Student s Learning Activities (Marie Sklodowska-Curie: Discoverer of two radioactive elements)
|
|
|
- Lawrence McDowell
- 5 years ago
- Views:
Transcription
1 Student s Learning Activities (Marie Sklodowska-Curie: Discoverer of two radioactive elements) Activity 1 You will watch a video with narration or listen to a story from your teacher about Maria Curie and her contribution to science. Please write the most important points of the story according to your view and discuss them in your group. Indicative important points: (the political and social situation prevailing at that time, the study the Becquerel rays, the discovery of two new chemical elements by Maria Curie with her husband, the Nobel Prize was given to Maria Curie for two times, the procedures of separation and processing which Maria Curie and her husband succeeded to isolate radium, the effects of radiation on his health,.). Activity 2 In the link: is presented more information about the life of Maria Curie. You can find information about the life, the contribution and the recognition of the work of Maria Curie. 1. Please compare the socio-economic environment in which she grew up and took the decisions about her future with those that prevail in your country now. What are the biggest differences? radioactive elements) 1
2 2. Based on information on the website above please create a timeline for the life and work of Maria Curie in order to be uploaded on the internet and posted in your classroom. Activity 3 Maria, Pierre and their collaborators, managed to isolate one decigramme of radium in the state of its permanent salt and to determine its atomic mass. Please research in the internet and write a text (300 words) in order to describe the procedures of separation and processing which they succeeded to isolate radium. Activity 4 Maria and Pierre Curie studied the "rays of Becquerel». Please look for online information about the discovery of Becquerel. (Indicatively at or ) What they added or further developed or researched the on the discovery of Becquerel? In order to organize your study you can use the table below The discovery of Becquerel and its meaning The discoveries of the Curies and their meaning The differences between the two discoveries 2 radioactive elements)
3 Activ ity 5 In the address you can find information about the radioactive elements, their properties and their uses. Please discuss in your group and write a text (300 words) about the radium... Activity 6 The study of the rays was the context in which there were realized important scientific achievements such as the cathode rays, the X-rays, the rays of Becquerel, the a,b,c, rays or the alpha, βeta, and γ rays. Please all groups made an agreement, so that each group to study one or two concepts and present the information to the whole class. Using the above concepts as key words into your search engine: a) identify the historical context, the date in which was officially announced their discovery, b) the scientist or scientists who made the discovery, c) the country and the institution they worked in. Working collaboratively in your group please place the date of the above discoveries in the axis of time. In modern science what is the accepted name and the nature of each of these rays? Activity 7 The concept of chemical element is initially displayed in the works of Aristotle (4th century BC), it was redefined by Dalton (1661) and Mendeleev (1871) with his work on the periodic table of elements. According to them the element is elementary and not further degradable in any simpler. Was the proposal of Maria Curie for the radioactive elements in agreement with the above position for the element or not? Who contemporary scientist to Maria Curie, according to the narrative, intervened in the debate on the meaning of the chemical element? radioactive elements) 3
4 Please discuss in your group to: a) determine the issue about the meaning of the chemical element, raised again b) search in textbooks or in the internet for the modern meaning of the chemical element and the location of radioactive elements in the modern periodic table (eg: Please write a text (300 words) in which the important points of the History of Science are included for the conceptual evolution of the term: chemical element from the ancient years until today. Activity 8 Using the list of ideas that describe the characteristics of science and the ways it develops, try to locate and write these ideas in the story you heard and the activities of this lesson. These ideas that scientists call Nature of Science (Nature Of Science-NOS- ) are: Characteristics of Nature of Science (NOS) 1. Science demands and relies on empirical evidence. 2. Knowledge production in science includes many common features and shared habits of mind. 3. Scientific knowledge is tentative but durable. 4. Laws and theories are related but distinct kinds of scientific knowledge. 5. Science is a highly creative endeavor. 6. Science has a subjective element. 7. There are historical, cultural, and social influences on science. 8. Science and technology impact each other, but they are not the same. 9. Science and its methods cannot answer all questions. Scientists argue that in order to learn science one must first understand what exactly science is. Because it is difficult to define science, scientists give a list of its characteristics. 4 radioactive elements)
5 .. Activity 9 Please research in the internet for information about the contribution of Maria Curie in Chemistry. Using the information you have collected please make a presentation in your class about Maria Curie and her work, or present it to a conference about the human rights or women scientists. Activity 10 In address is presented through comics the relation of Maria s Curie theories with those of Aristotle s, Lavoisier s, etc. For every comic you will find, please write a text on the differences between Maria s Curie theories and those of the respective scientists. radioactive elements) were written by Ioannis Vlachos and Aikaterini Rizaki with the support by the European Commission (Project LLP GR-COMENIUS- CMP) and the NKUA of Greece. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained there in. radioactive elements) 5
Student s Learning Activities (Mendeleev and the Periodic System)
 Student s Learning Activities (Mendeleev and the Periodic System) Activity 1 You will watch a video with narration or listen to a story from your teacher for the Mendeleev and his work about the classification
Student s Learning Activities (Mendeleev and the Periodic System) Activity 1 You will watch a video with narration or listen to a story from your teacher for the Mendeleev and his work about the classification
Student s Learning Activities (Democritus and the atoms)
 Student s Learning Activities (Democritus and the atoms) Activity 1 You will watch a video with narration or listen to a story from your teacher about the ancient philosophers Democritus and Plato. Please
Student s Learning Activities (Democritus and the atoms) Activity 1 You will watch a video with narration or listen to a story from your teacher about the ancient philosophers Democritus and Plato. Please
Madame Curie By Eve Curie
 Madame Curie By Eve Curie If you are searched for a book Madame Curie by Eve Curie in pdf format, then you have come on to correct site. We present full variant of this ebook in doc, txt, epub, PDF, DjVu
Madame Curie By Eve Curie If you are searched for a book Madame Curie by Eve Curie in pdf format, then you have come on to correct site. We present full variant of this ebook in doc, txt, epub, PDF, DjVu
In 1808 John Dalton proposed that:
 In 1808 John Dalton proposed that: all matter is made up of atoms which cannot be subdivided atoms of the same element are identical atoms of different elements have different masses atoms combine together
In 1808 John Dalton proposed that: all matter is made up of atoms which cannot be subdivided atoms of the same element are identical atoms of different elements have different masses atoms combine together
25.1. Nuclear Radiation
 Nuclear Radiation Marie Curie was a Polish scientist whose research led to many discoveries about radiation and radioactive elements. In 1934 she died from leukemia caused by her long-term exposure to
Nuclear Radiation Marie Curie was a Polish scientist whose research led to many discoveries about radiation and radioactive elements. In 1934 she died from leukemia caused by her long-term exposure to
The atomic theory explains
 The atomic theory explains radioactivity. Radioactive the photograph elements shown release here, energy radioactive as a result iron-59 of changes is used to in make their nuclei. an image In of the circulatory
The atomic theory explains radioactivity. Radioactive the photograph elements shown release here, energy radioactive as a result iron-59 of changes is used to in make their nuclei. an image In of the circulatory
Marie Curie: Radium, Polonium
 1 Chapter 5 Radium and Polonium Photographer unkown; copyright expired This photo of Marie and Pierre Curie was taken as they worked in their laboratory in 1904. Marie won two Nobel Prizes. The first she
1 Chapter 5 Radium and Polonium Photographer unkown; copyright expired This photo of Marie and Pierre Curie was taken as they worked in their laboratory in 1904. Marie won two Nobel Prizes. The first she
Marie and Pierre Curie. In 1903, the Nobel Prize was awarded to Marie and Pierre Curie for their work
 Molly Hellier Period 1 January 24,2011 Extra Credit Paper Marie and Pierre Curie In 1903, the Nobel Prize was awarded to Marie and Pierre Curie for their work with radiation. The couple made great strides
Molly Hellier Period 1 January 24,2011 Extra Credit Paper Marie and Pierre Curie In 1903, the Nobel Prize was awarded to Marie and Pierre Curie for their work with radiation. The couple made great strides
Introduction to Marie Curie. Paul Thompson, Vice-Chair Radiochemistry Group, Royal Society of Chemistry
 Introduction to Marie Curie Paul Thompson, Vice-Chair Radiochemistry Group, Royal Society of Chemistry Purpose of Meeting Welcome to this meeting on behalf of the Radiochemistry Group RSC. The meeting
Introduction to Marie Curie Paul Thompson, Vice-Chair Radiochemistry Group, Royal Society of Chemistry Purpose of Meeting Welcome to this meeting on behalf of the Radiochemistry Group RSC. The meeting
History of the Atom. Scientists and Their Contribution to the Model of an Atom
 History of the Atom Scientists and Their Contribution to the Model of an Atom 1700s 1800s 1900s History of the Atom Timeline 1766 1844 Antoine Lavoisier makes J.J. a substantial Thomson number discovers
History of the Atom Scientists and Their Contribution to the Model of an Atom 1700s 1800s 1900s History of the Atom Timeline 1766 1844 Antoine Lavoisier makes J.J. a substantial Thomson number discovers
Section 25 1 Nuclear Radiation Pages
 We have made it easy for you to find a PDF Ebooks without any digging. And by having access to our ebooks online or by storing it on your computer, you have convenient answers with section 25 1 nuclear
We have made it easy for you to find a PDF Ebooks without any digging. And by having access to our ebooks online or by storing it on your computer, you have convenient answers with section 25 1 nuclear
Section 25 1 Nuclear Radiation Answers
 We have made it easy for you to find a PDF Ebooks without any digging. And by having access to our ebooks online or by storing it on your computer, you have convenient answers with section 25 1 nuclear
We have made it easy for you to find a PDF Ebooks without any digging. And by having access to our ebooks online or by storing it on your computer, you have convenient answers with section 25 1 nuclear
The life and work of Marie Curie
 Reading Practice The life and work of Marie Curie Marie Curie is probably the most famous woman scientist who has ever lived. Born Maria Sklodowska in Poland in 1867, she is famous for her work on radioactivity,
Reading Practice The life and work of Marie Curie Marie Curie is probably the most famous woman scientist who has ever lived. Born Maria Sklodowska in Poland in 1867, she is famous for her work on radioactivity,
Radioactivity and Nuclear Reactions
 chapter 20 Radioactivity and Nuclear Reactions section 1 The Nucleus What You ll Learn what particles make up an atom and its nucleus how the nucleus is held together what radioactivity is Before You Read
chapter 20 Radioactivity and Nuclear Reactions section 1 The Nucleus What You ll Learn what particles make up an atom and its nucleus how the nucleus is held together what radioactivity is Before You Read
The life and work of Marie Curie
 The life and work of Marie Curie Marie Curie is probably the most famous woman scientist who has ever lived. Born Maria Sklodowska in Poland in 1867, she is famous for her work on radioactivity, and was
The life and work of Marie Curie Marie Curie is probably the most famous woman scientist who has ever lived. Born Maria Sklodowska in Poland in 1867, she is famous for her work on radioactivity, and was
Democritus of Abdera. John Dalton. Dalton s Atom. Dalton s Atomic Theory Ancient Greece - 4th century BC. Eaglesfield, England
 Democritus of Abdera Ancient Greece - 4th century BC first suggested the existence of tiny fundamental particles that make up matter. atoms = indestructible did not agree with the current sci theory -
Democritus of Abdera Ancient Greece - 4th century BC first suggested the existence of tiny fundamental particles that make up matter. atoms = indestructible did not agree with the current sci theory -
Beyond the Book. FOCUS Book Curious Marie Curie
 Refer to a periodic table of the elements to find radium (Ra). Use the table s key or other research to familiarize yourself with how to interpret the numbers in radium s box. FOCUS Book Curious Marie
Refer to a periodic table of the elements to find radium (Ra). Use the table s key or other research to familiarize yourself with how to interpret the numbers in radium s box. FOCUS Book Curious Marie
Atomic Theory Timeline
 Atomic Theory Timeline Democritus 450 B.C. Democritus was a Greek philosopher who came to the conclusion that everything was made up of tiny particles. He used the term atomos. Unfortunately, since Democritus
Atomic Theory Timeline Democritus 450 B.C. Democritus was a Greek philosopher who came to the conclusion that everything was made up of tiny particles. He used the term atomos. Unfortunately, since Democritus
Glencoe: Chapter 4. The Structure of the Atom
 Glencoe: Chapter 4 The Structure of the Atom Section One: Early Ideas about Matter Atomists and Democritus : 400 B.C. From Thrace in Greece. Atoms- Uncut-Table Indivisible parts which cannot be broken
Glencoe: Chapter 4 The Structure of the Atom Section One: Early Ideas about Matter Atomists and Democritus : 400 B.C. From Thrace in Greece. Atoms- Uncut-Table Indivisible parts which cannot be broken
A Correlation of. Eastern Hemisphere. Ohio s Learning Standards Social Studies: K-12 Grade 6
 A Correlation of Eastern Hemisphere To Grade 6 to,, Grade 6 Introduction This document demonstrates how, Eastern Hemisphere, Ohio Edition, meets : K-12 Social Studies for Grade 6. Correlation page references
A Correlation of Eastern Hemisphere To Grade 6 to,, Grade 6 Introduction This document demonstrates how, Eastern Hemisphere, Ohio Edition, meets : K-12 Social Studies for Grade 6. Correlation page references
Name: Row Period Due Date: 11/21/16
 Name: Row Period Due Date: 11/21/16 The History of the Atom Project I. A Cooperative Learning Activity In this project, each member of the team is responsible for a specific part of the activity. As experts
Name: Row Period Due Date: 11/21/16 The History of the Atom Project I. A Cooperative Learning Activity In this project, each member of the team is responsible for a specific part of the activity. As experts
HISTORY OF CHEMISTRY BY SARA C. MIGUEL M. JESSICA S.
 HISTORY OF CHEMISTRY BY SARA C. MIGUEL M. JESSICA S. A Little Info! Chemistry is a branch of science Dates back to prehistoric times ( looong time ago ) Is separated into four different time periods Prehistoric
HISTORY OF CHEMISTRY BY SARA C. MIGUEL M. JESSICA S. A Little Info! Chemistry is a branch of science Dates back to prehistoric times ( looong time ago ) Is separated into four different time periods Prehistoric
FAMOUS SCIENTISTS: LC CHEMISTRY
 FAMOUS SCIENTISTS: LC CHEMISTRY Study online at quizlet.com/_6j280 1. SVANTE AUGUST ARRHENIUS 4. ANTOINE HENRI BECQUEREL He developed a theory of acids and bases on how they form ions in solution. He also
FAMOUS SCIENTISTS: LC CHEMISTRY Study online at quizlet.com/_6j280 1. SVANTE AUGUST ARRHENIUS 4. ANTOINE HENRI BECQUEREL He developed a theory of acids and bases on how they form ions in solution. He also
Nuclear Physics. Physical Phase Change Material still has the same chemical properties. Types of Changes
 Nuclear Physics Project 2013 Nuclear Physics Chapters 39 and 40 Phase One Prepare a Position Poster In groups of 2 or 3, choose a topic from Chapter 39/40 that has economic, political, environmental or
Nuclear Physics Project 2013 Nuclear Physics Chapters 39 and 40 Phase One Prepare a Position Poster In groups of 2 or 3, choose a topic from Chapter 39/40 that has economic, political, environmental or
The origins of atomic theory
 Models of the atom It is important to realise that a lot of what we know about the structure of atoms has been developed over a long period of time. This is often how scientific knowledge develops, with
Models of the atom It is important to realise that a lot of what we know about the structure of atoms has been developed over a long period of time. This is often how scientific knowledge develops, with
History of the Atom. Say Thanks to the Authors Click (No sign in required)
 History of the Atom Say Thanks to the Authors Click http://www.ck12.org/saythanks (No sign in required) To access a customizable version of this book, as well as other interactive content, visit www.ck12.org
History of the Atom Say Thanks to the Authors Click http://www.ck12.org/saythanks (No sign in required) To access a customizable version of this book, as well as other interactive content, visit www.ck12.org
Isotopes of an element have the same symbol and same atomic number - Mass number refers to the protons plus neutrons in an isotope
 7.1 Atomic Theory and Radioactive Decay Natural background radiation exists all around us. This radiation consists of high energy particles or waves being emitted from a variety of materials Radioactivity
7.1 Atomic Theory and Radioactive Decay Natural background radiation exists all around us. This radiation consists of high energy particles or waves being emitted from a variety of materials Radioactivity
Lecture 21 Fundamentals of Physics Phys 120, Fall 2015 Nuclear Physics
 Lecture 21 Fundamentals of Physics Phys 120, Fall 2015 Nuclear Physics A. J. Wagner North Dakota State University, Fargo, ND 58102 Fargo, November 13, 2015 Overview Why care about nuclei? How do nuclei
Lecture 21 Fundamentals of Physics Phys 120, Fall 2015 Nuclear Physics A. J. Wagner North Dakota State University, Fargo, ND 58102 Fargo, November 13, 2015 Overview Why care about nuclei? How do nuclei
Inventors and Scientists: Marie Curie
 Inventors and Scientists: Marie Curie By Big History Project, adapted by Newsela staff on 06.15.16 Word Count 1,993 Level 920L Marie Curie in 1898. BELOW: Marie Curie in her laboratory in 1905. Bettmann/CORBIS
Inventors and Scientists: Marie Curie By Big History Project, adapted by Newsela staff on 06.15.16 Word Count 1,993 Level 920L Marie Curie in 1898. BELOW: Marie Curie in her laboratory in 1905. Bettmann/CORBIS
Section 4: Science and Me
 Section 4: Science and Me The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 4.01 Science in Everyday Life Chemistry (3)(B) Chemistry (3)(C)
Section 4: Science and Me The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 4.01 Science in Everyday Life Chemistry (3)(B) Chemistry (3)(C)
3.01 Understanding Atoms
 3.01 Understanding Atoms The Events Leading to the Discovery of the Building Block of Matter Dr. Fred Omega Garces Chemistry 111 Miramar College 1 3.02 Atomic Evolution Environmental Problems in our Lifetime
3.01 Understanding Atoms The Events Leading to the Discovery of the Building Block of Matter Dr. Fred Omega Garces Chemistry 111 Miramar College 1 3.02 Atomic Evolution Environmental Problems in our Lifetime
Name: Date: Period: #: Chapter 1: Outline Notes What Does a Historian Do?
 Name: Date: Period: #: Chapter 1: Outline Notes What Does a Historian Do? Lesson 1.1 What is History? I. Why Study History? A. History is the study of the of the past. History considers both the way things
Name: Date: Period: #: Chapter 1: Outline Notes What Does a Historian Do? Lesson 1.1 What is History? I. Why Study History? A. History is the study of the of the past. History considers both the way things
Background and Concept
 Background and Concept The concept of IYC 2011 started with the recognition by IUPAC that a number of scientific disciplines have achieved significant benefits from securing designation by the United Nations
Background and Concept The concept of IYC 2011 started with the recognition by IUPAC that a number of scientific disciplines have achieved significant benefits from securing designation by the United Nations
5.1. Developing the Atomic Theory. Science, Art, and Atoms
 5.1 Developing the Atomic Theory Here is a summary of what you will learn in this section: An element is a pure substance that cannot be broken down into other substances by chemical reactions. The smallest
5.1 Developing the Atomic Theory Here is a summary of what you will learn in this section: An element is a pure substance that cannot be broken down into other substances by chemical reactions. The smallest
SCIENCE INTERACTIVE NOTEBOOK
 Two atoms walk into a bar 1 SCIENCE INTERACTIVE NOTEBOOK Hey, I think I lost an electron! Are you sure? Yeah, I m positive. Dr. Erica Colón 2013 www.nittygrittyscience.blogspot.com Graphics www.djinkers.com
Two atoms walk into a bar 1 SCIENCE INTERACTIVE NOTEBOOK Hey, I think I lost an electron! Are you sure? Yeah, I m positive. Dr. Erica Colón 2013 www.nittygrittyscience.blogspot.com Graphics www.djinkers.com
What is Radiation? Física da Radiação MEBiom 2016/2017 Patrícia Gonçalves. Supporting Slides and images
 What is Radiation? Física da Radiação MEBiom 2016/2017 Patrícia Gonçalves Supporting Slides and images Radiation? Radiation is energy that comes from a source and travels through some material or through
What is Radiation? Física da Radiação MEBiom 2016/2017 Patrícia Gonçalves Supporting Slides and images Radiation? Radiation is energy that comes from a source and travels through some material or through
Geography. Programme of study for key stage 3 and attainment target (This is an extract from The National Curriculum 2007)
 Geography Programme of study for key stage 3 and attainment target (This is an extract from The National Curriculum 2007) Crown copyright 2007 Qualifications and Curriculum Authority 2007 Curriculum aims
Geography Programme of study for key stage 3 and attainment target (This is an extract from The National Curriculum 2007) Crown copyright 2007 Qualifications and Curriculum Authority 2007 Curriculum aims
The Nucleus Came Next
 The Nucleus Came Next Ernest Rutherford The New Zealand born British chemist and physicist who became known as the father of nuclear physics. He discovered the atomic nucleus, and thereby pioneered the
The Nucleus Came Next Ernest Rutherford The New Zealand born British chemist and physicist who became known as the father of nuclear physics. He discovered the atomic nucleus, and thereby pioneered the
The History of the Atom Project DUE NEXT FRIDAY (10/13) THAT IS WHEN YOU WILL PRESENT YOUR FINDINGS
 Name: Period: Date The History of the Atom Project DUE NEXT FRIDAY (10/13) THAT IS WHEN YOU WILL PRESENT YOUR FINDINGS A Cooperative Learning Activity In this jigsaw activity, each member of the team is
Name: Period: Date The History of the Atom Project DUE NEXT FRIDAY (10/13) THAT IS WHEN YOU WILL PRESENT YOUR FINDINGS A Cooperative Learning Activity In this jigsaw activity, each member of the team is
Atomic Theory. Introducing the Atomic Theory:
 Atomic Theory Chemistry is the science of matter. Matter is made up of things called atoms, elements, and molecules. But have you ever wondered if atoms and molecules are real? Would you be surprised to
Atomic Theory Chemistry is the science of matter. Matter is made up of things called atoms, elements, and molecules. But have you ever wondered if atoms and molecules are real? Would you be surprised to
ATOMS AND ELEMENTS. Democritus 400 B.C. Atomic Theory of Matter. Dalton s Postulates (1803) Page 1
 ATOMS AND ELEMENTS Democritus 400 BC Believed that matter was composed of invisible particles of matter he called atoms According to Democritus, atoms could not be broken into smaller particles Atomic
ATOMS AND ELEMENTS Democritus 400 BC Believed that matter was composed of invisible particles of matter he called atoms According to Democritus, atoms could not be broken into smaller particles Atomic
Structure of the Atom
 Structure of the Atom Questions about the examination of, what constitutes matter, come to us from the ancient Greeks. It was the Greeks, who first attempted to explain the nature of matter. The ancient
Structure of the Atom Questions about the examination of, what constitutes matter, come to us from the ancient Greeks. It was the Greeks, who first attempted to explain the nature of matter. The ancient
CHAPTER 1 INTRODUCTION TO RADIOBIOLOGY HDR 112 RADIATION BIOLOGY AND RADIATION PROTECTION MR KAMARUL AMIN BIN ABDULLAH
 HDR 112 RADIATION BIOLOGY AND RADIATION PROTECTION CHAPTER 1 INTRODUCTION TO RADIOBIOLOGY PREPARED BY: MR KAMARUL AMIN BIN ABDULLAH SCHOOL OF MEDICAL IMAGING FACULTY OF HEALTH SCIENCE LEARNING OUTCOMES
HDR 112 RADIATION BIOLOGY AND RADIATION PROTECTION CHAPTER 1 INTRODUCTION TO RADIOBIOLOGY PREPARED BY: MR KAMARUL AMIN BIN ABDULLAH SCHOOL OF MEDICAL IMAGING FACULTY OF HEALTH SCIENCE LEARNING OUTCOMES
University of Puerto Rico-Mayagüez Campus Department of Chemistry
 Syllabus for the 1 st Semester 2004-2005 Professor: Maritza De Jesús Echevarría, M.S. Room: Q-125 Office Hours: Monday, Wednesday, Friday Section: 131 8:30-9:30 am, 2:30-3:30 pm, Office: Q-274A I. General
Syllabus for the 1 st Semester 2004-2005 Professor: Maritza De Jesús Echevarría, M.S. Room: Q-125 Office Hours: Monday, Wednesday, Friday Section: 131 8:30-9:30 am, 2:30-3:30 pm, Office: Q-274A I. General
Understanding the Atom
 CHAPTER 7 Understanding the Atom LESSON 2 Protons, Neutrons, and Electrons How Atoms Differ What do you think? Read the three statements below and decide whether you agree or disagree with them. Place
CHAPTER 7 Understanding the Atom LESSON 2 Protons, Neutrons, and Electrons How Atoms Differ What do you think? Read the three statements below and decide whether you agree or disagree with them. Place
Atoms and Elements. Chapter 2. Chemistry 221 Professor Michael Russell
 Atoms and Elements Chapter 2 Chemistry 221 Professor Michael Russell ATOMS AND ELEMENTS Where Does Matter Come From? FROM THE Hydrogen and Helium important Also Carbon, Oxygen and Neon Element Abundance
Atoms and Elements Chapter 2 Chemistry 221 Professor Michael Russell ATOMS AND ELEMENTS Where Does Matter Come From? FROM THE Hydrogen and Helium important Also Carbon, Oxygen and Neon Element Abundance
TEACHERS GUIDELINES BUCKYBALLS
 NTSE - Nano Technology Science Education Project No: 511787-LLP-1-2010-1-TR-KA3-KA3MP TEACHERS GUIDELINES BUCKYBALLS Contents 1. Foreword for Teachers 2. Introduction 3. Learning Process 4. Conclusion
NTSE - Nano Technology Science Education Project No: 511787-LLP-1-2010-1-TR-KA3-KA3MP TEACHERS GUIDELINES BUCKYBALLS Contents 1. Foreword for Teachers 2. Introduction 3. Learning Process 4. Conclusion
A brief history of Chemistry. Science 9- Mr. Klasz
 A brief history of Chemistry Science 9- Mr. Klasz What is a pure substance? An element is a pure substance. A compound is made up of two or more elements which have been chemically compounded together.
A brief history of Chemistry Science 9- Mr. Klasz What is a pure substance? An element is a pure substance. A compound is made up of two or more elements which have been chemically compounded together.
Nuclear Science A Teacher s Guide to the Nuclear Science Wall Chart 1998 Contemporary Physics Education Project (CPEP)
 Nuclear Science A Teacher s Guide to the Nuclear Science Wall Chart 1998 Contemporary Physics Education Project (CPEP) Chapter 3 Radioactivity In radioactive processes, particles or electromagnetic radiation
Nuclear Science A Teacher s Guide to the Nuclear Science Wall Chart 1998 Contemporary Physics Education Project (CPEP) Chapter 3 Radioactivity In radioactive processes, particles or electromagnetic radiation
Lesson Plan Summary Magic Tree House #28: High Tide in Hawaii Standing on Solid Ground??
 Lesson Plan Summary Magic Tree House #28: High Tide in Hawaii Standing on Solid Ground?? DURING THIS BOOK STUDY, EACH STUDENT WILL: COMMON CORE STANDARDS ADDRESSED: Discover facts about the layers of the
Lesson Plan Summary Magic Tree House #28: High Tide in Hawaii Standing on Solid Ground?? DURING THIS BOOK STUDY, EACH STUDENT WILL: COMMON CORE STANDARDS ADDRESSED: Discover facts about the layers of the
Scientists to Know CHADWICK THOMSON RUTHERFORD DEMOCRITUS BOHR HEISENBERG DALTON
 Unit 1: Atoms Scientists to Know CHADWICK THOMSON RUTHERFORD DEMOCRITUS BOHR HEISENBERG DALTON The History of Discovering the Atom The Timeline of Discovery 1. Philosophical Era 2. Alchemical Era 3. Classical
Unit 1: Atoms Scientists to Know CHADWICK THOMSON RUTHERFORD DEMOCRITUS BOHR HEISENBERG DALTON The History of Discovering the Atom The Timeline of Discovery 1. Philosophical Era 2. Alchemical Era 3. Classical
Learning About Atoms. By SUSAN KNORR. COPYRIGHT 2004 Mark Twain Media, Inc. ISBN Printing No EB
 Learning About Atoms By SUSAN KNORR COPYRIGHT 2004 Mark Twain Media, Inc. ISBN 978-1-58037-894-9 Printing No. 1631-EB Mark Twain Media, Inc., Publishers Distributed by Carson-Dellosa Publishing Company,
Learning About Atoms By SUSAN KNORR COPYRIGHT 2004 Mark Twain Media, Inc. ISBN 978-1-58037-894-9 Printing No. 1631-EB Mark Twain Media, Inc., Publishers Distributed by Carson-Dellosa Publishing Company,
Vocabulary atom atomos Dalton's atomic theory law of constant composition law of definite proportions law of multiple proportions matter.
 1.3 Early Atomic Theory Lesson Objectives The student will: define matter and explain how it is composed of building blocks known as atoms. give a short history of how the concept of the atom developed.
1.3 Early Atomic Theory Lesson Objectives The student will: define matter and explain how it is composed of building blocks known as atoms. give a short history of how the concept of the atom developed.
Our Solar System Unit of Work
 Lesson 1: Introducing our Solar System Introduction In this lesson, students will be introduced to our Solar System. They will explore what it contains and use common items to create a scaled version of
Lesson 1: Introducing our Solar System Introduction In this lesson, students will be introduced to our Solar System. They will explore what it contains and use common items to create a scaled version of
Economic and Social Council 2 July 2015
 ADVANCE UNEDITED VERSION UNITED NATIONS E/C.20/2015/11/Add.1 Economic and Social Council 2 July 2015 Committee of Experts on Global Geospatial Information Management Fifth session New York, 5-7 August
ADVANCE UNEDITED VERSION UNITED NATIONS E/C.20/2015/11/Add.1 Economic and Social Council 2 July 2015 Committee of Experts on Global Geospatial Information Management Fifth session New York, 5-7 August
Physics 3150 WI Assignment #2 Winter 2005 Draft #1. Marie Curie and the 1903 Nobel Prize in Physics. Physics, University of Missouri Columbia
 Physics 3150 WI Assignment #2 Winter 2005 Draft #1 Marie Curie and the 1903 Nobel Prize in Physics Sarah Bird Physics, University of Missouri Columbia (November 26, 2007) Marie and Pierre Curie shared
Physics 3150 WI Assignment #2 Winter 2005 Draft #1 Marie Curie and the 1903 Nobel Prize in Physics Sarah Bird Physics, University of Missouri Columbia (November 26, 2007) Marie and Pierre Curie shared
DOWNLOAD OR READ : WORLD GEOGRAPHY READING AND VOCABULARY STUDY GUIDE 2007 PDF EBOOK EPUB MOBI
 DOWNLOAD OR READ : WORLD GEOGRAPHY READING AND VOCABULARY STUDY GUIDE 2007 PDF EBOOK EPUB MOBI Page 1 Page 2 world geography reading and vocabulary study guide 2007 world geography reading and pdf world
DOWNLOAD OR READ : WORLD GEOGRAPHY READING AND VOCABULARY STUDY GUIDE 2007 PDF EBOOK EPUB MOBI Page 1 Page 2 world geography reading and vocabulary study guide 2007 world geography reading and pdf world
Name: Period: Date: Applications of Nuclear Chemistry Activity
 Name: Period: Date: Applications of Nuclear Chemistry Activity Station 1: Chemical vs. Nuclear Reactions Directions: Pick up the Station 1 Cards and figure out which cards apply to chemical reactions and
Name: Period: Date: Applications of Nuclear Chemistry Activity Station 1: Chemical vs. Nuclear Reactions Directions: Pick up the Station 1 Cards and figure out which cards apply to chemical reactions and
Nuclear.15 now.notebook. February 05, 2016
 I can identify the types of radiation and write balanced nuclear equations. I can write balanced nuclear decay equations. I can explain the difference between fission and fusion. I can identify unknown
I can identify the types of radiation and write balanced nuclear equations. I can write balanced nuclear decay equations. I can explain the difference between fission and fusion. I can identify unknown
H 1. Nuclear Physics. Nuclear Physics. 1. Parts of Atom. A. Nuclear Structure. 2b. Nomenclature. 2. Isotopes. AstroPhysics Notes
 AstroPhysics Notes Nuclear Physics Dr. Bill Pezzaglia Nuclear Physics A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions Updated: 0Feb07 Rough draft A. Nuclear Structure. Parts of Atom. Parts of
AstroPhysics Notes Nuclear Physics Dr. Bill Pezzaglia Nuclear Physics A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions Updated: 0Feb07 Rough draft A. Nuclear Structure. Parts of Atom. Parts of
CHEMISTRY. Matter and Change. Table Of Contents. Section 4.1 Early Ideas About Matter. Unstable Nuclei and Radioactive Decay
 CHEMISTRY 4 Table Of Contents Matter and Change Section 4.1 Early Ideas About Matter Chapter 4: The Structure of the Atom Section 4.2 Section 4.3 Section 4.4 Defining the Atom How Atoms Differ Unstable
CHEMISTRY 4 Table Of Contents Matter and Change Section 4.1 Early Ideas About Matter Chapter 4: The Structure of the Atom Section 4.2 Section 4.3 Section 4.4 Defining the Atom How Atoms Differ Unstable
THE ATOM Pearson Education, Inc.
 THE ATOM Title and Highlight Right Side NOTES ONLY TN Ch 4.1-4.2 Topic: EQ: Date Reflect Question: Reflect on the material by asking a question (its not suppose to be answered from notes) NOTES: Write
THE ATOM Title and Highlight Right Side NOTES ONLY TN Ch 4.1-4.2 Topic: EQ: Date Reflect Question: Reflect on the material by asking a question (its not suppose to be answered from notes) NOTES: Write
Pre-AP Chemistry Dr. Brighid Corcoran, Ph.D. Lake Dallas High School Classroom Policies and Procedures
 Pre-AP Chemistry 2016-2017 Dr. Brighid Corcoran, Ph.D. Lake Dallas High School Classroom Policies and Procedures The following topics were covered with your child during class: Attendance/Make-Up Procedure
Pre-AP Chemistry 2016-2017 Dr. Brighid Corcoran, Ph.D. Lake Dallas High School Classroom Policies and Procedures The following topics were covered with your child during class: Attendance/Make-Up Procedure
H 1. Nuclear Physics. Nuclear Physics. 1. Parts of Atom. 2. Isotopes. AstroPhysics Notes. Dr. Bill Pezzaglia. Rough draft. A.
 AstroPhysics Notes Tom Lehrer: Elements Dr. Bill Pezzaglia Nuclear Physics Updated: 0Feb Rough draft Nuclear Physics A. Nuclear Structure A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions. Parts
AstroPhysics Notes Tom Lehrer: Elements Dr. Bill Pezzaglia Nuclear Physics Updated: 0Feb Rough draft Nuclear Physics A. Nuclear Structure A. Nuclear Structure B. Nuclear Decay C. Nuclear Reactions. Parts
Name: Period: Date: Applications of Nuclear Chemistry Activity
 Name: Period: Date: Applications of Nuclear Chemistry Activity Station 1: Chemical vs. Nuclear Reactions Directions: Pick up the Station 1 Cards and figure out which cards apply to chemical reactions and
Name: Period: Date: Applications of Nuclear Chemistry Activity Station 1: Chemical vs. Nuclear Reactions Directions: Pick up the Station 1 Cards and figure out which cards apply to chemical reactions and
An Introduction to Using Geographic Information Systems (GIS) in the Classroom
 Georgia Southern University Digital Commons@Georgia Southern Interdisciplinary STEM Teaching & Learning Conference Mar 24th, 10:00 AM - 11:30 AM An Introduction to Using Geographic Information Systems
Georgia Southern University Digital Commons@Georgia Southern Interdisciplinary STEM Teaching & Learning Conference Mar 24th, 10:00 AM - 11:30 AM An Introduction to Using Geographic Information Systems
0.G. Notational conventions for elementary geometry
 0.G. Notational conventions for elementary geometry Since concepts from elementary geometry play an important role in any account of the history of mathematics, we shall frequently discuss various geometrical
0.G. Notational conventions for elementary geometry Since concepts from elementary geometry play an important role in any account of the history of mathematics, we shall frequently discuss various geometrical
Grade 6 Social Studies
 Grade 6 Social Studies Social Studies Grade(s) 6th Course Overview This course focuses on the five Wisconsin Model Academic standards for Social Studies: Geography, History, Political Science and Citizenship,
Grade 6 Social Studies Social Studies Grade(s) 6th Course Overview This course focuses on the five Wisconsin Model Academic standards for Social Studies: Geography, History, Political Science and Citizenship,
Fiesta Ware. Nuclear Chemistry. 2009, Prentice-Hall, Inc.
 Fiesta Ware 2009, Prentice-Hall, Inc. Measuring Radioactivity One can use a device like this Geiger counter to measure the amount of activity present in a radioactive sample. The ionizing radiation creates
Fiesta Ware 2009, Prentice-Hall, Inc. Measuring Radioactivity One can use a device like this Geiger counter to measure the amount of activity present in a radioactive sample. The ionizing radiation creates
Women in Science. March 8, 2018 Escuela Oficial de Idiomas de Gijón John K. White
 Women in Science March 8, 2018 Escuela Oficial de Idiomas de Gijón John K. White Ada Lovelace Marie Curie Lise Meitner Maria Mayer Cecilia Payne Rachel Carson Mary Leakey Dorothy Hodgkin Rosalind Franklin
Women in Science March 8, 2018 Escuela Oficial de Idiomas de Gijón John K. White Ada Lovelace Marie Curie Lise Meitner Maria Mayer Cecilia Payne Rachel Carson Mary Leakey Dorothy Hodgkin Rosalind Franklin
Conceptual Physics 2006 (Hewitt) Correlated to: Nevada Science Content Standards (Grades 9-12)
 Nevada Science Content Standards (Grades 9-12) SCIENTIFIC INQUIRY (NATURE OF SCIENCE UNIFYING CONCEPT A) Scientific inquiry is the process by which humans systematically examine the natural world. Scientific
Nevada Science Content Standards (Grades 9-12) SCIENTIFIC INQUIRY (NATURE OF SCIENCE UNIFYING CONCEPT A) Scientific inquiry is the process by which humans systematically examine the natural world. Scientific
Chapter 7 - Radioactivity. Science 10 P
 Chapter 7 - Radioactivity Science 10 P286-328 What is Radiation? Radiation is: anything that radiates away from something. Radiation may be in the form of: particles (neutrons, alpha particles, and beta
Chapter 7 - Radioactivity Science 10 P286-328 What is Radiation? Radiation is: anything that radiates away from something. Radiation may be in the form of: particles (neutrons, alpha particles, and beta
Chapter 4. The structure of the atom. AL-COS Objectives 1, 2,3,4,7, 10, 15, 20, 21, 22, 27and 28
 Chapter 4 The structure of the atom AL-COS Objectives 1, 2,3,4,7, 10, 15, 20, 21, 22, 27and 28 You ll learn to Identify the experiments that led to the development of the nuclear model of atomic structure
Chapter 4 The structure of the atom AL-COS Objectives 1, 2,3,4,7, 10, 15, 20, 21, 22, 27and 28 You ll learn to Identify the experiments that led to the development of the nuclear model of atomic structure
CHEMISTRY Matter and Change. Chapter 4: The Structure of the Atom
 CHEMISTRY Matter and Change Chapter 4: The Structure of the Atom CHAPTER 4 Table Of Contents Section 4.1 Section 4.2 Section 4.3 Section 4.4 Early Ideas About Matter Defining the Atom How Atoms Differ
CHEMISTRY Matter and Change Chapter 4: The Structure of the Atom CHAPTER 4 Table Of Contents Section 4.1 Section 4.2 Section 4.3 Section 4.4 Early Ideas About Matter Defining the Atom How Atoms Differ
CHAPTER -4 STRUCTURE OF ATOM CONCEPT DETAILS
 CHAPTER -4 STRUCTURE OF ATOM CONCEPT DETAILS KEY CONCEPTS : [ *rating as per the significance of concept] 1. Dalton s Atomic theory ** 2. J J Thomson Experiments *** 3. Rutherford s Scattering Experiments
CHAPTER -4 STRUCTURE OF ATOM CONCEPT DETAILS KEY CONCEPTS : [ *rating as per the significance of concept] 1. Dalton s Atomic theory ** 2. J J Thomson Experiments *** 3. Rutherford s Scattering Experiments
Exploring the World s Cultures 90 Minute Social Studies Lesson Gallery Program Grades: 3-7. Exploring the World s Cultures. Description.
 TEACHER GUIDE Exploring the World s Cultures 90 Minute Social Studies Lesson Gallery Program Grades: 3-7 Exploring the World s Cultures Description Travel the world and connect with cultures with which
TEACHER GUIDE Exploring the World s Cultures 90 Minute Social Studies Lesson Gallery Program Grades: 3-7 Exploring the World s Cultures Description Travel the world and connect with cultures with which
Learning Outcomes 2. Key Concepts 2. Misconceptions and Teaching Challenges 3. Vocabulary 4. Lesson and Content Overview 5
 UNIT 3 GUIDE Table of Contents Learning Outcomes 2 Key Concepts 2 Misconceptions and Teaching Challenges 3 Vocabulary 4 Lesson and Content Overview 5 BIG HISTORY PROJECT / UNIT 3 GUIDE 1 Unit 3 Stars and
UNIT 3 GUIDE Table of Contents Learning Outcomes 2 Key Concepts 2 Misconceptions and Teaching Challenges 3 Vocabulary 4 Lesson and Content Overview 5 BIG HISTORY PROJECT / UNIT 3 GUIDE 1 Unit 3 Stars and
Social Studies Curriculum Sixth Grade
 Social Studies Curriculum Sixth Grade A. History Students will examine the key historic movements, events, and figures that contributed to the development of modern Europe and American nations from early
Social Studies Curriculum Sixth Grade A. History Students will examine the key historic movements, events, and figures that contributed to the development of modern Europe and American nations from early
Ancient Theories of Atoms
 Ancient Theories of Atoms The word atom was coined somewhere around 450 B.C. (it is an unfortunate side note about this period of history that it is usually quite difficult to give precise dates; one wishes
Ancient Theories of Atoms The word atom was coined somewhere around 450 B.C. (it is an unfortunate side note about this period of history that it is usually quite difficult to give precise dates; one wishes
Episode 1 - Discovering The Elements.
 Name: Period: Episode 1 - Discovering The Elements. 1. What element did Humphrey Davy discover? 2. What force did he use? 3. What is everything made of (and how many are there)? 4. What is an element?
Name: Period: Episode 1 - Discovering The Elements. 1. What element did Humphrey Davy discover? 2. What force did he use? 3. What is everything made of (and how many are there)? 4. What is an element?
AP Chemistry 1: Summer 2015 Assignment
 AP Chemistry 1: Summer 2015 Assignment TEXTBOOK: Chemistry, 2 nd Edition CK-12 Foundation (Berwick & Parsons); customized as AP Chemistry 1 Summer Reading Assignment The textbook & content outline can
AP Chemistry 1: Summer 2015 Assignment TEXTBOOK: Chemistry, 2 nd Edition CK-12 Foundation (Berwick & Parsons); customized as AP Chemistry 1 Summer Reading Assignment The textbook & content outline can
Course summary for "Albert Einstein and the Nature of Science" Meetings 1-3: Introduction to the Nobel Prize and to the nature of science
 Course summary for "Albert Einstein and the Nature of Science" Meetings 1-3: Introduction to the Nobel Prize and to the nature of science The course opened with a discussion of the questions: what is science
Course summary for "Albert Einstein and the Nature of Science" Meetings 1-3: Introduction to the Nobel Prize and to the nature of science The course opened with a discussion of the questions: what is science
HUMAN GEOGRAPHY AND CONTEMPORARY SOCIETY Global Patterns and Processes Spring 2009
 HUMAN GEOGRAPHY AND CONTEMPORARY SOCIETY Global Patterns and Processes Spring 2009 Professor: Reece Jones Office: 412 Saunders Hall Email: reecej@hawaii.edu Office hours: M, T, W, Th 4:30 5:00 or by appointment
HUMAN GEOGRAPHY AND CONTEMPORARY SOCIETY Global Patterns and Processes Spring 2009 Professor: Reece Jones Office: 412 Saunders Hall Email: reecej@hawaii.edu Office hours: M, T, W, Th 4:30 5:00 or by appointment
Chapter 6A Solving Exponential and Logarithmic Equations. Solve x+5 = x = 9 x x 2 = x 4. 5 x = 18
 Fry Texas A&M University!! Math 150!! Chapter 6!! Fall 2014! 1 Chapter 6A Solving Exponential and Logarithmic Equations Solve 1. 4 3x+5 = 16 2. 3 x = 9 x+5 3. 8 x 2 = 1 4 5 9 x 4. 5 x = 18 Fry Texas A&M
Fry Texas A&M University!! Math 150!! Chapter 6!! Fall 2014! 1 Chapter 6A Solving Exponential and Logarithmic Equations Solve 1. 4 3x+5 = 16 2. 3 x = 9 x+5 3. 8 x 2 = 1 4 5 9 x 4. 5 x = 18 Fry Texas A&M
Nuclear Reactions: Chemistry 5.1 AN INTRODUCTION TO NUCLEAR CHEMISTRY
 Nuclear Reactions: Chemistry 5.1 AN INTRODUCTION TO NUCLEAR CHEMISTRY Discovery of Radioactivity Roentgen In 1895 Wilhelm Konrad Roentgen discovered Xrays. Roentgen observed that a vacuum discharge tube
Nuclear Reactions: Chemistry 5.1 AN INTRODUCTION TO NUCLEAR CHEMISTRY Discovery of Radioactivity Roentgen In 1895 Wilhelm Konrad Roentgen discovered Xrays. Roentgen observed that a vacuum discharge tube
DIATHEMATIKON PROGRAMMA CROSS-THEMATIC CURRICULUM FRAMEWORK
 DIATHEMATIKON PROGRAMMA CROSS-THEMATIC CURRICULUM FRAMEWORK FOR STUDIES OF THE ENVIRONMENT The nature of Studies of the Environment Studies of the Environment is an interdisciplinary subject dealing with
DIATHEMATIKON PROGRAMMA CROSS-THEMATIC CURRICULUM FRAMEWORK FOR STUDIES OF THE ENVIRONMENT The nature of Studies of the Environment Studies of the Environment is an interdisciplinary subject dealing with
Augusta Canal National Heritage Area 8 Grade Lesson Plans. Stage 1 Desired Results
 Title: Falling into Place Stage 1 Desired Results Subject/Course: Grade: 8th Instructional Time: 10 hrs. Big Ideas: Geographic Regions, The Fall Line and Early Settlements, Rivers and Water Power, The
Title: Falling into Place Stage 1 Desired Results Subject/Course: Grade: 8th Instructional Time: 10 hrs. Big Ideas: Geographic Regions, The Fall Line and Early Settlements, Rivers and Water Power, The
Accelerated Chemistry Study Guide Atomic Structure, Chapter 3
 Accelerated Chemistry Study Guide Atomic Structure, Chapter 3 Terms and definitions atom ion law of constant composition isotope atomic theory of matter mass number cathode ray tube atomic mass electron
Accelerated Chemistry Study Guide Atomic Structure, Chapter 3 Terms and definitions atom ion law of constant composition isotope atomic theory of matter mass number cathode ray tube atomic mass electron
What happens during nuclear decay? During nuclear decay, atoms of one element can change into atoms of a different element altogether.
 When Henri Becquerel placed uranium salts on a photographic plate and then developed the plate, he found a foggy image. The image was caused by rays that had not been observed before. For his discovery
When Henri Becquerel placed uranium salts on a photographic plate and then developed the plate, he found a foggy image. The image was caused by rays that had not been observed before. For his discovery
Nuclear Chemistry Bravo 15,000 kilotons
 Nuclear Chemistry Nuclear Chemistry Bravo 15,000 kilotons Radioactive elements Radioactive elements Elements with atomic numbers greater than 83 are radioactive Why? These elements have too many protons
Nuclear Chemistry Nuclear Chemistry Bravo 15,000 kilotons Radioactive elements Radioactive elements Elements with atomic numbers greater than 83 are radioactive Why? These elements have too many protons
Ch05. Radiation. Energy and matter that comes from the nucleus of an atom. version 1.6
 Ch05 Radiation Energy and matter that comes from the nucleus of an atom. version 1.6 Nick DeMello, PhD. 2007-2016 Ch05 Radiation The Discovery of Radioactivity Phosphorescence Radioactive history Antoine
Ch05 Radiation Energy and matter that comes from the nucleus of an atom. version 1.6 Nick DeMello, PhD. 2007-2016 Ch05 Radiation The Discovery of Radioactivity Phosphorescence Radioactive history Antoine
Nuclear Physics and Astrophysics
 Nuclear Physics and Astrophysics PHY-302 Dr. E. Rizvi Lecture 1 - Course Organiser: Deputy: Dr E. Rizvi (room 401) Prof. J. Emerson My Office hours 1000 1100 Thursday 3 lecture slots per week Thursday
Nuclear Physics and Astrophysics PHY-302 Dr. E. Rizvi Lecture 1 - Course Organiser: Deputy: Dr E. Rizvi (room 401) Prof. J. Emerson My Office hours 1000 1100 Thursday 3 lecture slots per week Thursday
Chemistry Physical and Chemical Changes in Matter
 Physical and Chemical Changes in Matter Expanding Science Skills Series By Dr. Barbara R. Sandall Consultants: Schyrlet Cameron and Carolyn Craig COPYRIGHT 2010 Mark Twain Media, Inc. ISBN 978-1-58037-955-7
Physical and Chemical Changes in Matter Expanding Science Skills Series By Dr. Barbara R. Sandall Consultants: Schyrlet Cameron and Carolyn Craig COPYRIGHT 2010 Mark Twain Media, Inc. ISBN 978-1-58037-955-7
6. Atomic and Nuclear Physics
 6. Atomic and Nuclear Physics Chapter 6.2 Radioactivity From IB OCC, prepared by J. Domingues based on Tsokos Physics book Warm Up Define: nucleon atomic number mass number isotope. Radioactivity In 1896,
6. Atomic and Nuclear Physics Chapter 6.2 Radioactivity From IB OCC, prepared by J. Domingues based on Tsokos Physics book Warm Up Define: nucleon atomic number mass number isotope. Radioactivity In 1896,
10.1 RADIOACTIVE DECAY
 10.1 RADIOACTIVE DECAY When Henri Becquerel placed uranium salts on a photographic plate and then developed the plate, he found a foggy image. The image was caused by rays that had not been observed before.
10.1 RADIOACTIVE DECAY When Henri Becquerel placed uranium salts on a photographic plate and then developed the plate, he found a foggy image. The image was caused by rays that had not been observed before.
Field 046: Science Physical Science Assessment Blueprint
 Field 046: Science Physical Science Assessment Blueprint Domain I Foundations of Science 0001 The Nature and Processes of Science (Standard 1) 0002 Central Concepts and Connections in Science (Standard
Field 046: Science Physical Science Assessment Blueprint Domain I Foundations of Science 0001 The Nature and Processes of Science (Standard 1) 0002 Central Concepts and Connections in Science (Standard
The Scientific Revolution
 Chapter 18, Section 2 The Scientific Revolution (Pages 670 679) Setting a Purpose for Reading Think about these questions as you read: How did the Scientific Revolution change life in the 1600s? What is
Chapter 18, Section 2 The Scientific Revolution (Pages 670 679) Setting a Purpose for Reading Think about these questions as you read: How did the Scientific Revolution change life in the 1600s? What is
History of Science School Program
 Library, Art Collections, and Botanical Gardens History of Science School Program Week 3 Three Laws of Motion Theory of Gravity Theory of light and color Calculus Solving the problem of comets Prediction
Library, Art Collections, and Botanical Gardens History of Science School Program Week 3 Three Laws of Motion Theory of Gravity Theory of light and color Calculus Solving the problem of comets Prediction
Fill in the Following Table in your notes (assume an atom unless otherwise stated: Symbol Protons Electrons Neutrons Atomic # Mass # 24 Na
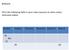 Bellwork: Fill in the Following Table in your notes (assume an atom unless otherwise stated: Symbol Protons Electrons Neutrons Atomic # Mass # 24 Na + 11 29 63 Bellwork: Fill in the Following Table in
Bellwork: Fill in the Following Table in your notes (assume an atom unless otherwise stated: Symbol Protons Electrons Neutrons Atomic # Mass # 24 Na + 11 29 63 Bellwork: Fill in the Following Table in
A Correlation of Prentice Hall World History The Modern Era 2014
 A Correlation of Prentice Hall World History The Modern Era 2014 To the College, Career, & Civic Life (C3) Framework for Social Studies State Standards Dimension 2 Disciplinary Indicators Introduction
A Correlation of Prentice Hall World History The Modern Era 2014 To the College, Career, & Civic Life (C3) Framework for Social Studies State Standards Dimension 2 Disciplinary Indicators Introduction
