Organic Chemistry. Second Edition. Chapter 19 Aromatic Substitution Reactions. David Klein. Klein, Organic Chemistry 2e
 Organic Chemistry Second Edition David Klein Chapter 19 Aromatic Substitution Reactions Copyright 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2e 19.1 Introduction to Electrophilic
Organic Chemistry Second Edition David Klein Chapter 19 Aromatic Substitution Reactions Copyright 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2e 19.1 Introduction to Electrophilic
PAPER No. 5: REACTION MECHANISM MODULE No. 2: Types of Organic Reaction Mechanisms
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper No. 5:Organic Chemistry-II Module No. 2: Overview of different types of Organic Reaction Mechanisms CHE_P5_M2 TABLE OF CONTENTS
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper No. 5:Organic Chemistry-II Module No. 2: Overview of different types of Organic Reaction Mechanisms CHE_P5_M2 TABLE OF CONTENTS
ORGANIC - CLUTCH CH. 3 - ACIDS AND BASES.
 !! www.clutchprep.com CONCEPT: OVERVIEW OF CHEMICAL REACTIONS There are 4 types of common chemical reactions that we need to be familiar with in organic chemistry 1. Acid-Base Reactions: Two molecules
!! www.clutchprep.com CONCEPT: OVERVIEW OF CHEMICAL REACTIONS There are 4 types of common chemical reactions that we need to be familiar with in organic chemistry 1. Acid-Base Reactions: Two molecules
Organic Mechanisms 1
 Organic Mechanisms 1 Concepts The key ideas required to understand this section are: Concept Book page Chemical properties of alkanes 314 Chemical properties of alkenes 318 Bonding in alkenes 320 Bonding
Organic Mechanisms 1 Concepts The key ideas required to understand this section are: Concept Book page Chemical properties of alkanes 314 Chemical properties of alkenes 318 Bonding in alkenes 320 Bonding
Substitution Reactions
 Substitution Reactions Substitution reactions are reactions in which a nucleophile displaces an atom or group of atoms (the leaving group) from a tetrahedral carbon atom. onsider the following general
Substitution Reactions Substitution reactions are reactions in which a nucleophile displaces an atom or group of atoms (the leaving group) from a tetrahedral carbon atom. onsider the following general
1.13 Acid-Base Reactions: Lone-Pair Donors & Acceptors
 1.13 Acid-Base Reactions: Lone-Pair Donors & Acceptors I, Cl, N 3, 3 P 4 pka 10 to 5 Super strong acids 3 + pka 1.7 RC 2 pka ~ 5 acids Ph pka ~ 10 get 2, R pka ~ 16 weaker RCC (alkynes) pka ~ 26 RN 2 pka
1.13 Acid-Base Reactions: Lone-Pair Donors & Acceptors I, Cl, N 3, 3 P 4 pka 10 to 5 Super strong acids 3 + pka 1.7 RC 2 pka ~ 5 acids Ph pka ~ 10 get 2, R pka ~ 16 weaker RCC (alkynes) pka ~ 26 RN 2 pka
